Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116365
Revised: December 5, 2025
Accepted: January 4, 2026
Published online: April 15, 2026
Processing time: 148 Days and 7.6 Hours
Angiosarcoma is rare and carries a poor prognosis, often presenting subtly both clinically and radiologically. In some cases, it manifests only as persistent unex
This case report describes a patient who suffered from severe anemia that required multiple transfusions, without a clear underlying cause despite multiple examinations, until the diagnosis of intestinal angiosarcoma was estab
This case underscores the importance of closely monitoring patients on TKIs for potential, subtle underlying malignancies and maintaining a high level of suspicion, particularly in the context of unexplained anemia or other atypical symptoms.
Core Tip: Angiosarcoma is rare and carries a poor prognosis, often presenting subtly both clinically and radiologically. In some cases, it manifests only as persistent unexplained anemia, making its early recognition difficult. The tumor may remain hidden, mimicking benign lesions, with symptoms masked or altered by concurrent treatments. In patients receiving tyrosine kinase inhibitors, such as for chronic myeloid leukemia, modulation of the vascular endothelial growth factor pathway and related mechanisms can further obscure tumor behavior and distort imaging findings, contributing to diagnostic delay.
- Citation: Najdi T, Seif S, Khoury D, Kahwagi A, Wehbe P, Karam R, El Halabi L, Slim R, Chakhtoura G, Trak V, Eid R. Occult intestinal angiosarcoma presenting as refractory anemia during tyrosine kinase inhibitor therapy for chronic myeloid leukemia: A case report. World J Gastrointest Oncol 2026; 18(4): 116365
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116365.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116365
Angiosarcomas are rare sarcomas arising from endothelial cells, representing 1%–2% of soft tissue sarcomas[1]. While these tumors can arise in various locations, the visceral type, especially involving the small intestine, is exceptionally rare, with only a few cases reported in the literature[2,3]. These tumors are aggressive, with a notably high mortality rate because of their delayed diagnosis. The visceral type is more challenging to diagnose than other forms owing to its nonspecific clinical manifestations such as nausea, vomiting, abdominal pain, gastrointestinal bleeding, anemia, fatigue, and weakness. Diagnosis is even more complicated in cases involving the epithelioid variant, which presents additional cytological diagnostic difficulties.
Anemia is a nonspecific finding that is also observed in numerous other hematologic diseases, such as chronic myeloid leukemia (CML)[4]. In CML, anemia may occur as a feature of the disease itself, particularly in the chronic phase, or may develop as an adverse effect of tyrosine kinase inhibitor (TKI) therapy, including agents such as nilotinib. Moreover, TKI treatment may alter the vascular microenvironment, potentially complicating radiologic interpretation and obscuring new angio-malignancies[5]. Here, we present a diagnostically challenging case of an intestinal epithelioid angiosarcoma in a patient with CML undergoing TKI therapy, whose sole presenting symptom was anemia and whose imaging was difficult to interpret owing to the effects of his treatment. This case highlights the critical importance of maintaining diagnostic vigilance when evaluating nonspecific symptoms in immunocompromised patients, emphasizing the need for integrated clinical, radiologic, and cytologic assessment.
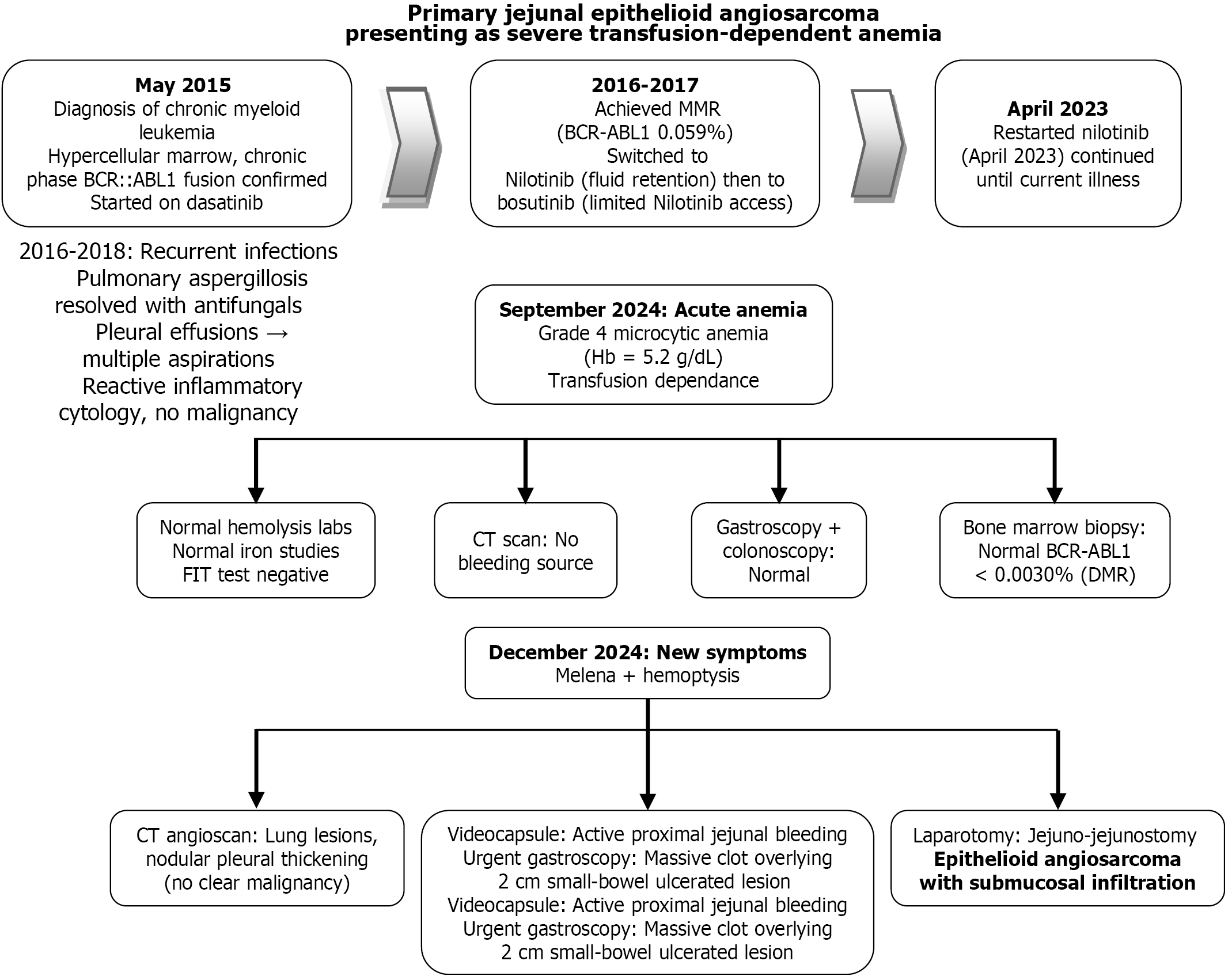
We present the case of a 55-year-old man who presented in September 2024 with severe grade 4 anemia (hemoglobin [Hb] = 5.2 g/dL) and extreme fatigue, prompting urgent hospitalization and initiation of the diagnostic workup discussed in this report.
The patient had a 9-year history of CML, which was initially diagnosed in May 2015 at the age of 45 years. At diagnosis, the bone marrow was hypercellular, with a marked myelemia observed on the peripheral blood smear, highly suggestive of chronic phase CML without signs of transformation or acceleration. Molecular biological analysis confirmed the presence of the BCR–ABL1 transcript. The patient was initially treated with dasatinib, and BCR–ABL transcript levels were regularly monitored, demonstrating a progressive decline. The patient remained stable until September 2024, when he began experiencing extreme fatigue and pallor.
The patient achieved a major molecular response in 2016, with a transcript level of 0.059%, and subsequently remained in molecular remission. In November 2017, therapy was switched to nilotinib (150 mg twice daily) owing to adverse effects, mainly fluid retention. In October 2021, the patient was transitioned to bosutinib (400 mg daily) owing to limited access to nilotinib at that time, before resuming nilotinib in April 2023, which he continued to receive until presentation.
Since the onset of CML, the patient had been clinically immunocompromised, experiencing multiple opportunistic infections despite normal and lymphocyte counts. Notably, in January 2016, he presented with a persistent cough and febrile episodes without dyspnea. A computed tomography (CT) scan performed as part of the diagnostic workup for persistent fever revealed bilateral pleural effusion and ascites, otherwise unremarkable pulmonary fields, mesenteric fat trabeculation, and loculated ascites. His clinical presentation and subsequent pleural fluid analysis led to the diagnosis of pulmonary aspergillosis, which was confirmed via PCR. The patient received antifungal therapy with voriconazole, resulting in the resolution of fever and improvement in pulmonary symptoms. The ascitic fluid was aspirated at that time for cytological analysis, and immunohistochemical staining (CK7, calretinin, BerEP4) was performed. Cytological examination did not reveal any malignancy but demonstrated reactive mesothelial cells, consistent with chronic inflammatory changes.
The persistent minimal pleural effusions required repeated bronchial aspirations in January and March 2018, which did not reveal any pathogenic organisms. The pleural fluid cytological analysis performed in January 2018 demonstrated a highly cellular sample, with a predominance of hematopoietic elements, including small mature lymphocytes, plasma cells, and both mononuclear and polymorphonuclear cells. Immature cells were also observed, along with vacuolated macrophages, indicating a reactive inflammatory process without evidence of malignancy. Special stains performed on subsequent samples, including Papanicolaou, DQ, Perls, PAS, Grocott, and Ziehl, revealed a high cellularity with macrophages (84%) and lymphocytes (16%). Minimal hemosiderin was noted, and no pathogenic organisms or malignant cells were identified, supporting a diagnosis of a nonspecific, non-malignant inflammatory pleural effusion.
In May 2022, a routine follow-up CT scan revealed no significant abnormalities. Incidental findings included focal spondylosis with disc space narrowing at the C5–C6 level, residually peripheral calcified lung lesions without evidence of active pulmonary cavities or infiltrates, multiple tiny bilateral peripheral pleural nodules, and small conglomerated calcified gallstones. Routine blood tests showed a Hb level of 13 g/dL, a white blood cell (WBC) count of 14.9 × 109/L with 20% lymphocytes, and a normal platelet count of 316 × 109/L. In May 2024, the Hb level was still 13.0 g/dL, the mean corpuscular volume (MCV) was 86 fL, ferritin was 137 ng/mL (normal), the WBC count was 14.9 × 109/L with 72% neutrophils and 20% lymphocytes, and the platelet count was 316 × 109/L, with no other abnormality.
The patient remained stable until September 2024, when he began experiencing extreme fatigue and pallor. Besides the icteric appearance, the patient appeared normal, conscious, cooperative, and oriented on clinical examination. Pulmonary auscultation was normal in both fields, and cardiac auscultation revealed slightly rapid but regular heart sounds. The abdomen was soft and non-tender on both deep and superficial palpation, with positive peristalsis. No evidence of gastrointestinal bleeding was observed; stools showed no macroscopic blood, with neither melena nor hematochezia.
Blood tests revealed a severe anemia (grade 4), and BCR–ABL levels were monitored. The BCR–ABL fusion transcript was quantified at < 0.0030%, indicating a stable deep molecular response (MR4.52). In view of this situation, nilotinib was suspended. A series of investigations were ordered to explore the cause of the anemia.
Laboratory examination revealed microcytic anemia (Hb = 5.2 g/dL, MCV = 68.7 fL, reticulocytes = 1.2%). Iron studies showed a serum iron level of 141.3 µg/dL and ferritin of 26.4 ng/mL (lower normal limit). Both direct and indirect Coombs tests were negative, and laboratory findings did not support a diagnosis of hemolytic anemia, with total bilirubin at 0.24 mg/dL (direct = 0.11 mg/dL, indirect = 0.13 mg/dL), haptoglobin at 365 mg/dL (high), and lactate dehydrogenase at 168 U/L. Liver function tests were normal, with serum glutamic pyruvic transaminase at 4.6 U/L, gamma-glutamyl transferase at 34.5 U/L, alkaline phosphatase at 65.4 U/L, and creatinine at 1.00 mg/dL. No vitamin deficiencies were detected: The patient’s vitamin B12 was 521.6 pg/mL, folic acid was 7.7 ng/mL, and thyroid function tests were within normal ranges (thyroid-stimulating hormone = 1.54 µU/mL, triiodothyronine = 2.52 pmol/L, thyroxine = 1.12 µg/dL). Clinically, there were no signs of bleeding or external hemorrhage, and a fecal immunochemical test (FIT) was negative (<50 ng Hb/mL) in October 2024.
Further investigations included serum protein electrophoresis, which revealed an inflammatory profile characterized by an increase in alpha globulins, although this finding was nonspecific. Viral etiologies were also investigated, including Parvovirus B19 and PCR testing for cytomegalovirus (CMV), which were all negative.
In addition to the blood tests, a total body CT scan (cervical–thoracic–abdominal–pelvic) was performed on September 24, 2024, with the following findings: Status post treatment of pulmonary and pleural aspergillosis with residual peripherally calcified cavitating lung lesion; no acute cavitating lung lesions or active lung infiltrates; multiple peripheral tiny bilateral pleural based lung nodules to be followed up according to Fleischner criteria; a single lower lung nodule measuring 9 mm × 11 mm; hepatomegaly with no focal mass lesions; and tiny conglomerated calcified gallstones. The scan was otherwise unremarkable.
Despite a negative FIT and unremarkable scan results, suspicion persisted. Therefore, a diagnostic gastro-colonoscopy was performed, which revealed no abnormalities at that time. Because of the patient’s multiple transfusion needs, with no signs of hemolytic anemia, hematologic disease, or CML relapse, close surveillance was maintained. A video capsule endoscopy was planned despite the normal CT scan, FIT, and endoscopic findings. However, it could not be performed at that time owing to insurance-related issues.
On October 16, 2024, the patient received his first intravenous iron transfusion (ferric carboxymaltose; Ferinject) owing to low ferritin levels. He tolerated the transfusion well, but by early November, he required repeated admissions for transfusions. During this time, his Hb levels were consistently low, and he required two units of blood every other day (on November 6, 14, 15, 19, 21, and 25), with only a modest increase in Hb to 7 g/dL. Table 1 summarizes the key differences in blood values before and after the patient’s acute presentation with severe anemia.
| Parameter | May 2024 (baseline) | September 2024 to October 2024 (acute presentation) |
| Hemoglobin (g/dL) | 13.0 | 5.2 |
| MCV (fL) | 86 | 68.7 (microcytic) |
| Ferritin (ng/mL) | 137 | 26.4 (low-normal) |
| Reticulocytes (%) | - | 1.2 |
| White blood count (× 109/L) | 14.9 | - (not significantly changed) |
| BCR-ABL1 (%) | 0.0061 | < 0.0030 (stable MR4.5) |
| Bilirubin (total/direct; mg/dL) | - | 0.24/0.11 |
| Haptoglobin (mg/dL) | - | 365 (high) |
| Lactate dehydrogenase (U/L) | - | 168 |
| FIT | - | Negative |
| Vitamin B12/folate | Normal | Normal |
The FIT was negative once again. To further investigate the underlying cause, a bone marrow biopsy performed on October 1, 2024, showed a normocellular marrow with mild granulocytic predominance. Bone marrow smear analysis demonstrated normal proportions and morphology of myeloid cells at all developmental stages, with no evidence of progression to the accelerated phase. Meanwhile, investigations continued to explore the cause of the patient’s anemia and frequent transfusion dependence, with continued efforts to obtain a video capsule endoscopy. The patient was not receiving any antiplatelet or anticoagulant therapy. He was empirically treated with tranexamic acid for suspected uncontrolled, undiagnosed bleeding, without any clinical benefit.
On December 5, 2024, the patient was admitted with anemia accompanied by melena and hemoptysis, which he reported had been occurring for the previous 2 weeks. A total-body CT angiographic scan performed on December 13 revealed a lobulated nodular lesion in the right lower lung lobe, with a spontaneous attenuation of 26 Hounsfield units (HU) and enhancement to 47 HU after contrast administration, with both peripheral and central calcifications. The lesion indented the right liver lobe and extended along the right diaphragm toward the right adrenal gland. Additional findings included left-sided nodular pleural thickening, a suspicious 2 cm × 2 cm nodule in the right upper lobe, and multiple bilateral subcentimetric parenchymal nodules compatible with infectious lesions. No active bleeding or definitive signs of malignancy were identified.
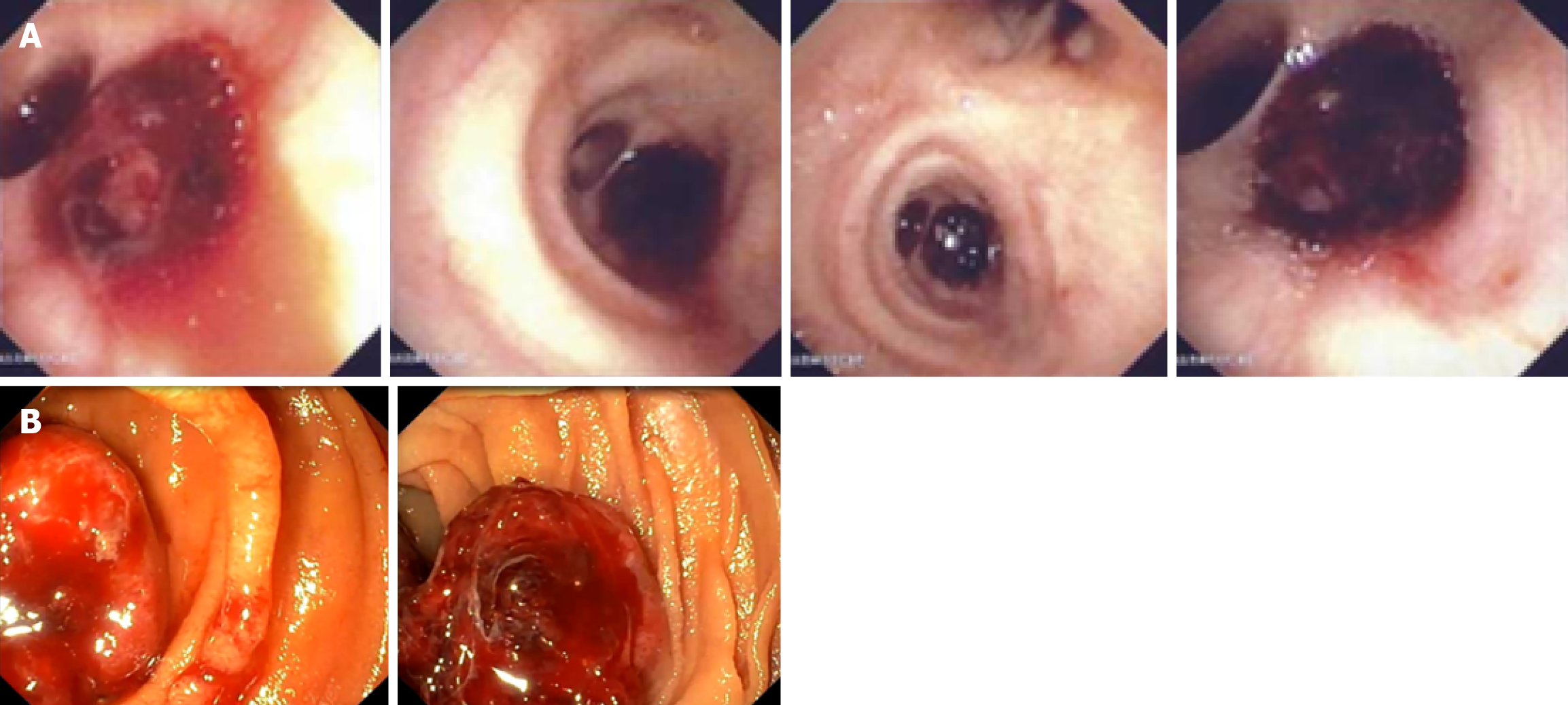
A video capsule endoscopy, performed a few days earlier as an outpatient, became available at the time of admission. It demonstrated active bleeding in the proximal jejunum with multiple clots arising from a single mucosal erosion, as well as a small amount of blood in the esophagus attributed to minor erosions near the tonsillar region. Images from the capsule endoscopy are shown in Figure 1. The ongoing melena necessitated an urgent repeat gastroscopy, despite a previously normal study. Gastroscopy showed a 3-mm congested nodule, covered by a clot, in the duodenum, of unclear origin, possibly a Dieulafoy lesion or a polyp. An endoclip was swiftly applied. As the examination progressed, the duodenum revealed a clot with bile-coated mucosa, which was aspirated to alleviate the obstruction. The most con
In light of the lesion characteristics and the ongoing bleeding requiring multiple transfusions, urgent surgical intervention was advised.
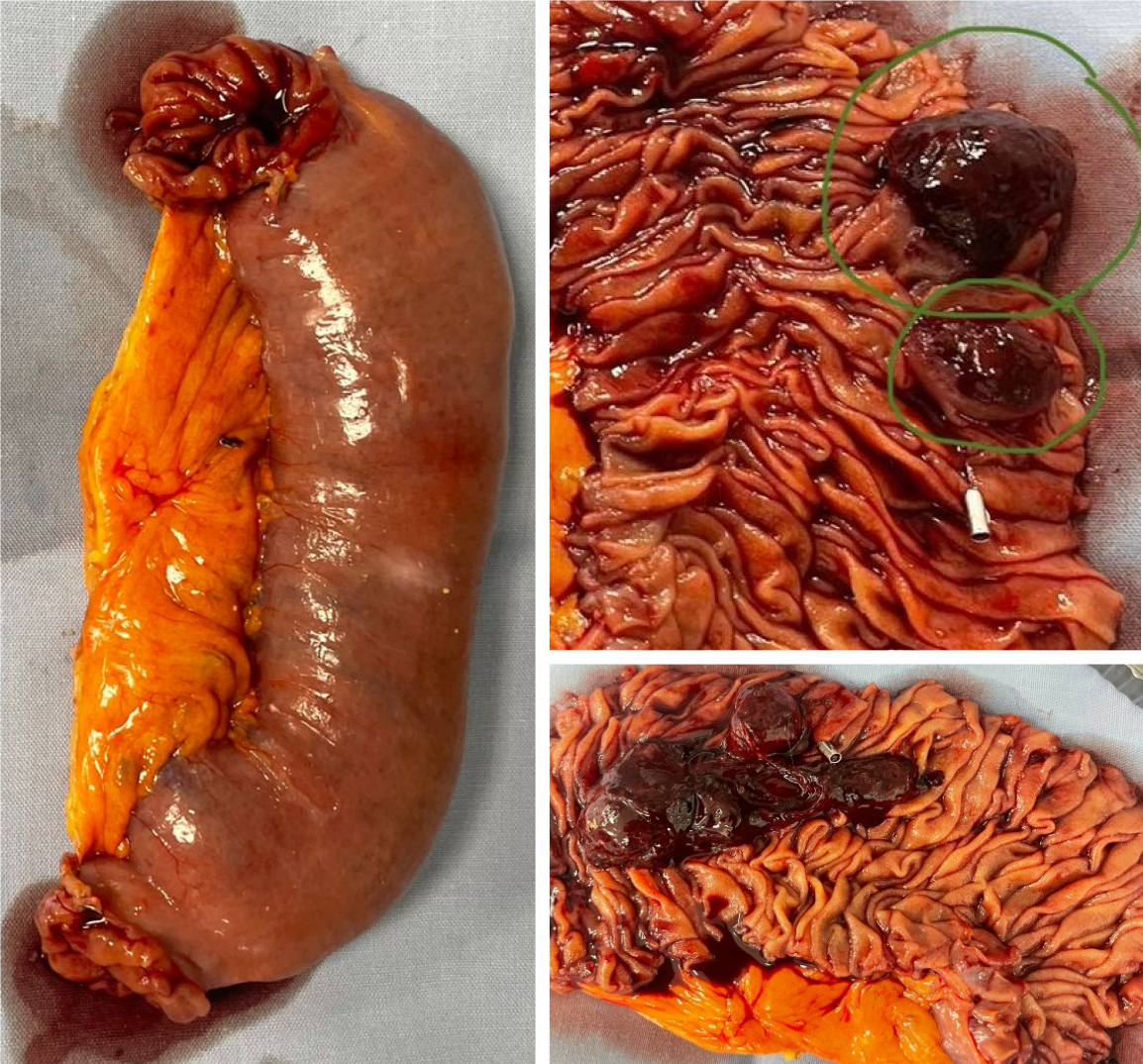
A median periumbilical laparotomy was therefore performed, allowing exploration of the abdominal cavity to identify two adjacent jejunal polyps. The surgeon proceeded with mesenteric resection of the affected jejunal loop and a latero-lateral jejuno-jejunostomy. Macroscopic examination of the resected specimen revealed two polyps in the jejunum, with clear surgical margins and no evidence of metastasis in the three lymph nodes identified (Figure 2). The patient was successfully stabilized postoperatively.
The macroscopic specimen consisted of a 12-cm segment of the intestine that had been longitudinally opened for examination. The serosal surface was smooth. The mucosa showed two polypoid lesions measuring 1.8 cm × 1.4 cm × 1.4 cm and 3 cm × 1.8 cm × 1.4 cm. Both had a similar hemorrhagic appearance with a beige rim at their base.
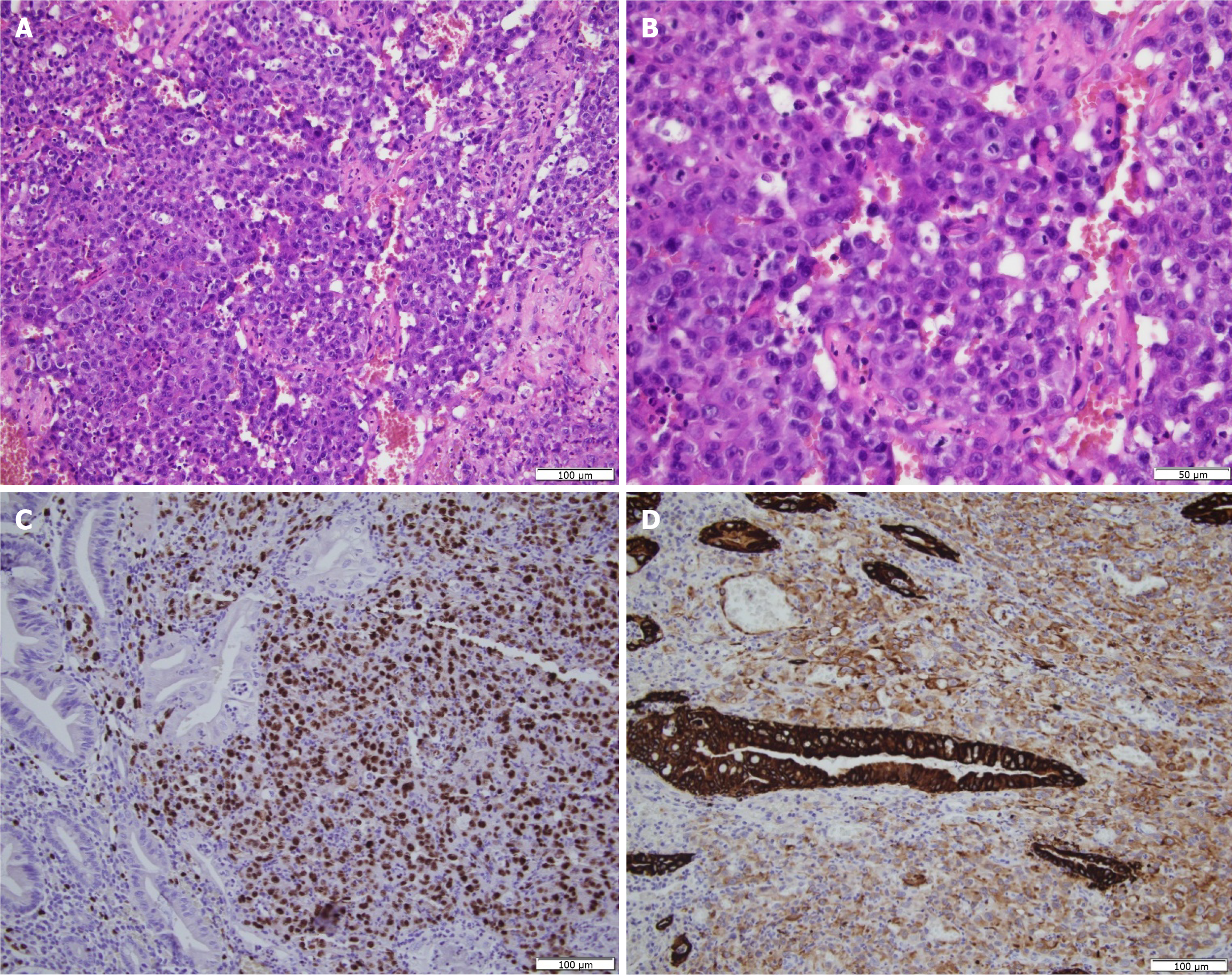
Histological examination of both polyps revealed similar features, consisting of an ulcerated tumor proliferation infiltrating the submucosa. The tumor was composed of sheets of large epithelioid cells, in areas forming slit-like, sometimes dilated vascular spaces containing papillary projections. The tumor cells were large, with prominent nucleoli and frequent mitotic figures. They were surrounded by extravasated red blood cells and showed neutrophilic exocytosis. Occasional tumor emboli were observed within peripheral lymphatic vessels. Surgical margins were free of tumor, and the three lymph nodes identified in the surrounding adipose tissue were negative for metastasis.
Immunohistochemically, the tumor cells showed strong expression of ERG and heterogeneous expression of cytoke
The endoscopic biopsy findings were also consistent with epithelioid angiosarcoma of the small intestine. Immunohistochemical analysis revealed that the tumor cells were positive for cytokeratin AE1/AE3 and ERG markers, showed overexpression of p53, and were negative for CMV and HHV8. The biopsy confirmed that the tumor proliferation invaded and ulcerated the mucosal surface. The complete clinical trajectory of the patient, from initial presentation to final diagnosis, is illustrated in Figure 4.
During the same admission, because the patient also presented with hemoptysis and the CT angiographic scan raised concern for possible lung metastases, a bronchoscopy was performed. The procedure was limited by a fibrinous clot obstructing the right lower bronchus. Pathological examination of the retrieved material showed only inflammatory changes, with no evidence of malignancy.
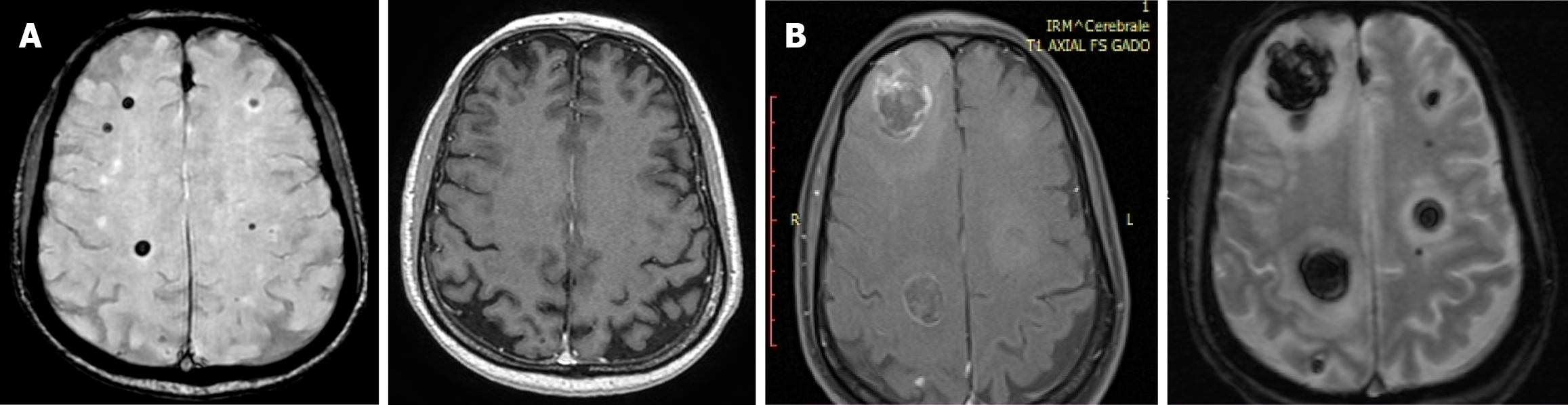
Neurologically, the patient remained asymptomatic. However, given the known tendency of epithelioid angiosarcoma to metastasize to the brain, brain magnetic resonance imaging (MRI) was performed on December 16, 2024, as part of the metastatic workup. The MRI revealed no acute stroke or intracranial hemorrhage but showed multiple small nodular lesions with susceptibility artifacts in both cerebral hemispheres and the cerebellum. Two right hemispheric lesions measured approximately 0.5 cm and 0.6 cm. There was no abnormal contrast enhancement, hydrocephalus, or extra-axial collection. These non-enhancing lesions were initially interpreted as cavernomas. Around this time, owing to loss of insurance coverage, the patient chose to transfer his care to another institution, resulting in a delay in initiating treatment.
Subsequently, following the development of neurological symptoms, a follow-up MRI on January 23, 2025, demonstrated significant interval progression compared to the previous one. The previously identified right hemispheric lesions had increased markedly in size, now measuring up to 2.5 cm and 2.0 cm, respectively. Despite their substantial growth, these lesions continued to exhibit only minimal contrast enhancement. The new findings, including numerous additional hemorrhagic intra-axial masses in both supratentorial and infratentorial regions, were highly suggestive of brain metastases. A slight midline shift to the left was observed, along with the presence of a cavum septum pellucidum cyst.
MRI studies from December 2024 and January 2025 are presented in Figure 5.
Following the diagnosis, the patient received 3D conformal radiotherapy with image-guided radiotherapy to the brain at a dose of 300 cGy per fraction, for a total dose of 3000 cGy, delivered from February 18, 2025, to March 4, 2025. The treatment was well tolerated. As a management plan, the regimen proposed consisted of weekly paclitaxel.
After completing the brain radiotherapy, the patient received approximately 4 months of weekly paclitaxel.
Follow-up thoracic CT angiography demonstrated disease progression, with widespread new nodular pulmonary lesions replacing the previously limited findings of a single left upper lobe lesion and a right upper lobe nodule. The peripheral calcified mass had enlarged to 12 cm × 6 cm, and new pleural involvement with nodular thickening was observed. Unfor
| Date | Event | Details/findings |
| January 2025 | Neurological deterioration | Brain MRI: Numerous new hemorrhagic intra-axial masses in supratentorial and infratentorial regions |
| February 2025 to March 2025 | Brain radiotherapy | 3D conformal IGRT was well-tolerated |
| March 2025 to January 2025 | Systemic therapy | Weekly paclitaxel, Bevacizumab was not added owing to the patient’s ongoing and recurrent bleeding risk |
| Follow-up | Disease progression | Thoracic CT scan: Extensive new pulmonary nodules; new pleural nodular involvement |
| Final decline | Clinical deterioration | Rapid thoracic disease progression despite ongoing management |
| Death | Cause of death | Uncontrolled thoracic hemorrhage before initiation of further therapy |
Intestinal angiosarcoma poses a diagnostic challenge because of its rare occurrence and non-specific clinical presentation. It should be suspected in patients presenting with gastrointestinal bleeding or anemia without signs of bleeding. Previous case reports have highlighted this challenge in the context of prior radiotherapy. Radiation can injure the gastrointestinal mucosa, leading to ulceration, friability, and chronic inflammation, which may cause slow, occult bleeding without visible clinical signs[6,7].
Anemia should prompt a thorough investigation and should not be automatically attributed to patients’ known underlying conditions without excluding other potential causes. A previous case of splenic angiosarcoma presenting with anemia mimicking myelofibrosis in the absence of underlying hematologic disease was reported and was associated with a poor prognosis[8]. In our case, despite the patient’s history of CML and a negative FIT, we proceeded with further investigations to exclude gastrointestinal causes.
The FIT is primarily designed to detect bleeding from the lower gastrointestinal tract. It is highly specific and has high overall diagnostic accuracy for detecting colorectal cancer, but is less sensitive for upper gastrointestinal bleeding sources[9]. This limitation was evident in our case, where the patient had significant bleeding from an upper gastrointestinal tumor requiring multiple transfusions, yet FIT results remained repeatedly negative.
Our patient also presented with a challenging lesion location—a 2-cm ulcerated lesion that was missed on imaging. Given the inconclusive correlation between clinical findings and imaging, and in view of the video capsule results as well as the second gastroscopy, we proceeded with surgical exploration. Indeed, laparotomy should be considered when the lesion is not accessible and noninvasive investigations fail to provide a definitive diagnosis or treatment.
In a study of 82 cases of gastrointestinal angiosarcoma, 26 patients were diagnosed through exploratory laparotomy—similar to our patient—after imaging and endoscopic evaluations failed to detect the lesions[10]. This underscores a significant diagnostic challenge, as these tumors often evade detection by conventional radiologic and endoscopic methods. In many instances, patients present acutely with gastrointestinal hemorrhage, acute abdomen, or even bowel perforation, requiring emergency surgical intervention. Given the heterogeneity of symptoms and the potential for severe, life-threatening complications, early and accurate diagnosis is essential. Therefore, a high index of suspicion should be maintained, and angiosarcoma should be considered in patients presenting with unexplained anemia, particularly in the context of underlying hematologic malignancies. While preoperative biopsy poses risks, particularly bleeding and potential tumor seeding, these must be carefully weighed against the aggressive nature and high mortality associated with angiosarcoma. In select cases with strong clinical suspicion, proceeding with biopsy may be justified despite the inherent risks, as timely diagnosis can significantly influence management and outcomes.
Accurate diagnosis of angiosarcoma necessitates a multidisciplinary approach, integrating clinical presentation, imaging studies, and histopathological analysis[11]. Cytologically, angiosarcoma typically presents with atypical, pleo
The cytologic presentation of epithelioid angiosarcoma can be misleading, as sheets of epithelioid endothelial cells may mimic other malignancies such as carcinomas[13]. Our patient’s prior pleural effusions were initially interpreted as reactive. However, cytology showing vacuolated macrophages, minimal hemosiderin, and no malignancy may have represented an early, undetected stage of disease, and retrospective application of immunostains such as ERG and cytokeratin might have revealed diagnostic clues. Although subsequent imaging revealed nodules suspicious for metastasis, pathological examination of bronchial aspirates remained negative. Moreover, cytological analysis of ascitic fluid revealed mesothelial reactive cells, which likely contributed to the early stages of angiosarcoma being overlooked, as appropriate diagnostic markers for vascular tumors were not applied at that time. Therefore, to facilitate rapid cytological diagnosis, detailed immunocytochemical staining panels for angiosarcoma should be considered early in the diagnostic workup when clinically suspected.
Vascular tumors such as angiosarcoma often contain a significant amount of blood, complicating the collection of adequate tumor samples for diagnosis through small biopsies[14]. Surgical specimens should therefore be considered not only in urgent settings, such as to control bleeding or prevent perforation, but also because they may provide a more reliable diagnosis than preoperative biopsies.
Vascular anomalies encompass a broad spectrum, ranging from benign vascular tumors such as hemangiomas and pyogenic granulomas to borderline lesions such as hemangioendothelioma and Kaposi sarcoma, and malignant tumors such as angiosarcoma and epithelioid hemangioendothelioma. In our case, despite the use of advanced imaging modalities such as CT scans, brain MRI, and bronchoscopy, determining the precise nature of the lesion and establishing its malignancy proved challenging. This reflects a well-documented limitation in the imaging of vascular tumors: Radiologic features of benign lesions are often nonspecific and may overlap with those of more aggressive pathologies. Therefore, histopathological confirmation remains essential, especially in adult patients presenting with soft tissue vascular tumors.
A study by Marcelin et al[15] further supports this notion, demonstrating that MRI could not reliably distinguish between atypical vascular tumors and benign malformations. Consequently, many authors advocate that the evaluation of soft tissue vascular tumors should adhere to the International Society for the Study of Vascular Anomalies classification, with the 2018 update serving as the current reference standard[16].
A 2025 study investigated the cytotoxic effects of imatinib and dasatinib on human aortic endothelial cells and revealed significant biochemical alterations in endothelial cells exposed to these TKIs, suggesting that even at clinically relevant concentrations, TKIs can induce sublethal stress in vascular endothelial cells through oxidative stress-mediated inflammation[17]. These findings underscore the importance of considering the potential angiogenic effects of TKIs such as nilotinib when evaluating unexplained anemia and ambiguous imaging findings.
In vitro studies have demonstrated that nilotinib may promote a pro-angiogenic tumor environment by enhancing the expression of key angiogenic mediators. Specifically, tumor cells exposed to nilotinib exhibited significantly elevated levels of VEGF, particularly at lower doses[5]. This upregulation of VEGF, along with increased interleukin-6 mRNA expression, suggests that nilotinib can influence tumor biology by fostering a microenvironment conductive to vascular proliferation and growth. In other studies, nilotinib has been found to have antiangiogenic effects[18]. It has shown to upregulate pro-atherogenic adhesion molecules such as ICAM-1, E-selectin, and VCAM-1 on human endothelial cells. Additionally, it suppresses key endothelial functions, including proliferation, migration, and tube formation, and binds to a distinct profile of target kinases. As a result, imaging findings in patients treated with nilotinib may vary—some vascular tumors may become more conspicuous owing to altered vascular architecture, while others, as in our case, may be obscured or misinterpreted[19]. In our patient, the initial lesion was missed because the lobulated lung nodules appeared calcified, likely owing to changes induced by TKI therapy, leading to a false sense of benignity.
Therefore, it is crucial to repeat imaging over time, as the radiologic features of vascular tumors in patients receiving nilotinib may evolve, and lesions that were initially subtle or ambiguous may become more apparent with disease progression or treatment-induced changes.
Moreover, given this potential risk associated with TKI therapy, we reviewed the rationale for treatment discontinuation in CML. Most studies require a minimum of 3 years on TKI and at least 1 year of sustained deep molecular response. Approximately 50% of patients relapse after discontinuation, regardless of the TKI used[20]. Clinical and biological factors do not yet allow precise stratification of patients at risk of relapse[21]. In select cases, such as when ongoing therapy may obscure imaging findings and delay the diagnosis of other malignancies, the risk–benefit balance could shift in favor of discontinuing treatment.
The absence of contrast enhancement on the initial MRI raised the question of whether the lesions represented caver
A case report described cerebrovascular disease in a patient treated with nilotinib using vessel wall MRI, which demonstrated concentric vessel wall thickening and enhancement indicative of vasculopathy[22]. This highlights a diagnostic challenge, as angiosarcoma can show varying degrees of differentiation. A similar case reported a cerebral angiosarcoma with both well-differentiated and poorly differentiated regions, leading to a misdiagnosis of cavernous angioma in the brain[23].
Similar findings have been reported in patients treated with bevacizumab, an anti-VEGF monoclonal antibody, where brain metastases lacked typical contrast enhancement. This phenomenon has been attributed to the antiangiogenic properties of bevacizumab, which restore the integrity of the blood–brain barrier and reduce extravascular leakage of contrast agent. Nilotinib targets BCR–ABL and also affects VEGF signaling. Accordingly, the non-enhancing lesions observed on the first MRI and the minimal enhancement seen on subsequent MRI in our patient, which is atypical for brain metastases, may be attributed to this effect of nilotinib on the integrity of the blood–brain barrier[24].
Sarcomas may also present with extensive infiltration of tumor-associated macrophages (TAMs) in the brain meta
These theories behind the absence of contrast enhancement in both the initial intestinal lesion and the brain lesion, atypical for metastatic disease, emphasize the need for a high index of suspicion. Imaging alone can be misleading, and histological evaluation remains essential to check for poorly differentiated areas when assessing atypical vascular brain lesions.
This case is limited by its single-patient design and the lack of long-term follow-up owing to the patient’s death soon after treatment initiation. Its strength lies in the comprehensive clinical, radiologic, and pathologic correlation, which emphasizes the diagnostic challenges of intestinal epithelioid angiosarcoma in the setting of TKI therapy.
This case highlights the diagnostic challenge posed by intestinal epithelioid angiosarcoma, particularly in a patient with a background of CML receiving long-term TKI. The patient’s acute anemia was never dismissed and prompted a thorough investigation, despite negative findings on FIT, normal gastro-colonoscopy, and unremarkable initial imaging. Radiologically, the tumor evaded detection, likely owing to alterations in vascular architecture induced by TKIs, which can obscure or distort typical imaging features, such as the expected contrast enhancement of brain metastases. Cytologically, the diagnosis was challenging as the effusion samples lacked definitive malignant features, and the tumor’s location made it inaccessible by biopsy without surgery.
In the presence of these three layers of complexity—clinical, radiologic, and cytologic—clinicians should maintain a high index of suspicion in patients presenting with unexplained anemia, particularly those receiving TKI therapy, and angiosarcoma should be considered in the differential diagnosis. When conventional diagnostic modalities are inconclusive, a multidisciplinary approach incorporating repeat imaging, endoscopic evaluation, and early histopathologic confirmation should be considered, while also considering early surgical intervention when suspicion remains high. In such cases, reliance on biopsy alone may not be sufficient, and gross surgical resection specimens can provide a definitive diagnosis and prevent further diagnostic delays. Thus, maintaining close interaction of the clinical, radiologic, and cytologic pillars is key to the timely and accurate diagnosis of such rare and aggressive malignancies.
| 1. | Nai Q, Ansari M, Liu J, Razjouyan H, Pak S, Tian Y, Khan R, Broder A, Bagchi A, Iyer V, Hamouda D, Islam M, Sen S, Yousif A, Hu M, Lou Y, Duhl J. Primary Small Intestinal Angiosarcoma: Epidemiology, Diagnosis and Treatment. J Clin Med Res. 2018;10:294-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 2. | Al Ali J, Ko HH, Owen D, Steinbrecher UP. Epithelioid angiosarcoma of the small bowel. Gastrointest Endosc. 2006;64:1018-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 3. | Allison KH, Yoder BJ, Bronner MP, Goldblum JR, Rubin BP. Angiosarcoma involving the gastrointestinal tract: a series of primary and metastatic cases. Am J Surg Pathol. 2004;28:298-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 84] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 4. | Eden RE, Coviello JM. Chronic Myelogenous Leukemia. 2023 Jan 16. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 5. | Zafarnia S, Bzyl-Ibach J, Spivak I, Li Y, Koletnik S, Doleschel D, Rix A, Pochon S, Tardy I, Koyadan S, van Zandvoort M, Palmowski M, Kiessling F, Lederle W. Nilotinib Enhances Tumor Angiogenesis and Counteracts VEGFR2 Blockade in an Orthotopic Breast Cancer Xenograft Model with Desmoplastic Response. Neoplasia. 2017;19:896-907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 6. | Parada Domínguez D, Peña González KB. Radiotherapy-associated intra-abdominal angiosarcoma after prostatic adenocarcinoma: Case reports. Mol Clin Oncol. 2018;9:592-596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Aitola P, Poutiainen A, Nordback I. Small-bowel angiosarcoma after pelvic irradiation: a report of two cases. Int J Colorectal Dis. 1999;14:308-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 8. | Wu M, Li Z, Luo L, Zhao W, Luo J. Metastatic splenic angiosarcoma presenting with anemia and bone marrow fibrosis mimicking primary myelofibrosis: A case report and literature review. Mol Clin Oncol. 2024;20:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160:171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 583] [Cited by in RCA: 527] [Article Influence: 43.9] [Reference Citation Analysis (3)] |
| 10. | Ma XM, Yang BS, Yang Y, Wu GZ, Li YW, Yu X, Ma XL, Wang YP, Hou XD, Guo QH. Small intestinal angiosarcoma on clinical presentation, diagnosis, management and prognosis: A case report and review of the literature. World J Gastroenterol. 2023;29:561-578. [PubMed] [DOI] [Full Text] |
| 11. | Young RJ, Brown NJ, Reed MW, Hughes D, Woll PJ. Angiosarcoma. Lancet Oncol. 2010;11:983-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 835] [Cited by in RCA: 703] [Article Influence: 43.9] [Reference Citation Analysis (0)] |
| 12. | Hart J, Mandavilli S. Epithelioid angiosarcoma: a brief diagnostic review and differential diagnosis. Arch Pathol Lab Med. 2011;135:268-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 13. | Mori T, Mizuguchi K, Shimaguchi C, Sakano K, Shimoda T, Okawa U, Okuda M, Usui M, Ikeda H. A Case of Epithelioid Angiosarcoma Diagnosed From Gross Examination of a Pulmonary Tumor Utilizing Imprint Cytology and Immunocytochemistry. Cancer Rep (Hoboken). 2024;7:e70014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Tabish N, Monaco SE. Epithelioid Vascular Lesions: The Differential Diagnosis and Approach in Cytology and Small Biopsies. Adv Anat Pathol. 2022;29:389-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Marcelin C, Dubois J, Kokta V, Giroux MF, Danino MA, Mottard S, Soulez G. Soft tissue vascular tumor-like lesions in adults: imaging and pathological analysis pitfalls per ISSVA classification. Insights Imaging. 2024;15:135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Wassef M, Borsik M, Cerceau P, Faucon B, Laurian C, Le Clerc N, Lemarchand-Venencie F, Massoni C, Salvan D, Bisdorff-Bresson A. [Classification of vascular tumours and vascular malformations. Contribution of the ISSVA 2014/2018 classification]. Ann Pathol. 2021;41:58-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 17. | Orleanska J, Krol W, Majzner K. Assessing endothelial cytotoxicity induced by tyrosine kinase inhibitors: insights from Raman and fluorescence imaging. Analyst. 2025;150:527-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Emir H, Albrecht-Schgoer K, Huber K, Grebien F, Eisenwort G, Schgoer W, Kaun C, Herndlhofer S, Theurl M, Cerny-Reiterer S, Hoermann G, Sperr WR, Uwe R, Wojta J, Wolf D, Superti-Furga G, Kirchmair R, Valent P. Nilotinib Exerts Direct Pro-Atherogenic and Anti-Angiogenic Effects On Vascular Endothelial Cells: A Potential Explanation For Drug-Induced Vasculopathy In CML. Blood. 2013;122:257. [RCA] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 19. | Hadzijusufovic E, Albrecht-Schgoer K, Huber K, Hoermann G, Grebien F, Eisenwort G, Schgoer W, Herndlhofer S, Kaun C, Theurl M, Sperr WR, Rix U, Sadovnik I, Jilma B, Schernthaner GH, Wojta J, Wolf D, Superti-Furga G, Kirchmair R, Valent P. Nilotinib-induced vasculopathy: identification of vascular endothelial cells as a primary target site. Leukemia. 2017;31:2388-2397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 110] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 20. | Saussele S, Richter J, Guilhot J, Gruber FX, Hjorth-Hansen H, Almeida A, Janssen JJWM, Mayer J, Koskenvesa P, Panayiotidis P, Olsson-Strömberg U, Martinez-Lopez J, Rousselot P, Vestergaard H, Ehrencrona H, Kairisto V, Machová Poláková K, Müller MC, Mustjoki S, Berger MG, Fabarius A, Hofmann WK, Hochhaus A, Pfirrmann M, Mahon FX; EURO-SKI investigators. Discontinuation of tyrosine kinase inhibitor therapy in chronic myeloid leukaemia (EURO-SKI): a prespecified interim analysis of a prospective, multicentre, non-randomised, trial. Lancet Oncol. 2018;19:747-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 529] [Cited by in RCA: 461] [Article Influence: 57.6] [Reference Citation Analysis (0)] |
| 21. | Inzoli E, Aroldi A, Piazza R, Gambacorti-Passerini C. Tyrosine Kinase Inhibitor discontinuation in Chronic Myeloid Leukemia: eligibility criteria and predictors of success. Am J Hematol. 2022;97:1075-1085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 22. | Suzuki K, Yamamoto J, Kakeda S, Takamatsu S, Miyaoka R, Kitagawa T, Saito T, Nakano Y, Nishizawa S. Vessel wall magnetic resonance imaging findings and surgical treatment in nilotinib-associated cerebrovascular disease: A case report. Mol Clin Oncol. 2019;10:239-243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Matsuno A, Nagashima T, Tajima Y, Sugano I. A diagnostic pitfall: Angiosarcoma of the brain mimicking cavernous angioma. J Clin Neurosci. 2005;12:688-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Karimi S, Lis E, Gilani S, D'Ambrosio N, Holodny A. Nonenhancing brain metastases. J Neuroimaging. 2011;21:184-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Klemm F, Maas RR, Bowman RL, Kornete M, Soukup K, Nassiri S, Brouland JP, Iacobuzio-Donahue CA, Brennan C, Tabar V, Gutin PH, Daniel RT, Hegi ME, Joyce JA. Interrogation of the Microenvironmental Landscape in Brain Tumors Reveals Disease-Specific Alterations of Immune Cells. Cell. 2020;181:1643-1660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 901] [Cited by in RCA: 782] [Article Influence: 130.3] [Reference Citation Analysis (0)] |
| 26. | Schulz M, Sevenich L. TAMs in Brain Metastasis: Molecular Signatures in Mouse and Man. Front Immunol. 2021;12:716504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Zhu C, Kros JM, Cheng C, Mustafa D. The contribution of tumor-associated macrophages in glioma neo-angiogenesis and implications for anti-angiogenic strategies. Neuro Oncol. 2017;19:1435-1446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 139] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 28. | Rogawski D, Wheeler J, Nie E, Zhu W, Villanueva E, Coffey G, Ma Q, Ganjoo K, Fischbein N, Iv M, Vogel H, Nagpal S. A rare non-gadolinium enhancing sarcoma brain metastasis with microenvironment dominated by tumor-associated macrophages. Acta Neuropathol Commun. 2024;12:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |