Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.116088
Revised: December 30, 2025
Accepted: February 2, 2026
Published online: April 14, 2026
Processing time: 152 Days and 16.7 Hours
Discriminating between gastrointestinal tuberculosis (GITB) and Crohn’s disease is a perplexing challenge for clinicians. The clinical presentation, endoscopic ap
To study the diagnostic accuracy of Xpert MTB/RIF Ultra (Xpert Ultra) and True
We prospectively included patients suspected to have GITB who underwent co
Of the 38 patients with complete follow-up, 26 had GITB, and 12 served as controls (n = 10 Crohn’s disease cases, n = 2 alternate diagnosis). GITB cases included three acid-fast bacilli culture-positive patients, four with granulomas, 17 with Xpert Ultra positivity and 15 with TrueNat plus positivity. Compared with the CRS, the overall sensitivity and specificity of Xpert Ultra for the diagnosis of GITB was 65.38% [95% confidence interval (CI): 44.33-82.79] and 83.33% (95%CI: 51.59-97.91), respectively. TrueNat plus had a sensitivity of 57.69% (95%CI: 36.92-76.65) and specificity of 50% (95%CI: 21.09-78.91).
TrueNat plus has a lower sensitivity and specificity compared with Xpert Ultra. However, the improved sensitivity of these tests compared with standard tests comes at the cost of reduced specificity.
Core Tip: This study compared Xpert Ultra and TrueNat plus assays for diagnosing gastrointestinal tuberculosis against a composite reference standard. Of 38 patients, 26 had gastrointestinal tuberculosis. Xpert Ultra showed higher sensitivity and specificity than TrueNat plus in intestinal tissue samples. The improved sensitivity of these tests compared with standard tests comes at the cost of reduced specificity.
- Citation: Sharma K, Jindal N, Sharma M, Mandavdhare HS, Gupta P, Sekar A, Dutta U, Sharma V. Xpert Ultra and TrueNat MTB plus assays for the diagnosis of gastrointestinal tuberculosis: A diagnostic accuracy study. World J Gastroenterol 2026; 32(14): 116088
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/116088.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.116088
Tuberculosis (TB) is the foremost cause of death from a single infectious agent and the second leading infectious disease[1]. Gastrointestinal TB (GITB) is difficult to diagnose primarily because it is a paucibacillary condition. Signs and symp
The diagnosis of GITB is usually based on microbiological positivity, using the acid-fast bacilli stain or mycobacterial culture. However, these tests lack sensitivity and are positive only in a subset of patients[2,4,5]. The use of histological analysis may be helpful if granulomatous inflammation, with or without caseation, is demonstrated[6]. However, because of the low yield of these tests, clinicians often resort to a diagnostic trial of anti-tuberculous therapy. This may result in delayed diagnosis of other conditions, including Crohn’s disease (CD). Therefore, there is a need for more sensitive diagnostic tests in the clinical armamentarium.
The World Health Organization has endorsed various nucleic acid amplification techniques [Xpert MTB/RIF (Xpert), Xpert Ultra, LAMP and TrueNat] for the diagnosis of pulmonary TB, but the data are limited for GITB[6]. Some of these recent point-of-care (POC) tests attempt to improve the diagnostic sensitivity by using multiple primers targeted against M. tuberculosis[7]. Here, we report about the use of Xpert MTB/RIF Ultra (Xpert Ultra) and TrueNat MTB Plus (TrueNat plus) assay in colonoscopic biopsies from patients with suspected GITB and their performance compared with a composite reference standard (CRS).
This study was a prospective diagnostic accuracy study conducted at the Departments of Gastroenterology and Micro
Consecutive patients suspected to have a possibility of GITB based on clinical, endoscopic, and radiological findings were enrolled in the study. All participants underwent routine clinical investigations, including imaging, colonoscopy, histopathology, and liquid mycobacterial culture (mycobacteria growth indicator tube). Intestinal tissue samples were obtained from patients with suspected intestinal TB using colonoscopic biopsy. As previously outlined, patients sus
Definite GITB: Patients with evidence of intestinal ulcers and microbiological positivity (acid-fast bacilli/M. tuberculosis culture positivity) from intestinal tissue.
Probable GITB: Patients with intestinal involvement consistent with GITB, histological findings (granuloma, caseating or not; chronic inflammation) consistent with GITB, and no evidence of alternative diagnosis. These patients were started on standard four-drug ATT and underwent a repeat colonoscopy after 2 months to look for early mucosal response, such as healing of ulcers[8]. The diagnosis of GITB was established if ulcer healing was observed.
Gold standard for GITB: Patients were diagnosed based on the CRS, which included microbiological positivity or an objective endoscopic response to ATT, i.e. definite or probable TB (see above). To allow unbiased assessment of Xpert Ultra and TrueNat, these assays were excluded from the CRS for GITB.
Diseased controls: Patients with intestinal lesions with alternate diagnosis, usually CD. The diagnosis was based on evidence of an alternative diagnosis as confirmed on histology and ancillary tests, and lack of endoscopic response to ATT and improvement with therapies directed at the underlying disease. The diagnosis of CD was established using standard European Crohn’s and Colitis Organization criteria, except in cases where diagnostic uncertainty with GITB led to the initial initiation of ATT[8]. Here, the diagnosis of CD was established by a lack of mucosal response followed by improvement with therapy directed at CD.
The patients underwent colonoscopy under sedation after a split-dose regimen using a total of 4 L of polyethylene glycol-based solution for bowel preparation. An attempt was made to reach the ileum in all cases, unless the presence of a narrowing/stricture did not allow the passage of the colonoscope. The biopsies were preferably obtained from ulcers, especially ulcer bases, and four pieces each were sent for histology, mycobacteria growth indicator tube, Xpert Ultra and TrueNat plus. For Xpert Ultra and TrueNat plus assays, tissue biopsies were homogenized and decontaminated by the N-acetyl-L-cysteine-NaOH method. Approximately 1 mL of homogenized lysed sample was used for Xpert ultra and 0.5 mL for the TrueNat plus assay.
Xpert MTB/RIF Ultra is a cartridge-based nucleic acid amplification test (nucleic acid amplification technique) that de
TrueNat MTB-PLUS is a chip-based molecular test that also detects M. tuberculosis (targets nrdZ and IS6110). Briefly, DNA was extracted using Trueprep extraction protocol of TrueNat plus (Molbio Diagnostics, Verna, India), and 6 μL of DNA was used for the TrueNat assay.
The data were entered into Microsoft Excel. Comparisons were made between the GITB and alternative diagnosis groups for clinical and endoscopic findings. Categorical variables were compared using the χ2 test, and continuous variables were compared using the independent sample T test or Mann-Whitney U test, depending on the normality of distribution. Comparison with the CRS for diagnosis was performed separately. The diagnostic accuracy (sensitivity, speci
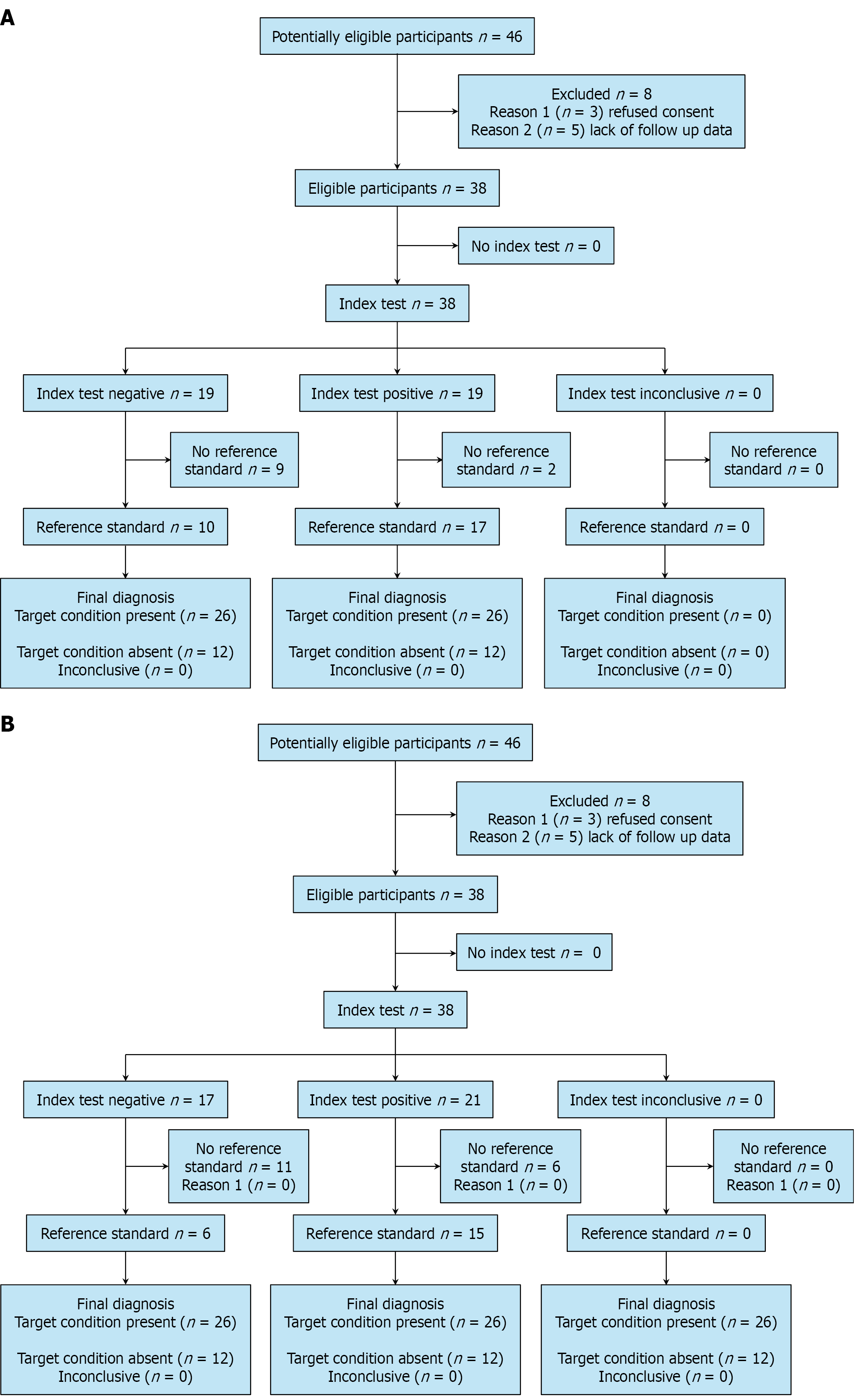
Of 46 eligible participants, three refused consent, and complete follow-up of five patients was not available. Therefore, 38 participants were included in the final analysis (Figure 1). Participants were aged 40 ± 16 years. Among the included participants, 21 (55%) were males and 17 (45%) were females (Table 1). Ultimately, 26 (57.8%) patients had a diagnosis of GITB, while 12 (31.5%) had an alternative diagnosis and were considered to be controls (n = 10 with CD, n = 2 with amoebic colitis). The diagnosis of GITB was based on microbiological positivity in 3 patients (11.5%) and objective response to ATT in 23 patients (88.5%).
| Clinical features | GITB (n = 26) | Controls (n = 12) | P value |
| Age, mean ± SD | 42.2 ± 17.8 | 36.4 ± 9.14 | 0.30 |
| Male gender | 13 | 8 | 0.48 |
| Abdominal pain | 20 (76.9) | 10 (83.3) | 1.00 |
| Diarrhea | 6 (23.1) | 6 (50) | 0.14 |
| Albumin (mg/dL) | 3.73 (3.4-4.13) | 3.94 (3.59-4.41) | 0.43 |
| Fever | 13 (50) | 3 (25) | 0.18 |
| Loss of appetite | 15 (57.7) | 7 (58.3) | 1.00 |
| Loss of weight | 21 (80.7) | 7 (58.3) | 0.23 |
| History of TB | 1 (4) | 2 (16.6) | 0.22 |
| Family history of TB | 7 (27) | 2 (16.6) | 0.68 |
We compared the clinical features between both groups. The most common symptoms in patients with GITB were abdominal pain (n = 20; 76.9%), loss of appetite (n = 15; 57.7%), weight loss (n = 21; 80.7%), diarrhea (n = 6; 23.1%), fever (n = 13; 50%). The most common symptoms in controls were abdominal pain (n = 10; 83.3%), loss of appetite (n = 7; 58.3%), weight loss (n = 7; 58.3%), diarrhea (n = 6; 50%), fever (n = 3; 25%) (Table 1). Amongst patients with GITB, one (4%) patient had a history of TB, and 7 (27%) had a family history of TB. GITB cases included three mycobacterial culture-positive patients and four with granulomas. In other patients with GITB, diagnosis was established based on mucosal healing in response to ATT (Table 2).
| GITB (n = 26) | Controls (n = 12) | ||
| Microbiology | Acid-fast bacilli smear | 1 | 0 |
| 3 | 0 | ||
| Histopathology-granuloma | 4 | 1-granuloma; 2-amoebiasis | |
| Response to antitubercular therapy | 23 | 4 (no response) and response to Crohn’s disease therapy; 6 (improvement with Crohn’s disease therapy) | |
In the 26 patients with GITB, 17 tested positive by Xpert Ultra and 15 tested positive by TrueNat Plus. In the control group, the (false) positivity for Xpert Ultra and TrueNat Plus was 2 and 6, respectively. Based on the CRS, the overall sensitivity and specificity of Xpert Ultra for GITB diagnosis was 65.38% (95%CI: 44.33-82.79) and 83.33% (95%CI: 51.59-97.91), respectively. TrueNat had a sensitivity of 57.69% (95%CI: 36.92-76.65) and a specificity of 50% (95%CI: 21.09-78.91). The sensitivity and specificity of both assays in definite GITB, probable GITB and definite + probable GITB are shown in Table 3. The performance of both tests using McNemar’s tests for the diseased group (GITB) had a two-tailed P value of 0.7518.
| Sensitivity | Specificity | PPV | NPV | Accuracy | ||
| Xpert Ultra | Definite GITB | 66.67 (9.4-99.2) | 83.33 (51.6-97.9) | 50 (18.3-81.7) | 90.91 (66.4-98) | 80 (51.9-95.7) |
| Probable GITB | 65.22 (42.7-83.6) | 88.24 (67.2-96.5) | 55.56 (40.4-69.8) | 71.43 (53.7-85.4) | ||
| Overall | 65.38 (44.3-82.8) | 89.47 (69.9-96.9) | 52.63 (38.2-66.6) | 71.05 (54.1-84.6) | ||
| TrueNat MTB Plus | Definite GITB | 66.67 (9.4-99.2) | 50 (21.1-78.9) | 25 (11.1-47) | 85.71 (52.4-97) | 53.33 (26.6-78.7) |
| Probable GITB | 56.52 (34.5-76.8) | 68.42 (52.6-80.9) | 37.5 (22.4-55.5) | 54.29 (36.7-71.2) | ||
| Overall | 57.69 (36.9-76.7) | 71.43 (56.5-82.8) | 35.29 (20.9-52.9) | 55.26 (38.3-71.4) |
In this study evaluating the role of Xpert Ultra in the diagnosis of GITB, we found that Xpert Ultra has a high sensitivity of 65.38% (95%CI: 44.33-82.79) with a specificity of 83.33% (95%CI: 51.59-97.91) for the diagnosis of GITB. This sensitivity compares favorably to the performance of the standard GeneXpert (Xpert MTB/RIF) in this setting. However, the im
The Xpert platform enables POC molecular diagnosis of TB. The standard GeneXpert test is based on detection of a single primer/target, the rpoB gene[10]. The Xpert Ultra seeks to improve the molecular diagnostic performance by utilizing three primers against rpoB and two multicopy amplification targets (IS6110 and IS1081)[11]. Indeed, the use of Xpert Ultra in pulmonary and some extrapulmonary TB has shown improved sensitivity[2,12]. In a study on central nervous system TB, the use of Xpert Ultra was associated with improved sensitivity for the diagnosis using CSF samples[7]. Therefore, we hypothesized that Xpert Ultra would help in improving the sensitivity. Previous studies with Xpert MTB/RIF suggest a modest performance with a pooled sensitivity as low as 23% but an excellent specificity[2]. Indeed, the current study suggests that the diagnostic sensitivity improves with the use of Xpert Ultra. However, this comes at a cost of decreased specificity, meaning that there were some false positives.
In the present study, we detected two false positives for Xpert Ultra; one had underlying amoebic colitis, while one had CD. The reasons for this false positivity are unclear, but this may be due to the nature of the sample (biopsy from the colon, which is exposed to many environmental antigens, including microorganisms), latent TB infection or the non-specific primers. Additional confirmatory tests such as sequencing should be performed in future studies. Recently, Ye et al[13] reported tissue next-generation sequencing for intestinal TB diagnosis and discrimination from CD with latent TB infection. The sensitivity and specificity of tissue next-generation sequencing for intestinal TB were 83% (72%-91%) and 100% (87%-100%)[13]. However, these exciting results need validation in other cohorts.
There is a dire need for better diagnosis of GITB because of the paucibacillary nature of the disease and low sensitivity of the currently available diagnostic tests. Underdiagnosis of GITB results in a delayed treatment and may result in sequelae and complicated phenotypes. In cases where GITB is misdiagnosed as CD, treatment with immunosuppressants can lead to dissemination of TB[14].
GeneXpert Ultra has a turnaround time of roughly 90 minutes, is more costly per test due to proprietary cartridges, and requires yearly calibration and consistent supply of electricity. Truenat Plus is usually less expensive, uses locally made consumables, and yields results in 60 minutes to 90 minutes. Truenat Plus, which is intended for decentralized use, is more useful in resource-constrained, endemic circumstances because it can operate on battery power and withstand higher temperature swings, whereas GeneXpert Ultra is better suited to well-equipped laboratories. The present study has multiple strengths: This is the first study to report the use of Xpert Ultra and TrueNat plus in GITB. Furthermore, this study reflects real-life clinical practice, with patients tested at presentation even as the clinical picture and diagnosis evolved during follow-up. The study ensures accurate diagnosis of the underlying condition by objectively documenting therapeutic response through colonoscopic evidence of ulcer healing. However, there are certain limitations of this work, including the fact that this was a single-center study and that the control group was heterogeneous. The major limitation of this study is the small sample size and lack of a priori sample size calculation. This was partly because this was a first study and there were no prior estimates of sensitivity of Xpert Ultra in the setting of GITB.
Xpert Ultra, a POC diagnostic test, performed better than TrueNat plus and has improved sensitivity over Xpert MTB/RIF, albeit at the cost of reduced specificity.
| 1. | World Health Organization. Global tuberculosis report 2024. Oct 29, 2024. [cited 11 Oct 2025]. Available from: https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2024. |
| 2. | Jha DK, Pathiyil MM, Sharma V. Evidence-based approach to diagnosis and management of abdominal tuberculosis. Indian J Gastroenterol. 2023;42:17-31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 3. | Mehta V, Desai D, Abraham P, Gupta T, Rodrigues C, Joshi A, Deshpande R, Sawant P, Ingle M, Rathi P, Mandot A. Do additional colonoscopic biopsies increase the yield of Mycobacterium tuberculosis culture in suspected ileo-colonic tuberculosis? Indian J Gastroenterol. 2018;37:226-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 4. | Kohli M, Schiller I, Dendukuri N, Yao M, Dheda K, Denkinger CM, Schumacher SG, Steingart KR. Xpert MTB/RIF Ultra and Xpert MTB/RIF assays for extrapulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2021;1:CD012768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 5. | Du J, Ma YY, Xiang H, Li YM. Confluent granulomas and ulcers lined by epithelioid histiocytes: new ideal method for differentiation of ITB and CD? A meta analysis. PLoS One. 2014;9:e103303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 6. | Shrivas A, Singh S. Tuberculosis Diagnosis and Management: Recent Advances. J Glob Infect Dis. 2025;17:3-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 7. | Donovan J, Thu DDA, Phu NH, Dung VTM, Quang TP, Nghia HDT, Oanh PKN, Nhu TB, Chau NVV, Ha VTN, Hang VTT, Trinh DHK, Geskus RB, Tan LV, Thuong NTT, Thwaites GE. Xpert MTB/RIF Ultra versus Xpert MTB/RIF for the diagnosis of tuberculous meningitis: a prospective, randomised, diagnostic accuracy study. Lancet Infect Dis. 2020;20:299-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 107] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 8. | El-Hussuna A, Hauer AC, Karakan T, Pittet V, Yanai H, Devi J, Yamamoto-Furusho JK, Sima AR, Desalegn H, Sultan MI, Sharma V, Shehab H, Mrabti L, Queiroz N, Jena A, Darma A, Davidson K, Avellaneda N, Elhadi M, Roslani A, Wickramasinghe D, Cajucom CA, Sebastian S. ECCO consensus on management of inflammatory bowel disease in low- and middle-income countries. J Crohns Colitis. 2026;20:jjaf125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 9. | Xpert® MTB-RIF Ultra. [cited 11 Oct 2025]. Available from: https://infomine.cepheid.com/sites/default/files/2023-04/Xpert%20MTB-RIF%20Ultra%20ENGLISH%20Package%20Insert%20for%20Indian%20Market%20302-2766%2C%20Rev.%20D.pdf. |
| 10. | Lawn SD, Nicol MP. Xpert® MTB/RIF assay: development, evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance. Future Microbiol. 2011;6:1067-1082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 381] [Cited by in RCA: 322] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 11. | World Health Organization. WHO consolidated guidelines on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 3rd ed. Mar 20, 2024. cited 11 Oct 2025]. Available from: https://www.who.int/publications/i/item/9789240089488. |
| 12. | Chakravorty S, Simmons AM, Rowneki M, Parmar H, Cao Y, Ryan J, Banada PP, Deshpande S, Shenai S, Gall A, Glass J, Krieswirth B, Schumacher SG, Nabeta P, Tukvadze N, Rodrigues C, Skrahina A, Tagliani E, Cirillo DM, Davidow A, Denkinger CM, Persing D, Kwiatkowski R, Jones M, Alland D. The New Xpert MTB/RIF Ultra: Improving Detection of Mycobacterium tuberculosis and Resistance to Rifampin in an Assay Suitable for Point-of-Care Testing. mBio. 2017;8:e00812-e00817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 519] [Article Influence: 57.7] [Reference Citation Analysis (0)] |
| 13. | Ye L, Cao Y, Fu Y, Tian C, Cao Q. Crohn's Disease With Latent Tuberculosis Infection or Intestinal Tuberculosis: Rapid Discrimination by Targeted Next-Generation Sequencing. Aliment Pharmacol Ther. 2025;61:1218-1225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 14. | Kedia S, Das P, Madhusudhan KS, Dattagupta S, Sharma R, Sahni P, Makharia G, Ahuja V. Differentiating Crohn's disease from intestinal tuberculosis. World J Gastroenterol. 2019;25:418-432. [PubMed] [DOI] [Full Text] |