Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.117278
Revised: December 30, 2025
Accepted: January 22, 2026
Published online: March 15, 2026
Processing time: 99 Days and 10.8 Hours
Recent advances in esophageal sponge cytology tests highlight their potential in esophageal squamous cell carcinoma (ESCC) screening, but their cost-effectiveness remains unclear.
To investigate the cost-effectiveness of the sponge cytology test for ESCC scree
We built a Markov model simulating 100000 participants aged 45 years in ESCC high-risk areas to project the cost-effectiveness of several screening strategies: No screening, endoscopic screening, and sponge cytology screening (with subsequent endoscopy for positive results) with different intervals. Outcomes included ESCC cases and deaths, cost, quality-adjusted life year (QALY), and incremental cost-effective ratio (ICER).
All screening strategies reduced ESCC cases and prevented deaths compared with no screening (373-2962 vs 3134 and 257-2305 vs 2409 per 100000 participants, respectively). Shorter screening intervals were associated with higher QALYs gained per person (one-time to per year: 532-5972 for endoscopy and 747-7162 for sponge cytology test). An annual sponge cytology test strategy was considered the most cost-effective with the greatest QALYs gained and an ICER of 6630 USD per QALY among all strategies. Endoscopy screening strategies were dominated by the annual sponge cytology strategy.
Our findings suggest that the sponge cytology screening strategy is cost-effective in ESCC high-risk areas and may inform policy decision-making.
Core Tip: This study is the first economic evaluation of a minimally invasive sponge cytology screening strategy in areas at high risk for esophageal squamous cell carcinoma (ESCC). We found that both endoscopic and sponge cytology screening programs were highly cost-effective compared with no screening. Among all evaluated strategies, annual sponge cytology screening provided the greatest health benefits at the most favorable cost-effectiveness, indicating that it may represent an optimal population-based screening approach for ESCC in high-risk regions.
- Citation: Xu YS, Gao Y, Jiao YF, Yu CT, Wang W, Wang TL, Chu C, Lin H, Xu JF, Xin L, Wang LW. Cost-effectiveness of sponge cytology test vs endoscopy for esophageal squamous cell carcinoma screening in high-risk areas. World J Gastrointest Oncol 2026; 18(3): 117278
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/117278.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.117278
Esophageal cancer ranks 11th and 7th in terms of incidence and mortality rates among all cancer types worldwide, and of those, approximately 85% of esophageal cancer cases had squamous cell carcinoma[1,2]. Esophageal squamous cell carcinoma (ESCC) mostly occurs in high-risk areas, such as Eastern and South Central Asia and South Africa[2]. As it has an asymptomatic onset, most cases are detected at late stages. The survival rate remains low in most countries[3] (with 3-year survival rates lower than 30%), thus early detection and curable treatment are crucial to prognostic improvement[4].
One-time upper gastrointestinal endoscopy is the current standard method for ESCC screening. Its effectiveness in reducing esophageal cancer incidence and mortality in high-risk regions has been widely confirmed[5-7]. However, large-scale endoscopic screening faces significant challenges, including unequal resource distribution and unavoidable discomfort. Notably, the novel esophageal sponge cytology test has emerged as a promising pre-endoscopic screening tool due to its minimally invasive nature and operational simplicity. The procedure involves swallowing a compressed sponge encapsulated within a gelatin capsule and attached to a thin string; when the capsule dissolves, the sponge expands in the stomach and is gently withdrawn through the esophagus, enabling the collection of exfoliated esophageal epithelial cells for cytological and biomarker analysis[8]. This test can facilitate risk stratification by identifying high-risk individuals for prioritized endoscopic referral and has already been implemented in real-world screening programs in China[8,9].
A series of studies have been conducted to evaluate the economic benefits of the sponge cytology test in Barrett’s esophagus and gastroesophageal reflux disease screening and surveillance in the United Kingdom and United States[10-13]. However, economic evaluation research on the sponge cytology test in ESCC screening is notably absent. Given that variations in disease subtypes and healthcare systems can significantly influence cost-effectiveness outcomes, further research is needed to assess the value of sponge cytology in the context of ESCC. In light of the globally uneven geo
The primary objective of this study was to evaluate the cost-effectiveness of sponge cytology screening in ESCC high-risk areas. We examined a range of screening strategies with varying modalities and intervals to identify the most optimal approach under the modeled conditions and to provide further evidence for ESCC prevention and control efforts.
This economic evaluation study was conducted based on a Markov model using TreeAge Pro version 2024 R1.1 (TreeAge Software; Williamstown, MA, United States), and the analyses were conducted based on Excel version 2021(Microsoft Corporation, Redmond, WA, United States). This study was reported based on the Consolidated Health Economic Evaluation Reporting Standards reporting guideline[16]. Ethical approval was not required as no human participants were involved in this study.
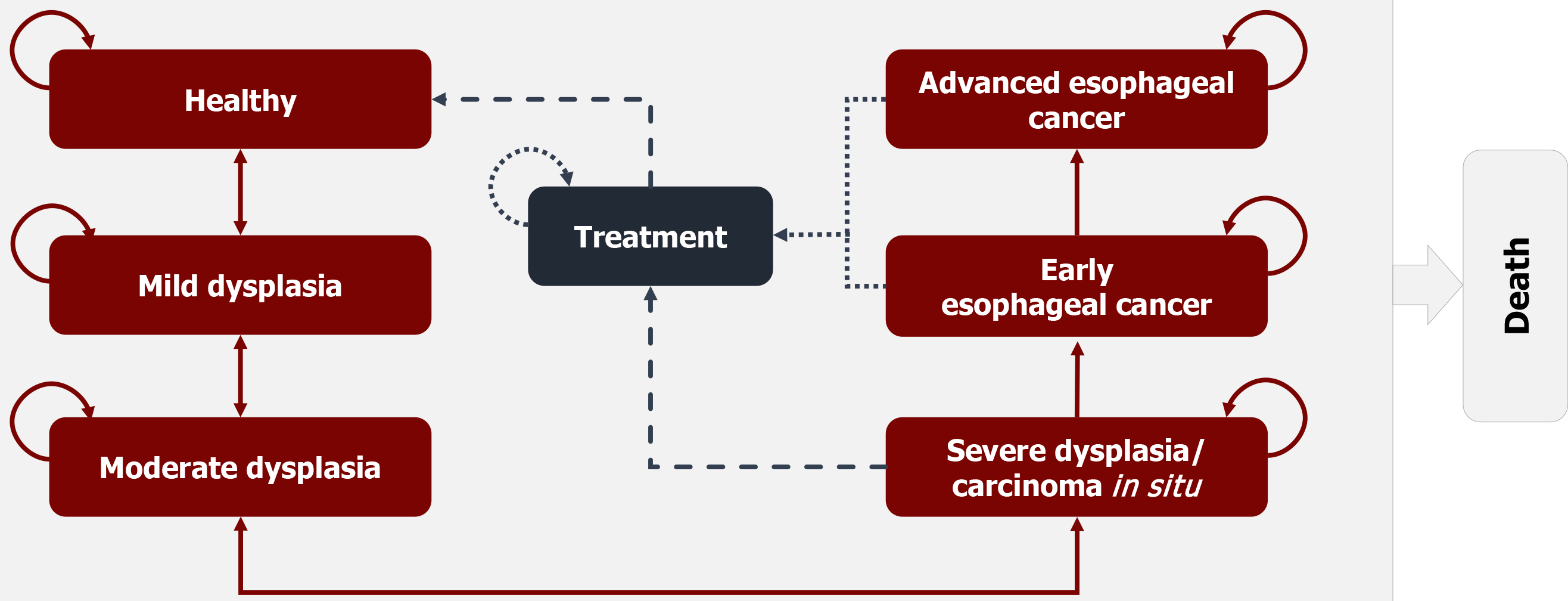
A Markov model was used in our analysis to simulate disease progression over time, providing a structured framework to evaluate the long-term outcomes and cost-effectiveness of different screening strategies. A hypothetical cohort of 100000 individuals aged 45 years residing in ESCC high-risk areas was projected. The natural history of ESCC is depicted in Figure 1. Initial prevalence and annual transition probabilities between health states are summarized in Table 1.
| Parameter | Base case | Range1 | Distribution2 | Ref. |
| Prevalence (%)3 | Zeng et al[14], 2020; Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| mD | 2.04 | 1.02-4.52 | Triangular (0.0102, 0.0204, 0.0452) | |
| MD | 0.22 | 0.11-0.59 | Triangular (0.0011, 0.0022, 0.0059) | |
| SD/CIS | 0.12 | 0.06-0.38 | Triangular (0.0006, 0.0012, 0.0038) | |
| EEC | 0.05 | 0.03-0.10 | Triangular (0.0003, 0.0005, 0.0010) | |
| AEC | 0.02 | 0.01-0.04 | Triangular (0.0001, 0.0002, 0.0004) | |
| Annual state transition rate4 | Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| Healthy to mD | 0.0120 | ± 50% | Triangular (0.006, 0.012, 0.018) | |
| mD to healthy | 0.0500 | ± 50% | Triangular (0.025, 0.05, 0.075) | |
| mD to MD | 0.0500 | ± 50% | Triangular (0.025, 0.05, 0.075) | |
| MD to mD | 0.0800 | ± 50% | Triangular (0.04, 0.08, 0.12) | |
| MD to SD/CIS | 0.1200 | ± 50% | Triangular (0.06, 0.12, 0.18) | |
| SD/CIS to MD | Age-specific | |||
| SD/CIS to EEC | ||||
| EEC to AEC | ||||
| PT-SD/CIS to SD/CIS | 0.0050 | ± 50% | Triangular (0.0025, 0.005, 0.0075) | |
| PT-EEC to EEC | 0.0500 | ± 50% | Triangular (0.025, 0.05, 0.075) | |
| PT-EEC to AEC | 0.1000 | ± 50% | Triangular (0.05, 0.10, 0.15) | |
| PT-AEC to AEC | 0.2304 | ± 50% | Triangular (0.115, 0.23, 0.345) | |
| Participation rate | Gao et al[8], 2023; Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| Endoscopy screening | 0.3352 | ± 50% | Triangular (0.30, 0.34, 0.67) | |
| Sponge cytology screening | 0.56 | ± 50% | Triangular (0.50, 0.56, 0.80) | |
| Endoscopy after sponge cytology test | 0.75 | ± 50% | Triangular (0.40, 0.75, 0.90) | |
| Probability of receiving treatment3 | Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| SD/CI | 0.7458 | 0.5625-0.9654 | Beta (20.75, 7.07) | |
| EEC | 0.9405 | 0.7149-1.0000 | Beta (5.13, 0.32) | |
| AEC | 0.9643 | 0.8393-1.0000 | Beta (11.08, 0.41) | |
| Probability of symptomatic detection | Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| SD/CIS | 0.01 | 0.005-0.02 | Triangular (0.005, 0.01, 0.02) | |
| EEC | 0.20 | 0.10-0.40 | Triangular (0.10, 0.20, 0.40) | |
| AEC | 0.70 | 0.56-0.90 | Triangular (0.56, 0.70, 0.90) | |
| Cost (USD) | ||||
| Endoscopic test | 74.31 | ± 50% | Triangular (50.36, 74.31, 108.69) | Xia et al[18], 2021; Li et al[19], 2019; Chen et al[37], 2017; and Xia et al[38], 2022 |
| Sponge cytology test | 35 | ± 50% | Triangular (17.5, 35, 52.5) | Estimated |
| Endoscopic complications | 119.61 | ± 50% | Gamma (9.75, 0.08) | Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 |
| Initial treatment | Xia et al[18], 2021; Chen et al[37], 2017; and Xia et al[38], 2022 | |||
| SD/CIS | 1687.61 | ± 50% | Gamma (3.33, 0.002) | |
| EEC | 8135.03 | ± 50% | Gamma (2.33, 0.0003) | |
| AEC | 7701.55 | ± 50% | Gamma (0.03, 0.00004) | |
| Annual treatment | ||||
| SD/CIS | 227.26 | ± 50% | Gamma (1.27, 0.006) | |
| EEC | 386.13 | ± 50% | Gamma (1.23, 0.003) | |
| AEC | 359.83 | ± 50% | Gamma (2.05, 0.006) | |
| Characteristics of tests | ||||
| Sensitivity of endoscopy | 100% | 0.88-1 | - | Xia et al[18], 2021; Nagami et al[39], 2014; and Dawsey et al[40], 1998 |
| Specificity of endoscopy | 100% | 0.59-1 | - | |
| Sensitivity of sponge cytology test | 94.5% | 0.888-0.975 | Triangular (0.888, 0.945, 0.975) | Gao et al[8], 2023 |
| Specificity of sponge cytology test | 91.9% | 0.912-0.925 | Triangular (0.912, 0.919, 0.925) | |
| Complication rate of endoscopic test | 0.00023 | 0-0.002 | Triangular (0, 0.00023, 0.0013) | Silvis et al[41], 1976 and Wang et al[42], 2019 |
| Health state5 | Xia et al[18], 2021 and Liu et al[20], 2018 | |||
| mD | 1 | 0.98-1 | Triangular (0.98, 1.00, 1.00) | |
| MD | 1 | 0.98-1 | Triangular (0.98, 1.00, 1.00) | |
| SD/CIS | 0.84 | 0.79-0.89 | Beta (3.57, 0.68) | |
| EEC | 0.70 | 0.66-0.74 | Beta (2.63, 1.13) | |
| AEC | 0.61 | 0.56-0.66 | Beta (1.12, 0.71) | |
| PT-SD/CIS | 0.90 | 0.88-0.92 | Beta (2.26, 0.25) | |
| PT-EEC | 0.81 | 0.79-0.83 | Beta (1.77, 0.41) | |
| PT-AEC | 0.74 | 0.71-0.77 | Beta (0.95, 0.33) | |
| Discount rate | 0.05 | 0-0.08 | Liu et al[21], 2022 | |
Patients diagnosed with esophageal dysplasia/carcinoma in situ or severe stages received appropriate therapy, such as endoscopic submucosal dissection, endoscopic mucosal resection, and radiofrequency ablation for esophageal dysplasia/carcinoma in situ and early esophageal cancer (including intramucosal carcinoma and submucosal carcinoma), or radical surgery, chemotherapy, radiotherapy, and other options for advanced esophageal cancer[17]. Among post-treatment patients, subsequent annual endoscopic surveillance was performed. Patients with esophageal dysplasia/carcinoma in situ were considered to be in a healthy state if no recurrence occurred within ten years[18], whereas those who refused treatment followed the natural progression of the disease.
The simulation continued until participants reached 75 years of age or died. In the model, it was assumed that participants with healthy, mild, or moderate dysplasia were assumed to die only from causes other than ESCC, those with severe dysplasia/carcinoma in situ or early ESCC followed all-cause mortality, and patients with advanced ESCC were subject to ESCC-specific mortality. A cycle length of one year and a half-cycle correction were applied in our simu
Model pathways are illustrated in Supplementary Figure 1. Screening strategies simulated in this study include no screening (natural history), endoscopic screening, and sponge cytology screening. Six screening intervals were evaluated: Once, every 10 years, every 5 years, every 3 years, every 2 years, and annually.
In the no screening pathway, participants followed the natural history of ESCC, and diagnoses were made only through incidental detection. The endoscopic screening pathway represented the current gold standard in the real world, and thus if a participant received a negative result, no further examination and treatment was performed. For participants with positive sponge cytology results (defined as high-grade intraepithelial lesions or more advanced stages), confir
The endoscopic test was assumed to be the gold standard test, with both sensitivity and specificity set at 100%, consistent with previous economic evaluation studies[10-12]. The sensitivity and specificity of the sponge cytology test were 94.5% and 91.9%, respectively[8]. Positive results of the sponge cytology test were defined as high-grade intraepithelial lesions or more advanced states, and confirmatory endoscopy was recommended for these individuals. The participation rates for screening were set at 33.52% for endoscopy and 56% for sponge cytology, based on population-based screening studies[5,8]. The probability of referral for confirmatory endoscopy following a positive sponge cytology result was 75%, as reported in the trial by Gao et al[8]. Further details are provided in Table 1.
A healthcare sector perspective was adopted in our study. Screening costs included equipment costs, examination fees, administration expenses, staff time costs, and related resources[19]. Costs for endoscopic screening were derived from a population-based screening study[19], and costs related to the sponge cytology test comprised screening mobilization and administrative expenses (5 USD/person), examination costs (20 USD/person), and manufacturing costs (10 USD/person). The costs of corresponding treatment were obtained from a previous study by Xia et al[18]. All costs were adjusted to 2022 USD according to the health and medical service component of the Consumer Price Index and the exchange rate (1 Chinese Yuan = 0.1484 USD). Based on published literature, utility scores for each stage were estimated and converted into quality-adjusted life years (QALYs; Table 1)[18,20]. An annual discount rate of 5% was applied to both costs and QALYs in this study, in accordance with the Chinese Guidelines for Pharmacoeconomic Evaluations[21].
Health outcomes included the number of ESCC cases and the number of ESCC deaths throughout the simulation. Cost-effectiveness outcomes included QALYs, costs, and the incremental cost-effective ratio (ICER). A QALY is a widely used outcome measure in health economic evaluations that integrates both the quantity and quality of life into a single metric. One QALY represents one year of life lived in perfect health, and utility values typically range from 0 (equivalent to death) to 1 (perfect health), with negative values possible for health states perceived as worse than death. The ICER is defined as the ratio of the incremental cost to the incremental health benefit, calculated as the difference in costs divided by the difference in QALYs gained between two strategies[22]: ICER = Incremental cost/QALYs gained. Comparisons of ICERs were assessed in two ways: (1) Each screening strategy was compared against the no screening strategy; and (2) All evaluated screening strategies, including both endoscopy and sponge cytology at different intervals, were compared to identify the optimal strategy.
A willingness-to-pay (WTP) threshold of one to three times per-capita gross domestic product (GDP) was adopted in this study, in accordance with the World Health Organization recommendations[23]. An ICER lower than one-time per-capita GDP was considered highly cost-effective, whereas an ICER between one to three times per-capita GDP was considered cost-effective. The per-capita GDP in high-risk areas of China was 9217 USD in 2022[24].
Model validation was performed using the Assessment of the Validation Status of Health-Economic Decision Models (Supplementary material)[25].
We further conducted scenario analyses across a range of possible screening ages to explore the cost-effectiveness of different screening strategies under various settings. A wide range of possible starting ages, from 40 years to 65 years, was examined in this analysis. Age-specific prevalence values and corresponding cycle lengths were varied, and the detailed parameter settings are summarized in Supplementary Table 1.
Deterministic one-way sensitivity analyses were performed for all parameters within predefined ranges to identify the most influential parameters, and the results are presented in Tornado diagrams. Probabilistic sensitivity analysis was further conducted using Monte Carlo simulation with 10000 iterations to evaluate the robustness of the model results and to identify the optimal strategy under different WTP thresholds; results are presented as a cost-effectiveness acceptability curve. Model parameters were varied based on 95% confidence interval, a 50% range, or ranges reported in the literature[11,12]. Utility values were assigned beta distributions, and costs were assigned gamma distributions, and a triangular distribution was used to represent uncertainty between minimum and maximum values when appropriate. Details of parameter uncertainties are summarized in Table 1.
Health outcomes are summarized in Table 2. All screening strategies resulted in fewer ESCC cases and deaths compared with no screening. Endoscopic screening reduced ESCC incidence by 6%-76%, corresponding to 191-2379 fewer cases per 100000 participants as screening frequency increased from once to annual screening. Similarly, endoscopic screening reduced ESCC-related mortality by 4%-79%, equivalent to 104-1895 fewer deaths per 100000 participants, compared with no screening.
| Strategy | Health outcomes (per 100000 participants) | |
| ESCC cases | ESCC deaths | |
| No screening | 3134 | 2409 |
| Sponge cytology screening | ||
| Once | 2962 | 2266 |
| Every 10 years | 2515 | 1874 |
| Every 5 years | 2011 | 1469 |
| Every 3 years | 1510 | 1088 |
| Every 2 years | 1060 | 750 |
| Every year | 373 | 257 |
| Endoscopic screening | ||
| Once | 2943 | 2305 |
| Every 10 years | 2606 | 2006 |
| Every 5 years | 2277 | 1685 |
| Every 3 years | 1867 | 1362 |
| Every 2 years | 1470 | 1052 |
| Every year | 755 | 514 |
For sponge cytology screening, ESCC incidence was reduced by 5%-88% and ESCC-related deaths by 6%-89%, corresponding to 172-2760 fewer ESCC cases and 143-2152 fewer deaths per 100000 participants, respectively. Among all strategies, annual sponge cytology screening yielded the greatest health benefit, preventing 2761 ESCC cases and 2152 deaths per 100000 participants compared with no screening.
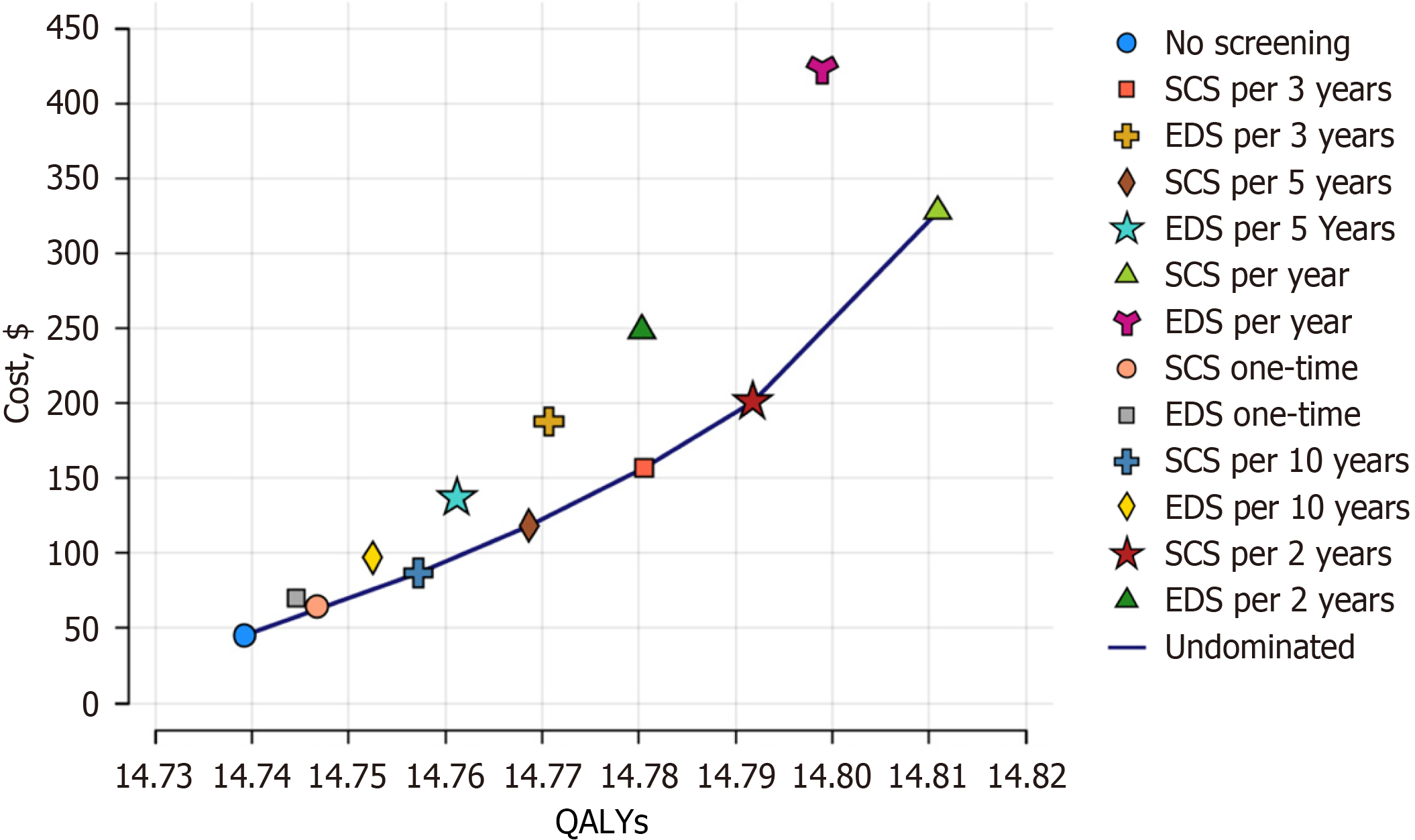
Cost-effectiveness analysis shows that all screening strategies incurred higher costs but yielded greater QALYs compared with the no-screening strategy (Table 3 and Figure 2). All screening strategies were considered highly cost-effective compared with no screening, with ICERs ranging from 2571 USD to 6318 USD.
| Strategy | Cost per 100000 participants (USD) | QALYs per 100000 participants | Incremental cost per 100000 participants (USD) | QALYs gained per 100000 participants | ICERs (USD per QALY gained) | |||
| Against no screening strategy | Among all strategies | Against no screening strategy | Among all strategies | Against no screening strategy | Among all strategies | |||
| No screening | 4532190 | 1473925 | - | - | - | - | - | - |
| Sponge cytology screening | ||||||||
| Once | 6452020 | 1474672 | 1919830 | 1919830 | 747 | 747 | 2571 | 2571 |
| Every 10 years | 8691075 | 1475723 | 4158885 | 4158885 | 1798 | 1798 | 2313 | 2313 |
| Every 5 years | 11823542 | 1476865 | 7291352 | 3132467 | 2940 | 1142 | 2480 | 2743 |
| Every 3 years | 15682730 | 1478055 | 11150540 | 3859188 | 4129 | 1190 | 2700 | 3244 |
| Every 2 years | 20115833 | 1479177 | 15583643 | 4433104 | 5252 | 1122 | 2967 | 3950 |
| Every year | 32781384 | 1481087 | 28249194 | 12665550 | 7162 | 1910 | 3944 | 6630 |
| Endoscopic screening | ||||||||
| Once | 7019587 | 1474458 | 2487397 | 567568 | 532 | -214 | 4671 | Dominated |
| Every 10 years | 9716978 | 1475250 | 5184788 | 1025903 | 1324 | -473 | 3915 | Dominated |
| Every 5 years | 13676309 | 1476120 | 9144119 | 185767 | 2194 | -745 | 4167 | Dominated |
| Every 3 years | 18773932 | 1477073 | 1424742 | 3091202 | 3147 | -982 | 4525 | Dominated |
| Every 2 years | 24851313 | 1478033 | 20319123 | 4735480 | 4108 | -1144 | 4946 | Dominated |
| Every year | 42263921 | 1479898 | 37731731 | 9482537 | 5972 | -1189 | 6318 | Dominated |
Under endoscopic screening, one additional QALY was gained at a cost of 3915-6318 USD, whereas under sponge cytology screening, the cost per QALY gained ranged from 2313-3944 USD. In both endoscopic and sponge cytology screening strategies, shorter screening intervals were associated with greater QALYs gained per 100000 participants, increasing from 532 to 5972 QALYs for endoscopy and from 747 to 7162 QALYs for the sponge cytology test as screening frequency increased from once to annual screening.
Further comparisons among all strategies indicated that annual sponge cytology screening was the optimal strategy, yielding the greatest QALYs gained (1910 per 100000 participants) with an ICER of 6630 USD. The endoscopy screening strategies were dominated by the annual sponge cytology strategy, as they produced fewer QALYs at higher or com
Comparisons between screening modalities within the same screening interval demonstrated that sponge cytology screening dominated endoscopic screening across all evaluated screening intervals, achieving higher QALYs at lower costs (Supplementary Table 2). Evaluation of screening intervals suggested that annual screening was the best option for both endoscopic and sponge cytology screening strategies, with ICERs of 6630 USD and 9340 USD, respectively, among all assessed strategies (Supplementary Table 3).
In scenario analyses, all screening strategies remained cost-effective compared with the no-screening strategy (Supplementary Figure 2A; ICERs ranged from 1397 USD to 6560 USD). Annual sponge cytology screening strategies consistently dominated among all assessed strategies, with ICERs ranging from 5400 USD to 6036 USD.
In addition, the results suggested that screening programs initiated at younger ages yielded greater QALYs in this setting (Supplementary Figure 2B), with total QALYs increasing from 718222 to 1591612 per 100000 participants as the starting age increased from 40 years to 65 years.
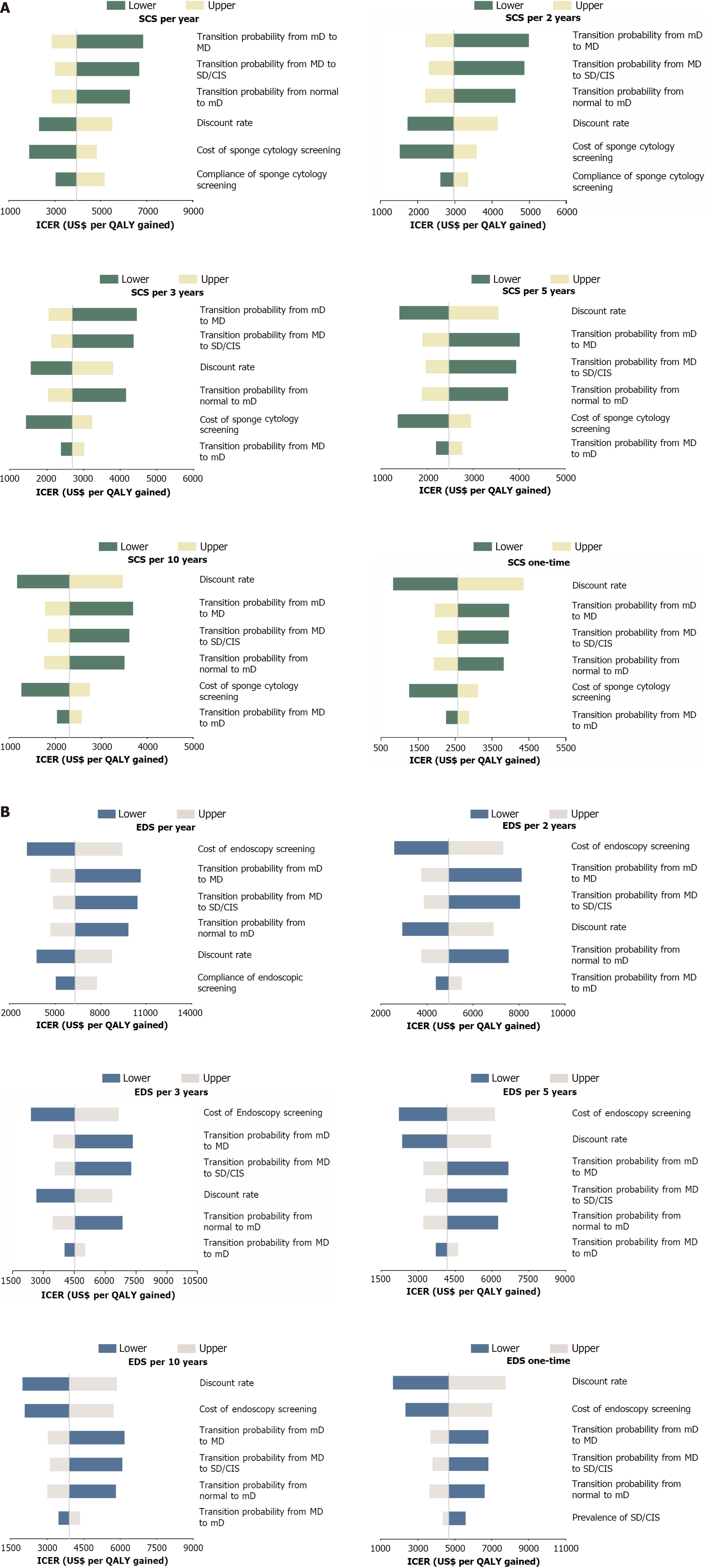
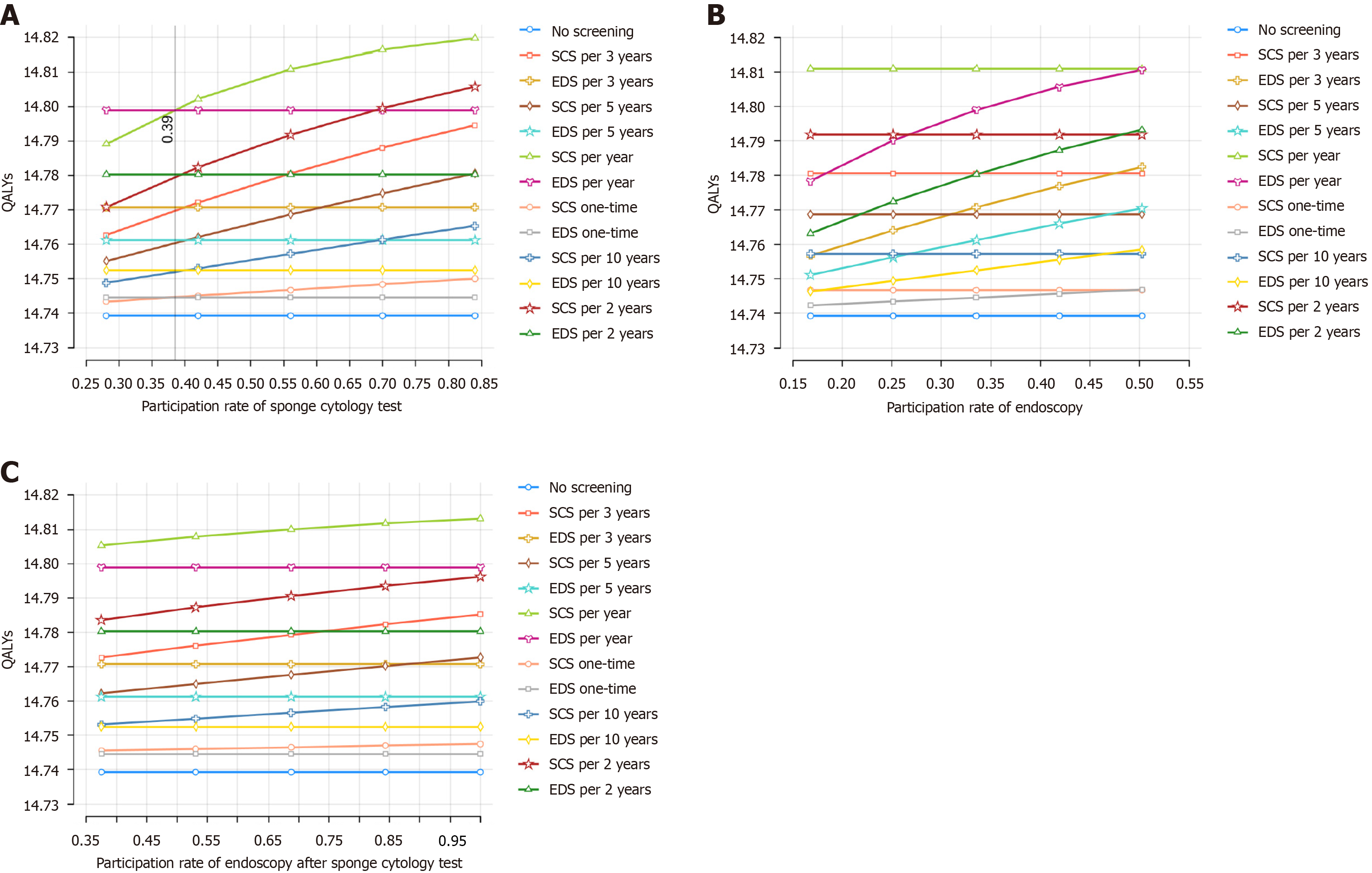
In deterministic one-way sensitivity, all screening strategies remained cost-effective in comparison with the no-screening strategy. The most influential parameters are summarized and presented in Tornado diagrams in Figure 3. Ranking results were sensitive to screening participation rates. When the participation rate in sponge cytology screening fell below 0.39, the annual endoscopic screening strategy yielded the highest QALYs (14.7990 per person) with an ICER of 22996 USD (Figure 4A and Supplementary Table 4). Although annual sponge cytology screening costs were approximately half as much as annual endoscopy screening (196 USD vs 423 USD per person), it was dominated by a sponge cytology participation rate of 0.28, as it yielded the second highest QALYs.
The annual sponge cytology screening strategy remained the best option when the participation rate in the endoscopy test varied within the predefined ranges (Figure 4B). At an endoscopy participation rate of 0.50, the annual endoscopy screening strategy achieved a similar health benefit to the annual sponge cytology test but at substantially higher costs (QALY/cost per person: 14.8105/598 USD for annual endoscopy screening vs 14.8109/328 USD for annual sponge cytology screening). The impact of participation rate on confirmation following a positive sponge cytology test is illu
We also examined the impact of endoscopy accuracy, which was assumed to be 100% in alignment with previous studies. The results demonstrated robustness, as the annual sponge cytology test remained the optimal strategy across a range of assumed endoscopy accuracy (Supplementary Tables 5 and 6).
The cost-effectiveness acceptability curve (Supplementary Figure 3) illustrates the result of 10000 iterations from the probabilistic sensitivity analysis. The probabilistic sensitivity analysis results are consistent with base-case findings and indicate that annual sponge cytology screening was cost-effective in 72.36% of iterations and 99.62% of iterations at the WTP thresholds of once and three-times per-capita GDP, respectively. Sponge cytology screening every 2 years was cost-effective in 23.98% of iterations at the once per-capita GDP threshold, whereas sponge cytology screening every 3 years was cost-effective in 2.85% iterations. Annual endoscopic screening was cost-effective in 0.26% of iterations at the WTP of three times the per-capita GDP.
To our knowledge, this is the first economic evaluation study of the minimally invasive sponge cytology test in ESCC high-risk areas. In this study, we found that for participants in ESCC high-risk areas, all screening programs were associated with a reduction in ESCC cases and deaths. Our study demonstrated that screening a targeted population was highly cost-effective, and an annual sponge cytology screening strategy was the most cost-effective of all the strategies assessed. Endoscopic screening resulted in increased costs, but fewer QALYs were gained compared to sponge cytology screening in this study. In scenario analyses, QALYs increased with younger screening age and screening intensity, representing the effectiveness of cancer screening.
All tested strategies remained consistently cost-effective compared with no screening in the deterministic one-way sensitivity analyses. Marginal disparity in ranking results occurred when the participation rate of screening varied, and this concurred with the findings of an earlier study comparing the cost-effectiveness of the sponge cytology test and endoscopy in Barrett’s esophagus screening[12]. Of note, sponge cytology tests are preferred over endoscopic tests without sedation due to their minimally invasive and less time-consuming nature[26], as the latter is currently the most performed modality in China. Examination using sponge cytology tests requires no facilities, and checks outside the hospital are thereby potentially suitable for population-based screening. Therefore, a higher participation rate of sponge cytology screening makes sense.
Previous studies have explored the cost-effectiveness of endoscopic screening strategies in ESCC high-risk areas like China[18,27,28]. All screening strategies remained cost-effective, and more frequent screenings yielded higher QALYs (from once to every 2 years: 1572532 to 1582334 per 100000 participants)[18], this is in line with our study. However, clinical resources are constrictive in ESCC high-risk areas for implementing mass endoscopic screening. Questionnaire-based risk-stratification tools prior to endoscopic screening are an alternative approach to promoting screening efficiency[29-31]. However, given their suboptimal performance in the screening population (areas under the receiver operating curve: 0.681-0.80), these tools were not included in our model. Our analyses focused on the sponge cytology test combined with endoscopic confirmation, which showed favorable discriminative abilities for ESCC in earlier studies[8,9,32]. This could potentially understate the benefits of sponge cytology screening, as its less invasive nature and time efficiency might make it more appealing for more frequent screening intervals.
Instead of implementing universal screening across all areas, the target population we focused on involved participants in ESCC high-risk areas given the unbalanced geographical distribution of ESCC. We believe that a targeted approach is more efficient as it yields higher positive cases and avoids unnecessary screening. Also, an extensive range of screening intervals and starting ages were included in this study to provide evidence for optimizing screening programs as tailored screening frequencies and starting ages were crucial for the balance of resources and risks. Another strength of our study is that the data incorporated in this study, e.g., initial prevalence, transition probabilities, and utility scores, were derived from several prospective trials. This allowed us to simulate scenarios reflecting real-world conditions more closely.
Notably, a combination of artificial intelligence (AI) with esophageal cytology further improved the cost-effectiveness of sponge cytology tests in this study, as AI-based automatic classification achieves comparative performance to expert cytologists[33-35], which leads to a reduction in expenditure due to extra labor. Reduced demand for professionals further promotes the fair distribution of screening opportunities, especially for regions with sparse medical resources. Besides, we believe that with the development of sponge cytology tests, e.g., scaled-up productivity and increased test accuracy, the overall costs are likely to decrease and the tests will be more affordable for wider use. Also, future advances in sponge cytology test data management and analytics can promote its applicability in different medical systems or settings.
There is no doubt that endoscopy can also detect gastric and duodenal lesions in addition to esophageal lesions. Encouragingly, sponge cytology has also shown remarkable performance in identifying cancerous conditions of the esophagogastric junction as well as gastric metaplasia and gastric atrophy[8,36]. Thus, sponge cytology could also suggest endoscopic referral for individuals at increased risk of gastric cancer. However, it should be noted that sponge cytology lacks evidence to detect stomach lesions as endoscopy does. We therefore recommend that the sponge cytology screening strategy should primarily be used for individuals with an increased risk of esophageal cancer, especially in high-risk areas or for those with known risk factors. For individuals with a combined risk of esophageal and gastric cancer, endoscopic screening should still be considered as the standard strategy. In such populations, a combined approach of sponge cytology and endoscopy may be a potential consideration. For example, if participants receive a negative endoscopy test in year 1, they might undergo sponge cytology tests in subsequent years; and when the next cycle begins, a repeat endoscopy test and subsequent sponge cytology tests should continue. This approach may offer a more comprehensive monitoring strategy for individuals at higher risk of both cancers. Nevertheless, further tailored research is imperative to verify the role of sponge cytology test in this population.
This study has limitations. First, adherence to all strategies across different screening intervals in this study was assumed to remain constant throughout the simulation due to model constraints, which does not fully reflect real-world variability. To address this, we conducted a wide range of sensitivity analyses to mitigate uncertainties in the parameters. Also, the available data on the accuracy of sponge cytology tests were limited by the lack of long-term follow-up. To better establish the test’s clinical utility, future studies incorporating extended follow-up periods are essential to evaluate its real-world effectiveness.
In summary, this study indicates that both endoscopic and sponge cytology screening programs are highly cost-effective compared with no screening in ESCC high-risk areas. Among the strategies evaluated, annual sponge cytology screening emerged as the optimal approach. These encouraging findings provide evidence for policymakers and healthcare sectors in terms of new paradigms for early ESCC detection and improved treatment.
We thank Lei Yuan from the Department of Health Management, Naval Medical University for his constructive comments on this work.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14861] [Article Influence: 7430.5] [Reference Citation Analysis (21)] |
| 2. | Morgan E, Soerjomataram I, Rumgay H, Coleman HG, Thrift AP, Vignat J, Laversanne M, Ferlay J, Arnold M. The Global Landscape of Esophageal Squamous Cell Carcinoma and Esophageal Adenocarcinoma Incidence and Mortality in 2020 and Projections to 2040: New Estimates From GLOBOCAN 2020. Gastroenterology. 2022;163:649-658.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 968] [Cited by in RCA: 812] [Article Influence: 203.0] [Reference Citation Analysis (3)] |
| 3. | Morgan E, Soerjomataram I, Gavin AT, Rutherford MJ, Gatenby P, Bardot A, Ferlay J, Bucher O, De P, Engholm G, Jackson C, Kozie S, Little A, Møller B, Shack L, Tervonen H, Thursfield V, Vernon S, Walsh PM, Woods RR, Finley C, Merrett N, O'Connell DL, Reynolds JV, Bray F, Arnold M. International trends in oesophageal cancer survival by histological subtype between 1995 and 2014. Gut. 2021;70:234-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 4. | Lao-Sirieix P, Fitzgerald RC. Screening for oesophageal cancer. Nat Rev Clin Oncol. 2012;9:278-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 128] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 5. | Chen R, Liu Y, Song G, Li B, Zhao D, Hua Z, Wang X, Li J, Hao C, Zhang L, Liu S, Wang J, Zhou J, Zhang Y, Li B, Li Y, Feng X, Li L, Dong Z, Wei W, Wang G. Effectiveness of one-time endoscopic screening programme in prevention of upper gastrointestinal cancer in China: a multicentre population-based cohort study. Gut. 2021;70:251-260. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 6. | Wei WQ, Chen ZF, He YT, Feng H, Hou J, Lin DM, Li XQ, Guo CL, Li SS, Wang GQ, Dong ZW, Abnet CC, Qiao YL. Long-Term Follow-Up of a Community Assignment, One-Time Endoscopic Screening Study of Esophageal Cancer in China. J Clin Oncol. 2015;33:1951-1957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 275] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 7. | Zhang N, Li Y, Chang X, Lei F, Ma H, Liu J, Yang J, Su M, Sun X, Zhao D, Sun Q, Wei W, Wang G, Wang J. Long-term effectiveness of one-time endoscopic screening for esophageal cancer: A community-based study in rural China. Cancer. 2020;126:4511-4520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Gao Y, Xin L, Lin H, Yao B, Zhang T, Zhou AJ, Huang S, Wang JH, Feng YD, Yao SH, Guo Y, Dang T, Meng XM, Yang ZZ, Jia WQ, Pang HF, Tian XJ, Deng B, Wang JP, Fan WC, Wang J, Shi LH, Yang GY, Sun C, Wang W, Zang JC, Li SY, Shi RH, Li ZS, Wang LW. Machine learning-based automated sponge cytology for screening of oesophageal squamous cell carcinoma and adenocarcinoma of the oesophagogastric junction: a nationwide, multicohort, prospective study. Lancet Gastroenterol Hepatol. 2023;8:432-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 9. | Gao Y, Xin L, Feng YD, Yao B, Lin H, Sun C, An W, Li ZS, Shi RH, Wang LW. Feasibility and Accuracy of Artificial Intelligence-Assisted Sponge Cytology for Community-Based Esophageal Squamous Cell Carcinoma Screening in China. Am J Gastroenterol. 2021;116:2207-2215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 10. | Heberle CR, Omidvari AH, Ali A, Kroep S, Kong CY, Inadomi JM, Rubenstein JH, Tramontano AC, Dowling EC, Hazelton WD, Luebeck EG, Lansdorp-Vogelaar I, Hur C. Cost Effectiveness of Screening Patients With Gastroesophageal Reflux Disease for Barrett's Esophagus With a Minimally Invasive Cell Sampling Device. Clin Gastroenterol Hepatol. 2017;15:1397-1404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 11. | Swart N, Maroni R, Muldrew B, Sasieni P, Fitzgerald RC, Morris S; BEST3 Consortium. Economic evaluation of Cytosponge®-trefoil factor 3 for Barrett esophagus: A cost-utility analysis of randomised controlled trial data. EClinicalMedicine. 2021;37:100969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 12. | Benaglia T, Sharples LD, Fitzgerald RC, Lyratzopoulos G. Health benefits and cost effectiveness of endoscopic and nonendoscopic cytosponge screening for Barrett's esophagus. Gastroenterology. 2013;144:62-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 13. | Gerson L, Lin OS. Cost-benefit analysis of capsule endoscopy compared with standard upper endoscopy for the detection of Barrett's esophagus. Clin Gastroenterol Hepatol. 2007;5:319-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Zeng H, Sun K, Cao M, Zheng R, Sun X, Liu S, Zhang Z, Liu Y, Guo G, Song G, Zhu Y, Wu X, Song B, Liao X, Chen Y, Song M, Giovannucci E, Zhuang G, Wei W, Chen W, He J. Initial results from a multi-center population-based cluster randomized trial of esophageal and gastric cancer screening in China. BMC Gastroenterol. 2020;20:398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 15. | Abnet CC, Arnold M, Wei WQ. Epidemiology of Esophageal Squamous Cell Carcinoma. Gastroenterology. 2018;154:360-373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1351] [Cited by in RCA: 1291] [Article Influence: 161.4] [Reference Citation Analysis (6)] |
| 16. | Husereau D, Drummond M, Augustovski F, de Bekker-Grob E, Briggs AH, Carswell C, Caulley L, Chaiyakunapruk N, Greenberg D, Loder E, Mauskopf J, Mullins CD, Petrou S, Pwu RF, Staniszewska S; CHEERS 2022 ISPOR Good Research Practices Task Force. Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) statement: updated reporting guidance for health economic evaluations. BMJ. 2022;376:e067975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 362] [Cited by in RCA: 297] [Article Influence: 74.3] [Reference Citation Analysis (0)] |
| 17. | He J, Chen WQ, Li ZS, Li N, Ren JS, Tian JH, Tian WJ, Hu FL, Peng J; Expert Group of China Guideline for the Screening, Early Detection and Early Treatment of Esophageal Cancer; Work Group of China Guideline for the Screening,Early Detection and Early Treatment of Esophageal Cancer. [China guideline for the screening, early detection and early treatment of esophageal cancer (2022, Beijing)]. Zhonghua Zhong Liu Za Zhi. 2022;44:491-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 18. | Xia R, Zeng H, Liu W, Xie L, Shen M, Li P, Li H, Wei W, Chen W, Zhuang G. Estimated Cost-effectiveness of Endoscopic Screening for Upper Gastrointestinal Tract Cancer in High-Risk Areas in China. JAMA Netw Open. 2021;4:e2121403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 19. | Li F, Li X, Guo C, Xu R, Li F, Pan Y, Liu M, Liu Z, Shi C, Wang H, Wang M, Tian H, Liu F, Liu Y, Li J, Cai H, Yang L, He Z, Ke Y. Estimation of Cost for Endoscopic Screening for Esophageal Cancer in a High-Risk Population in Rural China: Results from a Population-Level Randomized Controlled Trial. Pharmacoeconomics. 2019;37:819-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | Liu Q, Zeng H, Xia R, Chen G, Liu S, Zhang Z, Liu Y, Guo G, Song G, Zhu Y, Wu X, Song B, Liao X, Chen Y, Wei W, Chen W, Zhuang G. Health-related quality of life of esophageal cancer patients in daily life after treatment: A multicenter cross-sectional study in China. Cancer Med. 2018;7:5803-5811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Liu GE, Wu J, Xie F, Sun X, Li HC. China Guidelines for Pharmacoeconomic 2020. Beijing: China Market Publishing House, 2022. |
| 22. | Gray AM, Clarke PM, Wolstenholme J, Wordsworth S. Applied Methods of Cost-effectiveness Analysis in Healthcare (Handbooks in Health Economic Evaluation). Oxford: Oxford University Press, 2011. |
| 23. | Hutubessy R, Chisholm D, Edejer TT. Generalized cost-effectiveness analysis for national-level priority-setting in the health sector. Cost Eff Resour Alloc. 2003;1:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 402] [Cited by in RCA: 368] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 24. | National Bureau of Statistics. China Statistical Yearbook 2023. [cited 3 December 2025]. Available from: https://www.stats.gov.cn/sj/ndsj/. |
| 25. | Vemer P, Corro Ramos I, van Voorn GA, Al MJ, Feenstra TL. AdViSHE: A Validation-Assessment Tool of Health-Economic Models for Decision Makers and Model Users. Pharmacoeconomics. 2016;34:349-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 278] [Cited by in RCA: 266] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 26. | Januszewicz W, Tan WK, Lehovsky K, Debiram-Beecham I, Nuckcheddy T, Moist S, Kadri S, di Pietro M, Boussioutas A, Shaheen NJ, Katzka DA, Dellon ES, Fitzgerald RC; BEST1 and BEST2 study investigators. Safety and Acceptability of Esophageal Cytosponge Cell Collection Device in a Pooled Analysis of Data From Individual Patients. Clin Gastroenterol Hepatol. 2019;17:647-656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 73] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 27. | Yang J, Wei WQ, Niu J, Liu ZC, Yang CX, Qiao YL. Cost-benefit analysis of esophageal cancer endoscopic screening in high-risk areas of China. World J Gastroenterol. 2012;18:2493-2501. [PubMed] [DOI] [Full Text] |
| 28. | Hur C, Choi SE, Kong CY, Wang GQ, Xu H, Polydorides AD, Xue LY, Perzan KE, Tramontano AC, Richards-Kortum RR, Anandasabapathy S. High-resolution microendoscopy for esophageal cancer screening in China: A cost-effectiveness analysis. World J Gastroenterol. 2015;21:5513-5523. [PubMed] [DOI] [Full Text] |
| 29. | Chen W, Li H, Ren J, Zheng R, Shi J, Li J, Cao M, Sun D, He S, Sun X, Cao X, Feng S, Zhou J, Luo P, Zha Z, Jia S, Wang J, Ma H, Zeng H, Canfell K, He J. Selection of high-risk individuals for esophageal cancer screening: A prediction model of esophageal squamous cell carcinoma based on a multicenter screening cohort in rural China. Int J Cancer. 2021;148:329-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 30. | Liu M, Liu Z, Cai H, Guo C, Li X, Zhang C, Wang H, Hang D, Liu F, Deng Q, Yang X, Yuan W, Pan Y, Li J, Zhang C, Shen N, He Z, Ke Y. A Model To Identify Individuals at High Risk for Esophageal Squamous Cell Carcinoma and Precancerous Lesions in Regions of High Prevalence in China. Clin Gastroenterol Hepatol. 2017;15:1538-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 31. | Liu M, Zhou R, Liu Z, Guo C, Xu R, Zhou F, Liu A, Yang H, Li F, Duan L, Shen L, Wu Q, Zheng H, Tian H, Liu F, Liu Y, Pan Y, Chen H, Hu Z, Cai H, He Z, Ke Y. Update and validation of a diagnostic model to identify prevalent malignant lesions in esophagus in general population. EClinicalMedicine. 2022;47:101394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 32. | Roshandel G, Merat S, Sotoudeh M, Khoshnia M, Poustchi H, Lao-Sirieix P, Malhotra S, O'Donovan M, Etemadi A, Nickmanesh A, Pourshams A, Norouzi A, Debiram I, Semnani S, Abnet CC, Dawsey SM, Fitzgerald RC, Malekzadeh R. Pilot study of cytological testing for oesophageal squamous cell dysplasia in a high-risk area in Northern Iran. Br J Cancer. 2014;111:2235-2241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 33. | Berman AG, Tan WK, O'Donovan M, Markowetz F, Fitzgerald RC. Quantification of TFF3 expression from a non-endoscopic device predicts clinically relevant Barrett's oesophagus by machine learning. EBioMedicine. 2022;82:104160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 34. | Masqué-Soler N, Gehrung M, Kosmidou C, Li X, Diwan I, Rafferty C, Atabakhsh E, Markowetz F, Fitzgerald RC. Computational pathology aids derivation of microRNA biomarker signals from Cytosponge samples. EBioMedicine. 2022;76:103814. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 35. | Gehrung M, Crispin-Ortuzar M, Berman AG, O'Donovan M, Fitzgerald RC, Markowetz F. Triage-driven diagnosis of Barrett's esophagus for early detection of esophageal adenocarcinoma using deep learning. Nat Med. 2021;27:833-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 36. | Hadjinicolaou AV, Azizi AA, O'Donovan M, Debiram I, Fitzgerald RC, Di Pietro M. Cytosponge-TFF3 Testing can Detect Precancerous Mucosal Changes of the Stomach. Clin Gastroenterol Hepatol. 2022;20:1411-1412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Chen W, Zeng H, Chen R, Xia R, Yang Z, Xia C, Zheng R, Wei W, Zhuang G, Yu X, He J. Evaluating efficacy of screening for upper gastrointestinal cancer in China: a study protocol for a randomized controlled trial. Chin J Cancer Res. 2017;29:294-302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 38. | Xia R, Li H, Shi J, Liu W, Cao M, Sun D, He S, Yu Y, Li N, Lei L, Zhuang G, Chen W. Cost-effectiveness of risk-stratified endoscopic screening for esophageal cancer in high-risk areas of China: a modeling study. Gastrointest Endosc. 2022;95:225-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 39. | Nagami Y, Tominaga K, Machida H, Nakatani M, Kameda N, Sugimori S, Okazaki H, Tanigawa T, Yamagami H, Kubo N, Shiba M, Watanabe K, Watanabe T, Iguchi H, Fujiwara Y, Ohira M, Hirakawa K, Arakawa T. Usefulness of non-magnifying narrow-band imaging in screening of early esophageal squamous cell carcinoma: a prospective comparative study using propensity score matching. Am J Gastroenterol. 2014;109:845-854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 109] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 40. | Dawsey SM, Fleischer DE, Wang GQ, Zhou B, Kidwell JA, Lu N, Lewin KJ, Roth MJ, Tio TL, Taylor PR. Mucosal iodine staining improves endoscopic visualization of squamous dysplasia and squamous cell carcinoma of the esophagus in Linxian, China. Cancer. 1998;83:220-231. [PubMed] |
| 41. | Silvis SE, Nebel O, Rogers G, Sugawa C, Mandelstam P. Endoscopic complications. Results of the 1974 American Society for Gastrointestinal Endoscopy Survey. JAMA. 1976;235:928-930. [PubMed] [DOI] [Full Text] |
| 42. | Wang LW, Lin H, Xin L, Qian W, Wang TJ, Zhang JZ, Meng QQ, Tian B, Ma XD, Li ZS. Establishing a model to measure and predict the quality of gastrointestinal endoscopy. World J Gastroenterol. 2019;25:1024-1030. [PubMed] [DOI] [Full Text] |
| 43. | Briggs AH. Handling uncertainty in cost-effectiveness models. Pharmacoeconomics. 2000;17:479-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 623] [Cited by in RCA: 547] [Article Influence: 21.0] [Reference Citation Analysis (0)] |