Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.117465
Revised: December 28, 2025
Accepted: February 2, 2026
Published online: March 27, 2026
Processing time: 108 Days and 18.4 Hours
Hepatitis C virus (HCV) poses a formidable global health concern. Non-invasive assessment of hepatic fibrosis has gained increasing importance as liver biopsy, the current gold standard, is less favored as a routine test due to its invasiveness, cost, and risk of complications. Although several clinical scoring systems exist, the potential contribution of lipid biomarkers has not been sufficiently explored.
To develop and validate a novel diagnostic tool for non-invasive detection of significant hepatic fibrosis in HCV patients using a composite of serum bio
A retrospective of 316 chronic HCV patients was analyzed. Candidate predictors of significant hepatic fibrosis were screened by univariate analysis and selected using least absolute shrinkage and selection operator regression, followed by multivariate logistic regression to identify independent predictors. A novel fibrosis risk score (FRS) incorporating platelet count, liver function indices, and lipid biomarkers was developed and compared with three existing clinical scores. Six ML models were additionally developed, and diagnostic performance was assessed using area under the receiver operating characteristic curve (AUC), sensitivity, specificity, positive predictive value, negative predictive value, and F1-score.
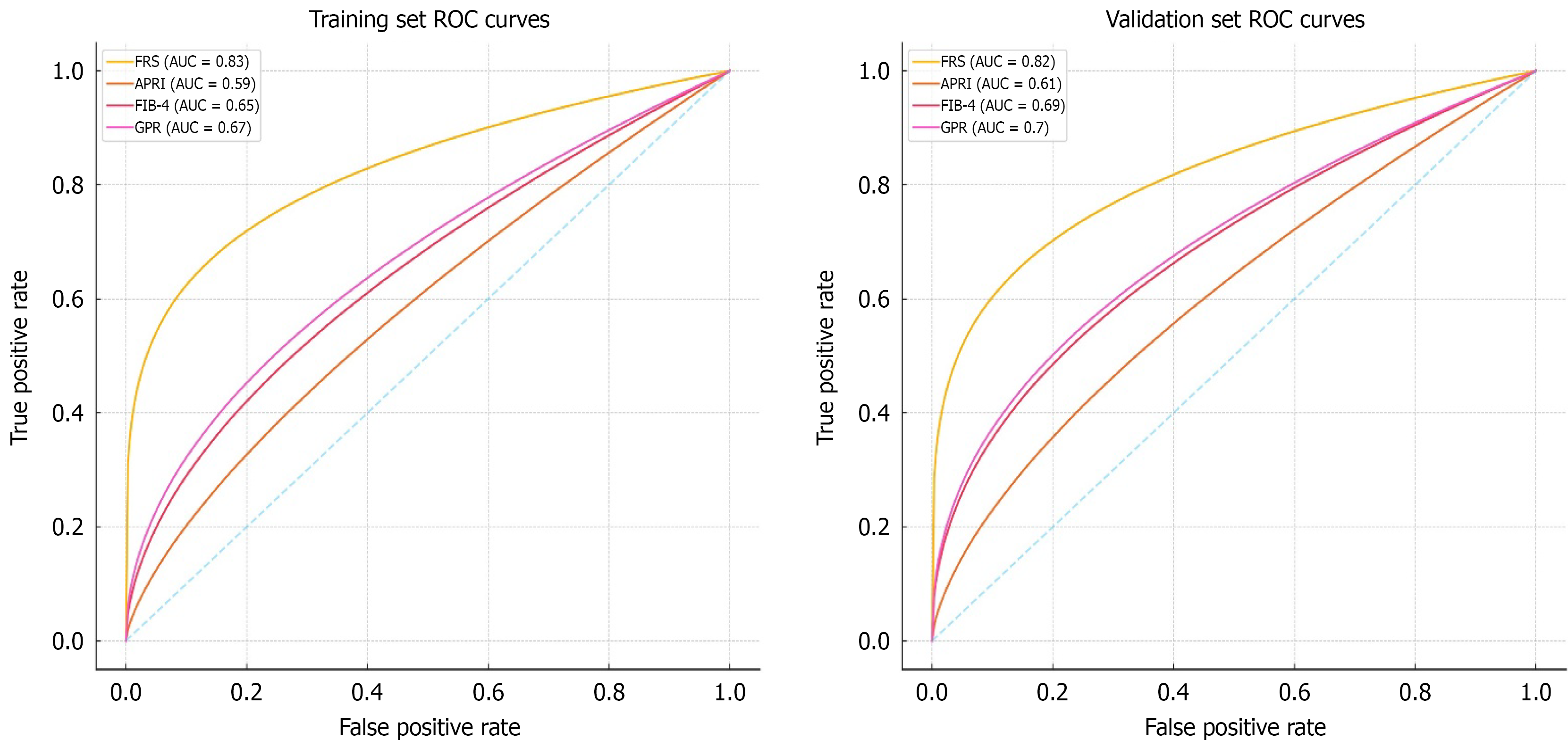
The newly developed FRS demonstrated excellent discriminatory performance for significant hepatic fibrosis, with scores ≥ 9 indicating higher risk and achieving an AUC of 0.83 (95% confidence interval: 0.75-0.88) in the training cohort. The model outperformed commonly used non-invasive indices, including aspartate aminotransferase-to-platelet ratio index (AUC = 0.59), fibrosis-4 (AUC = 0.65), and the gamma-glutamyl transpeptidase-to-platelet ratio (AUC = 0.67) at their respective optimal cutoff thresholds of 1.5, 1.45, and 0.4. Among the evaluated ML app
The FRS showed robust predictive accuracy for significant hepatic fibrosis in chronic HCV infection and offers a practical, reliable, and user-friendly non-invasive tool. Additionally, ML models substantially enhanced predictive performance. The random forest model demonstrated superior diagnostic potential among ML algorithms, supporting its applicability in fibrosis risk stratification and screening.
Core Tip: This study developed and validated a simple non-invasive fibrosis risk score for hepatitis C virus using platelet count, lipid markers, and liver function parameters. The score outperformed commonly used clinical tools for detecting significant hepatic fibrosis. Additionally, machine-learning models were evaluated, which outperformed the commonly used clinical risk scoring systems with random forest and AdaBoost demonstrating the highest diagnostic performance for detecting significant hepatic fibrosis in hepatitis C virus infection.
- Citation: Bashir A, Arora R, Mehrotra D, Bala M, Parry AH, Iqball A, Bhat SA, Wani ZA. Non-invasive prediction of significant hepatic fibrosis in individuals with chronic hepatitis C infection using fibrosis risk score and machine learning models. World J Hepatol 2026; 18(3): 117465
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/117465.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.117465
Hepatitis C virus (HCV) poses a substantial global public health challenge. According to the World Health Organization (WHO)’s 2024 report, roughly 50 million people are living with chronic HCV infection globally. Each year, an estimated 1 million new cases are detected, and in 2022 alone, the disease accounted for approximately 240000 deaths, predominantly owing to its complications, including cirrhosis and hepatocellular carcinoma (HCC)[1].
HCV infection triggers a continuous spectrum of hepatic injury, ranging from fibrosis to cirrhosis and, in some cases, HCC. Hepatic fibrosis represents an aberrant reparative response to persistent hepatocellular injury induced by chronic HCV infection[2,3]. The fibrotic process is marked by the excessive accumulation of extracellular matrix components - particularly collagen - synthesized by hepatic stellate cells that have transitioned from a quiescent to an activated state[4,5]. Importantly, fibrosis is dynamic and potentially reversible when inflammation is controlled and the etiologic agent is eliminated[6,7]. Hence, fibrosis staging remains central to patient management and prognostication[7].
Liver biopsy remains the gold standard for assessing the degree of hepatic fibrosis[7,8]. However, it is fraught with certain limitations, including its invasive nature, associated procedure-related complications, high cost, and the risk of sampling error inherent in examining only a small portion of hepatic tissue[8]. Consequently, there has been a growing interest to develop alternative non-invasive tools that can accurately predict hepatic fibrosis without the need for biopsy[9]. Direct serum fibrosis markers, such as hyaluronic acid and procollagen peptides, remain costly and are not widely accessible for routine use[10,11]. In contrast, indirect biomarkers offer a more affordable and practical approach for fibrosis screening and form the basis of several commonly used clinical scoring systems - such as the aspartate aminotransferase-to-platelet ratio index (APRI), fibrosis-4 (FIB-4), and the gamma-glutamyl transpeptidase-to-platelet ratio (GPR) scores[12,13].
A substantial body of evidence indicates that serum lipid levels are significantly reduced in patients with chronic viral hepatitis, particularly HCV[14,15]. The mechanisms underlying this serum lipid reduction are multifactorial and include impaired hepatic synthetic function, activation of pro-inflammatory and immune-mediated pathways, and direct viral interference with lipid metabolism[16,17]. Despite this biological plausibility, lipid parameters remain underutilized in fibrosis assessment, and their adjunctive role as non-invasive biomarkers is inadequately explored[18,19].
Non-invasive imaging modalities, including shear-wave elastography, transient elastography (FibroScan), and magnetic resonance elastography demonstrate high diagnostic performance[20-22]. Among these, transient elastography has emerged as the most widely used and well-validated tool for evaluating hepatic fibrosis. Transient elastography is a non-invasive, simple, and easy-to-perform imaging technique[23,24]. It has demonstrated high diagnostic accuracy in viral hepatitis, with meta-analysis studies reporting a high accuracy for detecting significant fibrosis (Meta-analysis of Histological Data in Viral Hepatitis ≥ F2) showing an area under the receiver operating characteristic curve (AUC) of 0.89. Its performance is even stronger for diagnosing cirrhosis (Meta-analysis of Histological Data in Viral Hepatitis F3-F4), with an AUC of around 0.94[25,26].
In this context, the present study aimed to integrate demographic data, hematological indices, liver function parameters and lipid profile indices to develop a reliable non-invasive tool for predicting significant hepatic fibrosis in chronic HCV patients. Additionally, the study assessed the added diagnostic value of machine-learning (ML) models in identifying significant hepatic fibrosis in chronic HCV patients.
This retrospective study comprised 316 patients with confirmed chronic HCV infection, defined by the presence of anti-HCV antibodies and a detectable HCV RNA assay for ≥ 6 months. Adults aged 18 years or older with chronic HCV infection with comprehensive clinical and laboratory records - including complete blood counts, liver function tests (LFT), lipid profiles, and documented transient elastography (FibroScan) assessments - were eligible for inclusion. Patients were excluded if they had hepatitis B co-infection or other etiologies of chronic liver disease, such as autoimmune hepatitis, metabolic-associated fatty liver disease, primary biliary cholangitis, primary sclerosing cholangitis, drug-induced liver injury, alcoholic liver disease, decompensated cirrhosis, or HCC. Patients with conditions that alter lipid metabolism, including those undergoing treatment with hypolipidemic drugs or pregnant individuals, were excluded from the study.
The study protocol was approved by the Institutional Review Board, and the requirement for written informed consent was waived owing to the retrospective nature of the study.
Transient elastography (FibroScan) was performed in strict accordance with established protocols, ensuring optimal measurement accuracy and methodological rigor[27,28]. Only scans with ≥ 10 valid measurements and an interquartile-range/median ratio ≤ 30% were considered valid and reliable. Liver fibrosis assessed by transient elastography was classified according to liver stiffness measurements expressed in kilopascals (kPa). Fibrosis was categorized as F0-F1 for stiffness values < 7.0 kPa, F2 for values between 7.0 kPa and 8.99 kPa, F3 for values ranging from 9.0 kPa to 12.49 kPa, and F4 for measurements ≥ 12.5 kPa. These cut-off points were selected based on widely accepted thresholds in the literature, which identify these values as optimal for detecting significant hepatic fibrosis in chronic viral hepatitis, including HCV. For analytical purposes, transient elastography scores of F0-F1 were considered non-significant fibrosis, whereas scores ≥ F2 (≥ 7.0 kPa) were classified as significant hepatic fibrosis[29,30].
Patient data were retrieved from the medical records system and included demographic variables such as age, gender, presence of diabetes mellitus, and body mass index (BMI). Laboratory parameters comprised complete blood count, LFT [serum bilirubin, total protein, serum albumin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), γ-glutamyl transferase (GGT)], lipid profiles [total cholesterol, triglycerides, low-density lipoprotein (LDL), high-density lipoprotein (HDL), and very low density lipoprotein (VLDL)]. Fasting venous blood samples were used for LFT and lipid profile using standardized laboratory protocols. Transient elastography measurements were also recorded. To ensure temporal consistency between the measurements, only cases in which the blood tests and transient elastography were conducted within seven days of each other were included. These variables were selected for analysis because they represent routinely available clinical indicators and are objective, easy to obtain, and closely associated with the severity of HCV infection. Serum biomarker–based fibrosis scores were calculated using standard formulas: The APRI was determined as [(AST/upper limit of normal) × 100]/platelet count (PLT); the FIB-4 index as (age × AST)/(PLT × √ALT); and the GPR as (GGT/upper limit of normal)/(PLT × 100)[11,12].
The statistical analyses were performed using Python 3 and IBM SPSS Statistics version 24 (IBM Corporation, Armonk, NY, United States). Data following a normal distribution are summarized as mean ± SD, while non-normal data are summarized as median (interquartile range). Differences between categorical variables were examined using the χ2 test. For continuous variables, comparisons were made using the independent Student’s t-test when data were normally distributed and the Mann-Whitney U test when data were non-normally distributed. Data normality was assessed with the Shapiro-Wilk test, and correlations with fibrosis stage were analyzed using Spearman rank correlation test. Variables demonstrating statistically significant differences between the two groups - non-significant fibrosis (F0-F1) and significant fibrosis (F2+) - were identified using univariate analysis. To minimize potential multicollinearity among predictors, feature selection was performed using least absolute shrinkage and selection operator regression with 10-fold cross-validation. Variables with non-zero coefficients at the optimal λ value were subsequently entered into a multivariate logistic regression analysis to determine their independent significance, expressed as adjusted odds ratios (ORs) with corresponding 95% confidence intervals (CIs). The regression coefficients were derived from the final multivariate analysis. Statistical significance was defined as P < 0.05.
A total of 316 patients with HCV were included in the study. The study was randomly split into a training cohort (n = 214; 68%) and a validation cohort (n = 102; 32%) using a simple random allocation method. Randomization was performed by assigning a random number to each patient using a computer-based random number generator. No stratification was employed during the randomization process, as the overall sample was deemed adequate and representative to ensure a balanced distribution of baseline characteristics between the two cohorts. The allocation ratio of approximately 2:1 was intentionally chosen to allocate a sufficient number of patients for model development in the training cohort while retaining an adequate number of patients in the validation cohort to allow for robust internal validation. The 2:1 train-validation partition ensures a larger training cohort for feature selection, optimal model fitting and model stability, whereas a sufficient number of patients in the validation cohort ensures meaningful evaluation of model performance and reduces optimism bias and overfitting.
The fibrosis risk score (FRS) was developed in the training cohort by using the regression coefficients obtained in multivariate model. Each variable was assigned a weighted score, and the cumulative score indicated the probability of an individual of having significant hepatic fibrosis. Cut-off values for variables were determined using the Youden Index. The FRS was subsequently applied to the validation cohort to assess its diagnostic performance by calculating the AUC, sensitivity, specificity, positive predictive value, and negative predictive value in both the training and validation cohorts. Diagnostic performance of FRS was compared with commonly used fibrosis scores (APRI, FIB-4, GPR).
Subsequently, several ML algorithms were developed to predict significant hepatic fibrosis using demographic and biochemical variables. The models evaluated included random forest (RF), AdaBoost, logistic regression, support vector machine (SVM), K-nearest neighbors (KNN), and Naive Bayes. These models were chosen because they represent diverse ML paradigms with unique strengths and weaknesses when applied in healthcare settings. To identify the optimal algorithm, a comparative evaluation was performed in both training and validation cohorts to identify the algorithm which is best suited to the demands of this clinical dataset.
RF and AdaBoost are robust ensemble models capable of handling complex clinical data and non-linear interactions. Logistic Regression is fast and easily interpretable but unable to capture non-linear relationships in complex clinical data. SVM performs well in handling high-dimensional data effectively through kernel functions but can be less efficient with larger datasets. KNN provides instance-based learning that adapts to local data structures; and naive Bayes can be efficient in limited sample sizes and provide probabilistic modeling. Evaluating multiple models together allows a balanced comparison between accuracy, interpretability, and generalizability in healthcare domain.
Five-fold cross-validation was used to optimize performance and mitigate class imbalance. Model performance was assessed using receiver operating characteristic curves, accuracy, precision, recall, and F1 score. The use of a validation set simulated real-world clinical conditions, allowing for internal validation and aids in assessment of model generalizability. The methodological workflow of the study is presented in Figure 1.
A total of 316 patients with confirmed HCV infection were enrolled in the study, of whom 182 (57.6%) had non-significant hepatic fibrosis (F0-F1) and 134 (42.4%) had significant fibrosis (F2-F4). There were no significant differences between the two groups with respect to some baseline characteristics including age, sex distribution, body mass index, some biochemical parameters (total protein, triglycerides, VLDL) (all P values > 0.05) (Table 1). However, the univariate analysis identified platelet count, ALT, AST, GGT, ALP, serum albumin, total bilirubin, total cholesterol, HDL, and LDL as parameters significantly associated with significant hepatic fibrosis (all P values < 0.05). A detailed comparison of variables between the non-significant hepatic fibrosis and significant hepatic fibrosis groups is provided in Table 1.
| Parameter | Univariate analysis | Multivariate analysis | ||||
| Non-significant fibrosis (F0-F1) (n = 182; 57.6%) | Significant fibrosis (F2-F4) (n = 134; 42.4%) | Spearman ρ | P value | OR (95%CI) | P value | |
| Gender | 0.049 | 0.38 | - | - | ||
| Males | 96 (52.7) | 64 (47.7) | ||||
| Females | 86 (47.3) | 70 (52.3) | ||||
| Age (year), mean ± SD | 46.23 ± 14.5 | 48.10 ± 10.93 | 0.070 | 0.192 | - | - |
| Body mass index, mean ± SD | 24.03 ± 3.7 | 23.96 ± 3.2 | 0.0500 | 0.659 | - | - |
| Diabetes | 0.082 | 0.146 | - | - | ||
| Yes | 22 (12) | 24 (17.91) | ||||
| No | 160 (88) | 110 (82.09) | ||||
| Platelet count (× 109/L) | 157.7 ± 30.93 | 127.15 ± 28.00 | -0.4421 | < 0.001 | 0.72 (0.60-0.86) | < 0.001 |
| Bilirubin (mg/dL) | 0.726 ± 0.29 | 0.818 ± 0.31 | 0.1056 | 0.007 | - | - |
| AST (U/L) | 33.2 (21, 50) | 49 (30, 66) | 0.4768 | < 0.001 | 1.18 (1.05-1.33) | 0.003 |
| ALT (U/L) | 36 (24, 49) | 47 (30, 64) | 0.33 | < 0.001 | - | - |
| GGT (U/L) | 26 (16, 33) | 46 (25, 79) | 0.50 | < 0.001 | 1.07 (1.02-1.13) | 0.01 |
| ALP (U/L) | 66 (52, 77) | 81 (64, 97) | 0.4298 | < 0.001 | 1.10 (1.02-1.13) | 0.01 |
| Serum albumin (g/dL) | 3.83 ± 0.29 | 3.16 ± 0.36 | -0.6278 | < 0.001 | 0.81 (0.68-0.96) | 0.02 |
| Total protein (g/dL) | 7.082 ± 1.23 | 6.912 ± 1.12 | -0.07 | 0.2 | - | - |
| Total cholesterol (mg/dL) | 160.4 ± 21.1 | 129.1 ± 23.9 | -0.6156 | < 0.001 | 0.84 (0.76-1.00) | 0.01 |
| Triglycerides (mg/dL) | 131 (120, 170) | 126 (115, 167) | -0.108 | 0.25 | - | - |
| HDL (mg/dL) | 37 (30, 54) | 30 (26, 44) | -0.4414 | < 0.001 | - | - |
| LDL (mg/dL) | 90.0 ± 4.3 | 65.4 ± 5.2 | -0.5992 | < 0.001 | 0.84 (0.76-1.00) | 0.01 |
| VLDL (mg/dL) | 24.97 ± 12.11 | 23.45 ± 18.50 | -0.1656 | 0.1424 | - | - |
Ten biochemical and hematological parameters - platelet count, AST, ALT, GGT, ALP, serum albumin, total bilirubin, total cholesterol, LDL, and HDL - were significantly different between the non-significant and significant hepatic fibrosis groups on univariate analysis. To mitigate multicollinearity and to identify the most relevant predictors these ten significant variables were subjected to least absolute shrinkage and selection operator regression with 10-fold cross-validation. The variables that demonstrated non-zero coefficients were used for multivariate modeling. The seven variables identified by multivariate logistic regression as independent predictors of significant hepatic fibrosis were used to develop the FRS. The predictors included: Platelet count (OR = 0.72; 95%CI: 0.60-0.86; P < 0.001), AST (OR = 1.18; 95%CI: 1.05-1.33; P = 0.003), GGT (OR = 1.07; 95%CI: 1.02-1.13; P = 0.01), ALP (OR = 1.10; 95%CI: 1.02-1.13; P = 0.01), serum albumin (OR = 0.81; 95%CI: 0.68-0.96; P = 0.02), total cholesterol (OR = 0.84; 95%CI: 0.76-1.00; P = 0.01), and LDL (OR = 0.84; 95%CI: 0.76-1.00; P = 0.01).
To develop a simple, practical and accurate FRS based on demographic and biochemical parameters, patients were allocated into a training set (n = 214; 68%) and a validation set (n = 102; 32%) using a random sampling method. There were no significant differences between the two groups with respect to demographic characteristics, biochemical parameters, or the proportion of patients with significant and non-significant hepatic fibrosis, indicating that both groups were comparable and suitable for subsequent model development and validation (Table 2).
| Parameters | Training dataset (n = 214) | Validation dataset (n = 102) | P value |
| Group | 0.815 | ||
| F0-F1 | 106 (49.5) | 49 (48) | |
| F2 | 36 (16.8) | 21 (20.5) | |
| F3 | 38 (17.8) | 18 (17.6) | |
| F4 | 34 (15.9) | 14 (13.7) | |
| Gender | 0.994 | ||
| Males | 109 (50.9) | 52 (51) | |
| Females | 105 (49.1) | 50 (49) | |
| Age (years) | 46.88 ± 13.5 | 47.12 ± 11.6 | 0.871 |
| Body mass index | 23.86 ± 3.44 | 24.01 ± 3.39 | 0.715 |
| Diabetes | 0.70 | ||
| Yes | 30 (14) | 16 (15.6) | |
| No | 184 (86) | 86 (84.3) | |
| Platelet count (× 109/L) | 145.44 ± 30.83 | 144.1 ± 29.2 | 0.708 |
| Bilirubin (mg/dL) | 0.75 ± 0.26 | 0.756 ± 0.27 | 0.852 |
| AST (U/L) | 36 (25, 60) | 40 (25, 63) | 0.55 |
| ALT (U/L) | 42 (23, 47) | 39 (25, 51) | 0.66 |
| GGT (U/L) | 34 (21, 55) | 36 (20, 53) | 0.90 |
| ALP (U/L) | 59 (48, 77) | 63.7 (54, 80) | 0.07 |
| Serum albumin (g/dL) | 3.53 ± 0.26 | 3.57 ± 0.33 | 0.284 |
| Total protein (g/dL) | 7.02 ± 1.21 | 7.01 ± 1.13 | 0.943 |
| Total cholesterol (mg/dL) | 148.33 ± 19.12 | 146.6 ± 22.61 | 0.505 |
| Triglycerides (mg/dL) | 128 (111, 167) | 133.2 (112, 174) | 0.38 |
| HDL (mg/dL) | 35 (27, 43) | 31.9 (25, 48) | 0.98 |
| LDL (mg/dL) | 80.11 ± 14.3 | 78.33 ± 15.2 | 0.323 |
| VLDL (mg/dL) | 24.32 ± 11.16 | 23.89 ± 17.68 | 0.822 |
Based on the results of the multivariate logistic regression, a simple and practical scoring model, FRS was developed using seven clinically relevant variables that demonstrated significant association with significant hepatic fibrosis. The components and scoring scheme of the FRS score are shown in Table 3. By summing the scores assigned to each variable, a total score is obtained that reflects the risk of having significant hepatic fibrosis.
| Parameter | Points | |
| Platelet count < 150 × 109/L | 5 | |
| AST > 45 U/L | 3 | |
| Serum albumin < 3.5 g/dL | 3 | |
| Total cholesterol < 140 mg/dL | 3 | |
| LDL < 80 mg/dL | 3 | |
| ALP > 75 U/L | 2 | |
| GGT > 40 U/L | 1 | |
| Interpretation of the score | ||
| Total score | Risk category | Interpretation |
| < 5 | Low risk | Minimal risk of significant fibrosis |
| ≥ 5 and < 9 | Intermediate risk | Borderline risk → further evaluation suggested |
| 9-20 | High risk | Strongly predictive of F2-F4 fibrosis |
Following the development of FRS the data from both the training and validation sets were analyzed to assess its predictive performance by comparing it against the established non-invasive fibrosis scores, including APRI, FIB-4, and GPR (Table 4). In the training set, the FRS demonstrated the highest predictive performance, yielding an AUC of 0.83 (95%CI: 0.75-0.88), which was significantly superior to APRI (AUC: 0.59, 95%CI: 0.49-0.66), FIB-4 (AUC: 0.65, 95%CI: 0.57-0.75), and GPR (AUC: 0.67, 95%CI: 0.55-0.76) for the same training set. A similar trend was observed in the validation set, where the FRS model developed in this study consistently outperformed these three clinical scoring systems, confirming its superior diagnostic accuracy for predicting significant hepatic fibrosis in HCV (Figure 2).
| Models | Cut-off | Training set | Validation set | ||||||||
| AUC (95%CI) | Sensitivity | Specificity | NPV | PPV | AUC (95%CI) | Sensitivity | Specificity | NPV | PPV | ||
| FRS | 9 | 0.83 (0.75-0.88) | 0.71 | 0.85 | 0.79 | 0.71 | 0.82 (0.76-0.90) | 0.72 | 0.84 | 0.79 | 0.72 |
| APRI | 1.5 | 0.59 (0.49-0.66) | 0.31 | 0.87 | 0.67 | 0.61 | 0.61 (0.48-0.73) | 0.31 | 0.83 | 0.65 | 0.62 |
| FIB-4 | 1.45 | 0.65 (0.57-0.75) | 0.50 | 0.78 | 0.72 | 0.59 | 0.69 (0.59-0.78) | 0.48 | 0.81 | 0.65 | 0.63 |
| GPR | 0.40 | 0.67 (0.55-0.76) | 0.49 | 0.83 | 0.71 | 0.68 | 0.70 (0.59-0.80) | 0.48 | 0.83 | 0.69 | 0.70 |
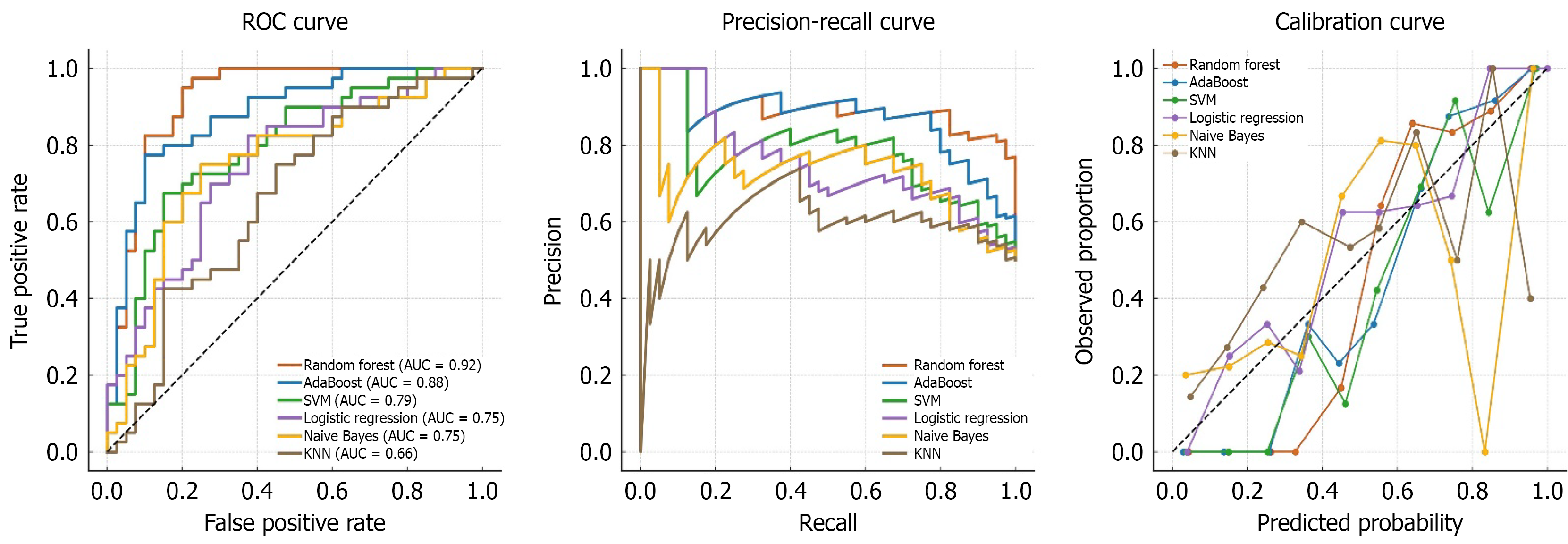
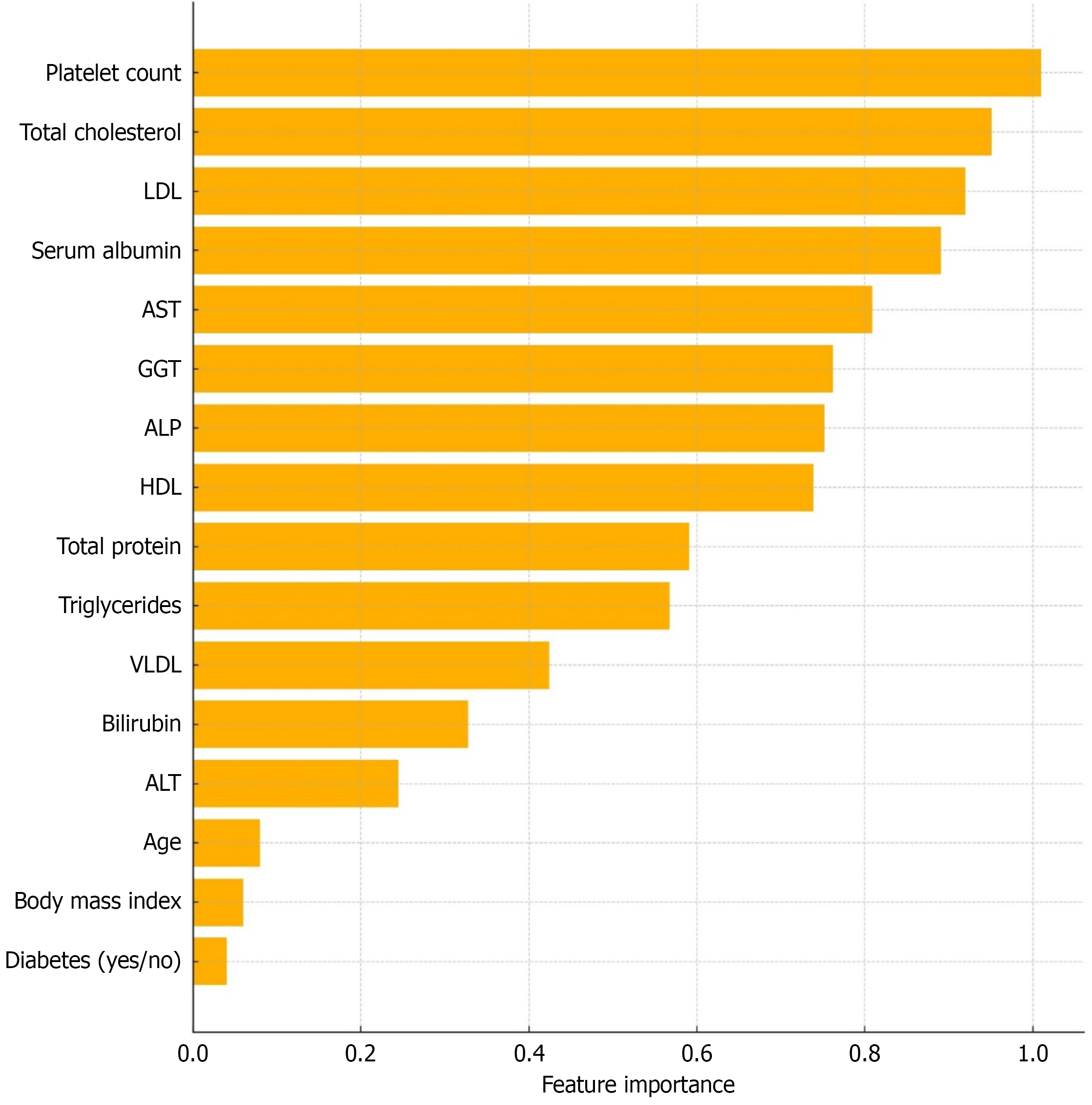
Finally, the predictive performance of multiple ML algorithms, including RF, AdaBoost, logistic regression, SVM, KNN and Naive Bayes was assessed to determine if they could perform better in predicting the significant hepatic fibrosis. The performance metrics for all models were evaluated in terms of AUC, sensitivity, specificity, positive predictive value, negative predictive value, F1 score, and Brier score, as summarized in Table 5. In the training dataset, among all models, RF demonstrated the best predictive performance with the highest AUC of 0.921, along with a sensitivity of 0.825, specificity of 0.90, positive predictive value of 0.89, negative predictive value of 0.83, F1 score of 0.85, and a Brier score of 0.14. AdaBoost was the second-best performing model, yielding an AUC of 0.881, a sensitivity of 0.80, specificity of 0.77, positive predictive value of 0.78, negative predictive value of 0.79, an F1 score of 0.79, and a Brier score of 0.15. Other ML algorithms showed relatively lower predictive efficiency in this dataset (Figure 3). The superior AUC value of the RF model highlights its strong discriminative ability. The relative importance of variables contributing to the ML prediction models is illustrated in Figure 4, with platelet count, total cholesterol, LDL, GGT, AST, ALP, and serum albumin emerging as the most influential predictors.
| Parameter | Model | AUC (95%CI) | Sensitivity | Specificity | PPV | NPV | F1 score | Brier score |
| Training | Random forest | 0.921 (0.889-0.953) | 0.825 | 0.900 | 0.892 | 0.837 | 0.857 | 0.141 |
| AdaBoost | 0.881 (0.842-0.921) | 0.800 | 0.775 | 0.780 | 0.795 | 0.790 | 0.157 | |
| SVM | 0.791 (0.742-0.842) | 0.675 | 0.850 | 0.818 | 0.723 | 0.740 | 0.188 | |
| Logistic regression | 0.750 (0.698-0.803) | 0.625 | 0.825 | 0.781 | 0.688 | 0.694 | 0.198 | |
| Naive Bayes | 0.751 (0.700-0.806) | 0.925 | 0.525 | 0.661 | 0.875 | 0.771 | 0.217 | |
| KNN | 0.658 (0.602-0.716) | 0.850 | 0.450 | 0.607 | 0.750 | 0.708 | 0.227 | |
| Validation | Random forest | 0.905 (0.870-0.940 | 0.800 | 0.880 | 0.870 | 0.820 | 0.835 | 0.152 |
| AdaBoost | 0.860 (0.820-0.903) | 0.770 | 0.750 | 0.760 | 0.765 | 0.765 | 0.169 | |
| SVM | 0.760 (0.710-0.812) | 0.650 | 0.820 | 0.790 | 0.700 | 0.710 | 0.205 | |
| Logistic regression | 0.735 (0.680-0.788) | 0.600 | 0.800 | 0.760 | 0.660 | 0.675 | 0.210 | |
| Naive Bayes | 0.740 (0.690-0.795) | 0.900 | 0.500 | 0.640 | 0.840 | 0.745 | 0.230 | |
| KNN | 0.630 (0.575-0.690) | 0.820 | 0.420 | 0.580 | 0.720 | 0.685 | 0.245 |
Liver biopsy remains the gold standard for diagnosing liver disease and grading hepatic fibrosis; however, its use has declined due to several limitations, including high cost, invasive nature, patient discomfort, potential procedural complications, and the requirement for specialized histopathological expertise. Consequently, a slew of non-invasive modalities has gained traction for the evaluation and staging of hepatic fibrosis and are advocated by the WHO. These methods incorporate either serum-based biomarkers - such as APRI, FIB-4, and GPR - or ultrasound-derived techniques, most notably transient elastography (FibroScan)[29,30]. The adoption of these approaches in clinical practice has substantially reduced the reliance on liver biopsy, particularly in individuals with an already established etiology of chronic liver disease[31,32].
The 2022 WHO guidelines for HCV emphasize on universal screening and widespread access to direct-acting antiviral therapy, in order to broaden treatment coverage irrespective of the stage of disease. These guidelines advocate the use of non-invasive tools (APRI, FIB-4, and FibroScan) to evaluate for significant hepatic fibrosis and cirrhosis, however, treatment eligibility is not dependent on degree of hepatic fibrosis, as treatment is recommended for all individuals with HCV[33,34].
In contrast, the WHO 2024 guidelines for chronic hepatitis B also advocate use of non-invasive tests such as APRI and FibroScan, particularly in resource-limited settings for the detection of significant hepatic fibrosis (≥ F2) or cirrhosis (F4), as it serves as an eligibility criterion to ensure that antiviral therapy is directed toward those at greatest risk of liver-related morbidity and mortality[35].
Numerous studies have demonstrated that serum lipid levels decline significantly in chronic viral hepatitis, particularly HCV. The hypolipidemic trend observed in HCV has a multifactorial etiology[36,37]. Despite this recognized association, lipid profiles are not routinely performed in such patients. As a consequence, the potential utility of lipid biomarkers, either alone or in combination with other biochemical parameters for evaluating hepatic fibrosis has not been adequately investigated. To date, most validated clinical scoring systems rely primarily on demographic factors (e.g., age), LFT and hematological parameters (platelet count), while the contribution of lipid markers in predicting hepatic fibrosis in HCV and other chronic liver diseases remains insufficiently explored[37,38]. To our knowledge, the Forns index is the only available non-invasive fibrosis score that incorporates a lipid profile parameter, namely serum cholesterol for estimation of risk of hepatic fibrosis. Developed for patients with HCV, it combines age, platelet count, GGT, and serum cholesterol to estimate the probability of significant hepatic fibrosis[39]. A Forns score < 4.2 indicates a low likelihood of significant fibrosis, whereas a score > 6.9 suggests a high probability of fibrosis. Values between 4.2 and 6.9 are con
The present study evaluated the predictive value of lipid biomarkers in conjunction with liver function parameters and platelet count in predicting significant hepatic fibrosis in patients with chronic HCV. The initial part of this study used conventional logistic regression to identify variables associated with hepatic fibrosis and subsequently derive a practical, reliable, and user-friendly FRS.
The FRS was developed by combining multiple parameters (platelet count, AST, ALP, GGT, serum albumin and serum lipid concentrations of total cholesterol and LDL), and its diagnostic performance for significant hepatic fibrosis was evaluated in the training cohort and internally validated against the validation cohort. The diagnostic performance of the FRS was subsequently compared with that of commonly used noninvasive fibrosis scores, including APRI, FIB-4 and GPR. In our study, the newly developed FRS demonstrated superior diagnostic performance, with an AUC of 0.83, compared with APRI (AUC = 0.59), FIB-4 (AUC = 0.65), and GPR (AUC = 0.67) at their respective optimal cutoff thresholds of 9, 1.5, 1.45, and 0.4.
The FIB-4 and APRI scores have demonstrated similar diagnostic performance for predicting significant hepatic fibrosis in patients with HCV in previous studies as they did in the current study. In a large retrospective cohort study comprising of 19510 chronic HCV patients, FIB-4 demonstrated the highest diagnostic performance (AUC = 0.70), followed by the King’s score (AUC = 0.70), while APRI showed a lower diagnostic performance (AUC = 0.68)[40].
This enhanced diagnostic performance of FRS may, in part, be due to integration of multiple complementary para
At a threshold of ≥ 9 points, the FRS yielded an optimal balance of sensitivity (71%) and specificity (85%). Clinically, this cut-off allows easy and meaningful risk stratification: Patients scoring ≥ 9 are more likely to have significant fibrosis and may warrant invasive assessment or close monitoring, whereas those below this value can be managed with routine follow-up. Although statistically derived, this threshold offers clear practical value for preliminary screening and prioritizing patients for further diagnostic workup.
In the second step, to further enhance diagnostic efficiency, we developed predictive models using ML algorithms. Given the potential concerns regarding overfitting and the potential optimism of ML approaches, 5-fold cross-validation was employed to improve model stability and robustness. Following multiple attempts, algorithms with consistently high diagnostic accuracy for significant hepatic fibrosis were identified. These models offer a concrete theoretical ground for subsequent development of practical, accurate, accessible, and clinically deployable fibrosis assessment tools, with pro
In comparison to existing conventional noninvasive fibrosis scores such as APRI, FIB-4, and GPR, both the logistic regression-derived FRS and the ML-based RF and AdaBoost models demonstrated superior diagnostic performance for detecting significant hepatic fibrosis. Moreover, the reliance of FRS on routine laboratory parameters enhances feasibility, accessibility and affordability particularly in low-resource settings, by reducing dependence on specialized and costly tests. These models may obviate the need for advanced diagnostic tests in low-risk patients, improving cost-effectiveness and accessibility, especially in resource-limited regions or economically disadvantaged populations.
Both the FRS and RF model yielded higher specificity than sensitivity, indicating strong utility in ruling out patients without significant fibrosis and minimizing misclassification. However, their moderately lower sensitivity suggests that mild cases of fibrosis may be under detected. The positive and negative predictive values of the models were comparable, indicating balanced diagnostic efficiency. Although high specificity is advantageous and desirable, sensitivity remains an area for improvement, potentially achievable through integration of emerging biomarkers or optimization and refinement of the algorithms in future investigations. It is plausible that owing to the complex interactions among liver function parameters, lipid profile indices, and platelet count, the non-linear ML models - RF and AdaBoost - outperformed linear models in predictive performance for fibrosis.
The results reflect that, within the context of this study, the RF model may be potentially more efficient, reliable, and precise method for diagnosing significant hepatic fibrosis in patients with HCV infection, compared with conventional scoring systems including FRS, APRI, FIB-4, and GPR.
Overall, the ML models in this study offered considerable promise as non-invasive tools for the assessment of significant hepatic fibrosis in HCV-infected individuals, particularly for screening high-risk populations and steering targeted diagnostic evaluation.
Wei et al[41] employed ML techniques to develop a non-invasive model for predicting hepatic fibrosis in patients with hepatitis B and C using same indicators as in FIB-4. They compared the diagnostic performance of multiple ML models and developed a novel fibrosis prediction tool, LiveBoost, which was evaluated against the conventional FIB-4 index. The authors reported that the gradient boosting ML model significantly outperformed FIB-4 in predicting advanced hepatic fibrosis and cirrhosis, achieving an AUC of 0.91, compared with 0.84 for FIB-4, thereby demonstrating higher discriminatory accuracy[41].
Song et al[42] investigated the application of ML models for the non-invasive prediction of advanced liver fibrosis in patients with chronic hepatitis B. Their findings showed that the RF algorithm achieved the highest predictive performance, with an AUC of 0.87, outperforming XGBoost, logistic regression, and SVM algorithms. Moreover, when compared with traditional fibrosis indices such as APRI (AUC of 0.58) and FIB-4 (AUC of 0.63), the RF model exhibited markedly superior diagnostic accuracy[42].
Some studies have also evaluated the role of ML-based algorithms for the non-invasive detection of liver fibrosis in non-alcoholic steatohepatitis, reporting that these models achieve significantly higher diagnostic accuracy than conventional clinical indices, thereby supporting their potential clinical utility in detection of hepatic fibrosis[43,44].
This study developed a novel FRS by integrating liver function indices, lipid profile markers, and platelet count, and validated its performance against three widely adopted non-invasive tools including APRI, FIB-4, and GPR. The FRS demonstrated superior diagnostic accuracy in comparison to these commonly used clinical scores. Furthermore, the use of ML approaches boosted diagnostic discrimination, with the RF algorithm yielding the highest diagnostic performance for detecting significant hepatic fibrosis among chronic HCV patients. Collectively, these findings suggest that the FRS offers a simple, practical, and clinically valuable alternative for estimating the likelihood of significant hepatic fibrosis, particularly within HCV-affected individuals.
The main limitation of the present study is the lack of liver biopsy as a reference standard to directly compare the diagnostic or predictive performance of the proposed score. Transient elastography is vulnerable to technical alterations and possesses inherent limitations that may impact fibrosis classification. Nevertheless, a growing shift in clinical practice over recent years has led to decline in the routine use of liver biopsy for detecting significant fibrosis, resulting in limited availability of biopsy as reference standard. Another limitation of the study is the absence of an external validation cohort, although internal validation was conducted by dividing the sample into training and validation sets. Another shortcoming of the study is retrospective design of the study. Additionally, the variations in laboratory testing protocols and the inherent heterogeneity of the disease may influence the model’s robustness and efficiency, potentially resulting in an overestimation of its generalizability.
Despite these limitations, the integration of lipid biomarkers with standard liver function indices and platelet count shows promise and can provide a practical and biologically plausible alternative tool for non-invasive fibrosis evaluation.
Future research should focus on multicenter external validation, include diverse patient populations to assess the real-world applicability of the model, and incorporate multidimensional factors - such as emerging molecular markers and imaging data - to develop a more comprehensive and robust predictive system.
| 1. | World Health Organization. Global hepatitis report 2024: action for access in low-and middle-income countries. 2024 Apr 9. [cited 7 December 2025]. Available from: https://digitallibrary.un.org/record/4043391?v=pdf. |
| 2. | Khatun M, Ray RB. Mechanisms Underlying Hepatitis C Virus-Associated Hepatic Fibrosis. Cells. 2019;8:1249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 121] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 3. | Fallowfield J, Hayes P. Pathogenesis and treatment of hepatic fibrosis: is cirrhosis reversible? Clin Med (Lond). 2011;11:179-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 4. | Chahal HS, Marseille EA, Tice JA, Pearson SD, Ollendorf DA, Fox RK, Kahn JG. Cost-effectiveness of Early Treatment of Hepatitis C Virus Genotype 1 by Stage of Liver Fibrosis in a US Treatment-Naive Population. JAMA Intern Med. 2016;176:65-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 118] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 5. | Rockey DC, Friedman SL. Fibrosis Regression After Eradication of Hepatitis C Virus: From Bench to Bedside. Gastroenterology. 2021;160:1502-1520.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (4)] |
| 6. | Elsharkawy A, Samir R, El-Kassas M. Fibrosis regression following hepatitis C antiviral therapy. World J Hepatol. 2022;14:1120-1130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 7. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis - 2021 update. J Hepatol. 2021;75:659-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1546] [Cited by in RCA: 1383] [Article Influence: 276.6] [Reference Citation Analysis (6)] |
| 8. | Stasi C, Milani S. Evolving strategies for liver fibrosis staging: Non-invasive assessment. World J Gastroenterol. 2017;23:191-196. [PubMed] [DOI] [Full Text] |
| 9. | Rungta S, Kumari S, Deep A, Verma K, Swaroop S. APRI and FIB-4 performance to assess liver fibrosis against predefined Fibroscan values in chronic hepatitis C virus infection. J Family Med Prim Care. 2021;10:4082-4088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 10. | Mendes LC, Stucchi RS, Vigani AG. Diagnosis and staging of fibrosis in patients with chronic hepatitis C: comparison and critical overview of current strategies. Hepat Med. 2018;10:13-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 11. | Kim WR, Berg T, Asselah T, Flisiak R, Fung S, Gordon SC, Janssen HL, Lampertico P, Lau D, Bornstein JD, Schall RE, Dinh P, Yee LJ, Martins EB, Lim SG, Loomba R, Petersen J, Buti M, Marcellin P. Evaluation of APRI and FIB-4 scoring systems for non-invasive assessment of hepatic fibrosis in chronic hepatitis B patients. J Hepatol. 2016;64:773-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 245] [Article Influence: 24.5] [Reference Citation Analysis (1)] |
| 12. | Liu X, Li H, Wei L, Tang Q, Hu P. Optimized cutoffs of gamma-glutamyl transpeptidase-to-platelet ratio, aspartate aminotransferase-to-platelet ratio index, and fibrosis-4 scoring systems for exclusion of cirrhosis in patients with chronic hepatitis B. Hepatol Commun. 2022;6:1664-1672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Surana P, Hercun J, Takyar V, Kleiner DE, Heller T, Koh C. Platelet count as a screening tool for compensated cirrhosis in chronic viral hepatitis. World J Gastrointest Pathophysiol. 2021;12:40-50. [PubMed] [DOI] [Full Text] |
| 14. | Elgretli W, Chen T, Kronfli N, Sebastiani G. Hepatitis C Virus-Lipid Interplay: Pathogenesis and Clinical Impact. Biomedicines. 2023;11:271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 15. | Tien PC. Hepatitis C Virus-Associated Alterations in Lipid and Lipoprotein Levels: Helpful or Harmful to the Heart? Clin Infect Dis. 2017;65:566-567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 16. | Bozdar K, Kumar P, Bajwa MA. Dyslipidemias in Hepatitis C Virus Infection at Isra University Hospital Hyderabad. Ann Pak Inst Med Sci. 2020;16:209-213. |
| 17. | Aizawa Y, Seki N, Nagano T, Abe H. Chronic hepatitis C virus infection and lipoprotein metabolism. World J Gastroenterol. 2015;21:10299-10313. [PubMed] [DOI] [Full Text] |
| 18. | Badawi R, Abu Rahma MZ, Ramadan HK, Soliman S, Mohareb DA, Hawash N, Elkafoury R, Abd-elsalam S. Lipid Profiles as Markers for the Severity of Liver Diseases in Cirrhotic Patients. Open Biomark J. 2021;11:93-98. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 19. | Miyazaki T, Honda A, Ikegami T, Saitoh Y, Hirayama T, Hara T, Doy M, Matsuzaki Y. Hepatitis C virus infection causes hypolipidemia regardless of hepatic damage or nutritional state: An epidemiological survey of a large Japanese cohort. Hepatol Res. 2011;41:530-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 20. | Ghadir MR, Riahin AA, Havaspour A, Nooranipour M, Habibinejad AA. The relationship between lipid profile and severity of liver damage in cirrhotic patients. Hepat Mon. 2010;10:285-288. [PubMed] |
| 21. | Baranova A, Lal P, Birerdinc A, Younossi ZM. Non-invasive markers for hepatic fibrosis. BMC Gastroenterol. 2011;11:91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 215] [Article Influence: 14.3] [Reference Citation Analysis (2)] |
| 22. | Kirk GD, Astemborski J, Mehta SH, Spoler C, Fisher C, Allen D, Higgins Y, Moore RD, Afdhal N, Torbenson M, Sulkowski M, Thomas DL. Assessment of liver fibrosis by transient elastography in persons with hepatitis C virus infection or HIV-hepatitis C virus coinfection. Clin Infect Dis. 2009;48:963-972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 122] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 23. | Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol. 2008;48:835-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1190] [Cited by in RCA: 1078] [Article Influence: 59.9] [Reference Citation Analysis (1)] |
| 24. | Castera L. Noninvasive methods to assess liver disease in patients with hepatitis B or C. Gastroenterology. 2012;142:1293-1302.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 446] [Article Influence: 31.9] [Reference Citation Analysis (2)] |
| 25. | Geng XX, Huang RG, Lin JM, Jiang N, Yang XX. Transient elastography in clinical detection of liver cirrhosis: A systematic review and meta-analysis. Saudi J Gastroenterol. 2016;22:294-303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 26. | Friedrich-Rust M, Ong MF, Martens S, Sarrazin C, Bojunga J, Zeuzem S, Herrmann E. Performance of transient elastography for the staging of liver fibrosis: a meta-analysis. Gastroenterology. 2008;134:960-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1164] [Cited by in RCA: 1059] [Article Influence: 58.8] [Reference Citation Analysis (3)] |
| 27. | Chon YE, Choi EH, Song KJ, Park JY, Kim DY, Han KH, Chon CY, Ahn SH, Kim SU. Performance of transient elastography for the staging of liver fibrosis in patients with chronic hepatitis B: a meta-analysis. PLoS One. 2012;7:e44930. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 229] [Article Influence: 16.4] [Reference Citation Analysis (1)] |
| 28. | Rinaldi L, Giorgione C, Mormone A, Esposito F, Rinaldi M, Berretta M, Marfella R, Romano C. Non-Invasive Measurement of Hepatic Fibrosis by Transient Elastography: A Narrative Review. Viruses. 2023;15:1730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (2)] |
| 29. | Liguori A, Zoncapè M, Casazza G, Easterbrook P, Tsochatzis EA. Staging liver fibrosis and cirrhosis using non-invasive tests in people with chronic hepatitis B to inform WHO 2024 guidelines: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2025;10:332-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 33.0] [Reference Citation Analysis (1)] |
| 30. | Lurie Y, Webb M, Cytter-Kuint R, Shteingart S, Lederkremer GZ. Non-invasive diagnosis of liver fibrosis and cirrhosis. World J Gastroenterol. 2015;21:11567-11583. [PubMed] [DOI] [Full Text] |
| 31. | Harrison SA. Utilization of FibroScan Testing in Hepatitis C Virus Management. Gastroenterol Hepatol (N Y). 2015;11:187-189. [PubMed] |
| 32. | Castéra L, Vergniol J, Foucher J, Le Bail B, Chanteloup E, Haaser M, Darriet M, Couzigou P, De Lédinghen V. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology. 2005;128:343-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1980] [Cited by in RCA: 1835] [Article Influence: 87.4] [Reference Citation Analysis (2)] |
| 33. | Gheorghe G, Bungău S, Ceobanu G, Ilie M, Bacalbaşa N, Bratu OG, Vesa CM, Găman MA, Diaconu CC. The non-invasive assessment of hepatic fibrosis. J Formos Med Assoc. 2021;120:794-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 34. | Agbim U, Asrani SK. Non-invasive assessment of liver fibrosis and prognosis: an update on serum and elastography markers. Expert Rev Gastroenterol Hepatol. 2019;13:361-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 97] [Article Influence: 13.9] [Reference Citation Analysis (1)] |
| 35. | Wong GL, Lemoine M. The 2024 updated WHO guidelines for the prevention and management of chronic hepatitis B: Main changes and potential implications for the next major liver society clinical practice guidelines. J Hepatol. 2025;82:918-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 46.0] [Reference Citation Analysis (2)] |
| 36. | Lai JC, Liang LY, Wong GL. Noninvasive tests for liver fibrosis in 2024: are there different scales for different diseases? Gastroenterol Rep (Oxf). 2024;12:goae024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (3)] |
| 37. | Joseph J. Serum Marker Panels for Predicting Liver Fibrosis - An Update. Clin Biochem Rev. 2020;41:67-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 38. | Schiavon Lde L, Narciso-Schiavon JL, de Carvalho-Filho RJ. Non-invasive diagnosis of liver fibrosis in chronic hepatitis C. World J Gastroenterol. 2014;20:2854-2866. [PubMed] [DOI] [Full Text] |
| 39. | Forns X, Ampurdanès S, Llovet JM, Aponte J, Quintó L, Martínez-Bauer E, Bruguera M, Sánchez-Tapias JM, Rodés J. Identification of chronic hepatitis C patients without hepatic fibrosis by a simple predictive model. Hepatology. 2002;36:986-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 793] [Cited by in RCA: 729] [Article Influence: 30.4] [Reference Citation Analysis (5)] |
| 40. | El-Kassas M, Elakel W, Elsharkawy A, Asem N, Abu-Elfatth A, Mostafa A, Abdelazeem A, El-Serafy M, Ibrahem M, Ghanem EA, Abdeen N, Doss W, Esmat G, Abdeltawab D. Comparison of different noninvasive scores for assessing hepatic fibrosis in a cohort of chronic hepatitis C patients. Sci Rep. 2024;14:29544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (5)] |
| 41. | Wei R, Wang J, Wang X, Xie G, Wang Y, Zhang H, Peng CY, Rajani C, Kwee S, Liu P, Jia W. Clinical prediction of HBV and HCV related hepatic fibrosis using machine learning. EBioMedicine. 2018;35:124-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Reference Citation Analysis (0)] |
| 42. | Song J, Ma N, Aini R, Yang Y. A machine learning model for non-invasive prediction of advanced liver fibrosis in patients with chronic hepatitis B. Am J Transl Res. 2025;17:4939-4951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 43. | Jamialahmadi T, Looha MA, Jangjoo S, Emami N, Abdalla MA, Ganjali M, Salehabadi S, Karav S, Sathyapalan T, Eid AH, Jangjoo A, Sahebkar A. Predictive performance of noninvasive factors for liver fibrosis in severe obesity: a screening based on machine learning models. J Diabetes Metab Disord. 2025;24:54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 44. | Xiong FX, Sun L, Zhang XJ, Chen JL, Zhou Y, Ji XM, Meng PP, Wu T, Wang XB, Hou YX. Machine learning-based models for advanced fibrosis in non-alcoholic steatohepatitis patients: A cohort study. World J Gastroenterol. 2025;31:101383. [PubMed] [DOI] [Full Text] |