Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114580
Revised: November 10, 2025
Accepted: January 19, 2026
Published online: March 7, 2026
Processing time: 153 Days and 17.2 Hours
Biologic therapies, including anti-tumor necrosis factor agents, have significantly improved the management of inflammatory bowel disease (IBD). However, their high cost can limit patient access. Biosimilars are a more affordable option than originator biologics and offer potential for improved accessibility, though real-world evidence comparing their effectiveness remains limited.
To describe real-world, comparative outcomes of IBD patients taking biologics and biosimilars, focusing on remi
We used data from a multicenter registry-based cohort, which includes participants from six Canadian IBD clinical centers. Adults with ulcerative colitis or Crohn’s disease who initiated biosimilar, or originator formulations of infliximab or adalimumab were included. The primary outcome was clinical remission; secondary outcomes included hospitalizations and emergency department (ED) visits. Kaplan-Meier survival analyses and multiva
A total of 258 individuals were analyzed (192 biosimilar initiators and 66 originator users). The median time to remission was similar between biosimilars (12.2 months) and originators (12.8 months). We did not detect diffe
This study suggests biosimilars and originators have similar remission, ED visits, and hospitalization in IBD. This reinforces confidence in their equivalence, improving access while supporting healthcare system sustainability.
Core Tip: Biologic therapies are effective for many people with inflammatory bowel disease, but they are expensive. Biosimilars are equivalent, lower-cost versions of biologic originators, yet real-world evidence comparing their effectiveness remains limited. We analyzed data from a Canadian multicenter registry including adults living with Crohn’s disease or ulcerative colitis, initiating originator or biosimilar. Outcomes of interest were remission and health care resource use. We found no clear differences in remission, hospitalizations, or emergency visits between the two groups. These findings support broader adoption of biosimilars and provide reassurance to patients and clinicians regarding their comparable effectiveness.
- Citation: Moura CS, Etingin A, Lukusa L, Singh H, Narula N, Targownik LE, Leung Y, Zezos P, Polewiczowska-Nowak B, Afif W, Bernatsky S. Comparable remission and health care use in real-world inflammatory bowel disease patients initiating originator biologics vs biosimilars. World J Gastroenterol 2026; 32(9): 114580
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/114580.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.114580
Inflammatory bowel disease (IBD), including Crohn's disease (CD) and ulcerative colitis (UC), is characterized by chronic inflammation of the gastrointestinal tract, resulting in symptoms such as abdominal pain, diarrhea, and fatigue. Due to its chronic and often severe nature, IBD often leads to a reduced quality of life and is also associated with hospitalizations and emergency department (ED) visits[1,2].
The goal of IBD treatment, including immunosuppressives and biologic therapies, is to achieve and maintain remission[3], which reduces the risk of disease complications, hospitalizations, and ED visits[4]. The introduction of biologic therapies, such as anti-tumor necrosis factor (TNF) agents, has significantly advanced IBD management, providing patients with the potential for sustained remission and an enhanced quality of life[5]. However, the high cost of biologics remains a significant barrier to their widespread adoption, limiting access for many patients and placing a financial strain on healthcare systems[1,6].
Biosimilars, which have structural and functional similarities to an already approved biologic (the ‘originator’), have emerged as a cost-effective alternative, with the potential to reduce healthcare expenditures and improve access to biologic therapies[7]. Despite their growing use, real-world evidence comparing the effectiveness of originator biologics and biosimilars in achieving remission and reducing hospitalizations and ED visits remains scarce.
Our objective was to evaluate time to remission, hospitalization and ED visit rates among people with IBD initiating originator TNF biologics or biosimilars. We hypothesized that biosimilars would be non-inferior to originator for these outcomes.
The CAnadian Network for Advanced Interdisciplinary Methods (CAN-AIM) biosimilar registry is an ongoing, multi-cohort initiative providing real-world data on originator biologics and biosimilars use. Established in 2018, the registry collects data from new and existing clinical cohorts, led by Canadian IBD Research Consortium (CIRC) investigators.
Participants in our cohort were prospectively enrolled during routine clinical assessment at six Canadian centres (Montreal, Vancouver, Winnipeg, Thunder Bay, Toronto, and Hamilton). We required individuals with established CD or UC to have either initiated or switched to either biosimilar or originator infliximab or adalimumab. Infliximab and adalimumab were the only biologics with approved biosimilar drugs in Canada during the study period.
Data in the CIRC cohort were captured using web-based standardized data entry forms. During the coronavirus disease 2019 pandemic (2020-2022), enrollment and follow-up visits were completed virtually; otherwise, data was collected in person. Data was cleaned and managed at the Research Institute of the McGill University Health Centre (the Institute).
Upon enrollment, participants underwent baseline assessments for disease activity, which were measured using the Partial Mayo Score (PMS), Harvey-Bradshaw Index (HBI), or Crohn's Disease Activity Index (CDAI). Baseline data collection included demographics (age, sex, race/ethnicity), smoking, disease duration, comorbidities (congestive heart failure, diabetes, hypertension, coronary artery disease, malignancy, renal failure), body mass index, past year hospitalizations, and current or past (12-month period before enrollment) IBD-related medications (corticosteroids, 5-aminosalicylates, 5-aminosalicylates, methotrexate, azathioprine).
After enrollment, participants were followed approximately every three months during the first year and every six months thereafter. Follow-up assessments included disease activity, medication use (including reasons for discontinuation, if applicable), and hospitalizations or ED visits occurring any time since the previous assessment.
This analysis included adult individuals (≥ 18 years old) diagnosed with IBD who started infliximab originator (IFX-O), infliximab biosimilar (IFX-B), adalimumab originator (ADA-O), or adalimumab biosimilar (ADA-B) between January 2018 and May 2024. Participants were required to have at least one follow-up visit to be included in the analysis. Because this was a pragmatic, registry-based study, the number of eligible patients was determined by all participants meeting the inclusion criteria within the CAN-AIM Biosimilar Registry during the study period. No formal a priori sample size or power calculation was performed, as all available data were included to maximize representativeness and precision of estimates.
The primary outcome was first clinical remission during follow-up defined as PMS score < 2 for UC, or CDAI < 150 or HBI < 5 for CD. The secondary outcomes included first hospitalization or ED visit after initiating infliximab therapy.
Baseline patient characteristics were summarized using descriptive statistics for the overall population and stratified by the biologic type at baseline (originator or biosimilar). Categorical variables were reported as absolute and relative frequencies, while continuous variables were summarized as mean ± SD or medians with interquartile ranges (IQR), depending on data distribution.
Steroid-sparing was explored descriptively by comparing the proportion of patients in each group with any corticosteroid use at baseline (within 12 months before biologic initiation) and within the first 12 months of follow-up among patients with available 12-month visit data.
Kaplan-Meier methods were used to estimate the probability of achieving the first remission and the occurrence of the first hospital or ED visit during the follow-up period. For the remission analysis, only individuals with sufficient disease activity score measures during follow-up were included. For all analyses, follow-up ended at the occurrence of the event of interest or was censored at the earliest occurrence of death, discontinuation of biosimilar/originator, loss to follow-up, or study end. Estimates were accompanied by 95% confidence interval (95%CI) and stratified by the type of biologic at enrollment (biosimilar or originator). Differences between survival curves were evaluated using the log-rank test.
Multivariate Cox proportional hazards models were adjusted to evaluate the association between the biologic type and (1) Remission; (2) Hospitalization; and (3) ED visit. All models were adjusted for the following baseline covariates: Age at IBD diagnosis, sex, IBD duration, moderate/high disease activity (PMS score ≥ 5 for UC, CDAI > 220 or HBI > 8 for CD), IBD diagnosis (CD vs UC), presence of one or more comorbidity, and current/previous use of prednisone or methotrexate/azathioprine. The remission model was further adjusted by considering past use of other biologics (biologic-naïve vs biologic-experienced individuals). For models where hospitalization or ED visit was the outcome, each model was additionally adjusted for the corresponding prior event (i.e., previous hospitalizations were included in the hospitalization model, and previous ED visits were included in the ED visit model) based on events occurring within the six months prior to enrolment. Both ED and hospitalization models were also adjusted for smoking. Results were presented as hazard ratios (HRs) with corresponding 95%CI. In sensitivity analyses, additional Cox regression models were fitted within the following subgroups: (1) Prior biologic exposure (naïve vs switchers); (2) IBD type (UC vs CD); (3) Prior hospitalization (yes vs no); and (4) Current/previous corticosteroid use (yes vs no). All analyses were conducted using R, version 4.4.3.
We studied 258 individuals, including 192 biosimilar initiators (157 IFX-B and 35 ADA-B) and 66 originator users (38 IFX-O and 28 ADA-O; Supplementary Figure 1). Just over half were male (51.2%) and the median age at study start was 38.3 years (IQR: 28.0-52.7; Table 1). At baseline, 34 individuals (13.2%) were biologic-experienced and 95 (36.8%) had previously been exposed to methotrexate and/or azathioprine. The median post-index follow-up period was 12 months (IQR: 4.4-27.0).
| Originator, n = 66 | Biosimilar, n = 192 | Overall, n = 258 | |
| Biologic at enrollment | |||
| Adalimumab | 28 (42.4) | 35 (18.2) | 63 (24.4) |
| Infliximab | 38 (57.6) | 157 (81.8) | 195 (75.6) |
| Age in years at IBD diagnosis, median (IQR) | 27.1 (19.0, 38.6) | 26.7 (20.1, 37.1) | 26.9 (19.9, 37.7) |
| Age in years at biologic initiation, median (IQR) | 37.3 (27.4, 55.3) | 38.7 (28.5, 52.3) | 38.3 (28.0, 52.7) |
| Sex | |||
| Female | 36 (54.5) | 96 (50.0) | 132 (51.2) |
| Male | 30 (45.5) | 96 (50.0) | 126 (48.8) |
| Race/ethnicity | |||
| Caucasian | 56 (84.8) | 158 (82.3) | 214 (82.9) |
| Others | 8 (12.1) | 30 (15.6) | 38 (14.7) |
| Missing | 2 (3.0) | 4 (2.1) | 6 (2.3) |
| Education | |||
| Secondary or less | 19 (28.8) | 55 (28.6) | 74 (28.7) |
| College or university | 43 (65.2) | 134 (69.8) | 177 (68.6) |
| Missing | 4 (6.1) | 3 (1.6) | 7 (2.7) |
| IBD diagnosis | |||
| CD | 44 (66.7) | 118 (61.5) | 162 (62.8) |
| UC | 22 (33.3) | 74 (38.5) | 96 (37.2) |
| Moderate or high disease activity1 | 26 (39.4) | 77 (40.1) | 103 (39.9) |
| At least one co-morbidity2 | 24 (36.4) | 68 (35.4) | 92 (35.7) |
| Smoking status at enrollment | |||
| Current smoker | 6 (9.1) | 18 (9.4) | 24 (9.3) |
| Ex-smoker | 16 (24.2) | 48 (25.0) | 64 (24.8) |
| Never smoker | 44 (66.7) | 126 (65.6) | 170 (65.9) |
| BMI (mean ± SD) | 25.8 (5.4) | 25.8 (5.8) | 25.8 (5.7) |
| Site | |||
| HHSC, Ontario | 35 (53.0) | 32 (16.7) | 67 (26.0) |
| McGill, Quebec | 13 (19.7) | 46 (24.0) | 59 (22.9) |
| MSH, Ontario | 2 (3.0) | 6 (3.1) | 8 (3.1) |
| PHCR, British Columbia | 0 (0.0) | 29 (15.1) | 29 (11.2) |
| TBRHSC, Ontario | 1 (1.5) | 13 (6.8) | 14 (5.4) |
| WRHA, Manitoba | 15 (22.7) | 66 (34.4) | 81 (31.4) |
| IBD duration at enrollment in years, (mean ± SD) | 9.6 ± 10.9 | 10.6 ± 10.9 | 10.3 ± 10.9 |
| Drug use at enrollment or in the 12-month period before | |||
| Other biologics | 15 (22.7) | 19 (9.9) | 34 (13.2) |
| Immunomodulators3 | 32 (48.5) | 63 (32.8) | 95 (36.8) |
| 5-ASA | 17 (25.7) | 49 (25.5) | 66 (25.6) |
| Corticosteroids | 33 (50.0) | 90 (46.9) | 123 (47.7) |
The proportion of patients using corticosteroids decreased in both groups: Among originator users, from 56.1% at baseline to 41.7% within 12 months; and among biosimilar users, from 52.6% to 23.7% over the same period.
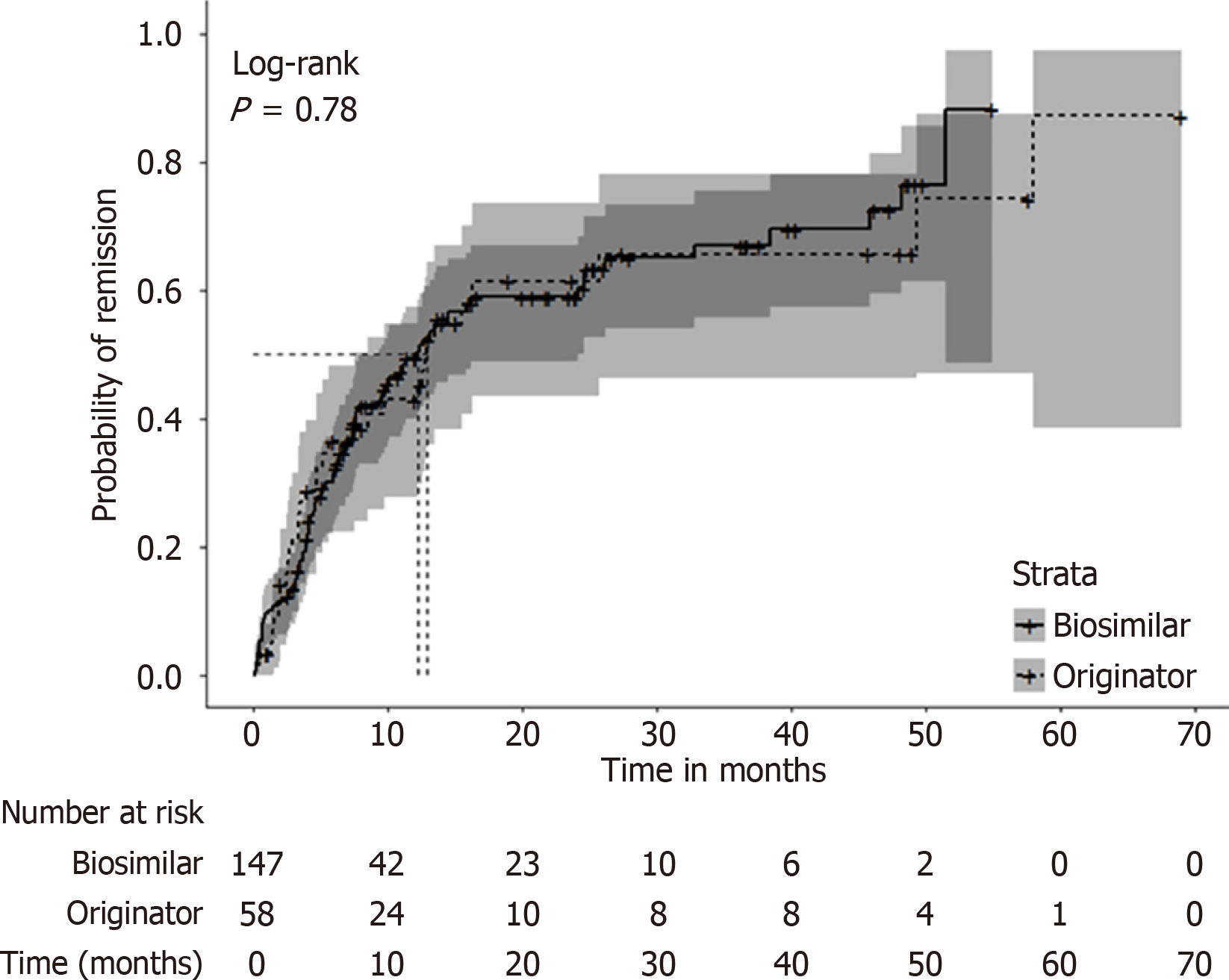
With 53 individuals already in remission at baseline, our remission analysis included the remaining 205 participants. The median time to clinical remission was 12.8 months (95%CI: 7.5-23.8 months) for originator users and 12.2 months (95%CI: 9.3-24.1 months) for biosimilar users. No significant difference in the time to achieve remission between biosimilar and originator users was detected (Figure 1). In the adjusted Cox regression model (Table 2), there was no clear difference in the likelihood of achieving remission when comparing biosimilar and originator treatments [adjusted HR (aHR): 1.49; 95%CI: 0.96-2.32]. This model showed that UC disease and moderate or high disease activity were associated with a longer time to achieving clinical remission.
| Characteristic | Unadjusted HR (95%CI) | Adjusted HR (95%CI) |
| Biosimilar at enrollment | 1.06 (0.70-1.60) | 1.49 (0.96-2.32) |
| Female sex | 0.96 (0.67-1.38) | 0.96 (0.66-1.40) |
| Age at IBD diagnosis | 0.99 (0.98-1.00) | 0.99 (0.98-1.01) |
| IBD duration at biologic start | 1.00 (0.98-1.02) | 0.98 (0.96-1.00) |
| UC disease | 0.50 (0.34-0.75) | 0.35 (0.22-0.55) |
| Moderate or high disease activity1 | 0.51 (0.35-0.74) | 0.36 (0.23-0.54) |
| Previous use of biologic2 | 1.10 (0.66-1.84) | 1.67 (0.93-2.99) |
| Corticosteroid use3 | 0.80 (0.55-1.16) | 1.00 (0.68-1.59) |
| Immunomodulator use4 | 1.40 (0.97-2.02) | 1.44 (0.97-2.13) |
| One or more comorbidity5 | 1.02 (0.71-1.48) | 1.41 (0.93-2.14) |
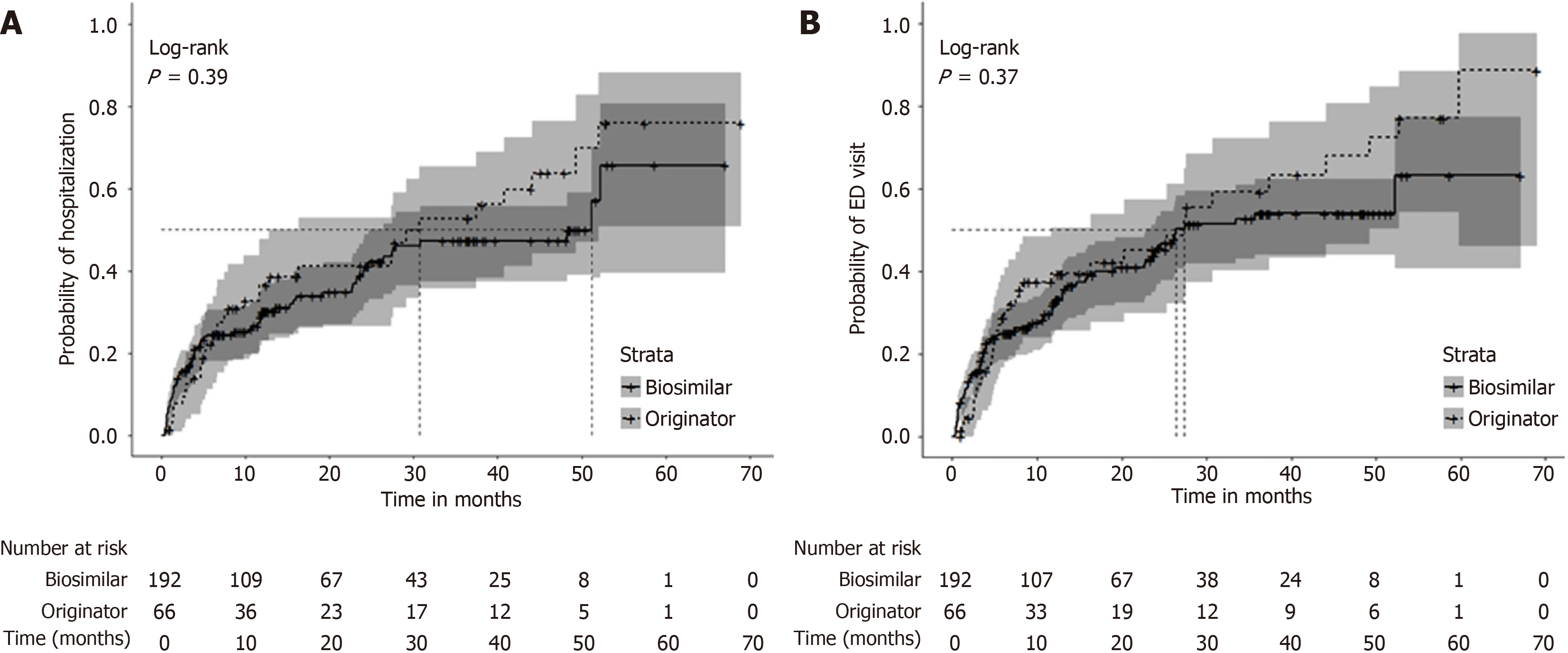
There were 106 hospitalizations during follow-up, with a rate of 2.3 events per 100 patient-months (95%CI: 1.9-2.9). Reasons for hospitalization included IBD flares (n = 59), IBD-related surgeries (n = 21), and any infection (n = 16). The median time to first hospitalization estimated by Kaplan-Meier analysis was 30.7 months (95%CI: 12.8-52.1 months) for originator users and 51.2 months (95%CI: 24.6-72.3 months) for biosimilar users with no statistically significant difference between groups (Figure 2A). We did not identify a clear difference in time to hospitalization comparing originator and biosimilar groups (aHR: 0.86, 95%CI: 0.56-1.32). Female sex, baseline systemic corticosteroids, and hospitalization in the 5 months before enrolment were significantly associated with a higher risk of hospitalization (Supplementary Table 1).
There were 116 ED visits during the follow-up, corresponding to 26.4 events per 1000 patient-months (95%CI: 21.8-31.7). Main reasons for ED visits included IBD flares (n = 64), IBD-related surgeries (n = 18) and infections (n = 15). The median time to first ED visit was 27.4 months (95%CI: 11.6-49.3 months) for originator users and 26.4 months (95%CI: 22.6-53.4 months) for biosimilar users (Figure 2B). In the multivariate hazards model, comparing biosimilar vs originator, the aHR for the first ED visit was 0.83 (95%CI: 0.86-1.30). Disease duration was inversely associated with the risk of ED visits, whereas corticosteroid use and ED visit within six months before enrollment was associated with higher risk of ED visits (Supplementary Table 2).
Subgroup analyses by prior biologic exposure, IBD type, prior hospitalization, and current/previous corticosteroid use yielded consistent results, with no significant differences between biosimilar and originator users across all three outcomes (Supplementary Figures 2-4).
In this real-world study, we found that biosimilar users achieved remission and healthcare use (hospital/ED) comparable to those on originator biologic. From a health-system perspective, the comparable safety and efficacy of biosimilars may enhance treatment accessibility and continuity of care while reducing socioeconomic disparities. The resulting cost-containment allows for reallocation of healthcare resources toward other areas of need, reinforcing system capacity and long-term sustainability. In addition, by incorporating data from multiple provinces, our analysis captures important inter-jurisdictional heterogeneity in prescribing practices and offers valuable perspectives on biosimilar uptake and effectiveness within real-world Canadian healthcare systems.
While our findings are consistent with existing evidence, particularly from Europe, our study provides novel insights into the North American context. Overall, our results are aligned with clinical trials and real-world evidence demon
Similarly, we were not able to demonstrate clear differences in rates of hospitalizations and ED visits when comparing the biosimilar and originator groups. Recent real-world data and clinical trials revealed comparable rates of hospitalizations and ED visits between biosimilar and originator biologics in IBD. A 2025 Canadian retrospective study of IBD patients mandated to switch from originator adalimumab to biosimilars reported no significant differences in hospitalization or ED visits (observed only in the originator group) over 30 months of follow-up[16]. These findings align with a 2024 United States Veterans Health Administration study of infliximab-naïve IBD patients, where biosimilars and originator infliximab had similar healthcare use patterns, including ED visits and hospitalizations[17].
Taking together, these results suggest that biosimilars offer a viable first-line option with respect to drug effectiveness and safety in IBD. This is particularly reassuring in the face of growing economic pressure on public healthcare systems to contain drug-related expenditures without compromising care quality. According to the Canadian Institute for Health Information’s 2023 report, biologics remain among the costliest drug classes for Canadian public drug programs, accounting for nearly 30% of total spending, or approximately CAD 4.7 billion in 2022[18]. Although overall spending on biologics has continued to rise in recent years, the uptake of biosimilars has helped to slow this growth. In fact, all Canadian jurisdictions have reported increased biosimilar use over the past few years. For example, British Columbia’s biosimilar-switching initiative (2019-2024), which involved transitioning more than 40000 patients from originators to biosimilars, is estimated to have saved CAD 732 million over five years while maintaining comparable patient outcomes[19]. These findings underscore the dual clinical and economic value of biosimilars.
Despite growing evidence supporting the comparability of biosimilars and originator products, hesitation around their use remains common among healthcare providers, particularly in the context of formulary-driven switches, which can trigger negative perceptions and skepticism toward biosimilars[20,21]. Much of this reluctance stems from persistent misconceptions about how biosimilars are developed and approved, as well as from misinformation or incomplete understanding of their efficacy and safety[22]. Addressing these concerns requires improving access to high-quality, trusted evidence, such as comparative effectiveness studies and patient-reported outcomes, and ensuring it reaches both healthcare professionals and patient advocacy groups. Doing so can promote informed decision-making and help reduce unwarranted hesitation[23].
This study has several strengths. First, it is a prospective, multicentric study utilizing data from the CAN-AIM/CIRC registry, which enhances the generalizability of findings. Minimal exclusion criteria ensure a wide range of patients are included, increasing the external validity of the findings. Moreover, data were collected prospectively using objective measures, minimizing the risk of recall bias, a limitation often present in observational studies. Finally, the inclusion of both intravenous and subcutaneous formulations of TNF inhibitors (specifically biosimilars of infliximab and adali
Several potential limitations should be acknowledged. The small sample size, particularly in the originator group, resulted in relatively wide confidence intervals and limited precision. The numerical imbalance between biosimilar and originator users primarily reflects the policy environment during the study period, when many Canadian provinces were transitioning to mandatory biosimilar initiation or switching. This may have constrained comparability between the two groups. Nevertheless, key prognostic factors for the studied outcome, such as disease activity and duration, were broadly comparable at baseline. Pooling data across different sites, biologic agents, IBD subtypes, and treatment histories (biologic-naïve and biologic-experienced) increased sample size but may have introduced heterogeneity and obscured subgroup-specific effects. However, the inclusion of sites from multiple jurisdictions, each with distinct biosimilar coverage policies and asynchronous adoption timelines, provides valuable insight into real-world biosimilar uptake and effectiveness at the provincial level. Moreover, consistent findings observed for multiple outcomes and confirmed through subgroup analyses support the overall robustness of the results. As with any observational study, unmeasured confounding cannot be entirely excluded, including potential physician- or patient-driven treatment preferences. Nonetheless, because most biosimilar initiations occurred under provincially mandated start or switch policies, the influence of such discretionary factors was likely limited. Finally, the median follow-up of approximately 12 months may be insufficient to fully assess the durability of response or long-term safety and immunogenicity - key considerations in chronic diseases such as IBD.
Looking ahead, several areas warrant further investigation. Long-term follow-up is essential to assess sustained remission, durability of response, and long-term safety profiles, including adverse events. From a policy and global health perspective, cost-effectiveness analyses should also be prioritized to inform resource allocation and reimbursement strategies. As the CAN-AIM registry continues to grow, its expanding sample size will provide greater statistical power to detect subtle but potentially meaningful differences. Future analyses could explore treatment effects across clinically relevant subgroups, including varying disease severity, and the presence of extra-intestinal manifestations, to help tailor treatment decisions more precisely.
This real-world study provides evidence that infliximab biosimilars are comparable to originators in achieving remission and reducing healthcare use among individuals with IBD. These findings, based on data from the CAN-AIM registry, reinforce the therapeutic value of biosimilars and their potential to reduce the financial burden on healthcare systems. Our results support broader adoption of biosimilars in IBD management and highlight the ongoing need for real-world monitoring to inform clinical and policy decision-making.
| 1. | Kuenzig ME, Coward S, Targownik LE, Murthy SK, Benchimol EI, Windsor JW, Bernstein CN, Bitton A, Jones JL, Lee K, Peña-Sánchez JN, Rohatinsky N, Ghandeharian S, Im JHB, Jogendran R, Meka S, Weinstein J, Jones May T, Jogendran M, Tabatabavakili S, Hazan E, Hu M, Osei JA, Khan R, Wang G, Browne M, Davis T, Goddard Q, Gorospe J, Latos K, Mason K, Kerr J, Balche N, Sklar A, Kaplan GG. The 2023 Impact of Inflammatory Bowel Disease in Canada: Direct Health System and Medication Costs. J Can Assoc Gastroenterol. 2023;6:S23-S34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 2. | Ding Z, Patel A, Izanec J, Pericone CD, Lin JH, Baugh CW. Trends in US emergency department visits and subsequent hospital admission among patients with inflammatory bowel disease presenting with abdominal pain: a real-world study from a national emergency department sample database. J Mark Access Health Policy. 2021;9:1912924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 3. | Torres J, Bonovas S, Doherty G, Kucharzik T, Gisbert JP, Raine T, Adamina M, Armuzzi A, Bachmann O, Bager P, Biancone L, Bokemeyer B, Bossuyt P, Burisch J, Collins P, El-Hussuna A, Ellul P, Frei-Lanter C, Furfaro F, Gingert C, Gionchetti P, Gomollon F, González-Lorenzo M, Gordon H, Hlavaty T, Juillerat P, Katsanos K, Kopylov U, Krustins E, Lytras T, Maaser C, Magro F, Marshall JK, Myrelid P, Pellino G, Rosa I, Sabino J, Savarino E, Spinelli A, Stassen L, Uzzan M, Vavricka S, Verstockt B, Warusavitarne J, Zmora O, Fiorino G. ECCO Guidelines on Therapeutics in Crohn's Disease: Medical Treatment. J Crohns Colitis. 2020;14:4-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1167] [Cited by in RCA: 1004] [Article Influence: 167.3] [Reference Citation Analysis (2)] |
| 4. | Khanna R, Bressler B, Levesque BG, Zou G, Stitt LW, Greenberg GR, Panaccione R, Bitton A, Paré P, Vermeire S, D'Haens G, MacIntosh D, Sandborn WJ, Donner A, Vandervoort MK, Morris JC, Feagan BG; REACT Study Investigators. Early combined immunosuppression for the management of Crohn's disease (REACT): a cluster randomised controlled trial. Lancet. 2015;386:1825-1834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 364] [Article Influence: 33.1] [Reference Citation Analysis (1)] |
| 5. | Baumgart DC, Le Berre C. Newer Biologic and Small-Molecule Therapies for Inflammatory Bowel Disease. N Engl J Med. 2021;385:1302-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 6. | Targownik LE, Kaplan GG, Witt J, Bernstein CN, Singh H, Tennakoon A, Aviña Zubieta A, Coward SB, Jones J, Kuenzig ME, Murthy SK, Nguyen GC, Peña-Sánchez JN, Benchimol EI. Longitudinal Trends in the Direct Costs and Health Care Utilization Ascribable to Inflammatory Bowel Disease in the Biologic Era: Results From a Canadian Population-Based Analysis. Am J Gastroenterol. 2020;115:128-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 7. | Burisch J, Claytor J, Hernandez I, Hou JK, Kaplan GG. The Cost of Inflammatory Bowel Disease Care: How to Make it Sustainable. Clin Gastroenterol Hepatol. 2025;23:386-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 58] [Article Influence: 58.0] [Reference Citation Analysis (0)] |
| 8. | Tapete G, Bertani L, Pieraccini A, Lynch EN, Giannotta M, Morganti R, Biviano I, Naldini S, Mumolo MG, De Nigris F, Calella F, Bagnoli S, Minciotti M, Maltinti S, Rentini S, Ceccarelli L, Lionetti P, Milla M, Costa F. Effectiveness and Safety of Nonmedical Switch From Adalimumab Originator to SB5 Biosimilar in Patients With Inflammatory Bowel Diseases: Twelve-Month Follow-Up From the TABLET Registry. Inflamm Bowel Dis. 2022;28:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 9. | Strik AS, van de Vrie W, Bloemsaat-Minekus JPJ, Nurmohamed M, Bossuyt PJJ, Bodelier A, Rispens T, van Megen YJB, D'Haens GR; SECURE study group. Serum concentrations after switching from originator infliximab to the biosimilar CT-P13 in patients with quiescent inflammatory bowel disease (SECURE): an open-label, multicentre, phase 4 non-inferiority trial. Lancet Gastroenterol Hepatol. 2018;3:404-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 10. | Goll GL, Jørgensen KK, Sexton J, Olsen IC, Bolstad N, Haavardsholm EA, Lundin KEA, Tveit KS, Lorentzen M, Berset IP, Fevang BTS, Kalstad S, Ryggen K, Warren DJ, Klaasen RA, Asak Ø, Baigh S, Blomgren IM, Brenna Ø, Bruun TJ, Dvergsnes K, Frigstad SO, Hansen IM, Hatten ISH, Huppertz-Hauss G, Henriksen M, Hoie SS, Krogh J, Midtgard IP, Mielnik P, Moum B, Noraberg G, Poyan A, Prestegård U, Rashid HU, Strand EK, Skjetne K, Seeberg KA, Torp R, Ystrøm CM, Vold C, Zettel CC, Waksvik K, Gulbrandsen B, Hagfors J, Mørk C, Jahnsen J, Kvien TK. Long-term efficacy and safety of biosimilar infliximab (CT-P13) after switching from originator infliximab: open-label extension of the NOR-SWITCH trial. J Intern Med. 2019;285:653-669. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 11. | Jørgensen KK, Olsen IC, Goll GL, Lorentzen M, Bolstad N, Haavardsholm EA, Lundin KEA, Mørk C, Jahnsen J, Kvien TK; NOR-SWITCH study group. Switching from originator infliximab to biosimilar CT-P13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52-week, randomised, double-blind, non-inferiority trial. Lancet. 2017;389:2304-2316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 556] [Cited by in RCA: 605] [Article Influence: 67.2] [Reference Citation Analysis (0)] |
| 12. | Smith JT, Velayos FS, Niu F, Liu V, Delate T, Pola S, Le K, Hui RL. Retrospective Cohort Study Comparing Infliximab-dyyb and Infliximab in Biologic-Naive Patients With Inflammatory Bowel Disease in the United States. Crohns Colitis 360. 2021;3:otab051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Huguet JM, Cortés X, Bosca-Watts MM, Aguas M, Maroto N, Martí L, Amorós C, Paredes JM. Real-world data on the infliximab biosimilar CT-P13 (Remsima(®)) in inflammatory bowel disease. World J Clin Cases. 2021;9:11285-11299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Derikx LAAP, Dolby HW, Plevris N, Lucaciu L, Rees CS, Lyons M, Siakavellas SI, Constantine-Cooke N, Jenkinson P, Su S, O'Hare C, Kirckpatrick L, Merchant LM, Noble C, Arnott ID, Jones GR, Lees CW. Effectiveness and Safety of Adalimumab Biosimilar SB5 in Inflammatory Bowel Disease: Outcomes in Originator to SB5 Switch, Double Biosimilar Switch and Bio-Naïve SB5 Observational Cohorts. J Crohns Colitis. 2021;15:2011-2021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Tursi A, Mocci G, Allegretta L, Aragona G, Bianco MA, Colucci R, Cuomo A, Della Valle N, Ferronato A, Forti G, Gaiani F, Giorgetti G, Graziani MG, Lofano K, Lorenzetti R, Larussa T, Penna A, Pica R, Pranzo G, Rodino' S, Scarcelli A, Zampaletta C, Bassotti G, Cazzato AI, Chiri S, Clemente V, Cocco A, De' Angelis G, Donnarumma L, Faggiani R, Graziosi C, Le Grazie M, Luzza F, Meucci C, Monterubbianesi R, Pagnini C, Perazzo P, Picchio M, Sacco R, Sebkova L, Serio M, Napolitano D, Pugliese D, Scaldaferri F, Schiavoni E, Turchini L, Armuzzi A, Elisei W, Maconi G, Papa A. Comparison of Performances of Adalimumab Biosimilars SB5, ABP501, GP2017, and MSB11022 in Treating Patients with Inflammatory Bowel Diseases: A Real-Life, Multicenter, Observational Study. Inflamm Bowel Dis. 2023;29:376-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Liu Chen Kiow J, Hoang T, Bedi HK, Majdzadeh Ardekani Z, Rosenfeld D, Reise-Filteau M, Bressler B, Leung Y, Rosenfeld G. Real-world experience and long-term outcomes of a mandatory non-medical switch of adalimumab originator to biosimilars in inflammatory bowel disease. World J Gastroenterol. 2024;30:4904-4913. [PubMed] [DOI] [Full Text] |
| 17. | Patel S, Walsh J, Pinnell D, Pei S, Chen W, Rojas J, Rathod A, Johnson J, Gawron A, Curtis JR, Baker JF, Cannon GW, Wu D, Lai M, Sauer BC. Real-world experience with biosimilar infliximab-adba and infliximab-dyyb among infliximab-naïve patients with inflammatory bowel disease in the Veterans Health Administration. Medicine (Baltimore). 2024;103:e39476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (10)] |
| 18. | Canadian Institute for Health Information. Prescribed Drug Spending in Canada, 2023. 2023. [cited 7 July 2025]. Available from: https://www.cihi.ca/en/prescribed-drug-spending-in-canada-2023. |
| 19. | Government of British Columbia. BC Biosimilars Initiative: A Report on Patient and Financial Impact. 2024. [cited 7 July 2025]. Available from: https://news.gov.bc.ca/files/BCBiosimilarsInitiativeAReportonPatientandFinancialImpact_June%202024.pdf. |
| 20. | Kaplan GG, Ma C, Seow CH, Kroeker KI, Panaccione R. The Argument Against a Biosimilar Switch Policy for Infliximab in Patients with Inflammatory Bowel Disease Living in Alberta. J Can Assoc Gastroenterol. 2020;3:234-242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 21. | Moayyedi P, Benchimol EI, Armstrong D, Yuan C, Fernandes A, Leontiadis GI. Joint Canadian Association of Gastroenterology and Crohn's Colitis Canada Position Statement on Biosimilars for the Treatment of Inflammatory Bowel Disease. J Can Assoc Gastroenterol. 2020;3:e1-e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Cohen HP, McCabe D. The Importance of Countering Biosimilar Disparagement and Misinformation. BioDrugs. 2020;34:407-414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 23. | Car E, Vandenplas Y, Lacosta TB, Simoens S, Huys I, Vulto AG, Barbier L. Mitigating the Nocebo Effect in Biosimilar Use and Switching: A Systematic Review. Pharmaceut Med. 2024;38:429-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/