Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. Mar 15, 2026; 18(3): 113757
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.113757
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.113757
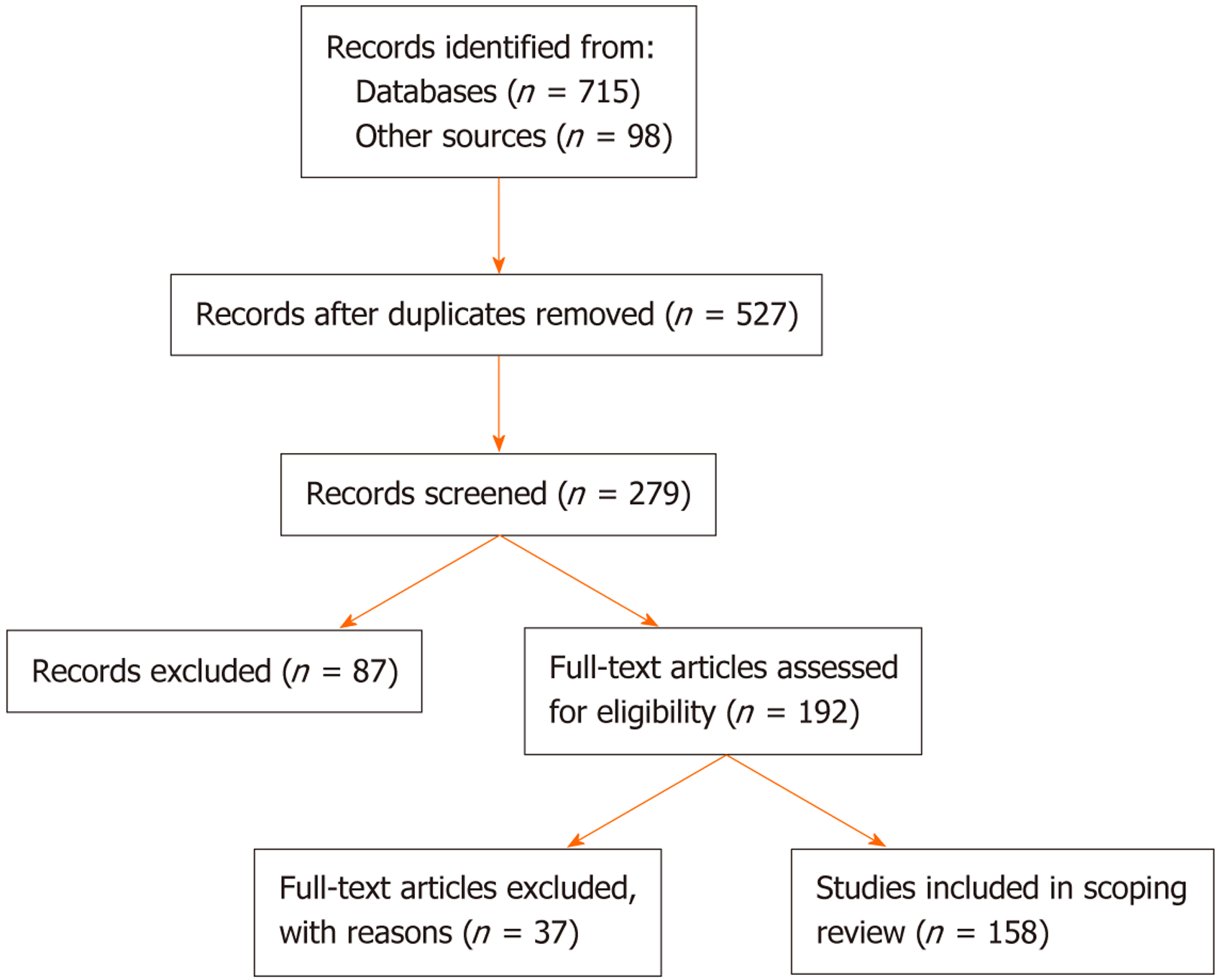
Figure 1 Literature screening and selection process for the scoping review.
A total of 813 records were identified (715 from databases and 98 from other sources). After removing duplicates, 527 records remained, of which 279 were screened. Following title and abstract screening, 192 full-text articles were assessed for eligibility. Of these, 37 were excluded with reasons, leaving 155 studies that were finally included in the review.
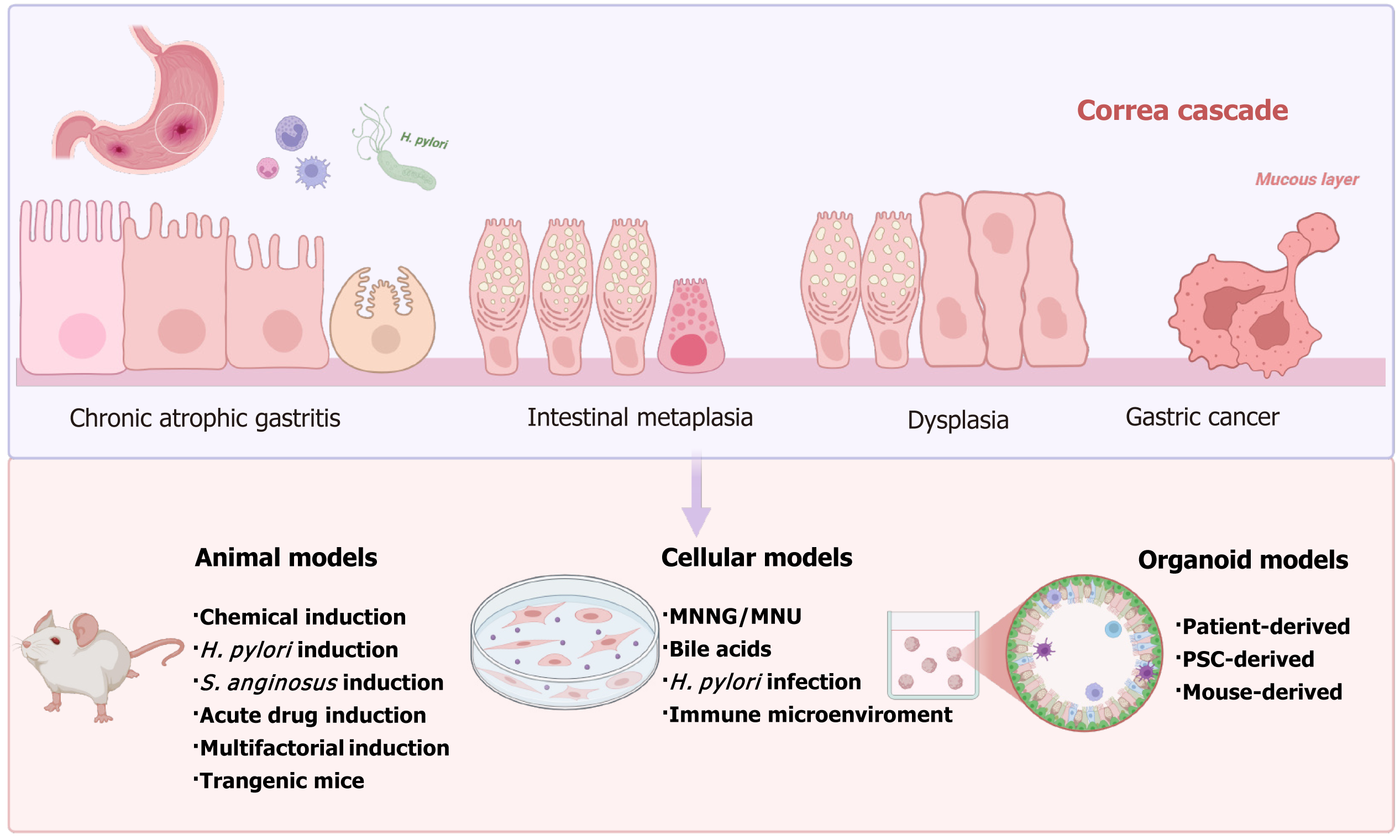
Figure 2 Representative experimental models for each stage of the Correa cascade, from chronic and atrophic gastritis to intestinal me
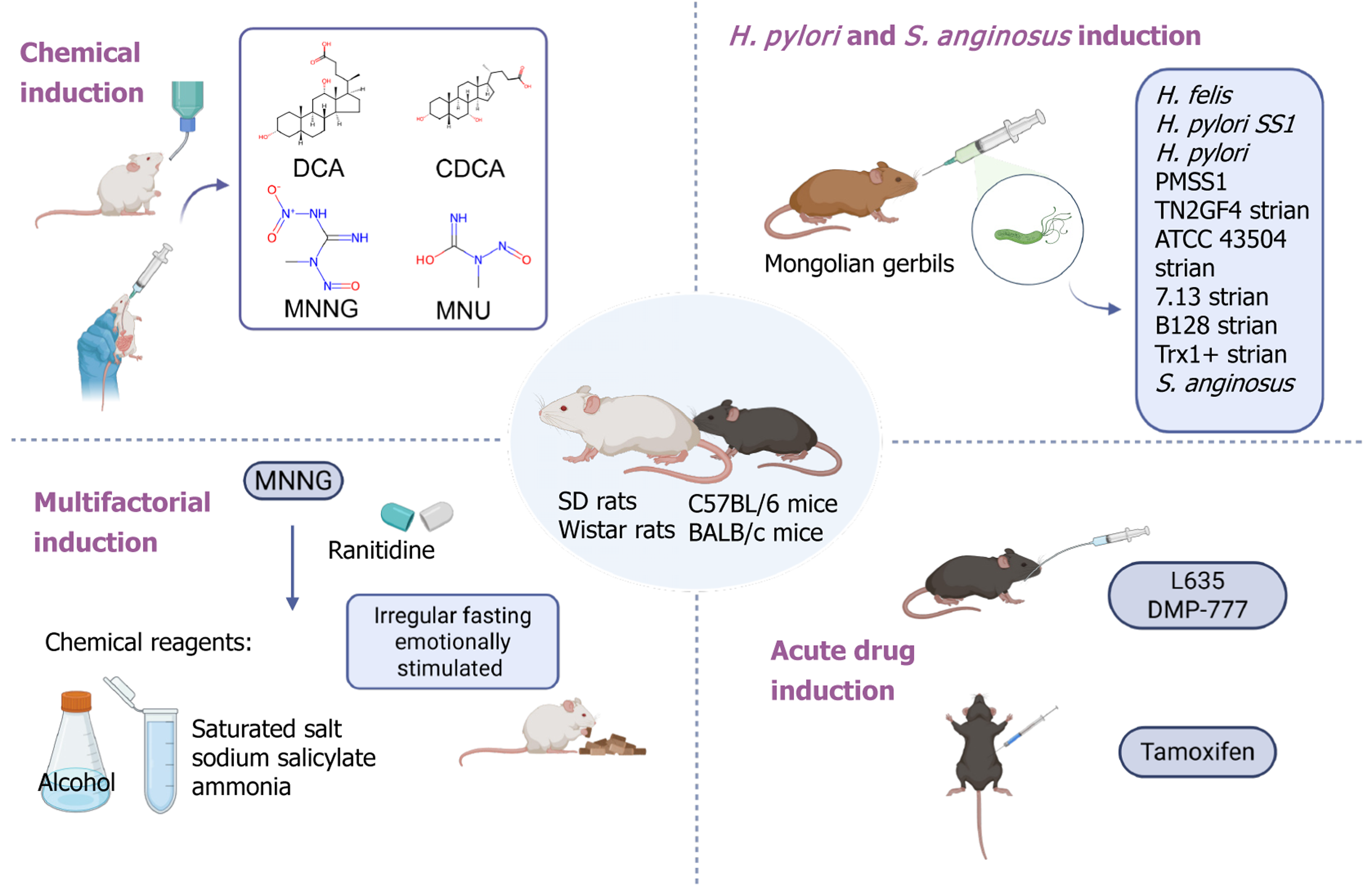
Figure 3 Commonly used strategies for constructing animal models of precancerous lesions of gastric cancer.
Including: (1) Chemical carcinogen-induced models using agents such as N-methyl-N’-nitro-N-nitrosoguanidine or N-Nitroso-N-methylurea in combination with high-salt diet, ethanol, bile acids, or sodium deoxycholate; (2) Infection-based models using Helicobacter pylori in mice or Mongolian gerbils to mimic inflammation-driven transformation; (3) Combination models integrating chemical exposure with Helicobacter pylori infection, high-salt feeding, ethanol administration, fasting, or ammonia water to accelerate disease progression; and (4) Spasmolytic polypeptide-expressing metaplasia models induced by tamoxifen, L635, or DMP-777, which recapitulate key features of gastric glandular cell transdifferentiation. DCA: Deoxycholic acid; CDCA: Chenodeoxycholic acid; MNNG: N-methyl-N’-nitro-N-nitrosoguanidine; MNU: N-Nitroso-N-methylurea; H. pylori: Helicobacter pylori; S. anginosus: Streptococcus anginosus; H. felis: Helicobacter felis; Trx1: Thioredoxin 1; SD: Sprague-Dawley.
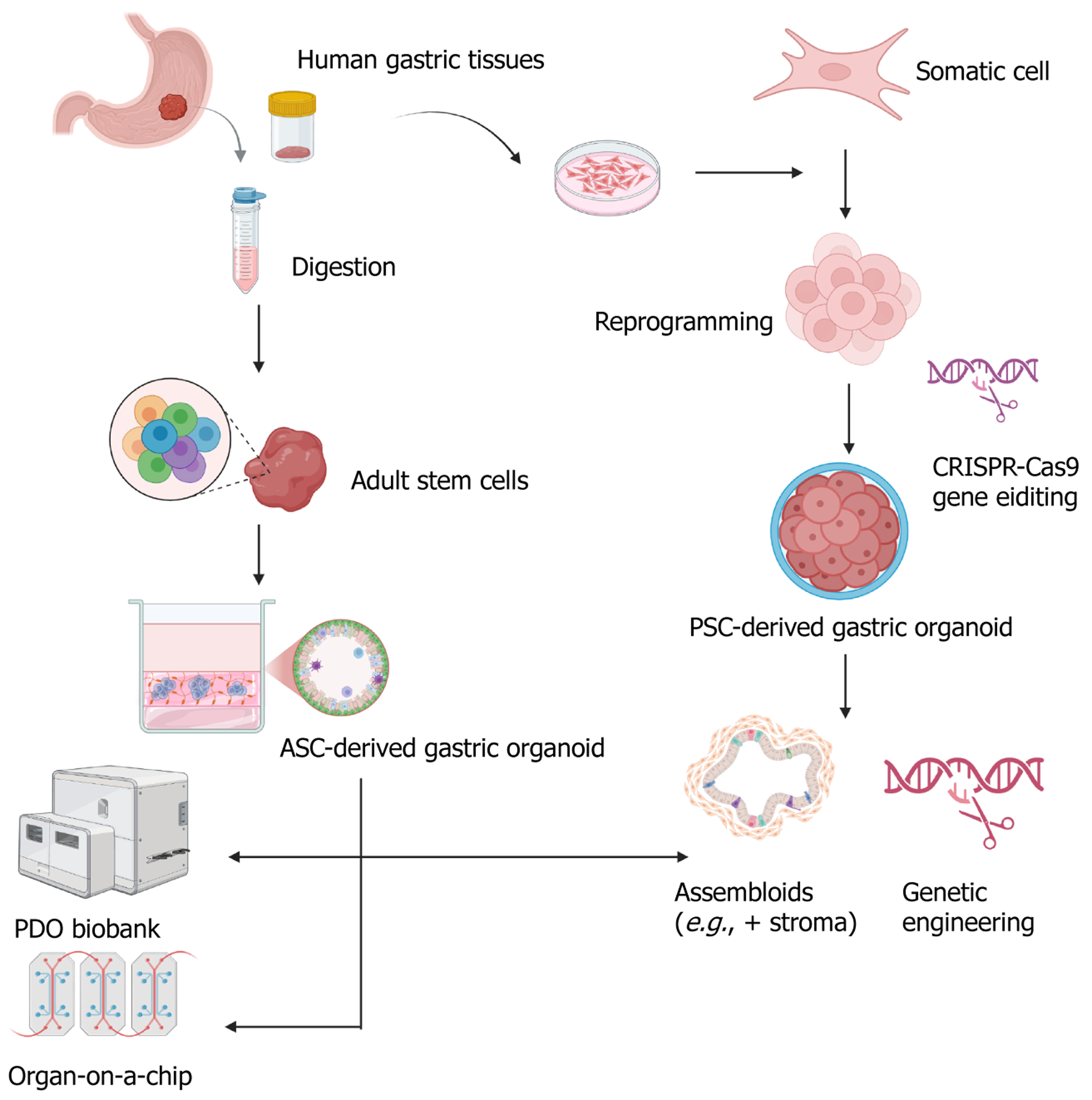
Figure 4 Human gastric organoids can be established either from adult stem cells isolated from gastric tissue biopsies or from plu
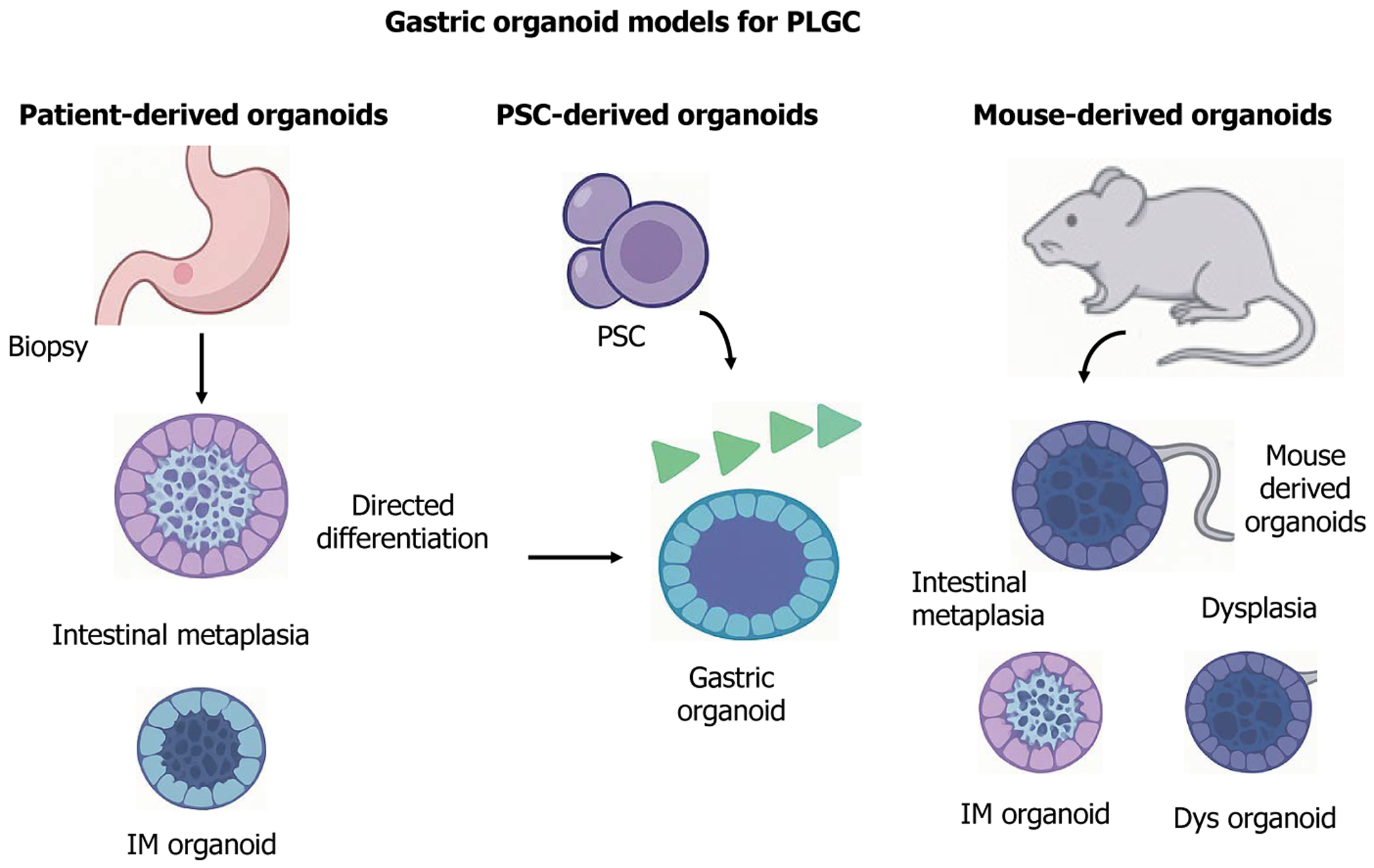
Figure 5 Construction and application of organoid models in precancerous lesions of gastric cancer research.
Organoids can be derived from patient tissues, pluripotent stem cells, or gastric tissues of genetically engineered mice: (1) Patient-derived organoids preserve key histological and molecular features of intestinal metaplasia (IM), dysplasia, and early gastric cancer, enabling personalized modeling of disease progression; (2) Pluripotent stem cell-derived organoids recapitulate gastric development and can be genetically modified to simulate metaplastic transitions (e.g., caudal-related homeobox transcription factor 2-driven IM); and (3) Mouse-derived organoids, including those from models like Mist1-Kras and leucine-rich repeat-containing G protein-coupled receptor 5-p53-/-, facilitate studies of genetic alterations in spasmolytic polypeptide-expressing metaplasia, IM, and dysplasia. Co-culture systems with immune cells or Helicobacter pylori further allow mechanistic investigations into inflammation, epithelial remodeling, and host-pathogen dynamics. These platforms provide versatile tools for in vitro modeling of the Correa cascade. PLGC: Precancerous lesions of gastric cancer; IM: Intestinal metaplasia; PSC: Pluripotent stem cell; Dys: Dysplasia.
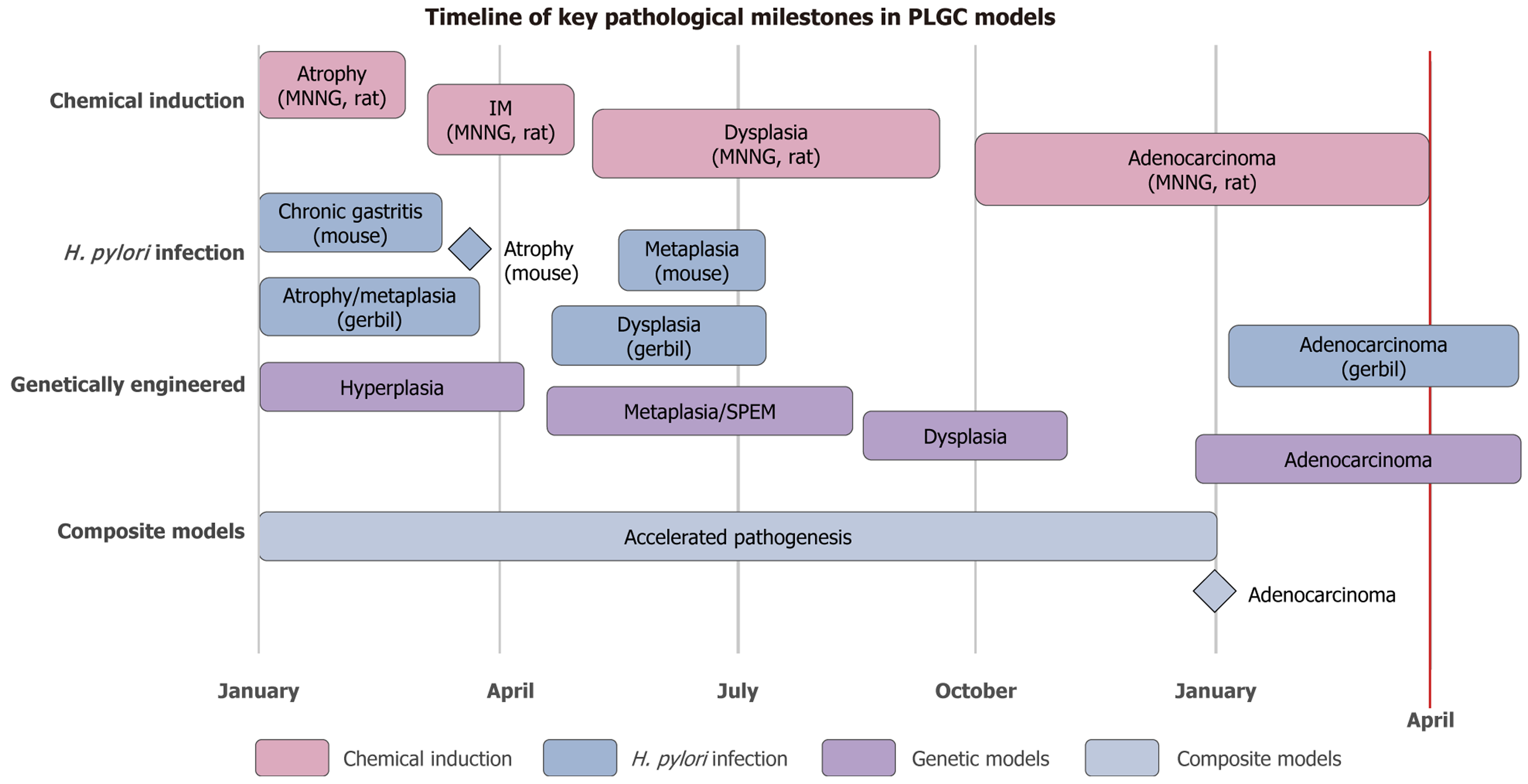
Figure 6 Integrates data from multiple studies to show approximate timeframes for the development of key stages of the Correa cascade.
Model systems are grouped by primary induction method. Note that the exact timing can vary based on specific protocol, species, strain, and dose. Temporal scale: Chemical induction (N-methyl-N’-nitro-N-nitrosoguanidine/N-Nitroso-N-methylurea) offers a relatively predictable and accelerated timeline for carcinogenesis, especially in rats. Natural history: Helicobacter pylori (H. pylori) infection models, particularly in Mongolian gerbils, best recapitulate the natural, multi-stage progression of human gastric carcinogenesis but require significantly longer experiments. Acceleration: Composite models (e.g., H. pylori + N-Nitroso-N-methylurea) and genetically engineered models (e.g., insulin-gastrin mice) can dramatically accelerate the development of advanced lesions and cancer. Model selection: This visual aid helps researchers align their experimental timeframe with their chosen model. For instance, a 6-month study might be sufficient to observe metaplasia in a composite model but would only capture early-stage gastritis in a standard H. pylori infection model in mice. MNNG: N-methyl-N’-nitro-N-nitrosoguanidine; IM: Intestinal metaplasia; H. pylori: Helicobacter pylori; SPEM: Spasmolytic polypeptide-expressing metaplasia.
- Citation: Zhou LJ, Hao XY, Wang C, Ren NN, Wang YG. Comprehensive modeling of the Correa cascade in precancerous lesions of gastric cancer: Leveraging animal, cellular, and organoid systems for translational insights. World J Gastrointest Oncol 2026; 18(3): 113757
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/113757.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.113757