Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.115131
Revised: November 11, 2025
Accepted: January 8, 2026
Published online: March 7, 2026
Processing time: 139 Days and 17.1 Hours
Pancreatic cancer, a highly malignant gastrointestinal tumor, has a five-year survival rate of only 10%. With the increasing aging population, its incidence is rising across East Asia, North America, and Europe. Chemotherapy remains a basic strategy of the current treatment regimen for pancreatic cancer. However, the development of multidrug resistance poses a significant challenge, drastically reducing the efficacy of chemotherapy agents. The mechanisms underlying resis
Core Tip: This review summarizes recent research on the role of plant-derived compounds in overcoming drug resistance in pancreatic cancer. It provides insights into the mechanisms of resistance and highlights the potential of plant-based compounds as alternative therapeutic strategies. Considering the limitations of current therapies and the growing issue of drug resistance, plant-derived compounds offer a promising direction for enhancing treatment outcomes. This review aims to inform future research and contribute to the development of more effective, safer, and patient-friendly treatment options for pancreatic cancer.
- Citation: Yu JQ, Yu SJ, He YH, Xue YZ, Yu XF, Chen W, Hu LY, Fan XF, Gao ZF, Zhou HK, Liu XR, He XS, Wang XG. Research progress of plant-derived chemical compounds for overcoming pancreatic cancer drug resistance. World J Gastroenterol 2026; 32(9): 115131
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/115131.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.115131
Pancreatic cancer is a highly lethal malignancy that has become the seventh leading cause of cancer-related deaths globally. It is expected that by 2030, pancreatic cancer will become the second leading cause of cancer-related deaths, only behind lung cancer[1]. North America, Europe, and Australia are regions with a high incidence of pancreatic cancer. Over the past 20 years, the number of diagnoses of pancreatic cancer has increased from 196000 cases in 1990 to 441000 cases in 2017, representing a twofold increase[2], because of the insidious nature of pancreatic cancer, more than 80% of diagnoses are made at an advanced stage, resulting in a five-year survival rate of only 11%[3]. It is widely recognized that smoking, alcohol abuse, diabetes, obesity, aging, family history, and genetics are high-risk factors influencing the incidence of pancreatic cancer[4]. In countries with a higher development index and an aging population (where the elderly population aged 65 and above exceeds 7%), there are generally more accurate medical examination tools, leading to a higher absolute number of cases of pancreatic cancer. In contrast, developing countries, especially in Africa, face challenges in introducing advanced imaging and accurate pathological diagnostic methods. This makes high-quality diagnosis of pancreatic cancer a luxury, and as a result, some global data on pancreatic cancer cases may be missing[5]. Although the absolute number of cases is high, patients in developed regions with well-established treatment systems tend to have better overall survival (OS) than patients in economically underdeveloped regions. Currently, the mainstream treatment involves R0 resection surgery combined with postoperative chemotherapy, with gemcitabine + nab-paclitaxel (AG) and fluorouracil, leucovorin, irinotecan, and oxaliplatin (FOLFIRINOX) regimens being the primary chemotherapy options. However, due to the highly resistant nature of pancreatic cancer, the benefits of chemotherapy regimens are suboptimal. As a result, scientists have made significant efforts to identify various resistance mechanisms in pancreatic cancer. On the basis of these mechanisms, new therapeutic approaches, such as neoadjuvant therapy, targeted immunotherapy, and cancer vaccines, have been explored. Many scientists believe that plant-derived compounds offer advantages, including the reduction of anticancer drug toxicity, having fewer side effects, and being cost-effective, making them a promising addition to cancer treatment regimens. Numerous studies have confirmed that medicinal plants and their active compounds help control diseases and inhibit carcinogenesis. Furthermore, active compounds from natural products or herbs have shown substantial effects in enhancing the efficacy of anticancer drugs while reducing their toxicity. Novel drug combinations consisting of traditional chemotherapy regimens and natural products are expected to achieve better outcomes than conventional chemotherapy drugs, while reducing adverse reactions[6].
The mainstream treatment regimen for pancreatic cancer remains surgery followed by postoperative chemotherapy. Due to the insidious nature of pancreatic cancer, only a small proportion of patients are eligible for surgical treatment. The median survival time is approximately 10-12 months for patients who undergo diagnostic evaluation and receive treatment, whereas untreated patients typically survive for only 5-6 months. The standard first-line treatment regimens are mFOLFIRINOX [a combination of 5-fluorouracil (5-FU), leucovorin, irinotecan, and oxaliplatin] or AG[7-10]. No other combination has demonstrated both superior survival benefits and reduced treatment toxicity compared with these two regimens. A recent study demonstrated that the modified version of the mFOLFIRINOX regimen, when used as adjuvant therapy, yields significantly longer survival compared to gemcitabine alone. The median disease-free survival in the modified mFOLFIRINOX group was significantly higher than in the gemcitabine group (21.6 months vs 12.8 months). However, the modified mFOLFIRINOX regimen is associated with greater toxicity, despite improvements compared to the traditional FOLFIRINOX regimen[11]. As a result, gemcitabine remains the most commonly used chemotherapy drug for pancreatic cancer in clinical practice. Unfortunately, approximately 80% of patients relapse and die within a year[12].
The disappointing outcomes are primarily attributed to factors such as inadequate lymph node dissection during surgery, undetected micro-metastases, chemotherapy drug resistance, and poor efficacy of postoperative adjuvant therapies, which result in suboptimal clinical outcomes. Given the low rate of complete R0 resection during surgery and the high likelihood of recurrence within a year after surgery, chemotherapy is generally considered the mainstream treatment for pancreatic cancer. Drug resistance remains a major factor influencing the effectiveness of chemotherapy regimens, with gemcitabine, the first-line chemotherapy drug for pancreatic cancer, being particularly affected. The specific mechanisms of resistance to gemcitabine are still a subject of ongoing debate.
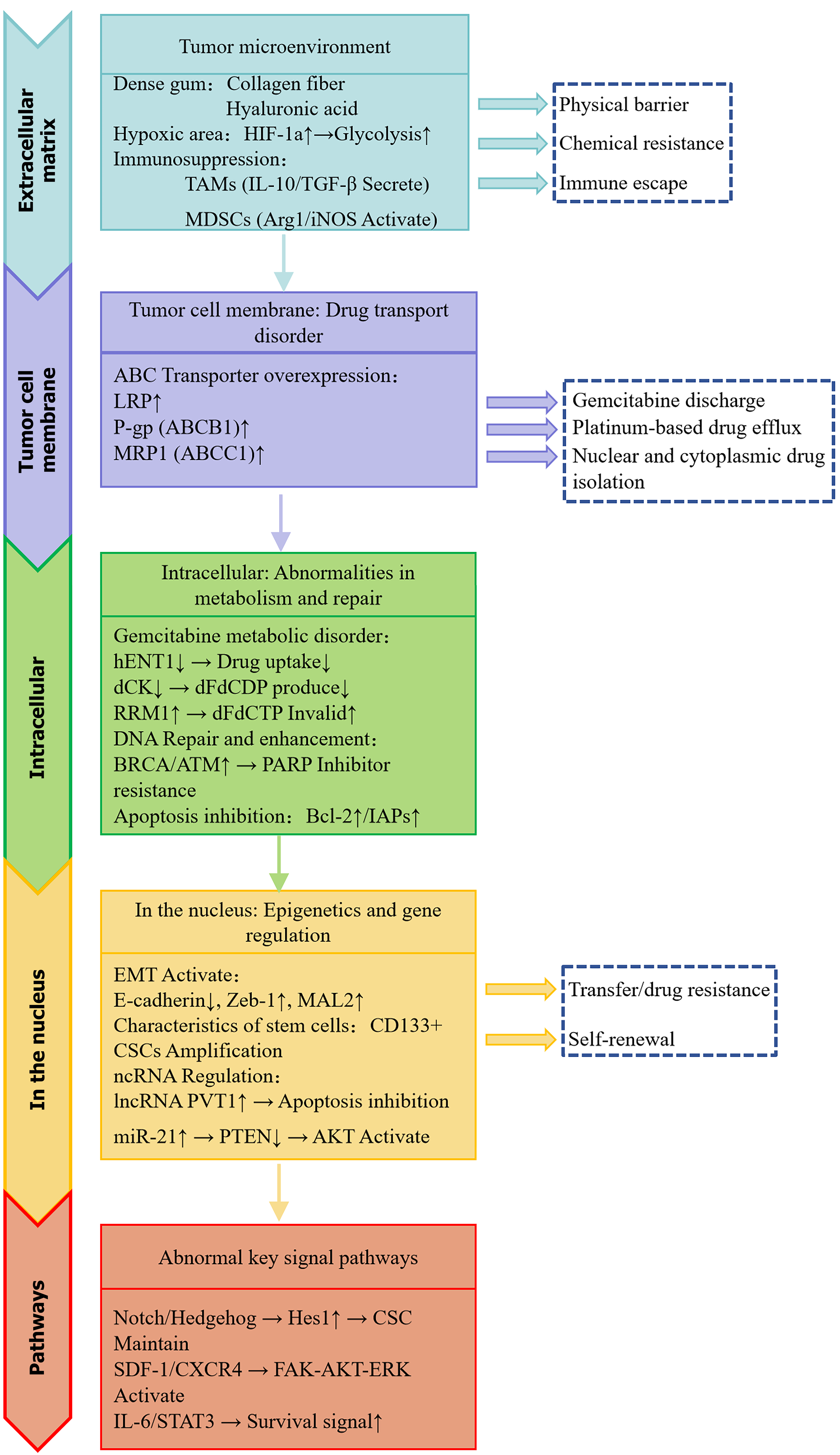
This review summarizes recent advances in understanding the mechanisms of drug resistance, including the following aspects: Drug transport disorders, key intracellular enzymes, epithelial-mesenchymal transition (EMT), the tumor microenvironment (TME), abnormal expression of resistance proteins, and the regulation of potential target genes and signaling pathways by non-coding RNAs (ncRNAs). Based on literature review, it is undeniable that pancreatic cancer cells exhibit higher resistance to gemcitabine compared to other chemotherapy drugs.
Chemotherapy drugs require nucleoside transporters (NTs) to pass through the plasma membrane and exert their effects[13]. Some studies have demonstrated that reducing human equilibrative nucleoside transporter 1 (hENT1) expression results in decreased cellular uptake of gemcitabine[14-18]. Patients with low expression of NTs have significantly shorter median survival times[19-24], it is inferred that a lack of hENT1 can lead to significant gemcitabine resistance[18]. Bhutia et al[25] showed that pharmacological inhibition of human concentrative nucleoside transporter 1 (hCNT1) degradation modestly increased the surface expression of hCNT1 in MIA PaCa-2 cells, and the cellular transport of gemcitabine. Constitutive expression of hCNT1 reduced the clonogenic survival of MIA PaCa-2 cells and significantly enhanced gemcitabine transport and chemoresistance[25].
Once chemotherapy drugs enter the cell via NTs, two nucleotidases play essential roles in deoxycytidine kinase (dCK) and ribonucleotide reductase (RR). dCK is the primary rate-limiting enzyme for the metabolism and activation of chemotherapy drugs within the cell[26]. Knockdown of dCK can induce drug resistance in pancreatic cancer[27]. Moreover, overexpression of dCK can restore drug sensitivity in resistant strains[28,29]. RR, another rate-limiting enzyme in the DNA synthesis pathway, primarily converts ribonucleotides into deoxyribonucleotide triphosphates, which are crucial for DNA replication and repair. RR consists of two subunits, M1 and M2[30]. The inhibition of RR by gemcitabine diphosphate (dFdCDP) activates gemcitabine[31,32]. Recent meta-analyses have shown that patients with low expression of RR subunit 1 (RRM1) have better OS and disease-free survival than those with high RRM1 expression[33-36]. This suggests that RRM1 expression is an indicator of poor survival in pancreatic cancer patients undergoing chemotherapy. Wang et al[37] and Nakahira et al[38] have experimentally demonstrated that overexpression of RRM1 and RRM2 proteins in pancreatic cancer cell lines can induce stable genetic resistance. Hence, both dCK and RR are key factors influencing drug resistance.
EMT is a phase of tumor cell phenotype transformation that facilitates the acquisition of a more invasive mesenchymal phenotype, often accompanied by morphological changes in cancer cells and alterations in genomic and proteomic levels[39]. The role of mesenchymal transcription factors in chemotherapy resistance has garnered increasing attention in recent years[40]. EMT is mediated by various key genes and signaling pathways. E-cadherin is strongly negatively correlated with zinc-finger E-box binding homeobox 1 (Zeb-1) and is closely related to resistance. Zeb-1 and other EMT regulators are implicated in maintaining drug resistance in human pancreatic cancer cells[41]. Silencing Zeb-1 upregulates E-cadherin expression and restores drug sensitivity by increasing the expression of epithelial markers such as epithelial-V-like antigen 1 and myelin and lymphocyte protein 2[42]. Signaling pathways, such as Notch, tumor necrosis factor α, transforming growth factor β, and hypoxia-inducible factor-1α (HIF-1α) pathways, are involved in EMT induction in pancreatic cancer cells[43]. Silencing Zeb-1 reverses EMT, restores the expression of epithelial marker genes, and enhances sensitivity to therapeutic agents, suggesting that Zeb-1 and other EMT regulators are involved in maintaining drug resistance in human pancreatic cancer cells.
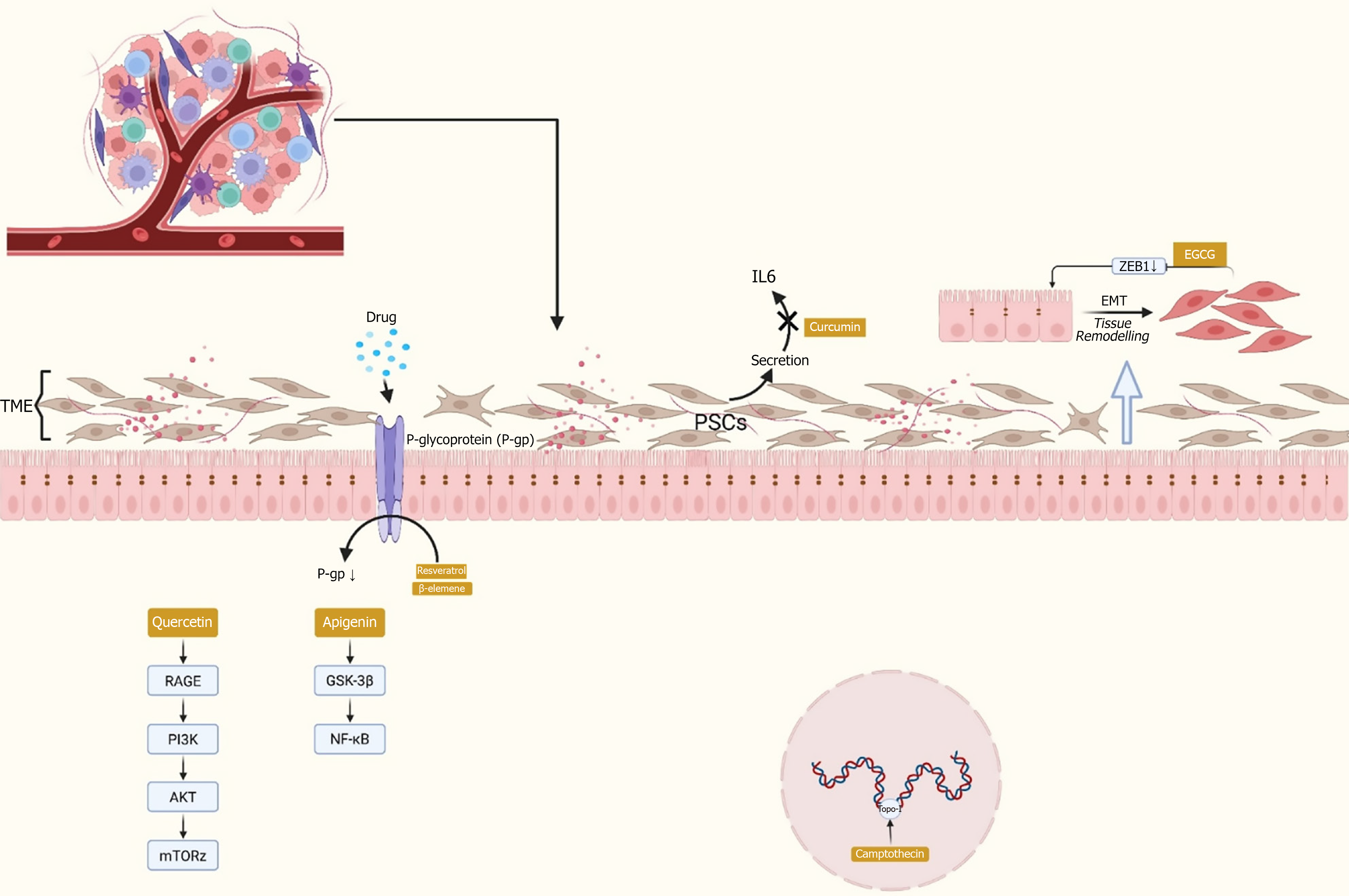
The TME encompasses blood vessels, immune cells, fibroblasts, signaling molecules, and the extracellular matrix[44]. Tumors can influence the microenvironment by releasing extracellular signals, promoting tumor angiogenesis, and inducing peripheral immune tolerance[45]. The microenvironment, in turn, can affect the efficacy of gemcitabine in treating cancer cells. The pancreatic cancer TME comprises pancreatic stellate cells (PSCs), fibroblasts, inflammatory cells, endothelial cells, and nerve cells[46,47]. These components contribute to drug resistance in pancreatic cancer[48-50]. Co-culture experiments confirmed that PSCs can enhance chemotherapy resistance to gemcitabine through the Notch signaling pathway by increasing Hes1 expression[51]. Previous studies have demonstrated that Notch signaling inhibitors or silencing Hes1 expression can reverse chemoresistance induced by PSCs[49]. Liu et al[50] discovered that periostin is overexpressed only in the stroma of pancreatic ductal adenocarcinoma (PDAC) and PSCs, suggesting its role in conferring resistance to gemcitabine. Zhang et al[52] demonstrated that PSCs activate the focal adhesion kinase-protein kinase B (AKT) and extracellular signal-regulated kinase 1/2 pathways and secrete interleukin-6 through the stromal cell-derived factor-1α/C-X-C receptor 4 axis, thereby promoting gemcitabine resistance. Emerging evidence indicates that aberrant activation of Hedgehog signaling is associated with tumor malignancy and drug resistance[53-55]. In vitro experiments have demonstrated that Hedgehog signaling promotes cancer progression by regulating cancer cell proliferation, malignancy, metastasis, and the expansion of cancer stem cells (CSCs)[56-59]. Hedgehog activation increases the stroma in the pancreatic cancer microenvironment, and its inhibition can reduce α-smooth muscle actin levels. Reduction of the stroma component can increase mean vascular density, leading to improved blood perfusion and drug delivery[60]. Blocking Hedgehog signaling with cyclopamine has been shown to reduce PDAC proliferation and reverse drug resistance[61]. Further exploration in mouse models suggests that inhibition of Hedgehog signaling leads to transient increases in tumor vasculature and gemcitabine concentration, resulting in temporary disease stabilization[60]. Resveratrol reprograms tumor-associated macrophages by inhibiting the signal transducer and activator of transcription 3 signaling pathway, thereby reducing the release of pro-tumorigenic factors and enhancing the penetration of chemotherapeutic agents[62]. Quercetin has been shown to promote the migration of dendritic cells to lymph nodes via the Toll-like receptor 4/receptor for advanced glycation end-products (RAGE) pathway, which subsequently initiates T cell responses and enhances the efficacy of chemotherapeutic drugs[63]. A considerable number of phytochemicals have a significant effect on remodeling the TME, which should not be underestimated.
Abnormal expression of transport proteins: Members of the ATP-binding cassette (ABC) transporter superfamily catalyze the translocation of substrates across cellular membranes and are widely expressed in all human tissues[64,65]. Multiple transporters are involved in resistance and disease pathways related to transporter dysfunction. Among them, ABCB1 is a multi-specific multidrug efflux pump involved in cellular detoxification, and cancer cells can exploit it to develop multidrug resistance (MDR) in response to chemotherapy. ABCG1 transports cholesterol and sphingolipids, playing a role in the reversal of cholesterol transport from peripheral tissues[66,67]. ABCG2 is a highly physiologically important multidrug efflux pump. Together with ABCB1, ABCG2 forms a pair of drug-resistant transmembrane transporters that act as gatekeepers in the blood-brain barrier. ABCG2 also transports various drugs and influence their pharmacokinetics. It plays a role in the transport of sulfated steroids and urate, and is reported to cause MDR in various cancer cells[68-70].
High expression of P-glycoprotein: P-glycoprotein (P-gp), encoded by the MDR1 gene, is a 1280-amino acid single-chain protein that acts as an efflux transporter, influencing the absorption, distribution, and elimination of various chemotherapy compounds. The exact mechanism of P-gp resistance remains unclear. It is generally believed that P-gp utilizes ATP hydrolysis to actively efflux drugs from cells, thereby reducing the intracellular drug concentration below the effective killing threshold. Some researchers have reported that P-gp expression is significantly higher in pancreatic cancer cells than in normal pancreatic tissue, which may contribute to the low efficacy of chemotherapy and the development of resistance[71].
Elevated expression of lung resistance-related protein: Lung resistance-related protein (LRP), initially known as LRP, was first identified by Scheper in 1993 in a non-small cell lung cancer cell line (SW-1573) that lacked P-gp expression. The LRP gene is located on chromosome 16, near the genes encoding MDR-associated proteins and protein kinase C-beta, and may mediate resistance through a transport mechanism[72]. LRP exhibits tissue-specific expression patterns and is widely detected in normal human tissues and tumor cells. The exact mechanism by which LRP contributes to resistance remains unclear. Some researchers believe that LRP may cause MDR by: (1) Preventing drugs from entering the cell nucleus through nuclear pores, or if drugs do enter, pumping them out before they can exert their effect; and (2) Facilitating the entry of drugs into vesicles in the cytoplasm, from where they are eventually excreted by exocytosis[72,73]. Other researchers suggest that LRP may not directly contribute to tumor MDR but works synergistically with multidrug resistance-associated protein in mediating tumor resistance[74]. Therefore, we believe that the increased expression of LRP may be associated with resistance to pancreatic cancer.
Low expression of topoisomerase II: Topoisomerases (Topo) are enzymes that correct DNA supercoiling by cleaving and rejoining the phosphodiester bonds in one or both strands of DNA. Topo II acts on both strands of DNA and usually requires ATP as an energy cofactor. Recent studies have shown that changes in Topo II activity and function are involved in mediating MDR or alternative mechanisms of resistance[75]. Research has shown that the expression of Topo II in pancreatic cancer cells decreases, which may be linked to the development of drug resistance[76].
Upregulated expression of glutathione S-transferases: Glutathione is the most important low-molecular-weight antioxidant synthesized in cells. Glutathione S-transferases (GSTs) catalyze the initial step in the glutathione conjugation reaction and are key enzymes in this process, existing in various forms and primarily localized in the cytoplasm. GSTs are overexpressed in multiple types of tumors, including pancreatic, ovarian, non-small cell lung, breast, colon, and lymphoma[77]. Studies have shown that GSTs protect cells from damage caused by exposure to both endogenous and exogenous electrophilic substances, exhibiting mutagenic and antitumor properties. They are highly expressed in tumor tissues and are associated with tumor resistance. In-depth research on GSTs lays the foundation for their application in cell protection and antitumor therapy in Table 1[64-92].
| Location | Expression trend | Primary function in drug resistance | Ref. | |
| ABC | Barrier-protected tissues (gut, blood-brain barrier, liver, kidney) | Upregulated | Pump hydrophobic drugs out of cells to reduce intracellular concentration and transport anionic compounds such as topoisomerase inhibitors | [64-70,79,80] |
| P-gp | Intestinal epithelium, liver, kidney, hematopoietic system | Upregulated | ATP-dependent efflux of chemotherapeutic agents, reducing their cytotoxic efficacy | [71,81-84] |
| LRP | Excretory and immune tissues | Upregulated | Mediates active exclusion of drugs via nuclear export and compartmentalization mechanisms | [72-74,85-87] |
| Topo II | Highly proliferative tissues (hematopoietic system, intestinal crypts, germ cells), with Topo IIα dominant in cycling cells and Topo IIβ in post-mitotic cells (e.g., neurons) | Downregulated | Alterations in Topo II expression levels or activity downregulation of expression: Tumor cells reduce the expression of Topo IIα (the primary target of chemotherapeutic agents), reducing the number of available drug-binding sites. Topo II gene mutations: Mutations within drug-binding domains (e.g., the ATP- or DNA-binding regions) can impair the affinity between the enzyme and its inhibitors | [75,76,88,89] |
| GSTs | Detoxifying organs (liver, kidney) | Upregulated | Conjugate GSH with electrophilic compounds, reducing drug-induced oxidative stress | [77,78,90-92] |
ncRNAs are a class of RNAs that lack protein-coding potential and were once considered non-essential components of the genome. However, increasingly diverse ncRNAs, including microRNAs (miRNAs), long ncRNAs (lncRNAs), and circular RNAs (circRNAs)[93], regulate tumor progression through chromatin remodeling, interactions with cytoplasmic mRNA, and the formation of competitive endogenous RNA networks[94,95]. In addition to their roles in cell proliferation, invasion, migration, and apoptosis, ncRNAs also coordinate metabolic reprogramming, such as glycolysis, mitochondrial function, glutamine, and lipid metabolism[94], thereby driving drug resistance in various types of cancer, including prostate cancer and breast cancer[96,97]. Therefore, ncRNAs have increasingly become recognized as important therapeutic targets and potential biomarkers in cancer treatment[93,97]. A growing body of evidence suggests that ncRNAs play a key role in regulating the chemosensitivity of pancreatic cancer to gemcitabine. Different ncRNAs can affect drug uptake, tumor cell cycle, apoptosis, and EMT by regulating the expression of potential target genes and related signaling pathways, thereby participating in pancreatic cancer resistance[98]. The following section summarizes relevant studies conducted over the past five years.
miRNA and chemoresistance in pancreatic cancer: Recent studies have shown that miRNA families play a significant role in the chemotherapy resistance process of pancreatic cancer. Overexpression of miR-210 enhances drug resistance by antagonizing gemcitabine-induced cell cycle arrest[99]. Elevated expression of miR-30a-3p suppresses intercellular communication by downregulating connexin 43, thereby hindering the intercellular transfer of cytotoxic agents such as gemcitabine through gap junctions and ultimately promoting drug resistance[100]. miR-146a-5p targets the 3’-untranslated region of tumor necrosis factor receptor-associated factor 6 and also downregulates the tumor necrosis factor receptor-associated factor 6/nuclear factor kappa B (NF-κB)-p65/P-gp axis, which collectively suppresses pancreatic cancer cell proliferation and promotes resistance[101]. Functional studies have shown that gemcitabine causes pancreatic cancer cells to downregulate miR-21 expression and upregulate FasL expression. After gemcitabine treatment, the increase in FasL expression induces apoptosis, while ectopic expression of miR-21 partially protects cancer cells from gemcitabine-induced apoptosis, confirming that FasL is a direct target of miR-21. Therefore, elevated serum levels of miR-21 have been proposed as a potential predictor of chemotherapy sensitivity in advanced pancreatic cancer[102]. Inhibition of miR-221/222 expression has been shown to arrest the cell cycle, promote apoptosis, and preserve gemcitabine sensitivity during treatment[103]. miR-126 targets vascular endothelial growth factor-A, influencing tumor angiogenesis and progression. Moreover, miR-126 plays a crucial role in cancer cell invasion and metastasis by regulating genes involved in cell adhesion and migration. In addition, dysregulation of miR-126 contributes to chemoresistance, affecting cancer cell survival and therapeutic response[104].
Inhibition of miR-497 significantly accelerates gemcitabine resistance, migration, and invasion in pancreatic CSCs. Overexpression of NFκB1 has been shown to promote gemcitabine-treated pancreatic CSC viability, migration, and invasion. However, miR-497 overexpression partially suppresses this process[105]. Ye et al[106] found that miR-7, by targeting polyadenosine-diphosphate-ribose polymerase 1/NF-κB signaling, acts as an important regulator of cellular senescence. miR-7 expression is decreased in gemcitabine-resistant cell lines, and its restoration makes pancreatic cancer cells sensitive to gemcitabine, reversing resistance and cellular senescence. miR-136-5p negatively regulates ZNF32, thereby reducing the proliferation and migration abilities of pancreatic cancer cells (PANC-1 and CFPAC-1), and reversing gemcitabine resistance[107]. The tumor suppressor miRNA-3662 inhibits chemotherapy resistance and aerobic glycolysis in PDAC cells via a negative feedback loop with HIF-1α[108].
In addition to gemcitabine, miRNAs have been reported to reverse resistance to other chemotherapy agent used in pancreatic cancer treatment. miR-499a-5p targets phosphatase and tensin homolog and activates the phosphatidylinositol 3-kinase (PI3K)/Akt pathway, promoting proliferation, migration, and 5-FU resistance in pancreatic cancer cells. Moreover, miR-499a-5p has been shown to affect the expression of MDR-related genes, including ABC subfamily B member 1 (P-gp), ABC subfamily C member 1 (MRP1), and ABC subfamily G member 2 (BCRP)[109]. The tumor sup
| Names of ncRNAs | Genomic location | Expression | Regulated chemotherapy drugs | Ref. |
| miR-210 | 11p15.5 | Upregulated | GEM | [99] |
| miR-30a-3p | 6q13 | Upregulated | GEM | [100] |
| miR-146a-5p | 5q33.3 | Downregulated | GEM | [101] |
| miR-21 | 17q23.2 | Upregulated | GEM | [102] |
| miR-221/222 | Xp11.3 | Upregulated | GEM | [103] |
| miR126 | 9q34.3 | Downregulated | GEM | [104] |
| miR-497 | 17p13.1 | Downregulated | GEM | [105] |
| miR-7 | 9q21.11 | Downregulated | GEM | [106] |
| miR-136-5p | 14q32.31 | Downregulated | GEM | [107] |
| miR-3662 | 14q32.31 | Downregulated | GEM | [108] |
| miR-499a-5p | 20q11.22 | Upregulated | 5-FU | [109] |
| miR-1291-5p | 12q24.31 | Downregulated | DDP | [110] |
| miR-181a-5p | 1q32.1 | Upregulated | FOLFIRINOX | [111] |
| miR-1307 | 15q21.1 | Downregulated | FOLFIRINOX | [112] |
Regulation of chemoresistance by lncRNA family members in pancreatic cancer: Studies have shown that certain members of the lncRNA family can regulate the process of chemotherapy resistance in pancreatic cancer. For example, lncRNA SLC7A11-AS1 is highly expressed in gemcitabine-resistant pancreatic cancer cell lines. It stabilizes nuclear factor erythroid-2-related factor 2 (Nrf2), a key regulator of antioxidant defense, through interaction with β-transducing repeat-containing protein 1. Nrf2, in turn, promotes chemoresistance by reducing the levels of reactive oxygen species in pancreatic cancer cells[113].
Similarly, lncRNA HOXA transcript at the distal tip (HOTTIP) is significantly upregulated in pancreatic cancer tissues and cell lines compared with non-cancerous pancreatic tissue and the non-tumor pancreatic cell line HPDE6. Functional inhibition of HOTTIP induces cell cycle arrest and suppresses cell invasion by suppressing EMT. Moreover, HOTTIP silencing enhances the antitumor effects of gemcitabine both in vitro and in vivo. These functions were partially mediated through the activation of HOXA13[114].
Plasmacytoma variant translocation 1 (PVT1), first discovered in 1986, has been shown to possess oncogenic potential in various malignancies[115]. However, studies on the PVT1 gene in pancreatic cancer only emerged recently[116], including research on the negative regulation of gemcitabine sensitivity in pancreatic cancer cells by PVT1. A susceptibility allele, rs1561927, was identified in PVT1[117]. Studies using quantitative reverse-transcription polymerase chain reaction to measure PVT1 expression in pancreatic tissues have revealed that PVT1 is expressed at significantly higher levels in pancreatic cancer tissues compared to non-cancerous tissues, and its expression is positively correlated with poor patient survival[118] (Table 3).
Role of circRNAs in chemoresistance in pancreatic cancer: Research on circRNAs in pancreatic cancer chemoresistance is relatively limited. Liu et al[119] found that circHIPK3 expression was elevated in both gemcitabine-resistant pancreatic cancer tumor tissues and gemcitabine-resistant pancreatic cancer cells. CircHIPK3 promotes gemcitabine resistance in pancreatic cancer cells by regulating RASSF1 through miR-330-5p targeting. Exosomal circZNF91, induced by hypoxia, was transferred to normoxic pancreatic cancer cells, where it competitively binds with miR-23b-3p, thus preventing miR-23b-3p from inhibiting the expression of the deacetylase sirtuin1. Upregulation of sirtuin1 enhances the deacetylation-dependent stability of HIF-1α protein, promoting glycolysis and conferring gemcitabine resistance-a finding further validated in mouse models[120]. Hong et al[121] also discovered that circZNF91 promotes pancreatic cancer growth and metastasis by sequestering miR-519 and targeting secreted modular calcium-binding protein 2, which increases the resistance of pancreatic cancer cells to gemcitabine. In another study, circ-MTHFD1 L was shown to promote DNA damage repair by upregulating the expression of RPN6, thereby enhancing gemcitabine resistance in pancreatic cancer cells. Remarkably, inhibition of circ-MTHFD1 L in combination with olaparib was able to reverse gemcitabine resistance in pancreatic cancer[122]. CircFARP1 plays a crucial role in CAF-induced gemcitabine resistance through activation of the caveolin 1/miR-660-3p/Leukemia inhibitory factor axis. Suppression of circFARP1 significantly inhibited PDAC growth and gemcitabine resistance in patient-derived xenograft (PDX) models[123]. CircLMTK2, an isoform of circZNF91, is overexpressed in gemcitabine-resistant pancreatic cancer cells and tissues. Knockdown of circLMTK2 promoted miR-485-5p sponge-mediated regulation of p21-regulated kinase 1, reducing gemcitabine resistance in pancreatic cancer cells[124] (Table 4 and Figure 1).
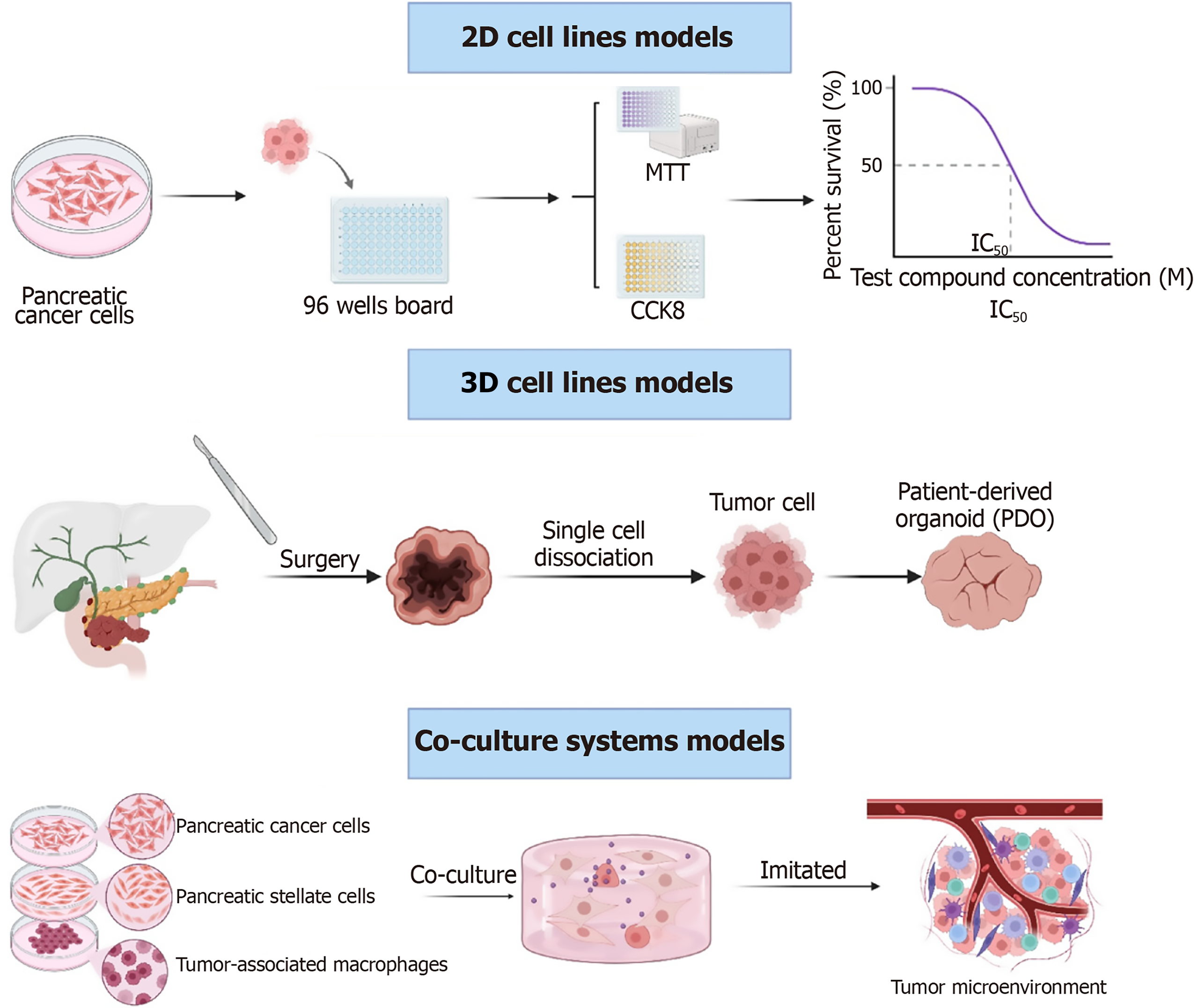
Here, we summarize the major experimental models used to investigate drug resistance in pancreatic cancer, based on extensive research and previous literature. These models can be broadly divided into four types: In vitro, in vivo, ex vivo, and specialized mechanistic models. Below, we provide a brief overview of these models, comparing their advantages, limitations, and applicability (Figure 2).
Two-dimensional cell line models: The classic experimental model for studying drug resistance. Examples of gemcitabine-resistant cell lines include PANC-1/gemcitabine[119], MIA PaCa-2/gemcitabine[125], and multi-drug-resistant cell lines such as Capan-1/P-gp (overexpressing P-gp)[126]. Researchers can use this model to select suitable cell lines based on the drugs identified in their experiments or conduct high-throughput screening of drugs in the pre-experiment phase. This is a bidirectional screening approach where methods such as CCK-8 or MTT assays can be used to observe changes in IC50 values. Researchers also explore the mechanisms of reversing drug resistance. The significant advantage of this model is its ability to study drug efficacy and mechanisms; however, it cannot replicate the TME, which is a notable limitation.
Three-dimensional organoid models: Three-dimensional (3D) organoid models are classified into patient-derived organoids (PDOs) and cell line-derived organoids. These models have gained considerable attention because they aim to retain tumor heterogeneity and stem cell properties, making them more representative of in vivo drug resistance phenotypes. PDO-based 3D organoid models are particularly useful for screening patient-specific drug combinations and predicting therapeutic responses[127]. Derived directly from patients’ tumor tissues, PDOs faithfully reproduce patient-specific molecular and phenotypic characteristics, enabling individualized testing of drug combinations and prediction of clinical therapeutic responses[128]. For instance, Tiriac et al[129] established a living biobank of pancreatic cancer organoids and demonstrated that their in-vitro drug-sensitivity profiles correlated strongly with patient outcomes in vivo, underscoring their potential as predictive diagnostic tools. Similarly, Huang et al[130] reported that organoid-based chemosensitivity assays successfully guided gemcitabine-based combination therapy, achieving improved clinical responses in PDAC patients. Moreover, recent advances in co-culture organoid models, which incorporate PSCs, immune components, or endothelial cells, have allowed researchers to more accurately recapitulate tumor-stroma interactions that drive chemoresistance[131]. Such integrated 3D systems are increasingly used to explore novel treatment strategies aimed at overcoming resistance and optimizing individualized therapy[132]. Collectively, 3D organoid models serve as a vital bridge between preclinical research and clinical application, offering a robust and adaptable platform for precision medicine in pancreatic cancer.
Co-culture systems models: Co-culture models simulate the TME by co-culturing selected pancreatic cancer cell lines with PSCs[125] or macrophages (tumor-associated macrophages)[133]. This model is used to study matrix-mediated drug resistance mechanisms, such as how PSCs secrete interleukin-6 to induce gemcitabine resistance in tumor cells.
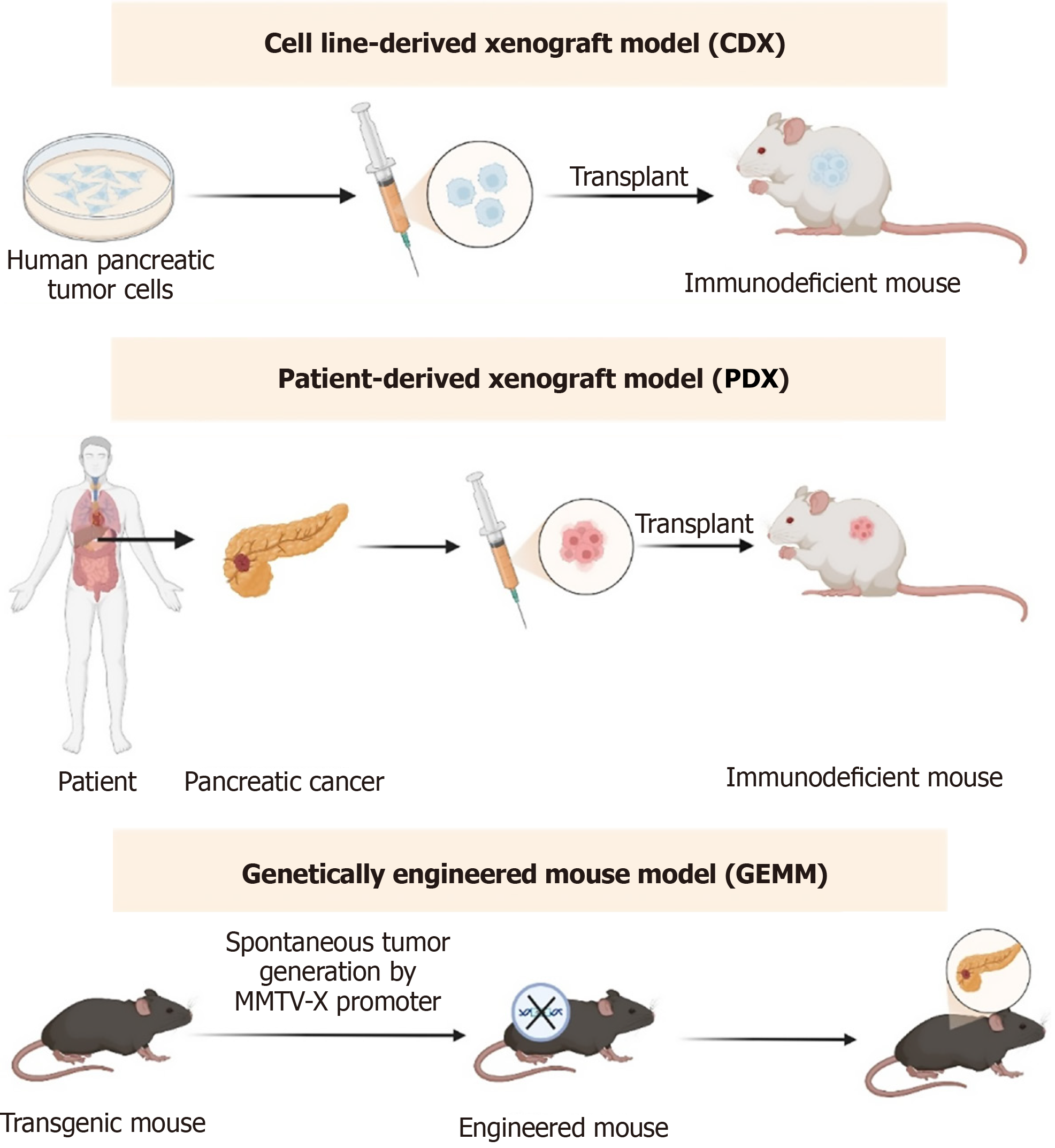
Xenograft models: Among the various experimental systems, xenograft models play a crucial role in in vivo studies of pancreatic cancer drug resistance. Drug-resistant cell lines, such as AsPC-1/gemcitabine, are commonly implanted into immunodeficient mice to assess the reversal of chemoresistance in vivo, typically through parameters such as tumor volume reduction and improved survival rates[134]. However, a major limitation of xenograft models is the lack of an immune microenvironment.
PDXs models: PDX models are derived from the patient’s own tumor, preserving the genetic and pathological characteristics of the primary tumor. These models are ideal for studying individualized drug resistance and play an irreplaceable role in testing targeted drugs (e.g., the reversal effect of polyadenosine-diphosphate-ribose polymerase inhibitors in BRCA-mutant PDX models)[135]. Essentially, 3D organoid models are derived from PDX models.
Genetically engineered mouse models: Genetically engineered mouse models enable the specific knockout or editing of genes to develop pancreatic cancer, thereby simulating the natural progression of drug resistance[136]. This model overcomes the limitations of xenograft models by incorporating an immune microenvironment; however, it incurs high financial and time costs, necessitating careful consideration by researchers (Figure 3).
Ex vivo models involve the excision of fresh tumor tissue from patients through surgical resection, which is then cultured in tissue slices. Drug sensitivity testing can be performed within a short period (72 hours)[137]. This model retains the original tumor’s cellular composition and spatial structure while maximizing the utilization of existing tissue. It can be widely used in clinical settings for short-term drug sensitivity tests. However, it is essential to note that tissue slices cannot be passaged, making long-term studies or mechanistic investigations unsuitable for this model.
In models where HIF-1α is known to mediate pancreatic cancer resistance, researchers have developed ways to simulate hypoxic environments to study how HIF-1α contributes to drug resistance. This is achieved by using hypoxic incubators (1% O2) to replicate the tumor’s hypoxic conditions[138]. Another specialized model focuses on exosome-mediated drug resistance. In this model, exosomes collected from drug-resistant cells are used to treat sensitive cells, thereby inducing cross-resistance and providing insights into the transfer of resistance mechanisms[139] (Table 5).
| Research objective | Recommended model |
| High-throughput compound screening | 2D cell lines or organoids |
| Microenvironment-mediated resistance mechanisms | Co-culture systems or PDX models |
| Personalized therapy validation | PDO, PDX, or organoids |
| Combination strategies with immunotherapy | Humanized immune system mice |
Dietary polyphenols, also referred to as phenolic compounds represent one of largest and most widely distributed groups of natural products in the plant kingdom. To date, over 8000 phenolic structures have been identified, including more than 4000 flavonoids[140-142]. The diversity and widespread distribution of polyphenols in plants increase their frequency of use. They can be classified into phenolic acids, flavonoids, polyphenolamides, and non-flavonoid polyphenols[143]. Representative substances include curcumin, resveratrol, and epigallocatechin gallate (EGCG).
Curcumin: Curcumin, a well-characterized polyphenol compound extracted from the turmeric plant, Curcuma longa. It was first isolated in 1815 and is also known as bisdemethoxycurcumin. Curcumin exhibits a broad spectrum of pharmacological activities, including anti-inflammatory, antioxidant, and anti-tumor properties, along with the ability to enhance radio- and chemotherapy sensitivity and protection of liver and kidney functions[144]. NF-κB is considered one of the major targets of curcumin’s activity, and much research has focused on this aspect[145]. Curcumin has poor bioavailability[146]. Clinical experiences using curcumin and the anti-metabolite gemcitabine in the treatment of advanced pancreatic cancer show that fewer than 10% of patients exhibit objective responses, with little impact on survival. However, the safety of this combination was demonstrated in phase I/II studies, with minimal toxic side effects[147,148]. The potent anti-proliferative activity of curcumin, which interacts with several intracellular signaling pathways, may enhance the antitumor effect of gemcitabine[149]. In addition to gemcitabine, curcumin has been reported to enhance the efficacy of other cytotoxic drugs, including cisplatin, oxaliplatin, and 5-FU[150,151]. While not all of these studies were focused on pancreatic cancer, they remain highly instructive.
Resveratrol: Resveratrol is a naturally occurring polyphenol synthesized by various plant species in response to damage, ultraviolet irradiation, and fungal attacks[152]. Among the more than 70 plant that contain resveratrol, red grape skins contain the highest amounts, with approximately 50 μg to 100 μg of resveratrol per gram of wet weight[153]. Interestingly, resveratrol maintains low biological toxicity to normal pancreatic cells[154,155]. This creates a foundation for its use in combination with chemotherapy drugs. Resveratrol sensitizes pancreatic cancer cells to various chemotherapy drugs, including gemcitabine, which has potential implications for cancer treatment. In an in situ human pancreatic cancer model, resveratrol significantly suppressed tumor growth (P < 0.001), and co-administration with gemcitabine further potentiated this inhibitory effect (P < 0.001). The combination of gemcitabine and resveratrol significantly downregulated markers of proliferation (Ki-67) and microvascular density (CD31) in tumor tissues (P < 0.001 vs control; P < 0.01 vs gemcitabine alone). Compared to the vehicle control, resveratrol suppressed NF-κB activation and reduced the expression of cyclin D1, cyclooxygenase-2, intercellular adhesion molecule-1, matrix metallopeptidase-9, and survivin. Overall, resveratrol can enhance the effect of gemcitabine by inhibiting markers of proliferation, invasion, angiogenesis, and metastasis[156]. The chemoresensitizing effect of resveratrol on tumor cells appears to be mediated through its regulation of various cell signaling molecules, including drug transporters, NF-κB, and signal transducer and activator of transcription 3[62,156,157].
EGCG: EGCG is the most abundant catechin in green tea. It is worth noting that both curcumin and EGCG are among the few plant-derived compounds approved for clinical trials. EGCG, when used alone, reduces the migration, invasion, and growth of pancreatic cancer cells. Moreover, when combined with gemcitabine, it enhances the therapeutic effects. EGCG and gemcitabine work synergistically by modulating EMT markers and inhibiting the Akt pathway, thereby suppressing pancreatic cancer cell growth, migration, and invasion[158]. A recent study investigated the effects and mechanisms of EGCG in combination with chemotherapeutic agents on pancreatic cancer cell growth, revealing that its primary mechanism involves the regulation of glycolysis. The results indicated that this specific catechin inhibits the growth of pancreatic cancer cells, and this inhibition is further enhanced under glucose-deprived conditions[159]. EGCG, when combined with bleomycin (an anticancer chemotherapy drug), exhibited anti-proliferative effects by inducing apoptosis in pancreatic cancer cells in vitro. EGCG also sensitized pancreatic cancer cells to various chemotherapy drugs, thereby overcoming drug resistance - a rare and valuable finding[160].
Flavonoids are essentially a subclass of polyphenolic compounds. After an extensive literature review, the editors have categorized them as a distinct group due to the large number of studies and detailed classifications. More than 10000 flavonoid compounds are known[161], which can be further divided into subgroups. These include flavanols, flavanones, flavonols, flavones, isoflavones, and anthocyanins[162]. The representative compounds selected in this review are quercetin and apigenin.
Quercetin: Quercetin is a flavonol[163]. Data support that commercially available quercetin can be administered orally at a daily dose of 1 g, with an absorption rate of up to 60%, thereby maintaining a sufficient blood concentration. This absorption efficiency is higher than that of EGCG and curcumin, both of which are considered safe and easily absorbed by the human body[164]. Quercetin has been shown to increase the sensitivity of pancreatic cancer cells to chemotherapy drugs, including BET inhibitors, doxorubicin, gemcitabine, sulforaphane (SFN), anthracyclines, and tumor necrosis factor-related apoptosis-inducing ligand[165]. Additionally, many signaling molecules are involved in oxidative and inflammatory pathways mediated by Toll-like receptor 4 during the development of chemotherapy resistance. One study investigated the anti-cancer effects and mechanisms of quercetin in gemcitabine-resistant cancer cells. In this study, BxPC-3, PANC-1, HepG2, and Huh-7 cell lines were tested. The cell proliferation trend showed that quercetin had a significant cytotoxic effect on resistant cell lines (including HepG2 and PANC-1). Compared with gemcitabine alone, combination therapy with quercetin resulted in greater anticancer benefits[166]. Another researcher focused on the RAGE, a major protein involved in drug resistance. It was found that silencing RAGE led to the downregulation of the classic PI3K/AKT/mechanistic target of rapamycin pathway, thus killing resistant cells. Quercetin showed a significant effect similar to si-RAGE silencing, effectively reducing RAGE expression and promoting gemcitabine sensitivity in MIA Paca-2 GEMR pancreatic cancer cells, suggesting an unexpected response when quercetin and gemcitabine were combined[63].
Apigenin: Apigenin possesses potent pharmacological activity and exhibits low biological toxicity[167]. However, its bioavailability is relatively low[168], at only 5%-10%[169]. Therefore, there is a need for research into more effective absorption derivatives and models. Recently, a study indirectly demonstrated that apigenin can overcome chemotherapy resistance in pancreatic cancer by inducing apoptosis in human pancreatic cancer cells through inhibiting the glycogen synthase kinase-3β/NF-κB signaling cascade[170]. However, the sensitizing effect of apigenin on chemotherapy drugs is limited and does not show a concentration-dependent pattern. A cell proliferation assay was performed using different concentrations (0-50 μM) of several chemotherapy drugs, combined with pre-treatment with apigenin at various time points (0, 6, 24, and 42 hours). Pre-treatment with 13 μM apigenin for 24 hours followed by gemcitabine a 36-hour pretreatment was found to be optimal for inhibiting cell proliferation, resulting in 59%-73% growth inhibition[171].
Since 1995, 162 terpenoid compounds have been discovered, and they are classified into five categories based on their structures: Monoterpenes, sesquiterpenes, diterpenes, triterpenes, and tetraterpenes. Terpenoid compounds are known for their anticancer, hepatoprotective, anti-inflammatory, antidiabetic, and neuroprotective properties[172]. A substantial of original research studies and reviews articles have highlighted the broad-spectrum anticancer activities and therapeutic potential of terpenoids[173]. The representative compounds selected for this review, which have been extensively studied, include β-elemene, paclitaxel (a semisynthetic derivative of taxol), and artemisinin.
β-elemene: β-elemene has been summarized by researchers as being effective in treating tumors across multiple systems (Integumentary System, Nervous System, Respiratory System, Immune System, Digestive System, Reproductive System, Endocrine System, Urinary System) in combination with various chemotherapy drugs (cisplatin, gefitinib, aldesleukin, oxaliplatin, doxorubicin, etc.) to enhance their effectiveness[174]. Xu’s study[175] found that β-elemene (30 μmol/L) significantly reduced the expression of P-gp. P-gp overexpression can cause MDR, and the reversal of drug resistance by β-elemene is likely related to this effect. Although there is a lack of studies specifically on β-elemene in pancreatic cancer, based on its sensitizing effects on chemotherapy drugs in many other types of tumors, we can reasonably speculate that β-elemene may have the potential to reverse drug resistance in pancreatic cancer.
Paclitaxel: Paclitaxel is a very special terpenoid compound that plays a key role in pancreatic cancer chemotherapy regimens. It is also one of the chemotherapy drugs that strengthens our belief in the great potential of plant-derived compounds in pancreatic cancer treatment. Paclitaxel is commonly used in combined with carboplatin or cisplatin as a first-line chemotherapeutic agent, and its most clinically accepted form is nab-paclitaxel (albumin-bound paclitaxel)[176]. This subtype has milder biological toxicity compared to solvent-based paclitaxel formulations. The phase III MPACT trial in metastatic pancreatic cancer patients showed that, compared to gemcitabine monotherapy, nab-paclitaxel combined with gemcitabine had superior efficacy, including the primary endpoint of OS (median OS: 8.7 months vs 6.6 months; P < 0.001)[177].
Artemisinin: Artemisinin was first isolated and for the treatment of malaria in China in 1972. It is a highly effective and cost-efficient drug for treating malaria and is classified as a sesquiterpene compound[178]. Subsequent research has demonstrated that artemisinin derivatives exhibit significant anticancer activity in various cancer cell lines and animal tumor models. Multiple clinical trials have confirmed its potential as an anticancer agent[179]. Furthermore, dihy
Alkaloids, derived from plants, are iconic specialized metabolites with numerous biological activities. Plants utilize these naturally occurring nitrogen-containing compounds as a defense mechanism to cope with various challenging environmental factors. These organic compounds have been used in traditional and modern medicine to treat various diseases. The diverse functional groups attached to their core structures are responsible for their various biological properties[183]. Alkaloids are generally categorized into six main categories: Piperidine alkaloids, isoquinoline alkaloids, indole alkaloids, terpenoid alkaloids, steroid alkaloids, and other alkaloids[184]. The representative compounds selected for this review are berberine, vinblastine, and camptothecin.
Berberine: Berberine is a naturally occurring plant-derived polyphenol found in several plants/herbs, including turmeric, goldenseal, Chinese goldthread, anemone, and Oregon grape[185]. As a widely used plant extract in clinical practice, a meta-analysis has shown that berberine significantly affects blood glucose levels, insulin resistance, inflammatory markers, colorectal adenomas, and Helicobacter pylori infection[186]. However, its poor solubility in water may hinder its effective absorption in the small intestine. Despite this, substantial progress has been made in improving its bioavailability[187]. Studies have demonstrated that berberine exhibits effective anti-inflammatory[188,189], antioxidant[189,190], antidiabetic[190-192], and anticancer properties[193-195]. In combination with lovastatin, berberine treatment of Panc02 cells resulted in a twofold increase in the percentage of cells in the G1 phase, accompanied by moderate increases in the number of G1 phase cells. Interestingly, although pretreatment with mevalonate pathway products (i.e., cholesterol synthesis) weakened the anticancer effect of lovastatin, it did not reduce the anticancer efficacy of berberine, suggesting that berberine enhanced the anticancer efficacy of lovastatin. In the Panc02 xenograft model in C57BL/6 mice, when treated with berberine (oral, 100 mg/kg/day) and lovastatin (intraperitoneal injection, 30 mg/kg/day), the tumor volume showed a slight reduction, with lovastatin alone showing a more pronounced effect. Still, it was significantly reduced with the combination therapy[196,197].
Vinblastine: Vinblastine and vincristine are representative members of the vinca alkaloid family and rank among the most significant plant-derived natural products contributing to cancer chemotherapy[198,199]. Both vinblastine and vincristine are effective clinical drugs that were introduced into clinical practice more than 50 years ago, used in combination to treat Hodgkin’s disease, testicular cancer, ovarian cancer, breast cancer, head and neck cancer, and non-Hodgkin lymphoma (vinblastine), or as curative treatment for pediatric lymphocytic leukemia and Hodgkin’s disease (vincristine). Four vinca alkaloid are used clinically, including vinblastine, vincristine, vindesine[200], and vinorelbine[201]. As a plant-derived compound directly used in cancer treatment, vinblastine, like paclitaxel, inevitably leads to drug resistance, which significantly hampers the treatment efficiency of vinca alkaloids. Consequently, many studies focus on restoring pancreatic cancer’s sensitivity to vinblastine. For example, inhibiting βIVb-tubulin in pancreatic cancer cells can increase sensitivity to vinca alkaloids (vincristine, vinorelbine, and vinblastine)[202]. Although research in this area is limited, we believe that vinblastine still has substantial potential to reverse drug resistance in pancreatic cancer.
Camptothecin: Camptothecin is derived from the Chinese tree Camptotheca acuminata, with mainland China being the only known source. It was originally isolated in 1966 as a potent anti-tumor agent[203]. Despite its good anticancer properties, camptothecin has extremely low bioavailability, which remains a critical limitation. As of June 2025, many researchers are working to improve its bioavailability and have made significant progress[204]. Topo-I serves as a key molecular target in the discovery and design of anticancer drugs. All clinically evaluated Topo-I inhibitors are camptothecin derivatives, including irinotecan (camptosar)[205] and topotecan (hycampitin)[206], which are Food and Drug Administration-approved for clinical use. Irinotecan, a component of the FOLFIRINOX regimen for pancreatic cancer, was shown in a global, multicenter phase III clinical trial (NAPOLI-1), which demonstrated improved survival in patients with metastatic pancreatic ductal adenocarcinoma who had previously received gemcitabine treatment, with a manageable safety profile[207]. Studies have found that phosphorylated aspirin (MDC-22) enhances the biological inhibition of irinotecan[208]. While much research focuses on the resistance issues related to the AG regimen for pancreatic cancer, only a few studies address FOLFIRINOX resistance. As an effective pancreatic cancer therapeutic compound, many studies aim to enhance the effects of camptothecin derivatives. It is worth considering whether camptothecin derivatives can be used as a variable in combination with existing drug treatment systems, which could be an interesting research direction for overcoming drug resistance.
Organosulfur compounds are found in foods like garlic, onions, broccoli, cabbage, and other vegetables from these plant families (Amaryllidaceae, Brassicaceae)[209,210]. Due to their ability to inhibit matrix metalloproteinases, suppress carcinogen-activating enzymes, and induce apoptosis, they have been investigated as potential antitumor agents and chemoresensitizers in combination therapies for colorectal cancer (CRC)[211-213]. Representative compounds include allicin and SFN.
Allicin: Garlic is widely used in cooking globally, and its active compound, allicin, is a potent organosulfur compound derived from garlic. Allicin is formed when the enzyme alliinase acts on another organic sulfur compound, alliin[214]. Studies have summarized that allicin has anticancer, antioxidant, anti-inflammatory, antifungal, and antibacterial properties[215]. Allicin is a lipophilic organosulfur compound and is unstable once synthesized, rapidly breaking down into a series of lipophilic organosulfur compounds, including diallyl disulfide, diallyl trisulfide, ajoene, allyl methyl trisulfide, diallyl sulfide, and others[216]. Among them, diallyl trisulfide has shown anticancer activity against pancreatic cancer. These compounds exert antitumor effects primarily by inhibiting proliferation, inducing apoptosis, suppressing migration and metastasis, and enhancing the chemotherapy sensitivity of tumor cells[217]. One study found that allicin affected a 3D drug-resistant model of pancreatic cancer[218]. Allicin has been found to reverse drug resistance in gastric cancer[219], hepatocellular carcinoma[220], CRC, and lung cancer[221] against 5-FU. Current research on allicin and its derivatives mainly focuses on gastric and CRC. As a safe and effective plant-derived compound, allicin warrants re-evaluated for its potential therapeutic effects against pancreatic cancer.
SFN: SFN is a natural, plant-derived compound found in cruciferous vegetables, such as broccoli and cauliflower, and has been widely studied for its neuroprotective and anticancer properties[222]. SFN demonstrates cytotoxic effects on pancreatic cancer, and a review of current experimental results suggests that SFN can regulate NF-κB-related target genes[223], induce cell cycle arrest, and increase reactive oxygen species levels to promote apoptosis[224]. Importantly, we found that combining SFN with gemcitabine in tumor xenografts overexpressing miR30a-3p reduced tumor volume and increased gemcitabine sensitivity[100]. This finding aligns with the ncRNA-mediated drug resistance mechanisms discussed earlier in this article. Despite these excellent preclinical results, no clinical trials have been conducted, and there is a need to fill this experimental gap.
Natural polysaccharides are large molecules with widespread biological activities, found in almost all plants. Due to the various methods of polysaccharide modification, numerous derivatives are available. Modern pharmacological research indicates that polysaccharides and their derivatives exhibit a variety of biological activities in vitro, including antioxidant, antitumor, immunomodulatory, antiviral, antibacterial, and anticoagulant functions[225-227]. Importantly, polysaccharides have been shown to have the ability to overcome drug resistance by targeting epigenetic regulators and components of the TME[228,229]. Depending on their biological source, polysaccharides can be classified into fungal polysaccharides, plant polysaccharides (originating from higher plants), animal polysaccharides, and bacterial polysaccharides. Representative substances include Astragalus polysaccharides (APS) and lentinan (LNT).
APS: Astragalus is a leguminous plant known as “Huangqi” in China. According to the Chinese Pharmacopoeia, the medicinal part of Astragalus is its dried root[230]. The main components of Astragalus include APS, flavonoids, saponins, and alkaloids. APS is one of the important active natural components derived from Astragalus[231]. Numerous pharmacological studies have demonstrated that APS possess diverse biological activities, such as the regulation of blood glucose and lipids, anticancer and anti-aging effects, and immunomodulatory functions[232]. The mainstream view is that APS exerts its antitumor effects by enhancing immunity, inducing tumor cell apoptosis, and inhibiting tumor cell proliferation and metastasis[233]. Recent retrospective studies have found that APS combined with gemcitabine and tegafur-otastat potassium capsules (S-1) significantly improves short-term efficacy and long-term survival in pancreatic cancer patients, and alleviates chemotherapy-induced immunosuppression and adverse reactions (P < 0.05)[234]. In 2018, Wu et al[235] first demonstrated that apatinib showed potential inhibitory effects on pancreatic cancer cells (ASPC-1 and PANC-1), and APS enhanced the antitumor effects of apatinib by further downregulating the phosphorylation of AKT, extracellular signal-regulated kinase, and matrix metallopeptidase-9. These excellent clinical and preclinical results confirm that APS can restore pancreatic cancer sensitivity to chemotherapy drugs. In the latest clinical guidelines for the integrated treatment of advanced pancreatic cancer using Chinese and Western medicine in China, APS have been combined with the AG regimen. However, no additional double-blind or prospective clinical studies, so whether this drug can reverse pancreatic cancer resistance remains unclear.
LNT: LNT is a key active extract from shiitake mushrooms. Numerous pharmacological and clinical studies have demonstrated that LNT has antioxidant, immunomodulatory, anticancer, blood sugar-lowering, and blood lipid-lowering biological activities[236-238]. LNT is a macromolecule with β-1,3-D-glucan, and its unique molecular structure is closely related to its pharmacological activity, with the β-glycosidic bond in glucan being the key structure for its antitumor function[239]. In Japan and China, LNT has been incorporated into cancer adjuvant therapy systems[237,240]. Sun et al[241] demonstrated that LNT, when combined with gemcitabine chemotherapy, significantly suppressed the proliferation of UBC cells-an effect confirmed by both in vivo and in vitro[242] experiments. There is a reported case in Japan of gastric and pancreatic cancer that showed a significant response to combination chemotherapy with S-1, paclitaxel, and LNT[243]. Although there are related literature reports, due to the age of the studies, their authenticity is questioned; therefore, the editors have chosen not to summarize this part of the content. Given the enhanced chemotherapeutic effect of LNT when combined with gemcitabine, it is conceivable that its combination with the AG regimen in pancreatic cancer could be highly promising, considering its stable source, high yield, and the unique advantages of fungal-derived compounds (Figures 4 and 5, Table 6).
| Names of compounds | The drug resistance mechanism or target | Experimental models | Regulated chemotherapy drugs |
| Curcumin | NF-κB | 2D cell, PDX, xenografts models | GEM, cisplatin, oxaliplatin, and 5-fluorouracil |
| Resveratrol | NF-κB, STAT3 | Co-culture systems models | GEM |
| EGCG | Akt | 2D cell, xenografts models | GEM |
| Quercetin | PI3K/AKT/mTOR | 2D cell, xenografts models | GEM |
| Apigenin | GSK-3β/NF-κB | 2D cell, xenografts models | GEM |
| β-elemene | P-gp | 2D cell, xenografts models | Cisplatin, gefitinib, aldesleukin, oxaliplatin, doxorubicin |
| Paclitaxel | Hedgehog | 2D cell, xenografts models | GEM |
| Artemisinin | / | 2D cell | GEM |
| Berberine | Rap1/PI3K-Akt | 2D cell, co-culture systems models | Lovastatin |
| Vinblastine | βIVb-tubulin | CDO | Vincristine |
| Camptothecin | Topo-I | 3D, PDX, xenografts models | GEM |
| Allicin | / | 2D cell | 5-fluorouracil |
| Sulforaphane | NF-κB | 2D cell, xenografts models | GEM |
| Astragalus polysaccharides | AKT/ERK/MMP-9 | 2D cell, xenografts models | GEM, S-1 |
| Lentinan | / | 2D cell | GEM, S-1, PTX |
Most plant-derived compounds, being naturally occurring substances, show varying degrees of efficacy against pan
A major issue with the natural compounds mentioned above is their poor bioavailability. This limitation can be primarily attributed to low oral bioavailability and inadequate systemic concentration. Despite numerous studies confirming their surprising effects in reversing pancreatic cancer resistance, the compounds’ effectiveness will be meaningless if their oral bioavailability or blood concentration cannot attain the efficacy observed in vitro experiments. Efforts to address this issue through high-doses administration often fail to enhance absorption efficiency and may lead to additional toxicity. Previous studies have shown that poor bioavailability is associated with limited absorption, rapid metabolism, and systemic elimination[146,244]. Numerous researchers have investigated synthetic derivatives and materials-based delivery systems to overcome these limitations and enhance bioavailability, thereby improving drug delivery to tumor sites and achieving encouraging outcomes[187,244-246].
The TME in pancreatic cancer is characterized by dense fibrotic stroma (with stroma accounting for > 70%) and immune-suppressive cell infiltration, which is not typically replicated in conventional in vitro cell line experiments. To simulate the TME, 3D organoid models are more appropriate. The weakening of drug efficacy is largely attributed to the physical barriers of the TME, immune evasion mechanisms, and high expression of MDR proteins. These factors contribute to the resistance of pancreatic cancer to chemotherapy. Recent research has shown that nanoparticle delivery systems can address this issue[247]. Through the application of advanced material technologies, these systems can sustain blood drug concentrations and achieve more precise and targeted delivery[248].
Plant-derived compounds can reverse resistance through multiple pathways[149]. For example, curcumin can enhance the efficacy of gemcitabine in combination by inhibiting NF-κB[249], and overcome pancreatic cancer chemoresistance through its actions on nuclear factor of activated T cells and Nrf2[250]. However, this multi-target approach is not always desirable. Multiple pathways can lead to unclear mechanisms and uncontrolled therapeutic effects, posing unpredictable risks. Achieving precision treatment remains a key hurdle for plant-derived compounds to become mainstream drugs.
The issue of standardization is particularly evident in polysaccharide-based compounds. Due to the uniqueness of polysaccharides, their chemical structures cannot always be easily depicted, which results in inefficiency when quantifying active substances and large batch-to-batch variability[251]. Chemical structural variations and the interference of metabolic products are other challenges. Many bioactive substances undergo metabolic transformation in the human digestive organs, particularly in the liver and small intestine. For example, curcumin’s double bonds are reduced to dihydrocurcumin, tetrahydrocurcumin, hexahydrocurcumin, and octahydrocurcumin by reductases in enterocytes and hepatocytes[252]. Resveratrol is reduced to dihydroresveratrol conjugates and other derivatives, and the extensive metabolism in the intestines and liver results in an oral bioavailability of less than 1%. Even with increasing doses and repeated administration, the situation doesn’t improve significantly[253]. Quercetin and kaempferol are rapidly metabolized in the liver and circulate as methyl, glucuronide, and sulfate conjugates[254]. Delivering plant-derived compounds to tumor sites in their native structures, as verified in vitro, remains a pressing issue.
Overcoming pancreatic cancer drug resistance using plant-derived compounds must address three major contradictions: Balancing multi-target specificity, TME penetration and immune remodeling synergy, and standardizing chemical heterogeneity. Future research should focus on integrating nanotechnology, biomarker stratification, and immune-metabolic reprogramming strategies to drive precision translation. To enhance research efficiency and facilitate the translation of in vitro and in vivo findings into clinical applications, selecting compounds that are it is advisable to prioritize compounds already approved by the Food and Drug Administration and National Medical Products Administration to ensure smoother clinical translation. Emerging delivery systems can be further optimized through nanotechnology-based approaches, including targeted liposomes and exosomes. Such systems may be activated by specific proteins in the cellular microenvironment or through biotransformation mediated by intestinal microbiota and hepatic enzymes, thereby enhancing bioavailability and overall delivery efficiency. At the same time, the rapid development of artificial intelligence technologies has brought new opportunities to researchers. For instance, platforms such as DeepPhytoScreen allow the construction of deep learning models using either public or user-provided datasets. These models facilitate the virtual screening of chemical probes or drug candidates against specific targets of interest. Through DeepScreening, users can conveniently build deep learning models and generate target-focused databases. The classification and regression models thus established can be employed for virtual screening the generated databases or existing chemical libraries. These libraries may include synthetic compounds, natural products, approved drugs, covalent agents, protein-protein interaction modulators, and allosteric regulators collected from various chemical suppliers. The entire workflow - from deep model training to virtual screening, and from database construction to target-focused de novo library generation - can be accomplished through DeepScreening. We believe that such deep learning-based web servers will greatly benefit biologists and chemists in fundamental research and drug discovery.
Pancreatic cancer, especially PDAC, remains a significant global health challenge due to delayed diagnosis, invasive metastasis, and stubborn treatment resistance. The current standard treatment regimens, based on gemcitabine (e.g., the AG regimen) or FOLFIRINOX, are limited by both intrinsic and acquired drug resistance, resulting in limited survival benefits for patients. This review systematically discusses the multidimensional mechanisms of chemotherapy resistance and highlights the potential of plant-derived compounds in overcoming these resistance barriers. Despite significant preclinical evidence, translational applications still face challenges, including improving compound bioavailability, systematically evaluating synergy with standard chemotherapy, and validating efficacy in advanced models (e.g., PDOs and genetically engineered mouse models). Future studies should focus on determining optimal dosing regimens, developing combination strategies, and establishing patient stratification biomarker systems. Plant-derived compounds provide valuable resources for overcoming challenges in pancreatic cancer therapy and show great potential as novel adjunctive strategies to improve clinical outcomes.
| 1. | Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913-2921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5860] [Cited by in RCA: 5398] [Article Influence: 449.8] [Reference Citation Analysis (6)] |
| 2. | Klein AP. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nat Rev Gastroenterol Hepatol. 2021;18:493-502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1039] [Cited by in RCA: 919] [Article Influence: 183.8] [Reference Citation Analysis (0)] |
| 3. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68:7-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11530] [Cited by in RCA: 13104] [Article Influence: 1638.0] [Reference Citation Analysis (3)] |
| 4. | Stoffel EM, Brand RE, Goggins M. Pancreatic Cancer: Changing Epidemiology and New Approaches to Risk Assessment, Early Detection, and Prevention. Gastroenterology. 2023;164:752-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 404] [Cited by in RCA: 366] [Article Influence: 122.0] [Reference Citation Analysis (7)] |
| 5. | Li Z, Zhang X, Sun C, Li Z, Fei H, Zhao D. Global, regional, and national burdens of early onset pancreatic cancer in adolescents and adults aged 15-49 years from 1990 to 2019 based on the Global Burden of Disease Study 2019: a cross-sectional study. Int J Surg. 2024;110:1929-1940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 74] [Article Influence: 37.0] [Reference Citation Analysis (1)] |
| 6. | Lin SR, Fu YS, Tsai MJ, Cheng H, Weng CF. Natural Compounds from Herbs that can Potentially Execute as Autophagy Inducers for Cancer Therapy. Int J Mol Sci. 2017;18:1412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 114] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 7. | Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de la Fouchardière C, Bennouna J, Bachet JB, Khemissa-Akouz F, Péré-Vergé D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M; Groupe Tumeurs Digestives of Unicancer; PRODIGE Intergroup. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817-1825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6475] [Cited by in RCA: 5911] [Article Influence: 394.1] [Reference Citation Analysis (4)] |
| 8. | Kang J, Hwang I, Yoo C, Kim KP, Jeong JH, Chang HM, Lee SS, Park DH, Song TJ, Seo DW, Lee SK, Kim MH, Hong SM, Shin SH, Hwang DW, Song KB, Lee JH, Kim SC, Ryoo BY. Nab-paclitaxel plus gemcitabine versus FOLFIRINOX as the first-line chemotherapy for patients with metastatic pancreatic cancer: retrospective analysis. Invest New Drugs. 2018;36:732-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 105] [Article Influence: 13.1] [Reference Citation Analysis (3)] |
| 9. | McBride A, Bonafede M, Cai Q, Princic N, Tran O, Pelletier C, Parisi M, Patel M. Comparison of treatment patterns and economic outcomes among metastatic pancreatic cancer patients initiated on nab-paclitaxel plus gemcitabine versus FOLFIRINOX. Expert Rev Clin Pharmacol. 2017;10:1153-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Peixoto RD, Ho M, Renouf DJ, Lim HJ, Gill S, Ruan JY, Cheung WY. Eligibility of Metastatic Pancreatic Cancer Patients for First-Line Palliative Intent nab-Paclitaxel Plus Gemcitabine Versus FOLFIRINOX. Am J Clin Oncol. 2017;40:507-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 11. | Conroy T, Hammel P, Hebbar M, Ben Abdelghani M, Wei AC, Raoul JL, Choné L, Francois E, Artru P, Biagi JJ, Lecomte T, Assenat E, Faroux R, Ychou M, Volet J, Sauvanet A, Breysacher G, Di Fiore F, Cripps C, Kavan P, Texereau P, Bouhier-Leporrier K, Khemissa-Akouz F, Legoux JL, Juzyna B, Gourgou S, O'Callaghan CJ, Jouffroy-Zeller C, Rat P, Malka D, Castan F, Bachet JB; Canadian Cancer Trials Group and the Unicancer-GI–PRODIGE Group. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N Engl J Med. 2018;379:2395-2406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2449] [Cited by in RCA: 2145] [Article Influence: 268.1] [Reference Citation Analysis (3)] |
| 12. | Catenacci DV, Junttila MR, Karrison T, Bahary N, Horiba MN, Nattam SR, Marsh R, Wallace J, Kozloff M, Rajdev L, Cohen D, Wade J, Sleckman B, Lenz HJ, Stiff P, Kumar P, Xu P, Henderson L, Takebe N, Salgia R, Wang X, Stadler WM, de Sauvage FJ, Kindler HL. Randomized Phase Ib/II Study of Gemcitabine Plus Placebo or Vismodegib, a Hedgehog Pathway Inhibitor, in Patients With Metastatic Pancreatic Cancer. J Clin Oncol. 2015;33:4284-4292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 488] [Cited by in RCA: 457] [Article Influence: 41.5] [Reference Citation Analysis (3)] |
| 13. | O'Reilly EM, Ko AH, Friedberg JW. Flashback Foreword: Gemcitabine for Advanced Pancreatic Cancer. J Clin Oncol. 2023;41:5479-5480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Schwenka J, Ignoffo RJ. Temozolomide. A new option for high-grade astrocytomas. Cancer Pract. 2000;8:311-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Abbruzzese JL, Grunewald R, Weeks EA, Gravel D, Adams T, Nowak B, Mineishi S, Tarassoff P, Satterlee W, Raber MN. A phase I clinical, plasma, and cellular pharmacology study of gemcitabine. J Clin Oncol. 1991;9:491-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 580] [Cited by in RCA: 579] [Article Influence: 16.5] [Reference Citation Analysis (3)] |
| 16. | Kelderman S, Schumacher TN, Haanen JB. Acquired and intrinsic resistance in cancer immunotherapy. Mol Oncol. 2014;8:1132-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 151] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 17. | She C, Wu C, Guo W, Xie Y, Li S, Liu W, Xu C, Li H, Cao P, Yang Y, Wang X, Chang A, Feng Y, Hao J. Combination of RUNX1 inhibitor and gemcitabine mitigates chemo-resistance in pancreatic ductal adenocarcinoma by modulating BiP/PERK/eIF2α-axis-mediated endoplasmic reticulum stress. J Exp Clin Cancer Res. 2023;42:238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 18. | Mini E, Nobili S, Caciagli B, Landini I, Mazzei T. Cellular pharmacology of gemcitabine. Ann Oncol. 2006;17 Suppl 5:v7-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 590] [Article Influence: 29.5] [Reference Citation Analysis (3)] |
| 19. | Nordh S, Ansari D, Andersson R. hENT1 expression is predictive of gemcitabine outcome in pancreatic cancer: a systematic review. World J Gastroenterol. 2014;20:8482-8490. [PubMed] [DOI] [Full Text] |
| 20. | Farrell JJ, Elsaleh H, Garcia M, Lai R, Ammar A, Regine WF, Abrams R, Benson AB, Macdonald J, Cass CE, Dicker AP, Mackey JR. Human equilibrative nucleoside transporter 1 levels predict response to gemcitabine in patients with pancreatic cancer. Gastroenterology. 2009;136:187-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 342] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 21. | Greenhalf W, Ghaneh P, Neoptolemos JP, Palmer DH, Cox TF, Lamb RF, Garner E, Campbell F, Mackey JR, Costello E, Moore MJ, Valle JW, McDonald AC, Carter R, Tebbutt NC, Goldstein D, Shannon J, Dervenis C, Glimelius B, Deakin M, Charnley RM, Lacaine F, Scarfe AG, Middleton MR, Anthoney A, Halloran CM, Mayerle J, Oláh A, Jackson R, Rawcliffe CL, Scarpa A, Bassi C, Büchler MW; European Study Group for Pancreatic Cancer. Pancreatic cancer hENT1 expression and survival from gemcitabine in patients from the ESPAC-3 trial. J Natl Cancer Inst. 2014;106:djt347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 205] [Article Influence: 17.1] [Reference Citation Analysis (1)] |
| 22. | Santini D, Perrone G, Vincenzi B, Lai R, Cass C, Alloni R, Rabitti C, Antinori A, Vecchio F, Morini S, Magistrelli P, Coppola R, Mackey JR, Tonini G. Human equilibrative nucleoside transporter 1 (hENT1) protein is associated with short survival in resected ampullary cancer. Ann Oncol. 2008;19:724-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 23. | Bird NT, Elmasry M, Jones R, Psarelli E, Dodd J, Malik H, Greenhalf W, Kitteringham N, Ghaneh P, Neoptolemos JP, Palmer D. Immunohistochemical hENT1 expression as a prognostic biomarker in patients with resected pancreatic ductal adenocarcinoma undergoing adjuvant gemcitabine-based chemotherapy. Br J Surg. 2017;104:328-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 24. | Elander NO, Aughton K, Ghaneh P, Neoptolemos JP, Palmer DH, Cox TF, Campbell F, Costello E, Halloran CM, Mackey JR, Scarfe AG, Valle JW, McDonald AC, Carter R, Tebbutt NC, Goldstein D, Shannon J, Dervenis C, Glimelius B, Deakin M, Charnley RM, Anthoney A, Lerch MM, Mayerle J, Oláh A, Büchler MW, Greenhalf W; European Study Group for Pancreatic Cancer. Expression of dihydropyrimidine dehydrogenase (DPD) and hENT1 predicts survival in pancreatic cancer. Br J Cancer. 2018;118:947-954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 25. | Bhutia YD, Hung SW, Patel B, Lovin D, Govindarajan R. CNT1 expression influences proliferation and chemosensitivity in drug-resistant pancreatic cancer cells. Cancer Res. 2011;71:1825-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (3)] |
| 26. | Azim HA Jr, Mok T. Systemic nonhormonal management of advanced prostate cancer and its likely impact on patients' survival and quality of life. Anticancer Drugs. 2008;19:645-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Maity G, Ghosh A, Gupta V, Haque I, Sarkar S, Das A, Dhar K, Bhavanasi S, Gunewardena SS, Von Hoff DD, Mallik S, Kambhampati S, Banerjee SK, Banerjee S. CYR61/CCN1 Regulates dCK and CTGF and Causes Gemcitabine-resistant Phenotype in Pancreatic Ductal Adenocarcinoma. Mol Cancer Ther. 2019;18:788-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 28. | Funamizu N, Okamoto A, Kamata Y, Misawa T, Uwagawa T, Gocho T, Yanaga K, Manome Y. Is the resistance of gemcitabine for pancreatic cancer settled only by overexpression of deoxycytidine kinase? Oncol Rep. 2010;23:471-475. [PubMed] |
| 29. | Tang K, Zhang Z, Bai Z, Ma X, Guo W, Wang Y. Enhancement of gemcitabine sensitivity in pancreatic cancer by co-regulation of dCK and p8 expression. Oncol Rep. 2011;25:963-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 30. | Cerqueira NM, Fernandes PA, Ramos MJ. Ribonucleotide reductase: a critical enzyme for cancer chemotherapy and antiviral agents. Recent Pat Anticancer Drug Discov. 2007;2:11-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 71] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 31. | Ruiz van Haperen VW, Veerman G, Vermorken JB, Peters GJ. 2',2'-Difluoro-deoxycytidine (gemcitabine) incorporation into RNA and DNA of tumour cell lines. Biochem Pharmacol. 1993;46:762-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 179] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 32. | Sui L, Wang J, Li BM. Role of the phosphoinositide 3-kinase-Akt-mammalian target of the rapamycin signaling pathway in long-term potentiation and trace fear conditioning memory in rat medial prefrontal cortex. Learn Mem. 2008;15:762-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 161] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 33. | Costantino CL, Witkiewicz AK, Kuwano Y, Cozzitorto JA, Kennedy EP, Dasgupta A, Keen JC, Yeo CJ, Gorospe M, Brody JR. The role of HuR in gemcitabine efficacy in pancreatic cancer: HuR Up-regulates the expression of the gemcitabine metabolizing enzyme deoxycytidine kinase. Cancer Res. 2009;69:4567-4572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 140] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 34. | Tatarian T, Jiang W, Leiby BE, Grigoli A, Jimbo M, Dabbish N, Neoptolemos JP, Greenhalf W, Costello E, Ghaneh P, Halloran C, Palmer D, Buchler M, Yeo CJ, Winter JM, Brody JR. Cytoplasmic HuR Status Predicts Disease-free Survival in Resected Pancreatic Cancer: A Post-hoc Analysis From the International Phase III ESPAC-3 Clinical Trial. Ann Surg. 2018;267:364-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 35. | Han QL, Zhou YH, Lyu Y, Yan H, Dai GH. Effect of ribonucleotide reductase M1 expression on overall survival in patients with pancreatic cancer receiving gemcitabine chemotherapy: A literature-based meta-analysis. J Clin Pharm Ther. 2018;43:163-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 36. | Aoyama T, Miyagi Y, Murakawa M, Yamaoku K, Atsumi Y, Shiozawa M, Ueno M, Morimoto M, Oshima T, Yukawa N, Yoshikawa T, Rino Y, Masuda M, Morinaga S. Clinical implications of ribonucleotide reductase subunit M1 in patients with pancreatic cancer who undergo curative resection followed by adjuvant chemotherapy with gemcitabine. Oncol Lett. 2017;13:3423-3430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 37. | Wang C, Zhang W, Fu M, Yang A, Huang H, Xie J. Establishment of human pancreatic cancer gemcitabineresistant cell line with ribonucleotide reductase overexpression. Oncol Rep. 2015;33:383-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 38. | Nakahira S, Nakamori S, Tsujie M, Takahashi Y, Okami J, Yoshioka S, Yamasaki M, Marubashi S, Takemasa I, Miyamoto A, Takeda Y, Nagano H, Dono K, Umeshita K, Sakon M, Monden M. Involvement of ribonucleotide reductase M1 subunit overexpression in gemcitabine resistance of human pancreatic cancer. Int J Cancer. 2007;120:1355-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 160] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 39. | Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20:69-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3195] [Cited by in RCA: 2824] [Article Influence: 403.4] [Reference Citation Analysis (4)] |
| 40. | Saitoh M. Transcriptional regulation of EMT transcription factors in cancer. Semin Cancer Biol. 2023;97:21-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 115] [Reference Citation Analysis (0)] |
| 41. | Seo J, Ha J, Kang E, Cho S. The role of epithelial-mesenchymal transition-regulating transcription factors in anti-cancer drug resistance. Arch Pharm Res. 2021;44:281-292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 42. | Arumugam T, Ramachandran V, Fournier KF, Wang H, Marquis L, Abbruzzese JL, Gallick GE, Logsdon CD, McConkey DJ, Choi W. Epithelial to mesenchymal transition contributes to drug resistance in pancreatic cancer. Cancer Res. 2009;69:5820-5828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 719] [Cited by in RCA: 722] [Article Influence: 42.5] [Reference Citation Analysis (4)] |
| 43. | Singh M, Yelle N, Venugopal C, Singh SK. EMT: Mechanisms and therapeutic implications. Pharmacol Ther. 2018;182:80-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 361] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 44. | Zhang J, Hu Z, Horta CA, Yang J. Regulation of epithelial-mesenchymal transition by tumor microenvironmental signals and its implication in cancer therapeutics. Semin Cancer Biol. 2023;88:46-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 71] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 45. | De Palma M, Biziato D, Petrova TV. Microenvironmental regulation of tumour angiogenesis. Nat Rev Cancer. 2017;17:457-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1595] [Cited by in RCA: 1407] [Article Influence: 156.3] [Reference Citation Analysis (3)] |
| 46. | Apte MV, Xu Z, Pothula S, Goldstein D, Pirola RC, Wilson JS. Pancreatic cancer: The microenvironment needs attention too! Pancreatology. 2015;15:S32-S38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (3)] |
| 47. | Pothula SP, Xu Z, Goldstein D, Pirola RC, Wilson JS, Apte MV. Key role of pancreatic stellate cells in pancreatic cancer. Cancer Lett. 2016;381:194-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (3)] |
| 48. | Delitto D, Black BS, Sorenson HL, Knowlton AE, Thomas RM, Sarosi GA, Moldawer LL, Behrns KE, Liu C, George TJ, Trevino JG, Wallet SM, Hughes SJ. The inflammatory milieu within the pancreatic cancer microenvironment correlates with clinicopathologic parameters, chemoresistance and survival. BMC Cancer. 2015;15:783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 49. | Cao F, Li J, Sun H, Liu S, Cui Y, Li F. HES 1 is essential for chemoresistance induced by stellate cells and is associated with poor prognosis in pancreatic cancer. Oncol Rep. 2015;33:1883-1889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 50. | Liu Y, Li F, Gao F, Xing L, Qin P, Liang X, Zhang J, Qiao X, Lin L, Zhao Q, Du L. Periostin promotes the chemotherapy resistance to gemcitabine in pancreatic cancer. Tumour Biol. 2016;37:15283-15291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 51. | Hwang HJ, Oh MS, Lee DW, Kuh HJ. Multiplex quantitative analysis of stroma-mediated cancer cell invasion, matrix remodeling, and drug response in a 3D co-culture model of pancreatic tumor spheroids and stellate cells. J Exp Clin Cancer Res. 2019;38:258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 98] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 52. | Zhang H, Wu H, Guan J, Wang L, Ren X, Shi X, Liang Z, Liu T. Paracrine SDF-1α signaling mediates the effects of PSCs on GEM chemoresistance through an IL-6 autocrine loop in pancreatic cancer cells. Oncotarget. 2015;6:3085-3097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 53. | Tostar U, Toftgård R, Zaphiropoulos PG, Shimokawa T. Reduction of human embryonal rhabdomyosarcoma tumor growth by inhibition of the hedgehog signaling pathway. Genes Cancer. 2010;1:941-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 54. | Hui M, Cazet A, Nair R, Watkins DN, O'Toole SA, Swarbrick A. The Hedgehog signalling pathway in breast development, carcinogenesis and cancer therapy. Breast Cancer Res. 2013;15:203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 55. | Scales SJ, de Sauvage FJ. Mechanisms of Hedgehog pathway activation in cancer and implications for therapy. Trends Pharmacol Sci. 2009;30:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 560] [Cited by in RCA: 527] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 56. | Sari IN, Phi LTH, Jun N, Wijaya YT, Lee S, Kwon HY. Hedgehog Signaling in Cancer: A Prospective Therapeutic Target for Eradicating Cancer Stem Cells. Cells. 2018;7:208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 164] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 57. | Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA, Baylin SB. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature. 2003;422:313-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 810] [Cited by in RCA: 821] [Article Influence: 35.7] [Reference Citation Analysis (0)] |
| 58. | Karhadkar SS, Bova GS, Abdallah N, Dhara S, Gardner D, Maitra A, Isaacs JT, Berman DM, Beachy PA. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature. 2004;431:707-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 786] [Cited by in RCA: 777] [Article Influence: 35.3] [Reference Citation Analysis (2)] |
| 59. | Giroux-Leprieur E, Costantini A, Ding VW, He B. Hedgehog Signaling in Lung Cancer: From Oncogenesis to Cancer Treatment Resistance. Int J Mol Sci. 2018;19:2835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 86] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 60. | Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, Madhu B, Goldgraben MA, Caldwell ME, Allard D, Frese KK, Denicola G, Feig C, Combs C, Winter SP, Ireland-Zecchini H, Reichelt S, Howat WJ, Chang A, Dhara M, Wang L, Rückert F, Grützmann R, Pilarsky C, Izeradjene K, Hingorani SR, Huang P, Davies SE, Plunkett W, Egorin M, Hruban RH, Whitebread N, McGovern K, Adams J, Iacobuzio-Donahue C, Griffiths J, Tuveson DA. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457-1461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2693] [Cited by in RCA: 2569] [Article Influence: 151.1] [Reference Citation Analysis (4)] |
| 61. | Huang FT, Zhuan-Sun YX, Zhuang YY, Wei SL, Tang J, Chen WB, Zhang SN. Inhibition of hedgehog signaling depresses self-renewal of pancreatic cancer stem cells and reverses chemoresistance. Int J Oncol. 2012;41:1707-1714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 62. | Gupta SC, Kannappan R, Reuter S, Kim JH, Aggarwal BB. Chemosensitization of tumors by resveratrol. Ann N Y Acad Sci. 2011;1215:150-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 234] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 63. | Lan CY, Chen SY, Kuo CW, Lu CC, Yen GC. Quercetin facilitates cell death and chemosensitivity through RAGE/PI3K/AKT/mTOR axis in human pancreatic cancer cells. J Food Drug Anal. 2019;27:887-896. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 121] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 64. | Dean M, Rzhetsky A, Allikmets R. The human ATP-binding cassette (ABC) transporter superfamily. Genome Res. 2001;11:1156-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 692] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 65. | Dean M, Annilo T. Evolution of the ATP-binding cassette (ABC) transporter superfamily in vertebrates. Annu Rev Genomics Hum Genet. 2005;6:123-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 541] [Cited by in RCA: 473] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 66. | Kobayashi A, Takanezawa Y, Hirata T, Shimizu Y, Misasa K, Kioka N, Arai H, Ueda K, Matsuo M. Efflux of sphingomyelin, cholesterol, and phosphatidylcholine by ABCG1. J Lipid Res. 2006;47:1791-1802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 164] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 67. | Yvan-Charvet L, Wang N, Tall AR. Role of HDL, ABCA1, and ABCG1 transporters in cholesterol efflux and immune responses. Arterioscler Thromb Vasc Biol. 2010;30:139-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 542] [Article Influence: 31.9] [Reference Citation Analysis (0)] |
| 68. | Kim HS, Kim E, Kim JW. Development of a breast self-examination program for the Internet: health information for Korean women. Cancer Nurs. 2001;24:156-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 69. | Ji N, Li H, Zhang Y, Li Y, Wang P, Chen X, Liu YN, Wang JQ, Yang Y, Chen ZS, Li Y, Wang R, Kong D. Lansoprazole (LPZ) reverses multidrug resistance (MDR) in cancer through impeding ATP-binding cassette (ABC) transporter-mediated chemotherapeutic drug efflux and lysosomal sequestration. Drug Resist Updat. 2024;76:101100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 70. | Leslie EM, Deeley RG, Cole SP. Multidrug resistance proteins: role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicol Appl Pharmacol. 2005;204:216-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1162] [Cited by in RCA: 1030] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 71. | Patel D, Sethi N, Patel P, Shah S, Patel K. Exploring the potential of P-glycoprotein inhibitors in the targeted delivery of anti-cancer drugs: A comprehensive review. Eur J Pharm Biopharm. 2024;198:114267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 72. | Scheffer GL, Wijngaard PL, Flens MJ, Izquierdo MA, Slovak ML, Pinedo HM, Meijer CJ, Clevers HC, Scheper RJ. The drug resistance-related protein LRP is the human major vault protein. Nat Med. 1995;1:578-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 423] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 73. | Pan LN, Zhang Y, Zhu CJ, Dong ZX. Kinesin KIF4A is associated with chemotherapeutic drug resistance by regulating intracellular trafficking of lung resistance-related protein. J Zhejiang Univ Sci B. 2017;18:1046-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 74. | Dalton WS, Scheper RJ. Lung resistance-related protein: determining its role in multidrug resistance. J Natl Cancer Inst. 1999;91:1604-1605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 75. | De Isabella P, Capranico G, Zunino F. The role of topoisomerase II in drug resistance. Life Sci. 1991;48:2195-2205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 76. | Takano H, Kohno K, Matsuo K, Matsuda T, Kuwano M. DNA topoisomerase-targeting antitumor agents and drug resistance. Anticancer Drugs. 1992;3:323-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 77. | Tew KD. Glutathione-Associated Enzymes In Anticancer Drug Resistance. Cancer Res. 2016;76:7-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 78. | McIlwain CC, Townsend DM, Tew KD. Glutathione S-transferase polymorphisms: cancer incidence and therapy. Oncogene. 2006;25:1639-1648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 422] [Cited by in RCA: 374] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 79. | Borst P, Elferink RO. Mammalian ABC transporters in health and disease. Annu Rev Biochem. 2002;71:537-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1164] [Cited by in RCA: 1113] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 80. | Higgins CF. ABC transporters: from microorganisms to man. Annu Rev Cell Biol. 1992;8:67-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2861] [Cited by in RCA: 2793] [Article Influence: 82.1] [Reference Citation Analysis (11)] |
| 81. | Tanigawara Y. Role of P-glycoprotein in drug disposition. Ther Drug Monit. 2000;22:137-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 169] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 82. | Fromm MF. P-glycoprotein: a defense mechanism limiting oral bioavailability and CNS accumulation of drugs. Int J Clin Pharmacol Ther. 2000;38:69-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 267] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 83. | Fromm MF. Importance of P-glycoprotein at blood-tissue barriers. Trends Pharmacol Sci. 2004;25:423-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 367] [Cited by in RCA: 402] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 84. | Guo Y, Ashrafizadeh M, Tambuwala MM, Ren J, Orive G, Yu G. P-glycoprotein (P-gp)-driven cancer drug resistance: biological profile, non-coding RNAs, drugs and nanomodulators. Drug Discov Today. 2024;29:104161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 52] [Reference Citation Analysis (0)] |
| 85. | Lehmann T, Torky AR, Stehfest E, Hofmann S, Foth H. Expression of lung resistance-related protein, LRP, and multidrug resistance-related protein, MRP1, in normal human lung cells in long-term cultures. Arch Toxicol. 2005;79:600-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 86. | Kitazono M, Sumizawa T, Takebayashi Y, Chen ZS, Furukawa T, Nagayama S, Tani A, Takao S, Aikou T, Akiyama S. Multidrug resistance and the lung resistance-related protein in human colon carcinoma SW-620 cells. J Natl Cancer Inst. 1999;91:1647-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 136] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 87. | Scheffer GL, Schroeijers AB, Izquierdo MA, Wiemer EA, Scheper RJ. Lung resistance-related protein/major vault protein and vaults in multidrug-resistant cancer. Curr Opin Oncol. 2000;12:550-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 126] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 88. | Dehshahri A, Ashrafizadeh M, Ghasemipour Afshar E, Pardakhty A, Mandegary A, Mohammadinejad R, Sethi G. Topoisomerase inhibitors: Pharmacology and emerging nanoscale delivery systems. Pharmacol Res. 2020;151:104551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 89. | Holm C, Goto T, Wang JC, Botstein D. DNA topoisomerase II is required at the time of mitosis in yeast. Cell. 1985;41:553-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 577] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 90. | Forman HJ, Zhang H, Rinna A. Glutathione: overview of its protective roles, measurement, and biosynthesis. Mol Aspects Med. 2009;30:1-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2200] [Cited by in RCA: 1783] [Article Influence: 104.9] [Reference Citation Analysis (0)] |
| 91. | Asantewaa G, Harris IS. Glutathione and its precursors in cancer. Curr Opin Biotechnol. 2021;68:292-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 108] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 92. | Dirr H, Reinemer P, Huber R. X-ray crystal structures of cytosolic glutathione S-transferases. Implications for protein architecture, substrate recognition and catalytic function. Eur J Biochem. 1994;220:645-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 340] [Cited by in RCA: 331] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 93. | Slack FJ, Chinnaiyan AM. The Role of Non-coding RNAs in Oncology. Cell. 2019;179:1033-1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1372] [Cited by in RCA: 1241] [Article Influence: 177.3] [Reference Citation Analysis (5)] |
| 94. | Nemeth K, Bayraktar R, Ferracin M, Calin GA. Non-coding RNAs in disease: from mechanisms to therapeutics. Nat Rev Genet. 2024;25:211-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 697] [Cited by in RCA: 582] [Article Influence: 291.0] [Reference Citation Analysis (1)] |
| 95. | Sanchez-Mejias A, Tay Y. Competing endogenous RNA networks: tying the essential knots for cancer biology and therapeutics. J Hematol Oncol. 2015;8:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 96. | Shih JW, Wang LY, Hung CL, Kung HJ, Hsieh CL. Non-Coding RNAs in Castration-Resistant Prostate Cancer: Regulation of Androgen Receptor Signaling and Cancer Metabolism. Int J Mol Sci. 2015;16:28943-28978. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 97. | Xu S, Wang L, Zhao Y, Mo T, Wang B, Lin J, Yang H. Metabolism-regulating non-coding RNAs in breast cancer: roles, mechanisms and clinical applications. J Biomed Sci. 2024;31:25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 98. | Lin Z, Lu S, Xie X, Yi X, Huang H. Noncoding RNAs in drug-resistant pancreatic cancer: A review. Biomed Pharmacother. 2020;131:110768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (3)] |
| 99. | Yang Z, Zhao N, Cui J, Wu H, Xiong J, Peng T. Exosomes derived from cancer stem cells of gemcitabine-resistant pancreatic cancer cells enhance drug resistance by delivering miR-210. Cell Oncol (Dordr). 2020;43:123-136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 155] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 100. | Georgikou C, Yin L, Gladkich J, Xiao X, Sticht C, Torre C, Gretz N, Gross W, Schäfer M, Karakhanova S, Herr I. Inhibition of miR30a-3p by sulforaphane enhances gap junction intercellular communication in pancreatic cancer. Cancer Lett. 2020;469:238-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 101. | Meng Q, Liang C, Hua J, Zhang B, Liu J, Zhang Y, Wei M, Yu X, Xu J, Shi S. A miR-146a-5p/TRAF6/NF-kB p65 axis regulates pancreatic cancer chemoresistance: functional validation and clinical significance. Theranostics. 2020;10:3967-3979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 126] [Article Influence: 21.0] [Reference Citation Analysis (4)] |
| 102. | Wang P, Zhuang L, Zhang J, Fan J, Luo J, Chen H, Wang K, Liu L, Chen Z, Meng Z. The serum miR-21 level serves as a predictor for the chemosensitivity of advanced pancreatic cancer, and miR-21 expression confers chemoresistance by targeting FasL. Mol Oncol. 2013;7:334-345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 165] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 103. | Park JK, Lee EJ, Esau C, Schmittgen TD. Antisense inhibition of microRNA-21 or -221 arrests cell cycle, induces apoptosis, and sensitizes the effects of gemcitabine in pancreatic adenocarcinoma. Pancreas. 2009;38:e190-e199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 232] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 104. | Jalil AT, Abdulhadi MA, Al-Ameer LR, Abbas HA, Merza MS, Zabibah RS, Fadhil AA. The emerging role of microRNA-126 as a potential therapeutic target in cancer: a comprehensive review. Pathol Res Pract. 2023;248:154631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 105. | Yu Q, Xiu Z, Jian Y, Zhou J, Chen X, Chen X, Chen C, Chen H, Yang S, Yin L, Zeng W. microRNA-497 prevents pancreatic cancer stem cell gemcitabine resistance, migration, and invasion by directly targeting nuclear factor kappa B 1. Aging (Albany NY). 2022;14:5908-5924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (4)] |
| 106. | Ye ZQ, Chen HB, Zhang TY, Chen Z, Tian L, Gu DN. MicroRNA-7 modulates cellular senescence to relieve gemcitabine resistance by targeting PARP1/NF-κB signaling in pancreatic cancer cells. Oncol Lett. 2021;21:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 107. | Xu Y, Qin Y, Cui JX, Xu J. MicroRNA-136-5p regulates gemcitabine resistance in pancreatic cancer via down-regulating ZNF32. Eur Rev Med Pharmacol Sci. 2020;24:10472-10482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 108. | Liu A, Zhou Y, Zhao T, Tang X, Zhou B, Xu J. MiRNA-3662 reverses the gemcitabine resistance in pancreatic cancer through regulating the tumor metabolism. Cancer Chemother Pharmacol. 2021;88:343-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 109. | Ouyang L, Liu RD, Lei DQ, Shang QC, Li HF, Hu XG, Zheng H, Jin G. MiR-499a-5p promotes 5-FU resistance and the cell proliferation and migration through activating PI3K/Akt signaling by targeting PTEN in pancreatic cancer. Ann Transl Med. 2021;9:1798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 110. | Tu MJ, Duan Z, Liu Z, Zhang C, Bold RJ, Gonzalez FJ, Kim EJ, Yu AM. MicroRNA-1291-5p Sensitizes Pancreatic Carcinoma Cells to Arginine Deprivation and Chemotherapy through the Regulation of Arginolysis and Glycolysis. Mol Pharmacol. 2020;98:686-694. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 111. | Meijer LL, Garajová I, Caparello C, Le Large TYS, Frampton AE, Vasile E, Funel N, Kazemier G, Giovannetti E. Plasma miR-181a-5p Downregulation Predicts Response and Improved Survival After FOLFIRINOX in Pancreatic Ductal Adenocarcinoma. Ann Surg. 2020;271:1137-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 112. | Carotenuto P, Amato F, Lampis A, Rae C, Hedayat S, Previdi MC, Zito D, Raj M, Guzzardo V, Sclafani F, Lanese A, Parisi C, Vicentini C, Said-Huntingford I, Hahne JC, Hallsworth A, Kirkin V, Young K, Begum R, Wotherspoon A, Kouvelakis K, Azevedo SX, Michalarea V, Upstill-Goddard R, Rao S, Watkins D, Starling N, Sadanandam A, Chang DK, Biankin AV, Jamieson NB, Scarpa A, Cunningham D, Chau I, Workman P, Fassan M, Valeri N, Braconi C. Modulation of pancreatic cancer cell sensitivity to FOLFIRINOX through microRNA-mediated regulation of DNA damage. Nat Commun. 2021;12:6738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 113. | Yang Q, Li K, Huang X, Zhao C, Mei Y, Li X, Jiao L, Yang H. lncRNA SLC7A11-AS1 Promotes Chemoresistance by Blocking SCF(β-TRCP)-Mediated Degradation of NRF2 in Pancreatic Cancer. Mol Ther Nucleic Acids. 2020;19:974-985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 114. | Li Z, Zhao X, Zhou Y, Liu Y, Zhou Q, Ye H, Wang Y, Zeng J, Song Y, Gao W, Zheng S, Zhuang B, Chen H, Li W, Li H, Li H, Fu Z, Chen R. The long non-coding RNA HOTTIP promotes progression and gemcitabine resistance by regulating HOXA13 in pancreatic cancer. J Transl Med. 2015;13:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 205] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 115. | Zhou C, Yi C, Yi Y, Qin W, Yan Y, Dong X, Zhang X, Huang Y, Zhang R, Wei J, Ali DW, Michalak M, Chen XZ, Tang J. LncRNA PVT1 promotes gemcitabine resistance of pancreatic cancer via activating Wnt/β-catenin and autophagy pathway through modulating the miR-619-5p/Pygo2 and miR-619-5p/ATG14 axes. Mol Cancer. 2020;19:118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 279] [Cited by in RCA: 312] [Article Influence: 52.0] [Reference Citation Analysis (6)] |
| 116. | Graham M, Adams JM. Chromosome 8 breakpoint far 3' of the c-myc oncogene in a Burkitt's lymphoma 2;8 variant translocation is equivalent to the murine pvt-1 locus. EMBO J. 1986;5:2845-2851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 102] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 117. | You L, Chang D, Du HZ, Zhao YP. Genome-wide screen identifies PVT1 as a regulator of Gemcitabine sensitivity in human pancreatic cancer cells. Biochem Biophys Res Commun. 2011;407:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 118. | Wolpin BM, Rizzato C, Kraft P, Kooperberg C, Petersen GM, Wang Z, Arslan AA, Beane-Freeman L, Bracci PM, Buring J, Canzian F, Duell EJ, Gallinger S, Giles GG, Goodman GE, Goodman PJ, Jacobs EJ, Kamineni A, Klein AP, Kolonel LN, Kulke MH, Li D, Malats N, Olson SH, Risch HA, Sesso HD, Visvanathan K, White E, Zheng W, Abnet CC, Albanes D, Andreotti G, Austin MA, Barfield R, Basso D, Berndt SI, Boutron-Ruault MC, Brotzman M, Büchler MW, Bueno-de-Mesquita HB, Bugert P, Burdette L, Campa D, Caporaso NE, Capurso G, Chung C, Cotterchio M, Costello E, Elena J, Funel N, Gaziano JM, Giese NA, Giovannucci EL, Goggins M, Gorman MJ, Gross M, Haiman CA, Hassan M, Helzlsouer KJ, Henderson BE, Holly EA, Hu N, Hunter DJ, Innocenti F, Jenab M, Kaaks R, Key TJ, Khaw KT, Klein EA, Kogevinas M, Krogh V, Kupcinskas J, Kurtz RC, LaCroix A, Landi MT, Landi S, Le Marchand L, Mambrini A, Mannisto S, Milne RL, Nakamura Y, Oberg AL, Owzar K, Patel AV, Peeters PH, Peters U, Pezzilli R, Piepoli A, Porta M, Real FX, Riboli E, Rothman N, Scarpa A, Shu XO, Silverman DT, Soucek P, Sund M, Talar-Wojnarowska R, Taylor PR, Theodoropoulos GE, Thornquist M, Tjønneland A, Tobias GS, Trichopoulos D, Vodicka P, Wactawski-Wende J, Wentzensen N, Wu C, Yu H, Yu K, Zeleniuch-Jacquotte A, Hoover R, Hartge P, Fuchs C, Chanock SJ, Stolzenberg-Solomon RS, Amundadottir LT. Genome-wide association study identifies multiple susceptibility loci for pancreatic cancer. Nat Genet. 2014;46:994-1000. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 311] [Cited by in RCA: 278] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 119. | Liu Y, Xia L, Dong L, Wang J, Xiao Q, Yu X, Zhu H. CircHIPK3 Promotes Gemcitabine (GEM) Resistance in Pancreatic Cancer Cells by Sponging miR-330-5p and Targets RASSF1. Cancer Manag Res. 2020;12:921-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 76] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 120. | Zeng Z, Zhao Y, Chen Q, Zhu S, Niu Y, Ye Z, Hu P, Chen D, Xu P, Chen J, Hu C, Hu Y, Xu F, Tang J, Wang F, Han S, Huang M, Wang C, Zhao G. Hypoxic exosomal HIF-1α-stabilizing circZNF91 promotes chemoresistance of normoxic pancreatic cancer cells via enhancing glycolysis. Oncogene. 2021;40:5505-5517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 122] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 121. | Hong C, Lishan W, Peng X, Zhengqing L, Yang Y, Fangfang H, Zeqian Y, Zhangjun C, Jiahua Z. Hsa_circ_0074298 promotes pancreatic cancer progression and resistance to gemcitabine by sponging miR-519 to target SMOC. J Cancer. 2022;13:34-50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 122. | Chen ZW, Hu JF, Wang ZW, Liao CY, Kang FP, Lin CF, Huang Y, Huang L, Tian YF, Chen S. Circular RNA circ-MTHFD1L induces HR repair to promote gemcitabine resistance via the miR-615-3p/RPN6 axis in pancreatic ductal adenocarcinoma. J Exp Clin Cancer Res. 2022;41:153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 123. | Hu C, Xia R, Zhang X, Li T, Ye Y, Li G, He R, Li Z, Lin Q, Zheng S, Chen R. circFARP1 enables cancer-associated fibroblasts to promote gemcitabine resistance in pancreatic cancer via the LIF/STAT3 axis. Mol Cancer. 2022;21:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 112] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 124. | Lu Y, Zhou S, Cheng G, Ruan Y, Tian Y, Lv K, Han S, Zhou X. CircLMTK2 Silencing Attenuates Gemcitabine Resistance in Pancreatic Cancer by Sponging miR-485-5p and to Target PAK1. J Oncol. 2022;2022:1911592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 125. | Amrutkar M, Berg K, Balto A, Skilbrei MG, Finstadsveen AV, Aasrum M, Gladhaug IP, Verbeke CS. Pancreatic stellate cell-induced gemcitabine resistance in pancreatic cancer is associated with LDHA- and MCT4-mediated enhanced glycolysis. Cancer Cell Int. 2023;23:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 126. | Miller DW, Fontain M, Kolar C, Lawson T. The expression of multidrug resistance-associated protein (MRP) in pancreatic adenocarcinoma cell lines. Cancer Lett. 1996;107:301-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 127. | Garcia PL, Miller AL, Yoon KJ. Patient-Derived Xenograft Models of Pancreatic Cancer: Overview and Comparison with Other Types of Models. Cancers (Basel). 2020;12:1327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 128. | Driehuis E, van Hoeck A, Moore K, Kolders S, Francies HE, Gulersonmez MC, Stigter ECA, Burgering B, Geurts V, Gracanin A, Bounova G, Morsink FH, Vries R, Boj S, van Es J, Offerhaus GJA, Kranenburg O, Garnett MJ, Wessels L, Cuppen E, Brosens LAA, Clevers H. Pancreatic cancer organoids recapitulate disease and allow personalized drug screening. Proc Natl Acad Sci U S A. 2019;116:26580-26590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 413] [Cited by in RCA: 355] [Article Influence: 50.7] [Reference Citation Analysis (4)] |
| 129. | Tiriac H, Belleau P, Engle DD, Plenker D, Deschênes A, Somerville TDD, Froeling FEM, Burkhart RA, Denroche RE, Jang GH, Miyabayashi K, Young CM, Patel H, Ma M, LaComb JF, Palmaira RLD, Javed AA, Huynh JC, Johnson M, Arora K, Robine N, Shah M, Sanghvi R, Goetz AB, Lowder CY, Martello L, Driehuis E, LeComte N, Askan G, Iacobuzio-Donahue CA, Clevers H, Wood LD, Hruban RH, Thompson E, Aguirre AJ, Wolpin BM, Sasson A, Kim J, Wu M, Bucobo JC, Allen P, Sejpal DV, Nealon W, Sullivan JD, Winter JM, Gimotty PA, Grem JL, DiMaio DJ, Buscaglia JM, Grandgenett PM, Brody JR, Hollingsworth MA, O'Kane GM, Notta F, Kim E, Crawford JM, Devoe C, Ocean A, Wolfgang CL, Yu KH, Li E, Vakoc CR, Hubert B, Fischer SE, Wilson JM, Moffitt R, Knox J, Krasnitz A, Gallinger S, Tuveson DA. Organoid Profiling Identifies Common Responders to Chemotherapy in Pancreatic Cancer. Cancer Discov. 2018;8:1112-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 942] [Cited by in RCA: 836] [Article Influence: 104.5] [Reference Citation Analysis (3)] |
| 130. | Huang L, Holtzinger A, Jagan I, BeGora M, Lohse I, Ngai N, Nostro C, Wang R, Muthuswamy LB, Crawford HC, Arrowsmith C, Kalloger SE, Renouf DJ, Connor AA, Cleary S, Schaeffer DF, Roehrl M, Tsao MS, Gallinger S, Keller G, Muthuswamy SK. Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell- and patient-derived tumor organoids. Nat Med. 2015;21:1364-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 567] [Cited by in RCA: 613] [Article Influence: 55.7] [Reference Citation Analysis (4)] |
| 131. | Tsai S, McOlash L, Palen K, Johnson B, Duris C, Yang Q, Dwinell MB, Hunt B, Evans DB, Gershan J, James MA. Development of primary human pancreatic cancer organoids, matched stromal and immune cells and 3D tumor microenvironment models. BMC Cancer. 2018;18:335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 351] [Cited by in RCA: 315] [Article Influence: 39.4] [Reference Citation Analysis (4)] |
| 132. | Tuveson D, Clevers H. Cancer modeling meets human organoid technology. Science. 2019;364:952-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 809] [Cited by in RCA: 677] [Article Influence: 96.7] [Reference Citation Analysis (3)] |
| 133. | Yu J, Xue Y, Gao Z, Hu L, Liu X, He X, Wang X. α-Asarone attenuates tumor-associated macrophages-induced gemcitabine resistance in pancreatic carcinoma via the transforming growth factor-beta 1/growth factor independent 1 axis. Anticancer Drugs. 2025;36:664-674. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 134. | Qiu F, Chen J, Cao J, Diao F, Huang P. Lowintensity lowfrequency ultrasound enhances the chemosensitivity of gemcitabineresistant ASPC1 cells via PI3K/AKT/NFκB pathwaymediated ABC transporters. Oncol Rep. 2020;44:1158-1168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 135. | Golan T, Atias D, Stossel C, Raitses-Gurevich M. Patient-derived xenograft models of BRCA-associated pancreatic cancers. Adv Drug Deliv Rev. 2021;171:257-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 136. | Westphalen CB, Olive KP. Genetically engineered mouse models of pancreatic cancer. Cancer J. 2012;18:502-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 99] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 137. | Steiner S, Pliego-Mendieta A, Haberecker M, Hussung S, Kollár A, Fritsch R, Arnold F, Lenggenhager D, Planas-Paz L, Pauli C. Ex vivo modeling of acquired drug resistance in BRAF - mutated pancreatic cancer organoids uncovers individual therapeutic vulnerabilities. Cancer Lett. 2024;584:216650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 138. | Kasuya K, Tsuchida A, Nagakawa Y, Suzuki M, Abe Y, Itoi T, Serizawa H, Nagao T, Shimazu M, Aoki T. Hypoxia-inducible factor-1α expression and gemcitabine chemotherapy for pancreatic cancer. Oncol Rep. 2011;26:1399-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 139. | Qi R, Bai Y, Li K, Liu N, Xu Y, Dal E, Wang Y, Lin R, Wang H, Liu Z, Li X, Wang X, Shi B. Cancer-associated fibroblasts suppress ferroptosis and induce gemcitabine resistance in pancreatic cancer cells by secreting exosome-derived ACSL4-targeting miRNAs. Drug Resist Updat. 2023;68:100960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Reference Citation Analysis (0)] |
| 140. | Bravo L. Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev. 1998;56:317-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2333] [Cited by in RCA: 1841] [Article Influence: 65.8] [Reference Citation Analysis (0)] |
| 141. | Harborne JB, Williams CA. Advances in flavonoid research since 1992. Phytochemistry. 2000;55:481-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2979] [Cited by in RCA: 1989] [Article Influence: 76.5] [Reference Citation Analysis (0)] |
| 142. | Cheynier V. Polyphenols in foods are more complex than often thought. Am J Clin Nutr. 2005;81:223S-229S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 370] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 143. | Tsao R. Chemistry and biochemistry of dietary polyphenols. Nutrients. 2010;2:1231-1246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1919] [Cited by in RCA: 1277] [Article Influence: 79.8] [Reference Citation Analysis (4)] |
| 144. | Hewlings SJ, Kalman DS. Curcumin: A Review of Its Effects on Human Health. Foods. 2017;6:92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2112] [Cited by in RCA: 1479] [Article Influence: 164.3] [Reference Citation Analysis (4)] |
| 145. | Basha R, Connelly SF, Sankpal UT, Nagaraju GP, Patel H, Vishwanatha JK, Shelake S, Tabor-Simecka L, Shoji M, Simecka JW, El-Rayes B. Small molecule tolfenamic acid and dietary spice curcumin treatment enhances antiproliferative effect in pancreatic cancer cells via suppressing Sp1, disrupting NF-kB translocation to nucleus and cell cycle phase distribution. J Nutr Biochem. 2016;31:77-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 146. | Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007;4:807-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4538] [Cited by in RCA: 3682] [Article Influence: 193.8] [Reference Citation Analysis (0)] |
| 147. | Kanai M, Yoshimura K, Asada M, Imaizumi A, Suzuki C, Matsumoto S, Nishimura T, Mori Y, Masui T, Kawaguchi Y, Yanagihara K, Yazumi S, Chiba T, Guha S, Aggarwal BB. A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer Chemother Pharmacol. 2011;68:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 282] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 148. | Sharma RA, Euden SA, Platton SL, Cooke DN, Shafayat A, Hewitt HR, Marczylo TH, Morgan B, Hemingway D, Plummer SM, Pirmohamed M, Gescher AJ, Steward WP. Phase I clinical trial of oral curcumin: biomarkers of systemic activity and compliance. Clin Cancer Res. 2004;10:6847-6854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1033] [Cited by in RCA: 903] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 149. | Bar-Sela G, Epelbaum R, Schaffer M. Curcumin as an anti-cancer agent: review of the gap between basic and clinical applications. Curr Med Chem. 2010;17:190-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 307] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 150. | Duarte VM, Han E, Veena MS, Salvado A, Suh JD, Liang LJ, Faull KF, Srivatsan ES, Wang MB. Curcumin enhances the effect of cisplatin in suppression of head and neck squamous cell carcinoma via inhibition of IKKβ protein of the NFκB pathway. Mol Cancer Ther. 2010;9:2665-2675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 151. | Jutooru I, Chadalapaka G, Lei P, Safe S. Inhibition of NFkappaB and pancreatic cancer cell and tumor growth by curcumin is dependent on specificity protein down-regulation. J Biol Chem. 2010;285:25332-25344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 150] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 152. | Penumathsa SV, Maulik N. Resveratrol: a promising agent in promoting cardioprotection against coronary heart disease. Can J Physiol Pharmacol. 2009;87:275-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 78] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 153. | Athar M, Back JH, Tang X, Kim KH, Kopelovich L, Bickers DR, Kim AL. Resveratrol: a review of preclinical studies for human cancer prevention. Toxicol Appl Pharmacol. 2007;224:274-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 553] [Cited by in RCA: 475] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 154. | Xin R, Shen B, Huang ZY, Liu JB, Li S, Jiang GX, Zhang J, Cao YH, Zou DZ, Li W, Li CG, Ma YS, Fu D. Research Progress in Elucidating the Mechanisms Underlying Resveratrol Action on Lung Cancer. Curr Pharm Biotechnol. 2023;24:427-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 155. | Shamim U, Hanif S, Albanyan A, Beck FW, Bao B, Wang Z, Banerjee S, Sarkar FH, Mohammad RM, Hadi SM, Azmi AS. Resveratrol-induced apoptosis is enhanced in low pH environments associated with cancer. J Cell Physiol. 2012;227:1493-1500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 156. | Harikumar KB, Kunnumakkara AB, Sethi G, Diagaradjane P, Anand P, Pandey MK, Gelovani J, Krishnan S, Guha S, Aggarwal BB. Resveratrol, a multitargeted agent, can enhance antitumor activity of gemcitabine in vitro and in orthotopic mouse model of human pancreatic cancer. Int J Cancer. 2010;127:257-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 122] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 157. | Fulda S, Debatin KM. Sensitization for tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by the chemopreventive agent resveratrol. Cancer Res. 2004;64:337-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 158. | Wei R, Penso NEC, Hackman RM, Wang Y, Mackenzie GG. Epigallocatechin-3-Gallate (EGCG) Suppresses Pancreatic Cancer Cell Growth, Invasion, and Migration partly through the Inhibition of Akt Pathway and Epithelial-Mesenchymal Transition: Enhanced Efficacy when Combined with Gemcitabine. Nutrients. 2019;11:1856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 159. | Wei R, Hackman RM, Wang Y, Mackenzie GG. Targeting Glycolysis with Epigallocatechin-3-Gallate Enhances the Efficacy of Chemotherapeutics in Pancreatic Cancer Cells and Xenografts. Cancers (Basel). 2019;11:1496. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 160. | Bimonte S, Leongito M, Barbieri A, Del Vecchio V, Barbieri M, Albino V, Piccirillo M, Amore A, Di Giacomo R, Nasto A, Granata V, Petrillo A, Arra C, Izzo F. Inhibitory effect of (-)-epigallocatechin-3-gallate and bleomycin on human pancreatic cancer MiaPaca-2 cell growth. Infect Agent Cancer. 2015;10:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 161. | Agati G, Brunetti C, Fini A, Gori A, Guidi L, Landi M, Sebastiani F, Tattini M. Are Flavonoids Effective Antioxidants in Plants? Twenty Years of Our Investigation. Antioxidants (Basel). 2020;9:1098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 149] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 162. | Di Pietro A, Conseil G, Pérez-Victoria JM, Dayan G, Baubichon-Cortay H, Trompier D, Steinfels E, Jault JM, de Wet H, Maitrejean M, Comte G, Boumendjel A, Mariotte AM, Dumontet C, McIntosh DB, Goffeau A, Castanys S, Gamarro F, Barron D. Modulation by flavonoids of cell multidrug resistance mediated by P-glycoprotein and related ABC transporters. Cell Mol Life Sci. 2002;59:307-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 164] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 163. | Kopustinskiene DM, Jakstas V, Savickas A, Bernatoniene J. Flavonoids as Anticancer Agents. Nutrients. 2020;12:457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1070] [Cited by in RCA: 718] [Article Influence: 119.7] [Reference Citation Analysis (1)] |
| 164. | Harwood M, Danielewska-Nikiel B, Borzelleca JF, Flamm GW, Williams GM, Lines TC. A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem Toxicol. 2007;45:2179-2205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 575] [Cited by in RCA: 622] [Article Influence: 32.7] [Reference Citation Analysis (3)] |
| 165. | Hu Y, Li R, Jin J, Wang Y, Ma R. Quercetin improves pancreatic cancer chemo-sensitivity by regulating oxidative-inflammatory networks. J Food Biochem. 2022;46:e14453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 166. | Liu ZJ, Xu W, Han J, Liu QY, Gao LF, Wang XH, Li XL. Quercetin induces apoptosis and enhances gemcitabine therapeutic efficacy against gemcitabine-resistant cancer cells. Anticancer Drugs. 2020;31:684-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 167. | Gupta S, Afaq F, Mukhtar H. Selective growth-inhibitory, cell-cycle deregulatory and apoptotic response of apigenin in normal versus human prostate carcinoma cells. Biochem Biophys Res Commun. 2001;287:914-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 203] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 168. | Gradolatto A, Basly JP, Berges R, Teyssier C, Chagnon MC, Siess MH, Canivenc-Lavier MC. Pharmacokinetics and metabolism of apigenin in female and male rats after a single oral administration. Drug Metab Dispos. 2005;33:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 127] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 169. | Cardona F, Andrés-Lacueva C, Tulipani S, Tinahones FJ, Queipo-Ortuño MI. Benefits of polyphenols on gut microbiota and implications in human health. J Nutr Biochem. 2013;24:1415-1422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1370] [Cited by in RCA: 1098] [Article Influence: 84.5] [Reference Citation Analysis (3)] |
| 170. | Johnson JL, de Mejia EG. Flavonoid apigenin modified gene expression associated with inflammation and cancer and induced apoptosis in human pancreatic cancer cells through inhibition of GSK-3β/NF-κB signaling cascade. Mol Nutr Food Res. 2013;57:2112-2127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 171. | Johnson JL, Gonzalez de Mejia E. Interactions between dietary flavonoids apigenin or luteolin and chemotherapeutic drugs to potentiate anti-proliferative effect on human pancreatic cancer cells, in vitro. Food Chem Toxicol. 2013;60:83-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 67] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 172. | Kuang Y, Li B, Wang Z, Qiao X, Ye M. Terpenoids from the medicinal mushroom Antrodia camphorata: chemistry and medicinal potential. Nat Prod Rep. 2021;38:83-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 173. | Huang M, Lu JJ, Huang MQ, Bao JL, Chen XP, Wang YT. Terpenoids: natural products for cancer therapy. Expert Opin Investig Drugs. 2012;21:1801-1818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 207] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 174. | Jiang Z, Jacob JA, Loganathachetti DS, Nainangu P, Chen B. β-Elemene: Mechanistic Studies on Cancer Cell Interaction and Its Chemosensitization Effect. Front Pharmacol. 2017;8:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 175. | Xu HB, Li L, Fu J, Mao XP, Xu LZ. Reversion of multidrug resistance in a chemoresistant human breast cancer cell line by β-elemene. Pharmacology. 2012;89:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 176. | Yardley DA. nab-Paclitaxel mechanisms of action and delivery. J Control Release. 2013;170:365-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 379] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 177. | Tehfe M, Dowden S, Kennecke H, El-Maraghi R, Lesperance B, Couture F, Letourneau R, Liu H, Romano A. nab-Paclitaxel Plus Gemcitabine Versus Gemcitabine in Patients with Metastatic Pancreatic Adenocarcinoma: Canadian Subgroup Analysis of the Phase 3 MPACT Trial. Adv Ther. 2016;33:747-759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 178. | Ma N, Zhang Z, Liao F, Jiang T, Tu Y. The birth of artemisinin. Pharmacol Ther. 2020;216:107658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 221] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 179. | Zeng ZW, Chen D, Chen L, He B, Li Y. A comprehensive overview of Artemisinin and its derivatives as anticancer agents. Eur J Med Chem. 2023;247:115000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 58] [Reference Citation Analysis (0)] |
| 180. | Zhou ZH, Chen FX, Xu WR, Qian H, Sun LQ, Lü XT, Chen L, Zhang J, Ji HC, Fei SJ. Enhancement effect of dihydroartemisinin on human γδ T cell proliferation and killing pancreatic cancer cells. Int Immunopharmacol. 2013;17:850-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 181. | Wang SJ, Gao Y, Chen H, Kong R, Jiang HC, Pan SH, Xue DB, Bai XW, Sun B. Dihydroartemisinin inactivates NF-kappaB and potentiates the anti-tumor effect of gemcitabine on pancreatic cancer both in vitro and in vivo. Cancer Lett. 2010;293:99-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 135] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 182. | Kong R, Jia G, Cheng ZX, Wang YW, Mu M, Wang SJ, Pan SH, Gao Y, Jiang HC, Dong DL, Sun B. Dihydroartemisinin enhances Apo2L/TRAIL-mediated apoptosis in pancreatic cancer cells via ROS-mediated up-regulation of death receptor 5. PLoS One. 2012;7:e37222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 183. | Bhambhani S, Kondhare KR, Giri AP. Diversity in Chemical Structures and Biological Properties of Plant Alkaloids. Molecules. 2021;26:3374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 402] [Cited by in RCA: 234] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 184. | Liu C, Yang S, Wang K, Bao X, Liu Y, Zhou S, Liu H, Qiu Y, Wang T, Yu H. Alkaloids from Traditional Chinese Medicine against hepatocellular carcinoma. Biomed Pharmacother. 2019;120:109543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 185. | Grycová L, Dostál J, Marek R. Quaternary protoberberine alkaloids. Phytochemistry. 2007;68:150-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 325] [Cited by in RCA: 289] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 186. | Li Z, Wang Y, Xu Q, Ma J, Li X, Yan J, Tian Y, Wen Y, Chen T. Berberine and health outcomes: An umbrella review. Phytother Res. 2023;37:2051-2066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 58] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 187. | Wang A, Yang W, Yang X, Mei X, Hu T, Liang R, Meng D, Yan D. MgAl monolayer hydrotalcite increases the hypoglycemic effect of berberine by enhancing its oral bioavailability. Biomed Pharmacother. 2020;127:110140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (3)] |
| 188. | Kuo CL, Chi CW, Liu TY. The anti-inflammatory potential of berberine in vitro and in vivo. Cancer Lett. 2004;203:127-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 478] [Cited by in RCA: 485] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 189. | Li Z, Geng YN, Jiang JD, Kong WJ. Antioxidant and anti-inflammatory activities of berberine in the treatment of diabetes mellitus. Evid Based Complement Alternat Med. 2014;2014:289264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 164] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 190. | Imenshahidi M, Hosseinzadeh H. Berberine and barberry (Berberis vulgaris): A clinical review. Phytother Res. 2019;33:504-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 241] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 191. | Chang W, Chen L, Hatch GM. Berberine as a therapy for type 2 diabetes and its complications: From mechanism of action to clinical studies. Biochem Cell Biol. 2015;93:479-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 192. | Lan J, Zhao Y, Dong F, Yan Z, Zheng W, Fan J, Sun G. Meta-analysis of the effect and safety of berberine in the treatment of type 2 diabetes mellitus, hyperlipemia and hypertension. J Ethnopharmacol. 2015;161:69-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 319] [Article Influence: 29.0] [Reference Citation Analysis (4)] |
| 193. | Sun Y, Xun K, Wang Y, Chen X. A systematic review of the anticancer properties of berberine, a natural product from Chinese herbs. Anticancer Drugs. 2009;20:757-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 306] [Cited by in RCA: 266] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 194. | Wang Y, Liu Y, Du X, Ma H, Yao J. The Anti-Cancer Mechanisms of Berberine: A Review. Cancer Manag Res. 2020;12:695-702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 138] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 195. | Rauf A, Abu-Izneid T, Khalil AA, Imran M, Shah ZA, Emran TB, Mitra S, Khan Z, Alhumaydhi FA, Aljohani ASM, Khan I, Rahman MM, Jeandet P, Gondal TA. Berberine as a Potential Anticancer Agent: A Comprehensive Review. Molecules. 2021;26:7368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 189] [Article Influence: 37.8] [Reference Citation Analysis (5)] |
| 196. | Issat T, Nowis D, Bil J, Winiarska M, Jakobisiak M, Golab J. Antitumor effects of the combination of cholesterol reducing drugs. Oncol Rep. 2011;26:169-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 197. | Okuno K, Xu C, Pascual-Sabater S, Tokunaga M, Han H, Fillat C, Kinugasa Y, Goel A. Berberine Overcomes Gemcitabine-Associated Chemoresistance through Regulation of Rap1/PI3K-Akt Signaling in Pancreatic Ductal Adenocarcinoma. Pharmaceuticals (Basel). 2022;15:1199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 198. | Neuss N, Johnson IS, Armstrong JG, Jansen CJ. The Vinca Alkaloids. Adv Chemother. 1964;12:133-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 199. | Kumar S, Singh B, Singh R. Catharanthus roseus (L.) G. Don: A review of its ethnobotany, phytochemistry, ethnopharmacology and toxicities. J Ethnopharmacol. 2022;284:114647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 200. | Budman DR. Vinorelbine (Navelbine): a third-generation vinca alkaloid. Cancer Invest. 1997;15:475-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 62] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 201. | Ng JS. Vinflunine: review of a new vinca alkaloid and its potential role in oncology. J Oncol Pharm Pract. 2011;17:209-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 202. | Sharbeen G, McCarroll J, Liu J, Youkhana J, Limbri LF, Biankin AV, Johns A, Kavallaris M, Goldstein D, Phillips PA. Delineating the Role of βIV-Tubulins in Pancreatic Cancer: βIVb-Tubulin Inhibition Sensitizes Pancreatic Cancer Cells to Vinca Alkaloids. Neoplasia. 2016;18:753-764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 203. | Schultz AG. Camptothecin. Chem Rev. 1973;73:385-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 93] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 204. | Kamle M, Pandhi S, Mishra S, Barua S, Kurian A, Mahato DK, Rasane P, Büsselberg D, Kumar P, Calina D, Sharifi-Rad J. Camptothecin and its derivatives: Advancements, mechanisms and clinical potential in cancer therapy. Med Oncol. 2024;41:263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 205. | Bailly C. Irinotecan: 25 years of cancer treatment. Pharmacol Res. 2019;148:104398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 340] [Article Influence: 48.6] [Reference Citation Analysis (2)] |
| 206. | Thomas A, Pommier Y. Targeting Topoisomerase I in the Era of Precision Medicine. Clin Cancer Res. 2019;25:6581-6589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 249] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 207. | Wang-Gillam A, Li CP, Bodoky G, Dean A, Shan YS, Jameson G, Macarulla T, Lee KH, Cunningham D, Blanc JF, Hubner RA, Chiu CF, Schwartsmann G, Siveke JT, Braiteh F, Moyo V, Belanger B, Dhindsa N, Bayever E, Von Hoff DD, Chen LT; NAPOLI-1 Study Group. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): a global, randomised, open-label, phase 3 trial. Lancet. 2016;387:545-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1017] [Cited by in RCA: 897] [Article Influence: 89.7] [Reference Citation Analysis (0)] |
| 208. | Rodriguez Lanzi C, Wei R, Luo D, Mackenzie GG. Phospho-Aspirin (MDC-22) inhibits pancreatic cancer growth in patient-derived tumor xenografts and KPC mice by targeting EGFR: Enhanced efficacy in combination with irinotecan. Neoplasia. 2022;24:133-144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 209. | Lu Y, Zhang M, Huang D. Dietary Organosulfur-Containing Compounds and Their Health-Promotion Mechanisms. Annu Rev Food Sci Technol. 2022;13:287-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 210. | Petropoulos S, Di Gioia F, Ntatsi G. Vegetable Organosulfur Compounds and their Health Promoting Effects. Curr Pharm Des. 2017;23:2850-2875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 211. | Alrumaihi F, Khan MA, Babiker AY, Alsaweed M, Azam F, Allemailem KS, Almatroudi AA, Ahamad SR, Alsugoor MH, Alharbi KN, Almansour NM, Khan A. Lipid-Based Nanoparticle Formulation of Diallyl Trisulfide Chemosensitizes the Growth Inhibitory Activity of Doxorubicin in Colorectal Cancer Model: A Novel In Vitro, In Vivo and In Silico Analysis. Molecules. 2022;27:2192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 212. | Kim DH, Kang DY, Sp N, Jo ES, Rugamba A, Jang KJ, Yang YM. Methylsulfonylmethane Induces Cell Cycle Arrest and Apoptosis, and Suppresses the Stemness Potential of HT-29 Cells. Anticancer Res. 2020;40:5191-5200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 213. | Wargovich MJ. Diallylsulfide and allylmethylsulfide are uniquely effective among organosulfur compounds in inhibiting CYP2E1 protein in animal models. J Nutr. 2006;136:832S-834S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 214. | Amagase H, Petesch BL, Matsuura H, Kasuga S, Itakura Y. Intake of garlic and its bioactive components. J Nutr. 2001;131:955S-962S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 629] [Cited by in RCA: 533] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 215. | El-Saadony MT, Saad AM, Korma SA, Salem HM, Abd El-Mageed TA, Alkafaas SS, Elsalahaty MI, Elkafas SS, Mosa WFA, Ahmed AE, Mathew BT, Albastaki NA, Alkuwaiti AA, El-Tarabily MK, AbuQamar SF, El-Tarabily KA, Ibrahim SA. Garlic bioactive substances and their therapeutic applications for improving human health: a comprehensive review. Front Immunol. 2024;15:1277074. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 40] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 216. | El-Saber Batiha G, Magdy Beshbishy A, G Wasef L, Elewa YHA, A Al-Sagan A, Abd El-Hack ME, Taha AE, M Abd-Elhakim Y, Prasad Devkota H. Chemical Constituents and Pharmacological Activities of Garlic (Allium sativum L.): A Review. Nutrients. 2020;12:872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 368] [Cited by in RCA: 371] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 217. | Zhou Y, Li X, Luo W, Zhu J, Zhao J, Wang M, Sang L, Chang B, Wang B. Allicin in Digestive System Cancer: From Biological Effects to Clinical Treatment. Front Pharmacol. 2022;13:903259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (4)] |
| 218. | Longati P, Jia X, Eimer J, Wagman A, Witt MR, Rehnmark S, Verbeke C, Toftgård R, Löhr M, Heuchel RL. 3D pancreatic carcinoma spheroids induce a matrix-rich, chemoresistant phenotype offering a better model for drug testing. BMC Cancer. 2013;13:95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 299] [Cited by in RCA: 290] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 219. | Khakbaz P, Panahizadeh R, Vatankhah MA, Najafzadeh N. Allicin Reduces 5-fluorouracil-resistance in Gastric Cancer Cells through Modulating MDR1, DKK1, and WNT5A Expression. Drug Res (Stuttg). 2021;71:448-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 220. | Zou X, Liang J, Sun J, Hu X, Lei L, Wu D, Liu L. Allicin sensitizes hepatocellular cancer cells to anti-tumor activity of 5-fluorouracil through ROS-mediated mitochondrial pathway. J Pharmacol Sci. 2016;131:233-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (3)] |
| 221. | Țigu AB, Toma VA, Moț AC, Jurj A, Moldovan CS, Fischer-Fodor E, Berindan-Neagoe I, Pârvu M. The Synergistic Antitumor Effect of 5-Fluorouracil Combined with Allicin against Lung and Colorectal Carcinoma Cells. Molecules. 2020;25:1947. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (4)] |
| 222. | Otoo RA, Allen AR. Sulforaphane's Multifaceted Potential: From Neuroprotection to Anticancer Action. Molecules. 2023;28:6902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 223. | Georgikou C, Buglioni L, Bremerich M, Roubicek N, Yin L, Gross W, Sticht C, Bolm C, Herr I. Novel Broccoli Sulforaphane-Based Analogues Inhibit the Progression of Pancreatic Cancer without Side Effects. Biomolecules. 2020;10:769. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 224. | Cho Y, Park MN, Choi M, Upadhyay TK, Kang HN, Oh JM, Min S, Yang JU, Kong M, Ko SG, Rahman MA, Harrath AH, Kim B. Sulforaphane regulates cell proliferation and induces apoptotic cell death mediated by ROS-cell cycle arrest in pancreatic cancer cells. Front Oncol. 2024;14:1442737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 225. | Zhao T, Yang M, Ma L, Liu X, Ding Q, Chai G, Lu Y, Wei H, Zhang S, Ding C. Structural Modification and Biological Activity of Polysaccharides. Molecules. 2023;28:5416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 84] [Reference Citation Analysis (0)] |
| 226. | Dedhia N, Marathe SJ, Singhal RS. Food polysaccharides: A review on emerging microbial sources, bioactivities, nanoformulations and safety considerations. Carbohydr Polym. 2022;287:119355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 227. | Chen F, Huang G. Preparation and immunological activity of polysaccharides and their derivatives. Int J Biol Macromol. 2018;112:211-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 201] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 228. | Nagaraj AS, Lahtela J, Hemmes A, Pellinen T, Blom S, Devlin JR, Salmenkivi K, Kallioniemi O, Mäyränpää MI, Närhi K, Verschuren EW. Cell of Origin Links Histotype Spectrum to Immune Microenvironment Diversity in Non-small-Cell Lung Cancer Driven by Mutant Kras and Loss of Lkb1. Cell Rep. 2017;18:673-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 229. | Feng B, Zhu Y, Su Z, Tang L, Sun C, Li C, Zheng G. Basil polysaccharide attenuates hepatocellular carcinoma metastasis in rat by suppressing H3K9me2 histone methylation under hepatic artery ligation-induced hypoxia. Int J Biol Macromol. 2018;107:2171-2179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 230. | Zhang CH, Yang X, Wei JR, Chen NM, Xu JP, Bi YQ, Yang M, Gong X, Li ZY, Ren K, Han QH, Zhang L, Li X, Ji MY, Wang CC, Li MH. Ethnopharmacology, Phytochemistry, Pharmacology, Toxicology and Clinical Applications of Radix Astragali. Chin J Integr Med. 2021;27:229-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 231. | Jin M, Zhao K, Huang Q, Shang P. Structural features and biological activities of the polysaccharides from Astragalus membranaceus. Int J Biol Macromol. 2014;64:257-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 238] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 232. | Zheng Y, Ren W, Zhang L, Zhang Y, Liu D, Liu Y. A Review of the Pharmacological Action of Astragalus Polysaccharide. Front Pharmacol. 2020;11:349. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 292] [Article Influence: 48.7] [Reference Citation Analysis (0)] |
| 233. | Li CX, Liu Y, Zhang YZ, Li JC, Lai J. Astragalus polysaccharide: a review of its immunomodulatory effect. Arch Pharm Res. 2022;45:367-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 204] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 234. | Li GY, Jiang J. Recent efficacy and long-term survival of Astragalus polysaccharide combined with gemcitabine and S-1 in pancreatic cancer. World J Clin Oncol. 2024;15:1404-1411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 235. | Wu J, Wang J, Su Q, Ding W, Li T, Yu J, Cao B. Traditional Chinese medicine Astragalus polysaccharide enhanced antitumor effects of the angiogenesis inhibitor apatinib in pancreatic cancer cells on proliferation, invasiveness, and apoptosis. Onco Targets Ther. 2018;11:2685-2698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (4)] |
| 236. | Sheng K, Wang C, Chen B, Kang M, Wang M, Liu K, Wang M. Recent advances in polysaccharides from Lentinus edodes (Berk.): Isolation, structures and bioactivities. Food Chem. 2021;358:129883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 100] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 237. | Meng Y, Lyu F, Xu X, Zhang L. Recent Advances in Chain Conformation and Bioactivities of Triple-Helix Polysaccharides. Biomacromolecules. 2020;21:1653-1677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 230] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 238. | Maeda YY, Chihara G. Lentinan, a new immuno-accelerator of cell-mediated responses. Nature. 1971;229:634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 100] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 239. | Akramiene D, Kondrotas A, Didziapetriene J, Kevelaitis E. Effects of beta-glucans on the immune system. Medicina (Kaunas). 2007;43:597-606. [PubMed] [DOI] [Full Text] |
| 240. | Ina K, Kataoka T, Ando T. The use of lentinan for treating gastric cancer. Anticancer Agents Med Chem. 2013;13:681-688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 140] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 241. | Sun M, Zhao W, Xie Q, Zhan Y, Wu B. Lentinan reduces tumor progression by enhancing gemcitabine chemotherapy in urothelial bladder cancer. Surg Oncol. 2015;24:28-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 242. | Sun M, Bu R, Zhang B, Cao Y, Liu C, Zhao W. Lentinan Inhibits Tumor Progression by Immunomodulation in a Mouse Model of Bladder Cancer. Integr Cancer Ther. 2020;19:1534735420946823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 243. | Kubota E, Kataoka H, Hayashi K, Kamiya T, Sasaki M, Ogasawara N, Yamada T, Wada T, Mori Y, Mizoshita T, Shimura T, Mizushima T, Okamoto Y, Ohara H, Joh T. Advanced stomach and pancreas cancer successfully treated with combination chemotherapy with S-1/paclitaxel/lentinan. Hepatogastroenterology. 2009;56:106-110. [PubMed] |
| 244. | Qutub M, Hussain UM, Tatode A, Premchandani T, Khan R, Umekar M, Taksande J, Singanwad P. Nano-Engineered Epigallocatechin Gallate (EGCG) Delivery Systems: Overcoming Bioavailability Barriers to Unlock Clinical Potential in Cancer Therapy. AAPS PharmSciTech. 2025;26:137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 245. | Alizadeh SR, Ebrahimzadeh MA. Quercetin derivatives: Drug design, development, and biological activities, a review. Eur J Med Chem. 2022;229:114068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 179] [Article Influence: 35.8] [Reference Citation Analysis (0)] |
| 246. | Li B, Hu Y, Wu T, Feng Y, Jiang C, Du H, Lu S. Apigenin-oxymatrine binary co-amorphous mixture: Enhanced solubility, bioavailability, and anti-inflammatory effect. Food Chem. 2022;373:131485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 247. | Dong X, Wang X, Zheng X, Jiang H, Liu L, Ma N, Wang S. Targeted nanoparticle delivery system for tumor-associated macrophage reprogramming to enhance TNBC therapy. Cell Biol Toxicol. 2025;41:58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 248. | Luo G, Li X, Lin J, Ge G, Fang J, Song W, Xiao GG, Zhang B, Peng X, Duo Y, Tang BZ. Multifunctional Calcium-Manganese Nanomodulator Provides Antitumor Treatment and Improved Immunotherapy via Reprogramming of the Tumor Microenvironment. ACS Nano. 2023;17:15449-15465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 71] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 249. | Kunnumakkara AB, Guha S, Krishnan S, Diagaradjane P, Gelovani J, Aggarwal BB. Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-kappaB-regulated gene products. Cancer Res. 2007;67:3853-3861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 443] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 250. | Arlt A, Schäfer H, Kalthoff H. The 'N-factors' in pancreatic cancer: functional relevance of NF-κB, NFAT and Nrf2 in pancreatic cancer. Oncogenesis. 2012;1:e35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 251. | Tang Z, Huang G. Extraction, structure, and activity of polysaccharide from Radix astragali. Biomed Pharmacother. 2022;150:113015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 101] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 252. | Dei Cas M, Ghidoni R. Dietary Curcumin: Correlation between Bioavailability and Health Potential. Nutrients. 2019;11:2147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 388] [Article Influence: 55.4] [Reference Citation Analysis (0)] |
| 253. | Walle T. Bioavailability of resveratrol. Ann N Y Acad Sci. 2011;1215:9-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 881] [Cited by in RCA: 756] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 254. | Dabeek WM, Marra MV. Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans. Nutrients. 2019;11:2288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 381] [Cited by in RCA: 553] [Article Influence: 79.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/