Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116420
Revised: December 17, 2025
Accepted: January 23, 2026
Published online: April 15, 2026
Processing time: 140 Days and 22.5 Hours
Post-intensive care syndrome (PICS) is a frequent complication following major gastrointestinal tumor surgery, characterized by persistent physical impairment associated with systemic inflammation, muscle catabolism, and neuromuscular dysfunction.
To investigate the impact of multimodal rehabilitation nursing (MRN) on physical functional recovery in post-operative gastrointestinal tumor patients with PICS, using mechanism-related outcome indicators.
This retrospective study enrolled 80 patients with PICS after gastrointestinal tumor surgery between August 2022 and August 2024. Patients who received an 8-week MRN program were compared with those receiving conventional care. Physical performance, inflammatory status, nutritional parameters, and neuromu
Compared with conventional care, MRN was associated with significant improve
MRN significantly improves physical recovery in post-operative PICS patients by modulating inflammation, mu
Core Tip: This retrospective analysis shows that early initiation of an 8-week multimodal rehabilitation nursing program significantly improves physical functional recovery in patients with gastrointestinal tumors and post-intensive care syndrome. Beyond enhancing handgrip strength, nutritional status, and mobility, multimodal rehabilitation nursing favorably influenced key pathophysiological processes by reducing systemic inflammation, as reflected by lower interleukin-6 levels, attenuating muscle catabolism through myostatin modulation, and improving neuromuscular electrophysiological function, as evidenced by increased compound muscle action potential amplitude/area ratios. These results support the incorporation of mechanism-based, multidisciplinary nursing interventions into routine post-operative care to improve long-term patient outcomes.
- Citation: Chen J, Zhang AQ. Impact of multimodal rehabilitation on physical function in post-intensive care syndrome patients after gastrointestinal tumor surgery. World J Gastrointest Oncol 2026; 18(4): 116420
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116420.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116420
With ongoing advances in surgical techniques and perioperative management, post-operative safety for patients with gastrointestinal tumors has improved substantially. However, advanced age, extensive radical resections, and the presence of multiple comorbidities continue to necessitate post-operative admission to the intensive care unit (ICU) for a subset of patients. During ICU hospitalization, exposure to mechanical ventilation, deep sedation, prolonged immobi
PICS is defined as newly acquired or worsening physical, cognitive, or psychological impairments that persist after ICU discharge. Among these domains, physical dysfunction is the most common in survivors of surgical intensive care. Its development is primarily driven by persistent systemic inflammation, accelerated skeletal muscle catabolism, and impairment of the neuromuscular junction, forming a pathological “inflammation-muscle-nerve” axis. Conventional post-operative nursing care primarily focuses on basic mobilization and general functional exercises, which are often insufficient to address the complex, multifactorial mechanisms underlying PICS. However, multimodal rehabilitation approaches integrate nutritional optimization, neuromuscular stimulation, motor-cognitive training, and autonomic regulation, enabling targeted intervention across the full spectrum of PICS pathophysiology.
Current rehabilitation nursing practice largely depends on macro-level functional assessment tools, such as the Barthel Index and the 6-minute walk test. While clinically convenient, these measures do not adequately reflect micro-level pathological changes in muscle metabolism, nerve conduction, or cellular function. Growing evidence suggests that physical impairment in PICS represents a multisystem disorder involving the “inflammation-muscle-nerve” axis, rendering single-modality interventions, such as early ambulation alone, insufficient to reverse its complex pathological processes[3,4]. Multimodal rehabilitation nursing (MRN) is based on a mechanism-oriented model that emphasizes multidisciplinary collaboration and dynamically tailored intervention strategies[5]. However, standardized intervention protocols and sensitive, mechanism-based evaluation systems specifically designed for gastrointestinal tumor patients with PICS remain limited.
This study aims to assess the effects of MRN on physical functional recovery in patients with gastrointestinal tumors and PICS and to elucidate its underlying mechanisms using comprehensive, multidimensional outcome measures, including novel biological and neurophysiological indicators. The results are expected to provide an evidence-based framework for the development of precise and individualized post-operative rehabilitation strategies in oncological care.
A total of 80 patients with gastrointestinal tumors who required post-operative ICU admission for at least 72 hours at Second People’s Hospital of Changzhou (Third Affiliated Hospital of Nanjing Medical University), between August 2022 and August 2024 were retrospectively included in this study. Inclusion criteria: (1) Histopathologically confirmed gastric or colorectal cancer; (2) Completion of radical surgical resection, including D2 lymphadenectomy or total mesorectal excision; (3) Diagnosis of PICS before discharge by a multidisciplinary team consisting of intensivists, rehabilitation physicians, nutritionists, and nursing staff, based on the Needham criteria (history of ICU stay plus at least one persistent physical dysfunction, with primary neuromuscular diseases excluded); (4) Age between 40 and 75 years; and (5) Availa
Exclusion criteria: (1) Preoperative sarcopenia, defined as a third lumbar vertebra (L3) skeletal muscle index < 43 cm2/m2 for men and < 41 cm2/m2 for women; (2) Preexisting conditions affecting motor function, such as peripheral neuro
Forty patients who received the MRN protocol implemented from March 2023 onward were allocated to the inter
Patients in the control group received routine post-operative care, primarily consisting of continuous monitoring of vital signs and drainage output, maintenance of fluid and electrolyte balance, and early initiation of enteral nutrition within 24 hours of surgery. Enteral feeding was initiated at 20 mL/hour and gradually advanced to a target energy intake of 25 kcal/kg/day and protein intake of 1.2 g/kg/day using a whole-protein formula. Beginning on post-operative day 3, nursing staff assisted patients with passive joint range-of-motion exercises and sitting at the bedside once or twice daily, with each session lasting 10-15 minutes. Following drain removal and stabilization of vital signs, patients progressed to standing at the bedside and short-distance ambulation. Before discharge, the responsible nurse provided routine health education, including guidance on wound care, dietary management, and follow-up visits; however, no structured exercise prescription or individualized training program was implemented.
In addition to conventional care, patients in the intervention group received an 8-week MRN program initiated on post-operative day 1. The program was coordinated by a critical care specialist nurse and delivered by a multidisciplinary team comprising rehabilitation therapists, nutritionists, and physical therapists, with interventions dynamically tailored according to the patient’s daily functional status. The MRN protocol included the following components: (1) Anti-inflammatory nutritional support: Enteral nutrition was commenced within 24 hours postoperatively, with targets of 30 kcal/kg/day and protein 1.8 g/kg/day, supplemented with ω-3 fatty acids (eicosapentaenoic acid + docosahexaenoic acid, 2.0 g/day), vitamin D3 (2000 IU/day), and leucine (3.0 g/day) to suppress inflammation and enhance muscle protein synthesis; (2) Neuromuscular electrical stimulation (NMES): From post-operative day 3, NMES was applied to the bilateral quadriceps muscles (50 Hz, 300 μs, 20 minutes per session, once daily) to preserve muscle fiber integrity and neuromuscular junction function; (3) Dual-task (motor-cognitive) training: Beginning on post-operative day 7, patients performed concurrent cognitive tasks (e.g., backward digit span, color recognition) during walking exercises three times per week for 30 minutes per session to improve central motor control; and (4) Biofeedback-assisted breathing training: Slow-paced diaphragmatic breathing (≤ 6 breaths per minute) was conducted using a Resperate® device for 15 minutes daily over a 4-week period to regulate autonomic function and attenuate inflammatory responses.
Throughout the intervention, the multidisciplinary team reviewed and adjusted the rehabilitation plan weekly based on key indicators, including interleukin (IL)-6 levels, phase angle (PhA), handgrip strength (HGS), and functional performance. Following hospital discharge, patients received continued guidance for home-based rehabilitation through telephone follow-up and an online platform to ensure continuity of care and adherence to the intervention.
Observation indicators were collected at the following time points as required: One day before surgery (T0), post-operative day 3 (T1), 8 weeks postoperatively (T2), and 3 months postoperatively (T3).
Muscle structure and metabolism-related indicators: HGS was assessed using a Jamar hydraulic hand dynamometer. Patients were seated with the elbow flexed at 90°, and maximal grip strength of the dominant hand was measured three times; the highest value was recorded (kg). Assessments were conducted at T0, T1, T2, and T3. Serum albumin. At each time point, 3 mL of fasting venous blood was collected in the morning. Samples were centrifuged at 3000 rpm for 10 minutes, after which the serum was separated and analyzed using an automated biochemical analyzer. The normal reference range for serum albumin is 35-50 g/L. Serum albumin serves as an indicator of nutritional reserve and inflammatory status, with reduced levels closely associated with impaired post-operative muscle protein synthesis and delayed functional recovery.
Neuro-motor control-related indicators: Timed up and go (TUG) test. The TUG test was performed with the patient seated in a standard armchair (seat height 46 cm). On command, the patient stood up, walked 3 m, turned around a marker, returned to the chair, and sat down. The total time required to complete the task was recorded in seconds, with longer durations indicating poorer balance and mobility. Assessments were conducted at T2 and T3.
Compound muscle action potential (CMAP) amplitude/area (A/A) ratio: Neuromuscular function was evaluated using an electromyography system (Keypoint Classic). The common peroneal nerve was electrically stimulated, and CMAPs were recorded from the tibialis anterior muscle. The ratio of CMAP amplitude (mV) to the negative phase area (mV millisecond) was calculated. This parameter minimizes the influence of muscle atrophy and provides a specific measure of neuromuscular junction transmission efficiency. Gait-related assessments, including the TUG test and CMAP A/A ratio, were performed only at T2 and T3, as these evaluations require patients to have achieved basic ambulatory capacity.
Systemic inflammation and cellular function indicators: Serum IL-6. Serum IL-6 levels were quantified using a double-antibody sandwich enzyme-linked immunosorbent assay, serving as a key indicator of the post-operative systemic inflammatory response. Limb PhA: PhA was assessed using a multi-frequency bioelectrical impedance analyzer (InBody S10; InBody Co., South Korea). Following an overnight fast and bladder emptying, patients were positioned supine with bare feet in contact with the electrodes. The device automatically generated PhA values (degrees) at 50 kHz, reflecting cellular membrane integrity and body cell mass.
Statistical analyses were conducted using SPSS version 27.0. Data normality was assessed with the Shapiro-Wilk test. Variables following a normal distribution are presented as mean ± SD and were compared using independent-samples t tests or repeated-measures analysis of variance, as appropriate. Non-normally distributed variables are reported as median (interquartile range) and were analyzed using the Mann-Whitney U test. Categorical data are expressed as n (%) and were compared using the χ2 test or Fisher’s exact test, as applicable. Multivariate analyses were performed using multiple linear regression models. All statistical tests were two-tailed, and a P value < 0.05 was considered statistically significant.
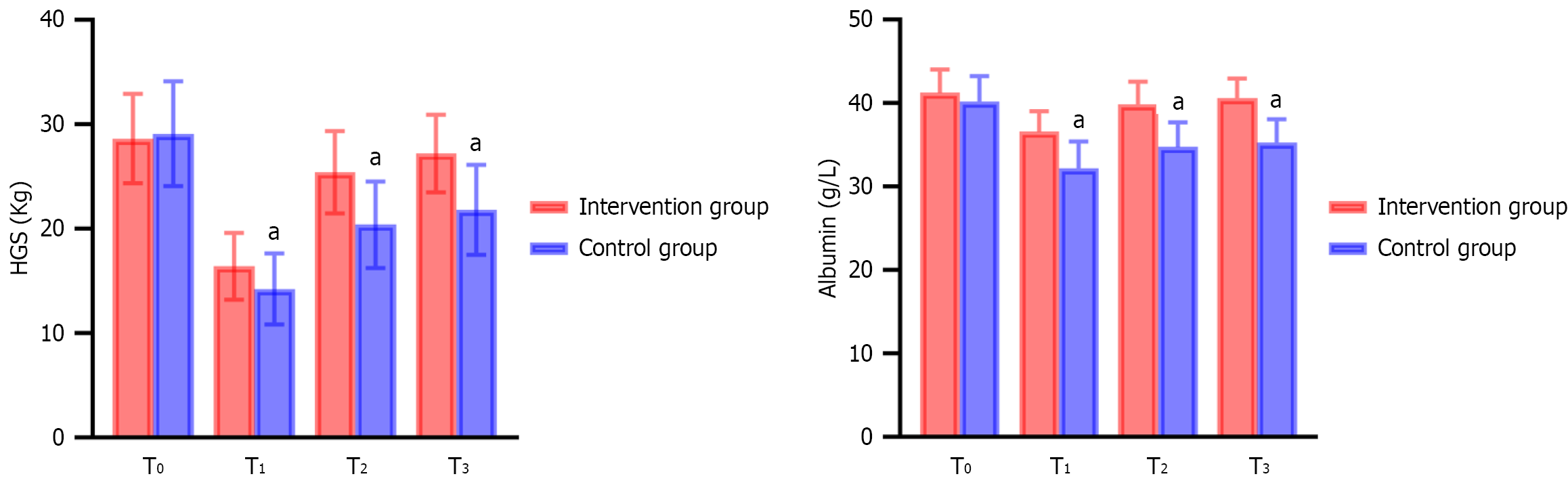
At baseline (T0), no significant differences in HGS or serum albumin levels were observed between the two groups (P > 0.05). However, significant between-group differences emerged at T1, T2, and T3 (all P < 0.05), with the intervention group consistently demonstrating higher HGS and serum albumin levels than the control group. These findings are summarized in Table 1 and illustrated in Figure 1.
| Indicator | Time point | Intervention group (n = 40) | Control group (n = 40) | F value (group/time/interaction) | P value |
| HGS (kg) | T0 | 28.62 ± 4.28 | 29.09 ± 5.02 | ||
| T1 | 16.38 ± 3.19 | 14.22 ± 3.41 | 44.87/156.30/9.21 | 0.00 | |
| T2 | 25.40 ± 3.96 | 20.37 ± 4.15 | |||
| T3 | 27.19 ± 3.72 | 21.80 ± 4.32 | |||
| Albumin (g/L) | T0 | 41.22 ± 2.81 | 40.91 ± 3.02 | ||
| T1 | 36.56 ± 2.46 | 32.19 ± 3.20 | 160.90/69.10/10.23 | 0.00 | |
| T2 | 39.87 ± 2.69 | 34.71 ± 2.98 | |||
| T3 | 40.59 ± 2.34 | 35.29 ± 2.76 |
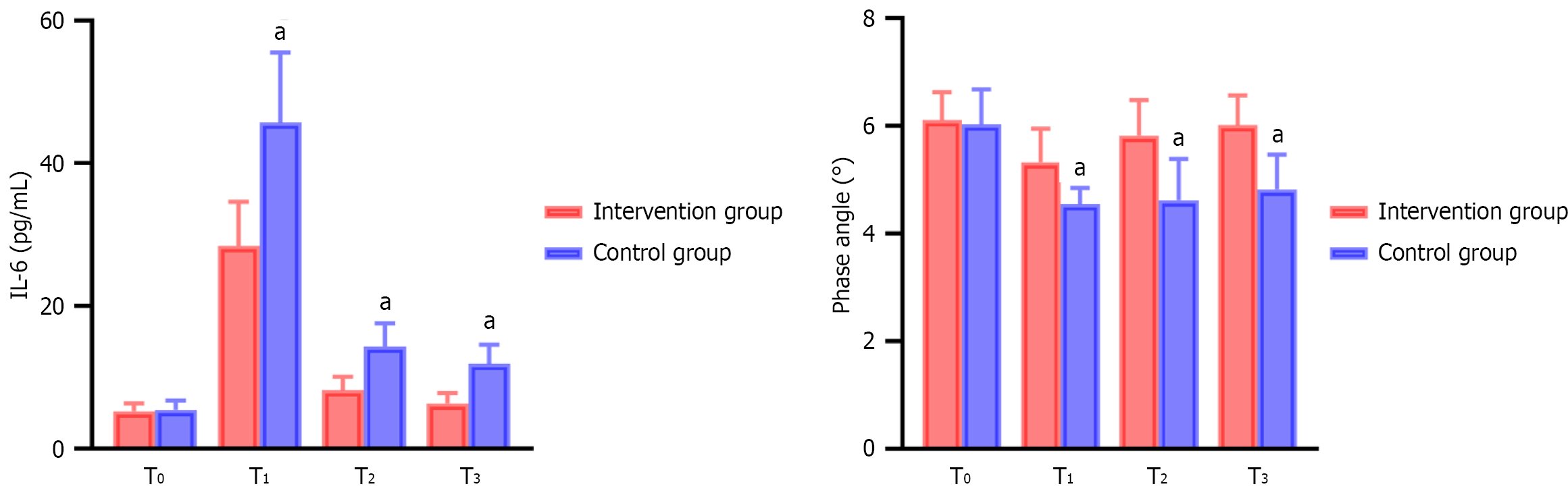
At T0, there were no significant differences in serum IL-6 levels or PhA between the two groups (P > 0.05). However, significant between-group differences were observed at T1, T2, and T3 (all P < 0.05), with the intervention group demonstrating lower IL-6 levels and higher PhA values than the control group. These results are presented in Table 2 and Figure 2.
| Indicator | Time point | Intervention group (n = 40) | Control group (n = 40) | F value (group/time/interaction) | P value |
| IL-6 (pg/mL) | T0 | 5.22 ± 1.10 | 5.40 ± 1.32 | ||
| T1 | 28.42 ± 6.21 | 45.73 ± 9.84 | |||
| T2 | 8.17 ± 1.93 | 14.32 ± 3.25 | 210.60/827.50/51.12 | 0.00 | |
| T3 | 6.32 ± 1.43 | 11.88 ± 2.70 | |||
| Phase angle (°) | T0 | 6.11 ± 0.52 | 6.03 ± 0.65 | ||
| T1 | 5.33 ± 0.62 | 4.55 ± 0.73 | 126.40/43.93/13.29 | 0.00 | |
| T2 | 5.82 ± 0.66 | 4.62 ± 0.77 | |||
| T3 | 6.02 ± 0.55 | 4.82 ± 0.65 |
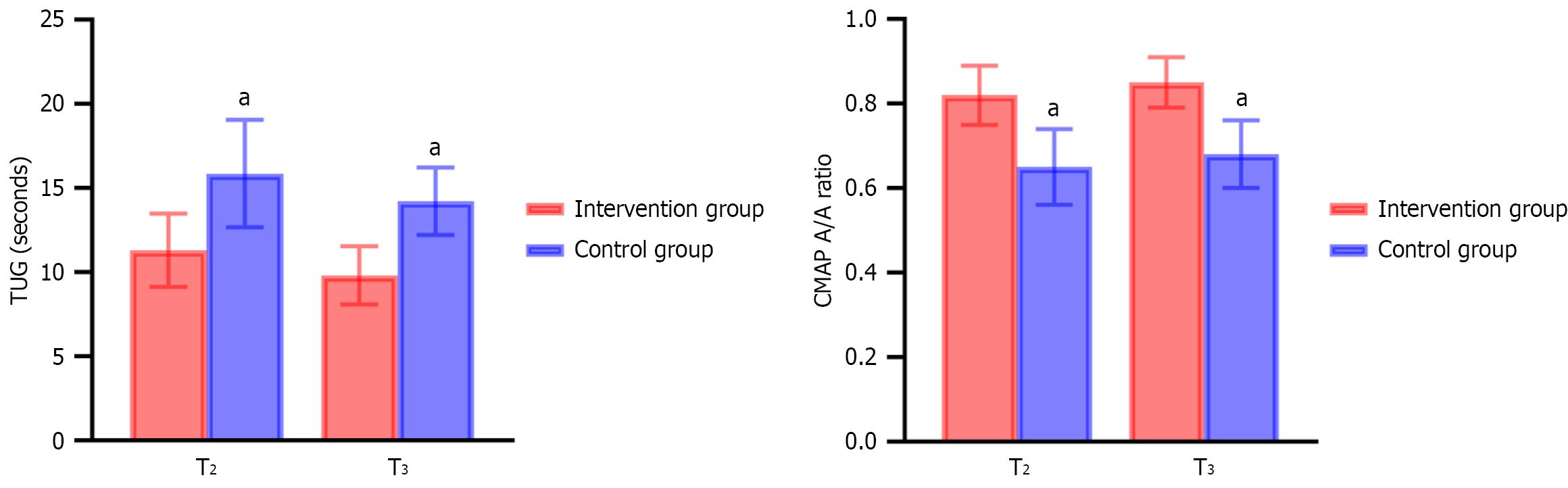
Significant differences between the two groups were identified in TUG performance and CMAP A/A ratio at both T2 and T3 (P < 0.05). The intervention group demonstrated shorter TUG times and higher CMAP A/A ratios than the control group (P < 0.05). These findings are detailed in Table 3 and illustrated in Figure 3.
| Indicator | Time point | Intervention group (n = 40) | Control group (n = 40) | t-value (group/time/interaction) | P value |
| TUG (seconds) | T2 | 11.29 ± 2.18 | 15.85 ± 3.20 | 7.45 | 0.00 |
| T3 | 9.80 ± 1.73 | 14.20 ± 2.01 | 10.49 | 0.00 | |
| CMAP A/A ratio | T2 | 0.82 ± 0.07 | 0.65 ± 0.09 | 9.43 | 0.00 |
| T3 | 0.85 ± 0.06 | 0.68 ± 0.08 | 10.75 | 0.00 |
The time to functional recovery was significantly shorter in the intervention group than in the control group, and the incidence of falls and hospital readmissions was also considerably lower in the intervention group (P < 0.05). These results are summarized in Table 4.
| Indicator | Intervention group (n = 40) | Control group (n = 40) | t/χ2 | P value |
| Time to recovery of somatic function (days), mean ± SD | 18.31 ± 4.24 | 25.76 ± 6.85 | 5.85 | 0.00 |
| Falls in the past 30 days | 1 (2.50) | 8 (20.00) | 0.03 | |
| Non-planned readmissions within 90 days | 2 (5.00) | 10 (25.00) | 0.03 |
Multiple linear regression analysis was conducted with time to functional recovery at 3 months postoperatively as the dependent variable. Independent variables included group assignment (intervention = 1, control = 0), age, surgical approach (open = 1, laparoscopic = 0), baseline tumor type, IL-6 level at T1, HGS at T2, TUG time at 8 weeks postoperatively, and the CMAP A/A ratio. The MRN intervention, T2 HGS, and CMAP A/A ratio emerged as independent negative predictors of time to functional recovery, whereas T1 IL-6 levels and TUG time were identified as positive predictors. The detailed results are presented in Table 5.
| Variable | Regression coefficient (β) | SE | 95% confidence interval | t-value | P value |
| Intervention group (intervention = 1) | -6.82 | 0.82 | -8.45 to -5.19 | -8.32 | 0.00 |
| Age (per 1-year increase) | 0.21 | 0.13 | -0.05 to 0.47 | 1.62 | 0.11 |
| Open surgery (yes = 1) | 1.84 | 0.97 | -0.10 to 3.78 | 1.90 | 0.06 |
| Gastric cancer | 0.63 | 1.12 | -1.60 to 2.86 | 0.56 | 0.58 |
| T1 IL-6 (per 1 pg/mL increase) | 0.78 | 0.31 | 0.16 to 1.40 | 2.52 | 0.01 |
| T2 HGS (per 1 kg increase) | -1.23 | 0.28 | -1.79 to -0.67 | -4.39 | 0.00 |
| T2 TUG (per 1 second increase) | 2.15 | 0.42 | 1.31 to 2.99 | 5.12 | 0.00 |
| T2 CMAP A/A ratio (per 0.1 increase) | -3.04 | 0.58 | -4.20 to -1.88 | -5.24 | 0.00 |
Using a retrospective design, this study systematically examined the effects of MRN on physical functional recovery in patients with PICS following gastrointestinal tumor surgery. The findings demonstrate that MRN not only significantly improves muscle integrity, neuromotor control, and systemic inflammatory status but also shortens time to functional recovery and reduces the incidence of falls and hospital readmissions. These results provide meaningful evidence for developing a mechanism-driven, precision-oriented post-operative rehabilitation framework.
HGS, a robust yet straightforward indicator of upper-limb muscle strength and overall muscular function, is widely recognized as a sensitive marker of post-operative sarcopenia and functional deterioration[6]. In this study, HGS values in the intervention group were significantly higher than those in the control group at both 8 weeks (T2) and 3 months (T3) after surgery. Moreover, multivariate regression analysis revealed that each 1-kg increase in T2 HGS was associated with a 1.23-day reduction in time to functional recovery (β = -1.23, 95% confidence interval: -1.79 to -0.67), indicating that MRN effectively preserves and enhances muscle strength recovery. This benefit is likely attributable to the synergistic nutritional strategy embedded in the MRN protocol, which includes high protein intake (1.8 g/kg/day), leucine supplementation (3.0 g/day), and ω-3 polyunsaturated fatty acids (eicosapentaenoic acid + docosahexaenoic acid, 2.0 g/day). Leucine stimulates muscle protein synthesis through activation of the mechanistic target of rapamycin complex 1 pathway[7,8]. At the same time, ω-3 fatty acids suppress nuclear factor-κB signaling and downregulate muscle-specific ubiquitin ligases, attenuating ubiquitin-proteasome system-mediated myofibrillar degradation[9,10].
Further analyses showed that serum albumin levels in the intervention group were significantly higher than those in the control group as early as post-operative day 3 (T1) and remained consistently elevated throughout subsequent follow-up assessments. This effect is attributable primarily to the MRN nutritional strategy, which includes high-protein intake (1.8 g/kg/day), leucine supplementation (3.0 g/day), and ω-3 fatty acids. These components not only directly enhance muscle protein synthesis but also support hepatic albumin production by attenuating systemic inflammatory responses, including reductions in IL-6 levels[11,12]. Early restoration of serum albumin reflects a metabolic shift from catabolism toward anabolism, providing a physiological foundation for muscle structural repair and functional rehabilitation. These findings further support serum albumin as a practical, readily accessible, and clinically meaningful biomarker for monitoring post-operative rehabilitation progress.
Post-operative systemic inflammation is a central driver of muscle catabolism[13]. In this study, serum IL-6 levels at T1 were significantly lower in the intervention group than in the control group (28.42 ± 6.21 pg/mL vs 45.73 ± 9.84 pg/mL), and T1 IL-6 emerged as an independent positive predictor of time to functional recovery (β = 0.78, P = 0.01), underscoring the critical importance of effective inflammatory control within the first 72 hours after surgery. PhA values in the intervention group were significantly higher at T1 (5.33 ± 0.62° vs 4.55 ± 0.73°) and remained consistently superior during subsequent follow-up.
PhA, derived from multi-frequency bioelectrical impedance analysis, reflects cellular membrane integrity, metabolic activity, and body cell mass[14-16]. The mitochondrial protection strategy incorporating coenzyme Q10 and L-carnitine improves oxidative phosphorylation efficiency and stabilizes mitochondrial membrane potential, improving cellular energy metabolism and contributing to higher PhA values[17-19]. The concurrent reduction in IL-6 and increase in PhA suggest that MRN preserves cellular functional integrity through complementary mechanisms of inflammation sup
Disrupted structural and functional remodeling of the neuromuscular junction represents a key microscopic mechanism underlying impaired motor control in patients with PICS[21]. In this study, TUG times in the intervention group were significantly shorter than those in the control group at both T2 and T3. Multiple regression analysis further demonstrated that each 1-second increase in T2 TUG time was associated with a 2.15-day prolongation of time to functional recovery. Shorter TUG times, therefore, reflect improved dynamic balance, gait stability, and overall mobility in patients receiving MRN.
The CMAP A/A ratio in the intervention group reached 0.82 ± 0.07 at T2, significantly exceeding that in the control group (0.65 ± 0.09) and emerged as a strong negative predictor of time to functional recovery. By accounting for the confounding effects of muscle atrophy on CMAP amplitude, the CMAP A/A ratio provides a specific measure of neuromuscular junction transmission efficiency[22,23]. NMES contributes to the preservation of acetylcholine receptor clustering[24,25], while dual-task (motor-cognitive) training improves corticospinal regulatory precision. These interven
In summary, by integrating multidimensional indicators, including HGS, serum albumin, IL-6, PhA, CMAP A/A ratio, and TUG performance, this study systematically elucidates the multi-target mechanisms by which MRN improves physical function in PICS patients following gastrointestinal tumor surgery. MRN effectively shortens time to functional recovery and reduces the risk of adverse events by coordinating the suppression of inflammation, regulation of muscle metabolism, preservation of cellular integrity, and restoration of neuromotor control. These findings not only provide a quantifiable, mechanism-based framework for rehabilitation assessment in clinical practice but also support the imple
| 1. | Pierre A, Favory R, Bourel C, Howsam M, Romien R, Lancel S, Preau S. Muscle weakness after critical illness: unravelling biological mechanisms and clinical hurdles. Crit Care. 2025;29:248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 2. | Lui KY, Luo G, Li S, Song X, Qian X, Dou R, Li L, Guan X, Cai C. Incidence and risk factors of Post-intensive care syndrome (PICS) in surgical ICU survivors: a prospective Chinese cohort study. BMC Public Health. 2024;24:3277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 3. | Chadda KR, Puthucheary Z. Persistent inflammation, immunosuppression, and catabolism syndrome (PICS): a review of definitions, potential therapies, and research priorities. Br J Anaesth. 2024;132:507-518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 61] [Article Influence: 30.5] [Reference Citation Analysis (1)] |
| 4. | Chadda KR, Blakey EE, Davies TW, Puthucheary Z. Risk factors, biomarkers, and mechanisms for persistent inflammation, immunosuppression, and catabolism syndrome (PICS): a systematic review and meta-analysis. Br J Anaesth. 2024;133:538-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 5. | Renner C, Jeitziner MM, Albert M, Brinkmann S, Diserens K, Dzialowski I, Heidler MD, Lück M, Nusser-Müller-Busch R, Sandor PS, Schäfer A, Scheffler B, Wallesch C, Zimmermann G, Nydahl P. Guideline on multimodal rehabilitation for patients with post-intensive care syndrome. Crit Care. 2023;27:301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 127] [Article Influence: 42.3] [Reference Citation Analysis (1)] |
| 6. | Vaishya R, Misra A, Vaish A, Ursino N, D'Ambrosi R. Hand grip strength as a proposed new vital sign of health: a narrative review of evidences. J Health Popul Nutr. 2024;43:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 184] [Article Influence: 92.0] [Reference Citation Analysis (2)] |
| 7. | Tamura K, Kitazawa H, Sugita S, Hashizume K, Iwashita M, Ishigami T, Minegishi Y, Shimotoyodome A, Ota N. Tyrosine Is a Booster of Leucine-Induced Muscle Anabolic Response. Nutrients. 2023;16:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Chen J, Liu X, Zou Y, Gong J, Ge Z, Lin X, Zhang W, Huang H, Zhao J, Saw PE, Lu Y, Hu H, Song E. A high-fat diet promotes cancer progression by inducing gut microbiota-mediated leucine production and PMN-MDSC differentiation. Proc Natl Acad Sci U S A. 2024;121:e2306776121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 79] [Article Influence: 39.5] [Reference Citation Analysis (1)] |
| 9. | Matsui R, Sagawa M, Sano A, Sakai M, Hiraoka SI, Tabei I, Imai T, Matsumoto H, Onogawa S, Sonoi N, Nagata S, Ogawa R, Wakiyama S, Miyazaki Y, Kumagai K, Tsutsumi R, Okabayashi T, Uneno Y, Higashibeppu N, Kotani J. Impact of Perioperative Immunonutrition on Postoperative Outcomes for Patients Undergoing Head and Neck or Gastrointestinal Cancer Surgeries: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Ann Surg. 2024;279:419-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 10. | Peris-Moreno D, Cussonneau L, Combaret L, Polge C, Taillandier D. Ubiquitin Ligases at the Heart of Skeletal Muscle Atrophy Control. Molecules. 2021;26:407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 11. | Haß U, Kochlik B, Herpich C, Rudloff S, Norman K. Effects of an Omega-3 Supplemented, High-Protein Diet in Combination with Vibration and Resistance Exercise on Muscle Power and Inflammation in Old Adults: A Pilot Randomized Controlled Trial. Nutrients. 2022;14:4274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 12. | Merchant RA, Chan YH, Anbarasan D, Seetharaman S, Au L, Nachammai V, Lai A, Ho V, Wong BLL, Pang E, Bhaskaran K. Impact of exercise and leucine-enriched protein supplementation on physical function, body composition, and inflammation in pre-frail older adults: a quasi-experimental study. Front Med (Lausanne). 2023;10:1204198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 13. | Ji Y, Li M, Chang M, Liu R, Qiu J, Wang K, Deng C, Shen Y, Zhu J, Wang W, Xu L, Sun H. Inflammation: Roles in Skeletal Muscle Atrophy. Antioxidants (Basel). 2022;11:1686. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 202] [Article Influence: 50.5] [Reference Citation Analysis (1)] |
| 14. | Gulin J, Ipavic E, Mastnak DM, Brecelj E, Edhemovic I, Kozjek NR. Phase angle as a prognostic indicator of surgical outcomes in patients with gastrointestinal cancer. Radiol Oncol. 2023;57:524-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 15. | da Silva BR, Orsso CE, Gonzalez MC, Sicchieri JMF, Mialich MS, Jordao AA, Prado CM. Phase angle and cellular health: inflammation and oxidative damage. Rev Endocr Metab Disord. 2023;24:543-562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 107] [Article Influence: 35.7] [Reference Citation Analysis (1)] |
| 16. | Sardinha LB, Rosa GB. Phase angle, muscle tissue, and resistance training. Rev Endocr Metab Disord. 2023;24:393-414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 17. | Cui W, Guo M, Liu D, Xiao P, Yang C, Huang H, Liang C, Yang Y, Fu X, Zhang Y, Liu J, Shi S, Cong J, Han Z, Xu Y, Du L, Yin C, Zhang Y, Sun J, Gu W, Chai R, Zhu S, Chu B. Gut microbial metabolite facilitates colorectal cancer development via ferroptosis inhibition. Nat Cell Biol. 2024;26:124-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 144] [Article Influence: 72.0] [Reference Citation Analysis (1)] |
| 18. | Zhang R, Kang R, Tang D. Gut Microbiome Mediates Ferroptosis Resistance for Colorectal Cancer Development. Cancer Res. 2024;84:796-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 19. | Cai Y, Zhao F, Cheng X. Gut Microbiota and Ferroptosis in Colorectal Cancer: A Comprehensive Review of Mechanisms and Therapeutic Strategies to Overcome Immune Checkpoint Resistance. Biomolecules. 2025;15:1546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 20. | Cioffi I, Scialò F, Di Vincenzo O, Gelzo M, Marra M, Testa A, Castiglione F, Vitale M, Pasanisi F, Santarpia L. Serum Interleukin 6, Controlling Nutritional Status (CONUT) Score and Phase Angle in Patients with Crohn's Disease. Nutrients. 2023;15:1953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 21. | Huang X, Jiang J, Xu J. Denervation-Related Neuromuscular Junction Changes: From Degeneration to Regeneration. Front Mol Neurosci. 2021;14:810919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 22. | Padilla CJ, Harrigan ME, Harris H, Schwab JM, Rutkove SB, Rich MM, Clark BC, Arnold WD. Profiling age-related muscle weakness and wasting: neuromuscular junction transmission as a driver of age-related physical decline. Geroscience. 2021;43:1265-1281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 23. | Waegaert R, Dirrig-Grosch S, Liu H, Boutry M, Luan P, Loeffler JP, René F. Alteration of the Neuromuscular Junction and Modifications of Muscle Metabolism in Response to Neuron-Restricted Expression of the CHMP2B(intron5) Mutant in a Mouse Model of ALS-FTD Syndrome. Biomolecules. 2022;12:497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 24. | Das P, Baloda V. Use of smooth muscle markers is better than the endothelial cell markers for identification of tumor venous invasion and extramural tumor deposits in gastrointestinal tract tumors. Indian J Pathol Microbiol. 2020;63:3-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 25. | Musarò A, Dobrowolny G, Cambieri C, Onesti E, Ceccanti M, Frasca V, Pisano A, Cerbelli B, Lepore E, Ruffolo G, Cifelli P, Roseti C, Giordano C, Gori MC, Palma E, Inghilleri M. Neuromuscular magnetic stimulation counteracts muscle decline in ALS patients: results of a randomized, double-blind, controlled study. Sci Rep. 2019;9:2837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (1)] |