Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.117083
Revised: December 14, 2025
Accepted: January 4, 2026
Published online: March 15, 2026
Processing time: 105 Days and 4.3 Hours
Helicobacter pylori (H. pylori)-negative early gastric cancer (GC) is an emerging clinical and epidemiological challenge that cannot be explained by the classical H. pylori-driven carcinogenic pathway. Growing evidence indicates that dietary factors play a critical role in modulating gastric mucosal integrity, inflammation, and carcinogenic progression in the absence of H. pylori infection. High salt intake and processed foods, particularly those rich in nitrates and nitrites, are associated with mucosal injury, oxidative stress, and endogenous N-nitroso compound for
Core Tip: Diet plays a critical role in the development of Helicobacter pylori (H. pylori)-negative early gastric cancer. High salt intake and processed foods increase carcinogenic risk, while fruits, vegetables, antioxidants, dietary fiber, and Mediterranean-style dietary patterns provide protective effects independent of H. pylori status. Understanding these dietary influences is essential for improving prevention strategies and refining risk assessment in H. pylori-negative populations.
- Citation: Fan D, Li RJ, Guo CY, Jiao Y. Dietary factors in Helicobacter pylori-negative early gastric cancer: Risk, protection, and mechanistic insights. World J Gastrointest Oncol 2026; 18(3): 117083
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/117083.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.117083
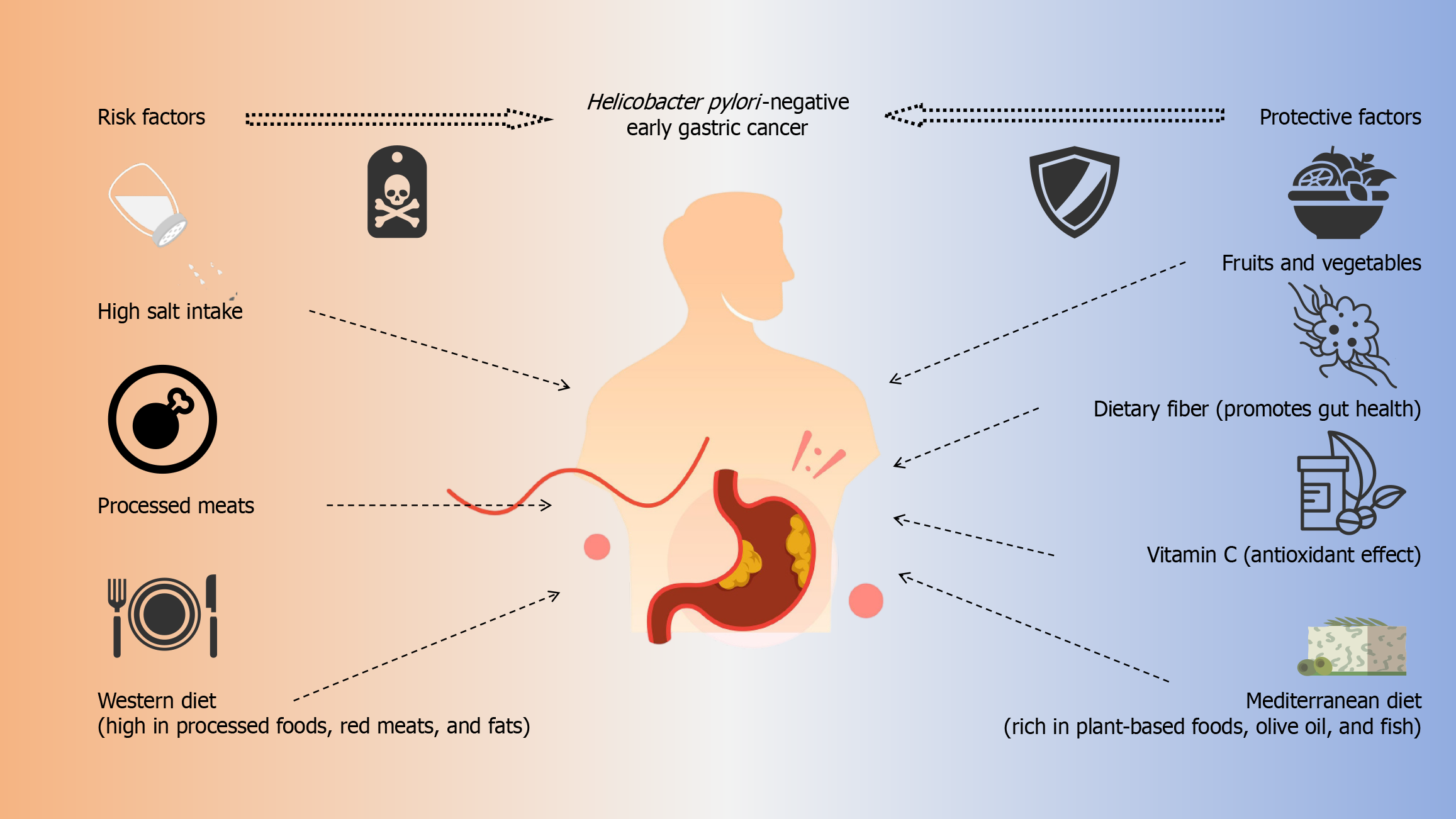
Gastric cancer (GC) is one of the leading causes of cancer-related deaths worldwide, despite significant decreases in incidence, particularly in high-income countries. However, in low- and middle-income countries, GC continues to represent a growing challenge, often linked to dietary habits, environmental exposures, and other lifestyle factors. Importantly, dietary habits in these settings are heterogeneous, and risk-promoting and protective dietary patterns may coexist within the same region or population. Helicobacter pylori (H. pylori) infection, a well-established risk factor for GC, has historically been central to the study of gastric carcinogenesis. Yet, the increasing recognition of H. pylori-negative early GC (EGC) has shifted the focus toward understanding additional factors contributing to GC development, particularly the role of dietary components. While H. pylori infection remains a key player in the pathogenesis of GC, a growing body of evidence has begun to explore how diet, particularly in individuals without H. pylori infection, modulates cancer risk. This minireview examines the critical dietary factors involved in H. pylori-negative EGC, synthesizing research findings related to the role of nutrients, dietary patterns, and specific food components (Figure 1; Table 1).
| Dietary component | Effect on gastric cancer risk |
| High salt intake | Increases risk; associated with mucosal damage and carcinogen promotion |
| Processed meats | Increases risk; linked to carcinogen formation |
| Fruits and vegetables | Protective effect; rich in antioxidants and micronutrients |
| Dietary fiber | Protective effect; aids in carcinogen excretion and gut health |
| Mediterranean diet | Protective effect; rich in anti-inflammatory foods |
| Western diet | Increases risk; high in processed foods, red meats, and fats |
| Vitamin C | Protective effect; reduces oxidative DNA damage |
| Vitamin D | Mixed evidence; some studies suggest protective effect |
One of the most consistently reported dietary risk factors for GC, including H. pylori-negative EGC, is high salt intake. Numerous studies have demonstrated a positive association between high dietary sodium and increased GC risk, regardless of H. pylori infection status[1,2]. Salt, particularly in preserved foods such as pickled vegetables, salted fish, and processed meats, has been linked to mucosal damage and the promotion of carcinogenic compounds such as N-nitroso compounds, with similar associations observed across different populations and dietary settings. Excessive salt exposure may directly impair gastric epithelial integrity and increase endogenous formation of N-nitroso compounds, thereby enhancing susceptibility to DNA damage and early malignant transformation. These compounds are produced during the preservation process and are thought to contribute to DNA damage in the gastric mucosa, which can lead to carcinogenesis[2,3].
In a large cohort study, Peleteiro et al[2] found that high salt intake was significantly associated with an increased risk of GC, even after adjusting for H. pylori infection status. Similarly, Takasu et al[1] identified salt as a prominent risk factor for GC, further highlighting its role in both H. pylori-positive and H. pylori-negative cases. This evidence underscores the importance of reducing salt intake as part of broader dietary interventions for GC prevention.
Conversely, diets rich in fruits and vegetables have consistently been associated with a reduced risk of GC, including H. pylori-negative EGC[4,5]. These foods are rich in antioxidants, fiber, and micronutrients, which have protective effects against oxidative stress and inflammation, two key contributors to gastric carcinogenesis. Antioxidant-rich diets may mitigate oxidative DNA damage and limit inflammation-driven epithelial injury, thereby reducing carcinogenic progression. This inverse association has been reported in diverse study populations, supporting a broadly protective role of plant-based dietary components. Beyond these general effects, such dietary components may also contribute to the maintenance of gastric mucosal barrier integrity and attenuation of chronic inflammatory signaling, thereby reducing vulnerability to malignant transformation.
Antioxidants such as vitamin C, vitamin E, and carotenoids (e.g., beta-carotene and lycopene) have shown inverse associations with GC risk, potentially through their ability to neutralize free radicals and prevent oxidative DNA damage[5,6]. However, reported effect sizes vary across studies, which may reflect differences in baseline nutritional status, dietary assessment methods, and population characteristics, rather than uniform biological effects. Moreover, dietary fiber, found abundantly in fruits, vegetables, and whole grains, has been shown to reduce the risk of GC by improving gut health and reducing exposure to carcinogens, with comparable trends observed across different geographic regions and study designs[4,5]. Fiber intake may further influence the gastric and intestinal microenvironment through mo
Dietary patterns, rather than isolated nutrients, have also been studied in relation to GC risk. The Mediterranean diet, characterized by high consumption of fruits, vegetables, legumes, whole grains, olive oil, and fish, has been consistently associated with a lower risk of GC, including in H. pylori-negative cases[7,8]. Although such dietary patterns are not universally adopted in low- and middle-income countries, they provide a useful reference framework for understanding how plant-based and anti-inflammatory dietary characteristics may confer protection against gastric carcinogenesis. This association appears to be relatively stable across populations with varying baseline dietary habits, suggesting that the protective effect is not confined to Mediterranean regions alone[9,10].
In contrast, Western dietary patterns, which are characterized by high consumption of red meat, processed foods, refined grains, and saturated fats, are associated with increased GC risk. The Western diet’s pro-inflammatory effects and high content of harmful fats and preservatives contribute to gastric carcinogenesis, both in H. pylori-positive and H. pylori-negative populations[1,11]. The association between Mediterranean dietary patterns and reduced GC risk was also supported by Castelló et al[10], who found that high adherence to Mediterranean and prudent diets, which share similarities in their focus on plant-based foods, was inversely related to GC risk. These findings emphasize the role of overall diet quality in modulating GC risk, independent of H. pylori status.
The potential role of micronutrients, including vitamin D, in GC prevention remains a topic of ongoing research. While vitamin D is known for its role in bone health, emerging evidence suggests it may also influence cancer-related pathways, including cell proliferation and immune regulation. These proposed mechanisms provide biological plausibility; however, inconsistent epidemiological findings indicate that such effects may be context-dependent rather than universal. Some studies have indicated that low vitamin D levels are associated with an increased risk of GC, particularly in regions with insufficient sunlight exposure[1,12].
However, the evidence on vitamin D’s protective effects against GC remains mixed. Several studies have reported null or inconsistent associations, suggesting that observed discrepancies may be driven by heterogeneity in study design, vitamin D exposure assessment, genetic background, sunlight availability, and residual confounding, rather than true biological contradiction. Consequently, current evidence does not allow a definitive conclusion regarding the role of vitamin D in H. pylori-negative gastric carcinogenesis.
Recent studies have explored the role of dietary inflammation in GC development. The Dietary Inflammatory Index (DII) is a tool used to assess the inflammatory potential of a diet based on the intake of specific nutrients and food groups. A higher DII score indicates a more pro-inflammatory diet, which has been linked to increased cancer risk, including GC[8,13]. Such pro-inflammatory dietary profiles may promote gastric carcinogenesis through sustained immune activation, oxidative stress, and disruption of epithelial homeostasis, even in the absence of H. pylori infection. This relationship has been observed across multiple populations, supporting the relevance of diet-induced inflammation beyond specific regional contexts. From a mechanistic perspective, pro-inflammatory dietary patterns may promote gastric carcinogenesis through sustained immune activation, oxidative stress, and disruption of epithelial homeostasis, even in the absence of H. pylori infection. Conversely, diets with lower DII scores tend to emphasize anti-inflammatory components that may counterbalance these processes[8].
While substantial evidence supports the role of diet in modulating GC risk, especially in H. pylori-negative populations, several methodological challenges remain. Differences in population characteristics, dietary assessment tools, and cultural eating patterns may partially account for variability in reported associations, highlighting the need for cautious interpretation of effect sizes. A major limitation in the current literature is the reliance on observational studies, which, despite their ability to identify associations, are prone to confounding and bias. In addition, overlapping dietary exposures and interrelated nutritional components complicate efforts to isolate individual effects, contributing to apparent inconsistencies across studies. These limitations underscore the importance of integrative analytical approaches that link dietary patterns with mechanistic biomarkers and stratified analyses, particularly in H. pylori-negative EGC[14,15].
Moreover, while dietary patterns such as the Mediterranean diet show promise in reducing GC risk, there is still considerable variability in how these patterns are defined and assessed. Standardized methodologies for evaluating dietary patterns are needed to improve comparability across studies and provide more robust evidence for dietary interventions. Finally, future research should focus on large-scale, longitudinal studies that explicitly stratify H. pylori-negative populations to better understand the long-term effects of dietary factors on GC development. Additionally, mechanistic studies are needed to explore the biological pathways through which specific nutrients and dietary patterns influence gastric carcinogenesis.
Dietary factors play a crucial role in the development of H. pylori-negative EGC. High salt intake, processed foods, and low consumption of fruits and vegetables have been consistently identified as risk factors, while diets rich in antioxidants, dietary fiber, and specific micronutrients offer protective effects. Mediterranean and other prudent dietary patterns have shown an inverse association with GC risk, independent of H. pylori infection. Despite these findings, significant gaps remain in understanding the precise mechanisms by which diet influences gastric carcinogenesis in H. pylori-negative populations. Future research should focus on clarifying these mechanisms and developing targeted dietary interventions to reduce GC risk in high-risk individuals.
| 1. | Takasu A, Gotoda T, Suzuki S, Kusano C, Goto C, Ishikawa H, Kogure H. Daily Diet and Nutrition Risk Factors for Gastric Cancer Incidence in a Japanese Population. Gut Liver. 2024;18:602-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 2. | Peleteiro B, Lopes C, Figueiredo C, Lunet N. Salt intake and gastric cancer risk according to Helicobacter pylori infection, smoking, tumour site and histological type. Br J Cancer. 2011;104:198-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 3. | Bouras E, Tsilidis KK, Triggi M, Siargkas A, Chourdakis M, Haidich AB. Diet and Risk of Gastric Cancer: An Umbrella Review. Nutrients. 2022;14:1764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 4. | Collatuzzo G, Cortez Lainez J, Pelucchi C, Negri E, Bonzi R, Palli D, Ferraroni M, Zhang ZF, Yu GP, Lunet N, Morais S, López-Carrillo L, Zaridze D, Maximovitch D, Guevara M, Santos-Sanchez V, Vioque J, Garcia de la Hera M, Ward MH, Malekzadeh R, Pakseresht M, Hernández-Ramírez RU, Turati F, Rabkin CS, Liao LM, Sinha R, López-Cervantes M, Tsugane S, Hidaka A, Camargo MC, Curado MP, Zubair N, Kristjansson D, Shah S, La Vecchia C, Boffetta P. The association between dietary fiber intake and gastric cancer: a pooled analysis of 11 case-control studies. Eur J Nutr. 2024;63:1857-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Lunet N, Valbuena C, Carneiro F, Lopes C, Barros H. Antioxidant vitamins and risk of gastric cancer: a case-control study in Portugal. Nutr Cancer. 2006;55:71-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Kim JH, Lee J, Choi IJ, Kim YI, Kwon O, Kim H, Kim J. Dietary Carotenoids Intake and the Risk of Gastric Cancer: A Case-Control Study in Korea. Nutrients. 2018;10:1031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 7. | Reytor-González C, Zambrano AK, Montalvan M, Frias-Toral E, Simancas-Racines A, Simancas-Racines D. Adherence to the Mediterranean Diet and its association with gastric cancer: health benefits from a Planeterranean perspective. J Transl Med. 2024;22:483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 8. | Luo MZ, Shu L, Ye XS. Association between priori and posteriori dietary patterns and gastric cancer risk: an updated systematic review and meta-analysis of observational studies. Eur J Cancer Prev. 2025;34:157-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 9. | Bai X, Li X, Ding S, Dai D. Adherence to the Mediterranean Diet and Risk of Gastric Cancer: A Systematic Review and Meta-Analysis. Nutrients. 2023;15:3826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 10. | Castelló A, Fernández de Larrea N, Martín V, Dávila-Batista V, Boldo E, Guevara M, Moreno V, Castaño-Vinyals G, Gómez-Acebo I, Fernández-Tardón G, Peiró R, Olmedo-Requena R, Capelo R, Navarro C, Pacho-Valbuena S, Pérez-Gómez B, Kogevinas M, Pollán M, Aragonés N; MCC-Spain researchers. High adherence to the Western, Prudent, and Mediterranean dietary patterns and risk of gastric adenocarcinoma: MCC-Spain study. Gastric Cancer. 2018;21:372-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 11. | Zhu Y, Jeong S, Wu M, Zhou JY, Jin ZY, Han RQ, Yang J, Zhang XF, Wang XS, Liu AM, Gu XP, Su M, Hu X, Sun Z, Li G, Yon Jung S, Li L, Mu L, Lu QY, La Vecchia C, Zhao JK, Zhang ZF. Index-based dietary patterns and stomach cancer in a Chinese population. Eur J Cancer Prev. 2021;30:448-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Kim JH, Lee J, Choi IJ, Kim YI, Kim J. Dietary patterns and gastric cancer risk in a Korean population: a case-control study. Eur J Nutr. 2021;60:389-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Zhai LL, Zhao SP, Yao P. [Meta-analysis of the Relationship between Dietary Inflammatory Index and Upper Gastrointestinal Cancer Risks]. Zhongguo Quanke Yixue. 2023;26:2286-2292, 2300. [DOI] [Full Text] |
| 14. | Horiuchi Y, Fujisaki J, Ishizuka N, Omae M, Ishiyama A, Yoshio T, Hirasawa T, Yamamoto Y, Nagahama M, Takahashi H, Tsuchida T. Study on Clinical Factors Involved in Helicobacter pylori-Uninfected, Undifferentiated-Type Early Gastric Cancer. Digestion. 2017;96:213-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 15. | Huang L, Chen L, Gui ZX, Liu S, Wei ZJ, Xu AM. Preventable lifestyle and eating habits associated with gastric adenocarcinoma: A case-control study. J Cancer. 2020;11:1231-1239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |