修回日期: 2026-02-09
接受日期: 2026-02-13
在线出版日期: 2026-02-28
肝细胞癌(hepatocellular carcinoma, HCC)的全程化管理有赖于生物标志物的辅助. 血清甲胎蛋白(alpha-fetoprotein, AFP)是最经典的HCC标志物, 但临床上约30%的患者呈AFP阴性, 无法满足临床需求. 异常凝血酶原(des-gamma-carboxy prothrombin, DCP)的发现及临床应用, 表明其在肝癌早筛、诊断、治疗及预后判断中不可替代实用价值. 此外, 整合DCP、AFP等指标的预测模型, 在临床实践中也具有重要意义. 本文系统梳理以DCP为核心的生物标志物在HCC筛查诊断、预后判断、疗效评价及复发监测等方面的应用现状, 为临床医生深入了解DCP的应用价值提供依据.
核心提要: 异常凝血酶原(des-gamma-carboxy prothrombin, DCP)在肝细胞癌全程管理中展现出核心价值. 它不仅弥补了肝癌患者甲胎蛋白阴性的诊断缺口, 更因其与肿瘤侵袭性生物学的高度关联, 成为预测微血管侵犯及术后复发的关键标志. 从辅助肝移植患者筛选, 到动态评估局部与系统治疗疗效, DCP及其参与的整合预测模型, 为肝癌的风险分层与个体化精准治疗实践提供了循证支持.
引文著录: 刘子菡, 王文俊, 党双锁. 异常凝血酶原在肝细胞癌全程管理中的价值. 世界华人消化杂志 2026; 34(2): 110-120
Revised: February 9, 2026
Accepted: February 13, 2026
Published online: February 28, 2026
The comprehensive management of hepatocellular carcinoma (HCC) relies on the assistance of biomarkers. Serum alpha-fetoprotein (AFP) is the most classic HCC biomarker, but approximately 30% of patients in clinical practice are AFP-negative, which fails to meet clinical needs. The discovery and clinical application of desgammacarboxy prothrombin (DCP) have demonstrated its indispensable role in the early screening, diagnosis, treatment, and prognosis assessment of HCC. Furthermore, predictive models integrating indicators such as DCP and AFP hold significant importance in clinical practice. This article systematically reviews the current applications of DCP in HCC screening, diagnosis, prognosis assessment, efficacy evaluation, and recurrence monitoring, aiming to provide a basis for clinicians to gain a deeper understanding of the value of DCP in managing HCC.
- Citation: Liu ZH, Wang WJ, Dang SS. Value of des-gamma-carboxy prothrombin in comprehensive management of hepatocellular carcinoma. Shijie Huaren Xiaohua Zazhi 2026; 34(2): 110-120
- URL: https://www.wjgnet.com/1009-3079/full/v34/i2/110.htm
- DOI: https://dx.doi.org/10.11569/wcjd.v34.i2.110
核心提要: 异常凝血酶原(des-gamma-carboxy prothrombin, DCP)在肝细胞癌全程管理中展现出核心价值. 它不仅弥补了肝癌患者甲胎蛋白阴性的诊断缺口, 更因其与肿瘤侵袭性生物学的高度关联, 成为预测微血管侵犯及术后复发的关键标志. 从辅助肝移植患者筛选, 到动态评估局部与系统治疗疗效, DCP及其参与的整合预测模型, 为肝癌的风险分层与个体化精准治疗实践提供了循证支持.
肝癌已成为全球癌症防控面临的重大挑战, 在所有恶性肿瘤中发病率居第六位, 死亡率居第三位[1]. 流行病学数据显示[2,3], 过去二十年间全球肝癌发病率上升了53.7%, 死亡率增加48.0%, 亚洲地区承担超过七成的疾病负担[4]. 肝细胞癌(hepatocellular carcinoma, HCC)是肝癌最主要的病理类型, 约占全部病例的90%[5]. 近年来HCC系统性治疗取得显著进展, 但是患者五年生存率仍普遍低于20%[6], 这一严峻现状与HCC早期诊断困难、治疗后易复发和高度的肿瘤异质性密切相关. 因此, 实现HCC的早期筛查、准确诊断与动态预后评估, 对改善患者生存结局具有关键意义.
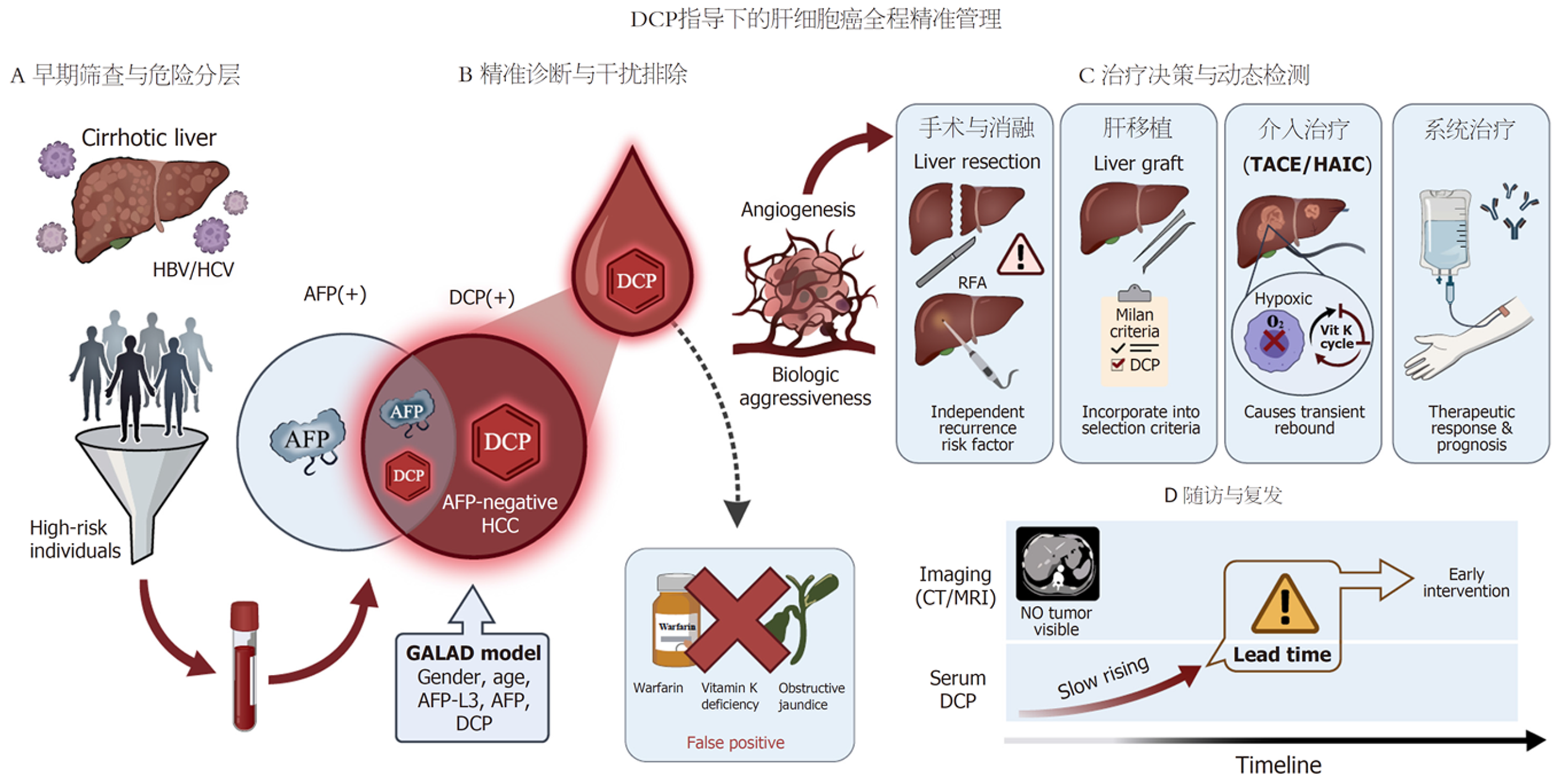
早期肝癌经过肝切除术、肝移植和局部消融等治疗可获得较长的生存期, 但是大多数HCC在发现时已为中晚期, 其疗效很大程度上依赖于对肝硬化等高危人群的定期监测与早期发现. 现行临床指南建议对高危人群每6 mo进行一次腹部超声检查, 可联合或不联合甲胎蛋白(alpha-fetoprotein, AFP)检测. 然而, 临床上约30%的HCC患者AFP阴性, 并且其诊断准确性受临界值设定、肝脏炎症活动度及基础肝病病因等多种因素影响[7]. 即便联合超声, 肝癌的检出率为74.1%[8], 意味着约四分之一的患者面临漏诊风险. 此外, 慢性肝炎和肝硬化等良性疾病也可能引起AFP非特异性升高, 导致假阳性结果[9]. 因此, 临床上亟需新型HCC标志物以弥补AFP的不足.
异常凝血酶原(des-gamma-carboxy prothrombin, DCP), 也被称为维生素K缺乏或拮抗剂诱导蛋白Ⅱ(protein Induced by vitamin K absence or antagonist-Ⅱ, PIVKA-Ⅱ), 是一种近年来备受关注的HCC潜在血清标志物[10]. 与AFP相比, DCP不易受妊娠或急性炎症等因素干扰, 且与肿瘤血管侵犯、分化程度及术后复发密切相关. 现有研究表明DCP在HCC诊断中表现出良好性能, 尤其对AFP阴性患者具有补充诊断信息, 此外, DCP在风险分层、疗效评估及复发预测方面也展现出独特优势. 目前, DCP正逐步整合进入HCC的全程管理体系, 然而关于DCP在肝癌管理中的整合路径尚未形成共识, 其在肝癌诊疗标准化应用仍缺乏系统总结与循证推荐. 为此, 本文系统梳理DCP的生物学特性与临床应用进展, 重点分析其在HCC早期筛查、鉴别诊断、疗效评价及复发预测中的具体效能, 结合最新循证医学证据与临床研究, 为DCP在HCC精准管理中的应用提供理论依据与实践参考.
DCP本质上是在维生素K代谢异常条件下生成的DCP, 其形成与肝细胞对凝血酶原的翻译后修饰障碍密切相关. 在正常生理状态下, 肝细胞合成的凝血酶原前体在N端结构域含有10个特定的谷氨酸残基, 这些残基在维生素K依赖性γ-谷氨酰羧化酶的催化下, 可完全羧化为γ-羧化谷氨酸(γ-carboxyglutamic acid, Gla)残基. 该结构是凝血酶原发挥正常功能的关键, 只有经过羧化酶的修饰, 凝血酶原才能与钙离子及磷脂膜结合, 进而参与凝血过程[11]. 然而, 当维生素K摄入不足、华法林等拮抗剂抑制或肝癌细胞中羧化酶功能受损时, 上述羧化反应受阻, 导致部分谷氨酸残基未能羧化, 从而生成羧化程度不等(含0-9个Gla残基)的DCP, 即DCP[12]. 从结构上看, 此类分子因缺乏完整的功能结构域, 无法有效结合钙离子, 不再具有凝血活性. 在早期实验室研究中, 其电泳迁移率由β区带移至γ区带的异常迁移, 是识别DCP的重要依据.
DCP的发现始于1963年, Hemker等[13]在慢性肝病及香豆素类药物治疗者血浆中首次发现无凝血活性的异常凝血因子, 揭示了维生素K与凝血功能之间的密切联系; 随后, Stenflo[14]纯化并解析了牛血浆中DCP的结构, 确立了其因Gla结构域缺陷导致功能丧失的理论基础; 直至1984年, Liebman等[15]的研究取得了突破性的进展, 他们首次报道HCC患者血清DCP水平显著升高, 且DCP的升高在维生素K补充后不可逆, 这将DCP与恶性肿瘤联系起来, 开启了DCP作为肿瘤标志物的研究新阶段.
DCP并非单一分子实体, 依据羧化程度差异, 主要可分为两种亚型: 在维生素K缺乏、服用维生素K拮抗剂或梗阻性黄疸等情况下产生的DCP变体通常仅含1-5个Gla残基, 被称为NX-DCP(NX-des-γ-carboxyprothrombin). 而在HCC患者中, 肿瘤细胞产生的DCP多数仍保留6-9个Gla残基, 即广义DCP[16]. 这两类亚型在血清学检测中具有重要区别, 目前常用的MU-3抗体可识别上述两类亚型, 但无法区分其Gla残基数量差异, 为解决鉴别难题, 研究表明NX-DCP可通过P-11或P-16特异性抗体进行检测[17]. 临床上常采用DCP与NX-DCP的比值进行鉴别诊断: 若比值>1.5, 提示HCC可能性较高; 若比值≤1.5, 则更可能源于外源性维生素K缺乏或胆汁淤积等非肿瘤因素[18]. 此外, 解读DCP升高时, 还需警惕非HCC恶性肿瘤的干扰, 胃癌、胰腺导管腺癌及胆管癌患者中也可能观察到DCP水平升高, 所以对于AFP阴性但DCP升高的患者, 仍需结合影像学排除消化道其他肿瘤.
现阶段, DCP的主流检测方法包括化学发光酶免疫测定、酶联免疫吸附测定、液相结合分析法以及放射免疫分析. 然而, 在梗阻性黄疸、维生素K缺乏、饮酒或服用华法林的非HCC人群中, 目前的检测方法并不能有效区分两种亚型. 为提升检测性能, 有研究者开发上转发光免疫层析检测卡[19]及免疫磁性微球检测法[20]等新型技术, 这些方法显示了优于化学发光酶免疫测定的抗干扰能力以及灵敏度, 但尚未在临床推广.
HCC中DCP的升高机制与良性维生素K缺乏存在本质差异, 后者在补充维生素K后DCP水平可迅速恢复至正常范围, 而HCC患者的DCP水平通常不受补充影响, 甚至持续上升, 这提示其升高源于肿瘤细胞自身的维生素K代谢紊乱[15]. HCC患者DCP升高的机制尚不明确, 可能涉及多个分子环节, 包括肝癌细胞中环氧化物还原酶复合体亚单位1表达下调, 导致维生素K循环障碍、多聚二磷酸腺苷核糖聚合酶-1等信号分子过度激活, 以及肿瘤内部缺氧等微环境因素共同作用, 引起γ-羧化酶功能障碍[21,22].
值得关注的是, HCC患者体内异常升高的DCP不仅是重要的血清学标志物, 本身还参与肿瘤的生物学进程. 研究表明[23], DCP可通过与肝细胞生长因子受体c-Met等相互作用, 激活JAK1-STAT3信号通路, 促进肿瘤细胞的DNA合成与增殖; 同时它能上调血管内皮生长因子等促血管生成因子表达, 刺激肿瘤新生血管形成[24]. 此外, DCP还直接参与肿瘤微环境的免疫调节, 抑制CD45+和CD3+免疫浸润, 通过增加活性氧产生诱导T细胞凋亡[25]. 因此, DCP不仅可作为HCC诊断与监测的生物标志物, 更通过激活促增殖与促血管生成信号通路参与肿瘤进展, 成为潜在的干预靶点.
筛查与监测是HCC全程管理的首要环节, 旨在从肝硬化等高危人群中早期发现肿瘤, 为根治性治疗争取时间窗. 多项研究证实DCP具有早期预警价值[26-29]. 与影像学检查相比, DCP往往在发现明确病灶前即呈现升高趋势[26]. Ricco等[27]的纵向研究观察到, 肝硬化患者在向HCC演变的过程中, 血清DCP水平呈持续上升趋势. 为明确其风险阈值, Caviglia等[28]对200例病毒性肝炎相关肝硬化患者进行了36 mo前瞻性随访显示, 发现最终发展为HCC的患者基线DCP水平显著高于未患癌组; 而当基线DCP>55 mAU/mL时, HCC的发生风险显著增加[危险比hazard ratio (HR) = 1.99]. 另一项纳入1187名肝硬化患者的大规模研究表明[29], 将DCP界值设定为50 mAU/mL, 其对HCC的筛查灵敏度可达80.0%. 除一般肝硬化人群外, 针对经直接抗病毒药物(direct-acting antiviral, DAA)治愈的丙型肝炎病毒(hepatitis C virus, HCV)相关肝硬化患者, DCP在残留致癌风险的监测中同样展现出独特价值[30]. 尽管病毒已清除, 但这部分人群仍面临癌变风险. 一项纳入400例HCV相关肝硬化患者的前瞻性研究显示[31], DCP与AFP均可独立预测HCC发生, DAA治疗结束时DCP≥41 mAU/mL的患者, 4年累积HCC发生率达24%; 而若DCP与AFP均为阳性, 则该风险升高18%. 这提示, DCP与AFP的联合检测是DAA治疗的有效监测策略.
鉴于DCP的预警潜力, 研究者将其与其它参数整合, 构建了多个综合预测模型以提升预测精度. Caviglia等[32]构建了包含年龄、性别、丙氨酸氨基转移酶(alanine aminotransferase, ALT)、天冬氨酸氨基转移酶、γ-谷氨酰转移酶、血小板计数、白蛋白、总胆红素和DCP的PIVKA-Ⅱ模型, 该模型在验证队列中预测12 mo内HCC发生的区分度良好, 其受试者工作曲线下面积(area under the curve, AUC)达0.84, 能有效区分HCC发生的低、中、高危人群, 三者HCC累计发病率呈现显著梯度差异, 分别为2.7%、4.0%和14.3%, 显示出良好的临床实用性. 在国际上, GALAD等权威评分系统将DCP列为核心组成部分[33], 该评分在肝硬化人群中表现出高灵敏度[34], 单时间点和纵向GALAD对早期HCC的敏感性分别为53.8%和69.2%[35], 且其性能受病因与抗病毒治疗的影响较小[36]. 然而, 美国一项Ⅲ期前瞻性队列研究指出, GALAD评分在提高敏感性的同时, 也可能伴随假阳性率的上升[37]. 考虑到AFP异质体比率(lens culinaris agglutinin-reactive alpha-fetoprotein percentage, AFP-L3%)检测成本较高且普及度有限, 为了在成本与效益间取得平衡, 研究者在GALAD模型的基础上排除AFP-L3%, 仅保留性别、年龄、AFP和DCP的简化模型, 如GAAD[38,39]、ASAP[40-42]、AALP[43]、GAAP[44]. 相关研究显示[45,46], 类GALAD模型的诊断准确性与GALAD模型相当, 简化的模型具备更好的成本效益比和临床可及性, 更适合在大规模人群筛查中推广应用. 最新版HES V2.0模型进一步整合了AFP、AFP-L3与DCP的动态变化, 以及年龄、ALT、血小板与病因等变量, 在HCC诊断前6-24 mo的预测中, 其真阳性率优于传统GALAD评分, 代表了未来动态监测的新方向[47].
基于上述证据, DCP在临床指南中的地位逐渐演变, 但全球共识尚未统一. 总体而言, DCP在HCC筛查与监测中表现出稳定甚至优于AFP的灵敏度, 其与AFP等指标的联合应用, 以及在GALAD、HES等预测模型中的整合, 不仅提前了早期肝癌的检出时间窗, 也为高危人群的分层管理与医疗资源优化提供了量化工具. 然而, 不同国家和地区指南对DCP在筛查中的定位尚存差异. 在指南推荐层面, 日本肝病学会指南率先将DCP与AFP、AFP-L3并列为"极高危人群"的必检项目, 推荐每3-4 mo同步检测[48]; 中国《原发性肝癌诊疗指南(2024)》[49]、《原发性肝癌的分层筛查与监测指南》[50]以及中国临床肿瘤学会[51]和中国台湾[52]等亦将DCP正式纳入筛查路径, 强调其与超声的联合应用. 相比之下, 欧洲肝脏研究协会[53]、美国肝病研究协会[54]、韩国肝癌协会[55]、欧洲肿瘤内科会[56]、英国胃肠病学会[57]等机构仍维持每6 mo一次腹部超声联合AFP的推荐, 认为除AFP外的血清标志物证据尚不充分, 仅建议在个体化评估中考虑使用. 各主要国际指南对HCC高危人群筛查手段的具体推荐总结如表1所示. 尽管各国指南不统一, 但随着GALAD等新型模型证据的积累以及检测标准化的推进, DCP在筛查策略中的地位逐步提升.
| 指南/学会 | 年份 | 推荐意见 |
| 国家卫健委《原发性肝癌诊疗指南》[49] | 2024 | 对肝癌高危人群的筛查与监测(超声显像联合血清AFP), 有助于肝癌的早期发现、早期诊断和早期治疗, 同时可以显著降低患者的死亡风险(1A). 基于性别、年龄、AFP、DCP和AFP-L3%构建的GALAD模型在诊断早期肝癌的灵敏度和特异度分别为85.6%和93.3%, 有助于AFP阴性肝癌的早期诊断(1A). 基于性别、年龄、AFP、DCP构建的简化的GAAD模型及ASAP模型与GALAD模型诊断效能类似(1A) |
| 《原发性肝癌分层筛查与监测指南》[50] | 2020 | 腹部US联合血清AFP是肝癌监测的一线工具(A1). 血清AFP联合AFP-L3%及PIVKA-II检测, 可提高早期肝癌的检出率(B2) |
| 欧洲肝病学会[53] | 2024 | 建议每6 mo行1次肝脏超声筛查HCC; US联合AFP可在降低特异度的同时提高灵敏度, 属合理选择. 目前尚无充分数据支持其他新兴影像(如简化MRI)或血清标志物常规用于监测(LoE 3, 强推荐, 共识) |
| 美国肝病学会[54] | 2023 | 建议使用超声联合AFP进行每半年(约6 mo)一次的HCC监测(证据等级2, 强推荐). 除AFP外, 不推荐对肝硬化或慢性HBV感染的高危患者常规使用基于CT或MRI的影像学检查及其他肿瘤标志物进行监测(证据等级5, 弱推荐) |
| 英国胃肠病学会[57] | 2024 | 对于肝硬化人群及特定亚群的慢性HBV感染者, 应考虑每6 mo进行一次超声检查及AFP检测(证据强; 推荐强) |
| 欧洲肿瘤内科学会[56] | 2025 | HCC监测应包含每6 mo一次的腹部超声(若条件允许可采用多期横断面影像学检查), 联合或不联合AFP评估(Ⅱ, A) |
| 日本肝病学会[48] | 2021 | 对于高危人群, 推荐每6 mo检测一次AFP、DCP和AFP-L3%; 对于极高危人群, 推荐每3-4 mo检测一次 |
| 韩国肝癌研究组[55] | 2022 | 建议每6 mo进行一次肝脏超声加血清AFP检测(A1) |
| 台湾肝癌协会[52] | 2022 | 临床实践中应联合超声与肿瘤标志物(AFP和/或DCP)进行HCC监测(A:100%; E:2; R:B) |
在肝癌的临床诊断流程中, 增强CT或MRI等影像学检查占据主导地位, 但对于直径小于2 cm的微小结节或伴有重度脂肪变性的病灶, 其定性诊断仍面临挑战. 在此背景下, DCP凭借其独特的生物学特性, 成为弥补影像学与传统血清学短板的关键工具. 大量循证医学证据证实, DCP在HCC的诊断中表现出优异效能[58-60]. Ji等[61]在HBV肝硬化队列中设定DCP临界值为45 mAU/mL, 结果显示其诊断HCC的AUC高达0.94, 灵敏度与特异度分别为77%和95%. 这一结果表明, 对于影像学特征不典型的疑难病例, 高水平DCP可作为支持诊断HCC的有力佐证, 有效缩短确诊时间窗.
DCP的一个重要诊断价值, 源于其与AFP的表达机制差异. AFP的升高主要与肝细胞的去分化及再生相关, 而DCP的产生则源于肿瘤细胞内维生素K循环障碍及缺氧微环境诱导的羧化酶活性缺陷[10]. 由于表达机制不同, 二者在血清水平上相关性弱, 使得DCP在AFP阴性人群中仍能维持较高的阳性率, 有效拓展了AFP的诊断盲区. 亚太地区专家共识指出, DCP是AFP阴性HCC患者最主要的补充诊断指标[62]. 因此, AFP联合DCP检测策略可产生的协同效应, 通过捕捉不同分子亚型HCC的生物学特征, 显著提升诊断的灵敏度与准确性. DCP与AFP在诊断效能的直接比较上, 学界观点尚未完全统一. 多数研究, 如Qian等[63]的结果, 显示DCP在诊断性能上优于AFP, 亦有如Hadi等[64]的研究认为二者表现相当. 尽管结论存在差异, 但真正推动DCP走向临床前沿的是其与AFP的联合检测策略[65], 二者联合可产生协同效应, 显著提升整体诊断性能.
在单一标志物联合应用的基础上, 为进一步追求诊断精准化, 研究者进而开发了多种整合DCP的多参数预测与诊断模型. 研究者针对非酒精性脂肪性肝病(non-alcoholic fatty liver disease, NAFLD)患者构建了一个综合预测模型, 该模型整合了DCP、年龄、性别、磷脂酰肌醇蛋白聚糖-3及脂联素五项指标[66]. 该模型在NAFLD人群中展现出AUC高达0.948的诊断效能. 更前沿的探索则集中于新的生物标志物组合, 如Jin等[67]提出的tsRNA-Thr-5-0015与AFP、DCP联用, 以及Xing等[68]开发的包含DCP的P4面板等, 这些模型在早期HCC检测中展现出极高潜力, 标志着肝癌诊断正从依赖单一影像或血清学指标, 迈向整合多维度信息的精准化新阶段.
HCC的治疗策略依赖于对肿瘤分期及生物学行为的精准评估. DCP不仅反映肿瘤负荷, 更与肿瘤的侵袭性、血管生成能力密切相关, 因此在不同治疗模式的疗效监测与预后判断中扮演着关键角色.
肝切除术是肝功能储备良好HCC患者获得根治性治疗的首选方案, 然而术后高复发率仍是限制患者长期生存的主要瓶颈. 微血管侵犯及血管包绕肿瘤细胞簇是术后复发的关键病理因素, 但由于缺乏术中识别手段, 其术前预测至关重要. 术前检测血清标志物为识别高复发风险人群提供了一种便捷、无创的手段, 有助于指导个体化手术方案的制定及术后辅助治疗的选择[69]. 研究表明[70], 高水平DCP与肿瘤低分化程度及微血管侵犯密切相关, 是HCC预测复发及不良预后的独立危险因素[71]. 一项基于多序列MRI影像组学的研究证实[72], 血清DCP>40 mAU/mL是血管包绕肿瘤细胞簇的独立预测因子, 该研究构建的DCP联合影像组学模型, 在预测高危病理特征方面展现出优异效能, 其AUC可达0.817, 为术前无创筛选高危复发人群提供了有力工具. 在长期预后方面, DCP的基线水平与患者的生存获益密切相关. 一项纳入792例根治性切除患者的回顾性分析显示[73], 术前DCP水平是总生存期和复发时间的独立预后因素. 极高DCP水平组(>399 mAU/mL)的复发风险较正常组增加了79%(HR = 1.790), 提示此类患者术后需接受更为密集的随访或辅助治疗. 这一结论在AFP阴性人群中尤为突出. 针对AFP阴性并接受根治性切除的HCC患者的多中心研究证实[74], 高DCP组(>600 mAU/mL)的5年总生存率相对于低DCP组显著较低(57.6% vs 76.3%, P<0.001), 且复发率高达61.0%. 更为重要的是, 利用DCP较短的血清半衰期进行术后动态监测, 可为评估手术效果与早期干预提供关键依据. 若术后DCP未如期降至正常范围, 往往提示存在残留微小病灶或早期复发风险. Wang等[75]的研究指出, DCP的清除延迟比AFP更能敏锐地反映手术彻底性, 为术后辅助治疗决策提供了重要依据. 为提升预测精度, DCP与肿瘤体积的比值被提出作为预测早期复发的新兴指标[76]. DCP与FIB-4指数的联合应用, 有助于评估癌症病例的肝纤维化状况来辅助评估预后[77]. 此外, 由γ-谷氨酰转移酶、AFP与DCP共同构建的复合指标, 甚至可将早期复发预测率提升至95.2%[78]. 这些综合评估体系的建立, 为实现HCC术后个体化管理提供了坚实的循证医学证据.
射频消融(radiofrequency ablation, RFA)是早期小肝癌(通常直径≤3 cm)的重要根治性手段. 由于RFA主要通过热效应诱导肿瘤凝固性坏死, 术后早期的炎性水肿及坏死组织吸收过程可能干扰影像学对残余病灶的判断, 血清学标志物因不受局部组织改变影响, 在疗效评估中具有独特优势[79]. 在术前风险分层方面, 一项针对162例接受RFA治疗的早期HCC患者的研究构建了综合临床评分系统, 将基线DCP>40 mAU/mL确定为术后复发的独立危险因素, 该系统同时整合了循环肿瘤细胞(circulating tumor DNA, ctDNA)、AFP及肿瘤大小与数目等指标[80]. 这一发现提示[81], 术前DCP升高的患者在RFA后面临更高的复发风险, 临床需对其制定更为密集的随访计划. 此外, DCP的短半衰期特性使其成为一个动态监测消融疗效的灵敏指标, Pan等[82]创新性地提出了"DCP动态速率"概念, 证实DCP下降速率的减缓与局部复发密切相关. 相较于传统影像学检查, 该指标在评估治疗反应方面展现出显著的时效性优势, 有助于在影像学发现明确复发灶之前识别出高危患者, 从而为早期补救性治疗争取宝贵的时间窗.
肝移植是合并肝硬化的早期HCC患者获得长期生存的理想根治性手段[83]. 然而, 以米兰标准为代表的传统体系主要依赖影像学肿瘤负荷, 虽能筛选出预后良好的患者, 却也因形态学限制过于严格, 可能排除部分生物学行为良好的潜在获益者. 对此, 引入DCP作为生物学标志物, 有助于优化受者筛选策略[84]. 将DCP纳入现有评估体系, 在不显著增加术后复发风险的前提下, 科学地拓展肝移植的适用人群. Wang等[85]的研究支持了这一观点, 他们发现将DCP纳入现有的肝移植标准可增加符合条件的HCC患者人数, 且不会影响移植后的长期预后. DCP优化筛选的效能, 源于其与不良病理特征及复发风险的强相关性. Bhatti等[86]研究证实, DCP是预测肝移植后微血管侵犯和肿瘤复发的强力因子. 正因如此, 移植前的DCP水平成为影响预后的关键危险因素. 当将其与AFP、肿瘤大小及数目等进行综合评估时, 构建预测效能极高的模型(AUC = 0.967), 从而精准识别出那些虽肿瘤负荷略超传统标准, 但生物学行为相对温和的潜在获益人群[87]. 此外, DCP与其他标志物的联合应用可提升预测复发的特异性. 一项纳入285例移植受者的前瞻性研究进一步显示, DCP与AFP-L3双阳性对肝癌复发的阳性预测值高达61.1%[88]. 这些证据表明, DCP在优化肝移植患者筛选与预后评估中提供了可靠的血清学依据.
经动脉化疗栓塞(transarterial chemoembolization, TACE)与肝动脉灌注化疗(hepatic arterial infusion chemotherapy, HAIC)是不可切除HCC应用最广泛的局部区域治疗手段, DCP是评估疗效与预测预后的关键血清学指标. Meta分析证实[89], 基线低水平DCP与TACE/HAIC治疗患者的更长总生存期显著相关. 更为关键的是, 治疗后的DCP应答是总生存期和无进展生存期延长的独立预测因素. 基于DCP这一动态监测特性, 研究者已将其整合入疗效评估体系. Lu等[90]联合DCP与AFP构建了术前风险评估评分; Wang等[91]进一步验证了DCP在预测TACE早期疗效中的价值, 确立了其在个体化治疗决策中的参考地位. 研究证实[92], 在TACE基础上联合PD-1抑制剂, 能更有效地降低患者血清DCP水平, 同时伴随血管内皮生长因子和成纤维细胞生长因子的显著下降. 这提示, DCP水平的动态变化可能作为评估联合治疗方案疗效的敏感生物标志物. 因此, 将DCP动态监测纳入介入治疗的管理, 已成为临床评估疗效及优化治疗策略的关键环节[93]. 需要特别注意的是, 在解读DCP变化时需警惕"假性反跳"现象, TACE以及抗血管内皮生长因子药物可能在肿瘤内部造成缺氧微环境, 暂时抑制维生素K循环, 导致部分治疗有效的患者出现一过性DCP水平反跳. 为避免误判疗效, 建议以4-6 wk内的动态变化趋势作为评估标准, 而非依赖单次检测的绝对值. DCP不仅是疗效监测的标志物, 其生成机制本身也提示了潜在的治疗靶点. 一项维生素K联合TACE的随机对照试验显示, 通过纠正羧化缺陷可有效降低DCP水平, 并增强TACE的抗肿瘤效果[94]. 这从机制上印证, 针对DCP产生路径的干预可能改善治疗结局.
在晚期肝癌的系统治疗中, DCP正逐步成为指导方案选择与疗效预测的关键生物标志物, 其价值贯穿于治疗前决策、治疗中监测与治疗后评估的全过程. 新近大样本回顾性分析表明[95], 基线DCP水平有助于指导一线免疫联合方案的决策. 当DCP>400 mAU/mL时, 患者可能从"度伐利尤单抗联合曲美木单抗"(Durvalumab/Tremelimumab, Durva/Treme)方案中获得更大获益; 而低DCP水平患者则可能更适合接受"阿替利珠单抗联合贝伐珠单抗"方案. 这一发现为晚期肝癌的个体化精准用药提供了重要参考. 此外, DCP的动态变化在疗效预测方面也展现出显著价值. 研究显示[96], DCP水平与Durva/Treme治疗的客观缓解率呈正相关, 其在治疗后4 wk的变化可能预测HCC抗肿瘤反应和疾病进展[97], 这一特性对于缺乏AFP表达的患者尤为重要[98]. 在更为复杂的联合治疗模式下, DCP仍保持良好的预测效能. 针对接受仑伐替尼单药或联合免疫检查点抑制剂治疗的不可切除HCC患者, 包含DCP的PIMET综合评分模型已被证实能有效进行预后分层[99]. 另有研究指出, 在立体定向放疗联合靶向及免疫治疗治疗后6-8 wk DCP较基线下降≥70%, 这提示患者的中位无进展生存期可显著延长至15.6 mo, 总生存期亦显著优于未达标者[100]. 值得注意的是, DCP不仅是预测标志物, 其生成机制本身也指向了潜在的治疗干预靶点. 索拉非尼与维生素K联合用药相比单用索拉非尼可获得更高的客观缓解率[101], 这一发现验证了纠正维生素K代谢的潜在治疗价值, 也进一步强化了DCP作为治疗反应监测指标和潜在治疗靶点的双重角色. 另一项对比"替雷利珠单抗联合仑伐替尼"与仑伐替尼单药治疗中晚期肝癌的研究发现[102], 联合治疗组不仅在客观缓解率和6 mo生存率上优于单药组, 其治疗后DCP的下降幅度也更为显著. 这提示DCP的动态清除可作为评价PD-1单抗联合靶向药物疗效的早期替代终点. 然而, 临床在解读DCP变化时需警惕药物引起的非肿瘤性升高. 一项基于OpenVigil数据库的挖掘研究发现[103], 索拉非尼治疗期间报告关联性最强的不良事件信号之一即DCP升高(OR = 502.46). 这提示, 某些酪氨酸激酶抑制剂本身可能干扰维生素K循环, 导致DCP水平出现与肿瘤负荷无关的药理学升高, 临床医生在评估疾病进展时需仔细甄别.
基于现有循证医学证据, DCP已广泛渗透至肝癌临床诊疗的各个环节, 为了更清晰地说明DCP在不同临床场景下的应用策略, 本文尝试梳理了DCP在HCC全程管理中的应用(图1), 为临床医生在肝癌的诊疗过程中提供参考. 在筛查领域, DCP在一定程度上弥补了AFP阴性肝癌的识别盲区, 提升了早期诊断的灵敏度, 特别是DCP被纳入的GALAD等整合预测模型, 高危人群风险分层窗口得以进一步前移. 而在治疗决策层面, DCP与肿瘤的血管侵犯、微小病灶残留以及恶性行为高度相关, 它可以用于预测肝癌术后的复发风险, 帮助优化肝移植患者的选择, 并能监测靶向联合免疫治疗的早期效果, 这些作用均肯定了其动态预警的辅助价值[10].
尽管DCP的临床效能日益受到认可, 但在全球范围内的标准化应用仍面临严峻挑战. 当前最主要的阻碍源于检测体系的差异性与干扰因素的控制, 包括临界值不统一、检测流程差异, 以及维生素K缺乏等因素所致的假阳性干扰. 值得注意的是, 各国指南对DCP的推荐力度存在差异, 这种差异可能源于各地区临床研究证据的多寡, 也可能与当地的诊疗习惯、医疗资源分布有关, 所以开展更多跨地区的高质量前瞻性研究, 是弥合这一认识差距的关键.
DCP从早期筛查、诊断辅助到治疗反应动态评估, 已展现出多层次的临床应用潜力. 然而, 若仅停留在价值肯定的层面, 可能无助于解决肝癌面临的临床困境. 未来的研究需要更关注DCP在真实世界诊疗流程中如何整合, 并深入分析其生物学意义如何转化为实际的临床预测能力. 在精准医疗的背景下, DCP能否真正融入HCC个体化诊疗体系, 不仅取决于技术本身的优化, 更依赖于我们在临床实践与学术对话中, 对其定位与局限保持清醒的审视.
| 1. | Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12-49. [PubMed] [DOI] |
| 2. | Hwang SY, Danpanichkul P, Agopian V, Mehta N, Parikh ND, Abou-Alfa GK, Singal AG, Yang JD. Hepatocellular carcinoma: updates on epidemiology, surveillance, diagnosis and treatment. Clin Mol Hepatol. 2025;31:S228-S254. [PubMed] [DOI] |
| 3. | Singal AG, Kanwal F, Llovet JM. Global trends in hepatocellular carcinoma epidemiology: implications for screening, prevention and therapy. Nat Rev Clin Oncol. 2023;20:864-884. [PubMed] [DOI] |
| 4. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [PubMed] [DOI] |
| 5. | Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A. Hepatocellular carcinoma. Lancet. 2022;400:1345-1362. [PubMed] [DOI] |
| 6. | Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Azevedo E Silva G, Chen WQ, Eser S, Engholm G, Stiller CA, Monnereau A, Woods RR, Visser O, Lim GH, Aitken J, Weir HK, Coleman MP; CONCORD Working Group. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018;391:1023-1075. [PubMed] [DOI] |
| 7. | Hanif H, Ali MJ, Susheela AT, Khan IW, Luna-Cuadros MA, Khan MM, Lau DT. Update on the applications and limitations of alpha-fetoprotein for hepatocellular carcinoma. World J Gastroenterol. 2022;28:216-229. [PubMed] [DOI] |
| 8. | Singal AG, Haaland B, Parikh ND, Ozbay AB, Kirshner C, Chakankar S, Porter K, Chhatwal J, Ayer T. Comparison of a multitarget blood test to ultrasound and alpha-fetoprotein for hepatocellular carcinoma surveillance: Results of a network meta-analysis. Hepatol Commun. 2022;6:2925-2936. [PubMed] [DOI] |
| 9. | Yeo YH, Lee YT, Tseng HR, Zhu Y, You S, Agopian VG, Yang JD. Alpha-fetoprotein: Past, present, and future. Hepatol Commun. 2024;8:e0422. [PubMed] [DOI] |
| 10. | Dong L, Qiu X, Gao F, Wang K, Xu X. Protein induced by vitamin K absence or antagonist II: Experience to date and future directions. Biochim Biophys Acta Rev Cancer. 2023;1878:189016. [PubMed] [DOI] |
| 11. | Uehara S, Gotoh K, Handa H, Honjo K, Hirayama A. Process of carboxylation of glutamic acid residues in the gla domain of human des-gamma-carboxyprothrombin. Clin Chim Acta. 1999;289:33-44. [PubMed] [DOI] |
| 12. | Zhang YS, Chu JH, Cui SX, Song ZY, Qu XJ. Des-γ-carboxy prothrombin (DCP) as a potential autologous growth factor for the development of hepatocellular carcinoma. Cell Physiol Biochem. 2014;34:903-915. [PubMed] [DOI] |
| 13. | Hemker HC, Veltkamp JJ, Hensen A, Loeliger EA. Nature of prothrombin biosynthesis: Preprothrombinaemia in vitamin K-deficiency. Nature. 1963;200:589-590. [PubMed] [DOI] |
| 14. | Stenflo J. Vitamin K and the biosynthesis of prothrombin. II. Structural comparison of normal and dicoumarol-induced bovine prothrombin. J Biol Chem. 1972;247:8167-8175. [PubMed] |
| 15. | Liebman HA, Furie BC, Tong MJ, Blanchard RA, Lo KJ, Lee SD, Coleman MS, Furie B. Des-gamma-carboxy (abnormal) prothrombin as a serum marker of primary hepatocellular carcinoma. N Engl J Med. 1984;310:1427-1431. [PubMed] [DOI] |
| 16. | Nakao A, Suzuki Y, Isshiki K, Kimura Y, Takeda S, Kishimoto W, Nonami T, Harada A, Takagi H. Clinical evaluation of plasma abnormal prothrombin (des-gamma-carboxy prothrombin) in hepatobiliary malignancies and other diseases. Am J Gastroenterol. 1991;86:62-66. [PubMed] |
| 17. | Sumi A, Akiba J, Ogasawara S, Nakayama M, Nomura Y, Yasumoto M, Sanada S, Nakashima O, Abe T, Yano H. Des-γ-carboxyprothrombin (DCP) and NX-DCP expressions and their relationship with clinicopathological features in hepatocellular carcinoma. PLoS One. 2015;10:e0118452. [PubMed] [DOI] |
| 18. | Toyoda H, Kumada T, Osaki Y, Tada T, Kaneoka Y, Maeda A. Novel method to measure serum levels of des-gamma-carboxy prothrombin for hepatocellular carcinoma in patients taking warfarin: a preliminary report. Cancer Sci. 2012;103:921-925. [PubMed] [DOI] |
| 19. | Huang Y, Zhang S, Zheng Q, Li Y, Yu L, Wu Q, Zheng J, Wu Y, Qiu F, Gao Q, Zhang J. Development of up-converting phosphor technology-based lateral flow assay for quantitative detection of serum PIVKA-II: Inception of a near-patient PIVKA-II detection tool. Clin Chim Acta. 2019;488:202-208. [PubMed] [DOI] |
| 20. | Chieh JJ, Huang KW, Chuang CP, Wei WC, Dong JJ, Lee YY. Immunomagnetic Reduction Assay on Des-Gamma-Carboxy Prothrombin for Screening of Hepatocellular Carcinoma. IEEE Trans Biomed Eng. 2016;63:1681-1686. [PubMed] [DOI] |
| 21. | Welsh J, Bak MJ, Narvaez CJ. New insights into vitamin K biology with relevance to cancer. Trends Mol Med. 2022;28:864-881. [PubMed] [DOI] |
| 22. | Liu Y, Li H, Dong J, Ma L, Liao A, Rong Z, Zhou Z, Cao L, Wang F, Wang J, Wang A. mTOR and ERK regulate VKORC1 expression in both hepatoma cells and hepatocytes which influence blood coagulation. Clin Exp Med. 2019;19:121-132. [PubMed] [DOI] |
| 23. | Yang Y, Li G, Lu Z, Liu Y, Kong J, Liu J. Progression of Prothrombin Induced by Vitamin K Absence-II in Hepatocellular Carcinoma. Front Oncol. 2021;11:726213. [PubMed] [DOI] |
| 24. | Wang SB, Cheng YN, Cui SX, Zhong JL, Ward SG, Sun LR, Chen MH, Kokudo N, Tang W, Qu XJ. Des-gamma-carboxy prothrombin stimulates human vascular endothelial cell growth and migration. Clin Exp Metastasis. 2009;26:469-477. [PubMed] [DOI] |
| 25. | Wan Y, Geng X, Liu Q, Lu S, Liu Y, Wang T, Zhang X, Li N, Li D, Li Z, Liu J, Wu H, Chen J. CTSE inhibits anti-tumor T cell response by promoting des-γ-carboxy prothrombin releasing in hepatocellular carcinoma. Cell Death Dis. 2025;16:434. [PubMed] [DOI] |
| 26. | Shahini E, Pasculli G, Solimando AG, Tiribelli C, Cozzolongo R, Giannelli G. Updating the Clinical Application of Blood Biomarkers and Their Algorithms in the Diagnosis and Surveillance of Hepatocellular Carcinoma: A Critical Review. Int J Mol Sci. 2023;24:4286. [PubMed] [DOI] |
| 27. | Ricco G, Cosma C, Bedogni G, Biasiolo A, Guarino M, Pontisso P, Morisco F, Oliveri F, Cavallone D, Bonino F, Plebani M, Brunetto MR. Modeling the time-related fluctuations of AFP and PIVKA-II serum levels in patients with cirrhosis undergoing surveillance for hepatocellular carcinoma. Cancer Biomark. 2020;29:189-196. [PubMed] [DOI] |
| 28. | Caviglia GP, Ciruolo M, Abate ML, Carucci P, Rolle E, Rosso C, Olivero A, Troshina G, Risso A, Nicolosi A, Ribaldone DG, Armandi A, Tandoi F, Saracco GM, Bugianesi E, Ciancio A, Gaia S. Alpha-Fetoprotein, Protein Induced by Vitamin K Absence or Antagonist II and Glypican-3 for the Detection and Prediction of Hepatocellular Carcinoma in Patients with Cirrhosis of Viral Etiology. Cancers (Basel). 2020;12:3218. [PubMed] [DOI] |
| 29. | Caviglia GP, Abate ML, Troshina G, Carucci P, Rolle E, Risso A, Burlone ME, Albè A, Crevola M, Musso EC, Rosso C, Armandi A, Olivero A, Minisini R, Saracco GM, Bugianesi E, Pirisi M, Ciancio A, Gaia S. Identification of the Best Cut-Off Value of PIVKA-II for the Surveillance of Patients at Risk of Hepatocellular Carcinoma Development. Biology (Basel). 2023;12:94. [PubMed] [DOI] |
| 30. | Su TH, Liao WC. Editorial: PIVKA-II Facilitates Risk Stratification for HCC Following HCV Cure. Aliment Pharmacol Ther. 2025;61:583-584. [PubMed] [DOI] |
| 31. | Degasperi E, Perbellini R, D'Ambrosio R, Uceda Renteria SC, Ceriotti F, Perego A, Orsini C, Borghi M, Iavarone M, Bruccoleri M, Rimondi A, De Silvestri A, Sangiovanni A, Lampertico P. Prothrombin induced by vitamin K absence or antagonist-II and alpha foetoprotein to predict development of hepatocellular carcinoma in Caucasian patients with hepatitis C-related cirrhosis treated with direct-acting antiviral agents. Aliment Pharmacol Ther. 2022;55:350-359. [PubMed] [DOI] |
| 32. | Caviglia GP, Fariselli P, D'Ambrosio R, Colombatto P, Degasperi E, Ricco G, Abate ML, Birolo G, Troshina G, Damone F, Coco B, Cavallone D, Perbellini R, Monico S, Saracco GM, Brunetto MR, Lampertico P, Ciancio A. Development and Validation of a PIVKA-II-Based Model for HCC Risk Stratification in Patients With HCV-Related Cirrhosis Successfully Treated With DAA. Aliment Pharmacol Ther. 2025;61:538-549. [PubMed] [DOI] |
| 33. | Eletreby R, Elsharkawy M, Taha AA, Hassany M, Abdelazeem A, El-Kassas M, Soliman A. Evaluation of GALAD Score in Diagnosis and Follow-up of Hepatocellular Carcinoma after Local Ablative Therapy. J Clin Transl Hepatol. 2023;11:334-340. [PubMed] [DOI] |
| 34. | Nartey YA, Yang JD, Zemla TJ, Ayawin J, Asibey SO, El-Kassas M, Bampoh SA, Duah A, Agyei-Nkansah A, Awuku YA, Afihene MY, Yamada H, Yin J, Plymoth A, Roberts LR. GALAD Score for the Diagnosis of Hepatocellular Carcinoma in Sub-Saharan Africa: A Validation Study in Ghanaian Patients. Cancer Res Commun. 2024;4:2653-2659. [PubMed] [DOI] |
| 35. | Singal AG, Tayob N, Mehta A, Marrero JA, El-Serag H, Jin Q, Saenz de Viteri C, Fobar A, Parikh ND. GALAD demonstrates high sensitivity for HCC surveillance in a cohort of patients with cirrhosis. Hepatology. 2022;75:541-549. [PubMed] [DOI] |
| 36. | Berhane S, Toyoda H, Tada T, Kumada T, Kagebayashi C, Satomura S, Schweitzer N, Vogel A, Manns MP, Benckert J, Berg T, Ebker M, Best J, Dechêne A, Gerken G, Schlaak JF, Weinmann A, Wörns MA, Galle P, Yeo W, Mo F, Chan SL, Reeves H, Cox T, Johnson P. Role of the GALAD and BALAD-2 Serologic Models in Diagnosis of Hepatocellular Carcinoma and Prediction of Survival in Patients. Clin Gastroenterol Hepatol. 2016;14:875-886.e6. [PubMed] [DOI] |
| 37. | Tayob N, Kanwal F, Alsarraj A, Hernaez R, El-Serag HB. The Performance of AFP, AFP-3, DCP as Biomarkers for Detection of Hepatocellular Carcinoma (HCC): A Phase 3 Biomarker Study in the United States. Clin Gastroenterol Hepatol. 2023;21:415-423.e4. [PubMed] [DOI] |
| 38. | Piratvisuth T, Hou J, Tanwandee T, Berg T, Vogel A, Trojan J, De Toni EN, Kudo M, Eiblmaier A, Klein HG, Hegel JK, Madin K, Kroeniger K, Sharma A, Chan HLY. Development and clinical validation of a novel algorithmic score (GAAD) for detecting HCC in prospective cohort studies. Hepatol Commun. 2023;7:e0317. [PubMed] [DOI] |
| 39. | Huang CF, Kroeniger K, Wang CW, Jang TY, Yeh ML, Liang PC, Wei YJ, Hsu PY, Huang CI, Hsieh MY, Lin YH, Huang JF, Dai CY, Chuang WL, Sharma A, Yu ML. Surveillance Imaging and GAAD/GALAD Scores for Detection of Hepatocellular Carcinoma in Patients with Chronic Hepatitis. J Clin Transl Hepatol. 2024;12:907-916. [PubMed] [DOI] |
| 40. | Beudeker BJB, Fu S, Balderramo D, Mattos AZ, Carrera E, Diaz J, Prieto J, Banales J, Vogel A, Arrese M, Oliveira J, Groothuismink ZMA, van Oord G, Hansen BE, de Man RA, Debes JD, Boonstra A. Validation and optimization of AFP-based biomarker panels for early HCC detection in Latin America and Europe. Hepatol Commun. 2023;7:e0264. [PubMed] [DOI] |
| 41. | Liu SY, Li C, Sun LY, Guan MC, Gu LH, Yin DX, Yao LQ, Liang L, Wang MD, Xing H, Zhu H, Pawlik TM, Lau WY, Shen F, Tong XM, Yang T. ASAP Score versus GALAD Score for detection of hepatitis C-related hepatocellular carcinoma: A multicenter case-control analysis. Front Oncol. 2022;12:1018396. [PubMed] [DOI] |
| 42. | Sun LY, Wang NY, Diao YK, Yan CL, Fan ZP, Wei LH, Li HJ, Guan MC, Wang MD, Pawlik TM, Lau WY, Shen F, Lv GY, Yang T. Comparison between models for detecting hepatocellular carcinoma in patients with chronic liver diseases of various etiologies: ASAP score versus GALAD score. Hepatobiliary Pancreat Dis Int. 2025;24:412-422. [PubMed] [DOI] |
| 43. | Ren T, Hou X, Zhang X, Chen D, Li J, Zhu Y, Liu Z, Yang D. Validation of combined AFP, AFP-L3, and PIVKA II for diagnosis and monitoring of hepatocellular carcinoma in Chinese patients. Heliyon. 2023;9:e21906. [PubMed] [DOI] |
| 44. | Liu M, Wu R, Liu X, Xu H, Chi X, Wang X, Zhan M, Wang B, Peng F, Gao X, Shi Y, Wen X, Ji Y, Jin Q, Niu J. Validation of the GALAD Model and Establishment of GAAP Model for Diagnosis of Hepatocellular Carcinoma in Chinese Patients. J Hepatocell Carcinoma. 2020;7:219-232. [PubMed] [DOI] |
| 45. | Hou J, Berg T, Vogel A, Piratvisuth T, Trojan J, De Toni EN, Kudo M, Malinowsky K, Findeisen P, Hegel JK, Schöning W, Madin K, Kroeniger K, Lik-Yuen Chan H, Sharma A. Comparative evaluation of multimarker algorithms for early-stage HCC detection in multicenter prospective studies. JHEP Rep. 2025;7:101263. [PubMed] [DOI] |
| 46. | Garay OU, Ambühl LE, Bird TG, Barnes E, Irving WL, Walkley R, Rowe IA. Cost-Effectiveness of Hepatocellular Carcinoma Surveillance Strategies in Patients With Compensated Liver Cirrhosis in the United Kingdom. Value Health. 2024;27:1698-1709. [PubMed] [DOI] |
| 47. | El-Serag HB, Jin Q, Tayob N, Salem E, Luster M, Alsarraj A, Khaderi S, Singal AG, Marrero JA, Asrani SK, Kanwal F. HES V2.0 outperforms GALAD for detection of HCC: A phase 3 biomarker study in the United States. Hepatology. 2025;81:465-475. [PubMed] [DOI] |
| 48. | Kudo M, Kawamura Y, Hasegawa K, Tateishi R, Kariyama K, Shiina S, Toyoda H, Imai Y, Hiraoka A, Ikeda M, Izumi N, Moriguchi M, Ogasawara S, Minami Y, Ueshima K, Murakami T, Miyayama S, Nakashima O, Yano H, Sakamoto M, Hatano E, Shimada M, Kokudo N, Mochida S, Takehara T. Management of Hepatocellular Carcinoma in Japan: JSH Consensus Statements and Recommendations 2021 Update. Liver Cancer. 2021;10:181-223. [PubMed] [DOI] |
| 49. | 中华人民共和国国家卫生健康委员会医政司. 原发性肝癌诊疗指南(2024年版). 肿瘤综合治疗电子杂志. 2024;10:17-68. [DOI] |
| 50. | 丁 惠国, 屠 红, 曲 春枫, 曹 广文, 庄 辉, 赵 平, 徐 小元, 杨 永平, 卢 实春. 原发性肝癌的分层筛查与监测指南(2020版). 临床肝胆病杂志. 2021;37:286-295. [DOI] |
| 51. | Li J, Jiang Z. Chinese Society of Clinical Oncology Breast Cancer (CSCO BC) guidelines in 2022: stratification and classification. Cancer Biol Med. 2022;19:769-773. [PubMed] [DOI] |
| 52. | Su TH, Wu CH, Liu TH, Ho CM, Liu CJ. Clinical practice guidelines and real-life practice in hepatocellular carcinoma: A Taiwan perspective. Clin Mol Hepatol. 2023;29:230-241. [PubMed] [DOI] |
| 53. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J Hepatol. 2025;82:315-374. [PubMed] [DOI] |
| 54. | Singal AG, Llovet JM, Yarchoan M, Mehta N, Heimbach JK, Dawson LA, Jou JH, Kulik LM, Agopian VG, Marrero JA, Mendiratta-Lala M, Brown DB, Rilling WS, Goyal L, Wei AC, Taddei TH. AASLD Practice Guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology. 2023;78:1922-1965. [PubMed] [DOI] |
| 55. | Korean Liver Cancer Association (KLCA) and National Cancer Center (NCC) Korea. 2022 KLCA-NCC Korea practice guidelines for the management of hepatocellular carcinoma. Clin Mol Hepatol. 2022;28:583-705. [PubMed] [DOI] |
| 56. | Vogel A, Chan SL, Dawson LA, Kelley RK, Llovet JM, Meyer T, Ricke J, Rimassa L, Sapisochin G, Vilgrain V, Zucman-Rossi J, Ducreux M; ESMO Guidelines Committee. Hepatocellular carcinoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2025;36:491-506. [PubMed] [DOI] |
| 57. | Suddle A, Reeves H, Hubner R, Marshall A, Rowe I, Tiniakos D, Hubscher S, Callaway M, Sharma D, See TC, Hawkins M, Ford-Dunn S, Selemani S, Meyer T. British Society of Gastroenterology guidelines for the management of hepatocellular carcinoma in adults. Gut. 2024;73:1235-1268. [PubMed] [DOI] |
| 58. | Syriha A, Pantzios S, Mandilara D, Galanis P, Stathopoulou I, Barla G, Elefsiniotis I. Diagnostic accuracy of serum protein induced by vitamin K absence (PIVKA-II), serum a-fetoprotein and their combination for hepatocellular carcinoma among Caucasian cirrhotic patients with diagnostic or non-diagnostic serum a-fetoprotein levels. Cancer Med. 2024;13:e6825. [PubMed] [DOI] |
| 59. | Guarneri V, Loggi E, Ramacieri G, Serra C, Vukotic R, Vitale G, Scuteri A, Cursaro C, Margotti M, Galli S, Caracausi M, Brodosi L, Gabrielli F, Andreone P. Diagnostic Performance of PIVKA-II in Italian Patients with Hepatocellular Carcinoma. Cancers (Basel). 2025;17:167. [PubMed] [DOI] |
| 60. | Chen Y, Yang X, Shao Y, Zhao H, Jiang J, Huang P, Lu Y, Xuan Z. Comparison of diagnostic performance of AFP, DCP and two diagnostic models in hepatocellular carcinoma: a retrospective study. Ann Hepatol. 2023;28:101099. [PubMed] [DOI] |
| 61. | Ji J, Liu L, Jiang F, Wen X, Zhang Y, Li S, Lou J, Wang Y, Liu N, Guo Q, Jia Y, Gao C. The clinical application of PIVKA-II in hepatocellular carcinoma and chronic liver diseases: A multi-center study in China. J Clin Lab Anal. 2021;35:e24013. [PubMed] [DOI] |
| 62. | Guan MC, Ouyang W, Liu SY, Sun LY, Chen WY, Tong XM, Zhu H, Yang T. Alpha-fetoprotein, protein induced by vitamin K absence or antagonist-II, lens culinaris agglutinin-reactive fraction of alpha-fetoprotein alone and in combination for early detection of hepatocellular carcinoma from nonalcoholic fatty liver disease: A multicenter analysis. Hepatobiliary Pancreat Dis Int. 2022;21:559-568. [PubMed] [DOI] |
| 63. | Qian XJ, Wen ZM, Huang XM, Feng HJ, Lin SS, Liu YN, Li SC, Zhang Y, Peng WG, Yang JR, Zheng ZY, Zhang L, Zhang DW, Lu FM, Liu LJ, Pan WD. Better performance of PIVKA-II for detecting hepatocellular carcinoma in patients with chronic liver disease with normal total bilirubin. World J Gastroenterol. 2023;29:1359-1373. [PubMed] [DOI] |
| 64. | Hadi H, Wan Shuaib WMA, Raja Ali RA, Othman H. Utility of PIVKA-II and AFP in Differentiating Hepatocellular Carcinoma from Non-Malignant High-Risk Patients. Medicina (Kaunas). 2022;58:1015. [PubMed] [DOI] |
| 65. | Tian S, Chen Y, Zhang Y, Xu X. Clinical value of serum AFP and PIVKA-II for diagnosis, treatment and prognosis of hepatocellular carcinoma. J Clin Lab Anal. 2023;37:e24823. [PubMed] [DOI] |
| 66. | Caviglia GP, Armandi A, Rosso C, Gaia S, Aneli S, Rolle E, Abate ML, Olivero A, Nicolosi A, Guariglia M, Ribaldone DG, Carucci P, Saracco GM, Bugianesi E. Biomarkers of Oncogenesis, Adipose Tissue Dysfunction and Systemic Inflammation for the Detection of Hepatocellular Carcinoma in Patients with Nonalcoholic Fatty Liver Disease. Cancers (Basel). 2021;13:2305. [PubMed] [DOI] |
| 67. | Jin K, Wu J, Yang J, Chen B, Xu J, Bao H, Zong W, Xie C, Chen L, Wang F. Identification of serum tsRNA-Thr-5-0015 and combined with AFP and PIVKA-II as novel biomarkers for hepatocellular carcinoma. Sci Rep. 2024;14:28834. [PubMed] [DOI] |
| 68. | Xing X, Cai L, Ouyang J, Wang F, Li Z, Liu M, Wang Y, Zhou Y, Hu E, Huang C, Wu L, Liu J, Liu X. Proteomics-driven noninvasive screening of circulating serum protein panels for the early diagnosis of hepatocellular carcinoma. Nat Commun. 2023;14:8392. [PubMed] [DOI] |
| 69. | Wang F, Xu Y, Chen Y, Wang X, Xu Q, Xia H, Yuan K, Dong Y, Liu L, Wang W. Patients with hepatocellular carcinoma with triple-positive tumor markers may benefit from postsurgery adjuvant treatment. J Gastrointest Surg. 2025;29:102075. [PubMed] [DOI] |
| 70. | Wu M, Yu H, Pang S, Liu A, Liu J. Application of CT-based radiomics combined with laboratory tests such as AFP and PIVKA-II in preoperative prediction of pathologic grade of hepatocellular carcinoma. BMC Med Imaging. 2025;25:51. [PubMed] [DOI] |
| 71. | Wang MD, Wang BD, Diao YK, Li C, Yao LQ, Liu H, Zeng YY, Chen Z, Wu H, Xu XF, Gu LH, Xu JH, Yin DX, Li YC, Chen FJ, Kow AWC, Pawlik TM, Shen F, Yang T. Tumor biology characteristics score based on alpha-fetoprotein and protein induced by vitamin K absence or antagonist II as a predictor for recurrence and survival after curative resection for hepatocellular carcinoma: a multicenter cohort study. J Gastrointest Surg. 2025;29:102038. [PubMed] [DOI] |
| 72. | 刘 子鑫, 闫 祖仪, 张 涛, 张 学琴, 顾 春燕, 瞿 琦, 姜 吉锋. 基于多序列MRI的多区域影像组学评分预测血管包绕肿瘤细胞簇和/或微血管侵犯阳性肝细胞癌. 中华肝胆外科杂志. 2024;30:886-892. [DOI] |
| 73. | 尹 东旭, 刁 永康, 王 明达, 李 超, 姚 岚清, 顾 丽慧, 徐 家豪, 徐 新飞, 沈 锋, 杨 田. 术前PIVKA-II水平对肝细胞癌患者行根治性切除术后生存和复发的预测价值. 肝胆胰外科杂志. 2024;36:709-715. [DOI] |
| 74. | Liu SY, Dai MG, Lu WF, Liang L, Liu JW, Li ZK, Ye B. Prognostic Value of Des-Gamma-Carboxy Prothrombin in AFP-Negative Hepatocellular Carcinoma Patients Following Liver Resection: A Multicenter Study. J Cancer. 2025;16:2680-2689. [PubMed] [DOI] |
| 75. | Wang M, Qian G, Xiao H, Liu X, Sun L, Chen Z, Lin K, Yao L, Li C, Gu L, Xu J, Sun X, Qiu W, Pawlik TM, Yee Lau W, Lv G, Shen F, Yang T. Prognostic significance of postoperative serological incomplete conversion of AFP and PIVKA-II after hepatic resection for hepatocellular carcinoma: a multicenter analysis of 1755 patients. Oncologist. 2024;29:e1723-e1733. [PubMed] [DOI] |
| 76. | Shimagaki T, Yoshizumi T, Itoh S, Iseda N, Tomiyama T, Morinaga A, Wang H, Kurihara T, Nagao Y, Toshima T, Harada N, Kinjo N, Maeda T, Mori M. The ratio of serum des-gamma-carboxy prothrombin to tumor volume as a new biomarker for early recurrence of resected hepatocellular carcinoma. Hepatol Res. 2022;52:381-389. [PubMed] [DOI] |
| 77. | Yanagaki M, Shirai Y, Hamura R, Taniai T, Tanji Y, Haruki K, Furukawa K, Onda S, Shiba H, Ikegami T. Novel combined fibrosis-based index predicts the long-term outcomes of hepatocellular carcinoma after hepatic resection. Int J Clin Oncol. 2022;27:717-728. [PubMed] [DOI] |
| 78. | Sun L, Gu M, Cai J, Yang W, Pan B, Wang B, Zhang C, Guo W. Combining γ-GT, PIVKA-II, and AFP to predict long-term prognosis in patients with hepatocellular carcinoma after hepatectomy. Clin Exp Pharmacol Physiol. 2023;50:287-297. [PubMed] [DOI] |
| 79. | Yao CC, Wang JH, Chen CH, Hung CH, Yen YH, Kee KM, Hu TH, Tsai MC, Kuo YH, Lu SN. Short Half-Life of Des-γ-Carboxy Prothrombin Is a Superior Factor for Early Prediction of Outcomes of Hepatocellular Carcinoma Treated with Radiofrequency Ablation. Diagnostics (Basel). 2023;13:696. [PubMed] [DOI] |
| 80. | He YZ, He K, Huang RQ, Liu LW, Ye SW, Qian JL, Peng P, Luo QJ, Wang ZL, Hu ZM. A clinical scoring system for predicting tumor recurrence after percutaneous radiofrequency ablation for 3 cm or less hepatocellular carcinoma. Sci Rep. 2021;11:8275. [PubMed] [DOI] |
| 81. | Zhu W, Wang W, Zheng W, Chen X, Wang X, Xie J, Jiang S, Chen H, Zhu S, Xue P, Jiang X, Li H, Wang G. Diagnostic performance of PIVKA-II in identifying recurrent hepatocellular carcinoma following curative resection: a retrospective cohort study. Sci Rep. 2024;14:8416. [PubMed] [DOI] |
| 82. | Pan X, Zhou Y, Li Z, Guo P, Zeng J, Dong X, Hu E, Qiu L, Cai Z, Chen G, Liu X. Des-γ-carboxy Prothrombin in hepatocellular carcinoma post-operative recurrence risk evaluation. Commun Med (Lond). 2025;5:65. [PubMed] [DOI] |
| 83. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on liver transplantation. J Hepatol. 2024;81:1040-1086. [PubMed] [DOI] |
| 84. | Kotwani P, Chan W, Yao F, Mehta N. DCP and AFP-L3 Are Complementary to AFP in Predicting High-Risk Explant Features: Results of a Prospective Study. Clin Gastroenterol Hepatol. 2022;20:701-703.e2. [PubMed] [DOI] |
| 85. | Wang K, Dong L, Lu Q, Yang Z, Fan X, Gao F, Ge W, Wang Z, Zhou Z, Lu D, Wei X, Wei Q, Zhuang L, Qin L, Ye Q, Yang J, Dong J, Zheng S, Xu X. Incorporation of protein induced by vitamin K absence or antagonist-II into transplant criteria expands beneficiaries of liver transplantation for hepatocellular carcinoma: a multicenter retrospective cohort study in China. Int J Surg. 2023;109:4135-4144. [PubMed] [DOI] |
| 86. | Bhatti ABH, Shafique U, Ahmed N, Abbas G, Atiq M, Zia HH, Khan NY, Rana A. Prothrombin-induced by vitamin K absence II as a prognostic factor in living donor liver transplantation for hepatocellular carcinoma. Sci Rep. 2025;15:21900. [PubMed] [DOI] |
| 87. | 吴 广东, 汤 睿, 李 昂, 童 翾, 于 里涵, 侯 昱丞, 阿卜杜萨拉木· 艾尼, 杨 威, 郝 华媛, 林 景熠, 卢 倩. 超UCSF标准且无大血管侵犯的肝细胞癌肝移植的预后危险因素分析. 中华普通外科杂志. 2024;39:339-343. [DOI] |
| 88. | Norman JS, Li PJ, Kotwani P, Shui AM, Yao F, Mehta N. AFP-L3 and DCP strongly predict early hepatocellular carcinoma recurrence after liver transplantation. J Hepatol. 2023;79:1469-1477. [PubMed] [DOI] |
| 89. | Yang M, Zhang X, Liu J. Prognostic value of des-γ-carboxy prothrombin in patients with hepatocellular carcinoma treated with transarterial chemotherapy: A systematic review and meta-analysis. PLoS One. 2019;14:e0225170. [PubMed] [DOI] |
| 90. | Lu SY, Sun HY, Zhou Y, Luo X, Liu S, Zhou WZ, Shi HB, Yang W, Tian W. Prognosis of Patients with Hepatocellular Carcinoma Treated with TACE: A New Score Combining Alpha-Fetoprotein and Des-γ-Carboxy Prothrombin. J Hepatocell Carcinoma. 2024;11:1979-1992. [PubMed] [DOI] |
| 91. | Wang SY, Su TH, Chen BB, Liu CJ, Liu CH, Yang HC, Tseng TC, Chen PJ, Kao JH. Prothrombin induced by vitamin K absence or antagonist-II (PIVKA-II) predicts complete responses of transarterial chemoembolization for hepatocellular carcinoma. J Formos Med Assoc. 2022;121:1579-1587. [PubMed] [DOI] |
| 93. | Xie H, Li Y, Yang J, Tan Y, Xu J, Yang X. DCP as a biomarker for TACE efficacy in hepatocellular carcinoma. Front Oncol. 2025;15:1560210. [PubMed] [DOI] |
| 94. | Haruna Y, Yakushijin T, Yamakawa M, Nakazawa T. Anticancer effects of vitamin K combined with transarterial chemoembolization in hepatocellular carcinoma, a randomized controlled trial. Br J Cancer. 2025;132:1141-1147. [PubMed] |
| 95. | Tanaka K, Tsuji K, Hiraoka A, Tada T, Hirooka M, Kariyama K, Tani J, Atsukawa M, Takaguchi K, Itobayashi E, Fukunishi S, Ishikawa T, Tajiri K, Ochi H, Toyoda H, Koshiyama Y, Ogawa C, Nishikawa H, Nishimura T, Hatanaka T, Kakizaki S, Noritake H, Kawata K, Naganuma A, Kosaka H, Matsui K, Matono T, Kuroda H, Yata Y, Tanaka H, Aoki T, Tamai H, Tada F, Ohama H, Kanayama Y, Nouso K, Morishita A, Tsutsui A, Nagano T, Itokawa N, Okubo T, Arai T, Yoshida O, Imai M, Nakamura S, Enomoto H, Kaibori M, Kudo M, Hiasa Y, Kumada T; Real-life Practice Experts for HCC (RELPEC) Study Group; HCC 48 Group (hepatocellular carcinoma experts from 48 clinics in Japan). Alpha-Fetoprotein and Des-Gamma-Carboxy Prothrombin-Based Tumor Marker Score for First-Line Immunotherapy Selection in Hepatocellular Carcinoma. Liver Cancer. 2025. [PubMed] [DOI] |
| 96. | Saeki I, Shimose S, Tomonari T, Ito T, Tani J, Takeuchi Y, Yoshioka N, Naito T, Takeuchi M, Kakizaki S, Hatanaka T, Sasaki K, Yasunaka T, Sakata M, Iwamoto H, Itano S, Shirono T, Tanabe N, Yamamoto T, Kanayama Y, Naganuma A, Nishina S, Otsuka M, Kobara H, Kawashima H, Takayama T, Kawaguchi T, Yamasaki T, Takami T; Hepatology InVestigator Experts in Japan (HIVE-J) Study Group. Alpha-fetoprotein and des-gamma-carboxy prothrombin can predict the objective response of patients with hepatocellular carcinoma receiving durvalumab plus tremelimumab therapy. PLoS One. 2024;19:e0311084. [PubMed] [DOI] |
| 97. | Kuzuya T, Kawabe N, Muto H, Wada Y, Komura G, Nakano T, Tanaka H, Nakaoka K, Ohno E, Funasaka K, Nagasaka M, Miyahara R, Hirooka Y. Early Changes in Alpha-Fetoprotein and Des-γ-Carboxy Prothrombin Are Useful Predictors of Antitumor Response to Durvalumab Plus Tremelimumab Therapy for Advanced Hepatocellular Carcinoma. Curr Oncol. 2024;31:4225-4240. [PubMed] [DOI] |
| 98. | Fang ZK, Xiao YT, Feng X, Shi ZJ, Liu SY, Yu Y, Jin LM, Huang DS, Zhang CW, Liu JW, Liang L. Early PIVKA-II Response Associated with Treatment Efficacy and Survival Outcomes for Patients with Advanced Hepatocellular Carcinoma Receiving Immune Checkpoint Inhibitors and Targeted Therapy. J Hepatocell Carcinoma. 2025;12:2235-2246. [PubMed] [DOI] |
| 99. | Guo DZ, Zhang SY, Dong SY, Yan JY, Wang YP, Cao Y, Rao SX, Fan J, Yang XR, Huang A, Zhou J. Prognostic model for predicting outcome and guiding treatment decision for unresectable hepatocellular carcinoma treated with lenvatinib monotherapy or lenvatinib plus immunotherapy. Front Immunol. 2023;14:1141199. [PubMed] [DOI] |
| 100. | Zhang T, Li W, Chen Q, He W, Sun J, Li D, Wang Q, Duan X. Prognostic significance of early alpha fetoprotein and des-gamma carboxy prothrombin responses in unresectable hepatocellular carcinoma patients undergoing triple combination therapy. Front Immunol. 2024;15:1508028. [PubMed] [DOI] |
| 101. | Haruna Y, Yakushijin T, Kawamoto S. Efficacy and safety of sorafenib plus vitamin K treatment for hepatocellular carcinoma: A phase II, randomized study. Cancer Med. 2021;10:914-922. [PubMed] [DOI] |
学科分类: 胃肠病学和肝病学
手稿来源地: 陕西省
同行评议报告学术质量分类
A级 (优秀): A
B级 (非常好): B
C级 (良好): C
D级 (一般): 0
E级 (差): 0
科学编辑: 刘继红 制作编辑:张砚梁