Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116097
Revised: November 28, 2025
Accepted: January 14, 2026
Published online: April 15, 2026
Processing time: 157 Days and 8.5 Hours
Three-dimensional (3D) culture systems closely recapitulate the tumor microenvironment, allowing the assessment of drug effects on cell-cell interactions and metabolic heterogeneity. Colorectal adenocarcinoma (CRAC) cells exhibit epithe
To elucidate the morphological and functional responses of 3D-cultured CRAC spheres to anticancer drugs and to characterize the residual CSC/EMT phenotypes.
Five CRAC cell lines were cultured as 3D spheres, treated with 5-fluorouracil, irinotecan, or oxaliplatin, and analyzed morphologically and molecularly.
All drugs significantly suppressed sphere growth, but the morphological and metabolic responses varied. Scanning electron microscopy revealed cell surface flattening or detachment in epithelial-type spheres, whereas mesenchy
Anticancer drugs induce distinct morphological and molecular adaptations, and residual CRAC spheres acquire hybrid epithelial-mesenchymal phenotypes associated with chemoresistance.
Core Tip: This study analyzed the effects of 5-fluorouracil, irinotecan, and oxaliplatin on three-dimensionally cultured colorectal adenocarcinoma (CRAC) spheres. Using cell viability assays, apoptosis detection, and quantitative reverse transcriptase PCR, we found that the residual spheres displayed increased expression of cancer stem cell and epithelial-mesenchymal transition markers, with distinct profiles between epithelial (HCT-15) and mesenchymal (SW480) types. These results indicate that CRAC spheres that survive anticancer drug treatment acquire hybrid epithelial-mesenchymal phenotypes and stem cell-like features, providing insights into the mechanisms of drug tolerance and cancer recurrence in colorectal cancer.
- Citation: Yoshimura Y, Shinji S, Ogawa Y, Yamada T, Matsuda A, Uehara K, Yokoyama Y, Takahashi G, Iwai T, Miyasaka T, Kanaka S, Matsui T, Hayashi K, Fujiwara M, Shichi Y, Arai T, Ishiwata T, Yoshida H. Morphological and functional responses of three-dimensional-cultured colorectal cancer spheres to anticancer drugs. World J Gastrointest Oncol 2026; 18(4): 116097
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116097.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116097
Colorectal cancer (CRC) is a leading cause of cancer-related morbidity and mortality worldwide[1]. By 2050, 6.9 million new patients aged ≥ 80 years (20.5% of all cancer cases) will be diagnosed with CRC worldwide[2]. Despite advances in surgical techniques and systemic chemotherapy, recurrence and resistance to anticancer drugs continue to pose major clinical challenges.
Neoadjuvant chemotherapy is increasingly applied in the management of advanced CRC, with the goal of tumor downstaging, improving resectability, and reducing recurrence risk[3,4]. Nevertheless, a subset of tumor cells often persists after treatment, forming residual masses that may harbor stem-like features and epithelial-mesenchymal plasticity, thereby contributing to relapse and therapeutic resistance[5,6]. The biological characteristics of these residual tumor spheres remain poorly understood, particularly with respect to their survival mechanisms, stemness potential, and epithelial-mesenchymal transition (EMT)-related traits.
Conventional two-dimensional (2D) culture models have substantially contributed to the understanding of CRC biology, therapeutic responses, cell-cell interactions, and the microenvironmental conditions of tumors in vivo[7]. Recently, three-dimensional (3D) culture systems, including spheroid and organoid models, have emerged as powerful tools for investigating tumor heterogeneity, drug sensitivity, and cancer stem cell (CSC) properties[8-12].
We recently reported significant differences in the cell morphology and behavior between 2D and 3D culture systems in five cultured colorectal adenocarcinoma (CRAC) cell lines (DLD-1, CACO-2, HCT-15, SW480, and COLO-320)[13]. The 2D-cultured cell lines exhibited similar cellular morphologies; however, DLD-1, CACO-2, and HCT-15 cells showed high expression of the epithelial marker E-cadherin (CDH1) and low expression of the mesenchymal marker VIM, whereas SW480 and COLO-320 cells exhibited opposite expression patterns. Epithelial-like DLD-1 and mesenchymal-like COLO-320 cells formed loosely connected granular spheres, whereas epithelial-like HCT-15 cells formed small solid spheres. Ki-67-positive proliferating cell counts fluctuated in both 2D and 3D epithelial cell cultures but remained consistently high in mesenchymal cells. These findings suggest the presence of epithelial-mesenchymal lineages within CRAC. However, unlike previous findings in pancreatic ductal adenocarcinoma (PDAC), no clear differences in sphere morphology or function were observed in 3D culture[14]. In PDAC, differences were observed in the effects of anticancer drugs on 3D-cultured spheres: Gemcitabine was more effective against epithelial PDAC spheres, whereas nab-paclitaxel was more effective against mesenchymal PDAC spheres.
This study aimed to evaluate the effects of currently used anticancer drugs on the proliferation and survival rates of spheres. Drug responses were compared using assays based on the spherical size reduction and cell death. The surviving spheres were analyzed for CSC markers and epithelial-mesenchymal-like phenotypes. By elucidating the characteristics of residual CRC spheres after drug exposure, this study aimed to provide new insights into the preoperative neoadjuvant effects of chemotherapy, the biological properties of unresectable tumors, and the potential for developing novel therapeutic strategies targeting drug-resistant tumor cell populations.
CACO-2 and COLO-320 human CRAC cells were provided by the RIKEN BioResource Research Center through the National Bioresource Project of the Ministry of Education, Culture, Sports, Science, and Technology, Japan. DLD-1, HCT-15, and SW480 human CRAC cell lines were obtained from the Cell Resource Center for Biomedical Research, Institute of Development, Aging, and Cancer, Tohoku University (Sendai, Japan). The cells were grown in growth medium (Roswell Park Memorial Institute-1640 medium containing 10% fetal bovine serum) at 37 °C in a humidified 5% CO2 atmosphere. Genomic DNA was extracted from CRAC cells using a DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany), according to the manufacturer’s instructions. Short tandem repeats were analyzed using the GenePrint 10 System (Promega, Madison, WI, United States) at BEX Co., Ltd., according to the manufacturer’s instructions. All CRAC cell lines were confirmed to be correctly genotyped and contamination-free[13].
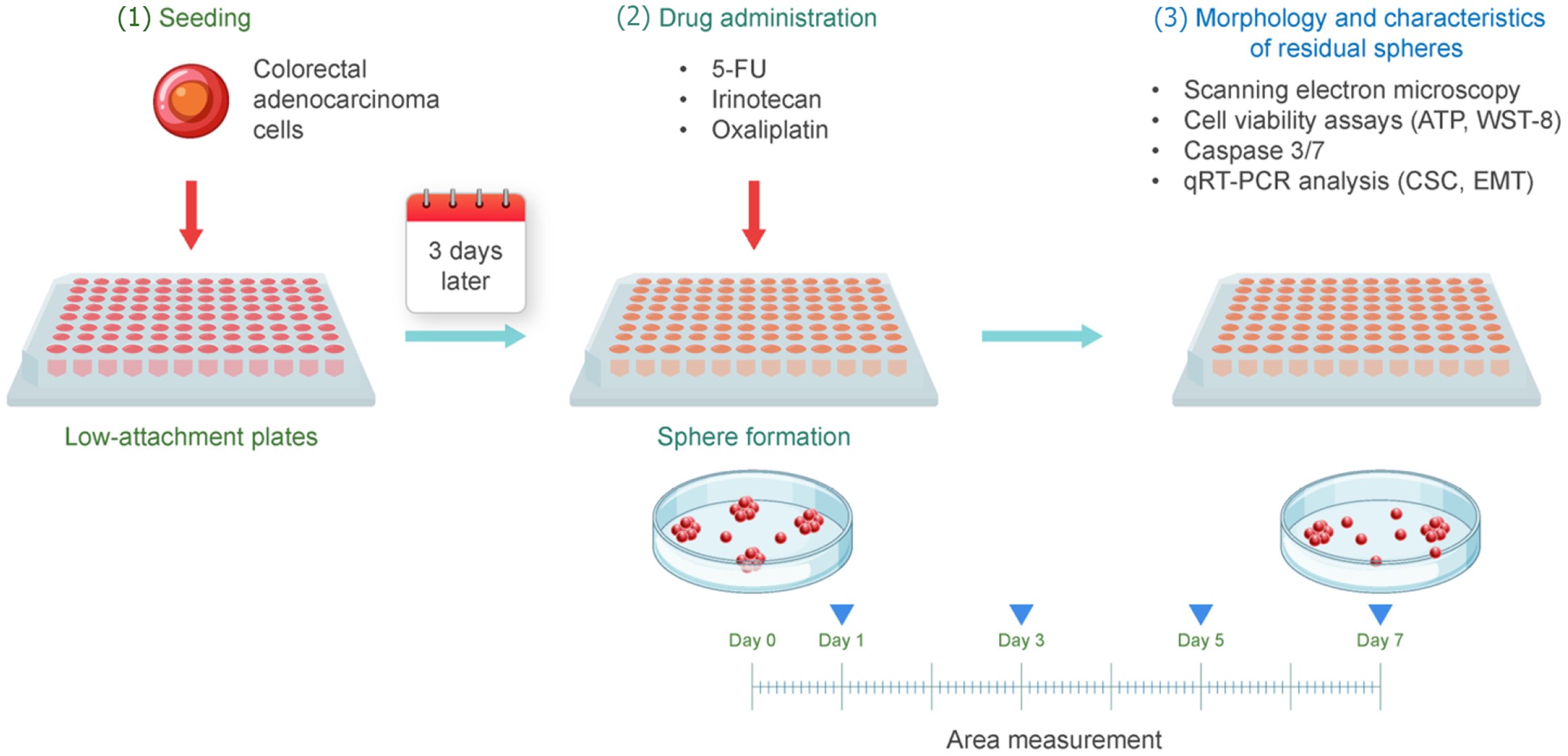
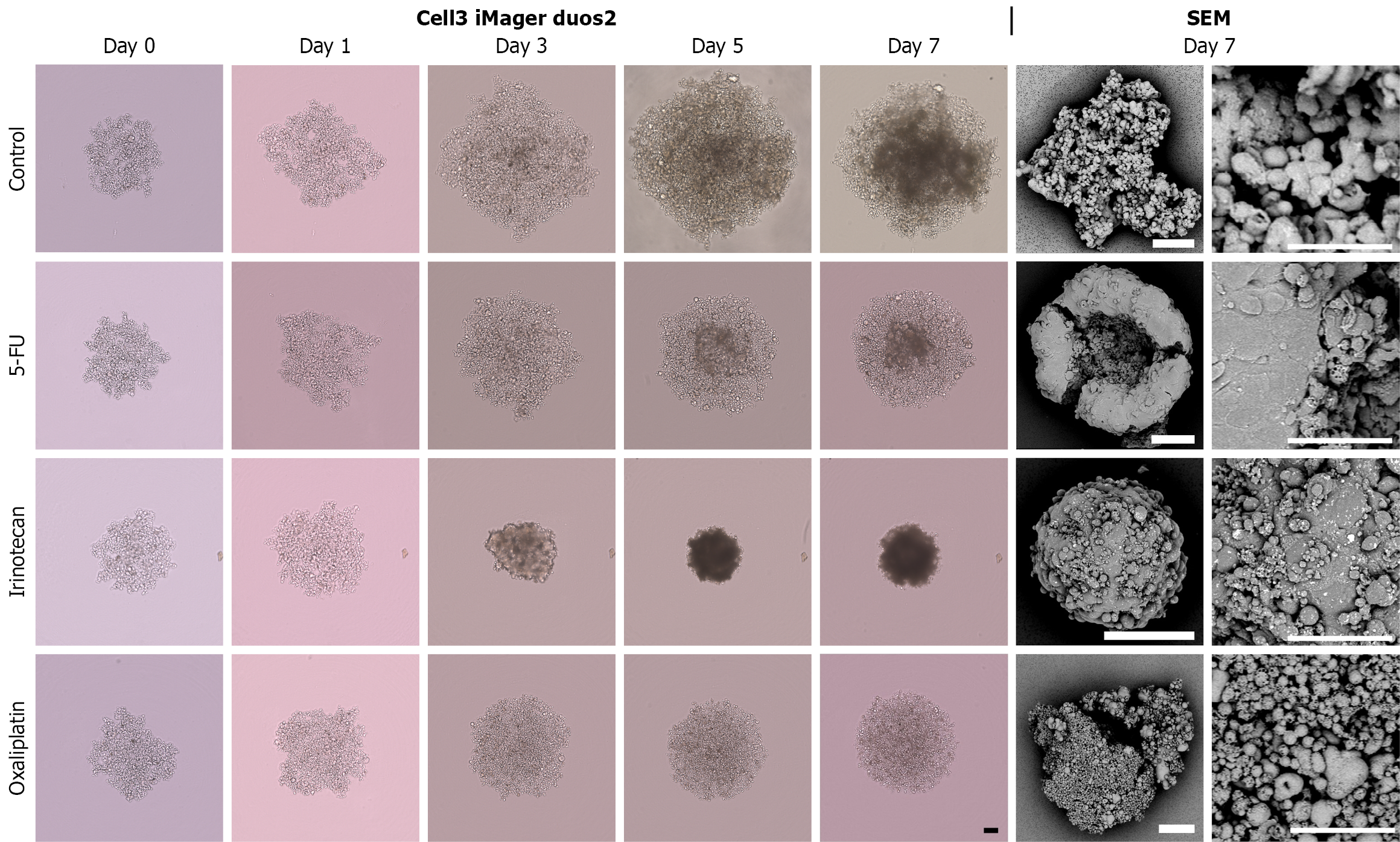
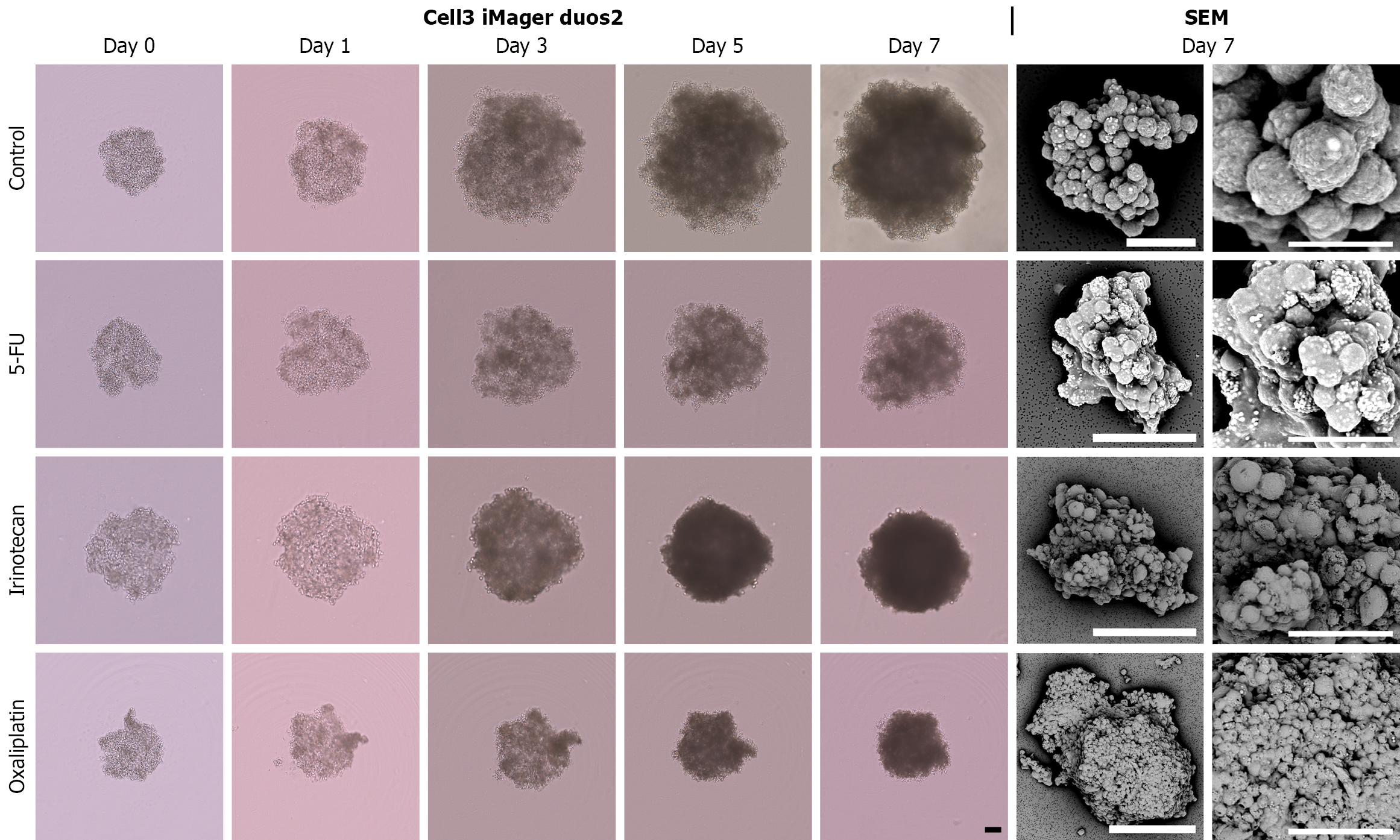
The effects of anticancer drugs on 3D-cultured CRC spheres were examined as follows. CRAC cells (3000 cells per 100 μL) were seeded into a low-attachment 96-well round-bottom plate (Thermo Fisher Scientific, Waltham, MA, United States) and cultured for 3 days to form spheres. Subsequently, 100 μL of anticancer drugs [5-fluorouracil (5-FU), irinotecan, and oxaliplatin] at a final concentration of 30 μM were added, and this time point was designated as day 0. The concentrations of the anticancer drugs effective against CRC organoids were determined based on previous studies[15]. 5-FU, irinotecan, and oxaliplatin were purchased from Selleckchem.com (Houston, TX, United States). The IC50 values of 5-FU, irinotecan, and oxaliplatin reported in 2D-cultured CRC cells generally range from 1-10 μM; however, higher concentrations are required in 3D spheroid models because of impaired drug penetration and microenvironmental resistance[9,11,16]. Therefore, we used 30 μM as an effective concentration to induce partial yet measurable responses in CRC spheres while allowing assessment of residual populations. To evaluate the inhibitory effects of anticancer drugs on cell proliferation, CRAC spheres were imaged on days 0, 1, 3, 5, and 7 using a cell imaging system (Cell3 iMager duos2, Screen Holdings Co., Ltd., Kyoto, Japan). The areas of the spheres were quantified from the images using an integrated artificial intelligence-based deep learning algorithm, as previously described[17]. In the control group, dimethyl sulfoxide for 5-FU and irinotecan or Milli-Q water for oxaliplatin was administered instead of anticancer drugs (Figure 1). Sphere growth assays were performed using three biological replicates, each measured in triplicate as technical replicates.
CRAC cells were cultured as described above, and the spheres were collected on day 7. Pretreatment was performed as previously reported for scanning electron microscopic (SEM) observations[13]. CRAC spheres were fixed overnight with 2.5% glutaraldehyde in 0.1 M phosphate buffer (pH = 7.4) at 23 °C. The glutaraldehyde solution was subsequently removed, and the cells were washed with phosphate-buffered saline. The CRAC spheres were postfixed with 1% OsO4 for 30 minutes. After complete dehydration in a graded ethanol series, the samples were suspended in 100% ethanol, air-dried, and coated with a platinum layer using an MSP-1S sputter coater (Vacuum Device Inc., Ibaraki, Japan). The CRAC spheres were examined and imaged using a Phenom Pro desktop scanning electron microscope with reflected electrons for spheres (Thermo Fisher Scientific). SEM analyses were performed on three independently prepared samples, each observed in triplicate.
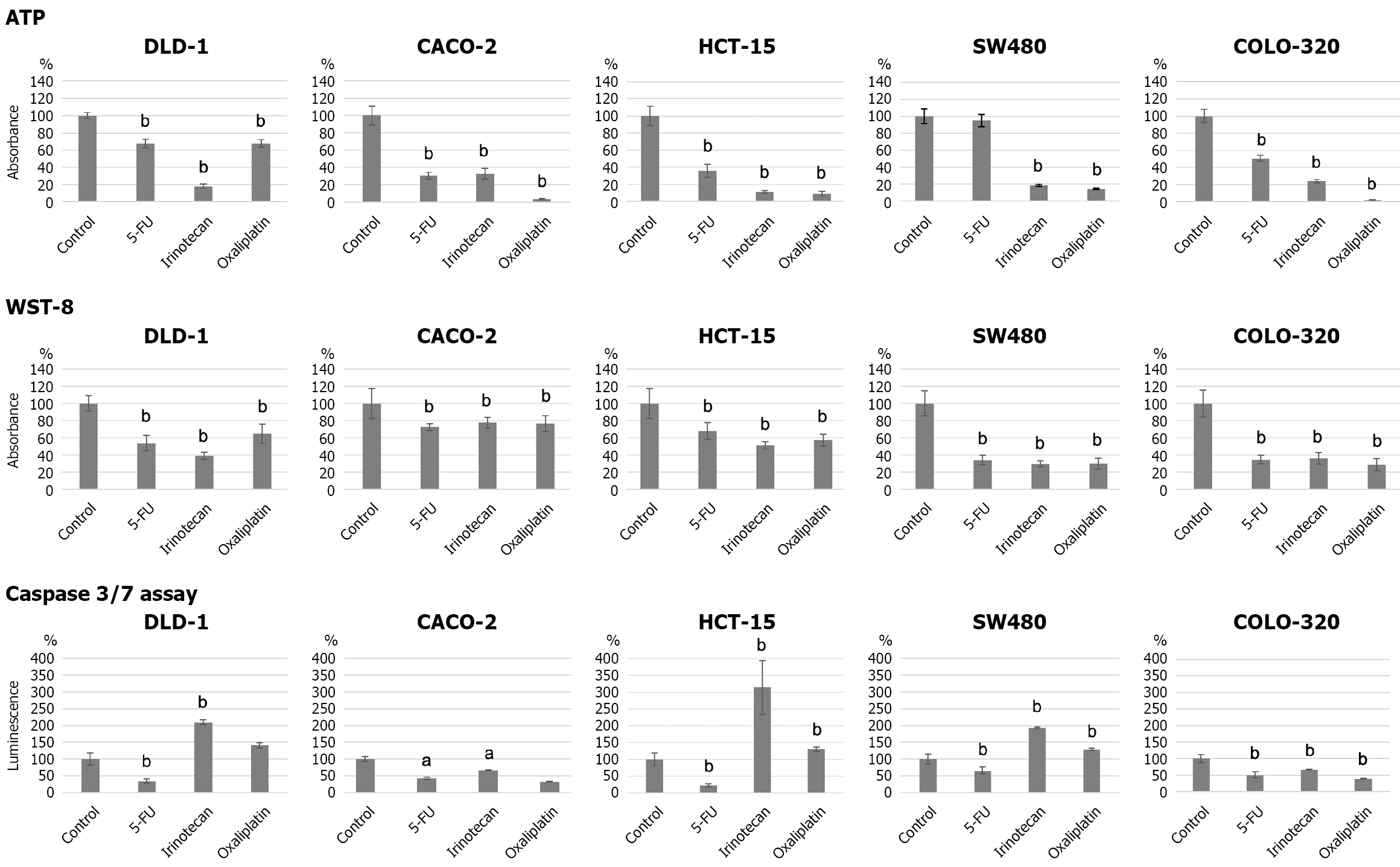
To evaluate cell viability by different principles, we performed both ATP and water-soluble tetrazolium 8 (WST-8) assays. The ATP assay measures intracellular ATP levels through a luciferase-catalyzed luminescent reaction, thereby reflecting cellular energy status[18]. In contrast, the WST-8 assay is a colorimetric method in which the tetrazolium salt WST-8 is reduced by cellular dehydrogenases to generate a soluble formazan dye, providing an estimate of metabolic activity and proliferative capacity[19]. CRAC cells were cultured, as described above. On day 7, the CRAC spheres were collected and transferred to CELLSTAR 96-well microplates (Greiner Bio-One International GmbH, Frickenhausen, Germany). ATP assays were performed using the CellTiter-Glo® 3D Reagent (Promega, Madison, WI, United States), according to the manufacturer’s instructions. Luminescence was measured using a Nivo Multimode Plate Reader (Revvity, Inc., Waltham, MA, United States). The ATP assay was performed in triplicate on three independent plates. For the WST-8 assay, the CRAC spheres were incubated with the WST-8 cell counting reagent (Cell Counting Reagent SF; Nacalai Tesque Inc., Kyoto, Japan) for 3 hours. Absorbance was measured at 450 nm using an iMark plate reader (Bio-Rad Laboratories, Hercules, CA, United States). The WST-8 assay was conducted in triplicate in three independent experiments[20,21].
To assess apoptosis activity within the 3D spheroids, Caspase-Glo® 3/7 3D Assay (Promega) was used to measure caspase-3/7 activity based on luminescent signal generation. The CRAC cells were cultured, as described above. Seven days after anticancer drug administration, 100 μL of culture medium was aspirated and removed, followed by the addition of 100 μL of Caspase-Glo 3/7 reagent. After incubating at 23 °C for 3 hours, the samples were transferred to CELLSTAR 96-well microplates (Greiner Bio-One International GmbH), and luminescence was detected using a Nivo Multimode Plate Reader (Revvity, Inc.). Caspase-3/7 assays were performed using three biological replicates, each measured in triplicate as technical replicates.
To examine the expression of CSC and EMT markers in spheres after anticancer drug treatment, quantitative reverse transcriptase PCR (qRT-PCR) was performed, as previously described[13]. Total RNA was isolated from cells using the NucleoSpin RNA Kit (Macherey-Nagel GmbH & Co. KG, Düren, Germany) and subsequently reverse-transcribed using the SuperScriptTM IV VILOTM Master Mix (Thermo Fisher Scientific). qRT-PCR was performed using the PowerTrackTM SYBR Green Master Mix (Thermo Fisher Scientific) on a QuantStudio 3 real-time PCR system (Thermo Fisher Scientific). Table 1 shows the primer sequences. GAPDH was used as an internal control. The threshold crossing value was noted for each transcript and normalized to that of the internal control. The relative quantification of each mRNA was performed using the comparative Ct (2-ΔΔCt), method. Three independent samples were prepared for PCR. Each sample was analyzed in duplicate, resulting in a biological replicate number of n = 3.
| Gene | Forward | Sequence |
| ALDH1A1 | F-primer | 5’-CGG-GAA-AAG-CAA-TCT-GAA-GAG-GG-3’ |
| R-primer | 5’-GAT-GCG-GCT-ATA-CAA-CAC-TGG-C-3’ | |
| CD44 | F-primer | 5’-CCA-GAA-GGA-ACA-GTG-GTT-TGG-C-3’ |
| R-primer | 5’-ACT-GTC-CTC-TGG-GCT-TGG-TGT-T-3’ | |
| CD133 (PROM1) | F-primer | 5’-CAC-TAC-CAA-GGA-CAA-GGC-GTT-C-3’ |
| R-primer | 5’-CAA-CGC-CTC-TTT-GGT-CTC-CTT-G-3’ | |
| E-cadherin (CDH1) | F-primer | 5’-GCC-TCC-TGA-AAA-GAG-AGT-GGA-AG-3′ |
| R-primer | 5’-TGG-CAG-TGT-CTC-TCC-AAA-TCC-G-3′ | |
| Vimentin (VIM) | F-primer | 5’-AGG-CAA-AGC-AGG-AGT-CCA-CTG-A-3′ |
| R-primer | 5’-ATC-TGG-CGT-TCC-AGG-GAC-TCA-T-3′ | |
| ZEB1 (AREB6) | F-primer | 5’-GGC-ATA-CAC-CTA-CTC-AAC-TAC-GG-3’ |
| R-primer | 5’-TGG-GCG-GTG-TAG-AAT-CAG-AGT-C-3’ | |
| GAPDH | F-primer | 5’-GTC-TCC-TCT-GAC-TTC-AAC-AGC-G-3′ |
| R-primer | 5’-ACC-ACC-CTG-TTG-CTG-TAG-CCA-A-3′ |
All estimations and group comparisons in Figures 2, 3, 4, 5, 6, and 7, were performed using a linear model (analysis of variance; ANOVA). For the ATP and WST-8 assays shown in Figure 7, a linear mixed-effects model was applied, in which plate-to-plate variability was incorporated as a random effect. When multiple comparisons were conducted, appropriate post hoc adjustments (e.g., Bonferroni or Tukey) were applied. Exact P values are reported in the figure legends. All statistical analyses were performed using SAS version 9.4 (SAS Institute Japan, Ltd., Tokyo, Japan).
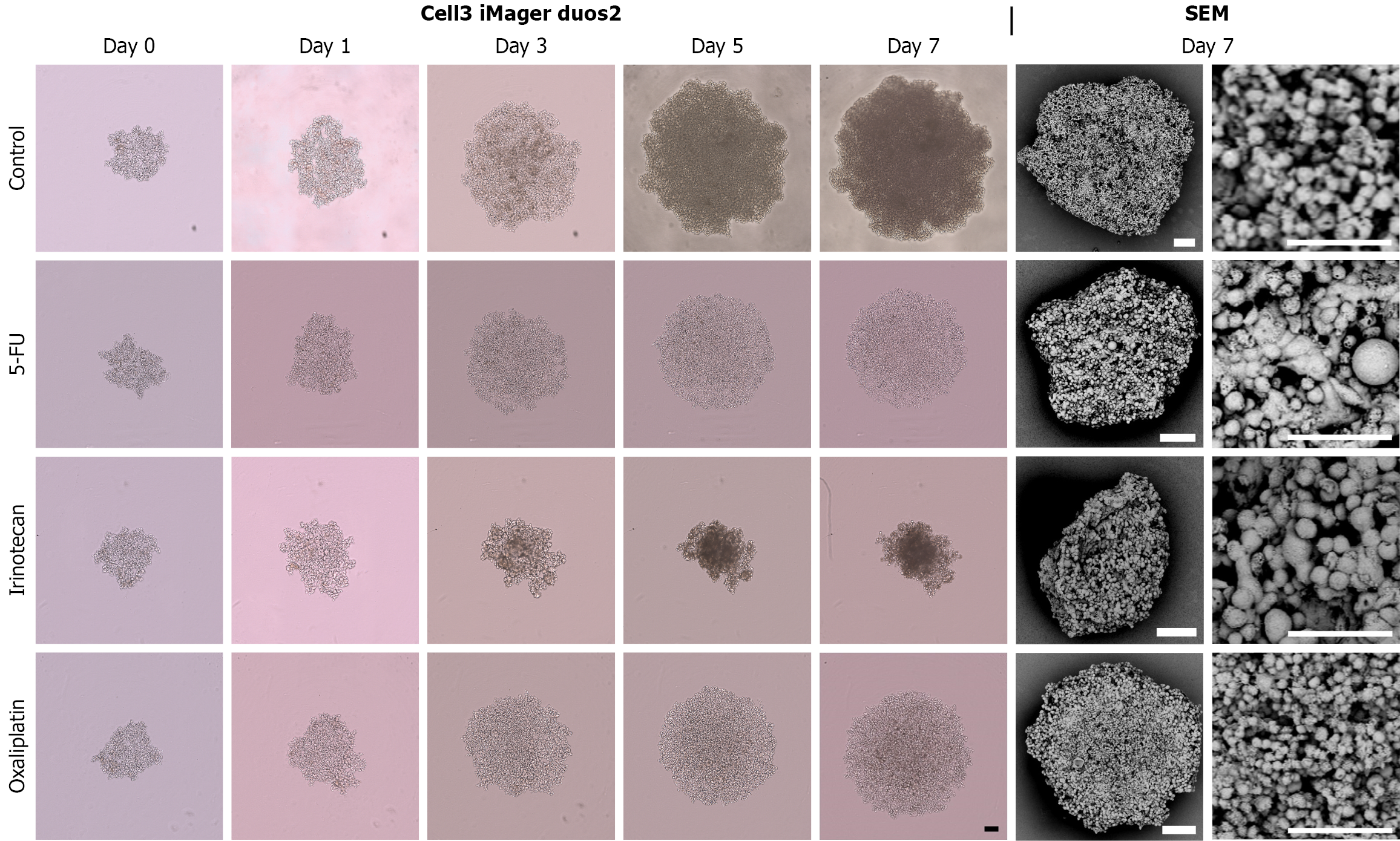
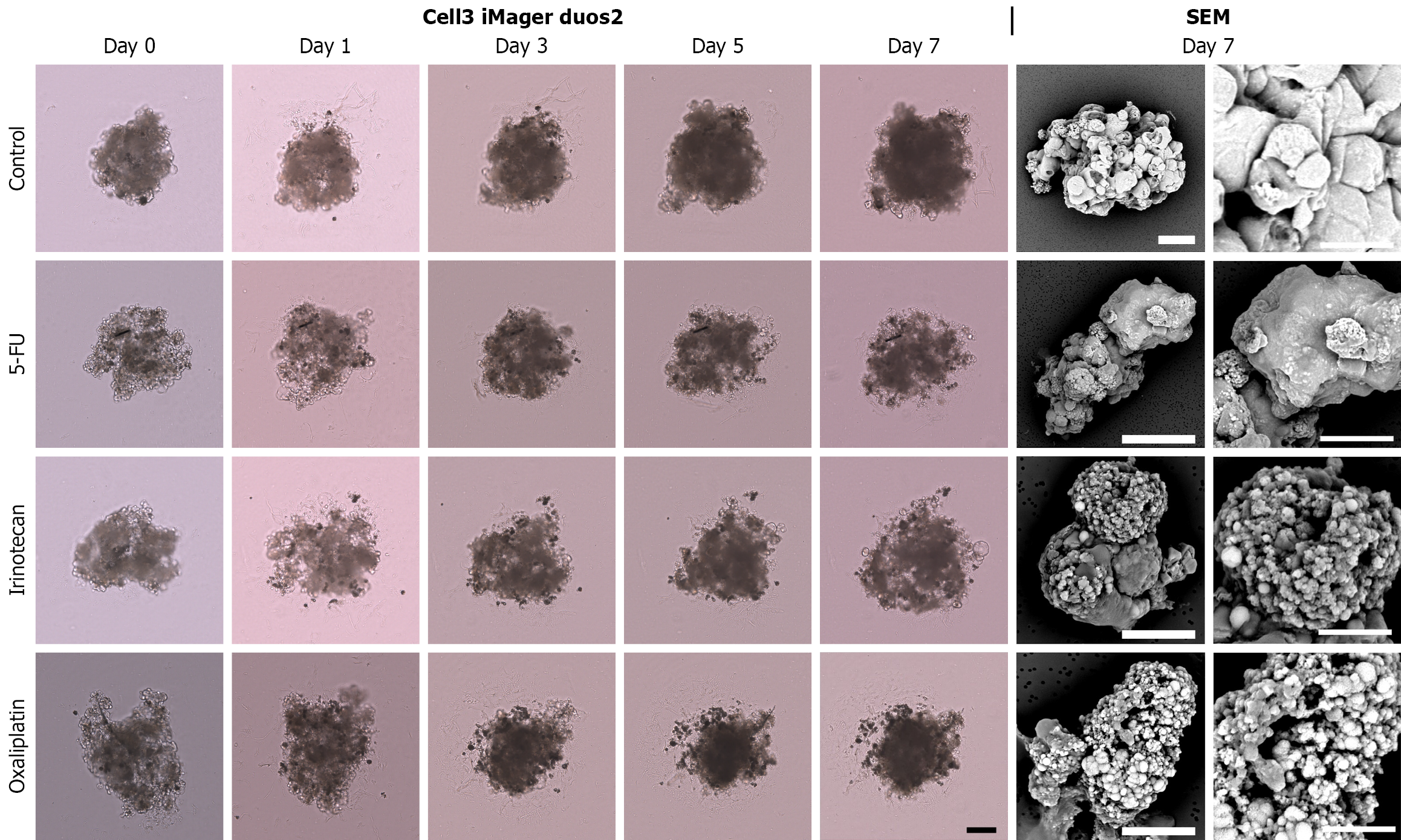
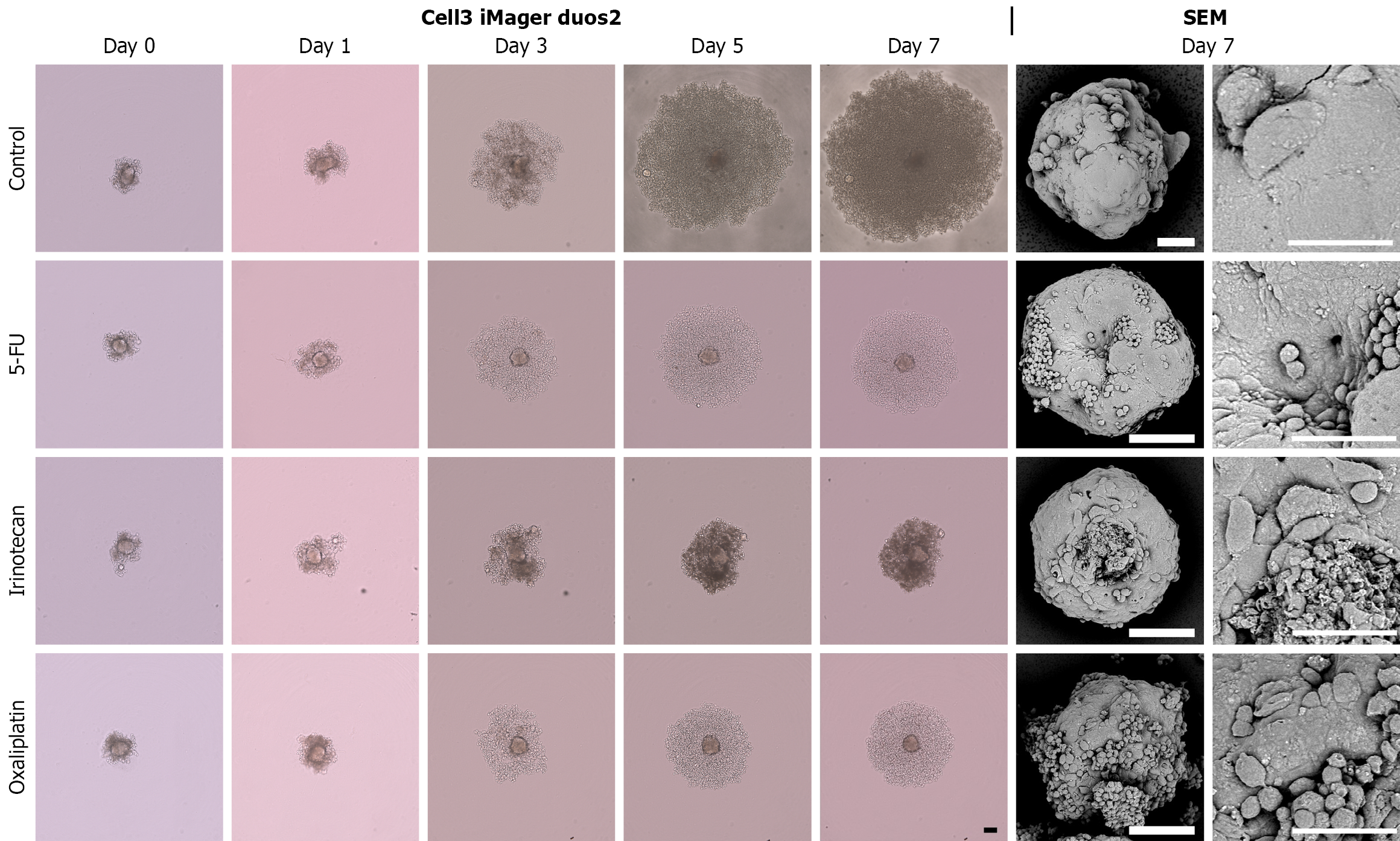
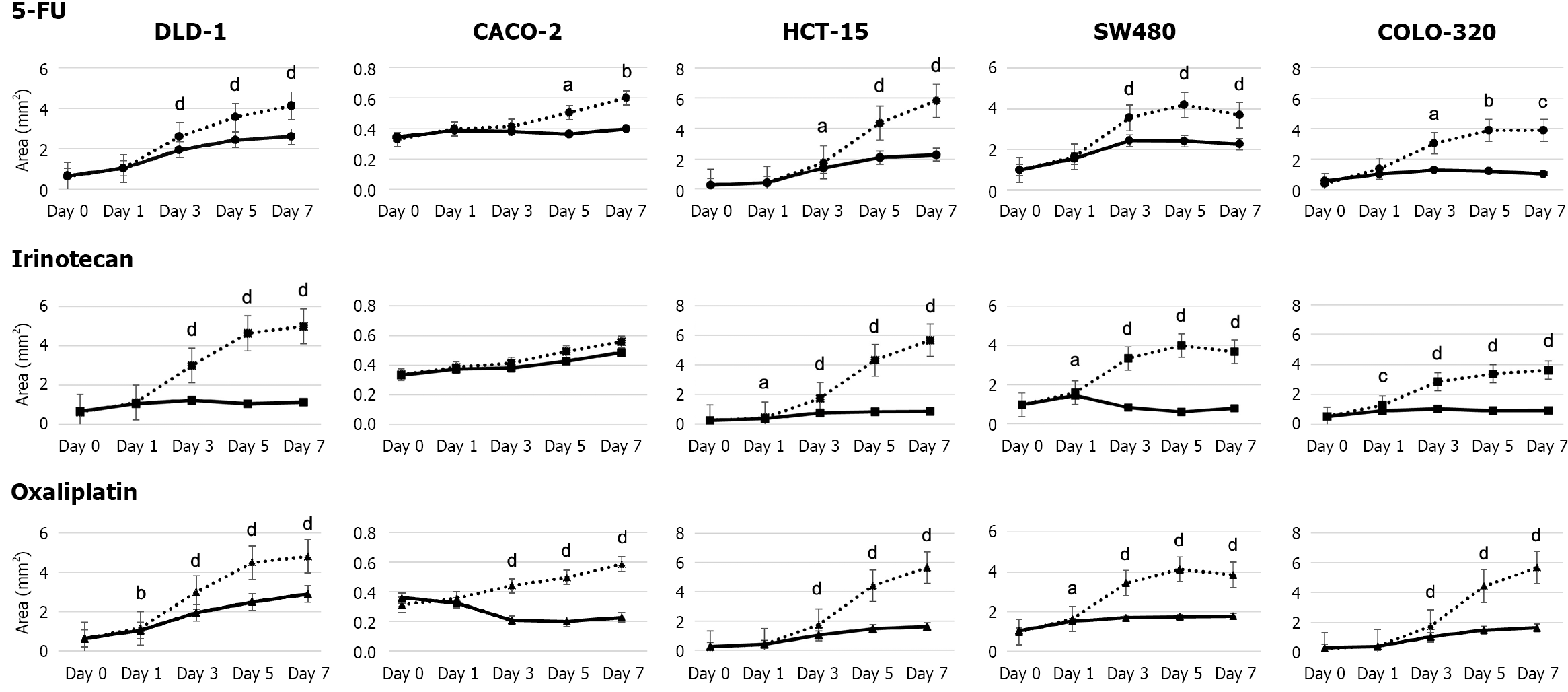
To investigate the effects of anticancer drugs on CRAC cells that had formed masses, experiments were conducted in which anticancer drugs were added to the spheres formed in 3D culture for 7 days (Figure 1). Three epithelial-like CRAC cell lines (DLD-1, CACO-2, and HCT-15) exhibiting high E-cadherin expression and low VIM expression and two CRAC cell lines (SW480 and COLO-320) exhibiting opposite expression patterns and mesenchymal-like properties were used[13]. In DLD-1 cells with the highest CDH1 (E-cadherin) expression, the three anticancer drugs showed significant growth-inhibitory effects (Figures 2 and 8). In particular, after irinotecan administration, the surface area of the spheres remained stable, showing only a slight increase compared with that before administration (Figure 8). However, the morphology of the spheres observed by SEM on day 7 after administration of the three types of anticancer drugs showed small spherical cancer cells clustered together, exhibiting a morphology similar to that of the control group (Figure 2). CACO-2 cell spheres showed no significant reduction in area following 5-FU administration (Figures 3 and 8). In the irinotecan treatment group, the sphere area showed a mild decrease on days 5 and 7 compared with the control group. In contrast, with oxaliplatin, the spherical area decreased markedly and progressively from days 3 to 7, with smaller spheres observed before treatment. Although the anticancer drugs exhibited different effects in suppressing sphere area, SEM revealed both granular and broad smooth areas on the surface of CACO-2 cells following treatment with any of the anticancer drugs (Figure 3). The spherical area of HCT-15 cells was significantly suppressed starting on day 3 after the administration of all three anticancer drugs (Figures 4 and 8). In particular, in irinotecan-treated HCT-15 cells, the spherical area remained almost unchanged from that before treatment (Figure 8). HCT-15 cell spheres, observed by SEM on day 7 after anticancer drug administration, were covered with a lining of cell-like cancer cells, exhibiting a smooth surface. Granular cancer cells were observed in some areas, displaying morphology similar to that of the control group (Figure 4). In SW480 cells exhibiting mesenchymal characteristics, the sphere size was significantly lower compared with that in the control group starting 1 day after the administration of all three anticancer drugs (Figures 5 and 8). In the irinotecan treatment group, the sphere area was lower from days 3 to 7 compared with that from pretreatment levels, and the spheres appeared darker in color on days 5 and 7 (Figure 8). The SEM image of SW-480 spheres on day 7 after 5-FU administration showed a characteristic bowl-shaped structure with a depressed center (Figure 5). SEM observation showed that on the 7th day after irinotecan administration, the spheres consisted of smooth cells closely adhering to each other and spherical cells with small granular substances (Figure 5). In COLO-320 cells, significant suppression of sphere area was observed on day 5 after 5-FU administration and on day 3 after irinotecan and oxaliplatin administration (Figures 6 and 8). In particular, in the irinotecan treatment group, the increase in sphere area was minimal, with little change observed before and after administration (Figure 8). SEM observation revealed that COLO-320 spheres in the control group consisted of small cancer cells clustered together. In the 5-FU-treated group, the cancer cells forming spheres exhibited prominent small smooth surface protrusions (Figure 6). In the irinotecan- and oxaliplatin-treated groups, numerous cancer cells with surface holes were observed (Figure 6).
To evaluate the cytotoxic effects of the anticancer drugs on CRAC spheres, two complementary cell viability assays were performed. The ATP assay reflects cellular energy status by measuring intracellular ATP levels, whereas the WST-8 assay indicates metabolic activity through the reduction of a tetrazolium salt by cellular dehydrogenases. Treatment with the three anticancer drugs significantly decreased the ATP levels in the five CRAC spheres (upper panels in Figure 7). Notably, ATP levels in SW480 cells remained unchanged after 5-FU treatment. Irinotecan markedly reduced ATP levels in DLD-1 cells, whereas oxaliplatin caused a greater decrease in CACO-2 and COLO-320 cells than the other two drugs. In HCT-15 and SW480 cells, both irinotecan and oxaliplatin induced a more pronounced reduction in ATP levels than 5-FU.
In the WST-8 assay, all CRAC spheres treated with the three anticancer drugs showed a significant decrease in WST-8 activity (middle panels in Figure 7). These findings indicate that, as observed in SW480 cells treated with 5-FU, some cells maintain ATP levels but exhibit reduced metabolic activity.
In the caspase-3/7 assay, which detects the execution phase of apoptosis, changes in caspase-3/7 activity varied depending on the type of anticancer drug and CRAC sphere (lower panels in Figure 7). Caspase-3/7 activity in HCT-15 spheres treated with irinotecan was approximately three-fold higher than that in the control and approximately two-fold higher in DLD-1 and SW480 spheres. Oxaliplatin treatment also resulted in a significant increase in caspase-3/7 activity in HCT-15 and SW480 cells.
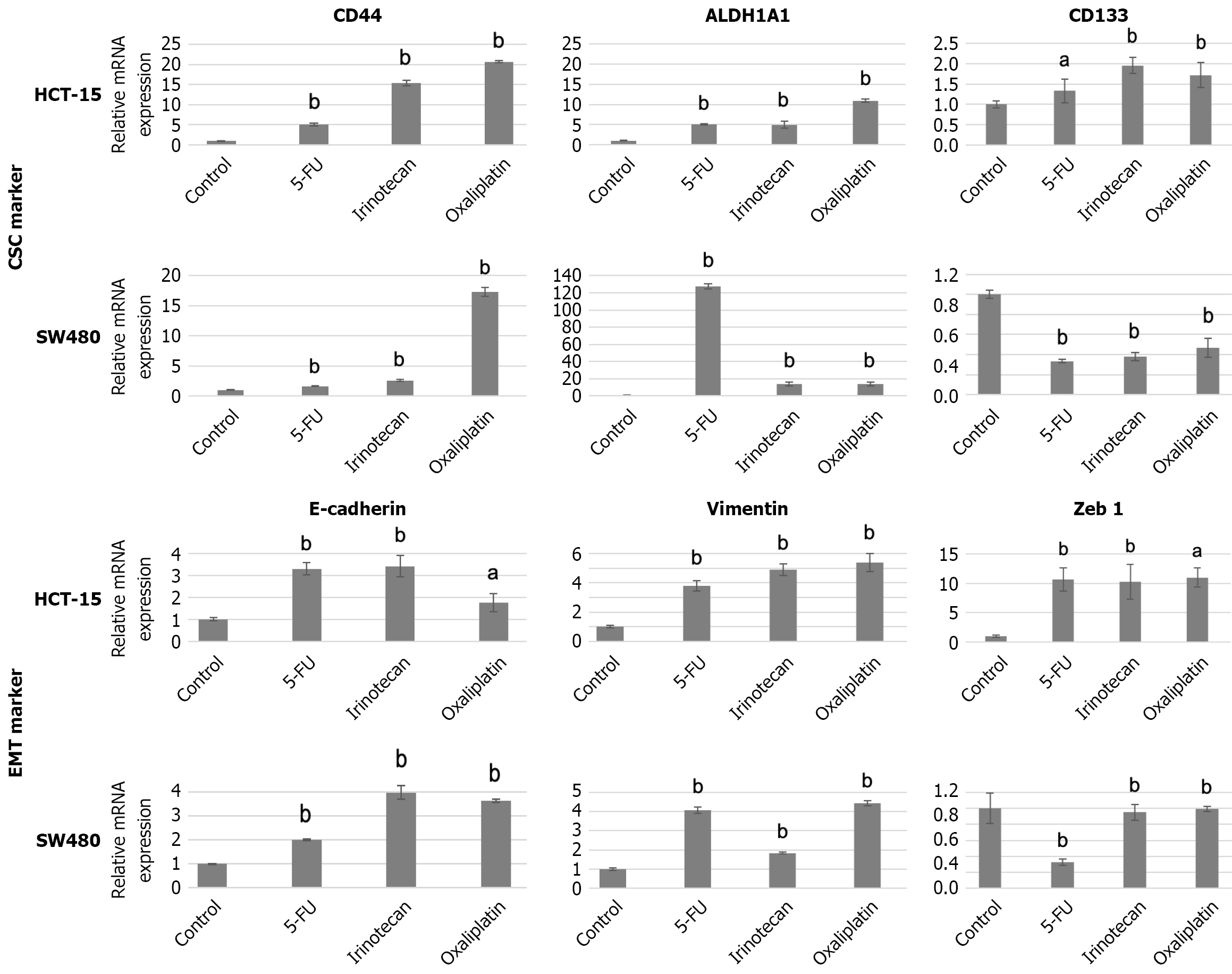
To investigate the CSC properties and EMT characteristics of residual CRAC after anticancer drug treatment, qRT-PCR was performed. The spheres of CRAC cells treated with anticancer drugs were small and tended to fragment easily, making it difficult to extract sufficient amounts of RNA. In this study, two representative CRAC cell lines were selected from the five cell types: HCT-15, an epithelial-type cell line, and SW480, a mesenchymal-type cell line. These lines formed smooth-surfaced spheres from which sufficient RNA could be obtained. In the spheres of HCT-15 cells treated with the three anticancer drugs, the major CSC markers of CRAC—CD44, ALDH1A1, and PROM1(CD133)—were significantly upregulated (upper two panels, Figure 9). In the SW480 cell spheres, anticancer drug treatment increased CD44 and ALDH1A1 expression, whereas PROM1 expression decreased. Notably, in SW480 spheres, CD44 expression was approximately 15-fold higher in the oxaliplatin-treated group and approximately 120-fold higher in the 5-FU-treated group than in the control group.
Next, the expression of epithelial and mesenchymal markers in the residual spheres after anticancer drug treatment was examined. In HCT-15 cells, the expression levels of epithelial marker CDH1 (E-cadherin) and the mesenchymal markers VIM and ZEB1 were higher than those in the control (third panels in Figure 9). In contrast, in SW480 cells, treatment with the three anticancer drugs increased both CDH1 and VIM expression, whereas ZEB1 expression decreased (fourth panel in Figure 9).
This study demonstrated that the responses of CRAC spheres to anticancer drugs differed according to the epithelial or mesenchymal phenotype of the originating cells. Using five CRAC cell lines, we showed that 5-FU, irinotecan, and oxaliplatin suppressed 3D sphere growth. However, the degree of metabolic inhibition, apoptosis induction, and stemness activation varied among the lines. In particular, the residual spheres that survived treatment displayed distinct expression profiles of CSC and EMT markers, suggesting the acquisition of a hybrid epithelial/mesenchymal (E/M) phenotype that may underlie drug resistance and recurrence.
These results emphasize the importance of 3D culture systems in modeling chemotherapy responses. Unlike 2D monolayers, 3D spheres recreate gradients of oxygen, nutrients, and drug diffusion that exist within solid tumors in vitro, enabling a more physiological assessment of metabolic adaptation and cell-cell interactions that influence resistance[22,23].
Epithelial-type HCT-15 and mesenchymal-type SW480 cells exhibited clearly different responses at both the morphological and molecular levels. In HCT-15 spheres, anticancer drug exposure led to an increased expression of epithelial (E-cadherin) and mesenchymal (VIM, ZEB1) markers, suggesting partial EMT activation. Conversely, SW480 spheres, initially low in CDH1 and high in VIM, displayed increased CDH1 and VIM expression but decreased ZEB1 expression after treatment. These findings indicate that the surviving CRAC spheres adopt a hybrid E/M phenotype in which epithelial and mesenchymal traits coexist. Such hybrid states confer both motility and adhesion, thereby enhancing stress tolerance and metastatic potential[24-26]. The downregulation of ZEB1, a key repressor of CDH1, suggests that SW480 spheres may partially revert to epithelial differentiation (mesenchymal-epithelial reverting transition), a reversible adaptation observed in several tumor models, including pancreatic and breast cancers[27-29]. This phenotypic plasticity provides a survival advantage: Cells can transiently modulate their adhesion and migration properties to withstand cytotoxic stress and subsequently repopulate after treatment. Thus, the hybrid E/M equilibrium observed in this study may represent an intermediate adaptive state between drug-sensitive epithelial cells and highly invasive mesenchymal cells, enabling the persistence of residual tumor spheres after chemotherapy.
Another notable aspect of this study was the dual evaluation of cell viability using ATP and WST-8 assays. Although ATP levels represent intracellular energy stores, WST-8 reduction reflects metabolic enzyme activity[18,19]. In all CRAC lines, except for 5-FU-treated SW480 cells, both parameters decreased in parallel. However, in SW480 spheres, ATP levels were maintained despite reduced WST-8 activity, indicating metabolic dormancy with preserved energy potential. This discrepancy is characteristic of drug-tolerant persister (DTP) cells, a slow-cycling subpopulation that survives cytotoxic stress via metabolic reprogramming rather than proliferation. DTP cells rely on mitochondrial oxidative phosphorylation to maintain low reactive oxygen species levels[30,31]. In CRC, 5-FU-resistant cells enter a metabolically suppressed but energy-sufficient state and escape apoptosis via mitochondrial compensation[16,32]. In our study, the preserved ATP in SW480 spheres may indicate a similar DTP-like phenotype. Their bowl-shaped structure under scanning electron microscopy, with necrotic centers and viable outer layers, supports the metabolic stratification typical of large 3D spheroids. Such dormant cells may later re-enter the cell cycle after drug removal, thereby contributing to relapse and metastasis.
Recent advances in patient-derived organoid (PDO) and patient-derived spheroid technologies have highlighted their ability to capture intratumoral heterogeneity and predict clinical drug response[15,33]. Notably, non-responder PDOs often exhibit increased stemness, metabolic reprogramming, and hybrid epithelial-mesenchymal features, closely resembling the adaptive phenotypes observed in our mesenchymal-type SW480 spheres. Although 3D cancer spheres do not fully recapitulate the architectural complexity of PDOs, they successfully reproduce key resistance-associated traits—including CD44/ALDH1A1 upregulation, partial EMT/mesenchymal-epithelial transition dynamics, and metabolic dormancy—while enabling more rapid and scalable functional assessment. From a translational perspective, these adaptive patterns mirror the biological characteristics reported in tumors with poor pathological response to neoadjuvant chemotherapy. Therefore, integrating sphere-derived EMT/CSC and metabolic signatures with organoid-based drug screening may help refine preoperative response prediction models and guide personalized chemotherapy selection.
In contrast, epithelial-type HCT-15 spheres showed reduced ATP and elevated caspase-3/7 activity, consistent with apoptosis activation. Epithelial cells generally exhibit higher apoptotic sensitivity, whereas mesenchymal cells tend to resist apoptosis through anti-apoptotic mechanisms, such as BCL2 and NF-κB pathway activation[34-36]. These observations highlight that the balance between energy conservation and apoptosis induction fundamentally differs between epithelial and mesenchymal phenotypes, reflecting their divergent stress response strategies.
The qRT-PCR analysis revealed that all three major CSC markers—CD44, ALDH1A1, and PROM1—were significantly upregulated in HCT-15 cells after drug exposure. In contrast, SW480 spheres showed a marked increase in CD44 and ALDH1A1 but a decrease in PROM1 expression. This pattern suggests that chemotherapy selectively enriches distinct CSC subpopulations, depending on the intrinsic cell phenotype. Their increased expression indicates that chemotherapy may not only fail to eradicate CSC-like cells but may also promote dedifferentiation into more resistant states. The significant 120-fold induction of CD44 expression in 5-FU-treated SW480 spheres underscores the ability of mesenchymal cells to enhance survival via CD44-mediated signaling. Conversely, PROM1 reduction may signify a transition from classical CD133+ stem-like progenitors toward a CD44+/ALDH1+ drug-tolerant phenotype, consistent with therapy-induced CSC reprogramming. Taken together, these findings suggest that surviving CRAC spheres acquire overlapping CSC and hybrid EMT features, creating a reservoir of adaptable, self-renewing cells capable of repopulating tumors after treatment.
The persistence of small, metabolically suppressed but viable spheres after chemotherapy may correspond to the residual tumor foci often observed in patients following neoadjuvant therapy. These cell populations maintain minimal metabolism and energy reserves, enabling survival under drug exposure and regrowth once therapy ceases[37,38]. Importantly, epithelial- and mesenchymal-type CRAC cells followed distinct adaptive trajectories; epithelial cells tended to undergo partial EMT, whereas mesenchymal cells showed partial reversal of epithelial traits. This bidirectional adaptability underscores the concept of epithelial-mesenchymal equilibrium, a hallmark of tumor plasticity, aggressiveness, and therapeutic resistance[39].
Clinically, these insights suggest that combining cytotoxic chemotherapy with agents that target plasticity and metabolic adaptation may improve treatment efficacy. Approaches that inhibit oxidative phosphorylation, block CD44 signaling, or limit EMT dynamics can prevent the survival of dormant residual cells. Moreover, 3D-culture-based models, as employed here, provide a robust preclinical platform to evaluate such combination therapies under physiological conditions.
In summary, anticancer drugs induce complex, phenotype-dependent responses in 3D-cultured CRC spheres. Surviving residual spheres display coordinated alterations in EMT, metabolism, and CSC marker expression, reflecting a hybrid E/M, stem-like, and metabolically adaptive state. This multifaceted plasticity allows tumor cells to persist under chemotherapy and may underlie recurrence. Future studies using PDOs, single-cell transcriptomics, and metabolic flux analyses will clarify the molecular mechanisms that drive this adaptive equilibrium. Therapeutic strategies that simultaneously target metabolic reprogramming and cellular plasticity may offer new avenues for overcoming chemoresistance in CRC.
Anticancer drugs induce marked morphological and functional alterations in 3D-cultured CRC spheres. Surviving residual spheres exhibit distinct CSC and EMT expression patterns, depending on the epithelial or mesenchymal subtype. These findings suggest that residual tumor cells with hybrid E/M characteristics may represent a key source of therapeutic resistance and tumor recurrence.
We thank Kazuo Onishi (SCREEN Holdings Co., Ltd.) for his technical assistance with image analysis using the Cell3iMager duos2.
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53448] [Cited by in RCA: 56090] [Article Influence: 7011.3] [Reference Citation Analysis (10)] |
| 2. | Pilleron S, Soto-Perez-de-Celis E, Vignat J, Ferlay J, Soerjomataram I, Bray F, Sarfati D. Estimated global cancer incidence in the oldest adults in 2018 and projections to 2050. Int J Cancer. 2021;148:601-608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 285] [Cited by in RCA: 226] [Article Influence: 45.2] [Reference Citation Analysis (0)] |
| 3. | Guo M, Jin N, Pawlik T, Cloyd JM. Neoadjuvant chemotherapy for colorectal liver metastases: A contemporary review of the literature. World J Gastrointest Oncol. 2021;13:1043-1061. [PubMed] [DOI] [Full Text] |
| 4. | Bhudia J, Glynne-Jones R, Smith T, Hall M. Neoadjuvant Chemotherapy without Radiation in Colorectal Cancer. Clin Colon Rectal Surg. 2020;33:287-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Dave B, Mittal V, Tan NM, Chang JC. Epithelial-mesenchymal transition, cancer stem cells and treatment resistance. Breast Cancer Res. 2012;14:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 206] [Cited by in RCA: 202] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 6. | Raimondi C, Gianni W, Cortesi E, Gazzaniga P. Cancer stem cells and epithelial-mesenchymal transition: revisiting minimal residual disease. Curr Cancer Drug Targets. 2010;10:496-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Esposito A, Ferraresi A, Vallino L, Garavaglia B, Dhanasekaran DN, Isidoro C. Three-Dimensional In Vitro Cell Cultures as a Feasible and Promising Alternative to Two-Dimensional and Animal Models in Cancer Research. Int J Biol Sci. 2024;20:5293-5311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 8. | Edmondson R, Broglie JJ, Adcock AF, Yang L. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay Drug Dev Technol. 2014;12:207-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2122] [Cited by in RCA: 1720] [Article Influence: 143.3] [Reference Citation Analysis (1)] |
| 9. | Friedrich J, Seidel C, Ebner R, Kunz-Schughart LA. Spheroid-based drug screen: considerations and practical approach. Nat Protoc. 2009;4:309-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1518] [Cited by in RCA: 1283] [Article Influence: 75.5] [Reference Citation Analysis (3)] |
| 10. | Han SJ, Kwon S, Kim KS. Challenges of applying multicellular tumor spheroids in preclinical phase. Cancer Cell Int. 2021;21:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 360] [Cited by in RCA: 295] [Article Influence: 59.0] [Reference Citation Analysis (0)] |
| 11. | Costa EC, Moreira AF, de Melo-Diogo D, Gaspar VM, Carvalho MP, Correia IJ. 3D tumor spheroids: an overview on the tools and techniques used for their analysis. Biotechnol Adv. 2016;34:1427-1441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 811] [Cited by in RCA: 635] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 12. | Weiswald LB, Bellet D, Dangles-Marie V. Spherical cancer models in tumor biology. Neoplasia. 2015;17:1-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 996] [Cited by in RCA: 846] [Article Influence: 76.9] [Reference Citation Analysis (0)] |
| 13. | Shinji S, Ogawa Y, Yamada T, Matsuda A, Uehara K, Yokoyama Y, Takahashi G, Iwai T, Miyasaka T, Kanaka S, Hayashi K, Shichi Y, Fujiwara M, Takahashi K, Arai T, Ishiwata T, Yoshida H. Morphological and functional analysis of colorectal cancer cell lines in 2D and 3D culture models. Sci Rep. 2025;15:3047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Minami F, Sasaki N, Shichi Y, Gomi F, Michishita M, Ohkusu-Tsukada K, Toyoda M, Takahashi K, Ishiwata T. Morphofunctional analysis of human pancreatic cancer cell lines in 2- and 3-dimensional cultures. Sci Rep. 2021;11:6775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 15. | van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, van Houdt W, van Gorp J, Taylor-Weiner A, Kester L, McLaren-Douglas A, Blokker J, Jaksani S, Bartfeld S, Volckman R, van Sluis P, Li VS, Seepo S, Sekhar Pedamallu C, Cibulskis K, Carter SL, McKenna A, Lawrence MS, Lichtenstein L, Stewart C, Koster J, Versteeg R, van Oudenaarden A, Saez-Rodriguez J, Vries RG, Getz G, Wessels L, Stratton MR, McDermott U, Meyerson M, Garnett MJ, Clevers H. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell. 2015;161:933-945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2105] [Cited by in RCA: 1860] [Article Influence: 169.1] [Reference Citation Analysis (5)] |
| 16. | Kurasaka C, Nishizawa N, Ogino Y, Sato A. Anticancer sensitivity and biological aspect of 5-fluorouracil-resistant human colorectal cancer HCT116 cells in three-dimensional culture under high- and low-glucose conditions. Nucleosides Nucleotides Nucleic Acids. 2024;43:870-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | Shichi Y, Gomi F, Hasegawa Y, Nonaka K, Shinji S, Takahashi K, Ishiwata T. Artificial intelligence-based analysis of time-lapse images of sphere formation and process of plate adhesion and spread of pancreatic cancer cells. Front Cell Dev Biol. 2023;11:1290753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 18. | Crouch SP, Kozlowski R, Slater KJ, Fletcher J. The use of ATP bioluminescence as a measure of cell proliferation and cytotoxicity. J Immunol Methods. 1993;160:81-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 741] [Cited by in RCA: 659] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 19. | Ishiyama M, Miyazono Y, Sasamoto K, Ohkura Y, Ueno K. A highly water-soluble disulfonated tetrazolium salt as a chromogenic indicator for NADH as well as cell viability. Talanta. 1997;44:1299-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 594] [Cited by in RCA: 510] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 20. | Urtasun N, Vidal-Pla A, Pérez-Torras S, Mazo A. Human pancreatic cancer stem cells are sensitive to dual inhibition of IGF-IR and ErbB receptors. BMC Cancer. 2015;15:223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 21. | Hagemann J, Jacobi C, Gstoettner S, Welz C, Schwenk-Zieger S, Stauber R, Strieth S, Kuenzel J, Baumeister P, Becker S. Therapy Testing in a Spheroid-based 3D Cell Culture Model for Head and Neck Squamous Cell Carcinoma. J Vis Exp. 2018;57012. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 22. | Chen YC, Lou X, Zhang Z, Ingram P, Yoon E. High-Throughput Cancer Cell Sphere Formation for Characterizing the Efficacy of Photo Dynamic Therapy in 3D Cell Cultures. Sci Rep. 2015;5:12175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 87] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 23. | Endoh D, Ishii K, Kohno K, Virgona N, Miyakoshi Y, Yano T, Ishida T. Chemoresistance related to hypoxia adaptation in mesothelioma cells from tumor spheroids. Exp Oncol. 2022;44:121-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Jolly MK, Boareto M, Huang B, Jia D, Lu M, Ben-Jacob E, Onuchic JN, Levine H. Implications of the Hybrid Epithelial/Mesenchymal Phenotype in Metastasis. Front Oncol. 2015;5:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 590] [Cited by in RCA: 534] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 25. | Liao TT, Yang MH. Hybrid Epithelial/Mesenchymal State in Cancer Metastasis: Clinical Significance and Regulatory Mechanisms. Cells. 2020;9:623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 26. | Tedja R, Alvero AB, Fox A, Cardenas C, Pitruzzello M, Chehade H, Bawa T, Adzibolosu N, Gogoi R, Mor G. Generation of Stable Epithelial-Mesenchymal Hybrid Cancer Cells with Tumorigenic Potential. Cancers (Basel). 2023;15:684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 27. | Chao YL, Shepard CR, Wells A. Breast carcinoma cells re-express E-cadherin during mesenchymal to epithelial reverting transition. Mol Cancer. 2010;9:179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 331] [Cited by in RCA: 318] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 28. | Reichert M, Bakir B, Moreira L, Pitarresi JR, Feldmann K, Simon L, Suzuki K, Maddipati R, Rhim AD, Schlitter AM, Kriegsmann M, Weichert W, Wirth M, Schuck K, Schneider G, Saur D, Reynolds AB, Klein-Szanto AJ, Pehlivanoglu B, Memis B, Adsay NV, Rustgi AK. Regulation of Epithelial Plasticity Determines Metastatic Organotropism in Pancreatic Cancer. Dev Cell. 2018;45:696-711.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 114] [Article Influence: 14.3] [Reference Citation Analysis (4)] |
| 29. | Sommariva M, Gagliano N. E-Cadherin in Pancreatic Ductal Adenocarcinoma: A Multifaceted Actor during EMT. Cells. 2020;9:1040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (4)] |
| 30. | Li Y, Chen H, Xie X, Yang B, Wang X, Zhang J, Qiao T, Guan J, Qiu Y, Huang YX, Tian D, Yao X, Lu D, Koeffler HP, Zhang Y, Yin D. PINK1-Mediated Mitophagy Promotes Oxidative Phosphorylation and Redox Homeostasis to Induce Drug-Tolerant Persister Cancer Cells. Cancer Res. 2023;83:398-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 104] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 31. | Zhang Z, Tan Y, Huang C, Wei X. Redox signaling in drug-tolerant persister cells as an emerging therapeutic target. EBioMedicine. 2023;89:104483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 57] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 32. | Pranzini E, Pardella E, Muccillo L, Leo A, Nesi I, Santi A, Parri M, Zhang T, Uribe AH, Lottini T, Sabatino L, Caselli A, Arcangeli A, Raugei G, Colantuoni V, Cirri P, Chiarugi P, Maddocks ODK, Paoli P, Taddei ML. SHMT2-mediated mitochondrial serine metabolism drives 5-FU resistance by fueling nucleotide biosynthesis. Cell Rep. 2022;40:111233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 77] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 33. | Neal JT, Li X, Zhu J, Giangarra V, Grzeskowiak CL, Ju J, Liu IH, Chiou SH, Salahudeen AA, Smith AR, Deutsch BC, Liao L, Zemek AJ, Zhao F, Karlsson K, Schultz LM, Metzner TJ, Nadauld LD, Tseng YY, Alkhairy S, Oh C, Keskula P, Mendoza-Villanueva D, De La Vega FM, Kunz PL, Liao JC, Leppert JT, Sunwoo JB, Sabatti C, Boehm JS, Hahn WC, Zheng GXY, Davis MM, Kuo CJ. Organoid Modeling of the Tumor Immune Microenvironment. Cell. 2018;175:1972-1988.e16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1311] [Cited by in RCA: 1145] [Article Influence: 143.1] [Reference Citation Analysis (4)] |
| 34. | Keitel U, Scheel A, Thomale J, Halpape R, Kaulfuß S, Scheel C, Dobbelstein M. Bcl-xL mediates therapeutic resistance of a mesenchymal breast cancer cell subpopulation. Oncotarget. 2014;5:11778-11791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 35. | An J, Lv J, Li A, Qiao J, Fang L, Li Z, Li B, Zhao W, Chen H, Wang L. Constitutive expression of Bcl-2 induces epithelial-Mesenchymal transition in mammary epithelial cells. BMC Cancer. 2015;15:476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 36. | Dieguez-Acuña FJ, Polk WW, Ellis ME, Simmonds PL, Kushleika JV, Woods JS. Nuclear factor kappaB activity determines the sensitivity of kidney epithelial cells to apoptosis: implications for mercury-induced renal failure. Toxicol Sci. 2004;82:114-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 37. | Liu S, Jiang A, Tang F, Duan M, Li B. Drug-induced tolerant persisters in tumor: mechanism, vulnerability and perspective implication for clinical treatment. Mol Cancer. 2025;24:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 38. | He J, Qiu Z, Fan J, Xie X, Sheng Q, Sui X. Drug tolerant persister cell plasticity in cancer: A revolutionary strategy for more effective anticancer therapies. Signal Transduct Target Ther. 2024;9:209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 62] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 39. | Lu J, Kornmann M, Traub B. Role of Epithelial to Mesenchymal Transition in Colorectal Cancer. Int J Mol Sci. 2023;24:14815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 100] [Article Influence: 33.3] [Reference Citation Analysis (0)] |