Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.116561
Revised: December 22, 2025
Accepted: January 15, 2026
Published online: March 15, 2026
Processing time: 114 Days and 7.3 Hours
Early gastric signet ring cell carcinoma (eGSRCC) is a distinct histological subtype of gastric cancer with unique biological behaviors. However, the impact of Helicobacter pylori (H. pylori) infection status on the clinical and endoscopic characteristics of endoscopically resectable eGSRCC remains uncertain.
To evaluate clinical and endoscopic characteristics of eGSRCC based on H. pylori status and to assess its implications for endoscopic management.
We retrospectively analyzed consecutive patients with histologically confirmed eGSRCC at Shenzhen People’s Hospital between January 2020 and December 2024. Patients were stratified into three groups: H. pylori-positive, H. pylori-eradicated and H. pylori-uninfected. Detailed clinical and endoscopic data were compared.
A total of 29 patients were included, with 12 (41.4%) in the H. pylori-positive group, 7 (24.1%) in the H. pylori-eradicated group, and 10 (34.5%) in the H. pylori-uninfected group. Significant differences in endoscopic features were observed based on H. pylori status; 88.9% of the lesions were located at the lower third of gastric body and 80% presented as type 0-IIb with whitish discoloration in H. pylori-uninfected group. In contrast, more lesions in H. pylori-positive group manifested 0-IIc type with reddish roughness with or without spontaneous bleeding. Notably, all three cases of delayed post-endoscopic submucosal dissection (ESD) bleeding occurred exclusively in the H. pylori-positive group. All the cases achieved en bloc resection with no recurrence.
H. pylori infection status significantly impacts the clinical and endoscopic presentations of eGSRCC. Recognizing the differences aids early detection and H. pylori eradication prior to ESD optimizes endoscopic management.
Core Tip: This study elucidates the different clinical and endoscopic features of early gastric signet ring cell carcinoma (eGSRCC) based on Helicobacter pylori (H. pylori) status. H. pylori-uninfected lesions were typically flat and pale in the lower gastric body, while H. pylori-positive lesions were often depressed and reddish. All delayed post-endoscopic submucosal dissection (ESD) bleeding events occurred in H. pylori-positive patients. Recognizing these patterns can aid in early detection. More importantly, H. pylori eradication prior to ESD should be considered to optimize endoscopic visualization and may reduce procedural bleeding risk, thereby refining the therapeutic approach for eGSRCC.
- Citation: Lu Q, Xiong F, Gu HC, Peng QZ, Yao J, Li DF, Huang BW, Wang LS. Clinical and endoscopic characteristics of early gastric signet ring cell carcinoma based on Helicobacter pylori infection status. World J Gastrointest Oncol 2026; 18(3): 116561
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/116561.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.116561
Although the incidence and mortality of gastric cancer have shown a gradual decline in recent epidemiological studies, this malignancy persistently maintains its position among the top five most prevalent cancers worldwide. Gastric signet-ring cell carcinoma (GSRCC), characterized by abundant intracytoplasmic mucin that displaces the nucleus, is classified as a unique subtype of poorly cohesive carcinoma according to the 5th edition of the World Health Organisation (WHO) classification. Although GSRCC accounts for a relatively small proportion of gastric cancers, its unique biological beha
ESD has been established as a first-line therapeutic modality for early gastric cancer (EGC). According to the latest Japanese gastric cancer guidelines, ESD is considered an absolute indication for the treatment of clinically intramucosal (cT1a) undifferentiated-type carcinomas that are without ulcerative findings (UL0) and measure 2 cm or less in long diameter[3,4]. Thus, eGSRCC fulfilling these criteria could achieve curative resection through ESD.
It’s widely acknowledged that Helicobacter pylori (H. pylori) infection is a well-established risk factor for gastric carcinogenesis, particularly for intestinal-type gastric cancer[5]. Despite several reports on the clinical and endoscopic features of eGSRCC, studies specifically focusing on its relationship with H. pylori infection status remain limitied[6-9]. Under
This retrospective cohort study included consecutive patients diagnosed with GSRCC pathologically at Shenzhen People’s Hospital between January 2020 and December 2024. Inclusion criteria required lesions meeting the indications of ESD in accordance with Japanese Gastric Cancer Association guidelines: (1) Histologically confirmed GSRCC based on the 5th edition of the WHO classification; (2) cT1a lesions by imaging and post-resection histology; (3) UL0; and (4) Long diameter ≤ 2 cm[3,4]. The study was approved by the Institutional Review Board of our hospital and informed consent was obtained from all patients before the procedures.
Patients were classified into three groups based on H. pylori status: (1) H. pylori-positive (confirmed by histology or urea breath test); (2) H. pylori-eradicated (previous infection with documented successful eradication); and (3) H. pylori-uninfected (no history of infection, confirmed by negative tests).
All esophagogastroduodenoscopy procedures were conducted by qualified gastroenterologists at our center using high-definition endoscopes (Olympus Optical; Fujifilm, Tokyo, Japan) combined with image-enhanced endoscopy modalities such as linked color imaging, narrow-band imaging (NBI), blue laser imaging (BLI) and magnifying endoscopy (ME). Histopathological evaluations of the final specimen following ESD or surgery were made by experienced pathologists according to the 5th edition of the WHO classification in the same hospital. Clinical and endoscopic variables recorded included demographics (age, gender), symptoms at presentation, H. pylori infection status, lesion location[10], size, morphology, surface color, vessel plus surface (VS) classification[11], clinical management and complications. Among these, the endoscopic assessments were performed independently by two experienced endoscopists who were blinded to the patients’ clinical information, histopathological findings, and H. pylori infection status. Follow-up endoscopy was performed at 2-, 6-, and 12-month post-resection and annually thereafter.
All statistical analyses were performed using SPSS version 25.0 (IBM, Armonk, NY, United States). Continuous variables were analyzed using the Kruskal-Wallis test and were presented as mean ± SD. Categorical variables were compared using the χ2 test or Fisher’s exact test and were expressed as n (%). A P value < 0.05 was considered statistically signi
A total of 29 patients were included, with 12 (41.4%) in the H. pylori-positive group, 7 (24.1%) in the H. pylori-eradicated group, and 10 (34.5%) in the H. pylori-uninfected group (Table 1). The cohort comprised 7 (24.1%) males and 22 (75.9%) females, with a mean age of 47.55 ± 12.19 years. All baseline demographics did not differ significantly among the groups. Notably, 31.0% of the patients presented as abdominal pain, while 51.7% of the patients were asymptomatic, and the lesions were incidentally discovered during routine screening endoscopy.
| Variables | Overall | H. pylori-positive | H. pylori-eradicated | H. pylori-uninfected | P value |
| n = 29 | 12 (41.4) | 7 (24.1) | 10 (34.5) | ||
| Age, years, mean ± SD | 47.55 ± 12.19 | 45.25 ± 10.26 | 54.14 ± 8.65 | 45.7 ± 15.75 | 0.347 |
| Sex | 0.542 | ||||
| Male | 7 (24.1) | 4 (33.3) | 2 (28.6) | 1 (10.0) | |
| Female | 22 (75.9) | 8 (66.7) | 5 (71.4) | 9 (90.0) | |
| Symptoms | 0.398 | ||||
| Abdominal pain | 9 (31.0) | 4 (33.3) | 1 (14.3) | 4 (40.0) | |
| Distension | 3 (10.3) | 2 (16.7) | 0 | 1 (10.0) | |
| Diarrhea/constipation | 2 (7.9) | 2 (16.7) | 0 | 0 | |
| Routine screening | 15 (51.7) | 4 (33.3) | 6 (85.7) | 5 (50.0) | |
| Management | 0.545 | ||||
| ESD | 19 (65.5) | 7 (58.3) | 6 (85.7) | 6 (60.0) | |
| Surgery | 10 (34.5) | 5 (41.7) | 1 (14.3) | 4 (40.0) | |
| En bloc resection | 29 (100.0) | 12 (100.0) | 7 (100.0) | 10 (100.0) | |
| Complication | 0.25 | ||||
| Bleeding | 3 (10.3) | 3 (25.0) | 0 | 0 | |
| Perforation | 0 | 0 | 0 | 0 | |
| Infection | 1 (3.4) | 0 | 0 | 1 (10.0) | |
| Anastomotic leakage | 0 | 0 | 0 | 0 | |
| Recurrence | 0 | 0 | 0 | 0 |
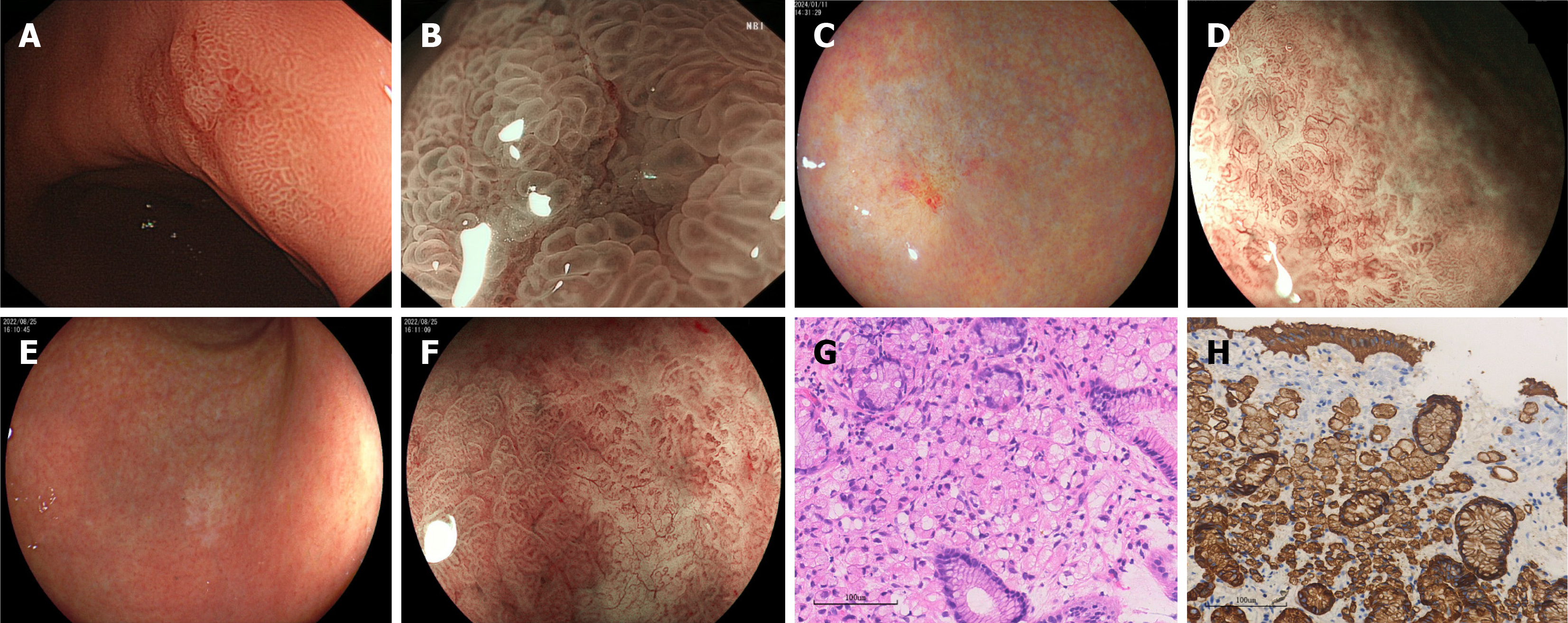
The overall mean tumor size was 12.69 ± 5.04 mm, which is not significantly different among three groups (Table 2). Tumor distribution was as follows: 82.8% were located in the gastric body, of which 70.8% in the lower third, 29.2% in the upper third. Additionally, 17.2% were located in the antrum, which were mostly in the H. pylori-positive group (80.0%). In H. pylori-uninfected group, almost all the lesions were located in the lower third of gastric body (88.9%). Morphologically, 44.8% of tumors appeared as flat (0-IIb), 34.5% as depressed (0-IIc), and 20.7% as slightly elevated and depressed (0-IIa + IIc/0-IIc + IIa) according to the Paris classification of superficial neoplastic lesions in the digestive tract (Figure 1). Notably, 50.0% of tumors in H. pylori-positive group presented as depressed (0-IIc) and 80.0% in H. pylori-uninfected group presented as flat (0-IIb). The surface colors of three groups were either whitish discoloration or subtle reddish roughness with or without spontaneous bleeding, which was significantly different (P = 0.002). 90.0% of the cases in H. pylori-uninfected group manifested as discoloration and 83.3% of the cases in H. pylori-positive group manifested as reddish roughness with or without spontaneous bleeding. With BLI/NBI-ME, demarcation line (DL) was observed in all the cases. Irregular microvascular patterns (MVP) and irregular microsurface patterns (MSP) were both observed in 89.7% of the cases. 86.2% of the tumors exhibited enlarged intervening part (IP).
| Variables | Overall | H. pylori-positive | H. pylori-eradicated | H. pylori-uninfected | P value |
| n | 29 | 12 (41.4) | 7 (24.1) | 10 (34.5) | |
| Tumor size/mm (mean ± SD) | 12.69 ± 5.04 | 13.2 ± 5.41 | 11.58 ± 4.57 | 13.47 ± 5.51 | 0.72 |
| Tumor location | 0.235 | ||||
| Gastric body | 24 (82.8) | 8 (66.7) | 7 (100.0) | 9 (90.0) | |
| Upper third | 7 (29.2) | 3 (37.5) | 3 (42.9) | 1 (11.1) | |
| Middle third | 0 | 0 | 0 | 0 | |
| Lower third | 17 (70.8) | 5 (62.5) | 4 (57.1) | 8 (88.9) | |
| Antrum | 5 (17.2) | 4 (33.3) | 0 | 1 (10.0) | |
| Morphology | 0.101 | ||||
| 0-IIb | 13 (44.8) | 3 (25.0) | 2 (28.6) | 8 (80.0) | |
| 0-IIc | 10 (34.5) | 6 (50.0) | 3 (42.9) | 1 (10.0) | |
| 0-IIa + IIc/0-IIc + IIa | 6 (20.7) | 3 (25.0) | 2 (28.6) | 1 (10.0) | |
| Surface color | 0.002 | ||||
| Whitish discoloration | 15 (51.7) | 2 (16.7) | 4 (57.1) | 9 (90.0) | |
| Reddish roughness/spontaneous bleeding | 14 (48.3) | 10 (83.3) | 3 (42.9) | 1 (10.0) | |
| VS classification | 0.999 | ||||
| DL | 29 (100.0) | 12 (100.0) | 7 (100.0) | 10 (100.0) | |
| Irregular MVP | 26 (89.7) | 10 (83.3) | 7 (100.0) | 9 (90.0) | |
| Irregular MSP | 26 (89.7) | 9 (75.0) | 7 (100.0) | 10 (100.0) | |
| Enlarged/absent IP | 25 (86.2) | 9 (75.0) | 7 (100.0) | 9 (90.0) |
Despite all lesions fulfilling the absolute indications of ESD, there were still 34.5% of the patients opted for surgical resection. Endoscopic resection (65.5%) achieved an en bloc resection rate of 100%, with negative lateral and vertical margins in all cases. Complications included delayed bleeding in 3 cases of H. pylori-positive group and postprocedural fever in 1 case of H. pylori-uninfected group. During a median follow-up period of 26 months, no local recurrence was observed (Table 2).
Our findings indicate that eGSRCC exhibits distinct clinical and endoscopic features based on H. pylori infection status, which can facilitate early diagnosis and appropriate endoscopic management. To our knowledge, this is the first study to specifically compare three groups (H. pylori-positive, H. pylori-eradicated, and H. pylori-uninfected patients), and evaluate various parameters including demographic characteristics, tumor size, location, morphology, surface color, VS classification, en bloc resection, complications, recurrence, and follow-up outcomes.
The majority of eGSRCC cases manifested nonspecific dyspeptic symptoms, clinically mimicking chronic gastritis or peptic ulcer disease, frequently leading to diagnostic delay in initial evaluations. Quite a few patients were diagnosed during routine health examination and some of them presented with abdominal pain, distension, and diarrhea. The high proportion of female patients (75.9%) in our cohort is consistent with previous studies[12]. However, it may also be influenced by the limited sample size inherent to this single-center retrospective study.
The role of H. pylori in gastric carcinogenesis has been extensively documented. Chronic infection leads to a persistent inflammatory response in the gastric mucosa, mediated by cytokines such as interleukin-1β and tumor necrosis factor-α, which in turn promote mucosal atrophy, intestinal metaplasia, and eventually neoplastic transformation[13,14]. Although undifferentiated-type EGC is predominant in H. pylori-uninfected cases, it has been reported that H. pylori infection could cause foveolar-type SRCC and displayed distinct histopathological features[15]. H. pylori eradication is now recognized as a primary prevention strategy for gastric carcinogenesis in high-risk populations[16]. However, the H. pylori-positive group did not demonstrate a significantly larger average tumor size compared to the H. pylori-uninfected group in our study, suggesting that there might be other factors related to tumor growth. Phalanusitthepha et al[17] reported a eGSRCC case series of 12 exhibiting pale, flat mucosal changes without other endoscopic evidence of ulceration, nodular elevation, or depression. Xu et al’s study[9] showed similar results that lesions were mostly presented as flat type, discoloration and located at the lower third. Our study similarly revealed the significant portion of the lesions in H. pylori-uninfected group were located at the lower third of gastric body and presented as type 0-IIb with whitish discoloration. While more lesions in H. pylori-positive group manifested 0-IIc type with reddish roughness with or without spontaneous bleeding. The differences in endoscopic manifestations may be rooted in the distinct mucosal microenvironments driven by H. pylori status. In H. pylori-positive patients, sustained chronic active inflammation leads to mucosal congestion, edema, and increased fragility, which may explain the more frequent presentation of lesions as reddish, rough, and even bleeding-prone depressed types (0-IIc). In contrast, in H. pylori-uninfected patients, the absence of this intense inflammatory background may allow the tumor to develop on normal-appearing mucosa, resulting in the characteristic flat and pale morphology (0-IIb). In the eradicated group, the resolution of inflammation may allow the mucosa to normalize while the neoplastic changes persist. Additionally, all 3 delayed bleeding cases post-ESD were H. pylori-positive and no complications were observed in H. pylori-eradicated group, which further revealed the role of H. pylori in perpetuating mucosal inflammation, highlighting the necessity of H. pylori eradication prior to ESD to mitigate peri-procedural bleeding risk.
ME is now established as a cornerstone modality for effective and accurate diagnosis of early EGC. The Japanese Gastroenterological Association (JGA) has established a unified algorithm for ME diagnosis of EGC (MESDA-G) worldwide to simplify the process of diagnosis and improve accuracy[18]. In Horiuchi et al’s study[19], white-light imaging provided conclusive diagnosis in 47.3% (35/74) of lesions, while 52.7% (39/74) were non-diagnostic. NBI-ME resolved 69.2% (27/39) of indeterminate cases, significantly enhancing the cumulative diagnostic yield to 83.8% (62/74). Following the diagnostic algorithm, all the cases of our study manifested distinct DL and all most all the cases showed irregular MVP, irregular MSP and enlarged IP. However, the VS classification of the lesions among three groups didn’t show significant difference. To the best of our knowledge, there have been no studies about the influences of H. pylori infection status on VS patterns in eGSRCC cases. Remarkably, all the cases in H. pylori-eradicated group showed distinct DL, irregular MVP, irregular MSP and enlarged IP. This demonstrates H. pylori eradication optimized endoscopic visualization for BLI/NBI-ME by reducing background mucosal inflammation and may improve the rate of curative resection with ESD.
It is noteworthy that 34.5% (10/29) of patients ultimately underwent surgical resection despite all lesions fulfilling absolute indications for ESD. This reflects the complexity of real-world clinical decision-making, influenced by multiple factors including patient preference for a perceived definitive surgical resection, varying levels of awareness, acceptance of ESD technique and its long-term outcomes, and the inherent selection bias of this retrospective study. This underscores the need for enhancing patient education and promoting ESD for indicated eGSRCC. All the cases of three groups achieved en bloc resection with no recurrence during a median follow-up of 26 months, indicating that ESD provides satisfying treatment and survival outcomes comparable to surgery for patients with eGSRCC. This result aligns with previous confirmative clinical trial about comparison between ESD and surgery for the curative resection of undifferentiated cancer[20].
This study has some limitations. First, the small sample size (n = 29), with particularly limited numbers in subgroups, may constrain the statistical power of our analyses. Second, the retrospective, single-center design inherently carries risks of selection and information bias. Third, several potential confounding variables were not accounted for, such as gastric mucosal background, medication use, and genetic background, which may independently influence endoscopic appearance or tumor biology. Fourth, the median follow-up period (26 months) remains insufficient to assess the long-term prognosis and recurrence risk of eGSRCC. The absence of observed recurrence may be attributable to the follow-up duration and sample size. Finally, as a single-center study, the patient population and management practices possess certain specificities, limiting the generalizability of our findings. Validation through multicenter studies involving broader populations is warranted.
This study demonstrates that H. pylori infection status plays a crucial role in determining the clinical and endoscopic features of endoscopically resectable eGSRCC. The findings advocate for meticulous endoscopic screening and suggest that advanced imaging techniques should be routinely employed in high-risk populations to enhance the early detection of subtle eGSRCC lesions. Crucially, H. pylori eradication prior to ESD should be considered not only to optimize endo
| 1. | Liu X, Cai H, Sheng W, Yu L, Long Z, Shi Y, Wang Y. Clinicopathological Characteristics and Survival Outcomes of Primary Signet Ring Cell Carcinoma in the Stomach: Retrospective Analysis of Single Center Database. PLoS One. 2015;10:e0144420. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (2)] |
| 2. | Wang X, Li X, Yang P, Qin X, Yu J, Cheng Y, Zhang G, Tang L. The differences in biological behavior and gene expression characteristics between pure and mixed early gastric signet ring cell carcinomas. Dig Liver Dis. 2023;55:815-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 3. | Japanese Gastric Cancer Association. Japanese Gastric Cancer Treatment Guidelines 2021 (6th edition). Gastric Cancer. 2023;26:1-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 976] [Cited by in RCA: 920] [Article Influence: 306.7] [Reference Citation Analysis (9)] |
| 4. | Ono H, Yao K, Fujishiro M, Oda I, Uedo N, Nimura S, Yahagi N, Iishi H, Oka M, Ajioka Y, Fujimoto K. Guidelines for endoscopic submucosal dissection and endoscopic mucosal resection for early gastric cancer (second edition). Dig Endosc. 2021;33:4-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 380] [Article Influence: 76.0] [Reference Citation Analysis (2)] |
| 5. | Sokic-Milutinovic A, Alempijevic T, Milosavljevic T. Role of Helicobacter pylori infection in gastric carcinogenesis: Current knowledge and future directions. World J Gastroenterol. 2015;21:11654-11672. [PubMed] [DOI] [Full Text] |
| 6. | Wang Y, Li Y, Wang B, Ran D, Zhu C, Li P, Jiang B, Wang S. Early onset, development and histological features of gastric signet-ring cell carcinoma. Front Oncol. 2023;13:1166549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 7. | Jin X, Wu W, Zhao J, Song S, Zhang C, Sun W, Lv B. Clinical Features and Risk Factors for Lymph Node Metastasis in Early Signet Ring Cell Gastric Cancer. Front Oncol. 2021;11:630675. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (2)] |
| 8. | Horiuchi Y, Hirasawa T, Fujisaki J. Endoscopic Features of Undifferentiated-Type Early Gastric Cancer in Patients with Helicobacter pylori-Uninfected or -Eradicated Stomachs: A Comprehensive Review. Gut Liver. 2024;18:209-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 9. | Xu J, Zhu J, Lin L, Li Z, Gu F, Wang F, Zhai H. Endoscopic and clinicopathologic features of early gastric signet ring cell carcinoma ≤20 mm: a retrospective observational study. Scand J Gastroenterol. 2023;58:38-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 10. | Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2962] [Cited by in RCA: 2911] [Article Influence: 194.1] [Reference Citation Analysis (7)] |
| 11. | Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy. 2009;41:462-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 341] [Article Influence: 20.1] [Reference Citation Analysis (1)] |
| 12. | Kao YC, Fang WL, Wang RF, Li AF, Yang MH, Wu CW, Shyr YM, Huang KH. Clinicopathological differences in signet ring cell adenocarcinoma between early and advanced gastric cancer. Gastric Cancer. 2019;22:255-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (5)] |
| 13. | Duan Y, Xu Y, Dou Y, Xu D. Helicobacter pylori and gastric cancer: mechanisms and new perspectives. J Hematol Oncol. 2025;18:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 144] [Article Influence: 144.0] [Reference Citation Analysis (2)] |
| 14. | Qadri Q, Rasool R, Gulzar GM, Naqash S, Shah ZA. H. pylori infection, inflammation and gastric cancer. J Gastrointest Cancer. 2014;45:126-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (2)] |
| 15. | Ma J, Meng Y, Zhou X, Guo L, Fu W. The Prognostic Significance and Gene Expression Characteristics of Gastric Signet-Ring Cell Carcinoma: A Study Based on the SEER and TCGA Databases. Front Surg. 2022;9:819018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 16. | Suzuki H, Mori H. World trends for H. pylori eradication therapy and gastric cancer prevention strategy by H. pylori test-and-treat. J Gastroenterol. 2018;53:354-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 17. | Phalanusitthepha C, Grimes KL, Ikeda H, Sato H, Sato C, Hokierti C, Inoue H. Endoscopic features of early-stage signet-ring-cell carcinoma of the stomach. World J Gastrointest Endosc. 2015;7:741-746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 18. | Muto M, Yao K, Kaise M, Kato M, Uedo N, Yagi K, Tajiri H. Magnifying endoscopy simple diagnostic algorithm for early gastric cancer (MESDA-G). Dig Endosc. 2016;28:379-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 206] [Article Influence: 20.6] [Reference Citation Analysis (2)] |
| 19. | Horiuchi Y, Fujisaki J, Yamamoto N, Shimizu T, Omae M, Ishiyama A, Yoshio T, Hirasawa T, Yamamoto Y, Tsuchida T, Igarashi M, Takahashi H. Accuracy of diagnostic demarcation of undifferentiated-type early gastric cancer for magnifying endoscopy with narrow-band imaging: surgical cases. Surg Endosc. 2017;31:1906-1913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 20. | Takizawa K, Ono H, Hasuike N, Takashima A, Minashi K, Boku N, Kushima R, Katayama H, Ogawa G, Fukuda H, Fujisaki J, Oda I, Yano T, Hori S, Doyama H, Hirasawa K, Yamamoto Y, Ishihara R, Tanabe S, Niwa Y, Nakagawa M, Terashima M, Muto M; Gastrointestinal Endoscopy Group (GIESG) and the Stomach Cancer Study Group (SCSG) of Japan Clinical Oncology Group. A nonrandomized, single-arm confirmatory trial of expanded endoscopic submucosal dissection indication for undifferentiated early gastric cancer: Japan Clinical Oncology Group study (JCOG1009/1010). Gastric Cancer. 2021;24:479-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 84] [Article Influence: 16.8] [Reference Citation Analysis (1)] |