Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.116436
Revised: December 9, 2025
Accepted: January 15, 2026
Published online: March 15, 2026
Processing time: 121 Days and 5.6 Hours
Pancreatic ductal adenocarcinoma (PDAC) is a highly lethal and aggressive malignancy. Although most cases are sporadic, hereditary pancreatitis (HP) represents an important predisposing condition. The substantial overlap in clinical symptoms and imaging findings between chronic pancreatitis and early PDAC often leads to diagnostic delay and missed opportunities for timely inter
A 53-year-old woman with genetically confirmed HP experienced recurrent episodes of pancreatitis and underwent multiple endoscopic retrograde cholangiopancreatography procedures. She was later admitted for evaluation of a markedly elevated carbohydrate antigen 19-9 level and diagnosed with PDAC. Surgical management included distal pancreatectomy, sigmoid colectomy, and total splenectomy, followed by six cycles of adjuvant chemotherapy. Postoperatively, tumor markers normalized, and clinical recovery was achieved.
For individuals with HP, lifelong surveillance and early intervention before malignant progression are strongly recommended.
Core Tip: Hereditary pancreatitis significantly increases the lifetime risk of pancreatic ductal adenocarcinoma and presents diagnostic challenges because of overlapping clinical and imaging features with chronic pancreatitis. Early recognition of hereditary pancreatitis as a cancer-predisposing condition, comprehensive germline testing, and individualized surveillance combining imaging and biomarker assessment are crucial. Timely intervention and multidisciplinary management can improve outcomes in this high-risk population.
- Citation: Tao ZT, Xu L, Chen YL, Gong B. Pancreatic ductal adenocarcinoma developing from hereditary pancreatitis: A case report. World J Gastrointest Oncol 2026; 18(3): 116436
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/116436.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.116436
Pancreatic ductal adenocarcinoma (PDAC) is an aggressive malignancy and the seventh leading cause of cancer-related deaths worldwide. It has a poor prognosis and typically follows a highly invasive clinical course. The age-standardized incidence rate is highest in North America and Western Europe (6.8-7.4 per 100000) and lower in Asia (3.2 per 100000)[1]. Although most cases occur sporadically, 5%-10% show a hereditary predisposition[2]. Hereditary pancreatitis (HP) is a form of chronic pancreatitis (CP) caused by autosomal dominant mutations. It is characterized by progressive exocrine and endocrine pancreatic insufficiency and a markedly increased lifetime risk of pancreatic cancer (PC)[3,4].Germline mutations in genes such as PRSS1 and SPINK1, which cause premature trypsin activation and pancreatic autodigestion, are closely associated with HP[5,6]. The chronic inflammation characteristic of HP creates a tumor-promoting microenvironment. Recent studies have shown a strong relationship between the tumor immune microenvironment and genes co-expressed with carboxyl ester lipase, suggesting that impaired transport of vitamins and trace elements may contribute to the development of CP and PDAC. However, the specific molecular mechanisms underlying malignant transformation remain unclear[7]. Notably, PDAC associated with HP tends to develop at a younger age than PDAC related to alcohol or other causes of pancreatitis, and individuals with HP face a higher lifetime risk of PC. These findings highlight the importance of early surveillance in high-risk populations[8].
We report a case of PDAC in an individual with confirmed HP, emphasizing the challenges in diagnosis and ma
Abdominal pain for one month.
A 53-year-old woman presented with left upper abdominal discomfort lasting one month, without obvious precipitating factors, accompanied by occasional dull pain radiating to the left posterior back. She denied nausea, vomiting, fever, jaundice, diarrhea, or hematochezia. The symptoms were initially untreated and showed no spontaneous improvement.
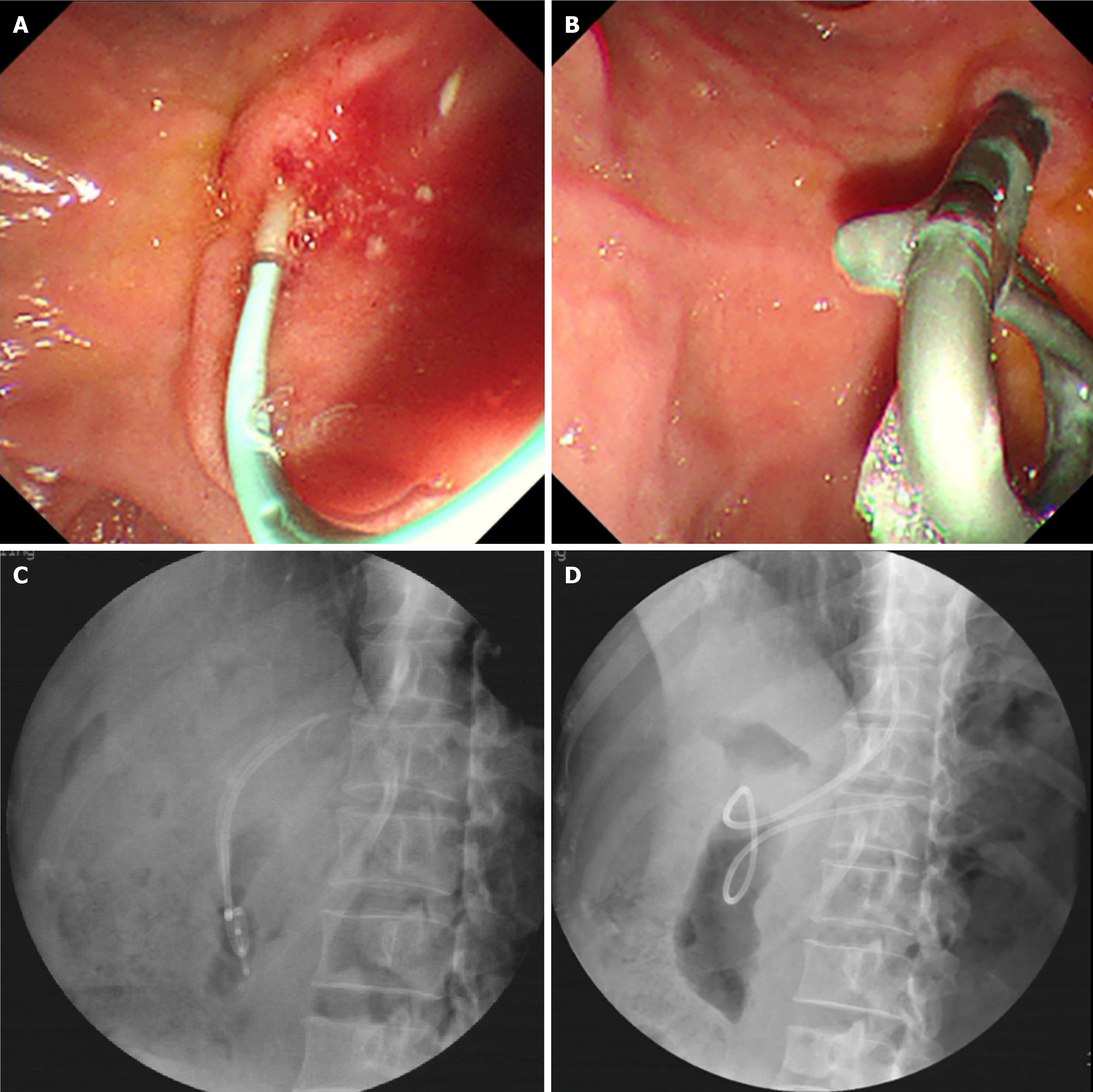
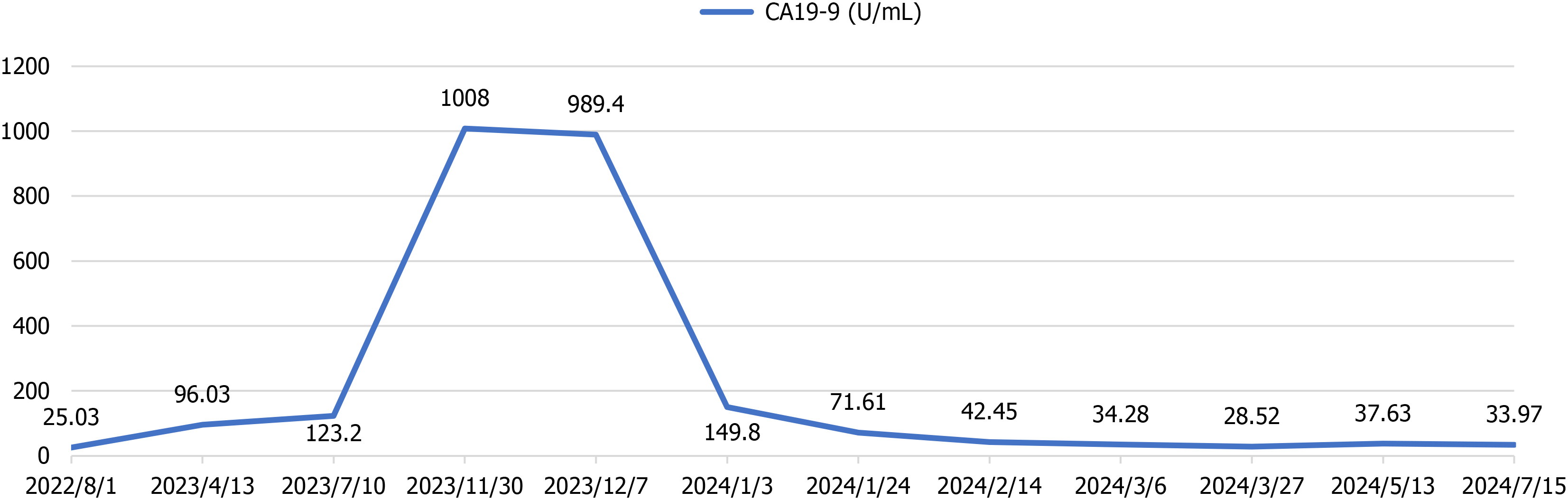
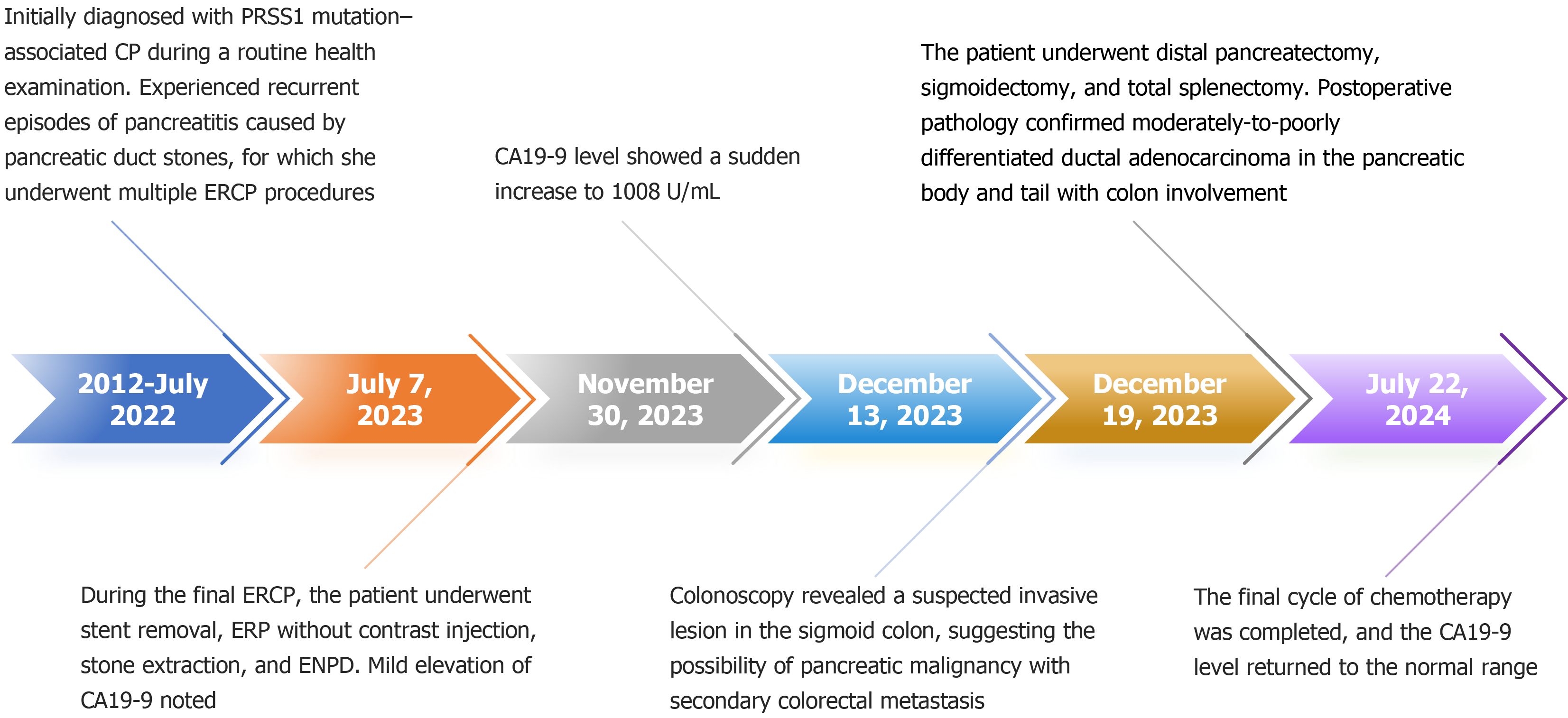
During a routine health examination in 2012, the patient was incidentally found to have pancreatic duct stones. She was asymptomatic at that time, with no abdominal pain, gastrointestinal symptoms, or weight loss. Genetic testing confirmed a PRSS1 mutation, leading to the diagnosis of HP. Magnetic resonance cholangiopancreatography (MRCP) revealed irregular dilation of the pancreatic duct, segmental stenosis, and multiple small filling defects. Between 2012 and 2016, she experienced recurrent episodes of pancreatitis with pancreatic duct stones and underwent several endoscopic retrograde cholangiopancreatography (ERCP) procedures, followed by adjuvant therapy with proton pump inhibitors, antibiotics, and pancreatic enzyme supplements. Her condition subsequently remained stable, with no recurrence of abdominal symptoms. A follow-up MRCP in July 2022 showed pancreatic atrophy, uneven dilation of the pancreatic duct, and a 3-mm nodular hypointense filling defect in the pancreatic head. The pancreatic contour and peripancreatic fat space were clear, with no evidence of retroperitoneal lymphadenopathy. In August 2022, she was admitted to the hospital for recurrent right upper abdominal pain and underwent four ERCP procedures during this period. The final ERCP, performed on July 7, 2023, confirmed CP with pancreatic duct stones (Figure 1). The therapeutic interventions included papillotomy, bilateral endoscopic retrograde pancreatic drainage with subsequent stent removal, endoscopic retrograde pancreatography without contrast injection, stone extraction, and endoscopic nasopancreatic drainage. Since 2022, carbohydrate antigen 19-9 (CA19-9) levels had been mildly elevated. A follow-up test on November 30, 2023, showed a marked increase in CA19-9 to 1008 U/mL.
The patient had a history of diabetes mellitus for two years, managed with miglitol for glycemic control, with fasting blood glucose maintained at 7.0 mmol/L. The patient denied any history of tobacco use or alcohol consumption.
Two sisters, one son, one nephew, and one niece had CP. The PRSS1 mutation was identified in her son and one of her sisters (a first-degree relative), as well as in the sister's son and daughter (second-degree relatives).
No clinically significant abnormalities were identified.
At admission, tumor marker levels were significantly elevated: CA19-9 > 1000.00 U/mL, carcinoembryonic antigen 13.35 ng/mL, carbohydrate antigen 50 62.96 U/mL, and carbohydrate antigen 242 > 200.0 U/mL.
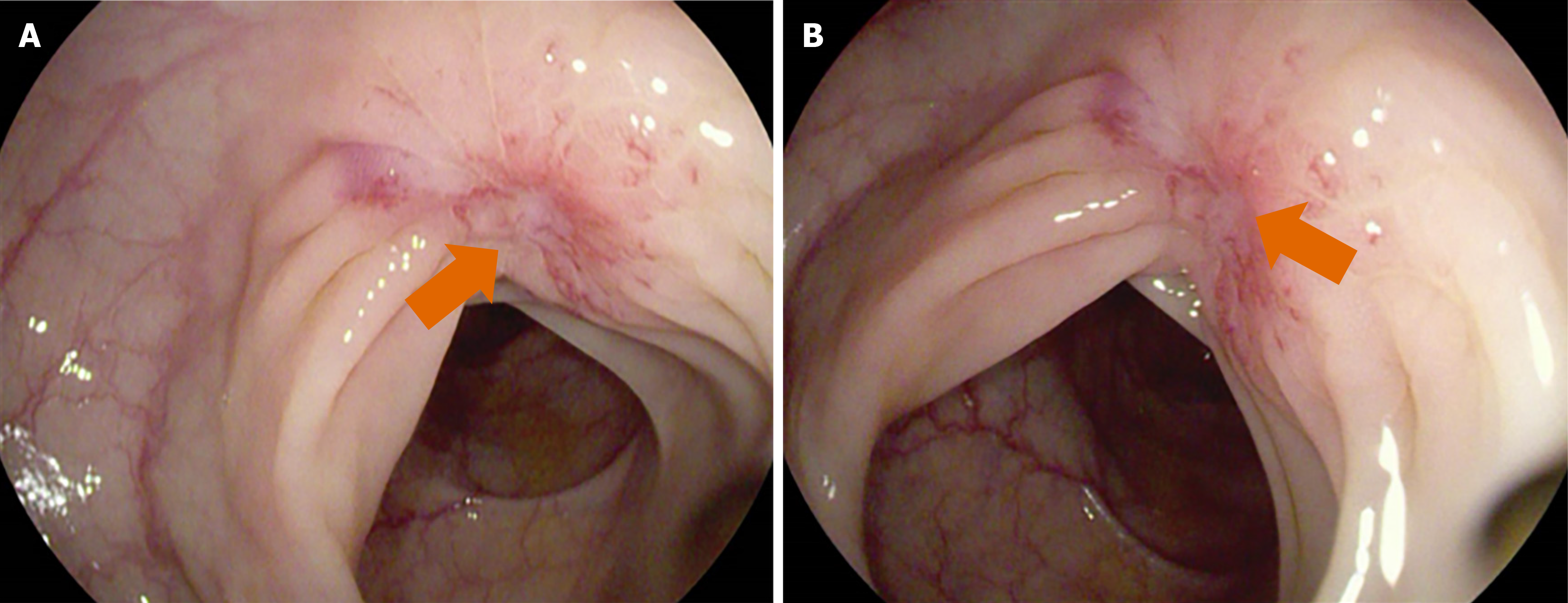
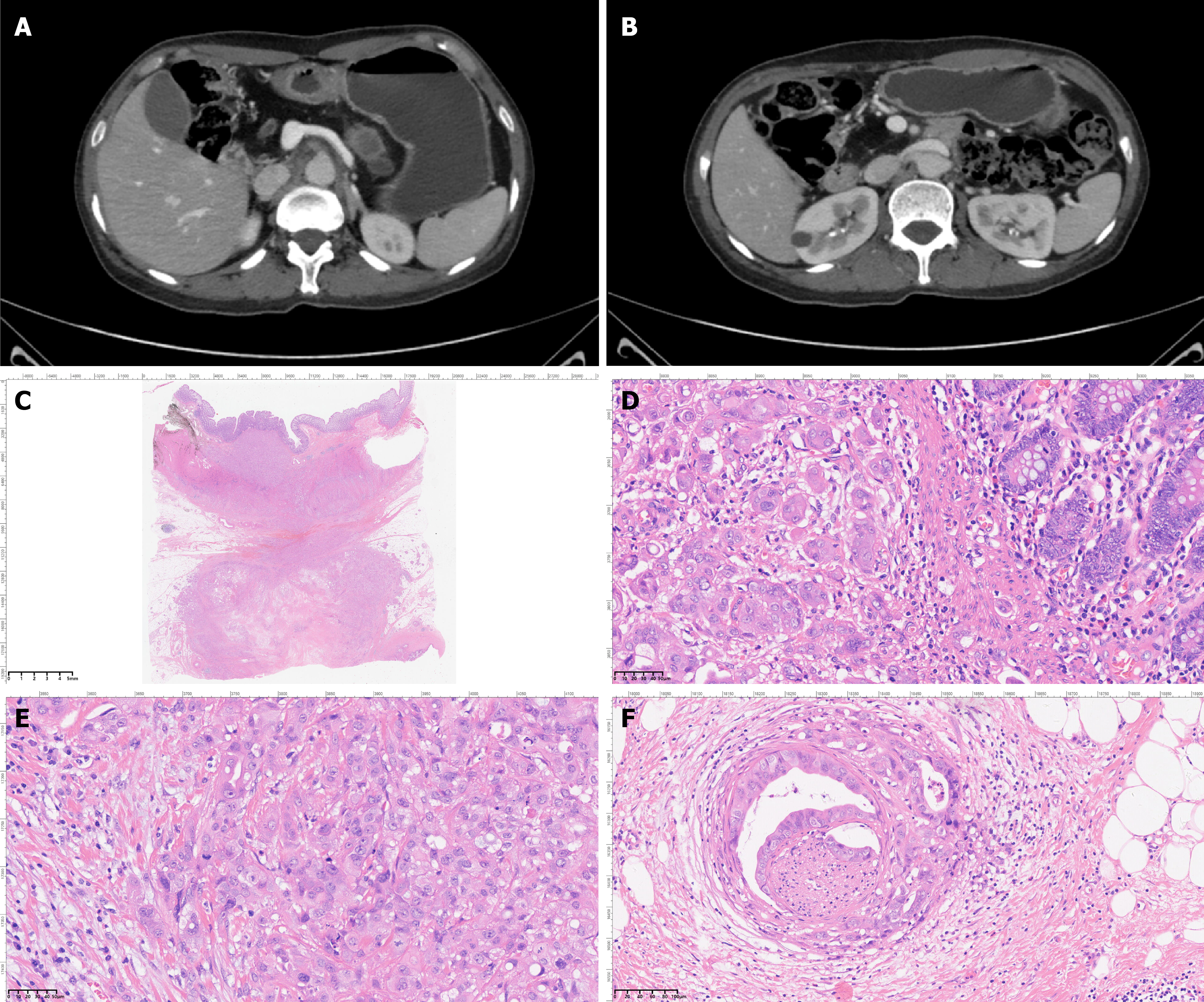
Colonoscopy on December 13, 2023, revealed a suspected invasive lesion in the sigmoid colon, with consideration of pancreatic malignancy and secondary colorectal metastasis (Figure 2). The upper abdomen contrast-enhanced dynamic computed tomography showed pancreatic atrophy with multiple calcifications in the pancreatic head and body, along with a cystic lesion in the pancreatic tail, consistent with chronic atrophic pancreatitis. No tumor tendencies were observed. However, postoperative histopathology confirmed a moderately to poorly differentiated ductal adenocarcinoma involving the pancreatic body and tail with extension to the colon (Figure 3).
PDAC with secondary malignant involvement of the colon.
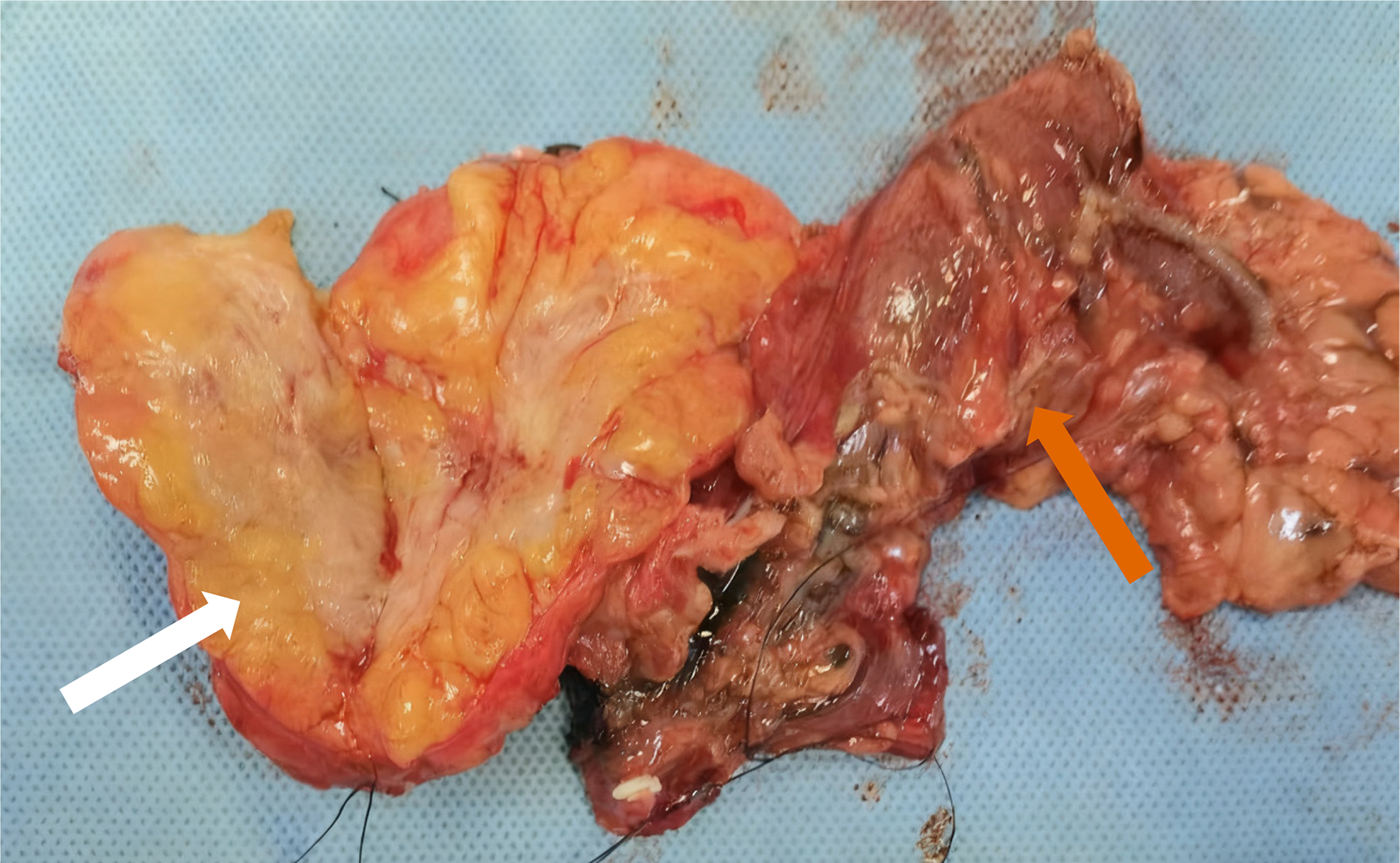
On December 19, 2023, distal pancreatectomy, sigmoidectomy, and total splenectomy were performed (Figure 4). Postoperatively, six cycles of adjuvant chemotherapy were administered using the following regimen: Gemcitabine 1.4 g on days 1 and 8, paclitaxel 150 mg on days 1 and 8, and sintilimab 200 mg on day 1, repeated every three weeks. The final cycle was completed on July 22, 2024.
At six months after surgery, tumor marker (CA19-9) levels had returned to the normal range (Figure 5). The timeline of the patient’s key clinical information was shown in Figure 6. During a recent telephone follow-up, the patient reported that routine re-examination in March 2025 revealed a significant elevation in serum CA19-9 levels (> 3000 U/mL). Local hospital clinicians diagnosed the patient with tumor recurrence. The patient is currently undergoing systemic chemotherapy for recurrent PC at a local hospital. The patient’s overall condition remained stable, and treatment is ongoing.
This report describes a case of PDAC with sigmoid colon metastasis that developed after more than 10 years of disease progression in a patient with HP caused by a PRSS1 gene mutation. The case outlines the full clinical course from pancreatic duct stones and recurrent inflammation to pancreatic atrophy and eventual malignant transformation. It aims to provide in-depth insights into the carcinogenic mechanisms associated with HP, critical warning signals during monitoring (such as dynamic changes in CA19-9 levels), and the clinical significance of achieving radical tumor resection in this high-risk context. Overall, this case offers valuable insights into the long-term management of individuals with HP, reinforcing the necessity of early detection strategies, regular imaging follow-up, and biomarker monitoring to reduce the risk of PC development and progression.
Distinguishing neoplastic transformation from chronic inflammatory changes in HP remains a major clinical challenge. The persistent inflammatory milieu - characterized by progressive ductal stenosis, calcifications, and fibrosis - creates a favorable environment for tumorigenesis while simultaneously masking the early radiological and metabolic signs of PDAC[4]. In this patient, preoperative upper abdominal contrast-enhanced computed tomography did not show any tumor-like alterations; however, postoperative pathological examination confirmed the presence of PDAC. A gradual increase in CA19-9 exceeding 1000 U/mL served as an early indicator of malignant transformation. However, repeated ERCP procedures and stent placements further complicated interpretation of the ductal anatomy. Consequently, even with advanced imaging modalities such as endoscopic ultrasonography and MRCP, identifying malignancy within the setting of HP is often difficult.
In this case, dynamic monitoring of CA19-9 levels proved highly informative. The mild yet persistent elevation noted since 2022 was likely related to benign biliary obstruction or inflammation secondary to CP. In contrast, the abrupt increase to 1008 U/mL in November 2023 represented a critical warning signal strongly suggestive of malignant transformation, even when imaging findings appeared stable. Our clinicians were astute in recognizing this possibility and proceeded with timely surgical intervention; the final postoperative pathology ultimately confirmed this suspicion. This observation highlights that, in high-risk individuals, a marked and rapid elevation in CA 19-9 should trigger prompt and thorough diagnostic evaluation, even in the absence of radiologic confirmation of a mass. Clinicians must therefore maintain a high index of suspicion for PDAC when new ductal irregularities, focal lesions, or biomarker elevations arise in patients with HP.
A large cohort study from the United States demonstrated a markedly increased cumulative risk of PC among individuals with HP. Compared with the general population, those carrying germline mutations in PRSS1 have a substantially higher likelihood of developing life-threatening PDAC[8]. By the age of 70, the cumulative risk of PC approaches 40%, particularly among individuals with paternal inheritance of the mutation[9]. Furthermore, a recent meta-analysis reported a standardized incidence ratio of approximately 63.36 for HP, with incidence rising in proportion to the duration of CP[10]. The latency period between the onset of pancreatitis and cancer development typically ranges from 10 years to 30 years, as illustrated by the present case. These findings reinforce that HP should be regarded as a high-risk pre-malignant condition rather than a benign recurrent inflammatory disorder, warranting proactive and lifelong surveillance to facilitate early detection of PDAC.
For families with HP, risk stratification and management should be guided by established genetic counseling and surveillance protocols. International guidelines recommend comprehensive germline genetic testing for all individuals with suspected HP or PDAC[11,12]. Screening for PRSS1, SPINK1, CFTR, and CTRC mutations supports accurate dia
The importance of such tailored surveillance is exemplified by the present case, which reflects more than a decade of CP preceding PDAC diagnosis and treatment. Given the familial clustering of CP among the siblings and offspring of the patient, earlier genomic analysis and proactive surveillance might have facilitated earlier detection or prevention of malignant transformation.
The management of PDAC arising in the setting of HP poses significant surgical and oncologic challenges. Repeated ductal interventions, extensive parenchymal fibrosis, and chronic inflammation alter normal pancreatic anatomy, making resection technically complex and increasing postoperative risks. In the present case, distal pancreatectomy, sigmoidectomy, and total splenectomy were successfully performed, followed by adjuvant chemotherapy. Although normalization of tumor markers six months after surgery represents a favorable short-term outcome, long-term surveillance remains crucial because HP-associated PDAC often exhibits aggressive biological behavior and a high risk of recurrence. This case’s recent follow-up with tumor recurrence further underscores the long-term pivotal role of tumor marker levels, systematic monitoring, and imaging examinations in PC management. As a widely used and highly relevant tumor marker in clinical practice, CA19-9 levels’ significant re-elevation provides an early and objective biological tumor recurrence warning. This finding strongly confirms that regular monitoring remains a key strategy for detecting occult recurrence and initiating timely intervention, even after initial treatment completion. This case reinforces the scientific rationale for incorporating CA19-9 dynamics into standardized follow-up protocols and alerts clinicians to the importance of closely monitoring its progressive increase, which signals disease progression risk. Continued multidisciplinary follow-up with periodic imaging and biomarker monitoring is therefore essential to ensure early detection of potential relapse and to optimize long-term prognosis.
Emerging evidence indicates that HP-associated PDAC differs molecularly from sporadic PDAC, showing earlier onset and distinct mutation patterns involving KRAS, CDKN2A, SMAD4, and TP53 genes[19]. These differences suggest unique carcinogenic pathways and highlight the need for precision-based approaches to risk assessment and early detection. Future research should focus on refining surveillance protocols and identifying reliable biomarkers capable of predicting malignant transformation in individuals with HP. Integrating molecular profiling, advanced imaging, and liquid biopsy techniques such as circulating tumor DNA analysis may further enhance early diagnosis. For clinicians, these findings emphasize that individuals with hereditary or familial pancreatitis should be managed proactively through structured and lifelong surveillance, rather than treated expectantly, to mitigate the substantial risk of PDAC development.
This case illustrates the natural course and malignant potential of PRSS1-related HP, demonstrating its progression to PDAC. It highlights that management should extend beyond treating acute exacerbations and complications to include a lifelong, proactive, and multidisciplinary surveillance strategy aimed at early detection of malignant transformation. Because the clinical, biochemical, and imaging features of CP often overlap with those of early PC, diagnosis of malignant change is frequently delayed. HP should therefore be recognized as a cancer-predisposing condition, warranting personalized risk reduction through comprehensive germline screening, targeted familial management, and individualized surveillance. Optimal outcomes require an integrated, multidisciplinary approach involving gastroenterology, surgery, oncology, and genetics.
We are grateful to all participants. We deeply appreciate the support of the patient and her family.
| 1. | Simoes PK, Olson SH, Saldia A, Kurtz RC. Epidemiology of pancreatic adenocarcinoma. Chin Clin Oncol. 2017;6:24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 2. | Vietri MT, D'Elia G, Caliendo G, Albanese L, Signoriello G, Napoli C, Molinari AM. Pancreatic Cancer with Mutation in BRCA1/2, MLH1, and APC Genes: Phenotype Correlation and Detection of a Novel Germline BRCA2 Mutation. Genes (Basel). 2022;13:321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 3. | Fujiwara M. Diagnosis of Hereditary Pancreatitis Following the Initial Acute Episode With Multiple Pseudocyst Complications. Cureus. 2024;16:e73653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 4. | Le Cosquer G, Maulat C, Bournet B, Cordelier P, Buscail E, Buscail L. Pancreatic Cancer in Chronic Pancreatitis: Pathogenesis and Diagnostic Approach. Cancers (Basel). 2023;15:761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 5. | Demcsák A, Sahin-Tóth M. Heterozygous Spink1 Deficiency Promotes Trypsin-dependent Chronic Pancreatitis in Mice. Cell Mol Gastroenterol Hepatol. 2024;18:101361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 6. | Németh BC, Sahin-Tóth M. Human cationic trypsinogen (PRSS1) variants and chronic pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2014;306:G466-G473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 7. | Guo K, Zhao Y, Cao Y, Li Y, Yang M, Tian Y, Dai J, Song L, Ren S, Wang Z. Exploring the key genetic association between chronic pancreatitis and pancreatic ductal adenocarcinoma through integrated bioinformatics. Front Genet. 2023;14:1115660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 8. | Shelton CA, Umapathy C, Stello K, Yadav D, Whitcomb DC. Hereditary Pancreatitis in the United States: Survival and Rates of Pancreatic Cancer. Am J Gastroenterol. 2018;113:1376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 73] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 9. | Lowenfels AB, Maisonneuve P, DiMagno EP, Elitsur Y, Gates LK Jr, Perrault J, Whitcomb DC. Hereditary pancreatitis and the risk of pancreatic cancer. International Hereditary Pancreatitis Study Group. J Natl Cancer Inst. 1997;89:442-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 772] [Cited by in RCA: 603] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 10. | Gandhi S, de la Fuente J, Murad MH, Majumder S. Chronic Pancreatitis Is a Risk Factor for Pancreatic Cancer, and Incidence Increases With Duration of Disease: A Systematic Review and Meta-analysis. Clin Transl Gastroenterol. 2022;13:e00463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 88] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 11. | Daly MB, Pal T, Maxwell KN, Churpek J, Kohlmann W, AlHilli Z, Arun B, Buys SS, Cheng H, Domchek SM, Friedman S, Giri V, Goggins M, Hagemann A, Hendrix A, Hutton ML, Karlan BY, Kassem N, Khan S, Khoury K, Kurian AW, Laronga C, Mak JS, Mansour J, McDonnell K, Menendez CS, Merajver SD, Norquist BS, Offit K, Rash D, Reiser G, Senter-Jamieson L, Shannon KM, Visvanathan K, Welborn J, Wick MJ, Wood M, Yurgelun MB, Dwyer MA, Darlow SD. NCCN Guidelines® Insights: Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic, Version 2.2024. J Natl Compr Canc Netw. 2023;21:1000-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 197] [Article Influence: 65.7] [Reference Citation Analysis (0)] |
| 12. | Stoffel EM, McKernin SE, Brand R, Canto M, Goggins M, Moravek C, Nagarajan A, Petersen GM, Simeone DM, Yurgelun M, Khorana AA. Evaluating Susceptibility to Pancreatic Cancer: ASCO Provisional Clinical Opinion. J Clin Oncol. 2019;37:153-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 137] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 13. | Panchoo AV, VanNess GH, Rivera-Rivera E, Laborda TJ. Hereditary pancreatitis: An updated review in pediatrics. World J Clin Pediatr. 2022;11:27-37. [PubMed] [DOI] [Full Text] |
| 14. | Daly MB, Pilarski R, Yurgelun MB, Berry MP, Buys SS, Dickson P, Domchek SM, Elkhanany A, Friedman S, Garber JE, Goggins M, Hutton ML, Khan S, Klein C, Kohlmann W, Kurian AW, Laronga C, Litton JK, Mak JS, Menendez CS, Merajver SD, Norquist BS, Offit K, Pal T, Pederson HJ, Reiser G, Shannon KM, Visvanathan K, Weitzel JN, Wick MJ, Wisinski KB, Dwyer MA, Darlow SD. NCCN Guidelines Insights: Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic, Version 1.2020. J Natl Compr Canc Netw. 2020;18:380-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 309] [Article Influence: 51.5] [Reference Citation Analysis (0)] |
| 15. | Ru N, Wu SY, Wang L, Zhu JH, Xu XN, Guo JY, Hu LH, Li ZS, Zou WB, Liao Z. SPINK1 mutations and risk of pancreatic cancer in a Chinese cohort. Pancreatology. 2021;21:848-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Greenhalf W, Lévy P, Gress T, Rebours V, Brand RE, Pandol S, Chari S, Jørgensen MT, Mayerle J, Lerch MM, Hegyi P, Kleeff J, Castillo CF, Isaji S, Shimosegawa T, Sheel A, Halloran CM, Garg P, Takaori K, Besselink MG, Forsmark CE, Wilcox CM, Maisonneuve P, Yadav D, Whitcomb D, Neoptolemos J; Working group for the International (IAP – APA – JPS – EPC) Consensus Guidelines for Chronic Pancreatitis. International consensus guidelines on surveillance for pancreatic cancer in chronic pancreatitis. Recommendations from the working group for the international consensus guidelines for chronic pancreatitis in collaboration with the International Association of Pancreatology, the American Pancreatic Association, the Japan Pancreas Society, and European Pancreatic Club. Pancreatology. 2020;20:910-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 17. | Ngamruengphong S, Canto MI. Screening for Pancreatic Cancer. Surg Clin North Am. 2016;96:1223-1233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | Shen MJ, Jamali A, Katona BW. Challenges of early detection of pancreatic cancer. J Clin Invest. 2025;135:e191942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Stratford JK, Yan F, Hill RA, Major MB, Graves LM, Der CJ, Yeh JJ. Genetic and pharmacological inhibition of TTK impairs pancreatic cancer cell line growth by inducing lethal chromosomal instability. PLoS One. 2017;12:e0174863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |