INTRODUCTION
Liver cancer ranks among the most prevalent malignancies globally and stands as a leading contributor to cancer-related mortality, surpassed only by lung cancer and colorectal cancer[1,2]. Characterized by high malignancy, challenges in early detection, and generally short survival expectancy, liver cancer poses significant clinical and public health burdens[3]. In recent years, percutaneous local ablation has emerged as a promising therapeutic modality for small hepatic tumors, typically measuring less than 3-4 cm in diameter. Commonly employed ablation techniques encompass temperature-based approaches, including radiofrequency ablation (RFA)[4], microwave ablation (MWA)[5], and cryoablation[6]. These modalities achieve tumor destruction through the induction of coagulative necrosis, which occurs when tissue temperatures exceed 60 °C or drop below -40 °C. However, a critical limitation of temperature-based ablation techniques is their lack of selectivity for tumor tissue, often resulting in collateral damage to adjacent structures such as blood vessels and bile ducts[7]. Furthermore, thermal or cryoablation exhibits reduced efficacy for lesions adjacent to major vasculature. This phenomenon is primarily attributed to the “heat sink effect”, where circulating blood dissipates thermal energy, thereby compromising the achievement of therapeutic temperatures within the target tissue[8].
Irreversible electroporation (IRE) is an innovative ablation modality that directly disrupts cell membranes without relying on thermal changes[9]. It achieves in situ tumor inactivation while sparing adjacent structures such as blood vessels, bile ducts, nerves, and intestinal tracts. Conventional IRE utilizes 80-120 unipolar pulses with durations ranging from 50 to 100 μs and an electric field intensity of 1500 V/cm[10]. However, prolonged electrical pulses carry risks, including interference with cardiac rhythm, induction of arrhythmias, and triggering of local or systemic muscle contractions. Recently, a novel technique termed high-frequency IRE (H-FIRE) has been developed. H-FIRE, a novel optimized derivative of IRE, employs alternating polarity pulses that allow nerves and muscles to return to a resting state, significantly reducing intraprocedural muscle contractions[11,12]. Studies have shown that, compared with conventional IRE, H-FIRE more effectively and uniformly elevates the induced transmembrane potential of cells to the threshold required for effective electroporation, resulting in a more homogeneous ablation zone[13]. H-FIRE has demonstrated promising tumor ablation efficacy and has been shown to inhibit tumor growth. Currently, it is recognized for its effectiveness and safety in the treatment of hepatocellular carcinoma[14], prostate lesions[15], intracranial tumors[16], lung cancer[17], and other malignancies.
Studies have demonstrated that H-FIRE can induce immunogenic cell death (ICD) in tumors, accompanied by the release of tumor-associated antigens and damage-associated molecular patterns (DAMPs), including high mobility group box 1 protein (HMGB1), calreticulin (CRT), and ATP[18]. Clinical studies in hepatocellular carcinoma patients have confirmed that H-FIRE activates systemic anti-tumor immunity by increasing local and peripheral blood levels of interleukins, tumor necrosis factors, and interferons, as well as enhancing the infiltration of activated cytotoxic T lymphocytes and dendritic cells (DC)[19]. Moreover, as a non-thermal modality, H-FIRE minimizes injury to tumor-surrounding blood vessels and lymphatic vessels[20], which facilitates the migration of immune cells into the tumor microenvironment (TME), laying a critical foundation for the combination of H-FIRE with immunotherapy[21].
Cancer immunotherapy represents an emerging therapeutic strategy that activates immune cells. Immunotherapy has the potential to eradicate solid tumors and circulating tumor cells by modulating immune responses, and it can even induce long-term immunological memory[22]. Monoclonal antibodies that block the programmed death-1 (PD-1)/programmed death-ligand 1 (PD-L1) interaction by binding to either the receptor or ligand have shown substantial clinical efficacy across various cancer types. Despite the systemic anti-tumor potential of PD-1/PD-L1 inhibitor, its efficacy is often limited by insufficient activation of the immune system and low response rates in many tumor types[23], while single-target therapies are further compromised by tumor heterogeneity[24]. As a key component of immune activation, DC orchestrates the generation of potent cytotoxic T lymphocytes and facilitates the uptake of tumor debris post-H-FIRE ablation[25]. Toll-like receptors (TLR), a major class of pattern recognition receptors, initiate innate immunity and regulate adaptive immunity by recognizing DAMPs[26]. Resiquimod (R848), a TLR7/8 agonist, exhibits robust immune adjuvant activity by potentiating DAMPs recognition, activating antigen-presenting cells (APC), and creating an inflammatory TME[27,28], making it an ideal candidate to enhance the anti-tumor immune response induced by H-FIRE.
Accordingly, integrating H-FIRE with PD-1/PD-L1 inhibitor BMS-1 and TLR7/8 agonist R848 constitutes a novel and promising combinatorial strategy. This triple therapy integrates the local ablation and ICD-inducing effects of H-FIRE, the immune checkpoint-blocking capacity of BMS-1, and the immune-adjuvant property of R848, synergistically addressing the limitations of single or dual therapies for liver cancer treatment.
MATERIALS AND METHODS
Reagents
BMS-1 (cat. no. HY-19991) and R848 (cat. no. HY-13740) were purchased from MedChemExpress (Monmouth Junction, NJ, United States). The following reagents and antibodies were obtained from Invitrogen (Carlsbad, CA, United States): APC anti-mouse CD3 (cat. no. 17-0032-80), FITC anti-mouse CD4 (cat. no. 11-0041-81), PE anti-mouse CD8a (cat. no. 12-0081-81), APC anti-mouse CD25 (cat. no. 17-0251-81), PE anti-mouse/rat FOXP3 (cat. no. 12-5773-82), PerCP/Cyanine5.5 anti-mouse F4/80 (cat. no. 45-4801-80), FITC anti-mouse CD11b (cat. no. 11-0112-81), PE anti-mouse CD206 (cat. no. 12-2061-80), APC anti-mouse CD86 (cat. no. 17-0826-81), FITC anti-mouse CD11c (cat. no. 11-0114-82), PE anti-mouse CD80 (cat. no. 12-0801-81), and anti-mouse CD16/32 antibody (cat. no. 14-0161-82). Triton X-100 (cat. no. 1139ML500) was supplied by BioFroxx (Einhausen, Germany). Collagenase IV (cat. no. C8160), Collagenase I (cat. no. C8140), Deoxyribonuclease I (DNase I, cat. no. D8071), red blood cell lysis buffer (cat. no. R1010), and phosphate-buffered saline (PBS) buffer (cat. no. P1022) were obtained from Solarbio (Beijing, China). The eosin staining solution (cat. no. ZLI-9613), hematoxylin staining solution (cat. no. ZLI-9610), and neutral balsam (cat. no. ZLI-9555) were purchased from Zhongshan Golden Bridge Biotechnology (Beijing, China). The TUNEL Detection Kit (cat. no. G1504-100T) was obtained from Servicebio (Wuhan, China). Anti-fluorescence quenching mounting medium (cat. no. 0100-20) was purchased from SouthernBiotech (Birmingham, AL, United States). The Ki67 antibody (cat. no. 27309-1-AP) was obtained from Proteintech (Rosemont, IL, United States). The HMGB1 antibody (cat. no. ET1601-2) was obtained from Hangzhou Huabio Biotechnology Co., Ltd. (Hangzhou, China). The CRT antibody (cat. no. bsm-51748M) was obtained from Beijing Biosynthesis Biotechnology Co., Ltd. (Beijing, China). The goat anti-rabbit IgG/FITC (cat. no. A0562) and goat anti-mouse IgG/FITC (cat. no. A0568) were obtained from Shanghai Beyotime Biotechnology Co., Ltd. (Shanghai, China).
Cell culture and animals
Murine hepatoma 22 (H22) cells were purchased from Wuhan Pricella Biotechnology Co., Ltd. (catalog no. CL-0341, Wuhan, China) and maintained in Roswell Park Memorial Institute 1640 medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin. Cultures were incubated at 37 °C in a humidified 5% CO2 atmosphere. Male BALB/c mice (20-22 g) were obtained from Beijing Huafukang Biotechnology Co., Ltd. (Beijing, China) and housed under controlled conditions: Temperature 22 ± 2 °C, relative humidity 50% ± 10%, and a 12-hour light/dark cycle. Standard chow and sterile water were provided ad libitum throughout the study. All the animal experiments complied with the guidelines of the Tianjin Medical Experimental Animal Care, and animal protocols were approved by the Institutional Animal Care and Use Committee of Yi Shengyuan Gene Technology (Tianjin) Co., Ltd., No. YSY-DWLL-2024756. Euthanasia was performed via cervical dislocation in accordance with ethical guidelines.
H-FIRE ablation combined with BMS-1 and R848
To establish primary liver cancer tumors, H22 cells (1 × 106) were subcutaneously injected into the right lower dorsal region of mice. Following injection, tumor growth was monitored, and the long diameter (a) and short diameter (b) of the maximum tumor cross-section were measured using an electronic digital caliper. Tumor volume was calculated using the formula: V = a × b2/2. When the longest diameter of the right tumor reached approximately 7-8 mm, tumor-bearing mice were randomly assigned to eight groups (control, BMS-1, R848, BMS-1 + R848, H-FIRE, H-FIRE + BMS-1, H-FIRE + R848, and H-FIRE + BMS-1 + R848) using a random number table method implemented by an independent researcher not involved in subsequent experimental operations. This randomization process ensured balanced distribution of baseline tumor volume and mouse body weight across groups. All subsequent procedures, including tumor volume measurement, general health status monitoring, survival assessment, and data recording, were performed by investigators blinded to the group assignments via a predefined coding system (each group was labeled with a unique alphanumeric code that was decoded only after data collection was completed).
For BMS-1 administration, each mouse received an intraperitoneal injection of 200 μg BMS-1 starting 1 day before H-FIRE treatment, with injections repeated every 2 days for a total of 4 doses. For R848 treatment, 30 μg R848 was intratumorally injected into the right tumor, initiated 1 day prior to H-FIRE treatment and repeated every 2 days for 4 total doses. For H-FIRE treatment, mice were first anesthetized via isoflurane inhalation. Once fully anesthetized, they were positioned prone, and the right tumor was treated using two sterilized ablation needles (Intelligent Health, Tianjin, China). The needles were spaced 0.5 cm apart and secured with plastic spacers, then connected to an H-FIRE generator (YTL-GM01, Intelligent Health, China). Ablation parameters included 1000 V asymmetric square-wave pulses, consisting of 100 pulse groups (10 pulses per group), with positive pulse width of 7 μs, negative pulse width of 3 μs, and an interval of 5 μs between adjacent sub-pulses[29]. To establish bilateral H22 subcutaneous tumors, 1 × 106 H22 cells were injected subcutaneously into the right lower dorsal region (primary tumor) and 0.5 × 106 H22 cells into the left lower dorsal region (distant secondary tumors, mimicking liver cancer metastases). Mice were monitored daily for general health status. Humane euthanasia was performed when excessive tumor burden was observed or animals exhibited moribund conditions, in accordance with humane endpoint criteria.
T2-weighted magnetic resonance imaging
Magnetic resonance (MR) imaging was performed on mice using a Siemens Magnetom Verio 3.0T scanner (Siemens, Germany) at two time points: 1 day before and 1 week after H-FIRE treatment. Prior to each imaging session, mice in all experimental groups were anesthetized via intraperitoneal injection of 1% pentobarbital sodium at a dose of 60 mg/kg. Animals were positioned prone on the MR scanning bed, with dedicated coils placed over their subcutaneous tumors. After secure immobilization relative to the coils, MR scanning was initiated. The specific parameters for the T2-weighted imaging (T2WI) sequence were as follows: Echo time = 77 ms; repetition time = 3000 ms; number of averages = 4; slice thickness = 1 mm; distance factor = 20%; field of view = 70 mm; field of view phase = 79.9%; slice oversampling = 50%; and slices per slab = 8.
Hematoxylin-eosin staining of paraffin sections
For Hematoxylin-eosin (HE) staining, 4 μm paraffin sections were prepared from embedded tissues, mounted on slides, and baked at 60 °C for 2 hours. Sections were immersed sequentially in xylene (I: 20 minutes; II: 20 minutes; III: 15 minutes), anhydrous ethanol (I: 5 minutes; II: 5 minutes), 85% ethanol (5 minutes), 75% ethanol (5 minutes), and distilled water. Sections were stained with Harris hematoxylin for 5-10 minutes, rinsed with tap water, differentiated in 1% hydrochloric acid-ethanol for several seconds, rinsed with tap water for 10 minutes, blued in bluing solution for 5 minutes, and washed with running water. Sections were stained with eosin for 1-3 minutes and rinsed with tap water. Sections were dehydrated in 75% ethanol (5 minutes), 85% ethanol (5 minutes), 95% ethanol (5 minutes), anhydrous ethanol (I: 5 minutes; II: 5 minutes), and xylene (5 minutes), then mounted with neutral balsam.
Immunohistochemistry of paraffin sections
Paraffin sections were deparaffinized in xylene (I: 20 minutes; II: 20 minutes), rehydrated through a graded ethanol series (anhydrous to 70%), and rinsed in distilled water. Antigen retrieval was performed by heating sections in EDTA buffer (pH 9.0) in a pressure cooker for 3 minutes once steam emerged, followed by cooling and 3 × 3 minutes PBS (pH 7.4) washes. Endogenous peroxidase activity was blocked with blocking reagent (15 minutes, room temperature, dark), followed by 3 × 3 minutes PBS washes. Sections were incubated with primary antibody in a humidified chamber at 4 °C overnight, then washed 3 × 3 minutes in PBS. After incubation with reaction enhancer (37 °C, 20 minutes) and enzyme-labeled polymer (37 °C, 20 minutes) with PBS washes between steps, sections were stained with freshly prepared DAB (color development was monitored microscopically) and rinsed in tap water to terminate the reaction. Nuclei were counterstained with hematoxylin (5 seconds), followed by tap water rinse. Finally, sections were dehydrated through a graded ethanol series (70% to anhydrous) and xylene (5 minutes), then mounted with neutral balsam. The semi-quantitative Ki67-positive rate was calculated as (positive nuclei/total nuclei) × 100% based on random non-overlapping high-power fields of the immunohistochemistry sections.
Immunofluorescent staining of paraffin sections
Paraffin sections were dewaxed sequentially in xylene (I, II, III; 20 minutes each), then rehydrated through a graded ethanol series (anhydrous, 90%, 80%, 70%; 3 minutes each) and rinsed with distilled water. Antigen retrieval was performed in EDTA buffer (pH 9.0) using a pressure cooker for 3 minutes (starting from steam emission), with care taken to prevent sections drying. After cooling, sections were washed in PBS (pH 7.4) three times (3 minutes each). Sections were circled with a hydrophobic histochemical pen, blocked with 1% bovine serum albumin at room temperature for 60 minutes, then incubated overnight at 4 °C with primary antibodies (HMGB1, 1:1000; CRT, 1:200) in a humidified chamber. After PBS washes (3 × 3 minutes), sections were incubated with the species-matched fluorescent secondary antibodies at room temperature for 1 hour in the dark. Following PBS washes (3 × 10 minutes), nuclei were counterstained with DAPI for 5 minutes at room temperature. Sections were washed in PBS (3 × 5 minutes), observed under a fluorescence microscope, and mounted with anti-fluorescence quenching medium. The mean fluorescence intensity (MFI) of HMGB1 and CRT in mouse tumor paraffin sections was quantified using ImageJ software.
Flow cytometric analysis of immune cells
For flow cytometric analysis, single-cell suspensions were prepared from fresh tumor and spleen tissues. Tissues were rinsed with ice-cold PBS, minced, and digested with collagenase I, collagenase IV, and DNase I at 37 °C for 50 minutes with shaking. Both tissues were passed through a 40 μm strainer into centrifuge tubes (cells released by gentle disruption with a syringe plunger), and residual cells were recovered by rinsing the strainer with serum-containing medium. Cells were pelleted by centrifugation at 300 × g for 5 minutes. Red blood cells were lysed by adding 3 mL lysis buffer to each sample, followed by gentle vortexing and 15-minute incubation on ice (with two additional vortexes). Leukocytes were pelleted at 450 × g for 5 minutes at 4 °C; supernatants were aspirated, and cells were washed once with PBS. Prior to staining, cells were blocked with anti-mouse CD16/32 antibody for 30 minutes.
T cell (CD3/CD4/CD8) and DC (CD11c/CD80/CD86) staining: Cells were resuspended in 100 μL PBS, incubated with APC anti-mouse CD3 (1:40), FITC anti-mouse CD4 (1:200), PE anti-mouse CD8a (1:80), FITC anti-mouse CD11c (1:200), PE anti-mouse CD80 (1:200), and APC anti-mouse CD86 (1:200) at 4 °C for 30 minutes in the dark, washed twice with PBS, resuspended in PBS, and analyzed by flow cytometry.
Tregs (CD4/CD25/FOXP3) staining: Cells were resuspended in 100 μL PBS and stained with FITC anti-mouse CD4 (1:200) and APC anti-mouse CD25 (1:100) at 4 °C for 30 minutes in the dark. After adding 1 mL True-Nuclear™ 1X Fix Concentrate, cells were vortexed and incubated at room temperature for 50 minutes in the dark. Following centrifugation (300 × g, 5 minutes) with 1 mL True-Nuclear™ 1X perm buffer, pellets were resuspended in 100 μL perm buffer, incubated with PE anti-mouse/rat FOXP3 (1:20) at 4 °C for 30 minutes in the dark, centrifuged again with perm buffer, washed twice with PBS, resuspended, and analyzed by flow cytometry.
Macrophage (F4/80/CD11b/CD86/CD206) staining: Cells were resuspended in 100 μL PBS, stained with PerCP/Cyanine5.5 anti-mouse F4/80 (1:40), FITC anti-mouse CD11b (1:100), and APC anti-mouse CD86 (1:200) at 4 °C for 30 minutes in the dark, and washed once with PBS. Cells were fixed with 500 μL paraformaldehyde (30 minutes, room temperature), washed with PBS, and permeabilized with 500 μL 0.5% Triton X-100 (10 minutes, room temperature). After centrifugation and washing, cells were resuspended in 100 μL PBS, stained with PE anti-mouse CD206 (1:100) at 4 °C for 30 minutes in the dark, washed twice with PBS, resuspended, and analyzed by flow cytometry.
TUNEL staining of paraffin sections
Paraffin sections were deparaffinized in xylene (I: 20 minutes; II: 20 minutes), rehydrated through a graded ethanol series (anhydrous to 70%), and rinsed in distilled water. Sections were digested with Proteinase K (20 μg/mL in PBS) at 37 °C for 20 minutes, followed by 3 × 5 minutes PBS washes. After equilibration with 50 μL equilibration buffer (10 minutes, room temperature), sections were incubated with TDT reaction mixture (recombinant TDT enzyme/CF488-dUTP labeling mix/equilibration buffer = 1:5:50) at 37 °C for 1 hour. Sections were then washed 4 × 5 minutes in PBS, counterstained with DAPI (5 minutes, room temperature, dark), and rinsed 3 × 5 minutes in PBS. Finally, sections were mounted with anti-fluorescence quenching medium. The images obtained were captured via confocal microscopy and subsequently analyzed using ImageJ software.
Safety assessment
Body weight was monitored at the indicated time points. Major organs, including the heart, liver, spleen, lung, and kidney, were subjected to HE staining. For blood biochemical analysis, blood samples were collected via right eyeball removal into heparinized 1.5 mL EP tubes. After standing at room temperature for 30 minutes, samples were centrifuged at 5000 rpm for 15 minutes at 4 °C. The supernatant was transferred to new EP tubes and analyzed using an automatic veterinary biochemical analyzer, with parameters set according to the manufacturer's instructions. The following indicators were assessed: Creatine kinase MB isoenzyme, a marker of cardiac tissue damage; renal function indicators, including creatinine and blood urea nitrogen; and hepatic function indicators, namely aspartate aminotransferase and alanine aminotransferase.
Statistical analysis
All statistical analyses and graphing were conducted using GraphPad Prism 10.4.1 (GraphPad Software Inc., San Diego, CA, United States). Continuous variables were presented as mean ± SD. Between two groups, comparisons were made using the unpaired Student’s t-test; for multiple groups, one-way analysis of variance (one-way ANOVA) was employed. Survival curves were constructed via the Kaplan-Meier approach, and differences in survival across groups were assessed using the log-rank test. Statistical significance was defined as a P value < 0.05.
RESULTS
H-FIRE induces immunogenic tumor cell death and releases DAMPs
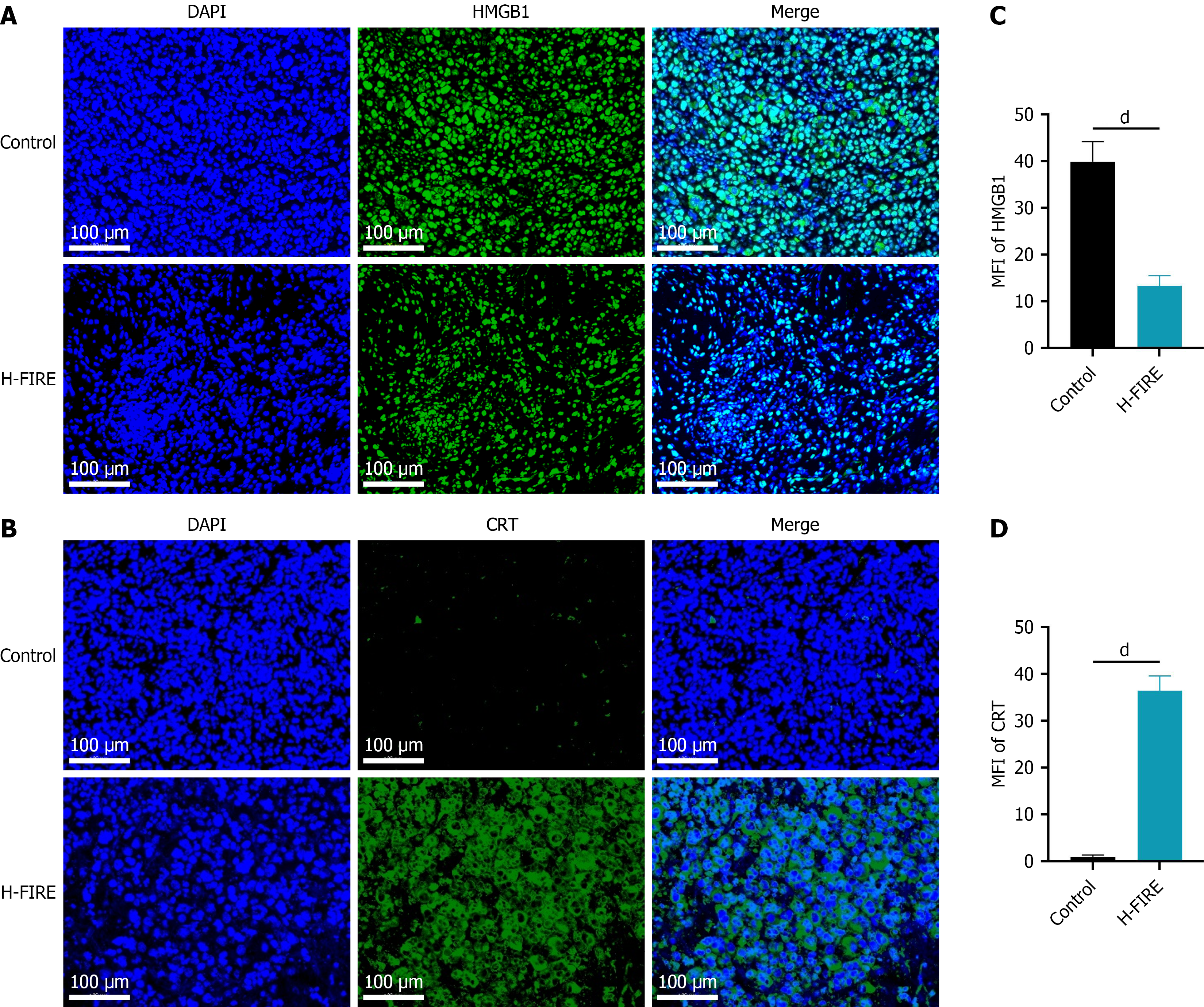
To assess the capacity of H-FIRE to induce ICD in tumor cells and release DAMPs, this study detected two key DAMPs, CRT and HMGB1, in subcutaneous tumor-bearing mice following H-FIRE treatment. HMGB1, a nuclear-binding protein primarily localized in the cell nucleus, is overexpressed in tumors. It is released into the extracellular space upon ICD activation. The translocation of HMGB1 from the nucleus to the extracellular fluid is also a hallmark of ICD, contributing to the recruitment and activation of immune cells as well as tissue repair. In normal cells, CRT is predominantly localized in the endoplasmic reticulum. Upon ICD activation, CRT translocates to the cell membrane surface via direct transmembrane transport or the endoplasmic reticulum-Golgi secretion pathway. As an “eat-me signal”, membrane-exposed CRT can be recognized by receptors on immune cells (e.g., DC and macrophages), facilitating the phagocytosis and clearance of damaged cells by immune cells.
In this study, immunofluorescent staining for HMGB1 (Figure 1A) and CRT (Figure 1B), together with quantitative analysis of MFI, was performed at 1 week post-treatment. The results demonstrated that the MFI of nuclear HMGB1 in the H-FIRE group was significantly lower than that in the control group (13.35 ± 2.17 vs 39.91 ± 4.27, P < 0.0001; Figure 1C), indicating that HMGB1 was markedly released into the extracellular space following H-FIRE treatment. Conversely, the MFI of extracellular CRT in the H-FIRE group was significantly higher than that in the control group (36.38 ± 3.19 vs 0.94 ± 0.36, P < 0.0001; Figure 1D), suggesting a substantial increase in extracellular CRT exposure compared with the control group.
Figure 1 High-frequency irreversible electroporation induces immunogenic cell death in subcutaneous tumor cells.
A and B: Immunofluorescence images of key immunogenic cell death markers (high mobility group box 1 protein/calreticulin) in tumors (DAPI for nuclear counterstaining); C and D: Quantitative analysis of their mean fluorescence intensity (n = 8). dP < 0.0001. H-FIRE: High-frequency irreversible electroporation; HMGB1: High mobility group box 1 protein; CRT: Calreticulin; MFI: Mean fluorescence intensity.
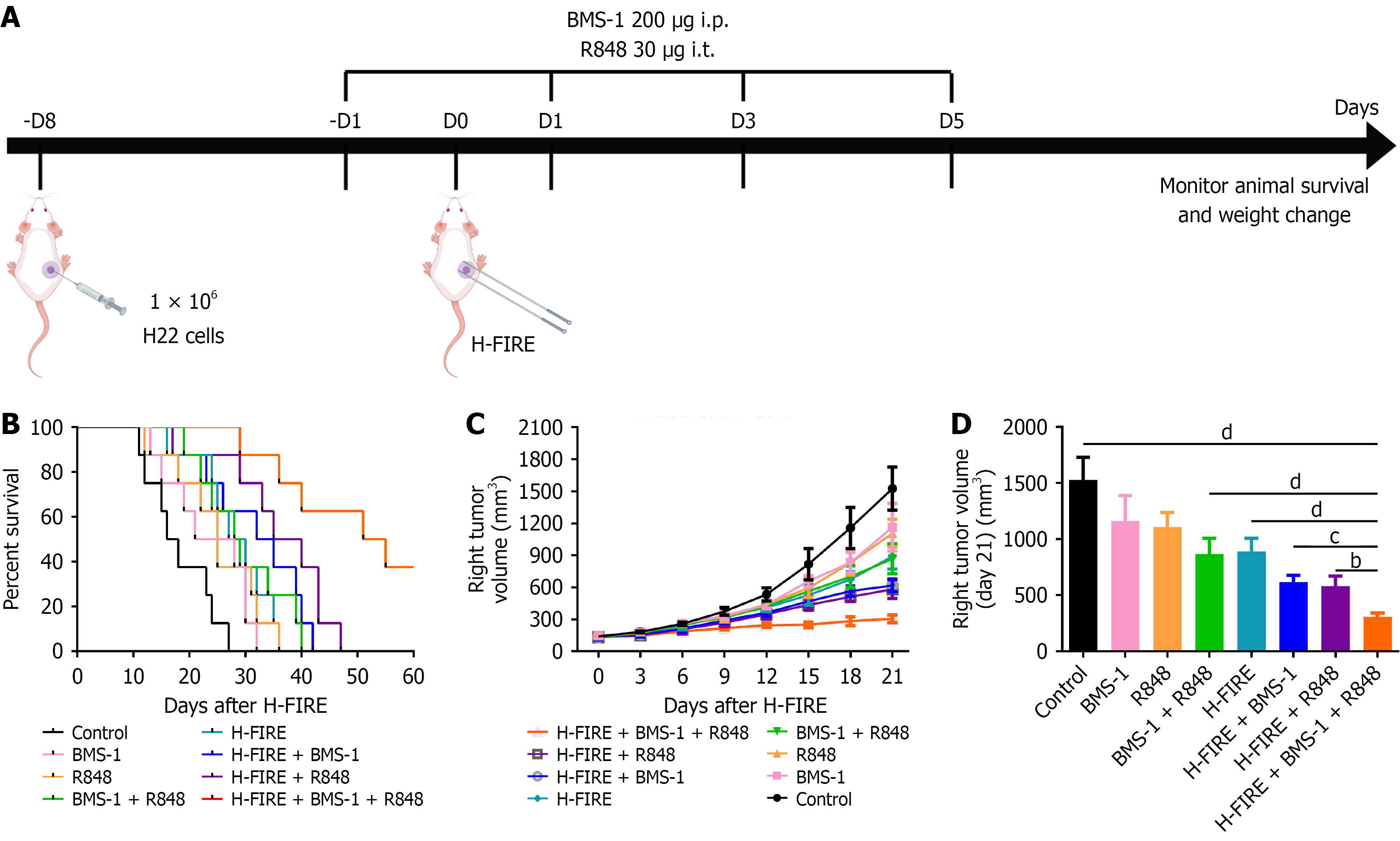
H-FIRE combined with BMS-1 and R848 inhibits tumor growth and improves survival
In this study, mice were randomly assigned to eight groups: Control, BMS-1, R848, BMS-1 + R848, H-FIRE, H-FIRE + BMS-1, H-FIRE + R848, and H-FIRE + BMS-1 + R848. Survival duration was monitored following different therapeutic interventions (Figure 2A). The results demonstrated that the median survival times of each group were 17, 24.5, 25, 28.5, 28.5, 33.5, 37.5, and 53 days, respectively. Compared with other treatment modalities, the H-FIRE + BMS-1 + R848 triple combination group significantly prolonged the survival of tumor-bearing mice (P < 0.05; Figure 2B). Analysis of subcutaneous tumor volume showed that baseline volumes were comparable across all eight groups before treatment. After treatment initiation, subcutaneous tumors in the control group grew rapidly, whereas tumor growth was markedly retarded in the H-FIRE + BMS-1 + R848 group (Figure 2C). On day 21 post-treatment, the tumor volume in the H-FIRE + BMS-1 + R848 group (305.3 ± 35.0 mm3) was significantly smaller than that in all other treatment groups (P < 0.05; Figure 2D).
Figure 2 Combinatorial therapy with high-frequency irreversible electroporation, BMS-1, and R848 prolongs survival and inhibits tumor growth in hepatoma 22 subcutaneous tumor-bearing mice.
A: Schematic of the hepatoma 22 subcutaneous tumor model and experimental treatment timeline; B: Kaplan-Meier survival curves across the different treatment groups (n = 8); C: Tumor growth kinetics presented as dynamic volume curves over the observation period (n = 8); D: Comparison of subcutaneous tumor volumes measured on day 21 post-treatment (n = 8). bP < 0.01, cP < 0.001, and dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
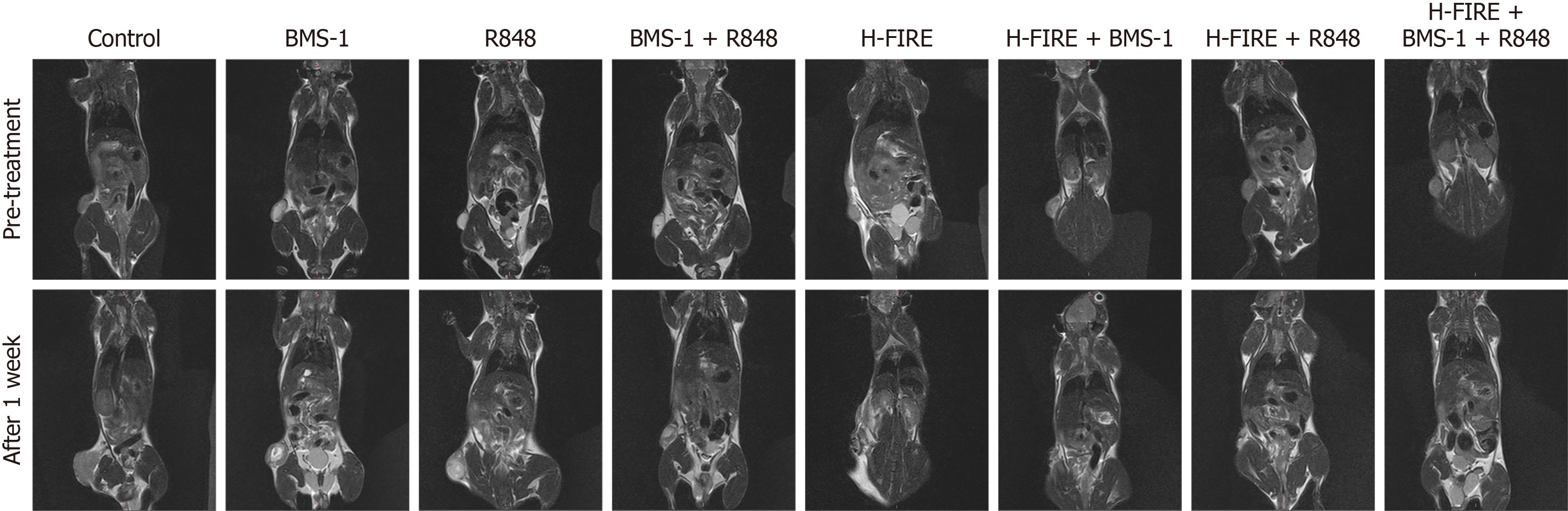
Coronal T2WI demonstrated dynamic changes in subcutaneous tumors among the eight groups of mice 1 day prior to H-FIRE treatment and on day 7 post-treatment (Figure 3). Specifically, T2WI of the control group revealed that the subcutaneous tumor on day 7 post-treatment was significantly larger than that before treatment, with a marked increase in tumor height. Meanwhile, subcutaneous tumors in the BMS-1, R848, and BMS-1 + R848 groups also exhibited varying degrees of enlargement on day 7 post-treatment compared with pre-treatment measurements. In contrast, tumor height in the H-FIRE, H-FIRE + BMS-1, H-FIRE + R848, and H-FIRE + BMS-1 + R848 groups showed no significant changes 1 week after treatment relative to pre-treatment values. Notably, the tumor height following H-FIRE + BMS-1 + R848 treatment was significantly reduced compared with that before treatment, whereas no significant change was observed in the tumor’s long diameter.
Figure 3 Representative coronal T2-weighted magnetic resonance scans of mouse tumors before treatment and 1 week post-treatment across all experimental groups.
H-FIRE: High-frequency irreversible electroporation.
H-FIRE combined with BMS-1 and R848 promotes tumor cell death and inhibits cell proliferation
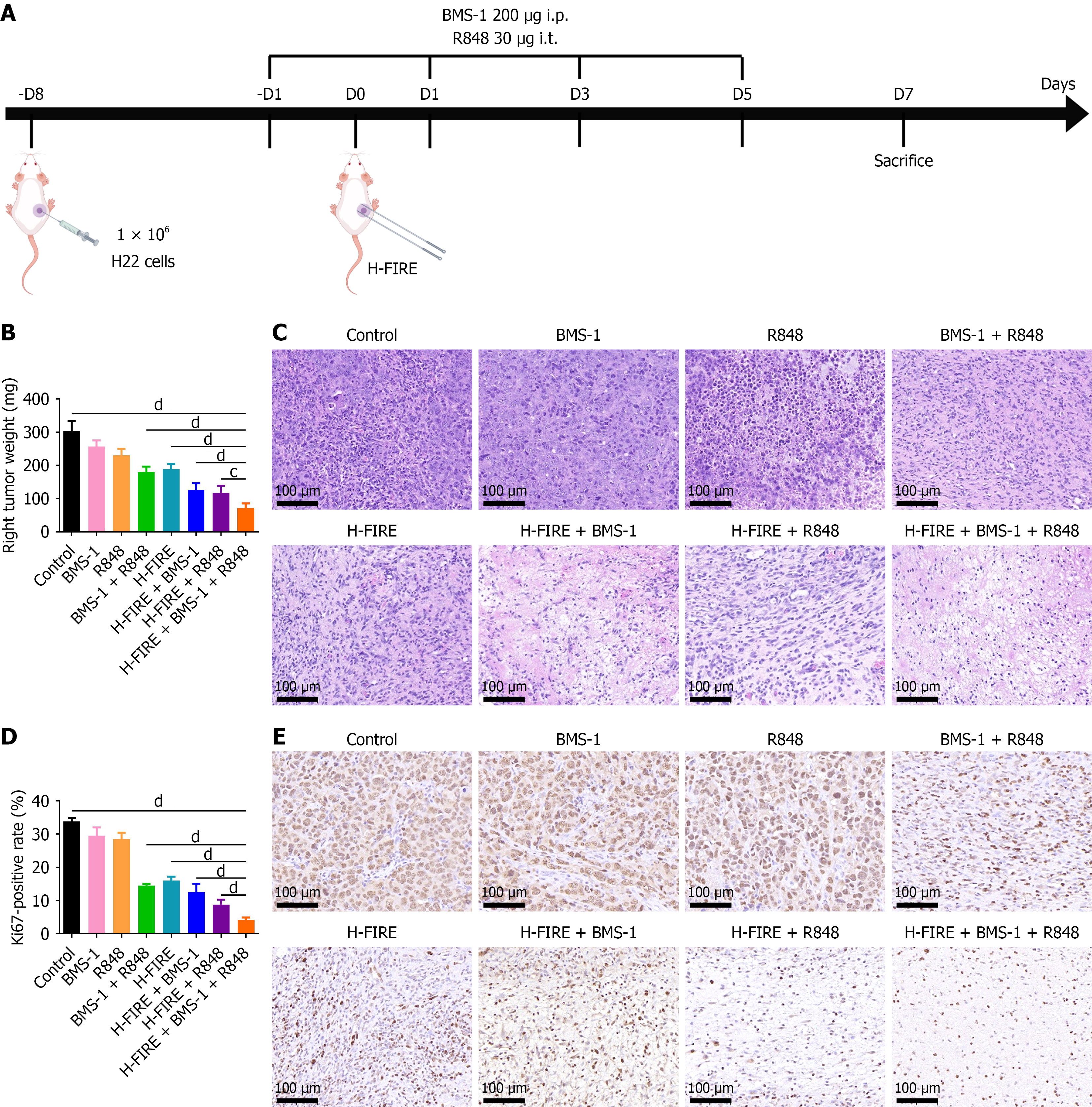
In this study, mice from each group were sacrificed at 1 week post-treatment, and subcutaneous tumors were weighed (Figure 4A). Notably, the tumor weight in the H-FIRE + BMS-1 + R848 group (71.47 ± 13.84 mg) was significantly lower than that in all other groups (P < 0.05; Figure 4B). Tumor tissue sections were subjected to HE staining and Ki67 immunohistochemical staining to evaluate post-treatment morphological changes and tumor cell proliferation.
Figure 4 High-frequency irreversible electroporation combined with BMS-1 and R848 impairs tumor cell viability and suppresses proliferation in hepatoma 22 subcutaneous tumors.
A: Schematic of the hepatoma 22 subcutaneous tumor-bearing mouse model and treatment regimen; B: Weight of excised subcutaneous tumors from the right flank at 1 week post-treatment; C: Hematoxylin-eosin staining of subcutaneous tumor sections across treatment groups (n = 8); D: Quantitative analysis of Ki67-positive rate (%) in right-sided subcutaneous tumors 1 week post-treatment (n = 8); E: Representative images of Ki67 immunohistochemical staining in right-flank subcutaneous tumors 1 week post-treatment. cP < 0.001, and dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
HE staining of tumor tissues revealed differences in intratumoral cell distribution across groups (Figure 4C). Specifically, tumors in the control group maintained an intact structure, with numerous densely packed cells exhibiting deep nuclear staining and marked atypia. In contrast, tumor tissues from the H-FIRE + BMS-1 + R848 group showed extensive necrosis, significantly sparse cell distribution, and characteristic degenerative changes, including vacuolation, cytoplasmic pallor, nuclear pyknosis and loss, and disruption of cellular architecture, indicating that the combination therapy exerted a potent cytotoxic effect on tumor cells. Ki67 immunohistochemical staining demonstrated the highest positive rate in the control group and the lowest in the H-FIRE + BMS-1 + R848 group. Quantitative analysis confirmed that the Ki67 positive rate in the latter group was 4.11% ± 0.78%, which was significantly lower than that in other treatment groups (P < 0.05; Figure 4D and E).
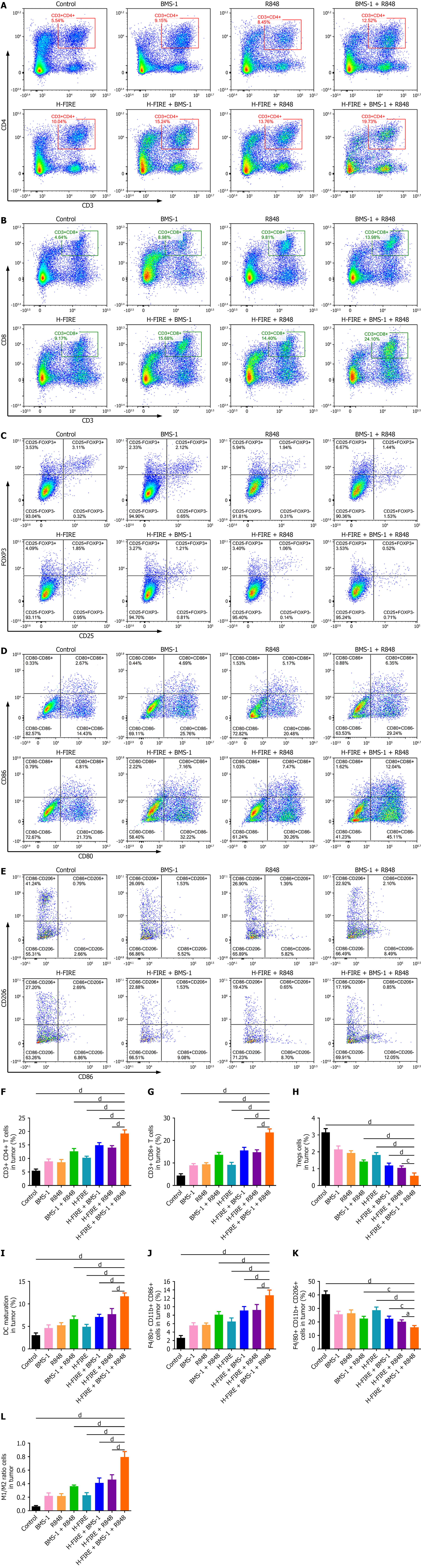
H-FIRE combined with BMS-1 and R848 improves the immune microenvironment of residual tumors
In this study, flow cytometry was used to analyze the proportion of immune cell subsets in tumors from each group at 1 week post-treatment, aiming to characterize the systemic immune response. The results demonstrated that the H-FIRE + BMS-1 + R848 group exhibited a significant increase in the proportions of CD3+ CD4+ T cells, CD3+ CD8+ T cells, CD11c+ CD80+ CD86+ DC, and F4/80+ CD11b+ CD86+ M1 macrophages, as well as an elevated M1/M2 macrophage ratio (all P < 0.05). Concurrently, the proportions of CD4+ CD25+ FOXP3+ Tregs and F4/80+ CD11b+ CD206+ M2 macrophages were significantly reduced (all P < 0.05; Figure 5). These findings indicate that the H-FIRE combined immunotherapy regimen can effectively activate anti-tumor immune responses in mice, thereby exerting therapeutic effects against tumors.
Figure 5 High-frequency irreversible electroporation combined with BMS-1 and R848 reprograms the tumor immune microenvironment in hepatoma 22 subcutaneous tumors.
A-E: Representative flow cytometry dot plots of intratumoral immune cell subsets post-treatment across all groups: CD3+ CD4+ T cells, CD3+ CD8+ T cells, CD4+ CD25+ FOXP3+ Tregs, CD11c+ CD80+ CD86+ dendritic cells, F4/80+ CD11b+ CD86+ M1 macrophages, and F4/80+ CD11b+ CD206+ M2 macrophages; F-K: Quantitative analysis of the proportions of the above-listed intratumoral immune cell subsets in each group post-treatment (n = 5); L: Quantitative analysis of the intratumoral M1/M2 macrophage ratio across groups post-treatment (n = 5). aP < 0.05, cP < 0.001, and dP < 0.0001. H-FIRE: High-frequency irreversible electroporation.
Evaluation of in vivo toxicity of H-FIRE combined with BMS-1 and R848
The aforementioned studies have demonstrated that H-FIRE combined with BMS-1 and R848 can significantly inhibit tumor growth and prolong survival in mice. However, in addition to evaluating therapeutic efficacy, the safety of different treatment modalities must also be considered. Therefore, throughout the experiment, the mental state, water intake, diet, defecation, and urination of each group were closely monitored and recorded, with no obvious abnormalities observed. Mice were weighed regularly every three days after H-FIRE treatment (Figure 6A), and comparisons of body weights on day 21 revealed no significant differences among groups (Figure 6B), indicating that H-FIRE combined with BMS-1 and R848 did not induce significant toxic side effects or damage in mice.
Figure 6 Safety assessment of high-frequency irreversible electroporation combined with BMS-1 and R848 in hepatoma 22 tumor-bearing mice.
A: Dynamic body weight changes of mice across all groups post-treatment (n = 8); B: Quantification of mouse body weights in each group at 21 days post-treatment (n = 8); C: Hematoxylin-eosin staining of major organs (heart, liver, spleen, lung, kidney) in mice from each group post-treatment, showing no obvious histopathological damage; D-H: Serum biochemical analysis shows no significant intergroup differences in liver function markers (aspartate aminotransferase, alanine aminotransferase), a cardiac injury marker (creatine kinase MB isoenzyme), and renal function parameters (creatinine, blood urea nitrogen) post-treatment (n = 8). H-FIRE: High-frequency irreversible electroporation; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; CK-MB: Creatine kinase MB isoenzyme; CREA: Creatinine; BUN: Blood urea nitrogen.
To further verify the biological safety of H-FIRE combined with BMS-1 and R848, paraffin-embedded pathological section analysis was performed on major organs (including the heart, liver, spleen, lung, and kidney) of mice in each group. HE staining results showed no obvious pathological changes, such as damage, metastasis, necrosis, or inflammation, in the major organs of any group (Figure 6C). Additionally, peripheral blood was collected via orbital sampling for blood biochemical analysis. The results demonstrated no significant intergroup differences in cardiac injury markers (creatine kinase MB isoenzyme), renal function indicators (creatinine, blood urea nitrogen), or liver function parameters (alanine aminotransferase, aspartate aminotransferase) (Figure 6D-H) after treatment. These findings indicate that H-FIRE combined with BMS-1 and R848 does not significantly impair the biological functions of major organs in mice.
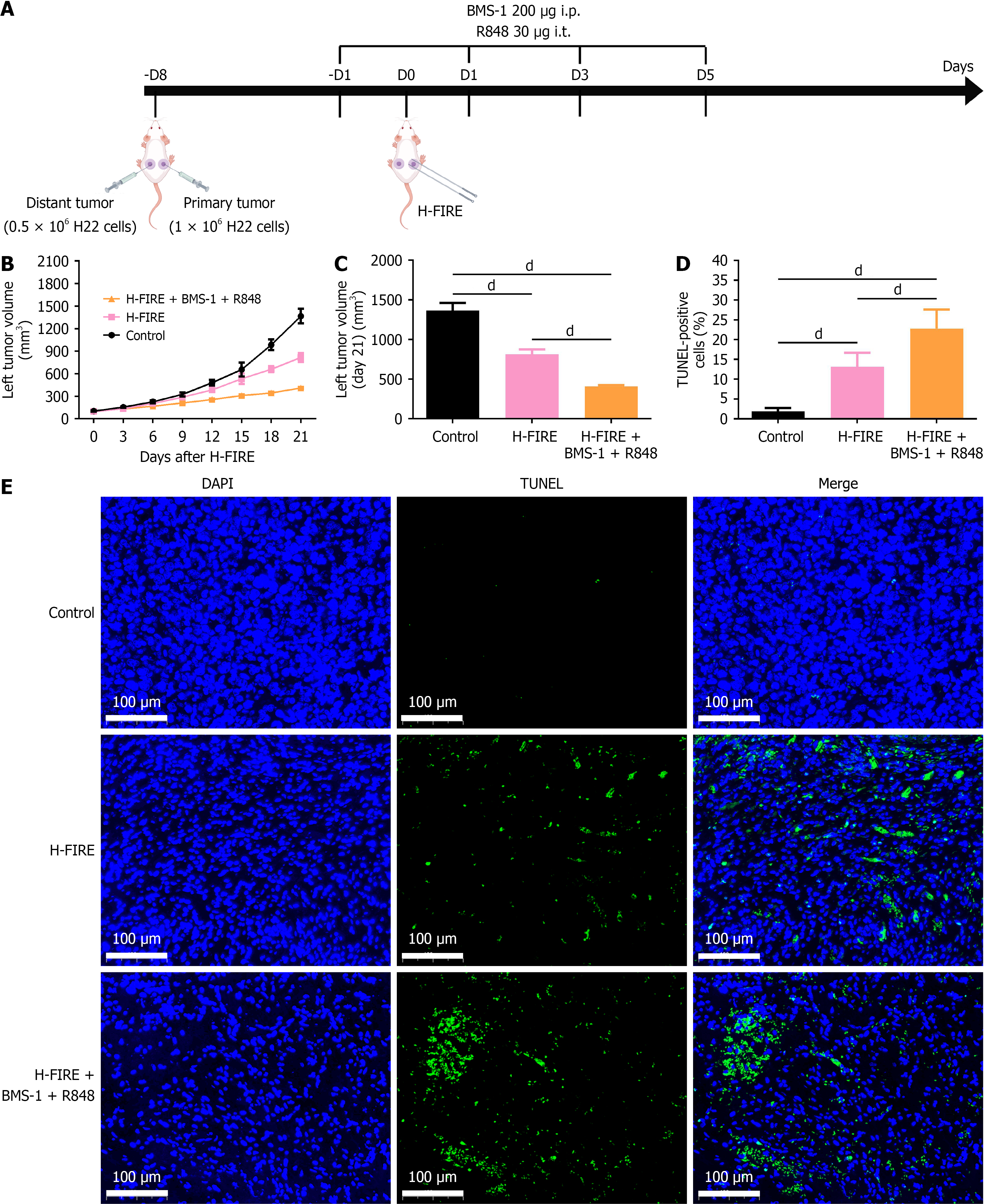
H-FIRE combined with BMS-1 and R848 induced a potent abscopal effect and systemic anti-tumor immunity
In this study, 1 × 106 H22 tumor cells were subcutaneously injected into the right dorsal region to simulate primary tumors, while 0.5 × 106 H22 cells were injected into the left dorsal region to model metastatic tumors (Figure 7A). Only the right tumors received designated treatments, whereas the left tumors remained untreated. Notably, despite the absence of direct H-FIRE or immunotherapeutic intervention on distant (left) tumors, the tumor volumes in both the H-FIRE monotherapy group and H-FIRE + BMS-1 + R848 group were significantly smaller than those in the control group. Furthermore, the H-FIRE + BMS-1 + R848 group exhibited a significantly stronger inhibitory effect on distant tumors compared with the H-FIRE monotherapy group (Figure 7B). On day 21, the mean tumor volume in the combined therapy group (409.5 ± 15.3 mm3) was significantly lower than that in the control group (1365.7 ± 96.5 mm3) and H-FIRE monotherapy group (816.0 ± 59.2 mm3) (Figure 7C; P < 0.05).
Figure 7 Therapeutic efficacy of high-frequency irreversible electroporation combined with BMS-1 and R848 in the hepatoma 22 bilateral tumor-bearing mice.
A: Schematic of the hepatoma 22 bilateral tumor-bearing mice and treatment regimen; B: Dynamic changes in left tumor volume of hepatoma 22 bilateral tumor-bearing mice post-treatment (n = 8); C: Comparison of left tumor volumes on day 21 post-treatment (n = 8); D: Quantitative analysis of TUNEL-positive cell proportion in left tumors 1 week post-treatment (n = 8); E: Representative TUNEL staining images of left-sided tumors from each group 1 week post-treatment. dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
Pathological assessment of left tumor cell apoptosis was performed at 1 week post-H-FIRE treatment. The results revealed that the apoptotic rate of distant tumor cells in the H-FIRE combined immunotherapy group (22.82% ± 4.76%) was significantly higher than that in the H-FIRE monotherapy group (13.20% ± 3.48%) and control group (1.88% ± 0.86%) (Figure 7D and E; P < 0.05). Additionally, the apoptotic rate in the H-FIRE monotherapy group was significantly elevated compared with the control group.
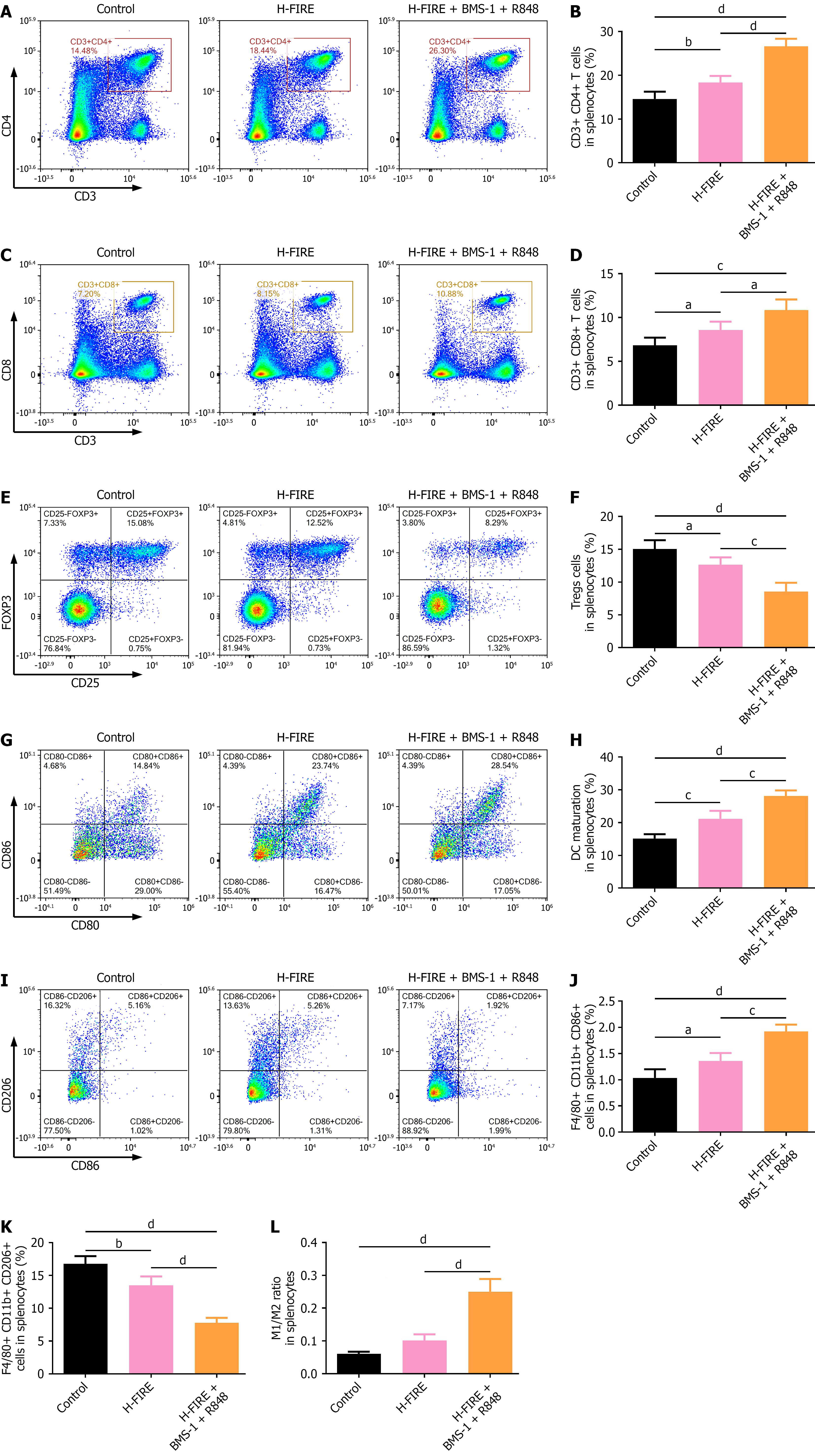
To evaluate systemic immune responses, flow cytometric analysis of splenic immune cell subsets was conducted at 1 week post-treatment. The results demonstrated that the H-FIRE combined immunotherapy group exhibited a significant increase in CD3+ CD4+ T cells and CD3+ CD8+ T cells, as well as in the proportions of DC and M1 macrophages, alongside an elevated M1/M2 macrophage ratio. Concurrently, the proportions of Tregs and M2 macrophages (Figure 8) were significantly reduced. These findings indicate that H-FIRE + BMS-1 + R848 can potently activate systemic anti-tumor immunity in mice and induce a robust abscopal effect.
Figure 8 High-frequency irreversible electroporation combined with BMS-1 and R848 elicits systemic antitumor immunity by modulating splenic immune cell subsets.
A and B: Proportion of splenic CD3+ CD4+ T cells; C and D: Proportion of splenic CD3+ CD8+ T cells; E and F: Proportion of splenic CD4+ CD25+ FOXP3+ Tregs; G and H: Proportion of splenic CD11c+ CD80+ CD86+ dendritic cells; I-L: Polarization of splenic macrophages: Proportion of F4/80+ CD11b+ CD86+ M1 and F4/80+ CD11b+ CD206+ M2 macrophages and the M1/M2 macrophage ratio. Each panel shows representative flow cytometry plots alongside quantitative data (n = 5). aP < 0.05, bP < 0.01, cP < 0.001, and dP < 0.0001. H-FIRE: High-frequency irreversible electroporation.
DISCUSSION
IRE is a non-thermal tissue ablation technique that has been progressively integrated into clinical practice globally, as evidenced by a systematic review and meta-analysis by Gupta et al[30] showing efficacy and safety in malignant liver tumors (25 studies, 776 subjects) with pooled 12-, 24-, and 36-month overall survival rates of 81.39%, 61.47%, and 40.88%, respectively, and 12- and 24-month progression-free survival rates of 64.19% and 49.05%, respectively. The overall complication rate was 23.7%, with most complications being minor. IRE holds promise as an effective adjuvant therapy for patients with advanced liver cancer. H-FIRE, a second-generation IRE modality, exhibits enhanced ablation efficacy and reduces muscle contractions compared to conventional IRE[16]. In cells subjected to either conventional IRE or H-FIRE, two distinct modes of cell death are observable: Accidental cell death and regulated cell death[29]. Accidental cell death can trigger the release of pro-inflammatory molecules but has limited capacity to activate adaptive immune responses. In contrast, regulated cell death, encompassing processes such as apoptosis, pyroptosis, ferroptosis, and necroptosis, can also induce inflammation while being more prone to stimulating adaptive immune responses[29]. Studies have demonstrated that H-FIRE significantly increases the proportion of cells undergoing regulated cell death relative to conventional IRE[31]. Our study’s detection of HMGB1 and CRT revealed that H-FIRE treatment promotes ICD in tumor cells, leading to increased nuclear release of HMGB1 and enhanced membrane exposure of CRT. As two representative DAMPs markers, the released HMGB1 and exposed CRT function as “eat-me signals” to recruit APC. These findings indicate that H-FIRE can augment immunogenicity within the TME, thereby converting “cold tumors” into “hot tumors”. This transformation is conducive to reversing the immunosuppressive TME, sensitizing tumors to immunotherapy, and establishing a foundation for subsequent immune responses. The findings of this study demonstrate that H-FIRE treatment effectively ablates primary tumors, leading to a significant increase in necrosis. Even partial ablation can shift the TME from an immunosuppressive state to a pro-inflammatory one. This transformation may be associated with the release of DAMPs from electroporated cells, recognized by the innate immune system, and tumor-associated antigens, which are presented by DC and macrophages[32]. Unlike thermal ablation modalities, H-FIRE does not generate sufficient heat to induce protein denaturation, thereby preserving the integrity of DAMPs and tumor-associated antigens released from electroporated cells. In contrast, the immune responses elicited by RFA and MWA are typically transient. Specifically, RFA results in an 87.5% reduction in cytotoxic T lymphocytes shortly after the procedure[33], while MWA leads to a decrease in T cells 72 hours post-ablation[34]. These short-lived T-cell responses may stem from the denaturation of tumor antigens released by thermal ablation, which subsequently diminishes the release of pro-inflammatory DAMPs.
Following H-FIRE ablation, critical structures in the treatment area, such as the extracellular matrix and blood vessels, remain preserved. The density and stiffness of the extracellular matrix may decrease, while tumor-associated vascular permeability may increase; these changes help stimulate local inflammation and enhance the infiltration of anti-tumor immune cells to target residual cancer cells[35]. In this study, 1 week after H-FIRE treatment, the levels of CD3+ CD4+ T cells and CD3+ CD8+ T cells remained significantly higher than those in the control group, reaching 1.87-fold and 2.07-fold of the control levels, respectively. Furthermore, H-FIRE exerts effects on both proliferating and non-proliferating cells within the electric field. Consequently, in addition to a marked reduction in tumor cells, persistent cells (e.g., cancer stem cells) and immunosuppressive cells (e.g., M2 macrophages, Tregs, and myeloid-derived suppressor cells) can also be eliminated[13]. The results of this study show that, following H-FIRE treatment, the levels of immunosuppressive cells (M2 macrophages and Tregs) were significantly lower compared to the control group. Consistent with these findings, clinical studies have reported that patients with localized pancreatic cancer who underwent IRE treatment exhibited a decrease in systemic Tregs and an increase in activated T cells two weeks post-treatment[36]. H-FIRE currently faces two major challenges: First, the limited volume of tissue it can ablate; second, the recurrence of inhibitory immune responses several weeks post-treatment. Large tumors not only diminish the ablation efficacy but also sustain the local immunosuppressive TME, which indirectly impairs therapeutic immune responses. This phenomenon involves the reaccumulation of Tregs, upregulation of PD-L1, and various other mechanisms that remain under investigation[37]. To ensure the clinical success of H-FIRE, extending beyond mere tumor shrinkage, it is critical to optimize its combination with immunosuppressive blockers and/or adjuvants.
PD-1 is expressed on the surface of cytotoxic T lymphocytes and binds to PD-L1 on tumor cells, leading to T-cell exhaustion and promoting tumor growth and metastasis. Recent studies comparing IRE with cryoablation and thermal ablation have demonstrated that combining anti-PD-1 therapy with IRE yields the most potent synergistic effect, resulting in prolonged tumor-free survival, increased CD8+ T-cell infiltration, and prevention of tumor recurrence. Additionally, modulation of the immunosuppressive TME has enhanced the efficacy of DC vaccination following IRE. In this study, H-FIRE combined with BMS-1 treatment enhanced the migratory capacity of CD8+ T cells, leading to a 1.70-fold increase in intratumoral CD8+ T cells 1 week post-treatment compared to H-FIRE alone. Moreover, a reduction of approximately 22.3% in M2 macrophages was observed compared to H-FIRE monotherapy. A retrospective clinical trial indicated that patients receiving IRE in combination with anti-PD-1 therapy exhibited improved overall survival (35.03 months vs 15.87 months) and progression-free survival (14.33 months vs 7.47 months) compared to those treated with IRE alone[38]. Importantly, no treatment-related mortality was reported in either group, and there were no statistically significant differences in complications or adverse events between the two groups.
PD-1/PD-L1 antibodies have been approved by the United States Food and Drug Administration for the treatment of various solid tumors and have exhibited favorable anti-tumor immune responses. However, several challenges persist: For instance, the response rate to immune checkpoint blockade (ICB) remains relatively low (ranging from 10% to 30%), and drug resistance represents a notable issue in clinical practice[39]. These limitations, low response rates and drug resistance, can be partially attributed to insufficient antigen presentation and the existence of an immunosuppressive TME[40]. The immunosuppressive TME comprises numerous tumor cells and various immune-regulatory cells that can inhibit the activity and infiltration of cytotoxic T cells. Additionally, it recruits immunosuppressive cells such as Tregs and myeloid-derived suppressor cells, while also accelerating tumor invasion and progression. Specifically, the deeper regions of tumor tissues often exhibit limited infiltration of cytotoxic T lymphocytes, accompanied by the accumulation of immunosuppressive immune cells[41]. To achieve effective tumor eradication and prevent recurrence and metastasis, enhancing the infiltration of cytotoxic T lymphocytes and reversing the immunosuppressive TME are regarded as crucial steps. In the early stages of tumor immunotherapy, tumor antigens must be released from tumor cells and cross-presented to CD8+ T cells by APC, such as DC, to enable appropriate recognition of the tumor by the host immune system. APC, including DC and macrophages, are typically in an undifferentiated state but can be activated upon antigen capture. They play a pivotal role in the complex immune network[42]. Upon receiving signals from APC, immature T cells can differentiate into various effector T cells, including CD4+ and CD8+ T cells. These T cells, together with natural killer cells, modulate the TME, ultimately contributing to the elimination of cancer cells.
TLR are innate inducers of immune responses, predominantly expressed on APC, with DC functioning as the most potent APC. TLR are also expressed at lower levels on epithelial cells and tumor cells. They possess the capacity to recognize a diverse array of exogenous and endogenous danger signals, a property that is particularly critical for immune cells infiltrating tumors. Given the roles of these immune cell populations in tumor growth and anti-tumor immune responses, TLR may represent viable targets for cancer immunotherapy[43]. Consequently, researchers have extensively explored immune adjuvants that activate TLR to potentiate immune responses against cancer cells. Among the notable therapeutic agents, TLR7 agonists (e.g., R837) and TLR7/8 dual agonists (e.g., R848) have demonstrated significant efficacy in enhancing anti-tumor immunity[44]. In particular, R848 exhibits greater potency than R837 and possesses improved water solubility. Additionally, R848 plays a pivotal role in promoting the maturation of DC and polarizing tumor-associated macrophages from the M2 to the M1 phenotype[45]. R848, a potent activator of DC, induces their maturation and stimulates T cells to potentiate anti-tumor responses, with demonstrated efficacy in both cutaneous malignancies[46] and leukemia[47]. However, systemic administration of TLR agonists is often associated with unfavorable pharmacokinetic profiles, including short half-lives, low therapeutic indices, and systemic cytokine release that can trigger toxic effects. Consequently, despite their significant anti-tumor activity in preclinical models, systemic administration of TLR agonists raises safety concerns in clinical settings. Recent studies indicate that intratumoral injection of TLR agonists achieves higher local drug concentrations and elicits robust anti-tumor responses[48]. Importantly, this route of administration not only impacts the treated tumor but also exerts potent anti-tumor effects on untreated lesions, occasionally leading to remission and the development of anti-tumor immune memory. In the present study, R848 was administered locally within the tumor.
The results showed that the combination of H-FIRE and R848 increased the M1/M2 macrophage ratio in tumors. Furthermore, this treatment significantly activated DC responses in vivo, with activation levels 2.53-fold and 1.57-fold higher than those in the control group and the H-FIRE monotherapy group, respectively. This activation was also notably greater than that observed in the H-FIRE + BMS-1 group (7.72% vs 7.11%), indicating a synergistic effect. Moreover, R848 effectively reduced the infiltration of inhibitory immune cells, including M2 macrophages and Tregs. CD4+ and CD8+ T cells were primarily recruited to the tumor site. These findings suggest that the R848 adjuvant sustains T-cell activity by reprogramming the TME through DC activation, thereby maintaining anti-tumor immunity. Mature DC mediate anti-tumor immunity by presenting tumor antigens and providing co-stimulatory signals essential for T-cell activation. Similar findings have been reported in lymphoma mouse models: Compared to IRE combined with anti-PD-1, IRE monotherapy and IRE combined with TLR3/9 agonists induced more robust antigen-specific CD8+ T-cell responses and inhibited tumor growth[49]. Additionally, Yang et al[50] demonstrated that IRE combined with a DC vaccine extended the median survival time of KPC tumor-bearing mice to 70 days. When paired with DC vaccines, IRE promotes ICD and reduces immunosuppressive components in the TME of locally advanced pancreatic cancer, leading to increased infiltration of CD8+ T cells and granzyme B+ cells.
H-FIRE requires a fully functional immune system to maximize its therapeutic efficacy. The combination of H-FIRE with R848 and anti-PD-1 antibodies is critical for inducing robust anti-tumor immune memory and achieving long-term tumor control[51]. In this study, we implemented a triple therapy regimen for liver cancer, utilizing H-FIRE in conjunction with BMS-1 and R848. The results revealed that this triple therapy not only enhanced the local effects of H-FIRE but also conferred significant therapeutic benefits against secondary tumors. Activation of the innate immune system via the TLR pathway facilitates more efficient processing of neoantigens released during H-FIRE. Additionally, checkpoint inhibition amplifies the neoantigen-specific adaptive immune response. This combinatorial strategy effectively suppresses primary tumor growth while exerting a significant inhibitory effect on secondary tumors, thereby achieving an abscopal effect.
Studies have indicated that following ICB treatment, the primary site of expansion for tumor-reactive CD8+ T cells is the splenic white pulp, where these cells differentiate into tumor-infiltrating clonotypes[52]. Upon expansion, tumor-reactive CD8+ T cells migrate from the spleen to the tumor site to exert their anti-tumor effects. Furthermore, alongside the increase in CD8+ T cell numbers induced by ICB treatment, studies have reported a reduction in the diversity of the T cell receptor repertoire[52]. This suggests that only a small subset of tumor-reactive CD8+ T cells, primarily derived from precursor T cells, undergo expansion. The critical phenotypic transformation required for this expansion occurs in the spleen, catalyzed by limited antigen cross-presentation by DC[52,53]. In our study, we investigated splenic immune cells and identified notable changes in immune cell subsets associated with both the spleen and tumors following H-FIRE treatment. Specifically, the H-FIRE combined immunotherapy group exhibited a significant increase in the percentage of CD3+ CD8+ T cells. Once activated, these antigen-specific T cells are capable of eliminating residual primary tumor cells and targeting secondary tumors. TUNEL assays further indicated a marked increase in apoptosis among tumor cells within these secondary tumors.