Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. Apr 15, 2026; 18(4): 113266
Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.113266
Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.113266
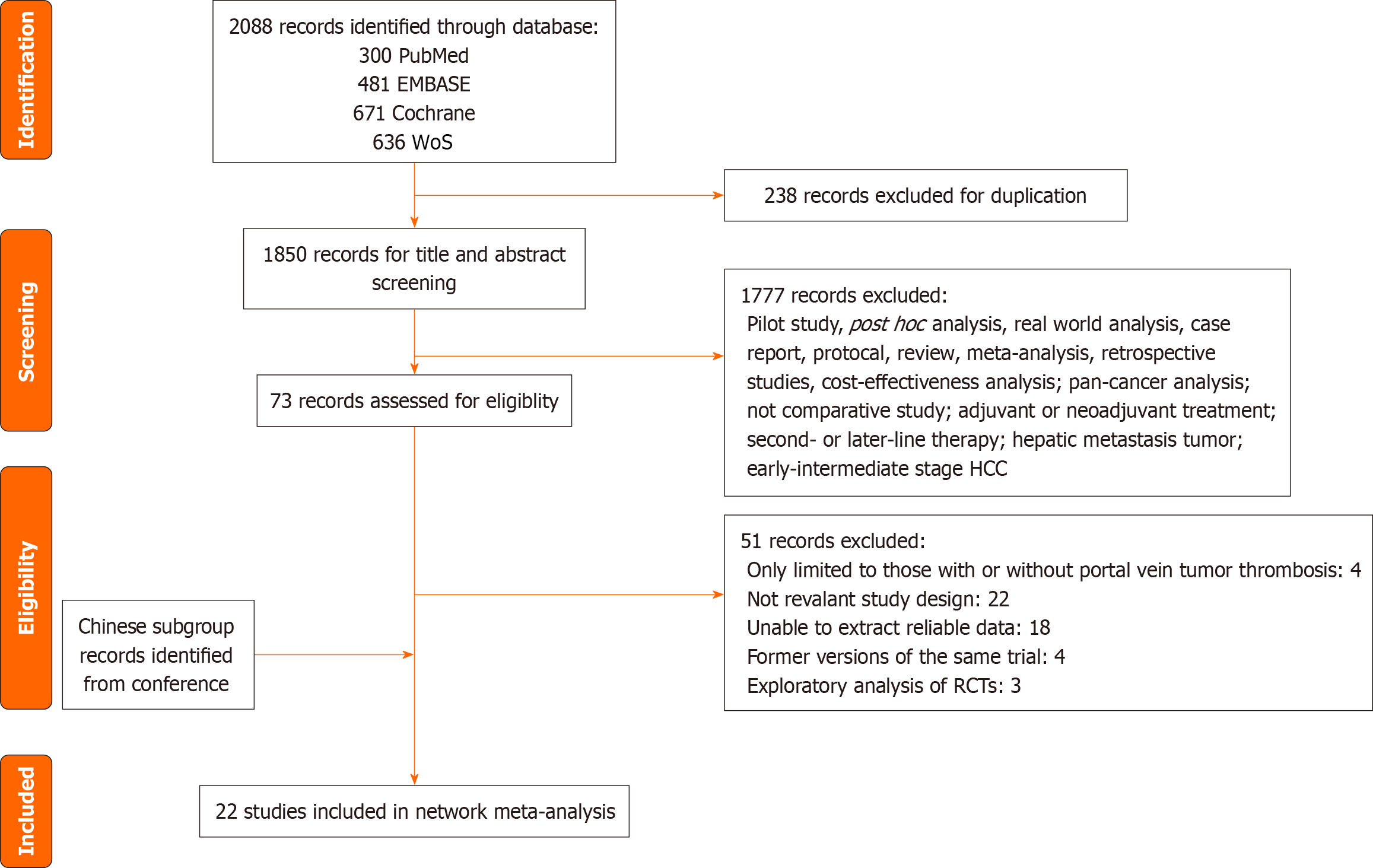
Figure 1 Flowchart of the literature search and selection process.
The REFLECT and LEAP-002 trials reported Chinese subgroup data at Chinese Society of Clinical Oncology 2017 and Asian Pacific Association for the Study of the Liver 2024, respectively. HCC: Hepatocellular carcinoma; RCT: Randomized controlled trial; WoS: Web of Science.
Figure 2 Partitioned survival model.
HCC: Hepatocellular carcinoma; TACE: Transarterial chemoembolization.
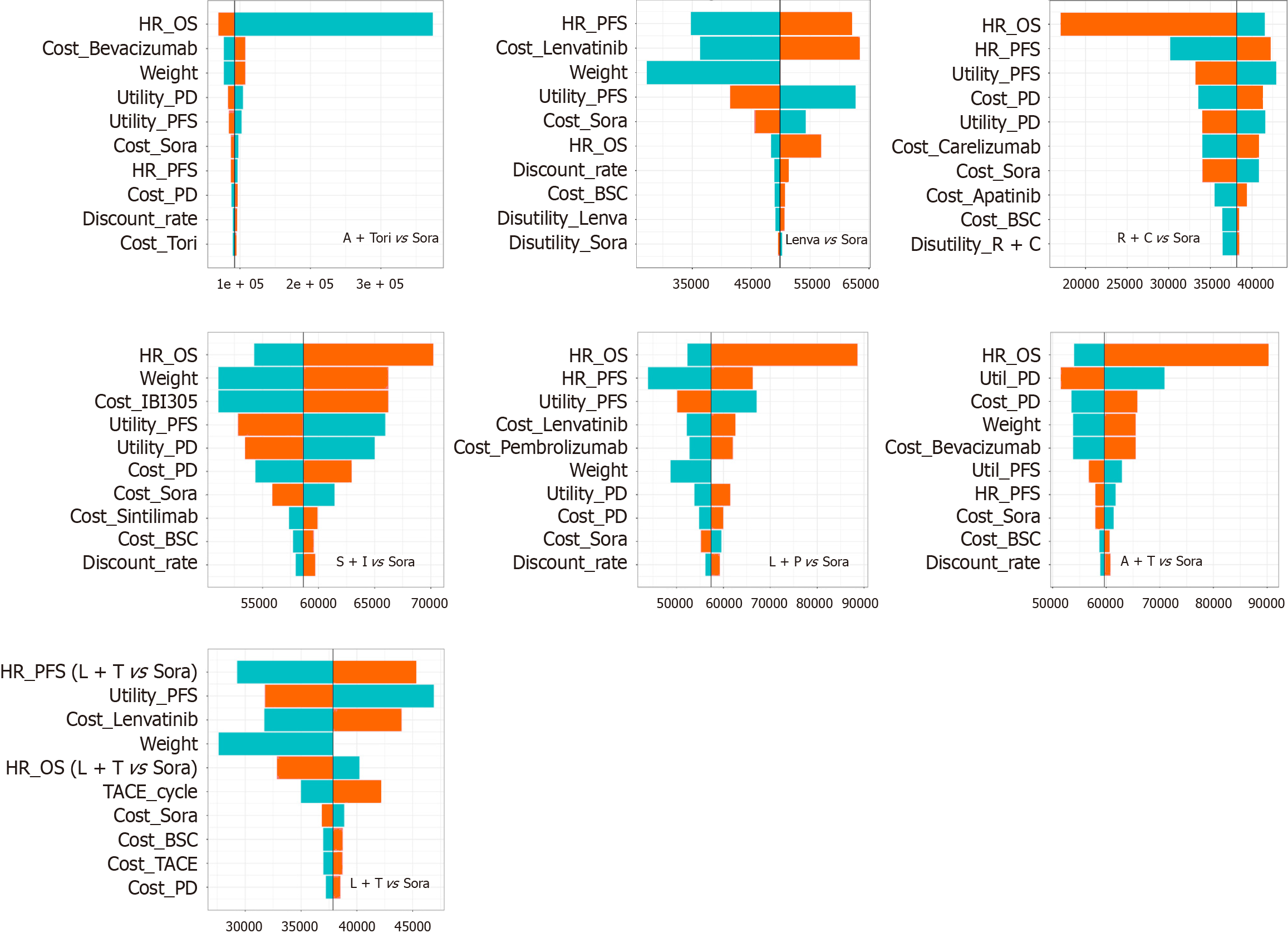
Figure 3 Tornado diagrams of one-way sensitivity analyses, using sorafenib as the comparator, the top ten variables were showed.
BSC: Best supportive care; HR: Hazard ratio; L + T: Lenvatinib plus transarterial chemoembolization; OS: Overall survival; PD: Progressive disease; PFS: Progression-free survival; R + C: Rivoceranib plus camrelizumab; Sora: Sorafenib; TACE: Transarterial chemoembolization; Tori: Toripalimab.
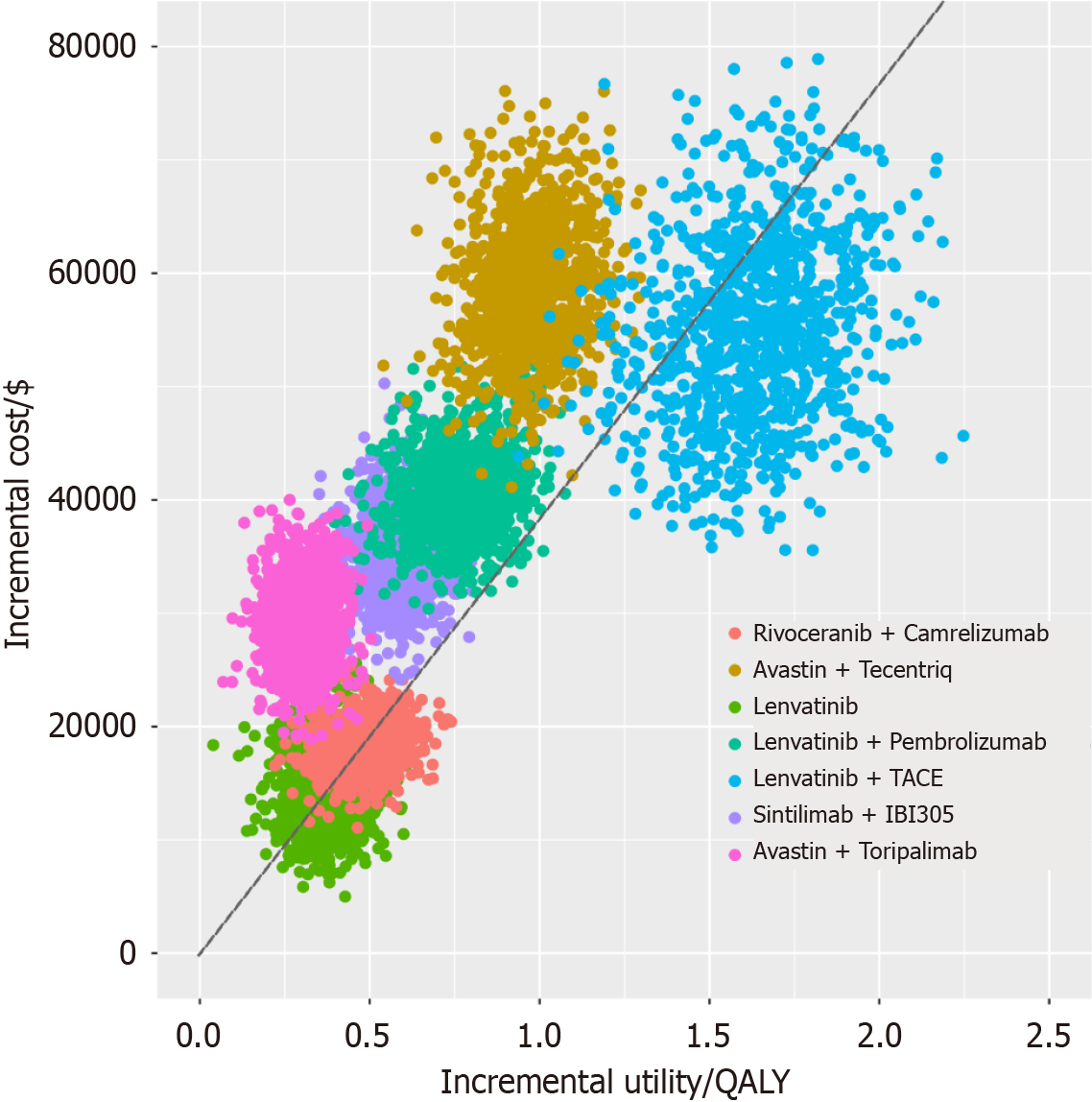
Figure 4 Probabilistic sensitivity analysis scatter plot for each treatment compared with sorafenib.
QALY: Quality-adjusted life year; TACE: Transarterial chemoembolization.
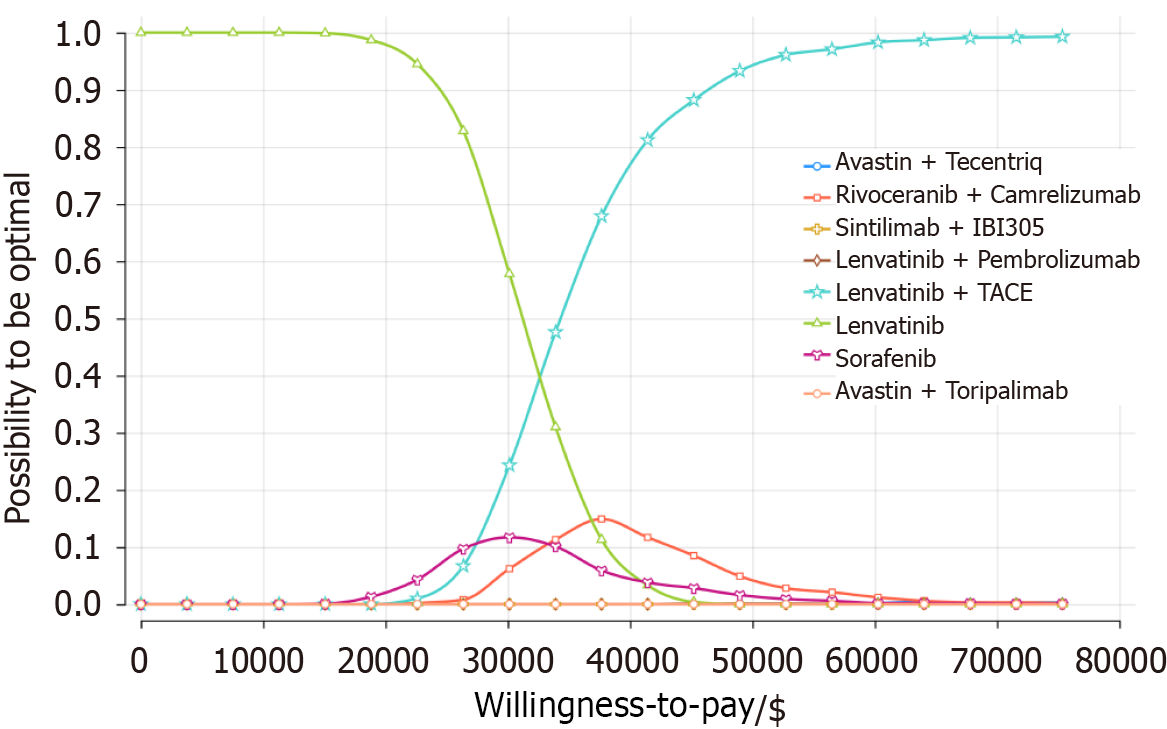
Figure 5 Cost-effectiveness acceptability curves under different willingness-to-pay thresholds.
TACE: Transarterial chemoembolization.
- Citation: Lin ZH, He Y, Xu HX, Xiao L. Cost-effectiveness analysis of seven treatments vs sorafenib as first-line therapy for advanced hepatocellular carcinoma in China. World J Gastrointest Oncol 2026; 18(4): 113266
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/113266.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.113266