Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.117367
Revised: January 27, 2026
Accepted: February 13, 2026
Published online: March 27, 2026
Processing time: 111 Days and 5 Hours
Autoimmune liver diseases, including autoimmune hepatitis, primary biliary cholangitis, and primary sclerosing cholangitis, have traditionally been regarded as disorders of young or middle-aged adults. However, the progressive aging of the global population has revealed a substantial and increasing number of cases diagnosed at ever older ages. Older adults frequently present with atypical or non-specific symptoms, are more likely to be diagnosed at advanced stages, and often exhibit a distinct biochemical profile compared with younger patients. In particular, the recent study by Delgado et al underscores these differences in late-onset autoimmune hepatitis, demonstrating a higher rate advanced fibrosis at presentation and diagnostic delay in elderly individuals. This editorial aims to highlight these observations, as these characteristics create unique diagnostic challenges and may negatively influence long-term outcomes. Treatment remains effective with standard first-line regimens; however, therapeutic decisions must be carefully individualized due to comorbidities, polypharmacy, and an increased vulnerability to treatment-related adverse events. Recognizing age-specific clinical patterns and adapting management strategies accordingly is essential to optimize care in this growing and frequently overlooked patient population.
Core Tip: Autoimmune liver diseases are increasingly relevant in older adults, with changing epidemiology and rising incidence, including presentation peaks in this age group. Clinical manifestations range from unusual presentations, acute liver failure, and acute hepatitis to more common indolent forms with minimal symptoms. Serologic testing does not differ from younger patients, but due to comorbidities, liver biopsy remains essential. First-line treatment generally does not change and is often effective, although older adults are more vulnerable to steroid-related complications and polypharmacy. Liver transplantation remains an option. However, this population is still understudied, and future research should incorporate comprehensive geriatric assessment.
- Citation: Hernández-Almonacid PG, Espejo-Amado JS, Marín-Quintero X. Autoimmune liver diseases in older adults: Clinical challenges and management considerations. World J Hepatol 2026; 18(3): 117367
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/117367.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.117367
This editorial refers to "Impact of age on autoimmune hepatitis: A comparative study of patients diagnosed before and after sixty" by Delgado et al, 2025; https://doi.org/10.4254/wjh.v17.i12.110312.
Autoimmune liver diseases (AILD) are a group of disorders characterized by immune-mediated responses directed against hepatocytes and cholangiocytes[1]. This group included patients with autoimmune hepatitis (AIH), primary biliary cholangitis (PBC), and primary sclerosing cholangitis (PSC). Recently, overlapping syndromes and immunoglobulin G4 (IgG4)-related cholangitis have been recognized as additional entities on this spectrum. These diseases were classically considered conditions in young or middle-aged adults. However, the diagnosis of AILD in individuals aged > 60 years has significantly increased[2].
Demographic shifts and population ageing are global phenomena. By 2030, one in six individuals worldwide will be aged ≥ 60 years[3]. Older adults constitute a distinct clinical group with different disease behaviors, therapeutic needs, and outcomes compared with younger patients. They often present with a more insidious yet aggressive course, con
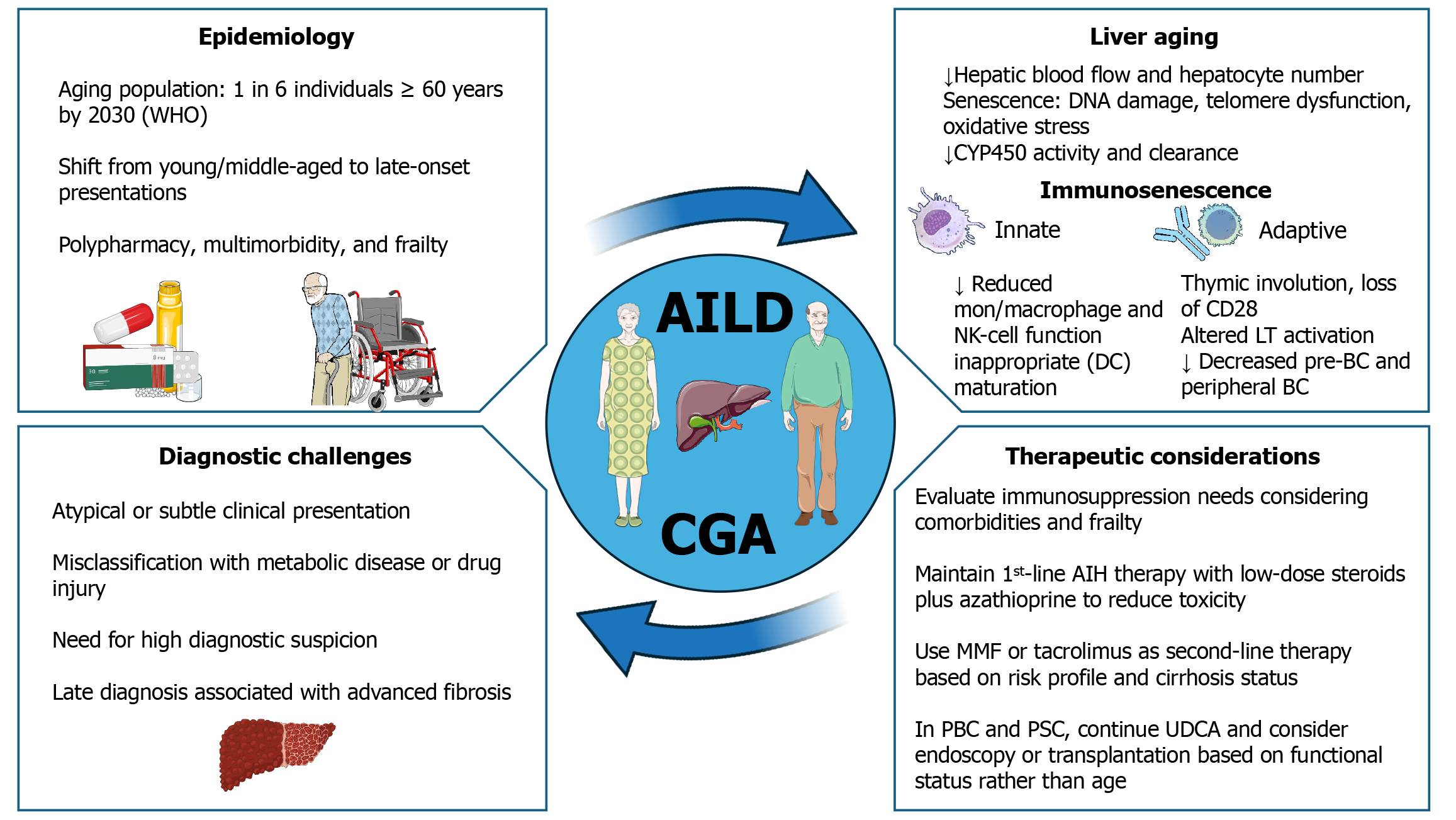
Delgado et al[9] provided important insights by identifying key differences in AIH among older adults. These findings offer a timely basis for examining the clinical challenges of AILD in elderly patients and outline the management considerations required for this growing population (Figure 1).
The study by Delgado et al[9] is a single-center retrospective cohort of adult patients with biopsy-confirmed AIH and ≥ 1 year of clinical follow-up. This is one of the largest series of studies conducted in Latin America. By analyzing de
The final cohort included 97 patients, approximately 40% of whom were aged > 60 years, highlighting the growing burden of this disease in the aging population. Older adults have a higher prevalence of metabolic and cardiovascular comorbidities, a more silent presentation with fewer classical symptoms, and lower transaminase and bilirubin levels. Despite the attenuated clinical profiles, these patients frequently present with ascites and advanced fibrosis. Importantly, this greater baseline severity did not result in a poorer treatment response, as older adults achieved biochemical outcomes comparable to those of younger patients, despite lower initial corticosteroid doses[9].
Although this study has limitations related to its retrospective, single-center design, moderate sample size, short follow-up period, and absence of key geriatric measures such as polypharmacy and frailty, it offers notable strengths. Considering the scarcity of regional data, the study population is valuable, and the diagnostic rigor is reinforced by the inclusion of exclusively biopsy-confirmed cases.
Therefore, this study makes a meaningful contribution by emphasizing the clinical and diagnostic features of late-onset AIH. These findings highlight the need for increased diagnostic suspicion of indolent presentations, frequent presence of advanced fibrosis at diagnosis in older adults, and influence of comorbidities that complicate therapeutic decision-making and outcomes.
Reports on AILD in older adults are limited; however, recent systematic reviews have shown an increasing incidence of AILD in older age groups[2,10]. Globally, AIH has an incidence of 1.37 and prevalence of 17.4 per 100000 individuals[11]. For PBC, pooled estimates reported an incidence of 1.76 and prevalence of 14.60 per 100000 individuals[12]. PSC data are scarce, with one large study reporting an incidence of 0.9 per 100000 individuals[13]. Age-specific patterns consistently revealed that older adults were most affected. The peak incidence of PBC occurs between the ages of 60 years and 79 years[12], mirroring AIH, in which presentation is most common after 60 years of age[11]. Overall, these data indicate a shift toward late-onset disease, highlighting the growing impact of population aging on AILD epidemiology.
Aging is a dynamic process that affects the tissues and organs at varying rates. Although the liver is believed to undergo minimal age-related changes, age-associated cellular and morphological alterations can lead to functional decline[14]. Senile atrophy, driven by reduced regenerative capacity, results in a 24%-37% decrease in liver weight along with re
Immunosenescence refers to the functional and quantitative remodeling of innate and adaptive immune cell popu
The primary challenge in the timely diagnosis of AILD in older adults is the wide variability in the clinical and serological phenotypes across populations. A systematic review by Chen et al[18] found that older adults are more often asy
Although AIH often has an indolent course, older adults may present with severe acute disease, with either de novo inflammation or exacerbation of an unrecognized chronic disease. In some studies, 41%-71% of older patients presented acutely, a pattern easily mistaken for drug-induced hepatotoxicity considering the high burden of polypharmacy[21]. The unusual symptoms include painless jaundice[22,23], hepatic encephalopathy[24], and acute liver failure[25]. Atypical features of PBC in older adults include initially normal liver test results[26], gastric antral vascular ectasia[27], recurrent hypoglycemic coma[28], and hemorrhage from a cholesterol embolus in the polyp[29]. These observations indicate that older adults may exhibit a broad and diverse clinical spectrum, reinforcing the need for a careful diagnostic evaluation.
The diagnostic criteria for AILD were the same for both the older and younger patients. In AIH, the autoantibody profile is similar across age groups, although the interpretation may be complicated by natural age-related autoantibodies that appear at low titers, have low affinity, and exhibit broad reactivity. Most older adults with AIH have type I disease, marked by ANA and anti-SM positivity with hypergammaglobulinemia, whereas < 4% have anti-LKM antibodies[21]. No age-specific serological patterns were identified for PBC or PSC. However, in PSCs, distinguishing the disease from IgG4-related sclerosing cholangitis, which is more common in older adults and more responsive to steroids, is essential, making serum IgG4 measurement a key step in evaluation[30].
The diagnostic landscape in older adults is complicated by the high prevalence of coexisting conditions such as metabolic dysfunction-associated steatotic liver disease (MASLD), viral hepatitis, drug-induced liver injury, and alcoholic liver disease, all of which commonly form part of the differential diagnosis in this population[4]. MASLD creates a complex ‘metabolic milieu’ that complicates clinical and biochemical monitoring; steatohepatitis may cause persistent aminotransferase elevations, potentially masking the biochemical response to immunosuppression or leading to an overestimation of AILD activity[31]. Histologically, this presents a diagnostic challenge as features such as macro
Overlapping phenomena between the different entities of AILD are not uncommon, with AIH-PBC Overlap syndrome being the most common type. Despite the lack of extensive epidemiological information, in some series, the mean age at presentation was 61 years, with a female predominance[34,35]. Furthermore, the age-related increase in extrahepatic autoimmune diseases poses an additional challenge to the diagnostic specificity. Sjögren’s syndrome and autoimmune thyroid disease are the most common; however, systemic lupus erythematosus, rheumatoid arthritis, ulcerative colitis, and Crohn’s disease are also frequent and may arise concurrently or even occur years following the diagnosis, reflecting the dynamic nature of the disease[31,36,37]. These diseases often generate non-specific autoantibody titers, reducing the positive predictive value of serological markers for AILD. This serological noise underscores the importance of a liver biopsy as an essential tool for establishing an accurate AILD diagnosis[4]. Notably, a wide range of metabolic and cardiovascular comorbidities shape the clinical landscape, influencing both the prognostic outlook and selection of therapeutic interventions.
AILD is associated with increased cancer risk; in PSC, the likelihood of cholangiocarcinoma, gallbladder cancer, and colon cancer is significantly elevated, whereas in AIH and PBC, the risk of hepatocellular carcinoma, especially within cirrhosis, is higher than that in the general population, reinforcing the need for appropriate oncologic surveillance[2,38].
Evidence comparing clinical outcomes across age groups in patients with AILD remains fragmented. Table 1 summarizes current studies evaluating these disparities, emphasizing the diagnostic challenges and phenotypic variations prevalent in the geriatric population[39-51].
| Ref. | Country | Total patients/elderly (%) | Comorbidities in the elderly | Biochemical profile | Serologic markers | Treatment response | Other characteristics |
| Autoimmune hepatitis | |||||||
| Czaja and Carpenter[39], 2006 | United States | n = 205/47 (23) > 60 years | AT (23%), RA (4%), SS (2%), SLE (2%) | No significant differences | No significant differences | 59% vs 61%; less steroid failure (5% vs 24%, P = 0.03) | Cirrhosis more common (33% vs 10%, P = 0.03); HLA-DR4 more frequent (47% vs 13%, P = 0.003) |
| Miyake et al[40], 2007 | Japan | n = 160/34 (21) > 65 years | 6 (18): CT, UC, GD, SS | Lower albumin; other labs similar | ANA/ASM 97% vs 94% (P = 0.53) | 95% vs 89% (P = 0.41) | Type II HLA: Frequency of DR2-DR4 was similar. Cirrhosis (F3-F4) more common in elderly (53% vs 30%, P = 0.014) |
| Morii et al[41], 2017 | Japan | n = 71/28 (39) > 70 years | SS, RA, CT | Lower albumin and cholesterol | Higher IgG and ANA titers (P = 0.02) | Both groups achieved remission | Fibrosis score similar |
| Baven-Pronk et al[42], 2018 | Netherlands | n = 359/73 (20.3) > 60 years | Higher autoimmune comorbidity (33% vs 20%, P < 0.05): CT, CD, UC, RA, SS, SLE | Lower ALAT (430 vs 670 IU/L, P < 0.001) | No significant differences | 81% vs 76% (P = 0.368) | Fewer acute onsets (10%), less jaundice (25%); no difference in cirrhosis |
| Díaz Ramírez et al[43], 2019 | Colombia | n = 214/29 (13.5) > 65 years | HBP (34.5%), dyslipidemia (20.7%), CVD (17.2%) | No significant differences | No significant differences | 83.9% vs 100% (P = 0.022) | Cirrhosis more common (55% vs 33%, P = 0.024); 1 elderly transplanted |
| Sonthalia et al[44], 2020 | India | n = 155/73 (20) > 60 years | DM, RA, CT | Higher ALAT and creatinine (P < 0.05) | ASM more common (45.5% vs 25.4%, P < 0.05) | 65.6% vs 51.5% | ACLF more frequent (39.4% vs 13.9%, P = 0.0024); cirrhosis more frequent (75.7% vs 56.6%, P = 0.045) |
| Dalekos et al[45], 2021 | Greece | n = 234/25 (10) > 70 years | CVD (72%), DM (32%) | Lower ALAT (P < 0.05) | No significant differences | 83.7% vs 88.9% (P = 0.566) | Cirrhosis more common (48% vs 27.3%, P = 0.03); no differences in presentation |
| Primary biliary cholangitis | |||||||
| Newton et al[46], 2000 | United Kingdom | n = 1023/397 (39) > 65 years | Not reported | No significant differences | No significant differences | Not evaluated | Similar clinical features; higher mortality (59% vs 33%, P = 0.001) |
| Muratori et al[47], 2008 | Italy | n = 234/49 (25) > 65 years | Not reported | Lower ALAT/ASAT (P = 0.0024) | No significant differences | Not evaluated | Elderly less symptomatic at diagnosis (26%, P = 0.02); male sex higher (40%, P = 0.002) |
| Franceschet et al[48], 2016 | Italy | n = 376/62 (16) > 65 years | Not reported | Lower ALAT, platelets, albumin | No significant differences | UDCA response similar (56.6% vs 67.7%, P = NS) | Higher mortality (41.9% vs 19.7%, P < 0.001) |
| Primary sclerosing cholangitis | |||||||
| Hirano et al[49], 2008 | Japan | n = 28/10 (25) > 50 years | UC | Not evaluated | Higher IgE in older group | Fewer liver transplants | Less CCA and less liver failure |
| Tanaka et al[50], 2014 | Japan | n = 197 PSC + 43 IgG4SC/98 (49) > 50 years | IBD | No significant differences | IgG and IgG4 similar in elderly vs young; IgG4-SC had higher IgG/IgG4; IgA higher in elderly PSC (P < 0.001) | Similar medical treatment; similar rate of stenting/balloon dilation; similar transplantation rates | Similar symptoms at presentation; 5-year survival 54.7% in elderly |
| Rupp et al[51], 2018 | Germany | n = 215/32 (15) > 50 years | IBD | Lower ALAT/ASAT (P = 0.03/0.05) | No significant differences | 6 elderly transplanted | CCA more frequent in elderly (12.5% vs 3.3%, P = 0.02); late onset associated with mortality; poorer transplant-free survival (10.5 years vs 20.8 years) |
The treatment of patients with AILD poses a significant challenge because of the frequent coexistence of multiple comorbidities, the high burden of polypharmacy, and the resulting risk of drug interactions, toxicity, and adverse effects. Additional difficulties arise in selecting and maintaining immunosuppressive therapy due to complications such as corticosteroid-induced osteoporosis[52], steroid-related psychosis, cognitive decline, delirium[53], increased cardio
Adding to this challenge is the scarcity of age-specific evidence, partly because of the exclusion of older adults from clinical trials and conditions more prevalent in this age group, such as chronic kidney disease, osteoporosis, cardio
Furthermore, osteoporosis is of particular interest. A key characteristic of glucocorticoid-induced osteoporosis is the rapid loss of bone mass (driven by increased bone resorption and reduced bone formation) together with an increased risk of fractures. It is estimated that within the first 3-6 months of therapy, the fracture risk is already significantly elevated, with reported incidences of up to 5.1% for vertebral fractures and 2.5% for non-vertebral fractures[59-61].
Three key points must be considered. First, the fracture risk is dose-dependent; even low doses such as 2.5 mg/day of prednisone are associated with an increased fracture risk[62]. Second, in patients receiving glucocorticoids, fractures tend to occur at higher bone mineral density (BMD) values compared with individuals not exposed to these medications[63-65], moreover, the underlying autoimmune disease itself should not be overlooked, as it contributes to a pro-inflammatory and pro-resorptive cytokine milieu and may be associated with malnutrition, weight loss, reduced functional capacity, and impaired mobility (factors that further predispose to the development of osteoporosis). Third, fracture risk assessment using the Fracture Risk Assessment Tool (FRAX) score, the main tool currently used to estimate fracture probability, tends to underestimate the risk when the total hip BMD is used, given that vertebral fractures are the predominant fractures in glucocorticoid users. These patients often exhibit spine-hip discordance in BMD; therefore, some authors have proposed adjustments to improve fracture risk estimation in this population[66]. Most clinical guidelines and expert consensus statements advocate the earlier initiation of therapy in high-risk patients. For example, the European Calcified Tissue Society and the International Osteoporosis Foundation recommend initiating treatment in patients with a ≥ 20% probability of fracture, a history of fragility fracture, those receiving prednisone doses ≥ 7.5 mg/day (or equivalent), or those with a T-score ≤ -1.5[67,68]. In contrast, the American College of Rheumatology recommends treatment initiation in patients with a history of fragility fracture, a T-score ≤ -2.5, a ≥ 10% probability of major osteoporotic fracture or ≥ 1% probability of hip fracture, and those receiving high-dose glucocorticoids, although specific dose thresholds are not clearly defined[69]. From our perspective, patients receiving glucocorticoids for > 3-6 months may benefit from earlier interventions. A T-score ≤ -1.5 appears to be a more appropriate threshold-considering the higher BMD at which fractures occur in this population-and a ≥ 10% risk of major osteoporotic fracture should be con
AIH is the condition under which the greatest amount of data is available for older adults. Most studies have employed standard corticosteroid therapy with or without azathioprine, as shown in a systematic review by Durazzo et al[70], in which the use of newer therapies was documented only in case series and reports that were excluded from formal analysis. This review evaluated 239 elderly patients, 151 of whom were candidates for immunosuppressive treatment. Of these, 87% received standard therapy, whereas the remaining 13% were treated with mycophenolate mofetil (MMF) in a single study[71]. One study has examined the effects of ursodeoxycholic acid (UDCA)[41].
The rates of complete or partial remission with standard therapy range from 90% to 95%[44,72,73], and are comparable to those reported in the general population[74]. Therapeutic failure was less frequent in older than in younger patients (9.1% vs 28.2%, P = 0.041; 5% vs 24%, P = 0.03)[39,75]. In the MMF study, the combined complete and incomplete response rates were 93.6%; however, this cohort included a higher proportion of patients with cirrhosis. Notably, 75% of the patients remained in remission for two years after treatment withdrawal. Fortunately, favorable therapeutic responses have been reported across different ethnic groups, with similar outcomes observed in Chinese and Caucasian cohorts[39,75].
Some studies have suggested lower relapse rates in older adults, such as a study by Al-Chalabi et al[73] (70% vs 42%, P = 0.002). Although standard therapy is highly effective, UDCA demonstrated a lower response rate, with only 50% of the patients achieving remission in a study by Morii et al[41].
In cases of incomplete response, guidelines recommend increasing corticosteroid and azathioprine doses before switching to second-line therapy[58,76], although this poses a challenge in older adults due to the higher risk of adverse effects and intolerance; therefore, we suggest considering the use of second-line agents rather than using higher doses of prednisolone. If second-line agents are required, options include MMF and tacrolimus. Although a few studies have specifically targeted older adults or provided geriatric subgroup analyses, evidence from the general population suggests a greater efficacy of tacrolimus in achieving remission in partial responders or in those with treatment failure (68.9% vs 59.6%), with comparable rates of adverse events (25.5% for MMF and 24.1% for tacrolimus). The most frequent side effects include neurotoxicity, gastrointestinal symptoms with tacrolimus, leukopenia, and gastrointestinal symptoms with MMF[77].
Regarding orthotopic liver transplantation in AIH, a European study reported five- and 10-year survival rates > 70%. However, age-stratified analyses showed better outcomes in younger recipients (78% in the 18-34 years group vs 61% in those aged > 50 years). This difference was attributed to the higher incidence of post-transplant infections in older patients (hazard ratio, 1.8; P = 0.002)[78]. Data from the European Liver Transplant Registry (1988-2016) showed that autoimmune cirrhosis accounted for 2% of all liver transplantation indications, totaling 2929 cases. The 10-year graft survival rate was 60%, and the patient survival rate was 68%. If the analysis were restricted to the past 15 years, these rates would have improved to 63% and 72%, respectively[79]. Age-stratified survival demonstrated a clear gradient; 75% for adults aged 18-45 years, 71% for those aged 46-60 years, 65% for those aged 60-70 years, and 60% for those aged > 70 years. Notably, the proportion of older transplant recipients has increased substantially, with adults aged > 60 years representing < 5% of liver transplant recipients in the 1980s and currently accounting for > 30%.
Regarding the treatment of PSC, a Japanese study involving 187 patients found no significant differences in the use of medical therapies between older and younger adults. In older and younger patients, UDCA was used in 76% and 77% of patients, bezafibrate in 19% and 20%, and prednisolone in 16% and 24% of patients, respectively. Endoscopic in
The Comprehensive Geriatric Assessment (CGA) is a multidimensional and dynamic appraisal process designed to assess the medical, functional, cognitive, and social capabilities and limitations of older adults. Rather than relying on chronological age, CGA serves as a framework to quantify biological reserves and predict critical outcomes, such as treatment-related toxicity and all-cause mortality[80,81]. CGA utilizes various domain-specific scales, including frailty indices such as the Liver Frailty Index and the Clinical Frailty Scale, which serve as the most robust predictors of mortality. Other essential domains include disease burden, typically assessed using the Charlson Comorbidity Index[81]; multidimensional tools such as the Multidimensional Prognostic Index integrate these parameters, offering superior prognostic precision by capturing the complex interplay between liver disease and geriatric status[80]; these assessments enable the classification of patients into fit (robust), vulnerable (pre-frail), and frail, offering a robust framework for individualized management and risk-stratified clinical decisions[82].
Establishing a CGA as a transversal pillar from the initial evaluation is crucial as it streamlines the entire clinical pathway in AILD. By stratifying patients into operational subgroups, this framework allows robust (fit) individuals to undergo standard, comprehensive diagnostic and therapeutic regimens, while identifying vulnerable (pre-frail) patients who may benefit from early, targeted interventions to preserve functional reserve. Conversely, in the frail population, the CGA prompts a critical reconsideration of aggressive management, shifting the focus toward safety, quality of life, and the avoidance of procedural futility[83,84]. This tailored approach informs high-stakes decisions, ranging from the intensity of immunosuppressive therapy to candidacy for liver transplantation, and facilitates the implementation of prehabilitation strategies aimed at improving or even reversing the frailty phenotype through nutritional and physical optimization[85]. Ultimately, efficient resource utilization can be achieved by minimizing unnecessary interventions and preventing treatment-related complications[81].
Despite the growing interest in AILD among older adults, important knowledge gaps persist. Most studies were retrospective and single-center, limiting external validity, and few systematically evaluated the influence of autoimmune, metabolic, or cardiovascular comorbidities on prognosis, fibrosis progression, or treatment response. The age cutoffs used to define older adults vary widely, further complicating the interpretation of the findings.
Dedicated cohorts and clinical trials with adequate representation of patients aged ≥ 70 years are needed, along with comprehensive geriatric assessments incorporating functional status, frailty, and polypharmacy, which are largely missing from the current research. Early diagnostic biomarkers, immune profiles related to immunosenescence, and the effects of frailty on outcomes should be prioritized. Evidence on liver transplantation and the eligibility criteria in older adults remains limited, representing an important area for future studies.
The peak incidence of AILD in patients aged > 60 years represents a significant epidemiological shift that demands a comprehensive approach. Clinicians must maintain a high index of suspicion because subtle clinical presentations often mask advanced underlying diseases. Considering the high prevalence of comorbidities, a low threshold for liver biopsy is essential to ensure diagnostic accuracy before therapy is initiated. Management must transition from age-based decisions to a frailty-centered framework, where integrating CGA is paramount for risk stratification, outcome prediction, and prevention of treatment-related futility. Furthermore, immunosuppressive therapy cannot be managed in isolation and must be strictly coupled with proactive preventive strategies that focus on cardiovascular optimization, bone health preservation, and the monitoring of infectious or cognitive complications. Ultimately, while standard therapies are effective, clinical success in older adults is measured not only by biochemical remission, but also by the preservation of functional reserve and quality of life.
| 1. | Invernizzi P. Geoepidemiology of autoimmune liver diseases. J Autoimmun. 2010;34:J300-J306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 69] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 2. | Trivedi PJ, Hirschfield GM. Recent advances in clinical practice: epidemiology of autoimmune liver diseases. Gut. 2021;70:1989-2003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 177] [Article Influence: 35.4] [Reference Citation Analysis (1)] |
| 3. | Noto S. Perspectives on Aging and Quality of Life. Healthcare (Basel). 2023;11:2131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 74] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 4. | Czaja AJ. Autoimmune liver disease. Curr Opin Gastroenterol. 2007;23:255-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 5. | Nicholson K, Liu W, Fitzpatrick D, Hardacre KA, Roberts S, Salerno J, Stranges S, Fortin M, Mangin D. Prevalence of multimorbidity and polypharmacy among adults and older adults: a systematic review. Lancet Healthy Longev. 2024;5:e287-e296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 141] [Article Influence: 70.5] [Reference Citation Analysis (0)] |
| 6. | Song M, Camargo MC, Derkach A, Rabkin CS, Engels EA. Associations Between Autoimmune Conditions and Gastric Cancer Risk Among Elderly Adults in the United States. Am J Gastroenterol. 2022;117:486-490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 7. | McGee EE, Castro FA, Engels EA, Freedman ND, Pfeiffer RM, Nogueira L, Stolzenberg-Solomon R, McGlynn KA, Hemminki K, Koshiol J. Associations between autoimmune conditions and hepatobiliary cancer risk among elderly US adults. Int J Cancer. 2019;144:707-717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Czaja AJ. Special clinical challenges in autoimmune hepatitis: the elderly, males, pregnancy, mild disease, fulminant onset, and nonwhite patients. Semin Liver Dis. 2009;29:315-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 9. | Delgado J, Fuentes M, Simian D, Poniachik J, Urzúa Á. Impact of age on autoimmune hepatitis: A comparative study of patients diagnosed before and after sixty. World J Hepatol. 2025;17:110312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (3)] |
| 10. | Wakil A, Muzahim Y, Awadallah M, Kumar V, Mazzaferro N, Greenberg P, Pyrsopoulos N. Trends of autoimmune liver disease inpatient hospitalization and mortality from 2011 to 2017: A United States nationwide analysis. World J Hepatol. 2024;16:1029-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Lv T, Li M, Zeng N, Zhang J, Li S, Chen S, Zhang C, Shan S, Duan W, Wang Q, Wu S, You H, Ou X, Ma H, Zhang D, Kong Y, Jia J. Systematic review and meta-analysis on the incidence and prevalence of autoimmune hepatitis in Asian, European, and American population. J Gastroenterol Hepatol. 2019;34:1676-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 12. | Lv T, Chen S, Li M, Zhang D, Kong Y, Jia J. Regional variation and temporal trend of primary biliary cholangitis epidemiology: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2021;36:1423-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 115] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 13. | Bambha K, Kim WR, Talwalkar J, Torgerson H, Benson JT, Therneau TM, Loftus EV Jr, Yawn BP, Dickson ER, Melton LJ 3rd. Incidence, clinical spectrum, and outcomes of primary sclerosing cholangitis in a United States community. Gastroenterology. 2003;125:1364-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 364] [Cited by in RCA: 302] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 14. | Michielsen P, Vandewoude M. Liver diseases in the older adult. Acta Gastroenterol Belg. 2010;73:1-4. [PubMed] |
| 15. | Bruguera M. [Liver diseases in the elderly]. Gastroenterol Hepatol. 2014;37:535-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Radonjić T, Dukić M, Jovanović I, Zdravković M, Mandić O, Popadić V, Popović M, Nikolić N, Klašnja S, Divac A, Todorović Z, Branković M. Aging of Liver in Its Different Diseases. Int J Mol Sci. 2022;23:13085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 17. | Watad A, Bragazzi NL, Adawi M, Amital H, Toubi E, Porat BS, Shoenfeld Y. Autoimmunity in the Elderly: Insights from Basic Science and Clinics - A Mini-Review. Gerontology. 2017;63:515-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 103] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 18. | Chen J, Eslick GD, Weltman M. Systematic review with meta-analysis: clinical manifestations and management of autoimmune hepatitis in the elderly. Aliment Pharmacol Ther. 2014;39:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | Tajiri K, Shimizu Y. Liver physiology and liver diseases in the elderly. World J Gastroenterol. 2013;19:8459-8467. [PubMed] [DOI] [Full Text] |
| 20. | Karlsen TH, Folseraas T, Thorburn D, Vesterhus M. Primary sclerosing cholangitis - a comprehensive review. J Hepatol. 2017;67:1298-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 758] [Cited by in RCA: 655] [Article Influence: 72.8] [Reference Citation Analysis (2)] |
| 21. | Czaja AJ. Clinical features, differential diagnosis and treatment of autoimmune hepatitis in the elderly. Drugs Aging. 2008;25:219-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 22. | Querejeta A, Hormigo-Sánchez AI, Fernández-Alonso C, Martín-Sánchez FJ. [Hyperbilirubinemia asymptomatic as presentation of autoimmune hepatitis in an elderly patient]. Rev Esp Geriatr Gerontol. 2016;51:358-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 23. | Madhani K, Leventhal R. 3170 Autoimmune Hepatitis in an Elderly Patient. Am J Gastroenterol. 2019;114:S1697-S1698. [DOI] [Full Text] |
| 24. | Yang S, Zhang N, Li X, Li Y, Zhou L, Wei Y, Zhou K, Pan H, Kang L. A Case of Autoimmune Hepatitis Initially Manifesting as Hepatic Encephalopathy. Cureus. 2024;16:e62890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Aziz AA, Aziz MA, Mehta D, Rashid MH. Autoimmune Hepatitis: An Unusual Presentation. J Community Hosp Intern Med Perspect. 2024;14:95-97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Dash A, Kragel P. Primary biliary cirrhosis in an elderly man with normal liver tests. South Med J. 2009;102:1272-1274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 27. | Biswas SN, Elhence A, Agrawal V, Ghoshal UC. Gastric Antral Vascular Ectasia as the First Presentation of Primary Biliary Cholangitis. Cureus. 2022;14:e21676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 28. | Liu Y, Xu J, Chen Y, Wang L, Chen W, Shen J. Recurrent Hypoglycemic Coma Episodes Associated With Primary Biliary Cirrhosis. JCEM Case Rep. 2025;3:luaf030. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Noguerón García A, Hernán Calvo F, Viguera Garrido L, Abizanda Soler P. [Gastrointestinal bleeding secondary to cholesterol embolus polyp as the first manifestation of primary biliary cirrhosis in an older adult]. Rev Esp Geriatr Gerontol. 2017;52:288-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 30. | Seo N, Kim SY, Lee SS, Byun JH, Kim JH, Kim HJ, Lee MG. Sclerosing Cholangitis: Clinicopathologic Features, Imaging Spectrum, and Systemic Approach to Differential Diagnosis. Korean J Radiol. 2016;17:25-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 31. | Kilani Y, Aldiabat M, Sirilan KYT, Nasir AB, Madi MY, Syn WK. The Impact of Metabolic Dysfunction-Associated Steatotic Liver Disease on Autoimmune Hepatitis Outcomes: A Nationwide Analysis of 2,880 Records. Clin Transl Gastroenterol. 2025;16:e00912. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Dalekos GN, Gatselis NK, Zachou K, Koukoulis GK. NAFLD and autoimmune hepatitis: Do not judge a book by its cover. Eur J Intern Med. 2020;75:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (2)] |
| 33. | Cazzaniga G, Bolis F, Caime C, Piazza R, Žigutytė L, Kather JN, Pagni F, Invernizzi P, Carbone M, Gerussi A. Diagnosing Autoimmune Hepatitis: Histological Correlations and Emerging Technologies. Liver Int. 2025;45:e70377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Sohal A, Nikzad N, Kowdley KV. Overlap syndromes in autoimmune liver disease: a review. Transl Gastroenterol Hepatol. 2025;10:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 35. | Silveira MG, Talwalkar JA, Angulo P, Lindor KD. Overlap of autoimmune hepatitis and primary biliary cirrhosis: long-term outcomes. Am J Gastroenterol. 2007;102:1244-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 107] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 36. | Guo L, Zhou L, Zhang N, Deng B, Wang B. Extrahepatic Autoimmune Diseases in Patients with Autoimmune Liver Diseases: A Phenomenon Neglected by Gastroenterologists. Gastroenterol Res Pract. 2017;2017:2376231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 37. | Tunio NA, Mansoor E, Sheriff MZ, Cooper GS, Sclair SN, Cohen SM. Epidemiology of Autoimmune Hepatitis (AIH) in the United States Between 2014 and 2019: A Population-based National Study. J Clin Gastroenterol. 2021;55:903-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 38. | Sy AM, Ferreira RD, John BV. Hepatocellular Carcinoma in Primary Biliary Cholangitis. Clin Liver Dis. 2022;26:691-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 39. | Czaja AJ, Carpenter HA. Distinctive clinical phenotype and treatment outcome of type 1 autoimmune hepatitis in the elderly. Hepatology. 2006;43:532-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 133] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 40. | Miyake Y, Iwasaki Y, Takaki A, Kobashi H, Sakaguchi K, Shiratori Y. Clinical features of Japanese elderly patients with type 1 autoimmune hepatitis. Intern Med. 2007;46:1945-1949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 41. | Morii K, Nagano Y, Yamamoto T, Nakamura S, Okushin H. Increasing incidence of elderly-onset autoimmune hepatitis. Geriatr Gerontol Int. 2017;17:1722-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 42. | Baven-Pronk MAMC, Biewenga M, van Silfhout JJ, van den Berg AP, van Buuren HR, Verwer BJ, van Nieuwkerk CMJ, Bouma G, van Hoek B. Role of age in presentation, response to therapy and outcome of autoimmune hepatitis. Clin Transl Gastroenterol. 2018;9:165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 43. | Díaz Ramírez GS, Martínez Casas OY, Marín Zuluaga JI, Donado Gómez JH, Muñoz Maya O, Santos Sánchez O, Restrepo Gutiérrez JC. Características diferenciales de la hepatitis autoinmune en adultos mayores colombianos: estudio de cohorte. Rev Colomb Gastroenterol. 2019;34:135-143. [DOI] [Full Text] |
| 44. | Sonthalia N, Jain S, Thanage R, Junare P, Chandnani S, Pawar V, Contractor Q, Rathi P. Clinical, serological, histopathological and treatment profile of autoimmune hepatitis in the elderly. Clin Exp Hepatol. 2020;6:13-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Dalekos GN, Azariadis K, Lygoura V, Arvaniti P, Gampeta S, Gatselis NK. Autoimmune hepatitis in patients aged 70 years or older: Disease characteristics, treatment response and outcome. Liver Int. 2021;41:1592-1599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 46. | Newton JL, Jones DE, Metcalf JV, Park JB, Burt AD, Bassendine MF, James OF. Presentation and mortality of primary biliary cirrhosis in older patients. Age Ageing. 2000;29:305-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 47. | Muratori P, Granito A, Pappas G, Muratori L, Lenzi M, Bianchi FB. Clinical and serological profile of primary biliary cirrhosis in young and elderly patients. QJM. 2008;101:505-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 48. | Franceschet I, Cazzagon N, Mangini C, Perini L, Floreani A. Primary biliary cholangitis in elderly patients. Digest Liver Dis. 2016;48:e53-e54. [DOI] [Full Text] |
| 49. | Hirano K, Tada M, Isayama H, Yashima Y, Yagioka H, Sasaki T, Kogure H, Togawa O, Arizumi T, Matsubara S, Nakai Y, Sasahira N, Tsujino T, Kawabe T, Omata M. Clinical features of primary sclerosing cholangitis with onset age above 50 years. J Gastroenterol. 2008;43:729-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 50. | Tanaka A, Tazuma S, Okazaki K, Tsubouchi H, Inui K, Takikawa H. Clinical profiles of patients with primary sclerosing cholangitis in the elderly. J Hepatobiliary Pancreat Sci. 2015;22:230-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 51. | Rupp C, Rössler A, Zhou T, Rauber C, Friedrich K, Wannhoff A, Weiss KH, Sauer P, Schirmacher P, Süsal C, Stremmel W, Gotthardt DN. Impact of age at diagnosis on disease progression in patients with primary sclerosing cholangitis. United European Gastroenterol J. 2018;6:255-262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 52. | Compston J. Glucocorticoid-induced osteoporosis: an update. Endocrine. 2018;61:7-16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 324] [Article Influence: 40.5] [Reference Citation Analysis (1)] |
| 53. | Judd LL, Schettler PJ, Brown ES, Wolkowitz OM, Sternberg EM, Bender BG, Bulloch K, Cidlowski JA, de Kloet ER, Fardet L, Joëls M, Leung DY, McEwen BS, Roozendaal B, Van Rossum EF, Ahn J, Brown DW, Plitt A, Singh G. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171:1045-1051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 158] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 54. | Maxwell CB, Jenkins AT. Drug-induced heart failure. Am J Health Syst Pharm. 2011;68:1791-1804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Ng MK, Celermajer DS. Glucocorticoid treatment and cardiovascular disease. Heart. 2004;90:829-830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 58] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 56. | Hu H, Zhang G, Chen T, Liu Y, Meng L, Holmdahl R, Dai L, Zhao Y. Immunosenescence in autoimmune diseases. Autoimmun Rev. 2025;24:103805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 57. | Liu Z, Liang Q, Ren Y, Guo C, Ge X, Wang L, Cheng Q, Luo P, Zhang Y, Han X. Immunosenescence: molecular mechanisms and diseases. Signal Transduct Target Ther. 2023;8:200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 714] [Cited by in RCA: 636] [Article Influence: 212.0] [Reference Citation Analysis (0)] |
| 58. | Mack CL, Adams D, Assis DN, Kerkar N, Manns MP, Mayo MJ, Vierling JM, Alsawas M, Murad MH, Czaja AJ. Diagnosis and Management of Autoimmune Hepatitis in Adults and Children: 2019 Practice Guidance and Guidelines From the American Association for the Study of Liver Diseases. Hepatology. 2020;72:671-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 793] [Cited by in RCA: 681] [Article Influence: 113.5] [Reference Citation Analysis (3)] |
| 59. | van Staa TP, Leufkens HG, Cooper C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int. 2002;13:777-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 933] [Cited by in RCA: 798] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 60. | Balasubramanian A, Wade SW, Adler RA, Lin CJF, Maricic M, O'Malley CD, Saag K, Curtis JR. Glucocorticoid exposure and fracture risk in patients with new-onset rheumatoid arthritis. Osteoporos Int. 2016;27:3239-3249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 61. | Amiche MA, Albaum JM, Tadrous M, Pechlivanoglou P, Lévesque LE, Adachi JD, Cadarette SM. Fracture risk in oral glucocorticoid users: a Bayesian meta-regression leveraging control arms of osteoporosis clinical trials. Osteoporos Int. 2016;27:1709-1718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 104] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 62. | Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C. Use of oral corticosteroids and risk of fractures. J Bone Miner Res. 2000;15:993-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1120] [Cited by in RCA: 916] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 63. | Van Staa TP, Laan RF, Barton IP, Cohen S, Reid DM, Cooper C. Bone density threshold and other predictors of vertebral fracture in patients receiving oral glucocorticoid therapy. Arthritis Rheum. 2003;48:3224-3229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 401] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 64. | Kanis JA, Johansson H, Oden A, Johnell O, de Laet C, Melton III LJ, Tenenhouse A, Reeve J, Silman AJ, Pols HA, Eisman JA, McCloskey EV, Mellstrom D. A meta-analysis of prior corticosteroid use and fracture risk. J Bone Miner Res. 2004;19:893-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 671] [Cited by in RCA: 542] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 65. | Lekamwasam S, Adachi JD, Agnusdei D, Bilezikian J, Boonen S, Borgström F, Cooper C, Diez Perez A, Eastell R, Hofbauer LC, Kanis JA, Langdahl BL, Lesnyak O, Lorenc R, McCloskey E, Messina OD, Napoli N, Obermayer-Pietsch B, Ralston SH, Sambrook PN, Silverman S, Sosa M, Stepan J, Suppan G, Wahl DA, Compston JE; Joint IOF-ECTS GIO Guidelines Working Group. A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos Int. 2012;23:2257-2276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 236] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 66. | Leslie WD, Lix LM, Johansson H, Oden A, McCloskey E, Kanis JA. Spine-hip discordance and fracture risk assessment: a physician-friendly FRAX enhancement. Osteoporos Int. 2011;22:839-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 102] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 67. | Messina OD, Vidal LF, Wilman MV, Bultink IEM, Raterman HG, Lems W. Management of glucocorticoid-induced osteoporosis. Aging Clin Exp Res. 2021;33:793-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 68. | Compston J, Cooper A, Cooper C, Gittoes N, Gregson C, Harvey N, Hope S, Kanis JA, McCloskey EV, Poole KES, Reid DM, Selby P, Thompson F, Thurston A, Vine N; National Osteoporosis Guideline Group (NOGG). UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos. 2017;12:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 687] [Cited by in RCA: 588] [Article Influence: 65.3] [Reference Citation Analysis (0)] |
| 69. | Buckley L, Guyatt G, Fink HA, Cannon M, Grossman J, Hansen KE, Humphrey MB, Lane NE, Magrey M, Miller M, Morrison L, Rao M, Robinson AB, Saha S, Wolver S, Bannuru RR, Vaysbrot E, Osani M, Turgunbaev M, Miller AS, McAlindon T. 2017 American College of Rheumatology Guideline for the Prevention and Treatment of Glucocorticoid-Induced Osteoporosis. Arthritis Rheumatol. 2017;69:1521-1537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 357] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 70. | Durazzo M, Lupi G, Scandella M, Ferro A, Gruden G. Autoimmune hepatitis treatment in the elderly: A systematic review. World J Gastroenterol. 2019;25:2809-2818. [PubMed] [DOI] [Full Text] |
| 71. | Zachou K, Gatselis NK, Arvaniti P, Gabeta S, Rigopoulou EI, Koukoulis GK, Dalekos GN. A real-world study focused on the long-term efficacy of mycophenolate mofetil as first-line treatment of autoimmune hepatitis. Aliment Pharmacol Ther. 2016;43:1035-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 84] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 72. | Granito A, Muratori L, Pappas G, Muratori P, Ferri S, Cassani F, Lenzi M, Bianchi FB. Clinical features of type 1 autoimmune hepatitis in elderly Italian patients. Aliment Pharmacol Ther. 2005;21:1273-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 58] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 73. | Al-Chalabi T, Boccato S, Portmann BC, McFarlane IG, Heneghan MA. Autoimmune hepatitis (AIH) in the elderly: a systematic retrospective analysis of a large group of consecutive patients with definite AIH followed at a tertiary referral centre. J Hepatol. 2006;45:575-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 142] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 74. | Gleeson D, Martyn-StJames M, Oo Y, Flatley S. What is the optimal first-line treatment of autoimmune hepatitis? A systematic review with meta-analysis of randomised trials and comparative cohort studies. BMJ Open Gastroenterol. 2025;12:e001549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 75. | Zhang Y, Sun WL, Jin DL, Jing-Hua D. Clinical features of elderly Chinese patients with autoimmune hepatitis. Turk J Gastroenterol. 2013;24:489-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 76. | Adis Medical Writers. Treat autoimmune hepatitis in the elderly with corticosteroids + azathioprine to reduce the risk of corticosteroid-related toxicity. Drugs Ther Perspect. 2019;35:129-134. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 77. | Abdollahi M, Ekrami NK, Ghojazadeh M, Boezen HM, Somi M, Alizadeh BZ. Tacrolimus and mycophenolate mofetil as second-line treatment in autoimmune hepatitis: Is the evidence of sufficient quality to develop recommendations? World J Gastroenterol. 2020;26:5896-5910. [PubMed] [DOI] [Full Text] |
| 78. | Schramm C, Bubenheim M, Adam R, Karam V, Buckels J, O'Grady JG, Jamieson N, Pollard S, Neuhaus P, Manns MM, Porte R, Castaing D, Paul A, Traynor O, Garden J, Friman S, Ericzon BG, Fischer L, Vitko S, Krawczyk M, Metselaar HJ, Foss A, Kilic M, Rolles K, Burra P, Rogiers X, Lohse AW; European Liver Intestine Transplant Association. Primary liver transplantation for autoimmune hepatitis: a comparative analysis of the European Liver Transplant Registry. Liver Transpl. 2010;16:461-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 79. | Adam R, Karam V, Cailliez V, O Grady JG, Mirza D, Cherqui D, Klempnauer J, Salizzoni M, Pratschke J, Jamieson N, Hidalgo E, Paul A, Andujar RL, Lerut J, Fisher L, Boudjema K, Fondevila C, Soubrane O, Bachellier P, Pinna AD, Berlakovich G, Bennet W, Pinzani M, Schemmer P, Zieniewicz K, Romero CJ, De Simone P, Ericzon BG, Schneeberger S, Wigmore SJ, Prous JF, Colledan M, Porte RJ, Yilmaz S, Azoulay D, Pirenne J, Line PD, Trunecka P, Navarro F, Lopez AV, De Carlis L, Pena SR, Kochs E, Duvoux C; all the other 126 contributing centers (www. eltr.org) and the European Liver and Intestine Transplant Association (ELITA). 2018 Annual Report of the European Liver Transplant Registry (ELTR) - 50-year evolution of liver transplantation. Transpl Int. 2018;31:1293-1317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 365] [Article Influence: 45.6] [Reference Citation Analysis (4)] |
| 80. | Pilotto A, Addante F, D'Onofrio G, Sancarlo D, Ferrucci L. The Comprehensive Geriatric Assessment and the multidimensional approach. A new look at the older patient with gastroenterological disorders. Best Pract Res Clin Gastroenterol. 2009;23:829-837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 81. | Dimitriadou I, Toska A, Eloranta S, Mört S, Korsström N, Lundberg A, Häger M, Melbarde-Kelmere A, Circenis K, Šteinmiller J, Skuladottir SS, Hjaltadóttir I, Fradelos EC. Comprehensive Geriatric Assessment: Addressing Unmet Healthcare Needs in Older Adults. Healthcare (Basel). 2025;13:2715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 82. | Morley JE, Vellas B, van Kan GA, Anker SD, Bauer JM, Bernabei R, Cesari M, Chumlea WC, Doehner W, Evans J, Fried LP, Guralnik JM, Katz PR, Malmstrom TK, McCarter RJ, Gutierrez Robledo LM, Rockwood K, von Haehling S, Vandewoude MF, Walston J. Frailty consensus: a call to action. J Am Med Dir Assoc. 2013;14:392-397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3267] [Cited by in RCA: 2882] [Article Influence: 221.7] [Reference Citation Analysis (4)] |
| 83. | Welsh TJ, Gordon AL, Gladman JR. Comprehensive geriatric assessment--a guide for the non-specialist. Int J Clin Pract. 2014;68:290-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 148] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 84. | Laube R, Wang H, Park L, Heyman JK, Vidot H, Majumdar A, Strasser SI, McCaughan GW, Liu K. Frailty in advanced liver disease. Liver Int. 2018;38:2117-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 89] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 85. | Lai JC, Sonnenday CJ, Tapper EB, Duarte-Rojo A, Dunn MA, Bernal W, Carey EJ, Dasarathy S, Kamath BM, Kappus MR, Montano-Loza AJ, Nagai S, Tandon P. Frailty in liver transplantation: An expert opinion statement from the American Society of Transplantation Liver and Intestinal Community of Practice. Am J Transplant. 2019;19:1896-1906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 182] [Article Influence: 26.0] [Reference Citation Analysis (1)] |