Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.117905
Revised: January 27, 2026
Accepted: March 23, 2026
Published online: April 27, 2026
Processing time: 123 Days and 17.7 Hours
Glucocorticoids (GC) are a potential therapy for acute liver failure (ALF). How
To assess the impact of GC therapy on 28-day survival of patients with ALF and identify early treatment-response factors.
In this single-centre retrospective cohort study, 179 patients with ALF from the past 12 years were included: 84 received GC treatment, and 95 served as non-GC controls. The primary outcome was 28-day survival. GC-treated patients were further stratified into responders and nonresponders to analyse the determinants of efficacy. Survival distributions were compared using Kaplan-Meier curves with the log-rank test. Independent predictors of GC response were identified through multivariate logistic regression. Statistical significance was set at P < 0.05.
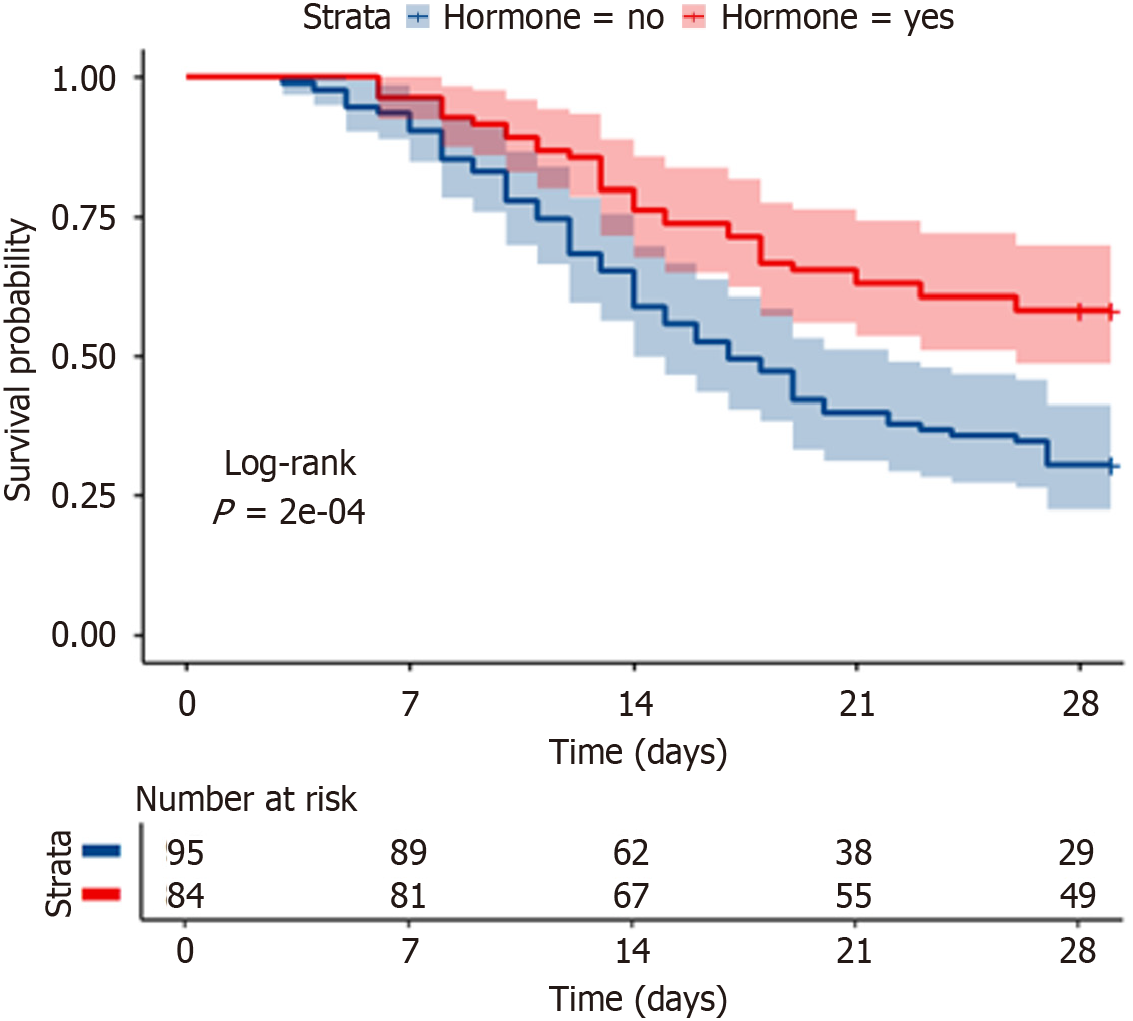
The 28-day survival rate was significantly greater in the GC group than in the control group (58.3% vs 30.5%, P < 0.001). An early increase in prothrombin activity (PTA ≥ 4.5% by day 3), along with baseline model for end-stage liver disease (MELD) score < 28.5 and blood ammonia concentration < 135.5 μg/dL, was independently associated with GC response. A model combining these factors predicted GC responsiveness with an accuracy of 95.2%.
GC therapy improves 28-day ALF survival. An early increase in the PTA, combined with baseline MELD score and blood ammonia level, effectively identifies patients who are most likely to benefit.
Core Tip: This retrospective cohort study of 179 acute liver failure patients demonstrates that glucocorticoid (GC) therapy significantly improves 28-day survival (58.3% vs 30.5%, P < 0.001). An early increase in prothrombin activity ≥ 4.5% by day 3, alongside baseline model for end-stage liver disease score < 28.5 and blood ammonia < 135.5 μg/dL, forms a highly accurate predictive model for GC response (95.2% accuracy). This model provides a practical tool to select patients most likely to benefit from GC treatment.
- Citation: He YL, Yin XH, Xu XT, Li J, Meng QH. Glucocorticoid therapy in acute liver failure: Survival outcomes, predictors, and a response prediction model. World J Hepatol 2026; 18(4): 117905
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/117905.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.117905
Acute liver failure (ALF) is a severe syndrome characterized by abrupt onset and rapid progression, manifesting as sudden hepatic injury, coagulopathy [international normalized ratio (INR) ≥ 1.5], and hepatic encephalopathy (HE) (≥ grade II) in patients without preexisting liver disease and is associated with extremely high mortality. The aetiology of ALF is diverse, and its pathogenesis is complex, with excessive inflammatory responses and immune activation con
Glucocorticoids (GCs) are a focal point in ALF management because of their potent immunomodulatory and antiinflammatory effects. Preclinical evidence has demonstrated that GCs mitigate immunemediated liver injury through multiple pathways, including by suppressing cytotoxic responses, modulating immune signalling, and inducing inflammatory cell apoptosis[4]. Clinically, early high-dose GC administration has been associated with reduced hepatocyte destruction and improved survival[5]. However, their utility is constrained by heterogeneous treatment responses and an increased risk of infections, underscoring the need for a highly individualized therapeutic strategy. Moreover, de
Controversy persists regarding the efficacy of GC therapy in ALF. This study aimed to determine whether GC treatment improves transplantfree survival in patients with ALF and to identify factors predictive of treatment response in GCtreated patients, ultimately informing early clinical identification of potential beneficiaries.
This single-center retrospective cohort study was conducted at Beijing YouAn Hospital, Capital Medical University. Hospitalized patients aged 18 years to 84 years who were diagnosed with ALF between September 2010 and December 2021 were consecutively enrolled. The manuscript was prepared according to the STROBE Statement. The diagnosis of ALF followed the Chinese Guidelines for the Diagnosis and Treatment of Liver Failure (2018 edition)[7], defined by acute onset with grade II or higher HE plus at least two of the following: (1) Severe gastrointestinal symptoms; (2) Serum total bilirubin (TBIL) ≥ 10 × upper limit of normal or a daily increase ≥ 17.1 μmol/L; (3) Prothrombin activity (PTA) ≤ 40% or INR ≥ 1.5; and (4) Imaging evidence of progressive liver shrinkage. Patients with concurrent shock, baseline infection, malignancy, or contraindications to GC therapy were excluded. The 28-day survival status was recorded.
Patients assigned to the GC treatment group received intravenous methylprednisolone (1 mg/kg/day), with the initiation and duration of therapy determined by the treating clinicians on the basis of the patient’s clinical course. The primary endpoint was 28-day transplant-free survival, which was determined through medical records and telephone follow-up. For patients who died during hospitalization, the date of death was confirmed via case record retrieval. Patients who were discharged were followed up by phone to determine their condition on day 28. The 28-day period was calculated from the date of admission. Demographic and clinical data, including laboratory parameters (complete blood count, liver and kidney function, and coagulation profile) at baseline and on day 3 of treatment, were extracted from the hospital’s electronic medical records. The model for end-stage liver disease (MELD) score was calculated as 11.2 × ln (INR) + 9.57 × ln (creatinine) + 3.78 × ln (TBIL) + 6.43. The MELD-Na score was calculated as MELD - [0.025 × MELD × (140 - Na)] + 140.
Statistical analyses were performed using R software (version 4.1.0), SPSS 23.0 and EasyR (https://www.easyr.cc Solutions, Inc., Shanghai). Continuous variables were compared between two independent groups using Student’s t test or the Mann-Whitney U test, as appropriate. Factors influencing GC response were analysed by logistic regression. The optimal cut-off points for continuous predictive factors were determined by maximizing the Youden index. Patients were then stratified on the basis of these cut-off points for early predictive indicators. Survival outcomes across subgroups were evaluated with Kaplan-Meier curves and compared using the log-rank test. To assess longitudinal changes, we employed generalized estimating equations. Preliminary assessment using histograms and Q-Q plots indicated that TBIL, INR, and PTA were approximately normally distributed on the original scale in our sample (Shapiro-Wilk test P > 0.05). Therefore, analyses were performed on the original scale to maintain clinical interpretability. For all three outcomes, we specified a Gaussian distribution with identity link. We evaluated three candidate correlation structures [independent, exchangeable, AR (1)] using the Quasi-likelihood under Independence Criterion; the exchangeable structure was retained as optimal. Robust sandwich variance estimators were used to ensure valid inference. Model diagnostics included plots of standardized residuals vs fitted values and Q-Q plots, which supported the adequacy of the Gaussian assumption. The primary focus was the group-by-time interaction effect. Two-sided P < 0.05 was considered statistically significant.
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki. The protocol was submitted for review, for which approval was obtained from the Ethics Committee of Beijing YouAn Hospital, Capital Medical University (No. LL-2024-139-K).
A total of 179 patients with ALF were included in the analysis. The most common aetiology was druginduced liver injury (42.46%), followed by viral hepatitis (36.88%). Among drug-induced cases, the three leading causes were traditional Chinese medicine (42.11%), antipyreticanalgesic drugs (21.05%), and antituberculosis agents (13.16%), as detailed in Supplementary Figure 1.
Among the total cohort, 84 patients received GC therapy, while 95 did not. Baseline characteristics of the two groups are presented in Supplementary Table 1. The median age differed significantly between the groups. After adjusting for age, the 28-day survival rate remained significantly greater in the GC-treated group than in the non-GC group (58.33% vs 30.53%, P < 0.001), as shown in Figure 1.
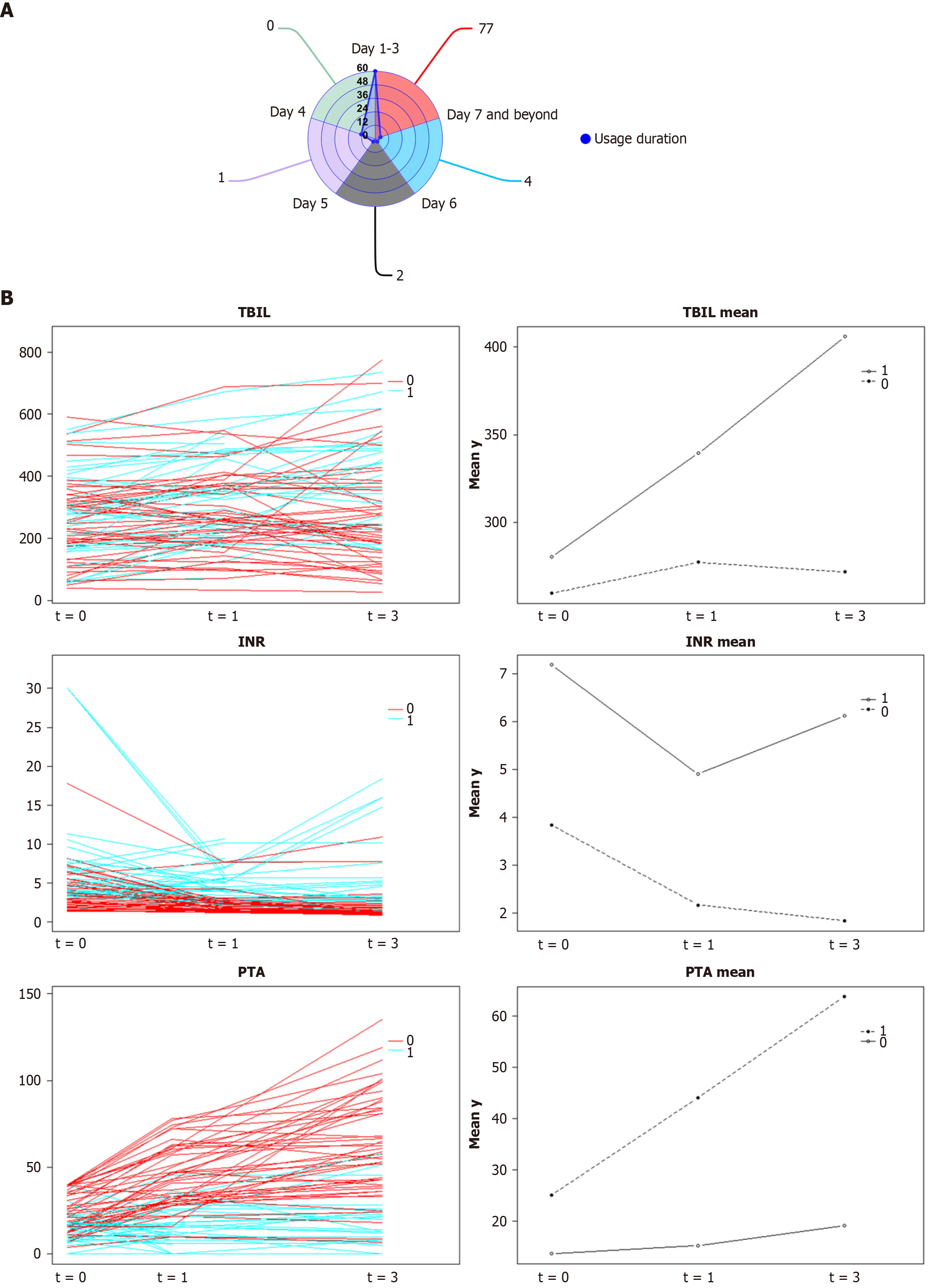
Among the total cohort, 84 patients received GC therapy. The timing and duration of treatment are summarized in Figure 2A. Most patients (94%) initiated GC within 1-3 days of disease onset, with the treatment course typically lasting less than one week, as shown in Figure 2A. On the basis of 28-day survival, these 84 patients were stratified into GC responders (survivors, n = 49) and GC non-responders (non-survivors, n = 35). Comparisons between groups revealed that compared with responders, non-responders had significantly worse baseline coagulation, higher blood ammonia levels, and higher MELD and MELD-Na scores, as detailed in Table 1.
| Parameters | Overall (n = 84) | Responders group (n = 49) | Nonresponders group (n = 35) | P value |
| Age | 38.00 (26.75-50.00) | 37.00 (28.00-49.00) | 41.00 (26.00-56.00) | 0.57 |
| Gender (male) | 43 (51.2) | 24 (49.0) | 19 (54.3) | 0.63 |
| ALT (U/L) | 1869.00 (720.78-3601.50) | 1379.00 (506.50-3347.00) | 2287.00 (810.50-4062.50) | 0.12 |
| AST (U/L) | 880.00 (289.50-2225.50) | 812.00 (216.00-1680.00) | 1342.00 (460.00-3178.40) | 0.06 |
| TBIL (μmol/L) | 255.85 (172.18-346.75) | 249.40 (175.90-327.20) | 280.20 (170.55-392.95) | 0.55 |
| DBIL (μmol/L) | 149.40 (90.55-217.50) | 137.60 (93.30-212.40) | 163.50 (89.10-219.35) | 0.73 |
| Albumin (g/L) | 32.35 (28.80-34.90) | 32.70 (29.40-35.40) | 31.60 (27.85-33.30) | 0.04 |
| UREA (mmol/L) | 4.14 (2.13-6.61) | 4.40 (2.13-6.90) | 3.74 (2.34-5.70) | 0.50 |
| Cr (μmol/L) | 62.50 (48.50-108.14) | 62.00 (47.00-108.14) | 68.00 (49.50-99.50) | 0.51 |
| NH3 (ug/dL) | 148.00 (110.00-192.25) | 133.00 (93.00-172.01) | 171.00 (141.50-259.50) | 0.00 |
| Na+ (mmol/L) | 136.95 (134.65-139.65) | 136.70 (134.70-139.10) | 137.20 (134.45-139.95) | 0.75 |
| K+ (mmol/L) | 3.79 (3.58-4.30) | 3.73 (3.53-4.22) | 3.84 (3.59-4.36) | 0.30 |
| PT (second) | 42.35 (28.20-70.58) | 30.00 (21.60-56.30) | 52.20 (40.65, 88.70) | < 0.001 |
| PTA (%) | 17.00 (10.75-30.85) | 25.00 (13.00-38.80) | 15.00 (7.80-18.00) | < 0.001 |
| INR | 3.76 (2.43-5.73) | 2.88 (1.89-4.98) | 4.55 (3.58-7.22) | 0.00 |
| FIB (g/L) | 1.04 (0.77-1.55) | 1.10 (0.80-1.57) | 0.98 (0.73-1.37) | 0.45 |
| WBC (109/L) | 9.57 (7.29-11.80) | 9.40 (7.05-11.52) | 10.15 (7.60-13.82) | 0.35 |
| NEUT (109/L) | 6.20 (5.16-9.22) | 5.91 (5.06-8.54) | 7.52 (5.49, 11.60) | 0.12 |
| HGB (g/L) | 125.00 (111.00-142.00) | 124.00 (113.00-142.00) | 126.00 (111.00-145.00) | 0.88 |
| PLT (109/L) | 150.55 (68.51) | 153.04 (65.77) | 147.06 (72.99) | 0.70 |
| AST/ALT | 0.62 (0.33-1.06) | 0.60 (0.31-1.03) | 0.67 (0.34-1.12) | 0.66 |
| NLR | 4.84 (2.85-7.96) | 4.07 (2.64-7.16) | 5.94 (3.88-10.60) | 0.04 |
| MEDL | 26.50 (20.75-33.00) | 23.00 (18.00-28.00) | 32.00 (27.00-37.00) | < 0.001 |
| MELD-Na | 28.50 (21.75-34.00) | 23.00 (19.00-30.00) | 34.00 (29.00-37.50) | < 0.001 |
In an effort to identify early indicators predictive of the response to GC therapy, longitudinal changes in TBIL, INR, and PTA were analyzed in GC responders and nonresponders at three time points, namely, baseline, day 3 of GC treatment, and 3 days after completion, as shown in Figure 2B. Responders exhibited downwards trajectories in TBIL and INR alongside an upwards trajectory in PTA. We have supplemented this section in the main text. For the indicator of TBIL, there was no significant difference between the two groups at baseline (P = 0.4969). However, after 3 days of hormone treatment, there was a significant difference between the two groups (P = 0.0263), but after hormone therapy, there was still a significant difference between the two groups (P = 0.0011).
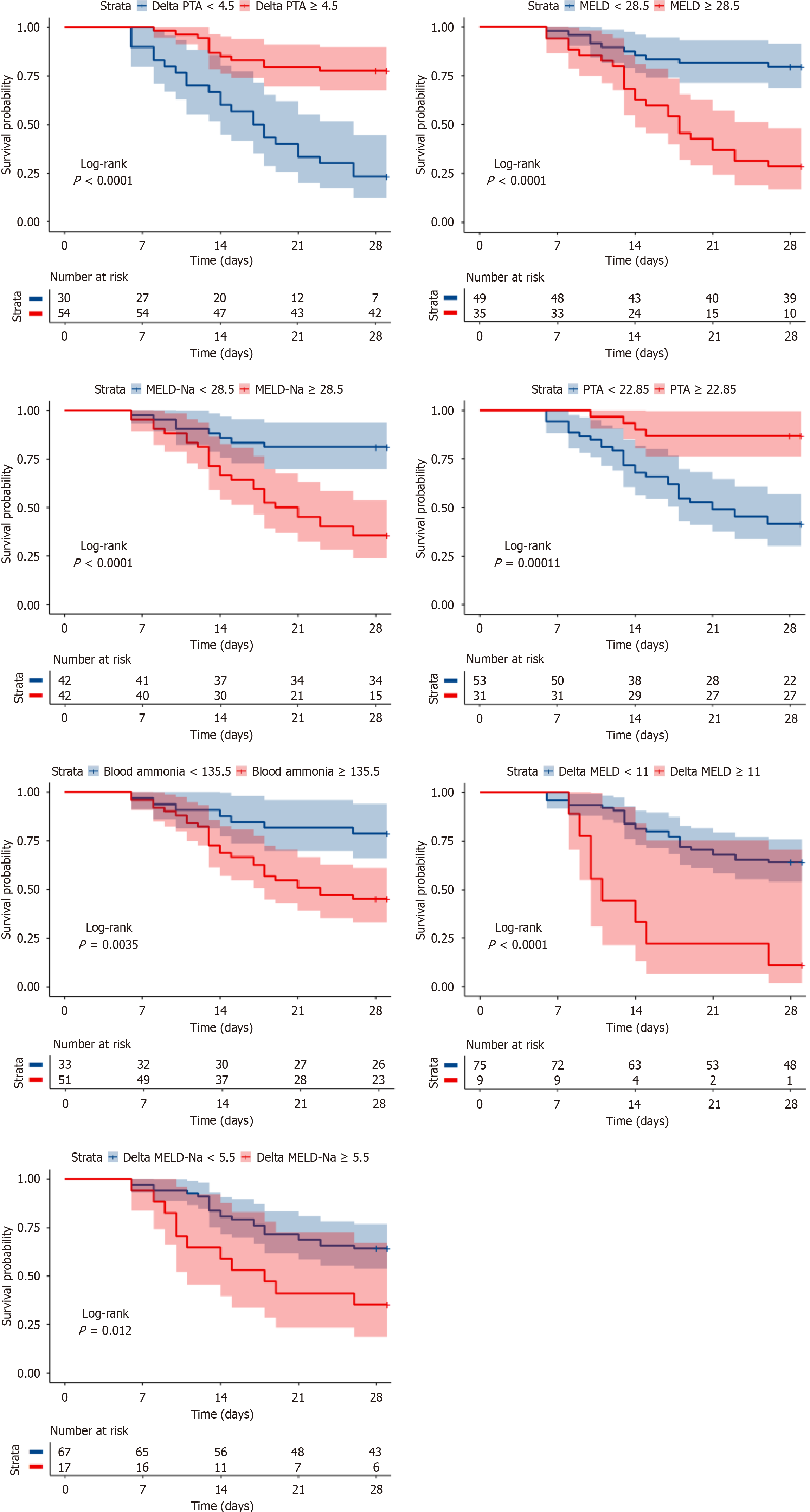
While decreasing trends were also observed for MELD and MELD-Na scores, the differences between groups did not reach statistical significance. Subsequent univariate and multivariate analyses revealed baseline PTA, ΔPTA, and blood ammonia level to be independent factors influencing the response to GC treatment (P < 0.05), as detailed in Table 2.
| Characteristic | Univariate analysis | Multivariate analysis | ||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age | 0.84 (0.54-1.29) | 0.42 | - | - |
| Gender | 1.24 (0.52-2.95) | 0.63 | - | - |
| WBC | 0.88 (0.57-1.35) | 0.55 | - | - |
| NEUT | 0.82 (0.53-1.27) | 0.37 | - | - |
| HGB | 1.20 (0.78-1.85) | 0.41 | - | - |
| NLR | 0.63 (0.39-1.01) | 0.05 | - | - |
| PLT | 1.09 (0.70-1.69) | 0.69 | - | - |
| ALT | 0.68 (0.43-1.08) | 0.10 | - | - |
| AST | 0.51 (0.24-1.08) | 0.08 | - | - |
| AST/ALT | 0.96 (0.62-1.48) | 0.85 | - | - |
| TBIL | 0.86 (0.56-1.33) | 0.50 | - | - |
| UREA | 0.77 (0.42-1.43) | 0.42 | - | - |
| Cr | 0.88 (0.57-1.36) | 0.58 | - | - |
| INR | 0.38 (0.16-0.92) | 0.03 | 1.28 (0.60-2.70) | 0.53 |
| PT | 0.39 (0.20-0.77) | 0.01 | 0.83 (0.39-1.77) | 0.63 |
| PTA | 3.24 (1.77-5.94) | < 0.001 | 4.21 (1.09-16.34) | 0.04 |
| ΔPTA | 4.96 (2.39-10.33) | < 0.001 | 7.78 (2.79-21.68) | < 0.001 |
| Blood ammonia | 0.46 (0.27-0.79) | 0.01 | 0.42 (0.19-0.96) | 0.04 |
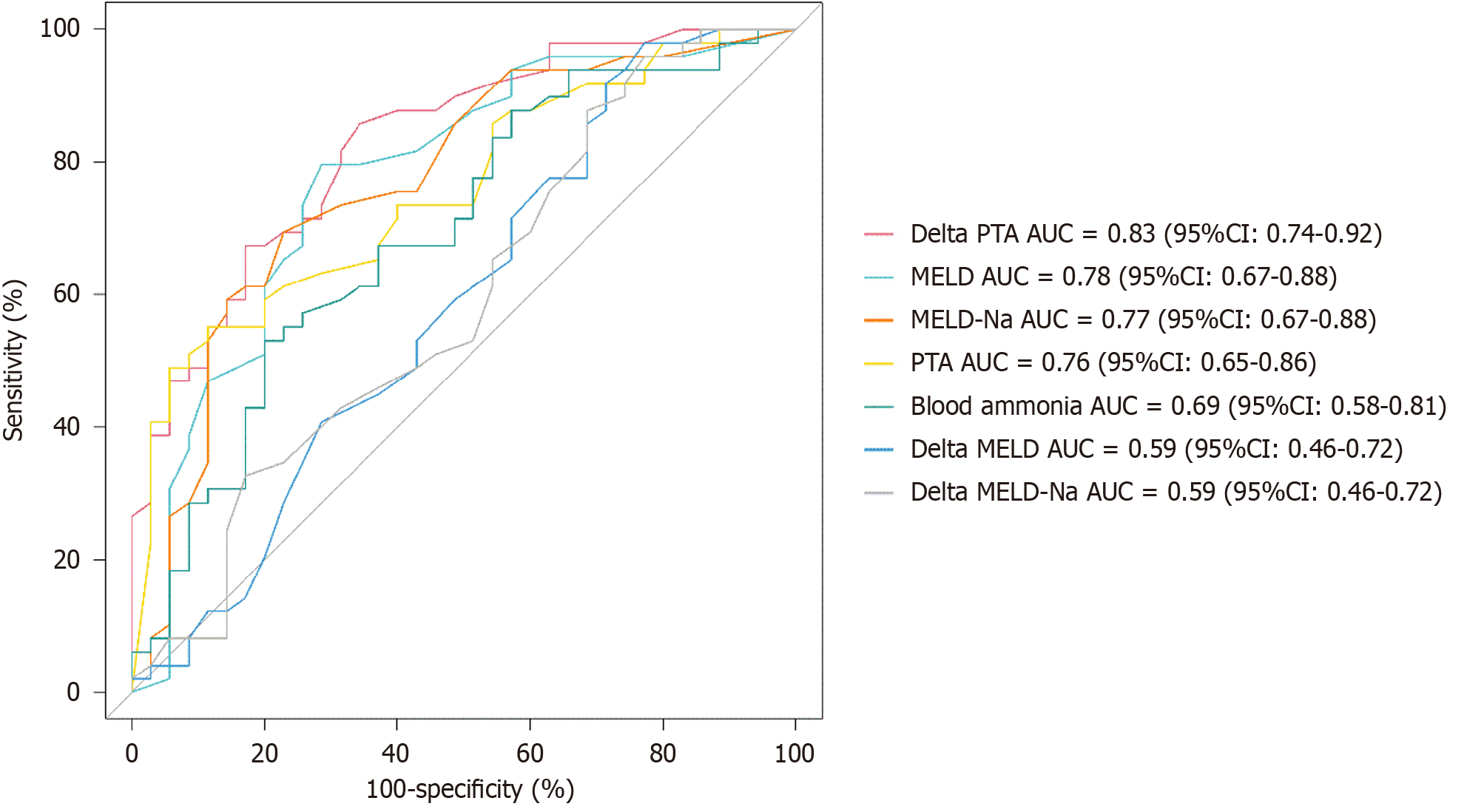
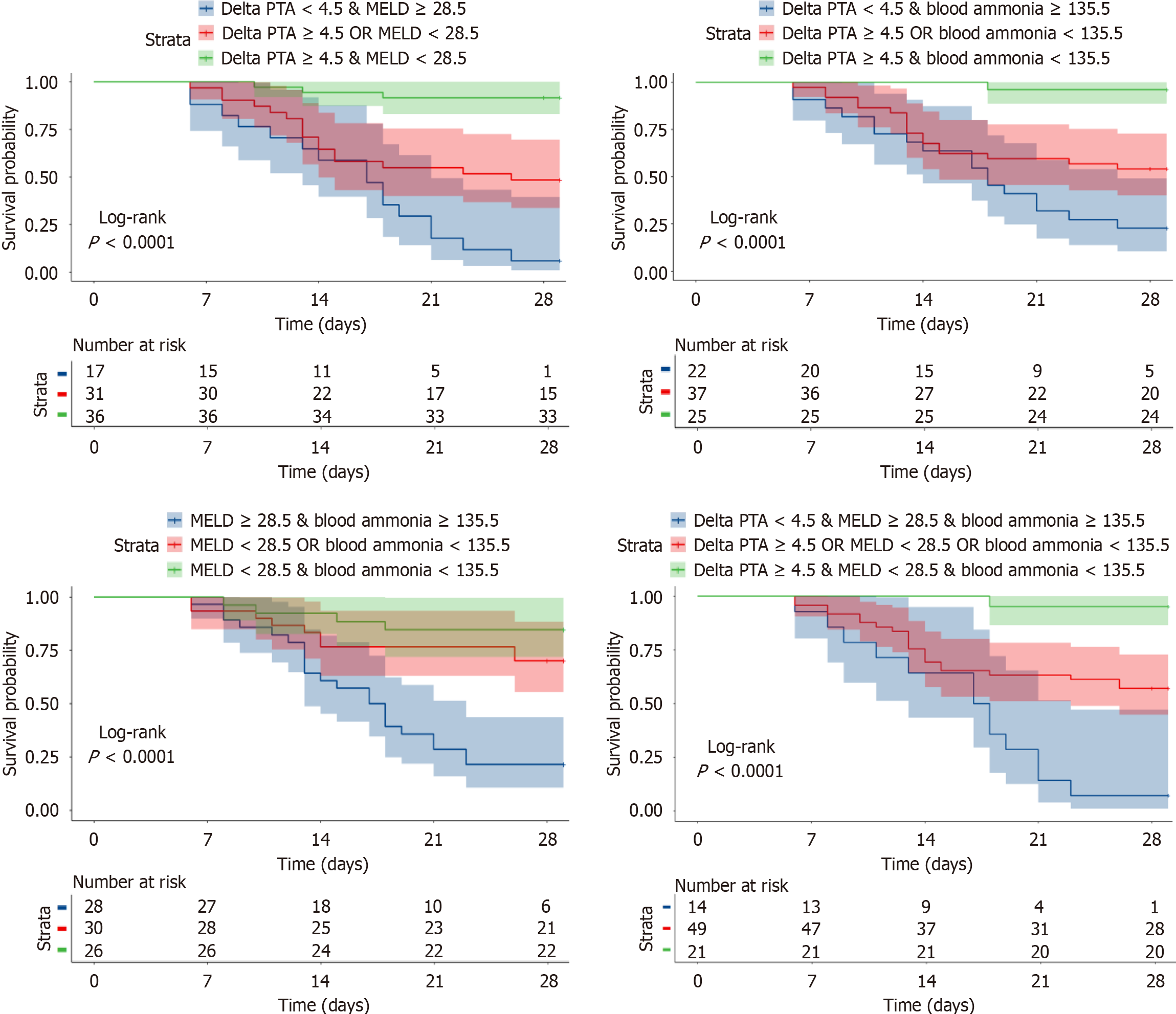
The optimal cut-off for ΔPTA was 4.5%, demonstrating the highest predictive performance, with an area under the receiver operating characteristic (AUROC) of 0.83 (sensitivity 85.71%, specificity 65.7%). This was followed by the baseline MELD score (cut-off of 28.5; AUROC of 0.78; sensitivity of 79.59%, and specificity of 71.43%), baseline PTA (cut-off of 22.85%; AUROC of 0.76; sensitivity of 55.1%, and specificity of 88.57%), and blood ammonia concentration (cut-off of 135.5 μg/dL; AUROC of 0.69; sensitivity of 53.06%, and specificity of 80.0%), as detailed in Figure 3 and Supple
Predictive accuracy was further improved by combining ΔPTA, MELD, and blood ammonia. In two-indicator models, patients meeting both favourable cut-offs had a GC response rate of 85%, whereas those meeting neither cut-off were 5.88%, as shown in Supplementary Table 3 and Figure 5. The three-indicator model provided even finer stratification: Patients who fulfilled all three criteria (ΔPTA ≥ 4.5%, MELD < 28.5, and blood ammonia concentration < 135.5 μg/dL) had a response rate of 95.24%, compared with only 7.14% for those who did not meet all three criteria, as shown in Supplementary Table 3. After bootstrap internal validation, the optimism-corrected AUC was 0.826, with a corrected sensitivity of 69.1% and specificity of 85.8%. The apparent performance (AUC = 0.977) was likely over-optimistic due to cutoff optimization within the same dataset, a known limitation of such approaches. The validated performance (AUC = 0.826) still represents good discrimination and supports the clinical utility of this triple-indicator model. Therefore, this triple-indicator model more precisely identifies patients with a low likelihood of benefit, offering greater clinical utility.
Although ALF has a low global incidence, it is characterized by rapid disease progression, extremely high early mortality, and an unpredictable clinical prognosis. Current management strategies-including hepatoprotective agents, plasma exchange, haemodiafiltration, and LT-aim to improve survival. However, these approaches (with the exception of LT) yield limited therapeutic benefits. Prior to the widespread adoption of LT, ALF was associated with a mortality rate of 80%-85%[8]. In recent years, advances in intensive care management have substantially improved ALF survival[2]. Even so, approximately half of patients still die without access to emergency LT, underscoring the critical role of LT in ALF management. However, LT has notable limitations: It is a costly intervention requiring major surgery and lifelong immunosuppression. Perioperative care for patients with ALF is particularly challenging, and their survival rates remain consistently lower than those of patients undergoing elective LT. Compounding these issues, organ donor shortages further restrict access to LT for the majority of patients with ALF[6].
Recent research has revealed that sustained and extensive hepatic inflammation is among the core pathological mechanisms driving the development and progression of ALF[9], providing a theoretical rationale for the use of GCs to suppress excessive immune-mediated inflammation. A prior study demonstrated that early GC administration can inhibit immune response-induced hepatocellular injury and microvascular dysfunction, thereby preserving liver regenerative capacity[10]. However, the clinical value of GCs in the management of patients with ALF remains controversial. Multiple randomized controlled trials (RCTs) conducted in the 20th century failed to confirm that GCs improve the survival of patients with ALF[11-13]. Similarly, earlier RCTs and observational studies from the United States and Europe also reported no significant survival benefit with GC use[11,13]. In contrast, our study revealed that GCs significantly improved the 28-day survival rate of patients with ALF, which aligns with findings from a Japanese study[5]. Notably, our study included a relatively larger sample size, whereas the United States and European RCTs referenced previously had a smaller sample (e.g., one key RCT enrolled only 40 patients[12]).
In our study, the main aetiologies of ALF were drug-induced (primarily herbal medicine) and hepatitis B virus infection. GC therapy was usually initiated after admission following the exclusion of contraindications, at a dosage of 0.5-1.0 mg/kg of body weight per day for an average duration of 3-7 days. In contrast, previous landmark studies (such as the 1993 EASL trial) primarily enrolled patients with viral hepatitis or ALF of indeterminate cause, and their GC regimens (timing, dosage, and duration) were not specified in detail. The low survival rates reported in those studies also suggest that their cohorts may have had more advanced disease. These factors, along with potential racial/ethnic differences, may partly explain the discrepancy between our findings and earlier results.
In recent years, several Japanese studies have consistently reported that early, high-dose, short-course GC therapy (e.g., 1000 mg/day methylprednisolone for 3 consecutive days) markedly increased survival of patients with ALF[5,14]. An analysis of our study population characteristics revealed that GC treatment was initiated within 1-3 days of hospital admission, corresponding to the early stage of ALF-which is consistent with the timing reported in the study by Yasui et al[15].
The immunosuppressive effects of GC inevitably raise concerns regarding infection risk. However, evidence linking GC therapy to a higher incidence of infections in ALF remains inconclusive. For instance, Yasui et al[15] reported that the use of GCs did not significantly increase infection rates. Notably, in patients who developed infections, the interval between GC initiation and LT was prolonged, with a reported median time to infection onset of 15 days[15]. In our cohort study, compared to the non-GC group, patients receiving GC therapy had a significantly lower incidence of gas
This shortcourse approach aligns with the concept proposed by Hisanaga et al[16], who suggested that GCs could serve as a transitional or adjuvant therapy to sustain patients awaiting LT or spontaneous recovery, provided that treatment duration is minimized. These observations collectively suggest that the efficacy and safety of GC therapy in patients with ALF may critically depend on timely initiation and precise control of treatment duration. A “shortcourse, rapidreview” strategy appears to be a relatively safe and practical option. In summary, while the conflicting results in previous studies may be attributed to factors such as limited sample size, delayed GC initiation, or prolonged treatment, our findings highlight the importance of optimized treatment timing and duration in maximizing therapeutic benefit while mini
The higher DAMA rate in the GC group represents an important source of potential bias, as DAMA is a complex phenomenon influenced by multiple factors and should not be equated with disease severity. While the worst-case analysis yielded a P value of 0.059, the consistent direction of benefit across all analyses-with absolute survival differences ranging from 14.1% to 49.1%-suggests that the observed treatment effect is unlikely to be entirely artifactual. The finding under the most extreme assumption did not reach statistical significance, but when considered alongside the highly significant results in more plausible scenarios, a true treatment effect cannot be ruled out.
Determining the optimal timing for LT in patients with ALF poses a significant clinical challenge. ALF is a highly dynamic condition, and early outcome prediction is difficult because of rapid clinical evolution. Excessive delay in decision-making may preclude LT as the disease progresses, leading to preventable mortality. Conversely, excessively early transplantation can result in patients who might otherwise recover from medical therapy, such as unnecessary major surgery, lifelong immunosuppression, and substantial resource expenditure, and may deprive other suitable candidates for a graft. The clinical window for LT in patients with ALF is narrow and is typically limited to 2-5 days after admission. Missing this window can result in loss of the survival opportunity afforded by transplantation[6]. Therefore, early application of reliable prognostic models is critical for optimizing outcomes. The MELD score and HE grade are key indicators of liver failure severity and prognosis. A MELD score ≥ 35 or grade 4 HE generally predicts end-stage disease with a poor prognosis, largely independent of therapeutic intervention. Consistent with this, the team led by Zhao et al[17] reported extremely low short-term survival in patients with MELD ≥ 35 and HE grade 4, irrespective of steroid use. Notably, among patients with MELD scores of 25-35 and no HE or HE grades 1-3, survival was significantly greater in those receiving GC than in those who did not[17].
Our findings also underscore the significant prognostic value of the MELD score and blood ammonia level in de
In conclusion, the dynamic stratification model established in this study enables the early identification (by day 3) of GC responders. This approach may help avoid unnecessary prolonged GC therapy and its associated risks (e.g., infection) in nonresponders while simultaneously minimizing either futile LT or missed transplant opportunities due to delayed assessment. We propose that a “short-course, rapid-assessment” GC strategy coupled with an effective early evaluation protocol could be a key determinant of successful GC therapy in patients with ALF.
In summary, the results of this study support the role of early, short-term GC therapy in improving survival for a subset of patients with ALF. We further propose, for the first time, a dynamic decisionmaking model centred on day-3 ΔPTA. This model provides a practical framework for individualized immunomodulatory management in patients with ALF and holds significant potential for clinical translation.
Although our three-parameter model demonstrated excellent predictive ability (AUC of 0.977), its clinical imple
This study had several limitations. First, the single-centre retrospective design may have introduced selection bias. Second, the absence of external validation limits the generalizability of our predictive model. Third, residual confounding from unmeasured factors, including care setting variations and discharge against medical advice, cannot be excluded. Finally, given the lack of international consensus on the use of GCs in ALF, our results require validation in prospective multicentre studies.
GC treatment significantly increased the 28-day survival rate of patients with ALF without increasing the incidence of adverse events. ΔPTA, baseline MELD score, and baseline blood ammonia level are independent early predictors of GC treatment response. A multivariable prediction model incorporating these three indicators can accurately identify patients who are likely to benefit from GC therapy while also effectively screening highrisk nonresponders, who may then be prioritized for timely LT assessment.
We express our gratitude to our colleagues who provided us with assistance.
| 1. | Maiwall R, Kulkarni AV, Arab JP, Piano S. Acute liver failure. Lancet. 2024;404:789-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 106] [Article Influence: 53.0] [Reference Citation Analysis (3)] |
| 2. | Stravitz RT, Lee WM. Acute liver failure. Lancet. 2019;394:869-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 655] [Article Influence: 93.6] [Reference Citation Analysis (3)] |
| 3. | Ling S, Jiang G, Que Q, Xu S, Chen J, Xu X. Liver transplantation in patients with liver failure: Twenty years of experience from China. Liver Int. 2022;42:2110-2116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 4. | Ye C, Li W, Li L, Zhang K. Glucocorticoid Treatment Strategies in Liver Failure. Front Immunol. 2022;13:846091. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 5. | Fujiwara K, Yasui S, Yonemitsu Y, Mikata R, Arai M, Kanda T, Imazeki F, Oda S, Yokosuka O. Efficacy of high-dose corticosteroid in the early stage of viral acute liver failure. Hepatol Res. 2014;44:491-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Kumar R, Anand U, Priyadarshi RN. Liver transplantation in acute liver failure: Dilemmas and challenges. World J Transplant. 2021;11:187-202. [PubMed] [DOI] [Full Text] |
| 7. | Liver Failure and Artificial Liver Group; Chinese Society of Infectious Diseases; Chinese Medical Association; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. [Guideline for diagnosis and treatment of liver failure]. Zhonghua Gan Zang Bing Za Zhi. 2019;27:18-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 64] [Reference Citation Analysis (1)] |
| 8. | Bernuau J, Rueff B, Benhamou JP. Fulminant and subfulminant liver failure: definitions and causes. Semin Liver Dis. 1986;6:97-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 392] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 9. | Wu Z, Han M, Chen T, Yan W, Ning Q. Acute liver failure: mechanisms of immune-mediated liver injury. Liver Int. 2010;30:782-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 227] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 10. | Fujiwara K, Hida S, Yasui S, Yokosuka O, Oda S. Corticosteroid might reduce serum levels of pro-inflammatory cytokines in fulminant hepatitis: A case series. Hepatol Res. 2018;48:106-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | Gregory PB, Knauer CM, Kempson RL, Miller R. Steroid therapy in severe viral hepatitis. A double-blind, randomized trial of methyl-prednisolone versus placebo. N Engl J Med. 1976;294:681-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 76] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 12. | Randomised trial of steroid therapy in acute liver failure. Report from the European Association for the Study of the Liver (EASL). Gut. 1979;20:620-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 83] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 13. | Karkhanis J, Verna EC, Chang MS, Stravitz RT, Schilsky M, Lee WM, Brown RS Jr; Acute Liver Failure Study Group. Steroid use in acute liver failure. Hepatology. 2014;59:612-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 146] [Article Influence: 12.2] [Reference Citation Analysis (4)] |
| 14. | Kotoh K, Enjoji M, Nakamuta M, Yoshimoto T, Kohjima M, Morizono S, Yamashita S, Horikawa Y, Yoshimitsu K, Tajima T, Asayama Y, Ishigami K, Hirakawa M. Arterial steroid injection therapy can inhibit the progression of severe acute hepatic failure toward fulminant liver failure. World J Gastroenterol. 2006;12:6678-6682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Yasui S, Fujiwara K, Haga Y, Nakamura M, Mikata R, Arai M, Kanda T, Oda S, Yokosuka O. Infectious complications, steroid use and timing for emergency liver transplantation in acute liver failure: analysis in a Japanese center. J Hepatobiliary Pancreat Sci. 2016;23:756-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Hisanaga T, Hidaka I, Sakaida I, Nakayama N, Ido A, Kato N, Takikawa Y, Inoue K, Shimizu M, Genda T, Terai S, Tsubouchi H, Takikawa H, Mochida S; Intractable Hepato-Biliary Disease Study Group Of Japan. Analysis of the safety of pretransplant corticosteroid therapy in patients with acute liver failure and late-onset hepatic failure in Japan. JGH Open. 2021;5:428-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Zhao B, Zhang HY, Xie GJ, Liu HM, Chen Q, Li RF, You JP, Yang S, Mao Q, Zhang XQ. Evaluation of the efficacy of steroid therapy on acute liver failure. Exp Ther Med. 2016;12:3121-3129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Kumar R, Shalimar, Sharma H, Goyal R, Kumar A, Khanal S, Prakash S, Gupta SD, Panda SK, Acharya SK. Prospective derivation and validation of early dynamic model for predicting outcome in patients with acute liver failure. Gut. 2012;61:1068-1075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 19. | Kuwano A, Okui T, Kohjima M, Kurokawa M, Goya T, Tanaka M, Aoyagi T, Takahashi M, Imoto K, Tashiro S, Suzuki H, Fujita N, Ushijima Y, Ishigami K, Tokunaga S, Kato M, Ogawa Y. Transcatheter arterial steroid injection therapy improves the prognosis of patients with acute liver failure. Medicine (Baltimore). 2023;102:e33090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |