Copyright: ©Author(s) 2026.
World J Hepatol. Apr 27, 2026; 18(4): 115582
Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.115582
Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.115582
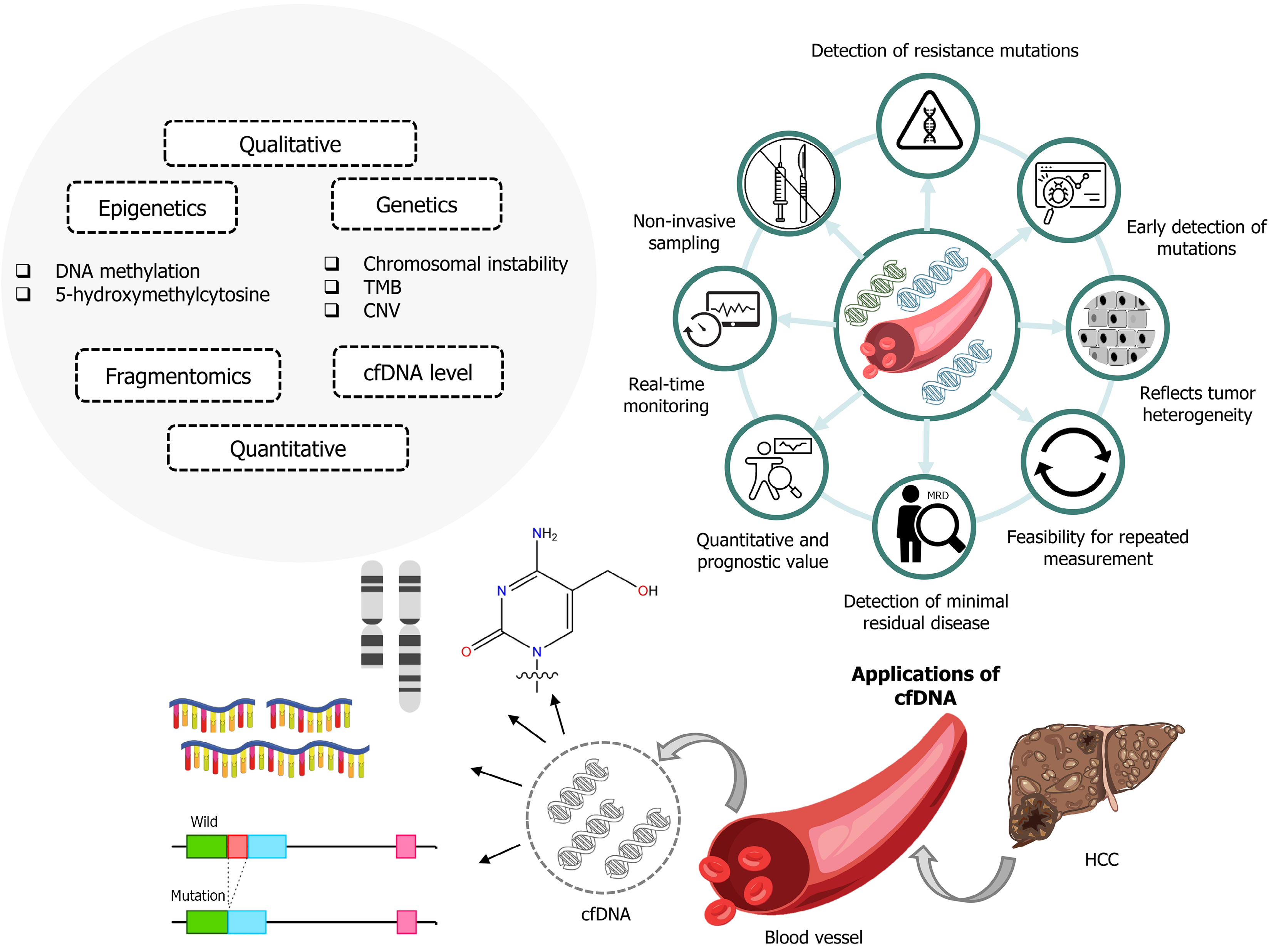
Figure 1 Circulating cell-free DNA as a tool for cancer diagnostics and monitoring response.
The left panel categorises cell-free DNA (cfDNA) assays into qualitative and quantitative domains: Epigenetic markers (DNA methylation, 5-hydroxymethylcytosine), genetic alterations (chromosomal instability, tumor mutation burden, copy number variation), and fragmentomics, with cfDNA level representing a quantitative burden metric. The central wheel illustrates clinical utilities of cfDNA, including noninvasive sampling, real time monitoring, quantitative and prognostic value, minimal residual disease detection, feasibility of repeated measurements, reflection of tumor heterogeneity, early detection of mutations, and identification of resistance mutations. The lower panel depicts sources and forms of tumour-derived DNA: Chromosomal changes, nucleotide level variants, copy number alterations, and structural variants/fusions, converging as circulating cfDNA released into blood; arrows indicate sampling from blood vessels and its application to hepatocellular carcinoma. Icons emphasise longitudinal use across therapy: Serial cfDNA enables dynamic response assessment and resistance surveillance without repeated tissue biopsy, aligning with use in hepatocellular carcinoma management pathways. cfDNA: Cell-free DNA; CNV: Copy number variation; TMB: Tumor mutation burden; HCC: Hepatocellular carcinoma.
- Citation: Sharma R, Singh SP, Bhatia G, Ramakrishna G. Cell-free DNA in hepatocellular carcinoma: Biology to treatment response. World J Hepatol 2026; 18(4): 115582
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/115582.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.115582