Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.115719
Revised: November 6, 2025
Accepted: January 19, 2026
Published online: April 15, 2026
Processing time: 167 Days and 8.2 Hours
Microsatellite instability (MSI) is a molecular subtype of gastric cancer caused by DNA mismatch repair defects, leading to mutations and neoantigen production. This profile influences tumor behavior, prognosis, and response to therapy, making it important for surgical decision-making. This article reviews the mole
Core Tip: This is an overview of the biological foundations, molecular mechanisms, diagnostic approaches, and clinical relevance of microsatellite instability in gastric cancer, emphasizing its role as a prognostic biomarker. The text also discusses therapeutic implications, including personalized surgical decision-making, treatment stratification, and the growing importance of integrating immunotherapy within multidisciplinary oncologic care.
- Citation: de Assumpção PP, Ishak G, de Assumpção PB, Casseb SMM, Acioli JF, Ramos SAA, Silva JMC, Guimarães KSP, de Assumpção CB, Kassab P, Mansfield P. What does the surgeon need to know about microsatellite instability in gastric cancers? World J Gastrointest Oncol 2026; 18(4): 115719
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/115719.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.115719
The increasing application of molecular biology to clinical practice is a welcome change to the landscape of therapeutic tools for our patients with gastric cancer. These changes require the surgeon to become more familiar with many of these innovations and terms[1-3]. Among gastric cancer surgeons, the molecular classification proposed by The Cancer Genome Atlas (TCGA) brought to the forefront the importance of fully understanding these new aspects of the surgeon's decision matrix.
Despite growing evidence on the molecular characterization of gastric cancer, there is a critical gap in literature specifically addressing the practical implications of microsatellite instability (MSI) for surgical decision-making, highlighting the need for a surgeon-oriented synthesis that bridges molecular oncology and operative management.
In this review, we aim to synthesize current evidence on the molecular basis, diagnostic strategies, and clinical implications of MSI in gastric cancer, emphasizing its impact on surgical decision-making and multidisciplinary management.
DNA replication is a dynamic process based on the capacity of an enzyme – DNA polymerase, to add specific nucleotides according to a template of DNA creating a complementary strand of the double helix DNA[4,5]. The process of adding nucleotides to the nascent strand of DNA has been compared to typing a text according to a template to be read almost in real time. Sometimes, typeset errors occur[6].
There are only four nucleotides composing DNA, these are T, C, A, and G. Since DNA is a double strand, each nucleotide is coupled with one specific partner. While working on DNA duplication, the polymerases need to add the correct partner to each nucleotide, i.e., if there is a T, the enzyme needs to add an A; if it is a G, a C will be added, in a very dynamic process[7].
Due to the sheer volume of events and the speed required, errors will occur, and a wrong nucleotide will be added. In such cases, one of the “quality control” systems often takes over[6]. The DNA repair system related to MSI cancers is composed of proteins that are able to identify errors and replace nucleotides with the correct ones[8,9].
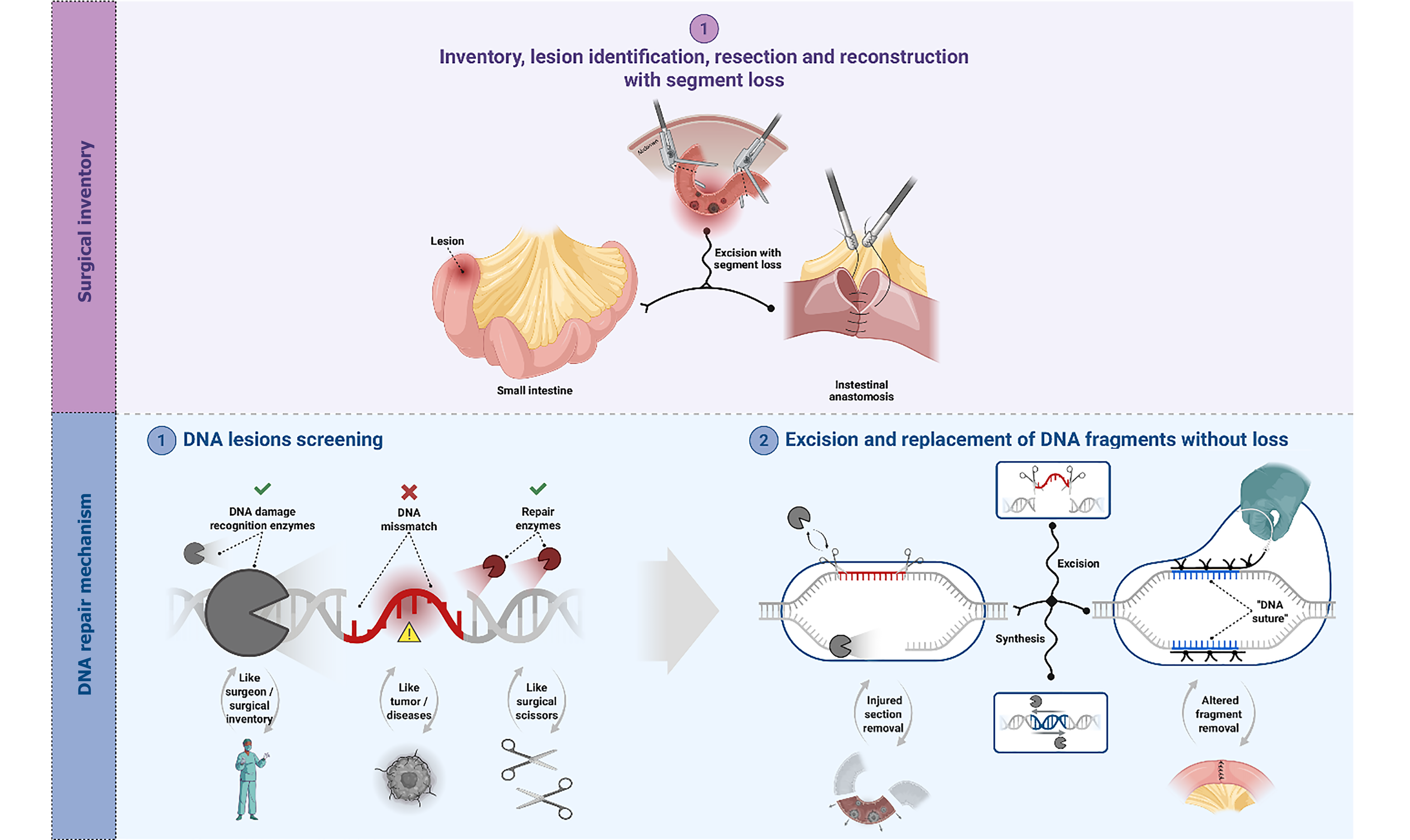
The DNA repair mechanism can be seen as analogous to a typical surgical procedure. This comparison is schematically illustrated in Figure 1, which shows similarities between the molecular repair machinery and surgical conduct in their operational mechanisms.
The surgeons usually perform an inventory to check every potential lesion in the abdominal cavity and if they identify an abnormality, like a bowel segment inflammation or tumor, this will be removed, and the remaining segments will be reconnected by an anastomosis, leading to an elimination of the “error” and reconstruction of the intestinal “sequence”, with a loss of the resected segment.
In the case of DNA repair mechanism, the involved proteins will check the DNA, find the error, remove it, re-synthase the correct DNA sequence, and insert de correct nucleotides into the DNA sequence, in this case, restoring it without loss of any DNA segment. Let’s wait for such evolution of not losing organs segments in the surgical field.
Using this analogy, the consequences of failing to identify and/or adequately correct any abdominal “error”, by the surgeon, may result in a wide possibility of troubles that may or may not threaten the patient’s health, according to what was lost.
Similarly, if the DNA repair fails in identifying and correct the DNA mismatches, the consequences for the cell, and for the patients’ health, will depend on what was lost. Fortunately, most of non-repaired DNA errors falls in regions of the DNA that are not transcribed or, if so, do not threaten the cell and the patients’ health. Actually, at each cell division a few DNA unrepaired errors persist, but usually do not cause any trouble.
The frequency of such unrepaired errors are considerable, even having an effective DNA repair apparatus, since the amount of DNA to be checked and, if necessary, removed, re-synthase and correctly relinked to the DNA sequence is enormous.
The continuous hard work of DNA synthesis, checking, and fixing errors can be compared to the production of an important textbook - the life textbook, in this case. Deletion and retyping events during the production of this long text occurs frequently, similarly to the errors generated in DNA replication that need to be identified and be restored by the DNA repair machinery. Should this system malfunction, either because it is unable to recognize the error or replace it with the correct one, the new DNA strand will retain the “wrong sequence”, just like the “wrong words or sentences” in a sent text.
Fortunately, as discussed, most such errors, although occurring in every cycle of DNA replication, rarely will cause troubles, as these “typeset” errors seldom provoke misinterpretation of the text meaning. Nonetheless, if the mismatched DNA base results in a significant modification of the encoded protein, a new epitope may be created which is different from those recognized by the immune system as “self”, may be produced[10,11].
MSI high gastric cancers are characterized by the incompetence of this DNA repair system, resulting in the production of plenty of new antigens that the immune system will recognize as foreign[9]. One of the ways of checking the occurrence of such deficiency in the mismatch repair system used to be by looking at microsatellites. Since microsatellites are repetitions of nucleotide sequences, they are prone to errors during the polymerase’s activity. While reading and typing a code, like the barcode numbers of a bank document, large repetitions, such as six or seven consecutive zeros, favor “typesetting” mistakes, like mismatch errors in microsatellites.
Backing to the surgical field, there are many signs that helps the surgeon to suspect that something is wrong in the abdominal cavity, like tachycardia, abdominal pain, distension and reaction, among others. These are not the cause of the real problem but guides the surgeon to identify the problem.
Similarly, to the cited concomitant but not causal events of the abdominal injury, MSI, instead of representing a causal mechanism of this tumor type, refers to a strategy for identifying a subgroup of cancers resulting from mismatch repair gene deficiencies. Conversely to what the subgroup's name may suggest, the microsatellites, usually, do not cause a molecular event important to the carcinogenesis but rather represents just a consequence of the actual causal mechanism. And, provides an accessible way of inferring the occurrence of the subjacent causal mechanism – the inability to identify and/or correct a type of DNA error[4,9].
The diagnosis and classification of the named MSI tumors moved from checking microsatellites to verifying the expression of the mismatch repair proteins by immunohistochemistry. Nevertheless, the molecular gastric cancer type will still be named MSI[1,12].
The molecular event peculiar to the MSI gastric cancer type is a deficiency in the mechanism that reduces the persistence of errors during DNA synthesis[12].
Thus, if a pathology report notes “lacking expression of mismatch repair proteins”, one must consider that multiple neoantigens may be produced and can thus serve as targets for an unleashed immune system. This is usually good for the patient and represents a status of probable immunocompetence, and the prognosis is often better than for those with stability of microsatellites[8].
According to retrospective studies, gastric cancers with MSI high have a better prognosis and seem not to benefit from neoadjuvant fluoropyrimidines-based chemotherapies[2,3,13]. Upfront surgery has emerged as a viable option for such cases.
Another important molecular feature affecting the management of MSI tumors is the programmed death 1 (PD-1) pathway expression[14]. Although the neoantigens created in MSI cancers can generate an immune response, some tumor cells escape immune destruction, taking advantage of a mechanism that regulates immune responses. These control mechanisms avoid excessive responses that might harm normal cells[12]. This is the case of the PD-1 pathway. Tumor cells that express or induce expression of programmed death ligand-1 (PD-L1), which is a PD-1 binding protein, activates this break of the immune response, resulting in the avoidance of these cancer cells’ immune destruction[15].
When an MSI high tumor escapes the immune surveillance that was triggered by the neoantigens produced due to the deficiency in mismatch repair genes, by overexpression of the PD-1 pathway, this is not necessarily the end of the story. Antibodies against PD-1 or anti-PD-L1 can avoid the activation of the PD-1 pathway by the cancer cells and improves clinical outcomes. In this case, science, and the patient may be the final winner. These antibodies are currently indicated for MSI-high (MSI-H) gastric cancers, according to approved protocols[16].
The molecular discoveries, while translated to clinical practice, deeply impact the medical management, including the surgical strategies. The current understanding of the mechanisms and impacts of MSI-H strongly modified traditional approaches including surgical management of these tumors.
The recently proposed modification on the indications of fluoropyrimidines-based neoadjuvant chemotherapy for MSI-type gastric cancers, is one of these novelties requiring moving the surgeons to increase their molecular understanding[1-4].
According to recent guidelines, these drugs may be avoided and replaced by upfront surgery, due to lack of clinical benefit in such molecular GC type, impacting clinical management.
In addition, immunotherapy is now indicated for treating MSI-H GC, including as a neoadjuvant approach[17].
Another potential application among the surgical treatment strategies is to considerer a less extended lymphadenectomy, since MSI-H gastric tumors may exempt radical lymphadenectomy in selected cases.
In many areas of the world, the surgeons are often the team leader in gastric cancer management. Therefore, surgeons must have a full comprehension of these new “molecular players” as relevant medical decisions will continue to evolve.
Among the potential future directions, avoiding surgery in cases of MSI-H tumors treated preoperatively and achieving complete pathological response are on the horizon. On the other hand, cases currently considered outside surgical therapeutic indications due to advanced stage may become potentially operable after immunotherapy. Between these extreme examples, tailoring each GC case to molecular features is a need that requires the surgeon, as team leader, to be involved.
MSI gastric cancer is the TCGA molecular type that has led to most of the current modifications in GC clinical management. This has impacted both the surgeon’s and the clinical oncologist's approaches to many patients, aiming for more favorable patient outcomes. Understanding the new and evolving molecular landscape and incorporating this knowledge into surgical oncology will benefit multidisciplinary teams. This will promote more harmonious decision-making in real-world practice and must be part of the GC surgeon’s repertoire.
| 1. | Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Cooke D, Corvera C, Das P, Enzinger PC, Enzler T, Fanta P, Farjah F, Gerdes H, Gibson MK, Hochwald S, Hofstetter WL, Ilson DH, Keswani RN, Kim S, Kleinberg LR, Klempner SJ, Lacy J, Ly QP, Matkowskyj KA, McNamara M, Mulcahy MF, Outlaw D, Park H, Perry KA, Pimiento J, Poultsides GA, Reznik S, Roses RE, Strong VE, Su S, Wang HL, Wiesner G, Willett CG, Yakoub D, Yoon H, McMillian N, Pluchino LA. Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022;20:167-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1229] [Article Influence: 307.3] [Reference Citation Analysis (5)] |
| 2. | Kohlruss M, Grosser B, Krenauer M, Slotta-Huspenina J, Jesinghaus M, Blank S, Novotny A, Reiche M, Schmidt T, Ismani L, Hapfelmeier A, Mathias D, Meyer P, Gaida MM, Bauer L, Ott K, Weichert W, Keller G. Prognostic implication of molecular subtypes and response to neoadjuvant chemotherapy in 760 gastric carcinomas: role of Epstein-Barr virus infection and high- and low-microsatellite instability. J Pathol Clin Res. 2019;5:227-239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 65] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 3. | Smyth EC. Chemotherapy for resectable microsatellite instability-high gastric cancer? Lancet Oncol. 2020;21:204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 4. | Smyth EC, Wotherspoon A, Peckitt C, Gonzalez D, Hulkki-Wilson S, Eltahir Z, Fassan M, Rugge M, Valeri N, Okines A, Hewish M, Allum W, Stenning S, Nankivell M, Langley R, Cunningham D. Mismatch Repair Deficiency, Microsatellite Instability, and Survival: An Exploratory Analysis of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) Trial. JAMA Oncol. 2017;3:1197-1203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 411] [Article Influence: 45.7] [Reference Citation Analysis (5)] |
| 5. | Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, Liu T, Schenker M, Yanez P, Tehfe M, Kowalyszyn R, Karamouzis MV, Bruges R, Zander T, Pazo-Cid R, Hitre E, Feeney K, Cleary JM, Poulart V, Cullen D, Lei M, Xiao H, Kondo K, Li M, Ajani JA. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2501] [Cited by in RCA: 2274] [Article Influence: 454.8] [Reference Citation Analysis (5)] |
| 6. | Song HY, Shen R, Mahasin H, Guo YN, Wang DG. DNA replication: Mechanisms and therapeutic interventions for diseases. MedComm (2020). 2023;4:e210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 7. | Watson JD, Crick FH. Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid. Nature. 1953;171:737-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10351] [Cited by in RCA: 5878] [Article Influence: 80.5] [Reference Citation Analysis (1)] |
| 8. | Li K, Luo H, Huang L, Luo H, Zhu X. Microsatellite instability: a review of what the oncologist should know. Cancer Cell Int. 2020;20:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 421] [Cited by in RCA: 360] [Article Influence: 60.0] [Reference Citation Analysis (6)] |
| 9. | Zhang Z, Liu Z, Gu Y, Luo R, Tang Z, Sun Y, Wang X. Microsatellite instability in gastric cancer: molecular features and clinical implications. Clin Cancer Bull. 2024;3:12. [DOI] [Full Text] |
| 10. | Hopkins JL, Lan L, Zou L. DNA repair defects in cancer and therapeutic opportunities. Genes Dev. 2022;36:278-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 149] [Article Influence: 37.3] [Reference Citation Analysis (1)] |
| 11. | Li GM. Mechanisms and functions of DNA mismatch repair. Cell Res. 2008;18:85-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1196] [Cited by in RCA: 1027] [Article Influence: 57.1] [Reference Citation Analysis (4)] |
| 12. | Yamamoto H, Watanabe Y, Arai H, Umemoto K, Tateishi K, Sunakawa Y. Microsatellite instability: A 2024 update. Cancer Sci. 2024;115:1738-1748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 13. | Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord JP, Geva R, Gottfried M, Penel N, Hansen AR, Piha-Paul SA, Doi T, Gao B, Chung HC, Lopez-Martin J, Bang YJ, Frommer RS, Shah M, Ghori R, Joe AK, Pruitt SK, Diaz LA Jr. Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol. 2020;38:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2532] [Cited by in RCA: 2268] [Article Influence: 378.0] [Reference Citation Analysis (9)] |
| 14. | Formica V, Morelli C, Fornaro L, Riondino S, Rofei M, Fontana E, Smyth EC, Roselli M, Arkenau HT. PD-L1 thresholds predict efficacy of immune checkpoint inhibition in first-line treatment of advanced gastroesophageal adenocarcinoma. A systematic review and meta-analysis of seven phase III randomized trials. ESMO Open. 2024;9:103967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 15. | Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11654] [Cited by in RCA: 10726] [Article Influence: 766.1] [Reference Citation Analysis (6)] |
| 16. | Huo G, Liu W, Chen P. Efficacy of PD-1/PD-L1 inhibitors in gastric or gastro-oesophageal junction cancer based on clinical characteristics: a meta-analysis. BMC Cancer. 2023;23:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 17. | Janjigian YY, Al-Batran SE, Wainberg ZA, Muro K, Molena D, Van Cutsem E, Hyung WJ, Wyrwicz L, Oh DY, Omori T, Moehler M, Garrido M, Oliveira SCS, Liberman M, Oliden VC, Smyth EC, Stein A, Bilici M, Alvarenga ML, Kozlov V, Rivera F, Kawazoe A, Serrano O, Heilbron E, Negro A, Kurland JF, Tabernero J; MATTERHORN Investigators. Perioperative Durvalumab in Gastric and Gastroesophageal Junction Cancer. N Engl J Med. 2025;393:217-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 168] [Article Influence: 168.0] [Reference Citation Analysis (1)] |