Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115108
Revised: November 10, 2025
Accepted: January 7, 2026
Published online: March 27, 2026
Processing time: 169 Days and 17.7 Hours
Clinically relevant postoperative pancreatic fistula (CR-POPF) is a critical com
To identify independent risk factors for CR-POPF after splenectomy in this cohort and to develop a clinically useful predictive nomogram.
This retrospective study included 186 patients who underwent splenectomy for cirrhotic splenomegaly with hypersplenism at our institution, between January 2019 and December 2024. PF was diagnosed and graded according to the 2016 International Study Group on Pancreatic Fistulas criteria. Univariate and mul
Of the 186 patients, 21 developed biochemical leak, and 6 developed CR-POPF, including 4 grade B and 2 grade C. Multivariate least absolute shrinkage and selection operator-penalized logistic analysis revealed that body mass index (BMI) [odds ratio = 3.578, 95% confidence interval (CI): 1.156-11.080; P = 0.027] and splenic thickness (odds ratio = 1.250; 95%CI: 1.014-1.542; P = 0.037) were independent risk factors for CR-POPF. The nomogram integrating these factors demonstrated excellent discriminative ability (area under the receiver operating characteristic curve = 0.816, 95%CI: 0.500-0.993), good calibration (Hosmer-Lemeshow statistic = 0.027, degrees of freedom = 3, P = 0.999 > 0.05), and favorable clinical utility (positive net benefit across a threshold probability range of approximately 0% to 99% in the decision curve analysis). PF grade analysis showed significant differences in BMI and pancreatic texture (P < 0.05).
This study identified BMI and splenic thickness as independent risk factors for CR-POPF after splenectomy in patients with cirrhosis with splenomegaly. The developed nomogram provides a reliable tool for individualized risk prediction, facilitating targeted perioperative management to reduce CR-POPF incidence and improve patient outcomes.
Core Tip: This retrospective study included 186 cirrhotic patients with splenomegaly who underwent splenectomy. Through multivariate least absolute shrinkage and selection operator-penalized logistic regression, body mass index and splenic thickness were confirmed as independent risk factors for clinically relevant postoperative pancreatic fistula. The nomogram built on these factors achieved an area under the receiver operating characteristic curve of 0.816, providing reliable individualized perioperative risk prediction to cut clinically relevant postoperative pancreatic fistula occurrence and boost patient recovery.
- Citation: Huang L, Li QL, Yu QS, Peng H, Zhen Z, Zhang Q, Shen Y. Predicting pancreatic fistula post-splenectomy in cirrhosis with splenomegaly: Risk factors and nomogram validation. World J Hepatol 2026; 18(3): 115108
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115108.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115108
Pancreatic fistula (PF) is a significant and complex complication with diverse etiologies. Pancreatic surgeries such as pancreaticoduodenectomy and distal pancreatectomy carry a high risk of PF, with reported incidence rates ranging from 3% to 45%, as shown in multiple studies[1]. This wide variation can be ascribed to the differences in surgical techniques, patient populations, and diagnostic criteria. Traumatic injuries, particularly pancreatic contusions, lacerations, or crush injuries, can also lead to PF because of the often underestimated extent of tissue damage and difficulties in managing pancreatic duct injuries[2]. In addition, acute pancreatitis, especially severe cases, is frequently associated with PF, where local ischemia and the digestive action of pancreatic enzymes can result in pancreatic tissue and duct necrosis, and approximately 20% of severe acute pancreatitis survivors develop PF[3]. The overall mortality rate related to PF is approximately 1%, mainly due to subsequent severe infections and multi-organ failure.
PF has emerged as a common complication of splenectomy, primarily due to iatrogenic injury to the pancreatic tail during surgical manipulation. As most cases of PF induced by splenectomy present with mild symptoms and achieve a favorable prognosis after adequate drainage and other supportive interventions, this complication has long been insufficiently emphasized in clinical practice. Notably, the pathogenesis of PF differs substantially between pancreatic surgery and splenectomies. In pancreatic surgery, PF typically arises from pancreatic-intestinal anastomotic leaks or defects on the pancreatic surface. In contrast, splenectomy-related PF is predominantly caused by iatrogenic injury to the pancreatic tail parenchyma during splenic mobilization or pedicle dissection. This mechanistic distinction indicates that the risk factors for pancreatic surgery-associated PF are irrelevant to splenectomy cohorts. Given that this study focused on patients with cirrhotic splenomegaly, it emphasizes cirrhosis-related factors, which are necessary for addressing the unmet clinical needs of this specific patient population. Massive splenomegaly, especially when the spleen is significantly enlarged, increases the complexity of the surgical procedure and the likelihood of accidental pancreatic injury. The main manifestations of PF after splenectomy are consistent with those in other scenarios, including persistent abdominal pain, fever, and elevated amylase levels in peritoneal drainage fluid. If not treated or managed properly, it can progress to serious complications, such as intra-abdominal abscess formation, sepsis, and even life-threatening hemorrhage, which significantly prolong hospital stays and escalate healthcare costs[4]. The existing literature has identified some risk factors for PF after splenectomy, such as the type of splenectomy and use of energy-based devices[5]. However, these studies often have limitations in terms of the sample size, patient heterogeneity, and inconsistent definitions of risk factors. Currently, there is a lack of comprehensive and unified nomogram-based analyses of risk factors for PF.
The effective management of PF depends on accurate monitoring and classification. According to the diagnostic criteria revised by the International Study Group on Pancreatic Fistulas (ISGPS) in 2016, the amylase level in the drainage fluid three days after surgery exceeded three times the normal serum value, which is in line with the diagnosis of PF[6,7]. The ISGPS classifies PF into three grades: Grade A (biochemical leak) is characterized by an increase in amylase without clinical symptoms; grade B is characterized by continuous drainage for more than 3 weeks accompanied by signs of infection; and grade C is characterized by organ failure or even requires reoperation. The incidence of these severe adverse outcomes further increases with the presence of grade B or C fistulas, as defined by the ISGPS. Therefore, investigators have focused on improving predictive models to assess the risk of developing clinically relevant postoperative pancreatic fistulas (CR-POPF). The harmful effects of CR-POPF can lead to severe complications in various systems. The leakage of pancreatic enzymes into the abdominal cavity can cause corrosive bleeding. The literature shows that approximately 12.7% of patients with grade B/C fistulas will have secondary erosion of abdominal blood vessels, and rupture of the splenic artery or short gastric artery can lead to fatal massive hemorrhage[8]. Pancreatic juice spillage can also disrupt the peritoneal barrier, and approximately 34.5% of cases progress to diffuse peritonitis, which in turn can induce sepsis and multiple organ failure, with a mortality rate of 8.3% to 15.6%. Among patients with CR-POPF complicated by liver cirrhosis and portal hypertension, 28.6% have an increased risk of bleeding due to coagulation disorders, and the incidence of infectious complications is 4.7 times higher than that in patients without CR-POPF[8]. These data underscore the clinical threat of CR-POPF, which not only directly impairs organ function but also triggers systemic inflammatory response syndrome and consumptive coagulopathy, thereby initiating a life-threatening pathological cascade[9].
Given the significant morbidity and potential mortality associated with CR-POPF, understanding its risk factors is crucial[10]. Although several risk factors have been identified in previous studies, comprehensive and integrated analyses that synthesize all these factors are still lacking. In addition, no nomogram prediction model has been specifically developed for CR-POPF after splenectomy. In this study, we conducted a thorough analysis of CR-POPF risk factors using a retrospective review of 186 cases collected over a 6-year period. We aimed to develop a more accurate predictive nomogram model by systematically evaluating both the patient- and surgery-related variables. This nomogram can help surgeons identify high-risk patients pre- or intra-operatively, enabling more proactive and personalized management strategies to prevent or better manage CR-POPF after splenectomy, thus contributing to clinical prevention efforts.
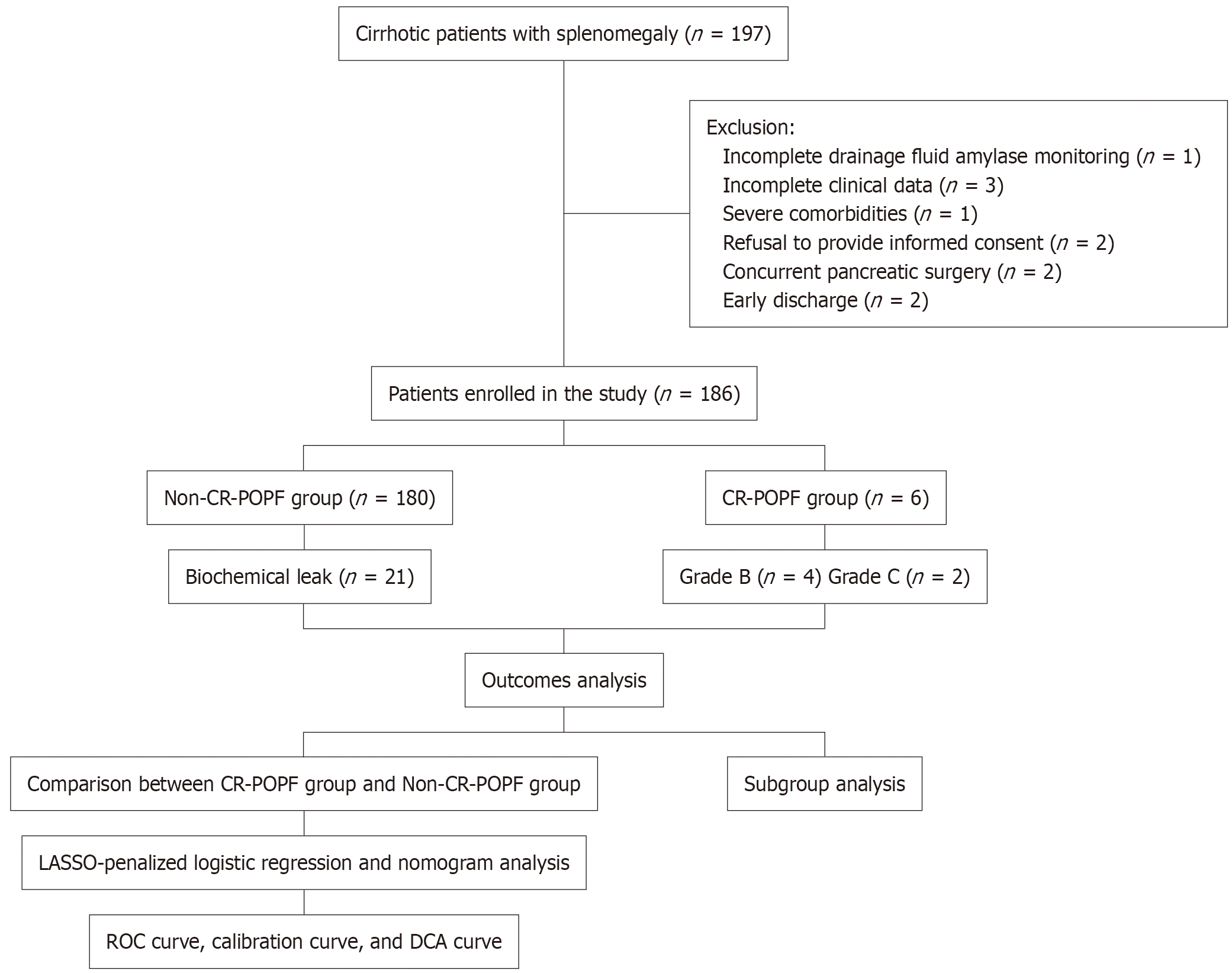
This retrospective study consecutively enrolled patients who underwent splenectomy for cirrhotic splenomegaly with hypersplenism at the First Affiliated Hospital of the Anhui University of Chinese Medicine between January 2019 and December 2024. In total, 197 patients were initially identified during the study period. After applying the strict inclusion and exclusion criteria, 186 patients were finally included in the analysis (Figure 1). Clinical data, including demographic characteristics, laboratory results, surgical details, postoperative outcomes, and complications, were collected from the electronic medical records and surgical databases. All the data were reviewed and verified by two independent re
Inclusion criteria were defined as follows: (1) Definite cirrhosis diagnosis per the European Association for the Study of the Liver Clinical Practice Guidelines[11], confirmed by: Clinical evidence of portal hypertension (splenomegaly, jaundice, ascites, varices, or encephalopathy); biochemical markers: Albumin < 35 g/L, bilirubin > 1.2 mg/dL; imaging: Liver stiffness ≥ 17.5 kPa by FibroTouch or nodular hepatic morphology on contrast-enhanced computed tomography (CT)/magnetic resonance imaging; (2) Presence of splenomegaly with hypersplenism: Ultrasound-proven splenic length > 13 cm or splenic thickness > 4 cm; cytopenia (platelets < 100 × 109/L, white blood cells < 3 × 109/L); clinical signs of portal hypertension, such as splenomegaly, jaundice, ascites, variceal bleeding history; (3) Underwent splenectomy; (4) Postoperative monitoring of drainage fluid amylase levels on days 2-3 after surgery; and (5) All patients, particularly adolescents aged ≤ 18 years, underwent hematology consultation and bone marrow aspiration to exclude hematological causes of splenomegaly before enrollment.
Exclusion criteria included: (1) Incomplete postoperative monitoring of drainage fluid amylase; (2) Incomplete clinical data (missing more than two key parameters); (3) Concurrent pancreatic surgery during splenectomy; (4) Severe co
Prior to commencing this study, the objectives and specific details were comprehensively explained to the participants, and informed consent was obtained from each participant or their respective representatives. This study was conducted in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki, revised in Fortaleza, Brazil, in October 2013). The study protocol was approved by the Institutional Review Board of the First Affiliated Hospital of Anhui University of Chinese Medicine, No. 2024AH-30. Patient data were de-identified and stored in a password-protected database compliant with the Health Insurance Portability and Accountability Act using Research Electronic Data Capture for secure data management (the raw data were shown in the Supplementary material).
All surgeries were performed by a single surgical team consisting of six experienced surgeons to ensure consistency in the operative techniques. Preoperative evaluations within 1 week before surgery included: (1) General lifestyle factors and medical conditions include smoking, alcohol consumption, hypertension, coronary artery disease, diabetes, dyslipidemia, body mass index (BMI), and American Society of Anesthesiologists Physical Status Classification (ASA classification); (2) Liver function assessment using the Child-Pugh classification system (class A, B, or C); (3) Routine hematological indices, including complete blood count (white blood cells, red blood cells, hemoglobin, and platelets), liver function tests (ala
Surgical procedures included splenectomy alone or in combination with periesophagogastric devascularisation. The postoperative diagnosis of PF was based on the 2016 ISGPS criteria: Grade A (biochemical leak), drainage fluid amylase > 3 times the upper limit of normal serum amylase without clinical symptoms and requiring no specific intervention. Grade B: Drainage fluid amylase > 3 times the upper limit of normal serum amylase, accompanied by prolonged drainage (> 3 weeks), clinical signs of infection (fever, leukocytosis), or need for imaging-guided intervention (percutaneous drainage). Grade C: Severe PF requiring reoperation, organ failure, or life-threatening complications (massive hemorrhage or sepsis). CR-POPF was defined as grade B or C.
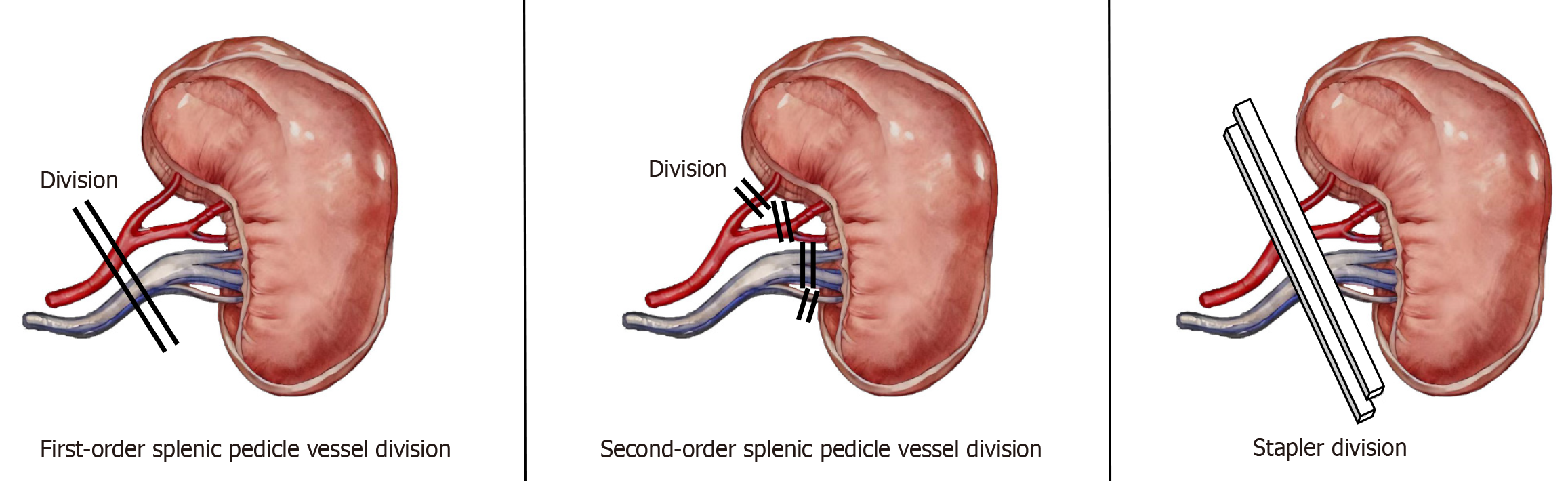
First-order splenic pedicle vessel division: The primary splenic pedicle was dissected, ligated, and sutured after dis
Second-order splenic pedicle vessel division: The serosa tissue in front of the secondary splenic pedicle space was separated and dissected after the bifurcation of the splenic pedicle was clinically determined; the secondary splenic pedicle or tertiary splenic pedicle space could be clearly visualized. Every secondary splenic pedicle vessel or tertiary splenic pedicle vessel was dissected and ligated under direct vision (Figure 2).
Stapler division: This indicates whether a surgical stapler is used to cut and seal the splenic pedicle vessels during the procedure (Figure 2).
Pancreatic texture: The pancreatic texture was evaluated intra-operatively by the surgeon via palpation, categorized as “soft”, “firm”, or “hard”. The hard texture was attributed to underlying chronic pancreatitis or pancreatic fibrosis. A soft texture is a normal physiological texture or a manifestation of pancreatic steatosis, and “firm” refers to a moderate consistency between soft and hard (Figure 2).
Blood loss: Calculated as the sum of the blood collected by the suction devices and the estimated amount of blood absorbed by the surgical gauze (1 g gauze = 1 mL blood) (Figure 2).
All statistical analyses were performed using the R software (version 4.5.1, R Foundation for Statistical Computing, Vienna, Austria).
Missing data: Missing values with < 5% missingness were handled using multiple imputations with ten imputed datasets via the mice package. Cases with > 5% of missing data were excluded from the analysis.
Outlier detection: Outliers in continuous variables were identified using the interquartile range (IQR) method (values > 1.5 × IQR above the 75th percentile or below the 25th percentile) and verified by visual inspection of boxplots. Outliers were retained after the confirmation of data accuracy.
Normality assessment: The normality of continuous variables was tested using the Shapiro-Wilk test. Normally dis
Group comparisons: Categorical variables were compared using the χ2 test (via χ2 test in the base package) or Fisher’s exact test (via Fisher’s test in the base package), as appropriate. Continuous variables were compared using the in
Risk factor analysis: Univariate and multivariate least absolute shrinkage and selection operator (LASSO)-penalized logistic regression analyses were performed using the glm function (base statistics package) and the glmnet function (glmnet package). Variables with a P-value < 0.05 in univariate analysis were included in the multivariate model. Additionally, the Bootstrap internal validation method was used to reduce optimistic bias of the reported area under the receiver operating characteristic (ROC) curve (AUC). Correlation analysis heatmaps were generated using R software (pheatmap package) to analyze key variables from the univariate analysis, and variables to be included in the mul
Nomogram construction: A nomogram was developed based on the multivariate LASSO-penalized logistic regression model using the nomogram function in the rms package. The nomogram was evaluated using the AUC, calibration curves, and decision curve analysis (DCA) using pROC, dplyr, and ggplot2 packages. Model performance was evaluated using the concordance index and calibration curves using the rms package. Statistical significance was defined as a two-tailed P-value of < 0.05.
Among the 186 patients included in the study, the mean age was 35.48 ± 14.25 years, with a range from 11 years to 78 years. The study included 106 male patients and 80 female patients. In terms of liver function, 145 patients were classified as Child-Pugh class A, 37 as class B, and four as class C. The mean splenic length was 188.08 ± 26.93 mm, the mean splenic thickness was 63.95 ± 11.21 mm, the mean portal vein diameter was 13.56 ± 2.17 mm, and the mean splenic vein diameter was 13.40 ± 2.53 mm.
This study identified 6 patients with CR-POPF and 180 without CR-POPF. In terms of demographic characteristics, there were no significant differences in age, sex, alcohol consumption, smoking, hypertension, coronary artery disease, dyslipidemia, diabetes, ASA classification, and Child-Pugh distribution between the two groups (P > 0.05). The CR-POPF group exhibited a significantly higher BMI than the non-CR-POPF group [26.9 (24.33, 28.53) kg/m2 vs 22.00 (20.30, 23.20) kg/m2, P < 0.05]. PT in the CR-POPF group was significantly lower than that in the non-CR-POPF group [11.75 (10.93, 12.20) seconds vs 13.50 (12.50, 14.88) seconds, P < 0.05]. Statistically significant differences were observed in splenic vein diameter [15 (15, 16.25) mm vs 13.00 (11.00, 15.00) mm] and splenic thickness (75.67 ± 8.55 mm vs 63.56 ± 11.10 mm) between the two groups (P < 0.05). The use of stapler divisions differed between the two groups, with a higher percentage of staplers used in the CR-POPF group (P < 0.05). The pancreatic texture also differed between the two groups (P < 0.05). In addition, the hospital stay was significantly longer in the CR-POPF group [14 (11.75, 33.25) days] than in the non-CR-POPF group [14.00 (12.00, 17.00) days, P < 0.05] (Table 1).
| Variables | CR-POPF group (n = 6) | Non-CR-POPF group (n = 180) | P value |
| Age (years) | 33.17 ± 21.78 | 35.56 ± 14.01 | 0.687 |
| Gender (male/female) | 0.701 | ||
| Male | 4 | 102 | |
| Female | 2 | 78 | |
| Alcohol consumption | 0.401 | ||
| Yes | 1 | 14 | |
| No | 5 | 166 | |
| Smoking | 0.185 | ||
| Yes | 2 | 23 | |
| No | 4 | 157 | |
| Hypertension | 0.552 | ||
| Yes | 1 | 22 | |
| No | 5 | 158 | |
| Coronary artery disease | 0.310 | ||
| Yes | 1 | 10 | |
| No | 5 | 170 | |
| Dyslipidemia | 0.518 | ||
| Yes | 1 | 20 | |
| No | 5 | 160 | |
| Diabetes | 0.249 | ||
| Yes | 2 | 28 | |
| No | 4 | 152 | |
| BMI (kg/m2) | 26.90 (24.33, 28.53) | 22.00 (20.30, 23.20) | 0 |
| ASA classification | 0.150 | ||
| I | 3 | 131 | |
| II | 2 | 39 | |
| III | 1 | 10 | |
| WBC (× 109/L) | 2.29 (1.27, 3.64) | 2.35 (1.72, 3.02) | 0.884 |
| RBC (× 1012/L) | 3.67 ± 0.65 | 3.87 ± 0.59 | 0.470 |
| HGB (g/L) | 102.50 ± 10.62 | 109.31 ± 18.80 | 0.379 |
| PLT (× 109/L) | 43.50 (33.00, 65.50) | 41.50 (32.00, 53.75) | 0.522 |
| ALT (U/L) | 26.40 (17.75, 45.50) | 25.05 (19, 33.95) | 0.920 |
| AST (U/L) | 26.50 (17.50, 42.50) | 28.50 (21, 36.75) | 0.934 |
| TB (μmol/L) | 14.63 (8.88, 17.85) | 17.90 (12.88, 27.23) | 0.123 |
| ALB (g/L) | 35.95 (30.25, 40.43) | 36.20 (32.90, 39.60) | 0.710 |
| PT (seconds) | 11.75 (10.93, 12.20) | 13.50 (12.50, 14.88) | 0.020 |
| INR | 1.09 (1.02, 1.18) | 1.23 (1.11, 1.36) | 0.067 |
| APTT (seconds) | 30.45 (27.73, 32.63) | 34.75 (31.70, 38.28) | 0.078 |
| D-Dimer (mg/L FEU) | 0.31 (0.19, 0.57) | 0.24 (0.19, 0.59) | 0.611 |
| Child-Pugh classification | 0.910 | ||
| A | 5 | 140 | |
| B | 1 | 36 | |
| C | 0 | 4 | |
| Ascite | 0.206 | ||
| No ascites | 3 | 132 | |
| Mild ascites | 2 | 24 | |
| Moderate to severe ascites | 1 | 24 | |
| Splenic length (mm) | 203.17 ± 7.41 | 187.58 ± 27.20 | 0.164 |
| Splenic thickness (mm) | 75.67 ± 8.55 | 63.56 ± 11.10 | 0.009 |
| Portal vein diameter (mm) | 14.00 (13.00, 15.50) | 13.00 (12.00, 15.00) | 0.380 |
| Splenic vein diameter (mm) | 15.00 (15.00, 16.25) | 13.00 (11.00, 15.00) | 0.038 |
| Main pancreatic duct diameter (mm) | 2.25 (1.85, 4.28) | 2.70 (2.20, 3.10) | 0.959 |
| Pancreatic CT value (HU) | 34.00 (30.25, 41.25) | 35.00 (29.25, 39.00) | 0.709 |
| Surgical method | 0.917 | ||
| Splenectomy | 5 | 147 | |
| Devascularization | 1 | 33 | |
| Splenic pedicle vessel division method | 0.092 | ||
| First-order splenic pedicle vessel division | 5 | 79 | |
| Second-order splenic pedicle vessel division | 1 | 101 | |
| Stapler division | 0.018 | ||
| Yes | 5 | 58 | |
| No | 1 | 122 | |
| Pancreatic texture | 0.048 | ||
| Soft | 4 | 87 | |
| Firm | 1 | 97 | |
| Hard | 1 | 2 | |
| Drainage fluid amylase | 20762.50 (10877.25, 42335.75) | 216.00 (100.00, 441.25) | 0 |
| Hospital stay (days) | 14.00 (11.75, 33.25) | 14.00 (12.00, 17.00) | 0.041 |
| Blood loss (mL) | 395.00 (290.00, 450.00) | 230.00 (200.00, 350.00) | 0.144 |
Of the 21 patients were classified as having biochemical leak, four as grade B, and two as grade C, according to the 2016 ISGPS criteria. Comparisons of clinical characteristics among the three grades revealed no significant differences in age, sex, alcohol consumption, smoking, hypertension, coronary artery disease, dyslipidemia, diabetes, ASA classification, or most laboratory indices, including white blood cells, hemoglobin, liver function parameters (alanine ami
| Variables | Biochemical leak (n = 21) | Grade B (n = 4) | Grade C (n = 2) | P value |
| Age (years, mean ± SD) | 34.19 ± 12.76 | 29.25 ± 20.85 | 41 ± 29.70 | 0.664 |
| Gender (male/female) | 0.648 | |||
| Male | 9 | 3 | 1 | |
| Female | 12 | 1 | 1 | |
| Alcohol consumption | 0.545 | |||
| Yes | 2 | 1 | 0 | |
| No | 19 | 3 | 2 | |
| Smoking | 0.813 | |||
| Yes | 9 | 1 | 1 | |
| No | 12 | 3 | 1 | |
| Hypertension | 0.338 | |||
| Yes | 7 | 0 | 1 | |
| No | 14 | 4 | 1 | |
| Coronary artery disease | 0.545 | |||
| Yes | 2 | 1 | 0 | |
| No | 19 | 3 | 2 | |
| Dyslipidemia | 0.752 | |||
| Yes | 4 | 1 | 0 | |
| No | 17 | 3 | 2 | |
| Diabetes | 0.542 | |||
| Yes | 4 | 1 | 1 | |
| No | 17 | 4 | 1 | |
| BMI (kg/m2) | 22.60 (21.15, 23.20) | 26.90 (24.95, 28.78) | 26.05 (23.80, 26.05) | 0 |
| ASA | 0.677 | |||
| I | 14 | 2 | 1 | |
| II | 5 | 1 | 1 | |
| III | 2 | 1 | 0 | |
| WBC (× 109/L) | 2.67 (2.05, 3.94) | 1.67 (1.24, 2.41) | 4.04 (3.24, 4.04) | 0.190 |
| RBC (× 1012/L) | 3.89 ± 0.59 | 3.33 ± 0.15 | 4.42 ± 0.70 | 0.082 |
| HGB (g/L) | 112.05 ± 19.60 | 98.25 ± 5.91 | 111 ± 15.56 | 0.397 |
| PLT (× 109/L) | 51.00 (32.00, 55.50) | 49.50 (34.00, 72.50) | 41.50 (33.00, 41.50) | 0.760 |
| ALT (U/L) | 24.00 (16.80, 38.00) | 26.40 (19.75, 53.45) | 28.50 (17.00, 28.50) | 0.962 |
| AST (U/L) | 28.00 (21.00, 35.50) | 31.00 (19.25, 49.50) | 23.00 (16.00, 23.00) | 0.570 |
| TB (μmol/L) | 16.20 (13.05, 31.17) | 11.60 (7.03, 15.25) | 18.70 (17.00, 18.70) | 0.392 |
| ALB (g/L) | 38.90 (33.85, 40.75) | 31.95 (29.15, 37.68) | 41.45 (38.80, 41.45) | 0.259 |
| PT (seconds) | 13.30 (12.00, 13.75) | 11.80 (10.48, 12.60) | 11.55 (11.20, 11.55) | 0.155 |
| INR | 1.16 (1.05, 1.26) | 1.14 (0.98, 1.21) | 1.06 (1.04, 1.06) | 0.423 |
| APTT (seconds) | 35.20 (31.80, 37.80) | 30.05 (26.78, 33.40) | 30.60 (29.00, 30.60) | 0.061 |
| D-Dimer (mg/L FEU) | 0.23 (0.19, 0.56) | 0.24 (0.19, 0.79) | 0.38 (0.32, 0.38) | 0.872 |
| Child-Pugh classification | 0.752 | |||
| A | 17 | 3 | 2 | |
| B | 4 | 1 | 0 | |
| C | 0 | 0 | ||
| Ascite | 0.227 | |||
| No ascites | 17 | 2 | 1 | |
| Mild ascites | 2 | 1 | 1 | |
| Moderate to severe ascites | 2 | 1 | 0 | |
| Splenic length (mm) | 188.62 ± 19.74 | 207 ± 5.72 | 195.5 ± 0.71 | 0.192 |
| Splenic thickness (mm) | 67.71 ± 11.89 | 76 ± 2.94 | 75 ± 18.38 | 0.344 |
| Portal vein diameter (mm) | 15.00 (13.00, 16.00) | 14.00 (13.25, 14.75) | 15.00 (13.00, 15.00) | 0.773 |
| Splenic vein diameter (mm) | 15.00 (14.00, 16.00) | 15.00 (15.00, 16.50) | 15.50 (15.00, 15.50) | 0.828 |
| Main pancreatic duct diameter (mm) | 2.80 (2.05, 3.05) | 2.25 (1.93, 3.70) | 3.25 (1.70, 3.25) | 0.742 |
| Pancreatic CT value (HU) | 32.00 (27.50, 35.00) | 32.50 (28.75, 37.75) | 41.00 (34.00, 41.00) | 0.093 |
| Surgical method | 0.162 | |||
| Splenectomy | 20 | 4 | 1 | |
| Devascularization | 1 | 0 | 1 | |
| Splenic pedicle vessel division method | 0.733 | |||
| First-order splenic pedicle vessel division | 16 | 3 | 2 | |
| Second-order splenic pedicle vessel division | 5 | 1 | 0 | |
| Stapler division | 0.497 | |||
| Yes | 12 | 3 | 2 | |
| No | 9 | 1 | 0 | |
| Pancreatic texture | 0.008 | |||
| Soft | 16 | 3 | 0 | |
| Firm | 5 | 1 | 0 | |
| Hard | 0 | 0 | 2 | |
| Drainage fluid amylase | 1485.00 (1294.50, 1961.00) | 12197.50 (10377.75, 24509.75) | 54671.50 (30000.00, 54671.50) | 0 |
| Hospital stay (days) | 14.00 (12.00, 16.50) | 22.50 (11.25, 33.75) | 14.00 (14.00, 14.00) | 0.079 |
| Blood loss (mL) | 340.00 (300.00, 372.50) | 380.00 (230.00, 447.50) | 400.00 (350.00, 400.00) | 0.460 |
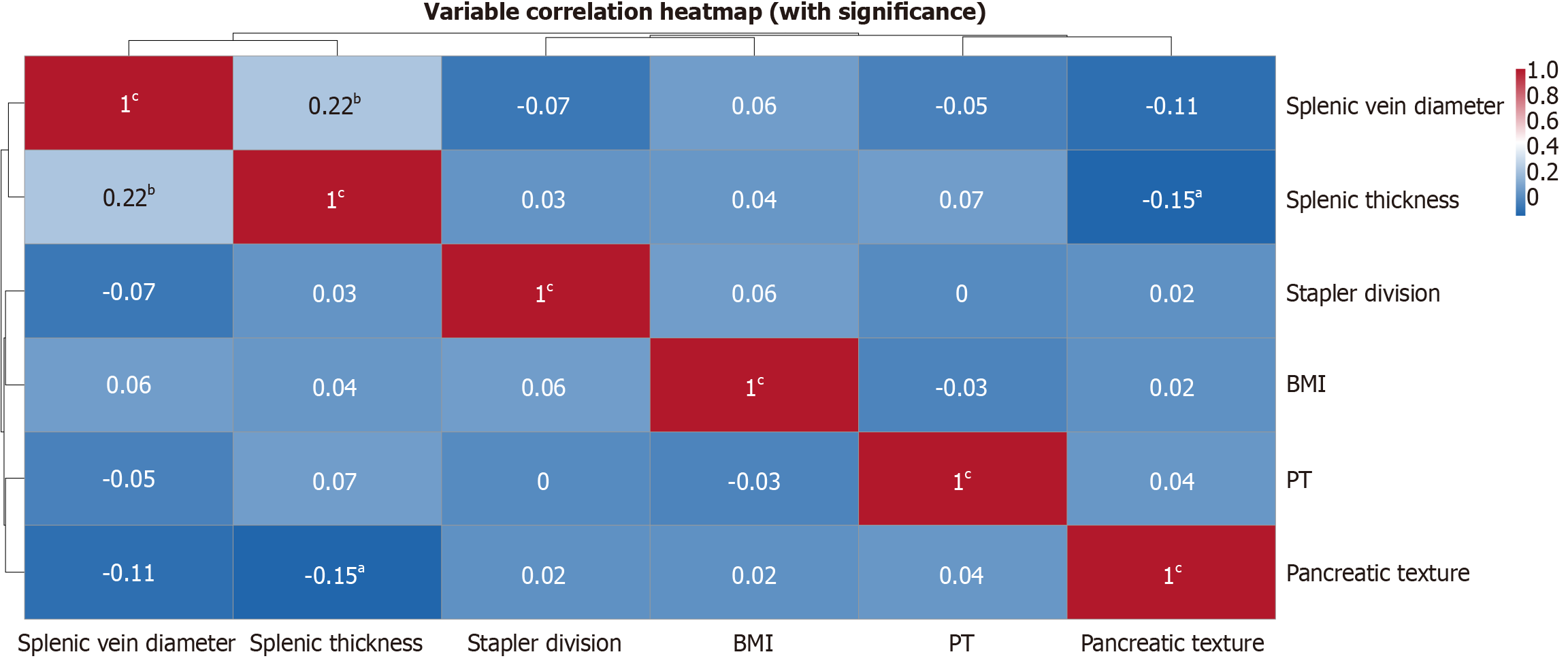
A univariate logistic regression analysis was performed to identify several potential risk factors for PF. Variables including BMI, PT, splenic thickness, splenic vein diameter, stapler division, and pancreatic texture showed significant differences in univariate analysis (P < 0.05). The correlation heatmap revealed significant collinearity among splenic thickness, splenic vein diameter, and pancreatic texture (Figure 3). Notably, splenic thickness showed a more significant difference (P = 0.009) between the two groups than the splenic vein diameter (P = 0.038) and pancreatic texture (P = 0.048). Furthermore, the total number of CR-POPF events (grades B/C) reported in the manuscript was substantially lower than the number of predictors that were initially planned for the multivariate model. To mitigate predictor re
| Variables | B | SE | Wald | df | Significance | Exp (B) | Lower 95%CI | Upper 95%CI |
| BMI | 1.275 | 0.577 | 4.887 | 1 | 0.027 | 3.578 | 1.156 | 11.080 |
| PT | -2.531 | 1.482 | 2.916 | 1 | 0.088 | 0.080 | 0.004 | 1.453 |
| Splenic thickness | 0.223 | 0.107 | 4.352 | 1 | 0.037 | 1.250 | 1.014 | 1.542 |
| Stapler division | 1.155 | 1.493 | 0.549 | 1 | 0.439 | 3.175 | 0.170 | 59.227 |
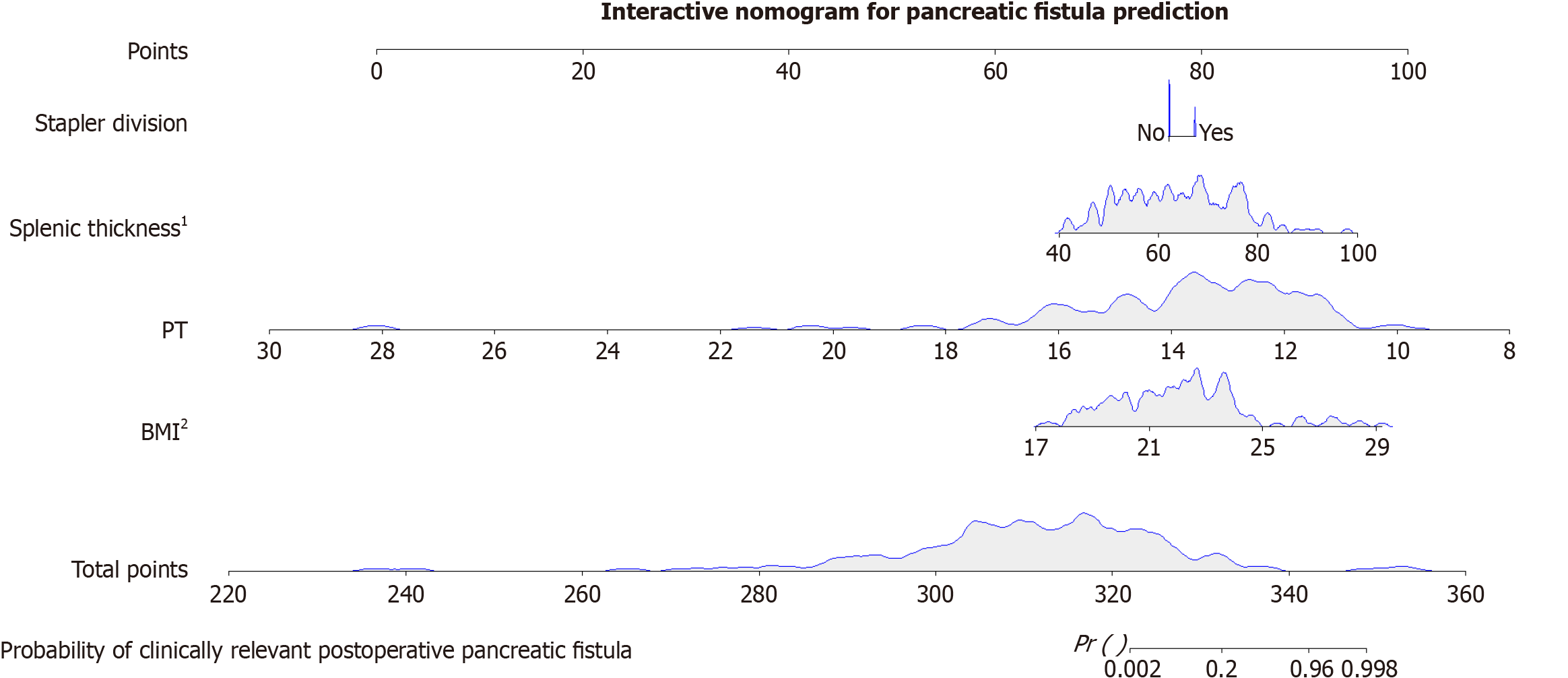
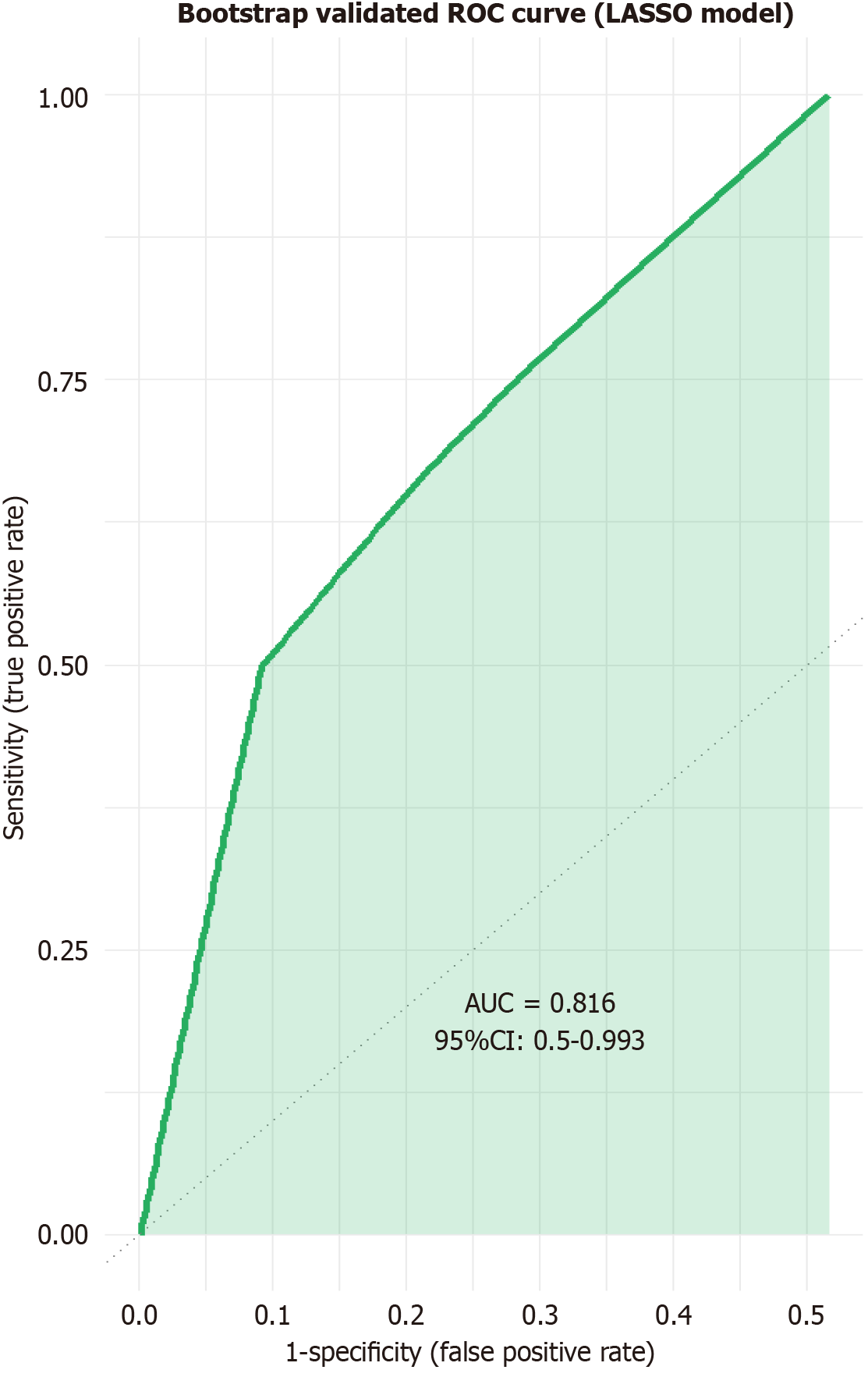
A nomogram was constructed to visually quantify the risk of CR-POPF based on LASSO-penalized logistic regression, with the optimal LASSO penalty coefficient determined as 0.0086 via 10-fold cross-validation, integrating the four independent risk factors identified: Stapler division, splenic thickness, PT, and BMI (Figure 4). The concordance index of the nomogram was 0.816 with a 95%CI: 0.500-0.993, further validating its robust predictive performance. To evaluate the discriminative efficacy of the nomogram in distinguishing between patients with and without CR-POPF, we plotted an ROC curve. Given that the nomogram was constructed based on LASSO-penalized logistic regression, 1000 bootstrap internal validations were performed to calibrate the AUC and reduce optimism bias. The AUC was 0.816 (consistent with the model performance), indicating excellent discriminative power, with a corresponding maximum Youden’s index of 0.903 (Figure 5).
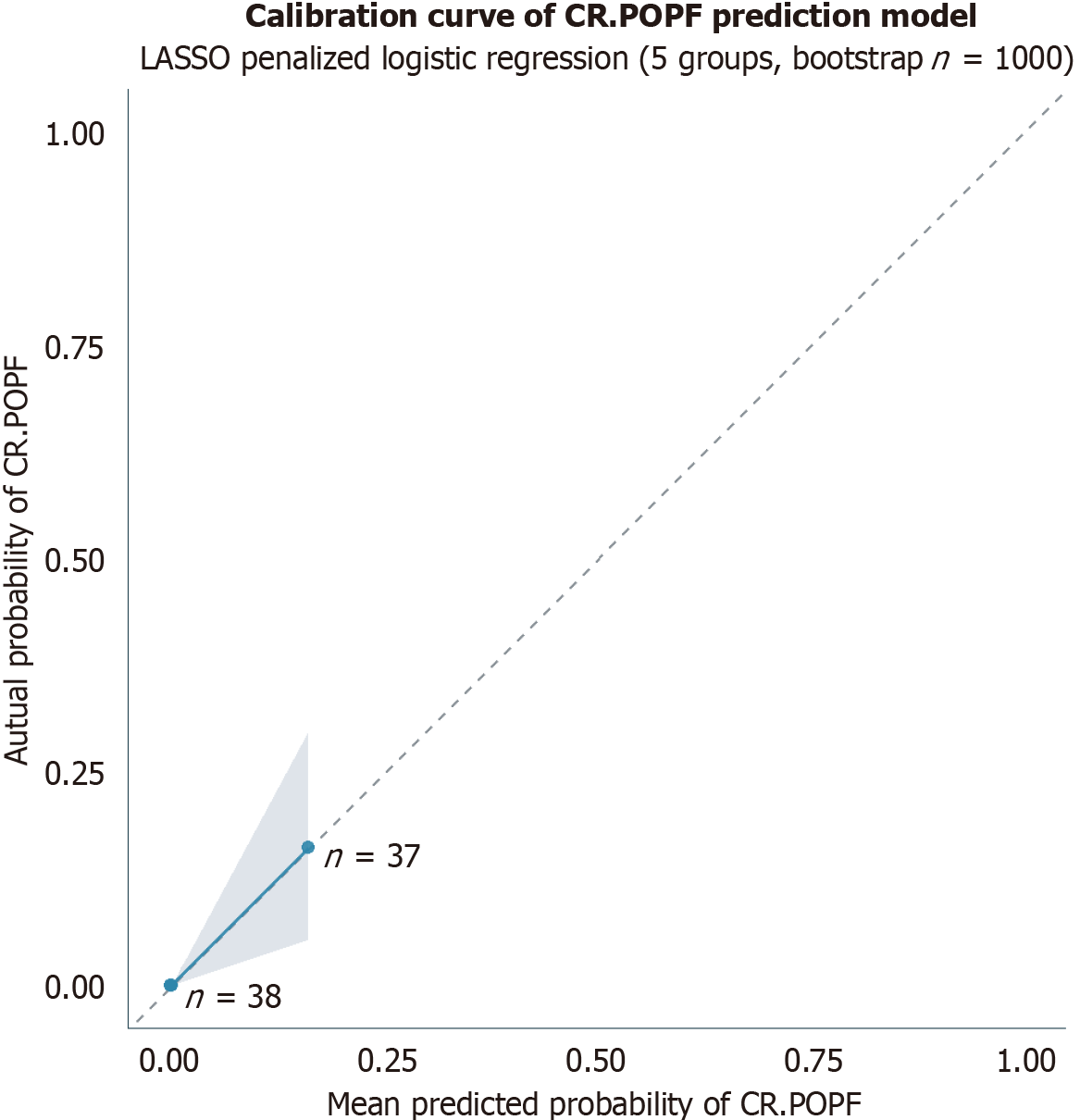
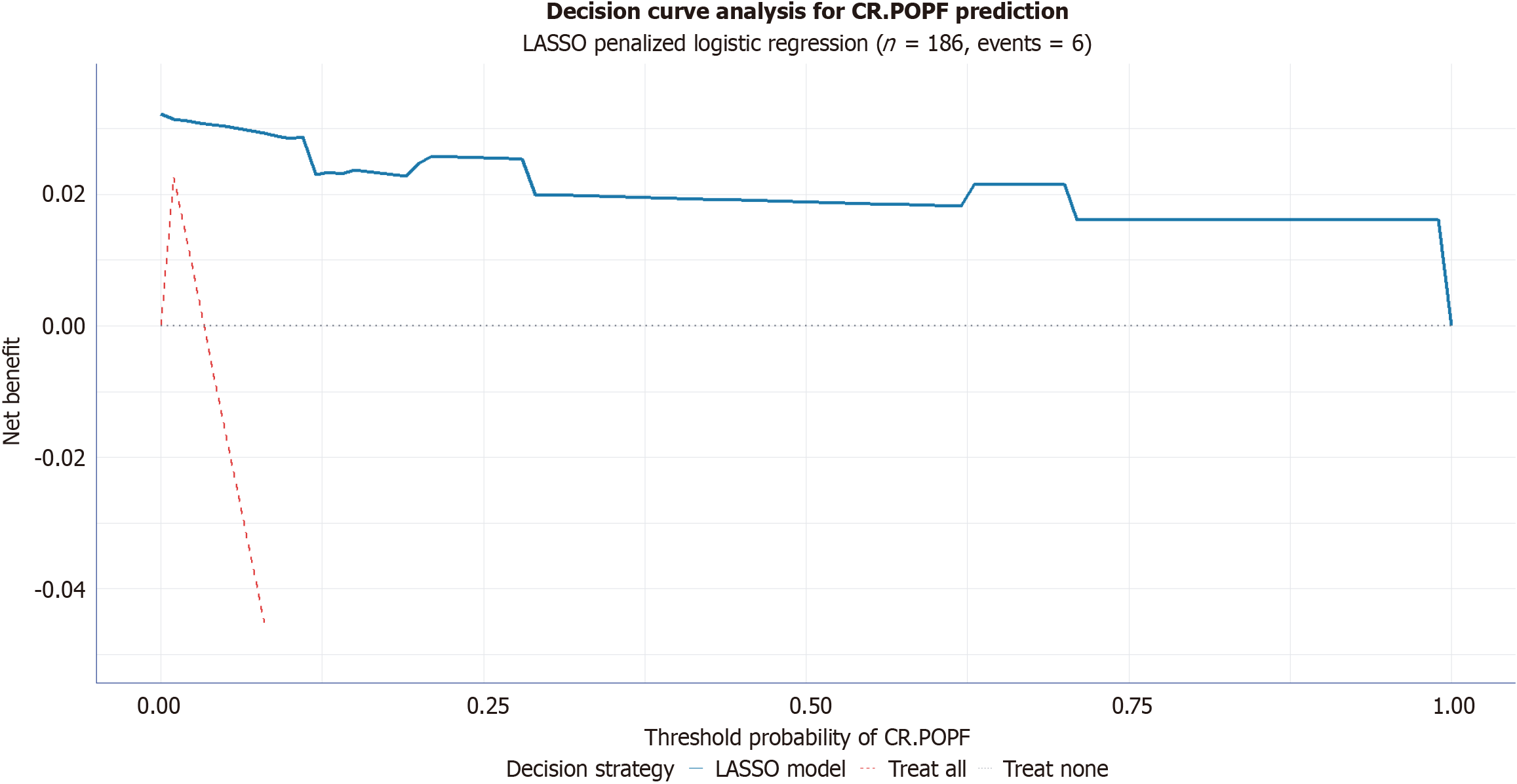
The calibration of the nomogram was evaluated using calibration curves with 1000 bootstrap resamples to correct for potential overfitting. The calibration curve showed good agreement between the predicted probabilities of CR-POPF and actual occurrence probabilities. The Hosmer-Lemeshow test indicated that the model had excellent calibration (Hosmer-Lemeshow statistic = 0.027, degrees of freedom = 3, P = 0.999 > 0.05), demonstrating no significant discrepancy between the predicted probabilities and the actual outcomes (Figure 6). DCA was performed to assess the clinical utility of the nomogram. The DCA curve showed that when the threshold probability of CR-POPF ranged from approximately 0% to 99%, the nomogram provided a positive net benefit that was superior to both the “treat all” and “treat none” strategies (Figure 7). This indicates that the nomogram can effectively assist clinicians in identifying high-risk patients who may benefit from targeted preventive interventions, thereby optimizing clinical decision-making.
CR-POPF (grade B/C) remains a significant complication following splenectomy, contributing to prolonged hospital stays, increased healthcare costs, and, in severe cases, life-threatening outcomes such as sepsis and multi-organ failure[12]. Our study aimed to identify independent risk factors for CR-POPF after splenectomy in patients with cirrhotic splenomegaly and hypersplenism, and to develop a clinically useful prediction tool. These findings provide novel insights into the pathogenesis of CR-POPF and offer practical guidance for its risk stratification and prevention. Uni
Furthermore, univariate analysis revealed several other variables associated with CR-POPF development, providing supplementary insights into the complex interplay between patient and surgical factors influencing CR-POPF risk. The PT [11.75 (10.93, 12.20) seconds vs 13.50 (12.50, 14.88) seconds, P = 0.020] was lower in the CR-POPF group. These findings may reflect altered coagulation dynamics in patients with cirrhosis and portal hypertension, potentially affecting pancreatic tissue perfusion and healing. Splenic thickness was greater in the CR-POPF group (75.67 ± 8.55 mm vs 63.56 ± 11.10 mm, P = 0.009), indicating that more severe splenomegaly might distort the peripancreatic anatomy, increasing the risk of iatrogenic pancreatic injury during dissection. BMI was significantly higher in the CR-POPF group [26.9 (24.33, 28.53) kg/m2 vs 22.00 (20.30, 23.20) kg/m2, P = 0.000], which could be attributed to increased technical difficulty in surgical exposure due to adipose tissue, potentially elevating trauma risk to adjacent pancreatic structures. The association of these variables with CR-POPF in the univariate and multivariate analyses highlights the multifaceted nature of CR-POPF pathogenesis, emphasizing the need for a comprehensive perioperative assessment of both patient characteristics and surgical variables.
The nomogram developed from these four risk factors, including BMI, PT, splenic thickness, and stapler division, provides a practical tool for individualized CR-POPF risk prediction. Its excellent discriminative ability (AUC = 0.816, 95%CI: 0.500-0.993), high calibration accuracy (Hosmer-Lemeshow statistic = 0.027, degrees of freedom = 3, P = 0.999 > 0.05), and positive net benefit across a broad range of threshold probabilities via DCA validated its clinical value. By integrating easily measurable variables (BMI and PT) and intra-operative observations (splenic thickness and stapler division), the nomogram allows surgeons to assess CR-POPF risk preoperatively or intra-operatively, enabling timely preventive measures[19]. High-risk patients could benefit from enhanced intraoperative care, including meticulous dissection near the pancreatic tail and cautious use of staplers, or postoperative monitoring with more frequent amylase testing in drainage fluid to detect early CR-POPF and initiate interventions, such as administration of pancreatic secretion inhibitors or targeted drainage.
The nomogram developed based on multivariate LASSO-penalized logistic regression results provides a valuable tool for predicting the risk of CR-POPF. Similar nomogram-based prediction models have been successfully used in other surgical fields to identify high-risk patients early[20]. Using our nomogram, clinicians could estimate the individual risk of CR-POPF in each patient before and during surgery. This early prediction is crucial as it allows for the timely implementation of preventive measures. Early monitoring of amylase in drainage fluid, as emphasized in international guidelines, can help detect CR-POPF at an early stage. Additionally, the early administration of appropriate medications, such as pancreatic secretion inhibitors, can potentially reduce the severity of CR-POPF. Previous studies have shown that timely intervention based on accurate risk prediction can significantly improve patient outcomes.
Our findings expand on prior research by identifying BMI and splenic thickness as novel risk factors for CR-POPF after splenectomy, which are not widely reported in the literature[21]. This differs from previous studies that merely emphasized “obesity” or “splenic volume” as qualitative risk factors[22,23]. Notably, BMI cannot be simply equated with obesity, which may be attributed to the specific pathophysiological characteristics of cirrhotic patients, such as latent edema and abnormal intra-abdominal fat distribution caused by portal hypertension; thus, BMI is a more sensitive quantitative indicator than the qualitative description of “obesity” in this population[24]. Splenic thickness and splenic volume are correlated, but not interchangeable. Previous studies have mostly focused on splenic volume; however, its clinical application is limited by complex measurements and high medical costs. In contrast, splenic thickness can be quickly obtained using preoperative ultrasound. While previous studies highlighted obesity, liver function, or surgical technique as potential risk factors, our multivariate analysis did not confirm Child-Pugh class as an independent predictor, likely because the specific cohort of cirrhotic patients exhibits portal hypertension-related anatomical changes such as splenic vein dilation, which may play a more direct role in CR-POPF pathogenesis than liver function alone[24]. Additionally, our focus on the stapler division provides actionable insights for intra-operative decision making, complementing prior work that emphasizes patient-related factors.
Currently, the treatment of CR-POPF mainly involves continuous drainage, antibiotic therapy, and the use of pancreatic secretion inhibitors[25]. In our experience, continuous drainage remains the cornerstone of CR-POPF treatment[26,27]. For most patients with biochemical leakage, no special treatment is required, and amylase levels in the drainage fluid return to normal within 7-10 days of splenectomy. For CR-POPF (grades B/C), while the effect of somatostatin remains controversial, our cases showed that after 2-3 weeks of drainage, the amylase level in the drainage fluid de
However, this study had several limitations. First, as this was a single-center retrospective study, the findings may be limited by institutional-specific surgical techniques and patient demographics, which reduce generalizability. Multicenter prospective studies with larger sample sizes are required to validate our nomogram. Second, the total number of CR-POPF events in this study was small, while the number of predictors was relatively large (events per variable < 10), which may lead to unstable model coefficients. Although penalized logistic regression was used to mitigate overfitting caused by excessive predictors, bootstrap internal validation was performed to calibrate that AUC optimism bias, residual bias, or instability in the results may still exist. Third, the definition of “soft, firm, and hard” pancreatic texture was based on intra-operative palpation (a standard clinical practice). Although the pancreatic CT values were integrated for supplementary assessment, there was no accurate correspondence between the two. Therefore, the development of histopathological or imaging-derived markers for pancreatic fibrosis may further optimize the risk stratification. Despite these limitations, the study identified BMI and splenic thickness as independent risk factors for CR-POPF in patients with cirrhotic splenomegaly and provided a practical nomogram, which has important clinical guiding value.
In summary, this study identified BMI and splenic thickness as independent risk factors of CR-POPF after splenectomy in patients with cirrhosis. The nomogram effectively predicted the individual CR-POPF risk, offering a valuable tool for personalized perioperative management. By implementing targeted preventive strategies based on risk assessments, clinicians can reduce the incidence of CR-POPF and improve patient outcomes.
| 1. | Kawaida H, Kono H, Hosomura N, Amemiya H, Itakura J, Fujii H, Ichikawa D. Surgical techniques and postoperative management to prevent postoperative pancreatic fistula after pancreatic surgery. World J Gastroenterol. 2019;25:3722-3737. [PubMed] [DOI] [Full Text] |
| 2. | Kang WS, Park YC, Jo YG, Kim JC. Pancreatic fistula and mortality after surgical management of pancreatic trauma: analysis of 81 consecutive patients during 11 years at a Korean trauma center. Ann Surg Treat Res. 2018;95:29-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 3. | Dronov OI, Kovalska IO, Horlach AI, Shchyhel IA. Prediction of external pancreatic fistula development in patients with acute infected necrotising pancreatitis. Wiad Lek. 2023;76:2365-2371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 4. | Fukada M, Murase K, Higashi T, Yasufuku I, Sato Y, Tajima JY, Kiyama S, Tanaka Y, Okumura N, Takahashi T, Matsuhashi N. Drain fluid and serum amylase concentration ratio is the most reliable indicator for predicting postoperative pancreatic fistula after distal pancreatectomy. BMC Surg. 2023;23:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 5. | Mehdorn AS, Schwieters AK, Mardin WA, Senninger N, Strücker B, Pascher A, Vowinkel T, Becker F. Pancreatic Fistula and Biochemical Leak after Splenectomy: Incidence and Risk Factors-A Retrospective Single-Center Analysis. Langenbecks Arch Surg. 2022;407:2517-2525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 6. | Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, Allen P, Andersson R, Asbun HJ, Besselink MG, Conlon K, Del Chiaro M, Falconi M, Fernandez-Cruz L, Fernandez-Del Castillo C, Fingerhut A, Friess H, Gouma DJ, Hackert T, Izbicki J, Lillemoe KD, Neoptolemos JP, Olah A, Schulick R, Shrikhande SV, Takada T, Takaori K, Traverso W, Vollmer CM, Wolfgang CL, Yeo CJ, Salvia R, Buchler M; International Study Group on Pancreatic Surgery (ISGPS). The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery. 2017;161:584-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3570] [Cited by in RCA: 3303] [Article Influence: 367.0] [Reference Citation Analysis (4)] |
| 7. | Furukawa K, Gocho T, Shirai Y, Iwase R, Haruki K, Fujiwara Y, Shiba H, Misawa T, Yanaga K. The Decline of Amylase Level of Pancreatic Juice After Pancreaticoduodenectomy Predicts Postoperative Pancreatic Fistula. Pancreas. 2016;45:1474-1477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 8. | Tian G, Li D, Yu H, Dong Y, Xue H. Splenic Bed Laparoscopic Splenectomy Approach for Massive Splenomegaly Secondary to Portal Hypertension and Liver Cirrhosis. Am Surg. 2018;84:1033-1038. [PubMed] |
| 9. | Guo CX, Shen YN, Zhang Q, Zhang XZ, Wang JL, Gao SL, Lou JY, Que RS, Ma T, Liang TB, Bai XL. Prediction of postoperative pancreatic fistula using a nomogram based on the updated definition. Ann Surg Treat Res. 2020;98:72-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 10. | Ouyang L, Liu RD, Ren YW, Nie G, He TL, Li G, Zhou YQ, Huang ZP, Zhang YJ, Hu XG, Jin G. Nomogram predicts CR-POPF in open central pancreatectomy patients with benign or low-grade malignant pancreatic neoplasms. Front Oncol. 2022;12:1030080. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (2)] |
| 11. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2259] [Cited by in RCA: 2059] [Article Influence: 257.4] [Reference Citation Analysis (6)] |
| 12. | Bassi C, Dervenis C, Butturini G, Fingerhut A, Yeo C, Izbicki J, Neoptolemos J, Sarr M, Traverso W, Buchler M; International Study Group on Pancreatic Fistula Definition. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138:8-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3690] [Cited by in RCA: 3344] [Article Influence: 159.2] [Reference Citation Analysis (4)] |
| 13. | Zhu X, Zhao Q, Xiong W, Luo L, Zheng Y, Huang H, Li J, Wan J, Xie W, Wang W. Anatomical observation and clinical significance of the left gastric vein in laparoscopic radical gastrectomy. J Gastrointest Oncol. 2021;12:1407-1415. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 14. | Huang L, Yu Q, Peng H, Zhen Z. Postoperative Outcomes Following a Modified Method of Surgical Division of the Splenic Pedicle in 719 Patients During Splenectomy for Portal Hypertension: A 12-Year, Retrospective, Single-Center Study. Med Sci Monit. 2022;28:e937763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 15. | Ausania F, Senra Del Rio P. How to close the pancreatic stump and the role of sealants. Minerva Chir. 2019;74:270-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 16. | Vecchio R, Cacciola E, Intagliata E. Using Stapler in Laparoscopic Splenectomy Does Not Affect the Risk of Pancreatic Tail Injury. J Indian Assoc Pediatr Surg. 2020;25:416-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 17. | Martin AN, Narayanan S, Turrentine FE, Bauer TW, Adams RB, Zaydfudim VM. Pancreatic duct size and gland texture are associated with pancreatic fistula after pancreaticoduodenectomy but not after distal pancreatectomy. PLoS One. 2018;13:e0203841. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (5)] |
| 18. | Teraoku H, Morine Y, Ikemoto T, Saito Y, Yamada S, Waki Y, Okikawa S, Miyazaki K, Nakasu C, Noma T, Takahashi A, Shimada M. Impact of pancreatic resection in patients with liver cirrhosis. J Med Invest. 2023;70:189-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 19. | Huang H, Wang C, Ji F, Han Z, Xu H, Cao M. Nomogram based on albumin and neutrophil-to-lymphocyte ratio for predicting postoperative complications after pancreaticoduodenectomy. Gland Surg. 2021;10:877-891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 20. | Mostafa A, Habeeb TA, Neri V, Elshahidy TM, A Fiad A. Risk factors for postoperative pancreatic fistula following non-traumatic, pancreatic surgery. Retrospective observational study. Ann Ital Chir. 2023;94:435-442. [PubMed] |
| 21. | Peng YP, Zhu XL, Yin LD, Zhu Y, Wei JS, Wu JL, Miao Y. Risk factors of postoperative pancreatic fistula in patients after distal pancreatectomy: a systematic review and meta-analysis. Sci Rep. 2017;7:185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (5)] |
| 22. | Miura Y, Sugiura T, Ashida R, Ohgi K, Kato Y, Otsuka S, Dei H, Uesaka K. Waist-to-Height Ratio as a Predictor Complementary to BMI for Postoperative Pancreatic Fistula After Pancreatoduodenectomy for Periampullary Tumors. Ann Surg Oncol. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (3)] |
| 23. | Abe H, Okamura Y, Yoshida N, Mitsuka Y, Aramaki O, Tago K, Okada M, Nakano N, Masuda S, Udagawa S. Pancreatic extracellular volume fraction on multiphasic contrast-enhanced computed tomography for predicting pancreatic fistula after pancreatoduodenectomy. Surg Today. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 24. | Chong E, Ratnayake B, Lee S, French JJ, Wilson C, Roberts KJ, Loveday BPT, Manas D, Windsor J, White S, Pandanaboyana S. Systematic review and meta-analysis of risk factors of postoperative pancreatic fistula after distal pancreatectomy in the era of 2016 International Study Group pancreatic fistula definition. HPB (Oxford). 2021;23:1139-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 25. | Kuscher S, Kiehl T, Kronberger IE, Moser P, Maier H, Maier S, Hautz T, Öfner D, Schneeberger S, Troppmair J. Distal Pancreatic Resection with Splenectomy in the Rat: A Pancreatic Fistula Model to Investigate Postsurgical Damage? Eur Surg Res. 2021;62:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 26. | Davidson TB, Yaghoobi M, Davidson BR, Gurusamy KS. Amylase in drain fluid for the diagnosis of pancreatic leak in post-pancreatic resection. Cochrane Database Syst Rev. 2017;4:CD012009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 27. | Perri G, Romandini E, Marchegiani G, Ghorbani P, Sahakyan M, Holmberg M, Cattelani A, Fretland Å, Montorsi R, Rodrigues ID, Kleive D, Bannone E, Edwin B, Gilg S, Labori KJ, Sparrelid E, Salvia R. Postoperative Hyperamylasemia (POH) Is an Early Predictor of Pancreatic Fistula Occurrence and Severity After Distal Pancreatectomy: Results from a European Multicentric Study. Ann Surg. 2025;282:291-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |