Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115221
Revised: November 7, 2025
Accepted: January 5, 2026
Published online: March 27, 2026
Processing time: 166 Days and 13.8 Hours
Liver transplantation (LT) in hyperimmunized recipients with pre-formed donor-specific antibodies (pDSA) or a positive crossmatch (CM) presents a significant immunological graft challenge.
To assess whether induction therapy with rabbit anti-T lymphocyte globulin (rATLG) and high-dose intravenous immunoglobulin (IVIG) reduces acute T-cell mediated rejection (TCMR), acute antibody-mediated rejection (aAMR), and graft loss in these patients.
This retrospective case-control study, conducted between 2016 and 2022, compared the outcomes of two groups of LT recipients: Forty-six hyperimmunized patients (high-risk, pDSA and/or CM positive at time of LT) were matched with 46 non-immunized (low-risk) recipients. High-risk patients received anti-T lymphocyte glo
The incidence of biopsy-proven TCMR was numerically lower in the high-risk group (19.6%) compared to the low-risk group (26.1%), with all cases classified as Banff mild or moderate. Of these, clinically significant rejections requiring treatment occurred in 10.9% of high-risk and 8.7% of low-risk recipients. A subset of the high-risk group (n = 4, 8.7%) developed aAMR vs none in the low-risk group. The one-year (high-risk: 83.6%, low-risk: 95.6%) and three-year (high-risk: 78.0%, low-risk: 91.2%) survival rates were comparable between the two groups (log-rank P = 0.051). Notably, no grafts were lost due to rejection in either group, and no adverse events were linked to the in
Short course of rATLG and IVIG induction therapy can be a valuable strategy for mitigating early immunological risks in hyperimmunized recipients, leading to comparable outcomes to non-immunized patients.
Core Tip: For hyperimmunized liver transplant recipients with high mean fluorescence intensity titer of pre-formed donor-specific antibodies and/or donor/recipient positive crossmatch, early induction with a short course of rabbit anti-T-lymphocyte globulins combined with high-dose intravenous immune-globulins can be a valuable strategy for mitigating early immunological risks (T-cell mediated rejection, acute antibody-mediated rejection severity and graft loss). If allograft dysfunction is suspected, closer serial donor-specific antibodies monitoring and early liver biopsy should be considered for timely diagnosis and management of antibody-mediated injury.
- Citation: EL-Domiaty N, Coilly A, Sebagh M, Taupin JL, Ibrahim W, Sacleux SC, Ichai P, Duhaut L, Pittau G, Ciacio O, Salloum C, Sa-Cunha A, Azoulay D, Feray C, Cherqui D, Shiha G, Samuel D, Saliba F. Induction with anti-T-lymphocyte globulins and human immunoglobulins: A strategy for hyperimmunized liver transplant patients. World J Hepatol 2026; 18(3): 115221
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115221.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115221
Liver transplantation (LT) is the established treatment for end-stage liver disease and liver cancer[1]. However, liver transplant recipients with pre-existing donor-specific antibodies (pDSA) or a positive donor-recipient crossmatch (D/R CM) defining a hyperimmunised state, have been reported to be associated with high risk of acute T-cell mediated rejection (TCMR), antibody-mediated rejection (aAMR) and subsequent graft failure in various studies[2-8]. This immunological challenge, affecting an estimated 7%-15% of LT candidates[3,4] often resulting from prior sensitization, arises from prior blood transfusions, pregnancies, or previous transplants, complicating donor matching and post-transplant immunosuppression[5]. Multiple studies have established the correlation between pDSA/positive CM and an increased risk of rejection and graft loss[6-8].
Induction therapy uses potent immunosuppressants during LT to modulate immune responses and prevent early rejection. In hyperimmunized patients, induction regimens often include polyclonal antibodies [e.g., antithymocyte globulins (ATG)], monoclonal antibodies (e.g., basiliximab, alemtuzumab), or targeted agents[7]. These aim to reduce TCMR and aAMR by depleting or inhibiting immune components[5]. In kidney transplantation, T-cell suppression via depleting or non-depleting agents is standard. ATGs, used since the 1990s, remain central in preventing acute rejection[9,10].
The two most used preparations are rabbit ATG (thymoglobulin; also known as ATG-Genzyme, Sanofi Genzyme, Cambridge, MA, United States) and rabbit anti-T-lymphocyte globulins (formally ATG-Fresenius Biotech GmbH, Munich, Germany, now ATLG, Neovii, Pharmaceuticals AG, Jona, Switzerland). Thymoglobulin is derived from immunized rabbits with fresh human thymocytes, and anti-T lymphocyte globulin (ATLG) is produced from the Jurkat T lymphoblastic cell line[10,11]. Despite improvements in immunosuppressive protocols, managing hyperimmunized liver transplant recipients continues to be challenging and associated with significant complications. The ideal induction regimen in LT remains a subject of ongoing research, with various combinations of agents, dosing strategies, and long-term outcomes under investigation. Moreover, balancing the risks of over-immunosuppression (including infection and malignancy) against the benefits of preventing rejection requires meticulous consideration and a personalized approach to patient care.
The optimal management of hyperimmunized patients remains a challenge. To address this challenge, this study investigates the efficacy of induction therapy with rabbit ATLG (rATLG) and high-dose intravenous immunoglobulin (IVIG) in high-risk hyperimmunized patients [defined by high mean fluorescence intensity (MFI) titers of pDSA and/or a positive D/R CM at the time of LT]. Our primary aim is to determine if this regimen can achieve comparable outcomes to non-immunized recipients and mitigate the inherent risks of hyperimmunization.
This is a case-control study from the Hepato-Biliary Centre of the Paul Brousse Hospital, France. From a database of over 910 patients who underwent LT between January 2016 and December 2022, 46 consecutive hyperimmunized recipients were retrospectively recruited for this study. These patients were specifically selected based on the presence of high pDSA titers and/or a positive complement-dependent cytotoxicity (CDC) CM at the time of LT, having undergone a first or second liver transplant, and having received induction therapy with short-term rATLG. Patients undergoing combined organ transplantation were excluded. A matched control group of 46 non-immunized LT recipients was recruited retrospectively. These controls were defined by the absence of pDSA at the time of LT and a negative CDC CM. To minimize confounding factors, the study employed a 1:1 matched case-control design where each hyperimmunized recipient was matched with a non-immunized recipient based on the following criteria: Age (matched within ± 3 years), sex (exact matching), primary indication for LT (exact matching) and calculated model for end-stage liver disease (MELD) score (matched within ± 3 points). Patients were divided into two groups according to their immunological status at time of LT: High-risk group (hyperimmunized group) and low-risk group (control non-immunized group).
The hyperimmunized status was defined by the presence of high pDSA titers (MFI ≥ 5000) and/or a positive CDC CM at the time of LT. Non-immunized status was defined by the absence or low pDSA (MFI < 5000) at the time of LT and negative CDC CM.
To minimize selection bias, we included: All eligible hyperimmunized recipients through consecutive case selection, applied rigorous matching, used blinded outcome assessment so pathologists had no access to biomarker data and ensured standardized post-transplant follow-up for all patients.
Data collected included recipient and donor’s demographics including age, sex, body mass index (BMI), history of hypertension, diabetes mellitus, etiology of underlying liver disease in addition to time on waiting list, number of previous liver transplants, MELD score, Child-Pugh score and cytomegalovirus (CMV) D/R mismatch. We collected data on the LT procedure: Duration, cold ischemia and type of transplant. Recipient and donor immunological status at time of the transplantation [human leukocyte antigen (HLA) class I and II antibodies, donor specific HLA antibodies], post-transplant immunosuppression protocol and induction therapy were also considered. Clinical outcomes [intensive care unit (ICU) stay, mortality, causes of mortality and complications] were recorded. The patients were followed up until December 2023.
Data were collected from charts and electronic databases after approval by the local institutional review board, in accordance with the International Guidelines for Ethical Review of Epidemiological Studies and principles of the Declaration of Helsinki.
The high-risk group received induction therapy consisting of a short course of rATLG (3 mg/kg/day on days 0, 1, 2) combined with high-dose human polyclonal IVIG, 2 g/kg over three days (administered as 1 g/kg on day 0 post-rATLG and 0.5 g/kg on days 1 and 2). This induction protocol with short-course ATLG was initially started in our centre in 2016. Maintenance immunosuppression was based on tacrolimus and started at a dose of 0.1 mg/kg per day between day 0 and day 3 and aiming for a trough level between 6 ng/mL and 10 ng/mL during the first post-transplant year, mycophenolate mofetil (1 g twice daily for 1 month and then dose adjusted according to clinical status) and steroids (500 mg pretransplant and then tapered progressively to 20 mg/day on day 7, and gradually withdrawn at month 6). Patients at risk of CMV infection, that is, seronegative recipients receiving a liver from a seropositive donor and seropositive recipients, received valganciclovir (900 mg/day) for 6 months and 3 months respectively.
Recipient HLA typing was conducted using high-resolution DNA typing (high resolution RSSO, One Lambda, West Hills, CA until end of 2017) or next-generation sequencing (NGS-go; GenDx, Utrecht, The Netherlands, from January 2018). Donor HLA typing was performed using high-resolution SSP (Linkage Biosciences, One Lambda) for local donors and about 2/3 of donors originating from areas outside metropolitan Paris, or other medium to high resolution SSP method.
Recipients were tested for the presence of circulating donor-specific anti-HLA class I A/B/Cw, and class II DR/DQ/DP IgG antibodies at baseline using Luminex single antigen (LSA) flow-bead assays (One Lambda). The pre-transplant assessment was performed within 48 hours prior to LT with a median time from most recent pre-transplant donor-specific antibodies (DSA) testing to LT was 24 hours [interquartile range (IQR) 12-36 hours]. DSA testing was not routinely performed post-transplant. It was performed for patients with known high titers (MFI > 10000) DSA and in case of biopsy findings suggestive of aAMR or severe graft injury of unexplained cause. Beads with a normalized mean MFI of greater than 1000 units according to the baseline formula (Fusion software, One Lambda) were considered positive according to the cut-off value in LT. Virtual CM relying on recipient’s pre-transplant HLA antibody levels (LSA results) and donor’s HLA typing, was almost systematically performed pre transplant as an initial tool to assess compatibility.
“For cause” based liver biopsies were performed when clinically indicated in relation to a progressive and steady increase in liver enzymes despite adjusting tacrolimus levels and after excluding a vascular or a biliary complication by Doppler ultrasound or magnetic resonance imaging. All liver biopsies were read locally by pathologists blinded to biomarker data and classified according to the type of allograft rejection using the international and standardized Banff classification[12]. Complement component 4d staining was performed by immunohistochemistry on paraffin-embedded tissue according to local practice. Biopsies were reviewed by one expert pathologist.
The primary outcome was the prevalence of TCMR and aAMR. Secondary outcomes included patient and graft survival, time to rejection, ICU stay, re-transplantation, major post-transplant complications and causes of mortality and graft failure. All patients had the same schedule of post-transplant follow-up. After discharge from the hospital, they were seen at our clinic at least monthly until 6 months after LT. After that, they were seen three, then two times a year.
All statistical analyses were performed using the statistical Package for the Social Sciences version 23 (SPSS, Inc., Chicago, IL, United States) and R software version 3.4.3 (R Foundation for Statistical Computing, Vienna, Austria). Categorical variables were expressed as n (%) and compared using the χ2 test with Fisher’s correction. Continuous variables were expressed as mean ± SD and compared using the t-test. Survival probabilities were calculated using the Kaplan-Meier method and compared using the log-rank test. Statistical significance was accepted with a P value ≤ 0.05 level and 95% confidence.
An a priori power calculation was not performed because this retrospective cohort included all consecutively available hyperimmunized recipients transplanted at our center during the study period. As a post-hoc estimate, based on the observed 3-year survival (78.0% vs 91.2%) and sample sizes of 46 patients per group, the study’s power to detect this difference (two-sided α = 0.05) is approximately 43%. This indicates limited power and a considerable risk of type II error for survival comparisons.
Ninety-two LT recipients (mean age 48.6 ± 15.2 years) were included, with a mean follow-up of 42.8 ± 25.5 months. Forty-six hyperimmunized recipients (with high pre-existing pDSA MFI and/or positive CDC crossmatch) were 1:1 matched with non-hyperimmunized controls. No significant differences were found in recipient age (47.6 years vs 49.6 years; P = 0.52), sex (female: 67.4% vs 63.0%; P = 0.66), BMI (25.8 vs 25.8; P = 0.96), MELD score (21.6 vs 19.9; P = 0.45), or indications for transplant (P = 0.94). A trend in CMV mismatching was observed (P = 0.06). Hyperimmunized patients had higher rates of prior LT (17.4% vs 4.3%; P = 0.04) and living donor grafts (8.7% vs 0%; P = 0.04). Donor age was similar (54.3 years vs 52.5 years; P = 0.70), and procedural factors (transplant duration, cold ischemia time) did not differ. Hyperimmunized recipients more often received female donor livers (60.9% vs 45.7%; P = 0.14), though not significantly. All data are summarized in Table 1.
| High-risk group (n = 46) | Low-risk group (n = 46) | P value | |
| Recipient characteristics | |||
| Gender | |||
| Female | 31 (67.4) | 29 (63.0) | 0.66 |
| Male | 15 (32.6) | 17 (37.0) | |
| Age (years) | 47.6 ± 14.5 | 49.6 ± 15.9 | 0.52 |
| BMI (kg/m2) | 25.8 ± 4.9 | 25.8 ± 6.8 | 0.96 |
| Pre-transplant cardiovascular disease | |||
| Hypertension | 13 (28.3) | 14 (30.4) | 0.82 |
| Diabetes mellitus | 9 (19.6) | 13 (28.3) | 0.33 |
| History for smoking | 12 (26.1) | 15 (32.6) | 0.53 |
| Brinkman index for smoking | |||
| 0 | 34 (73.9) | 31 (67.4) | |
| 201-400 | 8 (17.4) | 9 (19.6) | |
| > 400 | 4 (8.7) | 6 (13.0) | |
| Chronic kidney disease1 | 6 (13.0) | 2 (0.04) | 0.12 |
| Waiting time on waiting list (months) | 5.2 ± 7.5 | 6.9 ± 7.34 | 0.26 |
| MELD score | 21.6 ± 9.4 | 19.9 ± 11.0 | 0.45 |
| Child-Pugh score | 0.26 | ||
| A | 2 (4.3) | 6 (13.0) | |
| B | 23 (50.0) | 24 (52.2) | |
| C | 21 (45.7) | 16 (34.8) | |
| History of previous liver transplant | 8 (17.4) | 2 (4.3) | 0.04 |
| Indication of transplant | 0.94 | ||
| HCC | 11 (23.9) | 13 (28.3) | |
| Biliary cirrhosis | 8 (17.4) | 11 (23.9) | |
| Alcohol associated cirrhosis | 5 (10.9) | 5 (10.9) | |
| Viral hepatitis cirrhosis | 4 (8.7) | 3 (6.5) | |
| Vascular cirrhosis2 | 5 (10.9) | 1 (2.2) | |
| Acute fulminant hepatitis | 3 (6.5) | 3 (6.5) | |
| MASH cirrhosis | 3 (6.5) | 3 (6.5) | |
| Chronic rejection | 2 (4.3) | 2 (4.3) | |
| Autoimmune cirrhosis | 2 (4.3) | 2 (4.3) | |
| Others | 3 (6.5) | 3 (6.5) | |
| Transplant type | 0.04 | ||
| Deceased donor | 42 (91.3) | 46 (100.0) | |
| Living donor | 4 (8.7) | 0 (0.0) | |
| Transplant duration (minutes) | 455.2 ± 126.5 | 441.5 ± 122.2 | 0.60 |
| Cold ischemia time (minutes) | 408.5 ± 151.9 | 454.1 ± 150.5 | 0.15 |
| Donor characteristics | |||
| Gender | 0.14 | ||
| Female | 28 (60.9) | 21 (45.7) | |
| Male | 18 (39.1) | 25 (54.3) | |
| Age (years) | 54.3 ± 21.6 | 52.5 ± 23.3 | 0.70 |
| BMI (kg/m2) | 25.7 ± 4.4 | 24.9 ± 4.9 | 0.38 |
| CMV mismatch | 0.06 | ||
| D-/R- | 2 (4.3) | 11 (23.9) | |
| D-/R+ | 14 (30.4) | 11 (23.9) | |
| D+/R- | 14 (30.4) | 11 (23.9) | |
| D+/R+ | 16 (34.8) | 13 (28.3) | |
As expected, immunological profiles at transplant differed significantly between hyperimmunized (high-risk) and non-hyperimmunized (low-risk) groups. All high-risk patients had high MFI titers of class I or II pDSA; 80.4% (37/46) had a positive T-cell D/R CM, while the remaining 19.6% (9/46) had negative CM but high total pDSA MFI (median: 20834; IQR: 3835-49939). In contrast, all low-risk patients had negative T-cell D/R CM, with no or low-level pDSA (< 5000 MFI), below clinical significance (Table 2).
| High-risk group (n = 46) | Low-risk group (n = 46) | P value | |
| T-lymphocytes D/R CM | < 0.01a | ||
| Negative | 9 (19.6) | 46 (100.0) | |
| Positive | 37 (80.4) | 0 (0.0) | |
| Recipient HLA antibodies class I | < 0.01a | ||
| Negative | 7 (15.2) | 41 (89.1) | |
| Positive | 37 (80.4) | 5 (10.9) | |
| Missing data | 2 (4.3) | 0 (0) | |
| Recipient HLA antibodies class II | < 0.01a | ||
| Negative | 6 (13.0) | 42 (91.3) | |
| Positive | 38 (82.6) | 4 (8.7) | |
| Missing data | 2 (4.3) | 0 (2.2) | |
| DSA class I at transplant (MFI) | < 0.01a | ||
| Absent | 9 (19.6) | 42 (91.3) | |
| < 5000 | 13 (28.3) | 4 (8.7) | |
| 5000-10000 | 4 (8.7) | 0 (0.0) | |
| ≥ 10000 | 15 (32.6) | 0 (0.0) | |
| Missing data | 5 (10.8) | 0 (0.0) | |
| DSA class II at transplant (MFI) | < 0.01a | ||
| Absent | 8 (17.4) | 41 (89.1) | |
| < 5000 | 14 (30.4) | 5 (8.9) | |
| 5000-10000 | 3 (6.5) | 0 (0.0) | |
| ≥ 10000 | 16 (34.8) | 0 (0.0) | |
| Missing data | 5 (10.9) | 0 (0.0) | |
| Induction | < 0.01a | ||
| ATLG | 46 (100) | 0 (0.0) | |
| Basiliximab | 0 (0) | 3 (6.5) | |
| IVIG | 43 (93.5) | 0 (0.0) | < 0.01a |
| Immunosuppression at baseline | < 0.01a | ||
| MMF + corticosteroids (delayed introduction of CNI between day 2-3) | 32 (69.6) | 10 (28.3) | |
| MMF + corticosteroids+ CNI at day 0-1 | 14 (30.4) | 33 (71.7) | |
| Trough level of CNI (tacrolimus) (ng/mL) | |||
| At 3 months | 7.9 ± 3.3 | 10.7 ± 4.4 | 0.002 |
| At 6 months | 7.1 ± 2.1 | 7.1 ± 2.9 | 0.99 |
| At 1 year | 6.7 ± 3.3 | 7.2 ± 2.1 | 0.38 |
| At 3 years | 6.0 ± 2.7 | 6.3 ± 1.8 | 0.56 |
| At 5 years | 5.7 ± 1.9 | 6.0 ± 2.5 | 0.58 |
| Trough level of everolimus (ng/mL) | |||
| At 1 year | 4.5 ± 3.0 | 4.9 ± 1.6 | 0.62 |
| At 3 years | 5.7 ± 1.9 | 6.1 ± 2.5 | 0.59 |
Hyperimmunized recipients received rATLG plus high-dose IVIG (100% vs 0%; P < 0.01), while calcineurin inhibitor (CNI) initiation was delayed (post-operative days 2-3) more frequently in this group (69.6% vs 28.3%; P < 0.01). Low-risk patients typically received triple therapy (CNI, mycophenolate mofetil, corticosteroids), with only 6.5% (3) receiving basiliximab induction due to renal impairment (Table 2).
Maintenance immunosuppression regimens were similar between groups at all timepoints. Notably, high-risk recipients did not require higher CNI troughs despite their immunological risk (year 1 tacrolimus: 6.7 ng/mL vs 7.2 ng/mL, P = 0.38), suggesting ATLG + IVIG induction may facilitate standard-dose CNI use in sensitized patients. Significant early difference at 3 months (7.9 ng/mL vs 10.7 ng/mL, P = 0.002) likely reflects delayed CNI initiation in high-risk patients. By 6 months, troughs equalized (7.1 ng/mL in both groups, P = 0.99) with no long-term differences. This suggests ATLG + IVIG induction doesn't require higher maintenance immunosuppression long-term (Table 2).
In the rATLG/IVIG-treated high-risk group, postoperative leukocyte counts showed a slight elevation on day 1, followed by a decrease until day 30, with mean values remaining within the normal range [day 1: (10.3 ± 5.9) × 109/L, day 4: (7.3 ± 5.8) × 109/L, day 7: (9.3 ± 5.5) × 109/L, and day 30: (6.9 ± 5.1) × 109/L]. The total lymphocyte count demonstrated a significant decrease, reaching its nadir on postoperative days 1 and 4, followed by a gradual increase until month 1 [day 1: (0.28 ± 0.342) × 109/L, day 4: (0.43 ± 0.414) × 109/L, day 7: (0.72 ± 0.611) × 109/L, and day 30: (1.02 ± 0.759) × 109/L].
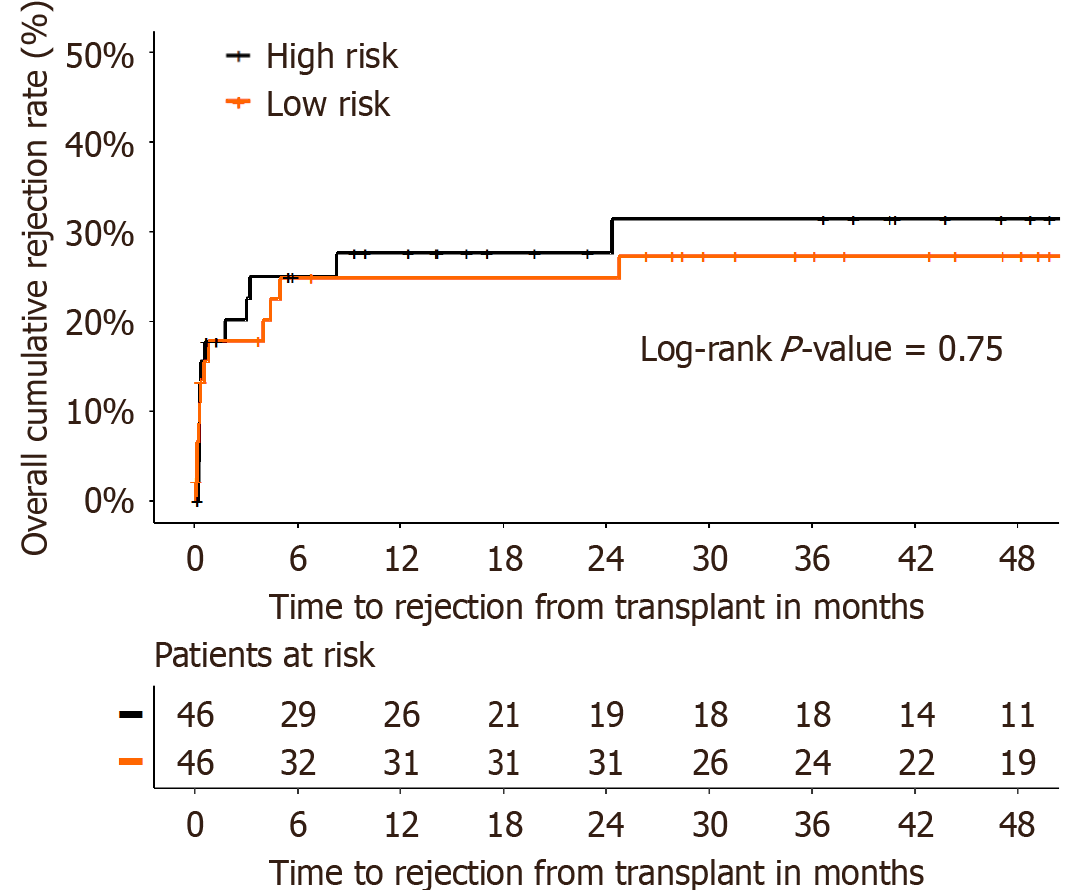
The overall incidence of biopsy-proven rejection was similar between the high-risk (28.3%, 13/46) and low-risk (26.1%, 12/46) groups (P = 0.82), with comparable time to first rejection (3.3 ± 6.7 months vs 3.4 ± 6.9 months; P = 0.98) (Table 3).
| High-risk group (n = 46) | Low-risk group (n = 46) | P value | |
| Overall rejection | 13 (28.3) | 12 (26.1) | 0.82 |
| Overall time to rejection (months) | 3.3 ± 6.7 | 3.4 ± 6.9 | 0.98 |
| Median IQR (Q1-Q3) | 0.367 (0.3-3.1) | 0.45 (0.2-4.3) | |
| Rejection type | 0.21 | ||
| TCMR | 9 (19.6) | 12 (26.1) | |
| aAMR | 2 (4.3) | 0 (0) | |
| aAMR + TCMR | 2 (4.3) | 0 (0) | |
| TCMR | |||
| BANFF classification | 0.21 | ||
| Mild (4-5) | 7/11 (63.6) | 10/12 (83.3) | |
| Moderate (6-7) | 4/11 (36.4) | 2/12 (16.7) | |
| Severe (8-9) | 0 (0) | 0 (0) | |
| Median time to TCMR (Q3-Q1) (days) | 7.9 (96.9-8) | 13.5 (129-4.8) | 0.87 |
| Treatment of TCMR (n = 9) | 0.14 | ||
| Steroid bolus | 5/46 (10.9) | 4/46 (8.7) | |
| Increase CNI dose | 1/46 (2.2) | 2/46 (4.3) | |
| Spontaneously resolved | 3/46 (6.5) | 6/46 (13.0) | |
| aAMR | |||
| aAMR | 4/46 (8.6) | 0 (0) | 0.06 |
| Median time to aAMR (Q3-Q1) (days) | 36 (81.8-10.5) | - | - |
| Treatment of aAMR | |||
| Switch cyclosporine to tacrolimus | 1/4 (25.0) | - | - |
| Steroid bolus + rituximab + 5 sessions PE | 1/4 (25.0) | - | - |
| Steroid bolus + IVIG | 2/4 (50.0) | - | - |
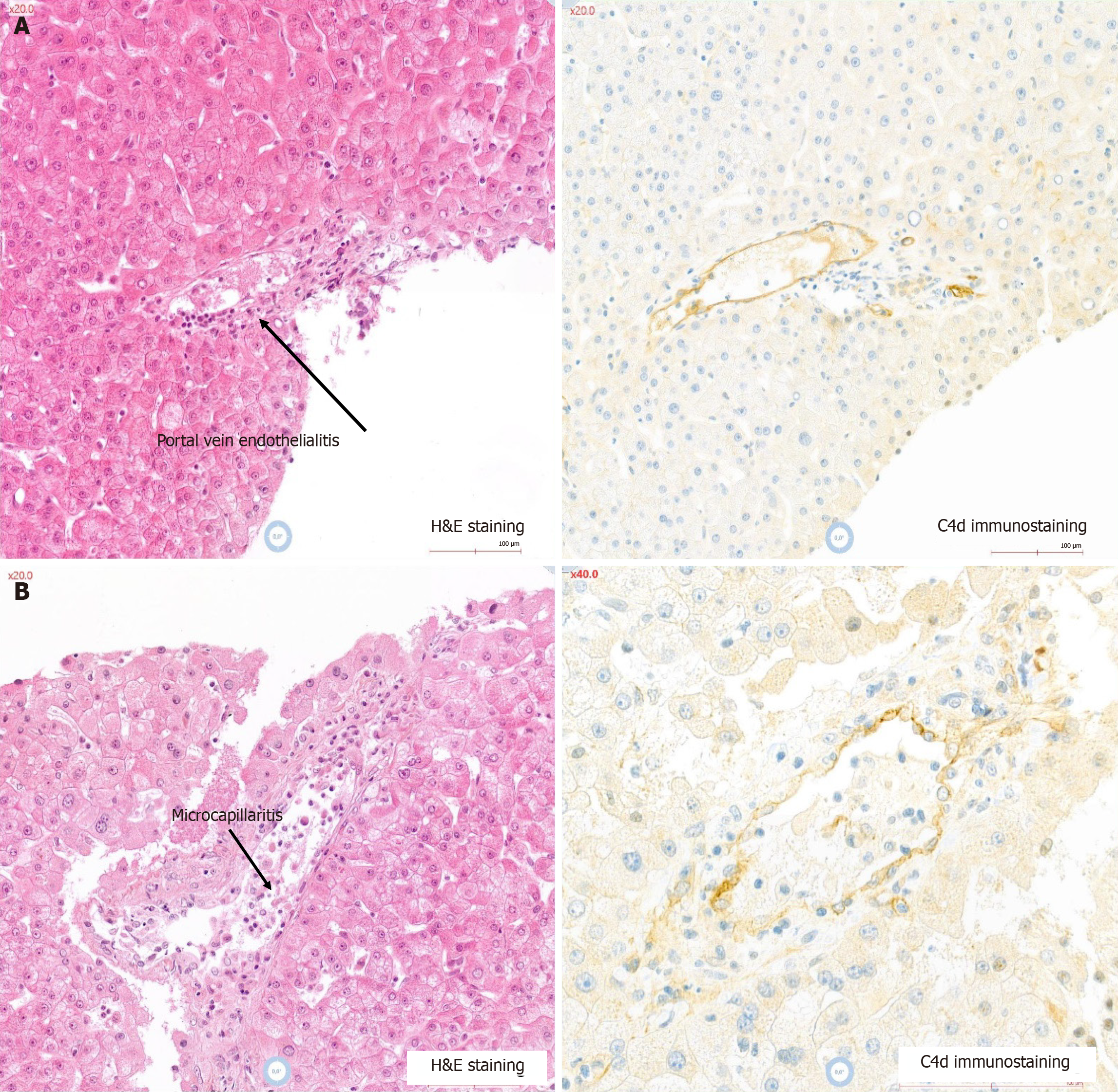
In the high-risk group, 19.5% (9/46) experienced TCMR (mild: n = 5, moderate: n = 4), managed with steroids (n = 5), increased CNI (n = 1), or spontaneously resolved without therapeutic intervention (n = 3). Therefore, 10.9% of the hyperimmunized patients had a biopsy-proven rejection and treated with a bolus of steroids. aAMR occurred in 8.7% (4/46 patients), with 50% (2/4) having concurrent severe TCMR (Banff 4-5). Pathological features are shown in Figure 1. All patients had highly elevated total MFI of pDSA (> 20000) at the time of rejection. Treatments for aAMR included steroids (n = 2), tacrolimus switch (n = 1), and steroids/rituximab/plasma exchange (n = 1) (Table 3).
In the low-risk group, 26.1% (12/46) developed TCMR (mild: n = 10, moderate: n = 2), resolving spontaneously resolved without therapeutic intervention (n = 6), with steroids (n = 4), or with CNI adjustment (n = 2). Steroid boluses were used for 8.7% of low-risk patients with rejection. Therefore, 8.7% of patients had biopsy-proven rejection that was treated with a bolus of steroids. No aAMR occurred in this group.
The cumulative time to first acute rejection (TCMR or aAMR) was similar between the groups (P = 0.75) (Figure 2). Table 4 details the characteristics and outcomes of the 13 rejected grafts in the high-risk group. In the high-risk group, two patients developed aAMR: The first had high-MFI of pDSA class I (> 10000; A2: 20648, A24: 14821, B56: 17979, B62: 14613), and the second had high MFI pDSA class II (> 10000; B1: 3965, DQ5: 22007, DQ6: 22594). Two patients experienced combined aAMR and TCMR. One had high MFI pDSA class I (> 10000; A33: 557, B65: 7008, B71: 3248, C8: 5015, C10: 5637). The other patient had high MFI pDSA class I and class II (> 10000; A24: 9500, B13: 12700, B7: 13141, DR15: 22100, DR51: 23300, DQ5: 2200, DR13: 2400) (Table 4).
| Rejection type | CM | DSA at LT (MFI) | Time to rejection from LT | BANFF score | C4d | Cholestasis | Other causes of cholestasis | DSA at time of rejection | Treatment | Follow up | |
| 1 | TCMR | Positive | - | Day 7 | 6 | Negative | Yes | No | - | Increase CNI dose | Died (sepsis; day 9 post-LT) |
| 2 | TCMR | Positive | I (9175); II (8575) | Year 2 | 4 | Negative | No | No | - | No | Normal |
| 3 | TCMR | Positive | I (22544); II (3314) | Day 7 | 4 | Negative | Yes | No | I (2176); II (1528) | Cs. bolus | Normal |
| 4 | TCMR | Positive | I (absent); II (2017) | Day 11 | 6 | Negative | No | No | I (absent); II (2700) | Cs. bolus + increase CNI dose | Normal |
| 5 | TCMR | Positive | I (19903); II (4539) | Month 3 | 5 | Positive | No | No | I (absent); II (596) | Cs. bolus | Normal |
| 6 | TCMR1 | Negative | I (728); II (8230) | Day 5 | 6 | Negative | Yes | No | - | Cs. bolus | Normal |
| 7 | TCMR1 | Negative | I (absent); II (6222) | Day 8 | 5 | Negative | Yes | No | - | No | Normal |
| 8 | TCMR | Positive | - | Day 7 | 5 | Negative | No | No | - | No | Normal |
| 9 | TCMR | Positive | I (14134); II (2368) | Month 8 | 7 | Negative | Yes | No | - | Cs. bolus | ECR (at month 18 post LT) |
| 10 | aAMR | Positive | I (57135)2; II (absent) | Day 54 | 3 | Positive | Yes | No | I (31075); II (absent) | Cs. bolus + IVIG | ECR (at month 19 post LT) |
| 11 | aAMR1 | Positive | I (3965)3; II (44601) | Day 91 | 3 | Positive | Yes | No | I (absent); II (4990) | Switch from cyclosporin to tacrolimus | ECR (at month 20 post LT) |
| 12 | aAMR TCMR | Positive | I (21465)4; II (2452) | Day 8 | 4 | Positive | Yes | No | I (83711); II (1484) | Cs. bolus + rituximab + 5 sessions of PE | Re-LT for biliary anastomotic stricture |
| 13 | aAMR TCMR | Positive | I (35341)5; II (50000) | Day 18 | 5 | Positive | Yes | No | I (4500); II (6000) | Cs. bolus + IVIG | Normal |
One-year post-transplant biopsies in high-risk recipients demonstrated steatohepatitis (n = 2), acute lobular hepatitis (n = 3), portal fibrosis (n = 3), regenerative nodules (n = 2), and hepatocellular carcinoma (HCC) (n = 1). Only one patient progressed to cirrhosis by 5 years. Low-risk recipients showed greater prevalence of steatohepatitis (n = 4) and regenerative nodules (n = 5), with minimal fibrosis (n = 1).
The mean overall follow-up duration was 42.8 ± 25.5 months. The mean ICU stay was significantly longer in the high-risk group compared to the low-risk group (10.9 ± 9.9 days vs 7.2 ± 5.8 days, P = 0.04) (Table 5).
| High-risk group (n = 46) | Low-risk group (n = 46) | P value | |
| Follow up period (months) | 35.8 ± 26.1 | 49.8 ± 25 | 0.01 |
| ICU stay (days) | 10.9 ± 9.9 | 7.2 ± 5.8 | 0.04 |
| Re-transplantation | 3 (6.5) | 2 (4.3) | 0.65 |
| Overall mortality rate | 11 (23.9) | 5 (10.9) | 0.09 |
| Cause of death | 0.07 | ||
| HCC recurrence | 3 (27.3) | 1 (20.0) | |
| Sepsis & multi organ failure | 3 (27.3) | 2 (40.0) | |
| Cardiovascular events | 5 (45.4) | 0 (0.0) | |
| Metastatic cancer | 0 (0.0) | 2 (40.0) | |
| 1-year post-transplant complications | |||
| Primary nonfunction graft | 1 (2.2) | 2 (4.5) | 0.53 |
| Small for size | 2 (4.3) | 0 (0.0) | 0.16 |
| Hepatic artery stenosis | 3 (6.5) | 1 (2.3) | 0.33 |
| Intestinal obstruction | 2 (4.3) | 0 (0.0) | 0.16 |
| Pancreatic fistula | 1 (2.2) | 0 (0.0) | 0.33 |
| Colonic perforation | 1 (2.2) | 0 (0.0) | 0.33 |
| Hematoma | 2 (4.3) | 3 (6.8) | 0.61 |
| Biliary stenosis | 2 (4.3) | 2 (4.5) | 0.96 |
| CMV reactivation | 3 (6.5) | 2 (4.5) | 0.68 |
| Viral infection | 1 (2.2) | 0 (0.0) | 0.33 |
| Bacterial infection | 4 (8.7) | 5 (11.4) | 0.321 |
| Fungal infection | 4 (8.7) | 1 (2.3) | 0.18 |
| Epilepsy | 1 (2.2) | 1 (2.3) | 0.98 |
| Hypertension | 1 (2.2) | 0 (0.0) | 0.33 |
| Acute kidney injury | 8 (17.4) | 2 (4.5) | 0.05 |
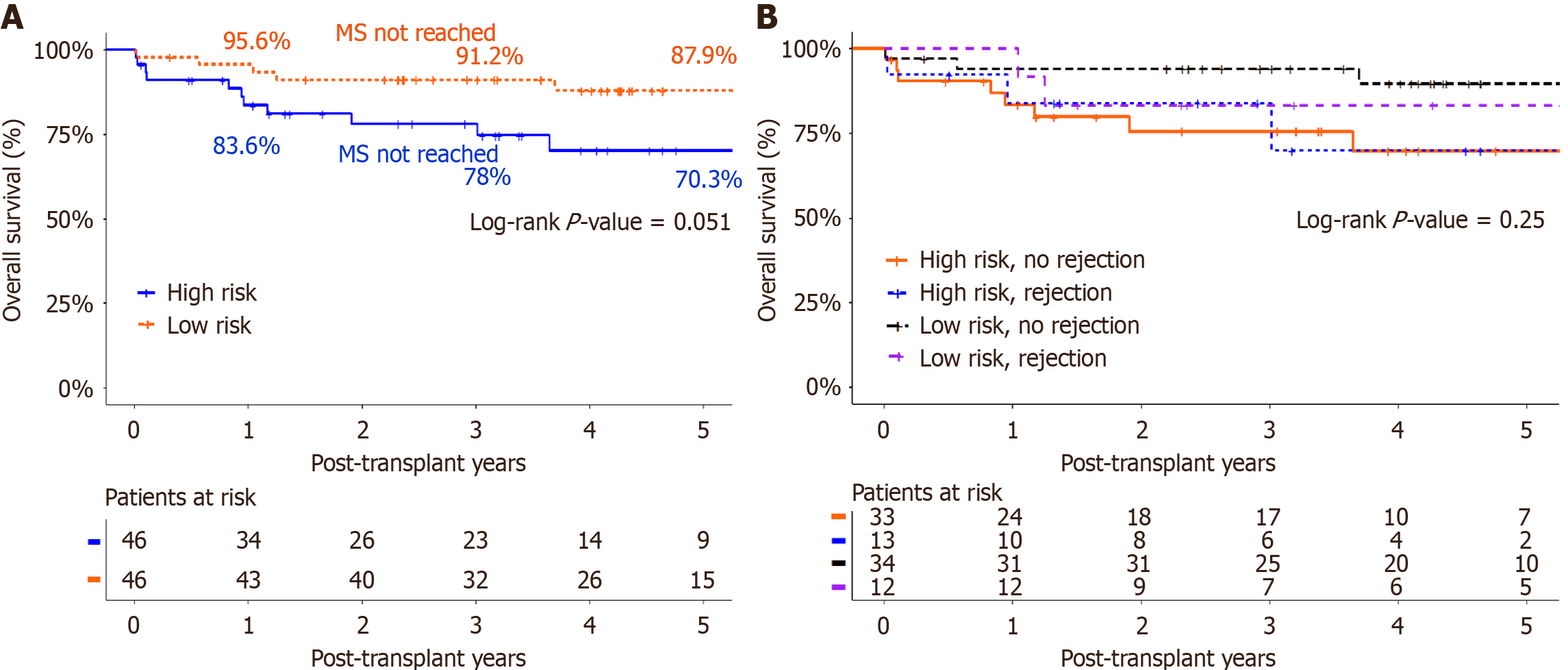
The survival curves suggest a potential clinically meaningful difference favoring non-immunized (low-risk) recipients (1-year: 95.6% vs 83.6%; 3-year: 91.2% vs 78.0%; 5-year: 87.9% vs 70.3%), though this did not reach statistical significance (log-rank P = 0.051) (Figure 3A). This borderline P-value likely reflects our limited sample size and non-rejection mortality risk rather than definitive equivalence. Overall patient survival did not differ significantly between patients who experienced graft rejection and those who did not, within either group (P = 0.25) (Figure 3B).
Re-transplantation occurred in 6.5% (3/46) of hyperimmunized patients due to a primary graft dysfunction (day 3), a small-for-size graft (day 30), and biliary stenosis (14 months). In the control group, 4.3% (2/46) underwent re-transplantation due to primary graft dysfunction (day 7) and hepatic artery stenosis (2 months) (Table 5).
The overall mortality rate was numerically higher in the high-risk group (23.9%, 11/46 patients) compared to the low-risk group (10.9%, 5/46 patients), although this difference did not reach statistical significance (P = 0.09). In the high-risk group, the leading causes of death were cardiovascular events (45.4%, 5 patients), which included two cases of cerebral hemorrhage (at 1 week and 2 years post-LT), two vascular aneurysm ruptures (at 1- and 3-year post-LT), and one severe mesenteric ischemia (at 1-month post-LT). Post-transplant HCC recurrence accounted for 27.3% of deaths (3 patients). For these HCC-related deaths, one patient died from cerebral metastasis at 4 years post-LT, and two patients died from liver and/or bone metastases at 1-year post-LT. All three patients with HCC recurrence had high pDSA titers and a positive CM at time of LT; notably, one of these patients also developed acute cellular rejection that was treated with bolus of corticosteroids. Septic complications accounted for the remaining 27.3% of deaths (3 patients) (Table 5). Among eight high-risk patients with previous transplants, one died from sepsis (pulmonary aspergillosis) 10 months post-LT while both patients in the low risk group survived.
The incidence of complications within the first year following LT was generally comparable between the high-risk and low-risk groups (Table 5). The only statistically significant difference observed was a higher incidence of acute kidney injury in the high-risk group (17.4%) compared to the low-risk group (4.5%, P = 0.05). Notably, no premedication was administered prior to ATLG infusion, and no induction-related allergic reactions or serious adverse events were reported in the high-risk group.
There were no cases of post-LT Epstein-Barr virus infection in either group. CMV reactivation occurred in 3 patients in the high-risk group (at 2 weeks, 1 month, and 1 year post-LT), compared to one patient in the low-risk group, who experienced reactivation at 1 year post-LT. Fungal infections were observed in 4 high-risk patients: Candida albicans infection within 4-7 days post-LT in two patients, invasive pulmonary aspergillosis at 1 month post-LT, and fungal sinusitis at 4 months post-LT. One low-risk patient developed invasive pulmonary aspergillosis at 1-month post-LT. Bacterial infections occurred in 4 high-risk patients, all within the first week after LT, whereas 5 low-risk patients developed bacterial infections at 3 months or later post-LT. While the rATLG/IVIG group had a slightly higher incidence of early infections, this did not translate into higher rates of severe infections or mortality, and all were manageable with standard treatment and monitoring (Table 5).
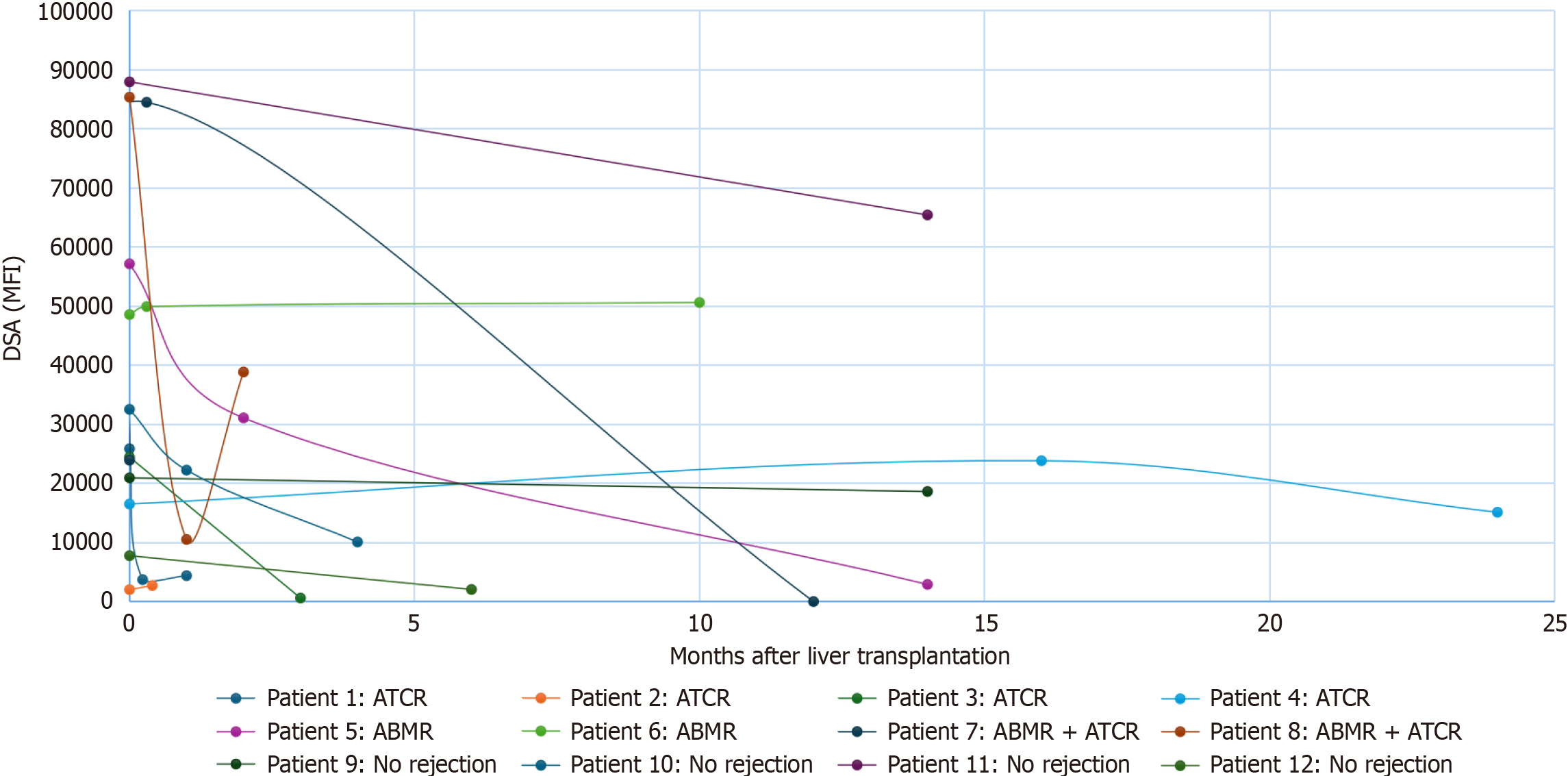
Serial DSA measurements were performed post-transplant in a subset of 12 high-risk patients, including 4 who developed aAMR, 4 with TCMR, and 4 who remained rejection-free. A decrease in DSA MFI levels post-transplant was observed in 75% of these patients. Specifically, DSA levels declined in all rejection-free patients, in two of the four patients with TCMR, and in three of the four patients with aAMR. These findings suggest a potential for DSA adsorption by the graft and highlight the potential importance of monitoring DSA dynamics in the surveillance of post-transplant rejection (Figure 4 and Table 6).
| Patient No. | Rejection status | DSA MFI sum at LT | Post-LT DSA MFI sum (months after LT) | Post-LT DSA MFI sum (months after LT) |
| 1 | TCMR | 25858 | 3704 (7 days)1 | 4382 (1 month) |
| 2 | TCMR | 2017 | 2708 (12 days)1 | - |
| 3 | TCMR | 24442 | 596 (3 months)1 | - |
| 4 | TCMR | 16502 | 23830 (16 months) | 15110 (24 months) |
| 5 | aAMR | 57138 | 31075 (2 months)1 | 2900 (14 months) |
| 6 | aAMR | 48566 | 49909 (3 months)1 | 50599 (at 10 months) |
| 7 | aAMR + TCMR | 23917 | 84474 (10 days)1 | 0 (12 months) |
| 8 | aAMR + TCMR | 85341 | 10500 (1 month)1 | 38841 (2 months) |
| 9 | No rejection | 20926 | 18624 (14 months) | - |
| 10 | No rejection | 32520 | 22236 (1 month) | 10081 (4 months) |
| 11 | No rejection | 87931 | 65414 (14 months) | - |
| 12 | No rejection | 7764 | 2045 (6 month) | - |
This study assesses rATLG and IVIG induction in hyperimmunized LT recipients with high pDSA MFI and/or positive D/R CM. Despite elevated immunological risk, biopsy-proven rejection (aTCMR/aAMR) rates were similar between hyperimmunized and non-immunized groups (28.3% vs 26.1%), aligning with prior studies reporting 23%-65% rejection in high-risk cohorts[13-16].
The incidence of biopsy-proven, treated aTCMR was 10.9% in hyperimmunized recipients vs 8.7% in controls. Only four hyperimmunized patients (8.7%) developed aAMR, with none in the control group. These rates are higher than the expected 2%-5% range, likely due to rigorous monitoring protocols, enhancing early detection[17-21]. Identifying DSAs and AMR early is vital, since timely changes to immunosuppression can improve graft survival and help avoid per
Interestingly, aAMR developed only in the high-risk group (8.7%), even though overall rejection rates were similar, indicating rATLG/IVIG induction therapy is more effective at preventing TCMR than aAMR in highly sensitized patients. Patients with very high-titer DSA (> 20000 MFI) remain susceptible to aAMR, but all cases were managed successfully without graft loss. This highlights the value of close post-transplant monitoring and prompt intervention for high-risk individuals.
Although hyperimmunization (high pDSA MFI > 10000 or positive CM) is linked to increased aAMR risk[4,7,17,23,24], its impact on long-term outcomes was minimal in our cohort, with comparable survival between groups (P = 0.25). Early rATLG induction may mitigate rejection risks with rejection rates similar to those in non-immunized recipients, aligning with findings showing ATG’s protective effect[21,25,26]. No severe TCMR or graft loss occurred, consistent with studies demonstrating ATG’s efficacy in reducing rejection and graft loss[7,17,27]. Re-LT rates were similar (6.5% vs 4.3%, P = 0.65). All 4 aAMR cases had high-titer (≥ 20000 MFI) class II pDSAs (HLA-DR/DQ/DP), corroborating evidence linking high MFI pDSAs to aAMR[17,21,28]. O’Leary et al[27] found class II pDSAs > 5000 MFI increased early rejection risk (HR = 1.58, P = 0.004). Biopsy with acid elution can confirm DSA binding to graft tissue, aiding diagnosis[4,17,29].
Some reports have investigated the efficacy and safety of different rabbit polyclonal anti-thymocyte induction regimens in renal transplantation and confirmed that there was a significant lower incidence of CMV infection, malignancy and death in patients treated with ATLG compared to thymoglobin[30-35]. Regarding induction therapy, our study's uti
Our data challenge the paradigm that hyperimmunized recipients universally require intensified maintenance immunisuppression regemin. Despite higher baseline risk, the high-risk group maintained comparable CNI troughs to controls, with no increase in rejection. This aligns with kidney transplant evidence that ATG induction permits lower long-term CNI exposure[32-34].
The reported impact of pDSA and a positive CM on graft and patient survival following LT remains a subject of ongoing debate, with some studies suggesting a detrimental effect[6,8,23,27,36,37] while others have found no significant association[3,13,14,38-40]. In this study, the hyperimmunized high-risk group exhibited overall survival rates of 83.6%, 78.0%, and 70.3% at 1 year, 3 years, and 5 years, respectively, compared to 95.6%, 91.2%, and 87.9% in the non-immunized low-risk group (P = 0.051). Although the P value (0.051) narrowly missed significance, the observed survival trends align with prior studies showing hyperimmunized recipients face higher non-rejection mortality risks[7]. The numerical difference may instead reflect more living donor transplants in high-risk patients (8.7% vs 0%, P = 0.04) and longer ICU stays (10.9 days vs 7.2 days, P = 0.04) suggesting greater perioperative complexity.
The overall mortality rates between the hyperimmunized and non-immunized control groups did not reach statistical significance (P = 0.09). Notably, the primary causes of death in the high-risk group were HCC recurrence and car
This study also confirmed the common notion that subclinical graft injury exhibits more hepatitis-like features over time after LT[12]. Subclinical graft injury and fibrosis progressed over time after LT. Thereby, biopsies without evidence of rejection declined, while indeterminate findings increased over time. The progression of these hepatitis features was accompanied by an increasing appearance of DSAs. Some studies[4,12,24,41] have reported an increase in DSAs over time. However, this association does not clarify whether DSAs are a cause or a consequence of graft injury.
This study is the first matched case-control analysis of long-term outcomes in hyperimmunized LT recipients receiving standardized rATLG and IVIG induction. Matching on key confounders (age, sex, MELD, indication) enhances comparability. A mean follow-up of 42.8 months allows assessment of long-term survival. Rigorous pre-transplant immunological profiling, including HLA typing and DSA testing, strengthens immunological validity. However, the retrospective design risks bias, and lack of routine post-transplant DSA monitoring limits understanding of DSA evolution and subclinical graft effects. Despite these limitations, the study provides valuable insights into outcomes of hyperimmunized recipients under targeted induction, offering a foundation for future prospective research. The impact of undetected subclinical rejection could be systematically evaluated in future trials employing protocol biopsies at standardized post-transplant intervals. This study offers mid-term data with an average follow-up of 42.8 months, but longer observation is needed to fully evaluate rATLG/IVIG induction’s effects on chronic rejection and late graft function. Although 5-year survival trends are not statistically significant, ongoing monitoring of these patients on the long term is of importance to capture chronic rejection and chronic graft dysfunction. We acknowledge that the single-center sample (n = 46 per group) limited statistical power for some outcomes. A post-hoc calculation based on observed 3-year survival rates (78.0% vs 91.2%) indicates only approximately 43% power to detect this difference (α = 0.05), implying a substantial risk of type II error. Consequently, the borderline P-value for overall survival (P = 0.051) may represent underpowering rather than absence of a true difference. The generalizability of our findings should be considered in the context of the patient population and study setting. The rATLG/IVIG strategy was tested across a spectrum of high-risk patients, but its efficacy may vary with DSA characteristics. The development of aAMR exclusively in patients with very high-titer DSA (> 20000 MFI) suggests that while this regimen is broadly applicable to hyperimmunized recipients, the highest-risk patients may require augmented therapy. Larger, multi-center prospective studies including a cohort of untreated hyperimmunized control group are warranted to validate these results and further optimize immunosuppressive strategies for this high-risk population.
aAMR mainly affected hyperimmunized recipients with high pDSA MFI and/or positive CM. A short-course rATLG and IVIG induction was associated with similar acute rejection rates and long-term patient and graft survival in both hyperimmunized and non-immunized recipients, supporting its use as a valuable strategy for mitigating early immunological risks in this high-risk group. Declining post-transplant DSA levels, potentially due to graft adsorption highlights the dynamic nature of DSA and the need for close monitoring. For suspected rejection, intensified DSA tracking and early biopsy are crucial for timely intervention for antibody-mediated injury. Overall, a tailored induction approach using rATLG and IVIG probably balanced the need for potent immunosuppression to prevent rejection with the risks of opportunistic infections and other adverse events in susceptible recipients.
| 1. | Kim WR, Lake JR, Smith JM, Skeans MA, Schladt DP, Edwards EB, Harper AM, Wainright JL, Snyder JJ, Israni AK, Kasiske BL. OPTN/SRTR 2015 Annual Data Report: Liver. Am J Transplant. 2017;17 Suppl 1:174-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 230] [Article Influence: 25.6] [Reference Citation Analysis (2)] |
| 2. | Lee BT, Fiel MI, Schiano TD. Antibody-mediated rejection of the liver allograft: An update and a clinico-pathological perspective. J Hepatol. 2021;75:1203-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (3)] |
| 3. | Ruiz R, Tomiyama K, Campsen J, Goldstein RM, Levy MF, McKenna GJ, Onaca N, Susskind B, Tillery GW, Klintmalm GB. Implications of a positive crossmatch in liver transplantation: a 20-year review. Liver Transpl. 2012;18:455-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (2)] |
| 4. | Del Bello A, Congy-Jolivet N, Muscari F, Lavayssière L, Esposito L, Cardeau-Desangles I, Guitard J, Dörr G, Suc B, Duffas JP, Alric L, Bureau C, Danjoux M, Guilbeau-Frugier C, Blancher A, Rostaing L, Kamar N. Prevalence, incidence and risk factors for donor-specific anti-HLA antibodies in maintenance liver transplant patients. Am J Transplant. 2014;14:867-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 5. | Cillo U, Bechstein WO, Berlakovich G, Dutkowski P, Lehner F, Nadalin S, Saliba F, Schlitt HJ, Pratschke J. Identifying risk profiles in liver transplant candidates and implications for induction immunosuppression. Transplant Rev (Orlando). 2018;32:142-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 6. | Levitsky J, Goldberg D, Smith AR, Mansfield SA, Gillespie BW, Merion RM, Lok AS, Levy G, Kulik L, Abecassis M, Shaked A. Acute Rejection Increases Risk of Graft Failure and Death in Recent Liver Transplant Recipients. Clin Gastroenterol Hepatol. 2017;15:584-593.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 188] [Article Influence: 20.9] [Reference Citation Analysis (1)] |
| 7. | Kubal CA, Mangus RS, Saxena R, Lobashevsky A, Higgins N, Agarwal A, Fridell JA, Tector AJ. Crossmatch-positive liver transplantation in patients receiving thymoglobulin-rituximab induction. Transplantation. 2014;97:56-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 8. | McCaughan JA, Robertson V, Falconer SJ, Cryer C, Turner DM, Oniscu GC. Preformed donor-specific HLA antibodies are associated with increased risk of early mortality after liver transplantation. Clin Transplant. 2016;30:1538-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 9. | Charlton M, Levitsky J, Aqel B, OʼGrady J, Hemibach J, Rinella M, Fung J, Ghabril M, Thomason R, Burra P, Little EC, Berenguer M, Shaked A, Trotter J, Roberts J, Rodriguez-Davalos M, Rela M, Pomfret E, Heyrend C, Gallegos-Orozco J, Saliba F. International Liver Transplantation Society Consensus Statement on Immunosuppression in Liver Transplant Recipients. Transplantation. 2018;102:727-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 221] [Article Influence: 27.6] [Reference Citation Analysis (2)] |
| 10. | Stevens RB, Wrenshall LE, Miles CD, Farney AC, Jie T, Sandoz JP, Rigley TH, Osama Gaber A. A Double-Blind, Double-Dummy, Flexible-Design Randomized Multicenter Trial: Early Safety of Single- Versus Divided-Dose Rabbit Anti-Thymocyte Globulin Induction in Renal Transplantation. Am J Transplant. 2016;16:1858-1867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 11. | Gharekhani A, Entezari-Maleki T, Dashti-Khavidaki S, Khalili H. A review on comparing two commonly used rabbit anti-thymocyte globulins as induction therapy in solid organ transplantation. Expert Opin Biol Ther. 2013;13:1299-1313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Demetris AJ, Bellamy C, Hübscher SG, O'Leary J, Randhawa PS, Feng S, Neil D, Colvin RB, McCaughan G, Fung JJ, Del Bello A, Reinholt FP, Haga H, Adeyi O, Czaja AJ, Schiano T, Fiel MI, Smith ML, Sebagh M, Tanigawa RY, Yilmaz F, Alexander G, Baiocchi L, Balasubramanian M, Batal I, Bhan AK, Bucuvalas J, Cerski CTS, Charlotte F, de Vera ME, ElMonayeri M, Fontes P, Furth EE, Gouw ASH, Hafezi-Bakhtiari S, Hart J, Honsova E, Ismail W, Itoh T, Jhala NC, Khettry U, Klintmalm GB, Knechtle S, Koshiba T, Kozlowski T, Lassman CR, Lerut J, Levitsky J, Licini L, Liotta R, Mazariegos G, Minervini MI, Misdraji J, Mohanakumar T, Mölne J, Nasser I, Neuberger J, O'Neil M, Pappo O, Petrovic L, Ruiz P, Sağol Ö, Sanchez Fueyo A, Sasatomi E, Shaked A, Shiller M, Shimizu T, Sis B, Sonzogni A, Stevenson HL, Thung SN, Tisone G, Tsamandas AC, Wernerson A, Wu T, Zeevi A, Zen Y. 2016 Comprehensive Update of the Banff Working Group on Liver Allograft Pathology: Introduction of Antibody-Mediated Rejection. Am J Transplant. 2016;16:2816-2835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 471] [Article Influence: 47.1] [Reference Citation Analysis (1)] |
| 13. | Taner T, Gandhi MJ, Sanderson SO, Poterucha CR, De Goey SR, Stegall MD, Heimbach JK. Prevalence, course and impact of HLA donor-specific antibodies in liver transplantation in the first year. Am J Transplant. 2012;12:1504-1510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 136] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 14. | Lunz J, Ruppert KM, Cajaiba MM, Isse K, Bentlejewski CA, Minervini M, Nalesnik MA, Randhawa P, Rubin E, Sasatomi E, de Vera ME, Fontes P, Humar A, Zeevi A, Demetris AJ. Re-examination of the lymphocytotoxic crossmatch in liver transplantation: can C4d stains help in monitoring? Am J Transplant. 2012;12:171-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 15. | Wiesner RH, Demetris AJ, Belle SH, Seaberg EC, Lake JR, Zetterman RK, Everhart J, Detre KM. Acute hepatic allograft rejection: incidence, risk factors, and impact on outcome. Hepatology. 1998;28:638-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 336] [Article Influence: 12.0] [Reference Citation Analysis (2)] |
| 16. | Dumortier J, Conti F, Hiriart JB, Dharancy S, Duvoux C, Besch C, Houssel-Debry P, Latournerie M, Chermak F, Meszaros M, Pageaux GP, Radenne S, Boillot O, Hardwigsen J, Kounis I, Kamar N, Saliba F, Erard D, Del Bello A. Treatment of donor-specific anti-HLA antibodies-mediated rejection after liver transplantation: A French nationwide retrospective study. Liver Transpl. 2023;29:1313-1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 17. | Del Bello A, Neau-Cransac M, Lavayssiere L, Dubois V, Congy-Jolivet N, Visentin J, Danjoux M, Le Bail B, Hervieu V, Boillot O, Antonini T, Kamar N, Dumortier J. Outcome of Liver Transplant Patients With Preformed Donor-Specific Anti-Human Leukocyte Antigen Antibodies. Liver Transpl. 2020;26:256-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 18. | Vandevoorde K, Ducreux S, Bosch A, Guillaud O, Hervieu V, Chambon-Augoyard C, Poinsot D, André P, Scoazec JY, Robinson P, Boillot O, Dubois V, Dumortier J. Prevalence, Risk Factors, and Impact of Donor-Specific Alloantibodies After Adult Liver Transplantation. Liver Transpl. 2018;24:1091-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 19. | Musat AI, Agni RM, Wai PY, Pirsch JD, Lorentzen DF, Powell A, Leverson GE, Bellingham JM, Fernandez LA, Foley DP, Mezrich JD, D'Alessandro AM, Lucey MR. The significance of donor-specific HLA antibodies in rejection and ductopenia development in ABO compatible liver transplantation. Am J Transplant. 2011;11:500-510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 142] [Article Influence: 9.5] [Reference Citation Analysis (2)] |
| 20. | Demetris AJ, Zeevi A, O'Leary JG. ABO-compatible liver allograft antibody-mediated rejection: an update. Curr Opin Organ Transplant. 2015;20:314-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 21. | Cicalese L, Walton ZC, Du X, Kulkarni R, Qiu S, El Hag M, Stevenson HL. Antibody-Mediated Rejection in Liver Transplantation: Immuno-Pathological Characteristics and Long-Term Follow-Up. Transpl Int. 2024;37:13232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 22. | Angelico R, Sensi B, Manzia TM, Tisone G, Grassi G, Signorello A, Milana M, Lenci I, Baiocchi L. Chronic rejection after liver transplantation: Opening the Pandora's box. World J Gastroenterol. 2021;27:7771-7783. [PubMed] [DOI] [Full Text] |
| 23. | O'Leary JG, Kaneku H, Demetris AJ, Marr JD, Shiller SM, Susskind BM, Tillery GW, Terasaki PI, Klintmalm GB. Antibody-mediated rejection as a contributor to previously unexplained early liver allograft loss. Liver Transpl. 2014;20:218-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 93] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 24. | Del Bello A, Congy-Jolivet N, Danjoux M, Muscari F, Lavayssière L, Esposito L, Cardeau-Desangles I, Guitard J, Dörr G, Milongo D, Suc B, Duffas JP, Alric L, Bureau C, Guilbeau-Frugier C, Rostaing L, Kamar N. De novo donor-specific anti-HLA antibodies mediated rejection in liver-transplant patients. Transpl Int. 2015;28:1371-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 25. | Baradaran H, Dashti-Khavidaki S, Taher M, Talebian M, Nasiri-Toosi M, Jafarian A. Antibody-Mediated Rejection in Adult Liver Transplant Recipients: A Case Series and Literature Review. J Clin Pharmacol. 2022;62:254-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 26. | O'Leary JG, Kaneku H, Susskind BM, Jennings LW, Neri MA, Davis GL, Klintmalm GB, Terasaki PI. High mean fluorescence intensity donor-specific anti-HLA antibodies associated with chronic rejection Postliver transplant. Am J Transplant. 2011;11:1868-1876. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 27. | O'Leary JG, Kaneku H, Jennings LW, Bañuelos N, Susskind BM, Terasaki PI, Klintmalm GB. Preformed class II donor-specific antibodies are associated with an increased risk of early rejection after liver transplantation. Liver Transpl. 2013;19:973-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (1)] |
| 28. | El Hag MI, Kaneku H, Jorgensen D, Zeevi A, Stevenson HL, Yadak N, Hassan M, Du X, Demetris AJ. Morphologic and immunophenotypic evaluation of liver allograft biopsies with contemporaneous serum DSA measurements. Clin Transplant. 2023;37:e14997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 29. | Neau-Cransac M, Le Bail B, Guidicelli G, Visentin J, Moreau K, Quinart A, Boueilh A, Laurent C, Taupin JL. Evolution of serum and intra-graft donor-specific anti-HLA antibodies in a patient with two consecutive liver transplantations. Transpl Immunol. 2015;33:58-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 30. | Ducloux D, Kazory A, Challier B, Coutet J, Bresson-Vautrin C, Motte G, Thalamy B, Rebibou JM, Chalopin JM. Long-term toxicity of antithymocyte globulin induction may vary with choice of agent: a single-center retrospective study. Transplantation. 2004;77:1029-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 57] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 31. | Bamoulid J, Crepin T, Gaiffe E, Laheurte C, Moulin B, Frimat L, Rieu P, Mousson C, Durrbach A, Heng AE, Rebibou JM, Saas P, Courivaud C, Ducloux D. Immune reconstitution with two different rabbit polyclonal anti-thymocytes globulins. Transpl Immunol. 2017;45:48-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 32. | Ray DS, Gang S, Khullar D, Bhalla AK, Sharma A, Abraham A, Raju SB, Siddhi V, Dutta AR, Abraham G, Shekar MS, Shah BV, Kothari J, Bahadur M, Ahmed I, Narula AS, Margabandhu S, Shetty S, Chodankar D, Mandrekar A, Salvi VS. Rabbit Anti-thymocyte Globulin (rATG) as Induction Immunosuppression Therapy in Patients Undergoing Renal Transplantation: Clinical Experience from RISE Registry. Indian J Transplant. 2023;17:294-300. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 33. | Brennan DC, Daller JA, Lake KD, Cibrik D, Del Castillo D; Thymoglobulin Induction Study Group. Rabbit antithymocyte globulin versus basiliximab in renal transplantation. N Engl J Med. 2006;355:1967-1977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 643] [Cited by in RCA: 550] [Article Influence: 27.5] [Reference Citation Analysis (2)] |
| 34. | Mourad G, Rostaing L, Legendre C, Garrigue V, Thervet E, Durand D. Sequential protocols using basiliximab versus antithymocyte globulins in renal-transplant patients receiving mycophenolate mofetil and steroids. Transplantation. 2004;78:584-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 135] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 35. | Kaden J, Völp A, Wesslau C. High graft protection and low incidences of infections, malignancies and other adverse effects with intra-operative high dose ATG-induction: a single centre cohort study of 760 cases. Ann Transplant. 2013;18:9-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 36. | Takaya S, Bronsther O, Iwaki Y, Nakamura K, Abu-Elmagd K, Yagihashi A, Demetris AJ, Kobayashi M, Todo S, Tzakis AG. The adverse impact on liver transplantation of using positive cytotoxic crossmatch donors. Transplantation. 1992;53:400-406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 101] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Kozlowski T, Rubinas T, Nickeleit V, Woosley J, Schmitz J, Collins D, Hayashi P, Passannante A, Andreoni K. Liver allograft antibody-mediated rejection with demonstration of sinusoidal C4d staining and circulating donor-specific antibodies. Liver Transpl. 2011;17:357-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 38. | den Dulk AC, Shi X, Verhoeven CJ, Dubbeld J, Claas FHJ, Wolterbeek R, Brand-Schaaf SH, Verspaget HW, Sarasqueta AF, van der Laan LJW, Metselaar HJ, van Hoek B, Kwekkeboom J, Roelen DL. Donor-specific anti-HLA antibodies are not associated with nonanastomotic biliary strictures but both are independent risk factors for graft loss after liver transplantation. Clin Transplant. 2018;32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 39. | Koch M, Marget M, Sterneck M, Fischer L, Thude H, Nashan B. Limited impact of pre-existing donor specific HLA-antibodies (DSA) on long term allograft survival after first adult liver transplantation. Hum Immunol. 2018;79:545-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 40. | Krendl FJ, Fodor M, Messner F, Balog A, Vales A, Cardini B, Resch T, Maglione M, Margreiter C, Riedmann M, Ulmer H, Öfner D, Oberhuber R, Schneeberger S, Weissenbacher A. Liver Transplantation in Recipients With a Positive Crossmatch: A Retrospective Single-Center Match-Pair Analysis. Transpl Int. 2023;36:11062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 41. | Höfer A, Jonigk D, Hartleben B, Verboom M, Hallensleben M, Manns MP, Jaeckel E, Taubert R. Non-invasive screening for subclinical liver graft injury in adults via donor-specific anti-HLA antibodies. Sci Rep. 2020;10:14242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |