Copyright: ©Author(s) 2026.
World J Stem Cells. Mar 26, 2026; 18(3): 113900
Published online Mar 26, 2026. doi: 10.4252/wjsc.v18.i3.113900
Published online Mar 26, 2026. doi: 10.4252/wjsc.v18.i3.113900
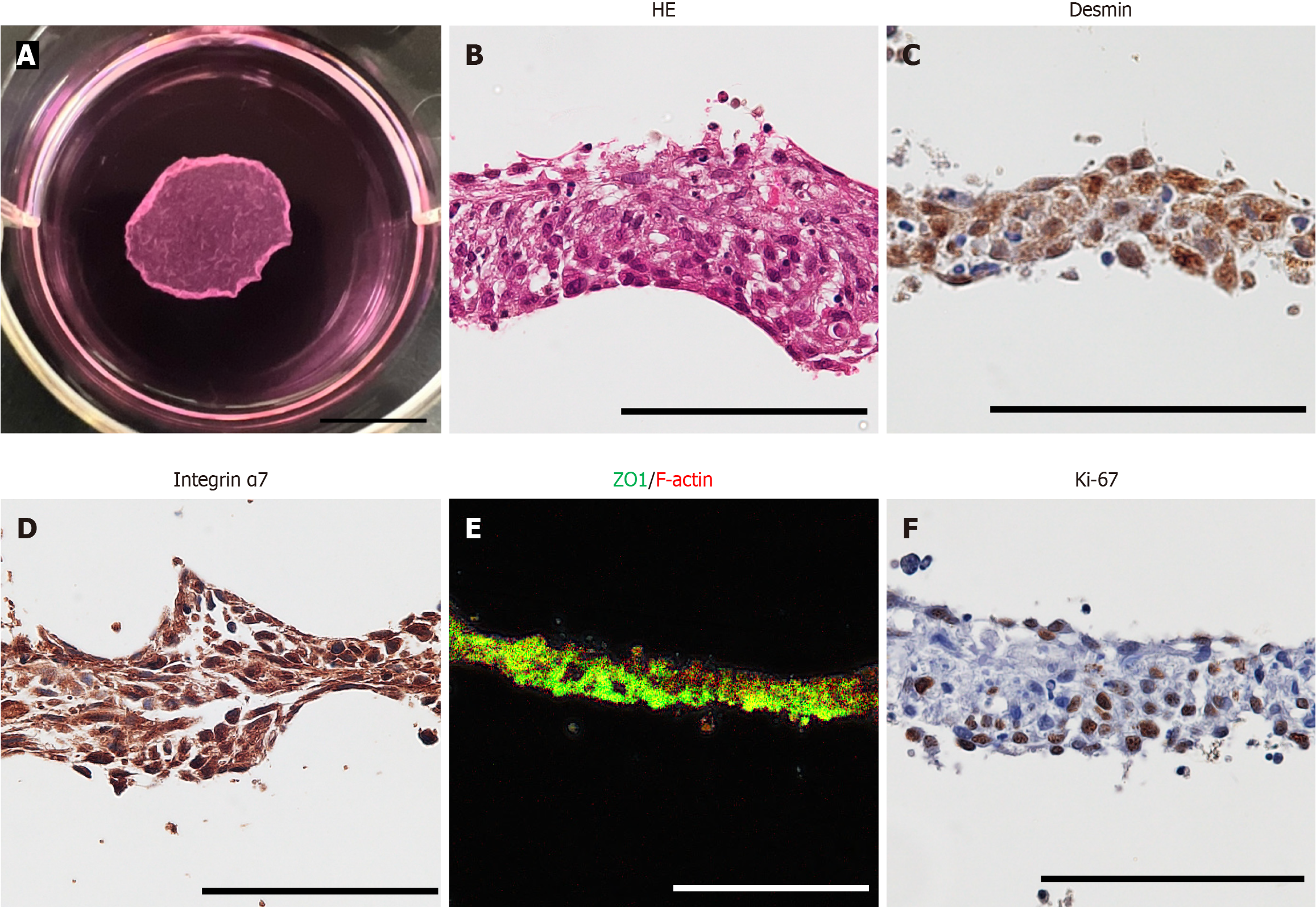
Figure 1 Characteristics of myoblast cell sheets.
A: Fabrication of myoblast cell sheets. The diameter of the myoblast cell sheet was approximately 10 mm (scale bar: 10 mm); B: Hematoxylin and eosin staining of the myoblast cell sheets (× 400, scale bar: 100 μm); C-F: Immunostaining of the myoblast cell sheets for desmin (C), Ki-67 (F), integrin α7 (D) (× 400, scale bar: 100 μm) and zonula occludens-1 (E) in myoblast cell sheets (× 200, scale bar: 100 μm). HE: Hematoxylin and eosin; ZO1: Zonula occludens-1.
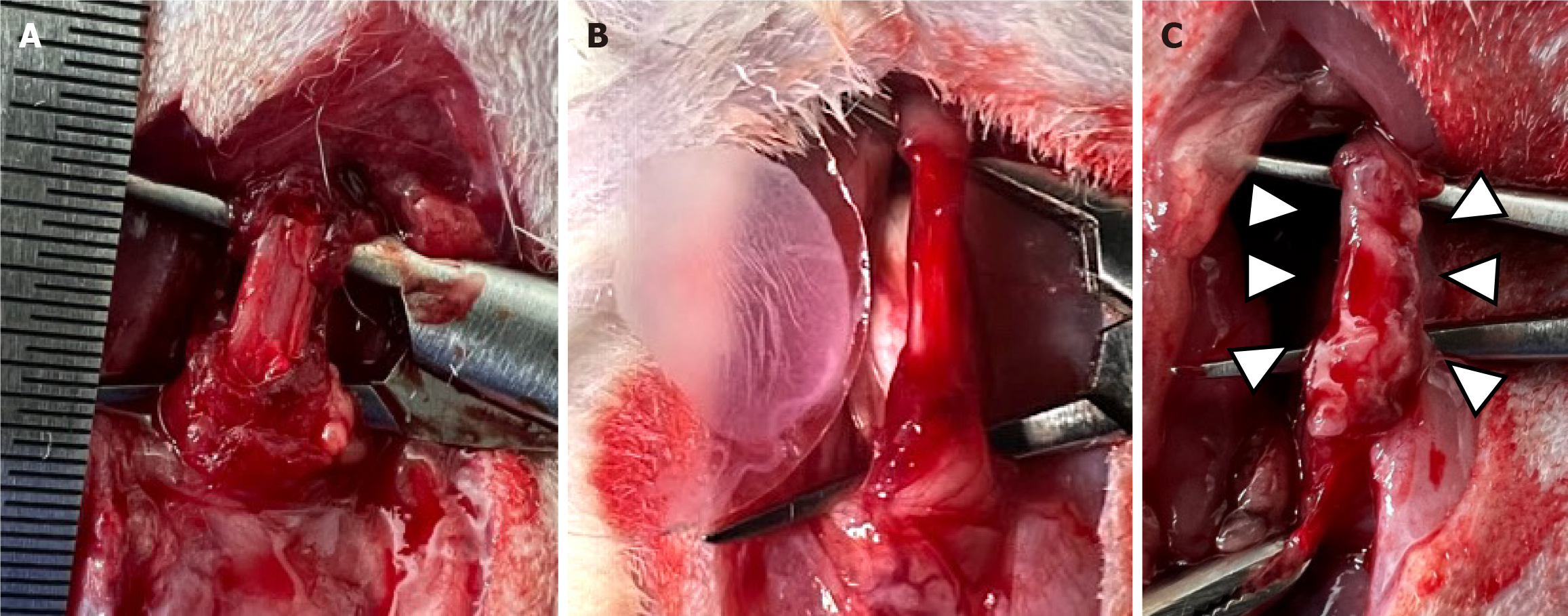
Figure 2 Animal experiments.
A: Resection of 67% of the abdominal esophageal muscularis propria. The major axis of the resected tissue measured 1 cm; B: We transplanted a myoblast cell sheet onto the resected esophageal muscularis propria using a circular glass sheet; C: Gross findings after myoblast cell sheet transplantation (△).
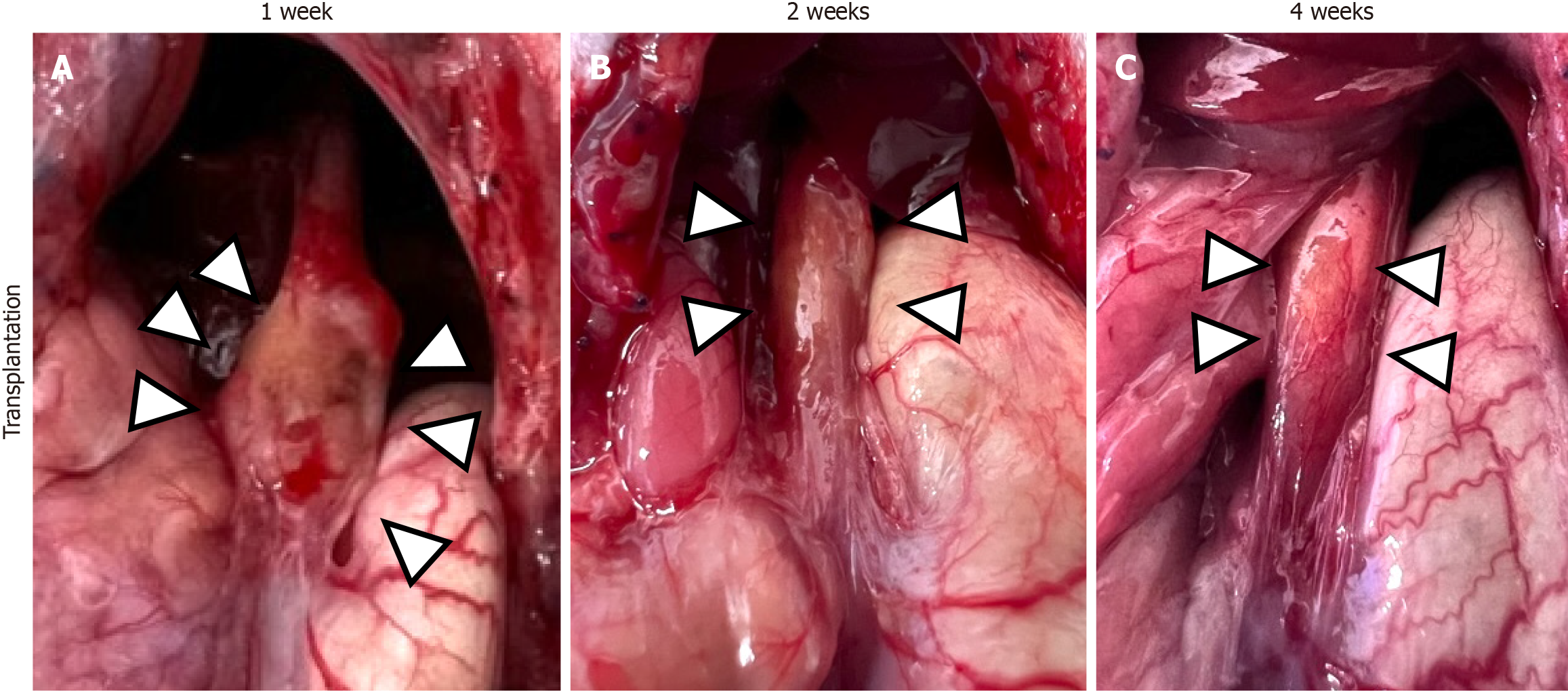
Figure 3 Macroscopic findings of grafted tissue in the transplant group.
A: At 1 week postoperatively, nodular thickening of the esophageal wall was observed at the site of myoblast cell sheet transplantation (△); B: At 2 weeks postoperatively, the nodules observed at week 1 had disappeared, and the transplantation site showed a smooth esophageal wall surface (△); C: At 4 weeks postoperatively, the macroscopic findings at the transplantation site remained consistent with those observed at week 2 (△).
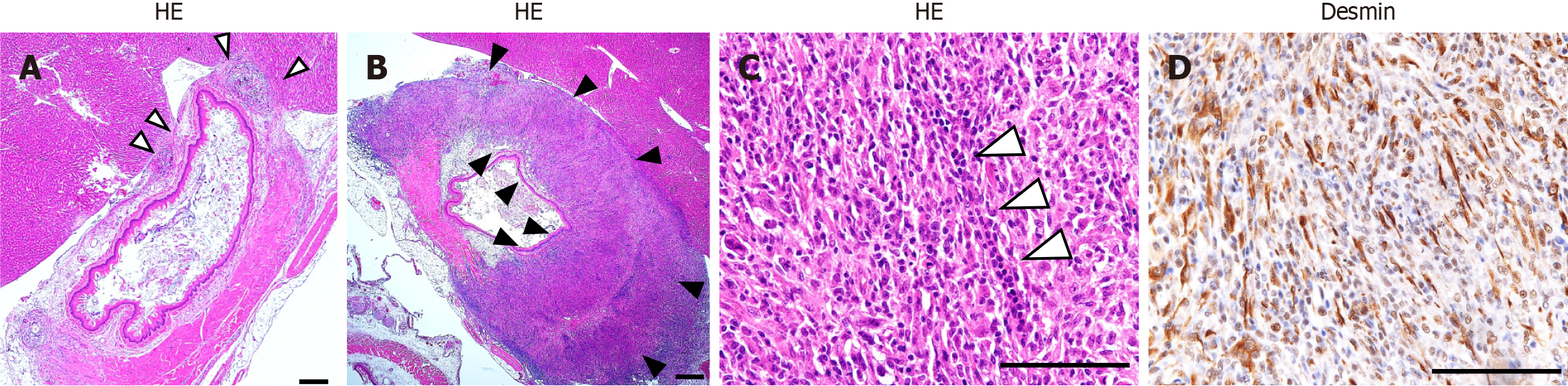
Figure 4 Histological evaluation at 1 week postoperatively.
A: Histological examination of the control group [hematoxylin and eosin (HE) staining, × 40] revealed no tissue replacement at the site of the esophageal muscularis propria resection. Fibrous adhesion was noted between the resected area and the liver (△); B: HE staining (× 40) of the transplantation group showed the myoblast cell sheet proliferating and replacing the resected esophageal muscularis propria (▲); C: HE staining (× 400, scale bar: 100 μm) of the transplantation group showed the cells in the myoblast sheet had partially formed a multinucleated myotube (△); D: Desmin staining (× 400, scale bar: 100 μm) of the transplantation group showed that the myoblast cell sheet contained fibrous desmin-positive cells, indicating proliferation of the myoblast cell sheet.
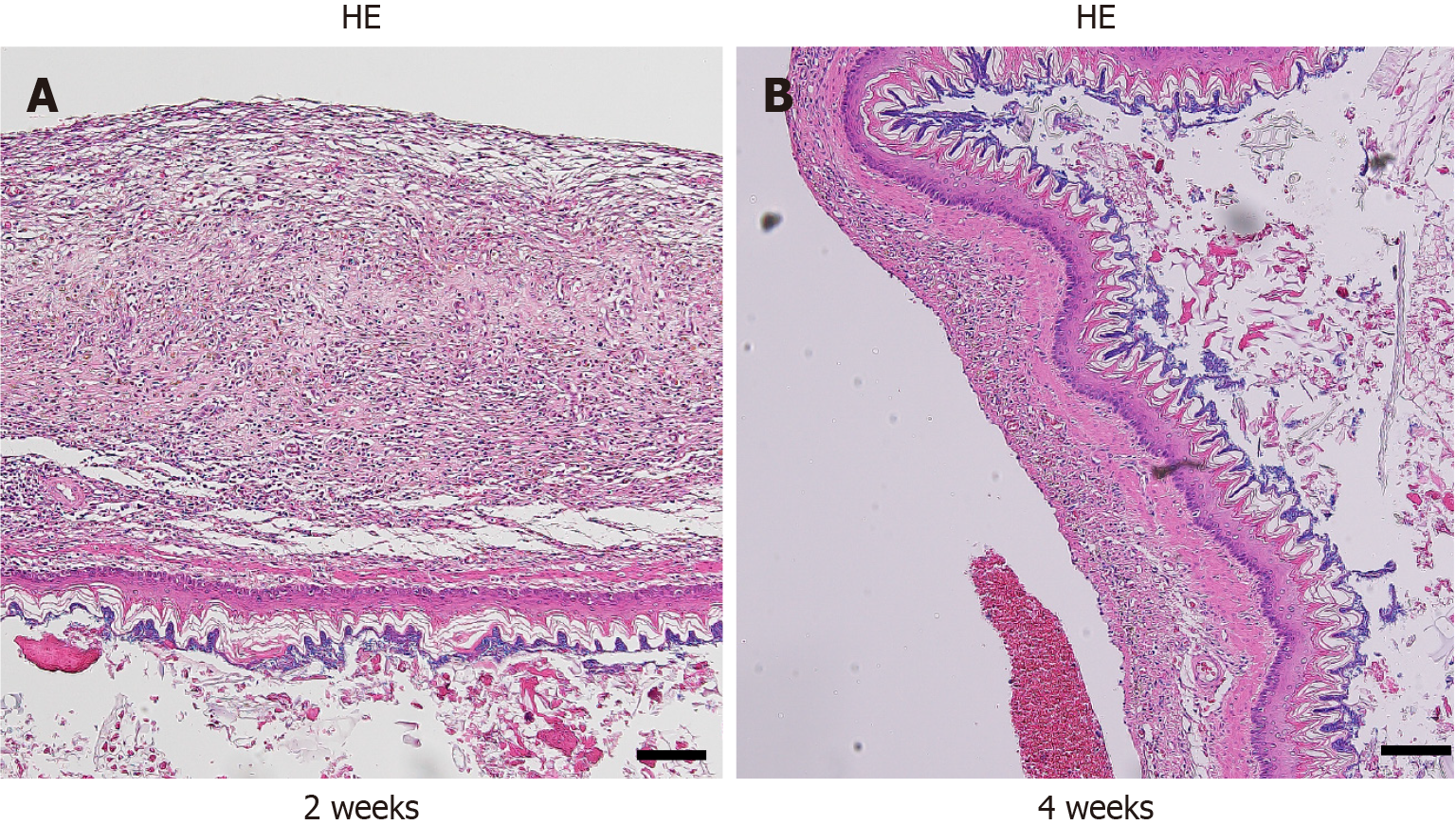
Figure 5 Histological evaluation of transplanted myoblast cell sheets at each time point.
A and B: Hematoxylin and eosin (HE) staining (× 100, scale bar: 100 μm) of the transplantation group at 2 weeks postoperatively revealed a decrease in the thickness of the cell sheet compared with the 1-week time point (A), and 4 weeks postoperatively revealed a further decrease in the thickness of the myoblast cell sheets (B).
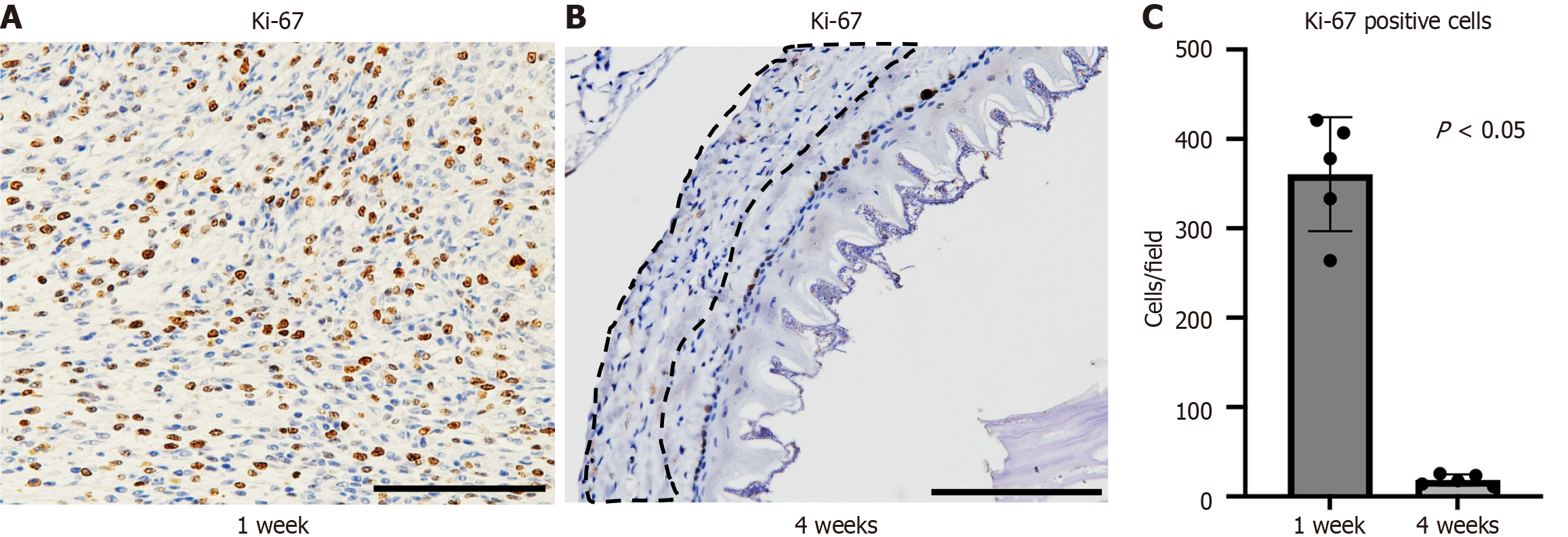
Figure 6 Ki-67-positive cells in the transplanted myoblast cell sheets at each time point.
A and B: Ki-67 immunostaining (× 400, scale bar: 100 μm) of the transplantation group at 1 week postoperatively showed that the layer formed by the myoblast cell sheet contained numerous Ki-67-positive cells (A), and 4 weeks postoperatively showed very few Ki-67 positive cells (proliferating cells) in the transplanted myoblast sheet layer (enclosed by a dotted line) (B); C: The number of Ki-67-positive cells per high-power field (× 400) detected via Ki-67 immunostaining showed that Ki-67-positive cells in the myoblast sheet-transplanted area at 4 weeks post-transplantation were significantly reduced compared to that at 1 week post-transplantation.
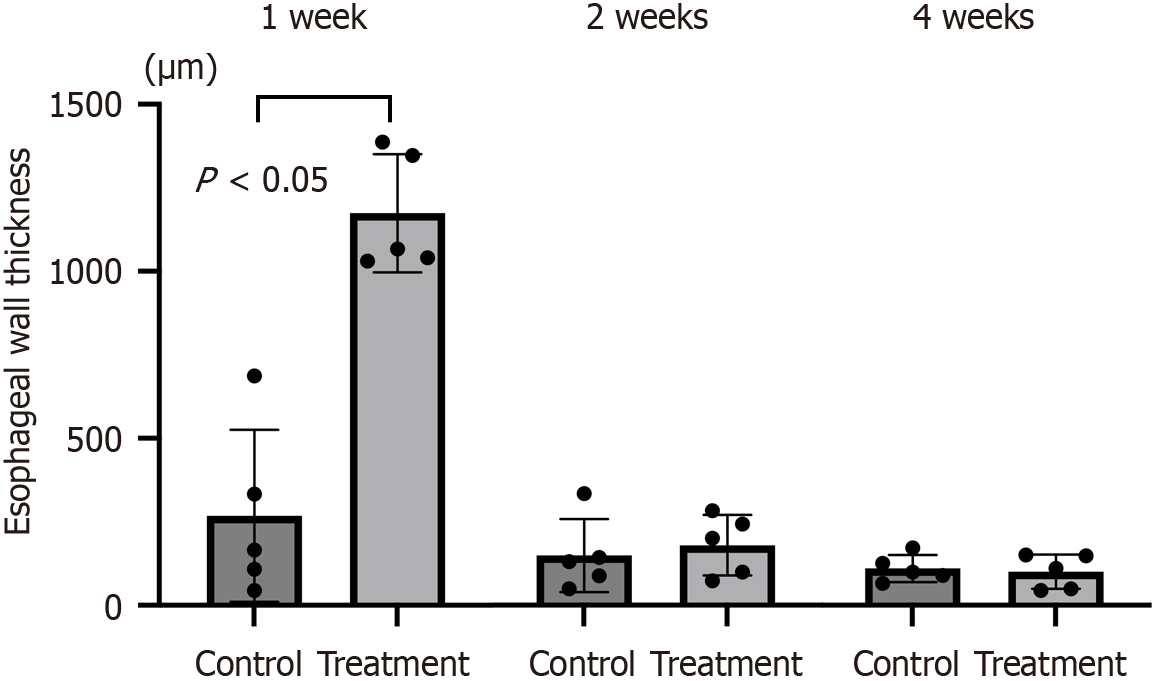
Figure 7 Esophageal wall thickness at the site of muscularis propria resection at each time point.
In the control group, the esophageal wall thickness remained constant at each time point. At 1 week postoperatively, the esophageal wall was significantly thicker in the transplantation group than in the control group (P < 0.05). The thickness of the esophageal wall decreased to a level comparable to that of the control group at 2 weeks postoperatively.
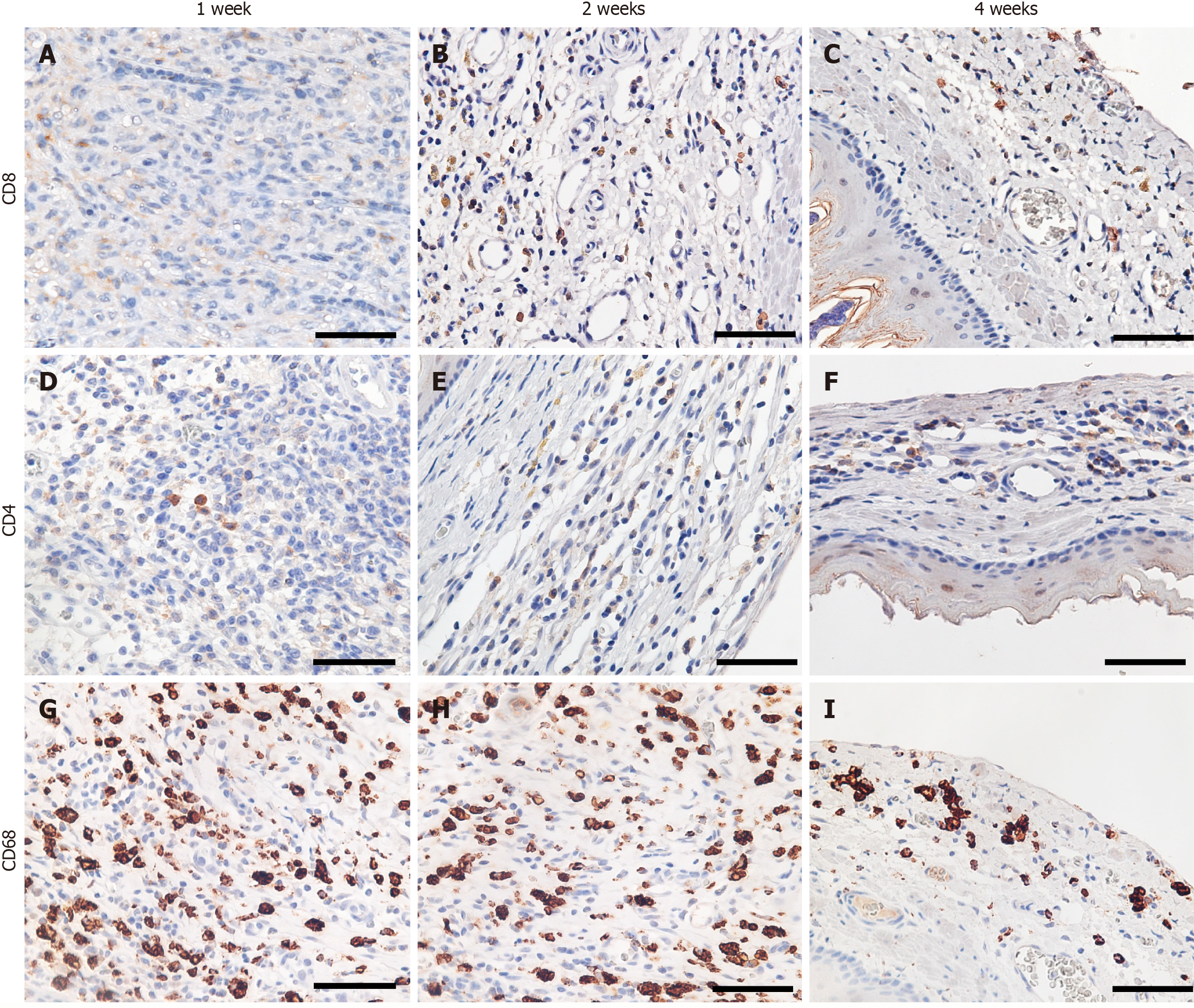
Figure 8 Immunostaining of immune cells in transplanted myoblast cell sheets at each time point.
A-C: CD8 immunostaining in the transplantation group showed infiltration of CD8+ lymphocytes in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm); D-F: CD4 immunostaining in the transplantation group showed infiltration of CD4+ lymphocytes in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm); G-I: CD68 immunostaining in the transplantation group showed infiltration of CD68+ macrophages in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm).
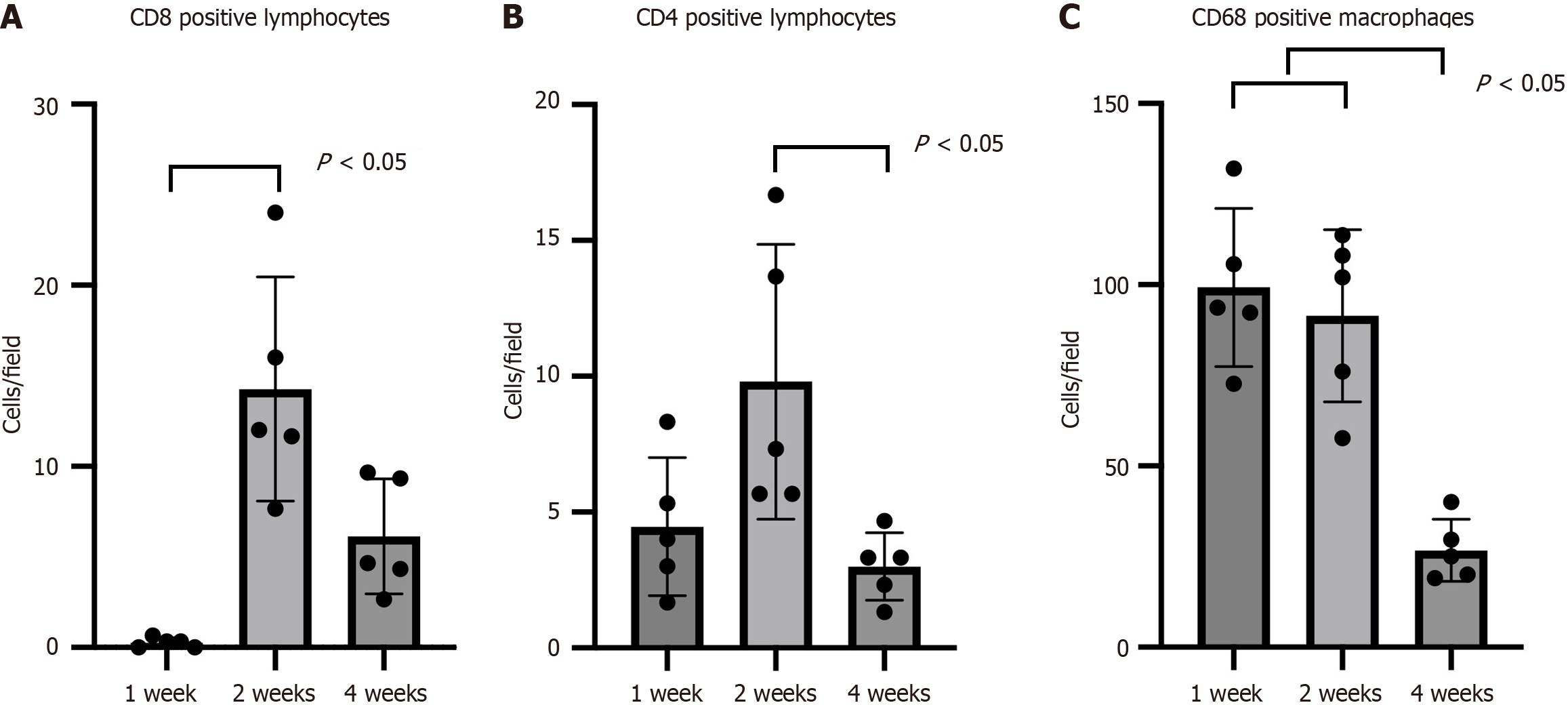
Figure 9 Quantitative analysis of immune cells.
A: The number of CD8+ cells per high-power field (× 400) detected via CD8 immunostaining showed mild infiltration of CD8+ cells at 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation; B: The number of CD4+ cells per high-power field (× 400) detected via CD4 immunostaining showed mild infiltration of CD4+ cells at 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation; C: The number of CD68+ cells per high-power field (× 400) detected via CD68 immunostaining showed mild infiltration of CD68+ cells at 1 week and 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation.
- Citation: Enjoji T, Kobayashi S, Miyamoto D, Ogawa S, Tetsuo H, Soyama A, Adachi T, Eguchi S, Kanetaka K. Allogeneic myoblast cell sheet transplantation for esophageal wall reinforcement. World J Stem Cells 2026; 18(3): 113900
- URL: https://www.wjgnet.com/1948-0210/full/v18/i3/113900.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i3.113900