Published online Sep 26, 2015. doi: 10.4252/wjsc.v7.i8.1118
Peer-review started: May 20, 2015
First decision: June 24, 2015
Revised: July 4, 2015
Accepted: July 29, 2015
Article in press: August 3, 2015
Published online: September 26, 2015
Processing time: 129 Days and 22.5 Hours
Stem cells are of global excitement for various diseases including heart diseases. It is worth to understand the mechanism or role of stem cells in the treatment of heart failure. Bone marrow derived stem cells are commonly practiced with an aim to improve the function of the heart. The majority of studies have been conducted with acute myocardial infarction and a few has been investigated with the use of stem cells for treating chronic or dilated cardiomyopathy. Heterogeneity in the treated group using stem cells has greatly emerged. Ever increasing demand for any alternative made is of at most priority for cardiomyopathy. Stem cells are of top priority with the current impact that has generated among physicians. However, meticulous selection of proper source is required since redundancy is clearly evident with the present survey. This review focuses on the methods adopted using stem cells for heart diseases and outcomes that are generated so far with an idea to determine the best therapeutic possibility in order to fulfill the present demand.
Core tip: Heart diseases are most common all over the world and any form of treatment would be well appreciated to enable successful well-being in the patients. Though there are enormous efforts attempted to handle heart diseases, there has been no propounded evidence so far to utilize a particular source of cells globally. In this regard, if the responses all over the world are collectively segregated to a global database, there may be options to identify the best suitable cell source that provides effective cure.
- Citation: Bilgimol JC, Ragupathi S, Vengadassalapathy L, Senthil NS, Selvakumar K, Ganesan M, Manjunath SR. Stem cells: An eventual treatment option for heart diseases. World J Stem Cells 2015; 7(8): 1118-1126
- URL: https://www.wjgnet.com/1948-0210/full/v7/i8/1118.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v7.i8.1118
Core tip: Heart diseases are most common all over the world and any form of treatment would be well appreciated to enable successful well-being in the patients. Though there are enormous efforts attempted to handle heart diseases, there has been no propounded evidence so far to utilize a particular source of cells globally. In this regard, if the responses all over the world are collectively segregated to a global database, there may be options to identify the best suitable cell source that provides effective cure.
Cardiac diseases have become a major cause of death worldwide[1-4]. Common causes of heart failure are ischemia, hypertension, coronary artery disease and idiopathic dilated cardiomyopathy. Up to 40% of cardiac patients are predominantly affected by diastolic dysfunction, although the left ventricular (LV) systolic function is mostly unaffected[5]. There are established medical and surgical procedures available for cardiac patients but these standard therapeutic modalities have not been much responded[1,6,7]. Cardiac risks are also associated with other complications[8-10], which thus gives an steady increase in the number of patients looking for an effective treatment option that can sustain their normal well-being. There is an urge for alternative therapies that can fulfill the present overwhelming demand[6,11]. Cardiac diseases trigger damage to cardiomyocytes that affects the heart muscles and there is a possibility of scar formation that will overload the blood flow and eventually stretch the cardiac cells. This ultimately leads to heart failure and commonly, deaths[12].
The current situation is therefore mostly favorable for alternative treatment possibilities that can improve the disease considerably at least with respect to the rate of survival or quality of life[13-15]. Recent evidence on cell based therapies has suggested improvement in cardiac function and regeneration of the damaged tissue[16]. It is stated that cell therapy has got potentials to do what standard drugs cannot do by replacing the damaged tissues with healthy tissue. Drugs have been demonstrated to slow the progression of heart failure, but none have shown their potential in reversing the process. However, the physiological perspectives have not shown any clear picture with cell therapies as it is difficult to estimate the cell survival and monitor cells in vivo[17,18] along with their low engraftment rate[18]. Valid doubts and concerns are raised for the existing use of stem cells for heart diseases and it is elaborated that although regenerative medicine has lots of hopes, results have not substantiated their importance in the in-house tentative studies[19]. There are some important factors that need to be considered such as appropriate selection of the cell type, number of cells required and appropriate route of administration that can help regeneration. Routes of injection generally include catheter-guided transendocardial, intracoronary, transvenous, and transepicardial injections[20]. The selected cells should have a control over their migration, proliferation and differentiation ex vivo and also in vivo[21]. Even though the mechanisms of action pertaining to cell therapies are not very clear, studies in comparison with routine therapies have shown promising results by significantly improving vessel density, inhibiting apoptosis of myocytes, and decreasing fibrosis and increasing survival rate even in non-ischemic heart failures[22,23]. There are other reports available with similar strategy to improve the survival of transplanted cardiac progenitor cells[24-27].
The progress of cell therapy is reported in Mayo Clinic proceedings as breakthroughs that have reached certain milestones and expectations that have a high prospect towards imminent benefits. The six major breakthroughs are summarized as: (1) safety establishment in intracoronary delivery; (2) possibility of therapeutic regeneration; (3) allogeneic cell therapy; (4) increasing mechanistic insights; (5) sparkling clinical efficacy; and (6) progress to phase 2 and 3 studies[28]. Cell therapy has shown enormous progress since its application in 1998 and this lightning speed is progressing towards eliminating the underlying cause of heart failures and handling the control over the damage[29]. Different cell types have been used to treat heart diseases[30-32]. There are potential benefits with the application of cardiac progenitor cells and induced pluripotent stem cells (iPSCs) as a promising approach to bring true myocardial regeneration[30].
The advent of iPSCs is also under remarkable consideration to model heart disease[33]. Stem cells have been suggested to be promising new therapeutic usage for heart diseases because of their remarkable proliferative and differentiation capacity that can supply specific cell types along with the viable functioning of heart muscles[12]. Evidence has shown that resident cardiac stem cells have got potentials to turn into cardiomyocytes in vivo[31]. There are other well-known and commonly available sources of progenitor cells from bone marrow, adipose tissue, and skeletal muscle or cells circulating in blood that are capable of improving myocardial function[11,34].
Although there are waves of research undertaken with stem cells or any cell therapy applications, in vitro studies are required to explore which is of high priority to advance with the strategies to tailor clinical applications and the task is highly challenging[35]. Appropriate selection of patients, informed consent and periodic patient monitoring are important factors to be reviewed by expert panel independent of investigators while opting for clinical trials[36,37]. While manufacturing stem cells, parameters like screening and testing for pathogens have to be carried out with utmost precision. Generation of consistent batches while manufacturing is highly recommended for therapeutic derivative of stem cells and this has to be carried out in aseptic conditions withstanding robust quality programs that qualify regulatory expectations which are suitable for clinical use in patients. It is also necessary to validate the procedures for transplant and confirm the regeneration of infarcted heart[38,39]. Overall, it is expected that the strategies adopted from routine therapies into stem cell-based therapies, have glorious potentials to increase the efficacy of treatment[40]. This review thus focuses on the studies attempting to assess various alternative therapeutic applications for heart diseases and to suggest the best suitable and effective source that has produced better success rate.
Heart disease continues to be the main cause of death though there is a considerable decline in the death rate with the advancement in medicine since 1999. Nonetheless, medical care has not been successful in reversing the loss of functional cardiomyocytes[41]. The surgeries being attempted for diagnostic cardiac catheterizations, coronary artery bypass graft procedures, cardiac pacemaker procedures, percutaneous transluminal coronary angioplasty procedures and heart valve procedures are all very expensive since they requires hospitalizations, post-surgical care, etc. They consume enormous time to handle the recovery process. Still, evidence suggests that cardiovascular diseases are not prevented though the lethality has been reduced considerably[42]. Presently, the treatment options vastly carried out for cardiovascular function include organ transplantation, surgical reconstruction, and usage of mechanical or artificial devices or administration of metabolically derived products[43]. Benefits have been associated with inotropic therapy, ventricular assist devices and hospice care. Recommendations are given towards the use of angotensin-converting enzyme inhibitors as a first line of treatment in patients with reduced LV systolic function with ejection fraction ≤ 35%-40%[44]. For normal ventricular ejection fraction, beta-blockers, isosorbide dinitrate plus hydralazine are being considered[45].
In this context, researchers have been encouraged to identify better strategies to image the detection and monitoring of cardiovascular disease progression. Biotech industries have also been progressing in different scenarios with other therapeutic possibilities which with mutual understanding between the pharmaceutical industries and medical specialists, may aim to bring options that provide immediate recovery along with cost effective strategies[3,46]. Interest relies on the targeted delivery of alternatives such as stem and progenitor cell populations that has also kindled the combinatorial use of genes to treat cardiovascular diseases. However, challenge depends on the host environment where it has poorly retained the survival of implanted cells and thus it requires the delivery of cells transfected or transduced ex vivo with genes that can enhance resistance to apoptosis in order to provide protection from the stressed microenvironment that can enable the improved function of the engrafted cells to appreciate better therapeutic outcome[47].
Even after maturation, cardiomyocytes have limited flexibility. The recovery of heart function after structural damage is less possible. Mesenchymal stem cell (MSC) therapy is predicted to be a promising area of research for regenerating damaged cardiac tissues. MSCs lack both major histocompatibility II and T-cell co-stimulatory signal expression and thus are immune-privileged. When MSCs are delivered systematically, they own a unique ability to enter into the specific sites of myocardial damage. There are mechanisms by which MSCs promote cardiac tissue regeneration. Data generated from several studies reveal that MSCs are therapeutically valuable and play a great role in cardiac diseases. In future, MSCs could be used for cell-based therapy for cardiac diseases.
MSCs are generally derived from any tissue sources. In the translational research MSCs used are mainly derived from bone marrow. Specifically, for cardiac repair or cardiac disorders, the MSCs used are derived from bone marrow[48]. Several studies are carried out to understand the mechanism of action of MSCs in cardiac repair. One of the studies conducted by Yang et al[49] demonstrated that a combination of statin along with bone marrow derived MSCs intramyocardial injections improved the efficiency of cardiomyocyte differentiation by 4-fold in a swine model.
Numerous preclinical studies are carried out using large animal models to understand the efficacy of MSC therapy for cardiac diseases. In these studies, stem cells are delivered to the heart via peripheral intravenous infusion during the open heart surgery. It can also be administered through catheter-based intracoronary infusion. The studies carried out after delivery of MSCs showed that intramyocardial injection had the highest retention rate of cells[50]. Among all the preclinical studies carried out with MSCs for cardiac diseases, surgical injection was the most commonly used method to deliver MSCs[51-53].
Though there are varieties of sources of MSCs, there has been particular interest in adipose lineage MSCs which were found to be easily available and successful in several preclinical studies. Several studies were carried out by the administration of adipose derived MSCs for myocardial infarction (MI), which were found to be successful[54].
Many clinical trials are carried out to improve the recovery of cardiac function. There were several favorable and unfavorable results during the course of time. In many of the studies, bone marrow mononuclear cells were injected. Nevertheless, detailed studies are to be carried out to understand the fate and effects of transplanted cells[55-58]. It is understood that bone marrow contains heterogeneous populations of cells, generally Hematopoietic stem cells (HSCs) and MSCs[59].
Apart from these, bone marrow also has different progenitors of myeloid and lymphoid lineages. Several studies indicate that both HSCs and MSCs are reported to acquire the phenotype of cardiomyocytes in syngeneic or xenogeneic recipients. Even though various studies were carried out, none of them quantitatively analyzed the regenerative capacity. Nevertheless, there is evidence shown by Norol et al[60] in nonhuman primate models that HSCs do not differentiate into cardiomyocytes or into endothelial cells in the infarcted myocardium. Apparently, the growth factors produced locally does not have any positive effect on the regeneration of myocardium[60]. It is well known that HSCs are a unique source of adult stem cells that are easily accessible. It is documented that HSCs were used to induce neoangiogenesis to treat ischemic vascular diseases. HSCs subsets also contain endothelial precursors that are AC133+, which are considered to be major requirements in patients with ischemic and refractory peripheral vascular or coronary artery diseases[61].
Endothelial progenitor cells (EPCs) consist of a heterogeneous group of cells which originates from multiple precursors within the bone marrow and are present in different stages of endothelial differentiation. The existence of endothelial dysfunction forecasts the consequences in patients who suffer with cardiovascular risk factors and persons with coronary artery disease. EPCs possess the ability to home to sites of vascular injury. Thus, EPCs could be easily adopted for therapeutic uses related to angiogenesis. Several studies conducted in coronary artery disease (CAD) patients revealed that they had reduced number of EPCs. They also had reduced number of circulating EPCs[62-65].
Bone marrow derived EPCs will be mobilized to stimulate angiogenesis and will attenuate tissue ischemia for CAD and peripheral arterial disease (PAD). Further to this, clinical trials carried out with different animal models favor the application of EPCs as a potential therapeutic agent in cardiovascular diseases[66,67].
Usage of EPC capture stent is also in practice. EPC capture stent is a device which uses the ability of bone marrow derived EPCs to repair damaged arterial segments. This instrument is manufactured in such a way that the surface of EPC antibody contains a covalently coupled polysaccharide intermediate coating with anti-human CD34 antibody and it is attached to a stainless steel stent. Once the stent is placed, the circulating EPCs will be attracted to CD34 antibodies and will be directed to differentiate into mature endothelial cells to form functional endothelium layer[68].
There are several studies ongoing to understand the effects of pharmacotherapies with EPCs in cardiac patients. EPC behavior and mechanisms are also elucidated in several studies conducted in patients with cardiovascular disease (CVD), CAD, heart failure and PAD. There are several promising studies showing that EPCs could be used as a novel therapy for CVD[69]. However, there are several limitations with EPCs due to its paucity of circulating cells. The efficacy of the treatment using EPCs can be obtained by increasing the cell number by concentrating and transplanting or by functional improvement using ex vivo augmentation.
Usage of iPSCs emerged as a novel therapy for the treatment of cardiac diseases. It has also attracted much attention and is considered to be the promising cell source for patient specific cell therapy[70]. Major advantage of iPSCs are that the clinically relevant number of cells could be generated easily when compared to adult stem cells and that they are associated with a less risk of immune rejection when compared to embryonic stem cells[71].
iPSCs are generally engendered from adult somatic tissues with precise reprogramming. Studies prove that iPSCs have many similarities to human embryonic stem cells especially in terms of morphology, proliferation, feeder dependence, surface markers, gene expression, epigenetic status, promoter activities, telomerase activities, etc.[72]. A study was conducted in a mouse model of MI, and human iPSCs were induced in this model. The results disclosed that the mice had regenerated myocardium, smooth muscles and endothelial tissue, restoring post ischaemic contractility performance and electric stability[73]. Even though, several studies and clinical trials were carried out to understand the efficacy of iPSCs in cardiac repair. When it comes to gene therapy, the integration of viral vectors in the genome remains unanswered. These viral vectors in the genome might promote malignancy. Apart from that, these virus-encoded genes can act as oncogenes. Thus, the clinical application of iPSC derived cardiomyocytes appears to be a distant possibility at present. However, the advent of different study models will resolve the issues in the future.
Although many cell types have been attempted, there have been promising outcomes with crude extracts of bone marrow derived cells. Utilization of such resources not only minimizes time but also may be useful in reducing the cost of expenses of application[74]. Recent use of bone marrow derived mononuclear cells (BMMNCs) in heart diseases has been able to improve LV function, and increase the myocardial perfusion, but has not produced any significant change in the scar tissue[75,76]. A double blind study of a total of 16 patients after previous placebo injection has described the improvement in angina symptoms and myocardial perfusion with the infusion of BMMNCs in chronic myocardial ischemia patients[77]. However, Heeschen et al[78] have shown that the application of BMMNCs to ischemic cardiomyopathy patients when injected, has profoundly reduced the neovascularization and thus limited its therapeutic potential for cell therapy. However, the reduced neovascularization is mostly reported to be impaired with aging, which is largely attributed to aged BMMNCs, but ageing does not affect the revitalization of neovascularization in the murine host with the transplantation of young BMMNCs[79]. Although the results have been more commented, scientists overall are not in favor of using BMMNCs because the small number of undifferentiated cells is unlikely to be effective[74].
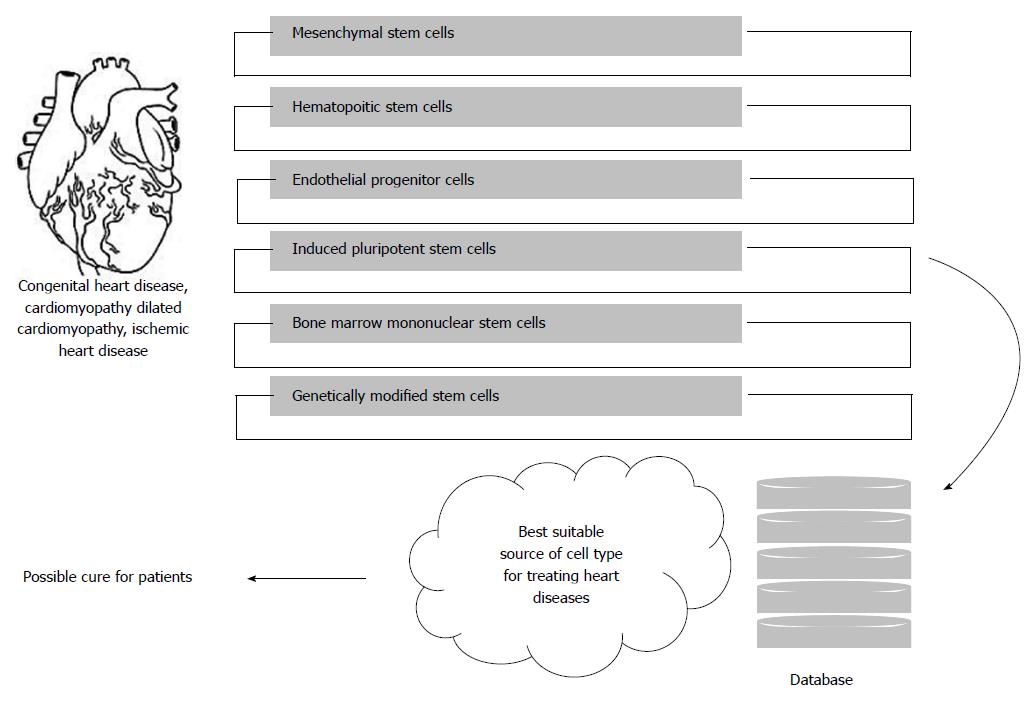
Genetic modifications of stem cells will result in enhanced efficacy. Genetically modified stem cells can be utilized for different diseases. A number of diseases can be treated now with them. Various strategies like pre-treatment with growth differentiation factors or cytokines, pre-conditioning like hypoxia and genetic modifications to overexpress anti-death signals are developed. It was Mangi et al[80] who first genetically modified MSCs to overexpress Akt which are resistant to apoptosis in vitro and in vivo. They proved that there was a drastic improvement in cardiac function in an MI rodent model[80]. Further work on this showed that Akt transduction stimulates MSCs to produce paracrine factors which are advantageous to infarcted heart post-engraftment[81,82]. In another experiment transfected MSCs with bFGF showed an increased survival and cytoprotection in hypoxic condition[83] (Figure 1).
It is clearly understood that there are underlying challenges for the identification of well-deserved type of regenerative therapy. The majority of cell types are under clinical trials but the results have been heterogeneous. Causes have been predicted to be poor survival, proliferation, engraftment and differentiation of engrafted cells. Ultimately, the objective is to increase the functional ability of stem cell before and after transplantation[4,84]. The concern thus lies in the knowledge on structural and functional characteristics of stem cell-derived cardiomyocytes, and their capability to integrate with host tissue[85,86]. Concerns need to be imparted for appropriate selection of patients with clear understanding about the variability of disease condition. Prospective outcomes have to be confirmed by the anatomical and histological examinations with a large number of studied patients in order to analyze significant observations[87]. With the prevailing methods for achieving an alternative approach to deal with heart diseases, the approach that would in future be available for the treatment is believed to possess more effective delivery methods and growth and storage methods[17].
Researchers are working on to effective management of the challenges for the utilization of stem cell therapy wherein, newer strategies are being adopted for the combination of cells with bioengineering techniques that may improve the efficacy considerably[88]. However, approaches dealing with the utilization of epicardium-derived cells and cardiomyocyte progenitor cells have synergistically shown better impact in cardiac function which is believed to be because of complementary paracrine actions[89]. Whatever the sources have been utilized or approached such as embryonic stem cells, iPSCs, adult stem cell lineages, bone marrow derived stem cells, or MSCs, it is widely acceptable that there is enormous hope underlying the usage of stem cell therapy as a clinically viable alternative for heart diseases[90] and the direction that the research is progressing is believed to be perfect which requires clear understanding and revival of the attempted trails to progress with newer ideas and accomplish the ultimate goal.
Challenges are still prevailing for the application of cell therapy to the patient. Considering the complications in delivery of the stem cells to the patient, three factors are to be taken into consideration: the type and nature of injury, timing of the therapy, and the ability of the cells to engraft the host myocardium[82]. There is a huge dilemma regarding the best time for stem cell delivery to patients. Several studies carried out in this field prove that early time points will be ideal for stem cell transplantation[91]. Choosing of best stem cell type also plays a crucial role in transplantation and engraftment ability to host myocardium. Recent studies indicated that transplanted cells are vulnerable to hostile ischemic environment. Thus, they will tend to disappear in a few days[92]. Several investigations are going on to improve the microenvironment and engraftment followed by survival.
Even though the mechanisms of action for all these stem cell based therapies are not well established, it remains a promising field of research. However, a major obstacle in this arena is the poor viability of implanted stem cells for cardiac repair. The strategies reviewed here are for the augmentation and development of appropriate approaches for the treatment of cardiac disorders. Attempts performed using a specific kind of cell type by any organization are claimed to provide efficient results in comparison to the other. This has created disputes in pursuing cell types that can generate positive outcomes and thus has deliberately given poor understanding amongst the physicians. Involvement of cell therapy possibilities has therefore anonymously diminished though it is under immediate purview. Future challenges pertaining to the application hence depend on the clear validation of the cell therapy with respect to source, mode of application, dosage and expected outcome. Reciprocation of these results is vital in order to pursue the technology.
Ethical constraints are also major hurdles to circumvent the use of alternative strategies which needs to be clearly framed considering the benefits that medical arena would acquire. Adult stem cells in this regard, have proven safe and generated decent outcomes. We suggest that, instead of exploiting research knowledge to fewer more decades, concentration towards the ultimate requirement is very important. The studies generated so far with various alternative treatment modalities for heart diseases worldwide can be collectively segregated in a common database and significance with the available data can be generated to find the best and viable source of approach that proves effective for heart diseases. In case of any discrepancy with the results, the reciprocation of the data can be attempted in order to obtain a solution.
| 1. | Kelly BB, Narula J, Fuster V. Recognizing global burden of cardiovascular disease and related chronic diseases. Mt Sinai J Med. 2012;79:632-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 2. | Celermajer DS, Chow CK, Marijon E, Anstey NM, Woo KS. Cardiovascular disease in the developing world: prevalences, patterns, and the potential of early disease detection. J Am Coll Cardiol. 2012;60:1207-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 310] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 3. | Cardiovascular disease. Nat Biotechnol. 2000;18 Suppl:IT15-IT17. [PubMed] [DOI] [Full Text] |
| 4. | Hoover-Plow J, Gong Y. Challenges for heart disease stem cell therapy. Vasc Health Risk Manag. 2012;8:99-113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 5. | Graham RM, Bishopric NH, Webster KA. Gene and cell therapy for heart disease. IUBMB Life. 2002;54:59-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 6. | Soran O. A new treatment modality in heart failure enhanced external counterpulsation (EECP). Cardiol Rev. 2004;12:15-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Haeck ML, Hoogslag GE, Rodrigo SF, Atsma DE, Klautz RJ, van der Wall EE, Schalij MJ, Verwey HF. Treatment options in end-stage heart failure: where to go from here? Neth Heart J. 2012;20:167-175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Ford ES. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care. 2005;28:1769-1778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1211] [Cited by in RCA: 1253] [Article Influence: 59.7] [Reference Citation Analysis (0)] |
| 9. | Huang Y, Wang S, Cai X, Mai W, Hu Y, Tang H, Xu D. Prehypertension and incidence of cardiovascular disease: a meta-analysis. BMC Med. 2013;11:177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 177] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 10. | Malvestutto CD, Aberg JA. Coronary heart disease in people infected with HIV. Cleve Clin J Med. 2010;77:547-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 11. | Janssens S. Stem cells in the treatment of heart disease. Annu Rev Med. 2010;61:287-300. [PubMed] [DOI] [Full Text] |
| 12. | Hotkar AJ, Balinsky W. Stem cells in the treatment of cardiovascular disease--an overview. Stem Cell Rev. 2012;8:494-502. [PubMed] |
| 13. | Orlic D. Stem cell repair in ischemic heart disease: an experimental model. Int J Hematol. 2002;76 Suppl 1:144-145. [PubMed] |
| 14. | Vrtovec B, Poglajen G, Haddad F. Stem cell therapy in patients with heart failure. Methodist Debakey Cardiovasc J. 2013;9:6-10. [PubMed] |
| 15. | Bernstein HS, Srivastava D. Stem cell therapy for cardiac disease. Pediatr Res. 2012;71:491-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 16. | Segers VF, Lee RT. Stem-cell therapy for cardiac disease. Nature. 2008;451:937-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 895] [Cited by in RCA: 857] [Article Influence: 47.6] [Reference Citation Analysis (0)] |
| 17. | Dinsmore JH, Dib N. Stem cells and cardiac repair: a critical analysis. J Cardiovasc Transl Res. 2008;1:41-54. [PubMed] |
| 18. | Seeger FH, Zeiher AM, Dimmeler S. Cell-enhancement strategies for the treatment of ischemic heart disease. Nat Clin Pract Cardiovasc Med. 2007;4 Suppl 1:S110-S113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Abbott A. Doubts over heart stem-cell therapy. Nature. 2014;509:15-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Fazel S, Tang GH, Angoulvant D, Cimini M, Weisel RD, Li RK, Yau TM. Current status of cellular therapy for ischemic heart disease. Ann Thorac Surg. 2005;79:S2238-S2247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Reza Mohammadhasani M, Hasanzad M, Mohammadhasani A, Samzadeh M, Eslami M. Some notes on stem cell therapy in cardiovascular diseases. ARYA Atheroscler. 2010;6:78-81. [PubMed] |
| 22. | Li TS, Mikamo A, Takahashi M, Suzuki R, Ueda K, Ikeda Y, Matsuzaki M, Hamano K. Comparison of cell therapy and cytokine therapy for functional repair in ischemic and nonischemic heart failure. Cell Transplant. 2007;16:365-374. [PubMed] |
| 23. | Ong SG, Lee WH, Huang M, Dey D, Kodo K, Sanchez-Freire V, Gold JD, Wu JC. Cross talk of combined gene and cell therapy in ischemic heart disease: role of exosomal microRNA transfer. Circulation. 2014;130:S60-S69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 182] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 24. | Huang M, Chen Z, Hu S, Jia F, Li Z, Hoyt G, Robbins RC, Kay MA, Wu JC. Novel minicircle vector for gene therapy in murine myocardial infarction. Circulation. 2009;120:S230-S237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 81] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 25. | Yockman JW, Kastenmeier A, Erickson HM, Brumbach JG, Whitten MG, Albanil A, Li DY, Kim SW, Bull DA. Novel polymer carriers and gene constructs for treatment of myocardial ischemia and infarction. J Control Release. 2008;132:260-266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Hu S, Huang M, Nguyen PK, Gong Y, Li Z, Jia F, Lan F, Liu J, Nag D, Robbins RC. Novel microRNA prosurvival cocktail for improving engraftment and function of cardiac progenitor cell transplantation. Circulation. 2011;124:S27-S34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 27. | Fischer KM, Cottage CT, Wu W, Din S, Gude NA, Avitabile D, Quijada P, Collins BL, Fransioli J, Sussman MA. Enhancement of myocardial regeneration through genetic engineering of cardiac progenitor cells expressing Pim-1 kinase. Circulation. 2009;120:2077-2087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 162] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 28. | Marbán E. Breakthroughs in cell therapy for heart disease: focus on cardiosphere-derived cells. Mayo Clin Proc. 2014;89:850-858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 29. | Sanganalmath SK, Bolli R. Cell therapy for heart failure: a comprehensive overview of experimental and clinical studies, current challenges, and future directions. Circ Res. 2013;113:810-834. [PubMed] [DOI] [Full Text] |
| 30. | Povsic TJ, O’Connor CM. Cell therapy for heart failure: the need for a new therapeutic strategy. Expert Rev Cardiovasc Ther. 2010;8:1107-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | Zhang H, Wang H, Li N, Duan CE, Yang YJ. Cardiac progenitor/stem cells on myocardial infarction or ischemic heart disease: what we have known from current research. Heart Fail Rev. 2014;19:247-258. [PubMed] |
| 32. | Oldroyd KG, Berry C, Bartunek J. Myocardial repair and regeneration: bone marrow or cardiac stem cells? Mol Ther. 2012;20:1102-1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 33. | Matsa E, Burridge PW, Wu JC. Human stem cells for modeling heart disease and for drug discovery. Sci Transl Med. 2014;6:239ps6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 163] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 34. | Templin C, Lüscher TF, Landmesser U. Cell-based cardiovascular repair and regeneration in acute myocardial infarction and chronic ischemic cardiomyopathy-current status and future developments. Int J Dev Biol. 2011;55:407-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 35. | Abdelwahid E, Siminiak T, Guarita-Souza LC, Teixeira de Carvalho KA, Gallo P, Shim W, Condorelli G. Stem cell therapy in heart diseases: a review of selected new perspectives, practical considerations and clinical applications. Curr Cardiol Rev. 2011;7:201-212. [PubMed] |
| 36. | Lodi D, Iannitti T, Palmieri B. Stem cells in clinical practice: applications and warnings. J Exp Clin Cancer Res. 2011;30:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 113] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 37. | Liras A. Future research and therapeutic applications of human stem cells: general, regulatory, and bioethical aspects. J Transl Med. 2010;8:131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 38. | Weber DJ. Manufacturing considerations for clinical uses of therapies derived from stem cells. Methods Enzymol. 2006;420:410-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 39. | Trounson A, Thakar RG, Lomax G, Gibbons D. Clinical trials for stem cell therapies. BMC Med. 2011;9:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 336] [Cited by in RCA: 295] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 40. | Srijaya TC, Ramasamy TS, Kasim NH. Advancing stem cell therapy from bench to bedside: lessons from drug therapies. J Transl Med. 2014;12:243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 41. | Doppler SA, Deutsch MA, Lange R, Krane M. Cardiac regeneration: current therapies-future concepts. J Thorac Dis. 2013;5:683-697. [PubMed] |
| 42. | Pearson TA. The prevention of cardiovascular disease: have we really made progress? Health Aff (Millwood). 2007;26:49-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | Nugent HM, Edelman ER. Tissue engineering therapy for cardiovascular disease. Circ Res. 2003;92:1068-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 107] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 44. | Friedrich EB, Böhm M. Management of end stage heart failure. Heart. 2007;93:626-631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 45. | Aronow WS. Treatment of heart failure with normal left ventricular ejection fraction. Compr Ther. 2007;33:223-230. [PubMed] |
| 46. | Kwok BW. Medical therapy in heart failure--is polypharmacy necessary? Ann Acad Med Singapore. 2004;33:444-450. [PubMed] |
| 47. | Fishbein I, Chorny M, Levy RJ. Site-specific gene therapy for cardiovascular disease. Curr Opin Drug Discov Devel. 2010;13:203-213. [PubMed] |
| 48. | da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119:2204-2213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1907] [Cited by in RCA: 1696] [Article Influence: 84.8] [Reference Citation Analysis (0)] |
| 49. | Yang YJ, Qian HY, Huang J, Li JJ, Gao RL, Dou KF, Yang GS, Willerson JT, Geng YJ. Combined therapy with simvastatin and bone marrow-derived mesenchymal stem cells increases benefits in infarcted swine hearts. Arterioscler Thromb Vasc Biol. 2009;29:2076-2082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 50. | Hou D, Youssef EA, Brinton TJ, Zhang P, Rogers P, Price ET, Yeung AC, Johnstone BH, Yock PG, March KL. Radiolabeled cell distribution after intramyocardial, intracoronary, and interstitial retrograde coronary venous delivery: implications for current clinical trials. Circulation. 2005;112:I150-I156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 331] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 51. | Shake JG, Gruber PJ, Baumgartner WA, Senechal G, Meyers J, Redmond JM, Pittenger MF, Martin BJ. Mesenchymal stem cell implantation in a swine myocardial infarct model: engraftment and functional effects. Ann Thorac Surg. 2002;73:1919-125; discussion 1926. [PubMed] |
| 52. | Schuleri KH, Feigenbaum GS, Centola M, Weiss ES, Zimmet JM, Turney J, Kellner J, Zviman MM, Hatzistergos KE, Detrick B. Autologous mesenchymal stem cells produce reverse remodelling in chronic ischaemic cardiomyopathy. Eur Heart J. 2009;30:2722-2732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 196] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 53. | Hamamoto H, Gorman JH, Ryan LP, Hinmon R, Martens TP, Schuster MD, Plappert T, Kiupel M, St John-Sutton MG, Itescu S. Allogeneic mesenchymal precursor cell therapy to limit remodeling after myocardial infarction: the effect of cell dosage. Ann Thorac Surg. 2009;87:794-801. [PubMed] [DOI] [Full Text] |
| 54. | Gimble JM, Katz AJ, Bunnell BA. Adipose-derived stem cells for regenerative medicine. Circ Res. 2007;100:1249-1260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1903] [Cited by in RCA: 1677] [Article Influence: 88.3] [Reference Citation Analysis (0)] |
| 55. | Schächinger V, Erbs S, Elsässer A, Haberbosch W, Hambrecht R, Hölschermann H, Yu J, Corti R, Mathey DG, Hamm CW. Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med. 2006;355:1210-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1540] [Cited by in RCA: 1306] [Article Influence: 65.3] [Reference Citation Analysis (0)] |
| 56. | Meyer GP, Wollert KC, Lotz J, Steffens J, Lippolt P, Fichtner S, Hecker H, Schaefer A, Arseniev L, Hertenstein B. Intracoronary bone marrow cell transfer after myocardial infarction: eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrOw transfer to enhance ST-elevation infarct regeneration) trial. Circulation. 2006;113:1287-1294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 793] [Cited by in RCA: 662] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 57. | Jeevanantham V, Butler M, Saad A, Abdel-Latif A, Zuba-Surma EK, Dawn B. Adult bone marrow cell therapy improves survival and induces long-term improvement in cardiac parameters: a systematic review and meta-analysis. Circulation. 2012;126:551-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 382] [Cited by in RCA: 360] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 58. | Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, Nygren JM, Fries JW, Tiemann K, Bohlen H, Hescheler J. Potential risks of bone marrow cell transplantation into infarcted hearts. Blood. 2007;110:1362-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 522] [Cited by in RCA: 459] [Article Influence: 24.2] [Reference Citation Analysis (1)] |
| 59. | Koide Y, Morikawa S, Mabuchi Y, Muguruma Y, Hiratsu E, Hasegawa K, Kobayashi M, Ando K, Kinjo K, Okano H. Two distinct stem cell lineages in murine bone marrow. Stem Cells. 2007;25:1213-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 60. | Norol F, Bonnet N, Peinnequin A, Chretien F, Legrand R, Isnard R, Herodin F, Baillou C, Delache B, Negre D. GFP-transduced CD34+ and Lin- CD34- hematopoietic stem cells did not adopt a cardiac phenotype in a nonhuman primate model of myocardial infarct. Exp Hematol. 2007;35:653-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 61. | Burt R, Pearce W, Luo K, Oyama Y, Davidson C, Beohar N, Gheorghiade M. Hematopoietic stem cell transplantation for cardiac and peripheral vascular disease. Bone Marrow Transplant. 2003;32 Suppl 1:S29-S31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 62. | Eizawa T, Ikeda U, Murakami Y, Matsui K, Yoshioka T, Takahashi M, Muroi K, Shimada K. Decrease in circulating endothelial progenitor cells in patients with stable coronary artery disease. Heart. 2004;90:685-686. [PubMed] |
| 63. | Wang HY, Gao PJ, Ji KD, Shen WF, Fan CL, Lu L, Zhu DL. Circulating endothelial progenitor cells, C-reactive protein and severity of coronary stenosis in Chinese patients with coronary artery disease. Hypertens Res. 2007;30:133-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 64. | Liguori A, Fiorito C, Balestrieri ML, Crimi E, Bruzzese G, Williams-Ignarro S, D’Amora M, Sommese L, Grimaldi V, Minucci PB. Functional impairment of hematopoietic progenitor cells in patients with coronary heart disease. Eur J Haematol. 2008;80:258-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 65. | Briguori C, Testa U, Riccioni R, Colombo A, Petrucci E, Condorelli G, Mariani G, D’Andrea D, De Micco F, Rivera NV. Correlations between progression of coronary artery disease and circulating endothelial progenitor cells. FASEB J. 2010;24:1981-1988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 66. | Kawamoto A, Gwon HC, Iwaguro H, Yamaguchi JI, Uchida S, Masuda H, Silver M, Ma H, Kearney M, Isner JM. Therapeutic potential of ex vivo expanded endothelial progenitor cells for myocardial ischemia. Circulation. 2001;103:634-637. [PubMed] |
| 67. | Kawamoto A, Tkebuchava T, Yamaguchi J, Nishimura H, Yoon YS, Milliken C, Uchida S, Masuo O, Iwaguro H, Ma H. Intramyocardial transplantation of autologous endothelial progenitor cells for therapeutic neovascularization of myocardial ischemia. Circulation. 2003;107:461-468. [PubMed] |
| 68. | Sedaghat A, Sinning JM, Paul K, Kirfel G, Nickenig G, Werner N. First in vitro and in vivo results of an anti-human CD133-antibody coated coronary stent in the porcine model. Clin Res Cardiol. 2013;102:413-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 69. | Losordo DW, Kibbe MR, Mendelsohn F, Marston W, Driver VR, Sharafuddin M, Teodorescu V, Wiechmann BN, Thompson C, Kraiss L. A randomized, controlled pilot study of autologous CD34+ cell therapy for critical limb ischemia. Circ Cardiovasc Interv. 2012;5:821-830. [PubMed] |
| 70. | Masumoto H, Matsuo T, Yamamizu K, Uosaki H, Narazaki G, Katayama S, Marui A, Shimizu T, Ikeda T, Okano T. Pluripotent stem cell-engineered cell sheets reassembled with defined cardiovascular populations ameliorate reduction in infarct heart function through cardiomyocyte-mediated neovascularization. Stem Cells. 2012;30:1196-1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 117] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 71. | Ptaszek LM, Mansour M, Ruskin JN, Chien KR. Towards regenerative therapy for cardiac disease. Lancet. 2012;379:933-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 659] [Reference Citation Analysis (2)] |
| 72. | Kong CW, Akar FG, Li RA. Translational potential of human embryonic and induced pluripotent stem cells for myocardial repair: insights from experimental models. Thromb Haemost. 2010;104:30-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 73. | Nelson TJ, Martinez-Fernandez A, Yamada S, Perez-Terzic C, Ikeda Y, Terzic A. Repair of acute myocardial infarction by human stemness factors induced pluripotent stem cells. Circulation. 2009;120:408-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 337] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 74. | Michler RE. Stem cell therapy for heart failure. Methodist Debakey Cardiovasc J. 2013;9:187-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 75. | Beeres SL, Bax JJ, Dibbets P, Stokkel MP, Zeppenfeld K, Fibbe WE, van der Wall EE, Schalij MJ, Atsma DE. Effect of intramyocardial injection of autologous bone marrow-derived mononuclear cells on perfusion, function, and viability in patients with drug-refractory chronic ischemia. J Nucl Med. 2006;47:574-580. [PubMed] |
| 76. | Beeres SL, Bax JJ, Kaandorp TA, Zeppenfeld K, Lamb HJ, Dibbets-Schneider P, Stokkel MP, Fibbe WE, de Roos A, van der Wall EE. Usefulness of intramyocardial injection of autologous bone marrow-derived mononuclear cells in patients with severe angina pectoris and stress-induced myocardial ischemia. Am J Cardiol. 2006;97:1326-1331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 77. | Rodrigo SF, van Ramshorst J, Beeres SL, Al Younis I, Dibbets-Schneider P, de Roos A, Fibbe WE, Zwaginga JJ, Schalij MJ, Bax JJ. Intramyocardial injection of bone marrow mononuclear cells in chronic myocardial ischemia patients after previous placebo injection improves myocardial perfusion and anginal symptoms: an intra-patient comparison. Am Heart J. 2012;164:771-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 78. | Heeschen C, Lehmann R, Honold J, Assmus B, Aicher A, Walter DH, Martin H, Zeiher AM, Dimmeler S. Profoundly reduced neovascularization capacity of bone marrow mononuclear cells derived from patients with chronic ischemic heart disease. Circulation. 2004;109:1615-1622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 454] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 79. | Sugihara S, Yamamoto Y, Matsuura T, Narazaki G, Yamasaki A, Igawa G, Matsubara K, Miake J, Igawa O, Shigemasa C. Age-related BM-MNC dysfunction hampers neovascularization. Mech Ageing Dev. 2007;128:511-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 80. | Mangi AA, Noiseux N, Kong D, He H, Rezvani M, Ingwall JS, Dzau VJ. Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med. 2003;9:1195-1201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1247] [Cited by in RCA: 1124] [Article Influence: 48.9] [Reference Citation Analysis (0)] |
| 81. | Kofidis T, de Bruin JL, Yamane T, Balsam LB, Lebl DR, Swijnenburg RJ, Tanaka M, Weissman IL, Robbins RC. Insulin-like growth factor promotes engraftment, differentiation, and functional improvement after transfer of embryonic stem cells for myocardial restoration. Stem Cells. 2004;22:1239-1245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 112] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 82. | Gnecchi M, He H, Noiseux N, Liang OD, Zhang L, Morello F, Mu H, Melo LG, Pratt RE, Ingwall JS. Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. FASEB J. 2006;20:661-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 968] [Cited by in RCA: 875] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 83. | Song H, Kwon K, Lim S, Kang SM, Ko YG, Xu Z, Chung JH, Kim BS, Lee H, Joung B. Transfection of mesenchymal stem cells with the FGF-2 gene improves their survival under hypoxic conditions. Mol Cells. 2005;19:402-407. [PubMed] |
| 84. | Dixit P, Katare R. Challenges in identifying the best source of stem cells for cardiac regeneration therapy. Stem Cell Res Ther. 2015;6:26. [PubMed] |
| 85. | Sun Q, Zhang Z, Sun Z. The potential and challenges of using stem cells for cardiovascular repair and regeneration. Genes Dis. 2014;1:113-119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 86. | Van Oorschot AA, Smits AM, Goumans MJ. Stem cells: the building blocks to repair the injured heart. Panminerva Med. 2010;52:97-110. [PubMed] |
| 87. | Henning RJ. Stem cells for cardiac repair: problems and possibilities. Future Cardiol. 2013;9:875-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 88. | Pelacho B, Mazo M, Gavira JJ, Prósper F. Adult stem cells: from new cell sources to changes in methodology. J Cardiovasc Transl Res. 2011;4:154-160. [PubMed] |
| 89. | Winter EM, van Oorschot AA, Hogers B, van der Graaf LM, Doevendans PA, Poelmann RE, Atsma DE, Gittenberger-de Groot AC, Goumans MJ. A new direction for cardiac regeneration therapy: application of synergistically acting epicardium-derived cells and cardiomyocyte progenitor cells. Circ Heart Fail. 2009;2:643-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 74] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 90. | Sheng CC, Zhou L, Hao J. Current stem cell delivery methods for myocardial repair. Biomed Res Int. 2013;2013:547902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 63] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 91. | Torella D, Ellison GM, Nadal-Ginard B, Indolfi C. Cardiac stem and progenitor cell biology for regenerative medicine. Trends Cardiovasc Med. 2005;15:229-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 92. | Cho HJ, Lee HJ, Youn SW, Koh SJ, Won JY, Chung YJ, Cho HJ, Yoon CH, Lee SW, Lee EJ. Secondary sphere formation enhances the functionality of cardiac progenitor cells. Mol Ther. 2012;20:1750-1766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Chawla M, Kounis NG S- Editor: Ji FF L- Editor: Wang TQ E- Editor: Jiao XK