Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.114435
Revised: November 20, 2025
Accepted: January 9, 2026
Published online: March 27, 2026
Processing time: 189 Days and 0.2 Hours
Malnutrition in chronic liver disease (CLD) is a critical, though often overlooked, complication. Results show that up to ninety percent of patients with advanced CLD suffer from protein calorie malnutrition. The study reviews existing li
Core Tip: Patients with chronic liver disease (CLD) may suffer from malnutrition, which may affect more than 90% of advanced CLD patients. The CLD review notes several mechanisms for nutritional deterioration including anorexia, malabsorption, and alterations of metabolism and outlines evidence-based, etiology-specific nutritional strategies. Inclusive of early assessment, multidisciplinary team care, individual-based intervention, nutritional management in CLD can greatly enhance patient outcomes and decrease disease burden.
- Citation: Hafez MM, Nasseem M, Boukhechem I, ElSheikh R, Tawheed A. Optimizing nutritional interventions in chronic liver disease: Etiology-specific strategies for enhanced clinical management. World J Hepatol 2026; 18(3): 114435
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/114435.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.114435
Nutritional support is important for chronic liver disease (CLD) management. It helps in disease progress and patient outcomes. According to studies, malnutrition in liver disease, particularly protein-calorie malnutrition (PCM), is common in approximately 65%-90% of patients with advanced liver disease. It is a well-known independent predictor of mor
The functions of the liver are important for digestion, metabolism, storage, and synthesis of nutrients. CLD impairs these functions, leading to energy imbalance, muscle wasting, and a deficiency of micronutrients. Sarcopenia, caused by changes in protein metabolism, is more common and complicates management. Disorders characterized by cholestatic liver disease specifically cause deficiencies of fat-soluble vitamins, while hepatocellular dysfunction is usually associated with changes in amino acid metabolism[1].
Cirrhosis patients experience symptoms such as vomiting, nausea, early satiety, and increased metabolic demands. When patients have these symptoms, they may reduce their dietary intake, which can worsen the disease and the overall condition. Signs of malnutrition may include impaired wound healing, prolonged recovery times, and functional decline. These signs and symptoms require a validated screening method such as Subjective Global Assessment (SGA), Mid-Upper Arm Circumference (MUAC), and handgrip strength, as malnutrition can be masked by conditions like fluid overload or obesity[2].
In cirrhotic patients, personalized nutritional strategies are crucial for disease staging. For example, branched-chain amino acids (BCAAs) are important for cirrhotic patients with related brain issues. Moreover, cirrhotic patients with poor digestion may need medium-chain triglycerides (MCTs) and fat-soluble vitamin supplements in their nutritional plan. In cirrhotic patients with very poor condition, enteral nutrition (EN) must be considered. EN is known to improve the cirrhotic patients’ conditions and lower mortality rates, especially when started as early as possible[3-5].
Effective management requires a multidisciplinary approach that includes hepatologists, dietitians, nurses, and pharmacists, aiming to provide individualized, evidence-based nutritional treatments for each patient. When patients are more educated and engaged, they are better able to follow dietary recommendations. Nutritional screening, early intervention, and ongoing follow-up should be standard practice for all patients suspected of malnutrition, as mal
CLD is a broad term that describes several conditions leading to the gradual damage of the liver. These diseases are progressive in nature and often result in cirrhosis. Having a comprehensive understanding of the common causes of CLD allows for effective management. Causes include alcoholic liver disease (ALD), non-alcoholic fatty liver disease (NAFLD), chronic viral hepatitis, cholestatic disorders, and many others. Alcohol-induced liver illness is one of the most common reasons for CLD. It develops a spectrum of diseases from alcoholic fatty liver to alcoholic hepatitis and, ultimately, cirrhosis. Liver damage correlates with the level and duration of alcohol consumption. People who drink heavily can develop inflammation, which can contribute to fat accumulation in liver cells. This condition is called steatosis. It may progress to two other serious conditions: Alcoholic steatohepatitis and cirrhosis[7]. Obesity and Metabolic Syndrome can lead to NAFLD becoming the most common CLD in developed countries. The disorder involves liver injuries that range from simple steatosis to non-alcoholic steatohepatitis (NASH). NASH can progress to fibrosis or cirrhosis. As obesity rates and other metabolic conditions such as type 2 diabetes continue to rise, NAFLD has become a public health concern due to its association with increased risks of liver complications and cardiovascular diseases[8].
The two most common and significant viral infections contributing to CLD worldwide are hepatitis B virus (HBV) and hepatitis C virus (HCV) infection. If not properly treated, these viruses can cause chronic inflammation and damage to liver cells. In East Asia and sub-Saharan Africa, the prevalence of chronic hepatitis B infection is high. Variations in hepatitis C genotypes also complicate the global epidemiology. Without appropriate therapy, chronic viral infections can eventually lead to cirrhosis and liver cancer. Cholestatic diseases are also important in the development of CLD. Damage or fibrosis of the bile ducts, as seen in primary biliary cholangitis and primary sclerosing cholangitis, disrupts bile drainage. This blockage results in the accumulation of bile acids in the liver, further harming liver cells and causing cholestasis. Over time, these conditions worsen liver damage. Other causes include genetic disorders like hereditary hemochromatosis, which involves excessive iron absorption, and Wilson's disease, characterized by copper buildup due to defects in copper metabolism. The interaction of inherited traits and environmental factors leads to liver damage[9].
In CLD patients, anorexia is a common symptom. The condition manifests as decreased food intake and loss of appetite. Patients may experience anorexia due to various factors, such as neurotransmitter changes related to liver failure. When the liver is healthy, it produces certain substrates that help regulate appetite. Additionally, inflammation of hepatocytes triggers the release of inflammatory cytokines that suppress appetite. Physiological changes also affect the patient’s taste and smell perception, leading to reduced pleasure from foods and increased refusal to eat. The disruption of these pathways occurs as the disease progresses[2,10].
Anorexia greatly impacts the nutritional health of CLD patients. Many patients with anorexia follow diets that are insufficient to prevent malnutrition. Reduced energy reserves cause muscle loss, or sarcopenia, making recovery more challenging. Therefore, nutritional interventions in hospitals have proven effective in managing malnutrition caused by anorexia in CLD patients. Providing nutrition early, whether orally or through a tube, can increase calorie intake and improve outcomes. Patients who receive adequate nutritional care generally recover better from hepatic decompensation than those who do not address nutritional deficiencies[11].
Structured meal plans with small, frequent meals can help reduce the effects of anorexia and increase calorie intake in people with CLD who have lost their appetite. The strategies emphasize high-energy foods but also consider individual preferences to promote consumption of foods that individuals will eat, not just what they should eat. The anorexia associated with CLD is complex and requires specific measures to stimulate appetite and nutrition, ensuring the patient receives enough calories without disrupting metabolism. Healthcare practitioners must be aware of other factors, such as medications prescribed for liver diseases, which can worsen anorexia. Therefore, the patient must be evaluated, and tailored nutritional strategies should be developed accordingly[12-15].
Malabsorption is a major issue for patients with CLD and severely exacerbates their nutrient deficiencies and outcomes. The causes of nutrient malabsorption in these patients are complex and often interconnected. Cholestasis, or impairment of bile flow, is a primary cause. Bile is essential for emulsifying and absorbing dietary fats; without it, fat malabsorption occurs, leading to deficiencies of essential fatty acids and fat-soluble vitamins A, D, E, and K. Liver dysfunction can significantly limit the liver's ability to process various nutrients. An example of metabolic imbalances is the disruption of protein, carbohydrate, and fat metabolism. Glucose levels in a patient may become unstable due to insulin resistance or reduced gluconeogenesis by the liver. This metabolic imbalance results in decreased insulin activity and increased lipolysis, causing fat deposits in the body despite nutrient deficiencies. People with CLD also experience malabsorption due to pancreatic insufficiency. Chronic alcoholic pancreatitis can impair the production of enzymes necessary for proper digestion of macronutrients, a condition known as pancreatic exocrine insufficiency[13,15]. As a result, patients with liver cirrhosis and pancreatic insufficiency may experience steatorrhea (excess fat in their stools). Iatrogenic causes significantly worsen malabsorption issues. Patients undergoing treatment for CLD face many gastrointestinal (GI) side effects that hinder nutrient absorption. Some commonly prescribed drugs, including lactulose for hepatic encephalopathy (HE), can cause diarrhea, leading to nutrient loss and dehydration. Due to these underlying problems, healthcare providers might observe various signs of malnutrition in patients; therefore, assessments are essential to detect them. Regular evaluations using anthropometric measures enable clinicians to monitor changes effectively. Managing nutrition in CLD is complex and requires personalized approaches, as the underlying causes can vary widely[15,16].
Children with CLD are an important group of patients because their metabolic demands are high due to rapid growth. Therefore, malabsorption and decreased caloric intake in this age group require an effective solution. MCTs can be effectively used in children with CLD, as MCTs are easily absorbed forms of nutrients that bypass the problematic pathways of absorption. Moreover, dietary modifications are necessary, taking into account the complex interactions between liver pathology and nutrient metabolism. Enteral or parenteral nutrition may be required when oral intake alone fails to meet caloric demands in severe cases[2].
The dysfunction of the liver in CLD can alter the synthesis of proteins, clotting factors, and others. Liver disorders thus lead to low albumin, fluid shifts, and edema. Moreover, high resting energy expenditure in CLD makes it harder to get adequate nutritional intake. The use of substances for energy metabolism in CLD patients is disturbed. The liver is important in glucose metabolism, but with the accumulation of fat and inflammation, insulin resistance hampers gluconeogenesis. The resulting unstable blood sugar complicates diet management and contributes to worsened malnutrition. Lipid processes are also disrupted. While a normal liver balances lipid synthesis and oxidation, lipogenesis occurs with decreased oxidation of fatty acids in CLD. This imbalance results in the buildup of triglycerides in the liver. It plays a role in the development of NAFLD and the worsening of alcohol-related liver disease[17].
The metabolism of micronutrients is similarly affected. The vitamins and minerals that our body lacks due to inadequate dietary intake and malabsorption syndromes adversely impact metabolism. Vitamin D deficiency occurs because the liver cannot convert vitamin D into its active form, while zinc deficiency is common due to urine loss and poor dietary intake. Amino acid metabolism is also disrupted. Patients often experience an imbalance between essential and non-essential amino acids because of impaired protein synthesis, leading to muscle wasting and sarcopenia. In CLD accompanied by encephalopathy, cognitive function is impaired, which complicates these metabolic changes. This can lead to reduced food intake or poor food choices due to confusion or fatigue[18].
Given the alterations in metabolism mentioned above in patients with CLD, it’s crucial to monitor them, as changes may occur with worsening or improvement of the disease, or with lifestyle modifications and pharmacotherapy. All recent studies show that having a personalized nutritional strategy is essential because it considers each patient’s metabolic status and the supplementations needed for every individual[2,18].
The iatrogenic factors prominently contribute to nutritional challenges in CLD. These challenges often happen after a medical procedure, a stay in a hospital, or medical treatment. One complication is the frequent hospital admissions of these patients, which often lead to extended periods of fasting or dietary restrictions due to diagnostic tests and surgical procedures. Furthermore, Inadequate access to calories and nutrients promptly can worsen nutritional health. Aside from that, some drugs affect nutrition. For instance, certain medications may cause nausea or changes in taste perception, leading to reduced appetite and food intake. Some drugs prescribed for complications of liver disease can occasionally cause malabsorption or impair metabolic processes. When drugs inhibit digestive enzymes or reduce bile secretion, this leads to fat malabsorption and nutrient deficiencies[17]. Dietary management can be complicated by iatrogenic malnutrition due to over-restriction imposed by healthcare professionals. In the past, clinicians recommended a reduced protein intake for patients suffering from HE, as this limited the worsening of the symptoms. Currently available guidelines suggest that most patients can tolerate normal or even higher protein levels if appropriate medical treatment is provided to manage encephalopathy. Imposing unsuitable dietary restrictions not only disregards nutritional needs but may also lead to muscle loss and deterioration of health[16,18]. Socio-economic problems, which affect their accessibility to healthy foods. Some people cannot afford food because it is either unavailable or too expensive for them. This can restrict their power to eat well and therefore affects their recovery.
Patients with CLD may require total parenteral nutrition (TPN) as an additional measure due to iatrogenic complications. Severely malnourished patients or those with significant liver issues might need TPN, which provides nutrition through an intravenous route. Inserting the catheter carries certain risks, such as infection. Prolonged use of TPN can impair liver function. Immunocompromised individuals or those with altered intestinal permeability and endotoxemia due to liver failure must be administered TPN very cautiously. In some cases of CLD, such as acute on chronic liver failure or acute alcoholic hepatitis, their support needs become more complex. The demand for enteral or parenteral nutrition increases during these times, but close monitoring of glucose levels and prevention of metabolic disturbances are essential due to the rapid changes in patients' nutritional status[19]. The best management plans involve a multidisciplinary approach and include the services of a dietitian and physicians, along with various healthcare providers. The patient's management will also require dietary modifications. However, any changes must align with the latest guidelines relevant to the specific patient, rather than generalized restrictions based on past practices[20].
CLD causes numerous metabolic changes that affect nutritional status. Therefore, patients need a special diet. A major concern is the lack of fat-soluble vitamins A, D, E, and K, often due to cholestasis and liver disease, which affect absorption. Vitamin D deficiency is common in advanced liver disease and is found in 64% to 92% of patients with chronic cholestatic conditions. Furthermore, vitamin D levels tend to inversely correlate with the severity of liver disease as measured by the Child-Pugh score. Due to reduced hepatic hydroxylation, vitamin D metabolism is decreased, which is necessary for its activation[17,20].
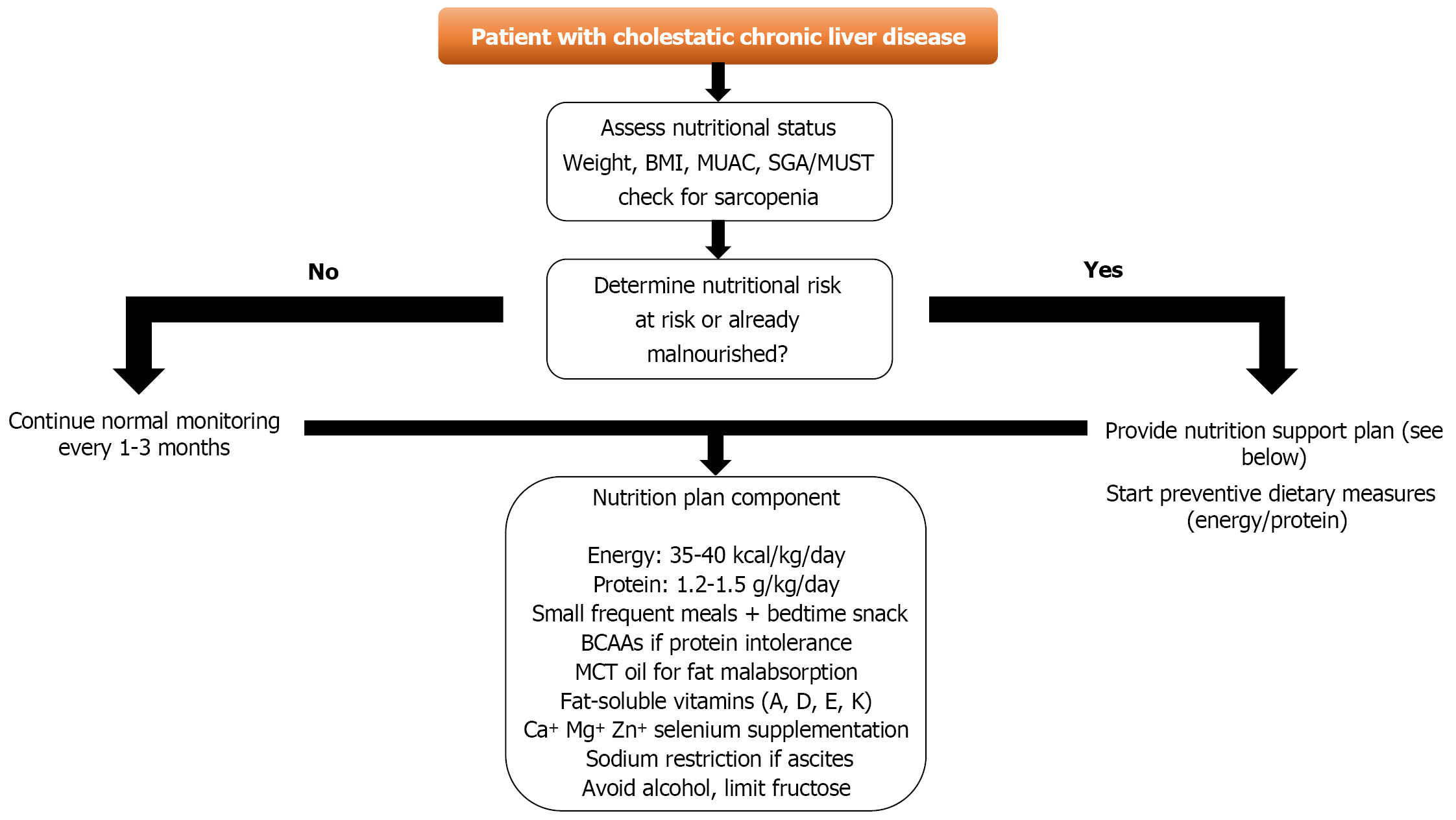
Thiamine (vitamin B1) deficiency is common in people with both alcoholic and non- ALDs due to inadequate intake, poor absorption, and limited hepatic reserves. Individuals lacking vitamin B1 (thiamine) are highly susceptible to developing serious neurological diseases like Wernicke encephalopathy and Korsakoff syndrome. This requires thiamine supplementation for affected individuals. Deficiencies in other water-soluble vitamins such as pyridoxine (B6), folate (B9), and cobalamin (B12) are also common in CLD because of reduced storage capacity in the diseased liver, which can cause anemia and complicate clinical management. Additionally, vitamin K deficiency is a significant concern in cholestatic liver diseases due to liver impairment and poor dietary absorption, increasing the risk of coagulopathy and bleeding (Figure 1). Changes in essential fatty acids and protein metabolism, along with vitamin deficiencies, are key features of nutritional disorders. Furthermore, patients with advanced liver disease often suffer from protein-energy malnutrition (PEM), which may result from reduced intake, increased energy expenditure due to inflammation, or malabsorption caused by portal hypertension or GI issues. Additionally, alcohol-related liver disease disrupts lipoprotein level metabolism[18,21]. In youngsters with CLD, along with severe protein and essential fatty acid deficiency, there must be concern for biliary atresia, which is the most common cause in this age group. For those patients, personalized nutrition interventions are crucial to support normal growth and development[21].
Assessing the nutritional status of patients with CLD presents significant challenges, primarily because conventional markers are often unreliable due to the disease's impact on fluid balance and metabolic function[4,22]. Likewise, standard laboratory markers such as serum albumin, prealbumin, and retinol-binding protein are often decreased due to impaired hepatic synthesis, fluid dilution, or protein loss[23-25]. Other lab parameters, such as lymphocyte count and prothrombin time, are also unreliable for malnutrition assessment in these patients. Given these limitations, a multi-parameter approach is uncommon. The body mass index (BMI) is not a reliable index in CLD due to ascites, edema, and fluid retention, thus artificially inflating body weight and masking a condition of true malnutrition. Patients with sarcopenic obesity may also have a normal or high BMI despite muscle loss. As a result, BMI should not be examined alone. It needs to be supplemented with SGA, MUAC, triceps skinfold, handgrip strength, and body composition for assessment[26,27].
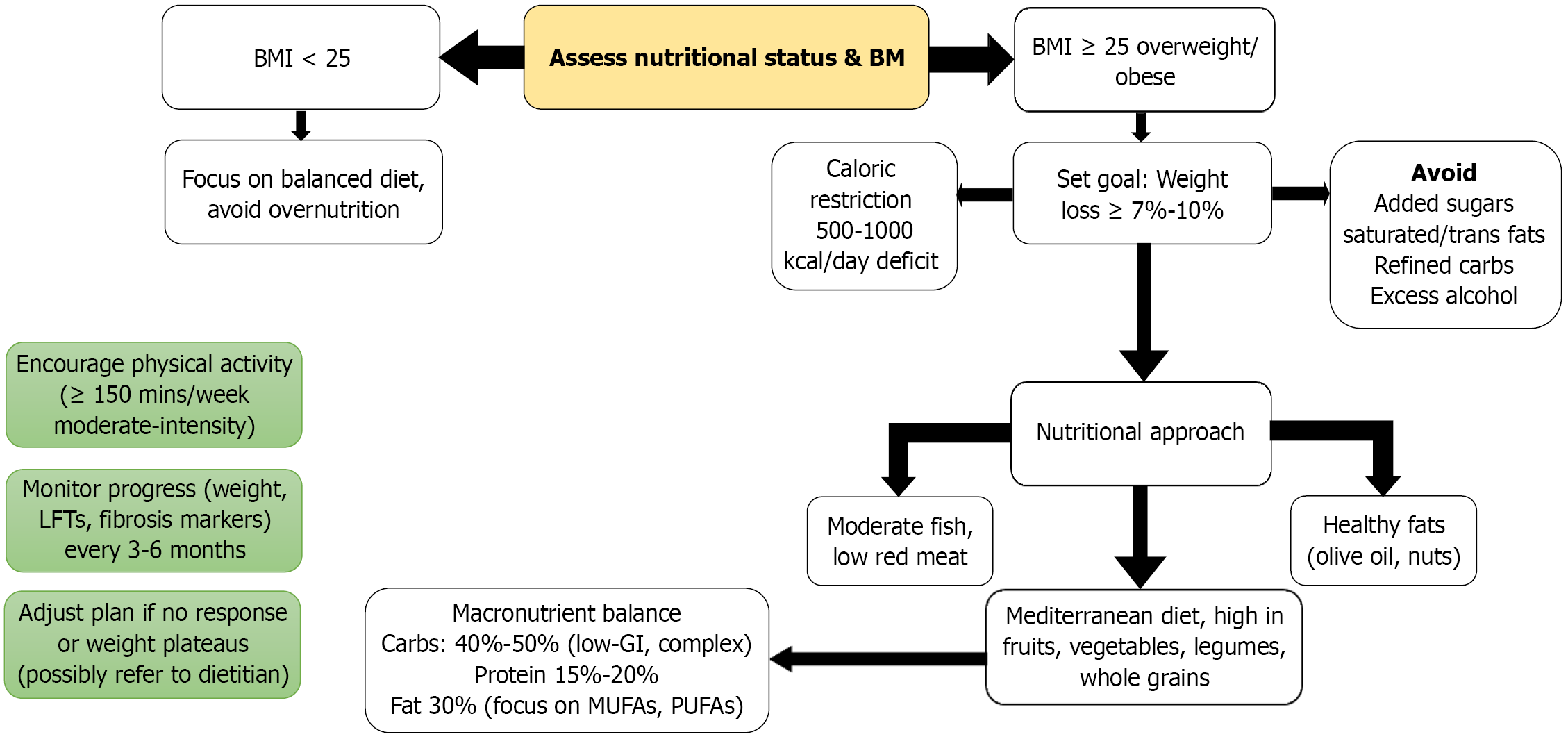
Clinical and anthropometric tools are regarded as practical, objective indicators. The SGA, a validated questionnaire and bedside tool, combines information from patient history (such as weight changes, dietary intake, and GI symptoms) with physical examination findings (including muscle wasting, subcutaneous fat loss, edema, and ascites) to determine nutritional status. It is considered reliable and useful for predicting outcomes[25]. Anthropometric measurements, like mid-arm muscle circumference and triceps skin-fold thickness-assessing muscle mass and fat stores respectively-are less influenced by fluid retention than total body weight and can aid in predicting mortality[27]. Functional assessments, especially handgrip strength measured with a dynamometer, are highly sensitive, useful for tracking patient progress, and can forecast complications and outcomes in patients with advanced liver disease[28] (Figure 2).
Body composition analysis offers more precise estimates of muscle mass and fat reserves. Although isotope dilution or neutron activation analysis are expensive methods, bioelectrical impedance analysis is more accessible but can be affected by fluid retention. Another evaluation technique is measuring body cell mass (BCM), which mainly assesses tissue metabolic activity. BCM is a crucial indicator, as its depletion signals a higher risk of mortality[29]. A 24-hour recall is a dietary survey used to evaluate daily food intake and identify deficiencies. All assessment methods aim to detect deficiencies in both micronutrients and macronutrients[4,27].
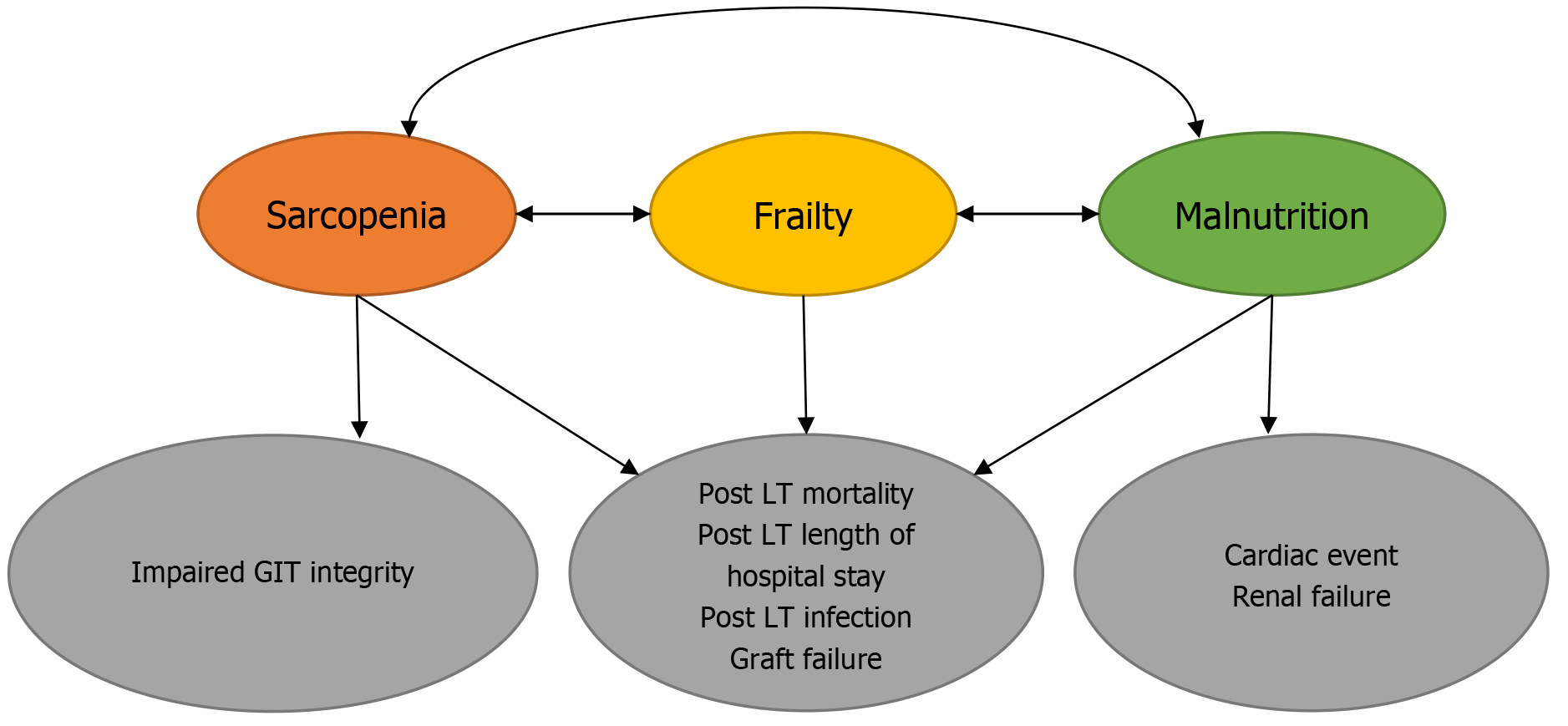
CLD patients, as mentioned, are at risk of malnutrition, which impacts their outcomes. In malnourished cirrhotic patients, there is a high risk of developing complications such as infections, primarily bacterial peritonitis. Additionally, CLD patients may develop ascites, HE, and hepatorenal syndrome. Patients might experience sarcopenia or PCM, which can affect muscle strength and reduce mobility (Figure 3). Moreover, malnutrition in CLD patients influences both functional and immune functions, increasing the risk of complications like bacterial infections. All of these factors can deteriorate quality of life, reduce patients’ tolerance to medical, nutritional, or procedural interventions, and ultimately lead to higher mortality and hospitalizations. Early detection of malnutrition in CLD is now an essential component of cirrhosis care. Routine nutrition screening and prompt intervention-including personalized dietary plans, correction of micronutrient deficiencies, and increased physical activity to prevent sarcopenia-are fundamental in managing liver cirrhosis[26,30,31].
The requirements for CLD patients to enhance liver function and nutritional status are approximately 35-40 kcal/kg/day[26,32]. Therefore, some guidelines suggest 25-35 kcal/kg/day for those without HE. The calculation should be based on dry body weight in cases of ascites and edema to avoid overestimation[28,33] (Table 1). Dietary restrictions should be avoided, as they can be harmful and decrease intake due to unpalatability. Sodium intake less than 2 g/day is re
| Guideline | Target population/scope | Energy (kcal/kg/day) | Protein (g/kg/day) | Meal timing/late-evening snack |
| EASL clinical practice guidelines | Adults with chronic liver disease/cirrhosis | 30-40 kcal/kg/day (use dry/ideal weight if ascites) | 1.2-1.5 g/kg/day (higher if sarcopenic) | Recommends small frequent meals and late-evening carbohydrate snack to reduce overnight catabolism |
| ESPEN guideline/practical guideline | Clinical nutrition for liver disease (comprehensive recommendations + practical flowcharts) | 30-40 kcal/kg/day (adjust for obesity/edema) | 1.2-1.5 g/kg/day (critically ill 12-2.0 g/kg/day as needed) | Recommends late-evening snacks and multiple small meals, practical guidance for implementation |
| INASL consensus statement (India) | Nutrition in CLD-adults and children; context-sensitive (resource settings) | Recommends individualized approximately 30-40 kcal/kg/day; use ideal/dry weight for ascites | 1.2-1.5 g/kg/day (higher for sarcopenia; adjust for comorbidity) | Advises small frequent meals and night snack (starch/CHO) to prevent fasting catabolism |
| AASLD practice guidance-malnutrition/frailty/sarcopenia in cirrhosis | Practical guidance for malnutrition, frailty and sarcopenia in cirrhosis | Aligns with approximately 30-40 kcal/kg/day goals; advises individualized targets | 1.2-1.5 g/kg/day stable patients; increase to approximately 1.5 g/kg if sarcopenic; critically ill targets higher | Supports avoidance of prolonged fasting; recommends late-evening snacks to reduce catabolism |
| ACG clinical guideline-malnutrition and nutritional recommendations in liver disease | New guidance covering malnutrition across liver disease spectrum | Recommends early screening and individualized energy (ranges like others; emphasises dry/ideal weight) | Recommends 1.2-1.5 g/kg/day (with adjustments for sarcopenia, critical illness) | Reiterates multiple small meals + late snack, practical implementation advice in United States practice settings |
CLD patients should eat multiple small meals about 5-7 times a day, as this helps reduce symptoms like nausea and vomiting, prevents protein overload, and minimizes prolonged fasting[28]. A key strategy is late-evening snacks, which offer many benefits such as improving nitrogen balance and decreasing the risk of sarcopenia by increasing muscle mass, since cirrhotic patients experience rapid muscle breakdown during overnight fasting due to accelerated starvation physiology[25,34]. This timing is effective because, during the postprandial period, protein breakdown is suppressed while synthesis is promoted, and nocturnal supplementation has been shown to produce greater gains in total body protein stores. The snack should be high in calories (at least 50 g of carbohydrates) and potentially enriched with BCAAs. According to clinical guidelines, this approach should be endorsed for all patients unless contraindicated.
The role of supplementation in cirrhotic patients is crucial because many patients with CLD experience deficiencies in micronutrients and macronutrients. In CLD, attention should be given to deficiencies in fat-soluble vitamins (A, D, E, K), B vitamins (thiamine, B12, folate, pyridoxine), and minerals such as zinc, potassium, magnesium, and phosphorus. Vitamin A deficiency may cause night blindness and hepatic fibrosis. Although vitamin A supplementation is important, caution is advised for patients who consume alcohol due to the high risk of hepatotoxicity[30-36]. Vitamin D deficiency is common and can lead to bone density loss. Recommended vitamin D supplementation includes 50000 IU weekly for 12 weeks, followed by 1000 IU daily with calcium intake of 800-1500 mg/day. Vitamin D also enhances immune function, aiding in the reduction of infections like spontaneous bacterial peritonitis[28,34]. Vitamin E deficiency may occur, so short-term supplementation can be initiated. However, long-term vitamin E supplementation is not recommended, as it can be harmful and may increase the risk of mortality[26,37].
Vitamin K is stored in the liver; therefore, in CLD patients, there may be a vitamin K deficiency, which can increase the bleeding risk in these patients. B vitamins-thiamine, folate, and pyridoxine-are commonly deficient due to poor intake, malabsorption, or impaired metabolism. Reduced zinc levels are also common and can affect HE. Deficiencies in potassium, magnesium, and phosphorus are frequently seen when diuretics are used. Conversely, manganese supplementation should be avoided because it accumulates in CLD, especially in the brain[37].
Supplements for those minerals and vitamins should be used once deficiencies are detected or empirically in high-risk patients[34]. Moreover, it is known to be beneficial in improving HE, nutritional status, and survival in advanced cirrhosis, particularly when administered at night. However, BCAA-enriched formulas are still controversial due to their high cost on patients[27,28]. In CLD, oral nutrition may be difficult or insufficient. In these conditions, EN via nasogastric tube should be established as early as possible to maintain nutritional support, as it is safe and helps improve nutritional status[28,34]. PN is the second-line method and is used if EN is contraindicated, as in HE or severely diseased GI tracts[27,35].
ALD has a poor nutritional profile, mainly due to the risk of PEM. PEM can significantly affect patient outcomes. In ALD, alcohol and the liver’s inability to metabolize nutrients efficiently disrupt normal metabolism. Since the liver is responsible for processing various nutrients, malnutrition is common during the development of ALD. Acute hepatitis (AH) occurs in a hypercatabolic and hypermetabolic state, caused by systemic inflammation and oxidative stress, which reduce the liver’s synthetic capacity. Patients often experience increased muscle protein breakdown, higher resting energy expenditure, and notable micronutrient deficiencies. Nutrition is thus a key component of therapy in AH, recommended alongside other pharmacological treatments, particularly in moderate and severe cases. Current guidelines suggest an intake of 35-40 kcal/kg/day and 1.2-1.5 g/kg/day of protein, emphasizing small, frequent meals and late-evening carbohydrate-rich snacks to prevent prolonged fasting and starvation-induced catabolism[32,37].
Attention should be given to fat-soluble vitamins (A, D, E, K), as liver dysfunction and alcohol exposure can significantly impair the absorption of these vitamins. A deficiency in vitamin A can cause metabolic disorders and lead to night blindness or xerophthalmia. Vitamin A levels should be carefully monitored to prevent toxicity, especially in already compromised livers. People suffering from chronic alcoholism are often deficient in magnesium, another important micronutrient. Its deficiency can lead to cardiovascular problems and severe neuromuscular issues. Common health effects include seizures and depression. Since alcohol consumption can increase magnesium loss through the kidneys, monitoring and supplementing magnesium is recommended. Similarly, it is crucial to obtain zinc supplements because zinc deficiency has been linked to weakened immunity and delayed wound healing. Zinc plays a role in over 300 metabolic enzyme functions that rebuild tissues, and replenishing this mineral may help with recovery from illness[15,37].
Eventually, managing nutritional care in patients with ALD requires a comprehensive approach that includes dietary changes and supplementation of both macro and micronutrients based on individual needs determined through clinical evaluation. Early identification of malnutrition risk helps improve health and reduces the risk of complications from advanced liver disease.
Due to the increasing rates of obesity and metabolic syndrome, NAFLD has become a widespread issue. Effectively managing this condition requires weight loss along with lifestyle changes, where even a small amount of weight loss can significantly benefit the liver. Studies show that losing 5%-10% of body weight can improve liver enzymes and reduce fatty liver. The Mediterranean diet or the dietary approaches to stop hypertension (DASH) plan appears to help patients with NAFLD[33,37]. These diets include fruits, vegetables, grains, nuts, and legumes, along with healthy fats like olive oil. They promote increased intake of mono- and polyunsaturated fats while reducing saturated and trans fats, sup
A key consideration is the urgent need to reduce saturated fats and simple sugars found in many Western diets. These elements decrease insulin sensitivity and increase the risk of fatty liver. Nutritional tips for NAFLD should focus on lowering saturated fats and full-fat dairy products, as well as reducing simple sugar intake. Another piece of advice is to consume foods high in fiber, as they enhance insulin sensitivity, help control blood sugar levels, and promote satiety, which can aid in weight loss[39]. Furthermore, NAFLD patients should adopt lifestyle modifications. Regular exercise helps with weight loss. Therefore, combining a healthy diet with physical activity improves liver health[36,40].
Nutritional management in patients with CLD due to viral hepatitis is essential to slow viral replication, hepatocellular injury, oxidative stress, and fibrosis. In patients with chronic viral hepatitis, inflammatory status impacts many metabolic pathways, including carbohydrate, lipid, and protein metabolism. These changes make patients with viral hepatitis more susceptible to protein-calorie malnutrition, sarcopenia, and vitamin deficiencies[37,40]. Additionally, detoxification abilities are impaired, leading to increased ammonia accumulation and by-products of metabolism. This buildup of metabolites can worsen symptoms such as anorexia, fatigue, and reduced food intake. Patients with HBV or HCV may also develop insulin resistance and diabetes, which can alter their overall nutritional needs. Overweight status and alcohol consumption, especially in HCV patients, can worsen the disease, accelerate hepatic fibrosis, and promote hepatic steatosis[38].
In viral hepatitis, the nutritional plan aims to decrease fibrosis and steatosis progression, preserve lean body mass, prevent sarcopenia, correct deficiencies in macronutrients and micronutrients, and address insulin resistance and obesity. Patients should follow their routine supplementation since deficiencies in vitamin D, folate, thiamine, and pyridoxine are common. Additionally, every nutritional plan should be initiated as early as possible and tailored to each patient to ensure better tolerance and a slower rate of liver complications. Guidelines recommend a daily caloric intake of 30-35 kcal/kg/day, with protein intake of about 1.2-1.5 g/kg/day. For those with insulin resistance, following specific diet plans like DASH combined with regular daily exercise to lose about 5%-10% of weight appears to improve their condition[41-43].
In cholestatic CLD, the main issue is fat-malabsorption caused by decreased bile secretion, which is essential for fat digestion and absorption. Additionally, patients with cholestatic CLD may develop portal hypertension, which can impair pancreatic enzyme secretion, cause intestinal dysmotility, and lead to bacterial overgrowth, all of which affect nutrient absorption. This results in lower levels of essential fatty acids and polyunsaturated fatty acids. Guidelines recommend a daily caloric intake of about 35-40 kcal/kg/day and 1.2-1.5 g/kg/day respectively. Fat intake should ideally make up 30%-35% of total calories, with caution to avoid excessive fat consumption and minimize adverse effects. If CLD patients experience steatorrhea, MCTs are recommended because they do not require hydrolysis for absorption[26,43]. Cholestatic CLD may also influence the progression of osteopenia and osteoporosis due to decreased vitamin D and calcium levels. Therefore, vitamin D supplements are advised, with daily calcium intake roughly 800-1500 mg/day[4,44] (Figure 1).
In autoimmune hepatitis (AIH), nutritional status is quite complex. Although patients with AIH may have normal BMI, body weight, midarm circumference, and triceps skinfold thickness-indicators that may suggest a normal nutritional state-AIH patients often have decreased levels of fat-soluble vitamins, especially vitamins A, D, and E. Energy tolerance in AIH patients is lower than usually expected for CLD, and dietary lipid intake may be about 29.82%. Guidelines recommend maintaining sufficient caloric intake in AIH patients and correcting any vitamin deficiencies[40-44].
Wilson disease (WD) is an autosomal-recessive disorder affecting copper metabolism. The goal of nutritional support in WD patients is to manage copper buildup and correct deficiencies. Since WD is a multisystem disorder impacting organs such as the liver, brain, bone, and endocrine system, it can significantly influence the body’s nutritional status. The key measure is to restrict copper in the patients’ diet. Foods high in copper include chocolate, nuts, liver, shellfish, and mushrooms. Zinc supplementation is vital in WD because it inhibits copper absorption by stimulating metallothionein production in enterocytes. Additionally, vitamin D is important because patients may develop osteopenia and oste
Pediatric patients: Children with long-term liver disease are special because their bodies need a lot of energy and they are still growing. Children with cystic fibrosis face a higher risk of growth failure, malnutrition, delayed puberty, and deficiency of fat-soluble vitamins. They usually need to eat more than most healthy peers, often requiring 120%-150% of their age-adjusted energy needs. To prevent developmental problems that are difficult to fix later, early nutritional interventions during the first years of life are crucial. Guidelines recommend supplementation with vitamins A, D, E, and K, using energy-dense formulas, and monitoring linear growth[44].
Geriatric patients: Fatigue, weakness, poor muscle health, movement problems, and deficiencies in vitamins and minerals are common in elderly patients with liver disease. Age-related anorexia in older adults results from various factors, such as altered taste perception and polypharmacy. Focus on adequate protein intake (≥ 1.2 g/kg/day); resistance or strength-training exercises when possible; small, frequent meals and late evening snacks to minimize fasting-related catabolism; and correcting micronutrient deficiencies, especially vitamin D, zinc, and B vitamins. Due to lower reserves, tailored strategies to support independence and improve quality of life are essential[44,45].
Liver transplant candidates: Poor outcomes after surgery are strongly linked to pre-transplant malnutrition. Increased risks include infections, prolonged ICU stays, and delayed wound healing. Improving nutritional practices before trans
HE: It is no longer recommended to prevent protein intake, even in cases of overt HE, as doing so can worsen sarcopenia and increase ammonia production from muscle degradation. Patients should receive 1.0-1.5 g/kg/day of protein, with an emphasis on plant-based and dairy proteins. Additionally, BCAA-enriched formulations can be administered when needed, especially in cases of recurrent or persistent HE. Long intervals between meals should be avoided. Maintaining a positive nitrogen balance, which supports metabolic functions and reduces catabolic stress, is essential. A late-evening snack should be included in the nutritional plan for every patient[45].
Upper GI bleeding: Patients recovering from variceal or non-variceal upper GI bleeding should be encouraged to re-initiate nutrition as soon as possible, usually within the following 24-48 hours of hemodynamic stability. Consuming enough calories that are high in protein helps mucosa to heal and reduces muscle loss. EN is preferred over parenteral routes because of low infection risk and maintenance of gut integrity. Once bleeding is controlled, it is generally safe to feed intubated or severely ill patients via nasogastric tube early on[44,45].
Customized nutritional strategies are essential for patients with CLD due to various challenges. A key concern is managing nutrition in HE. A transjugular intrahepatic portosystemic shunt (TIPS) in selected patients with cirrhosis can help reduce sarcopenia by lowering portal hypertension and enhancing splanchnic perfusion, nutrient absorption, and resting energy expenditure. Research indicates that muscle mass, strength, and function improve over 6-12 months, especially in patients with refractory ascites. TIPS supports this by decreasing hypertension, which relaxes blood vessels and can reduce pain. Although TIPS is not a primary nutritional treatment, it can be beneficial in managing sarcopenia when part of a comprehensive approach in patients with cirrhosis. Patients with HE should generally maintain adequate protein intake, usually 1.2-1.5 g/kg/day, to help preserve muscle mass. BCAA-rich diets might help patients with severe protein intolerance or to meet recommended nitrogen intake. BCAAs (leucine, isoleucine, and valine) are often deficient in CLD, while aromatic amino acids tend to be elevated[45].
Sodium restriction is critical for patients with ascites, usually limited to 2 g/day to control fluid retention. However, strict restrictions can reduce food palatability, leading to decreased intake and worsening malnutrition[44,45]. Fluid restriction is typically reserved for severe hyponatremia (serum sodium < 120-125 meq/L). Managing ascites often involves limiting sodium intake to less than 2 g/day, which can be challenging due to hidden sodium, low nutritional literacy, and cultural food practices. Some effective methods include using low-sodium flavorings (herbs, lemon, garlic, spices), avoiding high-sodium foods (processed meats, pickles, canned soups, fast food), and eating homemade meals. Patients should be provided with meal plans, shopping lists, and recipe modifications that are culturally familiar, which can improve long-term adherence. These strategies enhance sodium restriction and support better ascites management of ascites. Patients with CLD often experience obesity, which can mask malnutrition. Obesity may increase the risk of hepatic steatosis and fibrosis. Lifestyle modifications, including healthy diet plans and daily exercise, are essential[46]. Besides decreasing insulin sensitivity in CLD, obesity can progress to diabetes, which, as noted earlier, is common in CLD. Management may involve scheduled meals and regular follow-up to monitor blood glucose levels. In severe CLD cases with diabetes, insulin therapy might be necessary. Renal injury is also common in CLD and may require specific dietary adjustments, such as potassium and phosphorus restrictions. In cases of severe renal injury, protein restriction may be advised. As previously mentioned, if oral intake is inadequate, enteral or parenteral nutrition is recommended for CLD patients[43-46].
A multiprofessional team can help optimize the care of patients with CLD. However, personalized nutritional counseling is essential for the best outcomes. Since these patients have special needs, they must receive dietary advice tailored to their medical history, nutritional requirements, and lifestyles. A registered dietitian is an important professional who can assess a patient and design a meal plan according to clinical guidelines[21,46]. A multidisciplinary approach involving physicians, nurses, pharmacists, and dietitians is much more effective and improves outcomes more than individual practitioners. Multidisciplinary teams enhance communication and education, correcting errors of unnecessary dietary restrictions[24,47]. The goal is not only to give patients instructions but also to improve their knowledge by teaching them the specific daily amounts of each nutrient needed to support liver health. Counseling must be sensitive to both cultural and individual factors, such as the availability and preferences for food and multilingual resources. Regular follow-up is crucial; weekly check-ins allow for timely dietary adjustments[44-47].
Customizing dietary recommendations based on individual preferences of patients with CLDs can help improve meal adherence, enjoyment, and nutritional status. Allowing patients to express their food-related preferences and traditions enables dietitians to develop appealing and nutritious meal plans. Individuals with chronic conditions such as diabetes and hypertension can benefit greatly from personalized dietary plans. Using traditional recipes or preferred flavors may boost satisfaction without compromising medical goals. Strategies to make meals more enjoyable[3,47].
Eating is a fundamental source of human pleasure, and individuals often have a preferred meal they cherish. None
In CLD, continuous monitoring and follow-up are essential as they improve overall patient condition and management. The follow-up should begin with a comprehensive baseline assessment, including complete medical history, dietary habits, anthropometry, and drug history. SGA, MUAC, and triceps skinfold thickness are key tools in this process. For CLD patients, follow-up should occur every 2 weeks to 3 months, with a full examination to determine if the dietary plan remains appropriate. More severe cases may require more frequent assessments and follow-ups. Educating patients and using tools such as food diaries enable them to contribute to their care[47].
To address complications like ascites or GI bleeding, which might impair the patient's ability to eat or absorb food, specialized protocols are necessary. Enteral or parenteral nutrition may be required in such cases. Ultimately, any dietary changes should be documented and communicated with the entire care team to ensure continuity and the success of these modifications[1,48].
Nutritional management for CLD is an area of significant unmet needs and contains important gaps for further investigation. The most crucial gaps lie in the absence of standardized protocols for nutritional screening and assessment. Tools available today differ from institution to institution, which makes nutritional deficiency identification unclear. In the future, we should develop validated criteria that can be used universally in children and adults with CLD. This will help evaluate micronutrient deficiencies, sarcopenia, and malnutrition.
An important evidence gap exists regarding the long-term clinical effects of etiology-specific and personalized nutritional interventions. Although there is substantial evidence supporting personalized nutrition, randomized longitudinal trials are limited. More studies are needed to determine the best nutritional strategies to reduce conditions such as HE, ascites, and infections. Additionally, research should focus on whether individualized targets for macronutrients improve survival or reduce hospital admissions[47]. It must also establish how nutrition affects transplant-related outcomes and recovery after transplantation. Digital health and tech-based tools have become the latest innovations. Patients can be tracked more precisely and dynamically through remote dietary monitoring, mobile nutrition apps, and wearable devices. Future research should explore whether this technology can improve compliance, enable faster detection of eating problems, and allow for personalized, real-time adjustments. Conditions like anxiety and depression can influence eating behaviors and nutrient absorption. Investigating the effects of integrated psychosocial-nutritional interventions may reveal new pathways for patient-centered care, improving clinical outcomes. Collaborating to fill existing knowledge gaps will help enhance evidence-based dietary strategies and improve long-term patient outcomes[14,47].
More studies are examining the relationship between obesity, sarcopenia, and malnutrition in CLD. NAFLD continues to increase in prevalence, with evidence extending beyond simple measures like BMI, weight, and waist circumference. Advanced assessments such as dual-energy X-ray absorptiometry, magnetic resonance imaging-based quantification, and bioimpedance are now used for body composition analysis. Researchers are investigating how obesity and sarcopenia contribute to fibrosis progression and whether targeted nutrition, resistance exercise, and metabolic therapies can reverse muscle loss in obese patients with CLD.
Meanwhile, precision nutrition approaches incorporating metabolic profiling, microbiome signatures, and personalized diet plans are being tested for improved therapeutic outcomes. Factors such as food insecurity, socioeconomic barriers, cultural dietary practices, and limited access to specialized care remain important gaps to address in developing models of nutrition equity[48]. Artificial intelligence is also advancing, with machine-learning tools designed to automate body composition analysis, predict sarcopenia risk, and personalize nutritional recommendations-potentially enabling earlier, more targeted interventions[49].
In conclusion, effective nutritional management is a fundamental component of the few modifiable factors with the potential to improve clinical and quality-of-life outcomes in patients with CLD. Malnutrition, whether due to disease-related factors such as anorexia, malabsorption, or metabolic alterations, or external influences like socioeconomic challenges and outdated clinical practices, significantly impacts patient outcomes, including morbidity, transplantation eligibility, and mortality. Accurate assessment using tools like SGA, MUAC, and handgrip strength, alongside detailed diet histories, is essential for individualized intervention. Current guidelines recommend tailored caloric and protein intake, with nutritional support such as BCAAs, MCTs, and micronutrient supplementation employed as needed, and early consideration of enteral or parenteral nutrition in severe cases. A multidisciplinary approach-integrating medical, nutritional, psychological, and cultural care-is vital to optimize treatment adherence and overall health. Regular monitoring and flexible adjustment of dietary plans are crucial to address disease progression and complications like ascites or GI bleeding. Future research should focus on developing robust screening guidelines, exploring intervention outcomes, integrating technology, and investigating the connection between malnutrition, obesity, and sarcopenia, especially as NAFLD prevalence rises globally. Ultimately, shifting nutrition from a supportive role to a core therapeutic goal can improve patient outcomes, reduce healthcare burdens, and enhance quality of life in CLD management.
| 1. | Mouzaki M, Bronsky J, Gupte G, Hojsak I, Jahnel J, Pai N, Quiros-Tejeira RE, Wieman R, Sundaram S. Nutrition Support of Children With Chronic Liver Diseases: A Joint Position Paper of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr. 2019;69:498-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 72] [Article Influence: 10.3] [Reference Citation Analysis (3)] |
| 2. | Espina S, Casas-Deza D, Bernal-Monterde V, Domper-Arnal MJ, García-Mateo S, Lué A. Evaluation and Management of Nutritional Consequences of Chronic Liver Diseases. Nutrients. 2023;15:3487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (2)] |
| 3. | Shergill R, Syed W, Rizvi SA, Singh I. Nutritional support in chronic liver disease and cirrhotics. World J Hepatol. 2018;10:685-694. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 4. | Silva M, Gomes S, Peixoto A, Torres-Ramalho P, Cardoso H, Azevedo R, Cunha C, Macedo G. Nutrition in Chronic Liver Disease. GE Port J Gastroenterol. 2015;22:268-276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 5. | Mendez-Guerrero O, Carranza-Carrasco A, Chi-Cervera LA, Torre A, Navarro-Alvarez N. Optimizing nutrition in hepatic cirrhosis: A comprehensive assessment and care approach. World J Gastroenterol. 2024;30:1313-1328. [PubMed] [DOI] [Full Text] |
| 6. | Pelton M, Abdel-Meguid S, Goradia E, Bussetty A, Cohen D, Kesavarapu K. Role of Nutrition in the Management of Chronic Liver Disease. Gastro Hep Adv. 2025;4:100613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 7. | Kamran U, Towey J, Khanna A, Chauhan A, Rajoriya N, Holt A. Nutrition in alcohol-related liver disease: Physiopathology and management. World J Gastroenterol. 2020;26:2916-2930. [PubMed] [DOI] [Full Text] |
| 8. | Gan C, Yuan Y, Shen H, Gao J, Kong X, Che Z, Guo Y, Wang H, Dong E, Xiao J. Liver diseases: epidemiology, causes, trends and predictions. Signal Transduct Target Ther. 2025;10:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 137] [Article Influence: 137.0] [Reference Citation Analysis (1)] |
| 9. | Sharma A, Nagalli S. Chronic liver disease. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 10. | Tadokoro T, Morishita A, Himoto T, Masaki T. Nutritional Support for Alcoholic Liver Disease. Nutrients. 2023;15:1360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 11. | Rossi RE, Conte D, Massironi S. Diagnosis and treatment of nutritional deficiencies in alcoholic liver disease: Overview of available evidence and open issues. Dig Liver Dis. 2015;47:819-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 12. | Sierra L, Duong N. Fueling the failing liver-ACG clinical guideline: Malnutrition and nutritional recommendations in liver disease. Am J Gastroenterol. 2025;120:950-972. |
| 13. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on nutrition in chronic liver disease. J Hepatol. 2019;70:172-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 900] [Cited by in RCA: 789] [Article Influence: 112.7] [Reference Citation Analysis (2)] |
| 14. | Yang CH, Perumpail BJ, Yoo ER, Ahmed A, Kerner JA Jr. Nutritional Needs and Support for Children with Chronic Liver Disease. Nutrients. 2017;9:1127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 15. | Meyer F, Bannert K, Wiese M, Esau S, Sautter LF, Ehlers L, Aghdassi AA, Metges CC, Garbe LA, Jaster R, Lerch MM, Lamprecht G, Valentini L. Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis. Int J Mol Sci. 2020;21:5357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (2)] |
| 16. | Krenitsky J. Nutrition for patients with hepatic failure. Pract Gastroenterol. 2003;130:428-432. |
| 17. | Ali H, Shahzil M, Moond V, Shahzad M, Thandavaram A, Sehar A, Waseem H, Siddiqui T, Dahiya DS, Patel P, Tillmann H. Non-Pharmacological Approach to Diet and Exercise in Metabolic-Associated Fatty Liver Disease: Bridging the Gap between Research and Clinical Practice. J Pers Med. 2024;14:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 18. | Jamioł-Milc D, Gudan A, Kaźmierczak-Siedlecka K, Hołowko-Ziółek J, Maciejewska-Markiewicz D, Janda-Milczarek K, Stachowska E. Nutritional Support for Liver Diseases. Nutrients. 2023;15:3640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 19. | Matos C, Porayko MK, Francisco-Ziller N, DiCecco S. Nutrition and chronic liver disease. J Clin Gastroenterol. 2002;35:391-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 70] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 20. | Elsebaie EM, Abdel-Fattah AN, Bakr NA, Attalah KM, Aweas AA. Principles of Nutritional Management in Patients with Liver Dysfunction-A Narrative Review. Livers. 2023;3:190-218. [DOI] [Full Text] |
| 21. | Choe U. Nutritional Management for Pediatric Biliary Atresia Patients Preparing for Liver Transplantation. Children (Basel). 2025;12:391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 22. | Cheung K, Lee SS, Raman M. Prevalence and mechanisms of malnutrition in patients with advanced liver disease, and nutrition management strategies. Clin Gastroenterol Hepatol. 2012;10:117-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 245] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 23. | Bischoff SC, Bernal W, Dasarathy S, Merli M, Plank LD, Schütz T, Plauth M. ESPEN practical guideline: Clinical nutrition in liver disease. Clin Nutr. 2020;39:3533-3562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 242] [Article Influence: 40.3] [Reference Citation Analysis (5)] |
| 24. | Utakata Y, Miwa T, Hanai T, Aiba M, Unome S, Imai K, Shirakami Y, Takai K, Shimizu M. Usefulness of Retinol-Binding Protein in Predicting Mortality in Patients With Chronic Liver Disease. JGH Open. 2025;9:e70087. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 25. | Patton HM. Nutritional assessment of patients with chronic liver disease. Gastroenterol Hepatol (N Y). 2012;8:687-690. [PubMed] |
| 26. | Aller de la Fuente R. Nutrition and Chronic Liver Disease. Clin Drug Investig. 2022;42:55-61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 27. | Henkel AS, Buchman AL. Nutritional support in patients with chronic liver disease. Nat Clin Pract Gastroenterol Hepatol. 2006;3:202-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 91] [Article Influence: 4.6] [Reference Citation Analysis (2)] |
| 28. | Juakiem W, Torres DM, Harrison SA. Nutrition in cirrhosis and chronic liver disease. Clin Liver Dis. 2014;18:179-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 29. | Santangeli E, Abbati C, Chen R, Di Carlo A, Leoni S, Piscaglia F, Ferri S. Pathophysiological-Based Nutritional Interventions in Cirrhotic Patients with Sarcopenic Obesity: A State-of-the-Art Narrative Review. Nutrients. 2024;16:427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 30. | Singal AK, Charlton MR. Nutrition in alcoholic liver disease. Clin Liver Dis. 2012;16:805-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 31. | DiCecco SR, Francisco-Ziller N. Nutrition in alcoholic liver disease. Nutr Clin Pract. 2006;21:245-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 32. | Choi YR, Kim HS, Yoon SJ, Lee NY, Gupta H, Raja G, Gebru YA, Youn GS, Kim DJ, Ham YL, Suk KT. Nutritional Status and Diet Style Affect Cognitive Function in Alcoholic Liver Disease. Nutrients. 2021;13:185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 33. | Yasutake K, Kohjima M, Nakashima M, Kotoh K, Nakamuta M, Enjoji M. Nutrition therapy for liver diseases based on the status of nutritional intake. Gastroenterol Res Pract. 2012;2012:859697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 34. | Best SA, De Souza DP, Kersbergen A, Policheni AN, Dayalan S, Tull D, Rathi V, Gray DH, Ritchie ME, McConville MJ, Sutherland KD. Synergy between the KEAP1/NRF2 and PI3K Pathways Drives Non-Small-Cell Lung Cancer with an Altered Immune Microenvironment. Cell Metab. 2018;27:935-943.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 172] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 35. | Berná G, Romero-Gomez M. The role of nutrition in non-alcoholic fatty liver disease: Pathophysiology and management. Liver Int. 2020;40 Suppl 1:102-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 176] [Article Influence: 29.3] [Reference Citation Analysis (2)] |
| 36. | Whelan K, Staudacher H. Low FODMAP diet in irritable bowel syndrome: a review of recent clinical trials and meta-analyses. Curr Opin Clin Nutr Metab Care. 2022;25:341-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 37. | Puri P, Dhiman RK, Taneja S, Tandon P, Merli M, Anand AC, Arora A, Acharya SK, Benjamin J, Chawla YK, Dadhich S, Duseja A, Eapan CE, Goel A, Kalra N, Kapoor D, Kumar A, Madan K, Nagral A, Pandey G, Rao PN, Saigal S, Saraf N, Saraswat VA, Saraya A, Sarin SK, Sharma P, Shalimar, Shukla A, Sidhu SS, Singh N, Singh SP, Srivastava A, Wadhawan M. Nutrition in Chronic Liver Disease: Consensus Statement of the Indian National Association for Study of the Liver. J Clin Exp Hepatol. 2021;11:97-143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (4)] |
| 38. | Menta PL, Correia MI, Vidigal PV, Silva LD, Teixeira R. Nutrition status of patients with chronic hepatitis B or C. Nutr Clin Pract. 2015;30:290-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 39. | Nompleggi DJ, Bonkovsky HL. Nutritional supplementation in chronic liver disease: an analytical review. Hepatology. 1994;19:518-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 69] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 40. | Saron ML, Godoy HT, Hessel G. Nutritional status of patients with biliary atresia and autoimmune hepatitis related to serum levels of vitamins A, D and E. Arq Gastroenterol. 2009;46:62-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 41. | Saroli Palumbo C, Schilsky ML. Clinical practice guidelines in Wilson disease. Ann Transl Med. 2019;7:S65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 42. | von Schönfels W, Patsenker E, Fahrner R, Itzel T, Hinrichsen H, Brosch M, Erhart W, Gruodyte A, Vollnberg B, Richter K, Landrock A, Schreiber S, Brückner S, Beldi G, Sipos B, Becker T, Röcken C, Teufel A, Stickel F, Schafmayer C, Hampe J. Metabolomic tissue signature in human non-alcoholic fatty liver disease identifies protective candidate metabolites. Liver Int. 2015;35:207-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 43. | Lai JC, Tandon P, Bernal W, Tapper EB, Ekong U, Dasarathy S, Carey EJ. Malnutrition, Frailty, and Sarcopenia in Patients With Cirrhosis: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1611-1644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 575] [Cited by in RCA: 525] [Article Influence: 105.0] [Reference Citation Analysis (1)] |
| 44. | Iwasa M, Iwata K, Hara N, Hattori A, Ishidome M, Sekoguchi-Fujikawa N, Mifuji-Moroka R, Sugimoto R, Fujita N, Kobayashi Y, Takei Y. Nutrition therapy using a multidisciplinary team improves survival rates in patients with liver cirrhosis. Nutrition. 2013;29:1418-1421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 67] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 45. | Noureddin M, Harrison SA, Alkhouri N. MEFIB vs. MAST and FAST: Not a competition but useful tools. J Hepatol. 2024;80:e35-e36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 46. | Xian ZX, Wang X, Chen YC, Teng YS. Preliminary assessment of portable sleep monitoring for diagnosis of obstructive sleep apnea in children. Sleep Breath. 2024;28:419-425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 47. | Tandon P, Raman M, Mourtzakis M, Merli M. Sarcopenia in cirrhosis: from pathogenesis to clinical management. Hepatology. 2017;5:043-1057. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 48. | Rodríguez Villafuerte S. Editorial: Nutritional approaches in chronic liver diseases. Front Nutr. 2024;11:1480541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 49. | Jeong CW, Lim DW, Noh SH, Lee SH, Park C. Development of an artificial intelligence-based application for the diagnosis of sarcopenia: a retrospective cohort study using the health examination dataset. BMC Med Inform Decis Mak. 2025;25:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |