Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.117233
Revised: January 19, 2026
Accepted: March 13, 2026
Published online: April 16, 2026
Processing time: 132 Days and 18.6 Hours
Bronchogenic cysts (BCs) are congenital lesions typically located in the medi
We present two cases of young adults in whom lower esophageal submucosal lesions were incidentally detected during routine health examinations. Preope
Mediastinal cysts require comprehensive evaluations. Transesophageal tunneling provides a minimally invasive approach.
Core Tip: Mediastinal bronchogenic cysts adjacent to the esophagus can closely resemble submucosal esophageal lesions on preoperative imaging, posing diagnostic challenges. We describe two cases managed using a transesophageal mediastinal tunneling technique that integrates submucosal tunneling endoscopic resection with natural orifice transluminal endoscopic surgery. This innovative, minimally invasive approach allows precise intraoperative identification, complete en bloc cyst excision, and excellent postoperative recovery while avoiding thoracotomy.
- Citation: Ma JM, Ji CG, Liu L, Yin KG. Bronchogenic cysts treated with submucosal tunneling endoscopic resection and natural orifice transluminal endoscopy: Two case reports and review of literature. World J Gastrointest Endosc 2026; 18(4): 117233
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/117233.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.117233
Bronchogenic cysts (BCs) are congenital anomalies originating from the ventral aspect of the primitive foregut, resulting from abnormal development of the bronchial tree during embryogenesis[1]. BCs of the mediastinum have an incidence of approximately 6%, accounting for about 10% of primary mediastinal tumors and 50%-60% of mediastinal cysts[1,2]. These cysts are typically lined with respiratory epithelium and are commonly located in the middle and upper portions of the mediastinum. BC occurrence in the posterior mediastinum is relatively rare[3]. Furthermore, BCs may also be found in the lungs, neck, esophagus, and other anatomical locations.
BCs are generally benign lesions, yet nearly half of the patients may develop a range of symptoms and complications over time. The risk of malignant transformation is estimated at approximately 0.7%[4]. Therefore, once diagnosed, BCs should be managed appropriately after excluding contraindications. Surgical resection remains the cornerstone of treatment, such as thoracotomy or video-assisted thoracoscopic surgery[2]. These traditional techniques, although effective, are associated with considerable surgical trauma, postoperative pain, prolonged hospitalization, and extended recovery. Such limitations have fueled the search for less invasive yet equally effective alternatives for the management of mediastinal BCs. In recent years, minimally invasive endoscopic approaches for mediastinal cystic lesions have gained increasing attention, with several reports demonstrating the feasibility and safety of endoscopic resection for selected extraluminal esophageal cysts[5,6].
The submucosal tunneling technique was initially developed as a submucosal endoscopy with a mucosal flap safety valve[7] and was subsequently applied in peroral endoscopic myotomy to establish a submucosal working space across the gastroesophageal junction[8]. Building upon this foundation, submucosal tunneling endoscopic resection (STER) has emerged as a reliable therapeutic approach for submucosal tumors arising from the muscularis propria layer of the esophagus, stomach, or cardia. The technique is particularly suitable for lesions ≤ 3 cm in diameter that are well circumscribed and lack evidence of malignant invasion[9]. Beyond its established role in the treatment of solid submucosal tumors, multiple studies have also reported the application of STER in the management of intramural foregut-derived cysts of the esophagus[10]. In addition, STER has been combined with other endoscopic techniques, such as cyst fenestration, for the treatment of esophageal duplication cysts[11]. Overall, STER allows en bloc resection while maintaining mucosal integrity[12], thereby minimizing postoperative morbidity and promoting more rapid recovery compared with conventional surgical approaches.
Natural orifice transluminal endoscopic surgery (NOTES) encompasses endoscopic procedures in which the endoscope is introduced through a natural orifice, such as the mouth or anus, and then advanced via a transluminal incision to access anatomical regions beyond the reach of conventional endoscopy, including the abdominal and pelvic cavities[13]. Transesophageal approach, in particular, extends endoscopic access beyond the gastrointestinal lumen to mediastinal, thoracic, and even peritoneal structures without external incisions. This technique expands the therapeutic spectrum of endoscopy to encompass lesions located outside the gastrointestinal lumen, offering the potential for truly scarless, minimally invasive surgery.
Here, we present two cases of mediastinal BCs that were initially misdiagnosed as submucosal esophageal lesions originating from the muscularis propria. Additionally, we explore the combined use of STER and NOTES, emphasizing their safety and efficacy. This report highlights the diagnostic challenges and therapeutic potential of advanced endoscopic techniques in managing selected mediastinal lesions. This manuscript targets the dual clinical difficulties of diagnosis and treatment, exploring the application of the combined STER-NOTES technique. The timelines of the two cases are presented in Tables 1 and 2, and the clinical characteristics of these two cases are summarized in Table 3.
| Time | Information about the patient |
| March 15, 2023 | Routine health examination |
| March 27, 2023 | Received EUS |
| April 24, 2023 | Admitted to our hospital |
| April 24, 2023 to April 25, 2023 | Received laboratory and imaging examinations |
| April 26, 2023 | Underwent endoscopic surgery |
| April 27, 2023 | Pathological results confirmed BC |
| May 2, 2023 | Discharged from hospital |
| Time | Information about the patient |
| June 21, 2025 | Routine health examination |
| June 28, 2025 | Received imaging examinations |
| June 30, 2025 | Received EUS |
| July 4, 2025 | Admitted to our hospital |
| July 4, 2025 to July 6, 2025 | Received preoperative laboratory tests |
| July 7, 2025 | Underwent endoscopic surgery |
| July 8, 2025 | Pathological results confirmed BC |
| July 14, 2025 | Discharged from hospital |
| Category | Patient 1 | Patient 2 |
| Age/sex | 32/female | 36/male |
| Admission date | April 2023 | July 2025 |
| Symptom | Asymptomatic, detected during routine exam | Dysphagia for 1 month |
| Endoscopy | submucosal bulge located approximately 35 cm from the incisors, approximately 3.0 cm × 2.0 cm | submucosal bulge located approximately 38 cm from the incisors, approximately 2.0 cm × 2.0 cm |
| EUS | Hypoechoic mass from muscularis propria, approximately 2.7 cm × 1.3 cm, with no blood flow signals | Hypoechoic mass from muscularis propria, approximately 3.2 cm × 2.3 cm, no vascular signal, intermediate stiffness |
| CT | Low-density mass (approximately 2.8 cm × 2.5 cm) near EGJ, mild enhancement | Soft tissue nodule (approximately 3.1 cm × 3.1 cm) near EGJ, mild enhancement, luminal narrowing |
| Laboratory tests | Normal | Normal |
| Past history | None | None |
| Family history | No relevant family or malignancy history | No relevant family or malignancy history |
Case 1: A 32-year-old woman was referred to our hospital after a routine health examination, which revealed a sub
Case 2: A 36-year-old man complained of dysphagia for approximately one month. Besides dysphagia, he had no other symptoms, such as acid regurgitation and body weight loss.
Case 1: Upper gastrointestinal endoscopy identified a submucosal bulge located 35 cm from the incisors. Endoscopic ultrasonography (EUS) revealed the mass originating from the muscularis propria at the cardia; the preoperative diagnosis favored esophageal leiomyoma.
Case 2: Upper gastrointestinal endoscopy identified a submucosal bulge located 38 cm from the incisors. EUS revealed a mass originating from the muscularis propria at the cardia; the preoperative diagnosis favored esophageal leiomyoma.
Case 1: The patient had no remarkable past medical history.
Case 2: The patient had no remarkable past medical history.
Case 1: The patient’s personal and family history were unremarkable.
Case 2: The patient’s personal and family history were unremarkable.
Case 1: The patient’s temperature was 36.4 °C, heart rate was 64 bpm, respiratory rate was 17 breaths/minute, and blood pressure was 124/85 mmHg. The abdomen was soft, with no tenderness, no rebound pain, and no palpable mass.
Case 2: The patient’s temperature was 36.2 °C, heart rate was 82 bpm, respiratory rate was 20 breaths/minute, and blood pressure was 109/76 mmHg. The abdomen was soft, with no tenderness, no rebound pain, and no palpable mass.
Case 1: Laboratory tests, including carcinoembryonic antigen, carbohydrate antigen 19-9, and α-fetoprotein, were within normal ranges.
Case 2: Laboratory tests, including tumor markers, were within normal ranges.
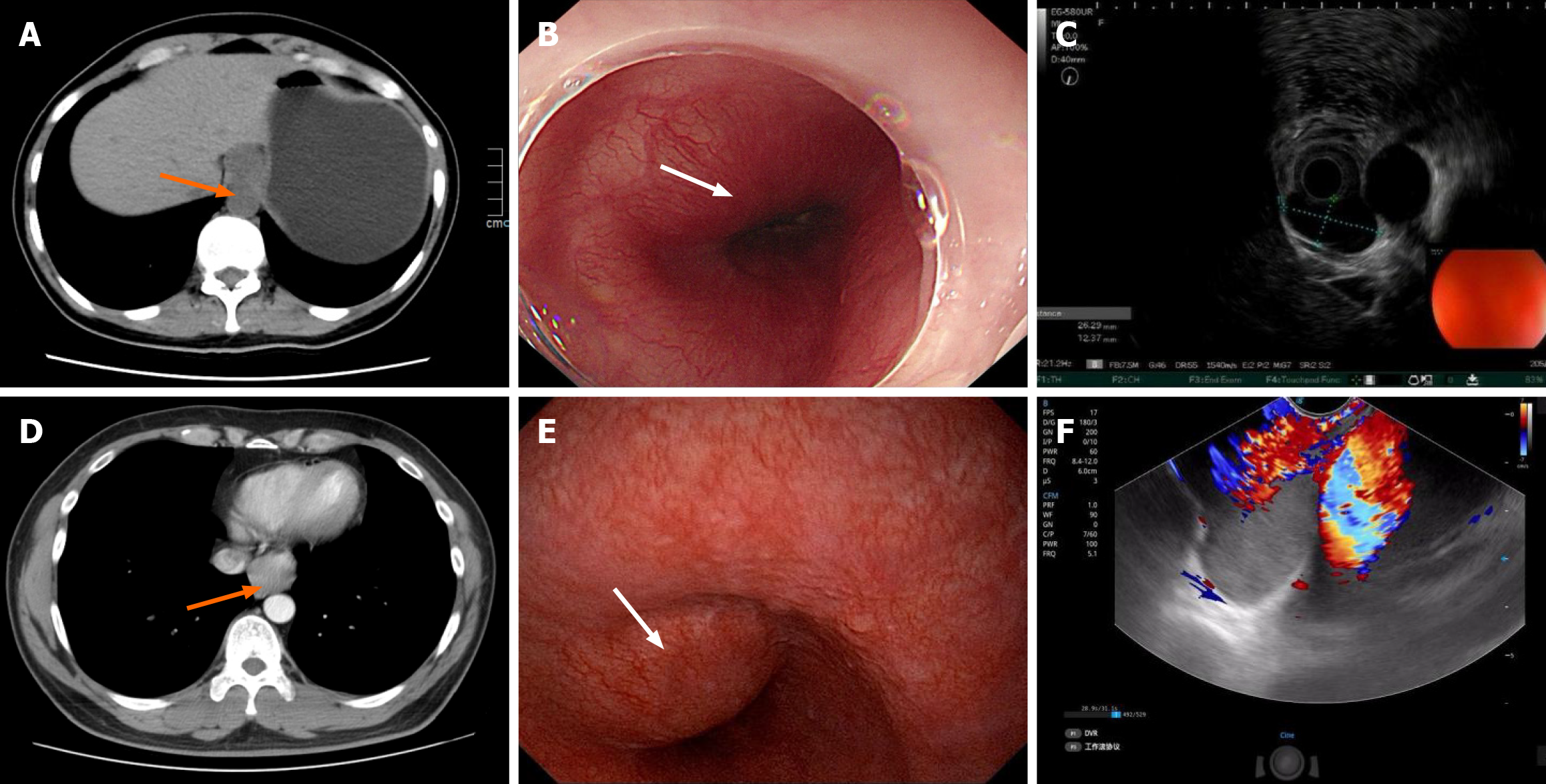
Case 1: Chest computed tomography (CT) revealed a well-defined, round, low-density mass measuring approximately 2.3 cm × 2.0 cm at the gastroesophageal junction. No obvious thickening of the esophageal wall was observed (Figure 1A). Upper gastrointestinal endoscopy (GIF-Q260J, Olympus, Tokyo, Japan) identified a submucosal bulge approximately 3.0 cm × 2.0 cm in size located 35 cm from the incisors (Figure 1B). EUS (EG-580UR, Fujifilm, Tokyo, Japan) revealed a hypoechoic, irregularly shaped mass originating from the muscularis propria at the cardia. The lesion had a clear margin, measuring approximately 2.7 cm × 1.3 cm; no intralesional or perilesional blood flow signals (Figure 1C) were detected on color Doppler flow imaging. Elastography suggested lesions of intermediate stiffness. All the imaging examinations were consistent with a benign submucosal lesion. The preoperative diagnosis favored esophageal leiomyoma.
Case 2: Contrast-enhanced CT showed that the location of the lesion was adjacent to the gastroesophageal junction, and the lesion size was 3.1 cm × 2.8 cm. The lesion exhibited mild enhancement, and the adjacent esophageal lumen appeared narrowed (Figure 1D). On upper gastrointestinal endoscopy (EG-UC5T, SonoScape, Shenzhen, China), a submucosal bulge was observed about 38 cm from the incisors, approximately 2.0 cm × 2.0 cm in size, with an intact and smooth surface (Figure 1E). EUS (EG-UC5T, SonoScape, Shenzhen, China) revealed a hypoechoic mass located 38 cm from the incisors, originating from the muscularis propria, with a local cross-sectional size of approximately 3.2 cm × 2.3 cm. Color Doppler flow imaging showed no vascular signal, and elastography indicated intermediate stiffness (Figure 1F). EUS suggested esophageal leiomyoma as a possible diagnosis.
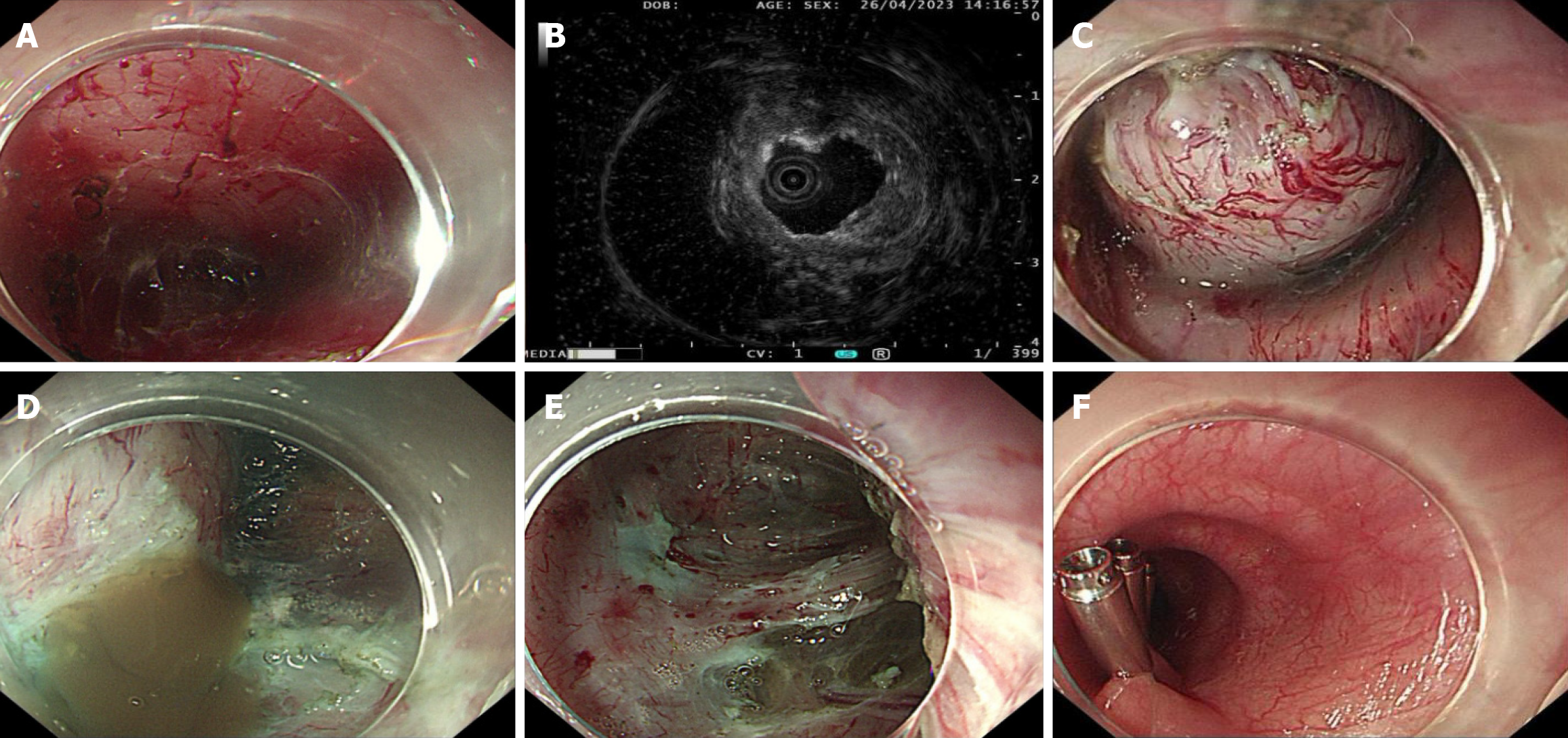
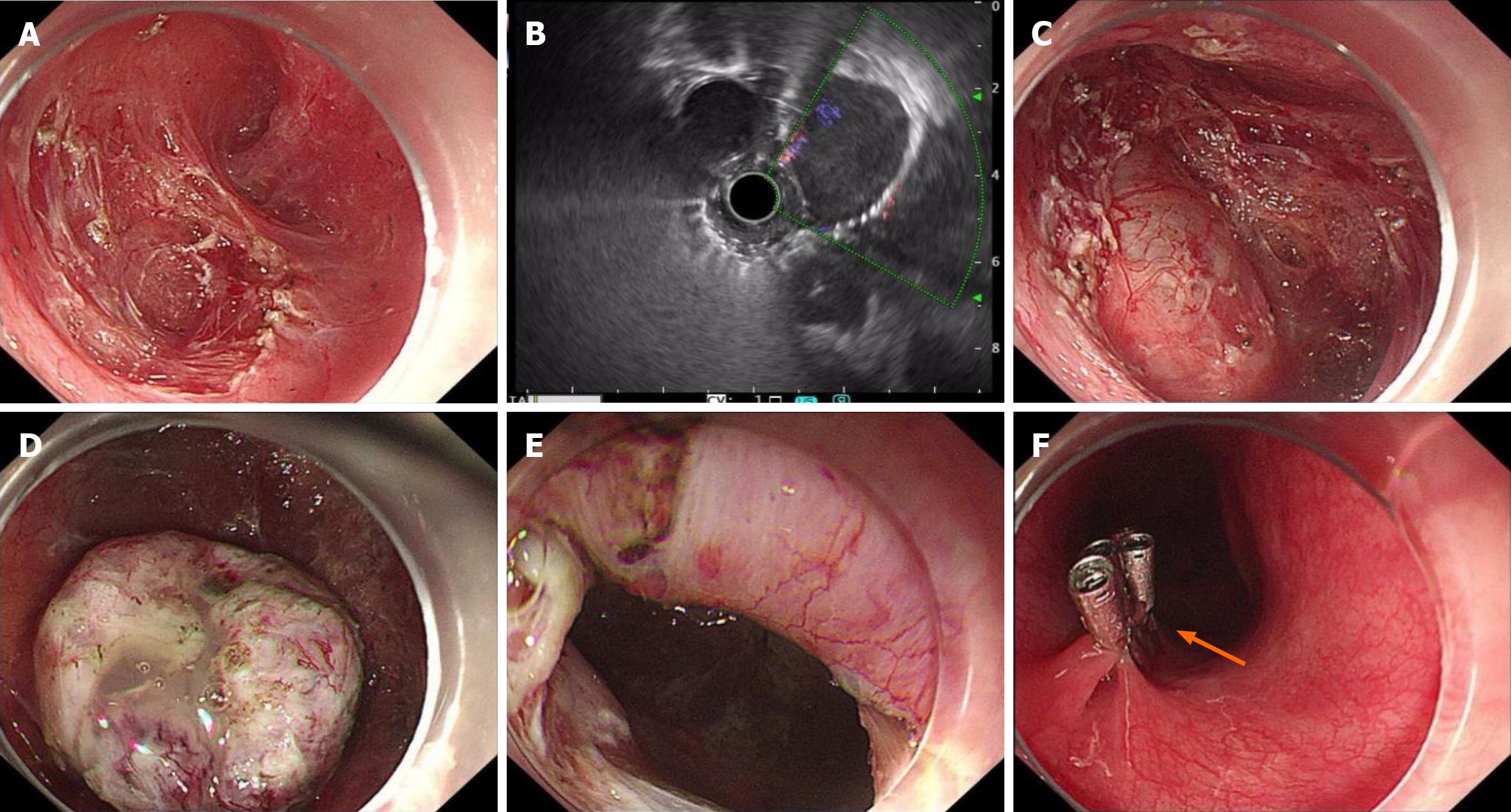
In both cases, preoperative EUS was performed during outpatient endoscopic examinations by experienced endoscopists, and detailed lesion characteristics were systematically documented in the EUS reports. The examinations revealed hypoechoic lesions originating from the muscularis propria with well-defined margins. Color Doppler imaging demonstrated an absence of intralesional vascularity, while elastography suggested lesions of intermediate stiffness. Contrast-enhanced CT showed mildly enhancing, hypovascular nodules with clear boundaries. These findings were reviewed during the preoperative assessment and were considered consistent with benign submucosal lesions by both endoscopists and radiologists. In the absence of alarming clinical symptoms and with unremarkable laboratory results, the preoperative diagnosis favored esophageal leiomyoma, and STER was therefore selected as the initial therapeutic modality (Figures 2 and 3).
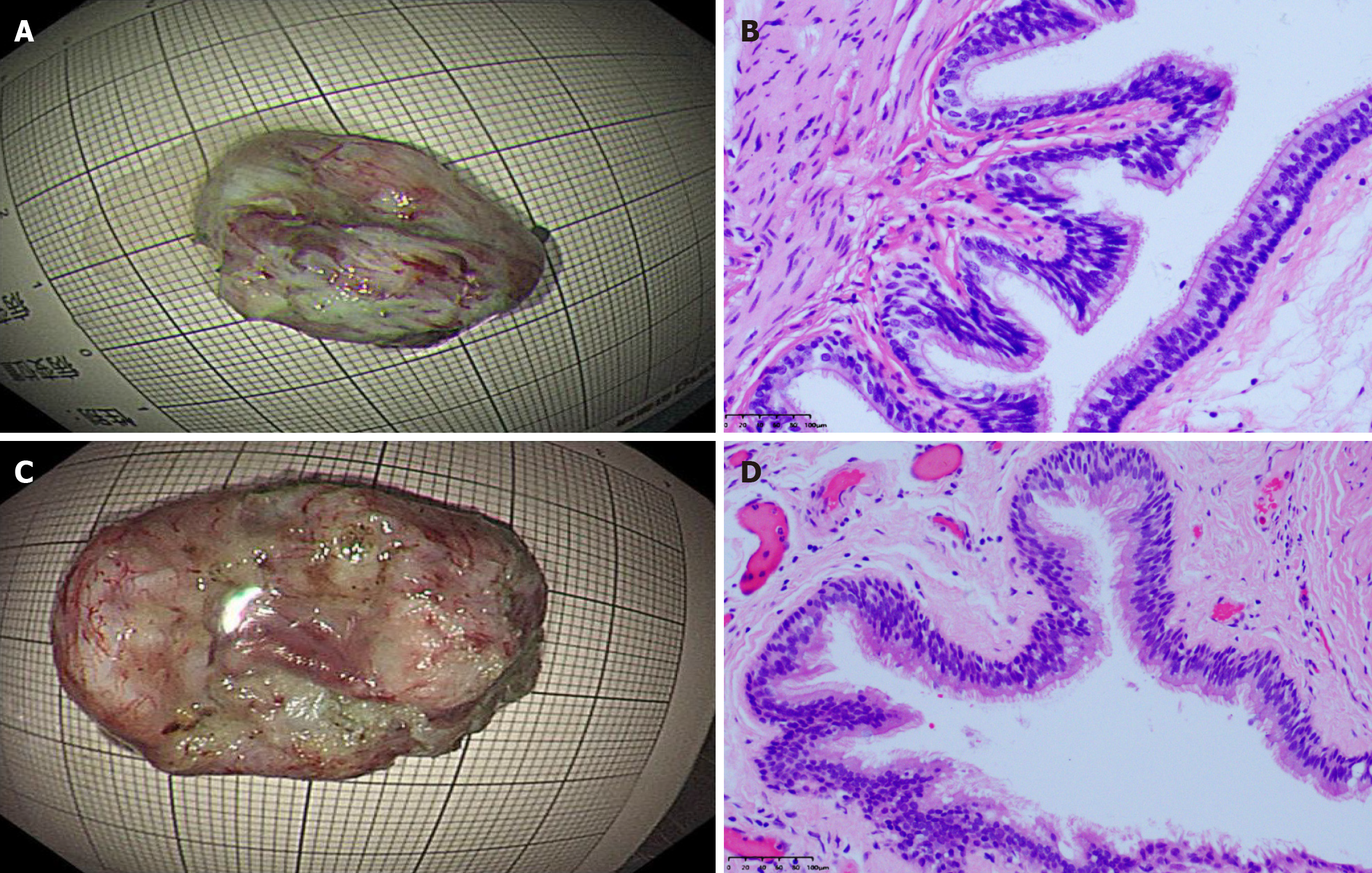
The gross and histopathological findings of the resected mediastinal BCs (Figure 4) demonstrate complete removal and confirm the diagnosis.
BCs tend to be treated when diagnosed, even if they are asymptomatic, because BCs may be complicated by infection or compression of adjacent structures. The cyst’s rupture impacts the pericardial cavity, the pleural cavity, or the trachea. Pneumothorax is another complication that may result in pleuritis. The main management of BC is resection. In our cases, the procedures were performed by three experienced endoscopists, each with more than 10 years of endoscopic expertise. All patients were placed in the left lateral decubitus position under general anesthesia with endotracheal intubation. Carbon dioxide (CO2) insufflation was routinely used throughout the procedure. All procedures were performed using a standard single-channel endoscope (GIF-Q260J, Olympus, Tokyo, Japan). EUS was performed using a high-frequency ultrasound miniprobe (UM-3R, Olympus, Tokyo, Japan) in case 1, and a linear-array echoendoscope (GF-UE260-AL5, Olympus, Tokyo, Japan) in case 2. A transparent distal cap (GF-UE260-AL5) was attached to the tip of the endoscope to enhance visualization and facilitate tissue manipulation.
A mixed solution containing 100 mL of normal saline and 2 mL to 3 mL of indigo carmine was prepared for sub
Upon reaching the lesion site, no abnormalities were identified within the submucosal layer (Figures 2 and 3). Therefore, intraoperative EUS examinations were repeatedly performed both inside and outside the submucosal tunnel. EUS confirmed the presence of the lesion beneath the muscularis propria, and consequently inferred that it originated from the posterior mediastinum (Figures 2 and 3). Although the imaging showed that the lesion was close to the thoracic aorta, a safe and adequate surgical field was established. Based on these findings, a full-thickness myotomy was per
Patients were kept nil per os during the first 48 hours after the procedure. Prophylactic intravenous antibiotics were routinely administered for 48 hours, and acid suppression therapy was provided daily throughout hospitalization. On postoperative day 1, Routine laboratory tests, including complete blood count, C-reactive protein, and procalcitonin, were performed to monitor systemic inflammation. In cases of postoperative chest pain, a chest CT scan was conducted to assess potential complications such as pneumomediastinum or pneumothorax. On postoperative day 3, patients were allowed to initiate a liquid diet, with gradual progression to a normal oral intake as tolerated.
After discharge, both patients were prescribed oral acid-suppressive agents and mucosal protective medications for one month. During follow-up, the first patient remained asymptomatic and experienced no adverse events. At the month-six follow-up, upper gastrointestinal endoscopy revealed a scar located 35 cm from the incisors at the previous resection site. EUS demonstrated no evidence of residual or recurrent cystic lesion, with well-preserved esophageal wall stratification and no abnormalities in the surrounding esophageal tissue. A chest CT scan showed no signs of esophageal wall thickening, luminal dilation, or enlarged mediastinal lymph nodes. The second patient remained free of symptoms or adverse events during follow-up. However, follow-up endoscopy had not yet been performed at the time of this report.
The mediastinum is a unique anatomical compartment located centrally within the thoracic cavity, bordered by the pleural cavities, diaphragm, and thoracic inlet. It contains multiple vital structures and pluripotent cells, providing a permissive environment for the development of various lesions. Mediastinal primary lesions represent a varied group of diseases, encompassing tumors, congenital anomalies, and inflammatory disorders[14,15]. Congenital mediastinal cysts represent a minority of mediastinal masses, accounting for approximately 12%-30% of cases, and are predominantly benign[16,17]. They comprise a variety of pathological types, including BCs, pericardial cysts, thymic cysts, thoracic duct cysts, pleural cysts, enteric cysts, thymic cysts, esophageal duplication cysts, and teratomatous cysts. Among these, BCs represent the most common subtype. They develop from aberrant budding of the tracheobronchial of the primitive foregut between the 3rd and 6th weeks of embryonic life. Detachment and migration of these buds may lead to mucus retention and subsequent cyst formation at the site of migration[18]. Histological examination showed that the cyst wall consisted of bronchial structures, comprising bronchial cartilage, ciliated columnar epithelium, and a few bronchial glands. Approximately 85% of BCs occur in the mediastinum, often close to the trachea, main bronchi, or carina, whereas about 15% are found within the lung parenchyma. In rare instances, BCs may become embedded within the esophageal wall during growth[17].
BCs are often asymptomatic and usually detected incidentally during imaging examinations. The management of asymptomatic BCs remains controversial[4]. While conservative observation may be considered in selected patients, several studies have shown that a substantial proportion of initially asymptomatic cysts may become symptomatic over time due to infection, progressive enlargement, compression, or rupture into adjacent vital thoracic organs. In particular, compression of the esophagus or trachea may lead to clinical manifestations such as chest pain, dysphagia, or respiratory distress. In severe cases presenting with life-threatening symptoms, prompt surgical intervention is required[19,20]. Moreover, delayed intervention may increase technical difficulty because of inflammation, adhesion, or fibrosis[21]. Therefore, prophylactic resection is generally recommended for medically fit patients once the diagnosis is established. Complete surgical resection is the best option to prevent future recurrence and complications, such as malignancy[22]. The present report included a case with asymptomatic BCs. The patient recovered uneventfully. In practice, management decisions should be individualized based on patient symptoms, cyst size and location, growth tendency, and procedural risk, with minimally invasive resection favored when feasible.
Early recognition and appropriate management of BCs are essential to prevent potential complications. Imaging modalities such as CT, magnetic resonance imaging (MRI), and EUS are essential for accurate localization, evaluation of involvement of adjacent structures, and delineation of the lesion. However, due to their diverse and nonspecific clinical presentations, coupled with the phenomenon that different diseases may share similar imaging characteristics, BCs are prone to being misdiagnosed as other mediastinal disorders. A single-center retrospective analysis of 129 patients with BCs systematically summarized their clinical and imaging characteristics[23]. In that series, BCs in two patients mimicked esophageal leiomyoma on EUS, a finding highly consistent with our two cases. These observations further illustrate the heterogeneous and nonspecific imaging appearances of BCs. In the present cases, the cysts were tightly adherent to the esophageal wall, and EUS failed to demonstrate clear extramural characteristics, making it difficult to distinguish them from other submucosal or extramural esophageal lesions, including esophageal leiomyoma, esophageal duplication cyst, pleural fibroma, and neurogenic tumors. Such diagnostic ambiguity complicates clinical decision-making and potentially delays appropriate management.
BCs typically present as solitary, well-circumscribed, round or ovoid cystic lesions on cross-sectional imaging, with smooth margins and sharply defined boundaries, reflecting their congenital cystic nature[24]. CT attenuation values vary widely depending on the composition of the cystic contents. Uninfected cysts usually contain water-like fluid mixed with proteinaceous mucus, resulting in attenuation values of approximately -20 Hounsfield units (HU) to 20 HU. In contrast, secondary infection or intracystic hemorrhage increases the protein concentration, leading to attenuation values exceeding 20 HU, which may further rise with increasing protein content or bleeding; calcification or milk-of-calcium layering within the cyst may also contribute to increased attenuation[25,26]. A subset of lesions may demonstrate irregular or lobulated contours rather than a classic round or ovoid appearance[25]. These atypical imaging features are probably associated with secondary changes, such as inflammation or repeated irritation, and may be accompanied by internal septations, multilocularity, or uneven wall thickening. Such content-dependent variations in attenuation can pose challenges in distinguishing BCs from solid mediastinal tumors on CT alone. In this context, MRI can be useful for demonstrating the cystic nature of the lesion, as the cysts typically show high signal intensity on T2-weighted images and variable signal intensity on T1-weighted images depending on proteinaceous or hemorrhagic content, thereby aiding confirmation when CT findings are equivocal[26].
In the present two cases, both cysts were located adjacent to the esophagus, on its left side above the diaphragm, and displayed a characteristic “teardrop” configuration. The lesions demonstrated soft-tissue attenuation with mild post-contrast enhancement, likely reflecting smooth muscle components within the cyst wall or granulation tissue hyperplasia secondary to repeated inflammation. Based on our experience, the unique anatomical location and variable internal composition of BCs necessitate careful differentiation from other lesions with overlapping clinical and imaging features, in order to ensure diagnostic accuracy and guide the most appropriate therapeutic strategy.
Thymic cysts typically present as well-circumscribed, unilocular or multilocular lesions in the anterior mediastinum and are usually asymptomatic. When symptoms occur, they are generally due to compression of adjacent structures, presenting as chest pain, dyspnea, or arrhythmias. Unlike BCs, thymic cysts rarely adhere to the esophageal wall. Pericardial cysts, most frequently located at the right cardiophrenic angle, may resemble BCs in imaging appearance but are distinguished by their characteristic anatomical location. Large cysts can cause chest pain, dyspnea, palpitations, or nonspecific ST-segment changes on electrocardiography. Esophageal duplication cysts usually appear as smooth, homogeneous paraesophageal lesions on CT. Their close relationship with the esophageal lumen, combined with the EUS finding of continuity between the cyst wall and the esophageal muscularis propria, is highly diagnostic. Neurenteric cysts, though rare, should also be considered. They typically arise in the posterior mediastinum, sometimes in association with vertebral anomalies, and can be differentiated based on their embryologic origin and characteristic location. Among solid tumors, esophageal leiomyomas are the most common benign intramural esophageal neoplasms[27-30]. On EUS, they present as solid hypoechoic masses without cystic components, a key feature that distinguishes them from BCs. Neurogenic tumors, such as schwannomas or neurofibromas, also arise predominantly in the posterior mediastinum and appear as well-defined, solid hypoechoic lesions with heterogeneous enhancement, in contrast to the fluid-filled nature of cysts. Lymphomas present as mediastinal solid masses with relatively high attenuation, frequently accompanied by multiple enlarged lymph nodes and systemic manifestations such as weight loss or cachexia[31-33].
Taken together, the substantial overlap in clinical presentation and imaging characteristics among mediastinal lesions underscores the diagnostic challenges associated with BCs. To reduce discrepancies between preoperative and intraoperative localization, a comprehensive evaluation integrating clinical findings with multimodal imaging is essential for accurate differentiation and clinical decision-making. In particular, careful and standardized EUS assessment plays a central role by providing precise layered characterization of lesion location, origin, size, echogenicity, margins, and vascularity, while cross-sectional imaging offers complementary anatomic information[34]. In selected cases with equivocal findings, MRI may further contribute to lesion characterization by providing additional information on cystic content and tissue properties. Moreover, thorough preoperative review of all imaging studies by the operating endo
Surgical resection remains the definitive treatment for mediastinal BCs, with minimally invasive techniques - including thoracoscopic and endoscopic approaches - gaining increasing preference due to reduced postoperative pain, shorter hospitalization, and faster recovery. In cases where cysts adhere to the esophageal wall or are closely associated with mediastinal structures, combined approaches such as STER and NOTES can enable safe and complete excision. The feasibility and efficacy of transesophageal mediastinal cyst resection using this combination have been demonstrated by Ma et al[35]. Several technical considerations are crucial for the success of this combined approach. Accurate lesion localization with EUS is essential both preoperatively and intraoperatively, particularly when initial exploration does not correlate with surgical findings or when the cyst’s origin and relationship to the esophageal wall layers are unclear. Appropriate tunnel orientation and entry site selection are also important to ensure adequate working space and facilitate secure closure of the mucosal incision. BCs often exhibit high wall tension and fragility, increasing the risk of rupture during dissection; controlled aspiration of cystic fluid may facilitate further dissection and reduce spillage. Careful, blunt, and sharp dissection within the mediastinum is required to avoid injury to adjacent vital structures, such as the trachea and major vessels, while ensuring complete excision to minimize recurrence risk. In addition, meticulous closure of the submucosal tunnel and mucosal entry site is critical, particularly after full thickness myotomy, to prevent postoperative leakage and infectious complications.
Postoperative management should include close monitoring for complications such as mediastinal emphysema, pneumothorax, hemorrhage, infection, and esophageal perforation. Prophylactic antibiotics are recommended for approximately 48 hours postoperatively due to the increased risk of infection following full thickness esophageal myotomy[36]. Careful intraoperative techniques and close postoperative monitoring are critical for minimizing their incidence. Regular follow-up with clinical assessment and imaging is recommended to detect potential recurrence[37].
Despite promising outcomes with minimally invasive strategies, standardized protocols for STER and NOTES are still evolving. Future research with larger patient cohorts and extended follow-up is required to optimize patient selection, refine surgical techniques, and validate long-term outcomes, thereby improving the safety and efficacy of these endoscopic interventions. Ultimately, these efforts will help establish evidence-based guidelines for the management of mediastinal BCs and expand the role of advanced endoscopic techniques in their treatment.
Distinguishing whether a lesion originates from the submucosal layer or from an extramural structure based on clinical features and preoperative imaging findings sometimes is inaccurate. Careful evaluation is therefore required, and intraoperative findings, combined with repeated EUS, provide crucial information for timely reassessment and ad
| 1. | Ali S, Rauf A, Meng LB, Sattar Z, Hussain S, Majeed U. Case Report: Severe back pain, epigastric distress and refractory nausea; an unusual presentation of mediastinal bronchogenic cyst. F1000Res. 2018;7:960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 2. | Strollo DC, Rosado-de-Christenson ML, Jett JR. Primary mediastinal tumors: part II. Tumors of the middle and posterior mediastinum. Chest. 1997;112:1344-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 213] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 3. | Thaller P, Blanchet C, Badr M, Mesnage R, Leboucq N, Mondain M, Cambonie G. Neonatal respiratory distress syndrome revealing a cervical bronchogenic cyst: a case report. BMC Pediatr. 2015;15:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 4. | Kirmani B, Kirmani B, Sogliani F. Should asymptomatic bronchogenic cysts in adults be treated conservatively or with surgery? Interact Cardiovasc Thorac Surg. 2010;11:649-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 5. | Elkholy S, El-Sherbiny M, Okasha HH, Abdallatef A, Haggag H, Zaher MA, Essam K. Endoscopic resection of an extraluminal esophageal duplication cyst. Endoscopy. 2025;57:E770-E771. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | Gao P, Li Q, Hu J, Xu J, Lin S, Cai M, Ma L, Chen W, Zhang Y, Chen SY, Zhong Y, Yao L, Zhou P. Transoesophageal endoscopic removal of a benign mediastinal tumour: a new field for endotherapy? Gut. 2020;69:1727-1729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 7. | Sumiyama K, Gostout CJ, Rajan E, Bakken TA, Knipschield MA, Marler RJ. Submucosal endoscopy with mucosal flap safety valve. Gastrointest Endosc. 2007;65:688-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 172] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 8. | Xu MD, Cai MY, Zhou PH, Qin XY, Zhong YS, Chen WF, Hu JW, Zhang YQ, Ma LL, Qin WZ, Yao LQ. Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos). Gastrointest Endosc. 2012;75:195-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 247] [Article Influence: 17.6] [Reference Citation Analysis (4)] |
| 9. | Inoue H, Ikeda H, Hosoya T, Onimaru M, Yoshida A, Eleftheriadis N, Maselli R, Kudo S. Submucosal endoscopic tumor resection for subepithelial tumors in the esophagus and cardia. Endoscopy. 2012;44:225-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 212] [Article Influence: 15.1] [Reference Citation Analysis (2)] |
| 10. | Sha H, Jiang ZD. Esophageal bronchogenic cyst treated with submucosal tunneling endoscopic resection: two case reports. J Med Case Rep. 2024;18:139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 11. | Chavan R, Nabi Z, Sud S, Gandhi C, Rajput S, Reddy DN. Advanced endoscopic techniques for esophageal duplication cyst treatment: beyond surgery. VideoGIE. 2025;10:326-332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 12. | Chen T, Zhou PH, Chu Y, Zhang YQ, Chen WF, Ji Y, Yao LQ, Xu MD. Long-term Outcomes of Submucosal Tunneling Endoscopic Resection for Upper Gastrointestinal Submucosal Tumors. Ann Surg. 2017;265:363-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 113] [Article Influence: 12.6] [Reference Citation Analysis (5)] |
| 13. | Baron TH. Natural orifice transluminal endoscopic surgery. Br J Surg. 2007;94:1-2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 14. | Macchiarini P, Ostertag H. Uncommon primary mediastinal tumours. Lancet Oncol. 2004;5:107-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 83] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 15. | Strollo DC, Rosado de Christenson ML, Jett JR. Primary mediastinal tumors. Part 1: tumors of the anterior mediastinum. Chest. 1997;112:511-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 205] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 16. | Takeda S, Miyoshi S, Minami M, Ohta M, Masaoka A, Matsuda H. Clinical spectrum of mediastinal cysts. Chest. 2003;124:125-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 156] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 17. | Esme H, Eren S, Sezer M, Solak O. Primary mediastinal cysts: clinical evaluation and surgical results of 32 cases. Tex Heart Inst J. 2011;38:371-374. [PubMed] |
| 18. | Sumiyoshi K, Shimizu S, Enjoji M, Iwashita A, Kawakami K. Bronchogenic cyst in the abdomen. Virchows Arch A Pathol Anat Histopathol. 1985;408:93-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 71] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 19. | Cheng L, Huang A, Jiang K, Xing S, Wang J, Nie J. Urgent Surgical Approach to Mediastinal Cyst Causing Life-Threatening Tracheal Obstruction. Ann Thorac Surg. 2018;105:e125-e128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 20. | Le Pimpec-Barthes F, Cazes A, Bagan P, Badia A, Vlas C, Hernigou A, Pricopi C, Riquet M. [Mediastinal cysts: clinical approach and treatment]. Rev Pneumol Clin. 2010;66:52-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 21. | Hayakawa T, Mitake M, Inaba H, Kobayashi M, Watanabe Y, Okabe A, Funai K. Bronchogenic Cyst with Degeneration of the Adjacent Membranous Portion of the Trachea: A Case Report. Surg Case Rep. 2025;11:25-0287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 22. | Alsmady M, Alshatnawi M, Al Bakri B, Sunoqrot M, Al-Na'san A, Alsabi’ AR, Alimoglu O. Mediastinal Bronchogenic Cysts: Clinical Presentation, Diagnosis, and Treatment Outcomes. Jordan Med J. 2024;58. [DOI] [Full Text] |
| 23. | Ma TT, Chen G, Wang D, Xu H, Zhang JG. Clinical and imaging characteristics of patients with bronchogenic cysts: a single-center retrospective analysis. BMC Med Imaging. 2023;23:128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 24. | McAdams HP, Kirejczyk WM, Rosado-de-Christenson ML, Matsumoto S. Bronchogenic cyst: imaging features with clinical and histopathologic correlation. Radiology. 2000;217:441-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 236] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 25. | Gu X, Zhu L, Li Y, Yin B, Wang Z. Imaging Findings and Misdiagnosis of Bronchogenic Cysts: A Study of 83 Cases. J Belg Soc Radiol. 2023;107:81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 26. | Jeon HG, Park JH, Park HM, Kwon WJ, Cha HJ, Lee YJ, Park CR, Jegal Y, Ahn JJ, Ra SW. Non-infected and Infected Bronchogenic Cyst: The Correlation of Image Findings with Cyst Content. Tuberc Respir Dis (Seoul). 2014;76:88-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 27. | Özdemir Ö. Anterior mediastinal masses and thymic cysts. World J Clin Cases. 2025;13:95035. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (2)] |
| 28. | Sorour AA, Maleszewski JJ, Schaff HV, Klarich KW. A Symptomatic Calcified Pericardial Cyst. Mayo Clin Proc. 2019;94:367-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 29. | Trojan J, Mousset S, Caspary WF, Hoepffner N. An infected esophageal duplication cyst in a patient with non-Hodgkin's lymphoma mimicking persistent disease. Dis Esophagus. 2005;18:287-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 30. | Vargas D, Suby-Long T, Restrepo CS. Cystic Lesions of the Mediastinum. Semin Ultrasound CT MR. 2016;37:212-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 31. | Chafik A, Benjelloun A, Qassif H, El Fikri A, El Barni R, Zrara I. Intramural esophageal bronchogenic cysts. Asian Cardiovasc Thorac Ann. 2011;19:69-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 32. | Tsai SJ, Lin CC, Chang CW, Hung CY, Shieh TY, Wang HY, Shih SC, Chen MJ. Benign esophageal lesions: endoscopic and pathologic features. World J Gastroenterol. 2015;21:1091-1098. [PubMed] [DOI] [Full Text] |
| 33. | Zackria R, Choi EH. Esophageal Schwannoma: A Rare Benign Esophageal Tumor. Cureus. 2021;13:e15667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (2)] |
| 34. | Hamouri S, Hatamleh M, Alaydi J, Alhadidi H, Alomari M, Aldaoud N, Darayseh B. Intra-thymic bronchogenic cyst an extremely rare tumor of anterior mediastinum in adults. J Cardiothorac Surg. 2018;13:120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 35. | Ma LY, Liu ZQ, Yao L, Wang Y, Li XQ, Zhong YS, Zhang YQ, Chen WF, Ma LL, Qin WZ, Hu JW, Cai MY, Li QL, Zhou PH. Transesophageal endoscopic resection of mediastinal cysts (with video). Gastrointest Endosc. 2022;95:642-649.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 36. | Chai NL, Li HK, Linghu EQ, Li ZS, Zhang ST, Bao Y, Chen WG, Chiu PW, Dang T, Gong W, Han ST, Hao JY, He SX, Hu B, Hu B, Huang XJ, Huang YH, Jin ZD, Khashab MA, Lau J, Li P, Li R, Liu DL, Liu HF, Liu J, Liu XG, Liu ZG, Ma YC, Peng GY, Rong L, Sha WH, Sharma P, Sheng JQ, Shi SS, Seo DW, Sun SY, Wang GQ, Wang W, Wu Q, Xu H, Xu MD, Yang AM, Yao F, Yu HG, Zhou PH, Zhang B, Zhang XF, Zhai YQ. Consensus on the digestive endoscopic tunnel technique. World J Gastroenterol. 2019;25:744-776. [PubMed] [DOI] [Full Text] |
| 37. | Asseri AA, Shati AA, Moshebah AY, Alshahrani OM, Saad RM, Alzuhari AM, Qout MMA, Al-Helal AS. Clinical Presentation and Surgical Management of Five Pediatric Cases with Bronchogenic Cysts: Retrospective Case Series. Children (Basel). 2022;9:1824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |