Copyright: ©Author(s) 2026.
World J Hepatol. Mar 27, 2026; 18(3): 117040
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.117040
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.117040
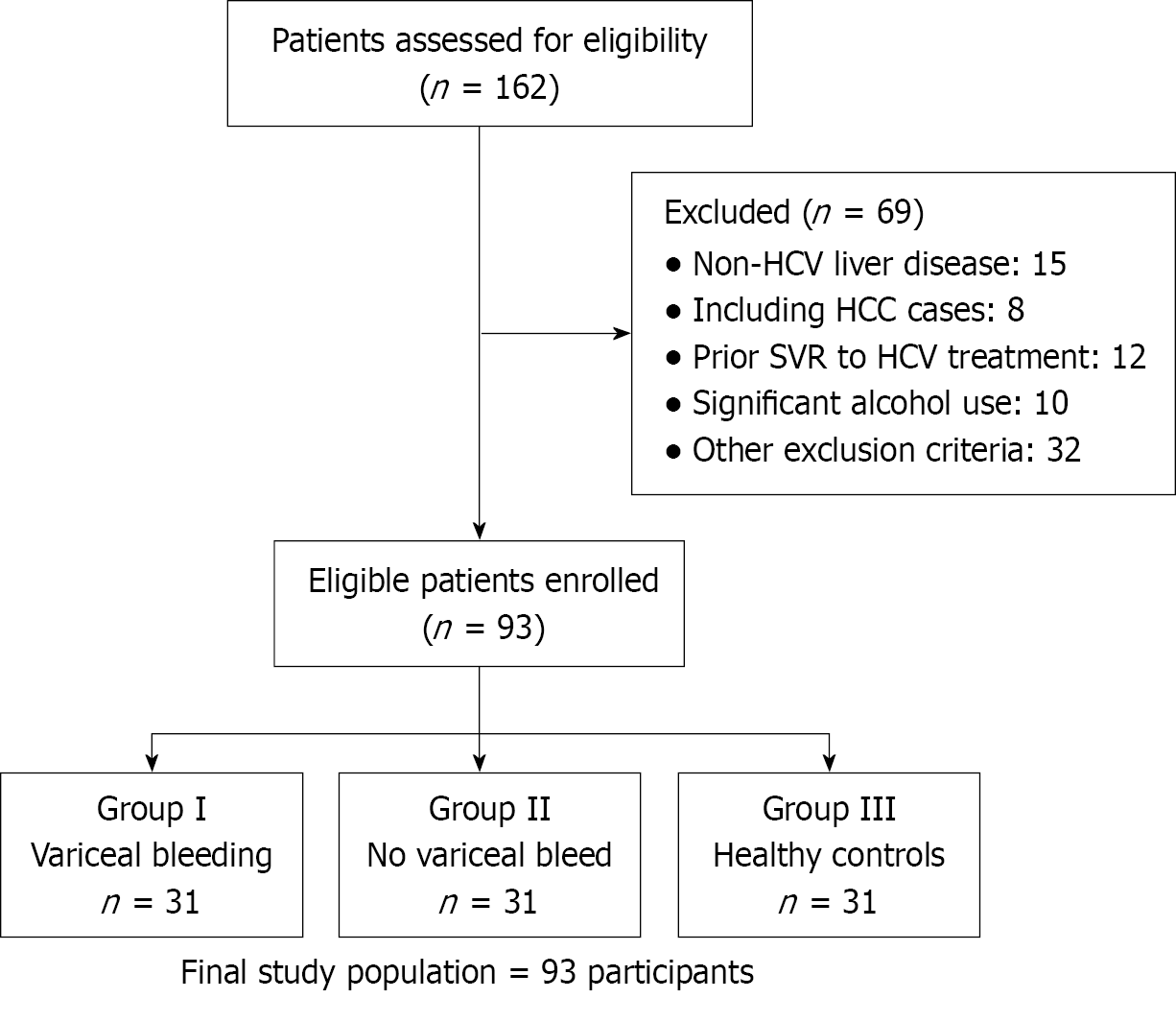
Figure 1 CONSORT flow diagram of participant selection.
Between December 2021 and December 2022, 162 patients with chronic hepatitis C virus (HCV) and liver cirrhosis were assessed for eligibility. A total of 69 patients were excluded based on predefined criteria, including non-HCV liver disease etiologies (n = 15; with 8 cases of hepatocellular carcinoma), prior sustained virological response to HCV treatment (n = 12), significant alcohol use (n = 10), and other exclusion criteria (n = 32). Consecutive enrollment continued until the target sample size was achieved, resulting in 93 eligible participants. These were allocated into three equal groups: Group I (variceal bleeding, n = 31), group II (no variceal bleeding, n = 31), and group III (healthy controls, n = 31). HCV: Hepatitis C virus; SVR: Sustained virological response.
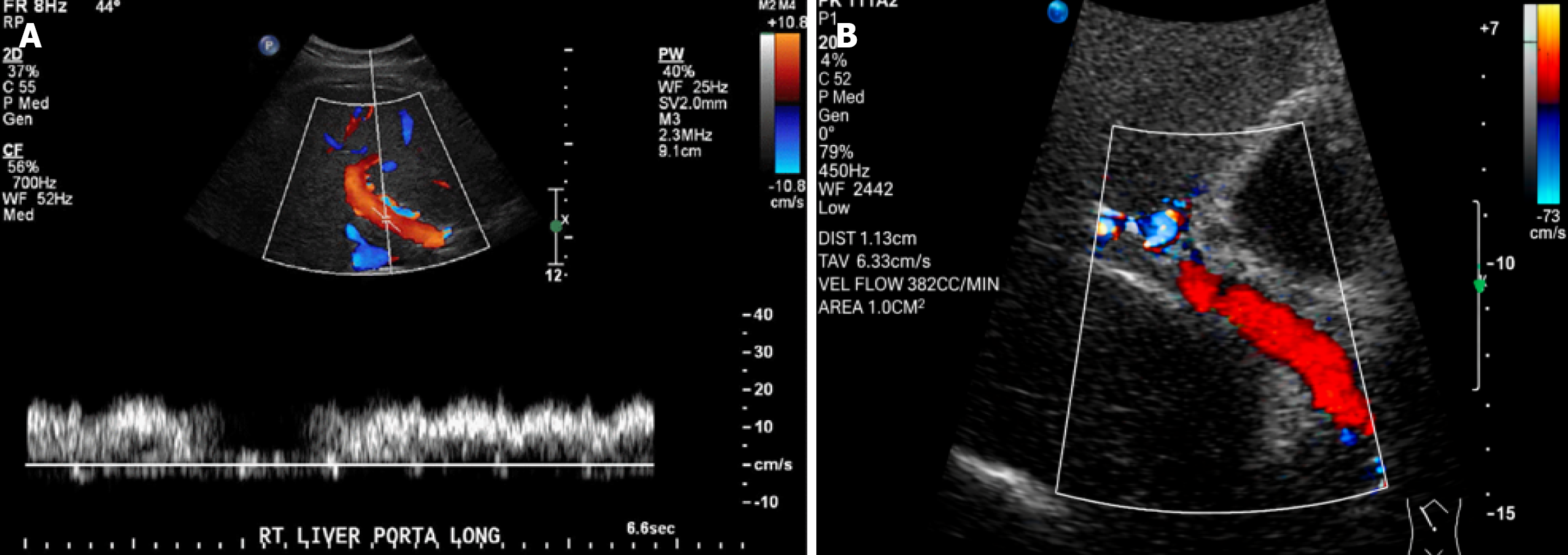
Figure 2 Doppler ultrasonography of the portal vein in a healthy control.
A: Portal vein diameter measuring 10 mm; B: Normal portal venous flow with a mean velocity of 25 cm/second and flow volume of 600 cc/minute.
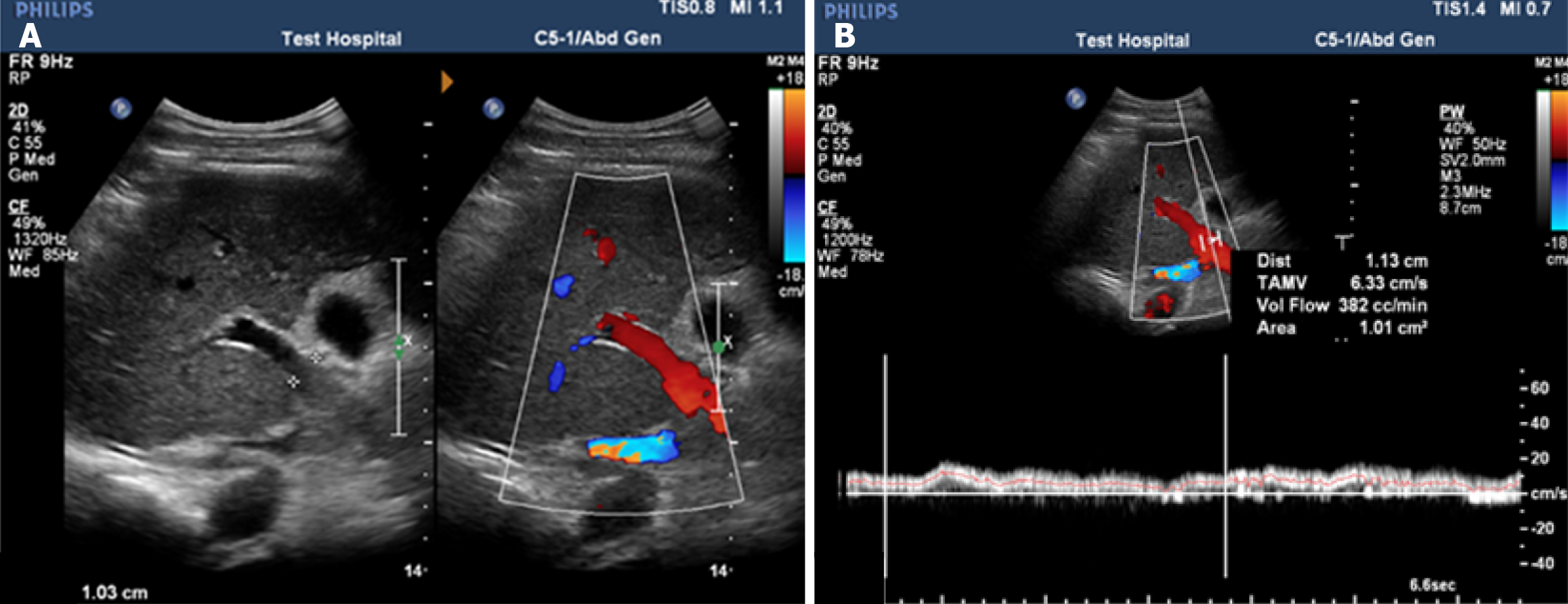
Figure 3 Doppler ultrasonography of the portal vein in a cirrhotic patient with portal hypertension.
A: Dilated portal vein diameter of 11.3 mm; B: Reduced mean flow velocity of 18 cm/second and flow volume of 382 cc/minute.
- Citation: Othman AAA, Elsharkawy L, Saleh MW, Mohamed MM, Elsayed FMA. Multimarker model for non-invasive assessment of variceal bleeding in chronic hepatitis C cirrhotic patients. World J Hepatol 2026; 18(3): 117040
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/117040.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.117040