Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115411
Revised: November 18, 2025
Accepted: January 19, 2026
Published online: March 27, 2026
Processing time: 161 Days and 16.9 Hours
Metabolic-associated steatohepatitis (MASH) is a prevalent metabolic condition affecting one-third of adults worldwide.
To evaluate the safety and efficacy of efruxifermin in patients with compensated MASH cirrhosis.
A comprehensive search was conducted up to May 2025. Outcomes assessed included fibrosis improvement, MASH resolution, liver stiffness, and metabolic indices such as high-density lipoprotein cholesterol and ho
The meta-analysis included five randomized controlled trials involving 450 patients. Efruxifermin led to significant improvement in fibrosis without worsening of MASH and MASH resolution without worsening of fibrosis (risk ratio = 3.00, 95% confidence interval: 2.05-4.38). Further, a significantly higher patients noted reduction in aspartate aminotransferase, alanine aminotransferase and homeostatic model assessment of insulin resistance levels, and increased high-density lipoprotein cholesterol. While efruxifermin was generally well-tolerated, gastrointestinal side effects such as nausea and diarrhea were more frequent in the efruxifermin group. The incidence of serious adverse events was comparable between groups.
Efruxifermin demonstrated significant efficacy in improving fibrosis, resolving MASH, and enhancing metabolic health in patients with compensated MASH cirrhosis. These findings support further investigation of efruxifermin in larger, long-term trials to confirm its therapeutic potential and safety profile.
Core Tip: This GRADE-assessed systematic review and meta-analysis of randomized controlled trials evaluates the efficacy and safety of efruxifermin, a long-acting fibroblast growth factor 21 analogue, in patients with compensated metabolic dysfunction-associated steatohepatitis (MASH) cirrhosis. Efruxifermin demonstrated significant improvements in liver fibrosis, MASH resolution, and metabolic parameters, with a tolerable safety profile. These findings highlight efruxifermin’s potential as a novel therapeutic option for compensated MASH cirrhosis, addressing a critical unmet need in this population.
- Citation: Alam U, Rath S, Ansab M, Kashif A, Sethi FA, Siddiqui HT, Afridi ZAK, Burhan M, Ali MA, Moiz A, Bacha Z, Khan A, Ahmed R, Alatise MB, Kamil KA. Efruxifermin in patients with metabolic dysfunction-associated steatohepatitis: A GRADE-assessed systematic review and meta-analysis. World J Hepatol 2026; 18(3): 115411
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115411.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115411
Metabolical dysfunction-associated fatty liver disease is extremely common, affecting about one in three adults worldwide[1]. Its progressive form, metabolic dysfunction-associated steatohepatitis (MASH), with steatosis along with hepatocellular injury and inflammation, is now the most common chronic liver disease globally[2]. A large fraction of patients with MASH will develop advanced fibrosis and ultimately, cirrhosis. Compensated cirrhosis (Child-Pugh A) will often go unrecognized clinically for years but represents a strong likelihood of decompensation or hepatocellular carcinoma risk. Importantly, currently there are no approved pharmacotherapies to treat MASH cirrhosis, and this represents a serious unmet medical need[3]. The increasing prevalence of obesity/metabolic syndrome suggests an even greater incidence of MASH cirrhosis in the future, and thus ultimately increases the need for effective intervention.
Fibroblast growth factor (FGF)-21 is a normal endocrine regulator of glucose and lipid metabolism and has known antifibrotic and hepatoprotective activities[4]. Efruxifermin, previously (AKR-001), is a recombinant, long-acting Fc-fusion engineered analogue of FGF-21 designed to take advantage of these beneficial effects on metabolism. Efruxifermin is a bivalent Fc-FGF-21 molecule that agonizes the FGF-21 receptor complex (FGF receptor 1c/2c/3c)[5], which has been shown to increase insulin sensitivity, lipid oxidative metabolism, decrease hepatic steatosis and hepatic inflammation. FGF-21 signaling has been shown in preclinical studies to decrease fibrogenesis and improve metabolic state in MASH models, so efruxifermin may provide additive benefits in MASH to tackle both metabolic dysregulation and fibrotic pathways[6].
Most recent systematic reviews of drug therapies for MASH have not included cirrhotic patients, with only biopsied cases of MASH indicated. In a network meta-analysis of 40 randomized controlled trials (RCTs; n approximately of 6600) of patients with confirmed MASH, Kovalic[7] identified obeticholic acid, resmetirom and pioglitazone for histological endpoints. Similarly, Jeong et al[8], recently published a study pooling RCTs of FGF-21 analogues with MASH (including efruxifermin) and determined when compared to placebo that fibrosis and MASH were better, but a higher rate of treatment emergent adverse events were also noted. In general, these meta-analyses [and others such as for glucagon-like peptide (GLP)-1 agonist or FGF-19 analogues] were not using cirrhotic patients. In fact, dedicated study in compensated MASH cirrhosis patients had frequently failed (e.g., semaglutide showed no fibrosis benefit in F4 patients)[9].
Also, to date, no meta-analysis has focused specifically on efruxifermin in patients with MASH cirrhosis. Most reviews emphasize endpoints of efficacy, with limited integration of safety. Thus, a meta-analysis targeting patients with compensated MASH cirrhosis integrating both efficacy and safety outcomes for efruxifermin, is a useful contribution to the field and is therefore the rationale for this meta-analysis.
This systematic review was carried out following the guidelines provided by the Cochrane Collaboration[10] and by Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines[11]. These guidelines were adhered to during the design, implementation, analysis, and reporting of the review. Additionally, the study protocol was registered in the International Prospective Register of Systematic Reviews under registration No. CRD420251059381.
An electronic search was performed on PubMed, EMBASE, and Web of Science, encompassing all available records from their inception up to May 2025. The keywords listed below were applied, without any language restrictions. The following keywords were used: “Efruxifermin”, “Fibroblast growth factor (FGF)-21 analogs”, “Metabolic dysfunction-associated steatotic liver disease (MASLD)”, “non-alcoholic fatty liver disease (NAFLD)” and “non-alcoholic steatohepatitis (NASH)”. Detailed search strategy is provided in Supplementary Table 1.
All studies retrieved from the online search were uploaded into Rayyan software for screening, and duplicate records were removed. The remaining studies were first evaluated based on their titles and abstracts. Full-text articles were retrieved for further evaluation if either reviewer deemed the abstract potentially relevant. Two independent reviewers (Rath S and Sajjad F) assessed the eligibility of each study based on predefined inclusion criteria. Conflicts were addressed through dialogue and consultation with a third reviewer (Alam U).
Included criteria: (1) Involved patients with MASH; (2) Efruxifermin as intervention; (3) Placebo as control; (4) Reported at least one relevant outcome; and (5) RCTs.
Exclusion criteria: (1) Observational studies; (2) Overlapping populations, defined by shared institutions and recruitment periods; (3) Populations outside the scope of interest; (4) Republished literature; (5) Protocols without reported results; (6) Reviews, abstracts, case reports, case series, background articles, expert opinions, or in vivo/in vitro studies; (7) Repeated data from the same clinical trial; and (8) Single armed studies.
Data from the included studies were extracted by two authors (Alam U and Siddiqui HT) into an Excel spreadsheet using a pre-piloted form; baseline data consisted of study characteristics (study type, country, sample size), demographics (age, male, female), clinical parameters (weight, body mass index, type 2 diabetes), liver health [alanine aminotransferase (AST), aspartate aminotransferase (ALT), procollagen type III N-terminal peptide, enhanced liver fibrosis (ELF) scores, liver stiffness], and metabolic parameters [glycated hemoglobin A1c, triglycerides, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, non HDL cholesterol, and homeostatic model assessment (HOMA) of insulin resistance (IR)]. Outcomes were categorized into fibrosis profile, hematological and serum parameters, and adverse events. The fibrosis profile outcomes of this study included achieving at least one stage of improvement in fibrosis without worsening of MASH, MASH resolution without worsening fibrosis, at least a two-point improvement in the non-alcoholic fatty liver disease activity score (NAS) with no worsening of fibrosis, > 5% normalized liver fat, ELF test score, liver stiffness measurement score (kPa). The hematological and serum parameters comprised change in AST, change in ALT, percentage change in adiponectin, HDL cholesterol, LDL cholesterol, change in triglycerides and HOMA index, adverse events included serious adverse events, adverse event leading to discontinuation, nausea, diarrhoea, and injection site bruising.
The revised Cochrane risk of bias tool for randomized trials[12] was used for evaluating bias across five domains in the RCTs randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selection of reported results. Each study’s overall risk of bias was categorized as low, some concerns, or high risk. This tool was similarly utilized in another study too[13], ensuring consistency and methodological rigor.
The GRADE tool was utilized by two independent authors (Alam U and Moiz A) with the assistance of the GRADEpro Guideline Development Tool[14] to evaluate the level of certainty of the evidence in this meta-analysis, with categorizations ranging from high to very low[15]. This approach was also utilized in a previous study, ensuring consistency in the appraisal of evidence quality[16]. Any disagreements were discussed and resolved through consensus.
Statistical analysis was conducted using R software and R Studio (version 4.4.2; R Core Team, Vienna, Austria), applying DerSimonian and Laird’s random-effects model to compute pooled estimates with 95% confidence intervals (CIs)[17]. The results were visualized in forest plots. Binary outcomes were analyzed using risk ratios (RRs), while continuous outcomes were assessed with mean differences (MDs), both presented in forest plots. Heterogeneity was evaluated using the Cochrane Q χ2 test and the I2 statistic, with P < 0.10 and I2 > 50% indicating significant heterogeneity[18]. To test the robustness of the pooled estimates, a leave-one-out analysis was performed, sequentially removing each study and reanalyzing the data ensuring that no individual study unduly impacted the overall effect estimates.
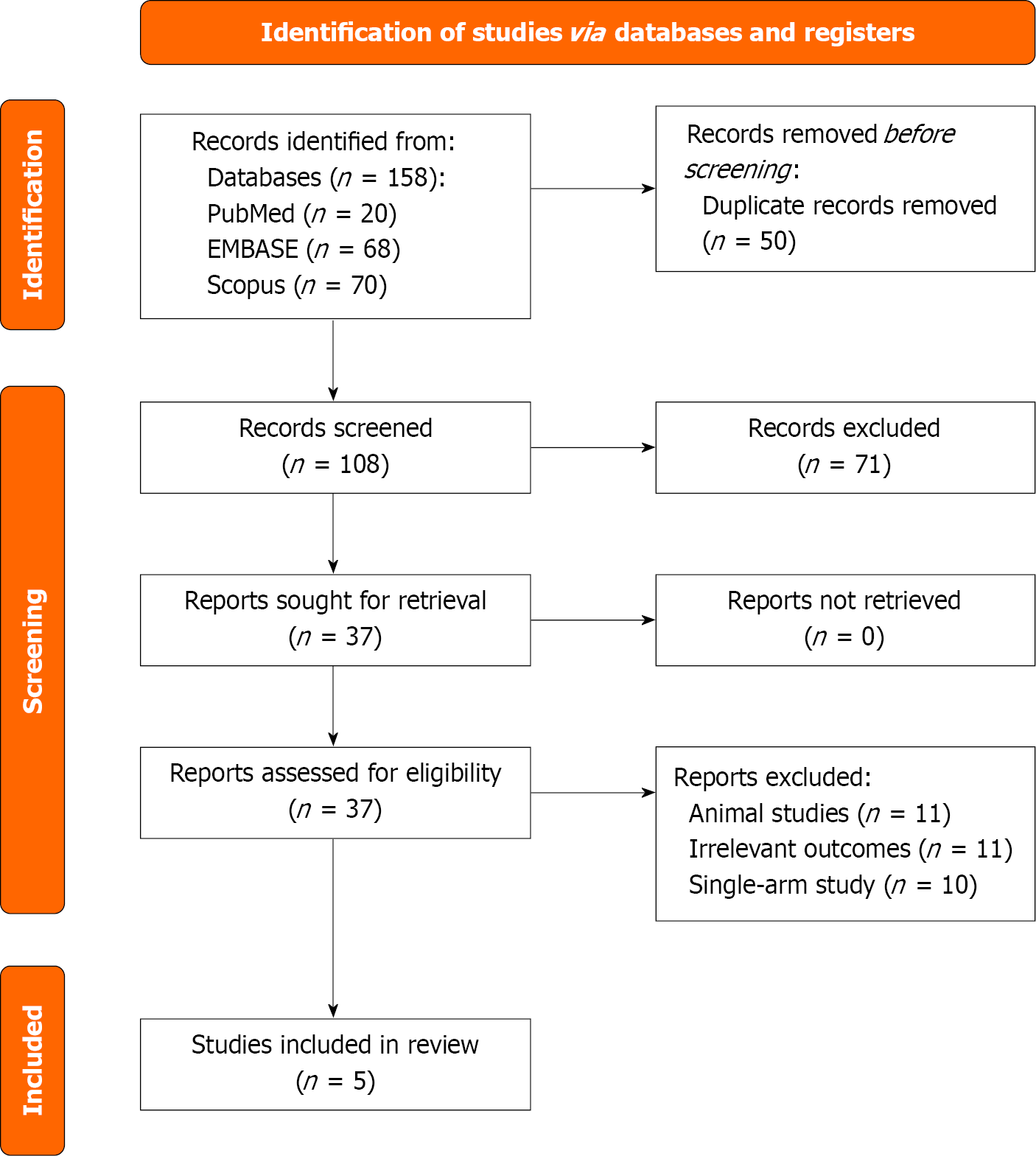
The search identified 158 records from 68 EMBASE, 20 from PubMed, and 70 from Scopus. After removing 50 duplicate records, 108 studies remained for the title and abstract screening. Following the screening process, 71 studies were excluded based on title and abstract evaluation. 37 full-text articles were assessed for eligibility; 5 studies were included in the qualitative and quantitative synthesis. These comprised five RCTs[19-23]. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram details the study selection process (Figure 1).
A total of 450 patients were included across the analyzed studies, with 305 patients in the efruxifermin group and 145 in the placebo group. The studies were published between 2024 and 2025, all conducted in the United States. The detailed baseline characteristics, demographics, clinical, and metabolic parameters are presented in Table 1.
| Baseline characteristics | Harrison et al[20] | Harrison et al[22] | Noureddin et al[21] | Harrison et al[19] | Harrison et al[23] | |||||
| Efruxifermin | Placebo | Efruxifermin | Placebo | Efruxifermin | Placebo | Efruxifermin | Placebo | Efruxifermin | Placebo | |
| Study type | RCT phase 2a | RCT phase 2b | RCT phase 2b | RCT phase 2a | RCT phase 2b | |||||
| Country | United States | United States | United States, Puerto Rico, Mexico | United States | United States | |||||
| Demographics | ||||||||||
| Sample size | 20 | 10 | 21 | 10 | 120 | 61 | 59 | 21 | 85 | 43 |
| Age (years) | 61.1 ± 10.0 | 57.1 ± 14.4 | 59 | 55 | 60.5 ± 8.6 | 61 ± 7.5 | 52.0 ± 13.3 | 52.4 ± 9.6 | 52.0 ± 13 | 52.4 ± 9.6 |
| Male | 4 (20.0) | 7 (70.0) | 12 (57) | 1 (10) | 37 (31) | 23 (38) | 28 (47) | 6 (29) | 33 (38) | 16 (37) |
| Female | 16 (80.0) | 3 (30.0) | 9 (43) | 9 (90) | 83 (69) | 38 (62) | 31 (53) | 15 (71) | 52 (62) | 27 (63) |
| Clinical parameters | ||||||||||
| Weight (kg) | 97.9 ± 19.8 | 119.1 ± 30.5 | 101 | 96 | NR | NR | 104.9 ± 23.6 | 99.6 ± 15.3 | 103.3 ± 21.8 | 107.6 ± 25.6 |
| BMI (kg/m2) | 36.0 ± 5.6 | 39.1 ± 8.2 | 35 | 35.4 | 35.3 ± 6.5 | 36.7 ± 6.8 | 37.5 ± 7.3 | 37.6 ± 4.8 | 37.7 ± 6.7 | 38.7 ± 7.7 |
| Type 2 diabetes | 10 (50) | 5 (50) | 21 (100) | 10 (100) | 95 (79) | 50 (82) | 27 (46) | 14 (67) | 62 (73) | 28 (65) |
| Liver profile | ||||||||||
| ALT (U/L) | 31.7 ± 16.8 | 32.7 ± 20.0 | 35 | 31 | 39.2 ± 21.6 | 40.3 ± 22.3 | 57.4 ± 33.3 | 50.7 ± 25.2 | 56.6 ± 30.0 | 62.2 ± 41.7 |
| AST (U/L) | 31.4 ± 13.7 | 28.9 ± 21.1 | 26 | 24 | 37.3 ± 18.7 | 35.5 ± 17.0 | 40.4 ± 21.7 | 38.6 ± 17.8 | 47.2 ± 25.3 | 57.0 ± 45.0 |
| Pro-C3 (μg/L) | 25.6 ± 27.5 | 22.6 ± 11.8 | 86 | 82 | 144.4 ± 71.8 | 131.7 ± 61.9 | 17.5 ± 7.6 | 16.1 ± 6.7 | 16.9 ± 7.0 | 16.5 ± 6.1 |
| ELF score | 10.4 ± 1.2 | 9.7 ± 0.8 | 9.2 | 9.6 | 10.5 ± 0.8 | 10.4 ± 0.8 | 9.5 ± 0.8 | 9.5 ± 1.0 | 9.75 ± 0.80 | 9.8 ± 0.7 |
| Liver stiffness (kPa) | 22.1 ± 10.8 | 25.8 ± 13.2 | 10 | 12 | 24.3 ± 12.8 | 24.7 ± 14.2 | NR | NR | 14.9 ± 6.3 | 14.5 ± 6.2 |
| Metabolic parameters | ||||||||||
| HbA1c (%) | 6.1 ± 1.0 | 6.6 ± 1.4 | 7 | 6.5 | 6.7 ± 1.1 | 6.8 ± 1.2 | 6.3 ± 1.1 | 6.49 ± 1.0 | 6.75 ± 1.10 | 6.8 ± 1.1 |
| Triglycerides (mg/dL) | 134.6 ± 62.8 | 121.7 ± 59.6 | 163 | 171 | 154.0 ± 74.3 | 143.3 ± 60.1 | 177.6 ± 85.8 | 208.3 ± 132.5 | 156.2 ± 59.6 | 169.7 ± 87.3 |
| HDL-C (mg/dL) | 50.4 ± 13.8 | 43.3 ± 12.4 | 42 | 50 | NR | NR | 41.3 ± 10.5 | 45.1 ± 9.6 | 41.1 ± 8.3 | 42.2 ± 9.7 |
| LDL-C (mg/dL) | 90.1 ± 34.4 | 89.5 ± 34.7 | 73 | 98 | NR | NR | 106.1 ± 35.6 | 116.0 ± 36.5 | 103.5 ± 33.3 | 94.2 ± 33.8 |
| Non-HDL-C (mg/dL) | 119.8 ± 12.4 | 119.8 ± 12.4 | 105 | 131 | NR | NR | 141.0 ± 38.7 | 159.3 ± 53.3 | NR | NR |
| HOMA-IR | 8.4 ± 3.7 | 12.5 ± 1.1 | 9.7 | 9.2 | NR | NR | 14.9 ± 19.3 | 10.5 ± 8.2 | NR | NR |
The risk of bias assessment categorized Harrison et al[19], Harrison et al[20], Noureddin et al[21], Harrison et al[22], and Harrison et al[23] as low risk providing the most robust evidence. Supplementary Figures 1 and 2 report the detailed bias assessment.
The certainty of evidence for comparing efruxifermin and placebo in the treatment of MASH was assessed using the GRADE framework via the GRADEpro Guideline Development Tool. Overall, the quality of evidence ranged from high to moderate. High-certainty evidence supported outcomes such as achieving > 1 stage improvement in fibrosis, MASH resolution without worsening fibrosis, and improvements in liver stiffness and fibrosis tests. In contrast, evidence for ALT and AST changes was of moderate certainty due to high heterogeneity among studies. The primary reasons for downgrading included imprecision due to small sample sizes in certain measures and variability across studies. Detailed ratings and explanations are provided in Supplementary Table 2.
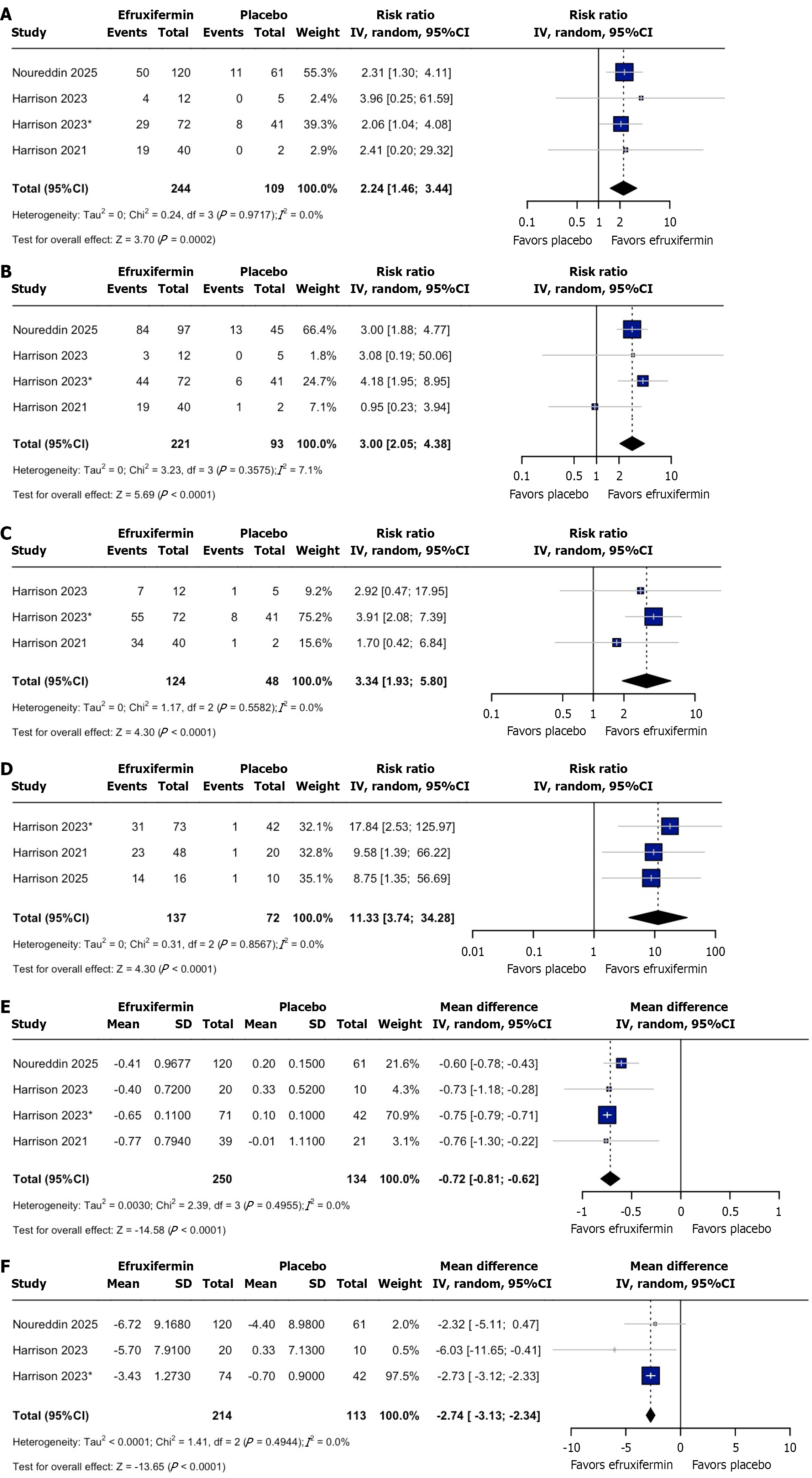
Achieving at least one stage of improvement in fibrosis without worsening of MASH: Pooled analysis of four studies concluded a significantly higher incidence of patients achieving > 1 stage improvement in fibrosis in the efruxifermin arm (RR = 2.24; 95%CI: 1.46-3.44; P = 0.0002), with homogeneity throughout (I2 = 0%; Figure 2A). Doi plot revealed a Lau-Feng-Khoo (LFK) index of 3.64, suggesting moderate publication bias (Supplementary Figure 3).
MASH resolution without worsening fibrosis: A significantly higher number of patients achieved resolution of MASH without worsening of fibrosis in the efruxifermin arm (RR = 3; 95%CI: 2.05-4.38; P < 0.0001) across four studies (Figure 2B). Pooled analysis demonstrated mild heterogeneity (I2 = 7.1%) and moderate publication bias, as suggested by an LFK index of -1.14 through the Doi plot (Supplementary Figure 4).
At least a two-point improvement in the NAS with no worsening of fibrosis: The pooled analysis of three studies demonstrated a significantly higher number of patients resulting in a 2-point improvement in NAS scores (RR = 3.34; 95%CI: 1.93-5.80), with homogenous estimates (Figure 2C). The Doi plot revealed an LFK index of -3.57, demonstrating moderate publication bias (Supplementary Figure 5).
Normalized liver fat (< 5%): The incidence of patients achieving normalization of their liver fat content (< 5%) was significantly higher in the efruxifermin arm (RR = 11.33; 95%CI: 3.74-34.28; P < 0.0001) across three studies, with homogenous estimates (Figure 2D). Doi plot relieved an LFK index of 1.26, suggesting mild publication bias (Supplementary Figure 6).
ELF test score: Pooled analysis of four studies revealed significantly lowered ELF scores in the efruxifermin arm (MD: -0.72; 95%CI: -0.81 to 0.62; P < 0.0001), with homogenous estimates (Figure 2E). Doi plot recorded an LFK index of 3.35, suggesting moderate publication bias (Supplementary Figure 7).
Liver stiffness measurement score (in kPa): Pooled analysis of three studies revealed a statistically significant lowering of liver stiffness scores in the efruxifermin arm (MD: -2.74; 95%CI: -3.13 to -2.35; P < 0.0001), with homogenous estimates (Figure 2F). Doi plot revealed an LFK index of -3.77, demonstrating moderate publication bias (Supplementary Figure 8).
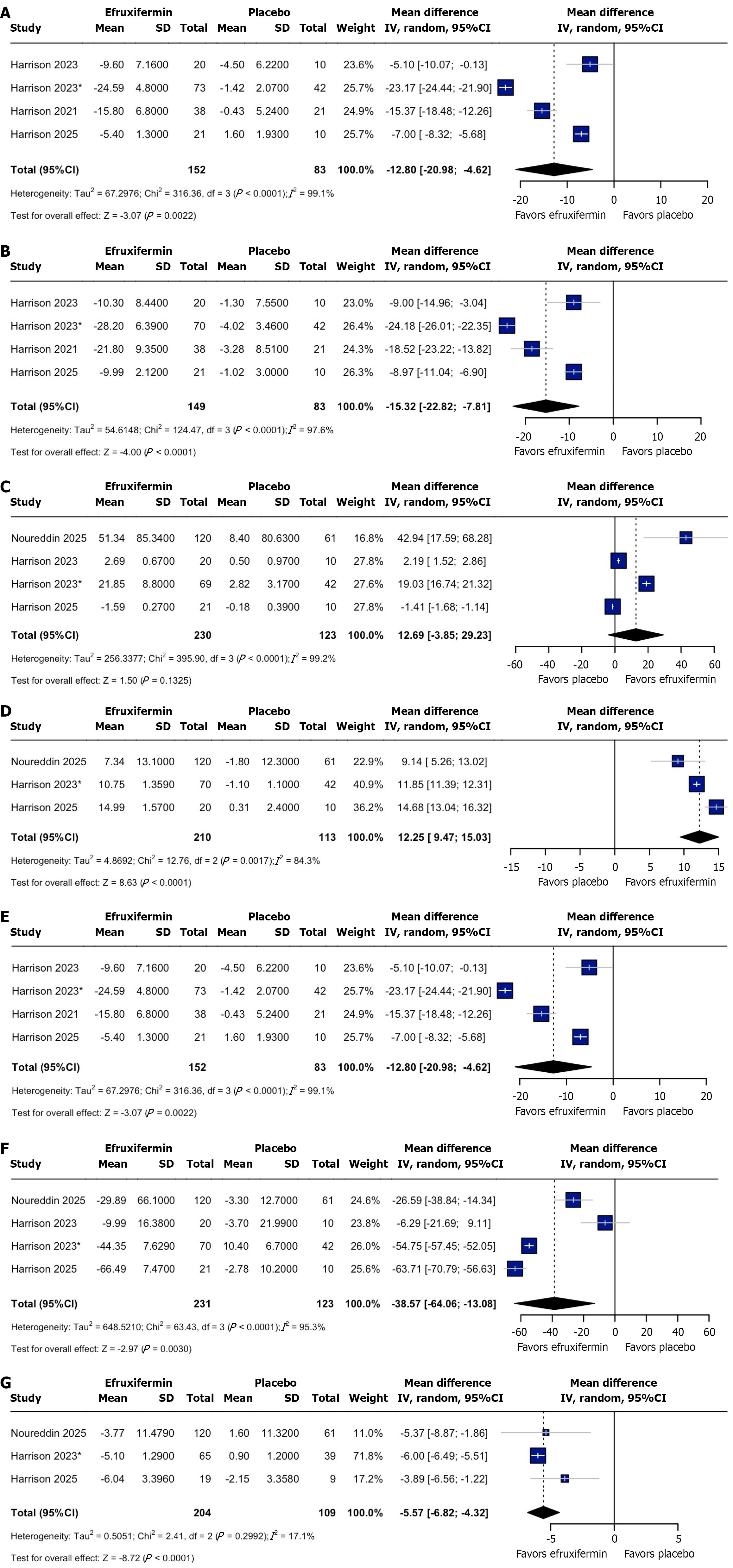
Change in AST: Pooled analysis of four studies demonstrated a significant lowering of AST in the efruxifermin arm (MD: -12.80; 95%CI: -20.98 to -4.62; P = 0.002; Figure 3A). The overall analysis demonstrated high heterogeneity (I2 = 99.1%, P < 0.0001), which remained unresolved following sensitivity analysis (Supplementary Figure 9). The Doi plot revealed an LFK index of 2.42, demonstrating moderate publication bias (Supplementary Figure 10).
Change in ALT: Pooled analysis of four studies demonstrated a significant lowering of ALT in the efruxifermin arm (MD: -15.32; 95%CI: -22.82 to -7.81; P < 0.0001; Figure 3B). The overall analysis demonstrated high heterogeneity (I2 = 97.6%, P < 0.0001), which remain unresolved following sensitivity analysis (Supplementary Figure 11). The Doi plot revealed an LFK index of 1.16, demonstrating mild publication bias (Supplementary Figure 12).
Percentage change in adiponectin: Pooled analysis of four studies demonstrated slightly higher but insignificant change in adiponectin levels in the efruxifermin arm (MD: 12.69; 95%CI: -3.85 to 29.23; P = 0.13; Figure 3C). Overall analysis demonstrated high heterogeneity (I2 = 99.2%, P < 0.0001), which remained unresolved following sensitivity analysis (Supplementary Figure 13). Doi plot revealed an LFK index of 8.71, suggesting high publication bias (Supplementary Figure 14).
HDL cholesterol: Pooled analysis of four studies indicated significantly higher HDL levels in the efruxifermin arm (MD: 12.25; 95%CI: 9.47-15.03; P < 0.0001). Pooled estimate demonstrated high heterogeneity (I2 = 84.3%), which remained unresolved on sensitivity analysis (Supplementary Figure 15). Doi plot revealed an LFK index of -0.56, indicating no publication bias (Figure 3D; Supplementary Figure 16).
LDL cholesterol: Pooled analysis of four studies revealed a significant lowering of LDL levels in the efruxifermin arm (MD: -12.80; 95%CI: -20.98 to -4.62; P = 0.002), with high heterogeneity (I2 = 99.1%, P < 0.0001; Figure 3E). Heterogeneity remained unresolved following sensitivity analysis (Supplementary Figure 17). Doi plot revealed an LFK index of 2.42, demonstrating mild to moderate publication bias (Supplementary Figure 18).
Change in triglycerides: Pooled analysis of three studies revealed significantly lowered triglycerides in the efruxifermin arm (MD: -38.57; 95%CI: -64.06 to -13.08; P = 0.003). Pooled estimate noted high heterogeneity (I2 = 95.3%), which remained unresolved following sensitivity analysis (Supplementary Figure 19). An LFK index of 4.32 was noted in the Doi plot indicating high publication bias (Figure 3F; Supplementary Figure 20).
HOMA index: Pooled analysis of three studies revealed a significantly reduced HOMA-IR levels in the efruxifermin arm (MD: -5.57; 95%CI: -6.82 to -4.32; P < 0.0001) (Figure 3G). Pooled analysis demonstrated mild heterogeneity (I2 = 17.1%), which was resolved on removal of either Harrison et al[22], and Harrison et al[23] (Supplementary Figure 21). Doi plot revealed an LFK index of 5.57 demonstrating high publication bias (Supplementary Figure 22).
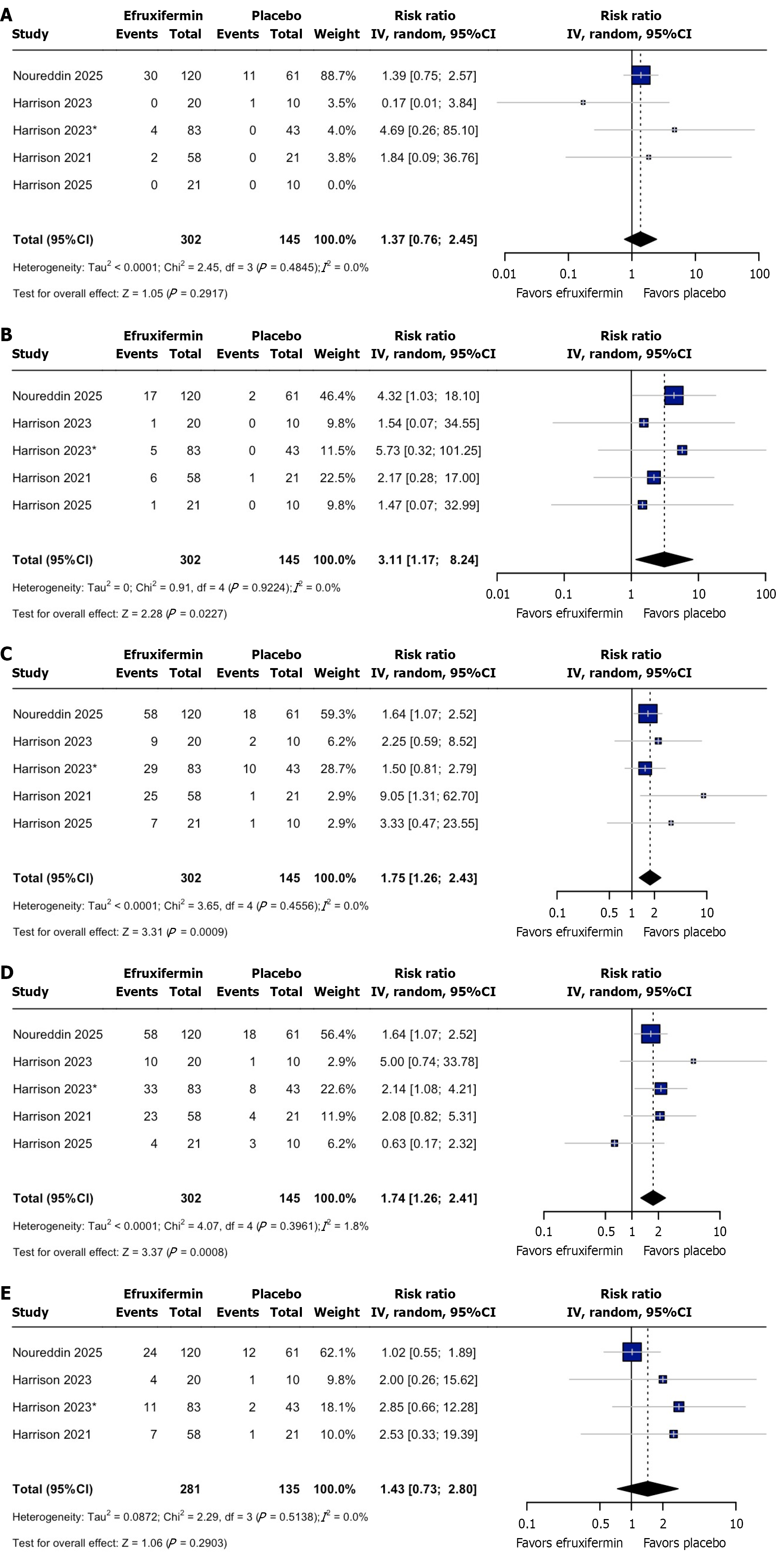
Serious adverse events: Pooled analysis of all five studies revealed similar incidences of SAEs in both arms (RR = 1.37; 95%CI: 0.76-2.45; P = 0.29), with homogenous estimate (Figure 4A). The Doi plot revealed an LFK index of 0.43, demonstrating no publication bias (Supplementary Figure 23).
Adverse event leading to discontinuation: Pooled analysis of all five studies recorded a significantly higher number of adverse events leading to discontinuation in the efruxifermin arm (RR = 3.11; 95%CI: 1.17-8.24; P = 0.023), with homogenous estimates (I2 = 0%; Figure 4B). Doi plot revealed an LFK index of -3.61, indicating moderate publication bias (Supplementary Figure 24).
Nausea: The overall analysis of all five studies revealed statistically significant risk of nausea in the efruxifermin arm (RR = 1.75; 95%CI: 1.26-2.43; P = 0.0009), with homogenous pooled estimate (Figure 4C). The Doi plot revealed an LFK index of 4.58, demonstrating high publication bias (Supplementary Figure 25).
Diarrhea: Pooled analysis of all five studies revealed statistically significant risks of diarrhea in the efruxifermin arm (RR = 1.74; 95%CI: 1.26-2.41; P = 0.0008), with a homogenous estimate (Figure 4D). Doi plot revealed an LFK index of 1.4, demonstrating mild publication bias (Supplementary Figure 26).
Injection site bruising: Pooled analysis of four studies revealed a similar incidence of injection site bruising in both arms (RR = 1.43; 95%CI: 0.73-2.80; P = 0.29), with a homogenous estimate (Figure 4E). Doi plot revealed an LFK index of 4.72, demonstrating moderate publication bias (Supplementary Figure 27).
Assessment of all outcomes using Egger’s plots revealed no significant publication bias (Supplementary Figures 28-43).
Our pooled analysis of five RCTs demonstrated that efruxifermin shows a favorable trend across key efficacy outcomes, including histological improvement in fibrosis, remission of MASH, and favorable changes in metabolic and non-invasive liver markers. While generally well-tolerated, efruxifermin was associated with a higher incidence of gastrointestinal side effects and treatment discontinuation.
Metabolic dysfunction-associated steatotic liver disease affects about 25% of the global population and is a leading cause of cirrhosis and hepatocellular carcinoma. It encompasses a spectrum from simple steatosis to MASH, characterized by necroinflammation and accelerated fibrosis progression. Metabolic dysfunction-associated steatotic liver disease is closely linked to metabolic syndrome, with type 2 diabetes increasing the risk of cirrhosis and its complications, in fact it is the most common cause of chronic liver disease and related morbidity and mortality[24].
Efruxifermin is a long-acting Fc-FGF-21 analogue that reduces cellular stress, enhances mitochondrial function, and activates antioxidant pathways[25]. With a half-life of 3-3.5 days, it outlasts most FGF-21 analogues and has demonstrated improvements in glycemic control and lipid profiles in patients with type 2 diabetes[26].
A key finding of this meta-analysis is efruxifermin’s significant histological impact on liver fibrosis. We observed a statistically significant increase in the proportion of patients achieving at least one-stage fibrosis improvement without worsening of MASH. Since MASH diagnosis relies on specific histological features from liver biopsy, the NAS was developed to quantify changes in therapeutic trials[27]. This finding is further supported by the phase 2b SYMMETRY trial by Noureddin et al[21], which, despite missing its primary endpoint at week 36, showed significant fibrosis reduction without MASH worsening at week 96 - especially with the 50 mg dose in patients with compensated MASH cirrhosis. Such improvement is particularly noteworthy given the challenges of reversing fibrosis in advanced liver disease.
However, the Doi plot revealed an LFK index of 3.64, indicating major asymmetry and suggesting moderate pub
To date, pharmacological options for MASH-related liver fibrosis remain limited. However, recent trials have evaluated several promising agents. In the MAESTRO-non-alcoholic steatohepatitis (NASH) phase 3 trial, resmetirom, a thyroid hormone receptor β agonist, achieved higher rates of NASH resolution and one-stage fibrosis improvement vs placebo, with reductions in LDL cholesterol, though gastrointestinal adverse events were more frequent[28]. The SYNERGY-NASH phase 2 trial of tirzepatide, a dual glucose-dependent insulinotropic polypeptide/GLP-1 receptor agonist, reported higher rates of MASH resolution and fibrosis improvement, predominantly with mild-to-moderate gastrointestinal events[29]. The FALCON 1 phase 2 study evaluated pegbelfermin, an FGF-21 analogue similar to efruxifermin, in patients with advanced fibrosis, it improved fibrosis, hepatic fat fraction, and noninvasive fibrosis markers and was generally well tolerated[30]. Collectively, these findings highlight both the promise and the challenges of pharmacological interventions for MASH.
Importantly, efruxifermin also significantly increased the likelihood of achieving at least a two-point improvement in the NAS without worsening fibrosis, underscoring its positive impact on liver health. This secondary endpoint captures meaningful reductions in hepatic fat, inflammation, and hepatocellular injury, reflecting efruxifermin’s influence on both metabolic and inflammatory pathways. Despite moderate publication bias, the consistency of this effect across studies - including results from Harrison et al[19] - supports its reliability. These findings strengthen the case for efruxifermin as a promising pharmacological treatment for MASH and highlight the need for further well-powered clinical trials to confirm its efficacy and safety.
Our meta also demonstrates that efruxifermin may improves multiple key outcomes in patients with MASH, including normalization of liver fat content, reduction in fibrosis as measured by the ELF test, and decreased liver stiffness scores assessed by elastography. Normalization of liver fat (< 5%) reflects partial reversal of hepatic steatosis, which is important for slowing disease progression, which is critical to halting disease progression[31]. Improvements in ELF scores and liver stiffness indicate meaningful antifibrotic effects, which are essential for reducing long-term risks such as cirrhosis and liver failure.
In evaluating the biochemical impact of efruxifermin in patients with MASH, significant reductions were observed in both AST and ALT levels, key markers of hepatocellular injury, in our metanalysis These findings suggest a notable hepatoprotective effect. Despite considerable heterogeneity, the consistent direction of effect across studies supports its role in mitigating liver inflammation and injury, central to MASH pathogenesis. Although the change in adiponectin - an anti-inflammatory adipokine associated with improved insulin sensitivity - did not reach statistical significance, a favorable trend was evident[32]. Given that adiponectin levels are influenced by other agents such as pioglitazone and GLP-1 receptor agonists, the modest rise observed with efruxifermin may still indicate partial metabolic improvement[33]. Collectively, these biochemical outcomes reinforce the therapeutic potential of efruxifermin in improving not only histological parameters but also systemic metabolic health in patients with MASH.
In our meta-analysis, efruxifermin was associated with improvements in key metabolic parameters, reinforcing its promise as a therapeutic agent for MASH. HDL levels were notably elevated, a clinically relevant finding given the high cardiovascular risk associated with MASH. This improvement in dyslipidemia suggests that long-term use of efruxifermin could help mitigate cardiovascular complications common in this population[34]. Additionally, a marked reduction in HOMA-IR reflects enhanced insulin sensitivity, addressing a central mechanism in MASH pathogenesis. The HOMA-IR index, calculated from fasting glucose and insulin, is a well-established surrogate for insulin resistance[35], which contributes to hepatic steatosis, inflammation, and fibrosis[36]. These effects parallel those of agents like GLP-1 receptor agonists and pioglitazone, highlighting efruxifermin’s potential as a comprehensive metabolic therapy in MASH.
Despite the consistency in histological endpoints, substantial heterogeneity was observed in serum biomarkers such as AST, ALT, LDL, HDL, and triglycerides (I2 > 95% in most analyses). This variability can be attributed to several study-level factors. First, the included trials enrolled distinct patient populations, ranging from F1-F3 NASH (Harrison et al[19]) to compensated F4 cirrhosis (Harrison et al[20]; Noureddin et al[21]), leading to differences in baseline enzyme levels and hepatic synthetic capacity. Second, trial designs varied significantly in treatment duration (12-96 weeks), dosing (28-70 mg), and combination regimens - such as the use of GLP-1RA in Harrison et al[22], which independently impacts metabolic markers. Third, methodological differences in laboratory measurements, endpoints (absolute vs percent change), and statistical approaches likely amplified inter-study variability.
Efruxifermin demonstrated an overall acceptable safety profile in patients with MASH. The incidence of serious adverse events was comparable between the efruxifermin and placebo groups, indicating no increased risk of major complications. However, gastrointestinal adverse events - particularly nausea and diarrhea - and treatment discontinuation due to side effects were more frequent in the efruxifermin arm. Despite this, injection site bruising occurred at similar rates across groups, reflecting good tolerability of its subcutaneous administration.
Unlike obeticholic acid, which is commonly associated with pruritus requiring active management[37], the adverse events linked to efruxifermin were typically mild to moderate and transient, closely resembling those seen with GLP-1 receptor agonists[38]. Importantly, the co-administration of efruxifermin with GLP-1 receptor agonists did not exacerbate gastrointestinal side effects, supporting its feasibility for combination regimens[20].
Key strengths of this meta-analysis include a rigorous search strategy, low risk of bias in included trials, and robust synthesis of histological, biochemical, and safety outcomes. However, limitations include the small number of studies, short follow-up durations, and significant heterogeneity in metabolic markers. Publication bias was noted in several outcomes, suggesting possible underreporting of negative findings. In addition, the pooled data may not fully capture long-term outcomes or differences in response based on baseline FGF-21 sensitivity, a factor increasingly recognized in advanced liver disease. Therefore, while the results suggest promising efficacy, these findings should be interpreted with caution given the preliminary nature of the available evidence and the limited duration of follow-up in most studies. Larger, long-term trials are warranted to confirm these effects and establish clinical durability.
Efruxifermin shows significant promise as a treatment for compensated MASH cirrhosis. Its ability to improve fibrosis, resolve MASH, reduce hepatic fat, and enhance metabolic health addresses multiple pathogenic pathways. While gastrointestinal side effects are common, they are generally mild and manageable. These findings support further investigation in larger, long-term trials. Future studies should address remaining gaps in the pharmacological management of MASH. This includes exploring combination strategies, personalized dosing, and mechanisms of FGF-21 resistance. Head-to-head trials comparing efruxifermin with other FGF-21 analogues, such as pegbelfermin, or GLP-1 receptor agonists like tirzepatide or semaglutide, would clarify comparative efficacy and safety. Investigations into predictive biomarkers, such as baseline FGF-21, circulating adiponectin, hepatic FGF-21 receptor expression, or HOMA-IR, could enable more individualized therapy. Trials with long-term, clinically meaningful endpoints, including decompensation-free survival, progression to hepatocellular carcinoma, or need for liver transplantation, are warranted to establish durability and real-world impact.
| 1. | Amini-Salehi E, Letafatkar N, Norouzi N, Joukar F, Habibi A, Javid M, Sattari N, Khorasani M, Farahmand A, Tavakoli S, Masoumzadeh B, Abbaspour E, Karimzad S, Ghadiri A, Maddineni G, Khosousi MJ, Faraji N, Keivanlou MH, Mahapatro A, Gaskarei MAK, Okhovat P, Bahrampourian A, Aleali MS, Mirdamadi A, Eslami N, Javid M, Javaheri N, Pra SV, Bakhsi A, Shafipour M, Vakilpour A, Ansar MM, Kanagala SG, Hashemi M, Ghazalgoo A, Kheirandish M, Porteghali P, Heidarzad F, Zeinali T, Ghanaei FM, Hassanipour S, Ulrich MT, Melson JE, Patel D, Nayak SS. Global Prevalence of Nonalcoholic Fatty Liver Disease: An Updated Review Meta-Analysis comprising a Population of 78 million from 38 Countries. Arch Med Res. 2024;55:103043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 94] [Article Influence: 47.0] [Reference Citation Analysis (1)] |
| 2. | Zhai M, Liu Z, Long J, Zhou Q, Yang L, Zhou Q, Liu S, Dai Y. The incidence trends of liver cirrhosis caused by nonalcoholic steatohepatitis via the GBD study 2017. Sci Rep. 2021;11:5195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 3. | Feng G, Valenti L, Wong VW, Fouad YM, Yilmaz Y, Kim W, Sebastiani G, Younossi ZM, Hernandez-Gea V, Zheng MH. Recompensation in cirrhosis: unravelling the evolving natural history of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2024;21:46-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 144] [Article Influence: 72.0] [Reference Citation Analysis (1)] |
| 4. | Zhou C, Pan X, Huang L, Wu T, Zhao T, Qi J, Wu J, Mukondiwa AV, Tang Y, Luo Y, Tu Q, Huang Z, Niu J. Fibroblast growth factor 21 ameliorates cholestatic liver injury via a hepatic FGFR4-JNK pathway. Biochim Biophys Acta Mol Basis Dis. 2024;1870:166870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 5. | Puengel T, Tacke F. Efruxifermin, an investigational treatment for fibrotic or cirrhotic nonalcoholic steatohepatitis (NASH). Expert Opin Investig Drugs. 2023;32:451-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 6. | Yadav P, Khurana A, Bhatti JS, Weiskirchen R, Navik U. Glucagon-like peptide 1 and fibroblast growth factor-21 in non-alcoholic steatohepatitis: An experimental to clinical perspective. Pharmacol Res. 2022;184:106426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 7. | Kovalic AJ. Pharmacotherapeutic Impact on Nonalcoholic Steatohepatitis Histology: A Systematic Review and Network Meta-analysis. J Clin Exp Hepatol. 2022;12:1057-1068. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 8. | Jeong C, Han N, Jeon N, Rhee SJ, Staatz CE, Kim MS, Baek IH. Efficacy and Safety of Fibroblast Growth Factor-21 Analogs for the Treatment of Metabolic Dysfunction-Associated Steatohepatitis: A Systematic Review and Meta-Analysis. Clin Pharmacol Ther. 2024;116:72-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 9. | Chang Y, Jeong SW, Jang JY. Recent updates on pharmacologic therapy in non-alcoholic fatty liver disease. Clin Mol Hepatol. 2024;30:129-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 10. | Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, Thomas J. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10:ED000142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2342] [Cited by in RCA: 3589] [Article Influence: 512.7] [Reference Citation Analysis (2)] |
| 11. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 12. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20150] [Article Influence: 2878.6] [Reference Citation Analysis (5)] |
| 13. | Rath S, Girgis M, Gupta I, Chauhan A, Ud Din Z, Hotchandani H, Hasan A, Ahmed R, Qureshi R. Assessing Efficacy of Interleukin-1 Blockade in Patients with Alcoholic Hepatitis: A Comprehensive Systematic Review of Emerging Evidence. Life (Basel). 2025;15:1106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 14. | Santesso N, Carrasco-Labra A, Langendam M, Brignardello-Petersen R, Mustafa RA, Heus P, Lasserson T, Opiyo N, Kunnamo I, Sinclair D, Garner P, Treweek S, Tovey D, Akl EA, Tugwell P, Brozek JL, Guyatt G, Schünemann HJ. Improving GRADE evidence tables part 3: detailed guidance for explanatory footnotes supports creating and understanding GRADE certainty in the evidence judgments. J Clin Epidemiol. 2016;74:28-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 106] [Article Influence: 10.6] [Reference Citation Analysis (2)] |
| 15. | Brignardello-Petersen R, Guyatt GH. Assessing the certainty of the evidence in systematic reviews: importance, process, and use. Am J Epidemiol. 2025;194:1681-1686. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 16. | Rath S, Alam U, Kashif A, Ansab M, Siddiqui HT, Burhan M, Afridi ZAK, Khan A, Gulalay A, Aslam B, Ahmed R. Intravenous thrombolysis as an adjunct to endovascular thrombectomy in acute basilar artery occlusion: a GRADE-assessed systematic review and meta-analysis. Naunyn Schmiedebergs Arch Pharmacol. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 17. | DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32714] [Cited by in RCA: 30771] [Article Influence: 769.3] [Reference Citation Analysis (4)] |
| 18. | Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557-560. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Harrison SA, Ruane PJ, Freilich BL, Neff G, Patil R, Behling CA, Hu C, Fong E, de Temple B, Tillman EJ, Rolph TP, Cheng A, Yale K. Efruxifermin in non-alcoholic steatohepatitis: a randomized, double-blind, placebo-controlled, phase 2a trial. Nat Med. 2021;27:1262-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 278] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 20. | Harrison SA, Frias JP, Neff G, Abrams GA, Lucas KJ, Sanchez W, Gogia S, Sheikh MY, Behling C, Bedossa P, Shao L, Chan D, Fong E, de Temple B, Shringarpure R, Tillman EJ, Rolph T, Cheng A, Yale K; HARMONY Study Group. Safety and efficacy of once-weekly efruxifermin versus placebo in non-alcoholic steatohepatitis (HARMONY): a multicentre, randomised, double-blind, placebo-controlled, phase 2b trial. Lancet Gastroenterol Hepatol. 2023;8:1080-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 183] [Article Influence: 61.0] [Reference Citation Analysis (0)] |
| 21. | Noureddin M, Rinella ME, Chalasani NP, Neff GW, Lucas KJ, Rodriguez ME, Rudraraju M, Patil R, Behling C, Burch M, Chan DC, Tillman EJ, Zari A, de Temple B, Shringarpure R, Jain M, Rolph T, Cheng A, Yale K. Efruxifermin in Compensated Liver Cirrhosis Caused by MASH. N Engl J Med. 2025;392:2413-2424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 58] [Article Influence: 58.0] [Reference Citation Analysis (1)] |
| 22. | Harrison SA, Frias JP, Lucas KJ, Reiss G, Neff G, Bollepalli S, Su Y, Chan D, Tillman EJ, Moulton A, de Temple B, Zari A, Shringarpure R, Rolph T, Cheng A, Yale K. Safety and Efficacy of Efruxifermin in Combination With a GLP-1 Receptor Agonist in Patients With NASH/MASH and Type 2 Diabetes in a Randomized Phase 2 Study. Clin Gastroenterol Hepatol. 2025;23:103-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 61] [Article Influence: 61.0] [Reference Citation Analysis (1)] |
| 23. | Harrison SA, Ruane PJ, Freilich B, Neff G, Patil R, Behling C, Hu C, Shringarpure R, de Temple B, Fong E, Tillman EJ, Rolph T, Cheng A, Yale K. A randomized, double-blind, placebo-controlled phase IIa trial of efruxifermin for patients with compensated NASH cirrhosis. JHEP Rep. 2023;5:100563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 69] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 24. | Chan WK, Chuah KH, Rajaram RB, Lim LL, Ratnasingam J, Vethakkan SR. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): A State-of-the-Art Review. J Obes Metab Syndr. 2023;32:197-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 617] [Cited by in RCA: 515] [Article Influence: 171.7] [Reference Citation Analysis (10)] |
| 25. | Tillman EJ, Brock WJ, Rolph T. Efruxifermin, a long-acting Fc-fusion FGF21 analogue, reduces body weight gain but does not increase sympathetic tone or urine volume in Sprague Dawley rats. Br J Pharmacol. 2022;179:1384-1394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 26. | Kaufman A, Abuqayyas L, Denney WS, Tillman EJ, Rolph T. AKR-001, an Fc-FGF21 Analog, Showed Sustained Pharmacodynamic Effects on Insulin Sensitivity and Lipid Metabolism in Type 2 Diabetes Patients. Cell Rep Med. 2020;1:100057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 27. | Brunt EM, Kleiner DE, Wilson LA, Belt P, Neuschwander-Tetri BA; NASH Clinical Research Network (CRN). Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology. 2011;53:810-820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1072] [Cited by in RCA: 998] [Article Influence: 66.5] [Reference Citation Analysis (4)] |
| 28. | Harrison SA, Bedossa P, Guy CD, Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW, Rinella ME, Anstee QM, Abdelmalek MF, Younossi Z, Baum SJ, Francque S, Charlton MR, Newsome PN, Lanthier N, Schiefke I, Mangia A, Pericàs JM, Patil R, Sanyal AJ, Noureddin M, Bansal MB, Alkhouri N, Castera L, Rudraraju M, Ratziu V; MAESTRO-NASH Investigators. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med. 2024;390:497-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1457] [Cited by in RCA: 1304] [Article Influence: 652.0] [Reference Citation Analysis (2)] |
| 29. | Loomba R, Hartman ML, Lawitz EJ, Vuppalanchi R, Boursier J, Bugianesi E, Yoneda M, Behling C, Cummings OW, Tang Y, Brouwers B, Robins DA, Nikooie A, Bunck MC, Haupt A, Sanyal AJ; SYNERGY-NASH Investigators. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis. N Engl J Med. 2024;391:299-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 671] [Cited by in RCA: 567] [Article Influence: 283.5] [Reference Citation Analysis (1)] |
| 30. | Loomba R, Sanyal AJ, Nakajima A, Neuschwander-Tetri BA, Goodman ZD, Harrison SA, Lawitz EJ, Gunn N, Imajo K, Ravendhran N, Akahane T, Boone B, Yamaguchi M, Chatterjee A, Tirucherai GS, Shevell DE, Du S, Charles ED, Abdelmalek MF. Pegbelfermin in Patients With Nonalcoholic Steatohepatitis and Stage 3 Fibrosis (FALCON 1): A Randomized Phase 2b Study. Clin Gastroenterol Hepatol. 2024;22:102-112.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 72] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 31. | Day JW, Rosenberg WM. The enhanced liver fibrosis (ELF) test in diagnosis and management of liver fibrosis. Br J Hosp Med (Lond). 2018;79:694-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Lihn AS, Pedersen SB, Richelsen B. Adiponectin: action, regulation and association to insulin sensitivity. Obes Rev. 2005;6:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 481] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 33. | Simental-Mendía LE, Sánchez-García A, Linden-Torres E, Simental-Mendía M. Impact of glucagon-like peptide-1 receptor agonists on adiponectin concentrations: A meta-analysis of randomized controlled trials. Br J Clin Pharmacol. 2021;87:4140-4149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 34. | Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, Harrison SA, Brunt EM, Sanyal AJ. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5962] [Cited by in RCA: 5317] [Article Influence: 664.6] [Reference Citation Analysis (2)] |
| 35. | Salgado AL, Carvalho Ld, Oliveira AC, Santos VN, Vieira JG, Parise ER. Insulin resistance index (HOMA-IR) in the differentiation of patients with non-alcoholic fatty liver disease and healthy individuals. Arq Gastroenterol. 2010;47:165-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 208] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 36. | Zhao X, An X, Yang C, Sun W, Ji H, Lian F. The crucial role and mechanism of insulin resistance in metabolic disease. Front Endocrinol (Lausanne). 2023;14:1149239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 389] [Cited by in RCA: 289] [Article Influence: 96.3] [Reference Citation Analysis (1)] |
| 37. | Jang H, Han N, Staatz CE, Kwak JH, Baek IH. Effect on lipid profile and clinical outcomes of obeticholic acid for the treatment of primary biliary cholangitis and metabolic dysfunction-associated steatohepatitis: A systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2023;47:102227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 38. | Wharton S, Davies M, Dicker D, Lingvay I, Mosenzon O, Rubino DM, Pedersen SD. Managing the gastrointestinal side effects of GLP-1 receptor agonists in obesity: recommendations for clinical practice. Postgrad Med. 2022;134:14-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 152] [Article Influence: 38.0] [Reference Citation Analysis (3)] |