Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115539
Revised: November 12, 2025
Accepted: January 21, 2026
Published online: March 27, 2026
Processing time: 158 Days and 6.5 Hours
Dyslipidemia in liver cirrhosis represents a dynamic consequence of progressive hepatocellular failure rather than a conventional metabolic disorder, arising from coordinated disruptions in lipoprotein synthesis, remodeling, and clearance. Cirrhotic liver remodeling is associated with reduced apolipoprotein production, impaired very low-density lipoprotein export, downregulation of low-density lipoprotein receptor-mediated uptake, and cholestasis-driven accumulation of atypical lipoproteins, including lipoprotein X and lipoprotein Z, which distort standard lipid metrics and contribute to oxidative and inflammatory signaling. These molecular perturbations are further shaped by disease etiology, insulin resistance, and cytokine-mediated inhibition of lipid-processing pathways, generating stage-specific lipid phenotypes that correlate with prognosis and systemic complications. Given the altered hepatic handling of lipid-modifying drugs and the limited applicability of statins in advanced disease, this review synthesizes current mechanistic and translational evidence on non-statin lipid-lowering therapies in cirrhosis. Available data indicate that ezetimibe and fibrates modulate intestinal cholesterol flux and peroxisome proliferator-activated receptor-α signaling, respectively, but their effects on cirrhotic lipid networks remain incompletely defined. In contrast, proprotein convertase subtilisin/kexin type 9 inhibitors and RNA-based therapies such as inclisiran reduce circulating atherogenic lipoproteins through receptor-dependent pathways with minimal hepatic biotransformation, although clinical and molecular data are largely restricted to compensated cirrhosis. Emerging agents targeting upstream cholesterol synthesis and intrahepatic thyroid hormone receptor-β signaling further highlight the potential to influence hepatic lipid oxidation and fibrogenic signaling, yet evidence in cirrhosis is sparse. Collectively, the data underscore dyslipidemia as an integrated molecular feature of cirrhosis and identify non-statin therapies as tools to interrogate, rather than simply correct, disrupted hepatic lipid biology, emphasizing the need for stage- and mechanism-specific investigation.
Core Tip: Dyslipidemia in liver cirrhosis reflects fundamental alterations in hepatic lipid biology, including impaired apolipoprotein synthesis, defective lipoprotein export, receptor downregulation, and cholestasis-driven accumulation of atypical particles such as lipoprotein X and lipoprotein Z. These molecular disturbances generate stage-specific lipid phenotypes that challenge conventional cardiovascular risk assessment and drug targeting. Non-statin lipid-lowering agents provide mechanistically informative tools to probe these disrupted pathways; however, their effects in cirrhosis remain largely defined by pharmacologic rationale rather than direct clinical evidence. Understanding how disease stage and etiology reshape hepatic lipid handling is essential for rational application and future development of lipid-modifying strategies in cirrhotic populations.
- Citation: Fuentes-Mendoza JM, Concepción-Zavaleta MJ, Mendoza-Godoy JJ, Concepción-Urteaga LA, Martínez-Gutiérrez CO, Paz-Ibarra J. Dyslipidemia in liver cirrhosis: Pathophysiology and emerging therapeutic approaches. World J Hepatol 2026; 18(3): 115539
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115539.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115539
Liver cirrhosis represents the advanced stage of chronic liver injury and is defined by progressive fibrosis, nodular regeneration, and irreversible distortion of hepatic architecture[1]. It arises from heterogeneous etiologies, including viral infections, toxic exposures, metabolic causes, hereditary disorders, and immune-mediated liver diseases[2]. Given the liver’s central role in lipid synthesis, lipoprotein assembly, and metabolic regulation, cirrhosis is consistently accom
Statins remain the reference therapy for dyslipidemia; however, their applicability in cirrhosis, particularly in de
These limitations have renewed interest in non-statin lipid-lowering therapies for patients with cirrhosis. Although statins confer well-established cardiovascular benefit, treatment discontinuation or dose reduction occurs in a substantial proportion of patients due to intolerance, prompting guideline-supported use of non-statin agents when additional low-density lipoprotein (LDL) cholesterol (LDL-C) reduction is required in high-risk populations[12]. Moreover, several non-statin therapies, particularly inhibitors of proprotein convertase subtilisin/kexin type 9 (PCSK9), target discrete molecular pathways involved in lipoprotein turnover, inflammatory signaling, and hepatic lipid handling, raising important mechanistic and translational questions in cirrhotic liver disease[13].
This narrative mini-review was conducted to synthesize current evidence on the prevalence, pathophysiology, and management of dyslipidemia in patients with liver cirrhosis, with a specific focus on non-statin lipid-lowering therapies. A comprehensive literature search was performed in PubMed/MEDLINE, EMBASE, Scopus, and Web of Science databases, covering publications from January 2000 to August 2025. Search terms included combinations of “liver cirrhosis”, “dyslipidemia”, “non-statin therapy”, “ezetimibe”, “fibrates”, “PCSK9 inhibitors”, “bempedoic acid”, “omega-3 fatty acids”, “thyroid hormone receptor agonists”, and “RNA-based lipid-lowering therapies”. Additional relevant studies were identified through manual screening of reference lists from key reviews and original articles.
Eligible studies included clinical trials, observational cohort studies, systematic reviews, meta-analyses, and selected translational or mechanistic studies evaluating lipid abnormalities or non-statin lipid-lowering interventions in adults with cirrhosis or chronic liver disease. Editorials, case reports, and studies without liver-specific data were excluded. Two reviewers independently screened titles and abstracts, extracted relevant data, and resolved discrepancies by consensus. Owing to heterogeneity in study designs, patient populations, and therapeutic exposures, findings were synthesized narratively and organized by therapeutic class and disease stage (compensated vs decompensated cirrhosis), emphasizing pharmacological mechanisms, hepatic handling, safety considerations, and clinical applicability. In total, 93 studies were included in the qualitative synthesis.
Liver cirrhosis remains a major global health burden, ranking among the leading causes of chronic disease-related mortality and accounting for approximately 1 million deaths annually worldwide, with a substantial contribution to disability-adjusted life years[14,15]. The epidemiologic framework of cirrhosis-associated dyslipidemia has been refined by the recent reclassification of non-alcoholic fatty liver disease into steatotic liver disease, encompassing metabolic dysfunction-associated steatotic liver disease (MASLD), alcohol-associated liver disease, and the overlapping metabolic dysfunction and alcohol-related liver disease phenotype[16]. In population-based cohorts, MASLD affects up to 18.5% of men and 10.3% of women, and now represents the most prevalent etiologic context in which dyslipidemia coexists with cirrhosis, cardiometabolic disease, and systemic complications, while metabolic dysfunction and alcohol-related liver disease and alcohol-associated liver disease remain less prevalent but disproportionately affect male populations[17-21]. Beyond prevalence, contemporary studies consistently demonstrate that dyslipidemia in cirrhosis is primarily linked to disease severity rather than lipid excess: As hepatic function deteriorates, patients with advanced chronic liver disease or higher Child-Pugh class exhibit marked reductions in total cholesterol, LDL-C, and high-density lipoprotein (HDL) cholesterol (HDL-C), whereas triglyceride and very LDL (VLDL) levels show weaker or inconsistent associations with fibrosis severity[22]. These patterns are reproducible across viral, metabolic, and alcohol-related etiologies, supporting a paradigm in which hypocholesterolemia and hypolipidemia serve as epidemiologic signatures of impaired hepatic metabolic capacity and clinically relevant markers of disease stage and prognosis, rather than conventional indicators of cardiovascular risk[14,22].
Cirrhosis encompasses heterogeneous etiologies that converge on a common endpoint: Progressive disruption of hepatic lipid handling. Viral hepatitis exemplifies this interaction, as hepatitis B virus promotes hepatic fatty acid synthesis, whereas hepatitis C virus directly alters intracellular cholesterol trafficking and lipoprotein assembly[23]. Similarly, alcohol-related liver disease evolves from steatosis to cirrhosis primarily as a function of cumulative alcohol exposure, with female sex, obesity, and high-fat diets amplifying metabolic injury[24]. In parallel, MASLD, defined by hepatic steatosis in the presence of cardiometabolic risk factors, has emerged as a dominant cause of cirrhosis and hepatocellular carcinoma, while cardiovascular disease remains the leading cause of death in this population[25]. Autoimmune and cholestatic liver diseases further contribute to end-stage liver disease, in which impaired bile flow profoundly distorts cholesterol excretion and lipoprotein composition[26,27].
However, beyond etiology, cardiometabolic comorbidities and hepatic failure jointly shape the dyslipidemia pheno
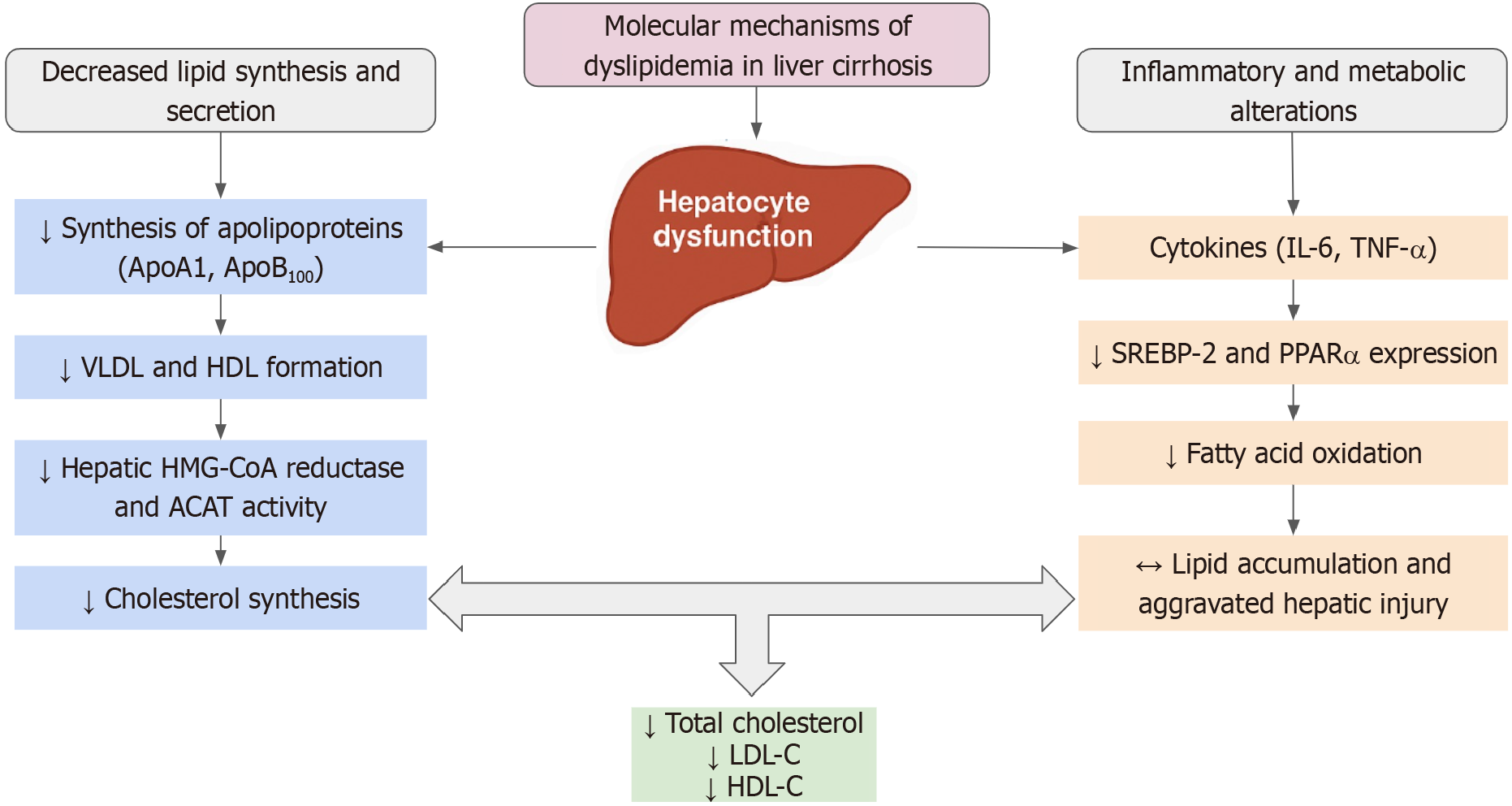
The liver serves as the central regulator of lipid homeostasis through coordinated apolipoprotein synthesis, VLDL assembly and secretion, and receptor-mediated LDL clearance[45-47]. In cirrhosis, progressive loss of functional hepatocyte mass disrupts these processes, resulting in impaired apolipoprotein production, reduced VLDL export, and downregulation of LDL receptors, with consequent declines in total cholesterol, LDL, and HDL that closely track disease severity[35,48]. Accordingly, lipid concentrations show consistent inverse associations with Child-Pugh and Model for End-Stage Liver Disease scores, positioning hypocholesterolemia not merely as a biochemical abnormality but as a surrogate of hepatic reserve and prognosis (Figure 1)[48]. Yet this pattern is not linear: Cholestasis fundamentally alters cholesterol excretion and lipid absorption, producing paradoxical hypertriglyceridemia via impaired lipoprotein lipase activity and reduced clearance of triglyceride-rich lipoproteins[49,50]. In this setting, accumulation of abnormal lipoproteins, particularly lipoprotein X and the hepatotoxic LDL-like lipoprotein Z, distorts conventional lipid metrics, mimics an atherogenic profile, and independently predicts worse survival[51-53].
By contrast, viral and metabolic liver diseases impose distinct molecular perturbations that further reshape the cirrhotic lipid landscape. Hepatitis C virus hijacks the VLDL assembly pathway to form lipoviral particles, suppresses microsomal triglyceride transfer protein, and selectively lowers LDL while enriching triglyceride fractions within HDL[54,55]. Con
| Risk factor | Pathophysiological mechanisms | Clinical manifestations | Ref. |
| Cardiometabolic comorbidities (diabetes, hypertension, obesity, MASLD) | (1) Insulin resistance promotes hepatic de novo lipogenesis and VLDL secretion; (2) Adipokine imbalance (↑leptin, ↓adiponectin); and (3) PNPLA3 and TM6SF2 variants impair triglyceride mobilization and VLDL secretion, fostering steatosis and atherogenic dyslipidemia | (1) Overlap of MetS and cirrhosis (up to 60% of patients); and (2) Central obesity, hypertriglyceridemia and low HDL-C accelerate fibrosis and CV events (“liver-heart-metabolism” axis) | [4,20,24-26,37-41] |
| Hepatic dysfunction and impaired lipid handling | (1) Loss of hepatocyte mass reduces apolipoprotein synthesis (ApoA-I and ApoB), VLDL secretion and LDL receptor activity to ↓total cholesterol, LDL-C, HDL-C; and (2) Cholestasis to paradoxical hypertriglyceridemia (impaired LPL activity) | (1) In compensated cirrhosis: Modest lipid reductions and near-normal TG; (2) In decompensated cirrhosis: Lowest TC and HDL-C in Child-Pugh C; and (3) Hypocholesterolemia predicts poor survival and transplant-free mortality | [8,9,11,20-23,28,31-33] |
| Chronic systemic inflammation and viral/metabolic injury | (1) Oxidative LDL uptake by Kupffer cells to cytokine release (TNF-α, IL-6); (2) Stellate cell activation to fibrosis; (3) HCV alters VLDL assembly and lowers LDL-C; (4) MASLD dyslipidemia promotes lipotoxicity and carcinogenesis; and (5) Lipidomic signatures in HCC | (1) Increased sd-LDL and oxidized LDL despite low absolute LDL-C; (2) Paradoxical ↑CAD incidence in cirrhosis; and (3) Dyslipidemia linked to fibrogenesis, HCC risk and extra-hepatic morbidity | [24,25,27,29,30,34-36,41] |
The clinical expression of dyslipidemia in cirrhosis is stage dependent and closely mirrors hepatic reserve rather than conventional cardiovascular risk. In compensated cirrhosis, lipid profiles typically show low-normal total cholesterol, LDL-C, and HDL-C, with preserved or mildly elevated triglycerides; however, progression to decompensation is marked by progressive hypocholesterolemia, most pronounced in Child-Pugh C, while triglycerides may paradoxically increase, in part due to portal hypertension-associated insulin resistance[22,58,61]. Importantly, contemporary studies identify low cholesterol levels as a robust prognostic marker, independently associated with reduced transplant-free survival, higher short-term mortality after acute gastrointestinal bleeding, and adverse outcomes in critical illness, whereas preserved or elevated cholesterol reflects retained synthetic capacity and improved survival[62,63]. Nevertheless, despite declining absolute lipid concentrations, qualitative molecular alterations persist: Enrichment of small dense and oxidized LDL particles confers heightened atherogenic potential, helping to explain the comparable or increased burden of coronary artery disease observed in cirrhosis, particularly among patients with cardiometabolic comorbidities[41,64,65]. Metabolic syndrome, affecting up to 60% of cirrhotic patients, further amplifies risk through central obesity, hypertriglyceridemia, low HDL-C, and insulin resistance[31,66,67], while insulin resistance mechanistically promotes hepatic steatosis, accelerates fibrogenesis, and reinforces systemic inflammation, consolidating the bidirectional “liver-heart-metabolism” axis[68-70]. Emerging lipidomic studies corroborate these observations by demonstrating disease-specific alterations in fatty acid composition and apolipoprotein expression that translate into distinct circulating lipid signatures with prognostic relevance for both hepatic and cardiovascular outcomes[70].
In advanced cirrhosis, progressive reductions in total cholesterol and LDL-C undermine their reliability as cardiovascular risk markers, complicating both risk stratification and therapeutic decision-making[71]. Consequently, attention has shifted toward molecularly informative biomarkers that better reflect atherogenic burden, including apolipoprotein B (ApoB), which captures the total number of atherogenic particles, and lipoprotein(a), which confers cardiovascular risk even at low LDL-C concentrations[72]. More recently, multi-omics approaches, integrating lipidomic, proteomics, and HDL functional assays, have revealed qualitative alterations in lipoprotein composition and function that are not apparent on standard lipid panels, offering a more precise framework for risk assessment in cirrhosis.
Despite growing therapeutic options, dyslipidemia management in cirrhosis remains constrained by altered pharmacokinetics and a paucity of cirrhosis-specific evidence. Most non-statin agents were validated in non-cirrhotic po
| Therapy | Main mechanism of action | Use in compensated cirrhosis | Main risk in cirrhosis (advanced/decompensated) |
| Ezetimibe | Inhibits intestinal cholesterol absorption (NPC1 L1 transporter) | Possible, with caution | Increased drug exposure in Child-Pugh B/C, higher risk of hepatotoxicity |
| Fibrates | Activate PPAR-α to ↓triglycerides, ↑HDL | Possible, under monitoring | Elevation of liver enzymes, cholestasis, rhabdomyolysis (especially if combined with statins) |
| PCSK9 inhibitors (alirocumab, evolocumab) | Monoclonal antibodies inhibiting PCSK9 to ↑LDL receptor recycling, ↓LDL-C | Data limited; theoretically safer since not hepatically metabolized | Limited clinical data in cirrhosis, safety in advanced liver disease not established |
| Bempedoic acid | Inhibits ATP-citrate lyase (upstream of HMG-CoA reductase) | Potential option, but limited data in cirrhosis | No robust studies in advanced liver disease; potential risk of hepatotoxicity |
| Inclisiran | Small interfering RNA targeting hepatic PCSK9 synthesis, leading to sustained LDL receptor upregulation and LDL-C reduction | Potential option; limited but favorable pharmacologic profile given minimal hepatic metabolism | Very limited clinical data in cirrhosis; safety in Child-Pugh B/C not established |
Mechanistically distinct non-statin therapies enable pathway-specific modulation of hepatic and systemic lipid metabolism. However, ezetimibe, by inhibiting Niemann-Pick C1-Like 1-mediated intestinal cholesterol absorption, lowers LDL-C, whereas MASLD studies report inconsistent effects on steatosis and inflammation despite modest improvements in liver enzymes and fibrosis[73,78]. Similarly, fibrates activate peroxisome proliferator activated receptor-α (PPAR-α) to reduce triglycerides and raise HDL-C and appear safe in primary biliary cirrhosis; nevertheless, data in other cholestatic disorders remain limited[74,79]. By contrast, bempedoic acid, an adenosine triphosphate-citrate lyase inhibitor acting upstream of 3-hydroxy-3-methylglutaryl-coenzyme A reductase, achieves moderate LDL-C reductions but still lacks direct evidence in cirrhotic populations[75,80]. In sharp contrast, PCSK9 inhibitors enhance hepatic LDL receptor recycling and produce profound LDL-C lowering with proven cardiovascular benefit in high-risk patients[81]. Notably, omega-3 fatty acids exert pleiotropic effects by activating PPAR-α and hepatocyte nuclear factor-4α (HNF4α), enhancing β-oxidation via CPT1A and acyl-CoA oxidase 1, suppressing sterol regulatory element-binding protein 1c (SREBP-1c)-driven lipogenesis, and attenuating nuclear factor kappa B–mediated inflammation; clinically, they are safe in compensated cirrhosis and effective for isolated hypertriglyceridemia[82]. Finally, emerging RNA-based therapies, including siRNA, antisense oligonucleotides, and gene-editing platforms, offer liver-specific lipid modulation; however, evidence in cirrhosis remains minimal outside early-phase studies[83-85].
Disrupted intrahepatic thyroid hormone signaling has emerged as a key molecular driver of lipid dysregulation in liver disease. Although first-generation thyroid hormone receptor β-agonists such as sobetirome and eprotirome effectively reduced LDL-C but were limited by off-target skeletal toxicity[86,87]. In contrast, second-generation, liver-directed agents demonstrate improved selectivity: VK2809 enhances hepatic fatty acid oxidation, reduces steatosis, and lowers LDL and triglycerides with favorable safety profiles in compensated cirrhosis[88,89]. More importantly, resmetirom, now approved for metabolic dysfunction-associated steatohepatitis, restores hepatic thyroid hormone receptor β signaling, reduces liver fat, and improves systemic lipid profiles with good tolerability, positioning thyroid hormone analogues as a mechanistically precise strategy for dyslipidemia in liver disease[90,91].
Current evidence supports a paradigm shift from LDL-C-centric assessment toward molecular risk markers such as ApoB, non-HDL-C, and lipoprotein(a), complemented by omics-based profiling[72]. Accordingly, among non-statin therapies, PCSK9 inhibitors and inclisiran appear most promising in Child-Pugh A-B patients, particularly in statin intolerance or when substantial LDL reduction is required, with genetic data suggesting favorable hepatic safety[80,81,92]. Nevertheless, robust evidence in advanced cirrhosis remains scarce: Bempedoic acid is under investigation in MASLD[93], ezetimibe trials largely exclude cirrhotic patients, and PCSK9-targeted therapies lack data in Child-Pugh C. Therefore, the next critical step is the development of dedicated, stage-specific trials to define safety, efficacy, and molecular predictors of response, enabling evidence-based integration of non-statin therapies into cirrhosis care.
Dyslipidemia in cirrhosis reflects a stage-dependent disruption of hepatic lipid regulation rather than a conventional cardiovascular risk state. As liver function deteriorates, molecular lipid markers, such as ApoB, no-HDL-C, lipoprotein(a), and emerging omics signatures, provide more accurate risk stratification than LDL-C. Among non-statin therapies, PCSK9 inhibitors, inclisiran, and liver-directed thyroid hormone receptor β agonists emerge as the most mechanistically grounded options in compensated cirrhosis, while evidence in advanced disease remains limited. Progress in this field will depend on dedicated, stage-specific clinical trials to establish safety, efficacy, and precision-guided treatment strategies.
| 1. | Yoo HJ, Jung KJ, Kim M, Kim M, Kang M, Jee SH, Choi Y, Lee JH. Liver Cirrhosis Patients Who Had Normal Liver Function Before Liver Cirrhosis Development Have the Altered Metabolic Profiles Before the Disease Occurrence Compared to Healthy Controls. Front Physiol. 2019;10:1421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (5)] |
| 2. | Ginès P, Krag A, Abraldes JG, Solà E, Fabrellas N, Kamath PS. Liver cirrhosis. Lancet. 2021;398:1359-1376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1345] [Cited by in RCA: 1160] [Article Influence: 232.0] [Reference Citation Analysis (6)] |
| 3. | Memon MY, Ali G, Bukhari SI, Sandeelo N, Alvi H, Talib A, Imran P, Baloch I, Shah SZA. Dyslipidemia is an Independent Predictor of Rapid Progressive Disease in Patientswith Chronic Liver Disease. J Pharm Res Int. 2022;34:12-18. [DOI] [Full Text] |
| 4. | Patel SB, Belalcazar LM, Afreen S, Balderas R, Hegele RA, Karpe F, Ponte-Negretti CI, Rajpal A. American Association of Clinical Endocrinology Consensus Statement: Algorithm for Management of Adults with Dyslipidemia - 2025 Update. Endocr Pract. 2025;31:1207-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 5. | Jan A, Narwaria M, Mahawar KK. A Systematic Review of Bariatric Surgery in Patients with Liver Cirrhosis. Obes Surg. 2015;25:1518-1526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 90] [Article Influence: 8.2] [Reference Citation Analysis (1)] |
| 6. | Ress C, Kaser S. Mechanisms of intrahepatic triglyceride accumulation. World J Gastroenterol. 2016;22:1664-1673. [PubMed] [DOI] [Full Text] |
| 7. | Li X, Wang L, Li D, Niu J, Gao P. Dyslipidemia is a Risk Factor for the Incidence and Severity of Drug-Induced Liver Injury (DILI): A Retrospective Population-Based Study in China. Med Sci Monit. 2019;25:3344-3353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 8. | Yun B, Ahn SH, Yoon JH, Kim BK. Statin use and risk of progression to liver cirrhosis in chronic hepatitis B independent of conventional risk factors: A nationwide study. Hepatol Commun. 2022;6:2455-2464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 9. | Gratacós-Ginès J, Pose E. Review of the role of statins in cirrhosis and portal hypertension. Clin Liver Dis (Hoboken). 2023;22:50-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (2)] |
| 10. | Pirillo A, Catapano AL. Statin intolerance: diagnosis and remedies. Curr Cardiol Rep. 2015;17:27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Banach M, Rizzo M, Toth PP, Farnier M, Davidson MH, Al-Rasadi K, Aronow WS, Athyros V, Djuric DM, Ezhov MV, Greenfield RS, Hovingh GK, Kostner K, Serban C, Lighezan D, Fras Z, Moriarty PM, Muntner P, Goudev A, Ceska R, Nicholls SJ, Broncel M, Nikolic D, Pella D, Puri R, Rysz J, Wong ND, Bajnok L, Jones SR, Ray KK, Mikhailidis DP. Statin intolerance - an attempt at a unified definition. Position paper from an International Lipid Expert Panel. Arch Med Sci. 2015;11:1-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 293] [Article Influence: 26.6] [Reference Citation Analysis (2)] |
| 12. | Chang FM, Wang YP, Lang HC, Tsai CF, Hou MC, Lee FY, Lu CL. Statins decrease the risk of decompensation in hepatitis B virus- and hepatitis C virus-related cirrhosis: A population-based study. Hepatology. 2017;66:896-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 99] [Article Influence: 11.0] [Reference Citation Analysis (2)] |
| 13. | Hung TH, Tsai CC, Lee HF. Statin use in cirrhotic patients with infectious diseases: A population-based study. PLoS One. 2019;14:e0215839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 14. | Asrani SK, Devarbhavi H, Eaton J, Kamath PS. Burden of liver diseases in the world. J Hepatol. 2019;70:151-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3074] [Cited by in RCA: 2523] [Article Influence: 360.4] [Reference Citation Analysis (7)] |
| 15. | Xiao S, Xie W, Zhang Y, Lei L, Pan Y. Changing epidemiology of cirrhosis from 2010 to 2019: results from the Global Burden Disease study 2019. Ann Med. 2023;55:2252326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 16. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966-1986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2198] [Cited by in RCA: 2022] [Article Influence: 674.0] [Reference Citation Analysis (8)] |
| 17. | Ji H, Cheng S; Heart-Liver Axis Research Collaboration. Sex differences in prevalence and prognosis of steatotic liver disease phenotypes: Biological sex matters. J Hepatol. 2024;80:e68-e69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 18. | Lo GH. Did Statins Really Reduce the Risk of Cirrhosis and Decompensation in Chronic Hepatitis B Patients? Am J Gastroenterol. 2017;112:386-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 19. | Slim M, Ruiz-Cabello E, Robles-Díaz M, Lucena MI, Andrade RJ. A New Hepatoprotective Effect of Statins: Are They Always Safe for the Liver? Am J Gastroenterol. 2017;112:384-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 20. | Hidalgo I, Fonseca-Coronado S, Ceballos G, Meaney E, Nájera N. Dyslipidemias, fatty liver, and cardiovascular disease. Cardiovasc Metab Sci. 2022;33:134-139. [RCA] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 21. | Gallo G, Nalli G, Baratta F, Desideri G, Savoia C. Metabolic Dysfunction-Associated Steatotic Liver Disease: A Silent Driver of Cardiovascular Risk and a New Target for Intervention. Int J Mol Sci. 2025;26:8081. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (5)] |
| 22. | Badawi R, Abu Rahma MZ, Ramadan HK, Soliman S, Mohareb DA, Hawash N, Elkafoury R, Abd-Elsalam S. Lipid Profiles as Markers for the Severity of Liver Diseases in Cirrhotic Patients. Open Biomark J. 2021;11:93-98. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 23. | Liou JW, Mani H, Yen JH. Viral Hepatitis, Cholesterol Metabolism, and Cholesterol-Lowering Natural Compounds. Int J Mol Sci. 2022;23:3897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 24. | Patel R, Mueller M. Alcohol-Associated Liver Disease. 2023 Jul 13. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 25. | Girish V, John S. Metabolic Dysfunction-Associated Steatotic Liver Disease (MΑSLD). 2025 Aug 9. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 26. | Penz-Österreicher M, Österreicher CH, Trauner M. Fibrosis in autoimmune and cholestatic liver disease. Best Pract Res Clin Gastroenterol. 2011;25:245-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 62] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 27. | Manns MP, Czaja AJ, Gorham JD, Krawitt EL, Mieli-Vergani G, Vergani D, Vierling JM; American Association for the Study of Liver Diseases. Diagnosis and management of autoimmune hepatitis. Hepatology. 2010;51:2193-2213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1210] [Cited by in RCA: 1025] [Article Influence: 64.1] [Reference Citation Analysis (2)] |
| 28. | Bjornstad P, Eckel RH. Pathogenesis of Lipid Disorders in Insulin Resistance: a Brief Review. Curr Diab Rep. 2018;18:127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 187] [Cited by in RCA: 156] [Article Influence: 19.5] [Reference Citation Analysis (2)] |
| 29. | Akhtar DH, Iqbal U, Vazquez-Montesino LM, Dennis BB, Ahmed A. Pathogenesis of Insulin Resistance and Atherogenic Dyslipidemia in Nonalcoholic Fatty Liver Disease. J Clin Transl Hepatol. 2019;7:362-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 30. | Freeman AM, Acevedo LA, Pennings N. Insulin Resistance. 2023 Aug 17. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 31. | Martin A, Lang S, Goeser T, Demir M, Steffen HM, Kasper P. Management of Dyslipidemia in Patients with Non-Alcoholic Fatty Liver Disease. Curr Atheroscler Rep. 2022;24:533-546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 32. | Muzurović E, Peng CC, Belanger MJ, Sanoudou D, Mikhailidis DP, Mantzoros CS. Nonalcoholic Fatty Liver Disease and Cardiovascular Disease: a Review of Shared Cardiometabolic Risk Factors. Hypertension. 2022;79:1319-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 105] [Article Influence: 26.3] [Reference Citation Analysis (1)] |
| 33. | Asrih M, Jornayvaz FR. Metabolic syndrome and nonalcoholic fatty liver disease: Is insulin resistance the link? Mol Cell Endocrinol. 2015;418 Pt 1:55-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 235] [Article Influence: 21.4] [Reference Citation Analysis (2)] |
| 34. | Januario E, Barakat A, Rajsundar A, Fatima Z, Nanda Palienkar V, Bullapur AV, Singh Brar S, Kharel P, Koyappathodi Machingal MM, Backosh A. A Comprehensive Review of Pathophysiological Link Between Non-alcoholic Fatty Liver Disease, Insulin Resistance, and Metabolic Syndrome. Cureus. 2024;16:e75677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (2)] |
| 35. | Behera BP. A cross-sectional observational study of lipid profile of cirrhosis of liver patients in a teaching hospital in North Odisha, India. Int J Adv Med. 2020;7:783-789. [DOI] [Full Text] |
| 36. | Unger LW, Forstner B, Schneglberger S, Muckenhuber M, Eigenbauer E, Scheiner B, Mandorfer M, Trauner M, Reiberger T. Patterns and prevalence of dyslipidemia in patients with different etiologies of chronic liver disease. Wien Klin Wochenschr. 2019;131:395-403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 37. | Charlat M, Poltiyelova E, Silverman J, Lit A, Frishman WH, Lebovics E. Hypercholesterolemia of Cholestasis. Cardiol Rev. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 38. | Kalra DK, Shotwell M, Garg A, Duell PB, Wilson DP, Martin SS, Soffer DE, Rosenson RS, Shapiro MD, Ahmad Z, Underberg J, Sperling L, Jortani SA, Remaley A. Lipoprotein X - Pathophysiology, diagnosis, and management. J Clin Lipidol. 2025;19:759-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 39. | Stojkoski N, Bertrand M, Messaoudi K, Bendavid C, Al-Shami R, Moreau C. Biochemical exploration of cholestasis: interpretation, traps and interferences. Clin Biochem. 2025;135:110852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 40. | Shoaib N, Khan Z, Ibrahim M, Hafeez A, Fatima A, Imran H, Saleem F, Hassan Askari SM, Gull S. Dyslipidemia and impaired liver function biomarkers in patients with hepatitis B liver cirrhosis. J Taibah Univ Med Sci. 2023;18:748-754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 41. | Gîrleanu I, Trifan A, Huiban L, Muzîca C, Petrea OC, Sîngeap AM, Cojocariu C, Chiriac S, Cuciureanu T, Costache II, Stanciu C. Ischemic Heart Disease and Liver Cirrhosis: Adding Insult to Injury. Life (Basel). 2022;12:1036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 42. | Boldys A, Buldak L. Metabolic dysfunction-associated steatotic liver disease: Navigating terminological evolution, diagnostic frontiers and therapeutic horizon-an editorial exploration. World J Gastroenterol. 2024;30:2387-2390. [PubMed] [DOI] [Full Text] |
| 43. | Song Z, Miao X, Liu S, Hu M, Xie X, Sun Y, Leng S. Associations between cardiometabolic indices and the onset of metabolic dysfunction-associated steatotic liver disease as well as its progression to liver fibrosis: a cohort study. Cardiovasc Diabetol. 2025;24:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 44. | Kim HY, Rinella ME. Emerging therapies and real-world application of metabolic dysfunction-associated steatotic liver disease treatment. Clin Mol Hepatol. 2025;31:753-770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 45. | Arvind A, Osganian SA, Cohen DE, Corey KE. Lipid and Lipoprotein Metabolism in Liver Disease. 2019 Jul 21. In: Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. [PubMed] |
| 46. | Perez-Matos MC, Sandhu B, Bonder A, Jiang ZG. Lipoprotein metabolism in liver diseases. Curr Opin Lipidol. 2019;30:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 47. | Sira MM, Sira AM. Lipid-Liver Crosstalk in Health and Disease. J Liver Res Disord Ther. 2016;2:00016. [DOI] [Full Text] |
| 48. | Ghadir MR, Riahin AA, Havaspour A, Nooranipour M, Habibinejad AA. The relationship between lipid profile and severity of liver damage in cirrhotic patients. Hepat Mon. 2010;10:285-288. [PubMed] |
| 49. | Nemes K, Åberg F, Gylling H, Isoniemi H. Cholesterol metabolism in cholestatic liver disease and liver transplantation: From molecular mechanisms to clinical implications. World J Hepatol. 2016;8:924-932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 50. | Huang YW, Lee CL, Yang SS, Fu SC, Chen YY, Wang TC, Hu JT, Chen DS. Statins Reduce the Risk of Cirrhosis and Its Decompensation in Chronic Hepatitis B Patients: A Nationwide Cohort Study. Am J Gastroenterol. 2016;111:976-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 91] [Article Influence: 9.1] [Reference Citation Analysis (2)] |
| 51. | Gurevitz C, Shalaurova I, Connelly MA, Rosenson RS. LpX and LpZ associated hypercholesterolemia in a patient with primary sclerosing cholangitis - A case report. J Clin Lipidol. 2025;19:1171-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 52. | van den Berg EH, Flores-Guerrero JL, Gruppen EG, Garcia E, Connelly MA, de Meijer VE, Bakker SJL, Blokzijl H, Dullaart RPF. Profoundly Disturbed Lipoproteins in Cirrhotic Patients: Role of Lipoprotein-Z, a Hepatotoxic LDL-like Lipoprotein. J Clin Med. 2022;11:1223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 53. | van den Berg EH, Flores-Guerrero JL, Dullaart RPF. Lipoprotein Z, an abnormal LDL-like lipoprotein, independently predicts mortality in cirrhosis. Eur J Intern Med. 2022;101:128-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 54. | Felmlee DJ, Hafirassou ML, Lefevre M, Baumert TF, Schuster C. Hepatitis C virus, cholesterol and lipoproteins--impact for the viral life cycle and pathogenesis of liver disease. Viruses. 2013;5:1292-1324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 110] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 55. | Nagano T, Seki N, Tomita Y, Sugita T, Aida Y, Itagaki M, Sutoh S, Abe H, Tsubota A, Aizawa Y. Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions. Int J Mol Sci. 2015;16:20576-20594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 56. | Borén J, Adiels M, Björnson E, Matikainen N, Söderlund S, Rämö J, Ståhlman M, Ripatti P, Ripatti S, Palotie A, Mancina RM, Hakkarainen A, Romeo S, Packard CJ, Taskinen MR. Effects of TM6SF2 E167K on hepatic lipid and very low-density lipoprotein metabolism in humans. JCI Insight. 2020;5:e144079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 57. | Méndez-Sánchez N, Cerda-Reyes E, Higuera-de-la-Tijera F, Salas-García AK, Cabrera-Palma S, Cabrera-Álvarez G, Cortez-Hernández C, Pérez-Arredondo LA, Purón-González E, Coronado-Alejandro E, Panduro A, Rodríguez-Hernández H, Cruz-Ramón VC, Valencia-Rodríguez A, Qi X, Hamdan-Pérez N, Aguilar-Olivos NE, Barranco-Fragoso B, Ramírez-Pérez O, Vera-Barajas A. Dyslipidemia as a risk factor for liver fibrosis progression in a multicentric population with non-alcoholic steatohepatitis. F1000Res. 2020;9:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 58. | Singh M, Khan MI, Gupta AC, Giri R, Alam A. Role of lipid profile in assessment of severity of cirrhosis. Int J Adv Med. 2019;6:1303-1307. [DOI] [Full Text] |
| 59. | Buechler C, Aslanidis C. Role of lipids in pathophysiology, diagnosis and therapy of hepatocellular carcinoma. Biochim Biophys Acta Mol Cell Biol Lipids. 2020;1865:158658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 60. | Zhang QQ, Lu LG. Nonalcoholic Fatty Liver Disease: Dyslipidemia, Risk for Cardiovascular Complications, and Treatment Strategy. J Clin Transl Hepatol. 2015;3:78-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 120] [Article Influence: 10.9] [Reference Citation Analysis (2)] |
| 61. | Bassani L, Fernandes SA, Raimundo FV, Harter DL, Gonzalez MC, Marroni CA. Lipid profile of cirrhotic patients and its association with prognostic scores: a cross-sectional study. Arq Gastroenterol. 2015;52:210-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 62. | Cheng R, Tan N, Kang Q, Luo H, Chen H, Pan J, Han Y, Yang Y, Xu X. High-density lipoprotein cholesterol is a predictor of survival in cirrhotic patients with acute gastrointestinal bleeding: a retrospective study. BMC Gastroenterol. 2020;20:381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 63. | Kaplan DE, Serper MA, Mehta R, Fox R, John B, Aytaman A, Baytarian M, Hunt K, Albrecht J, Njei B, Taddei TH; VOCAL Study Group. Effects of Hypercholesterolemia and Statin Exposure on Survival in a Large National Cohort of Patients With Cirrhosis. Gastroenterology. 2019;156:1693-1706.e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 122] [Article Influence: 17.4] [Reference Citation Analysis (1)] |
| 64. | Gu C, Dong L, Chai L, Tong Z, Gao F, Ageno W, Romeiro FG, Qi X. Risk of Coronary Artery Disease in Patients with Liver Cirrhosis: A Systematic Review and Meta-analysis. J Clin Transl Hepatol. 2025;13:93-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 65. | Ismaiel A, Dumitraşcu DL. Cardiovascular Risk in Fatty Liver Disease: The Liver-Heart Axis-Literature Review. Front Med (Lausanne). 2019;6:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 139] [Article Influence: 19.9] [Reference Citation Analysis (1)] |
| 66. | Habib S, Murakami T, Takyar V, Patel K, Dominguez C, Zhan Y, Mehrpour O, Hsu CH. The Impact of Metabolic Syndrome on the Prognosis of High-Risk Alcoholic Hepatitis Patients: Redefining Alcoholic Hepatitis. Gastroenterology Res. 2023;16:25-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 67. | Alnees M, Awwad M, Najajra D, Darwish A, Heih O, Al-Ali T, Hamdeh NA, Abdullah A, Ahmad J, Jarrar K, Al-Ihribat A, Salloum O, Abdoh Q, Khadija HA. Predictive factors for metabolic syndrome in patients with metabolic dysfunction-associated steatotic liver disease (MASLD). BMC Gastroenterol. 2025;25:562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 68. | Hliwa A, Lange-Andrzejewska O, Laski D, Sledzinski M, Remiszewski P, Drobinska A, Mika A, Sledzinski T. Alterations of the Fatty Acid Profile and the Expression of Genes Related to FA Metabolism in Cirrhotic Liver Tissue. Int J Mol Sci. 2024;25:8115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 69. | Islam MS, Wei P, Suzauddula M, Nime I, Feroz F, Acharjee M, Pan F. The interplay of factors in metabolic syndrome: understanding its roots and complexity. Mol Med. 2024;30:279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 46.0] [Reference Citation Analysis (1)] |
| 70. | Yu L, Liu Y, Guo R, Yang T, Pan G, He Y, Gao S, Yang R, Li Z, Li L, Yu C. The metabolic syndrome-insulin resistance index: a tool for identifying dyslipidemia across varied glucose metabolic score in patients with cardiovascular disease. Front Endocrinol (Lausanne). 2025;16:1473308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 71. | Kreidieh M, Hamadi R, Alsheikh M, Al Moussawi H, Deeb L. Statin Use in Patients With Chronic Liver Disease and Cirrhosis: Current Evidence and Future Directions. Gastroenterology Res. 2022;15:1-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 72. | Berberich AJ, Hegele RA. A Modern Approach to Dyslipidemia. Endocr Rev. 2022;43:611-653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 282] [Article Influence: 70.5] [Reference Citation Analysis (1)] |
| 73. | Kamanu C, Karalis DG. The Role of Non-Statin Lipid Lowering Therapies to Reduce ASCVD Events in Primary Prevention. Curr Atheroscler Rep. 2025;27:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 74. | Pouwels S, Sakran N, Graham Y, Leal A, Pintar T, Yang W, Kassir R, Singhal R, Mahawar K, Ramnarain D. Non-alcoholic fatty liver disease (NAFLD): a review of pathophysiology, clinical management and effects of weight loss. BMC Endocr Disord. 2022;22:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 716] [Cited by in RCA: 613] [Article Influence: 153.3] [Reference Citation Analysis (7)] |
| 75. | Fernández-Friera L, Fuster V, López-Melgar B, Oliva B, García-Ruiz JM, Mendiguren J, Bueno H, Pocock S, Ibáñez B, Fernández-Ortiz A, Sanz J. Normal LDL-Cholesterol Levels Are Associated With Subclinical Atherosclerosis in the Absence of Risk Factors. J Am Coll Cardiol. 2017;70:2979-2991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 250] [Article Influence: 27.8] [Reference Citation Analysis (1)] |
| 76. | Castellote J, Ariza J, Rota R, Girbau A, Xiol X. Serious drug-induced liver disease secondary to ezetimibe. World J Gastroenterol. 2008;14:5098-5099. [PubMed] [DOI] [Full Text] |
| 77. | Frühwald L, Fasching P, Dobrev D, Kaski JC, Borghi C, Wassmann S, Huber K, Semb AG, Agewall S, Drexel H. Management of dyslipidaemia in patients with comorbidities-facing the challenge. Eur Heart J Cardiovasc Pharmacother. 2025;11:164-173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 78. | Boskabadi AR, Khodabandelu S, Rahimi Y, Motamedi A, Asili P, Ghasempour A, Keshavarzian A, Noori S, Rahmanian M. Efficacy and Safety of Ezetimibe for Non-Alcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Curr Rev Clin Exp Pharmacol. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 79. | Carrion AF, Lindor KD, Levy C. Safety of fibrates in cholestatic liver diseases. Liver Int. 2021;41:1335-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 80. | Laufs U, Banach M, Mancini GBJ, Gaudet D, Bloedon LT, Sterling LR, Kelly S, Stroes ESG. Efficacy and Safety of Bempedoic Acid in Patients With Hypercholesterolemia and Statin Intolerance. J Am Heart Assoc. 2019;8:e011662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 306] [Article Influence: 43.7] [Reference Citation Analysis (1)] |
| 81. | Feder S, Wiest R, Weiss TS, Aslanidis C, Schacherer D, Krautbauer S, Liebisch G, Buechler C. Proprotein convertase subtilisin/kexin type 9 (PCSK9) levels are not associated with severity of liver disease and are inversely related to cholesterol in a cohort of thirty eight patients with liver cirrhosis. Lipids Health Dis. 2021;20:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 82. | Speliotes EK, Balakrishnan M, Friedman LS, Corey KE. Treatment of Dyslipidemia in Common Liver Diseases. Clin Gastroenterol Hepatol. 2018;16:1189-1196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 83. | Jakielska E, Głuszak P, Walczak M, Bryl W. Effects of PCSK9 inhibitors on metabolic-associated fatty liver disease: a short review. Prz Gastroenterol. 2023;18:148-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 84. | Lawitz EJ, Shevell DE, Tirucherai GS, Du S, Chen W, Kavita U, Coste A, Poordad F, Karsdal M, Nielsen M, Goodman Z, Charles ED. BMS-986263 in patients with advanced hepatic fibrosis: 36-week results from a randomized, placebo-controlled phase 2 trial. Hepatology. 2022;75:912-923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 85. | Bhatia HS, Bajaj A, Goonewardena SN, Moriarty PM. Pelacarsen: Mechanism of action and Lp(a)-lowering effect. J Clin Lipidol. 2025;19:1208-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 86. | Sinha RA, Bruinstroop E, Yen PM. Actions of thyroid hormones and thyromimetics on the liver. Nat Rev Gastroenterol Hepatol. 2025;22:9-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 87. | Freund MET, van der Most F, Groeneweg S, van Geest FS, Visser WE. Thyroid Hormone Analogs: Recent Developments. Thyroid. 2025;35:990-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 88. | Soares De Oliveira L, Ritter MJ. Thyroid hormone and the Liver. Hepatol Commun. 2025;9:e0596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 89. | Ratziu V, Scanlan TS, Bruinstroop E. Thyroid hormone receptor-β analogues for the treatment of metabolic dysfunction-associated steatohepatitis (MASH). J Hepatol. 2025;82:375-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 51.0] [Reference Citation Analysis (1)] |
| 90. | Polyzos SA, Targher G. Hepatic thyroid hormone receptor-β signalling: Mechanisms and recent advancements in the treatment of metabolic dysfunction-associated steatohepatitis. Diabetes Obes Metab. 2025;27:1635-1647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 91. | Kuchay MS, Isaacs S, Misra A. Intrahepatic hypothyroidism in MASLD: Role of liver-specific thyromimetics including resmetirom. Diabetes Metab Syndr. 2024;18:103034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 92. | Vujkovic M. Assessing Long-Term Liver Safety of Statins and PCSK9 Inhibitors Using Human Genetics. Cell Mol Gastroenterol Hepatol. 2024;17:173-174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 93. | Butera E, Termite F, Esposto G, Galasso L, Mignini I, Borriello R, Ainora ME, Miele L, Gasbarrini A, Zocco MA. Exploring the Role of Bempedoic Acid in Metabolic Dysfunction Associated Steatotic Liver Disease: Actual Evidence and Future Perspectives. Int J Mol Sci. 2024;25:6938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |