Copyright: ©Author(s) 2026.
World J Stem Cells. Apr 26, 2026; 18(4): 118404
Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.118404
Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.118404
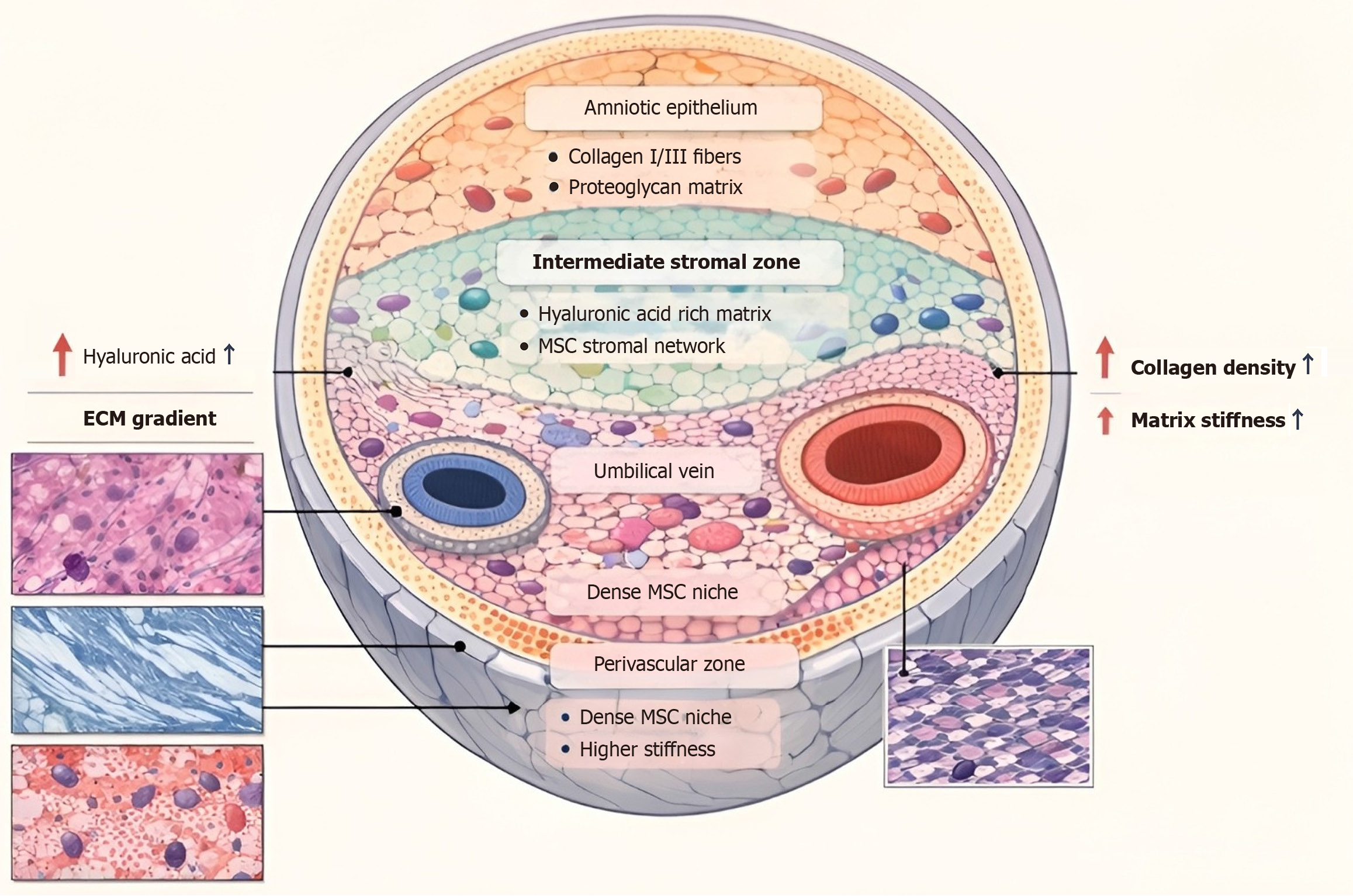
Figure 1 Cross-sectional view of the human umbilical cord.
Structural and cellular organization of Wharton’s jelly (WJ) within the human umbilical cord. The umbilical cord is composed of two arteries and one vein, and the whole is surrounded by WJ, with the latter covered by amniotic epithelium. The composition of WJ includes the subamniotic, intermediate stromal, and perivascular regions, each with distinct cell populations and biomechanical properties. The perivascular region is composed of more mesenchymal stem cells, resulting in the increased amount of collagen and the stiffness of the matrix, whereas the intermediate stromal region is composed mainly of a hyaluronic acid-based matrix. The histological images show the microstructural features of the matrix that contribute to the biomechanical properties of the WJ. ECM: Extracellular matrix; MSC: Mesenchymal stem cell.
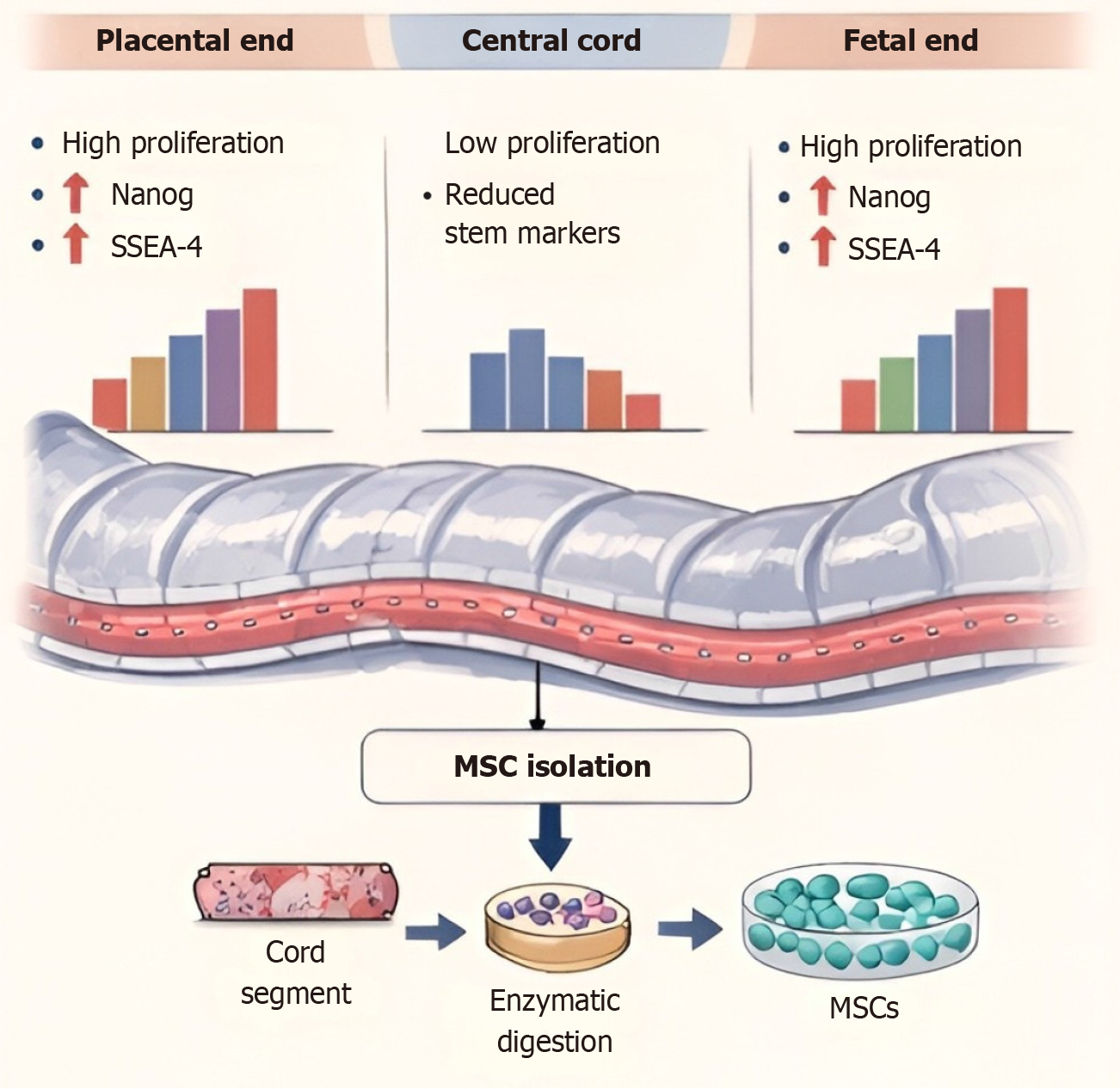
Figure 2 Regional heterogeneity in stemness features of Wharton’s jelly-derived mesenchymal stromal cells along the umbilical cord axis.
Mesenchymal stem cells (MSCs) obtained from different parts of the umbilical cord tissue are functionally and molecularly heterogeneous. MSCs obtained from the placental-fetal segment exhibit higher proliferative potential and higher expression of stemness-associated genes, such as Nanog and SSEA-4, compared to MSCs obtained from the central part of the umbilical cord, which exhibit lower proliferative activity and lower stemness-associated gene expression. The scheme also presents the general procedure for MSC isolation from umbilical cord tissue, including enzymatic digestion and expansion of isolated MSCs. MSC: Mesenchymal stem cell.
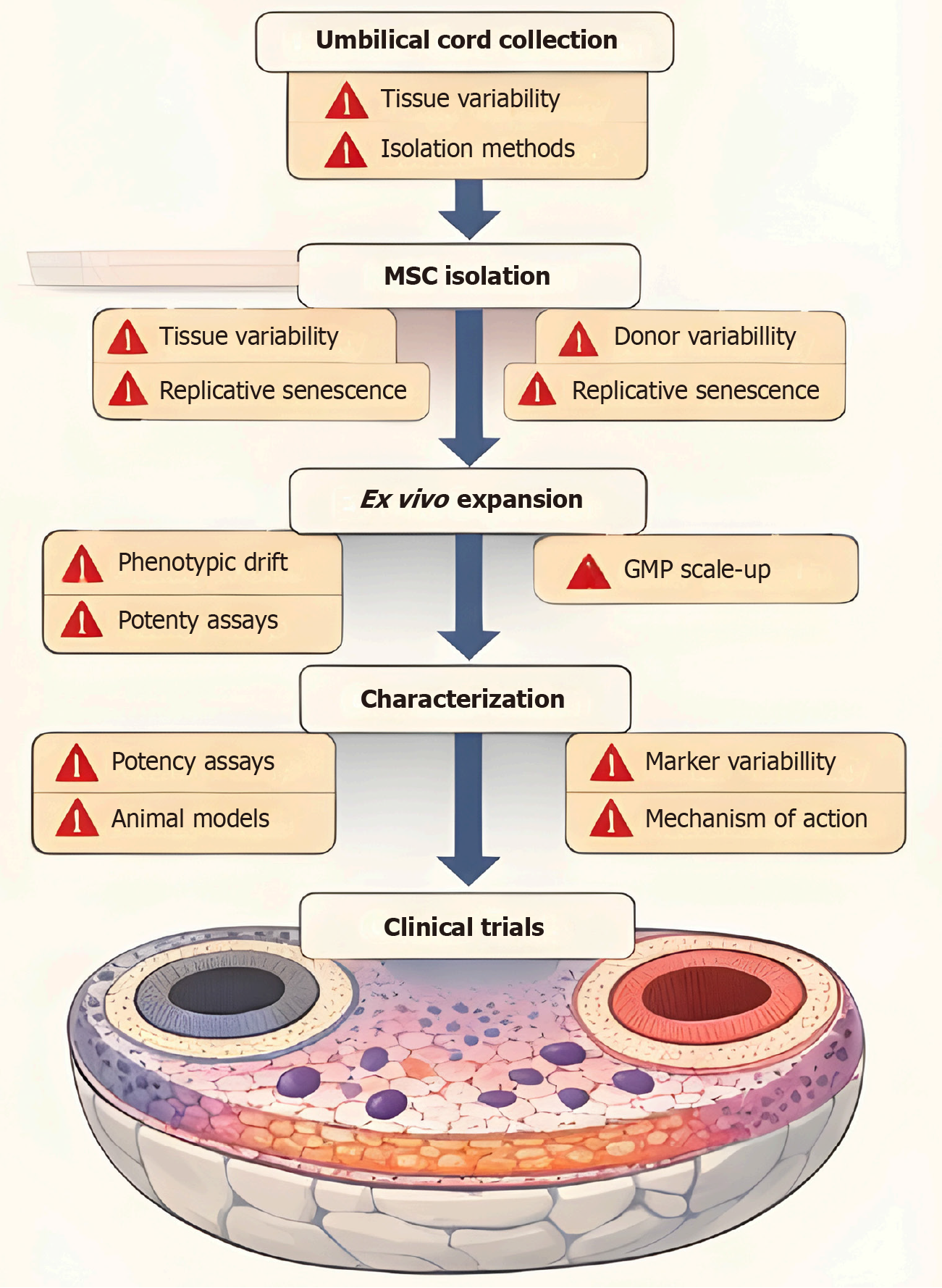
Figure 3 Translational pathway and bottlenecks in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy.
There are several steps in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy, including tissue procurement, cell isolation and expansion, cell characterization, pre-clinical validation, and clinical validation. However, there are several bottlenecks in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy, including donor variability, lack of standardization in cell isolation and potency, difficulties in large-scale manufacturing, and limited understanding of long-term safety and the mechanism of action. MSC: Mesenchymal stem cell.
- Citation: Habiba UE, Greene DL, Shamim S. Anatomy, function, biomechanics, and regenerative properties of Wharton’s jelly in the umbilical cord. World J Stem Cells 2026; 18(4): 118404
- URL: https://www.wjgnet.com/1948-0210/full/v18/i4/118404.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i4.118404