Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.114279
Revised: December 17, 2025
Accepted: January 22, 2026
Published online: March 27, 2026
Processing time: 192 Days and 6.9 Hours
Frailty has been associated with mortality among patients referred for liver trans
To investigate the effect of physical frailty, measured by the liver frailty index (LFI), on the likelihood of LT, pre- and early post-LT outcomes, in an Australian cohort.
Data were collected on adults with cirrhosis referred for LT and had their baseline LFI assessment. Outcomes of interest included: Receiving a LT, pre-LT unplanned hospitalizations, LT surgical complications, intensive care unit (ICU) and hospital length of stay. Cox proportional hazards modelling determined associations between LFI and outcomes, adjusting for age, sex, hepatocellular carcinoma, and model for end-stage liver disease score. Competing risk analysis explored reasons for not being transplanted including waitlist mortality [sub-hazards ratio (HR)].
Among 266 patients [median model for end-stage liver disease 16 (interquartile range 11-19)], the median LFI was 3.7 (3.3-4.1); 19% were robust, 68% pre-frail, and 14% frail. After adjustment, each 1-point increase in the LFI was associated with a 29% lower likelihood of receiving a LT [HR = 0.71, 95% confidence interval (CI): 0.54-0.94, P = 0.020]. Competing-risk analysis showed higher LFI increased waitlist mortality (sub-HR = 1.90, 95%CI: 1.33-2.70). Each 1-point rise also conferred a 69% higher risk of unplanned pre-LT hospitalization (HR = 1.69, 95%CI: 1.09-2.62, P = 0.020). Among transplanted patients, higher LFI predicted prolonged ICU stay (> 4 days; odds ratio = 3.24, 95%CI: 1.06-9.85). Frailty was not associated with surgical complications or hospital length of stay.
Physical frailty independently predicts reduced LT likelihood, higher waitlist mortality, greater pre-LT unplanned hospitalizations, and prolonged ICU stay. This study provides the first Australian validation, extending LFI evi
Core Tip: Frailty is increasingly recognized as a predictor of adverse outcomes in liver transplantation (LT), yet evidence using the liver frailty index (LFI) outside the United States is limited. This study is the first to evaluate the LFI in an Australian LT cohort, demonstrating its ability to identify patients at higher risk of waitlist mortality, unplanned hospitalizations, and prolonged intensive care unit stay after LT. These findings validate the LFI as a valuable tool for risk stratification, supporting its integration into LT assessment to guide surveillance and resource allocation for interventions.
- Citation: Johnston HE, Mayr HE, Andelkovic ME, Takefala TG, Chen Y, Thrift AP, Hickman IJ, Macdonald GA. Clinical burden of physical frailty in patients evaluated for liver transplantation: A retrospective cohort study. World J Hepatol 2026; 18(3): 114279
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/114279.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.114279
Physical frailty is increasingly recognized as an important prognostic factor in patients with advanced liver disease (cirrhosis). Frailty is a state of global physiological decline and vulnerability to stressors[1], and is linked to reduced strength, poor physical functioning and mobility, impaired balance and gait speed[2], and elements of cognitive dys
While frailty was initially described in geriatrics, it is now being examined in chronic diseases like cirrhosis, where malnutrition and cognitive impairment lead to the emergence of a frail phenotype[6]. The current practice guidance on frailty, recommends patients with cirrhosis should be assessed for frailty[2], with an expert opinion panel emphasizing the importance of this in patients undergoing liver transplantation (LT) evaluation[7]. Despite this, the implementation of a validated frailty instrument into clinical practice is not universal[7].
There are several instruments that could be used to assess physical frailty in patients with cirrhosis. At present the liver frailty index (LFI) is the most widely studied and expert opinion from the transplant community supports the use of the LFI for assessing physical frailty[7]. The LFI is a sex-specific composite metric incorporating measures of hand grip strength; HGS (the average of three readings of the dominant hand), time taken to complete five chair stands and three balance positions[8]. It provides both a continuous index score (0-7) with higher scores indicating a greater degree of frailty and three distinct categories robust (< 3.20), pre-frail (3.20-4.50), or frail (> 4.50).
Baseline LFI is associated with increased LT waitlist mortality independent of model for end-stage liver disease (MELD) scores[8] and is an accurate and objective assessment of a patient’s health status compared to a clinician’s subjective assessment[9]. Furthermore, the LFI is a simple tool that can be incorporated into clinical assessments, with excellent inter and intra-rater reliability and reproducibility[10,11]. While there is a link between physical frailty and waitlist mortality, the link with other important clinical outcomes is less clear. Additionally, most current research on the utility of the LFI has come from one large United States cohort[12]. The aims of this study therefore were to explore the impact of physical frailty (LFI), on pre and early post-LT outcomes in an Australian cohort of patients referred for LT.
This single center cohort study included patients from the Queensland Liver Transplant Service, Australia. The study was approved by the Medical Ethics Committee of Metro South Health, approval No. HREC/2018/QMS/46728 in accordance with the Declaration of Helsinki and Istanbul. As data collected was part of routine care, a waiver of consent was provided. This study was reported in accordance with the STROBE guidelines[13]. Adults (> 18 years) assessed for LT between March 2018 and September 2022 were eligible. Patients were included if they had the LFI[8] completed at their baseline dietetic consultation. Excluded patients were re-transplant, multiple simultaneous organ transplants, or those who did not have cirrhosis. Inpatients were excluded unless the primary indication for admission was for LT evaluation, and they were medically stable.
Primary outcomes were: (1) The likelihood of receiving a LT (and competing risks related to not being transplanted); and (2) Unplanned admissions to hospital pre-LT. Secondary outcomes were intensive care unit (ICU) and hospital length of stay (LOS) and major surgical transplant complications[8]. Electronic medical records were accessed to obtain clinical and laboratory data, baseline dietetic assessments, data on unplanned admissions and the LT admission. Clinical data included age, sex, etiology of liver disease, MELD score[14], presence of hepatocellular carcinoma (HCC), ascites, and a documented diagnosis of type 2 diabetes. Anthropometric assessments included weight in kilograms (digital scales, Tanita, Japan) and height in meters (wall-mounted stadiometer) to allow calculation of body mass index (kg/m2). If ascites was present, dry weight was estimated from last weight post-paracentesis, or by subtracting 5%, 10%, or 15% of total body weight for mild, moderate or severe ascites, respectively as previously described[15]. An additional 5% was subtracted if oedema was present. Malnutrition was determined by the modified subjective global assessment for cirrhosis[16].
The LFI was calculated using an online calculator[8]. The LFI uses the average of three measurements of HGS; time spent standing in three balance positions (side by side, semi-tandem and tandem); and the time taken to complete five chair-stands. A calibrated digital Jamar dynamometer was used to take the HGS. The LFI is a continuous variable (0-7) that classifies patients into three categories as well as provides a continuous index: Robust (< 3.20), pre-frail (3.20-4.50), or frail (> 4.50). We have demonstrated dietitians can reliably perform the LFI in clinical settings[10].
Pre-LT data included the impact of frailty on the likelihood of receiving a LT and unplanned admissions whilst un
If patients had an unplanned admission pre-LT, the time in days to first unplanned admission was recorded. Un
For LT recipients, the durations of ICU and total hospital LOS, and the presence of major surgical complications during transplant admission were recorded. Complications were categorized according to the Clavien-Dindo grading system for post-operative surgical complications[18]. ICU and hospital LOS were grouped into ≤ or > 4 days and ≤ or > 18 days, respectively, as per proposed benchmarks for prolonged LOS after LT[19].
Descriptive analyses were conducted on demographic and clinical variables. Values presented as n (%), and either mean ± SD (normally distributed data), or median [interquartile range (IQR)] (non-normally distributed). Normality was assessed using Shapiro Wilks test with visual interpretation of normality plots. The primary predictor was frailty (LFI score and category). Mann-Whitney U and χ2 tests were used to explore associations with frailty. One way analysis of variance compared continuous variables across frailty categories. Binary logistic regression analyses were performed for the LFI with the pre and early post-LT outcomes. Multivariable analyses adjusted for confounders (age, sex, presence of HCC and MELD score).
Survival analysis included Kaplan-Meier curves and log-rank tests to examine the relationship between frailty and: (1) Time from initial clinic appointment until LT; and (2) Time from initial clinic appointment until first unplanned ad
Significance was set at P < 0.05. P values are reported to three decimal places, with P < 0.001 shown for values below this threshold. Statistical analyses were carried out using IBM SPSS Statistics, version 28.0 (IBM Armonk, NY: IBM Corp) and R Studio statistical software (version 4.1.2, R Core Team 2021, Austria).
Of the 274 patients seen following LT referral, 266 (97%) patients had baseline LFI data available and were included in the final study sample. Table 1 provides the demographic and clinical characteristics of the cohort.
| Characteristic | Study population (n = 266) |
| Age (years) | 58 (50-63) |
| Sex, male | 200 (75) |
| MELD score | 16 (11-19) |
| Dry weight (kg) | 83 (72-99) |
| BMI kg/m2 [BMI categories (based on estimated dry weight1)] | 27 (24-32) |
| Underweight (≤ 18.49 kg/m2) | 7 (3) |
| Healthy (18.5-24.9 kg/m2) | 82 (30) |
| Overweight (25-29.9 kg/m2) | 88 (33) |
| Obese (≥ 30 kg/m2) | 89 (33) |
| Aetiology of liver disease | |
| Alcohol-related liver disease | 103 (39) |
| Chronic viral hepatitis (HCV/HBV) | 61 (23) |
| MASLD | 46 (17) |
| PBC/PSC | 36 (13) |
| AIH | 6 (2) |
| Cryptogenic | 4 (2) |
| Other2 | 10 (4) |
| Those with hepatocellular carcinoma | 73 (27) |
| Type two diabetes | 64 (24) |
| Ascites present | 91 (34) |
| SGA score | |
| A (well nourished) | 179 (67) |
| B (moderately malnourished) | 78 (30) |
| C (severely malnourished) | 9 (3) |
| Frailty | |
| LFI | 3.7 (3.3-4.1) |
| Robust (< 3.2) | 50 (19) |
| Pre-frail (3.2-4.5) | 179 (67) |
| Frail (> 4.5) | 37 (14) |
| Liver transplant recipients3 | 144 (54) |
Table 2 details the median LFI and frailty categories according to various demographic and clinical variables. There were significant differences in LFI and frailty categories in patients with and without HCC and type 2 diabetes, and those malnourished vs well nourished. The median MELD score increased with degree of frailty (P < 0.001). There was no significant association between LFI and sex, age, dry weight or body mass index.
| Characteristic grouping | Median LFI (IQR) | P value | Robust | Pre-frail | Frail | P value |
| Categorical variables | ||||||
| With HCC (n = 73) | 3.5 (3.2-3.8) | < 0.001 | 23 (46) | 45 (25) | 5 (14) | 0.002 |
| Without HCC (n = 193) | 3.8 (3.4-4.2) | 27 (54) | 134 (75) | 32 (86) | ||
| Well nourished (n = 179) | 3.5 (3.2-3.9) | < 0.001 | 45 (90) | 122 (68) | 12 (32) | < 0.001 |
| Moderately-severely malnourished | 4.2 (3.7-4.6) | 5 (10) | 57 (32) | 25 (68) | ||
| Type two diabetes (n = 64) | 3.9 (3.5-4.3) | 0.020 | 4 (8) | 47 (26) | 13 (35) | 0.007 |
| No type two diabetes (n = 202) | 3.6 (3.2-4.0) | 46 (92) | 132 (74) | 24 (65) | ||
| Male (n = 199) | 3.7 (3.3-4.1) | 0.750 | 39 (78) | 131 (73) | 29 (78) | 0.680 |
| Female (n = 67) | 3.8 (3.3-4.2) | 11 (22) | 48 (27) | 8 (22) | ||
| Obese (BMI > 30 kg/m2) (n = 89) | 3.8 (3.4-4.2) | 0.210 | 14 (28) | 59 (33) | 16 (43) | 0.320 |
| Non-obese (BMI < 30 kg/m2) | 3.6 (3.3-4.1) | 36 (72) | 120 (67) | 21 (57) | ||
| Continuous variables | ||||||
| MELD score, median (IQR) | - | - | 12 (9-16) | 16 (11-19) | 19 (15-23) | < 0.001 |
| Age (years), median (IQR) | - | - | 57 (49-63) | 58 (50-62) | 60 (51-64) | 0.270 |
| Dry weight (kg), median (IQR) | - | - | 82 (73-95) | 83 (71-100) | 85 (75-101) | 0.770 |
One hundred and forty-four (54%) patients received a LT with two patients dying at the time of LT. Of the remaining 122 patients, 61 (23%) were in the waitlist mortality group, 29 (11%) were discharged from the service when their liver disease improved, and 32 (12%) left the service for other reasons (personal choice, return to substance use or still awaiting LT). The baseline LFI in LT recipients was significantly lower, indicating a lesser degree of frailty, than in those who were not transplanted [3.6 (3.3-4.0) vs 3.8 (3.4-4.2), P = 0.010].
Kaplan-Meier analysis (Supplementary Figure 1) demonstrated a significant difference across the frailty categories in the time to receive a LT or otherwise leave the Queensland Liver Transplant Service (P = 0.040). Adjusted Cox proportional hazards analysis found that for every 1-point increase in the LFI, the likelihood of LT decreased by 29% (HR = 0.71, 95%CI: 0.54-0.94, P = 0.020). When analysed with frailty categories, the likelihood of receiving a LT for pre-frail and frail patients was significantly lower than for robust patients (HR = 0.63, 95%CI: 0.45-0.86, P = 0.004). Cox proportional hazards analysis also identified the MELD score was an independent factor influencing the likelihood of LT. Specifically, every 1-point increase in the MELD was associated with an increased likelihood of receiving a LT (HR = 1.04, 95%CI: 1.00-1.08, P = 0.020).
Competing risk analysis provided reasons for the lower likelihood of LT (Supplementary Table 1). A positive association with LFI and waitlist mortality was demonstrated. For every 1-point increase in the LFI, there was a 90% increased risk of waitlist mortality (sHR = 1.90, 95%CI: 1.33-2.70, P < 0.001). Frail patients also had a significantly greater risk of waitlist mortality compared to robust patients (sHR = 4.92, 95%CI: 1.79-13.50, P = 0.002). Pre-frail patients showed a similar but not significant trend towards higher waitlist mortality risk (sHR = 1.99, 95%CI: 0.83-4.77, P = 0.100). Physical frailty was not associated with the competing risk of being discharged from the service because of improvement in liver disease (sHR = 1.26, 95%CI: 0.67-2.34, P = 0.470), or for leaving the service for other reasons (sHR = 0.64, 95%CI: 0.36-1.12, P = 0.120).
Thirty-nine percent of patients experienced at least one unplanned hospital admission prior to LT. Unplanned ad
| Total cohort (n = 266) | Continuous LFI median (IQR) | Univariable analysis | Multivariable analysis | ||||
| Beta value/co-efficient (SE) | OR (95%CI) | P value | Beta value/co-efficient (SE) | OR (95%CI) | P value | ||
| Pre transplant | |||||||
| LT recipient | -0.47 (0.19) | 0.63 (0.44-0.90) | 0.010 | -0.62 (0.21) | 0.54 (0.36-0.81) | 0.003 | |
| Yes (n = 144) | 3.6 (3.3-4.0) | ||||||
| No (n = 122) | 3.8 (3.4-4.2) | ||||||
| Unplanned hospital admission | 0.85 (0.20) | 2.34 (1.56-3.49) | < 0.001 | 0.52 (0.22) | 1.69 (1.09-2.62) | 0.020 | |
| No (n = 161) | 3.6 (3.2-3.9) | ||||||
| Yes (n = 105) | 3.9 (3.5-4.3) | ||||||
| Early post-transplant complications | |||||||
| Grade ≥ 3 post op complication1,2 | 0.37 (0.26) | 1.45 (0.87-2.41) | 0.200 | 0.71 (0.30) | 1.19 (0.66-2.12) | 0.600 | |
| No (n = 72) | 3.5 (3.1-4.0) | ||||||
| Yes (n = 72) | 3.7 (3.4-4.1) | ||||||
| ICU LOS > 4 days3 | 0.87 (0.48) | 2.38 (0.94-6.05) | 0.070 | 1.17 (0.56) | 3.24 (1.06-9.85) | 0.040 | |
| No (n = 129) | 3.6 (3.3-4.0) | ||||||
| Yes (n = 13) | 3.9 (3.5-4.8) | ||||||
| Hospital LOS > 18 days3 | 0.77 (0.35) | 2.16 (1.09-4.28) | 0.030 | 0.68 (0.39) | 1.98 (0.92-4.26) | 0.080 | |
| No (n = 114) | 3.5 (3.2-4.0) | ||||||
| Yes (n = 28) | 3.8 (3.6-4.2) | ||||||
Multivariable analysis of frailty categories (Table 4) demonstrated frail patients had a 3.07-fold (95%CI: 1.06-8.90) and pre-frail patients a 2.49-fold (95%CI: 1.08-5.72) increased odds of having an unplanned admission compared to robust patients (P = 0.040 and P = 0.030 respectively). There was no association between LFI and unplanned admissions for specific indications (hepatic encephalopathy, etc.).
| Clinical outcome across frailty groups total cohort (n = 266) | Univariable analysis | Multivariable analysis | ||||
| Beta value/co-efficient (SE) | OR (95%CI) | P value | Beta value/co-efficient (SE) | OR (95%CI) | P value | |
| Pre transplant | ||||||
| LT recipient (yes/total n = 144/266) | ||||||
| Frail (n = 14/37) | -1.16 (0.45) | 0.33 (0.13-0.76) | 0.010 | -1.44 (0.49) | 0.24 (0.90-0.62) | 0.003 |
| Pre-frail (n = 97/179) | -0.50 (0.33) | 0.61 (0.32-1.17) | 0.140 | -0.62 (0.35) | 0.54 (0.27-1.08) | 0.040 |
| Robust (n = 33/50) | Reference | Reference | Reference | Reference | Reference | - |
| Unplanned hospital admission (yes/total n = 105/266) | ||||||
| Frail (n = 21/37) | 1.79 (0.50) | 6.00 (2.27-15.79) | < 0.001 | 1.12 (0.54) | 3.07 (1.06-8.90) | 0.04 |
| Pre-frail (n = 75/179) | 1.19 (0.40) | 3.29 (1.51-7.17) | 0.003 | 0.91 (0.42) | 2.49 (1.08-5.72) | 0.030 |
| Robust (n = 9/50) | Reference | Reference | - | Reference | Reference | - |
| Early post-transplant complications | ||||||
| Grade ≥ 3 post op complication1,2 (yes/total n = 72/144) | ||||||
| Frail (n = 7/14) | 0.43 (0.64) | 1.54 (0.44-5.42) | 0.500 | 0.09 (0.70) | 1.10 (0.28-4.28) | 0.900 |
| Pre-frail (n = 52/97) | 0.58 (0.41) | 1.78 (0.80-3.97) | 0.160 | 0.33 (0.43) | 1.39 (0.60-3.25) | 0.400 |
| Robust (n = 13/33) | Reference | Reference | - | Reference | Reference | - |
| ICU LOS > 4 days3 (yes/total n = 13/142) | ||||||
| Frail (n = 4/14) | 1.79 (0.94) | 6.00 (0.95-37.86) | 0.060 | 2.24 (1.09) | 9.45 (1.11-80.82) | 0.040 |
| Pre-frail (n = 7/96) | 0.21 (0.80) | 1.18 (0.23-5.99) | 0.840 | 0.20 (0.86) | 1.22 (0.22-6.65) | 0.810 |
| Robust (n = 2/32) | Reference | Reference | - | Reference | Reference | - |
| LOS > 18 days3 (yes/total n = 28/142) | ||||||
| Frail (n = 5/14) | 2.12 (0.92) | 8.33 (1.38-50.47) | 0.020 | 1.90 (0.96) | 6.66 (1.01-43.80) | 0.050 |
| Pre-frail (n = 21/96) | 1.44 (0.83) | 4.20 (0.93-19.03) | 0.060 | 1.31 (0.79) | 3.71 (0.80-17.28) | 0.100 |
| Robust (n = 2/32) | Reference | Reference | - | Reference | Reference | - |
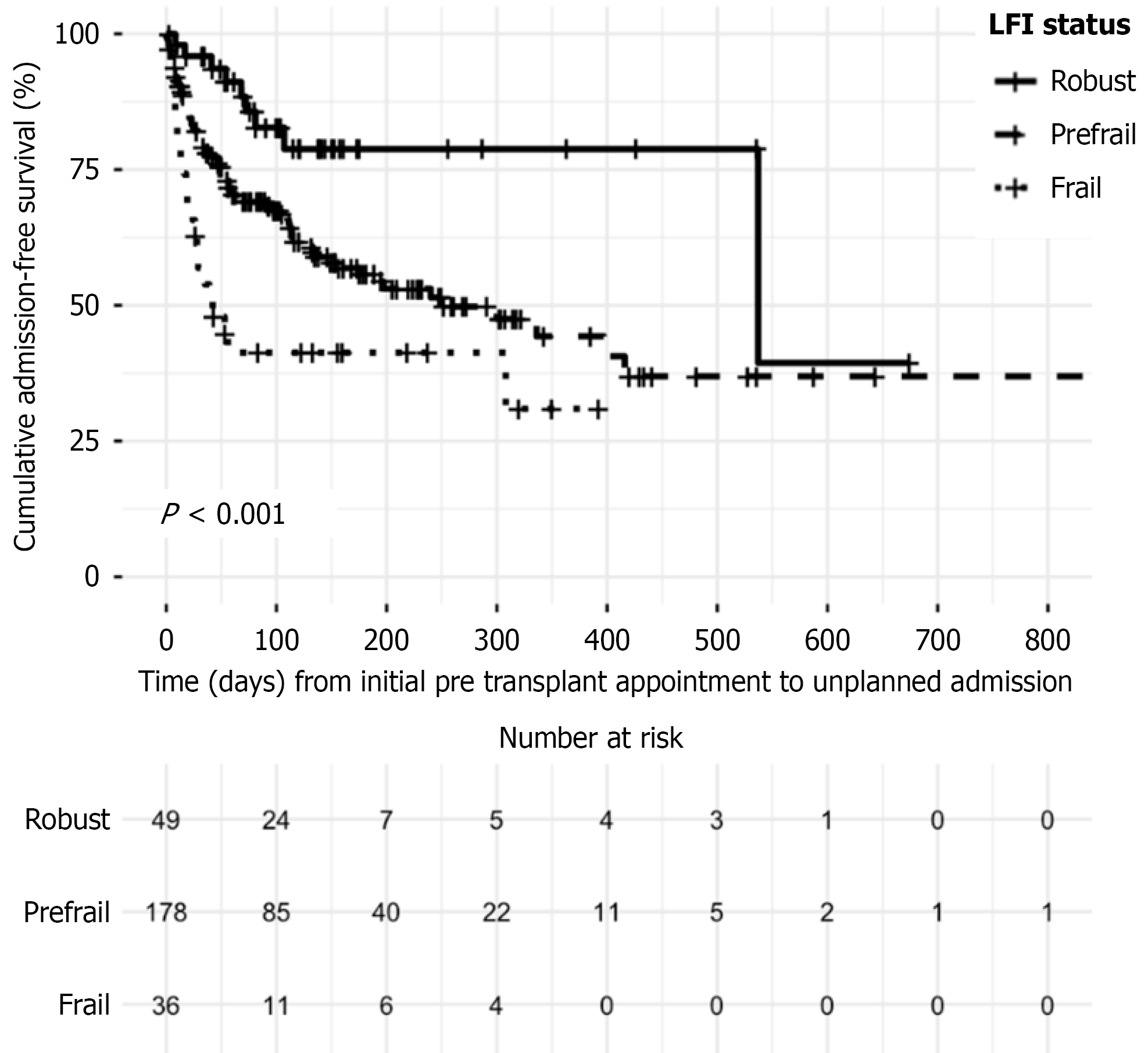
Kaplan-Meier analysis (Figure 1) demonstrated that not only were frail patients more likely to have an unplanned admission, but that these admissions occurred sooner than in other frailty categories (P < 0.001). For the 105 patients who experienced an unplanned admission, the median time to first unplanned admission was 19 (IQR 4-38) days for frail patients, 37 (IQR 19-114) days for pre-frail patients and 68 (IQR 41-81) days for robust patients. Cox proportional hazards analysis demonstrated that both LFI and MELD were independently associated with unplanned admissions. For every 1-point increase in the LFI, the risk of an unplanned admission increased by 42% (HR = 1.42, 95%CI: 1.06-1.90, P = 0.020), while for each one step worsening in frailty category (pre-frail compared to robust; and frail compared to pre-frail) there was a 48% increased risk of unplanned admissions before LT (HR = 1.48, 95%CI: 1.03-2.12, P = 0.030). Each 1-point increase in MELD score was associated with a 10% increase in the risk of unplanned admissions (HR = 1.10, 95%CI: 1.06-1.15, P < 0.001).
Following LT, the median ICU LOS was 1 (IQR 0.9-2) day, while the median hospital LOS was 12 (IQR 9-16) days. Nine percent of transplanted patients had an ICU LOS > 4 days while 20% had a hospital LOS > 18 days. Multivariable analysis (Tables 3 and 4) indicated that every 1-point increase in the LFI was associated with a 3.24-fold (95%CI: 1.06-9.85) increased odds of having an ICU admission > 4 days (P = 0.040). Frail patients had 9.45-fold (95%CI: 1.11-80.82) higher likelihood of an extended ICU LOS than robust patients (P = 0.040). Similarly for hospital LOS, frail patients were 6.66 times (95%CI: 1.01-43.80) more likely to have a LOS > 18 days compared to robust patients (P = 0.050) (Table 4). A similar trend was seen for LFI score, although this was not statistically significant (Table 3). Fifty percent of LT recipients experienced a major surgical complication (Clavien-Dindo ≥ 3) during their transplant admission, including two peri-transplant deaths, however the LFI was not associated with the risk of major surgical complications (Tables 3 and 4).
Understanding the impact of physical frailty in patients undergoing LT evaluation provides valuable insights to optimize outcomes for transplantation. In this Australian study of 266 patients referred for LT, we demonstrated that physical frailty (using the LFI) was strongly associated with several meaningful clinical outcomes, including the likelihood of progressing to transplantation or not, unplanned hospitalizations and longer peri-transplant ICU LOS. Importantly, these associations persisted independent of disease severity assessed by the MELD, underscoring frailty as a distinct and complementary construct in risk stratification.
Our findings align with the seminal work by Lai et al[8]and Kardashian et al[20] from the United States, which de
A novel aspect of the current study was that it examined the impact of baseline LFI on a broad range of clinical outcomes in patients’ transplant journeys including time to first unplanned admission and early post-transplant resource utilization, and in an Australian cohort, extending evidence beyond the predominance of United States based research to-date. Nearly 40% of patients experienced an unplanned admission, and frail patients were at a substantially higher risk (3-fold increased risk of unplanned admissions and occurring sooner than robust patients). This is consistent with smaller studies that used other frailty measures[22,23]. Our results extend this evidence by demonstrating that the LFI, a tool specific for liver cirrhosis, captures this vulnerability effectively. Being pre-frail also conferred a risk, demonstrating a higher risk of unplanned admissions compared to robust patients, without a significant impact on the likelihood of progressing to LT. This highlights a clinically relevant vulnerability that may be detectable prior to progression to frailty.
Following LT, frailty was most strongly associated with prolonged ICU stay, without an impact on LOS or major transplant surgical complications. This finding is consistent with the observations from Lai et al[24] using the LFI who also reported a prolonged ICU stay among frail patients. Notably, the results diverge from studies using the Fried Frailty Index, where frailty predicted surgical complications and overall LOS[25]. This may reflect the differences in the frailty constructs measured by each tool.
Collectively, these findings have clear implications for clinical practice. Firstly, they support the use of routine frailty assessment. While a specific tool into clinical practice is yet to be determined, the LFI is recommended for use in LT centers[2,7,26]. The LFI is an inexpensive, rapid assessment that is reproducible[10]. Frailty assessment can also assist with risk stratification, providing information beyond the MELD for patients who may benefit from prehabilitation and increased surveillance or expedited evaluation times. As we identified that pre-frail patients also experience increased risk, this enables an opportunity for early targeted intervention for preventative strategies before progressing to frailty.
Physical frailty is a potentially modifiable risk factor. However, high quality intervention studies targeting frailty in patients with cirrhosis are limited[2]. Several small studies in this cohort targeting muscle health demonstrate that improvements in muscle mass, physical performance and frailty markers are possible[27,28]. At present, results are from independent exercise[29], and nutrition[30] interventions, however, multimodal therapies are likely to give greater benefits[28]. Current guidance statements recommend combined approaches, but clearly there is more research needed in this area[2,26,31].
The lower prevalence of frailty in the current study (14%), compared to previous studies (19% to 33%)[21,32-34], raised questions about the optimal thresholds for clinical use. During the development of the LFI[8] thresholds for defining frailty categories (robust < 3.2, pre-frail > 3.2-4.5 and frail > 4.5) were determined from the LFI scores at the 20th and 80th centiles in that cohort. More recently, the LFI scores of a larger cohort of pre-LT patients identified thresholds of 4.4 and 4.2 shown to predict 3 months and 6 months waitlist mortality risk respectively[20]. In the current study, a substantially larger proportion of patients would be categorized at risk, suggesting that existing cut points may under-identify vulnerable individuals. Further investigation is needed to establish thresholds for clinical decision making, particularly for targeted pre-habilitation interventions. Relaxing the thresholds would potentially enhance earlier identification but may increase resource demand for those requiring intervention.
Overall, this study demonstrates that the LFI is clinically meaningful and applicable in settings outside the United States. Our findings contribute important non-United States data to further validate the LFI, addressing a gap previously highlighted in the literature[6].
Firstly, the LFI measures physical frailty and does not capture cognitive or psychological impairments of frailty which can significantly impact independence and quality of life. Secondly, frailty was measured at a single time point, with the associations relating to baseline frailty. There is some evidence that deteriorating frailty in pre-LT populations increases the likelihood of adverse outcomes[17]. Further longitudinal studies would help understand the trajectory of frailty pre-LT and the impact on outcomes. Finally, while this single center study may limit the generalizability, it provides a comprehensive analysis of a range of clinical outcomes related to frailty status at baseline. It demonstrates the utility of incorporating LFI measurements into real-life clinical practice. Given the realities of routine clinical practice, MELD rather than newer iterations such as MELD-Na or MELD 3.0 was collected, as this score was more consistently documented in clinical records. This approach ensured data completeness and uniformity across the cohort. Future research directions would benefit from multicenter studies, and longitudinal analyses of LFI trajectories across pre- and post-transplant care.
This research underscores the value of incorporating frailty assessments such as the LFI into baseline liver transplant evaluation. The LFI is a practical, office-based test that can easily be repeated. It identifies patients at risk of poor outcomes, enabling proactive intervention and monitoring over time. Identifying frailty in clinical practice allows clinicians to tailor surveillance, prioritize prehabilitation, and inform transplant decision-making alongside traditional measures of disease severity. These findings support LFI use in routine care and confirm its validity beyond United States centers, reinforcing its global applicability.
We acknowledge the staff and patients of the Queensland Liver Transplant Service for their times during the study period.
| 1. | Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA; Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:M146-M156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19956] [Cited by in RCA: 17082] [Article Influence: 683.3] [Reference Citation Analysis (26)] |
| 2. | Lai JC, Tandon P, Bernal W, Tapper EB, Ekong U, Dasarathy S, Carey EJ. Malnutrition, Frailty, and Sarcopenia in Patients With Cirrhosis: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1611-1644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 575] [Cited by in RCA: 525] [Article Influence: 105.0] [Reference Citation Analysis (1)] |
| 3. | Ney M, Tangri N, Dobbs B, Bajaj J, Rolfson D, Ma M, Ferguson T, Bhardwaj P, Bailey RJ, Abraldes J, Tandon P. Predicting Hepatic Encephalopathy-Related Hospitalizations Using a Composite Assessment of Cognitive Impairment and Frailty in 355 Patients With Cirrhosis. Am J Gastroenterol. 2018;113:1506-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (2)] |
| 4. | Lai JC, Feng S, Terrault NA, Lizaola B, Hayssen H, Covinsky K. Frailty predicts waitlist mortality in liver transplant candidates. Am J Transplant. 2014;14:1870-1879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 445] [Cited by in RCA: 407] [Article Influence: 33.9] [Reference Citation Analysis (1)] |
| 5. | Tandon P, Tangri N, Thomas L, Zenith L, Shaikh T, Carbonneau M, Ma M, Bailey RJ, Jayakumar S, Burak KW, Abraldes JG, Brisebois A, Ferguson T, Majumdar SR. A Rapid Bedside Screen to Predict Unplanned Hospitalization and Death in Outpatients With Cirrhosis: A Prospective Evaluation of the Clinical Frailty Scale. Am J Gastroenterol. 2016;111:1759-1767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 168] [Article Influence: 16.8] [Reference Citation Analysis (1)] |
| 6. | Van Jacobs AC. Frailty Assessment in Patients with Liver Cirrhosis. Clin Liver Dis (Hoboken). 2019;14:121-125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 7. | Lai JC, Sonnenday CJ, Tapper EB, Duarte-Rojo A, Dunn MA, Bernal W, Carey EJ, Dasarathy S, Kamath BM, Kappus MR, Montano-Loza AJ, Nagai S, Tandon P. Frailty in liver transplantation: An expert opinion statement from the American Society of Transplantation Liver and Intestinal Community of Practice. Am J Transplant. 2019;19:1896-1906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 182] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 8. | Lai JC, Covinsky KE, Dodge JL, Boscardin WJ, Segev DL, Roberts JP, Feng S. Development of a novel frailty index to predict mortality in patients with end-stage liver disease. Hepatology. 2017;66:564-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 473] [Cited by in RCA: 425] [Article Influence: 47.2] [Reference Citation Analysis (5)] |
| 9. | Lai JC, Covinsky KE, McCulloch CE, Feng S. The Liver Frailty Index Improves Mortality Prediction of the Subjective Clinician Assessment in Patients With Cirrhosis. Am J Gastroenterol. 2018;113:235-242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 143] [Article Influence: 17.9] [Reference Citation Analysis (2)] |
| 10. | Johnston HE, de Crom T, Hargrave C, Adhyaru P, Woodward AJ, Pang S, Ali A, Coombes JS, Keating SE, McLean K, Mayr HL, Macdonald GA, Hickman IJ. The inter- and intrarater reliability and feasibility of dietetic assessment of sarcopenia and frailty in potential liver transplant recipients: A mixed-methods study. Clin Transplant. 2021;35:e14185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 11. | Wang CW, Lebsack A, Chau S, Lai JC. The Range and Reproducibility of the Liver Frailty Index. Liver Transpl. 2019;25:841-847. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 12. | Williams FR, Milliken D, Lai JC, Armstrong MJ. Assessment of the Frail Patient With End-Stage Liver Disease: A Practical Overview of Sarcopenia, Physical Function, and Disability. Hepatol Commun. 2021;5:923-937. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (3)] |
| 13. | von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61:344-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13633] [Cited by in RCA: 12408] [Article Influence: 689.3] [Reference Citation Analysis (6)] |
| 14. | Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D'Amico G, Dickson ER, Kim WR. A model to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4208] [Cited by in RCA: 3749] [Article Influence: 150.0] [Reference Citation Analysis (5)] |
| 15. | Tandon P, Ney M, Irwin I, Ma MM, Gramlich L, Bain VG, Esfandiari N, Baracos V, Montano-Loza AJ, Myers RP. Severe muscle depletion in patients on the liver transplant wait list: its prevalence and independent prognostic value. Liver Transpl. 2012;18:1209-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 447] [Article Influence: 31.9] [Reference Citation Analysis (3)] |
| 16. | Morgan MY, Madden AM, Jennings G, Elia M, Fuller NJ. Two-component models are of limited value for the assessment of body composition in patients with cirrhosis. Am J Clin Nutr. 2006;84:1151-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 17. | Lai JC, Dodge JL, Kappus MR, Dunn MA, Volk ML, Duarte-Rojo A, Ganger DR, Rahimi RS, McCulloch CE, Haugen CE, McAdams-DeMarco M, Ladner DP, Segev DL, Verna EC; Multi-Center Functional Assessment in Liver Transplantation (FrAILT) Study. Changes in frailty are associated with waitlist mortality in patients with cirrhosis. J Hepatol. 2020;73:575-581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 133] [Article Influence: 22.2] [Reference Citation Analysis (2)] |
| 18. | Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibañes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9980] [Cited by in RCA: 9341] [Article Influence: 549.5] [Reference Citation Analysis (5)] |
| 19. | Muller X, Marcon F, Sapisochin G, Marquez M, Dondero F, Rayar M, Doyle MMB, Callans L, Li J, Nowak G, Allard MA, Jochmans I, Jacskon K, Beltrame MC, van Reeven M, Iesari S, Cucchetti A, Sharma H, Staiger RD, Raptis DA, Petrowsky H, de Oliveira M, Hernandez-Alejandro R, Pinna AD, Lerut J, Polak WG, de Santibañes E, de Santibañes M, Cameron AM, Pirenne J, Cherqui D, Adam RA, Ericzon BG, Nashan B, Olthoff K, Shaked A, Chapman WC, Boudjema K, Soubrane O, Paugam-Burtz C, Greig PD, Grant DR, Carvalheiro A, Muiesan P, Dutkowski P, Puhan M, Clavien PA. Defining Benchmarks in Liver Transplantation: A Multicenter Outcome Analysis Determining Best Achievable Results. Ann Surg. 2018;267:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 216] [Article Influence: 27.0] [Reference Citation Analysis (4)] |
| 20. | Kardashian A, Ge J, McCulloch CE, Kappus MR, Dunn MA, Duarte-Rojo A, Volk ML, Rahimi RS, Verna EC, Ganger DR, Ladner D, Dodge JL, Boyarsky B, McAdams-DeMarco M, Segev DL, Lai JC. Identifying an Optimal Liver Frailty Index Cutoff to Predict Waitlist Mortality in Liver Transplant Candidates. Hepatology. 2021;73:1132-1139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 21. | Haugen CE, McAdams-DeMarco M, Holscher CM, Ying H, Gurakar AO, Garonzik-Wang J, Cameron AM, Segev DL, Lai JC. Multicenter Study of Age, Frailty, and Waitlist Mortality Among Liver Transplant Candidates. Ann Surg. 2020;271:1132-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 22. | Dunn MA, Josbeno DA, Tevar AD, Rachakonda V, Ganesh SR, Schmotzer AR, Kallenborn EA, Behari J, Landsittel DP, DiMartini AF, Delitto A. Frailty as Tested by Gait Speed is an Independent Risk Factor for Cirrhosis Complications that Require Hospitalization. Am J Gastroenterol. 2016;111:1768-1775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 115] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 23. | Sinclair M, Poltavskiy E, Dodge JL, Lai JC. Frailty is independently associated with increased hospitalisation days in patients on the liver transplant waitlist. World J Gastroenterol. 2017;23:899-905. [PubMed] [DOI] [Full Text] |
| 24. | Lai JC, Shui AM, Duarte-Rojo A, Ganger DR, Rahimi RS, Huang CY, Yao F, Kappus M, Boyarsky B, McAdams-Demarco M, Volk ML, Dunn MA, Ladner DP, Segev DL, Verna EC, Feng S; from the Multi‐Center Functional Assessment in Liver Transplantation (FrAILT) Study. Frailty, mortality, and health care utilization after liver transplantation: From the Multicenter Functional Assessment in Liver Transplantation (FrAILT) Study. Hepatology. 2022;75:1471-1479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (1)] |
| 25. | DeMaria S Jr, Khromava M, Schiano TD, Lin HM, Kim S. Standardized measures of frailty predict hospital length of stay following orthotopic liver transplantation for hepatocellular carcinoma. Clin Transplant. 2019;33:e13746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 26. | Mohan R, Kalra A. Nutrition, frailty assessment, and interventions for the liver transplant candidate. Clin Liver Dis (Hoboken). 2023;22:23-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (4)] |
| 27. | Kruger C, McNeely ML, Bailey RJ, Yavari M, Abraldes JG, Carbonneau M, Newnham K, DenHeyer V, Ma M, Thompson R, Paterson I, Haykowsky MJ, Tandon P. Home Exercise Training Improves Exercise Capacity in Cirrhosis Patients: Role of Exercise Adherence. Sci Rep. 2018;8:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 28. | Johnston HE, Takefala TG, Kelly JT, Keating SE, Coombes JS, Macdonald GA, Hickman IJ, Mayr HL. The Effect of Diet and Exercise Interventions on Body Composition in Liver Cirrhosis: A Systematic Review. Nutrients. 2022;14:3365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 29. | Lin FP, Visina JM, Bloomer PM, Dunn MA, Josbeno DA, Zhang X, Clemente-Sanchez A, Tevar AD, Hughes CB, Jakicic JM, Duarte-Rojo A. Prehabilitation-Driven Changes in Frailty Metrics Predict Mortality in Patients With Advanced Liver Disease. Am J Gastroenterol. 2021;116:2105-2117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (2)] |
| 30. | Siramolpiwat S, Limthanetkul N, Pornthisarn B, Vilaichone RK, Chonprasertsuk S, Bhanthumkomol P, Nunanan P, Issariyakulkarn N. Branched-chain amino acids supplementation improves liver frailty index in frail compensated cirrhotic patients: a randomized controlled trial. BMC Gastroenterol. 2023;23:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 31. | Williams FR, Berzigotti A, Lord JM, Lai JC, Armstrong MJ. Review article: impact of exercise on physical frailty in patients with chronic liver disease. Aliment Pharmacol Ther. 2019;50:988-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 32. | Lai JC, Rahimi RS, Verna EC, Kappus MR, Dunn MA, McAdams-DeMarco M, Haugen CE, Volk ML, Duarte-Rojo A, Ganger DR, O'Leary JG, Dodge JL, Ladner D, Segev DL. Frailty Associated With Waitlist Mortality Independent of Ascites and Hepatic Encephalopathy in a Multicenter Study. Gastroenterology. 2019;156:1675-1682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 205] [Cited by in RCA: 194] [Article Influence: 27.7] [Reference Citation Analysis (2)] |
| 33. | Fozouni L, Mohamad Y, Lebsack A, Freise C, Stock P, Lai JC. Frailty Is Associated With Increased Rates of Acute Cellular Rejection Within 3 Months After Liver Transplantation. Liver Transpl. 2020;26:390-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 34. | Haugen CE, McAdams-DeMarco M, Verna EC, Rahimi RS, Kappus MR, Dunn MA, Volk ML, Gurakar A, Duarte-Rojo A, Ganger DR, O'Leary JG, Ladner D, Garonzik-Wang J, Segev DL, Lai JC. Association Between Liver Transplant Wait-list Mortality and Frailty Based on Body Mass Index. JAMA Surg. 2019;154:1103-1109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (1)] |