Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116104
Revised: November 19, 2025
Accepted: January 4, 2026
Published online: April 15, 2026
Processing time: 157 Days and 3.8 Hours
Hepatocellular carcinoma (HCC) remains one of the leading causes of cancer-related deaths, with high recurrence and metastasis rates after treatment. Incom
To investigate whether miR-206 regulates the HIF-1α/PFKFB3/glycolysis axis in the recurrence and metastasis of HCC following iRFA.
A clinical study was conducted in 45 patients with HCC undergoing RFA, compa
Clinically, iRFA was associated with increased HIF-1α and pyruvate, decreased glucose, and altered miR-206 levels compared with complete ablation. In Td-ECs, thermal stimulation enhanced proliferation, migration, glycolysis, and HIF-1α/PFKFB3 expression, while miR-206 overexpression significantly attenuated these effects. Dual-luciferase assays confirmed that miR-206 directly binds the 3′UTR of HIF-1α. In animal models, incomplete ablation increased microvessel density, α-SMA, HIF-1α, and PFKFB3, while miR-206 expression was reduced.
miR-206 suppresses HIF-1α-driven PFKFB3-mediated glycolysis, thereby limiting angiogenesis, cell migration, and recurrence after iRFA. These findings suggest that miR-206 is a potential therapeutic target to reduce HCC re
Core Tip: Incomplete radiofrequency ablation (iRFA) creates a hypoxic microenvironment that drives recurrence of hepatocellular carcinoma. This study reveals that microRNA-206 (miR-206) directly targets hypoxia-inducible factor-1α and suppresses 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3-mediated glycolysis, thereby reducing angiogenesis, migration, and metastasis after iRFA. Using patient samples, endothelial cell models, and a rabbit VX2 tumor model, we demonstrate that restoring miR-206 expression counteracts post-ablation metabolic reprogramming. These findings highlight miR-206 as a promising therapeutic target to prevent recurrence and improve long-term outcomes after local ablation therapy in hepatocellular carcinoma.
- Citation: Lu D, Wang LJ, Chai J, Jiang J, Tang YL. MicroRNA-206 suppresses hypoxia-inducible factor-1α/PFKFB3-mediated glycolysis to inhibit recurrence and metastasis of hepatocellular carcinoma after incomplete radiofrequency ablation. World J Gastrointest Oncol 2026; 18(4): 116104
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116104.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116104
Hepatocellular carcinoma (HCC) is one of the most common malignancies worldwide and remains a leading cause of cancer-related deaths[1-3]. Despite advances in surgical and interventional therapies, recurrence and metastasis continue to limit long-term survival[4]. Radiofrequency ablation (RFA) is widely used as a minimally invasive therapy for small HCC, with outcomes comparable to those of surgical resection in carefully selected patients[5,6]. However, incomplete RFA (iRFA) remains a major clinical challenge, particularly for tumors larger than 3 cm, where residual tumor tissue can promote local recurrence and distant metastasis. Understanding the molecular mechanisms that drive recurrence after iRFA is essential for improving treatment outcomes.
iRFA produces a hypoxic microenvironment in residual tumor tissue[7]. Hypoxia-inducible factor-1α (HIF-1α), a master regulator of cellular adaptation to hypoxia, is frequently upregulated under these conditions and promotes tumor progression[8,9]. One of its key downstream targets is 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3), a glycolytic enzyme that enhances glucose metabolism, endothelial sprouting, and angiogenesis[10-13]. Activation of the HIF-1α/PFKFB3 axis has been linked to increased tumor proliferation, migration, and invasion, suggesting a central role in post-ablation recurrence[14,15].
Recent studies identify microRNA-206 (miR-206) as a tumor suppressor that regulates proliferation, migration, and metabolic activity in several cancers, partly through modulation of HIF-1α-regulated pathways[16]. Computational predictions and experimental data suggest that HIF-1α may be a direct target of miR-206, raising the possibility that miR-206 could suppress tumor recurrence after iRFA by downregulating HIF-1α and its downstream glycolytic signaling.
Although hypoxia-driven metabolic reprogramming has been implicated in HCC recurrence after iRFA, the role of miR-206 in regulating this process has not been fully defined. In particular, whether miR-206 directly targets HIF-1α, modulates PFKFB3-dependent glycolysis, and influences tumor angiogenesis and metastasis in the setting of iRFA remains unclear. In this study, we investigated the function of miR-206 in regulating the HIF-1α/PFKFB3/glycolysis pathway. Using patient samples, cell-based assays, and a rabbit VX2 tumor model, we evaluated whether miR-206 suppresses glycolysis-driven tumor progression after iRFA. Our findings provide new insights into the molecular mechanisms of HCC recurrence and suggest that miR-206 may represent a novel therapeutic target for preventing disease progression.
A total of 45 patients with HCC who underwent RFA were prospectively recruited from the First Hospital of the University of Science and Technology of China between January 2019 and December 2021. Inclusion criteria were: (1) Age 18-75 years; (2) A diagnosis of HCC confirmed by imaging and/or pathology; and (3) Eligibility for RFA according to international guidelines. Exclusion criteria included prior systemic therapy, concurrent malignancies, and incomplete clinical records. Of these patients, 25 were classified as tumor-node-metastasis stage II and 20 as stage III. Computed tomography (CT) and magnetic resonance imaging (MRI) examinations conducted at the 1-month postoperative follow-up showed that 35 patients achieved complete ablation, while 10 patients showed imaging evidence of residual enhan
Serum samples were collected preoperatively and at 1 week postoperatively. Levels of HIF-1α, miR-206, glucose, and pyruvate were quantified using ELISA kits (Beyotime, Shanghai, China), qRT-PCR, and biochemical assays, as described below. A flowchart summarizing patient enrollment, grouping, and sample collection timepoints is provided in Figure 1 to facilitate understanding of the clinical study design. Written informed consent was obtained from all patients before enrolment in the study. The study was approved by the institutional ethics committee.
RFA was performed using a monopolar electrode ( Rita Medical Systems, AngioDynamics, Latham, New York, United States) under ultrasound guidance. Ablation power was set at 60-80 W for 6-12 min, depending on tumor size. The needle tip temperature was continuously monitored (maintained at 90 °C ± 5 °C). Complete ablation was defined as a necrotic area extending at least 0.5-1.0 cm beyond the tumor margin. Incomplete ablation was defined as a necrotic margin <0.5 cm or the presence of residual enhancement on follow-up imaging.
A total of 54 healthy New Zealand white rabbits (male or female, weight: 2.0-2.5 kg) and 1 VX2 tumor-bearing donor rabbit were used in this study. The 54 experimental rabbits were randomly allocated into three groups of 18 animals each, corresponding to sham treatment (Group A), complete RFA (Group B), and iRFA (Group C). Complete ablation was defined as an ablation zone extending 0.5-1.0 cm beyond the tumor margin on contrast-enhanced CT or MRI performed 1 month after RFA. Incomplete ablation was defined as an ablation margin less than 0.5 cm or persistent contrast enhancement on the 1-month follow-up imaging. A sham group was included to provide a baseline for assessing thermal injury independent of tumor destruction, although such a control group does not exist in clinical practice. Each group contained 18 rabbits, and the animals were sacrificed at three prespecified timepoints: Immediately after treatment (0 week), at 1 week, and at 3 weeks. At each timepoint, six rabbits per group were euthanized for tissue collection and analysis.
One VX2 tumor-bearing rabbit served solely as the donor for tumor fragments and was not included in any experimental group. The VX2 donor rabbit was anesthetized with sodium pentobarbital (100 mg/kg, intravenous), and the hind-limb subcutaneous VX2 tumor was surgically excised. Necrotic tissue and fascia were removed, and viable, fish-flesh-like tumor tissue from the actively growing edge was cut into approximately 2-mm fragments and placed in sterile saline containing gentamicin (20000 units/50 mL).
For implantation, each experimental rabbit was anesthetized and placed in a supine position. A 2-cm midline subxiphoid incision was made to expose the left medial hepatic lobe. A 4F percutaneous hepatic puncture sheath was inserted obliquely through the liver capsule to a depth of about 5 mm, and 3-5 tumor fragments (about 2 mm each) were gently pushed into the liver parenchyma using the inner stylet. The puncture tract was sealed with a 2 mm × 2 mm × 5 mm gelatin sponge to prevent bleeding. The liver was returned to the abdominal cavity, and the incision was closed in layers.
Tumor formation was confirmed 2 weeks after implantation by contrast-enhanced CT, and only rabbits with successful tumor establishment were included in the subsequent experimental procedures. After successful tumor establishment, rabbits were randomized into three groups (n = 18 per group): (1) Group A (sham): Needle insertion without ablation; (2) Group B (complete RFA): Ablation margin ≥0.5-1.0 cm beyond the tumor; and (3) Group C (iRFA): Partial ablation leaving residual viable tumor.
Each group was further subdivided so that six animals were sacrificed at each of the three timepoints (0, 1, and 3 weeks). RFA was performed using the same system and settings as in the clinical protocol. Tumor and adjacent liver tissues were harvested for histology, immunohistochemistry, and biochemical analyses. All procedures followed NIH guidelines for the care and use of laboratory animals (NIH publication No. 85-23, revised 1985).
Human umbilical vein endothelial cells (Chinese Academy of Sciences, Shanghai, China) were cultured in DMEM supplemented with 20% FBS, 10 ng/mL VEGF, and antibiotics. Human HCC HepG2 cells (ATCC, United States) were cultured in serum-free RPMI-1640 for 48 h. Conditioned medium from HepG2 cells was then mixed in a 1:1 ratio with endothelial cell medium to induce differentiation into tumor-derived endothelial cells (Td-ECs). Expression of TEM1 and TEM8 confirmed successful induction[16].
Td-ECs were exposed to thermal stimulation in a water bath at 37 °C (control), 41 °C, 44 °C, or 47 °C for 10 min to simulate RFA conditions. Functional assays were performed at 12, 24, 48, and 72 h after stimulation. To model the sublethal heat exposure occurring in the periablation zone during RFA, we selected temperatures of 41 °C, 44 °C, and 47 °C based on published thermal distribution data. Clinical and experimental studies show that the central ablation core reaches 60 °C-100 °C, while the surrounding viable zone typically remains within 40 °C-50 °C, which promotes endothelial activation, glycolytic reprogramming, and angiogenic signaling. Temperatures around 41 °C represent mild sublethal stress, whereas temperatures of 44 °C-47 °C reflect the upper physiologic threshold before irreversible injury. Therefore, this gradient reproduces the biologically relevant thermal conditions associated with incomplete ablation.
PFKFB3 knockdown: Three siRNA sequences were designed and screened; the most effective sequence (5′-GCUCC
HEK293 cells were co-transfected with wild-type or mutant HIF-1α 3′UTR constructs and miR-206 mimics or controls. Luciferase activity was measured 48 h later using the Dual-Luciferase Reporter Assay System (Promega, Madison, Wisconsin, United States).
Cell proliferation was assessed with the CCK-8 assay (MCE, New York, United States). Migration and invasion were analyzed using wound-healing and Transwell assays. Apoptosis was measured by TUNEL staining. Tube formation was assessed on Matrigel-coated plates. Glucose uptake and pyruvate production were measured using commercial kits (Nanjing Jiancheng, Nanjing, Jiangsu, China).
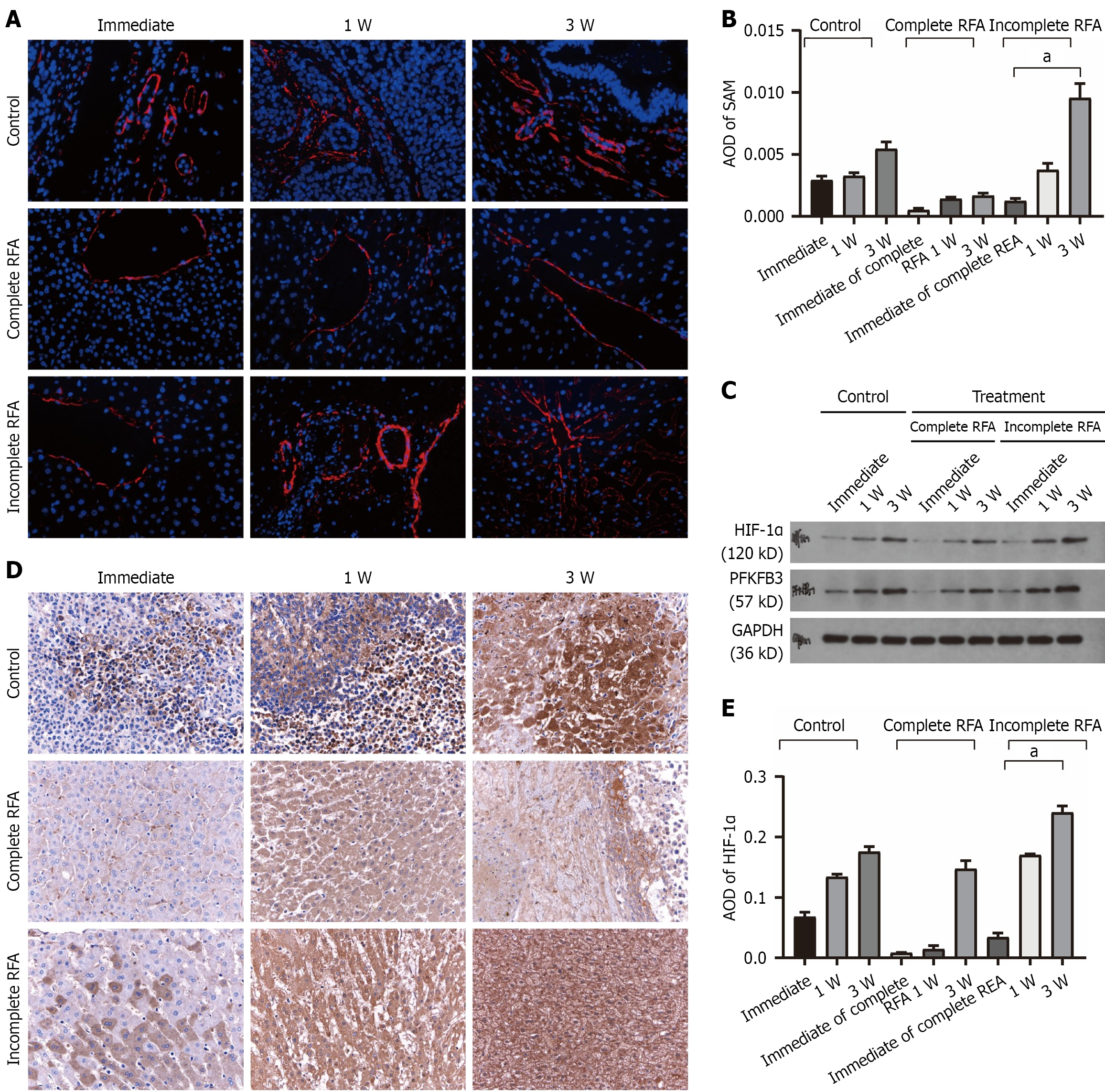
Tumor sections were stained for α-SMA and CD34 to assess angiogenesis. Microvessel density was quantified in five random high-power fields. HIF-1α, PFKFB3, and GAPDH expression were analyzed by western blot and immunohistochemistry. miR-206 levels were measured by qRT-PCR.
All experiments were independently repeated at least three times. Data are expressed as mean ± SD. Comparisons between two groups were performed using Student’s t-test, and comparisons among multiple groups were conducted using one-way ANOVA followed by Tukey’s post hoc test. Survival analysis in patients was performed using Kaplan-Meier curves and log-rank tests. Cox proportional hazards regression was used to identify independent prognostic factors. A two-sided P < 0.05 was considered statistically significant.
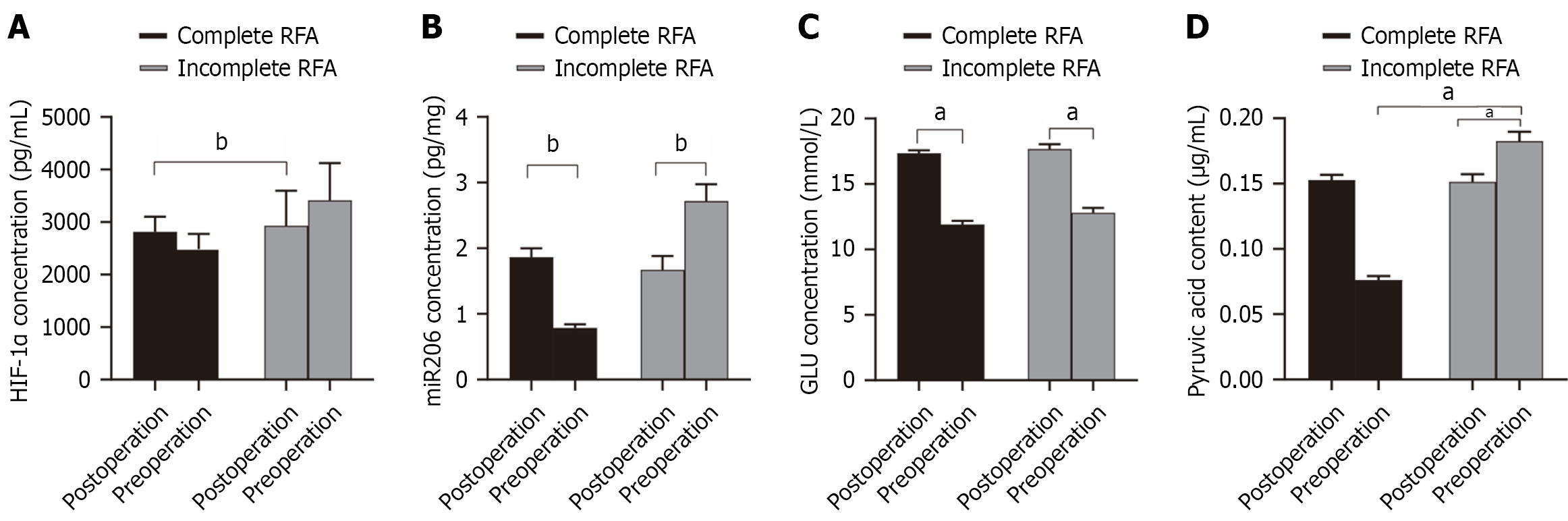
Among 45 patients with HCC undergoing RFA, 35 achieved complete ablation, and 10 developed local recurrence or metastasis owing to incomplete ablation. Postoperative serum HIF-1α levels were significantly higher in the incomplete ablation group compared with the complete ablation group (Figure 2A). Serum miR-206 expression was significantly lower in the incomplete ablation group after RFA (Figure 2B). Serum glucose decreased and pyruvate increased after RFA, with more pronounced changes in the incomplete group (Figures 2C and 2D). These data suggest that iRFA is associated with reduced miR-206 expression, enhanced HIF-1α/PFKFB3 signaling, and increased glycolytic activity.
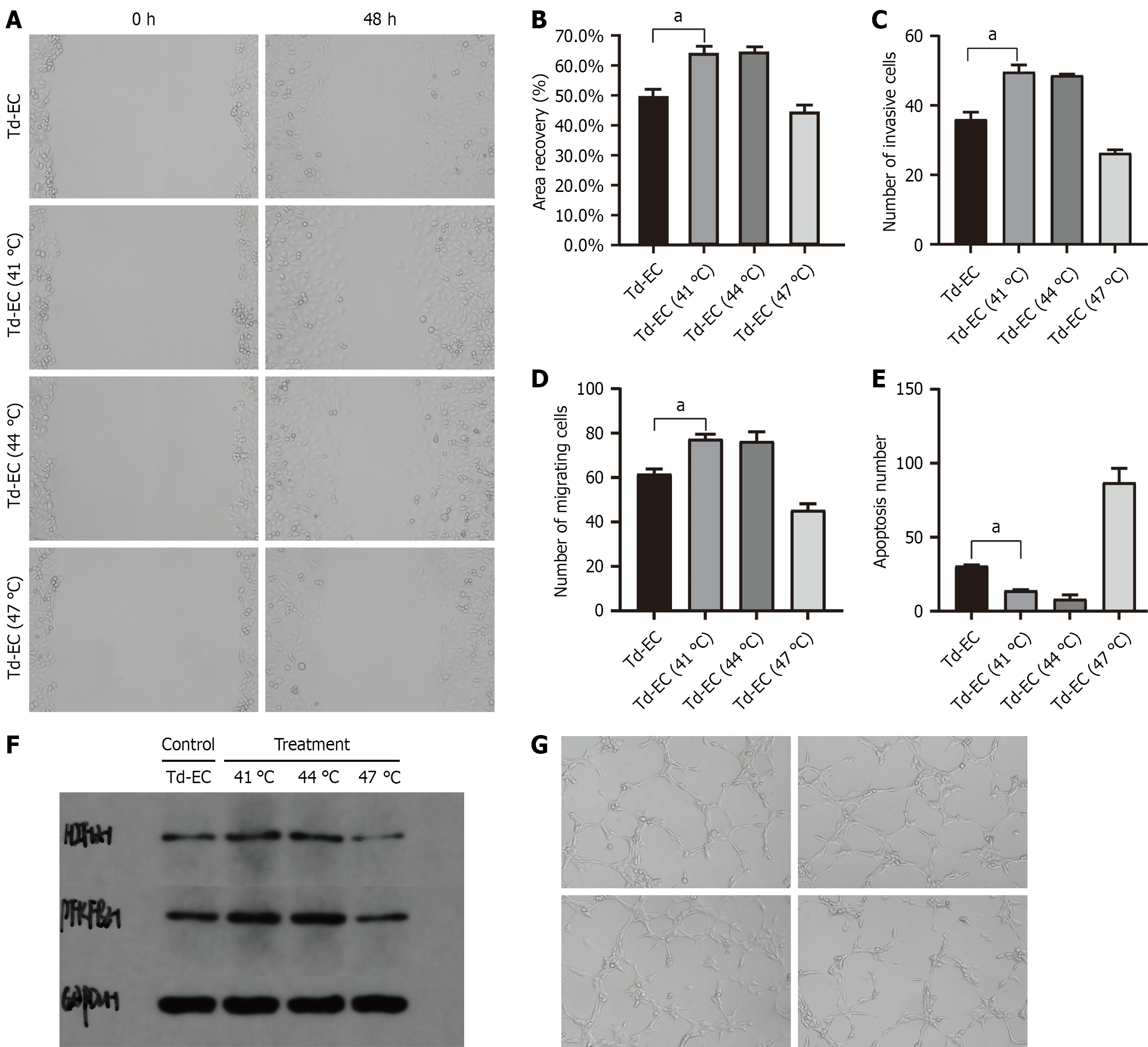
Td-ECs exposed to 41 °C exhibited enhanced proliferation (Figure 3A), migration, invasion (Figures 3B and 3C), and tube formation compared with control cells at 37 °C (all P < 0.05; Figure 3D). These cells also showed increased glucose uptake and pyruvate production and higher expression of HIF-1α and PFKFB3 by western blot (Figure 3E). At higher temperatures (44 °C-47 °C), cell viability decreased and apoptosis increased (Figures 3E and 3F), consistent with cytotoxic stress (Figure 3G).
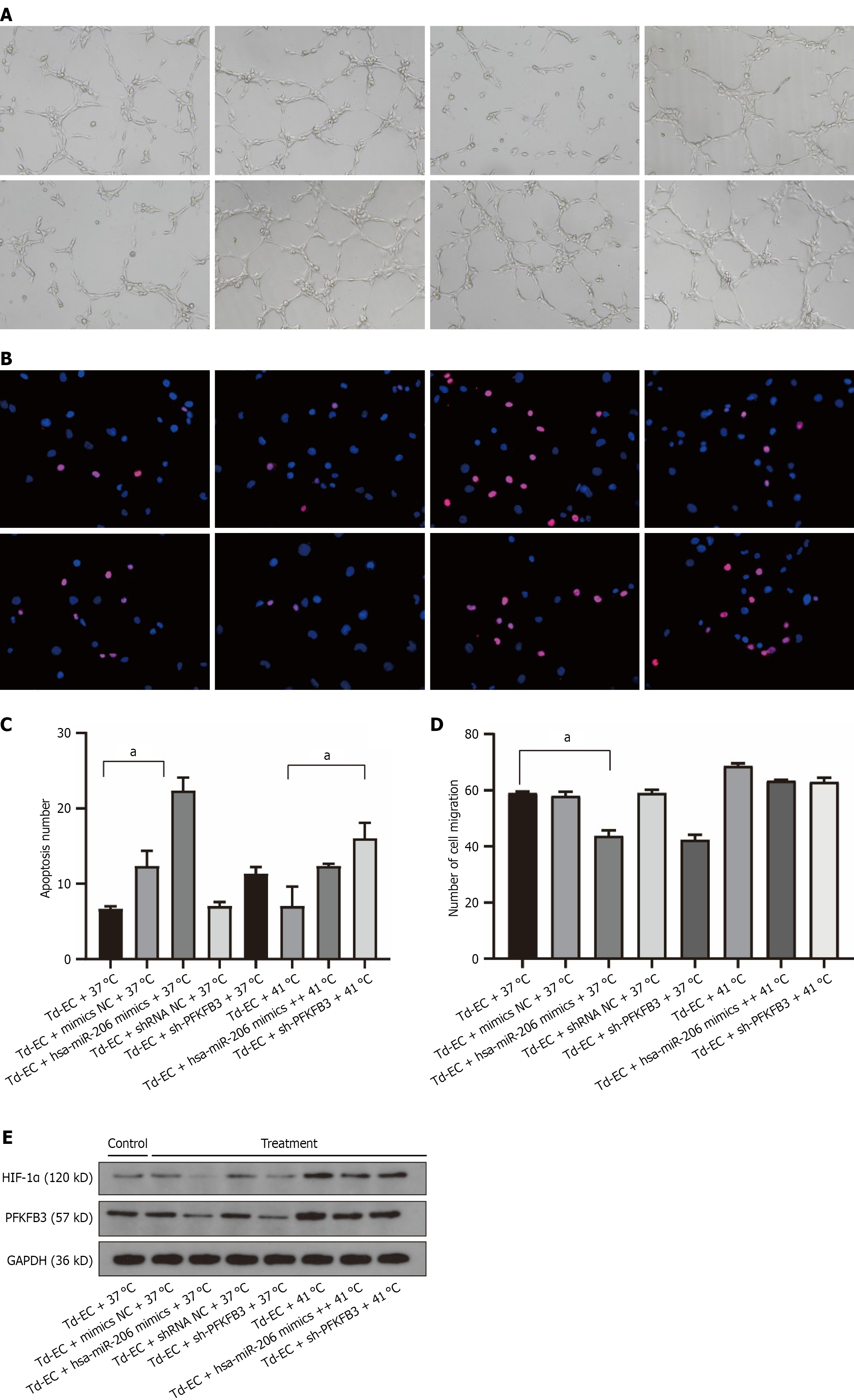
Dual-luciferase reporter assays confirmed that miR-206 binds the 3′UTR of HIF-1α, significantly reducing luciferase activity compared with the mutant construct (P < 0.01, data not shown). In Td-ECs, transfection with miR-206 mimics downregulated HIF-1α and PFKFB3 protein and mRNA expression, suppressed tube formation (Figure 4A), induced apoptosis (Figures 4B and 4C), and reduced migration (Figures 4D and 4E). Conversely, inhibition of miR-206 produced the opposite effects, confirming its regulatory role.
PFKFB3 silencing using shRNA markedly reduced glycolysis and endothelial proliferation, migration, and angiogenic capacity under thermal stimulation (Figure 4). The effects of PFKFB3 knockdown closely resembled those of miR-206 overexpression, indicating that PFKFB3 is a functional mediator of the miR-206/HIF-1α axis.
To further validate the mechanism, rescue assays were performed. Co-overexpression of HIF-1α with miR-206 mimics partially restored glycolysis, migration, and tube formation in Td-ECs (Figure 3). Similarly, co-overexpression of PFKFB3 reversed the suppressive effects of miR-206 on cell phenotypes. These results confirm that miR-206 acts upstream of HIF-1α and PFKFB3 in regulating glycolysis and tumor-promoting phenotypes.
In the rabbit VX2 model, incomplete ablation resulted in significantly increased α-SMA expression (Figure 5A), higher microvessel density (Figure 5B), and greater glycolytic activity (glucose and pyruvate changes, not shown) compared with complete ablation at 3 weeks (P < 0.01; Figures 5C and 5D). Western blotting and immunohistochemistry demonstrated elevated HIF-1α and PFKFB3 expressions in incompletely ablated tumors (Figure 5E). In contrast, miR-206 expression was significantly lower in incompletely ablated tissues compared with completely ablated tissues, supporting its inverse relationship with HIF-1α/PFKFB3 activation. These animal groups were selected to mirror the clinical categories used in patient analyses, allowing direct comparison of molecular and functional changes between complete and incomplete ablation conditions.
In this study, we investigated the role of miR-206 in the recurrence and metastasis of HCC following iRFA. Our findings highlight miR-206 as a crucial regulator of the HIF-1α/PFKFB3 axis, which controls glycolysis and tumor progression after iRFA. Specifically, miR-206 downregulated HIF-1α expression, suppressed PFKFB3-mediated glycolysis, and inhibited tumor cell migration and angiogenesis in both in vitro and animal models. These results suggest that miR-206 plays a protective role in preventing recurrence and metastasis of HCC after incomplete ablation, offering a potential therapeutic target for improving patient outcomes in this context.
miR-206 has emerged as a tumor suppressor in various cancers, including HCC, where it is known to regulate cell cycle progression, apoptosis, and metastasis[16-20]. Our study supports these findings by showing that miR-206 directly targets HIF-1α, inhibiting the HIF-1α/PFKFB3 glycolytic axis, which is critical for promoting aggressive tumor pheno
Our findings are consistent with previous studies implicating the HIF-1α/PFKFB3 axis in promoting cancer progression through glycolytic reprogramming[21-25]. HIF-1α is well established as a key mediator of the cellular response to hypoxia, and its activation is known to drive glycolysis, angiogenesis, and metastasis in various cancers, including HCC[14,26-29]. PFKFB3, a critical regulator of glycolysis, has also been shown to support endothelial cell proliferation and migration in the context of tumor angiogenesis[11,24-27]. However, the role of miR-206 in regulating this pathway in HCC, particularly after iRFA, has not been previously reported[28-31]. Our study is the first to demonstrate that miR-206 directly targets HIF-1α, thereby suppressing glycolysis and tumor progression after thermal ablation. This novel mechanism provides new insights into how miR-206 could be harnessed as a therapeutic strategy to prevent recurrence and metastasis of HCC following incomplete ablation.
One strength of this study is its multitiered design, which combines clinical data, in vitro experiments, and an animal model to examine the role of miR-206 in HCC recurrence after incomplete ablation. By integrating molecular mechanisms with functional outcomes, we provide a more comprehensive understanding of how miR-206 may influence tumor progression. The consistency of findings across multiple experimental systems also strengthens the validity and relevance of our results[32-35].
Our study has several limitations. First, although we enrolled 45 patients, the sample size was modest, particularly in the incomplete ablation group (n = 10). Larger studies with multicenter cohorts are needed to validate these findings and determine the clinical value of miR-206 as a prognostic biomarker. Second, we did not perform gradient ablation experiments to define the exact thermal threshold at which incomplete ablation activates the HIF-1α/PFKFB3 pathway. Such work may help establish clearer criteria for iRFA failure. Third, although both in vitro and in vivo assays were conducted, the role of miR-206 in other HCC subtypes or in response to other ablative therapies remains uncertain. In addition, long-term follow-up data were not available to evaluate the durability of miR-206-related effects on recurrence.
Although our in vitro data and animal findings consistently support the involvement of the miR-206/HIF-1α/PFKFB3 axis in glycolytic activation after insufficient ablation, this mechanistic relationship has not been directly validated in vivo. We did not perform rescue experiments, such as delivering miR-206 mimics or inhibitors in the animal model, to determine whether altering this pathway reverses pro-angiogenic or pro-glycolytic changes. For this reason, the observed association between reduced miR-206 expression and activation of HIF-1α/PFKFB3 signaling after incomplete ablation should be interpreted as supportive rather than conclusive evidence of causality[36-40]. Future studies incorporating in vivo gain- and loss-of-function approaches will be essential to confirm this mechanistic link.
Future studies should incorporate in vivo gain- and loss-of-function experiments, such as treating tumor-bearing animals with miR-206 mimics or inhibitors, to directly confirm the causal role of this pathway in recurrence after RFA. Additional work is also needed to evaluate the therapeutic potential of miR-206, whether through direct miRNA replacement or by targeting upstream regulators that modulate its expression. Gradient ablation studies coupled with longitudinal sampling will be important for defining the thermal and temporal conditions under which miR-206 is most effective in limiting tumor progression. Finally, given the central role of metabolic reprogramming in cancer, future investigations should explore how other pathways, including mTOR and AMPK signaling, interact with miR-206 in regulating tumor metabolism and recurrence.
In conclusion, our study reveals a novel mechanism by which miR-206 regulates the HIF-1α/PFKFB3 glycolytic axis to inhibit tumor recurrence and metastasis following iRFA of HCC. These findings identify a potential therapeutic target for preventing recurrence and improving outcomes in patients with HCC after iRFA. Further validation in larger clinical cohorts and additional preclinical studies are required to translate these findings into clinical practice.
| 1. | Lu H, Gao L, Lv J. Circ_0078710 promotes the development of liver cancer by upregulating TXNDC5 via miR-431-5p. Ann Hepatol. 2022;27:100551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 2. | Ma C, Zhang Q, Greten TF. MDSCs in liver cancer: A critical tumor-promoting player and a potential therapeutic target. Cell Immunol. 2021;361:104295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 3. | Song L, Zhang JG, Zheng L, Feng X, Hou J, Zhang HL, Liu SF. Establishment of rat liver cancer cell lines with different metastatic potential. Sci Rep. 2020;10:8329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 4. | Chen TW, Yin FF, Yuan YM, Guan DX, Zhang E, Zhang FK, Jiang H, Ma N, Wang JJ, Ni QZ, Qiu L, Feng J, Zhang XL, Bao Y, Wang K, Cheng SQ, Wang XF, Wang X, Li JJ, Xie D. CHML promotes liver cancer metastasis by facilitating Rab14 recycle. Nat Commun. 2019;10:2510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 5. | Xia S, Li X, Xu S, Ni X, Zhan W, Zhou W. Sublethal heat treatment promotes breast cancer metastasis and its molecular mechanism revealed by quantitative proteomic analysis. Aging (Albany NY). 2022;14:1389-1406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Yap S, Ooi EH, Foo JJ, Ooi ET. Bipolar radiofrequency ablation treatment of liver cancer employing monopolar needles: A comprehensive investigation on the efficacy of time-based switching. Comput Biol Med. 2021;131:104273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Tong Y, Yang H, Xu X, Ruan J, Liang M, Wu J, Luo B. Effect of a hypoxic microenvironment after radiofrequency ablation on residual hepatocellular cell migration and invasion. Cancer Sci. 2017;108:753-762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 8. | Yamada S, Utsunomiya T, Morine Y, Imura S, Ikemoto T, Arakawa Y, Kanamoto M, Iwahashi S, Saito Y, Takasu C, Ishikawa D, Shimada M. Expressions of hypoxia-inducible factor-1 and epithelial cell adhesion molecule are linked with aggressive local recurrence of hepatocellular carcinoma after radiofrequency ablation therapy. Ann Surg Oncol. 2014;21 Suppl 3:S436-S442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Xu M, Zheng YL, Xie XY, Liang JY, Pan FS, Zheng SG, Lü MD. Sorafenib blocks the HIF-1α/VEGFA pathway, inhibits tumor invasion, and induces apoptosis in hepatoma cells. DNA Cell Biol. 2014;33:275-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 10. | Teuwen LA, Draoui N, Dubois C, Carmeliet P. Endothelial cell metabolism: an update anno 2017. Curr Opin Hematol. 2017;24:240-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 11. | Cruys B, Wong BW, Kuchnio A, Verdegem D, Cantelmo AR, Conradi LC, Vandekeere S, Bouché A, Cornelissen I, Vinckier S, Merks RM, Dejana E, Gerhardt H, Dewerchin M, Bentley K, Carmeliet P. Glycolytic regulation of cell rearrangement in angiogenesis. Nat Commun. 2016;7:12240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 142] [Article Influence: 14.2] [Reference Citation Analysis (4)] |
| 12. | Simon-Molas H, Arnedo-Pac C, Fontova P, Vidal-Alabró A, Castaño E, Rodríguez-García A, Navarro-Sabaté À, Lloberas N, Manzano A, Bartrons R. PI3K-Akt signaling controls PFKFB3 expression during human T-lymphocyte activation. Mol Cell Biochem. 2018;448:187-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 13. | De Bock K, Georgiadou M, Carmeliet P. Role of endothelial cell metabolism in vessel sprouting. Cell Metab. 2013;18:634-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 321] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 14. | Suyama K, Silagi ES, Choi H, Sakabe K, Mochida J, Shapiro IM, Risbud MV. Circadian factors BMAL1 and RORα control HIF-1α transcriptional activity in nucleus pulposus cells: implications in maintenance of intervertebral disc health. Oncotarget. 2016;7:23056-23071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Obach M, Navarro-Sabaté A, Caro J, Kong X, Duran J, Gómez M, Perales JC, Ventura F, Rosa JL, Bartrons R. 6-Phosphofructo-2-kinase (pfkfb3) gene promoter contains hypoxia-inducible factor-1 binding sites necessary for transactivation in response to hypoxia. J Biol Chem. 2004;279:53562-53570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 204] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 16. | Pan JY, Sun CC, Bi ZY, Chen ZL, Li SJ, Li QQ, Wang YX, Bi YY, Li DJ. miR-206/133b Cluster: A Weapon against Lung Cancer? Mol Ther Nucleic Acids. 2017;8:442-449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 17. | Ringelhan M, McKeating JA, Protzer U. Viral hepatitis and liver cancer. Philos Trans R Soc Lond B Biol Sci. 2017;372:20160274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 251] [Article Influence: 27.9] [Reference Citation Analysis (2)] |
| 18. | Huang F, Wang BR, Wang YG. Role of autophagy in tumorigenesis, metastasis, targeted therapy and drug resistance of hepatocellular carcinoma. World J Gastroenterol. 2018;24:4643-4651. [PubMed] [DOI] [Full Text] |
| 19. | Wang K, Wang C, Jiang H, Zhang Y, Lin W, Mo J, Jin C. Combination of Ablation and Immunotherapy for Hepatocellular Carcinoma: Where We Are and Where to Go. Front Immunol. 2021;12:792781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 20. | Kim BH, Lee D, Jung KW, Won YJ, Cho H. Cause of death and cause-specific mortality for primary liver cancer in South Korea: A nationwide population-based study in hepatitis B virus-endemic area. Clin Mol Hepatol. 2022;28:242-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 21. | Liu CY, Chen KF, Chen PJ. Treatment of Liver Cancer. Cold Spring Harb Perspect Med. 2015;5:a021535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 326] [Cited by in RCA: 277] [Article Influence: 25.2] [Reference Citation Analysis (1)] |
| 22. | Lee S, Kang TW, Song KD, Lee MW, Rhim H, Lim HK, Kim SY, Sinn DH, Kim JM, Kim K, Ha SY. Effect of Microvascular Invasion Risk on Early Recurrence of Hepatocellular Carcinoma After Surgery and Radiofrequency Ablation. Ann Surg. 2021;273:564-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (1)] |
| 23. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6763] [Cited by in RCA: 6564] [Article Influence: 820.5] [Reference Citation Analysis (5)] |
| 24. | Mohkam K, Dumont PN, Manichon AF, Jouvet JC, Boussel L, Merle P, Ducerf C, Lesurtel M, Rode A, Mabrut JY. No-touch multibipolar radiofrequency ablation vs. surgical resection for solitary hepatocellular carcinoma ranging from 2 to 5 cm. J Hepatol. 2018;68:1172-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (3)] |
| 25. | Parisi A, Desiderio J, Trastulli S, Castellani E, Pasquale R, Cirocchi R, Boselli C, Noya G. Liver resection versus radiofrequency ablation in the treatment of cirrhotic patients with hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2013;12:270-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (3)] |
| 26. | Reiter RJ, Sharma R, Rosales-Corral S. Anti-Warburg Effect of Melatonin: A Proposed Mechanism to Explain its Inhibition of Multiple Diseases. Int J Mol Sci. 2021;22:764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 27. | Collin LJ, Maliniak ML, Cronin-Fenton DP, Ahern TP, Christensen KB, Ulrichsen SP, Damkier P, Hamilton-Dutoit S, Yacoub R, Christiansen PM, Sørensen HT, Lash TL. Hypoxia-inducible factor-1α expression and breast cancer recurrence in a Danish population-based case control study. Breast Cancer Res. 2021;23:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 28. | Chen Z, Wang J, Lin Y. Comparison of the efficacy and safety of repeated hepatectomy and radiofrequency ablation in the treatment of primary recurrent liver cancer: a meta-analysis. World J Surg Oncol. 2022;20:182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 29. | Su T, Huang M, Liao J, Lin S, Yu P, Yang J, Cai Y, Zhu S, Xu L, Peng Z, Peng S, Chen S, Kuang M. Insufficient Radiofrequency Ablation Promotes Hepatocellular Carcinoma Metastasis Through N6-Methyladenosine mRNA Methylation-Dependent Mechanism. Hepatology. 2021;74:1339-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 108] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 30. | Biagioni A, Laurenzana A, Menicacci B, Peppicelli S, Andreucci E, Bianchini F, Guasti D, Paoli P, Serratì S, Mocali A, Calorini L, Del Rosso M, Fibbi G, Chillà A, Margheri F. uPAR-expressing melanoma exosomes promote angiogenesis by VE-Cadherin, EGFR and uPAR overexpression and rise of ERK1,2 signaling in endothelial cells. Cell Mol Life Sci. 2021;78:3057-3072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 31. | Shin SU, Cho HM, Das R, Gil-Henn H, Ramakrishnan S, Al Bayati A, Carroll SF, Zhang Y, Sankar AP, Elledge C, Pimentel A, Blonska M, Rosenblatt JD. Inhibition of Vasculogenic Mimicry and Angiogenesis by an Anti-EGFR IgG1-Human Endostatin-P125A Fusion Protein Reduces Triple Negative Breast Cancer Metastases. Cells. 2021;10:2904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Li XY, Wang DP, Li S, Xue WH, Qian XH, Liu KL, Li YH, Lin QQ, Dong G, Meng FH, Jian LY. Discovery of N-(1,3,4-thiadiazol-2-yl)benzamide derivatives containing a 6,7-methoxyquinoline structure as novel EGFR/HER-2 dual-target inhibitors against cancer growth and angiogenesis. Bioorg Chem. 2022;119:105469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 33. | Zhang N, Li H, Qin C, Ma D, Zhao Y, Zhu W, Wang L. Insufficient radiofrequency ablation promotes the metastasis of residual hepatocellular carcinoma cells via upregulating flotillin proteins. J Cancer Res Clin Oncol. 2019;145:895-907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 34. | Teng W, Liu KW, Lin CC, Jeng WJ, Chen WT, Sheen IS, Lin CY, Lin SM. Insufficient ablative margin determined by early computed tomography may predict the recurrence of hepatocellular carcinoma after radiofrequency ablation. Liver Cancer. 2015;4:26-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 35. | Song H, Li D, Wang X, Fang E, Yang F, Hu A, Wang J, Guo Y, Liu Y, Li H, Chen Y, Huang K, Zheng L, Tong Q. HNF4A-AS1/hnRNPU/CTCF axis as a therapeutic target for aerobic glycolysis and neuroblastoma progression. J Hematol Oncol. 2020;13:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 36. | Pelicano H, Martin DS, Xu RH, Huang P. Glycolysis inhibition for anticancer treatment. Oncogene. 2006;25:4633-4646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1226] [Cited by in RCA: 1084] [Article Influence: 54.2] [Reference Citation Analysis (0)] |
| 37. | Zhao X, Zhu Y, Hu J, Jiang L, Li L, Jia S, Zen K. Shikonin Inhibits Tumor Growth in Mice by Suppressing Pyruvate Kinase M2-mediated Aerobic Glycolysis. Sci Rep. 2018;8:14517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (4)] |
| 38. | Hu C, Liu T, Han C, Xuan Y, Jiang D, Sun Y, Zhang X, Zhang W, Xu Y, Liu Y, Pan J, Wang J, Fan J, Che Y, Huang Y, Zhang J, Ding J, Yang S, Yang K. HPV E6/E7 promotes aerobic glycolysis in cervical cancer by regulating IGF2BP2 to stabilize m(6)A-MYC expression. Int J Biol Sci. 2022;18:507-521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 112] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 39. | Chen L, Ying X, Zhang D, Lai L, Wu F, Tu J, Ji J. Iodine-125 Brachytherapy Can Prolong Progression-Free Survival of Patients with Locoregional Recurrence and/or Residual Hepatocellular Carcinoma After Radiofrequency Ablation. Cancer Biother Radiopharm. 2021;36:820-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 40. | Ampferl R, Rodemann HP, Mayer C, Höfling TTA, Dittmann K. Glucose starvation impairs DNA repair in tumour cells selectively by blocking histone acetylation. Radiother Oncol. 2018;126:465-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |