Published online Mar 26, 2026. doi: 10.4252/wjsc.v18.i3.116298
Revised: January 12, 2026
Accepted: February 12, 2026
Published online: March 26, 2026
Processing time: 106 Days and 0.3 Hours
Chronic meniscal injuries pose a significant challenge in orthopedic medicine, frequently resulting in enduring pain, functional limitations, and progression to osteoarthritis. Existing treatment modalities have limitations, particularly for tears in the avascular zone.
To evaluate the clinical efficacy of autologous bone marrow-derived mesen
A prospective clinical trial was conducted between September 2023 and December 2024, enrolling 80 patients with chronic meniscal injuries identified through magnetic resonance imaging (MRI). The patients were administered an intra-articular injection of autologous BM-MSCs (40 × 106 cells). Primary outcomes included Visual Analog Scale pain, International Knee Documentation Committee and Lysholm Knee scores. Secondary outcomes comprised MRI T2 mapping values and meniscal morphology evaluations. Patients were evaluated at baseline and at 3 months, 6 months, and 12 months after injection.
Seventy-six patients completed the 12-month follow-up period. Visual Analog Scale pain scores significantly decreased from 6.8 ± 1.3 at baseline to 2.1 ± 0.9 at 12 months (P < 0.001). International Knee Documentation Committee scores improved from 42.3 ± 8.7 to 71.5 ± 10.2 (P < 0.001), while Lysholm scores improved from 58.4 ± 11.3 to 85.2 ± 9.8 (P < 0.001). MRI T2 mapping revealed a significant reduction in T2 relaxation times in the treated meniscus (38.5 ± 4.2 milliseconds to 31.2 ± 3.8 milliseconds, P < 0.001), indicating enhanced tissue quality. No serious adverse events were observed.
Autologous BM-MSC injection effectively reduces pain and improves function in patients with chronic meniscal injuries. Moreover, it appears to be a safe alternative to surgery in selected patients.
Core Tip: This prospective clinical study examined the therapeutic efficacy of autologous bone marrow-derived mesenchymal stem cell injections in patients with chronic meniscal injuries. Significant pain reduction and functional improvement were observed over 12 months. This was supported by enhanced Visual Analog Scale pain, International Knee Documentation Committee, and Lysholm scores, as well as magnetic resonance imaging T2 mapping, indicating enhanced meniscal tissue quality. The intervention proved to be safe, minimally invasive, and well-tolerated, suggesting that bone marrow-derived mesenchymal stem cell therapy could offer a viable regenerative option to surgery for chronic meniscal injuries. This approach may facilitate both structural restoration and clinical recovery.
- Citation: Fu X, Song H, Wu JS, Chen X. Prospective evaluation of autologous bone marrow-derived mesenchymal stem cell injection in meniscal injury. World J Stem Cells 2026; 18(3): 116298
- URL: https://www.wjgnet.com/1948-0210/full/v18/i3/116298.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i3.116298
The meniscus is vital in knee joint biomechanics as it distributes loads, absorbs shocks, stabilizes the joint, and protects articular cartilage[1]. Meniscal injuries are prevalent in orthopedic conditions, with an annual incidence of about 66 per 100000 individuals, greatly impacting quality of life and potentially hastening osteoarthritis development[2]. The restricted blood supply to the inner two-thirds of the meniscus poses a considerable healing challenge, particularly in the avascular zone where most degenerative tears occur[3].
The current treatment strategies for meniscal injuries vary from conservative management to surgical intervention. Recent evidence indicates that arthroscopic partial meniscectomy, previously the gold standard, may not yield better outcomes compared with conservative treatment and could hasten joint degeneration[4]. This shift in perspective has led to the investigation of regenerative medicine methods, notably mesenchymal stem cell (MSC) therapy, which presents the opportunity for tissue regeneration rather than solely symptom management[5].
Bone marrow-derived MSCs (BM-MSCs) have become a promising therapeutic strategy due to their multilineage differentiation potential, immunomodulatory effects, and reparative paracrine activity[6]. These cells can transform into meniscal fibrochondrocytes and generate crucial extracellular matrix components necessary for meniscal structure and function[7]. Additionally, BM-MSCs exhibit paracrine effects by secreting growth factors, cytokines, and extracellular vesicles, which regulate inflammation, enhance angiogenesis, and activate endogenous repair mechanisms[8].
Previous studies have shown the feasibility and safety of MSC therapy for meniscal injuries in both preclinical and clinical settings. Animal studies have indicated that MSCs improve meniscal healing, decrease inflammatory markers, and enhance the biomechanical properties of repaired tissues[9]. Clinical trials have documented enhancements in pain, function, and quality of life after MSC treatment. However, the majority of studies have concentrated on MSCs as a complement to surgical repair rather than a standalone treatment[10].
The mechanism of action of MSCs in meniscal repair involves both direct and indirect pathways. Direct differentiation into meniscal cells contributes to tissue regeneration, while paracrine signaling promotes endogenous repair by recruiting and activating resident progenitor cells[11]. MSCs also modulate the inflammatory microenvironment by suppressing proinflammatory cytokines and promoting anti-inflammatory mediators, thereby creating conditions favorable for tissue healing[12].
Recent advances in imaging technology, especially quantitative magnetic resonance imaging (MRI) techniques such as T2 mapping, have facilitated noninvasive evaluation of meniscal tissue quality and composition[13]. The T2 relaxation time is associated with collagen organization and water content, offering objective indicators of tissue integrity that enhance clinical assessments[14]. These imaging biomarkers provide valuable insights into the biological impacts of regenerative therapies, extending beyond conventional morphological evaluations.
Despite the increasing interest in MSC therapy for meniscal injuries, numerous questions remain unanswered. The optimal cell dose, delivery method, and patient selection criteria necessitate further clarification[15]. Additionally, investigating the long-term durability of treatment effects and the potential for disease modification is essential. This study aimed to fill these gaps by assessing the clinical efficacy of autologous BM-MSC injections in a well-characterized cohort of patients with chronic meniscal injuries. The evaluation utilized both clinical and advanced imaging outcomes to comprehensively assess treatment response.
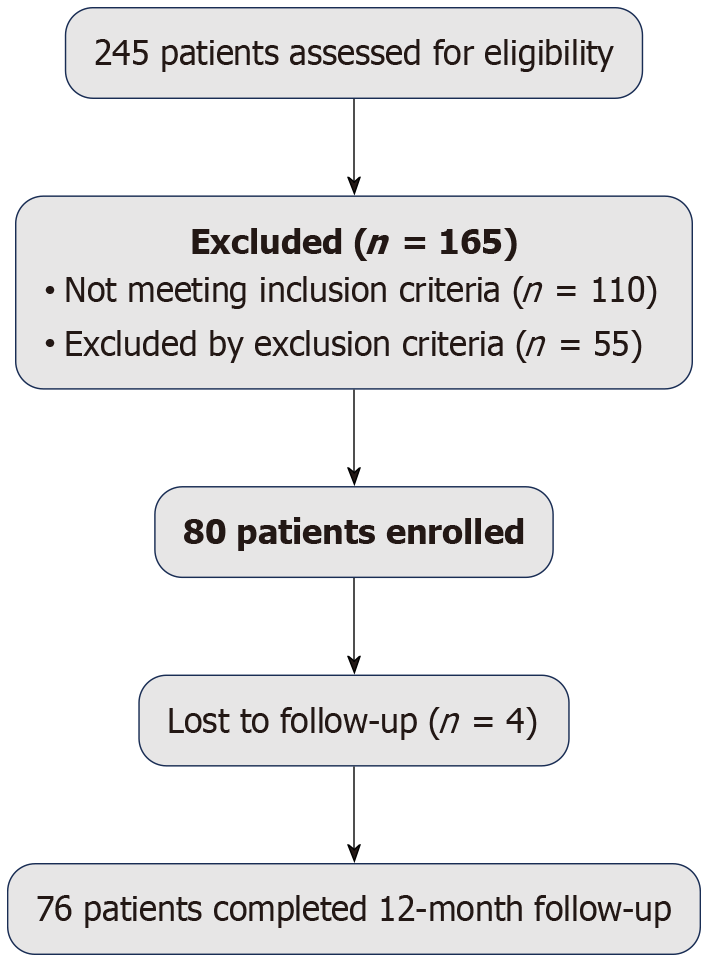
This prospective, single-arm clinical trial was conducted at our institution between September 2023 and December 2024. The study was approved by the Institutional Review Board and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants after a detailed explanation of the study procedures, potential risks, and anticipated benefits. The study design followed recommendations from the International Society for Cellular Therapy regarding MSC characterization. Moreover, it adhered to the Minimum Information for Studies Evaluating Biologics in Orthopaedics guidelines to ensure standardized reporting of biologic interventions. Patients were recruited through the orthopedic and sports medicine clinics at our institution, including referrals from primary care physicians and self-referrals in response to study advertisements. A total of 245 patients presenting with knee pain and suspected meniscal pathology were screened for eligibility. Following a comprehensive evaluation, including clinical examination, MRI, and laboratory testing, 80 patients who met the inclusion criteria were enrolled. The patient screening and enrollment process is summarized in Figure 1. Sample size calculation was based on previously reported effect sizes for pain reduction following MSC therapy, providing 80% statistical power to detect a clinically meaningful difference of 2 points on the Visual Analog Scale (VAS) and accounting for an anticipated dropout rate of 10%.
Inclusion criteria were: (1) Age between 30 years and 65 years to ensure adequate bone marrow cellularity while minimizing age-related confounding; (2) Chronic meniscal injury confirmed by 3.0T MRI demonstrating grade II or III meniscal signal changes according to the Crues classification, with symptoms persisting for at least three months; (3) Failure of conservative management, including a minimum of six weeks of physical therapy and oral anti-inflammatory medication; (4) A VAS pain score of at least 4 during daily activities; (5) Body mass index (BMI) between 20 kg/m2 and
Exclusion criteria included: (1) Complete meniscal root tears or bucket-handle tears requiring immediate surgical intervention; (2) Kellgren-Lawrence grade III or IV osteoarthritis indicating advanced degenerative disease; (3) Significant ligamentous instability, including complete anterior cruciate ligament rupture or grade III collateral ligament injury; (4) Previous knee surgery within the preceding 12 months or a history of total knee arthroplasty; (5) Systemic inflammatory arthropathies such as rheumatoid arthritis, psoriatic arthritis, or systemic lupus erythematosus; (6) Active infection or prior septic arthritis; (7) Malignancy within the past five years, excluding completely excised non-melanoma skin cancer; (8) Use of immunosuppressive therapy or systemic corticosteroids within three months before enrolment; (9) Intra-articular injections, including corticosteroids or hyaluronic acid, within six months; (10) Coagulopathy or anticoagulant therapy that could not be safely discontinued; (11) Pregnancy or lactation; (12) Significant medical comorbidities, including uncontrolled diabetes mellitus (glycated hemoglobin > 8%), severe cardiovascular disease, or advanced chronic kidney disease (estimated glomerular filtration rate < 30 mL/minute/1.73 m2); and (13) Contraindications to MRI, including non-compatible implants or severe claustrophobia.
Bone marrow aspiration was conducted under sterile conditions in a procedural room with continuous cardiac monitoring and pulse oximetry. Following standard preparation and draping, the posterior superior iliac spine was located and infiltrated with 10 mL of 1% lidocaine for local anesthesia, ensuring sufficient periosteal anesthesia to reduce procedural discomfort. Utilizing an 11-gauge Jamshidi needle, multiple small-volume aspirations (2-3 mL each) were obtained from various sites within the iliac crest to optimize MSC yield and minimize peripheral blood contamination. A total of 60 mL of bone marrow aspirate was collected using heparinized syringes.
The aspirated bone marrow was promptly transported to our Good Manufacturing Practice-certified cell-processing facility, while maintaining a chain of custody documentation. Mononuclear cells were isolated through density gradient centrifugation using Ficoll-Paque (density, 1.077 g/mL) at 400 × g for 30 minutes at room temperature. The mononuclear cell layer was meticulously collected, washed twice with phosphate-buffered saline containing 2% human serum albumin, and then suspended in alpha-minimum essential medium supplemented with 10% autologous serum, 1% penicillin-streptomycin, and 2 mmol/L L-glutamine. Cells were seeded at a density of 1 × 105 cells/cm2 in T-175 flasks and incubated at 37 °C in 5% CO2 with 95% humidity.
The culture medium was changed every three days, and non-adherent cells were removed with the initial medium change at 72 hours. Upon reaching 80%-85% confluence (usually within 14-17 days), cells were harvested using 0.25% trypsin-ethylenediaminetetraacetic acid, washed, and passaged at a 1:3 ratio. MSCs were expanded to passage 3 to achieve adequate cell numbers while preserving stemness characteristics, with regular morphological assessments confirming a spindle-shaped fibroblastic appearance. Quality control testing at each passage involved evaluating cell viability through trypan blue exclusion (required > 95%), calculating population doubling time, and conducting mycoplasma testing using polymerase chain reaction.
The expanded MSCs underwent comprehensive characterization following the criteria set by the International Society for Cellular Therapy. Flow cytometry analysis utilized a panel of surface markers, confirming high expression (> 95%) of CD73, CD90, and CD105, and low expression (< 2%) of CD14, CD19, CD34, CD45, and HLA-DR. Trilineage differentiation potential was confirmed by culturing MSCs in adipogenic, osteogenic, and chondrogenic differentiation media for 21 days, followed by staining with Oil Red O, Alizarin Red, and Alcian Blue, respectively. Differentiation markers were quantitatively assessed using reverse transcription-polymerase chain reaction. Sterility testing included aerobic and anaerobic bacterial cultures maintained for 14 days, fungal cultures, and endotoxin testing using the Limulus Amebocyte Lysate assay (required < 0.5 EU/mL). Karyotype analysis involved examining 20 metaphase chromosomes to exclude chromosomal abnormalities. Cell senescence was evaluated through beta-galactosidase staining, with less than 5% senescent cells considered acceptable. The final cell product was suspended in 3 mL of lactated Ringer’s solution supplemented with 1% human serum albumin and 10% autologous serum, maintaining cell viability above 90% for up to 4 hours at 4 °C.
All injections were administered by the same experienced orthopedic surgeon to ensure procedural consistency. The procedure took place in a sterile environment with the patient in a supine position and the knee flexed at 90 degrees. The superolateral approach was utilized for intra-articular access, followed by skin preparation with chlorhexidine and sterile draping. The injection site was numbed with 1% lidocaine (2 mL) for local anesthesia. Under ultrasound guidance using a high-frequency linear transducer, a 21-gauge needle was inserted into the joint space, confirming intra-articular placement by aspiration of synovial fluid when present. The prepared MSC suspension containing 40 × 106 cells in 3 mL was slowly injected over 30 seconds to minimize shear stress on cells and ensure even distribution within the joint space. This cell dose was chosen based on previous clinical studies demonstrating efficacy and safety of similar doses (20 × 106 to 50 × 106 cells) in knee joint regenerative therapies, balancing therapeutic potential with practical cell expansion considerations. After injection, the needle was removed, and a gentle passive range of motion was performed to facilitate cell distribution. A sterile dressing was applied, and the patients were monitored for 30 minutes for any immediate adverse reactions. Post-injection activity modification included avoiding weight-bearing for 48 hours, followed by progressive weight-bearing as tolerated with the use of crutches for one week if necessary.
Clinical evaluations were conducted at baseline (within 2 weeks before injection) and at 3 months, 6 months, and 12 months post-injection by independent assessors who were blinded to the injection parameters. The VAS for pain was utilized during weight-bearing activities, where patients indicated their pain level on a 100-mm horizontal line ranging from “no pain” to “worst imaginable pain”. The VAS was consistently administered with standardized instructions, prompting patients to rate their average pain over the past week for enhanced reliability. The International Knee Documentation Committee (IKDC) Subjective Knee Form was used to assess symptoms, function, and sports activities, with scores on a scale from 0 to 100, where higher scores denoted better function. The questionnaire was self-admi
MRI examinations were conducted on a 3.0T scanner (Siemens Magnetom Prisma, Erlangen, Germany) using a dedicated 15-channel knee coil at baseline and at 6 months and 12 months post-injection. The standardized protocol comprised: (1) Sagittal proton density-weighted sequences with and without fat suppression [repetition time/echo time (TR/TE): 3000/30 milliseconds, slice thickness: 3 mm, field of view: 160 mm, matrix: 384 × 384]; (2) Coronal T2-weighted sequences with fat suppression (TR/TE: 3500/70 milliseconds); (3) Axial proton density-weighted sequences (TR/TE: 3000/30 milliseconds); and (4) Three-dimensional double-echo steady-state sequences for morphological assessment (TR/TE: 14.5/5 milliseconds, slice thickness: 0.7 mm). Quantitative T2 mapping was performed using a multi-echo spin-echo sequence with eight echo times (TE: 10 milliseconds, 20 milliseconds, 30 milliseconds, 40 milliseconds, 50 milliseconds, 60 milliseconds, 70 milliseconds, and 80 milliseconds), a TR of 2000 milliseconds, a slice thickness of 3 mm, and an in-plane resolution of 0.5 mm × 0.5 mm. T2 relaxation times were calculated using a mono-exponential fitting algorithm with noise correction. The meniscus was manually segmented by an experienced musculoskeletal radiologist, blinded to the clinical outcomes, into the anterior horn, body, and posterior horn regions. Mean T2 values were calculated for each region and for the entire meniscus, with lower T2 values indicating better tissue organization and reduced water content. The MRI Osteoarthritis Knee Score was utilized for the semiquantitative evaluation of meniscal morphology, signal intensity, and extrusion. Meniscal extrusion was measured as the distance between the peripheral meniscal margin and the tibial plateau edge on coronal images, with more than 3 mm considered pathological extrusion. The presence of bone marrow lesions, cartilage defects, and synovitis has also been documented as potential confounding factors affecting the clinical outcomes.
A standardized progressive rehabilitation protocol was implemented for all patients under the supervision of experienced physical therapists. During phase 1 (weeks 0-2), the focus was on protection and inflammation control. Interventions included non-weight-bearing or toe-touch weight-bearing with crutches, gentle passive range of motion exercises within pain-free limits, quadriceps isometric exercises, and cryotherapy for 20 minutes three times daily. Patients were advised against using anti-inflammatory medications during this phase to prevent interference with the MSC-mediated healing response. Phase 2 (weeks 3-6) emphasized progressive loading and range of motion restoration. Patients advanced to partial weight-bearing (50% body weight) using a bathroom scale for feedback, performed active-assisted range of motion exercises targeting full extension and 120° of flexion, started closed kinetic chain exercises such as mini-squats and heel raises, and commenced stationary cycling with minimal resistance once achieving 100° of flexion. Proprioceptive training with balance boards was introduced as tolerated. Phase 3 (weeks 7-12) focused on strength and functional recovery, advancing to full weight-bearing and discontinuation of assistive devices upon normalized gait. Progressive resistance training was initiated using elastic bands and weight machines to target the quadriceps, hamstrings, and hip muscles. Functional exercises like step-ups, lunges, and single-leg squats were introduced based on individual capacity. Aquatic therapy was included for patients experiencing persistent pain during land-based exercise. Low-impact aerobic conditioning, including cycling, elliptical training, and swimming, was recommended. Phase 4 (months 4-6) focused on return to activities, incorporating sport-specific training for athletic patients, including agility drills, plyometric exercises, and gradual return to running programs following established guidelines. Patients were cleared for return to sports upon achieving a limb symmetry index greater than 90% on functional testing, full pain-free range of motion, and absence of joint effusion. Education on activity modification, weight management, and long-term joint protection strategies was provided throughout the rehabilitation period.
Statistical analysis was conducted using SPSS version 28.0 (IBM Corporation, Armonk, NY, United States) with a significance level set at P < 0.05 for all tests. Continuous variables were presented as mean ± SD for normally distributed data or median with interquartile range (IQR) for non-normally distributed data. Normality was assessed using the Shapiro-Wilk test and visual inspection of histograms and Q-Q plots. Categorical variables were reported as frequencies and percentages. Primary outcome measures (VAS, IKDC, and Lysholm scores) were analyzed using repeated-measures ANOVA with Greenhouse-Geisser correction for violations of sphericity. Post hoc pairwise comparisons with Bonferroni adjustments were conducted to identify specific time points showing significant changes. Effect sizes were calculated using Cohen’s d to quantify treatment effects, with values of 0.2, 0.5, and 0.8 representing small, medium, and large effects, respectively. Secondary outcomes, including MRI T2 mapping values, were analyzed using paired t-tests for comparisons between baseline and follow-up time points. Correlation analyses using Pearson or Spearman coefficients were employed to examine the relationship between clinical outcomes and imaging parameters. Subgroup analyses based on age (< 50 years vs ≥ 50 years), BMI (< 30 kg/m2 vs ≥ 30 kg/m2), meniscal tear location (medial vs lateral), and baseline symptom severity were conducted using mixed-effects models to identify factors influencing treatment response. Missing data were addressed using multiple imputation with 10 imputed datasets for patients with incomplete follow-up, assuming data were missing at random. Sensitivity analyses comparing the complete case analysis with the imputed data were performed to assess the robustness of the findings. The number needed to treat was calculated based on the proportion of patients achieving minimal clinically important differences in primary outcomes. All analyses were carried out following intention-to-treat principles, with a per-protocol analysis conducted as a sensitivity analysis.
Among the 245 patients initially screened, 80 met all inclusion criteria and were enrolled between September 2023 and March 2024. Four patients (5%) were lost to follow-up (two relocated, one withdrew consent, and one underwent unrelated surgery), resulting in 76 patients (95%) completing the 12-month assessment. The study cohort included 42 males (52.5%) and 38 females (47.5%), with a mean age of 48.3 ± 9.7 years (range: 30-65 years). The mean BMI was 27.2 ± 3.8 kg/m2, with 31 patients (38.8%) classified as overweight (BMI = 25-30 kg/m2) and 18 patients (22.5%) classified as obese (BMI ≥ 30 kg/m2). The median duration of symptoms prior to enrollment was 14.5 months (IQR: 8-24 months), with 58 patients (72.5%) reporting symptoms for more than 12 months. Regarding meniscal pathology, 52 patients (65%) had medial meniscal tears, 21 patients (26.3%) had lateral meniscal tears, and 7 patients (8.7%) had both medial and lateral involvement. On MRI, 45 patients (56.3%) demonstrated grade II signal changes and 35 patients (43.7%) demonstrated grade III changes. The posterior horn was involved in 61 patients (76.3%), the meniscal body in 38 patients (47.5%), and the anterior horn in 15 patients (18.8%), with 28 patients (35%) showing multi-regional involvement.
The VAS pain scores showed significant improvement at all follow-up time points (Table 1). The average baseline VAS score of 6.8 ± 1.3 decreased to 4.2 ± 1.5 at 3 months (P < 0.001), 3.1 ± 1.2 at 6 months (P < 0.001), and 2.1 ± 0.9 at 12 months (P < 0.001). The effect size for pain reduction was substantial (Cohen’s d = 3.92) after 12 months. Notably, 68 patients (89.5%) experienced a reduction of ≥ 2 points on the VAS scale, which is considered clinically significant, and 42 patients (55.3%) achieved ≥ 50% pain reduction from baseline.
| Outcome measure | Baseline | 3 months | 6 months | 12 months | P value1 |
| VAS pain score (0-10) | 6.8 ± 1.3 | 4.2 ± 1.5a | 3.1 ± 1.2a | 2.1 ± 0.9a | < 0.001 |
| IKDC score (0-100) | 42.3 ± 8.7 | 55.6 ± 9.3a | 64.8 ± 10.1a | 71.5 ± 10.2a | < 0.001 |
| Lysholm score (0-100) | 58.4 ± 11.3 | 69.2 ± 10.8a | 78.3 ± 10.2a | 85.2 ± 9.8a | < 0.001 |
| KOOS pain | 45.2 ± 12.1 | 58.3 ± 11.5a | 68.4 ± 10.8a | 75.6 ± 11.2a | < 0.001 |
| KOOS symptoms | 48.6 ± 13.4 | 60.2 ± 12.7a | 69.8 ± 11.9a | 76.3 ± 12.1a | < 0.001 |
| KOOS ADL | 52.3 ± 14.2 | 64.5 ± 13.1a | 73.2 ± 12.5a | 79.8 ± 11.8a | < 0.001 |
| KOOS Sports/Rec | 35.4 ± 15.6 | 45.2 ± 14.8a | 56.3 ± 15.2a | 65.4 ± 14.9a | < 0.001 |
| KOOS QoL | 38.7 ± 16.2 | 51.3 ± 15.4a | 62.5 ± 14.7a | 70.2 ± 13.8a | < 0.001 |
| ROM flexion (degrees) | 118.5 ± 12.3 | 125.3 ± 10.2a | 131.2 ± 8.7a | 134.5 ± 7.2a | < 0.001 |
| ROM extension (degrees) | -3.2 ± 2.1 | -1.8 ± 1.5a | -0.8 ± 1.2a | -0.2 ± 0.8a | < 0.001 |
IKDC scores demonstrated a progressive enhancement from 42.3 ± 8.7 at baseline to 71.5 ± 10.2 at 12 months (P < 0.001), with 62 patients (81.6%) achieving the minimal clinically important difference of 8.8 points. Similarly, Lysholm scores increased from 58.4 ± 11.3 to 85.2 ± 9.8 (P < 0.001), with 65 patients (85.5%) surpassing the 8.9-point minimum clinically important difference threshold. The percentage of patients with “good” or “excellent” Lysholm scores (≥ 84) rose from 0% at baseline to 68.4% at 12 months.
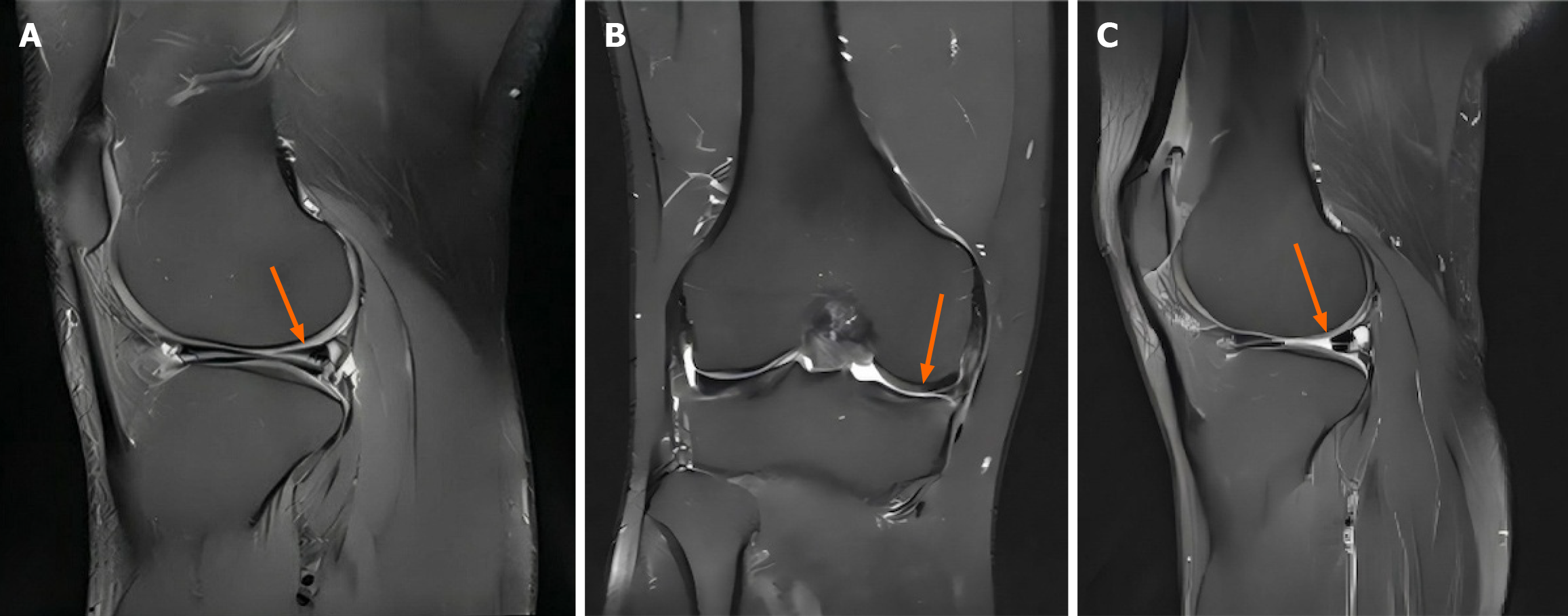
Quantitative T2 mapping demonstrated significant alterations in meniscal tissue composition post-MSC therapy (Table 2, Figure 2). The average T2 relaxation time for the complete meniscus decreased from 38.5 ± 4.2 milliseconds initially to 34.8 ± 3.9 milliseconds at 6 months (P < 0.001) and 31.2 ± 3.8 milliseconds at 12 months (P < 0.001), indicating enhanced tissue structure and reduced moisture content. Regional assessment indicated that the posterior horn, initially exhibiting the highest T2 values (41.2 ± 5.1 milliseconds), displayed the most substantial enhancement (reduction to 32.5 ± 4.2 milliseconds at 12 months, P < 0.001).
| Meniscal region | Baseline (milliseconds) | 6 months (milliseconds) | 12 months (milliseconds) | Change from baseline (milliseconds) |
| Anterior horn | 35.8 ± 3.9 | 33.2 ± 3.5b | 30.1 ± 3.2a | -15.9 |
| Body | 37.4 ± 4.1 | 34.5 ± 3.8b | 31.3 ± 3.5a | -16.3 |
| Posterior horn | 41.2 ± 5.1 | 36.8 ± 4.5a | 32.5 ± 4.2a | -21.1 |
| Overall meniscus | 38.5 ± 4.2 | 34.8 ± 3.9a | 31.2 ± 3.8a | -19.0 |
Morphological assessment using the MRI Osteoarthritis Knee Score criteria indicated an enhancement in meniscal signal intensity in 48 patients (63.2%), among whom 31 patients (40.8%) exhibited complete resolution of intrasubstance signal abnormalities. Initially observed in 34 patients (mean: 3.8 ± 1.2 mm), meniscal extrusion reduced to 2.9 ± 1.0 mm at 12 months (P = 0.003), with 12 patients (35.3%) achieving a reduction below the 3 mm threshold. There was no progression of existing meniscal tears or development of new tears during the follow-up period.
Age-stratified analysis indicated that younger patients (aged < 50 years, n = 42) exhibited more substantial enhancements in IKDC scores (mean difference: 32.4 ± 11.2) compared to older patients (aged ≥ 50 years, n = 38; mean difference: 25.8 ± 10.5, P = 0.024). However, both age groups achieved clinically meaningful improvements. Analysis by BMI subgroups revealed no significant disparity in pain reduction between patients with BMI < 30 kg/m2 and those with BMI ≥ 30 kg/m2 (P = 0.182). However, individuals with lower BMI demonstrated a tendency toward superior functional outcomes. Patients with isolated medial meniscus tears showed slightly better outcomes compared to those with lateral or combined tears, achieving higher final Lysholm scores (86.8 ± 9.2 vs 82.5 ± 10.1, P = 0.048). Baseline symptom duration did not significantly affect the treatment response, with similar improvements observed in patients with symptoms at < 12 months vs ≥ 12 months (P = 0.364).
No significant adverse events associated with the MSC injections were documented throughout the study. Minor adverse events comprised transient pain at the injection site in 18 patients (23.7%), mild swelling in the knee lasting less than 48 hours in 12 patients (15.8%), and temporary stiffness in eight patients (10.5%). All minor events resolved spontaneously, without requiring intervention. There were no instances of infections, allergic reactions, or systemic complications observed. Laboratory assessments at 3 months and 12 months revealed no notable alterations in the complete blood count, inflammatory markers (erythrocyte sedimentation rate, C-reactive protein), or metabolic parameters.
At the 12-month follow-up, 58 patients (76.3%) indicated satisfaction with the treatment outcomes, with the majority reporting being “satisfied” or “very satisfied”. Out of the 32 patients who engaged in recreational sports before the injury, 24 (75%) successfully resumed their previous activity levels, although 8 patients opted for lower-impact sports. The limb symmetry index for the single-leg hop test showed improvement from 68.4% ± 12.3% at baseline to 89.2% ± 8.7% at 12 months (P < 0.001) among patients capable of performing the test.
Pearson’s correlation analysis demonstrated moderately negative correlations between T2 relaxation times and the following clinical scores at 12 months: VAS pain (r = -0.482, P < 0.001), IKDC (r = 0.524, P < 0.001), and Lysholm (r = 0.498, P < 0.001). Patients with more than 20% reduction in T2 values exhibited significantly better clinical outcomes compared to those with less than 20% reduction (mean IKDC difference: 75.3 ± 9.1 vs 66.8 ± 10.5, P = 0.002).
This study demonstrates that autologous BM-MSC injection offers substantial and lasting enhancements in pain and functional outcomes for patients with chronic meniscal injuries. The observed clinical advantages, together with measurable enhancements in meniscal tissue quality revealed by MRI T2 mapping, indicate that BM-MSC therapy has regenerative effects. These findings suggest that BM-MSC therapy may present a promising treatment option for selected patients with meniscal pathology who have not responded to conservative management.
The significant pain reduction observed in our group, with average VAS scores decreasing from 6.8 to 2.1 over 12 months, is consistent with recent meta-analyses demonstrating the effectiveness of MSC therapy for musculoskeletal conditions[16]. The extent of improvement observed in this study is greater than that reported for other non-surgical interventions, such as platelet-rich plasma and hyaluronic acid injections. A recent systematic review by Li and Weng[17] revealed that VAS reductions of 0.76 points were typically achieved with platelet-rich plasma injections for meniscal tears, which is significantly lower than the 4.7-point reduction noted in our study. The superior outcomes of MSCs may be attributed to their broader mechanisms of action, which include the release of growth factors, direct cellular differentiation, and potent immunomodulatory effects[18].
The notable progression in functional scores during the 12-month follow-up period is particularly noteworthy. In contrast to corticosteroid injections that provide temporary relief and surgical debridement that may accelerate degeneration, MSC therapy promotes ongoing tissue healing[19]. The sustained enhancement between 6 months and 12 months indicates that the regenerative processes initiated by MSCs extend beyond the initial treatment phase. This aligns with preclinical studies illustrating that transplanted MSCs can persist and remain metabolically active in the joint environment for several months, continuously releasing bioactive factors, and potentially differentiating into meniscal tissue[20].
Quantitative T2 mapping has shown clear evidence of enhanced meniscal tissue quality post-MSC treatment. A 19% decrease in the total T2 relaxation times indicates reduced water content and enhanced collagen structure, indicative of healthier meniscal tissue[21]. These results hold significance as prior T2 mapping studies on meniscal injuries have mainly concentrated on diagnostic uses rather than treatment monitoring. Our findings propose that T2 mapping could be a valuable biomarker for monitoring responses to regenerative therapies. The notably greater enhancement in the posterior horn, where tears are most frequent and healing is typically limited due to poor vascularity, implies that MSCs might be particularly effective in addressing injuries in anatomical regions that are conventionally challenging[22].
The correlation between imaging improvements and clinical outcomes enhances the evidence for the true biological effect of MSC therapy, rather than just a placebo response. Patients with larger reductions in T2 values demonstrated better functional improvements, indicating a direct link between clinical benefits and tissue regeneration. This association has significant implications for patient selection and prognosis, as baseline T2 values can assist in identifying individuals most likely to benefit from MSC therapy[23].
Our safety profile compares favorably with those of surgical interventions and other injection therapies. The lack of serious adverse events and the minor, self-limiting nature of the observed side effects support the safety of intra-articular MSC injections when conducted with appropriate cell processing and quality control measures[24]. This is particularly relevant considering the current apprehensions regarding the potential risks associated with cell-based therapies, such as the theoretical risks of tumorigenicity or ectopic tissue formation. Our thorough cell characterization protocol, which includes karyotype analysis and senescence assessment, probably played a role in the outstanding safety profile noted in this study.
The subgroup analysis reveals superior outcomes in younger patients. This finding is consistent with the recognized age-related decrease in endogenous repair capability and might indicate enhanced integration of transplanted cells with the host tissue in younger individuals[25]. However, the observation that older patients attained clinically significant improvements implies that age by itself is not a contraindication for MSC therapy. The absence of a substantial impact of BMI on outcomes differs from surgical literature that demonstrates inferior results in obese patients, potentially due to MSC therapy targeting biological rather than solely mechanical factors[26].
Comparison of surgical outcomes is particularly relevant considering the current shift away from arthroscopic debridement for degenerative meniscal tears. Recent randomized controlled trials have indicated that arthroscopic partial meniscectomy does not yield superior outcomes compared to sham surgery or physical therapy alone for degenerative tears[27]. In contrast, our findings exhibited distinct superiority over the initial status, showing sustained enhancement over time. Moreover, unlike meniscectomy, which involves tissue removal and may hasten osteoarthritis, MSC therapy seeks to enhance tissue integrity and alter disease progression[28].
Mechanisms underlying the observed clinical improvements likely involve multiple pathways. Although the relative contribution of this mechanism to paracrine effects remains unclear, the direct differentiation of MSCs into meniscal fibrochondrocytes may contribute to tissue regeneration[29]. Prolonged clinical improvement suggests that beyond immediate anti-inflammatory effects, MSCs may establish a regenerative microenvironment that promotes ongoing healing. Recent evidence indicates that MSC-derived extracellular vesicles carrying microRNAs, proteins, and lipids play crucial roles in intercellular communication and tissue repair[30]. These vesicles modulate inflammation, promote angiogenesis, and stimulate the proliferation of resident progenitor cells, which may persist even after the transplanted cells are no longer viable.
This study has several limitations. The single-arm design without a control group limits our ability to definitively attribute improvements to MSC therapy rather than to placebo effects or natural history. However, the magnitude and duration of the improvements, coupled with objective imaging changes, suggest true biological effects beyond the placebo. The relatively short follow-up period of 12 months precluded the assessment of long-term durability and potential disease-modifying effects. Long-term studies are needed to determine whether MSC therapy can prevent or delay the progression to osteoarthritis and to confirm its long-term safety beyond 12 months. Heterogeneity in the meniscal tear patterns and locations may have influenced the outcomes, although our sample size was limited for more detailed subgroup analyses. Additionally, all injections were performed by a single surgeon, which ensured procedural consistency but may limit generalizability and reproducibility across different practitioners. We did not perform second-look arthroscopy to directly visualize meniscal healing; instead, we relied on noninvasive imaging assessment.
This study provides convincing evidence that autologous BM-MSC injection is a safe and effective treatment for chronic meniscal injuries, leading to significant improvements in pain, function, and tissue quality over 12 months. The treatment targets the underlying pathology through regenerative mechanisms rather than solely offering symptomatic relief, presenting a paradigm shift in meniscal injury management. Although additional research is necessary to refine protocols and validate long-term benefits, MSC therapy signifies a promising advancement in the nonsurgical management of meniscal pathology and could aid in maintaining joint health and preventing progression to osteoarthritis.
| 1. | Mameri ES, Dasari SP, Fortier LM, Verdejo FG, Gursoy S, Yanke AB, Chahla J. Review of Meniscus Anatomy and Biomechanics. Curr Rev Musculoskelet Med. 2022;15:323-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 95] [Reference Citation Analysis (1)] |
| 2. | Gee SM, Tennent DJ, Cameron KL, Posner MA. The Burden of Meniscus Injury in Young and Physically Active Populations. Clin Sports Med. 2020;39:13-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 3. | Yan W, Dai W, Cheng J, Fan Y, Wu T, Zhao F, Zhang J, Hu X, Ao Y. Advances in the Mechanisms Affecting Meniscal Avascular Zone Repair and Therapies. Front Cell Dev Biol. 2021;9:758217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 4. | Sihvonen R, Paavola M, Malmivaara A, Itälä A, Joukainen A, Nurmi H, Kalske J, Ikonen A, Järvelä T, Järvinen TAH, Kanto K, Karhunen J, Knifsund J, Kröger H, Kääriäinen T, Lehtinen J, Nyrhinen J, Paloneva J, Päiväniemi O, Raivio M, Sahlman J, Sarvilinna R, Tukiainen S, Välimäki VV, Äärimaa V, Toivonen P, Järvinen TLN; FIDELITY (Finnish Degenerative Meniscal Lesion Study) Investigators. Arthroscopic partial meniscectomy versus placebo surgery for a degenerative meniscus tear: a 2-year follow-up of the randomised controlled trial. Ann Rheum Dis. 2018;77:188-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 109] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 5. | Bian Y, Wang H, Zhao X, Weng X. Meniscus repair: up-to-date advances in stem cell-based therapy. Stem Cell Res Ther. 2022;13:207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 6. | Sekiya I, Koga H, Otabe K, Nakagawa Y, Katano H, Ozeki N, Mizuno M, Horie M, Kohno Y, Katagiri K, Watanabe N, Muneta T. Additional Use of Synovial Mesenchymal Stem Cell Transplantation Following Surgical Repair of a Complex Degenerative Tear of the Medial Meniscus of the Knee: A Case Report. Cell Transplant. 2019;28:1445-1454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 7. | Chen K, Man C, Zhang B, Hu J, Zhu SS. Effect of in vitro chondrogenic differentiation of autologous mesenchymal stem cells on cartilage and subchondral cancellous bone repair in osteoarthritis of temporomandibular joint. Int J Oral Maxillofac Surg. 2013;42:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 8. | Kou M, Huang L, Yang J, Chiang Z, Chen S, Liu J, Guo L, Zhang X, Zhou X, Xu X, Yan X, Wang Y, Zhang J, Xu A, Tse HF, Lian Q. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool? Cell Death Dis. 2022;13:580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 507] [Cited by in RCA: 437] [Article Influence: 109.3] [Reference Citation Analysis (6)] |
| 9. | Qi Y, Chen G, Feng G. Osteoarthritis prevention and meniscus regeneration induced by transplantation of mesenchymal stem cell sheet in a rat meniscal defect model. Exp Ther Med. 2016;12:95-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 10. | Hatsushika D, Muneta T, Nakamura T, Horie M, Koga H, Nakagawa Y, Tsuji K, Hishikawa S, Kobayashi E, Sekiya I. Repetitive allogeneic intraarticular injections of synovial mesenchymal stem cells promote meniscus regeneration in a porcine massive meniscus defect model. Osteoarthritis Cartilage. 2014;22:941-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 124] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 11. | Gorgun C, Ceresa D, Lesage R, Villa F, Reverberi D, Balbi C, Santamaria S, Cortese K, Malatesta P, Geris L, Quarto R, Tasso R. Dissecting the effects of preconditioning with inflammatory cytokines and hypoxia on the angiogenic potential of mesenchymal stromal cell (MSC)-derived soluble proteins and extracellular vesicles (EVs). Biomaterials. 2021;269:120633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 88] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 12. | Tan SHS, Wong JRY, Sim SJY, Tjio CKE, Wong KL, Chew JRJ, Hui JHP, Toh WS. Mesenchymal stem cell exosomes in bone regenerative strategies-a systematic review of preclinical studies. Mater Today Bio. 2020;7:100067. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 107] [Article Influence: 17.8] [Reference Citation Analysis (1)] |
| 13. | Zhao H, Li H, Liang S, Wang X, Yang F. T2 mapping for knee cartilage degeneration in young patients with mild symptoms. BMC Med Imaging. 2022;22:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 14. | Kajabi AW, Zbýň Š, Smith JS, Hedayati E, Knutsen K, Tollefson LV, Homan M, Abbasguliyev H, Takahashi T, Metzger GJ, LaPrade RF, Ellermann JM. Seven tesla knee MRI T2*-mapping detects intrasubstance meniscus degeneration in patients with posterior root tears. Radiol Adv. 2024;1:umae005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 15. | Mazy D, Lu D, Leclerc S, Laor B, Wang J, Pinvicy A, Moldovan F, Nault ML. Animal models used in meniscal repair research from ex vivo to in vivo: A systematic review. J Orthop. 2024;55:23-31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 16. | Matas J, García C, Poblete D, Vernal R, Ortloff A, Luque-Campos N, Hidalgo Y, Cuenca J, Infante C, Cadiz MI, Khoury M, Luz-Crawford P, Espinoza F. A Phase I Dose-Escalation Clinical Trial to Assess the Safety and Efficacy of Umbilical Cord-Derived Mesenchymal Stromal Cells in Knee Osteoarthritis. Stem Cells Transl Med. 2024;13:193-203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 17. | Li Z, Weng X. Platelet-rich plasma use in meniscus repair treatment: a systematic review and meta-analysis of clinical studies. J Orthop Surg Res. 2022;17:446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (1)] |
| 18. | Caplan AI. There Is No "Stem Cell Mess". Tissue Eng Part B Rev. 2019;25:291-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 19. | Jacob G, Shimomura K, Krych AJ, Nakamura N. The Meniscus Tear: A Review of Stem Cell Therapies. Cells. 2019;9:92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 70] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 20. | Ozeki N, Kohno Y, Kushida Y, Watanabe N, Mizuno M, Katano H, Masumoto J, Koga H, Sekiya I. Synovial mesenchymal stem cells promote the meniscus repair in a novel pig meniscus injury model. J Orthop Res. 2021;39:177-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 21. | Chaudhari AS, Black MS, Eijgenraam S, Wirth W, Maschek S, Sveinsson B, Eckstein F, Oei EHG, Gold GE, Hargreaves BA. Five-minute knee MRI for simultaneous morphometry and T(2) relaxometry of cartilage and meniscus and for semiquantitative radiological assessment using double-echo in steady-state at 3T. J Magn Reson Imaging. 2018;47:1328-1341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 22. | Qu F, Li Q, Wang X, Cao X, Zgonis MH, Esterhai JL, Shenoy VB, Han L, Mauck RL. Maturation State and Matrix Microstructure Regulate Interstitial Cell Migration in Dense Connective Tissues. Sci Rep. 2018;8:3295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 23. | Li C, Hu X, Meng Q, Zhang X, Zhu J, Dai L, Cheng J, Zhong M, Shi W, Ren B, Zhang J, Fu X, Duan X, Ao Y. The potential of using semitendinosus tendon as autograft in rabbit meniscus reconstruction. Sci Rep. 2017;7:7033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 24. | Rai MF, Brophy RH, Sandell LJ. Osteoarthritis following meniscus and ligament injury: insights from translational studies and animal models. Curr Opin Rheumatol. 2019;31:70-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 25. | Han J, Wang C, Yang H, Luo J, Zhang X, Zhang XA. Novel Insights into the Links between N6-Methyladenosine and Regulated Cell Death in Musculoskeletal Diseases. Biomolecules. 2024;14:514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 26. | Kim KI, Kim MS, Kim JH. Intra-articular Injection of Autologous Adipose-Derived Stem Cells or Stromal Vascular Fractions: Are They Effective for Patients With Knee Osteoarthritis? A Systematic Review With Meta-analysis of Randomized Controlled Trials. Am J Sports Med. 2023;51:837-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (1)] |
| 27. | Noorduyn JCA, van de Graaf VA, Willigenburg NW, Scholten-Peeters GGM, Kret EJ, van Dijk RA, Buchbinder R, Hawker GA, Coppieters MW, Poolman RW; ESCAPE Research Group. Effect of Physical Therapy vs Arthroscopic Partial Meniscectomy in People With Degenerative Meniscal Tears: Five-Year Follow-up of the ESCAPE Randomized Clinical Trial. JAMA Netw Open. 2022;5:e2220394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 28. | van der Graaff SJA, Eijgenraam SM, Meuffels DE, van Es EM, Verhaar JAN, Hofstee DJ, Auw Yang KG, Noorduyn JCA, van Arkel ERA, van den Brand ICJB, Janssen RPA, Liu WY, Bierma-Zeinstra SMA, Reijman M. Arthroscopic partial meniscectomy versus physical therapy for traumatic meniscal tears in a young study population: a randomised controlled trial. Br J Sports Med. 2022;56:870-876. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 29. | Moran CJ, Busilacchi A, Lee CA, Athanasiou KA, Verdonk PC. Biological augmentation and tissue engineering approaches in meniscus surgery. Arthroscopy. 2015;31:944-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 30. | Wong C, Stoilova I, Gazeau F, Herbeuval JP, Fourniols T. Mesenchymal stromal cell derived extracellular vesicles as a therapeutic tool: immune regulation, MSC priming, and applications to SLE. Front Immunol. 2024;15:1355845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |