©The Author(s) 2021.
World J Stem Cells. Nov 26, 2021; 13(11): 1667-1695
Published online Nov 26, 2021. doi: 10.4252/wjsc.v13.i11.1667
Published online Nov 26, 2021. doi: 10.4252/wjsc.v13.i11.1667
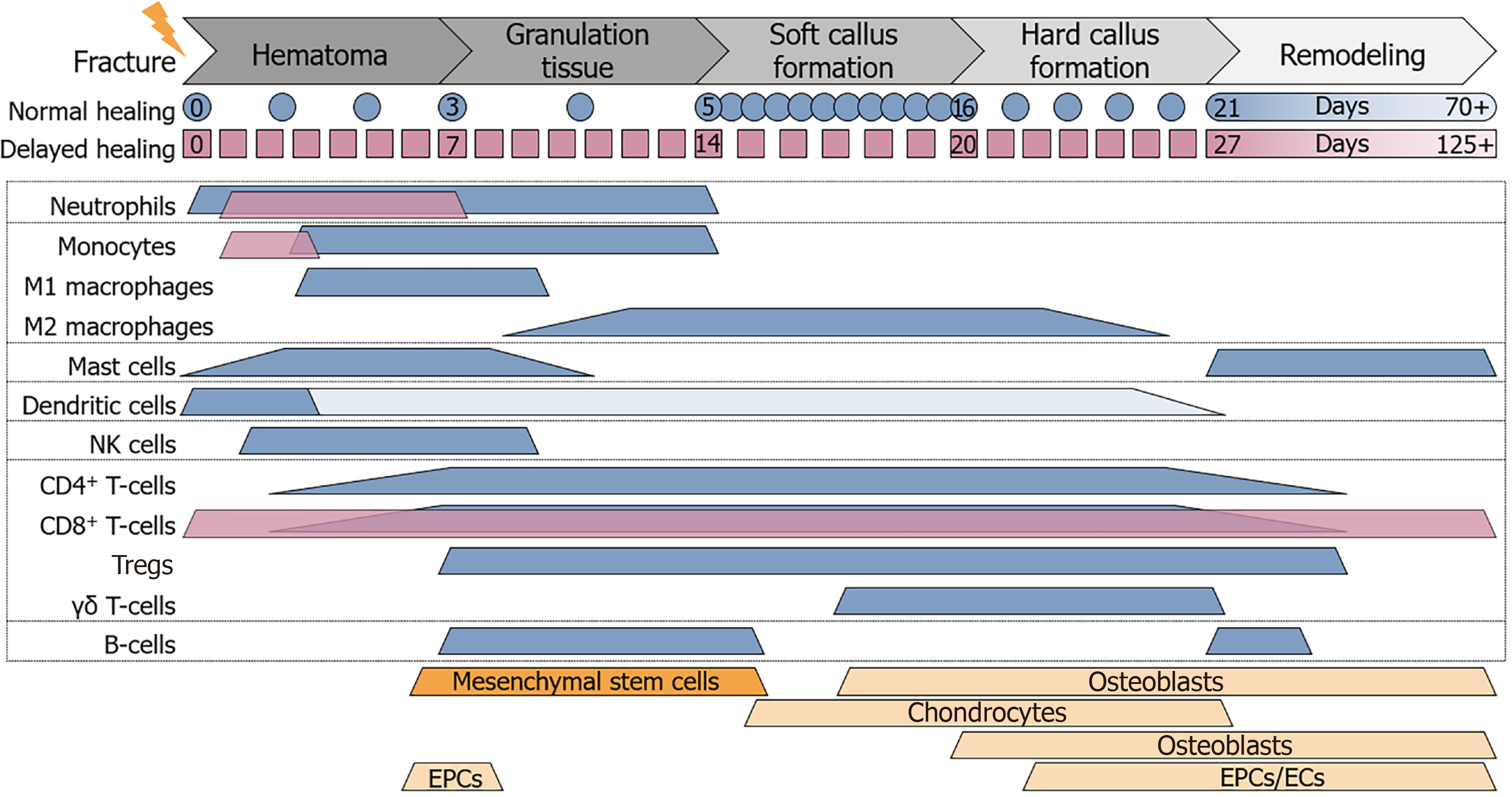
Figure 1 Cell composition at the site of fracture.
During the different phases of fracture healing the cell composition at the site of fracture changes. Expected timeline of normal (blue) and delayed (magenta) fracture healing is depicted below the phases of fracture healing. Colored (blue and magenta) beams representing the timeframe where immune cells are expected to be active at the site of fracture, based on different in vivo studies. Orange beams representing the timeframe where mesenchymal stem cells, chondrocytes, osteoblasts, osteoclasts, endothelial progenitor cells, and endothelial cells are involved in the fracture healing process. CD: Cluster of differentiation; EPCs: Endothelial progenitor cells; ECs: Endothelial cells; NK: Natural killer.
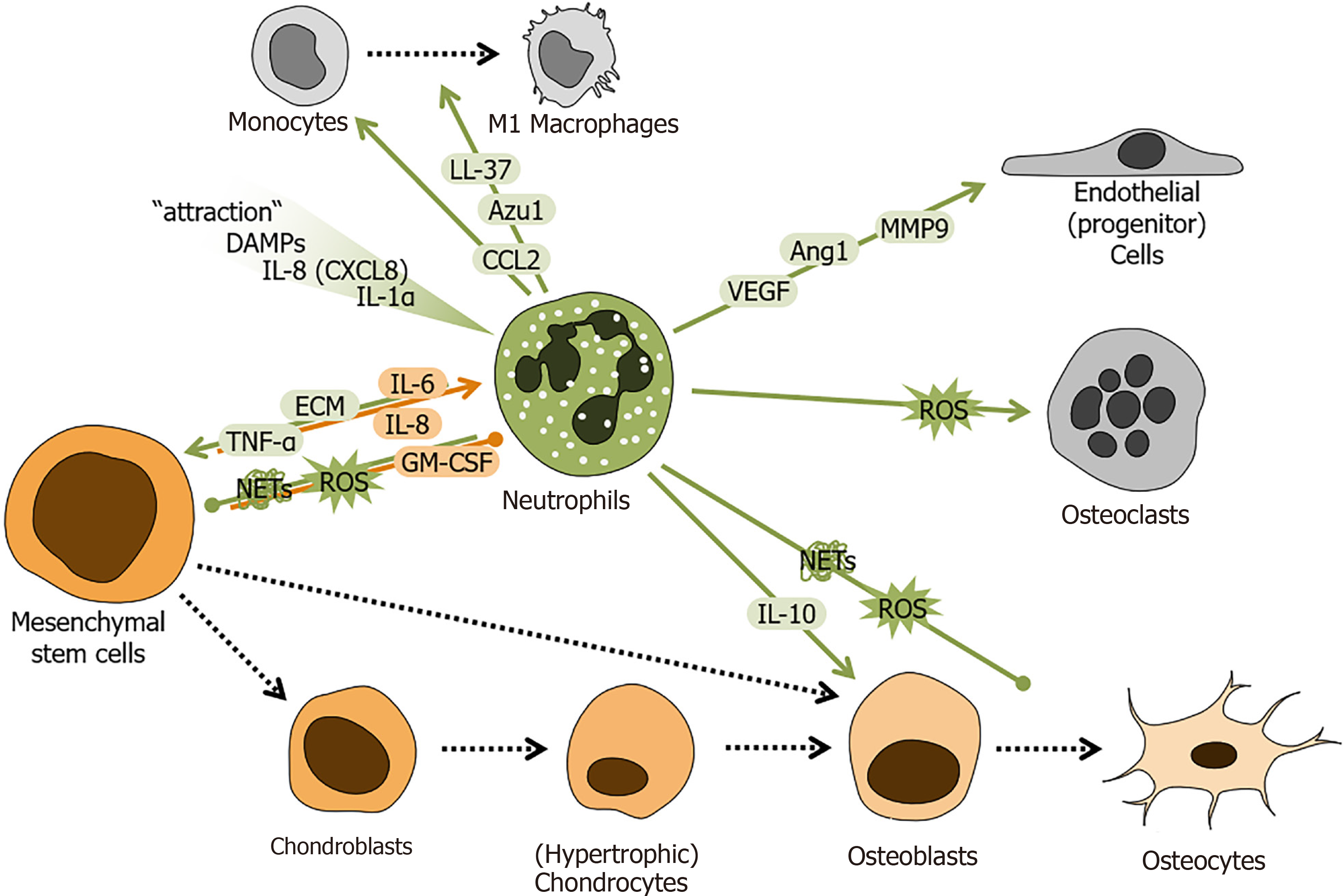
Figure 2 Schematic overview of the regulatory role of neutrophils during fracture healing.
Factors secreted/released from neutrophils and mesenchymal stem cells (MSCs) regulate cellular responses during fracture healing. Neutrophil-derived factors are marked in green. MSC-derived factors are marked in orange. Local increase in danger associated molecular patterns, interleukins 8 (IL-8 or CXCL8) and 1 alpha (IL-1α) attract neutrophils to the fracture site. There neutrophils secrete /release factors e.g. tumor necrosis factor alpha, IL-10, C-C-motif chemokine ligand 2, azurocidin 1, cathelicidin (LL-37), vascular endothelial growth factor, angiopoietin 1, matrix metalloproteinase 9, reactive oxygen species, neutrophil extracellular traps, and extracellular matrix components to interact with other cells at the site of fracture. MSCs secrete/release factors e.g., granulocyte-macrophage colony-stimulating factor, IL-6 and 8 to affect neutrophils. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. DAMPs: Danger associated molecular patterns; IL: Interleukin; TNF-α: Tumor necrosis factor alpha; CCL2: C-C-motif chemokine ligand 2; CXCL: C-X-C motif chemokine ligand; Azu1: Azurocidin 1; VEGF: Vascular endothelial growth factor; Ang1: Angiopoietin 1; MMP9: Matrix metalloproteinase 9; ROS: Reactive oxygen species; NETs: Neutrophil extracellular traps; ECM: Extracellular matrix; GM-CSF: Granulocyte-macrophage colony-stimulating factor.
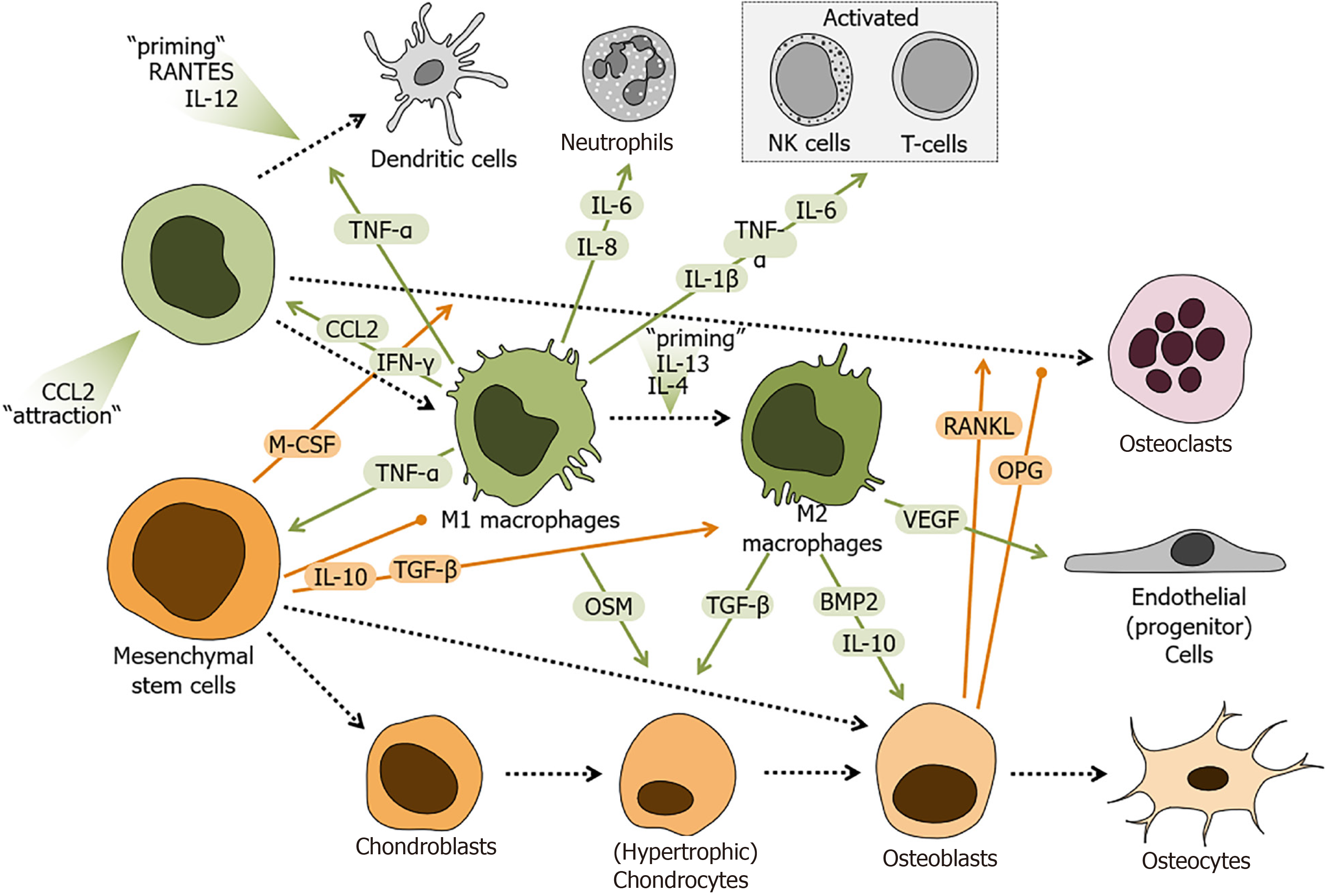
Figure 3 Schematic overview of the regulatory role of monocytes and macrophages during fracture healing.
Factors secreted/released from mesenchymal stem cells (MSCs), monocytes, and monocyte-derived cells regulate cellular responses during fracture healing. Factors derived from monocytes and macrophages are marked in green. MSC-derived factors are marked in orange. Local increase in C-C-motif chemokine ligand 2 (CCL2) attract monocytes to the site of fracture, which in the presence of C-C-motif chemokine ligand 5 (RANTES or CCL5) and interleukin 12 (IL-12) differentiate into dendritic cells, in an inflammatory environment are primed towards pro-inflammatory M1 macrophages, or in the presence of IL-4 and IL-13 transform into anti-inflammatory M2 macrophages. Depending on their differentiation state, monocyte-derived cells secrete/release factors e.g., tumor necrosis factor alpha, interferon gamma, CCL2, vascular endothelial growth factor, bone morphogenetic protein 2, transforming growth factor beta (TGF-β), oncostatin M (OSM), IL-1β, IL-6, IL-8, and IL-10 to interact with other cells in the site of fracture. MSCs and MSC-derived cells secrete /release factors e.g., macrophage colony-stimulating factor, IL-10, TGF-β, receptor activator of nuclear factor kappa-Β ligand, or its antagonist osteoprotegerin to affect the monocyte derived-cells. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. CCL2: C-C-motif chemokine ligand 2; IL: Interleukin; TNF-α: Tumor necrosis factor alpha; IFN-γ: Interferon gamma; VEGF: Vascular endothelial growth factor; BMP2: Bone morphogenetic protein 2; TGF-β: Transforming growth factor beta; OSM: Oncostatin M; M-CSF: Macrophage colony-stimulating factor; RANKL: Receptor activator of nuclear factor kappa-Β ligand; OPG: Osteoprotegerin.
Figure 4 Schematic overview of the regulatory role of mast cells during fracture healing.
Cellular responses during fracture healing are orchestrated by factors secreted/released from mast cells and mesenchymal stem cells (MSCs). Factors derived from mast cells are marked in green. MSC-derived factors are marked in orange. Mast cells communicate with other cells during fracture healing by producing and releasing a large variety of factors e.g., tumor necrosis factor alpha, interleukin 1 beta (IL-1β), IL-6, and IL-8, keratinocytes-derived chemokine (KC or CXCL1), macrophage inflammatory protein 2 (MIP-2 or CXCL2), fibroblast growth factors, vascular endothelial growth factor (VEGF), transforming growth factor beta (TGF-β), receptor activator of nuclear factor kappa-Β ligand, or histamines. MSCs in turn affect mast cells by secreted factors e.g., IL-6, TGF-β, or VEGF. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. TNF-α: Tumor necrosis factor alpha; IL: Interleukin; FGF: Fibroblast growth factor; TGF-β: Transforming growth factor beta; VEGF: Vascular endothelial growth factor; RANKL: Receptor activator of nuclear factor kappa-Β ligand.
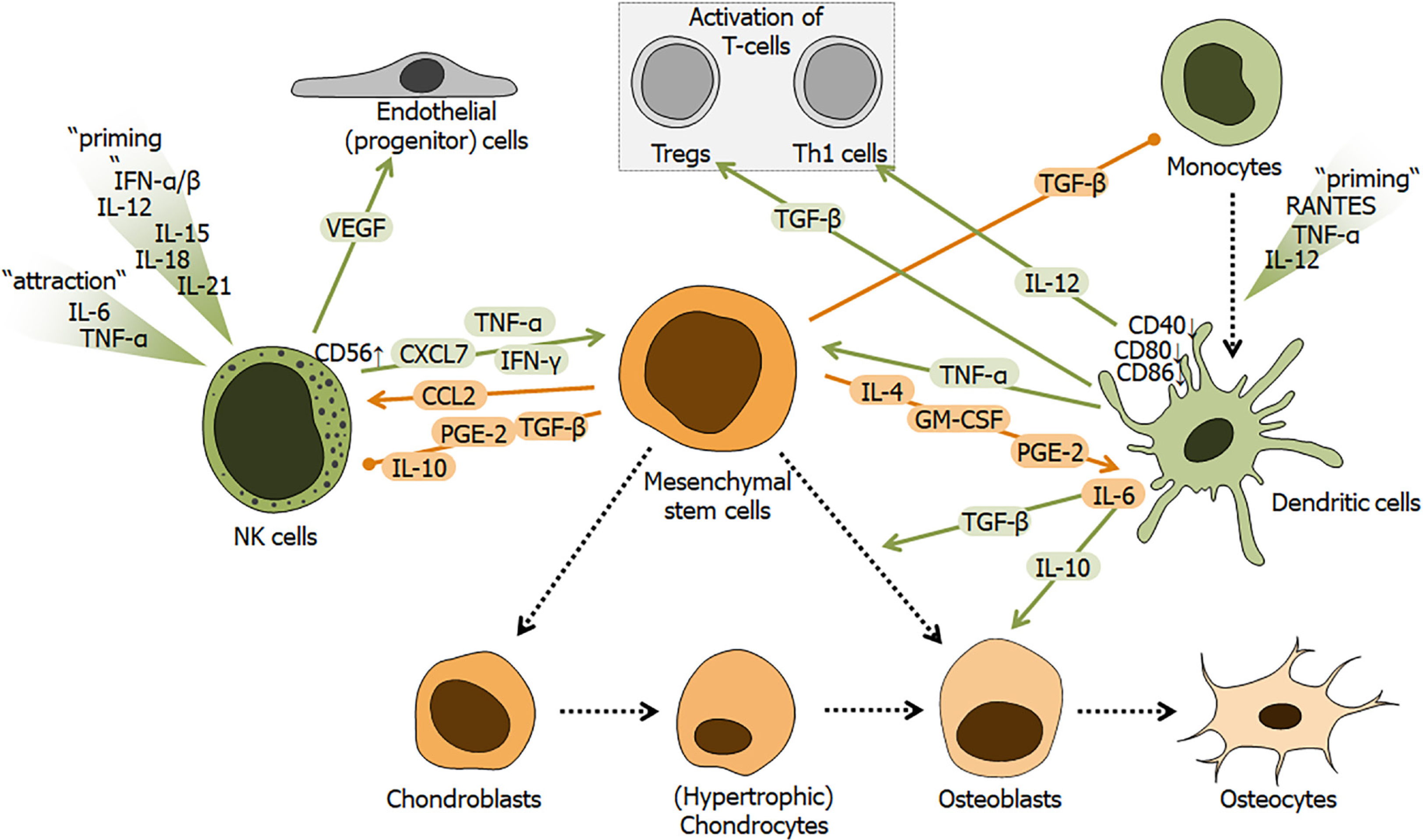
Figure 5 Schematic overview of the regulatory role of dendritic cells and natural killer cells during fracture healing.
Fracture healing is affected by factors secreted / released from mesenchymal stem cells (MSCs), natural killer cells (NK cells) or monocyte-derived dendritic cells. Factors derived from NK cells and dendritic cells are marked in green. MSC-derived factors are marked in orange. NK cells are attracted and primed by local increase in factors, e.g., tumor necrosis factor alpha (TNF-α), interferons alpha and beta (IFN-β), and interleukin 6 (IL-6), IL-12, IL-15, IL-18, or IL-21. NK cells, depending on their activation status, secrete factors, e.g., TNF-α, interferon gamma, vascular endothelial growth factor, or pro-platelet basic protein (PPBP or CXCL7). Dendritic cells differentiate from monocytes in the presence of TNF-α, IL-12, and C-C-motif chemokine ligand 5 (RANTES or CCL5). Dendritic cells then secrete factors, e.g., TNF-α, transforming growth factor beta (TGF-β), and IL-10 and IL-12. Both cell types are strongly affected by factors secreted/released from MSCs, e.g., C-C-motif chemokine ligand 2 (CCL2), prostaglandin E2 (PGE2), granulocyte-macrophage colony-stimulating factor, TGF-β, or IL-4, IL-6 and IL-10. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. CD: Cluster of differentiation; NK: Natural killer; TNF-α: Tumor necrosis factor alpha; IL: Interleukin; IFN-α: Interferon alpha; IFN-γ: Interferon gamma; VEGF: Vascular endothelial growth factor; CCL: C-C-motif chemokine ligand; TGF-β: Transforming growth factor beta; PGE2: Prostaglandin E2; GM-CSF: Granulocyte-macrophage colony-stimulating factor.
Figure 6 Schematic overview of the regulatory role of T-cells during fracture healing.
Fracture healing is affected by interactions between mesenchymal stem cells (MSCs) and T-cells. However, the effect strongly depends on the activation or differentiation status of the T-cells, which is in CD4+ T-cells strongly dependent on activation of signal transducer and activator of transcription (STAT) signaling. Th1 cells are primed by interferon gamma (IFN-γ) and interleukin 12 (IL-12), which activate STAT-1 and STAT4 signaling in these cells. Th1 cells then secrete factors e.g., tumor necrosis factor alpha (TNF-α), IFN-γ, or IL-2. Th17 cells are primed by transforming growth factor beta (TGF-β), IL-1β and IL-6, which activate STAT3 signaling in the cells. Th17 cells then secrete factors e.g., IL-17 and receptor activator of nuclear factor kappa-Β ligand (RANKL). Th2 cells, characterized by activated STAT-6 and GATA3 (GATA Binding Protein 3) signaling, are primed by IL-2 and IL-4, and secrete factors e.g., IL-4, IL-10 and IL-13. Regulatory T cells (Tregs) are attracted and primed by factors, e.g., C-C-motif chemokine ligand 22 (CCL22), TGF-β, and IL-2, which activate forkhead box P3, STAT-3 and STAT-5 signaling in these cells. Tregs then secrete factors e.g., IL-4, IL-10, and TGF-β, to regulate osteoblast and osteoclast function, but also activation of T-cells. The same factors (IL-4, IL-10, and TGF-β) are also released by γδ T-cells. CD8+ cytotoxic T-cells enhance the pro-inflammatory reaction by releasing factors, e.g., TNF-α and IFN-γ. The different T-cell subsets, are influenced by MSCs and osteoblasts, which secrete / release factors, e.g. TGF-β, IL-4, prostaglandin E2, heme oxygenase 1 (HO-1), RANKL, or delta like ligand 4. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. CD: Cluster of differentiation; NK: Natural killer; TNF-α: Tumor necrosis factor alpha; IL: Interleukin; IFN: Interferon; CCL: C-C-motif chemokine ligand; TGF-β: Transforming growth factor beta; PGE2: Prostaglandin E2; RANKL: Receptor activator of nuclear factor kappa-Β ligand; STAT: Signal transducer and activator of transcription; Tregs: Regulatory T cells; Foxp3: Forkhead box P3; DLL4: Delta like ligand 4.
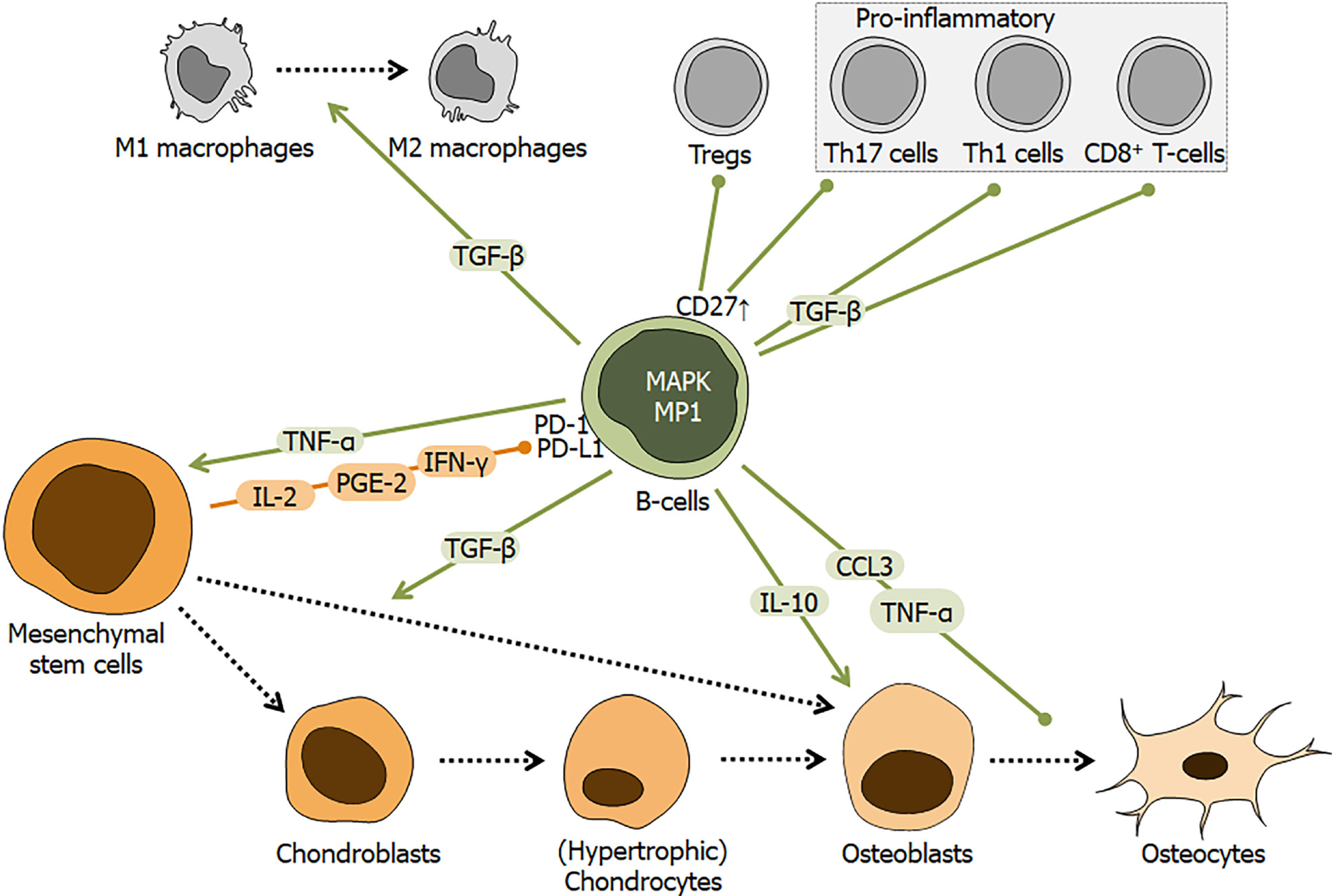
Figure 7 Schematic overview of the regulatory role of B-cells during fracture healing.
Fracture healing is affected by interactions between mesenchymal stem cells (MSCs) and B-cells. MSCs may affect B-cells by factors, e.g., interleukin 2 (IL-2), prostaglandin E2, and interferon gamma (IFN-γ), which interact with the programmed cell death 1 receptor and its ligand. Resulting in activation of mitogen-activated protein kinase and its downstream target MEK partner 1, induces B-cells to secrete/release factors, e.g., tumor necrosis factor alpha, transforming growth factor beta, IL-10, and C-C-motif chemokine ligand 3. Colored arrows depict stimulation and blunt end lines inhibition. Dashed black arrows indicate differentiation processes. CD: Cluster of differentiation; TNF-α: Tumor necrosis factor alpha; IL: Interleukin; IFN: Interferon; CCL: C-C-motif chemokine ligand; TGF-β: Transforming growth factor beta; PGE2: Prostaglandin E2; Tregs: Regulatory T cells; MAPK: Mitogen-activated protein kinase; PD-1: Programmed cell death 1; PD-L1: Programmed cell death ligand 1; MP1: MEK partner 1.
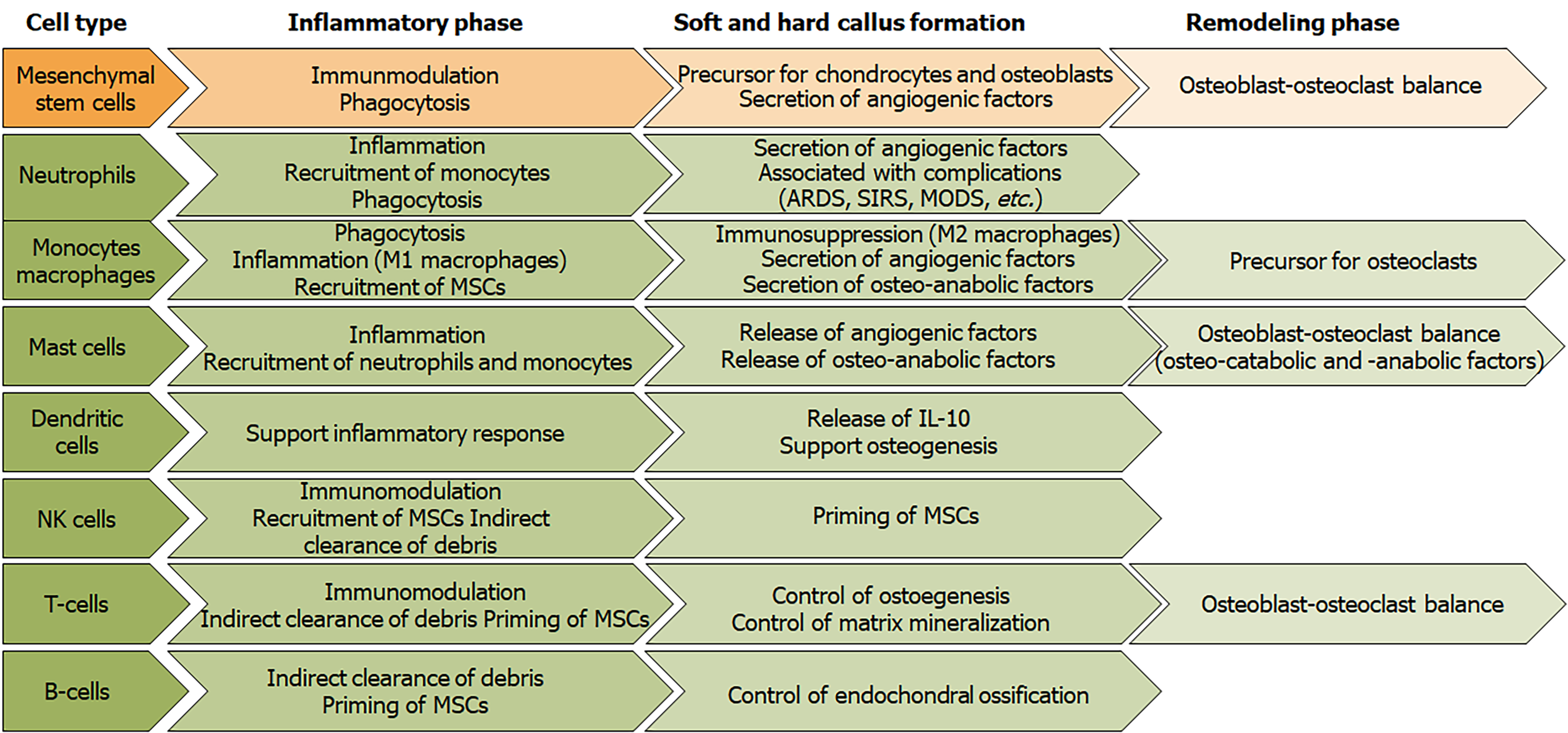
Figure 8 Overview of the roles of immune cells and mesenchymal stem cells during the different phases of fracture healing.
NK cells: Natural killer cells; MSCs: Mesenchymal stem cells; ARDS: Acute respiratory distress syndrome; SIRS: Systemic inflammatory response syndrome; MODS: Multi-organ dysfunction syndrome; IL-10: Interleukin 10.
- Citation: Ehnert S, Relja B, Schmidt-Bleek K, Fischer V, Ignatius A, Linnemann C, Rinderknecht H, Huber-Lang M, Kalbitz M, Histing T, Nussler AK. Effects of immune cells on mesenchymal stem cells during fracture healing. World J Stem Cells 2021; 13(11): 1667-1695
- URL: https://www.wjgnet.com/1948-0210/full/v13/i11/1667.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v13.i11.1667