Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. Apr 15, 2026; 18(4): 116104
Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116104
Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116104
Figure 1 Flowchart of the clinical study design.
miR-206: MicroRNA-206; HIF-1α: Hypoxia-inducible factor-1α.
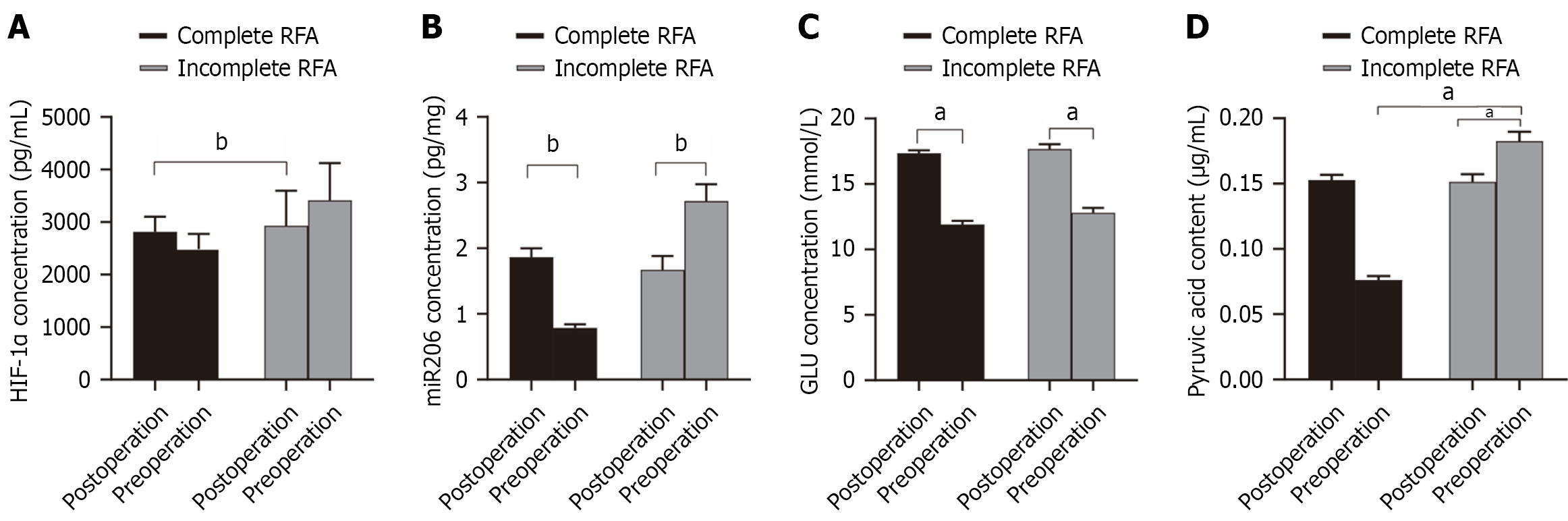
Figure 2 Changes in serum biomarker levels in patients undergoing complete or incomplete radiofrequency ablation.
A: Serum hypoxia-inducible factor-1α levels measured preoperatively and 1 week after radiofrequency ablation (RFA) in the complete ablation group (n = 35) and incomplete ablation group (n = 10); B: Serum microRNA-206 levels in the same patient cohorts (n = 35 vs n = 10); C: Changes in serum glucose levels before and after RFA (n = 45); D: Changes in serum pyruvate levels before and after RFA (n = 45). Data are presented as mean ± SD. Student’s t-test was used for comparisons. aP < 0.05; bP < 0.01. RFA: Radiofrequency ablation; miR-206: MicroRNA-206; HIF-1α: Hypoxia-inducible factor-1α.
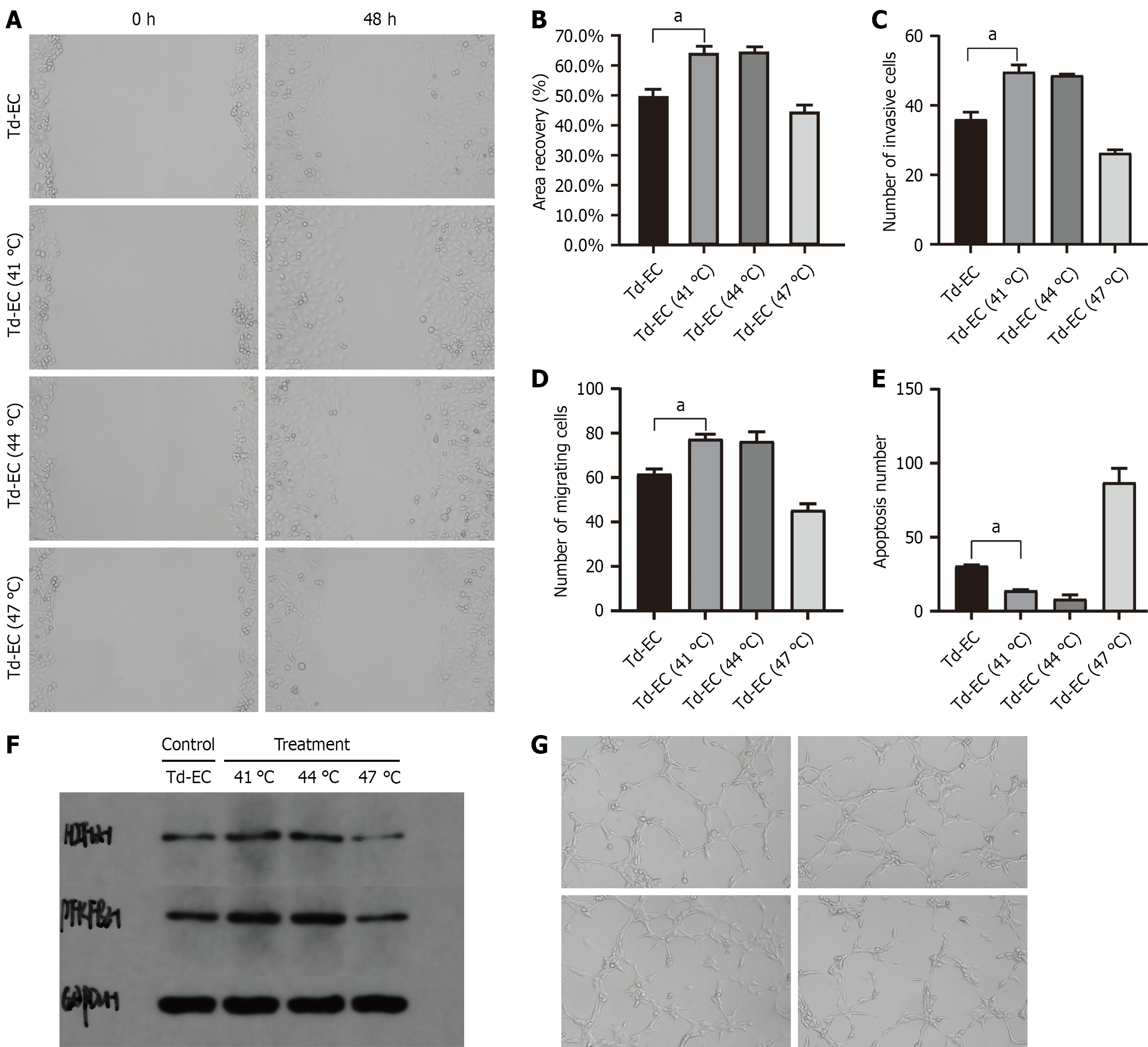
Figure 3 Functional changes in tumor-derived endothelial cells after thermal stimulation.
A: Wound-healing migration assay at 0 h and 48 h under 37 °C or 41 °C (n = 3); B: Quantification of wound-closure rate (n = 3); C: Transwell migration assay (n = 3); D: Transwell invasion assay (n = 3); E: Apoptosis measured by TUNEL staining at 37 °C, 41 °C, 44 °C, and 47 °C (n = 3); F: Western blot analysis of hypoxia-inducible factor-1α, 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3, and GAPDH under each temperature condition (n = 3); G: Tube-formation assay under 37 °C, 41 °C, 44 °C, and 47 °C (n = 3). Data are expressed as mean ± SD; one-way ANOVA was used for statistical analysis. aP < 0.05. Td-EC: Tumor-derived endothelial cells.
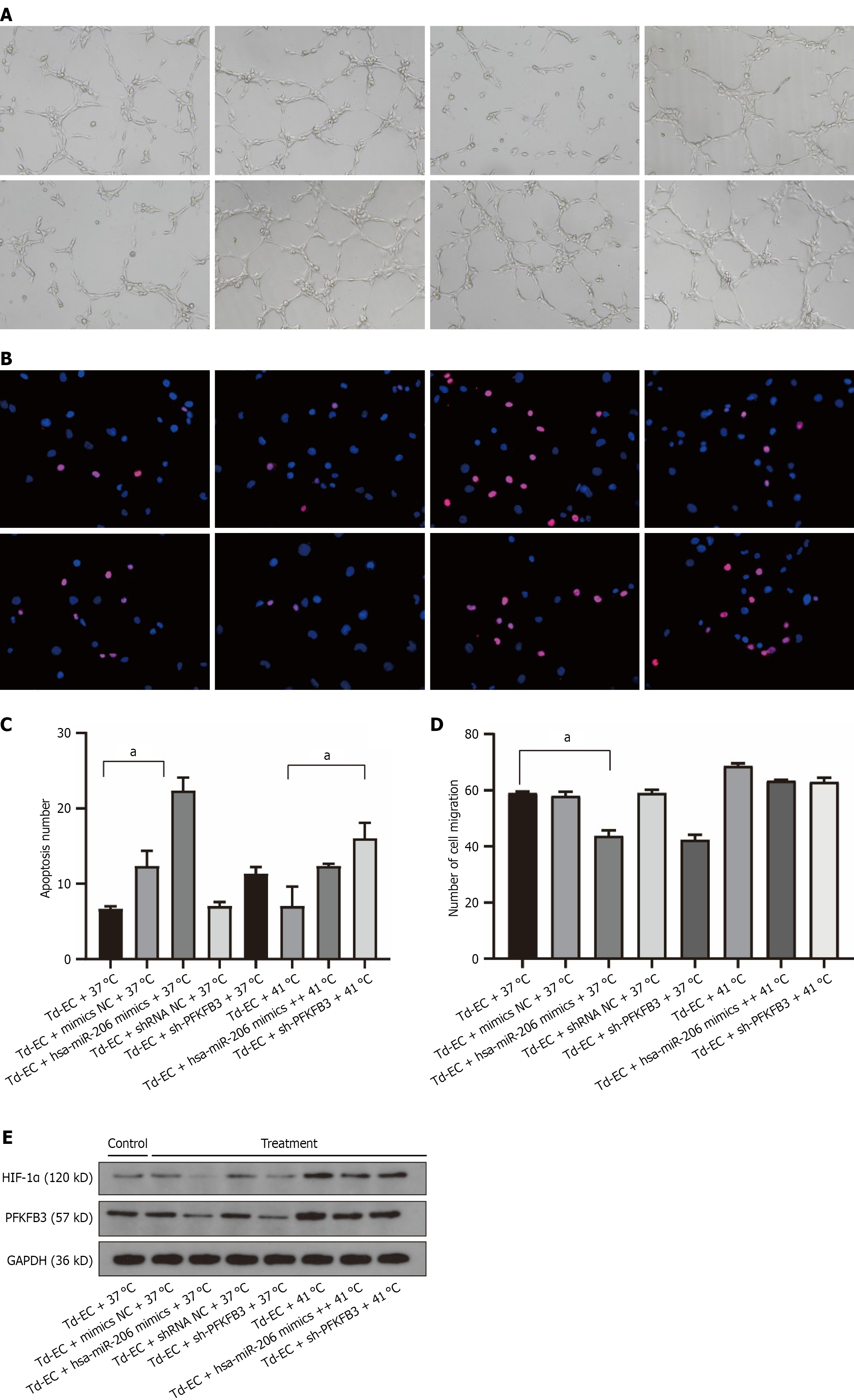
Figure 4 Effects of microRNA-206 overexpression and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 knockdown on endothelial function and glycolysis under thermal stimulation.
A: Tube-formation assay under 37 °C and 41 °C in control, mimic NC, microRNA-206 (miR-206) mimics, shRNA NC, and sh-6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3) groups (n = 3); B and C: TUNEL apoptosis staining and quantification under the same conditions (n = 3); D: Transwell migration assay results comparing miR-206 and sh-PFKFB3 effects (n = 3); E: Western blot analysis of hypoxia-inducible factor-1α, PFKFB3, and GAPDH under all conditions (n = 3). Data represent mean ± SD; one-way ANOVA was used. aP < 0.05. Td-EC: Tumor-derived endothelial cells; HIF-1α: Hypoxia-inducible factor-1α; PFKFB3: 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3.
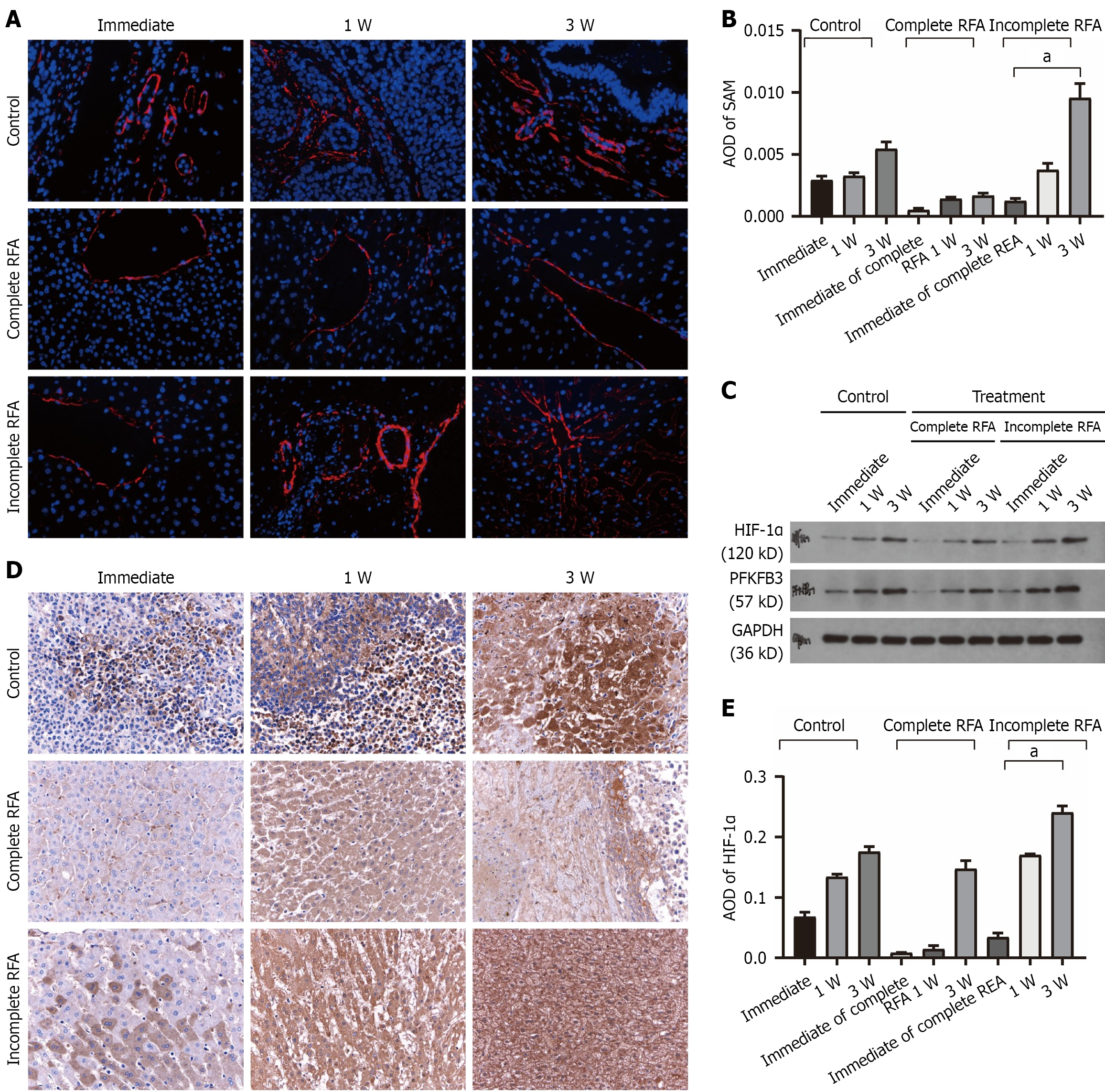
Figure 5 Angiogenesis, glycolytic signaling, and protein expression in rabbit VX2 liver tumors after complete or incomplete radio
- Citation: Lu D, Wang LJ, Chai J, Jiang J, Tang YL. MicroRNA-206 suppresses hypoxia-inducible factor-1α/PFKFB3-mediated glycolysis to inhibit recurrence and metastasis of hepatocellular carcinoma after incomplete radiofrequency ablation. World J Gastrointest Oncol 2026; 18(4): 116104
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116104.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116104