Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.114768
Revised: November 5, 2025
Accepted: January 13, 2026
Published online: March 27, 2026
Processing time: 179 Days and 23.8 Hours
Splenic stiffness measurement (SSM) via transient elastography is an emerging, non-invasive method for assessing portal hypertension and liver disease severity in cirrhosis. Its role is particularly relevant in low-resource settings where access to endoscopic services is limited.
To assess splenic stiffness patterns among cirrhotic patients using transient elastography and determine their relationship with liver disease severity.
In a comparative cross-sectional study at a tertiary hospital in Katsina, Nigeria, 60 patients with clinically diagnosed liver cirrhosis and 60 age- and sex-matched healthy controls underwent SSM using transient elastography. Liver disease severity was classified using the Child-Pugh score. Data were analyzed using Mann-Whitney U, Kruskal-Wallis, and Spearman’s correlation tests.
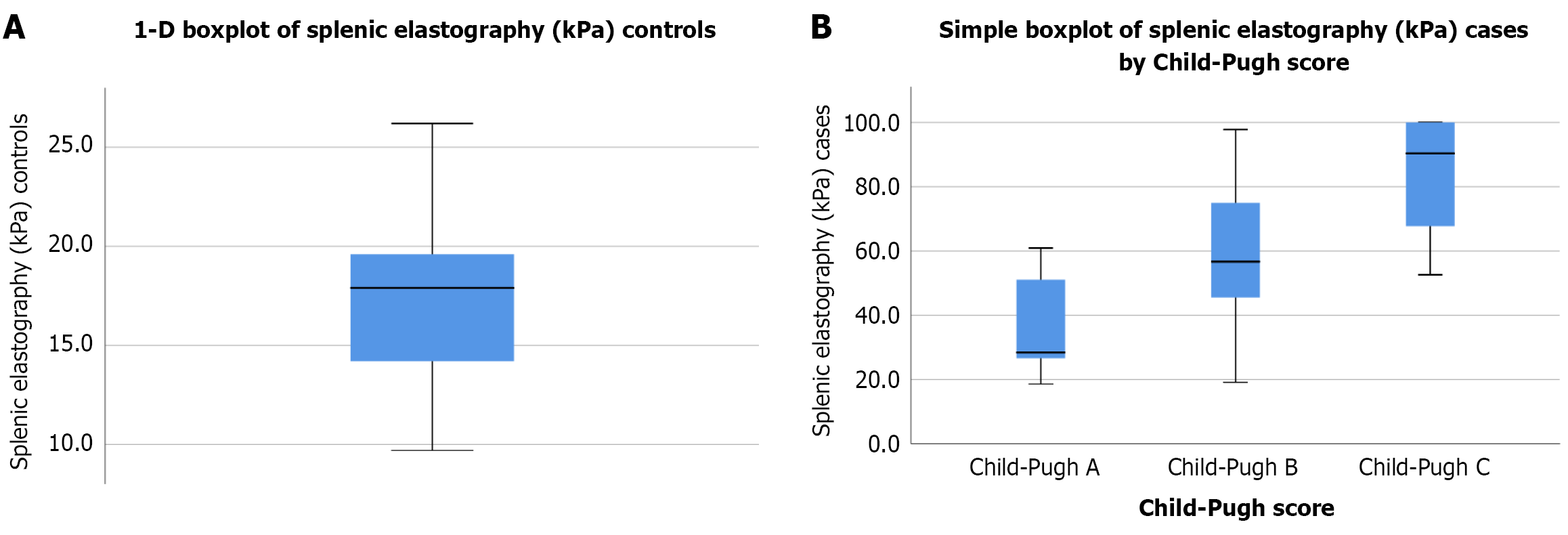
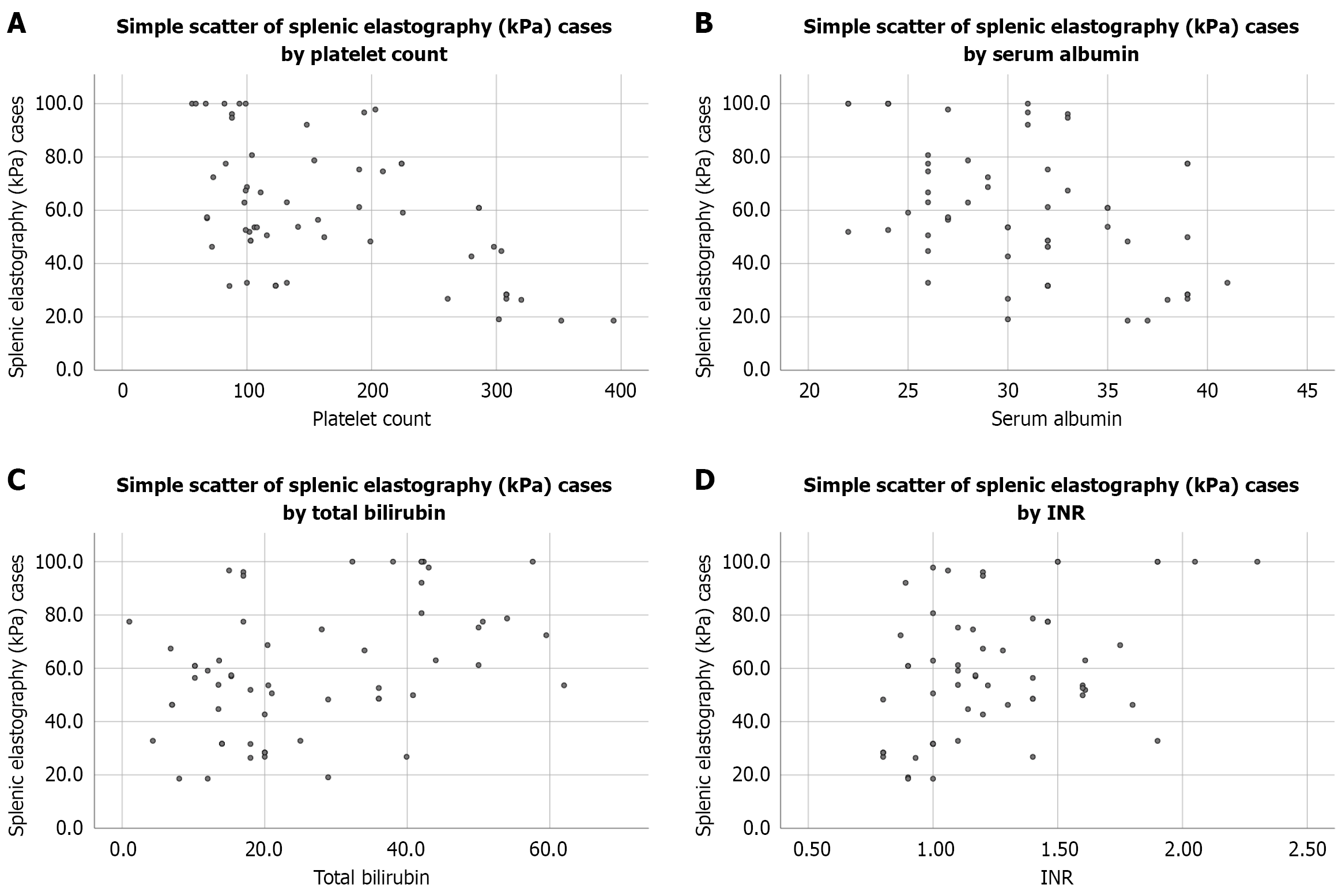
Median SSM was significantly higher in cirrhotic patients compared with controls (56.8 kPa vs 17.9 kPa; P < 0.001). SSM increased progressively with worsening Child-Pugh class (A: 28.4 kPa; B: 56.7 kPa; C: 90.6 kPa; P < 0.001). SSM correlated positively with total bilirubin (r = 0.384; P = 0.002) and international normalized ratio (r = 0.441; P < 0.001), and negatively with serum albumin (r = -0.468; P < 0.001) and platelet count (r = -0.533; P < 0.001).
SSM correlates with liver disease severity and offers a practical, non-invasive alternative for assessing portal hypertension, potentially reducing reliance on endoscopy in resource-limited settings.
Core Tip: Splenic stiffness measurement (SSM) by transient elastography is a promising, non-invasive proxy for portal hypertension in cirrhosis, especially where endoscopy access is limited. In this North-Western Nigerian cohort, SSM was significantly higher in cirrhotic patients and rose with advancing Child-Pugh class. Strong correlations emerged between SSM and indicators of liver dysfunction (bilirubin, international normalized ratio, albumin, platelets), supporting its utility as an objective severity marker. Implementing SSM could streamline risk stratification, monitor disease progression, and alleviate the burden on scarce endoscopic services in low-resource settings, offering a scalable tool for improving clinical decision-making.
- Citation: Saleh HT, Musa Y, Aminu AS, Abdullahi HZ, Samaila AA, Borodo MM. Splenic elastography in cirrhosis: A practical, non-invasive portal hypertension assessment from a Nigerian tertiary center. World J Hepatol 2026; 18(3): 114768
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/114768.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.114768
Liver cirrhosis is the end-stage of chronic liver disease, characterized by architectural distortion, fibrosis, and regenerative nodule formation[1]. Globally, it accounts for about two million deaths annually[2] with complications such as variceal bleeding contributing significantly to mortality[3]. Portal hypertension is a key pathophysiological hallmark of cirrhosis, defined as a portal pressure gradient exceeding 5 mmHg[4]. Clinically significant portal hypertension (CSPH) develops when the gradient exceeds 10 mmHg, at which point varices commonly form[5]. In low-resource regions like sub-Saharan Africa, hepatitis B virus infection is a leading cause of cirrhosis[6], and variceal bleeding remains a major cause of morbidity and mortality[3]. Upper gastrointestinal endoscopy is the gold standard for detecting varices[7], but its utility is limited in many low-resource settings due to high costs, lack of trained personnel, and limited infrastructure[8]. These barriers make routine endoscopic screening of all cirrhotic patients impractical.
Non-invasive tools for assessing portal hypertension include platelet count, spleen diameter, platelet count-to-spleen diameter ratio, and liver stiffness measurement (LSM)[9,10]. Recently, splenic stiffness measurement (SSM) using tr
This study aimed to characterize splenic stiffness patterns using transient elastography among cirrhotic patients in North-Western Nigeria and examine their relationship with liver disease severity, thereby evaluating its potential as a non-invasive tool for risk stratification in resource-limited settings.
This study was conducted at the Federal Teaching Hospital Katsina, a 500-bed tertiary referral center in North-Western Nigeria. The Gastroenterology Unit is equipped with a FibroScan Expert 630 device (Echosens, Paris, France). Approximately 40 patients with cirrhosis are managed monthly, providing a consistent recruitment base.
We conducted a comparative cross-sectional study from March 2024 to September 2024. Sixty adult patients (≥ 18 years) with liver cirrhosis and sixty age- and sex-matched healthy controls were recruited in a 1:1 ratio.
Inclusion criteria for case: Diagnosis of cirrhosis established by ≥ 2 of the following: (1) Clinical and/or biochemical features of chronic liver disease (e.g., ascites, variceal bleeding, hypoalbuminemia with reversed albumin/globulin ratio); (2) Imaging suggestive of cirrhosis (e.g., coarse echotexture, nodular surface); (3) Liver stiffness > 15 kPa on transient elastography in accordance with EASL 2021 Clinical Practice Guidelines[15]; and (4) Histological confirmation (METAVIR F4), where available.
Exclusion criteria for cases: (1) Age < 18 years; (2) Pregnancy; (3) Hepatocellular carcinoma (> 2 cm lesion with alpha-fetoprotein > 400 ng/mL); (4) Focal splenic lesions or massive ascites; (5) Participants with splenomegaly due to he
Inclusion criteria for controls: (1) No clinical or imaging evidence of chronic liver disease; and (2) Negative serology for hepatitis B surface antigen and anti-hepatitis C virus antibody.
Exclusion criteria for controls: (1) Patients with esophageal varices from non-cirrhotic portal hypertension; (2) Significant fluid overload from non-hepatic causes; (3) Obesity (body mass index ≥ 30 kg/m2); and (3) Participants with splenomegaly due to hematologic diseases, malaria, or congenital splenic disorders.
All participants underwent detailed history-taking and physical examination. For cases, the severity of liver disease was assessed using the Child-Pugh classification, incorporating serum albumin, total bilirubin, international normalized ratio (INR), presence of ascites, and encephalopathy grade[16]. Blood samples were analyzed for: (1) Liver function tests (total bilirubin, direct bilirubin, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, albumin, total protein); (2) Hematology (platelet count); (3) Coagulation profile (prothrombin time and INR); and (4) Viral serology (hepatitis B surface antigen, anti-hepatitis C virus antibody).
SSM was performed using transient elastography (FibroScan Expert 630, Echosens, Paris). The patient was positioned supine with slight right rotation to expose the posterolateral aspect of the left lower costal region. The spleen was identified via ultrasound guidance in the 7th-9th intercostal space along the posterior axillary line.
The Gastroenterology Unit comprises two consultant gastroenterologists and two fellows. One of the consultants had formal certification training in transient elastography (FibroScan®) and supervised all procedures. At least ten valid measurements were taken with the M+ probe, ensuring an interquartile range (IQR) to median ratio ≤ 30% and a success rate ≥ 60%[17]. The median value was expressed in kPa.
All statistical analyses were performed using IBM SPSS Statistics, version 30.0 (IBM Corp., Armonk, NY, United States). Continuous variables were assessed for normality using the Shapiro-Wilk test and summarized as mean ± SD or median with IQR, as appropriate. Categorical variables were presented as n (%). Comparisons between cirrhotic patients and healthy controls were conducted using the Mann-Whitney U test for non-normally distributed variables. Comparisons across Child-Pugh classes were evaluated using the Kruskal-Wallis test, with post hoc pairwise comparisons where applicable.
Associations between categorical variables were assessed using the χ2 test or Fisher’s exact test, as appropriate. Correlations between splenic stiffness and laboratory parameters (platelet count, albumin, total bilirubin, and INR) were examined using Spearman’s rank correlation coefficient. Boxplots were used to illustrate group differences in splenic stiffness between cirrhotic patients and healthy controls, as well as across Child-Pugh classes. Scatterplots were used to depict correlations between splenic stiffness and key laboratory indices. All tests were two-tailed, and P < 0.05 was considered statistically significant.
Ethical approval was obtained from the Human Research Ethics Committee of Federal Teaching Hospital Katsina (approval No. FTHKTNHREC.REG.24/06/22C/051). Written informed consent was obtained from all participants. The study adhered to the principles of the Declaration of Helsinki (2013 revision).
A total of 120 participants were enrolled: 60 patients with liver cirrhosis and 60 age- and sex-matched healthy controls. The overall mean age was 44.4 ± 12.0 years (range: 19-71), with males comprising 65% of the cohort (male-to-female ratio, 1.86:1). The age and sex distribution did not differ significantly between groups (P = 1.000; Table 1). Hepatitis B virus was the predominant etiology of cirrhosis (70%), followed by hepatitis C virus (10%). All patients reported herbal medication use, while none reported significant alcohol consumption.
| Variable(s) | Liver disease category | Total | χ2 | P value | |
| Control | Cases (cirrhosis) | ||||
| Age of participants (in years) | |||||
| ≤ 30 | 9 (15.0) | 9 (15.0) | 18 (15.0) | 0.000 | 1.000 |
| 31-40 | 10 (16.7) | 10 (16.7) | 20 (16.7) | ||
| 41-50 | 19 (31.7) | 19 (31.7) | 38 (31.7) | ||
| 51-60 | 14 (23.3) | 14 (23.3) | 28 (23.3) | ||
| ≥ 61 | 8 (13.3) | 8 (13.3) | 16 (13.3) | ||
| Range | 19-71 | 19-71 | 19-71 | ||
| mean ± SD | 44.32 ± 11.96 | 44.57 ± 12.11 | 44.44 ± 11.99 | ||
| Gender of participants | |||||
| Female | 21 (35.0) | 21 (35.0) | 42 (35.0) | 0.000 | 1.000 |
| Male | 39 (65.0) | 39 (65.0) | 78 (65.0) | ||
Median SSM was markedly elevated in cirrhotic patients compared with controls [56.8 kPa (IQR: 36.4-88.9) vs 17.9 kPa (IQR: 14.2-20.6); P < 0.001; Table 2, Figure 1]. SSM values increased progressively with worsening liver disease severity: Child-Pugh A: 28.4 kPa; B: 56.7 kPa; and C: 90.6 kPa (P < 0.001 for trend; Figure 1B). Compared to controls, cirrhotic patients demonstrated significantly lower platelet counts and serum albumin levels, and higher total bilirubin and INR values (P < 0.001 for all comparisons; Table 2).
| Parameter | Cirrhotics (n = 60) | Controls (n = 60) | 1P value |
| Splenic stiffness (kPa) | 56.8 (36.4-88.9) | 17.9 (14.2-20.6) | < 0.001 |
| Platelet count (× 109/L) | 85 (65-110) | 220 (190-250) | < 0.001 |
| Serum albumin (g/L) | 30.2 ± 5.1 | 42.8 ± 3.6 | < 0.001 |
| Total bilirubin (μmol/L) | 40.5 (22.3-78.6) | 12.6 (9.8-15.4) | < 0.001 |
| INR | 1.6 ± 0.3 | 1.1 ± 0.1 | < 0.001 |
Among cirrhotic patients, SSM showed strong negative correlations with platelet count (r = -0.53, P < 0.001; Figure 2A) and serum albumin (r = -0.47, P < 0.001; Figure 2B). Positive correlations were observed with total bilirubin (r = 0.38, P = 0.002; Figure 2C) and INR (r = 0.44, P < 0.001; Figure 2D). These findings indicate that splenic stiffness increases with worsening synthetic function and portal hypertension severity.
Our study demonstrated a markedly elevated SSM in patients with liver cirrhosis compared to healthy controls, which increases progressively with worsening Child-Pugh class. Furthermore, SSM correlates significantly with key laboratory indicators of hepatic decompensation, including serum albumin, total bilirubin, INR, and platelet count. These findings underscore SSM’s potential as a non-invasive surrogate marker for portal hypertension and liver disease severity when used independently in low-resource settings.
Our results are consistent with studies from Europe and Asia, where SSM has shown strong associations with portal hypertension and cirrhosis severity[11,18-20]. Colecchia et al[11] reported that SSM was significantly higher in cirrhotic patients compared with controls and increased with worsening liver function, findings echoed in our cohort. Similarly, Siahaan et al[21] observed that SSM was superior to liver stiffness in predicting complications of portal hypertension.
African data remain limited. M Hussein et al[22] reported SSM among 50 Egyptians with cirrhosis; it was found to be significantly associated with the presence and grade of esophageal varices and correlated with thrombocytopenia, hypoalbuminemia, hyperbilirubinemia, and elevated INR, mirroring our findings. Our study expands this evidence base as the largest West African dataset to date, featuring a control group and a standardized elastography protocol.
While LSM is a well-validated marker of fibrosis and early portal hypertension, its sensitivity declines at advanced stages when hepatic fibrosis reaches a plateau. SSM, in contrast, reflects splenic congestion and fibrosis secondary to elevated portal pressure, providing a more direct index of portal hypertension. Comparative studies have shown SSM to outperform LSM in predicting CSPH and varices[11,18,21]. These findings support the potential complementary role of SSM in evaluating cirrhosis severity, especially in low-resource settings where invasive measurements are not feasible.
Several studies have proposed threshold values of SSM for identifying CSPH and esophageal varices. Colecchia et al[11] in 2012 reported a cut-off of 46 kPa for CSPH with an area under the receiver operating characteristic curve of 0.93, while Zykus et al[18] in 2015 found 47.6 kPa to predict the presence of esophageal varices in chronic liver disease. More recently, Liu et al[17] in 2023 identified 43.4 kPa as the optimal threshold for high-risk varices in cirrhotic patients using a spleen-dedicated transient elastography device. In our cohort, the median SSM among cirrhotic patients (56.8 kPa) and particularly in those with Child-Pugh C disease (90.6 kPa) exceeded these published cut-offs, suggesting that most of our participants likely had CSPH. Although the present study was not designed to determine a specific diagnostic threshold, these comparative data highlight the potential of SSM to serve as a practical, non-invasive indicator of portal hy
The spleen enlarges and stiffens in cirrhosis due to a combination of passive congestion from elevated portal venous pressure, hyperplasia of the reticuloendothelial system, and fibrotic changes within the splenic parenchyma[23]. These structural alterations increase tissue elasticity modulus, measurable as higher kPa values on transient elastography. The progressive rise in SSM from Child-Pugh A to C in our study likely reflects worsening portal hypertension and splenic congestion with advancing disease.
The inverse correlation between SSM and platelet count aligns with the well-established link between hypersplenism and thrombocytopenia in cirrhosis[24]. Similarly, the negative association with serum albumin and positive association with bilirubin and INR reflect the intertwined pathophysiology of portal hypertension and hepatic synthetic dysfunction.
In many sub-Saharan African settings, endoscopic variceal screening is limited by availability, long wait times, and high out-of-pocket costs[8,25]. SSM offers a portable, rapid, and non-invasive alternative that could help triage patients for endoscopy, focusing scarce resources on those most likely to have advanced portal hypertension. Although the current study does not report variceal prediction cut-offs (reserved for a separate analysis), the strong correlation between SSM and markers of decompensation suggests that it could be integrated into clinical algorithms for cirrhosis management in low-resource contexts. Additionally, transient elastography requires minimal patient preparation, can be performed at the bedside or outpatient clinic, and yields immediate results. These features make it particularly appealing in rural and underserved areas where follow-up may be challenging.
Cross-sectional design precludes assessment of longitudinal changes in SSM or its predictive value for outcomes such as bleeding or mortality. Single-center study may limit generalizability to broader populations. Measurement variability: While transient elastography is user-friendly, variability can occur, especially in patients with narrow intercostal spaces or high body mass index. Diagnostic confirmation: Although we included a well-matched control group, histological confirmation of cirrhosis was not available for all patients, introducing potential diagnostic misclassification. Findings in this study primarily reflects viral related etiology.
Future multicenter prospective studies in Africa should evaluate SSM alongside other non-invasive markers to develop region-specific predictive algorithms. Integration of SSM into existing screening protocols could be tested for cost-effectiveness and impact on patient outcomes.
Splenic stiffness is significantly increased in patients with cirrhosis compared with healthy individuals and demonstrates a strong association with the severity of liver disease. In regions with limited endoscopic capacity, SSM provides a practical, non-invasive tool to inform clinical decision-making and optimize the allocation of endoscopic services. Broader implementation and rigorous validation of this approach have the potential to enhance liver disease assessment and improve resource utilization in low-resource settings.
| 1. | Asrani SK, Devarbhavi H, Eaton J, Kamath PS. Burden of liver diseases in the world. J Hepatol. 2019;70:151-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3074] [Cited by in RCA: 2528] [Article Influence: 361.1] [Reference Citation Analysis (7)] |
| 2. | GBD 2017 Cirrhosis Collaborators. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5:245-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1329] [Cited by in RCA: 1172] [Article Influence: 195.3] [Reference Citation Analysis (10)] |
| 3. | Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1775] [Cited by in RCA: 1553] [Article Influence: 172.6] [Reference Citation Analysis (5)] |
| 4. | Bosch J, Abraldes JG, Berzigotti A, García-Pagan JC. The clinical use of HVPG measurements in chronic liver disease. Nat Rev Gastroenterol Hepatol. 2009;6:573-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 625] [Cited by in RCA: 547] [Article Influence: 32.2] [Reference Citation Analysis (4)] |
| 5. | de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2652] [Cited by in RCA: 2371] [Article Influence: 215.5] [Reference Citation Analysis (2)] |
| 6. | Musa BM, Bussell S, Borodo MM, Samaila AA, Femi OL. Prevalence of hepatitis B virus infection in Nigeria, 2000-2013: a systematic review and meta-analysis. Niger J Clin Pract. 2015;18:163-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 96] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 7. | Garcia-Tsao G, Sanyal AJ, Grace ND, Carey W; Practice Guidelines Committee of the American Association for the Study of Liver Diseases; Practice Parameters Committee of the American College of Gastroenterology. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology. 2007;46:922-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1387] [Cited by in RCA: 1198] [Article Influence: 63.1] [Reference Citation Analysis (2)] |
| 8. | Alatise OI, Allen H, Barton-Burke M, Abiodun AA, Ajayi AO, Ajayi N; Nigerian Endoscopy Collaborative, Knapp GC, Kingham PT, Murthy SS. Endoscopy capacity in Nigeria: a national needs assessment and recommendations. Surg Endosc. 2025;39:5698-5713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 9. | Giannini E, Botta F, Borro P, Risso D, Romagnoli P, Fasoli A, Mele MR, Testa E, Mansi C, Savarino V, Testa R. Platelet count/spleen diameter ratio: proposal and validation of a non-invasive parameter to predict the presence of oesophageal varices in patients with liver cirrhosis. Gut. 2003;52:1200-1205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 364] [Cited by in RCA: 309] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 10. | Kazemi F, Kettaneh A, N'kontchou G, Pinto E, Ganne-Carrie N, Trinchet JC, Beaugrand M. Liver stiffness measurement selects patients with cirrhosis at risk of bearing large oesophageal varices. J Hepatol. 2006;45:230-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 262] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 11. | Colecchia A, Montrone L, Scaioli E, Bacchi-Reggiani ML, Colli A, Casazza G, Schiumerini R, Turco L, Di Biase AR, Mazzella G, Marzi L, Arena U, Pinzani M, Festi D. Measurement of spleen stiffness to evaluate portal hypertension and the presence of esophageal varices in patients with HCV-related cirrhosis. Gastroenterology. 2012;143:646-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 392] [Article Influence: 28.0] [Reference Citation Analysis (3)] |
| 12. | Stefanescu H, Procopet B. Noninvasive assessment of portal hypertension in cirrhosis: liver stiffness and beyond. World J Gastroenterol. 2014;20:16811-16819. [PubMed] [DOI] [Full Text] |
| 13. | Kim NJ, Su F, Biggins SW. CON: Noninvasive Modalities Are Preferred to Screen for the Diagnosis of Esophageal and Gastric Varices When the Diagnosis of Cirrhosis Is Made. Clin Liver Dis (Hoboken). 2020;16:48-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 14. | Fraquelli M, Giunta M, Pozzi R, Rigamonti C, Della Valle S, Massironi S, Conti CB, Aghemo A, Ronchi G, Iurlo A, Primignani M, Conte D, Colombo M. Feasibility and reproducibility of spleen transient elastography and its role in combination with liver transient elastography for predicting the severity of chronic viral hepatitis. J Viral Hepat. 2014;21:90-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 15. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis - 2021 update. J Hepatol. 2021;75:659-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1546] [Cited by in RCA: 1388] [Article Influence: 277.6] [Reference Citation Analysis (6)] |
| 16. | Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 1973;60:646-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6342] [Cited by in RCA: 5745] [Article Influence: 108.4] [Reference Citation Analysis (9)] |
| 17. | Liu J, Xu H, Liu W, Zu H, Ding H, Meng F, Zhang J. Spleen stiffness determined by spleen-dedicated device accurately predicted esophageal varices in cirrhosis patients. Ther Adv Chronic Dis. 2023;14:20406223231206223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 18. | Zykus R, Jonaitis L, Petrenkienė V, Pranculis A, Kupčinskas L. Liver and spleen transient elastography predicts portal hypertension in patients with chronic liver disease: a prospective cohort study. BMC Gastroenterol. 2015;15:183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 19. | Shanka NY, Pavlov CS, Mekonnen NL. Non-invasive methods for diagnosing portal hypertension and variceal bleeding due to liver cirrhosis secondary to NAFLD/MASLD: systematic review. Front Med (Lausanne). 2024;11:1459569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 20. | Majumdar A, Marasco G, Vestito A, Pinzani M, Festi D. The Assessment of Portal Hypertension. In: Elastography of the Liver and Beyond. Cham: Springer, 2021: 159-171. [DOI] [Full Text] |
| 21. | Siahaan BS, Kurniawan J, Simadibrata M, Rizka A, Sanityoso A, Rumende CM, Rachman A, Shatri H. Spleen and Liver Stiffness as Noninvasive Predictors of Varices in HBV Cirrhosis. Indones J Gastroenterol Hepatol Dig Endosc. 2023;24:137-142. [DOI] [Full Text] |
| 22. | M Hussein A, Badr B, Abdelsameea E. Role of Liver and Spleen Stiffness in Predicting the Degree and Bleeding Risk of Esophageal Varices in Cirrhotic Patients. Benha Med J. 2022. [DOI] [Full Text] |
| 23. | Bolognesi M, Merkel C, Sacerdoti D, Nava V, Gatta A. Role of spleen enlargement in cirrhosis with portal hypertension. Dig Liver Dis. 2002;34:144-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 183] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 24. | Mitchell O, Feldman DM, Diakow M, Sigal SH. The pathophysiology of thrombocytopenia in chronic liver disease. Hepat Med. 2016;8:39-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 25. | Mwachiro M, Topazian HM, Kayamba V, Mulima G, Ogutu E, Erkie M, Lenga G, Mutie T, Mukhwana E, Desalegn H, Berhe R, Meshesha BR, Kaimila B, Kelly P, Fleischer D, Dawsey SM, Topazian MD. Gastrointestinal endoscopy capacity in Eastern Africa. Endosc Int Open. 2021;9:E1827-E1836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |