Copyright: ©Author(s) 2026.
World J Hepatol. Mar 27, 2026; 18(3): 115411
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115411
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115411
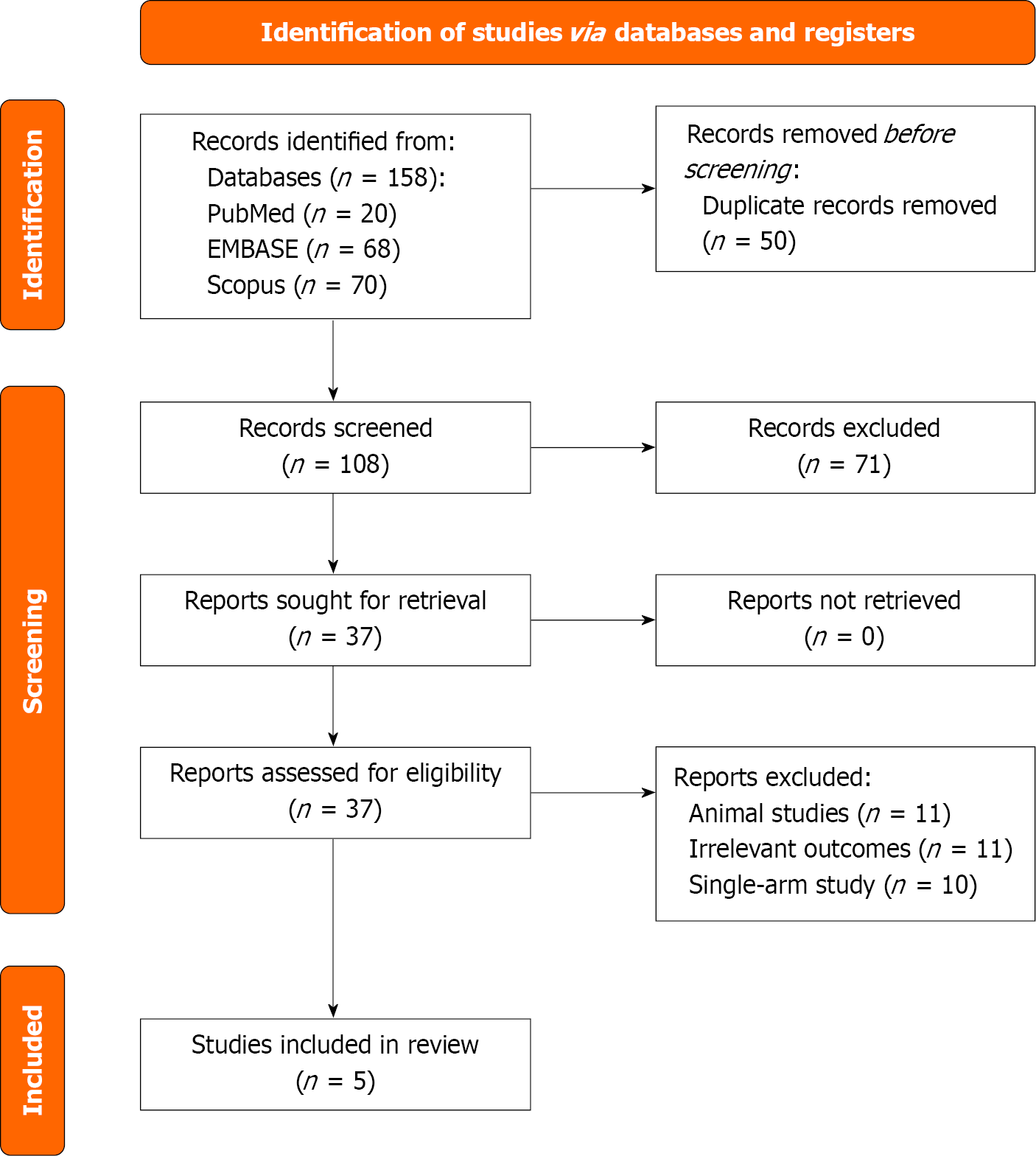
Figure 1
Preferred Reporting Items for Systematic Reviews and Meta-Analyses chart of included studies.
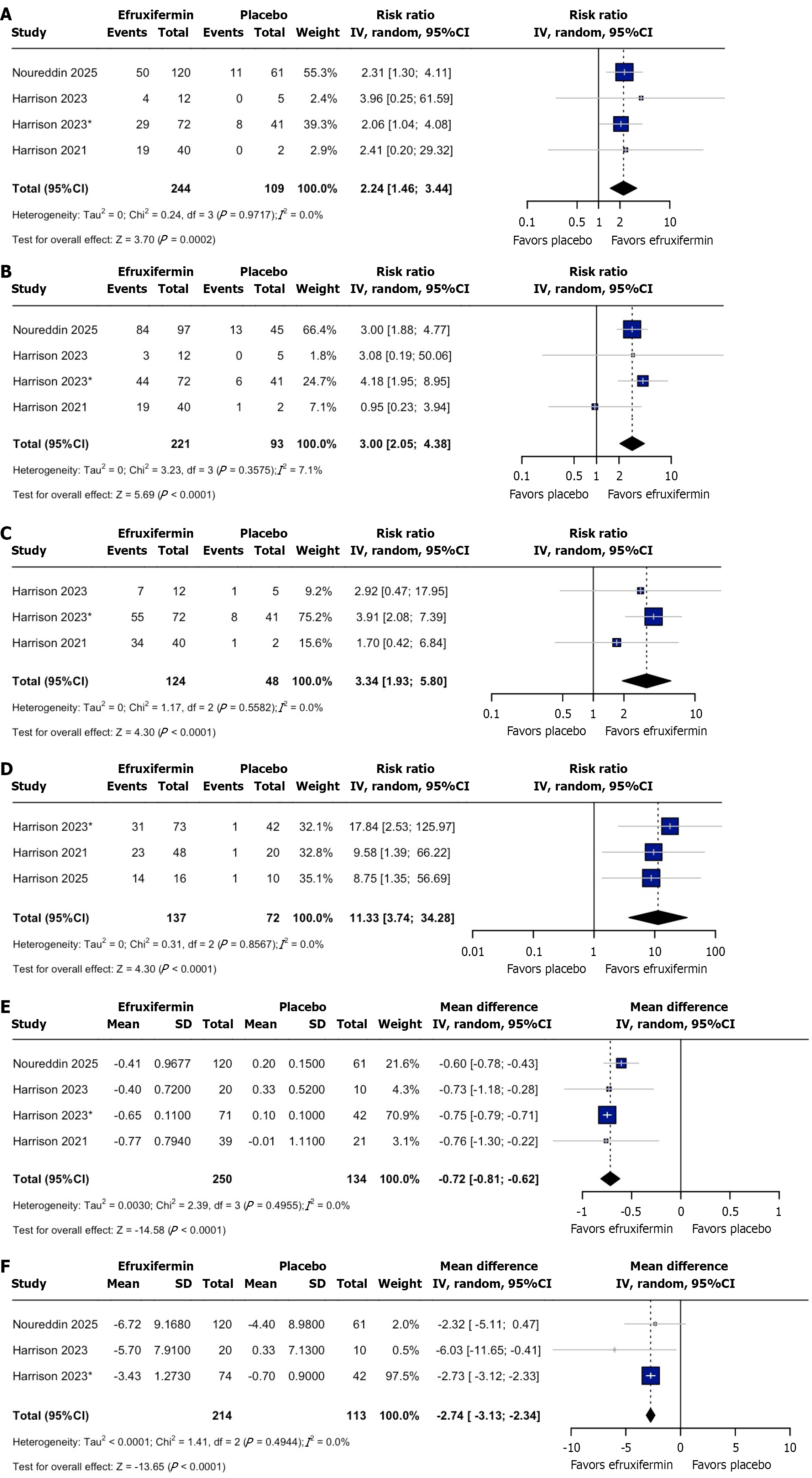
Figure 2 Forest plot of outcomes.
A: Achieving at least one stage of improvement in fibrosis without worsening of metabolic-associated steatohepatitis; B: Metabolic-associated steatohepatitis resolution without worsening fibrosis; C: At least a two-point improvement in the non-alcoholic fatty liver disease activity score with no worsening of fibrosis; D: Normalized liver fat; E: Enhanced liver fibrosis test score; F: Liver stiffness measurement score. CI: Confidence interval.
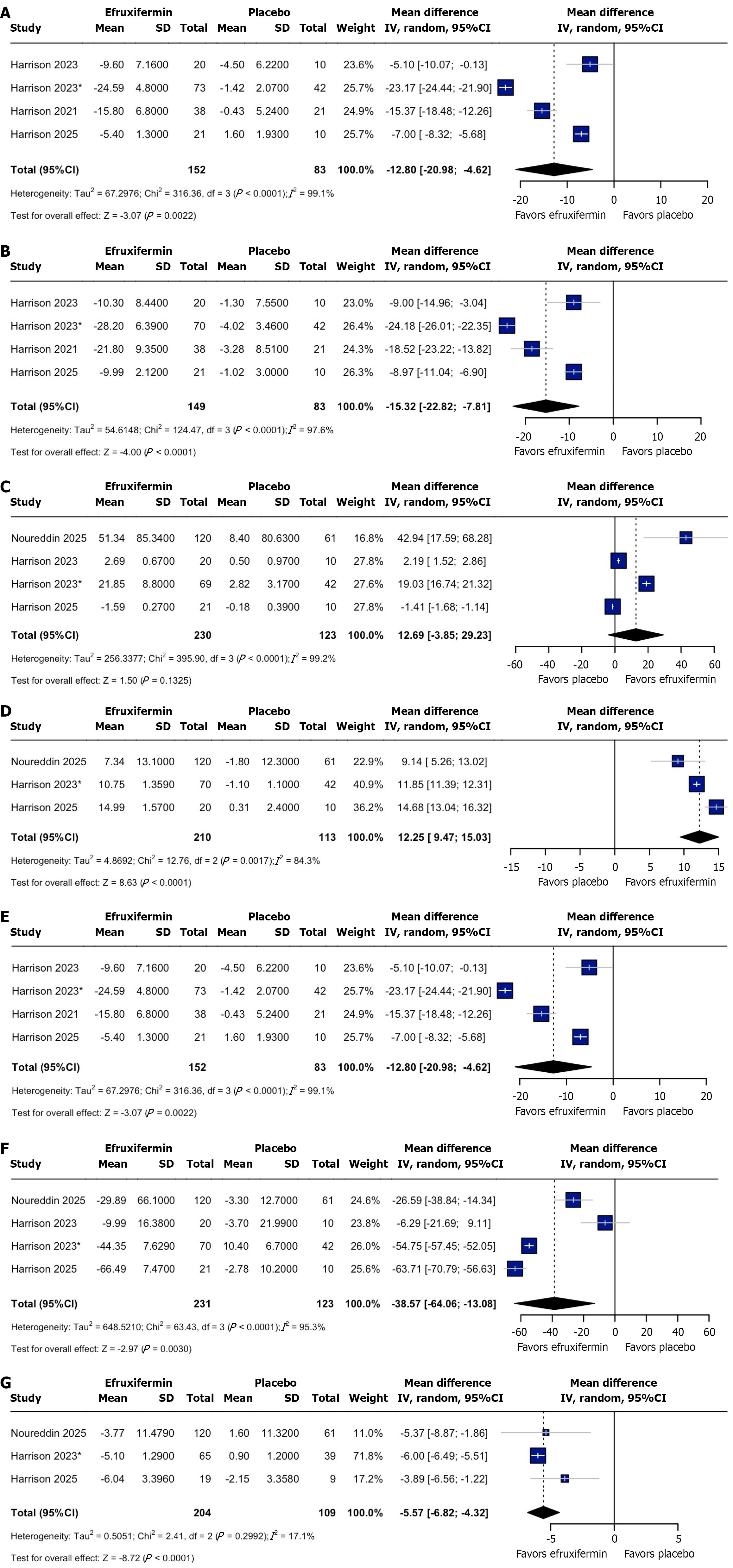
Figure 3 Forest plot of outcomes.
A: Change in alanine aminotransferase; B: Change in aspartate aminotransferase; C: Percentage change in adiponectin; D: High-density lipoprotein cholesterol; E: Low-density lipoprotein cholesterol; F: Triglycerides; G: Homeostatic model assessment index. CI: Confidence interval.
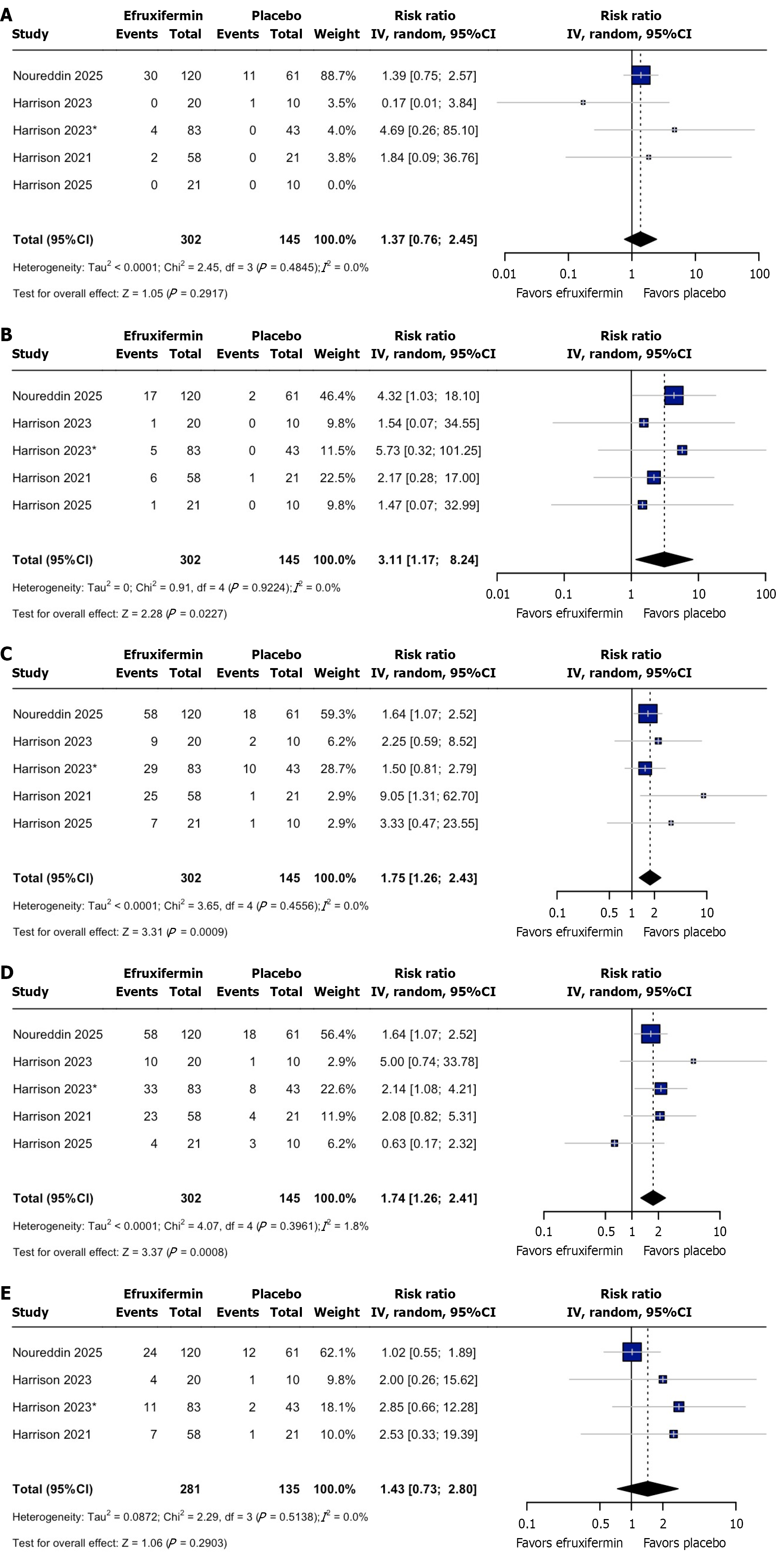
Figure 4 Forest plot of outcomes.
A: Serious adverse events; B: Adverse events leading to discontinuation; C: Nausea; D: Diarrhea; E: Injection site reaction. CI: Confidence interval.
- Citation: Alam U, Rath S, Ansab M, Kashif A, Sethi FA, Siddiqui HT, Afridi ZAK, Burhan M, Ali MA, Moiz A, Bacha Z, Khan A, Ahmed R, Alatise MB, Kamil KA. Efruxifermin in patients with metabolic dysfunction-associated steatohepatitis: A GRADE-assessed systematic review and meta-analysis. World J Hepatol 2026; 18(3): 115411
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115411.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115411