Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.117040
Revised: December 17, 2025
Accepted: January 22, 2026
Published online: March 27, 2026
Processing time: 119 Days and 22.8 Hours
Despite the success of direct-acting antivirals, a large global population with es
To evaluated a multimarker panel, including free triiodothyronine (FT3), platelet count (PLT), international normalized ratio (INR), and portal vein diameter (PVD), for the non-invasive stratification of variceal bleeding risk.
A case-control study was conducted on 93 participants, categorized into three groups: 31 chronic HCV cirrhotic patients with variceal bleeding (group I), 31 chronic HCV cirrhotic patients without variceal bleeding (group II), and 31 healthy controls (group III). Thyroid function tests (thyroid-stimulating hormone, FT3, free thyroxine), PLT, INR, spleen size, and PVD were assessed. Doppler ultrasound was used to evaluate portal venous blood flow, while upper gastrointestinal endoscopy determined the variceal grades. Statistical analysis included one-way analysis of variance, Pearson correlation, multivariate ordinal logistic regression, and receiver operating characteristic curve analysis.
FT3 levels were significantly lower in cirrhotic patients, with the lowest levels in group I (P < 0.01). Higher variceal grades (III/IV) were associated with lower FT3, lower PLT, and higher INR (P < 0.01). In multivariate analysis, lower FT3, lower PLT, and higher INR were independent predictors of variceal severity (all P < 0.01). FT3 < 2.5 pmol/L predicted high-grade varices with an area under the curve (AUC) of 0.88 (sensitivity 85.7%, specificity 78.3%). PVD ≥ 13 mm also showed strong predictive value (AUC = 0.85). A novel Variceal Risk Score (VRS) integrating FT3, PLT, INR, and PVD demonstrated outstanding accuracy (AUC = 0.94).
This study validates a multimarker approach, identifying a core panel of non-invasive predictors-FT3, PLT, and INR for variceal bleeding risk. The derived VRS can accurately identify high-risk cirrhotic patients, suggesting its potential use as a triage tool to make endoscopic screening programs more efficient.
Core Tip: This study validates a novel, non-invasive multimarker approach for identifying chronic hepatitis C cirrhotic patients at high risk for variceal bleeding. We identified a core panel of independent predictors: Low free triiodothyronine levels, low platelet count, and high international normalized ratio. A composite Variceal Risk Score integrating these with portal vein diameter demonstrated outstanding accuracy (area under the curve = 0.94). This strategy can efficiently triage patients for endoscopic screening, potentially reducing procedural burden in resource-limited settings.
- Citation: Othman AAA, Elsharkawy L, Saleh MW, Mohamed MM, Elsayed FMA. Multimarker model for non-invasive assessment of variceal bleeding in chronic hepatitis C cirrhotic patients. World J Hepatol 2026; 18(3): 117040
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/117040.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.117040
While the advent of direct-acting antivirals has dramatically reduced the incidence of new hepatitis C virus (HCV) infections[1], a substantial global population with established HCV-related cirrhosis persists. These patients remain at lifelong risk for the complications of portal hypertension, such as variceal bleeding, regardless of virological cure[2,3]. In Egypt, despite a highly successful public health campaign[4], the legacy of a high historical prevalence has left a large cohort of patients with advanced liver disease requiring long-term monitoring[5]. This reality underscores the critical need for accessible, non-invasive tools to stratify bleeding risk.
Thyroid dysfunction, particularly low triiodothyronine (T3) syndrome, is a well-documented extrahepatic mani
Varices result directly from portal hypertension, which involves increased intrahepatic resistance and hyperdynamic circulation[10]. Thyroid hormones, particularly T3, influence vascular tone and hemodynamics[11]. However, the potential role of thyroid dysfunction in variceal pathophysiology remains underexplored, with few studies investigating this relationship and none specifically in the Egyptian population, where HCV-related liver disease burden is ex
Given the exceptionally high historical burden of HCV in Egypt, this population represents a critical group for refining risk stratification strategies. To establish a proof-of-concept within a homogenous cohort and minimize confounding from mixed etiologies [such as alcohol-related or metabolic dysfunction-associated steatotic liver disease (MASLD)] or virological status, we focused exclusively on patients with HCV-related cirrhosis and active viremia.
Current methods for assessing variceal severity, such as endoscopy, are invasive and may not be feasible in resource-limited settings. This study, therefore, aimed to develop and validate a non-invasive multimarker model incorporating thyroid function [free T3 (FT3) levels], hematological parameters [platelet count (PLT), international normalized ratio (INR)], and portal hemodynamics [portal vein diameter (PVD), spleen size] to improve the non-invasive assessment of variceal severity. We hypothesize that lower FT3 levels, reduced PLT, and increased PVD and spleen size are inde
This was a collaborative study conducted at Zagazig University Hospital, with contributing investigators from the Faculty of Medicine at Suez University, Egypt, between December 2021 and December 2022. The study included 93 participants divided into three groups.
Group I: Included 31 chronic HCV cirrhotic patients with variceal bleeding admitted to the emergency gastroenterology unit. Their ages ranged from 44 years to 78 years with a mean ± SD of 57 ± 9.46. There were 9 female (29%) and 22 male (71%). Furthermore, none of these patients were on secondary prophylaxis with non-selective beta-blockers (NSBB) before the index bleeding event. For this group, all laboratory investigations [including thyroid function tests, complete blood count (CBC), and INR], radiological assessments (ultrasound and Doppler), and blood sample collection were performed upon admission, before any therapeutic endoscopic intervention (band ligation or sclerotherapy) during the same hospital stay. This ensured that the measured biomarkers reflected the pre-intervention physiological state associated with acute variceal hemorrhage. Patients presenting with concurrent acute systemic infection (e.g., spontaneous bacterial peritonitis, pneumonia) or hemodynamic instability primarily attributable to causes other than acute variceal hemorrhage (e.g., septic or cardiogenic shock) were excluded from group I.
Group II: Included 31 chronic HCV cirrhotic patients without variceal bleeding who follow up in the outpatient clinic. Critically, no patients in group II were receiving primary prophylaxis with NSBB or nitrates at the time of enrollment, ensuring that the assessed portal hemodynamics and variceal status reflected the natural, unmodified history of the disease. Their ages ranged from 49 years to 76 years with a mean ± SD of 60 ± 6.59. There were 12 female (38.7%) and 19 male (61.3%). For all participants in group II, blood sampling and clinical assessments were performed during a scheduled, routine outpatient visit. To ensure measurements reflected a stable baseline state, patients were excluded if they had a history of acute decompensation (e.g., new-onset ascites, hepatic encephalopathy), overt infection, or blood transfusion within the 4 weeks preceding enrollment.
Group III: Included 31 apparently healthy control persons. Their ages ranged from 45 years to 73 years with a mean ± SD of 56 ± 6.52. There were 17 female (54.9%) and 14 male (45.1%). The inclusion of a healthy control group was essential for two primary reasons: First, to establish a robust, local reference range for FT3 and other parameters, as 'normal' levels can vary between populations and assay methods; and second, to quantitatively demonstrate the progressive decline in FT3 from health (group III) to compensated cirrhosis (group II) and finally to decompensated cirrhosis with variceal bleeding (group I). This gradient strengthens the argument for FT3 as a marker of disease progression, not just a binary indicator of cirrhosis.
The sample size was calculated using the z-test for two proportions, based on an expected prevalence of thyroid dysfunction in cirrhotic patients of 30%[12], a margin of error of 5%, and a confidence level of 95%. The minimum required sample size was estimated to be 31 participants per group to ensure adequate statistical power. Consecutive eligible patients who presented during the study period were screened and enrolled until the pre-specified sample size for each group was fulfilled.
The two cirrhotic patient groups were well-matched at baseline, with no significant difference in age or non-invasive liver fibrosis scores [fibrosis-4 (FIB-4) index and aspartate aminotransferase (AST) to platelet ratio index (APRI)], ensuring comparability for the analysis of complications.
The participant selection process is detailed in Figure 1. Between December 2021 and December 2022, 162 patients with chronic HCV and liver cirrhosis were assessed for eligibility. After applying the pre-defined exclusion criteria, 69 patients were excluded to establish a homogeneous study cohort. The most frequent reasons for exclusion were non-HCV liver disease etiologies [n = 15, including 8 cases of hepatocellular carcinoma (HCC)], a prior sustained virological response (SVR) to HCV treatment (n = 12), and significant alcohol use (n = 10). Consecutive enrollment continued until the predetermined sample size of 31 participants per group was met, resulting in a final study population of 93 participants.
The study design and reporting adhered to the STROBE guidelines for case-control studies to ensure methodological transparency and reproducibility
The study protocol was approved by the Institutional Review Board of Zagazig University (No. ZU-IRB#1897). Before enrolling, all participants provided written informed consent. The study adhered to the principles of the Declaration of Helsinki and ensured the confidentiality of all patient data.
Participants in groups I and II were included if they had a confirmed diagnosis of chronic HCV infection, as evidenced by detectable HCV RNA using polymerase chain reaction (PCR), and liver cirrhosis diagnosed based on clinical, biochemical, and radiological criteria, including ultrasound findings of a shrunken liver with a nodular surface and splenomegaly. For group I, a history of variceal bleeding confirmed by upper gastrointestinal endoscopy was required. For group II, participants had no history of variceal bleeding or previous endoscopy. Group III consisted of healthy control individuals. To rigorously confirm their health status, all control participants underwent the same laboratory, virological (including negative HCV RNA by PCR and HBsAg), and radiological investigations as the patient groups. This confirmed the absence of liver disease, thyroid dysfunction, and any other major active medical conditions. They served as age- and sex-matched controls.
The rationale for HCV-only (homogeneity to avoid confounding by different etiologies), relevance to the Egyptian population, and note that extrapolation to other etiologies is a limitation and future direction.
Participants were excluded based on the following criteria to ensure a homogenous study cohort and to eliminate potential confounding factors, including, but not limited to.
Etiology of liver disease: Participants were excluded if they had non-HCV-related liver disease, including hepatitis B virus infection, autoimmune hepatitis, bilharzial infection, drug-induced liver injury, or HCC. This exclusion was critical to maintain a cohort with a single, uniform etiology (active HCV viremia), as different liver diseases have distinct pathophysiologies and natural histories that could confound the relationship between the studied biomarkers and portal hypertensive complications. The exclusion of HCC was particularly important as it significantly alters systemic and portal hemodynamics, synthetic function, and clinical prognosis. Furthermore, patients with a history of HCV who had achieved SVR after antiviral therapy were excluded to specifically investigate biomarker relationships in the context of ongoing viral replication and its associated inflammatory milieu.
Concurrent metabolic and endocrine conditions: Individuals with a history of significant alcohol-related liver disease (defined as alcohol intake > 30 g/day for men and > 20 g/day for women), MASLD (indicated by a body mass index > 30 kg/m² or uncontrolled type 2 diabetes with HbA1c > 8%), thyroid dysfunction, thyroidectomy, the use of thyroid hormone replacement therapy, or diabetes mellitus were excluded. To definitively rule out primary autoimmune thyroid disease as a cause of thyroid dysfunction, all participants with abnormal thyroid function tests were screened for anti-thyroid peroxidase antibodies, and any patient with positive antibodies was excluded. These conditions can directly alter thyroid hormone metabolism, insulin resistance pathways, and systemic inflammation, all of which could independently influence the progression of portal hypertension and variceal development, thereby confounding the study's primary aims.
Hemodynamic and procedural confounders: To ensure that the assessed portal hemodynamics reflected the natural history of HCV-related cirrhosis, we excluded patients with conditions associated with hyperdynamic circulation independent of portal hypertension (e.g., aortic regurgitation, arteriovenous fistulae) and those with a history of previous interventions for portal hypertension or varices (such as endoscopic sclerotherapy, band ligation, transjugular intrahepatic portosystemic shunt, or splenectomy). These interventions and conditions directly and permanently alter portal pressure, collateral formation, and splenic dynamics.
Pharmacological confounders: Patients taking medications known to significantly alter thyroid function or systemic hemodynamics, such as amiodarone or systemic corticosteroids, were excluded. Additionally, to prevent confounding of coagulation parameters, patients on any anticoagulant (e.g., warfarin, direct oral anticoagulants) or antiplatelet (e.g., aspirin, clopidogrel) therapy were also excluded.
Acute clinical conditions: Patients with recent acute decompensation (within 4 weeks), active systemic infection, recent blood transfusion, or hemodynamic instability were excluded to prevent acute confounding of biomarker levels.
Special populations: Pregnant women were excluded due to the profound physiological hormonal changes during pregnancy that can significantly confound thyroid function test results.
All participants underwent a thorough clinical evaluation, including.
Medical history: Focus on HCV diagnosis, liver disease duration, and variceal bleeding history.
Physical examination: Assessment for signs of chronic liver disease (e.g., jaundice, ascites, spider angiomas) and thyroid dysfunction (e.g., goiter, tremors).
Child-Pugh score: Used to classify the severity of liver dysfunction based on clinical and laboratory parameters (e.g., ascites, encephalopathy, bilirubin, albumin, INR)[13].
Five mL of fresh venous blood were collected from each participant after an overnight fast, with 2.5 mL of ethylenediaminetetraacetic acid for CBC analysis and HbA1c analysis, and 2.5 mL drawn without anticoagulants for other serum testing. The samples were then centrifuged at 3000 rpm for 15 minutes, and serum aliquots were stored at -20 °C until further analysis. All samples were promptly sent to the laboratory.
Liver and renal function tests: Serum levels of total and direct bilirubin, albumin, alanine aminotransferase (ALT), AST, and creatinine were measured using an automated clinical chemistry analyzer (COBAS 500c, Roche Diagnostics).
Hematological tests: CBC including PLT was performed using an automated hematology analyzer (Sysmex KX-21). The coagulation profile: Prothrombin time and INR was assessed using an automated coagulation analyzer (Sysmex CS-2100i).
Thyroid function tests: Serum levels of thyroid-stimulating hormone (TSH), FT3, and free thyroxine (FT4) were measured using electrochemiluminescence immunoassay on a COBAS e 411 analyzer (Roche Diagnostics). To rule out primary thyroid disease, anti-thyroid peroxidase antibodies were also assessed for all participants; any patient with positive antibodies was excluded from the study.
Diagnosis of diabetes mellitus: Fasting and postprandial blood glucose levels were measured. HbA1c was assessed using a COBAS 6000 analyzer to exclude diabetic patients.
Virological and autoimmune markers: Anti-HCV antibodies and HBsAg were tested using enzyme-linked immu
Assessment of liver disease severity: To objectively verify the comparability of the two cirrhotic groups (group I and group II) in terms of their underlying fibrotic burden, the FIB-4 index and APRI were calculated from baseline laboratory data. The FIB-4 index was calculated using the standard formula: [Age (years) × AST (U/L)]/[PLT (109/L) × ALT1/2 (U/L)]. The APRI score was calculated as: [(AST/upper limit of normal)/PLT (109/L)] × 100. These indices were used as surrogates for disease chronicity, acknowledging the challenge in precisely determining the duration of HCV infection.
Abdominal ultrasound and doppler: All subjects underwent examination with a 3.5 MHz transducer (Philips iU22). Liver size, spleen bipolar diameter, and PVD were measured. Liver enlargement was defined as > 2 cm below the right costal margin without deep inspiration. Cirrhotic morphology was identified by a coarse nodular echo pattern, shrunken size, and predominant caudate lobe[14]. PVD was measured where it crosses the inferior vena cava during quiet respiration; > 13 mm indicated portal hypertension[15]. Splenomegaly was defined as a bipolar diameter > 130 mm at the left midaxillary line[16]. Hilar varices and prominent splenic veins (> 10 mm) were noted. The abdomen and pelvis were scanned for ascites. Portal venous flow parameters (peak systolic velocity, flow volume) were assessed via Doppler ultrasound.
Upper gastrointestinal endoscopy: All patients in group I and group II underwent examination using a Pentax EPK-I 5000 videoscope. The grading of esophageal varices was performed according to the Thakeb classification[17], which is a recognized endoscopic classification system in Egyptian clinical practice and was the standardized institutional protocol during the study period. This system classifies varices as: Grade I (small, straight), grade II (moderate-sized, clubbed), grade III (large, tortuous), and grade IV (varices with dilated capillaries). For the purpose of this study and in line with the principle of risk stratification in contemporary guidelines, high-grade or high-risk varices were defined as Thakeb grade III or IV. For the purpose of this study and in alignment with contemporary international risk stratification principles, high-grade or high-risk varices were defined as Thakeb grade III or IV, corresponding to large varices warranting prophylaxis in guidelines such as Baveno/EASL.
Classification of gastric varices: Gastric varices were classified according to the Sarin classification[18]. For this study, fundal varices (FV) were defined as those located in the gastric fundus, encompassing both gastroesophageal varices type 2 (GOV2) and isolated gastric varices type 1 (IGV1).
Data were analyzed using SPSS version 23 (IBM Corp., Armonk, NY, United States). Continuous variables were expressed as mean ± SD, and categorical variables as n (%). The normality of continuous variables was assessed using the Shapiro-Wilk test. Data with non-normal distribution were analyzed using non-parametric tests. Comparisons between groups were performed using one-way analysis of variance (ANOVA) for continuous variables, followed by Scheffe’s post-hoc test for pairwise comparisons due to its robustness in controlling type I error rates. The χ2 test was used for categorical variables. Spearman’s rank correlation was used to assess relationships between thyroid hormone levels, variceal grades, and other clinical parameters, as it is appropriate for ordinal data and does not assume normality.
The model for end-stage liver disease (MELD) score was calculated for all cirrhotic patients using the standard formula: MELD = 3.78 × ln [serum bilirubin (mg/dL)] + 11.2 × ln (INR)+ 9.57 × ln [serum creatinine (mg/dL)] + 6.43.
Multivariate ordinal logistic regression was performed to identify independent predictors of variceal severity. Initial models included established scores (Child-Pugh, MELD), while the final model was constructed to avoid multicollinearity and included FT3, PLT, INR, bleeding status, age, and sex. Multicollinearity was assessed using variance inflation factors (VIF). To ensure the robustness of our findings, we performed several validation steps. All multivariate models were adjusted for age and sex as standard covariates. A sensitivity analysis was conducted by re-running the final multivariate model after excluding patients with documented non-hepatic chronic comorbidities (n = 4) to assess potential confounding. Internal validation of the Variceal Risk Score (VRS) was performed using 1000 bootstrap samples with bias correction, which also served as an assessment of model stability against sampling variability. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the diagnostic accuracy of FT3 levels, variceal grade, and PVD in predicting high-grade varices. The optimal cut-off values were determined by maximizing sensitivity and specificity, and the area under the curve (AUC) was calculated to assess overall diagnostic performance. Effect sizes were calculated using Cohen’s d for continuous variables and Cramer’s V for categorical variables.
To synthesize the identified independent predictors into a clinically applicable tool, a composite VRS was developed. A binary logistic regression model was constructed with the presence of high-grade varices (grades III/IV) as the dependent variable and FT3, PLT, INR, and PVD as covariates. The VRS was calculated as a weighted combination based on the logistic regression coefficients. The diagnostic performance of the VRS for predicting high-grade varices was sub
The demographic and clinical characteristics of the participants are summarized in Table 1. The two cirrhotic patient groups (I and II) were similar in age and sex distribution. Furthermore, both groups had similarly elevated non-invasive fibrosis scores (FIB-4: 4.5 vs 4.3, P = 0.48; APRI: 2.1 vs 1.9, P = 0.25), confirming that all patients had established, severe cirrhosis. As anticipated in this case-control design, group I (with variceal bleeding) exhibited features of more advanced liver dysfunction, including a significantly different distribution of Child-Pugh classes (P < 0.01) and higher MELD scores (16.8 ± 4.5 vs 12.1 ± 3.8, P < 0.01; see Table 2). These differences in overall liver function were rigorously accounted for in the subsequent multivariate regression analyses to identify independent predictors specific to variceal severity.
| Parameter | Group I (n = 31) | Group II (n = 31) | Group III (n = 31) | P value1 |
| Age (years) | 57 ± 9.46 | 60 ± 6.59 | 56 ± 6.52 | 0.06 |
| FIB-4 index | 4.5 ± 1.2 | 4.3 ± 1.1 | NA | 0.48 |
| APRI score | 2.1 ± 0.7 | 1.9 ± 0.6 | NA | 0.25 |
| Sex (male/female) | 22/9 | 19/12 | 14/17 | 0.08 |
| Child-Pugh score | < 0.001 | |||
| Class A | 7 (22.6) | 9 (29.1) | ||
| Class B | 18 (58.0) | 17 (54.8) | ||
| Class C | 6 (19.4) | 5 (16.1) |
| Parameter | Group I (n = 31) | Group II (n = 31) | Group III (n = 31) | P value (I vs II)1 | P value (I vs III)1 | P value (II vs III)1 |
| WBCs (× 103/μL) | 8.78 ± 5.75 | 8.55 ± 6.94 | 7.58 ± 2.24 | 0.89 (NS) | 0.28 (NS) | 0.46 (NS) |
| Hb (g/dL) | 9.10 ± 1.34 | 9.83 ± 1.49 | 12.31 ± 1.86 | 0.04 (S) | < 0.01 (HS) | < 0.01 (HS) |
| PLT (× 103/μL) | 97.71 ± 35.82 | 123.00 ± 51.61 | 267.97 ± 71.24 | 0.03 (S) | < 0.01 (HS) | < 0.01 (HS) |
| INR | 1.47 ± 0.28 | 1.39 ± 0.37 | 1.04 ± 0.15 | 0.36 (NS) | < 0.01 (HS) | < 0.01 (HS) |
| Albumin (g/dL) | 2.34 ± 0.53 | 2.62 ± 0.68 | 4.05 ± 0.58 | 0.08 (NS) | < 0.01 (HS) | < 0.01 (HS) |
| Bilirubin (mg/dL) | 2.14 ± 2.00 | 2.44 ± 2.64 | 0.88 ± 1.15 | 0.62 (NS) | 0.004 (HS) | 0.004 (HS) |
| ALT (U/L) | 46.2 ± 15.8 | 42.5 ± 13.9 | 30.1 ± 9.8 | 0.28 (NS) | < 0.01 (HS) | 0.02 (S) |
| AST (U/L) | 58.7 ± 18.4 | 55.1 ± 16.2 | 33.9 ± 10.7 | 0.37 (NS) | < 0.01 (HS) | 0.01 (S) |
| Creatinine (mg/dL) | 1.32 ± 0.41 | 0.94 ± 0.22 | 0.82 ± 0.18 | < 0.01 (HS) | < 0.01 (HS) | 0.03 (S) |
| MELD score | 16.8 ± 4.5 | 12.1 ± 3.8 | < 0.01 (HS) |
Table 2 presents the laboratory findings across the three groups. Significant differences were observed in hemoglobin (Hb), PLT, INR, albumin, bilirubin, ALT, AST, and white blood cell count (WBCs) between the cirrhotic groups (groups I and II) and the control group (group III). Specifically, group I had a significantly lower Hb (9.10 ± 1.34 g/dL) and PLT [(97.71 ± 35.82) × 103/μL] compared to group III [Hb: 12.31 ± 1.86 g/dL, PLT: (267.97 ± 71.24) × 103/μL; P < 0.01]. Similarly, INR was significantly higher in group I (1.47 ± 0.28) compared to group III (1.04 ± 0.15; P < 0.01). Albumin levels were significantly lower in group I (2.34 ± 0.53 g/dL) compared to group III (4.05 ± 0.58 g/dL; P < 0.01). Bilirubin levels were also significantly higher in group I and group II compared to group III (P < 0.01). ALT and AST levels were significantly higher in group I (ALT: 46.2 ± 15.8 U/L; AST: 58.7 ± 18.4 U/L) compared to group III (ALT: 30.1 ± 9.8 U/L; AST: 33.9 ± 10.7 U/L; P < 0.01). No significant differences were observed in WBC counts across the groups (P > 0.05). Pairwise comparisons using Scheffe’s post-hoc test revealed that group I and group II differed significantly in Hb (P = 0.04) and PLT (P = 0.03), but not in INR, albumin, bilirubin, ALT, or AST (P > 0.05). As expected, serum creatinine and the calculated MELD score were also significantly higher in group I compared to group II (P < 0.01), reflecting the more advanced liver and potential renal dysfunction in patients presenting with variceal bleeding (Table 2).
Thyroid function tests revealed significant alterations in FT3 levels among cirrhotic patients. Group I had significantly lower FT3 levels (2.34 ± 0.53 pmol/L) compared to group III (4.05 ± 0.58 pmol/L; P < 0.01). Similarly, group II exhibited lower FT3 levels (2.62 ± 0.68 pmol/L) compared to controls (P < 0.01). No significant differences were observed in TSH or FT4 levels across the groups (P > 0.05). Pairwise comparisons using Scheffe’s post-hoc test confirmed that FT3 levels in group I and group II were significantly lower than in group III (P < 0.01), but there was no significant difference between group I and group II (P = 0.83) (Table 3).
| Parameter | Group I (n = 31) | Group II (n = 31) | Group III (n = 31) | P value (I vs II)1 | P value (I vs III)1 | P value (II vs III)1 |
| TSH (μIU/mL) | 1.22 ± 1.47 | 1.41 ± 1.75 | 1.70 ± 1.40 | 0.65 (NS) | 0.47 (NS) | 0.72 (NS) |
| FT3 (pmol/L) | 2.34 ± 0.53 | 2.62 ± 0.68 | 4.05 ± 0.58 | 0.83 (NS) | < 0.01 (HS) | < 0.01 (HS) |
| FT4 (pmol/L) | 14.35 ± 2.87 | 13.22 ± 5.48 | 15.29 ± 2.28 | 0.31 (NS) | 0.15 (NS) | 0.06 (NS) |
Table 4 summarizes the radiological findings, including spleen size and PVD. Group I had a significantly larger spleen size (16.95 ± 1.99 cm) and PVD (13.33 ± 2.05 mm) compared to group III (spleen size: 12.47 ± 0.68 cm, PVD: 10.45 ± 0.59 mm; P < 0.01). Similarly, group II had a larger spleen size (14.10 ± 1.87 cm) and PVD (11.95 ± 2.01 mm) compared to group III (P < 0.01). Pairwise comparisons using Scheffe’s post-hoc test revealed significant differences between group I and group II in spleen size (P < 0.01) and PVD (P < 0.01), indicating progressive changes in portal hemodynamics with disease severity (Table 4).
The distribution and classification of variceal patterns in the cirrhotic cohort are detailed in Table 5. Esophageal varices were the most common pattern in both groups. The prevalence of FV, encompassing both IGV1 and GOV2, was significantly higher in patients with a history of bleeding (group I, 29.0%) compared to those without (group II, 12.9%). Notably, the combined pattern of both esophageal and FV (GOV2) was over twice as frequent in group I. All patients in group I presented with acute variceal hemorrhage confirmed on endoscopy, with the bleeding source identified as esophageal varices in all cases where they were present.
| Variceal pattern | Group I (with bleeding) (n = 31) | Group II (without bleeding) (n = 31) | Total (n = 62) |
| Esophageal varices only | 22 (71.0) | 27 (87.1) | 49 (79.0) |
| Fundal varices only (IGV1) | 2 (6.5) | 1 (3.2) | 3 (4.8) |
| Both esophageal and fundal varices (GOV2) | 7 (22.6) | 3 (9.7) | 10 (16.1) |
| Total patients with fundal varices | 9 (29.0) | 4 (12.9) | 13 (21.0) |
Endoscopic findings revealed that group I had higher grades of oesophageal varices (OV) and a higher prevalence of FV compared to group II. Specifically, 38.7% of group I patients had grade III varices, compared to only 9.7% in group II (P < 0.01). Additionally, 29% of group I patients had FV, compared to 12.9% in group II (P < 0.01) (Table 6).
All 31 patients in group I presented with acute variceal hemorrhage confirmed on urgent endoscopy. As per standard management protocols, therapeutic intervention was required for hemostasis in all cases. During the index endoscopy, 28 patients (90.3%) underwent endoscopic band ligation for esophageal varices, while the remaining 3 patients (9.7%) received sclerotherapy for the management of FV. These interventions were performed after all study-related biomarker assessments were completed.
Relationship between OV grades and different parameters in group I and group II: To evaluate how key clinical parameters varied with increasing variceal severity, we analyzed their distribution across OV grades within each cirrhotic group (Table 6). The analysis revealed significant trends for FT3 levels, PLT, INR, spleen size, and PVD across different OV grades, with post-hoc tests confirming the most pronounced changes at higher grades (III/IV). These consistent patterns provided the rationale for selecting these specific parameters for inclusion in subsequent multivariate regression analyses to identify independent predictors (Table 7).
| Parameter | Group I | Group II | ||||
| F value | P value | Significant pairs (Scheffe test) | F value | P value | Significant pairs (Scheffe test) | |
| TSH (μU/mL) | 1.16 | 0.35 | NS | 1.76 | 0.18 | NS |
| FT3 (pmol/L) | 7.96 | 0.001 | Grade 1 vs 3, grade 2 vs 3, grade 3 vs 4 (P < 0.05) | 4.55 | 0.01 | Grade 0 vs 3, grade 1 vs 3 (P < 0.05) |
| FT4 (pmol/L) | 1.62 | 0.21 | NS | 1.32 | 0.29 | NS |
| PLT (× 103/μL) | 3.96 | 0.02 | Grade 1 vs 3 (P < 0.05) | 7.60 | 0.00 | Grade 0 vs 3, grade 1 vs 3 (P < 0.05) |
| INR | 3.52 | 0.03 | Grade 2 vs 4 (P < 0.05) | 6.88 | 0.00 | Grade 1 vs 4, grade 2 vs 4 (P < 0.05) |
| Spleen size (cm) | 3.31 | 0.04 | Grade 1 vs 3 (P < 0.05) | 6.58 | 0.00 | Grade 0 vs 3, grade 1 vs 3 (P < 0.05) |
| PVD (mm) | 3.64 | 0.02 | Grade 1 vs 4 (P < 0.05) | 8.40 | 0.00 | Grade 0 vs 3, grade 1 vs 3 (P < 0.05) |
Relationship between FV and different parameters in group I and group II:Table 8 shows no statistically significant relationship between the presence of FV and thyroid function parameters (TSH, FT3, and FT4) or liver-related parameters (PLT, INR, spleen size, and PVD) in both group I and group II. However, it is important to note that this analysis was likely underpowered due to the small number of patients with FV (n = 13), and thus the absence of a significant association should be interpreted with caution (Table 8).
| Parameter | Group I1 | P value2 | Group II1 | P value2 | ||
| FV present | FV absent | FV present | FV absent | |||
| TSH (μIU/mL) | 1.62 ± 2.51 | 1.06 ± 0.77 | 0.35 | 2.65 ± 3.23 | 1.23 ± 1.44 | 0.45 |
| FT3 (pmol/L) | 2.40 ± 0.93 | 2.72 ± 0.91 | 0.39 | 3.01 ± 1.38 | 2.51 ± 0.91 | 0.34 |
| FT4 (pmol/L) | 13.82 ± 1.35 | 14.56 ± 3.29 | 0.39 | 17.45 ± 6.72 | 12.59 ± 5.13 | 0.09 |
| PLT (× 103/μL) | 100.56 ± 37.12 | 96.55 ± 36.06 | 0.78 | 100.75 ± 30.61 | 126.29 ± 53.64 | 0.36 |
| INR | 1.46 ± 0.12 | 1.48 ± 0.33 | 0.84 | 1.74 ± 0.62 | 1.35 ± 0.31 | 0.40 |
| Spleen size (cm) | 17.69 ± 1.79 | 16.65 ± 2.02 | 0.19 | 15.07 ± 2.33 | 13.96 ± 1.80 | 0.27 |
| PVD (mm) | 13.66 ± 1.67 | 13.19 ± 2.21 | 0.58 | 12.38 ± 1.60 | 11.89 ± 2.08 | 0.66 |
The correlation analysis for FV (Table 8) was performed on the combined group of all patients with FV (n = 13), which included those with both isolated FV and FV concurrent with esophageal varices. A subgroup analysis comparing these two patterns was not performed due to the limited sample size.
Relationship between Child-Pugh scores and thyroid function tests in group I and group II: Our results highlight a significant association between Child-Pugh class and FT3 levels in both group I and group II, with the lowest levels observed in patients with Child-Pugh C. Post hoc analysis confirms significant differences in FT3 levels between Child-Pugh A vs C and B vs C. In contrast, no significant associations were found between the Child-Pugh class and TSH or FT4 levels. These results reinforce the potential role of FT3 as an indicator of liver disease severity, as progressive hepatic dysfunction appears to contribute to reduced FT3 levels in cirrhotic patients, regardless of variceal bleeding history (Table 9).
| Child-Pugh class | Group I1 | P value2 | Significant pairs (Scheffe test) | Group II1 | P value2 | Significant pairs (Scheffe test) | ||||
| TSH | FT3 | FT4 | TSH | FT3 | FT4 | |||||
| Class A | 1.22 ± 1.47 | 2.99 ± 0.91 | 14.35 ± 2.87 | < 0.01 | FT3 A vs C (P < 0.01) | 1.35 ± 1.52 | 3.25 ± 0.85 | 14.80 ± 2.45 | < 0.01 | FT3 A vs C (P < 0.01) |
| Class B | 1.41 ± 1.75 | 2.55 ± 0.89 | 13.22 ± 5.48 | < 0.01 | FT3 B vs C (P < 0.01) | 1.58 ± 1.68 | 2.75 ± 0.78 | 13.65 ± 5.10 | < 0.01 | FT3 B vs C (P < 0.01) |
| Class C | 1.70 ± 1.40 | 1.75 ± 0.77 | 15.29 ± 2.28 | < 0.01 | FT3 A vs C, B vs C (P < 0.01) | 1.85 ± 1.60 | 2.05 ± 0.70 | 15.50 ± 2.15 | < 0.01 | FT3 A vs C, B vs C (P < 0.01) |
Our results present the Spearman’s rank correlation between thyroid function tests and clinical parameters in group I and group II. FT3 levels showed a significant negative correlation with OV grades, INR, and Child-Pugh scores in both groups. Similarly, OV grades correlated negatively with PLT and positively with spleen size and PVD. No significant correlations were observed between TSH or FT4 levels and clinical parameters. These findings suggest that FT3 levels are closely associated with liver disease severity and variceal grades, regardless of variceal bleeding history (Table 10).
| Parameter | TSH1 | FT31 | FT41 | TSH2 | FT32 | FT42 | ||||||
| ρ | P value | ρ | P value | ρ | P value | ρ | P value | ρ | P value | ρ | P value | |
| OV grades | 0.25 | 0.17 | -0.61 | < 0.001 | -0.22 | 0.23 | 0.38 | 0.03 | -0.51 | < 0.001 | -0.32 | 0.07 |
| INR | -0.11 | 0.55 | -0.35 | 0.04 | -0.22 | 0.22 | 0.17 | 0.35 | 0.10 | 0.57 | 0.16 | 0.37 |
| Child-Pugh score | 0.26 | 0.15 | -0.43 | 0.01 | -0.16 | 0.37 | 0.33 | 0.06 | -0.33 | 0.05 | -0.31 | 0.08 |
| PLT (× 103/μL) | -0.03 | 0.86 | 0.28 | 0.12 | 0.21 | 0.25 | -0.23 | 0.20 | 0.40 | 0.02 | 0.19 | 0.29 |
| Spleen size (cm) | -0.19 | 0.29 | -0.05 | 0.77 | -0.15 | 0.41 | 0.05 | 0.77 | -0.15 | 0.40 | 0.04 | 0.81 |
Our results show statistically significant negative correlations between FT3, PLT, and OV grades in both group I and group II. Additionally, there were statistically significant positive correlations between INR, spleen size, PVD, Child-Pugh score, MELD score, and OV grades. The MELD score showed a strong positive correlation with OV grades (group I: Ρ = 0.56, P < 0.001; group II: Ρ = 0.45, P = 0.01), reinforcing its role as a composite marker of liver dysfunction and portal hypertension severity. These findings suggest that FT3 levels, along with established prognostic scores and portal hemodynamic markers, are closely associated with variceal severity, while FV development is less influenced by these parameters (Table 11).
| Parameter | OV1 | FV1 | OV2 | FV2 | ||||
| ρ | P value | ρ | P value | ρ | P value | ρ | P value | |
| TSH (μIU/mL) | 0.25 | 0.17 | 0.15 | 0.42 | 0.38 | 0.03 | -0.25 | 0.16 |
| FT3 (pmol/L) | -0.61 | < 0.001 | 0.14 | 0.44 | -0.51 | < 0.001 | -0.15 | 0.41 |
| FT4 (pmol/L) | -0.22 | 0.23 | 0.10 | 0.58 | -0.32 | 0.07 | -0.27 | 0.13 |
| PLT (× 103/μL) | -0.52 | < 0.001 | 0.10 | 0.58 | -0.52 | < 0.001 | 0.14 | 0.43 |
| INR | 0.35 | 0.04 | 0.02 | 0.90 | 0.36 | 0.04 | -0.33 | 0.05 |
| Spleen size (cm) | 0.44 | 0.01 | 0.02 | 0.90 | 0.43 | 0.01 | 0.18 | 0.32 |
| PVD (mm) | 0.49 | 0.005 | 0.10 | 0.58 | 0.46 | 0.008 | 0.06 | 0.73 |
| Child-Pugh score | 0.54 | 0.001 | 0.16 | 0.38 | 0.38 | 0.03 | 0.06 | 0.73 |
| MELD score | 0.56 | < 0.001 | 0.13 | 0.47 | 0.45 | 0.01 | 0.08 | 0.65 |
Our results present the multivariate ordinal logistic regression analysis identifying independent predictors of higher esophageal variceal grade. To comprehensively assess the contribution of our proposed markers alongside established liver disease severity scores, we constructed two multivariate models for each group (Table 12). Model 1 was adjusted for age, sex, and the Child-Pugh score. Model 2 was adjusted for age, sex, and the MELD score, which provides a more objective and continuous assessment of liver dysfunction.
| Variable | Group I-model 1 (Child-Pugh) | Group I-model 2 (MELD) | Group II-model 1 (Child-Pugh) | Group II-model 2 (MELD) | ||||
| aOR (95%CI) | P value | aOR (95%CI) | P value | aOR (95%CI) | P value | aOR (95%CI) | P value | |
| FT3 (per 1 pmol/L decrease) | 2.25 (1.52-3.33) | < 0.001 | 2.18 (1.48-3.21) | < 0.001 | 2.10 (1.45-3.05) | < 0.001 | 2.05 (1.42-2.98) | < 0.001 |
| 1.75 (1.28-2.39) | < 0.001 | 1.70 (1.25-2.32) | 0.001 | 1.65 (1.22-2.24) | 0.001 | 1.62 (1.20-2.19) | 0.002 | |
| INR (per 0.1 unit increase) | 1.15 (1.03-1.28) | 0.012 | 1.13 (1.02-1.26) | 0.023 | 1.14 (1.02–1.27) | 0.018 | 1.12 (1.01-1.25) | 0.038 |
| Child-Pugh score | 1.45 (1.08-1.95) | 0.013 | 1.40 (1.05-1.88) | 0.023 | ||||
| MELD score | 1.12 (1.02-1.23) | 0.015 | 1.10 (1.01-1.20) | 0.035 | ||||
| Age (years) | 1.02 (0.98-1.06) | 0.280 | 1.02 (0.98-1.06) | 0.305 | 1.01 (0.97-1.05) | 0.550 | 1.01 (0.97-1.05) | 0.585 |
| Sex (male) | 1.20 (0.87-1.66) | 0.260 | 1.18 (0.85-1.63) | 0.322 | 1.15 (0.84–1.58) | 0.380 | 1.14 (0.83-1.56) | 0.415 |
In both group I and group II, lower FT3 levels and lower PLT remained strong, independent predictors of higher variceal grades across both model 1 and model 2. For instance, in group I (model 1), each 1 pmol/L decrease in FT3 was associated with a 2.25-fold increase in the odds of a more severe variceal grade (95%CI: 1.52-3.33, P < 0.001). INR also remained a significant predictor in all models (P < 0.05). Notably, radiological parameters such as PVD and spleen size, which were significant in univariate analyses, did not retain independent significance in the multivariate models that included FT3, PLT, and INR. The consistent significance of this core panel of biomarkers, regardless of whether Child-Pugh or MELD score was included in the model, underscores their robust and independent value in predicting variceal severity.
These findings suggest that FT3 levels, PLT, and INR are strong independent predictors of variceal severity in cirrhotic patients with HCV, even after accounting for established prognostic scores. The integration of these non-invasive biomarkers is associated with enhanced early risk assessment, which could help clinicians prioritize high-risk patients for timely intervention.
To develop a single, generalizable predictive model for clinical use and to address potential multicollinearity between the proposed biomarkers and established liver disease severity scores, we performed a multivariate ordinal logistic regression analysis on the combined cohort of all cirrhotic patients (group I and II, n = 62). A history of variceal bleeding was included as a binary covariate. To avoid the statistical instability caused by the high correlation between our core biomarkers (e.g., INR) and the composite scores (Child-Pugh and MELD), which was confirmed by elevated VIF (VIF > 5), we constructed a final, parsimonious model.
This final model excluded the composite scores and focused on the core non-invasive panel. In this model, lower FT3 levels, lower PLT, and higher INR persisted as strong, independent predictors of more severe varices. As expected, a history of variceal bleeding was itself a powerful independent predictor of underlying variceal severity. The consistency of FT3, PLT, and INR in this stable model underscores their fundamental role in risk stratification across the entire spectrum of patients with HCV-related cirrhosis (Table 13).
| Variable1 | aOR (95%CI) | P value |
| FT3 (per 1 pmol/L decrease) | 2.25 (1.68-3.01) | < 0.001 |
| PLT (per 50 × 103/μL decrease) | 1.72 (1.38-2.15) | < 0.001 |
| INR (per 0.1 unit increase) | 1.15 (1.06-1.25) | 0.001 |
| Bleeding status (yes) | 2.85 (1.90-4.28) | < 0.001 |
| Age (years) | 1.01 (0.98-1.04) | 0.480 |
| Sex (male) | 1.15 (0.91-1.45) | 0.240 |
To assess the robustness of our findings against potential confounding from comorbidities, a sensitivity analysis was performed by excluding patients with documented nonhepatic chronic conditions (n = 4). The results remained virtually unchanged, confirming the stability of our core predictors.
Our results present the diagnostic performance of FT3 levels in predicting high-grade varices (grade III/IV) in group I and group II based on the ROC curve analysis. In group I, an FT3 cut-off value of 2.5 pmol/L provided a sensitivity of 85.7% (95%CI: 72.8%-93.9%) and a specificity of 78.3% (95%CI: 64.2%-88.7%). The positive predictive value (PPV) was 76.9%, and the negative predictive value (NPV) was 86.7%. The AUC was 0.88 (95%CI: 0.79-0.93), indicating excellent diagnostic accuracy. In group II, an FT3 cut-off value of 2.6 pmol/L yielded a sensitivity of 84.0% (95%CI: 70.0%-93.0%) and a specificity of 77.0% (95%CI: 62.0%-88.0%). The PPV was 75.0%, and the NPV was 85.0%. The AUC was 0.86 (95%CI: 0.77-0.92), reflecting a strong predictive value for high-grade varices (Table 14).
| Parameter | Group I | Group II | ||
| Value | 95%CI | Value | 95%CI | |
| Cut-off FT3 (pmol/L) | 2.5 | 2.6 | ||
| Sensitivity (%) | 85.7 | 72.8-93.9 | 84.0 | 70.0-93.0 |
| Specificity (%) | 78.3 | 64.2-88.7 | 77.0 | 62.0-88.0 |
| PPV (%) | 76.9 | 63.2-87.5 | 75.0 | 60.0-86.0 |
| NPV (%) | 86.7 | 73.2-94.9 | 85.0 | 70.0-94.0 |
| AUC | 0.88 | 0.79-0.93 | 0.86 | 0.77-0.92 |
These findings confirm that FT3 levels serve as a reliable non-invasive marker for identifying patients at risk of high-grade varices in both groups, supporting their potential role for assessing variceal severity in cirrhotic patients.
Our results present the diagnostic performance of variceal grade in predicting high-risk varices (grade III/IV with red signs) in group I and group II, based on ROC curve analysis. In group I, a cut-off variceal grade of II demonstrated a sensitivity of 85.0% (95%CI: 72.0%-93.0%) and a specificity of 80.0% (95%CI: 66.0%-90.0%). The PPV was 82.0%, and the NPV was 83.0%. The AUC was 0.88 (95%CI: 0.80-0.94), indicating excellent diagnostic accuracy. In group II, a cut-off variceal grade of II provided a sensitivity of 84.0% (95%CI: 70.0%-93.0%) and a specificity of 78.0% (95%CI: 63.0%-89.0%). The PPV was 80.0%, and the NPV was 82.0%. The AUC was 0.87 (95%CI: 0.78-0.93), confirming a strong predictive value for identifying high-risk varices (Table 15).
| Parameter | Group I | Group II | ||
| Value | 95%CI | Value | 95%CI | |
| Cut-off variceal grade | Grade II | Grade II | ||
| Sensitivity (%) | 85.0 | 72.0-93.0 | 84.0 | 70.0-93.0 |
| Specificity (%) | 80.0 | 66.0-90.0 | 78.0 | 63.0-89.0 |
| PPV (%) | 82.0 | 69.0-91.0 | 80.0 | 65.0-90.0 |
| NPV (%) | 83.0 | 69.0-92.0 | 82.0 | 67.0-92.0 |
| AUC | 0.88 | 0.80-0.94 | 0.87 | 0.78-0.93 |
These results indicate that variceal grade ≥ II is a reliable predictor of high-risk varices in both groups, supporting its role as an endoscopic marker for assessing variceal severity in cirrhotic patients.
Our results present the diagnostic performance of PVD in predicting high-grade varices (grade III/IV) in group I and group II, based on ROC curve analysis. In group I, a PVD cut-off of 13 mm demonstrated a sensitivity of 80.0% (95%CI: 66.0%-90.0%) and a specificity of 75.0% (95%CI: 60.0%-86.0%). The PPV was 78.0%, and the NPV was 77.0%. The AUC was 0.85 (95%CI: 0.77-0.91), indicating strong diagnostic accuracy. In group II, a PVD cut-off of 12.5 mm provided a sensitivity of 78.0% (95%CI: 63.0%-89.0%) and a specificity of 74.0% (95%CI: 59.0%-86.0%). The PPV was 76.0%, and the NPV was 76.0%. The AUC was 0.84 (95%CI: 0.76-0.90), confirming a strong predictive value for identifying high-grade varices (Table 16).
| Parameter | Group I | Group II | ||
| Value | 95%CI | Value | 95%CI | |
| Cut-off PVD (mm) | 13 | 12.5 | ||
| Sensitivity (%) | 80.0 | 66.0-90.0 | 78.0 | 63.0-89.0 |
| Specificity (%) | 75.0 | 60.0-86.0 | 74.0 | 59.0-86.0 |
| PPV (%) | 78.0 | 64.0-88.0 | 76.0 | 61.0-87.0 |
| NPV (%) | 77.0 | 62.0-88.0 | 76.0 | 61.0-87.0 |
| AUC | 0.85 | 0.77-0.91 | 0.84 | 0.76-0.90 |
These findings suggest that PVD is a reliable non-invasive marker for predicting high-grade varices in cirrhotic patients, supporting its role in risk assessment and early detection.
To integrate the core independent predictors into a single diagnostic tool, a VRS was developed using binary logistic regression (Table 17). The model, which included FT3, PLT, INR, and PVD to predict high-grade varices (grades III/IV), yielded the following formula: VRS = [-1.15 × FT3 (pmol/L) + -0.018 × PLT (× 103/μL)] + (1.92 × INR) + (0.28 × PVD (mm). PVD was selected for the final VRS over spleen size due to its marginally stronger independent association with high-grade varices in the initial model-building steps. The VRS demonstrated outstanding diagnostic accuracy for identifying highgrade varices, with an apparent AUC of 0.94. Internal validation using 1000 bootstrap samples, which also served as a comprehensive sensitivity analysis for model stability, corrected for overoptimism, yielded a biascorrected AUC of 0.92 (95%CI: 0.86-0.96).
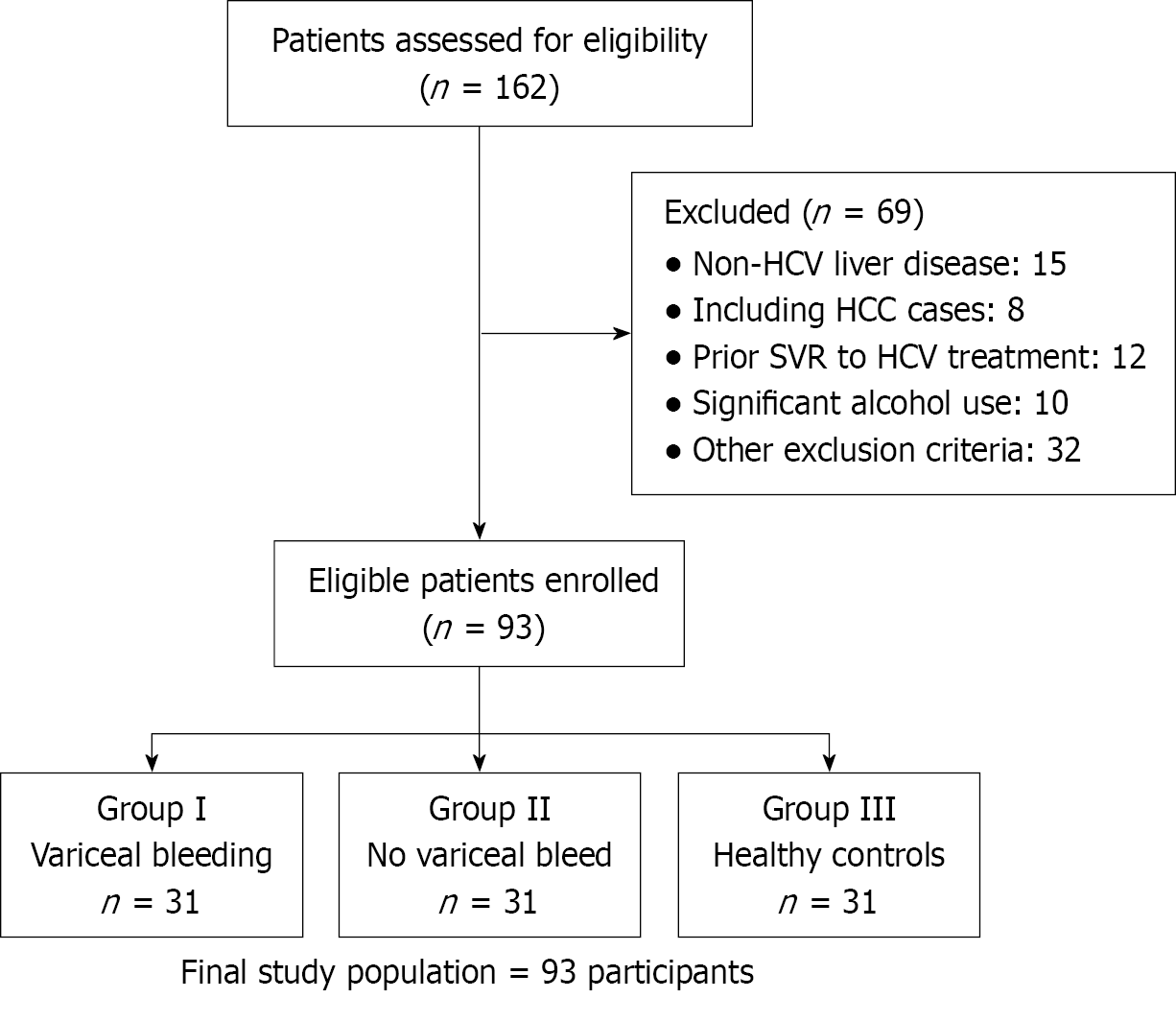
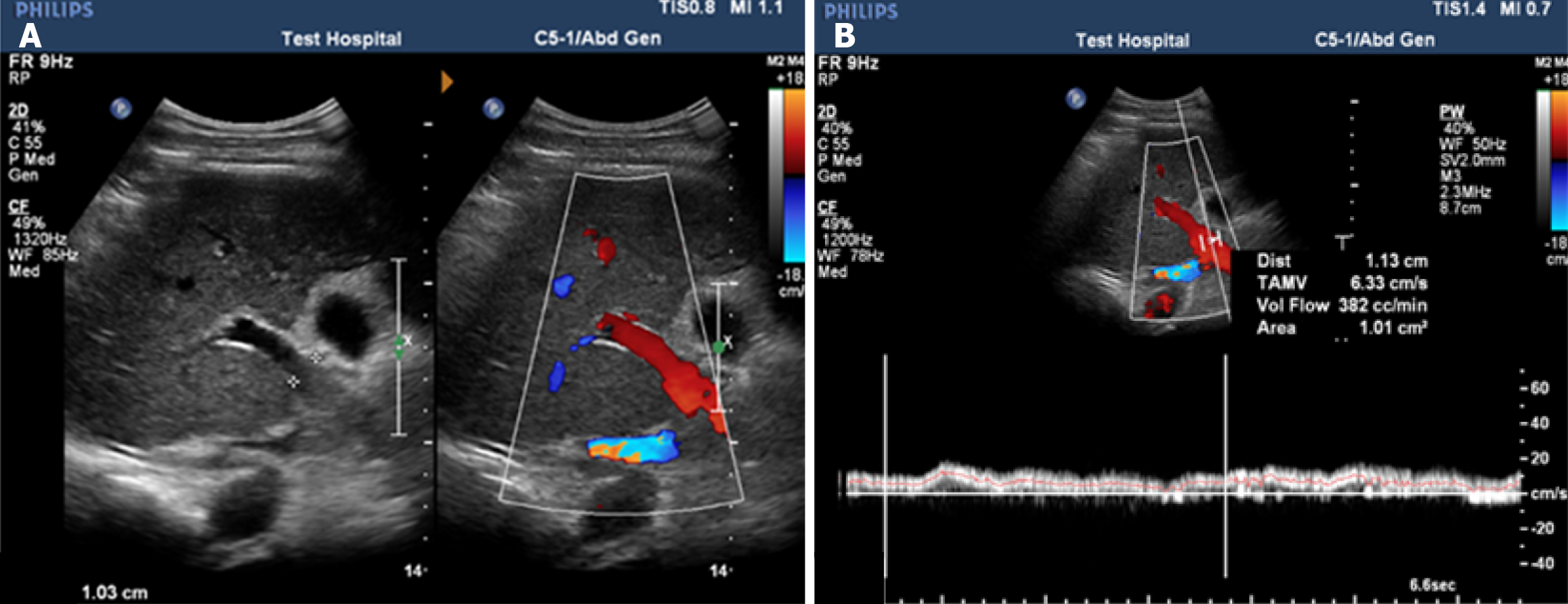
Doppler ultrasonography revealed significant hemodynamic changes in cirrhotic patients. The PVD and flow velocity in a healthy control are illustrated in Figure 2. In contrast, portal hypertension in a cirrhotic patient, characterized by an increased PVD and reduced flow velocity, is demonstrated in Figure 3.
Chronic HCV infection has historically been a significant global health burden, with liver cirrhosis and its complications, such as portal hypertension and gastroesophageal varices, being major contributors to morbidity and mortality worldwide[1-5]. In Egypt, a massive public health effort has led to the treatment of millions of patients, dramatically reducing the prevalence of viremic HCV[19]. However, this successful campaign has also identified a large, pre-existing cohort of patients with established cirrhosis who remain at risk for decompensation[20]. This legacy of advanced liver disease continues to be a major clinical and public health challenge in Egypt, with significant implications for long-term monitoring[21].
While low T3 syndrome is a recognized marker of hepatic dysfunction and poor prognosis in cirrhosis[22-25], its specific association with gastroesophageal variceal severity, particularly in HCV-related cirrhosis, has received limited attention. Our finding of significantly reduced FT3 levels in cirrhotic patients, especially those with variceal bleeding, extends this relationship to portal hypertensive complications. This observation gains additional relevance when considered alongside established noninvasive predictors of variceal risk, such as thrombocytopenia and increased PVD[26-30]. The integration of thyroidal, hematological, and hemodynamic markers in a unified model addresses a critical clinical gap in resourcelimited settings like Egypt, where endoscopic surveillance remains challenging.
We acknowledge that the hepatic venous pressure gradient (HVPG) is the gold standard for quantifying portal hypertension. However, HVPG measurement is an invasive and specialized procedure not routinely available in many resource-limited settings, including our center. The primary objective of this study was to develop a completely non-invasive, multimarker model that could be widely applicable in such contexts. Therefore, our model was benchmarked against endoscopic variceal grading, which is the current clinical standard for determining the need for primary prophylaxis.
The demographic and clinical characteristics of the study participants revealed no significant differences in age or sex distribution among the three groups, ensuring homogeneity for comparative analysis. The mean age was 57 ± 9.46 years in group I, 60 ± 6.59 years in group II, and 56 ± 6.52 years in group III (P = 0.06). The sex distribution was also comparable, with males constituting 71% of group I, 61.3% of group II, and 45.1% of group III (P = 0.08). Furthermore, an analysis of disease severity markers revealed a key observation: The two cirrhotic cohorts, with and without variceal bleeding, presented with comparable baseline severity of liver fibrosis as quantified by FIB-4 and APRI scores. This suggests that among patients with established severe cirrhosis, the occurrence of variceal bleeding was not a direct function of a further increase in fibrotic burden as quantified by FIB-4 and APRI, but rather of other factors reflected in the Child-Pugh and MELD scores. Instead, it highlights the role of other factors, such as individual variations in portal pressure or vascular anatomy, in triggering this specific decompensating event. The use of these objective fibrosis markers strengthens the validity of the comparison between the two patient groups (Table 1).
The absence of significant age differences among the groups is crucial for minimizing confounding factors in the analysis. Age is a known risk factor for the progression of liver disease and the development of complications such as portal hypertension and variceal bleeding[31]. However, the comparable age ranges in this study suggest that the observed differences in thyroid function, portal hemodynamics, and variceal severity are more likely attributable to the disease state rather than age-related factors. This finding aligns with previous studies that have reported similar age distributions in cirrhotic populations[32-34]. The higher proportion of males in the cirrhotic groups (group I and II) is consistent with the global epidemiology of HCV, where males are more frequently affected due to higher exposure to risk factors such as intravenous drug use, occupational hazards, and higher rates of alcohol consumption[35]. This sex disparity has been well-documented in the literature, with studies showing that males are more likely to develop advanced liver disease, and its complications compared to females[36]. This demographic consistency strengthens the generalizability of our findings to similar populations and ensures that the observed differences in thyroid function and variceal severity are not biased by sex-related factors.
Our findings confirm a distinct laboratory profile associated with decompensated cirrhosis. Patients with variceal bleeding exhibited significant hematological and synthetic dysfunction, characterized by profound anemia, thrombocytopenia, elevated INR, and hypoalbuminemia compared to healthy controls. This pattern highlights the convergence of portal hypertensive sequelae—such as bleeding and hypersplenism-and failing hepatic synthetic capacity in driving the complications of advanced liver disease (Table 2).
The significantly lower Hb levels in group I reflect the hematological consequences of chronic liver disease, including chronic blood loss from variceal bleeding and anemia of chronic disease. Anemia in cirrhotic patients is multifactorial, resulting from gastrointestinal bleeding, hypersplenism, and impaired erythropoiesis due to reduced hepatic production of erythropoietin[37]. The lower Hb levels in group I compared to group II (P = 0.04) further underscore the impact of variceal bleeding on hematological parameters. This finding aligns with previous studies that have identified anemia as a common complication in cirrhotic patients with variceal bleeding[27]. Thrombocytopenia is a hallmark of chronic liver disease and is primarily attributed to hypersplenism, reduced thrombopoietin production by the liver, and immune-mediated platelet destruction[38]. The significantly lower PLT in group I compared to group III (P < 0.01) and group II (P = 0.03) highlight the role of thrombocytopenia as a marker of advanced liver disease and portal hypertension. PLT has been widely used as a non-invasive predictor of variceal severity, with lower counts being associated with higher variceal grades and increased risk of bleeding[39]. This finding is consistent with previous studies that have identified thrombocytopenia as an independent predictor of variceal bleeding in cirrhotic patients[40-43].
The significantly higher INR in group I reflects impaired hepatic synthetic function, as the liver produces clotting factors. Elevated INR is a well-established marker of liver dysfunction and is included in the Child-Pugh and MELD scoring systems for assessing liver disease severity[44]. The higher INR in group I compared to group III (P < 0.01) underscores the association between advanced liver disease and variceal bleeding. This finding aligns with previous studies that have identified elevated INR as a predictor of variceal severity and bleeding risk[45,46]. Albumin is synthesized exclusively by the liver, and its levels are a reliable indicator of hepatic synthetic function. The significantly lower albumin levels in group I compared to group III (P < 0.01) reflect the impaired synthetic capacity of the liver in cirrhotic patients. The lower albumin levels in group I compared to group II (P = 0.08) further highlight the progressive nature of liver dysfunction in patients with variceal bleeding. Hypoalbuminemia is associated with worse clinical outcomes in cirrhosis, including increased risk of ascites, spontaneous bacterial peritonitis, and mortality[47]. Bilirubin is a byproduct of Hb metabolism and is primarily excreted by the liver. Elevated bilirubin levels in group I and group II compared to group III (P < 0.01) reflect impaired bilirubin metabolism and excretion due to hepatocellular dysfunction and cholestasis. The higher bilirubin levels in group I compared to group II (P = 0.62) suggest that variceal bleeding is more common in patients with advanced liver dysfunction. Hyperbilirubinemia is a key component of the Child-Pugh score and is associated with worse outcomes in cirrhotic patients[48,49]. ALT and AST are markers of hepatocellular injury, with elevated levels indicating ongoing liver inflammation and necrosis. The significantly higher ALT and AST levels in group I compared to group III (P < 0.01) reflect the active hepatocellular injury associated with chronic HCV infection. The higher AST levels compared to ALT in group I (AST/ALT ratio > 1) are consistent with the pattern observed in advanced liver disease, where mitochondrial dysfunction and hepatocyte necrosis lead to increased AST release[50]. This finding aligns with previous studies that have identified elevated transaminases as markers of disease activity and progression in HCV-related cirrhosis[51].
A key finding was the pronounced reduction in FT3 levels across both cirrhotic groups, consistent with the non-thyroidal illness syndrome (NTIS), while TSH and FT4 remained unaffected. This isolated low T3 state suggests a targeted impairment in peripheral hormone conversion-a process heavily reliant on hepatic function-rather than a primary thyroid axis failure. The numerical gradient from controls to compensated (group II) and decompensated (group I) cirrhosis implies that FT3 may serve as a sensitive barometer of overall hepatic metabolic health (Table 3).
The similarly elevated FIB 4 and APRI scores in both cirrhotic groups confirm that all patients had advanced fibrosis. This suggests that low FT3 levels primarily reflect overall hepatic disease severity rather than being a variceal-specific marker. Nevertheless, in our multivariate models adjusted for established severity scores (Child Pugh, MELD), FT3 remained an independent predictor of variceal grade, supporting its additive value in risk stratification beyond general disease severity.
The significantly lower FT3 levels in cirrhotic patients (groups I and II) compared to healthy controls (group III) are characteristic of low T3 syndrome, a condition commonly observed in chronic liver disease. Low T3 syndrome results from impaired hepatic deiodination of T4 to the biologically active T3, a process mediated by type 1 deiodinase in the liver[52]. The lowest FT3 levels in group I (patients with variceal bleeding) suggest that thyroid dysfunction is more pronounced in patients with advanced liver disease and complications such as portal hypertension and variceal bleeding. In cirrhotic patients, hepatic dysfunction disrupts this enzymatic activity, leading to reduced T3 levels and elevated reverse T3 levels[53]. Previous studies have consistently reported low T3 levels in cirrhotic patients, with the lowest levels observed in those with advanced liver disease and complications such as variceal bleeding. The association between low T3 levels and worse clinical outcomes has been well-documented, reinforcing the prognostic value of thyroid function tests in cirrhosis[9,54,55]. The absence of significant differences in TSH and FT4 levels across the groups indicates that the hypothalamic-pituitary-thyroid axis remains intact in cirrhotic patients. This preservation of TSH and FT4 levels, despite reduced FT3 levels, suggests a compensatory mechanism to maintain euthyroidism in the context of chronic liver disease[56]. The normal TSH levels reflect the body’s attempt to stimulate thyroid hormone production, while the normal FT4 levels indicate that the thyroid gland is functioning adequately. However, the liver's impaired ability to convert T4 to T3 results in low T3 levels, a hallmark of NTIS[57]. This pattern has been well-documented in cirrhotic patients and is associated with worse clinical outcomes, including increased mortality and complications such as hepatic encephalopathy and ascites[6,58]. The significant reduction in FT3 levels in cirrhotic patients, particularly those with variceal bleeding, with preservation of normal TSH and FT4 levels suggests that the observed thyroid dysfunction is primarily due to impaired hepatic metabolism rather than primary thyroid disease and highlights the potential of FT3 levels as a non-invasive marker for assessing liver disease severity and variceal risk in cirrhotic patients.
Radiologically, we observed a stepwise increase in both spleen size and PVD, progressing from healthy controls to compensated cirrhosis and culminating in the most significant enlargement in patients with variceal bleeding. This progressive splenomegaly and portal venous dilation provide direct anatomical evidence of escalating portal hypertension, confirming that these ultrasound parameters effectively mirror the hemodynamic burden of advancing liver disease (Table 4). The Doppler ultrasound findings are visually demonstrated in Figures 2 and 3, where the PV’s progressive dilation and portal flow velocity reduction indicate the hemodynamic alterations associated with portal hypertension (Figures 2 and 3).
The significantly larger spleen size in cirrhotic patients (groups I and II) compared to healthy controls (group III) is a direct consequence of portal hypertension. Portal hypertension leads to congestion of the splenic vein, resulting in splenomegaly due to increased blood flow and pressure within the splenic vasculature[27]. The larger spleen size in group I compared to group II (P < 0.01) reflects the more advanced portal hypertension and hypersplenism in patients with variceal bleeding. Splenomegaly is a well-established marker of portal hypertension and is often associated with thrombocytopenia due to platelet sequestration in the enlarged spleen[59]. Previous studies have consistently identified splenomegaly as a marker of portal hypertension and hypersplenism in cirrhotic patients, with larger spleen sizes being associated with higher variceal grades and increased bleeding risk. The progressive increase in spleen size observed in this study aligns with these findings, reinforcing the clinical relevance of splenomegaly as a predictor of variceal severity[39,60,61]. The significantly larger PVD in cirrhotic patients (group I and II) compared to healthy controls (group III) is another hallmark of portal hypertension. Increased intrahepatic vascular resistance in cirrhosis leads to dilation of the PV as the body attempts to compensate for the elevated pressure within the portal system[26]. The larger PVD in group I compared to group II (P < 0.01) underscores the association between advanced portal hypertension and variceal bleeding. PVD has been widely used as a non-invasive marker for assessing portal hypertension, with diameters > 13 mm being strongly predictive of variceal severity and bleeding risk[62]. The association between PVD and portal hypertension has been well-documented, with larger diameters being predictive of variceal severity and bleeding risk. The findings in this study are consistent with previous reports, which have identified PVD > 13 mm as a reliable marker for assessing portal hemodynamics in cirrhotic patients[63,64]. The progressive increase in spleen size and PVD from group III to group II and group I is indicative of the increasing severity of portal hypertension and its complications in cirrhotic patients. By incorporating these radiological markers into clinical practice, clinicians can better identify patients at high risk of variceal bleeding and implement timely interventions to prevent complications. Spleen size and PVD can be considered non-invasive predictors of variceal severity and bleeding risk in cirrhotic patients.
Endoscopic evaluations in our study revealed a more severe portal hypertensive burden in group I, characterized by both higher grades of OV and a greater prevalence of FV compared to group II. Specifically, the prevalence of high-grade (grade III) OV was significantly higher in group I (Table 6). The pattern of varices also differed significantly between the groups (Table 5). Group I had over twice the prevalence of FV (29.0% vs 12.9%), with a notably higher proportion of patients presenting with both oesophageal and FV (GOV2 pattern: 22.6% vs 9.7%). This finding aligns with the understanding that the development of complex variceal patterns, particularly GOV2, is a marker of more advanced and severe portal hypertension. The concurrent presence of high-grade OV and FV in group I underscores the association between the overall burden of portosystemic collaterals and the risk of variceal bleeding.
The significantly higher prevalence of high-grade OV (grade III) in group I (38.7%) compared to group II (9.7%) underscores the association between variceal bleeding and advanced portal hypertension. High-grade varices are a direct consequence of increased intrahepatic vascular resistance and elevated portal pressure, which are hallmarks of advanced liver cirrhosis[27]. The presence of grade III varices, characterized by large, tortuous veins, is a strong predictor of variceal bleeding, as these varices are more prone to rupture due to increased wall tension[40]. The higher prevalence of grade III varices in group I aligns with previous studies that have identified variceal size as a critical risk factor for bleeding, emphasizing the importance of endoscopic surveillance in cirrhotic patients[39]. Previous studies have consistently identified high-grade OV as a strong predictor of variceal bleeding in cirrhotic patients. For example, Garcia-Tsao et al[10] reported that patients with grade III varices have a significantly higher risk of bleeding compared to those with lower-grade varices[27] in addition, variceal size is a critical determinant of bleeding risk, with larger varices being more prone to rupture due to increased wall tension. The findings in this study align with these reports, reinforcing the importance of endoscopic evaluation in assessing variceal risk[65,66]. The higher prevalence of FV in group I (29%) compared to group II (12.9%) further highlights the severity of portal hypertension in patients with variceal bleeding. FV are often associated with spontaneous portosystemic shunts, which develop as a compensatory mechanism to alleviate elevated portal pressure. However, these varices are also at high risk of bleeding, particularly in patients with advanced liver disease[67]. The presence of FV in group I suggests more severe portal hypertension and a higher likelihood of complications, such as variceal hemorrhage. The association between FV and severe portal hypertension has been well-documented in literature and is often associated with spontaneous portosystemic shunts and is more common in patients with advanced cirrhosis[68-70]. The endoscopic findings in this study provide critical insights into the relationship between variceal severity and the risk of bleeding in cirrhotic patients. The higher prevalence of high-grade oesophageal and FV in group I underscores the importance of endoscopic evaluation for assessing variceal severity in cirrhotic patients and its management. These findings suggest that patients with high-grade varices, particularly those with FV, are at significantly higher risk of bleeding and require closer monitoring and timely intervention, such as endoscopic band ligation or beta-blocker therapy[26]. The results also highlight the utility of endoscopy as a diagnostic tool for identifying patients at high risk of variceal bleeding, which is crucial for preventing life-threatening complications.
Our results demonstrate distinct relationships between variceal type and clinical parameters. A consistent and significant association was observed between OV severity and markers of disease progression. In both patient groups, higher OV grades were strongly associated with lower FT3 levels, lower PLT, and higher INR, spleen size, and PVD (P < 0.05) (Table 7). Correlation analyses further confirmed significant negative associations of FT3 and PLT with OV grades, and positive correlations of INR, spleen size, PVD, and Child-Pugh score with OV grades (Tables 10 and 11). In stark contrast, no statistically significant relationship was found between the presence of FV and any of the studied thyroid or liver-related parameters in either group (Table 8). This lack of association should be interpreted cautiously, as the analysis was likely underpowered due to the small subgroup of patients with FV (n = 13). Therefore, while our findings hint that the pathophysiology of FV may involve distinct hemodynamic pathways less directly influenced by these conventional markers, this requires confirmation in larger, adequately powered studies.
The significant negative correlation between FT3 levels and OV grades in both group I and group II highlights the potential role of thyroid dysfunction in the progression of portal hypertension and variceal development. Low FT3 levels, characteristic of low T3 syndrome, are associated with impaired hepatic deiodination of T4 toT3, a process that is disrupted in cirrhotic patients due to hepatocellular dysfunction[12]. Low T3 levels have also been associated with endothelial dysfunction and increased intrahepatic vascular resistance, which are key drivers of portal hypertension[9,71]. The inverse relationship between FT3 levels and OV grades suggests that thyroid dysfunction is associated with more advanced portal hypertension, leading to more severe varices[27,56]. The negative correlation between PLT and OV grades is consistent with thrombocytopenia being a marker of hypersplenism and advanced liver disease. Thrombocytopenia in cirrhotic patients is primarily due to platelet sequestration in the enlarged spleen, reduced thrombopoietin production by the liver, and immune-mediated platelet destruction[28]. Lower PLT are associated with higher variceal grades and increased bleeding risk, as platelets are crucial in maintaining vascular integrity and preventing variceal rupture[39,72]. This finding aligns with previous studies that have identified thrombocytopenia as an independent predictor of variceal severity[72,73]. The positive correlations between INR, spleen size, PVD, and OV grades further underscore the importance of these parameters in assessing variceal risk. Elevated INR reflects impaired hepatic synthetic function, while increased spleen size and PVD are direct consequences of portal hypertension[26]. These findings are consistent with previous studies that have identified these markers as reliable predictors of variceal severity and bleeding risk in cirrhotic patients[62].
In contrast to OV, no significant relationship was found between FV and thyroid function parameters (TSH, FT3, FT4) or liver-related parameters (PLT, INR, spleen size, PVD). While this may suggest that the development of FV is less influenced by these specific markers compared to OV, this analysis was likely underpowered due to the small number of patients with FV (n = 13), and thus firm conclusions cannot be drawn. FV are often associated with spontaneous portosystemic shunts, which may develop independently of the severity of portal hypertension or liver dysfunction. The established pathophysiology of FV differs from that of OV, as FV is more likely to result from specific vascular anomalies or collateral pathways that bypass the liver, rather than generalized portal hypertension[67-70,74]. This distinct pathophysiology is a plausible explanation for why FV development might not correlate strongly with conventional markers of liver dysfunction and portal hemodynamics. This reinforces the clinical consensus that FV requires a different approach to risk assessment and management compared to OV, as highlighted by Sarin et al[18], who emphasized the role of endoscopic ultrasound (EUS), multidetector computed tomography, and multi-detector row CT angiography in detecting and grading FV[75,76].
Our correlation analyses reveal a distinct pathophysiological landscape for different variceal types. For OV, the significant negative correlations of FT3 with OV grades, INR, and Child-Pugh scores in both patient groups (Table 9), coupled with positive correlations of INR, spleen size, PVD, and MELD score with OV grades (Table 11), paint a coherent picture. This pattern suggests that OV severity is integrally linked to the progression of liver dysfunction and portal hypertension. The strong association with low FT3 levels is particularly notable and aligns with proposed mechanisms where thyroid hormone deficiency may contribute to endothelial dysfunction and increased intrahepatic vascular resistance[77]. The robust correlation with the MELD score further underscores that OV are a manifestation of overall liver disease severity.
In stark contrast, the absence of significant correlations between FV and these same parameters (Table 10) implies a different pathogenesis, one less dependent on the conventional markers of portal hypertensive severity and more related to the development of specific spontaneous portosystemic shunts[67-70]. The collective validation of these non-invasive markers[78] supports their use in a multimarker risk stratification strategy, with the core panel of FT3, PLT, and INR reflecting the underlying synthetic and metabolic dysfunction driving OV risk.
The findings from Tables 6, 7, 9, and 10 highlight the distinct pathophysiological mechanisms underlying OV and FV development in cirrhotic patients. While OV progression is closely associated with markers of liver dysfunction (e.g., low FT3 levels, thrombocytopenia), portal hypertension (e.g., increased spleen size, PVD), and thyroid dysfunction, FV development appears to be influenced by different factors, such as spontaneous portosystemic shunts. These results emphasize the importance of differentiating between OV and FV when assessing variceal risk and planning interventions. For OV, markers such as FT3 levels, PLT, and portal hemodynamic parameters (e.g., spleen size and PVD) can serve as non-invasive indicators for identifying patients at higher risk of bleeding and guiding clinical management. For FV, our study did not identify a reliable non-invasive biomarker from the panel we tested. Therefore, until future larger studies prove otherwise, the identification of high-risk FV continues to rely on alternative diagnostic approaches, such as EUS. By integrating these findings into clinical practice, clinicians can better tailor risk assessment and intervention strategies to the specific type of varices, ultimately improving patient outcomes.
Our results highlight the relationship between Child-Pugh scores and thyroid function parameters (FT3, TSH, FT4) in chronic HCV cirrhotic patients with variceal bleeding (group I) and without variceal bleeding (group II). In both groups, FT3 levels were significantly lower in patients with Child-Pugh class C compared to classes A and B (P < 0.05). No significant differences were observed in TSH or FT4 levels across Child-Pugh classes, suggesting that progressive hepatic dysfunction primarily affects FT3 levels, while TSH and FT4 remain relatively stable (Table 9).
The significant reduction in FT3 levels in patients with Child-Pugh class C underscores the potential of FT3 as a non-invasive biomarker for assessing liver disease severity and predicting complications in cirrhotic patients. Low FT3 levels, characteristic of low T3 syndrome, are linked to impaired hepatic conversion of T4 to T3, a process disrupted in advanced cirrhosis. The lowest FT3 levels observed in Child-Pugh class C patients indicate that thyroid dysfunction is more pronounced in those with severe liver disease, reinforcing FT3’s role in disease monitoring and prognostic assessment[52-55]. The preservation of normal TSH and FT4 levels across Child-Pugh classes indicates that the hypothalamic-pituitary-thyroid axis remains intact in cirrhotic patients. This suggests that the observed thyroid dysfunction is primarily due to impaired hepatic metabolism rather than primary thyroid disease. The normal TSH levels reflect the body's attempt to stimulate thyroid hormone production, while the normal FT4 levels indicate that the thyroid gland is functioning adequately. However, the liver's impaired ability to convert T4 to T3 results in low T3 levels, a hallmark of NTIS[56]. By incorporating FT3 levels into clinical practice, clinicians can better identify patients with advanced liver disease and implement timely interventions that may improve outcomes. For example, patients with low FT3 levels and high Child-Pugh scores may benefit from closer monitoring and early interventions, such as beta-blockers or endoscopic band ligation, to prevent variceal bleeding[27]. Additionally, FT3 levels could be used as part of a multimarker panel that includes other non-invasive markers (e.g., PLT, PVD, and others) to improve risk prediction and guide treatment decisions. The progressive decline in FT3 levels with worsening Child-Pugh scores underscores the prognostic value of thyroid dysfunction in cirrhotic patients, as low T3 levels have been associated with worse clinical outcomes, including increased mortality and complications such as hepatic encephalopathy and ascites[79-81]. These findings align with previous studies that have identified low T3 levels as a predictor of poor outcomes in cirrhosis, reinforcing the im
The multivariate ordinal logistic regression provide compelling evidence for our proposed biomarkers. The initial separate models for group I and II (Table 12) revealed a remarkable consistency, identifying the same trio of predictors, FT3, PLT, and INR, in both patients with and without a bleeding history. For instance, each 1 pmol/L decrease in FT3 was associated with approximately a 2.2-fold increase in the odds of more severe varices across all models. This internal validation strongly suggests these markers are fundamental to the pathophysiology of portal hypertensive complications. The subsequent final unified model on the combined cohort (Table 13), which was designed to avoid multicollinearity, further strengthens this conclusion, providing a single, stable, and applicable tool for risk stratification. The persistence of these three parameters in this robust model, even after controlling for the strong confounding effect of bleeding status itself, underscores their fundamental and independent value. This convergence of evidence from multiple analytical approaches solidifies the role of this multimarker panel in the noninvasive assessment of variceal severity.
The diagnostic performance of these and other key parameters was subsequently evaluated. ROC curve analysis confirmed that FT3 levels < 2.5 pmol/L (Table 14), variceal grade ≥ II (Table 15), and PVD ≥ 13 mm (Table 16) demon
The most significant advancement offered by this study is the development of a composite VRS (Table 17). By integrating the four strongest non-invasive predictors, FT3, PLT, INR, and PVD, into a single model, we achieved a superior diagnostic performance (AUC = 0.94) that surpassed any individual marker. This finding robustly validates our core hypothesis that a multimarker approach is more powerful for risk stratification. The VRS provides a quantitative and practical framework that could be easily implemented in clinical practice. A patient's data can be input into the simple formula, and the resulting score would offer a highly accurate, non-invasive estimate of their risk for harboring high-grade varices, thereby optimizing the allocation of endoscopic resources.
The precise mechanistic link between low FT3 levels and the severity of portal hypertension remains to be fully elucidated. While our study demonstrates a strong associative relationship, it does not provide mechanistic proof. The observed correlation could be multifactorial. Low T3 syndrome is a well-known marker of critical illness and may simply reflect the severity of hepatic dysfunction. However, a potential pathophysiological contribution is plausible. Experimental data suggest that thyroid hormones, particularly T3, possess vasodilatory properties and play a role in main
This study represents a significant contribution to the field of hepatology, particularly in the context of Egypt, where the burden of HCV-related liver disease is among the highest globally. By employing a multimarker approach that integrates thyroid dysfunction (FT3 levels), portal hemodynamics (PVD, spleen size), and hematological parameters (PLT), this study provides a comprehensive framework for non-invasive assessment of variceal severity in cirrhotic patients. The inclusion of thyroid dysfunction as a predictor of variceal severity is particularly novel, as it highlights the role of low T3 syndrome in the pathophysiology of portal hypertension and variceal development, an underexplored relationship, especially in the Egyptian population. Furthermore, the study's focus on FV and their distinct patho
This study provides a clinically actionable framework for non-invasive risk stratification of variceal bleeding in cirrhotic patients, particularly in resource-limited settings where endoscopic surveillance is challenging. By demonstrating that low FT3 levels (< 2.5 pmol/L), thrombocytopenia, and increased PVD (≥ 13 mm) independently predict high-risk varices, our findings enable clinicians to prioritize patients for early intervention, such as beta-blocker therapy or endoscopic band ligation, while safely deferring endoscopy in low-risk cases. This approach could sig
While this study provides valuable insights, it is important to acknowledge its limitations. First, by design, we excluded patients with SVR to create a homogenous cohort with active HCV viremia. While this strengthens the internal validity for this specific group, it limits the generalizability of our findings to the large and growing population of patients with post-SVR cirrhosis, who remain at risk for portal hypertensive complications. Second, the study was conducted at a single center, which may limit the generalizability of the findings to other populations or healthcare settings, particularly in regions with different HCV epidemiology or healthcare resources. Third, the sample size, though adequate for the primary analysis, was relatively small for robust subgroup analyses. This was particularly evident in the cohort of patients with FV (n = 13), which limited the power of our correlation analysis for this specific subgroup and precluded a meaningful comparison between isolated and concurrent FV. Fourth, the study relied on ultrasonographic and endoscopic measurements, which, while standard, may introduce variability due to differences in operator technique and interpretation. Fifth, the study did not explore the potential impact of thyroid hormone replacement therapy on variceal risk or liver disease progression, which could be an important area for future research. Sixth, HVPG measurement was not available for the retrospective cohort and therefore direct comparison with the invasive gold-standard HVPG could not be performed. This limitation is acknowledged, and future prospective studies with HVPG correlation are warranted. Finally, another limitation of our study is the absence of liver stiffness measurement (LSM), for instance via transient elastography (FibroScan®), which is a well-validated non-invasive parameter for assessing fibrosis and portal hypertension. The inclusion of LSM could have further strengthened our predictive model. Unfortunately, this technology was not available at our institution during the study period. We recommend that future studies integrate LSM with our proposed multimarker panel to evaluate for incremental predictive value.
The present study was specifically designed to investigate the cross-sectional relationship between non-invasive markers and variceal bleeding risk. It was not powered to assess correlations with other complications of cirrhosis (such as hepatic encephalopathy or hepatorenal syndrome) or mortality. The potential of FT3 as a predictor for overall prognosis and other complications is an important area for future longitudinal research.
This study demonstrates the utility of a multimarker approach for the non-invasive identification of cirrhotic patients at high risk of variceal bleeding. A final multivariate model, which controlled for bleeding status, identified a core set of independent predictors: Lower FT3 levels, lower PLT, and a higher INR. This core model is complemented by radiological markers; specifically, a PVD ≥ 13 mm demonstrated excellent standalone diagnostic accuracy for high-grade varices. Therefore, a practical risk-stratification strategy could prioritize patients based on the core hematological and hormonal biomarkers, with portal hemodynamic measurements providing additional supportive evidence. This approach could help optimize the allocation of endoscopic resources in resource-limited settings.
| 1. | Schiano Moriello N, Pinchera B, Gentile I. Personalized care approaches to hepatitis C therapy: recent advances and future directions. Expert Rev Anti Infect Ther. 2024;22:139-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 2. | Lens S, Baiges A, Alvarado-Tapias E, LLop E, Martinez J, Fortea JI, Ibáñez-Samaniego L, Mariño Z, Rodríguez-Tajes S, Gallego A, Bañares R, Puente Á, Albillos A, Calleja JL, Torras X, Hernández-Gea V, Bosch J, Villanueva C, García-Pagán JC, Forns X. Clinical outcome and hemodynamic changes following HCV eradication with oral antiviral therapy in patients with clinically significant portal hypertension. J Hepatol. 2020;73:1415-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 134] [Article Influence: 22.3] [Reference Citation Analysis (1)] |
| 3. | Semmler G, Binter T, Kozbial K, Schwabl P, Hametner-Schreil S, Zanetto A, Gavasso S, Chromy D, Bauer DJM, Simbrunner B, Scheiner B, Bucsics T, Stättermayer AF, Pinter M, Steindl-Munda P, Schöfl R, Russo FP, Simioni P, Trauner M, Ferenci P, Reiberger T, Mandorfer M. Noninvasive Risk Stratification After HCV Eradication in Patients With Advanced Chronic Liver Disease. Hepatology. 2021;73:1275-1289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 4. | Waked I, Esmat G, Elsharkawy A, El-Serafy M, Abdel-Razek W, Ghalab R, Elshishiney G, Salah A, Abdel Megid S, Kabil K, El-Sayed MH, Dabbous H, El Shazly Y, Abo Sliman M, Abou Hashem K, Abdel Gawad S, El Nahas N, El Sobky A, El Sonbaty S, El Tabakh H, Emad E, Gemeah H, Hashem A, Hassany M, Hefnawy N, Hemida AN, Khadary A, Labib K, Mahmoud F, Mamoun S, Marei T, Mekky S, Meshref A, Othman A, Ragab O, Ramadan E, Rehan A, Saad T, Saeed R, Sharshar M, Shawky H, Shawky M, Shehata W, Soror H, Taha M, Talha M, Tealaab A, Zein M, Hashish A, Cordie A, Omar Y, Kamal E, Ammar I, AbdAlla M, El Akel W, Doss W, Zaid H. Screening and Treatment Program to Eliminate Hepatitis C in Egypt. N Engl J Med. 2020;382:1166-1174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 202] [Article Influence: 33.7] [Reference Citation Analysis (2)] |
| 5. | Gomaa A, Gomaa M, Allam N, Waked I. Hepatitis C Elimination in Egypt: Story of Success. Pathogens. 2024;13:681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 6. | Antonelli A, Ferri C, Fallahi P, Ferrari SM, Ghinoi A, Rotondi M, Ferrannini E. Thyroid disorders in chronic hepatitis C virus infection. Thyroid. 2006;16:563-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 81] [Article Influence: 4.1] [Reference Citation Analysis (2)] |
| 7. | Van den Berghe G. Non-thyroidal illness in the ICU: a syndrome with different faces. Thyroid. 2014;24:1456-1465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 202] [Article Influence: 16.8] [Reference Citation Analysis (4)] |
| 8. | Piantanida E, Ippolito S, Gallo D, Masiello E, Premoli P, Cusini C, Rosetti S, Sabatino J, Segato S, Trimarchi F, Bartalena L, Tanda ML. The interplay between thyroid and liver: implications for clinical practice. J Endocrinol Invest. 2020;43:885-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 9. | Xiong Y, Xia Z, Yang L, Huang J. Low T3 syndrome is associated with poor prognosis in patients with hepatitis B virus-related acute-on-chronic liver failure. Expert Rev Gastroenterol Hepatol. 2022;16:681-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 10. | Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1775] [Cited by in RCA: 1553] [Article Influence: 172.6] [Reference Citation Analysis (5)] |
| 11. | Mazzaro C, Quartuccio L, Adinolfi LE, Roccatello D, Pozzato G, Nevola R, Tonizzo M, Gitto S, Andreone P, Gattei V. A Review on Extrahepatic Manifestations of Chronic Hepatitis C Virus Infection and the Impact of Direct-Acting Antiviral Therapy. Viruses. 2021;13:2249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (2)] |
| 12. | Gionfra F, De Vito P, Pallottini V, Lin HY, Davis PJ, Pedersen JZ, Incerpi S. The Role of Thyroid Hormones in Hepatocyte Proliferation and Liver Cancer. Front Endocrinol (Lausanne). 2019;10:532. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 13. | El-Khateeb E, Darwich AS, Achour B, Athwal V, Rostami-Hodjegan A. Review article: time to revisit Child-Pugh score as the basis for predicting drug clearance in hepatic impairment. Aliment Pharmacol Ther. 2021;54:388-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 14. | Leigh N, Hammill CW. Liver ultrasound: Normal anatomy and pathologic findings. Surg Open Sci. 2024;19:178-198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 15. | Al-Nakshabandi NA. The role of ultrasonography in portal hypertension. Saudi J Gastroenterol. 2006;12:111-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 16. | Benter T, Klühs L, Teichgräber U. Sonography of the spleen. J Ultrasound Med. 2011;30:1281-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 33] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 17. | Tag-Adeen M, Alsenbesy M, Ghweil AA, Abd Elrazek MAH, Elgohary EA, Sallam MM, Ismael A, Nawara A. Liver stiffness measurement and spleen diameter as predictors for the presence of esophageal varices in chronic hepatitis C patients. Medicine (Baltimore). 2017;96:e8621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 18. | Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992;16:1343-1349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1011] [Cited by in RCA: 864] [Article Influence: 25.4] [Reference Citation Analysis (2)] |
| 19. | El-Akel W, El-Sayed MH, El Kassas M, El-Serafy M, Khairy M, Elsaeed K, Kabil K, Hassany M, Shawky A, Yosry A, Shaker MK, ElShazly Y, Waked I, Esmat G, Doss W. National treatment programme of hepatitis C in Egypt: Hepatitis C virus model of care. J Viral Hepat. 2017;24:262-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 143] [Article Influence: 15.9] [Reference Citation Analysis (1)] |
| 20. | Yameny AA. Hepatocellular carcinoma (HCC) in Egypt: Prevalence, risk factors, diagnosis and prevention: A Review. J Biosci Appl Res. 2024;10:879-890. [RCA] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 21. | Elbahrawy A, Ibrahim MK, Eliwa A, Alboraie M, Madian A, Aly HH. Current situation of viral hepatitis in Egypt. Microbiol Immunol. 2021;65:352-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 22. | Li C, Zhao C, Yu Z, Yang W, Sun M, Li Y, Guo G, Hui Y, Wang X, Fan X, Wang B, Zhang J, Sun C. Low free triiodothyronine levels are associated with frail phenotype in hospitalized inpatients with cirrhosis. Postgrad Med. 2022;134:516-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 23. | Visser TJ. Regulation of Thyroid Function, Synthesis and Function of Thyroid Hormones. In: Vitti P, Hegedus L, editors. Thyroid Diseases. Cham: Springer, 2018: 1-30. [DOI] [Full Text] |
| 24. | Vargas-Beltran AM, Armendariz-Pineda SM, Martínez-Sánchez FD, Martinez-Perez C, Torre A, Cordova-Gallardo J. Interplay between endocrine disorders and liver dysfunction: Mechanisms of damage and therapeutic approaches. World J Gastroenterol. 2025;31:108827. [PubMed] [DOI] [Full Text] |
| 25. | Nardin G, Colombo BDS, Ronsoni MF, Silva PESE, Fayad L, Wildner LM, Bazzo ML, Dantas-Correa EB, Narciso-Schiavon JL, Schiavon LL. Thyroid hormone profile is related to prognosis in acute decompensation of cirrhosis. Arch Endocrinol Metab. 2024;68:e230249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 26. | Berzigotti A, Seijo S, Reverter E, Bosch J. Assessing portal hypertension in liver diseases. Expert Rev Gastroenterol Hepatol. 2013;7:141-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 213] [Article Influence: 16.4] [Reference Citation Analysis (1)] |
| 27. | Kaplan DE, Ripoll C, Thiele M, Fortune BE, Simonetto DA, Garcia-Tsao G, Bosch J. AASLD Practice Guidance on risk stratification and management of portal hypertension and varices in cirrhosis. Hepatology. 2024;79:1180-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 283] [Article Influence: 141.5] [Reference Citation Analysis (18)] |
| 28. | Mitchell O, Feldman DM, Diakow M, Sigal SH. The pathophysiology of thrombocytopenia in chronic liver disease. Hepat Med. 2016;8:39-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 29. | Kayacetin E, Efe D, Doğan C. Portal and splenic hemodynamics in cirrhotic patients: relationship between esophageal variceal bleeding and the severity of hepatic failure. J Gastroenterol. 2004;39:661-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 30. | Berzigotti A, Piscaglia F. Ultrasound in portal hypertension--part 1. Ultraschall Med. 2011;32:548-68; quiz 569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 31. | Tsochatzis EA, Bosch J, Burroughs AK. Liver cirrhosis. Lancet. 2014;383:1749-1761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1536] [Cited by in RCA: 1355] [Article Influence: 112.9] [Reference Citation Analysis (4)] |
| 32. | El-Serag HB. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology. 2012;142:1264-1273.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2629] [Cited by in RCA: 2553] [Article Influence: 182.4] [Reference Citation Analysis (10)] |
| 33. | Mohamoud YA, Mumtaz GR, Riome S, Miller D, Abu-Raddad LJ. The epidemiology of hepatitis C virus in Egypt: a systematic review and data synthesis. BMC Infect Dis. 2013;13:288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 249] [Article Influence: 19.2] [Reference Citation Analysis (1)] |
| 34. | El-Sawy AA, Mohamed AER. Low serum free and total tri-iodothyronine hormones as possible prognostic factors in liver cirrhotic patients due to chronic hepatitis C. Thyroid Res Pract. 2015;12:87-92. [DOI] [Full Text] |
| 35. | Gower E, Estes C, Blach S, Razavi-Shearer K, Razavi H. Global epidemiology and genotype distribution of the hepatitis C virus infection. J Hepatol. 2014;61:S45-S57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1506] [Cited by in RCA: 1347] [Article Influence: 112.3] [Reference Citation Analysis (2)] |
| 36. | Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16:589-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3422] [Cited by in RCA: 3209] [Article Influence: 458.4] [Reference Citation Analysis (9)] |
| 37. | Gkamprela E, Deutsch M, Pectasides D. Iron deficiency anemia in chronic liver disease: etiopathogenesis, diagnosis and treatment. Ann Gastroenterol. 2017;30:405-413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 38. | Afdhal NH, Giannini EG, Tayyab G, Mohsin A, Lee JW, Andriulli A, Jeffers L, McHutchison J, Chen PJ, Han KH, Campbell F, Hyde D, Brainsky A, Theodore D; ELEVATE Study Group. Eltrombopag before procedures in patients with cirrhosis and thrombocytopenia. N Engl J Med. 2012;367:716-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 242] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 39. | Sharma SK, Aggarwal R. Prediction of large esophageal varices in patients with cirrhosis of the liver using clinical, laboratory and imaging parameters. J Gastroenterol Hepatol. 2007;22:1909-1915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 54] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 40. | de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2652] [Cited by in RCA: 2371] [Article Influence: 215.5] [Reference Citation Analysis (2)] |
| 41. | Sigal SH, Sherman Z, Jesudian A. Clinical Implications of Thrombocytopenia for the Cirrhotic Patient. Hepat Med. 2020;12:49-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 42. | Alvaro D, Caporaso N, Giannini EG, Iacobellis A, Morelli M, Toniutto P, Violi F; PReBRiC (Procedure-Related Bleeding Risk in Cirrhosis) group. Procedure-related bleeding risk in patients with cirrhosis and severe thrombocytopenia. Eur J Clin Invest. 2021;51:e13508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 43. | Chen Y, Yang C, Huang S, Liu J, Wang Y, Zhou C, Li T, Wang C, Ju S, Bai Y, Yao W, Xiong B. The impact of thrombocytopenia on variceal bleeding in cirrhotic patients with transjugular intrahepatic portosystemic shunt. Sci Rep. 2023;13:1633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 44. | Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 1973;60:646-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6342] [Cited by in RCA: 5745] [Article Influence: 108.4] [Reference Citation Analysis (9)] |
| 45. | Hshieh TT, Kaung A, Hussain S, Curry MP, Sundaram V. The international normalized ratio does not reflect bleeding risk in esophageal variceal hemorrhage. Saudi J Gastroenterol. 2015;21:254-258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 46. | Faisal MS, Singh T, Esfeh JM. Performance of elevated PT/INR as a risk factor for re-bleeding after band ligation in patients with acute variceal hemorrhage. Dig Liver Dis. 2020;52:1213-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 47. | Bai Z, Méndez-Sánchez N, Romeiro FG, Mancuso A, Philips CA, Tacke F, Basaranoglu M, Primignani M, Ibrahim M, Wong YJ, Nery FG, Teschke R, Ferreira CN, Muñoz AE, Pinyopornpanish K, Thevenot T, Singh SP, Mohanty A, Satapathy SK, Ridola L, Maruyama H, Cholongitas E, Levi Sandri GB, Yang L, Shalimar, Yang Y, Villa E, Krag A, Wong F, Jalan R, O'Brien A, Bernardi M, Qi X; Liver Cirrhosis-related Complications (LCC)-International Special Interest Group. Use of albumin infusion for cirrhosis-related complications: An international position statement. JHEP Rep. 2023;5:100785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 48. | Miyata T, Yamashita YI, Arima K, Higashi T, Hayashi H, Imai K, Nitta H, Chikamoto A, Beppu T, Baba H. Alteration of prognostic efficacy of albumin-bilirubin grade and Child-Pugh score according to liver fibrosis in hepatocellular carcinoma patients with Child-Pugh A following hepatectomy. Ann Gastroenterol Surg. 2022;6:127-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 49. | da Fonsecaa LG, de Melob MAZ, da Silveirac THM, Yamamotod VJ, Hashizumee PHS, Sabbagaf J. Prognostic role of albumin-bilirubin (ALBI) score and Child-Pugh classification in patients with advanced hepatocellular carcinoma under systemic treatment. Ecancermedicalscience. 2024;18:1748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 50. | Giannini EG, Testa R, Savarino V. Liver enzyme alteration: a guide for clinicians. CMAJ. 2005;172:367-379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1498] [Cited by in RCA: 1242] [Article Influence: 59.1] [Reference Citation Analysis (1)] |
| 51. | Rumi MG, De Filippi F, Donato MF, Del Ninno E, Colombo M. Progressive hepatic fibrosis in healthy carriers of hepatitis C virus with a transaminase breakthrough. J Viral Hepat. 2002;9:71-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 52. | Sabatino L, Vassalle C, Del Seppia C, Iervasi G. Deiodinases and the Three Types of Thyroid Hormone Deiodination Reactions. Endocrinol Metab (Seoul). 2021;36:952-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (2)] |
| 53. | Marino L, Kim A, Ni B, Celi FS. Thyroid hormone action and liver disease, a complex interplay. Hepatology. 2025;81:651-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 30] [Article Influence: 30.0] [Reference Citation Analysis (1)] |
| 54. | Agiasotelli D, Alexopoulou A, Vasilieva L, Dourakis SP. Low free T3 levels are related to early mortality in patients with decompensated cirrhosis and acute-on chronic liver failure. J Hepatol. 2014;61:1446-1447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 55. | Hartl L, Simbrunner B, Jachs M, Wolf P, Bauer DJM, Scheiner B, Balcar L, Semmler G, Schwarz M, Marculescu R, Dannenberg V, Trauner M, Mandorfer M, Reiberger T. Lower free triiodothyronine (fT3) levels in cirrhosis are linked to systemic inflammation, higher risk of acute-on-chronic liver failure, and mortality. JHEP Rep. 2024;6:100954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 56. | Punekar P, Sharma AK, Jain A. A Study of Thyroid Dysfunction in Cirrhosis of Liver and Correlation with Severity of Liver Disease. Indian J Endocrinol Metab. 2018;22:645-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 57. | Haymart MR, Papaleontiou M. Updates in Thyroidology. Endocrin Metab Clin North Am. 2022;51:i. [DOI] [Full Text] |
| 58. | Feng HL, Li Q, Cao WK, Yang JM. Changes in thyroid function in patients with liver failure and their clinical significance: A clinical study of non-thyroidal illness syndrome in patients with liver failure. Hepatobiliary Pancreat Dis Int. 2020;19:561-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 59. | Gordon SC, Gonzalez HC. Hepatic Manifestations of Systemic Disorders. In: E.R. Schiff ER, Maddrey WC, Reddy KR, editors. Schiff's Diseases of the Liver. United States: John Wiley & Sons, 2017: 218-243. [DOI] [Full Text] |
| 60. | Dib N, Oberti F, Calès P. Current management of the complications of portal hypertension: variceal bleeding and ascites. CMAJ. 2006;174:1433-1443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 61. | Sarwar S, Khan AA, Alam A, Butt AK, Shafqat F, Malik K, Ahmad I, Niazi AK. Non-endoscopic prediction of presence of esophageal varices in cirrhosis. J Coll Physicians Surg Pak. 2005;15:528-531. [PubMed] |
| 62. | Salman A, Salman MA, Ismaeel Saadawy AM, Tourky M, Shawkat M. Portal venous hemodynamics as predictors for the development and grades of esophageal varices in Post‐HCV cirrhotic patients: An Egyptian center study. Adv Dig Med. 2021;8:146-154. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 63. | Maruyama H, Tobari M, Nagamatsu H, Shiina S, Yamaguchi T. Contrast-enhanced ultrasonography for the management of portal hypertension in cirrhosis. Front Med (Lausanne). 2022;9:1057045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 64. | Choi G, Kim KA, Lee J, Park YS, Lee J, Choi JW, Lee CH. Ultrasonographic atlas of splenic lesions. Ultrasonography. 2022;41:416-429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 65. | Raffetto JD, Khalil RA. Mechanisms of varicose vein formation: valve dysfunction and wall dilation. Phlebology. 2008;23:85-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 133] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 66. | Henry Z, Patel K, Patton H, Saad W. AGA Clinical Practice Update on Management of Bleeding Gastric Varices: Expert Review. Clin Gastroenterol Hepatol. 2021;19:1098-1107.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 122] [Article Influence: 24.4] [Reference Citation Analysis (2)] |
| 67. | Juncu S, Minea H, Girleanu I, Huiban L, Muzica C, Chiriac S, Timofeiov S, Mihai F, Cojocariu C, Stanciu C, Trifan A, Singeap AM. Clinical Implications and Management of Spontaneous Portosystemic Shunts in Liver Cirrhosis. Diagnostics (Basel). 2024;14:1372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 68. | Siramolpiwat S. Transjugular intrahepatic portosystemic shunts and portal hypertension-related complications. World J Gastroenterol. 2014;20:16996-17010. [PubMed] [DOI] [Full Text] |
| 69. | Boike JR, Thornburg BG, Asrani SK, Fallon MB, Fortune BE, Izzy MJ, Verna EC, Abraldes JG, Allegretti AS, Bajaj JS, Biggins SW, Darcy MD, Farr MA, Farsad K, Garcia-Tsao G, Hall SA, Jadlowiec CC, Krowka MJ, Laberge J, Lee EW, Mulligan DC, Nadim MK, Northup PG, Salem R, Shatzel JJ, Shaw CJ, Simonetto DA, Susman J, Kolli KP, VanWagner LB; Advancing Liver Therapeutic Approaches (ALTA) Consortium. North American Practice-Based Recommendations for Transjugular Intrahepatic Portosystemic Shunts in Portal Hypertension. Clin Gastroenterol Hepatol. 2022;20:1636-1662.e36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 196] [Cited by in RCA: 179] [Article Influence: 44.8] [Reference Citation Analysis (2)] |
| 70. | Rathi S, Brocco S, Formentin C, Zarantonello L, Mangini C, Meneghello G, Merkel C, Angeli P, Stramare R, Montagnese S, Amodio P. Spontaneous portosystemic shunts in cirrhosis: Detection, implications, and clinical associations. Dig Liver Dis. 2021;53:1468-1475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 71. | Sahin T, Oral A, Turker F, Kocak E. Can hypothyroidism be a protective factor for hepatocellular carcinoma in cirrhosis? Medicine (Baltimore). 2020;99:e19492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 72. | Afsar A, Nadeem M, Shah SAA, Hussain H, Rani A, Ghaffar S. Platelet count can predict the grade of esophageal varices in cirrhotic patients: a cross-sectional study. F1000Res. 2021;10:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 73. | Eldesoky AE, Aeriba M, Abd Elfattah F, Elhawary G, Awad M. Platelet count/spleen diameter ratio as a predictor of high-risk esophageal varices in patients with liver cirrhosis. Med J Viral Hepatitis. 2022;6.2:25-30. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 74. | Nardelli S, Riggio O, Gioia S, Puzzono M, Pelle G, Ridola L. Spontaneous porto-systemic shunts in liver cirrhosis: Clinical and therapeutical aspects. World J Gastroenterol. 2020;26:1726-1732. [PubMed] [DOI] [Full Text] |
| 75. | Willmann JK, Weishaupt D, Böhm T, Pfammatter T, Seifert B, Marincek B, Bauerfeind P. Detection of submucosal gastric fundal varices with multi-detector row CT angiography. Gut. 2003;52:886-892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 76. | Elrabat A, El-gamal S, Kashwaa MM, El-rakhawy MM. Detection of fundic varices obturation by endoscopic ultrasound versus multidetector computed tomography. Egypt J Intern Med. 2019;31:487-494. [DOI] [Full Text] |
| 77. | Iwakiri Y. Endothelial dysfunction in the regulation of cirrhosis and portal hypertension. Liver Int. 2012;32:199-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 145] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 78. | Ahmad AK, Atzori S, Maurice J, Taylor-Robinson SD, Lim AK. Non-invasive splenic parameters of portal hypertension: Assessment and utility. World J Hepatol. 2020;12:1055-1066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 79. | Sarangapani A, Shanmugam C, Kalyanasundaram M, Rangachari B, Thangavelu P, Subbarayan JK. Noninvasive prediction of large esophageal varices in chronic liver disease patients. Saudi J Gastroenterol. 2010;16:38-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 80. | El-Naggar YAF, Abdallah IAM, Attia HA. Prediction of Large Esophageal Varices by Ultrasound Doppler and Serum Markers in Portal Hypertensive Cirrhotic Patients in Sharkia. Egypt J Hosp Med. 2021;84:2619-2624. [DOI] [Full Text] |
| 81. | Liang H, Si H, Liu M, Yuan L, Ma R, Zhang G, Yang J, Mo Z, Zhao Q. Non-Invasive Prediction Models for Esophageal Varices and Red Signs in Patients With Hepatitis B Virus-Related Liver Cirrhosis. Front Mol Biosci. 2022;9:930762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |