Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114544
Revised: November 6, 2025
Accepted: December 25, 2025
Published online: March 7, 2026
Processing time: 151 Days and 5.3 Hours
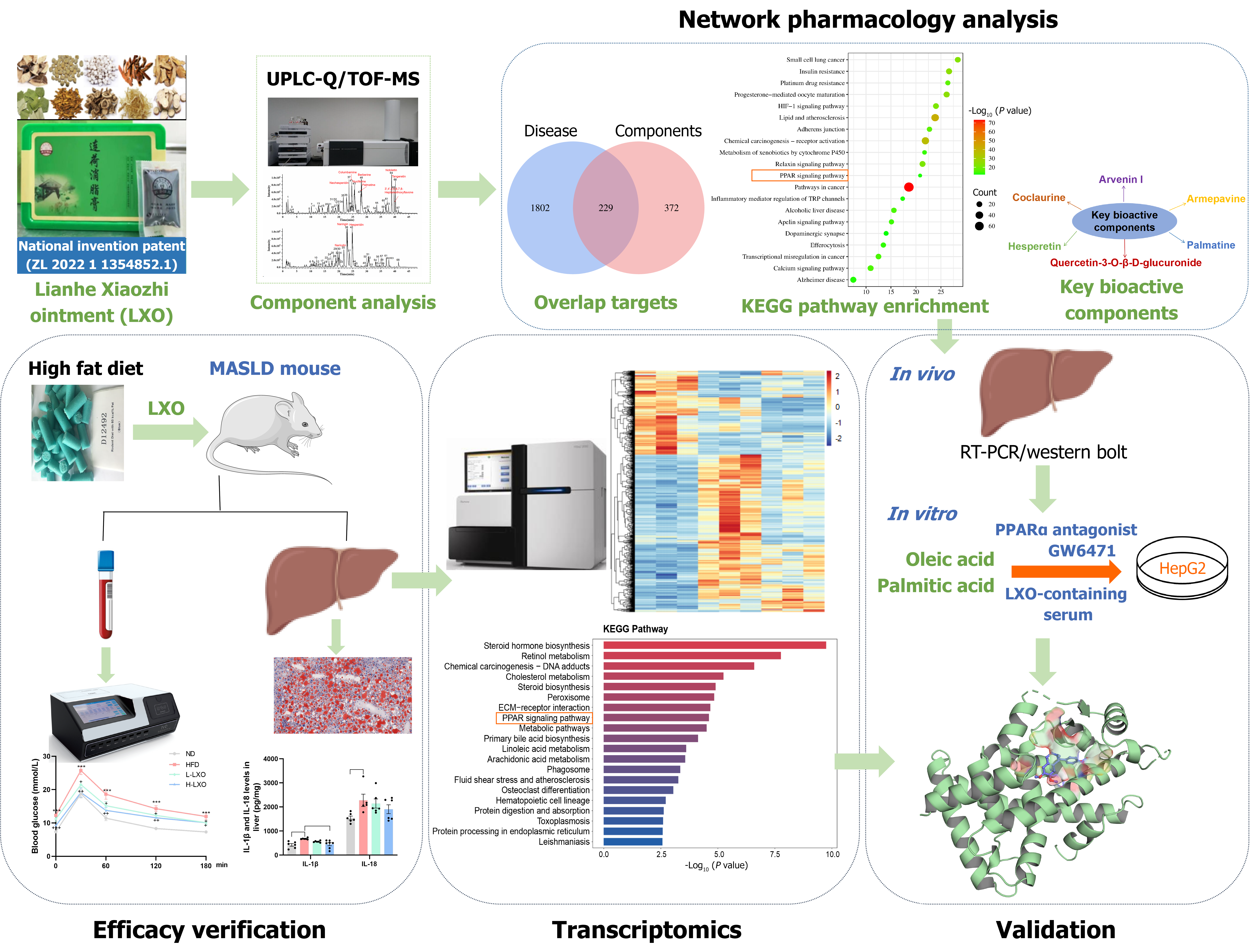
Lianhe Xiaozhi ointment (LXO), an innovative formulation derived from the classic Huanglian Wendan decoction, has been granted a national invention patent. With increasing years of clinical expertise within the Jiangsu Province Hospital of Chinese Medicine in China, LXO has become increasingly recognized as a potent remedy for metabolic disorders, particularly metabolic dysfunction-associated steatotic liver disease (MASLD). To date, specific bioactive components and underlying mechanisms remain unclear.
To determine the bioactive components of LXO and clarify its mechanisms of action relevant to management of MASLD.
We used ultra-high performance liquid chromatography with quadrupole time-of-flight mass spectrometry and network pharmacology approaches to systematically determine the key bioactive components of LXO and elucidate biological pathways modulated in the treatment of MASLD. Critical signaling pathways were also elucidated via hepatic transcriptomic analysis, and an exhaustive and rigorous exploration of the therapeutic efficacy and underlying mechanisms of LXO in MASLD was conducted via a combination of in vivo and in vitro high-fat model experiments.
Network pharmacology analysis revealed six pivotal bioactive components within LXO that collectively serve as the cornerstone for their efficacy against MASLD. LXO exerts multiple therapeutic effects, including weight gain retardation, amelioration of glucose and lipid metabolism disturbances, liver injury mitigation, hepatic inflammation alleviation, and correction of gut microbiota disorders. Multiple platform analyses, including hepatic trans
Collectively, our findings suggest that LXO exerts therapeutic effects on high-fat diet-induced MASLD at least in part via PPARα pathway activation.
Core Tip: Lianhe Xiaozhi ointment effectively alleviates metabolic dysfunction-associated steatotic liver disease by im
- Citation: Nie LJ, Wang GX, Yang XY, Sun J, Cao YT, Lou Y, Lu YF, Yu JY, Zhou XQ. Lianhe Xiaozhi ointment ameliorates metabolic dysfunction-associated steatotic liver disease via peroxisome proliferator-activated receptor alpha pathway activation. World J Gastroenterol 2026; 32(9): 114544
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/114544.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.114544
Metabolic dysfunction-associated steatotic liver disease (MASLD) is a chronic metabolic disorder that is intricately associated with type 2 diabetes, obesity, and hyperlipidemia[1]. Its global prevalence is currently 30% and is projected to reach 55.7% by 2040, imposing a considerable burden on global healthcare systems[2,3]. The complex and multifaceted etiological and pathological mechanisms governing MASLD initiation and progression remain largely unknown. Moreover, insufficient effective therapeutic approaches have been discovered to date[4]. Consequently, there exists an immediate necessity to develop efficacious therapeutic drugs for the management of MASLD.
Peroxisome proliferator-activated receptors (PPARs) serve as the principal regulators of systemic and hepatic meta
In China, traditional Chinese medicines (TCM) is extensively used to treat chronic liver diseases[15,16]. TCM can treat MASLD through the enhancement of lipid metabolism, reduction of hepatic inflammation, improvement of fibrosis, and regulation of the gut microbiota[17]. Lianhe Xiaozhi ointment (LXO), derived from the classic Huanglian Wendan decoction, displays favorable therapeutic efficacy in patients with MASLD[18]. This formulation has been awarded a national invention patent (No. ZL 2022 1 1354852.1). LXO has been clinically prescribed for several years at the Jiangsu Province Hospital of Chinese Medicine to manage patients with insulin-resistant diabetes, MASLD, or obesity. Our cli
The present study employed ultra-high performance liquid chromatography with quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) and network pharmacology to identify core bioactive components and signaling pathways relevant to MASLD treatment modulated by LXO. We also investigated the therapeutic effects of LXO in MASLD mice, explored underlying mechanisms, and validated findings using HepG2 cells. These results highlight the promising therapeutic potential of LXO for MASLD management and suggest a novel approach to developing a defined treatment strategy. An overview of the experimental design is provided in Figure 1.
Assay kits for total cholesterol (TC) (cat. A111-1-1), TG (cat. A110-1-1), non-esterified fatty acids (NEFA) (cat. A042-2-1), alanine aminotransferase (ALT) (cat. C009-2-1), and aspartate aminotransferase (AST) (cat. C010-2-1) were procured from the Nanjing Jiancheng Bioengineering Institute (Nanjing, Jiangsu Province, China). Mouse IL-1β (cat. AF2040-A) and IL-18 (cat. AF2169-A) enzyme-linked immunosorbent assay (ELISA) kits were acquired from Aifang Biological (Changsha, Hunan Province, China). Beta-hydroxybutyrate (BHB) (cat. RXJ99926) and lipopolysaccharide (LPS) (cat. RXJ202425M) ELISA kits were purchased from RUIXIN BIOTECH (Quanzhou, Fujian Province, China). Culture medium optimized for HepG2 cells (cat. CM-0103) was purchased from Wuhan Pricella Biotechnology Co., Ltd. (Wuhan, Hubei Province, China). Sodium palmitate (PA) and sodium oleate (OA) kits (cat. KT002) were procured from Kunchuang Biotechnology (Xi’an, Shaanxi Province, China). GW6471 (cat. HY-15372) was purchased from MedChemExpress LLC (Shanghai, China). Fenofibrate (cat. F6020) was purchased from Merck KGaA (Darmstadt, Germany). An RNA extraction kit (cat. 302031AX) was procured from GENENODE (Wuhan, Hubei Province, China). Cell counting kit 8 kits (cat. G4103) and other reagents were obtained from Servicebio (Wuhan, Hubei Province, China). The details of the antibodies used are provided in Supplementary Table 1.
LXO incorporates ten Chinese herbs: Coptidis rhizoma, Pinelliae rhizoma, bamboo shavings, Pericarpium trichosanthis, tangerine peel, lotus leaf, poria, immature bitter orange, licorice root, and Polygonum cuspidatum. Details of LXO herb use are presented in Table 1. The herbs were procured from Jiangsu Province Hospital of Chinese Medicine and authenticated by professional Chinese pharmacists. To prepare the LXO decoction, the herbs were immersed in water for 1 hour prior to boiling for 2 hours in a programmed electric herbal cooker. The supernatant was decanted and the procedure was repeated after adding fresh water. The two batches of liquid were combined and concentrated to 2.834 g/mL. The recommended human dose is 109 g (for a 70 kg adult). Using a standard conversion factor of 9.1 for mice, this equates to 14.17 g/kg[20]. The low-dose LXO (L-LXO, equivalent dosage) group and the high-dose LXO (H-LXO, 2 × equivalent dosage) group were administered 14.17 g/kg and 28.34 g/kg doses, respectively[20].
| Chinese name | Latin name | Plant part | Usage (g/day) |
| Huanglian | Coptis chinensis Franch. | Rhizoma | 3 |
| Fabanxia | Pinellia ternata (Thunb) Breit. | Tuber | 10 |
| Zhuru | Bambusa tuldoides Munro | Culm shavings | 10 |
| Gualoupi | Trichosanthes kirilowii Maxim. | Pericarpium | 15 |
| Chenpi | Citrus reticulata Blanco | Pericarpium | 10 |
| Heye | Nelumbo nucifera Gaertn. | Leaf | 15 |
| Fuling | Poria cocos (Schw.) Wolf | Sclerotium | 10 |
| Zhishi | Citrus aurantium L. | Fructus immaturus | 15 |
| Gancao | Glycyrrhiza uralensis Fisch. | Root and rhizome | 6 |
| Huzhang | Polygonum cuspidatum Sieb.et Zucc. | Root and rhizome | 15 |
The LXO decoction was first mixed with 50% methanol solution to achieve complete dissolution. Subsequently, the mixture was centrifuged at high speed. The clear supernatant was then harvested for mass spectrometric analysis. The detailed instrumental parameters for the liquid chromatography and mass spectrometry are provided in Supplementary Tables 2 and 3. For compound identification, the mass-spectrometry data were first matched to entries in the natural products high resolution mass spectrometry/mass spectrometry spectral library 1.0. Components were initially subjected to a strict screening procedure using scoring metrics for each chromatographic peak. A meticulous verification step was subsequently performed, involving in-depth examination of molecular ion peaks, fragment ion details, and other relevant indicators. Identification of components not found in the database was accomplished through analysis of mass spectrometry fragmentation patterns and cross-referencing with literature reports.
Relevant targets of LXO decoction components: The active components of the LXO decoction were screened for oral bioavailability and drug-likeness using the TCMSP database (https://old.tcmsp-e.com/tcmsp.php)[21]. Subsequently, the putative targets were refined by retaining only those with a Z-score ≥ 0 from PharmMapper (http://www.lilab-ecust.cn/pharmmapper/) or a probability ≥ 0.1 from SwissTarget (http://swisstargetprediction.ch/). The datasets were subsequently merged and overlapping entries were deduplicated. To guarantee consistency, the UniProt database (https://www.uniprot.org/) was used to obtain gene IDs for all targets.
Therapeutic targets of MASLD: The keywords “nonalcoholic fatty liver disease” and “metabolic dysfunction-associated steatotic liver disease” were respectively input into the DisGeNet (https://www.disgenet.org/) and GeneCards (https://www.genecards.org/) databases. Filtration criteria were implemented to preserve DisGeNet targets with gda scores ≥ 0.1 and GeneCards targets with relevance scores ≥ 10. These datasets were subsequently integrated and duplicate overlapping entries were eliminated.
Network construction and analysis: To identify potential therapeutic targets of LXO in MASLD, we integrated its active component targets with known disease targets using a bioinformatics platform (http://www.bioinformatics.com.cn/). The resulting common targets were subsequently analyzed using the STRING database (https://string-db.org) under default settings for “homo sapiens” to generate a protein-protein interaction (PPI) network. To elucidate the mechanism of LXO in MASLD, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analyses were conducted using the Metascape platform (https://www.metascape.org) under stringent criteria (min overlap = 3, min enrichment = 1.5, P < 0.01). The key active components were further screened employing degree, betweenness, and closeness centrality of the network topology.
After the 2-dimensional structures of key active components were obtained from PubChem database (https://pubchem.ncbi.nlm.nih.gov) and optimized into 3-dimensional models, molecular docking was performed to assess their binding affinity with PPARα. The PPARα structure (PDB: 3VI8) was sourced from the Research Collaboratory for Structural Bioinformatics Protein Data Bank and converted, along with the ligands, into the PDBQT format with the addition of polar hydrogens. The docking grid was established as a 48 Å × 50 Å × 50 Å cubic pocket (center: 12.095, 5.473, -7.416; grid spacing: 0.05 nm). Autodock Vina 1.2.2 was employed to predict the binding poses and energies of the protein-ligand complexes. The resulting interactions were comprehensively analyzed using PLIP and LigPlus, and the structural details were visualized using PyMOL 2.5.
A cohort of 48 male C57BL/6J mice was procured from GemPharmatech Co., Ltd. (Nanjing, Jiangsu Province, China) and housed in a specific pathogen-free level experimental animal center. Using stratified randomization by body weight, mice were allocated to receive either a normal diet (ND) (n = 12) or a high-fat diet (HFD) (Research Diets; cat. D12492, n = 36). Following an 8-week HFD feeding period, the mice were randomly assigned to three subgroups: A L-LXO group (n = 12, 14.17 g/kg/day), an H-LXO group (n = 12, 28.34 g/kg/day), and an HFD control group (n = 12). Each group was provided an equivalent volume of drinking water. Each therapeutic regimen was administered daily for a duration of 12 weeks. Throughout the treatment phase, mouse body weight, water intake, food consumption, behavioral patterns, and mental states were meticulously observed and systematically recorded. After humane euthanasia, blood, liver tissue, and cecal contents were harvested from the animals, and the blood was centrifuged to isolate serum for storage at -80 °C. All experiments adhered to the World Medical Association Statement regarding the use of animals in biomedical research and were approved by the Ethics Committee of Nanjing University of Chinese Medicine (No. 202212A024; approval date: January 11, 2023).
HepG2 cells were sourced from Servicebio (Wuhan, Hubei Province, China) and cultivated in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin. Subsequently, a high-fat condition was established by treating the cells with a free fatty acid (FFA) combination (OA: PA = 2:1). The culture duration was approximately 48 hours after the addition of FFA- and H-LXO-derived serum.
Mice were orally administered a 2 g/kg glucose solution and fasting blood glucose (FBG) levels were precisely measured at 0, 30, 60, 120, and 180 minutes after administration. The area under the curve for the oral glucose tolerance test (AUCOGTT) was calculated based on the collected data.
Following manufacturer’s instructions, detection kits were used to determine serum ALT, AST, TG, TC, LPS, and BHB levels. A fully automatic biochemical analyzer was utilized to quantify the serum concentrations of high-density lipoprotein cholesterol and low-density lipoprotein cholesterol. Liver tissue was precisely weighed and nine volumes of physiological saline (9 mL/g) were added. The mixture was mechanically homogenized in an ice-water bath to yield a 10% homogenate. Following centrifugation of the homogenate, the supernatant was harvested and quantified for IL-18, IL-1β, and NEFA.
Following standard processing, liver tissues were used for two staining protocols: Hematoxylin and eosin on 4-μm paraffin sections and Oil red O on 8-μm frozen optimal cutting temperature compound-embedded sections. An Olympus CX23 microscope was used for image documentation.
Total proteins were extracted from liver tissue and HepG2 cells in radio immunoprecipitation assay buffer (supplemented with phenyl methane sulfonyl fluoride), quantified using the bicinchoninic acid assay, and then separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Following transfer to polyvinylidene difluoride membranes and a 30-minute block with a rapid-sealing solution, the membranes were incubated overnight at 4 °C with primary antibodies (Supplementary Table 1). After thorough washing, the membranes were probed with a horseradish pero
Form each group, three liver samples were randomly selected and entrusted to Hangzhou Lianchuan Biotechnology Co., Ltd. for RNA sequencing. The Illumina HiSeqTM sequencing platform was employed for transcriptome sequencing and analysis, which encompassed RNA extraction, library preparation, data evaluation and quality control, transcriptome splicing, fragments per kilobase of exon model per million mapped fragments quantification, sequencing evaluation, differential gene analysis, and gene enrichment analysis.
Following total RNA extraction with TRIzol reagent and quality assessment via a Thermo NanoDrop 2000, complementary DNA (cDNA) was generated using a first strand cDNA synthesis kit (GENENODE, cat. 302447AX). The polymerase chain reaction (PCR) protocol involved an initial 3-minute denaturation at 95 °C, followed by 40 cycles of 10 seconds at 95 °C and 30 seconds at 60 °C. Actb was used as a reference gene to standardize target gene expression. Relative expression levels were computed via the 2-ΔΔCt method. The primers used are listed in Supplementary Table 4.
For comprehensive analysis, we randomly selected six cecal content samples from each group. The entire process, including extraction, library preparation, sequencing, and analysis, was performed by Metabo-Profile Biotechnology Co., Ltd. (Shanghai, China). Cecal content samples (0.2-0.5 g) were collected and transferred into precooled 1.5 mL centrifuge tubes containing extraction lysis buffer, followed by thorough homogenization via a tissue grinder. For 16S rRNA gene amplicon sequencing, bioinformatics, and statistical analyses, genomic DNA was first extracted from the processed samples using an OMEGA Soil DNA kit (cat. D5635-02). Prior to analysis, its quality was confirmed by assessing concentration and purity with a NanoDrop NC2000 and integrity on a 1% agarose gel.
Liver tissue, 1 mL of extraction solution (containing isopropanol, n-hexane, and an internal standard), and steel beads were added to a 2 mL Eppendorf (EP) tube. The mixture was centrifuged after three cycles of vortexing, grinding, and ultrasonication in an ice water bath. Approximately 800 μL of the supernatant was transferred to a new EP tube, dried under nitrogen gas, and then derivatized with 500 μL of a methanol: Trimethylsilyl diazomethane solution (1:2) for 30 minutes. Samples were then dried under nitrogen gas and mixtures were reconstituted with 160 μL n-hexane and centrifuged at high speed. Finally, the supernatants were analyzed by gas chromatography-mass spectrometry. The entire process, including extraction, detection, and analysis, was meticulously performed by Shanghai BIOTREE Biomedical Technology Co., Ltd. (Shanghai, China).
Statistical processing and graph generation were conducted using SPSS 21.0 and GraphPad Prism 8.0, respectively. Differences between the two groups were evaluated using Student’s t-test, while one-way analysis of variance with Tukey’s post-hoc comparison was used to assess differences among multiple groups. Spearman’s correlation analysis was performed to elucidate the complex relationships among gut microbiota, metabolites, and clinical-related characteristics. A P value < 0.05 was regarded as statistically significant.
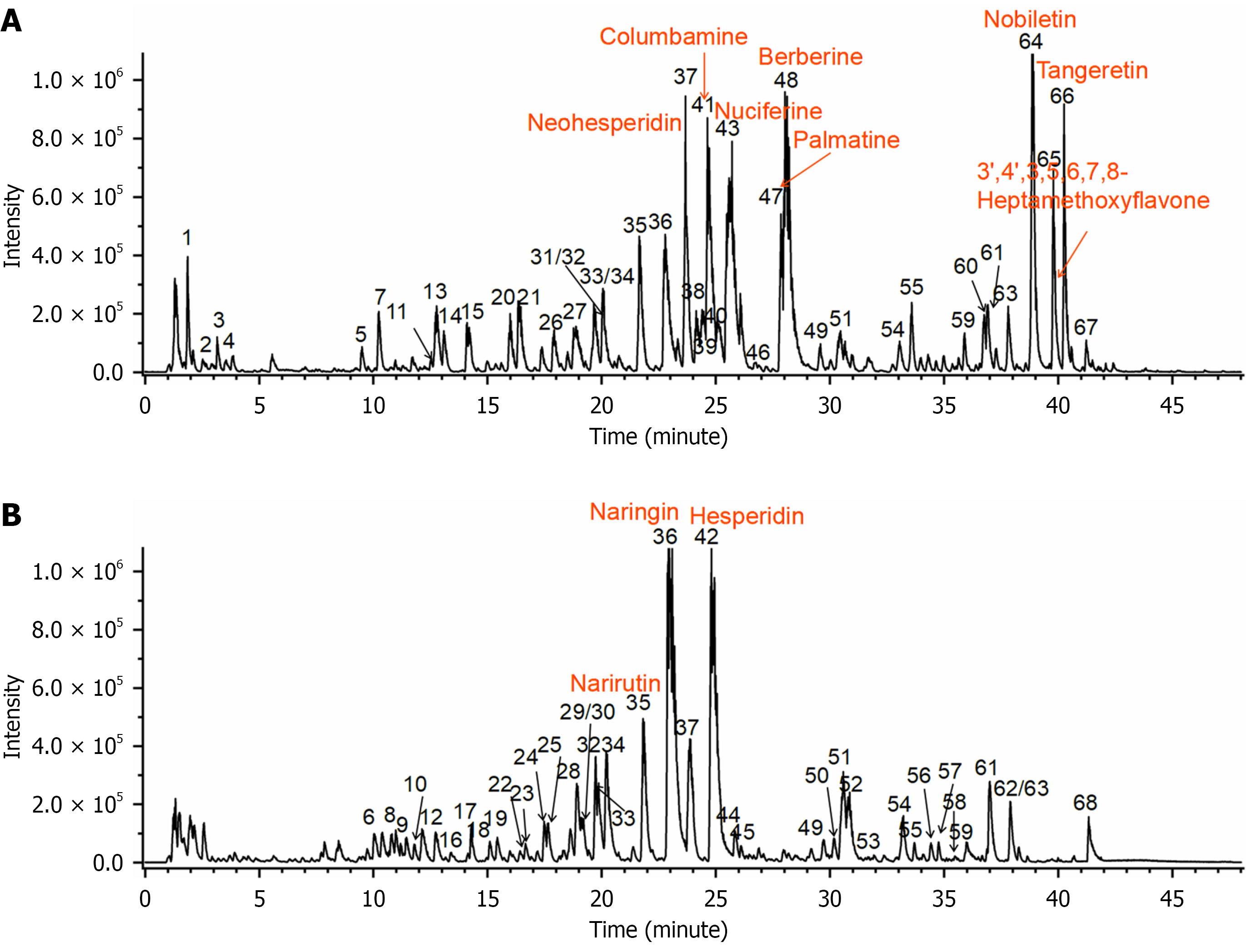
Using multilevel mass spectrometry data and relevant literature, 69 LXO decoction components were identified (Figure 2, Supplementary Table 5). Naringin, hesperidin, neohesperidin, nobiletin, tangeretin, and berberine were present in high abundance (Figure 2, Supplementary Table 5).
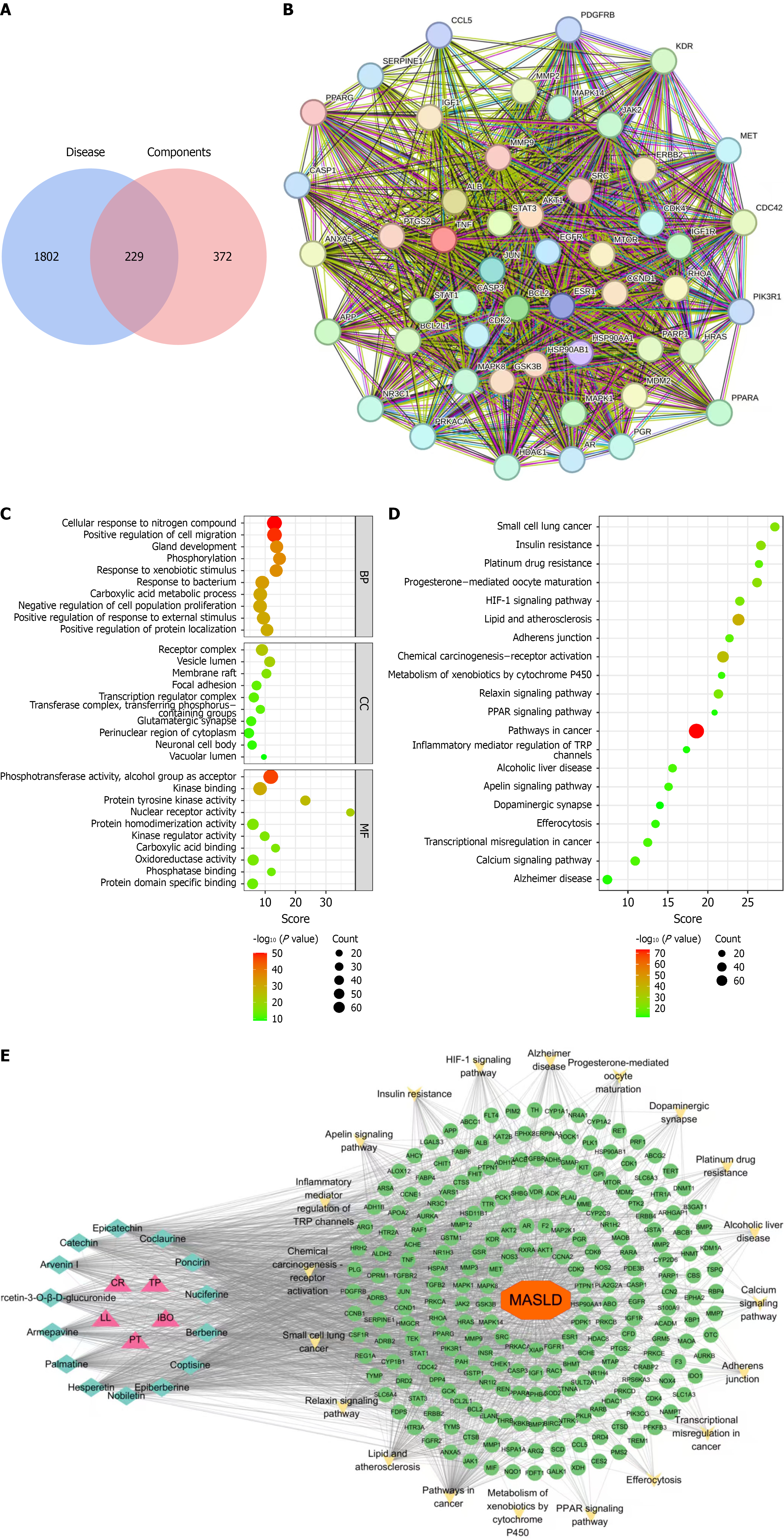
Fourteen active LXO decoction components (Supplementary Table 6) were selected for their oral bioavailability (≥ 30%) and drug likeness (≥ 0.18). A total of 601 potential targets of these active components and 2031 unique disease targets were identified. After integrating the two target types, 229 overlapping genes were identified (Figure 3A, Supplementary Table 7). These genes are potential targets for the efficacy of LXO against MASLD. The generated PPI network contained 229 nodes and 4435 edges, with the top 50 targets ranked by closeness shown in Figure 3B. GO analysis revealed that the key targets of LXO in the treatment of MASLD were primarily involved in the cellular response to nitrogen compounds and positive regulation of cell migration (Figure 3C). These processes are directly implicated in the progression of fatty liver disease from simple steatosis to fibrosis, cirrhosis, and hepatocellular carcinoma[22,23].
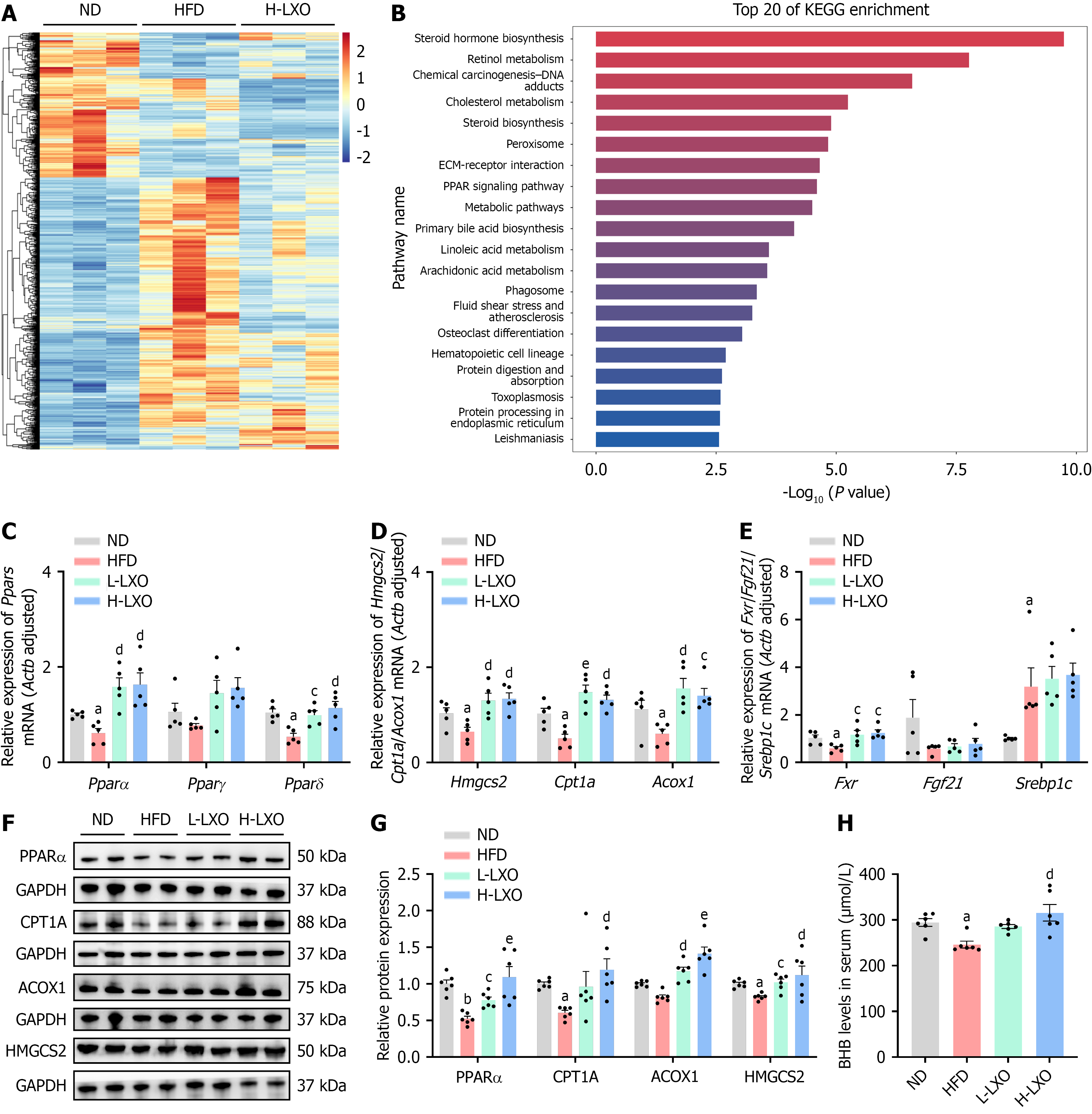
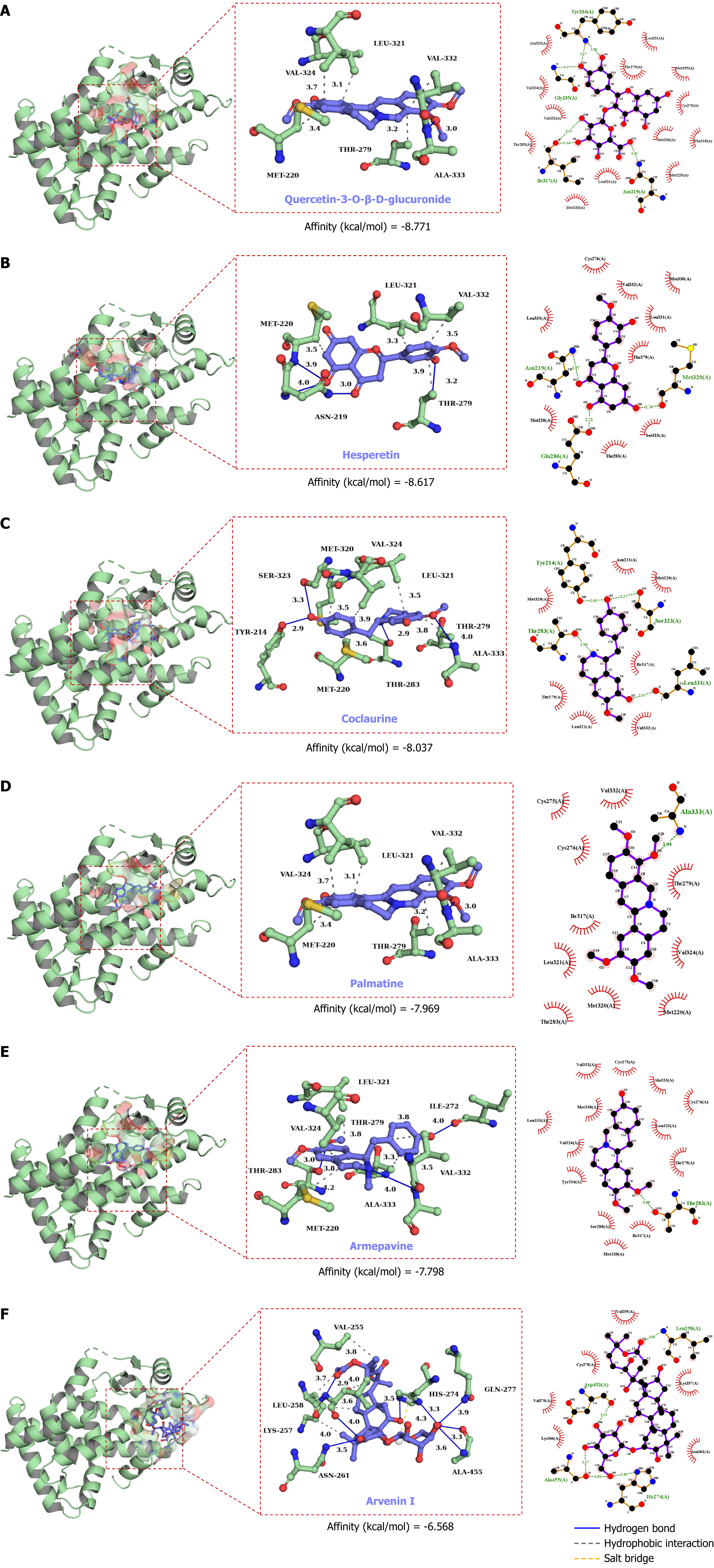
KEGG pathway enrichment analysis revealed that the primary targets of LXO in MASLD were significantly enriched in pathways associated with cancer, lipid metabolism, and atherosclerosis (Figure 3D). Among these pathways, insulin resistance, PPAR and hypoxia inducible factor (HIF)-1 signaling are closely associated with MASLD treatment efficacy. To more intuitively demonstrate the associations between herbs, components, diseases, pathways, and targets, an “herb-ingredient-disease-pathway-target” network relationship diagram was constructed via the Cytoscape 3.8.0 software (Figure 3E). To further prioritize the key bioactive components, we analyzed topological indicators, such as betweenness centrality, closeness centrality, and degree. As shown in Supplementary Table 8, hesperetin, palmatine, armepavine, arvenin I, coclaurine, and quercetin-3-O-β-D-glucuronide showed greater than average representation at three nodes. These components are considered crucial for the therapeutic efficacy of LXO against MASLD. Our findings identified the key bioactive components and signaling pathways of LXO in MASLD treatment, providing valuable insights for further drug development and research.
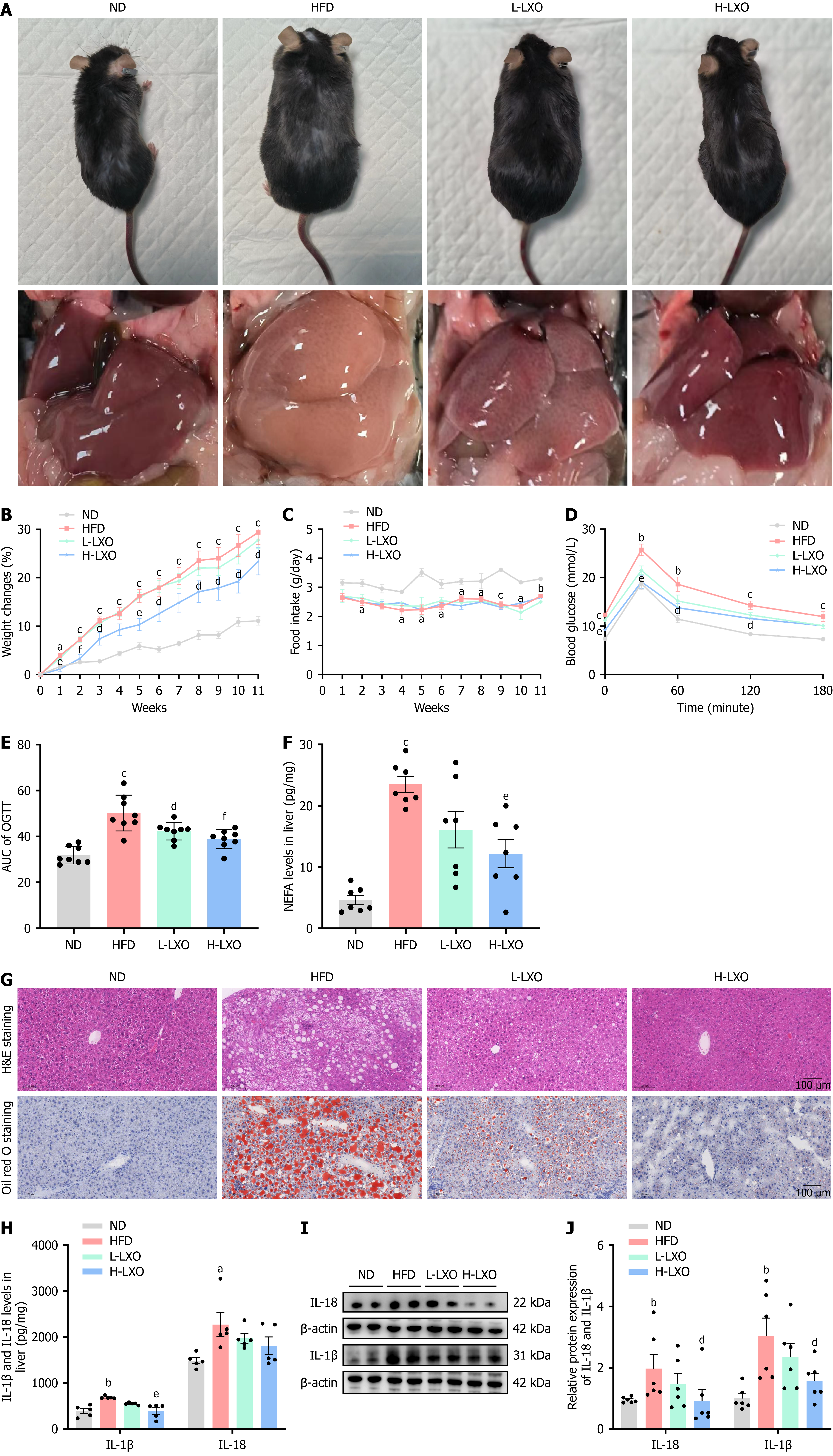
The HFD group experienced significant increases in body weight, FBG, ALT, AST, TG, TC, and other relevant indices. Mice treated with LXO exhibited reduced body size, a healthier hepatic appearance with diminished lipid accumulation (Figure 4A), and a remarkable delay in weight gain progression (Figure 4B). These alterations could not be ascribed to disparities in food intake (Figure 4C). LXO significantly decreased FBG levels in MASLD mice (Table 2) and increased glucose tolerance, as evidenced by lower AUCOGTT values (Figure 4D and E). ALT and AST levels revealed that LXO significantly improved hepatic function in MASLD mice (Table 2). Both L-LXO and H-LXO effectively reduced serum TG levels in MASLD mice (Table 2). Although H-LXO treatment tended to lower TC and low-density lipoprotein cholesterol levels, these differences were not statistically significant (Table 2). LXO also significantly reduced NEFA levels and lipid accumulation in the liver (Figure 4F and G).
| Variables | ND | HFD | L-LXO | H-LXO |
| ALT (U/L) | 23.32 ± 2.34 | 44.98 ± 3.07c | 32.10 ± 2.68c,e | 28.10 ± 1.56a,f |
| AST (U/L) | 11.60 ± 0.93 | 59.42 ± 4.31c | 39.75 ± 5.60e | 27.87 ± 2.86f |
| TG (mmol/L) | 0.58 ± 0.07 | 0.84 ± 0.04a | 0.61 ± 0.06d | 0.66 ± 0.04d |
| TC (mmol/L) | 2.57 ± 0.10 | 5.60 ± 0.25c | 5.06 ± 0.42c | 4.86 ± 0.54b |
| LDL-C (mmol/L) | 0.41 ± 0.03 | 1.30 ± 0.09c | 1.34 ± 0.19c | 1.14 ± 0.18b |
| HDL-C (mmol/L) | 2.34 ± 0.04 | 2.98 ± 0.16b | 3.02 ± 0.14b | 2.88 ± 0.14a |
| FBG (mmol/L) | 8.70 ± 0.12 | 11.03 ± 0.28c | 10.23 ± 0.36b | 9.23 ± 0.21f |
Following a 20-week HFD regimen, the mice displayed MASH symptoms accompanied by hepatic inflammation. ELISA kits and western blotting were used to quantitatively assess inflammatory responses. LXO intervention significantly decreased hepatic IL-1β and IL-18 levels in MASLD mice (Figure 4H-J). These findings emphasize that LXO can effectively alleviate HFD-induced weight gain, glucose and lipid metabolic disorders, liver injury, and inflammation.
After RNA-sequencing analysis of mouse liver with stringent statistical thresholds of false discovery rate < 0.05, a comprehensive panorama of 797 differentially expressed genes was revealed across the ND, HFD, and H-LXO groups. Hierarchical clustering analysis revealed substantial disparities in the expression profiles of these differentially expressed genes, significantly differentiated the groups (Figure 5A). To conduct a more in-depth exploration of the functional implications associated with these findings, transcriptomics-guided KEGG pathway analyses were performed. These analyses shed light on complex molecular functions and pathways. Notably, the PPAR signaling pathway was highly enriched, a finding consistent with our network pharmacology analysis (Figure 3D and Figure 5B). These findings underscore the importance of the PPAR pathway in the treatment of MASLD with LXO. Upon in-depth scrutiny, LXO was shown to exert a considerable stimulatory effect on hepatic expression of the PPARα, and PPARδ genes in MASLD mice (Figure 5C). LXO also elevated the transcript levels of crucial enzymes and receptors implicated in lipid catabolism, including Hmgcs2, Acox1, Cpt1a, and Fxr (Figure 5D and E). Notably, the expression of Srebp1c, a key transcription factor in lipogenesis, and Fgf21, a hormone that regulates systemic energy expenditure and fatty acid oxidation, remained unaltered (Figure 5E). Collectively, these findings suggest that LXO selectively facilitates lipid catabolism rather than lipogenesis or cholesterol synthesis, with PPAR signaling playing a central role.
Leveraging the knowledge obtained via network pharmacology, transcriptomics, and quantitative real-time-PCR, we conducted a more in-depth exploration of the role of PPARα signaling in MASLD treatment with LXO. Our findings indicate that LXO can upregulate hepatic PPARα protein expression in MASLD mice, facilitating fatty acid β-oxidation. This upregulation was mainly evidenced by increased expression of carnitine palmitoyl transferase 1a (CPT1A), acyl-CoA oxidase 1 (Acox1), and 3-hydroxy-3-methylglutaryl-CoA synthase 2 (HMGCS2) proteins, with increased generation of ketone bodies, particularly BHB (Figure 5F-H). PPARα is also capable of modulating hepatic inflammation through NF-κB inhibition[12]. In the present study, LXO significantly reduced IL-1β and IL-18 levels in the livers of mice with MASLD (Figure 4H-J). Another notable observation was an increase in serum LPS levels in MASLD mice, which activated Toll-like receptor (TLR) 4/NF-κB signaling. LXO treatment mitigated these LPS elevations in MASLD mice, effectively inhibiting the TLR4/NF-κB signaling cascade (Supplementary Figure 1). Collectively, these results underscore the potential advantageous role of LXO-mediated PPARα modulation for ameliorating hepatic lipid metabolism and hepatic inflammation.
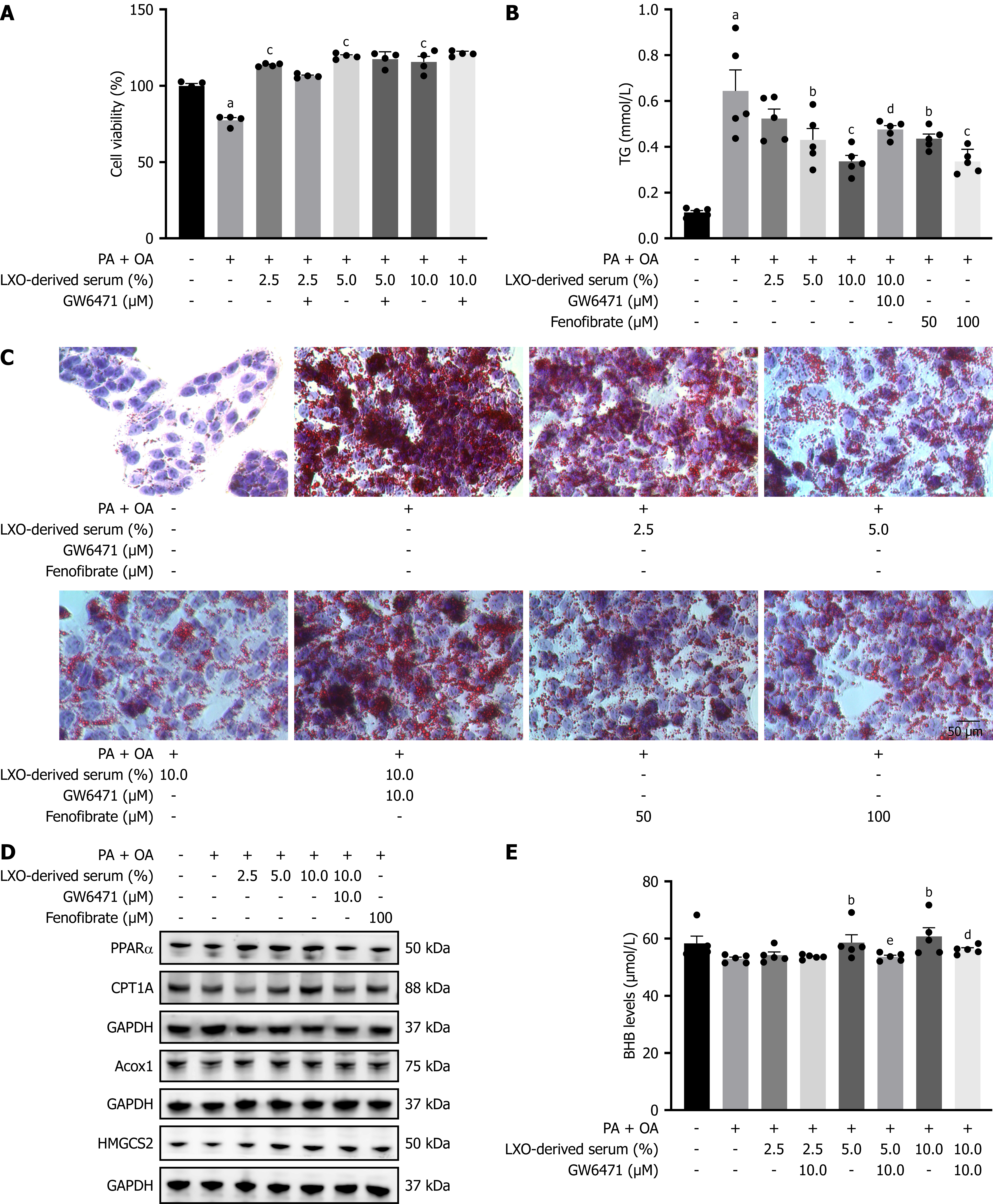
To further corroborate the crucial role of PPARα signaling in the lipid-lowering effects of LXO, a high-fat model of HepG2 cells was established using FFAs. Based on relevant literature[24], the manufacturer’s instructions, and our preliminary results, the final modeling concentrations for OA and PA were set at 200 μM and 100 μM, respectively. The concentration of the PPARα inhibitor GW6471 was 10 μM. As depicted in Figure 6A, PA decreased HepG2 cell viability, whereas LXO-derived serum notably increased the cell viability. GW6471 did not exert any perceptible effect on this phenomenon. Similar to fenofibrate (100 μM), a PPARα agonist with lipid-lowering properties, LXO notably decreased TG levels in HepG2 cells. This effect was partially countered by GW6471 treatment (Figure 6B). Oil red O staining further validated this result (Figure 6C). Further investigations demonstrated that LXO enhanced PPARα, Acox1, CPT1A, and HMGCS2 protein levels in HepG2 cells, and BHB levels in cell supernatant (Figure 6D and E). GW6471 countered these effects. The PPARα signaling pathway thus plays a critical role in the lipid-lowering effect of LXO.
Molecular docking was performed to predict the interactions between six active components and specific amino acid residues within the PPARα protein structure. All six key active compounds established stable complexes with PPARα via hydrophobic interactions and hydrogen bonds. Among them, quercetin-3-O-β-D-glucuronide demonstrated the best binding effect with PPARα (Figure 7).
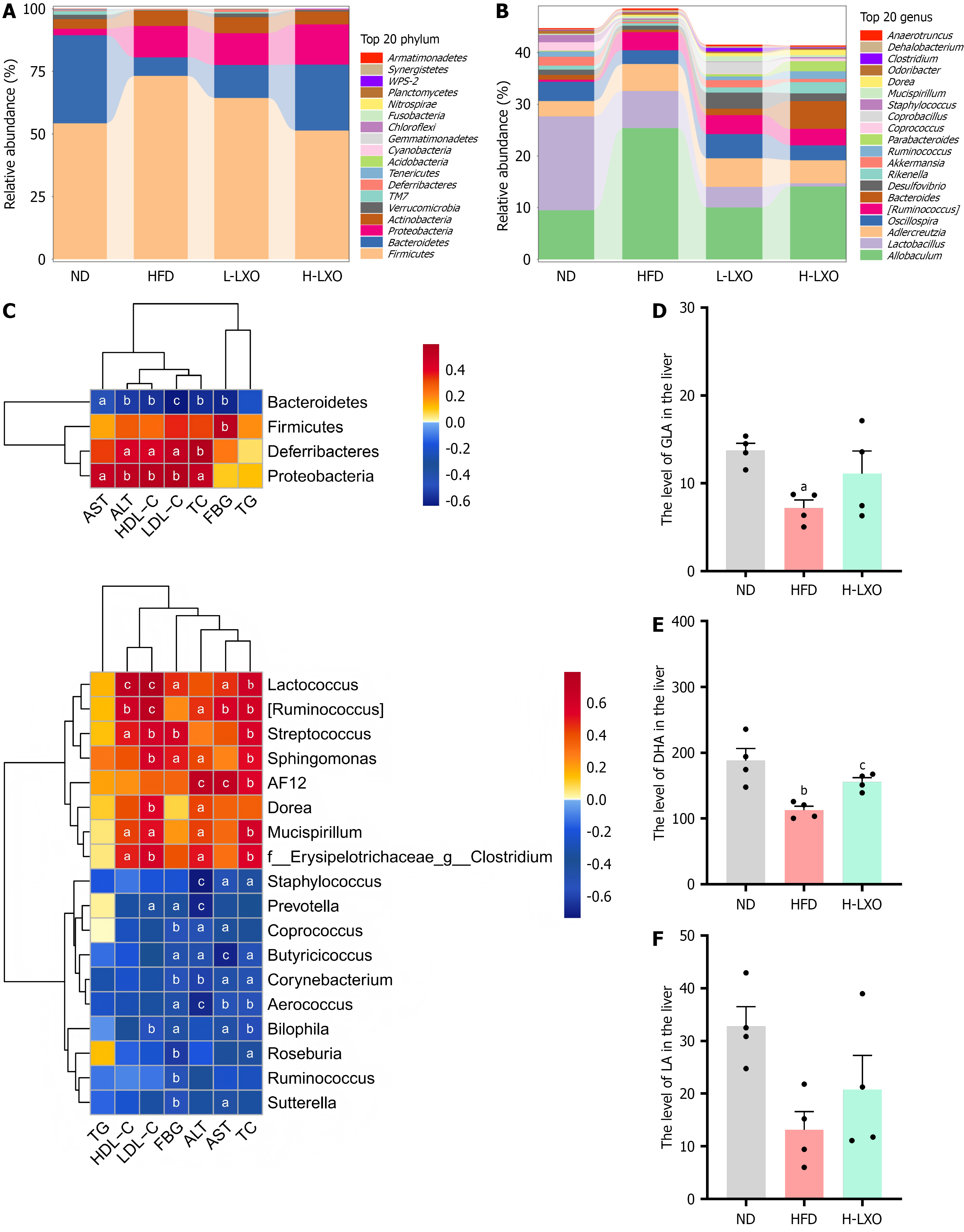
Considering that PPARα can be activated by endogenous ligands, including fatty acids and bile acids[25], we hypothesized that LXO might affect production of these ligands via gut microbiota modulation, thereby enhancing PPARα signaling. 16S rRNA sequencing was performed on cecal contents. Analysis of α diversity indicated that LXO treatment improved gut microbiota richness, diversity, and evenness (Supplementary Figure 2A). Principal coordinate analysis of β diversity revealed distinct clustering of gut microbiota among the four groups (Supplementary Figure 2B). To pinpoint the specific taxa responsible for these changes, linear discriminant analysis effect size was conducted, which identified significant alterations at multiple taxonomic levels (Supplementary Figure 2C). LXO reversed the abundances of Firmicutes and Bacteroidetes at the phylum level (Figure 8A, Supplementary Figure 2C and Supplementary Table 9) and the abundances of Clostridiaceae (family)/Clostridium (genus), Akkermansia, Ruminococcus, Coprococcus, Bilophila, Butyricicoccus, Sutterella, Aerococcus, Corynebacterium, Pseudomonas, Streptococcus, Jeotgalicoccus, and Dehalobacterium at the genus level (Figure 8B, Supplementary Figure 2C and Supplementary Table 10).
Subsequent studies and analyses revealed notable correlations between specific differentially expressed microorganisms and serum indicators relevant to MASLD. Strong associations were observed between Bacteroidetes and related serum indicators (Figure 8C). LXO significantly increased Parabacteroides distasonis abundance (ND: 0.33%; HFD: 0.00%; L-LXO: 0.67%; H-LXO: 2.83%). This species is recognized for its ability to counter MASLD symptoms by activating PPARα via its metabolite fatty acids[4]. Subsequent investigations revealed increased levels of hepatic fatty acids gamma-linolenic acid (GLA), docosahexaenoic acid (DHA), and linolenic acid (LA) (Figure 8D-F). These findings suggest that LXO-induced PPARα activation may be modulated by the gut microbiota and its metabolites, with fatty acids serving as pi
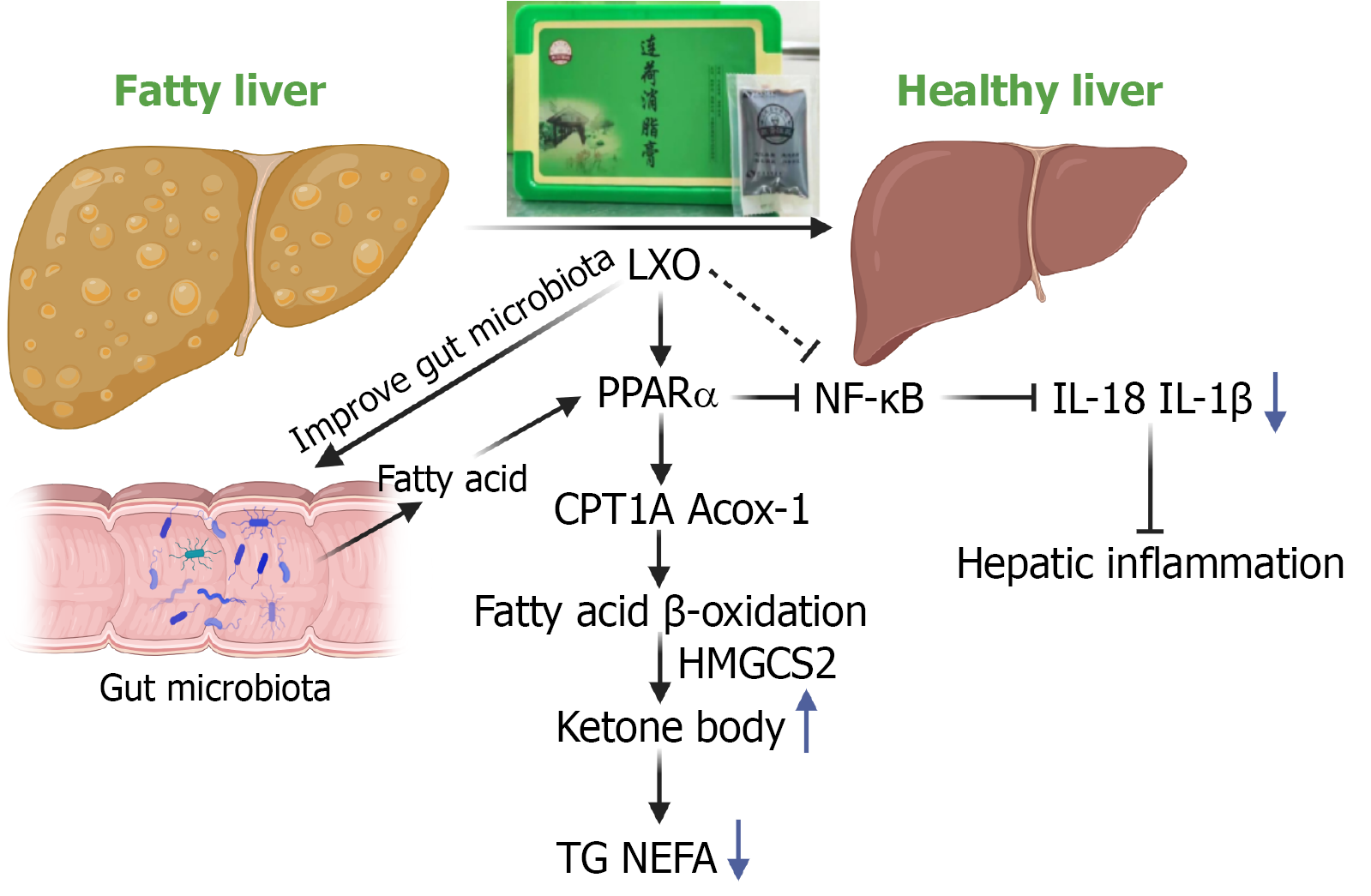
The search for effective therapies for MASLD is imperative, given its prevalence as a metabolic disorder driven by hepatic steatosis and associated cardiometabolic risks. Our study focused on established biomarkers that reflect core disease processes, including FBG for insulin resistance, TG/TC for lipid metabolism, and ALT/AST for liver injury. The marked improvement in these parameters by LXO confirmed its therapeutic efficacy at the clinical phenotypic level. Further investigation revealed that LXO alleviated hepatic inflammation and corrected the gut microbiota imbalance in MASLD mice. Via UPLC-Q/TOF-MS and network pharmacology analysis, six key bioactive components of LXO implicated in its efficacy against MASLD (hesperetin, palmatine, armepavine, arvenin I, coclaurine, and quercetin-3-O-β-D-glucuronide) were identified. Multiomics analysis further pinpointed the PPARα signaling pathway as pivotal to LXO’s therapeutic mechanism. In summary, this study not only demonstrates the therapeutic potential of LXO against MASLD but also elucidates its underlying mechanisms (Figure 9).
Using network pharmacology, we predicted the key active components, their targets, and signaling pathways associated with MASLD that are modulated by LXO. This analysis identified 229 common targets and six promising components. Among the enriched pathways, the PPAR, HIF-1, and insulin resistance signaling pathways were closely associated with MASLD pathogenesis and treatment. Network pharmacology predictions are susceptible to potential biases, necessitating integration with other omics technologies such as transcriptomics or metabolomics to bolster prediction robustness[26]. Our liver transcriptome analyses revealed significant enrichment in PPAR signaling pathway genes among the identified differentially expressed genes. We thus further explored the PPAR signaling pathway.
The PPAR receptor family encompasses three subtypes: PPARα, PPARβ/δ, and PPARγ. Among these subtypes, PPARα is highly expressed in liver tissue and participates in regulating fatty acid oxidation and inflammatory responses[6,13]. In the present study, LXO activated hepatic PPARα, consequently facilitating fatty acid β-oxidation by upregulating the downstream genes Cpt1a and Acox1. Simultaneously, it activated Hmgcs2, synergistically augmenting ketogenesis. These findings were corroborated in in vivo and in vitro experiments. Notably, all six key bioactive components screened from LXO displayed strong PPARα binding affinities. LXO notably elevated the relative abundance of Parabacteroides distasonis, which has been proven to activate PPARα receptors via fatty acid metabolism[4]. LXO also increased GLA, DHA, and LA levels in the liver, all of which serve as endogenous PPARα agonists[4,25]. These findings imply that PPARα assumes a pivotal role in the therapeutic effect of LXO against MASLD, possibly through direct or indirect activation of PPARα. These results require more rigorous experimental verification.
Increasing evidence supports a crucial role of gut microbiota dysregulation in inducing immune imbalance and metabolic disturbances, leading to MASLD initiation and progression[27]. Mounting evidence suggests that the gut microbiota can be therapeutically targeted by TCM to manage MASLD/MASH, suggesting novel strategies for disease regulation and prevention[28,29]. In line with previous studies[30,31], HFD disrupted the gut microbiota. In this study, LXO partially reversed this gut microbiota dysbiosis. H-LXO treatment nearly fully restored normal relative abundances of Firmicutes and Bacteroidetes at the phylum level. LXO also altered the relative abundances of 13 bacterial genera. Several identified genera have been correlated with MASLD/MASH. Certain probiotics, including Akkermansia[32], Coprococcus[33], Butyricicoccus[34] and Aerococcus[35], have been demonstrated to be beneficial in MASLD/MASH treatment. In contrast, pathogenic bacteria, including Bilophila[36], Pseudomonas[37] and Streptococcus[38] have been implicated in MASLD/MASH initiation and progression. Moreover, H-LXO notably increased Rikenella, Bacteroides, and Parabacteroides abundance. Augmentation of these bacteria in MASLD/MASH treatment has been proven beneficial[39,40].
Alterations in the gut microbiota can impair the integrity of the intestinal epithelial barrier, permitting bacteria and LPS to translocate into circulation and subsequently reach the liver via the portal vein[41]. This process upregulates TLR4 receptors, initiating downstream inflammatory signal transduction, especially via NF-κB signaling[42]. NF-κB functions as a crucial regulator of inflammatory responses. In the present study, LXO effectively ameliorated gut microbiota dysbiosis, suppressed hepatic TLR4 and NF-κB activity, and decreased IL-1β production and release. As a critical mediator in MASH, IL-1β drives hepatic TG accumulation and subsequent cell death. Modulation of NF-κB signaling by LXO may be realized directly through key bioactive constituents such as hesperetin[43], palmatine[44] and quercetin-3-O-β-D-glucuronide[45], or indirectly via activation of PPARα[11,12]. Notably, our study revealed that LXO activated PPARα signaling in vivo and in vitro experimental models. The results suggest that LXO mitigate liver inflammation by directly or indirectly modulating NF-κB signaling. Elucidating the exact effects of various LXO active components on NF-κB signaling and alleviation of MASLD require rigorous experimental verification.
This study has some limitations. First, this study represents a preliminary exploration of the mechanisms underlying the therapeutic effects of LXO on MASLD. Direct validation of proposed key targets and pathways is lacking. Additional experiments are necessary to clarify the specific mechanisms involved, such as validation via PPARα gene knockout mouse models. Second, our molecular docking results are preliminary predictions requiring experimental validation (e.g., affinity assays or structural biology studies) to confirm the interactions between key components and PPARα. Third, gut microbiota analysis predominantly focuses on alterations in microbial composition. In-depth exploration of functional metabolites (e.g., short-chain fatty acids) and their interactions with host metabolism is lacking. This limitation restricts a comprehensive understanding of gut microbiota’s contribution to the therapeutic effects of LXO on MASLD. Fourth, there is a deficiency in serum pharmacochemical analysis, which results in the actual bioactive components absorbed into the systemic circulation remaining unverified. The truly bioavailable components represent a key goal for our future research. Finally, although this study demonstrated certain effects in animal models, a preliminary exploration of the pharmacokinetics, bioavailability, and clinical efficacy of LXO in humans is lacking, impeding its potential for clinical translation.
Collectively, the present findings indicate that LXO mitigates hepatic lipid metabolism and inflammation in MASLD mice, in part by activating PPARα signaling. These results highlight the potential of LXO as a promising candidate for MASLD treatment. Further investigations are necessary to elucidate precise mechanisms and validate LXO's efficacy and safety in clinical settings in multicenter trials.
| 1. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966-1986. [PubMed] [DOI] [Full Text] |
| 2. | Le MH, Yeo YH, Zou B, Barnet S, Henry L, Cheung R, Nguyen MH. Forecasted 2040 global prevalence of nonalcoholic fatty liver disease using hierarchical bayesian approach. Clin Mol Hepatol. 2022;28:841-850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 276] [Cited by in RCA: 248] [Article Influence: 62.0] [Reference Citation Analysis (0)] |
| 3. | Wong VW, Ekstedt M, Wong GL, Hagström H. Changing epidemiology, global trends and implications for outcomes of NAFLD. J Hepatol. 2023;79:842-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 591] [Cited by in RCA: 539] [Article Influence: 179.7] [Reference Citation Analysis (0)] |
| 4. | Kuang J, Wang J, Li Y, Li M, Zhao M, Ge K, Zheng D, Cheung KCP, Liao B, Wang S, Chen T, Zhang Y, Wang C, Ji G, Chen P, Zhou H, Xie C, Zhao A, Jia W, Zheng X, Jia W. Hyodeoxycholic acid alleviates non-alcoholic fatty liver disease through modulating the gut-liver axis. Cell Metab. 2023;35:1752-1766.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 244] [Article Influence: 81.3] [Reference Citation Analysis (6)] |
| 5. | Cooreman MP, Vonghia L, Francque SM. MASLD/MASH and type 2 diabetes: Two sides of the same coin? From single PPAR to pan-PPAR agonists. Diabetes Res Clin Pract. 2024;212:111688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 6. | Guixé-Muntet S, Biquard L, Szabo G, Dufour JF, Tacke F, Francque S, Rautou PE, Gracia-Sancho J. Review article: vascular effects of PPARs in the context of NASH. Aliment Pharmacol Ther. 2022;56:209-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Ruppert PMM, Kersten S. Mechanisms of hepatic fatty acid oxidation and ketogenesis during fasting. Trends Endocrinol Metab. 2024;35:107-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 60] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 8. | Francque S, Verrijken A, Caron S, Prawitt J, Paumelle R, Derudas B, Lefebvre P, Taskinen MR, Van Hul W, Mertens I, Hubens G, Van Marck E, Michielsen P, Van Gaal L, Staels B. PPARα gene expression correlates with severity and histological treatment response in patients with non-alcoholic steatohepatitis. J Hepatol. 2015;63:164-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 303] [Article Influence: 27.5] [Reference Citation Analysis (1)] |
| 9. | Yahaghi L, Yaghmaei P, Hayati-Roodbari N, Irani S, Ebrahim-Habibi A. Betanin effect on PPAR-α and SREBP-1c expression in NMRI mice model of steatohepatitis with fibrosis. Physiol Int. 2020;107:67-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Abdelmegeed MA, Yoo SH, Henderson LE, Gonzalez FJ, Woodcroft KJ, Song BJ. PPARalpha expression protects male mice from high fat-induced nonalcoholic fatty liver. J Nutr. 2011;141:603-610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 217] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 11. | Wang M, Luo W, Yu T, Liang S, Sun J, Zhang Y, Han X, Long X, Liang G, Li G. Corynoline protects ang II-induced hypertensive heart failure by increasing PPARα and Inhibiting NF-κB pathway. Biomed Pharmacother. 2022;150:113075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 12. | Vanden Berghe W, Vermeulen L, Delerive P, De Bosscher K, Staels B, Haegeman G. A paradigm for gene regulation: inflammation, NF-kappaB and PPAR. Adv Exp Med Biol. 2003;544:181-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 182] [Article Influence: 7.9] [Reference Citation Analysis (3)] |
| 13. | Linghu L, Zong W, Liao Y, Chen Q, Meng F, Wang G, Liao Z, Lan X, Chen M. Herpetrione, a New Type of PPARα Ligand as a Therapeutic Strategy Against Nonalcoholic Steatohepatitis. Research (Wash D C). 2023;6:0276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 14. | Yoo J, Jeong IK, Ahn KJ, Chung HY, Hwang YC. Fenofibrate, a PPARα agonist, reduces hepatic fat accumulation through the upregulation of TFEB-mediated lipophagy. Metabolism. 2021;120:154798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 93] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 15. | Liu Q, Li X, Pan Y, Liu Q, Li Y, He C, Zheng N, Wang Y, Wang H, Wang Y, Sheng L, Zhang B, Shen T, Wu G, Li H, Wang X, Zhang W, Hu Y, Zhao Y. Efficacy and safety of Qushi Huayu, a traditional Chinese medicine, in patients with nonalcoholic fatty liver disease in a randomized controlled trial. Phytomedicine. 2024;130:155398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Zhou TT, Zhu WJ, Feng H, Ni Y, Li ZW, Sun DD, Li L, Tan JN, Yu CT, Shen WX, Cheng HB. A network pharmacology integrated serum pharmacochemistry strategy for uncovering efficacy of YXC on hepatocellular carcinoma. J Ethnopharmacol. 2024;319:117125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 17. | Chen M, Xie Y, Gong S, Wang Y, Yu H, Zhou T, Huang F, Guo X, Zhang H, Huang R, Han Z, Xing Y, Liu Q, Tong G, Zhou H. Traditional Chinese medicine in the treatment of nonalcoholic steatohepatitis. Pharmacol Res. 2021;172:105849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 18. | Qian X, Zhou J, Li S, Yuan S. [Effects of Huanglian Wendan Decoction on Blood Glucose and Blood Lipid Indexes in Diabetes Mellitus Type 2 Patients with Non-Alcoholic Fatty Liver Disease]. Liaoning Zhongyi Zazhi. 2023;50:155-159. [DOI] [Full Text] |
| 19. | Zhang YJ. [Clinical effect of Lianhe Xiaozhi Ointment on Type2 diabetes mellitus with metabolism associated fatty liver disease syndrome of phlegm-heat]. M.Sc. Thesis, Nanjing University of Chinese Medicine. 2024. Available from: https://x.cnki.net/web/trilalread/article/trialPdfRead.html?fileName=1025016561NH&tableName=CMFDTOTAL&dbCode=CMFD&fileSourceType=1&groupId=&appId=CRSP_BASIC_PSMC&SharedTextId=FF2F782BE90D0E85616754&act=&f=tr. |
| 20. | Miao R, Zhang Y, Zhang Y, Fang X, Yin R, Tian J. Jiangtang Tiaozhi formula ameliorates MASLD by regulating liver ABCD2/PEX2/ATGL axis-mediated fatty acid metabolic reprogramming. Phytomedicine. 2025;145:157032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 21. | Ma L, Wang Y, Liu Y, Wang J, Han Y, Qin D, Yan H. Analysis of the Network Pharmacology Mechanism and Clinical Efficacy of Qingre Jiedu Decoction (QJD) in the Treatment of Infectious Mononucleosis (IM) in Children. Drug Des Devel Ther. 2025;19:6283-6300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Song Q, Hwang CL, Li Y, Wang J, Park J, Lee SM, Sun Z, Sun J, Xia Y, Nieto N, Cordoba-Chacon J, Jiang Y, Dou X, Song Z. Gut-derived ammonia contributes to alcohol-related fatty liver development via facilitating ethanol metabolism and provoking ATF4-dependent de novo lipogenesis activation. Metabolism. 2024;151:155740. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 23. | Xiao Y, Batmanov K, Hu W, Zhu K, Tom AY, Guan D, Jiang C, Cheng L, McCright SJ, Yang EC, Lanza MR, Liu Y, Hill DA, Lazar MA. Hepatocytes demarcated by EphB2 contribute to the progression of nonalcoholic steatohepatitis. Sci Transl Med. 2023;15:eadc9653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 24. | Chen S, Che S, Li S, Ruan Z. The combined impact of decabromodiphenyl ether and high fat exposure on non-alcoholic fatty liver disease in vivo and in vitro. Toxicology. 2021;464:153015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 25. | Kliewer SA, Sundseth SS, Jones SA, Brown PJ, Wisely GB, Koble CS, Devchand P, Wahli W, Willson TM, Lenhard JM, Lehmann JM. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci U S A. 1997;94:4318-4323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1783] [Cited by in RCA: 1634] [Article Influence: 56.3] [Reference Citation Analysis (4)] |
| 26. | Huang J, Shi R, Chen F, Tan HY, Zheng J, Wang N, Li R, Wang Y, Yang T, Feng Y, Zhong Z. Exploring the anti-hepatocellular carcinoma effects of Xianglian Pill: Integrating network pharmacology and RNA sequencing via in silico and in vitro studies. Phytomedicine. 2024;133:155905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Long Q, Luo F, Li B, Li Z, Guo Z, Chen Z, Wu W, Hu M. Gut microbiota and metabolic biomarkers in metabolic dysfunction-associated steatotic liver disease. Hepatol Commun. 2024;8:e0310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 53] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 28. | Han R, Qiu H, Zhong J, Zheng N, Li B, Hong Y, Ma J, Wu G, Chen L, Sheng L, Li H. Si Miao Formula attenuates non-alcoholic fatty liver disease by modulating hepatic lipid metabolism and gut microbiota. Phytomedicine. 2021;85:153544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 29. | Hu Q, Zhang W, Wu Z, Tian X, Xiang J, Li L, Li Z, Peng X, Wei S, Ma X, Zhao Y. Baicalin and the liver-gut system: Pharmacological bases explaining its therapeutic effects. Pharmacol Res. 2021;165:105444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 183] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 30. | Hernández-Ceballos W, Cordova-Gallardo J, Mendez-Sanchez N. Gut Microbiota in Metabolic-associated Fatty Liver Disease and in Other Chronic Metabolic Diseases. J Clin Transl Hepatol. 2021;9:227-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 31. | Kang K, Sun Y, Pan D, Sang LX, Sun MJ, Li YL, Chang B. Distinctive gut microbial dysbiosis between chronic alcoholic fatty liver disease and metabolic-associated fatty liver disease in mice. Exp Ther Med. 2021;21:418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 32. | Song L, Li Y, Qu D, Ouyang P, Ding X, Wu P, Guan Q, Yang L. The regulatory effects of phytosterol esters (PSEs) on gut flora and faecal metabolites in rats with NAFLD. Food Funct. 2020;11:977-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 33. | Li F, Ye J, Shao C, Zhong B. Compositional alterations of gut microbiota in nonalcoholic fatty liver disease patients: a systematic review and Meta-analysis. Lipids Health Dis. 2021;20:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 34. | Mu H, Zhou Q, Yang R, Zeng J, Li X, Zhang R, Tang W, Li H, Wang S, Shen T, Huang X, Dou L, Dong J. Naringin Attenuates High Fat Diet Induced Non-alcoholic Fatty Liver Disease and Gut Bacterial Dysbiosis in Mice. Front Microbiol. 2020;11:585066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 142] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 35. | Jian J, Nie MT, Xiang B, Qian H, Yin C, Zhang X, Zhang M, Zhu X, Xie WF. Rifaximin Ameliorates Non-alcoholic Steatohepatitis in Mice Through Regulating gut Microbiome-Related Bile Acids. Front Pharmacol. 2022;13:841132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 36. | Jian C, Luukkonen P, Sädevirta S, Yki-Järvinen H, Salonen A. Impact of short-term overfeeding of saturated or unsaturated fat or sugars on the gut microbiota in relation to liver fat in obese and overweight adults. Clin Nutr. 2021;40:207-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 37. | Sun C, Qiu C, Zhang Y, Yan M, Tan J, He J, Yang D, Wang D, Wu L. Lactiplantibacillus plantarum NKK20 Alleviates High-Fat-Diet-Induced Nonalcoholic Fatty Liver Disease in Mice through Regulating Bile Acid Anabolism. Molecules. 2023;28:4042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 38. | Huang ZR, Chen M, Guo WL, Li TT, Liu B, Bai WD, Ai LZ, Rao PF, Ni L, Lv XC. Monascus purpureus-fermented common buckwheat protects against dyslipidemia and non-alcoholic fatty liver disease through the regulation of liver metabolome and intestinal microbiome. Food Res Int. 2020;136:109511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 39. | Carbajo-Pescador S, Porras D, García-Mediavilla MV, Martínez-Flórez S, Juarez-Fernández M, Cuevas MJ, Mauriz JL, González-Gallego J, Nistal E, Sánchez-Campos S. Beneficial effects of exercise on gut microbiota functionality and barrier integrity, and gut-liver crosstalk in an in vivo model of early obesity and non-alcoholic fatty liver disease. Dis Model Mech. 2019;12:dmm039206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 114] [Article Influence: 16.3] [Reference Citation Analysis (4)] |
| 40. | Kang Y, Kuang X, Yan H, Ren P, Yang X, Liu H, Liu Q, Yang H, Kang X, Shen X, Tong M, Li L, Wang X, Guo L, Ma J, Zhang F, Fan W. A Novel Synbiotic Alleviates Autoimmune Hepatitis by Modulating the Gut Microbiota-Liver Axis and Inhibiting the Hepatic TLR4/NF-κB/NLRP3 Signaling Pathway. mSystems. 2023;8:e0112722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 67] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 41. | Matsushita N, Osaka T, Haruta I, Ueshiba H, Yanagisawa N, Omori-Miyake M, Hashimoto E, Shibata N, Tokushige K, Saito K, Tsuneda S, Yagi J. Effect of Lipopolysaccharide on the Progression of Non-Alcoholic Fatty Liver Disease in High Caloric Diet-Fed Mice. Scand J Immunol. 2016;83:109-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 42. | Cao Y, Ren G, Zhang Y, Qin H, An X, Long Y, Chen J, Yang L. A new way for punicalagin to alleviate insulin resistance: regulating gut microbiota and autophagy. Food Nutr Res. 2021;65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 43. | Ma H, Feng X, Ding S. Hesperetin attenuates ventilator-induced acute lung injury through inhibition of NF-κB-mediated inflammation. Eur J Pharmacol. 2015;769:333-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 44. | Xu Y, Shen B, Pan X, Liu C, Wang Y, Chen X, Wang T, Chen G, Chen J. Palmatine ameliorated lipopolysaccharide-induced sepsis-associated encephalopathy mice by regulating the microbiota-gut-brain axis. Phytomedicine. 2024;124:155307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 45. | Mao H, Feng Y, Feng J, Yusufu Y, Sun M, Yang L, Jiang Q. Quercetin-3-O-β-D-glucuronide attenuates osteoarthritis by inhibiting cartilage extracellular matrix degradation and inflammation. J Orthop Translat. 2024;45:236-246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/