Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.116567
Revised: November 20, 2025
Accepted: December 24, 2025
Published online: March 15, 2026
Processing time: 118 Days and 19.2 Hours
The early detection of hepatocellular carcinoma (HCC) in patients with liver cir
To evaluate a dual-parameter liquid biopsy approach by comparing a tumor-derived circulating microRNA and an immune-related marker for discriminating HCC from cirrhosis.
A prospective observational case-control study included 211 participants: 70 with HCC; 60 with hepatitis C virus-related liver cirrhosis; and 81 healthy controls. Pla
Plasma levels of hsa-miR-21-5p, hsa-miR-155-5p, and LAIR-1 expression were significantly elevated in the HCC group compared with the cirrhosis and control groups (P < 0.001). For distinguishing HCC from cirrhosis, both hsa-miR-21-5p and LAIR-1 MFI demonstrated outstanding diagnostic performance with AUCs of 0.990 [95% confidence interval: 0.974-1.000] and 0.997 (95% confidence interval: 0.990-1.000), respectively. At optimal cutoffs hsa-miR-21-5p showed a sensitivity of 98.6% and specificity of 96.7% while LAIR-1 MFI showed 100% sensitivity and 98.3% specificity. hsa-miR-155-5p and the percentage of LAIR-1+ T cells showed lower but significant per
This dual-parameter approach, integrating plasma hsa-miR-21-5p and T cell LAIR-1 MFI, achieved exceptional diagnostic accuracy for HCC in a high-risk cohort. While their individual performance was comparable, LAIR-1 MFI provides unique, independent diagnostic information. These findings nominate a promising, integrated liquid biopsy strategy to improve HCC surveillance.
Core Tip: Early, accurate, and noninvasive diagnosis of hepatocellular carcinoma (HCC) remains a major clinical challenge, particularly in Egypt, where hepatitis C virus-related cirrhosis is prevalent. This study introduced a novel dual-parameter liquid biopsy combining plasma miR-21-5p (a tumor-derived oncogenic microRNA) and T cell leukocyte-associated immunoglobulin-like receptor-1 mean fluorescence intensity (an immune checkpoint marker). In a prospective Egyptian cohort, this approach achieved exceptional diagnostic accuracy (area under the curve > 0.99) and maintained high sensitivity in early-stage and α-fetoprotein-negative HCC. The findings proposed an integrated molecular-immune diagnostic pathway that could substantially improve HCC surveillance and early detection in high-risk populations.
- Citation: Sherief DE, Shehata HH, Nosair N, Othman AAA, Sadaka E, Elgamal R. Dual-parameter liquid biopsy using plasma miR-21-5p and T cell LAIR-1 mean fluorescence intensity for hepatocellular carcinoma diagnosis in a high-risk Egyptian cohort. World J Gastrointest Oncol 2026; 18(3): 116567
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/116567.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.116567
Hepatocellular carcinoma (HCC) remains one of the most lethal cancers worldwide and a leading cause of cancer-related deaths. Global estimates from the GLOBOCAN 2020 report millions of new cancer cases and show that liver cancer is among the top contributors to cancer mortality, reflecting a growing global burden of HCC driven by viral hepatitis, metabolic disease, and environmental exposures[1]. Clinically, the challenge is blunt and familiar: Curative therapies (resection, ablation, transplantation) are effective primarily when HCC is detected at early stages, yet most patients are diagnosed late when therapeutic options and survival are limited[2].
Early detection depends on accurate surveillance of at-risk populations, principally patients with advanced fibrosis or established cirrhosis. Current standard surveillance relies on periodic abdominal ultrasounds with or without serum α-fetoprotein (AFP) testing. While ultrasound is noninvasive and widely available, its sensitivity for small, early HCC is modest and highly operator-dependent; the addition of AFP improves sensitivity but remains imperfect, leaving an urgent need for better, noninvasive biomarkers to improve early detection and guide timely therapy[3].
The clinical imperative is especially acute in Egypt where the epidemiologic profile amplifies the problem. National registry data show that liver cancer constitutes a major proportion of cancer incidence in Egypt, reflecting a high population prevalence of chronic hepatitis C virus (HCV) infection and its downstream burden of cirrhosis and HCC[4]. Several Egyptian cohorts have likewise documented an older median age at presentation, high rates of cirrhosis, and frequent comorbid metabolic disease, all of which complicate surveillance and management strategies in real-world practice.
In parallel with technical advances in molecular diagnostics, circulating microRNAs (miRNAs) have emerged as robust, stable, and clinically attractive biomarkers. miRNAs are short, noncoding RNAs that modulate gene expression at the post-transcriptional level and are readily detectable in plasma and serum. Among them, miR-21 is one of the most extensively investigated oncogenic miRNAs (oncomiR). It has been mechanistically linked to hepatocarcinogenesis through suppression of tumor suppressors such as PTEN and PDCD4 and promotion of prosurvival signaling pathways[5,6]. Multiple meta-analyses and systematic reviews have reported that circulating miR-21 demonstrates good diagnostic performance for HCC with pooled sensitivities and specificities that support its role as a promising noninvasive marker, yet heterogeneity between studies and differences in sample preparation, normalizers, and cutoffs temper generalizability[7,8].
Regionally, several Egyptian studies have interrogated circulating miRNAs in HCC. Notably, Eldosoky et al[9] recently evaluated panels that included hsa-miR-21-5p and hsa-miR-155-5p in Egyptian patients infected with HCV and reported improved diagnostic accuracy when combining miRNAs with AFP, underscoring the local promise of miRNA-based assays. Other local investigations have replicated significant upregulation of miR-21 in Egyptian HCC cohorts, sup
Beyond molecular signatures, mounting evidence points to the importance of immune-checkpoint and inhibitory receptors in the tumor microenvironment as both mechanistic drivers and potential biomarkers. Leukocyte-associated immunoglobulin-like receptor-1 (LAIR-1, CD305), an inhibitory collagen receptor expressed on T cells and myeloid cells, modulates immune activation and has been implicated in tumor immune evasion across several cancers[11]. Recent translational studies describe LAIR-1 expression in HCC tissues and its association with tumor biology; experimental work suggests LAIR-1 may influence proliferation and invasion through pathways including PI3K-AKT-mTOR[12,13]. Importantly, a case-control study from an Egyptian cohort reported higher LAIR-1 expression on cytotoxic T cells in HCC compared with patients with cirrhosis and healthy controls and suggested that combining LAIR-1 indices with AFP could improve discrimination of malignant transformation in HCV-related cirrhosis[14]. These data support the concept that immune phenotyping when paired with circulating molecular markers may provide a complementary diagnostic signal.
Despite these advances a clear and actionable knowledge gap remains. Most prior studies have evaluated either circulating miRNAs or immune markers independently; head-to-head comparisons of circulating miR-21 vs other candidate miRNAs (for example, miR-155) and immune markers, such as LAIR-1, within the same well-characterized Egyptian cohort are scarce. Furthermore, formal diagnostic accuracy metrics [area under the curve (AUC), optimal cutoffs, sensitivity, specificity] reported in Egyptian populations are limited, and few studies have directly contrasted molecular and immune biomarkers to identify the single or combined marker with the greatest translational potential for local surveillance programs.
The scientific rationale for the present work, therefore, rests on two pillars. First, miR-21 captures tumor-intrinsic oncogenic signaling that is directly tied to tumorigenesis and its shedding into the circulation. Second, LAIR-1 captures immune-microenvironmental suppression that accompanies malignant transformation. A comparative evaluation of these orthogonal biomarker classes in the same clinical population may identify a superior, clinically implementable test or a simple combined algorithm that improves early HCC detection beyond current standards.
Accordingly, we conducted a prospective case-control study in an Egyptian cohort to: (1) Quantify plasma hsa-miR-21-5p and hsa-miR-155-5p levels and LAIR-1 mean fluorescence intensity (MFI) expression in patients with HCC, patients with HCV-related cirrhosis, and healthy controls; (2) Compare the diagnostic performance of these markers using re
This prospective, observational, case-control study was conducted to evaluate and compare the diagnostic performance of plasma hsa-miR-21-5p, hsa-miR-155-5p, and T cell LAIR-1 expression in distinguishing HCC from liver cirrhosis. This prospective observational case-control study was designed and reported in accordance with the STROBE statement[15].
The study was conducted at the Clinical Pathology, Internal Medicine, and Oncology Departments of Kafrelsheikh University Hospitals, Egypt in collaboration with the Internal Medicine and Clinical Pathology Departments of Suez University Hospital between January 2022 and June 2024. Both institutions are tertiary referral centers providing advanced hepatology, oncology, and laboratory diagnostic services.
A total of 211 participants were enrolled and categorized into three groups: 70 patients with newly diagnosed, treatment-naïve HCC; 60 patients with HCV-related liver cirrhosis without evidence of malignancy; and 81 age-matched and sex-matched healthy controls. The sample size was estimated using MedCalc software (MedCalc Software Ltd., Ostend, Belgium) for ROC curve analysis, assuming an expected AUC of 0.85 compared with a null hypothesis of 0.70 with a power of 90% and a significance level of 0.05[16]. This calculation required a minimum of 60 participants per group. Our final sample size met or exceeded this threshold (HCC: n = 70, cirrhosis: n = 60, control: n = 81) to enhance statistical robustness. Missing data represented less than 3% of the dataset and were handled using pairwise deletion after confirming randomness by Little’s MCAR test (P > 0.20). No variable had more than 3% missing values, and all analyses were conducted on complete-case data after verification of missingness at random.
This study received approval from the Medical Research Ethics Committee of Kafrelsheikh University (Reference No. KFSIRB200-107). Written informed consent was obtained from all participants before enrollment. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki (2013 revision) and complied with the ethical standards of the Egyptian Ministry of Health research regulations.
Inclusion criteria: (1) Adults aged 18 years or older; (2) HCC was diagnosed radiologically or histopathologically in accordance with the 2022 American Association for the Study of Liver Diseases guidelines[17]; (3) The cirrhosis group included patients with established HCV-related liver cirrhosis without any radiologic or biomarker evidence of HCC; and (4) Healthy controls were individuals with no clinical or laboratory evidence of liver disease and negative viral hepatitis markers.
Exclusion criteria: (1) Previous or concurrent malignancy, autoimmune or hematologic disorder, significant alcohol intake, recent infection within the preceding 4 weeks, or receiving immunosuppressive therapy; and (2) Samples that were hemolyzed or insufficient for analysis. These criteria ensured that potential confounders affecting circulating miRNA or immune marker expression were minimized.
All participants underwent comprehensive clinical assessment and baseline investigations, including a detailed medical history, physical examination, and imaging evaluation with abdominal ultrasonography and when indicated triphasic CT. Laboratory tests included liver function profile, complete blood count, coagulation parameters, HCV antibody, HBV surface antigen, and serum AFP. All clinical and laboratory data were recorded on standardized case report forms to ensure consistency.
Blood sampling and processing: Venous blood samples (5 mL) were drawn from each participant after an overnight fast. Plasma samples were collected in EDTA tubes, centrifuged at 3000 rpm for 15 min at 4 °C, and aliquoted for storage at
Routine biochemical and hematological parameters were determined using standard automated analyzers. All laboratory procedures were conducted in accordance with the manufacturer’s guidelines and under strict internal quality control. To minimize potential bias laboratory personnel performing the biomarker assays were blinded to participants’ clinical group assignments (HCC, cirrhosis, or control).
RNA extraction and quantitative real-time PCR: Total RNA, including small RNAs, was isolated from 200 μL of plasma using the miRNeasy Serum/Plasma Kit (Qiagen, Germany; Cat. No. 217184) according to the manufacturer’s protocol. Samples were processed in randomized batches to control for technical batch effects. To control for extraction efficiency, 3.5 μL of miScript miRNA Mimic (Syn-cel-miR-39, Qiagen, Germany) was spiked into each sample during the lysis step. RNA purity and concentration were verified spectrophotometrically, accepting samples with an A260/A280 ratio between 1.8 and 2.1. Complementary DNA synthesis was performed using the miScript II RT Kit (Qiagen, Germany; Cat. No. 218161). Reverse transcription (RT) and quantitative PCR (qPCR) for all samples were performed using master mixes to ensure reaction consistency, and a calibrator sample was included in every qPCR run to allow for inter-run calibration. Quantitative real-time PCR was carried out using the miScript SYBR Green PCR Kit (Qiagen, Germany; Cat. No. 218073) on a Rotor-Gene Q thermal cycler (Qiagen, Germany).
Specific primers were employed for hsa-miR-21-5p (Assay ID MS00009079) and hsa-miR-155-5p (Assay ID MS00031486) with SNORD68 serving as the endogenous reference control[18]. Thermal cycling conditions comprised an initial activation step at 95 °C for 15 min, followed by 40 cycles of denaturation at 94 °C for 15 s, annealing at 55 °C for 30 s, and extension at 70 °C for 30 s. A melting curve analysis was performed at the end of each run to confirm am
Flow cytometric analysis of LAIR-1 expression: Surface expression of LAIR-1 on T lymphocytes was analyzed using anti-human CD3 FITC (BioLegend, Cat. No. 300306) and anti-human LAIR-1 PE (BioLegend, Cat. No. 337604) antibodies, along with matched isotype controls. Antibody concentrations were preoptimized via titration, and the same antibody lots were used throughout the study to ensure consistency.
For staining 100 μL of whole blood was incubated with the antibody cocktail for 20 min at 4 °C in the dark. Red blood cells were then lysed with BD FACS™ Lysing Solution, washed twice in PBS, and resuspended for immediate analysis on a BD FACSVerse™ flow cytometer. Instrument performance was calibrated daily using BD Cytometer Setup & Tracking Beads. Fluorescence compensation was performed using single-stained UltraComp eBeads™ (Thermo Fisher Scientific) to minimize spectral overlap. A minimum of 50000 CD3+ T cell events was acquired per sample. Data acquisition were performed using FACSuite™ software, and analyses were carried out in FlowJo v10 (Tree Star, United States).
LAIR-1 expression was expressed as the percentage of LAIR-1-positive T cells (LAIR-1+ TC %) and the median fluorescence intensity (MedFI) of LAIR-1 on CD3+ T cells. The MedFI, representing the median receptor density per cell, was prioritized as it provides a robust quantitative measure that is less sensitive to outlier values and gating strategy compared with MFI. These standardization procedures were implemented to ensure data accuracy, reproducibility, and compliance with MIFlowCyt guidelines[20].
The primary outcome measure was the diagnostic accuracy of plasma hsa-miR-21-5p for differentiating HCC from cirrhosis, expressed as AUC with 95% confidence interval (CI), sensitivity, and specificity. Secondary outcomes included the diagnostic performance of hsa-miR-155-5p and LAIR-1 parameters, comparison of correlated AUCs using DeLong’s test, and evaluation of combined biomarker models through multivariable logistic regression.
Data were analyzed using IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, NY, United States) and MedCalc version 22.017 (MedCalc Software Ltd., Ostend, Belgium). The normality of continuous variables was examined using the Shapiro-Wilk test. Continuous variables were presented as median and interquartile range (IQR) and compared among groups using the Kruskal-Wallis test, followed by Dunn’s post hoc test for pairwise comparisons with Bonferroni adjustment. Categorical variables were expressed as n (%) and compared using the χ2 test or Fisher’s exact test as appropriate.
Diagnostic performance was evaluated using ROC curve analysis, and optimal cutoff points were determined using the Youden index[21]. Comparisons between correlated AUCs were performed using the DeLong nonparametric approach[22]. Multivariable logistic regression analysis was conducted to identify independent predictors of HCC diagnosis, adjusting for age, sex, and AFP levels. Model calibration was tested using the Hosmer-Lemeshow goodness-of-fit test while discrimination was quantified by AUC. Multicollinearity among continuous predictor variables in the logistic regression model was assessed using the variance inflation factor (VIF). A VIF value greater than 5 was considered indicative of significant collinearity. Internal validation of model performance was assessed using bootstrap resampling (500 iterations). Correlations between continuous variables were analyzed using Spearman’s rank correlation coefficient (ρ).
All tests were two-tailed, and statistical significance was defined as P < 0.05. Graphical presentations, including boxplots, ROC curves, and correlation scatterplots, were generated using R software version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria) with the ggplot2 and pROC packages to ensure reproducible and high-quality visualizations.
A total of 211 participants were included in the final analysis: 70 patients with HCC; 60 with HCV-related liver cirrhosis; and 81 healthy controls. The HCC group was significantly older [median age 66.0 years (IQR: 6)] than both the cirrhosis [60.0 years (IQR: 6)] and control groups [57.5 years (IQR: 7)] (Kruskal-Wallis P < 0.001). Sex distribution was similar across groups. As expected serum AFP levels were substantially higher in the HCC group compared with the cirrhosis and control groups (P < 0.001, Table 1). Data completeness were high across all variables with less than 3% missing data overall; missing observations were confirmed to be missing completely at random (Little’s test, P > 0.20) and handled by pairwise deletion.
| Parameter | HCC (n = 70) | Cirrhosis (n = 60) | Control (n = 81) | P value |
| Age in years | 66 (6) | 60 (6) | 57.5 (7) | < 0.0011 |
| Female sex | 24 (34.3) | 20 (33.3) | 31 (38.3) | 0.3502 |
| AFP in ng/mL | 305 (720) | 18 (32) | 4.5 (3) | < 0.0011 |
| ALT in U/L | 65 (38) | 58 (35) | 28 (10) | < 0.0011 |
| AST in U/L | 75 (42) | 62 (38) | 25 (9) | < 0.0011 |
| Albumin in g/dL | 3.1 (0.6) | 3.4 (0.5) | 4.2 (0.3) | < 0.0011 |
Tumor characteristics of the patients with HCC are detailed in Table 2. The cohort included a range of disease stages with 47.1% (n = 33) presenting with a solitary, small lesion. Notably, a majority of patients (60.0%) had regional lymph node metastasis, and 42.9% had distant metastasis, indicating a cohort with a significant proportion of advanced disease.
| Tumor feature | n (%) |
| Solitary lesion (< 1.9 cm; no invasion) | 33 (47.1) |
| Multiple lesions (no vascular invasion) | 20 (28.6) |
| Multiple lesions (with vascular invasion) | 17 (24.3) |
| Regional lymph-node metastasis | 42 (60.0) |
| Distant metastasis | 30 (42.9) |
To evaluate the potential influence of disease stage on our leading biomarkers, we stratified the HCC cohort (n = 70) according to the Barcelona Clinic Liver Cancer (BCLC) staging system. The cohort included 38 patients (54.3%) with early-stage disease (BCLC 0/A) and 32 patients (45.7%) with advanced-stage disease (BCLC B/C). As anticipated plasma levels of both hsa-miR-21-5p and LAIR-1 MFI were significantly higher in patients with advanced-stage HCC compared with those with early-stage disease (Table 3). This confirms that these biomarkers reflect increasing tumor burden and disease progression.
| Biomarker | BCLC stage 0/A (early) (n = 38) | BCLC stage B/C (advanced) (n = 32) | P value |
| hsa-miR-21-5p | 28.4 (25.8-34.1) | 39.2 (31.5-48.9) | 0.021 |
| LAIR-1 MFI | 39.8 (36.2-44.3) | 46.5 (41.8-50.1) | 0.007 |
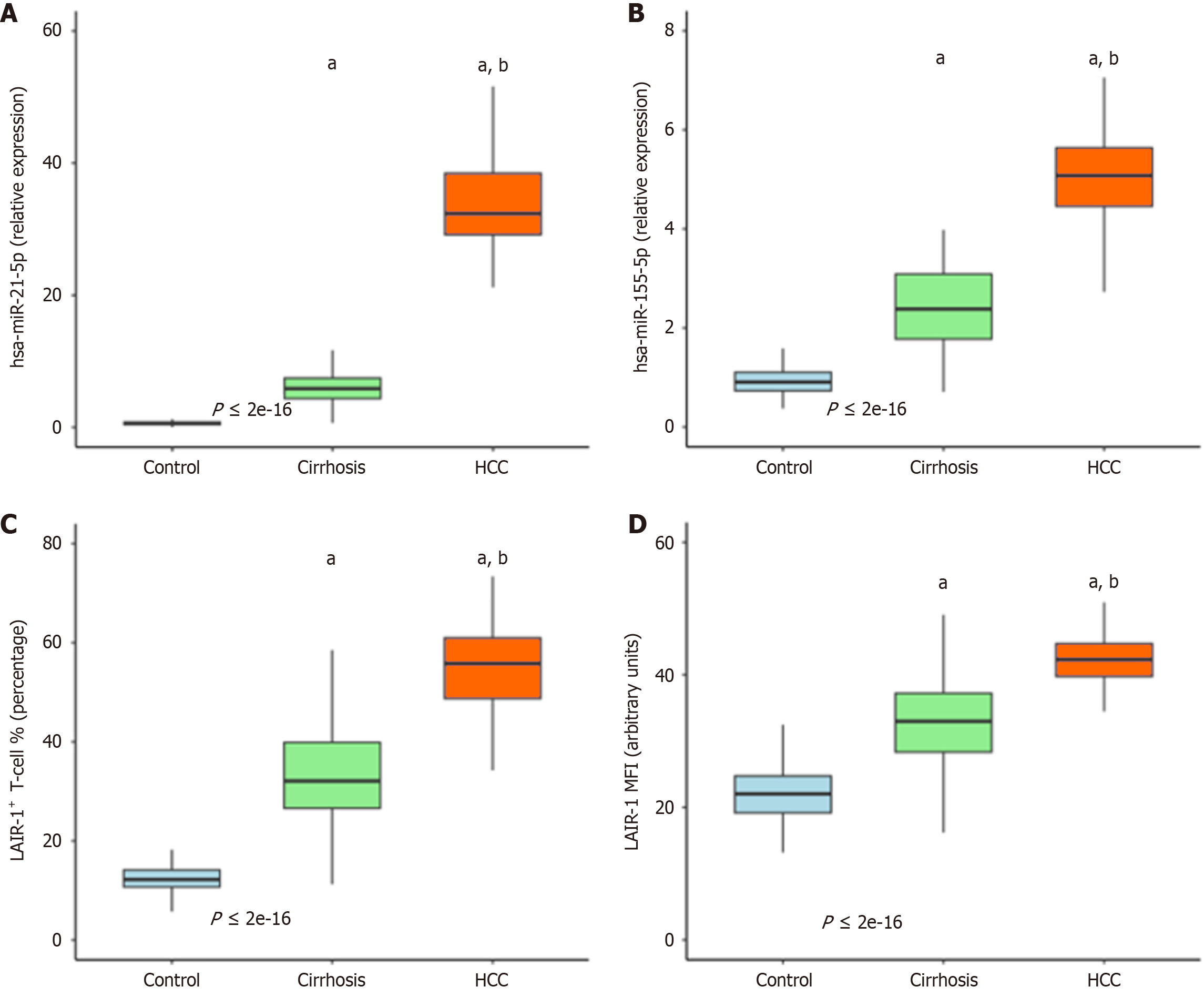
Circulating miRNA quantification and flow cytometric immune phenotyping revealed significant dysregulation across the study groups with a progressive increase from controls to patients with cirrhosis to those with HCC (Tables 4 and 5, Figure 1). Plasma hsa-miR-21-5p was the most markedly elevated biomarker, showing a significant progressive increase (Kruskal-Wallis P < 0.001). A similar though less pronounced pattern was observed for hsa-miR-155-5p (P < 0.001). This pattern of progressive increase was mirrored in the immune checkpoint marker LAIR-1 for both the percentage of positive T cells (LAIR-1+ TC %) and the MFI (P < 0.001 for both), indicating robust immune dysregulation during hepatocarcinogenesis. Post hoc analysis with Bonferroni correction confirmed that these differences were statistically significant for all pairwise comparisons (HCC vs cirrhosis, HCC vs control, cirrhosis vs control) for all biomarkers (adjusted P < 0.01 for all comparisons).
| Biomarker | HCC (n = 70) | Cirrhosis (n = 60) | Control (n = 81) | P value |
| hsa-miR-21-5p | 34.0 (32.5) | 6.0 (5.25) | 0.6 (0.6) | < 0.001 |
| hsa-miR-155-5p | 5.1 (2.2) | 2.35 (1.23) | 0.9 (0.5) | < 0.001 |
| Parameter | HCC (n = 70) | Cirrhosis (n = 60) | Control (n = 81) | P value |
| LAIR-1+ T cell % | 56.7 (31.33) | 33.0 (21.1) | 11.75 (5.33) | < 0.001 |
| LAIR-1 MFI | 42.3 (7.95) | 30.0 (16.0) | 22.4 (8.13) | < 0.001 |
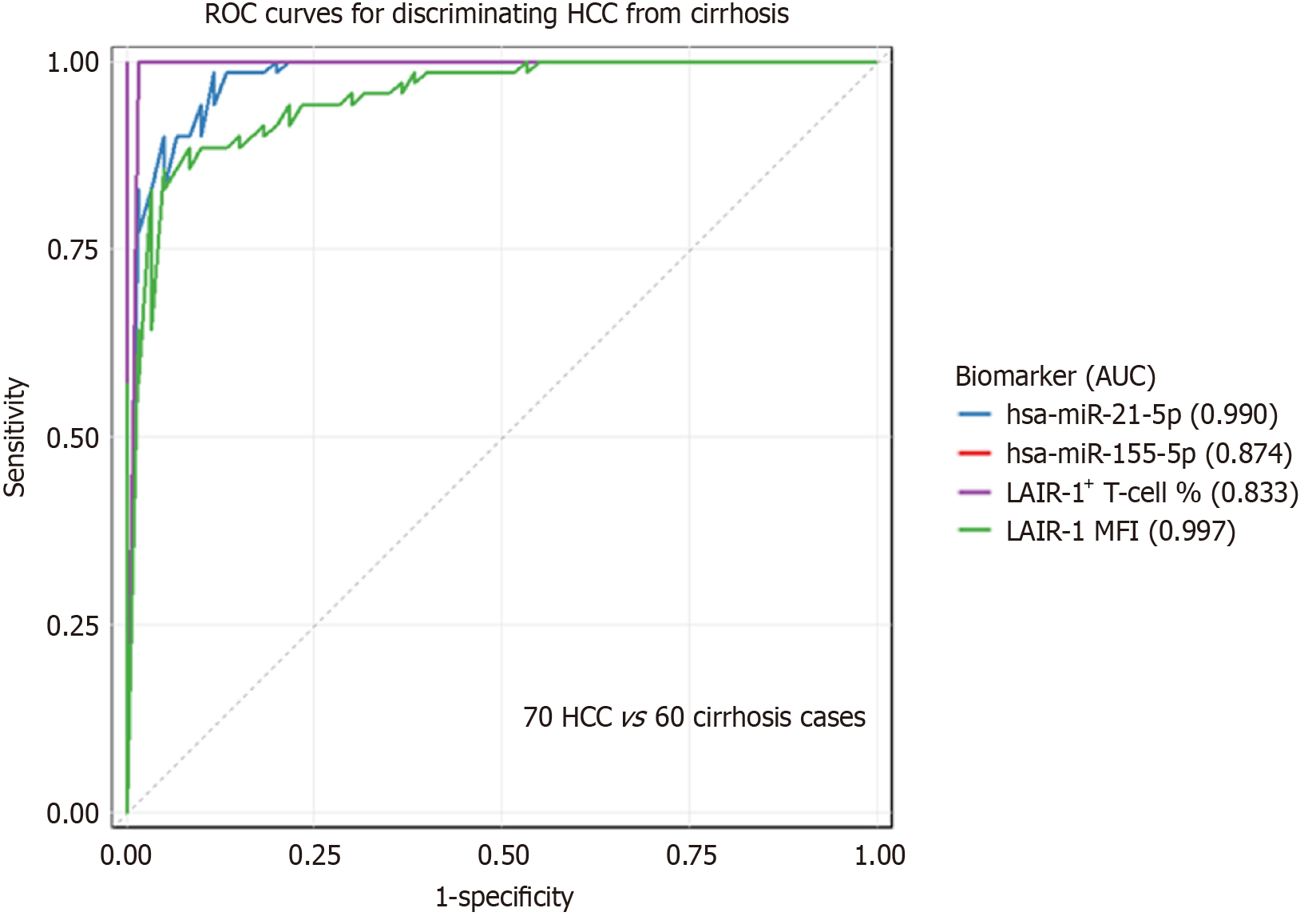
The diagnostic performance of all biomarkers for the critical task of distinguishing HCC from cirrhosis was evaluated using ROC curve analysis (Table 6). Two biomarkers demonstrated exceptional, near-perfect discrimination: Plasma hsa-miR-21-5p (AUC = 0.990; 95%CI: 0.974-1.000) and LAIR-1 MFI (AUC = 0.997; 95%CI: 0.990-1.000). At their optimal cutoffs, hsa-miR-21-5p provided a sensitivity of 98.6% and a specificity of 96.7% while LAIR-1 MFI achieved 100% sensitivity and 98.3% specificity. In contrast, the other biomarkers showed lower albeit significant diagnostic accuracy: Hsa-miR-155-5p (AUC = 0.874); and LAIR-1+ T cell % (AUC = 0.833). The latter operated as a high-specificity rule-in test (100% specificity) but with limited sensitivity (52.9%) (Figure 2).
| Biomarker | AUC (bootstrap 95%CI) | Optimal cutoff | Sensitivity (%) | Specificity (%) |
| hsa-miR-21-5p | 0.990 (0.974-1.000) | > 14.0 | 98.6 | 96.7 |
| hsa-miR-155-5p | 0.874 (0.809-0.930) | > 3.8 | 75.7 | 93.3 |
| LAIR-1+ T cell % | 0.833 (0.767-0.895) | > 55.9 | 52.9 | 100.0 |
| LAIR-1 MFI | 0.997 (0.990-1.000) | > 29.3 | 100.0 | 98.3 |
Pairwise comparisons of the AUC were performed using a bootstrap approach to approximate DeLong’s test (Table 7). The AUC of plasma hsa-miR-21-5p was statistically superior to that of hsa-miR-155-5p (0.990 vs 0.874; z = 4.025, P = 5.7 × 10-5). Most importantly, no significant difference was observed between the diagnostic accuracy of hsa-miR-21-5p and LAIR-1 MFI (0.990 vs 0.997; z = -1.218, P = 0.223), confirming their equivalent performance. Furthermore, LAIR-1 MFI demonstrated a significantly larger AUC than hsa-miR-155-5p (0.997 vs 0.874; z = -4.275, P = 1.9 × 10-5).
| Comparison | AUC1 | AUC2 | z statistic | P value |
| hsa-miR-21-5p vs hsa-miR-155-5p | 0.990 | 0.874 | 4.025 | < 0.001 |
| hsa-miR-21-5p vs LAIR-1 MFI | 0.990 | 0.997 | -1.218 | 0.223 |
| hsa-miR-155-5p vs LAIR-1 MFI | 0.874 | 0.997 | -4.275 | < 0.001 |
To identify independent predictors and assess the potential synergy of a multimarker approach, a multivariable logistic regression model was constructed incorporating hsa-miR-21-5p, hsa-miR-155-5p, LAIR-1 MFI, and AFP. To formally assess potential multicollinearity, VIF analysis was performed. As shown in Table 8, all VIF values were below the conservative threshold of 5, indicating that multicollinearity was not a major source of bias in the model. In this model only LAIR-1 MFI remained an independent predictor of HCC (OR per unit increase = 1.484; 95%CI: 1.043-2.111; P = 0.028) while hsa-miR-21-5p, hsa-miR-155-5p, and AFP were not statistically significant (Table 8). The Hosmer-Lemeshow goodness-of-fit test confirmed excellent model calibration (χ² = 6.24, P = 0.620). The combined model demonstrated exceptional discrimination with an apparent AUC of 0.998. Bootstrap internal validation (500 replicates) confirmed robust performance (corrected 95%CI: 0.994-1.000). Decision curve analysis across a range of clinical probability thresholds (5%-30%) indicated that the combined model provided a superior net benefit compared with strategies of referring all or no patients with cirrhosis for HCC diagnostics.
| Predictor | Odds ratio | 95%CI | P value | Variance inflation factor |
| hsa-miR-21-5p (per unit) | 1.013 | 0.831-1.235 | 0.899 | 4.8 |
| hsa-miR-155-5p (per unit) | 0.911 | 0.463-1.795 | 0.789 | 1.5 |
| LAIR-1 MFI (per unit) | 1.484 | 1.043-2.111 | 0.028 | 3.1 |
| AFP (per ng/mL) | 1.053 | 0.946-1.173 | 0.345 | 2.9 |
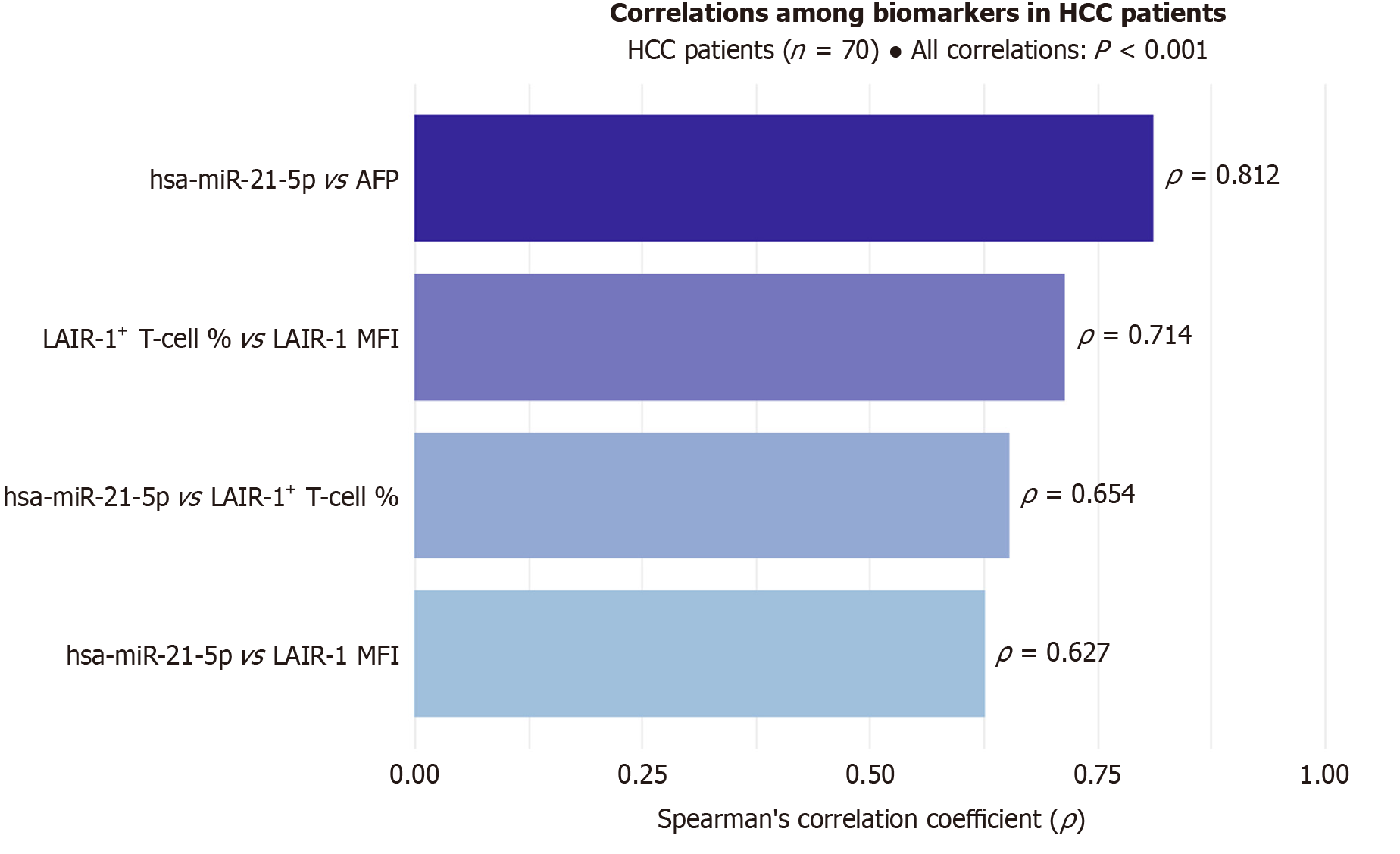
Spearman correlation analysis within the HCC cohort revealed significant associations between the candidate biomarkers and indicators of disease severity (Table 9). A very strong positive correlation was observed between plasma hsa-miR-21-5p and serum AFP (ρ = 0.812, P < 0.001). Furthermore, hsa-miR-21-5p showed strong positive correlations with both measures of LAIR-1 expression (ρ = 0.654 with LAIR-1+ TC % and ρ = 0.627 with LAIR-1 MFI; P < 0.001 for both). Consistent with their functional link, the two LAIR-1 parameters were also strongly correlated with each other (ρ = 0.714, P < 0.001). Clinically, higher plasma levels of hsa-miR-21-5p and LAIR-1 expression were significantly associated with the presence of vascular invasion and distant metastasis (P < 0.01 for all associations), suggesting their potential role not only in diagnosis but also as indicators of aggressive tumor biology (Figure 3).
| Correlation pair | Spearman’s ρ | P value |
| hsa-miR-21-5p vs AFP | 0.812 | < 0.001 |
| hsa-miR-21-5p vs LAIR-1+ T cell % | 0.654 | < 0.001 |
| hsa-miR-21-5p vs LAIR-1 MFI | 0.627 | < 0.001 |
| LAIR-1+ T cell % vs LAIR-1 MFI | 0.714 | < 0.001 |
To evaluate the clinical utility of the leading biomarkers in the most challenging and relevant scenarios, we conducted subgroup analyses focusing on early-stage HCC and AFP-negative tumors (Table 10). For the discrimination of early-stage HCC (solitary lesion < 1.9 cm; n = 33) from cirrhosis, both hsa-miR-21-5p and LAIR-1 MFI maintained exceptional discriminative ability with AUCs of 0.981 (95%CI: 0.949-1.000) and 0.992 (95%CI: 0.975-1.000), respectively. The sensitivity of hsa-miR-21-5p was 96.9%, and LAIR-1 MFI again achieved 100% sensitivity in this early-stage cohort. Furthermore, we analyzed the 22 patients with HCC with AFP-negative disease (AFP < 20 ng/mL). In this diagnostically difficult subgroup in which the standard biomarker fails, hsa-miR-21-5p correctly identified 21 of 22 cases (95.5% sensitivity), and LAIR-1 MFI identified all 22 (100% sensitivity). The performance of both biomarkers was not significantly different from their performance in the overall HCC cohort (DeLong test, P > 0.05 for both).
| Biomarker | Subgroup | AUC (95%CI) | Sensitivity (%) | Specificity (%) |
| hsa-miR-21-5p | Early-stage HCC (n = 33) vs cirrhosis | 0.981 (0.949-1.000) | 96.9 | 96.7 |
| LAIR-1 MFI | Early-stage HCC (n = 33) vs cirrhosis | 0.992 (0.975-1.000) | 100.0 | 98.3 |
| hsa-miR-21-5p | AFP-negative HCC (n = 22) vs cirrhosis | 0.986 (0.961-1.000) | 95.5 | 96.7 |
| LAIR-1 MFI | AFP-negative HCC (n = 22) vs cirrhosis | 0.998 (0.990-1.000) | 100.0 | 98.3 |
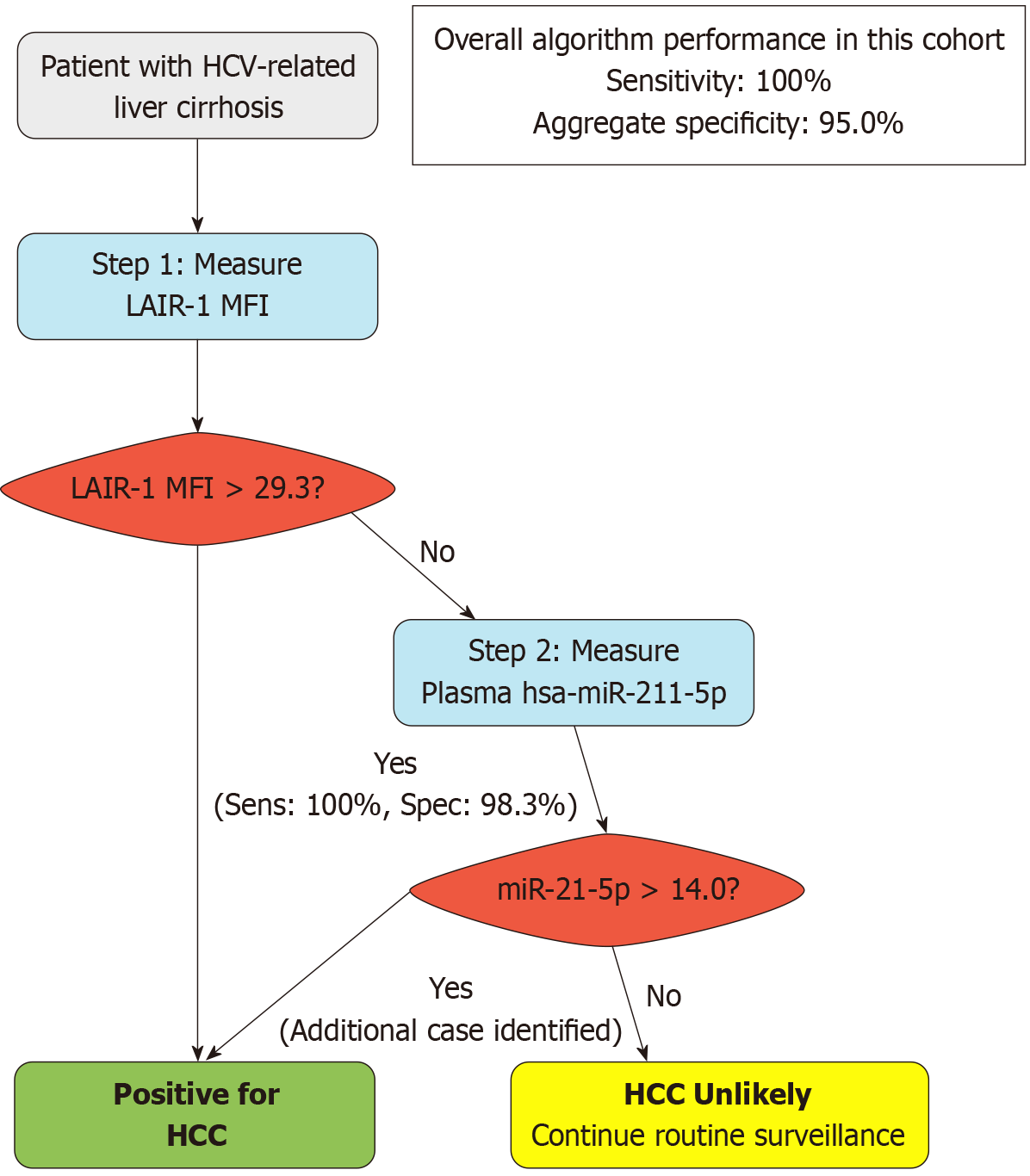
Based on the complementary diagnostic strengths of the leading biomarkers, we proposed a sequential algorithm for clinical decision-making. Leveraging the perfect sensitivity (100%) of LAIR-1 MFI, we modeled a two-step pathway in which patients with cirrhosis were first tested for LAIR-1 MFI with positive results (MFI > 29.3) triggering immediate diagnostic imaging for HCC. For the minority of patients testing negative in the first step, a subsequent plasma hsa-miR-21-5p test would be performed. In our cohort this integrated strategy would have successfully identified all 70 patients with HCC, yielding a perfect aggregate sensitivity of 100% while maintaining a high aggregate specificity of 95.0% for distinguishing HCC from cirrhosis. This proposed workflow synthesizes the robust immune signal of LAIR-1 with the potent molecular signal of hsa-miR-21-5p into a practical and highly accurate diagnostic pathway.
The paradigm of liquid biopsy for cancer detection is rapidly evolving from a singular focus on tumor-derived analytes toward a more holistic integration of tumor-intrinsic and host-immune signals. Early, accurate, and noninvasive detection of HCC remains a cornerstone for improving survival, particularly in regions such as Egypt where hepatitis C-related cirrhosis drives high disease burden. Conventional tools such as ultrasound and serum AFP lack the necessary sensitivity and specificity for small or AFP-negative tumors, underscoring the pressing need for reliable noninvasive molecular biomarkers that can complement imaging and identify malignant transformation at its inception[3]. In this context circulating miRNAs and immune-checkpoint-related molecules represent promising candidates because they can be repeatedly measured, reflect tumor biology and host immune modulation, and can be integrated into population-level surveillance strategies.
Our study, therefore, aimed to perform a head-to-head comparison of a quintessential tumor-derived oncomiR, plasma hsa-miR-21-5p, and a functional readout of host immune dysregulation, LAIR-1 MFI expression, for distinguishing HCC from HCV-related cirrhosis in an Egyptian cohort. This approach allowed us to evaluate the orthogonal diagnostic contributions of molecular and immune biomarker classes. We found that both hsa-miR-21-5p and LAIR-1 MFI exhibited exceptional diagnostic accuracy (AUC = 0.990 and 0.997, respectively), far surpassing hsa-miR-155-5p. Importantly, their diagnostic performance remained outstanding in early-stage and AFP-negative HCC subgroups. These findings bridge tumor-intrinsic molecular signaling and immune-phenotypic modulation, providing a novel, dual-parameter diagnostic framework within a well-characterized Egyptian cohort in which such integrated analyses are scarce[23-25].
We acknowledge that the exceptional diagnostic accuracy (AUC > 0.99) observed for our leading biomarkers must be interpreted in the context of our study cohort. The inclusion of patients with advanced-stage HCC (BCLC B/C), who demonstrated significantly higher levels of both biomarkers (Table 3), likely contributed to these near-perfect discrimination metrics. This spectrum bias may lead to an overestimation of performance relative to a pure surveillance population. However, the critical finding is that both biomarkers retained outstanding accuracy (AUC > 0.98) in the predefined subgroup of patients with early-stage HCC vs patients with cirrhosis (Table 10). This demonstrates that while advanced disease elevates biomarker levels, a powerful and diagnostically useful signal is already present in early, potentially curable tumors.
Our analysis revealed a striking, stepwise elevation in plasma hsa-miR-21-5p levels from healthy controls to patients with cirrhosis and most markedly to those with HCC (Table 4 and Figure 1). This biomarker demonstrated near-perfect discrimination between HCC and cirrhosis (AUC = 0.990) with a sensitivity of 98.6% and specificity of 96.7% (Table 6 and Figure 2). Its diagnostic power remained robust in critical subgroup analyses, accurately identifying both early-stage (AUC = 0.981) and AFP-negative HCC (95.5% sensitivity) (Table 10).
The exceptional performance of hsa-miR-21-5p is firmly grounded in its well-characterized role as a potent oncomiR. It directly represses key tumor suppressors like PTEN and PDCD4, thereby activating prosurvival and proliferative pathways such as PI3K/AKT[5,6]. Beyond these established targets, miR-21-5p has been shown to suppress hepatocyte nuclear factor 4α, a master regulator of hepatocyte differentiation and a critical tumor suppressor in HCC[25]. The loss of hepatocyte nuclear factor 4α promotes dedifferentiation and is a key event in the initiation and progression of hepatocarcinogenesis, providing a direct mechanistic link between elevated miR-21-5p and the development of HCC. Its significant upregulation in our cohort aligns with prior Egyptian studies reporting elevated miR-21 in HCV-related HCC[9,10], reinforcing its biological plausibility and regional relevance. The very strong positive correlation we observed with serum AFP (ρ = 0.812, P < 0.001; Table 9) suggests that miR-21-5p levels reflect tumor burden. However, its paramount clinical value lies in its ability to detect AFP-negative tumors with high sensitivity, effectively compensating for a major weakness of the current standard. This positions hsa-miR-21-5p as a powerful noninvasive molecular companion biomarker capable of uncovering biologically aggressive but serologically elusive HCC. While our findings are robust, it is noteworthy that the diagnostic accuracy (AUC = 0.990) in our homogeneous cohort exceeds the pooled AUC of about 0.86 reported in broader meta-analyses[7]. This may be attributed to our focus on a high-risk HCV population and the direct, clinically relevant comparison to controls with cirrhosis rather than healthy individuals, who present a more challenging diagnostic benchmark.
It is also noteworthy that miR-21 functions at the intersection of inflammation, fibrosis, and oncogenesis. Its induction by NF-κB and STAT3 signaling during chronic hepatitis enhances hepatic stellate cell activation, promoting fibrogenesis that sets the stage for malignant transformation. The reciprocal activation of immune checkpoints such as LAIR-1 may thus represent a downstream consequence of this chronic inflammatory milieu, reinforcing immune tolerance and tumor escape[26].
Mirroring the molecular findings, T cell expression of the immune checkpoint LAIR-1 was significantly dysregulated in HCC. Both the LAIR-1+ TC % and the LAIR-1 MFI showed a progressive increase from controls to the HCC group (Table 5; Figure 1). A critical insight from our analysis is that LAIR-1 MFI (AUC = 0.997) demonstrated far superior diagnostic power compared with the mere percentage of positive cells (AUC = 0.833) (Table 6). This result proposed that quantitative receptor density rather than qualitative cellular prevalence was the more powerful metric for assessing immune checkpoint activity in cancer diagnostics. In a multivariable model adjusting for all key biomarkers, LAIR-1 MFI emerged as a significant independent predictor of HCC. Although the VIF analysis confirmed that severe multicollinearity was not present (all VIF < 5, Table 8), the strong bivariate correlations observed between hsa-miR-21-5p, AFP, and LAIR-1 parameters (Table 9) suggest that these biomarkers capture overlapping aspects of the underlying biology, namely a synergy between high tumor burden and an immunosuppressive phenotype. Therefore, the independence of LAIR-1 MFI in the statistical model likely reflects its role as a highly sensitive integrator of this shared pathophysiology rather than representing a completely unrelated biological process, and its expression was associated with vascular invasion and metastasis (Table 11).
| Biomarker | Diagnostic performance (AUC) | Key strength | Role in multivariable model | Correlation with disease severity |
| hsa-miR-21-5p | 0.990 (exceptional) | Very high sensitivity (98.6%) and specificity (96.7%) | Not an independent predictor (P = 0.899) | Strong correlation with AFP (ρ = 0.812); associated with metastasis |
| hsa-miR-155-5p | 0.874 (good) | Good specificity (93.3%) | Not an independent predictor (P = 0.789) | Not assessed in this context |
| LAIR-1+ T cell % | 0.833 (good) | Perfect specificity (100%) as a rule-in test | Not tested in the primary model | Strong correlation with LAIR-1 MFI (ρ = 0.714) |
| LAIR-1 MFI | 0.997 (exceptional) | Perfect sensitivity (100%) and high specificity (98.3%) | Sole independent predictor (OR = 1.48, P = 0.028) | Associated with vascular invasion and metastasis |
| Combined model | 0.998 (near-perfect) | Superior net benefit for clinical decision-making | N/A | N/A |
The superior performance of MFI over cell frequency is biologically profound. While the percentage of LAIR-1+ cells indicates the size of a potentially immunosuppressed T cell pool, the MFI reflects the receptor density per cell, which is a more direct measure of the intensity of the inhibitory signal and the degree of T cell dysfunction or exhaustion[27]. This finding suggests that the functional state of the immune system, quantified at the level of receptor density, is a more sensitive indicator of malignant transformation than a simple cellular count. Our results provide compelling clinical corroboration for experimental studies linking LAIR-1 signaling to oncogenic pathways like PI3K-AKT-mTOR in HCC cells[12,13]. Furthermore, a recent study by Pan et al[27] demonstrated that LAIR-1 expression in HCC cells promoted immune evasion via a GSK-3β/β-catenin/MYC/PD-L1 axis. Our data on peripheral T cells complement this, suggesting a systemic immune landscape where high LAIR-1 expression on T cells facilitates this tumor immune escape, leading to the observed metastatic phenotype. Our work builds upon the foundational report by Hammad et al[14] in an Egyptian cohort by introducing and validating the quantitative dimension of MFI, establishing it as a more discriminative parameter and a statistically independent predictor in a combined diagnostic model.
Our formal comparison established a clear diagnostic hierarchy: LAIR-1 MFI and miR-21-5p demonstrated equivalent, outstanding performance (AUCs > 0.99), both significantly outperforming hsa-miR-155-5p (AUC = 0.874) and LAIR-1+ T cell % (AUC = 0.833) (Tables 6 and 7). The most critical insight came from the multivariable model (Table 8), which provided the statistical foundation for integration by revealing that LAIR-1 MFI captures unique variance not explained by the other biomarkers. Specifically, LAIR-1 MFI emerged as the sole independent predictor of HCC while the powerful oncomiR miR-21-5p did not. This indicates that the systemic immune dysregulation captured by LAIR-1 MFI provides a diagnostic signal that is orthogonal and non-redundant to the tumor-derived molecular signal of miR-21-5p. The correlation matrix (Table 9 and Figure 3) further revealed interconnected biology, showing the strong correlation between miR-21, AFP, and LAIR-1 parameters suggests a synergy between tumor burden and immune suppression. Capitalizing on these complementary strengths, we proposed a sequential diagnostic algorithm (Figure 4) that achieves a theoretical aggregate sensitivity of 100% and specificity of 95%.
The underperformance of hsa-miR-155-5p, another recognized oncomiR, warrants scientific explanation. While miR-155 is involved in oncogenesis, its expression is also highly activated in general inflammatory processes, and recent reviews have detailed the broader miRNA landscape in HCC, including its metabolic and therapeutic implications[28,29]. In a cohort with underlying HCV-related cirrhosis, a state of chronic inflammation, the signal of miR-155 derived from the tumor may be confounded by the noise of its expression from activated immune cells, thereby reducing its specificity for cancer detection. The equivalent performance and statistical independence of miR-21-5p and LAIR-1 MFI are highly significant. They represent two fundamental hallmarks of cancer: Sustaining proliferative signaling (miR-21); and avoiding immune destruction (LAIR-1)[30]. Our study provides clinical evidence that these hallmarks are co-expressed and detectable in circulation. This multimodal approach aligns with the cutting-edge liquid biopsy paradigm in oncology, seeking to combine multiple analyte classes (e.g., circulating tumor DNA, proteins, cells) for a more comprehensive picture[31]. Our model is a novel iteration for HCC, effectively combining a tumor-derived molecular signal with a host immune signal to maximize diagnostic power.
The ultimate test for any new biomarker is its performance in real-world clinical scenarios. Our cohort included a range of disease stages, including a significant proportion of early-stage (47.1%, Table 2) and AFP-negative (31.4%) HCC cases. It was in these subgroups that our leading biomarkers proved most compelling, maintaining outstanding accuracy (Table 10). The ability of both markers to identify AFP-negative HCC with near-perfect sensitivity is not just a statistical result but a potential solution to a persistent clinical dilemma, directly addressing a key limitation outlined in current management guidelines[17].
The robust performance in early-stage disease is biologically plausible. Dysregulation of miR-21 is recognized as an early event in hepatocarcinogenesis, and the process of immune editing and the establishment of an immunosuppressive microenvironment begin early in tumor development[32]. Therefore, it is mechanistically sound that these signals are detectable in the periphery before a tumor reaches a size easily identifiable by conventional imaging. The proposed se
This comprehensive analysis provides a head-to-head comparison of circulating miRNAs and an immune checkpoint marker for HCC diagnosis in a high-risk Egyptian cohort. Our findings can be summarized in three key points. First, both plasma hsa-miR-21-5p and LAIR-1 MFI demonstrated exceptional, and statistically equivalent, diagnostic accuracy for distinguishing HCC from cirrhosis (AUCs of 0.990 and 0.997, respectively), significantly outperforming hsa-miR-155-5p. Second, in a multivariable model adjusting for all key biomarkers, only LAIR-1 MFI remained an independent predictor of HCC, suggesting it captures unique variance in the malignant transformation process. Third, the strong positive correlations between hsa-miR-21-5p, AFP, and LAIR-1 expression, alongside their association with vascular invasion and metastasis, indicate that these biomarkers are linked to both tumor burden and an immunosuppressive phenotype. Collectively, these results nominate a dual-parameter approach combining the molecular signal of hsa-miR-21-5p with the immune signal of LAIR-1 MFI as a highly promising strategy for non-invasive HCC surveillance.
Building on this summary, our data delineate a coherent biological narrative of hepatocarcinogenesis in the Egyptian context: The malignant transformation of cirrhotic liver tissue drives concurrent and correlated alterations in both the molecular and immune compartments, detectable peripherally. The progressive increase of hsa-miR-21-5p and LAIR-1 MFI from control through cirrhosis to HCC (Tables 4 and 5; Figure 1) illustrates the co-evolution of tumor-driven signa
These findings also resonate with the emerging systems-level understanding of HCC as a disease of disrupted tumor-immune-microenvironmental crosstalk. Integrating miRNA profiles with immune checkpoint phenotyping echoes the multi-omic strategies of modern liquid biopsy frameworks, which aim to capture both tumor-derived and host-response signals in a single blood draw. This dual-axis model could therefore form the foundation of a more holistic, precision-based surveillance paradigm. This concept aligns with emerging multi-omic frameworks integrating transcriptomic, epigenetic, and immune-profiling data for HCC risk stratification and early detection[33-35].
The excellent calibration of the logistic model (Hosmer-Lemeshow P > 0.05) and minimal optimism in bootstrap validation support the internal validity of these findings, yet generalizability must be confirmed in geographically and etiologically diverse populations.
This work provides several novel contributions. First, it is among the first to perform a direct, within-cohort head-to-head diagnostic comparison of circulating hsa-miR-21-5p, hsa-miR-155-5p, and peripheral T cell LAIR-1 (both percent and MFI) in an Egyptian HCV-related population using rigorous assay protocols and bootstrap-validated ROC analyses. Second, by showing equivalent, near-perfect performance of an immune MFI readout and a circulating miRNA and by proposing a practical sequential algorithm, this study bridges molecular liquid biopsy and immune phenotyping approaches while providing an immediately testable diagnostic pathway for surveillance programs in Egypt and similar high-burden settings. Finally, the demonstration that both markers retain high accuracy in early and AFP-negative HCC highlights their potential to address a pressing clinical need that standard surveillance (ultrasound ± AFP) does not fully meet. These points add complementary evidence to recent Egyptian and international reports that emphasize the diagnostic promise of miR-21 and the role of LAIR-1 in HCC biology.
Clinically, the results nominate two pragmatic paths. First, miR-21 quantification, using standardized plasma collection, SNORD68 normalization, and RT-qPCR as implemented here, could be evaluated as a reflex test in cirrhosis surveillance for patients with indeterminate imaging or low AFP. Second, LAIR-1 MFI measurement if reproducibly validated on harmonized cytometry platforms could function as a high-sensitivity triage test to prioritize diagnostic imaging. The complementary sequential algorithm we modeled would be particularly attractive in resource-constrained settings where imaging capacity is limited. A blood-based triage could concentrate imaging resources on the highest-probability cases while preserving case-capture for early, treatable lesions.
For research our findings support prioritizing: (1) External validation cohorts (multicenter, population-based); (2) Prospective longitudinal surveillance studies to assess lead-time gain and effects on stage at diagnosis and outcomes; and (3) Mechanistic work to clarify whether LAIR-1 upregulation is a driver or a response to tumor-derived signals and how it interacts with miR-21-mediated pathways (PTEN, PDCD4, PI3K/AKT). Integration with other liquid-biopsy modalities (cell free DNA methylation, protein panels) and cost-effectiveness modelling will also be necessary to build an imple
From a translational perspective both assays used in this study, RT-qPCR quantification of plasma miR-21-5p and flow-cytometric measurement of T cell LAIR-1 MFI, are technically feasible in most tertiary laboratories. Implementation in surveillance programs would require standardization of preanalytical procedures, inter-laboratory calibration, and cost-effectiveness evaluation in population-level studies.
We acknowledge several limitations. First, the case-control design, drawn from tertiary referral centers, carries the risk of spectrum bias. Our HCC group included a substantial fraction of advanced cases that may inflate measured accuracy relative to a general surveillance population. Second, although internal bootstrap validation was performed, there was no external validation cohort; therefore, over-optimism cannot be excluded, and replication in independent Egyptian and multiethnic cohorts is mandatory. Third, while we used SNORD68 as an endogenous control and adhered to recom
This study demonstrated that a dual-axis liquid biopsy approach, integrating a tumor-intrinsic molecular marker (plasma hsa-miR-21-5p) with a host-immune functional marker (T cell LAIR-1 MFI) achieved exceptional diagnostic performance for discriminating HCC from HCV-related cirrhosis. Our findings challenged the siloed evaluation of biomarkers by revealing that the malignant state is most accurately captured at the intersection of oncogenic drive and systemic immune dysfunction. The superiority of LAIR-1 MFI over cellular prevalence sets a new methodological standard for immune phenotyping in this context. The proposed sequential algorithm synthesized these signals into a practical and highly accurate diagnostic pathway with significant potential for surveillance programs in Egypt and similar high-burden regions. External multicenter validation, assay harmonization, and longitudinal outcome studies are now warranted to confirm the clinical utility of this integrated paradigm and establish its place in future precision-hepatology workflows.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69513] [Article Influence: 13902.6] [Reference Citation Analysis (40)] |
| 2. | Rizzo A, Cusmai A, Gadaleta-Caldarola G, Palmiotti G. Which role for predictors of response to immune checkpoint inhibitors in hepatocellular carcinoma? Expert Rev Gastroenterol Hepatol. 2022;16:333-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 72] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 3. | Tzartzeva K, Obi J, Rich NE, Parikh ND, Marrero JA, Yopp A, Waljee AK, Singal AG. Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients With Cirrhosis: A Meta-analysis. Gastroenterology. 2018;154:1706-1718.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 975] [Cited by in RCA: 925] [Article Influence: 115.6] [Reference Citation Analysis (2)] |
| 4. | Ibrahim AS, Khaled HM, Mikhail NN, Baraka H, Kamel H. Cancer incidence in egypt: results of the national population-based cancer registry program. J Cancer Epidemiol. 2014;2014:437971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 443] [Cited by in RCA: 388] [Article Influence: 32.3] [Reference Citation Analysis (1)] |
| 5. | Meng F, Henson R, Wehbe-Janek H, Ghoshal K, Jacob ST, Patel T. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology. 2007;133:647-658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2258] [Cited by in RCA: 2172] [Article Influence: 114.3] [Reference Citation Analysis (0)] |
| 6. | Zhou T, Liu L, Lan H, Fang D. Effects of LAIR-1 on hepatocellular carcinoma cell proliferation and invasion via PI3K-AKT-mTOR pathway regulation. Immun Inflamm Dis. 2023;11:e982. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 7. | Qu J, Yang J, Chen M, Cui L, Wang T, Gao W, Tian J, Wei R. MicroRNA-21 as a diagnostic marker for hepatocellular carcinoma: A systematic review and meta-analysis. Pak J Med Sci. 2019;35:1466-1471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (2)] |
| 8. | Alemayehu E, Belete MA, Walle M, Getu F, Mulatie Z, Teshome M, Anley DT, Weldehanna DG, Gedefie A, Ebrahim H. Diagnostic accuracy of circulating miRNAs to discriminate hepatocellular carcinoma from liver cirrhosis: a systematic review and meta-analysis. Front Med (Lausanne). 2024;11:1359414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 9. | Eldosoky MA, Hammad R, Elmadbouly AA, Aglan RB, Abdel-Hamid SG, Alboraie M, Hassan DA, Shaheen MA, Rushdi A, Ahmed RM, Abdelbadea A, Abdelmageed NA, Elshafei A, Ali E, Abo-Elkheir OI, Zaky S, Hamdy NM, Lambert C. Diagnostic Significance of hsa-miR-21-5p, hsa-miR-192-5p, hsa-miR-155-5p, hsa-miR-199a-5p Panel and Ratios in Hepatocellular Carcinoma on Top of Liver Cirrhosis in HCV-Infected Patients. Int J Mol Sci. 2023;24:3157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 10. | Elabd WK, Elbakry MMM, Hassany M, Baki AA, Seoudi DM, El Azeem EMA. Evaluation of miRNA-7, miRNA-10 and miRNA-21 as diagnostic non-invasive biomarkers of hepatocellular carcinoma. Clin Exp Hepatol. 2023;9:221-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (2)] |
| 11. | Meyaard L. The inhibitory collagen receptor LAIR-1 (CD305). J Leukoc Biol. 2008;83:799-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 229] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 12. | Cao Q, Fu A, Yang S, He X, Wang Y, Zhang X, Zhou J, Luan X, Yu W, Xue J. Leukocyte-associated immunoglobulin-like receptor-1 expressed in epithelial ovarian cancer cells and involved in cell proliferation and invasion. Biochem Biophys Res Commun. 2015;458:399-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 13. | Pan B, Ke X, Qiu J, Ye D, Zhang Z, Zhang X, Luo Y, Yao Y, Wu X, Wang X, Tang N. LAIR1-mediated resistance of hepatocellular carcinoma cells to T cells through a GSK-3β/β-catenin/MYC/PD-L1 pathway. Cell Signal. 2024;115:111039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 14. | Hammad R, Aglan RB, Mohammed SA, Awad EA, Elsaid MA, Bedair HM, Khirala SK, Selim MA, Abo Elqasem AA, Rushdi A, Ali M, Abo-Elkheir OI, Sanad EF, Hamdy NM. Cytotoxic T Cell Expression of Leukocyte-Associated Immunoglobulin-Like Receptor-1 (LAIR-1) in Viral Hepatitis C-Mediated Hepatocellular Carcinoma. Int J Mol Sci. 2022;23:12541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 57] [Reference Citation Analysis (1)] |
| 15. | von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12:1495-1499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8394] [Cited by in RCA: 7610] [Article Influence: 634.2] [Reference Citation Analysis (6)] |
| 16. | Hanley JA, McNeil BJ. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology. 1982;143:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16948] [Cited by in RCA: 12314] [Article Influence: 279.9] [Reference Citation Analysis (2)] |
| 17. | Singal AG, Llovet JM, Yarchoan M, Mehta N, Heimbach JK, Dawson LA, Jou JH, Kulik LM, Agopian VG, Marrero JA, Mendiratta-Lala M, Brown DB, Rilling WS, Goyal L, Wei AC, Taddei TH. AASLD Practice Guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology. 2023;78:1922-1965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1428] [Cited by in RCA: 1332] [Article Influence: 444.0] [Reference Citation Analysis (3)] |
| 18. | Kroh EM, Parkin RK, Mitchell PS, Tewari M. Analysis of circulating microRNA biomarkers in plasma and serum using quantitative reverse transcription-PCR (qRT-PCR). Methods. 2010;50:298-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 983] [Cited by in RCA: 925] [Article Influence: 57.8] [Reference Citation Analysis (0)] |
| 19. | Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158539] [Cited by in RCA: 138942] [Article Influence: 5557.7] [Reference Citation Analysis (4)] |
| 20. | Oldaker T, Bakke A. Quality Assurance and Validation in Clinical Flow Cytometry. In: Schmitz JL, Detrick B, O'Gorman MRG, editors. Manual of Molecular and Clinical Laboratory Immunology. United States: John Wiley & Sons, Inc, 2024. [DOI] [Full Text] |
| 21. | Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3:32-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 228] [Reference Citation Analysis (2)] |
| 22. | DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44:837-845. [PubMed] [DOI] [Full Text] |
| 23. | Mourad L, El-Ahwany E, Zoheiry M, Abu-Taleb H, Hassan M, Ouf A, Rahim AA, Hassanien M, Zada S. Expression analysis of liver-specific circulating microRNAs in HCV-induced hepatocellular Carcinoma in Egyptian patients. Cancer Biol Ther. 2018;19:400-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 24. | Rahdan F, Saberi A, Saraygord-Afshari N, Hadizadeh M, Fayeghi T, Ghanbari E, Dianat-Moghadam H, Alizadeh E. Deciphering the multifaceted role of microRNAs in hepatocellular carcinoma: Integrating literature review and bioinformatics analysis for therapeutic insights. Heliyon. 2024;10:e39489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 25. | Hatziapostolou M, Polytarchou C, Aggelidou E, Drakaki A, Poultsides GA, Jaeger SA, Ogata H, Karin M, Struhl K, Hadzopoulou-Cladaras M, Iliopoulos D. An HNF4α-miRNA inflammatory feedback circuit regulates hepatocellular oncogenesis. Cell. 2011;147:1233-1247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 391] [Article Influence: 26.1] [Reference Citation Analysis (1)] |
| 26. | Zhang T, Yang Z, Kusumanchi P, Han S, Liangpunsakul S. Critical Role of microRNA-21 in the Pathogenesis of Liver Diseases. Front Med (Lausanne). 2020;7:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 27. | Pan B, Shen S, Zhao J, Zhang Z, Ye D, Zhang X, Yao Y, Luo Y, Wang X, Tang N. LAIR1 promotes hepatocellular carcinoma cell metastasis and induces M2-macrophage infiltration through activating AKT-IKKβ-p65 axis. Mol Carcinog. 2024;63:1827-1841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 28. | Liapopoulos D, Sarantis P, Biniari T, Bousou TE, Trifylli EM, Anastasiou IA, Kokkali S, Korakaki D, Pantzios S, Koustas E, Elefsiniotis I, Karamouzis MV. MicroRNA Landscape in Hepatocellular Carcinoma: Metabolic Re-Wiring, Predictive and Diagnostic Biomarkers, and Emerging Therapeutic Targets. Biomedicines. 2025;13:2243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 29. | Seddiki N, Brezar V, Ruffin N, Lévy Y, Swaminathan S. Role of miR-155 in the regulation of lymphocyte immune function and disease. Immunology. 2014;142:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 145] [Article Influence: 12.1] [Reference Citation Analysis (1)] |
| 30. | Hanahan D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022;12:31-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7883] [Cited by in RCA: 6430] [Article Influence: 1607.5] [Reference Citation Analysis (4)] |
| 31. | Heitzer E, Haque IS, Roberts CES, Speicher MR. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat Rev Genet. 2019;20:71-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 969] [Cited by in RCA: 1058] [Article Influence: 151.1] [Reference Citation Analysis (4)] |
| 32. | Ringelhan M, Pfister D, O'Connor T, Pikarsky E, Heikenwalder M. The immunology of hepatocellular carcinoma. Nat Immunol. 2018;19:222-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 861] [Cited by in RCA: 804] [Article Influence: 100.5] [Reference Citation Analysis (2)] |
| 33. | Chen G, Zhang J, Fu Q, Taly V, Tan F. Integrative analysis of multi-omics data for liquid biopsy. Br J Cancer. 2023;128:505-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 34. | Galun D, Mijac D, Filipovic A, Bogdanovic A, Zivanovic M, Masulovic D. Precision Medicine for Hepatocellular Carcinoma: Clinical Perspective. J Pers Med. 2022;12:149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 35. | Llovet JM, Montal R, Sia D, Finn RS. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat Rev Clin Oncol. 2018;15:599-616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1582] [Cited by in RCA: 1479] [Article Influence: 184.9] [Reference Citation Analysis (1)] |