Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.115263
Revised: November 23, 2025
Accepted: February 5, 2026
Published online: April 21, 2026
Processing time: 184 Days and 18.6 Hours
Aggressive angiomyxoma (AA) is a rare, slow-growing, locally invasive mesen
We report a case of AA located in the lesser curvature of the gastric antrum in a middle-aged female who presented with a palpable abdominal mass of one week’s duration. The patient subsequently underwent en bloc tumor resection, with histological confirmation of the diagnosis of gastric AA. Currently, there are no signs of tumor recurrence, and the patient’s general condition is satisfactory.
To our knowledge, this represents the second reported case of gastric-origin AA worldwide. AA arising in the stomach remains difficult to differentiate radiologically, and further clinical data are needed for comprehensive analysis in the future.
Core Tip: Aggressive angiomyxoma is a rare, slow-growing, locally invasive mesenchymal tumor. We present a case of aggressive angiomyxoma located in the stomach in a female, with a comprehensive review of its imaging features of endoscopic ultrasound, computed tomography, and magnetic resonance imaging.
- Citation: Wang JJ, Xu WL, Li AQ. Aggressive angiomyxoma occurring in the stomach: A case report and review of literature. World J Gastroenterol 2026; 32(15): 115263
- URL: https://www.wjgnet.com/1007-9327/full/v32/i15/115263.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i15.115263
Aggressive angiomyxoma (AA) is a rare, slow-growing, locally aggressive mesenchymal tumor classified by the World Health Organization as being of uncertain differentiation[1]. AA most frequently occurs in the perineal region of women of reproductive age, but may also arise in the vagina, vulva, and other pelvic soft tissues. However, AA may also develop in males, with a male-to-female prevalence ratio of approximately 1:6[2]. The clinical manifestations of AA are highly variable, ranging from painless masses to nonspecific symptoms such as dyspareunia. Due to its rarity, AA is often misdiagnosed as other conditions, including lipomas, Bartholin gland cysts, and hernias.
AA can occur in extrapelvic and extraperineal sites, such as the larynx[3], maxilla[4], floor of the mouth[5], scrotum[6], prostate[7], thigh[8], axilla and chest wall[9], lung[10], and jejunum[11]. Gastric origin of AA is exceptionally rare, and to our knowledge, the case we report here represents the second documented instance worldwide[12].
We present a case of AA localized to the stomach in a female, with a comprehensive review of its endoscopic ultrasound (EUS), computed tomography (CT), and magnetic resonance imaging (MRI) features.
A 48-year-old female presented to our hospital with “recurrent epigastric pain for several years, recurrent with nausea after consuming large amounts of cold beverages and grilled fish for 13 days”.
Thirteen days prior to admission, the patient developed severe epigastric pain with nausea after consuming large amounts of cold beverages and grilled fish, prompting emergency evaluation.
Over the past several years, the patient experienced recurrent epigastric pain triggered by improper diet or fasting, without associated diarrhea, vomiting, or gastrointestinal bleeding. Repeated gastroscopies in 2021 and 2024 revealed multiple small polyps in the gastric body and shallow duodenal ulcers. Laboratory investigations during this period showed an erythrocyte sedimentation rate of 34 mm/hour, with no other significant abnormalities.
The patient had no significant medical history of chronic diseases (e.g., hypertension or diabetes) and no significant family history.
Past medical history was non-contributory, including no history of surgery, trauma, or infectious diseases.
Physical examination findings were unremarkable.
Emergency laboratory tests revealed an elevated high-sensitivity C-reactive protein level of 19.77 mg/L. Other pa
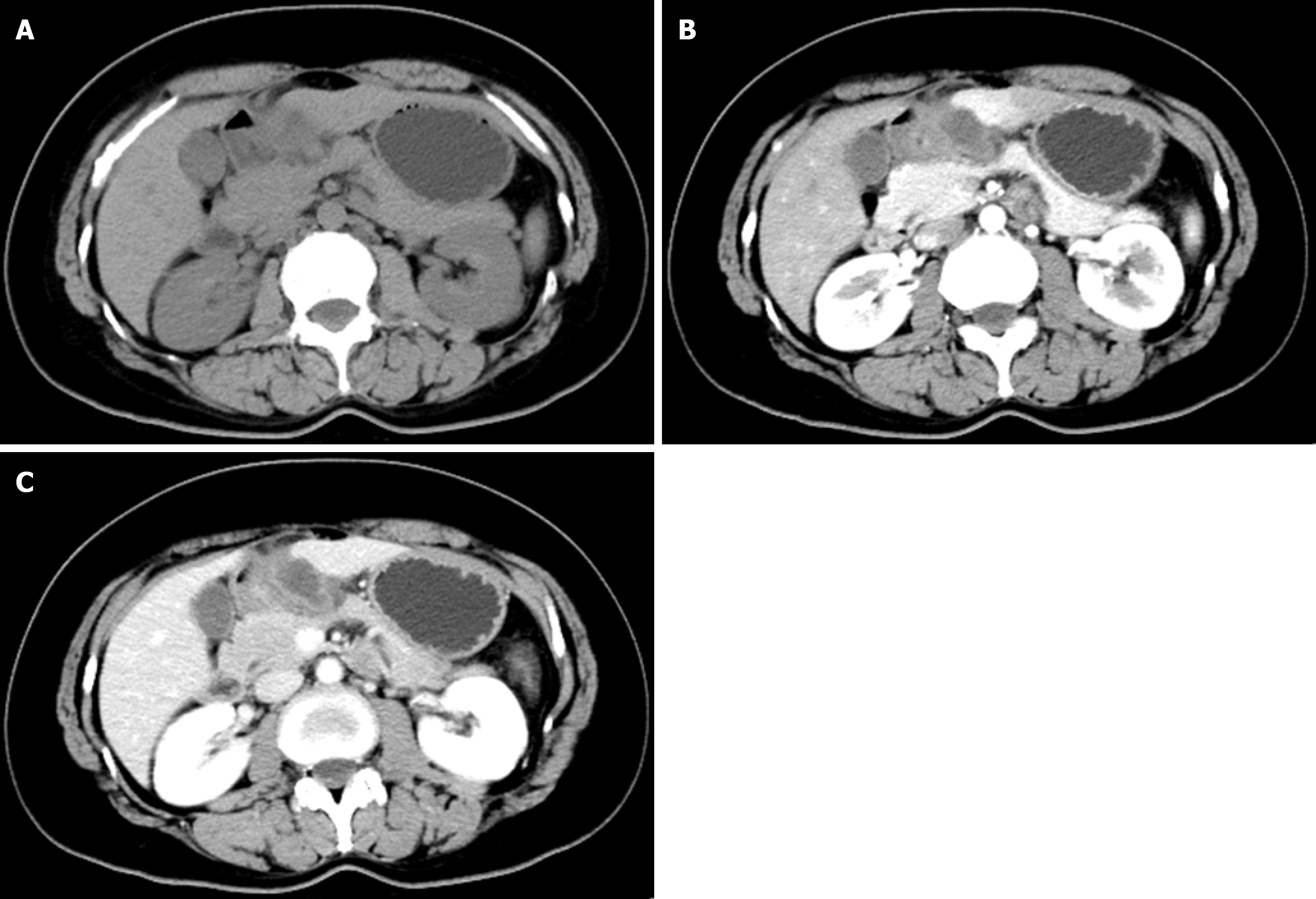
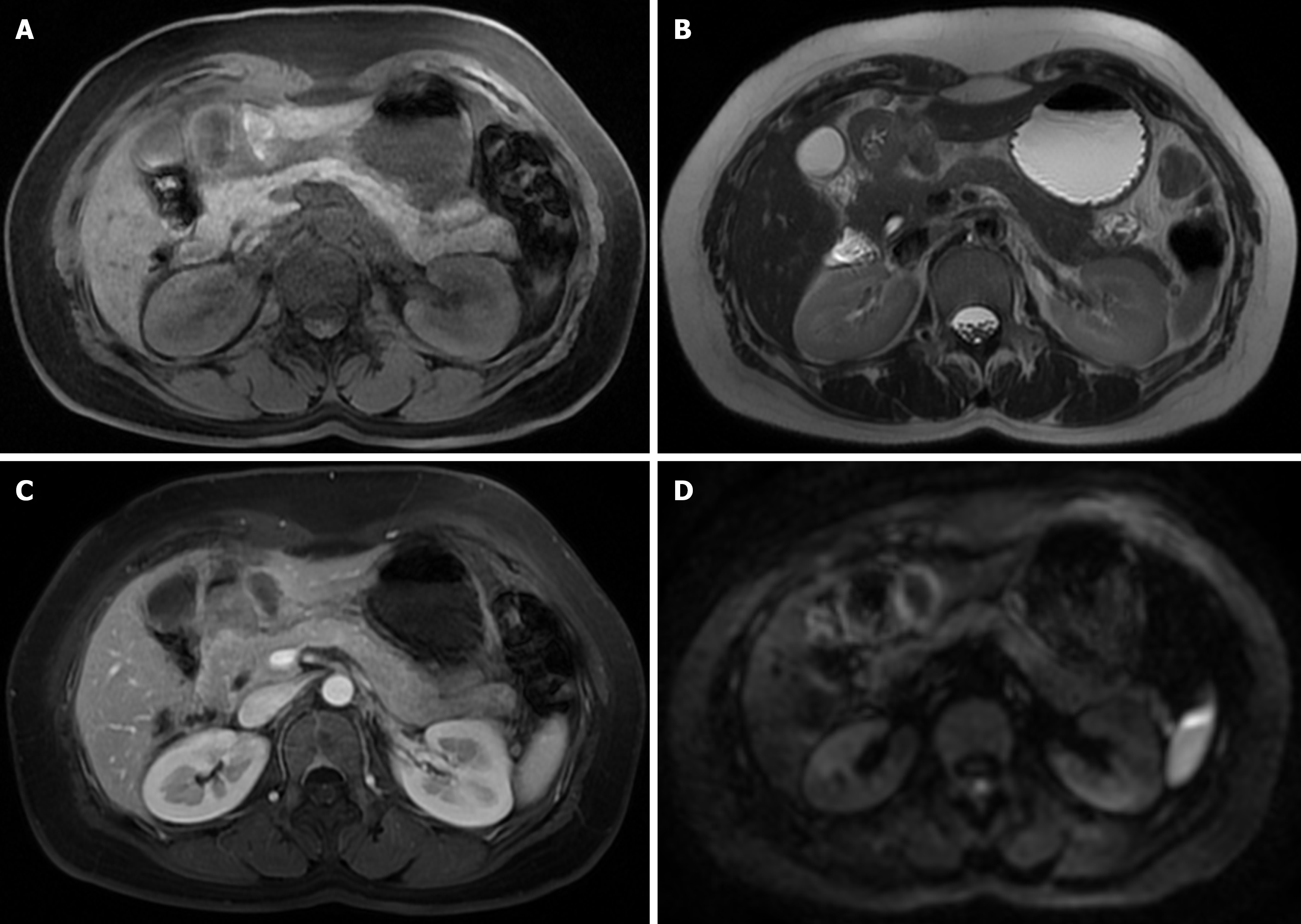
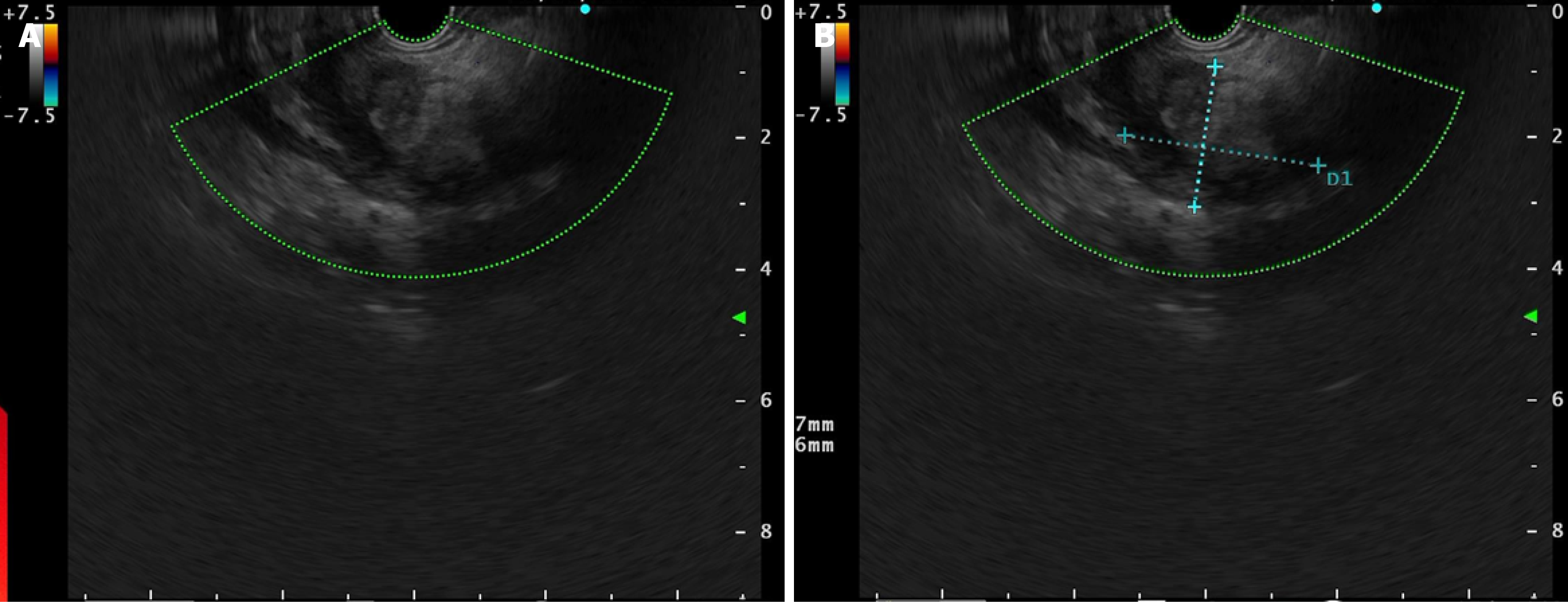
Contrast-enhanced abdominal CT demonstrated irregular thickening of the gastric antral wall with heterogeneous enhancement. A cystic lesion (3.4 cm × 2.2 cm) was found adjacent to the lesser curvature of the gastric antrum, exhibiting thick-walled morphology, blurred margins, and mild enhancement post-contrast (Figure 1). Two omental nodules near the inferior liver margin and enlarged lymph nodes in the hepatogastric ligament and mesentery were observed. MRI revealed an abnormal signal lesion (2.8 cm × 1.5 cm) lateral to the lesser curvature of the gastric antrum, showing hyperintensity on T1 weighted imaging (T1WI) (Figure 2), hypointensity on T2 weighted imaging (T2WI) (Figure 2), and no enhancement post-contrast (Figure 2). The vertical-axis ultrasound gastroscopy scan shows a uniform echo in the gastric body. A low-echo mass-like change approximately 3.0 cm × 2.2 cm in size is observed on the outer wall of the gastric antrum (Figure 3), with uneven internal echoes and visible high-echo structures (Figure 3). Based on the imaging findings, a hematoma or inflammatory lesion was suspected, with malignancy not excluded.
The final diagnosis of AA was established based on the surgical pathology findings.
Laparoscopic resection of the abdominal masses and adhesiolysis were performed. Gross pathology showed a well-circumscribed, encapsulated tumor with a raised surface. Histopathology demonstrated spindle cells dispersed within a myxocollagenous stroma. Immunohistochemistry findings showed positive cytokeratin (pan) (+), desmin (+), cluster of differentiation (CD) 34 (vascular +), β-catenin (cytoplasmic +), H3K27Me3 (+), RB1 (+), CD68 (scattered +), endoplasmic reticulum (focal +), Fli-1 (vascular +), CD31 (vascular +), and INI-1 (+); and negative CD117 (-), S-100 (-), smooth muscle actin (-), DOG-1 (-), ALK (-), SOX10 (-), STAT6 (-), EMA (-), NTRK (-), mesothelin (-), calretinin (-), TFE3 (-), and PR (-).
The patient underwent uneventful surgery and was discharged on postoperative day 2. Follow-up contrast-enhanced abdominal CT at 3 months showed no evidence of recurrence, and the patient is currently in a stable condition.
AA is a rare mesenchymal tumor that is classified as an aggressive tumor due to its local invasiveness and high recur
Usually, AA lesions are discovered through imaging examinations after consultation, so it is particularly important to identify AA during imaging. In the past, scholars have studied the manifestations of AA on CT, MRI, and B-ultrasound. Five female patients with AA underwent CT and MRI studies, which showed a well-defined mass that displaced adjacent structures. The tumor had low attenuation relative to muscle on CT. On MRI, the tumor was isointense relative to muscle on T1WI, hyperintense on T2WI, and enhanced after gadolinium contrast with a characteristic “whorl” internal pattern. MRI demonstrated the extent of the tumor and its relationship to the pelvic floor. The appearance of recurrent tumors was similar to that of the primary lesion[19].
On MRI, AA is hypodense relative to muscle and appears layered or swirling after contrast injection. AA is isointense relative to muscle on T1WI and hyperintense on T2WI, which is enhanced after gadolinium injection, indicating rich blood vessels. The tumor fibrovascular stroma is enhanced, showing a characteristic layered or swirling appearance[20].
AA may be identified on enhanced CT or MRI due to the characteristic internal structural features on contrast-enhanced CT or T1-weighted MRI, namely “swirling”, and the low-signal band structure of fibrovascular tissue can also be shown on T2WI. However, compared with CT, MRI can show the margins of the tumor relative to the surrounding structures, especially the dynamic contrast-enhanced sequence, so it is more helpful for preoperative assessment of the scope of surgery[21].
In this case, AA grew outside the gastric wall, and no characteristic “whirlpool” pattern was seen on CT and MRI. In addition, AA showed cystic lesions on CT, without the characteristic whirlpool sign of AA in the perineum. Therefore, we speculate that the previous typical AA imaging findings cannot be used for AA in rare sites such as the gastric wall.
In our case, we showed the image of AA under ultrasound gastroscopy for the first time, where low-echo mass-like changes outside the gastric antrum wall, uneven internal echoes, and high-echo structures were seen. This is different from the imaging manifestations of AA with medium-high echoes and rich blood vessels[22,23] under conventional percutaneous B-ultrasound. This may also suggest that the ultrasound images of AA in rare sites and those in common sites may be different.
AA is a benign tumor that usually grows slowly, has a tendency to local invasion and recurrence, but has a good overall prognosis and is rarely life-threatening[21]. Distant metastasis and even death are rarely reported[24-26].
When feasible, surgical resection is the most effective treatment for AA, but local lesions are prone to recurrence. However, if conditions permit after local recurrence, local surgical resection of the recurrence can still be performed[27]. Therefore, it is very important to evaluate the extent of the tumor through imaging examinations before surgery. In addition, gonadotropin-releasing hormone agonists (GnRH-a) can be used before surgery to reduce the size of the tumor and prevent postoperative recurrence. For patients who are unwilling to accept or cannot tolerate surgical treatment, GnRH-a alone can be tried[28]. In male patients, if the androgen receptor (AR) is positive, AR blocking drugs (bica
In summary, AA remains a condition prone to misdiagnosis, especially when it occurs in atypical locations such as non-perineal areas. Preoperative imaging remains the primary method for identifying the lesion. Although CT and MRI exhibit characteristic findings, the imaging manifestations of AA in non-typical locations remain atypical. The EUS features of AA occurring in the stomach also require further case accumulation for a more comprehensive understanding. Surgery remains the primary treatment modality. Postoperative follow-up with regular imaging is necessary to assess possible recurrence.
| 1. | Fletcher CD. The evolving classification of soft tissue tumours - an update based on the new 2013 WHO classification. Histopathology. 2014;64:2-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 355] [Article Influence: 27.3] [Reference Citation Analysis (1)] |
| 2. | Karwacki GM, Stöckli M, Kettelhack C, Mengiardi B, Studler U. Radiographic diagnosis and differentiation of an aggressive angiomyxoma in a male patient. J Radiol Case Rep. 2013;7:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 3. | Sylvester DC, Kortequee S, Moor JW, Woodhead CJ, Maclennan KA. Aggressive angiomyxoma of larynx: case report and literature review. J Laryngol Otol. 2010;124:793-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 4. | Wang Z, Liu Y, Yang L, Gu L, He Y, Huang D, Zhu G. Maxillary aggressive angiomyxoma showing ineffective to radiotherapy: a rare case report and review of literature. Int J Clin Exp Pathol. 2015;8:1063-1067. [PubMed] |
| 5. | Yamashita Y, Tokunaga O, Goto M. Aggressive angiomyxoma of the oral floor: report of a case. J Oral Maxillofac Surg. 2004;62:1429-1431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 6. | Nyandwi L, Idrissa S, Moustapha H, Ada MOA, Boubacar E, Boubacar I, Abdelfatteh Z, Amine K, Habibou A. Aggressive angiomyxoma as a rare cause of scrotum enlargement in a 10-month-old boy: a case report. J Med Case Rep. 2022;16:293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Zhu Z, Yan J, Tang G. Aggressive angiomyxoma of the prostate: A case report. Medicine (Baltimore). 2018;97:e13716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 8. | Heffernan EJ, Hayes MM, Alkubaidan FO, Clarkson PW, Munk PL. Aggressive angiomyxoma of the thigh. Skeletal Radiol. 2008;37:673-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 9. | Nayal B, Rao L, Rao AC, Sharma S, Shenoy R. Extragenital aggressive angiomyxoma of the axilla and the chest wall. J Clin Diagn Res. 2013;7:718-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 10. | Choi YD, Kim JH, Nam JH, Choi C, Na KJ, Song SY. Aggressive angiomyxoma of the lung. J Clin Pathol. 2008;61:962-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 11. | Tiwari N, Magu SK, Ghoshal UC, Sharma AK. Aggressive angiomyxoma of the jejunum: a rare cause of obscure gastrointestinal bleeding. Trop Gastroenterol. 2007;28:183-185. [PubMed] |
| 12. | Narang M, Das P, Singh AN, Madhusudhan KS. Aggressive angiomyxoma of the stomach: a novel site of a rare neoplasm. BMJ Case Rep. 2022;15:e249182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Sutton BJ, Laudadio J. Aggressive angiomyxoma. Arch Pathol Lab Med. 2012;136:217-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 95] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 14. | Nucci MR. Mesenchymal Lesions of the Lower Genital Tract. Surg Pathol Clin. 2009;2:603-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 15. | Akram H, Tran D, Rehman R, Al-Wahab Z. Aggressive angiomyxoma of left buttock. BMJ Case Rep. 2021;14:e241550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 16. | Sozutek A, Irkorucu O, Reyhan E, Yener K, Besen AA, Erdogan KE, Gonlusen G, Doran F. A Giant Aggressive Angiomyxoma of the Pelvis Misdiagnosed as Incarcerated Femoral Hernia: A Case Report and Review of the Literature. Case Rep Surg. 2016;2016:9256749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 17. | Korecka KK, Hyla-Klekot LE, Kudela GP, Paleń PA, Kajor MW, Koszutski TK. Aggressive Angiomyxoma in an 11-Year-Old Boy - Diagnostic and Therapeutic Dilemmas: An Unusual Case Report and Review of the Literature. Urology. 2020;144:205-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 18. | Nucci MR, Weremowicz S, Neskey DM, Sornberger K, Tallini G, Morton CC, Quade BJ. Chromosomal translocation t(8;12) induces aberrant HMGIC expression in aggressive angiomyxoma of the vulva. Genes Chromosomes Cancer. 2001;32:172-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 60] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 19. | Jeyadevan NN, Sohaib SA, Thomas JM, Jeyarajah A, Shepherd JH, Fisher C. Imaging features of aggressive angiomyxoma. Clin Radiol. 2003;58:157-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 69] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Al-Umairi RS, Kamona A, Al-Busaidi FM. Aggressive Angiomyxoma of the Pelvis and Perineum: A Case Report and Literature Review. Oman Med J. 2016;31:456-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 21. | Cardenas Lara FJ, Bauzon JS, Perry WRG, Kelley SR. Aggressive Angiomyxoma of the Pelvis: 35-Year Experience. Dis Colon Rectum. 2024;67:514-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Ismail MI, Wong YP, Tan GH, Fam XI. Paratesticular aggressive angiomyxoma: A rare case. Urol Ann. 2017;9:197-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 23. | Sakata K, Ishikawa S, Tokue A, Hirota N. Aggressive angiomyxoma of the scrotum. Urol Int. 1997;58:247-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 24. | Siassi RM, Papadopoulos T, Matzel KE. Metastasizing aggressive angiomyxoma. N Engl J Med. 1999;341:1772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 108] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 25. | Blandamura S, Cruz J, Faure Vergara L, Machado Puerto I, Ninfo V. Aggressive angiomyxoma: a second case of metastasis with patient's death. Hum Pathol. 2003;34:1072-1074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 119] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 26. | Geng J, Cao B, Wang L. Aggressive angiomyxoma: an unusual presentation. Korean J Radiol. 2012;13:90-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 27. | Aminimoghaddam S, Sarchami N, Mahboub SS. Second recurrence of aggressive vulvar angiomyxoma: a case report. J Int Med Res. 2023;51:3000605231189366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 28. | Wang Y, Bu X, Liu Y, Xing Y, Tong Q. Characteristics and treatment strategies of aggressive angiomyxoma in women: A retrospective review of 87 cases. Front Surg. 2023;10:966971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 29. | Franza A, Gusmaroli E, Fabbroni C, Vigorito R, Pasquali S, Casali PG, Sanfilippo RG. Long-term disease stability with bicalutamide in a man with aggressive angiomyxoma: case report and state of art. Front Oncol. 2023;13:1260668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 30. | Rhomberg W, Jasarevic Z, Alton R, Kompatscher P, Beer G, Breitfellner G. Aggressive angiomyxoma: irradiation for recurrent disease. Strahlenther Onkol. 2000;176:324-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (2)] |
| 31. | Li L, Chuang H, He-Nan L, Dong-Yuan L, Qing-Hua L, Wei L, Liang-Shan L, Ting-Yuan L, Xue-Quan H. Aggressive angiomyxoma of the pelvis and abdominal wall: Dramatic response to chemical ablation therapy. Front Oncol. 2023;13:1154283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |