Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.114657
Revised: December 4, 2025
Accepted: February 13, 2026
Published online: April 7, 2026
Processing time: 183 Days and 15.9 Hours
Cardiovascular and liver metabolic diseases are interconnected conditions, asso
To compare liver and cardiovascular outcomes between lean and non-lean indi
We conducted a systematic review and meta-analysis of studies published in the past 20 years comparing seven outcomes between lean and non-lean with dysfunction-associated steatotic liver disease individuals. A random-effects model was applied to calculate pooled odds ratios (pORs) with 95% con
A total of 31 studies comprising 10735550 participants were included. Non-lean individuals with MASLD had higher odds of liver fibrosis (pOR = 2.0, 95%CI: 1.0, 3.9, P = 0.04) and hepatic steatosis (pOR = 2.1, 95%CI: 1.5, 2.9, P < 0.001) compared to lean individuals with MASLD. Lean individuals with MASLD had lower odds of cirrhosis (pOR = 0.7, 95%CI: 0.5, 0.9, P = 0.007) and hypertension (pOR = 0.7, 95%CI: 0.6, 0.9, P = 0.002) compared to non-lean individuals with MASLD. There was no significant statistical difference in mortality between lean and non-lean MASLD groups (pOR = 1.4, 95%CI: 1.0, 2.0, P = 0.06). Also, no statistically significant differences were found for metabolic dysfunction-associated steatohepatitis (pOR = 1.2, 95%CI: 0.7, 2.0, P = 0.5) and cardiovascular di
In our study, lean individuals with MASLD have the similar odds of metabolic dysfunction-associated steatohepatitis, cardiovascular diseases and mortality compared to non-lean MASLD patients. Therefore, there is a need to expand MASLD screening and risk stratification strategies beyond obesity-based metrics alone.
Core Tip: Metabolic dysfunction-associated steatotic liver disease affects both lean and non-lean individuals. This meta-analysis of over 10 million participants found that non-lean patients had greater odds of hepatic steatosis, fibrosis, cirrhosis, and hypertension. Lean patients, however, showed similar risks of nonalcoholic steatohepatitis, cardiovascular disease, and mortality. These results support expanding metabolic dysfunction-associated steatotic liver disease screening and risk assessment beyond obesity-related criteria.
- Citation: Mapouka M, Pabingui E, Tazinkeng NN, Gurmessa M, Vickos U, Ndemazie NB, Camengo Police SM. Outcomes of liver and cardiovascular metabolic diseases among lean vs non-lean individuals with metabolic dysfunction-associated steatotic liver disease. World J Gastroenterol 2026; 32(13): 114657
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/114657.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.114657
Liver conditions such as steatosis, fibrosis, cirrhosis, and Metabolic dysfunction-associated steatohepatitis (MASH) have been reported in the literature as common complications of patients living with obesity and metabolic dysfunction-associated steatotic liver disease (MASLD)[1]. In addition, obesity and MASLD represent indirect and often under recognized risk factors for a group of cardiovascular metabolic diseases (CMD) such as atherosclerotic cardiovascular disease (ASCVD), heart failure (HF), and hypertension; thus showing that heart conditions are also complications of MASLD (Table 1)[2]. Therefore, MASLD and ASCVD are recognized as interconnected conditions that share common pathophysiological mechanisms and contribute to simultaneous disease progression and outcomes. These conditions are frequently encountered in clinical practice. Whether diagnosed independently or concurrently in a single patient, limited evidence exists regarding their interactions and outcomes[2]. Increasing interest within the clinical community has focused on how these conditions manifest in lean and non-lean individuals with MASLD[3].
| Stages | Definition |
| Stage 0: No CKM risk factors | Individuals with normal BMI and waist circumference, normoglycemia, normotension, a normal lipid profile, and no evidence of CKD or subclinical or clinical CVD |
| Stage 1: Excess or dysfunctional adiposity | Individuals with overweight/obesity, abdominal obesity, or dysfunctional adipose tissue, without the presence of other metabolic risk factors or CKD. BMI ≥ 25 kg/m2 (or ≥ 23 kg/m2 if Asian ancestry), waist circumference ≥ 88/102 cm in women/men (or if Asian ancestry ≥ 80/90 cm in women/men), or fasting blood glucose ≥ 100-124 mg/dL or HbA1c between 5.7% and 6.4% |
| Stage 2: Metabolic risk factors and CKD | Individuals with metabolic risk factors, hypertriglyceridemia (≥ 135 mg/dL), hypertension, MetS, diabetes, or CKD |
| Stage 3: Subclinical CVD in CKM | Subclinical ASCVD or subclinical HF among individuals with excess/dysfunctional adiposity, other metabolic risk factors, or CKD. Subclinical ASCVD to be principally diagnosed by coronary artery calcification (subclinical atherosclerosis by coronary catheterization/computed tomography angiography also meets criteria). Subclinical HF diagnosed by elevated cardiac biomarkers (NT-proBNP ≥ 125 pg/mL, hs-troponin T ≥ 14 ng/L for women and ≥ 22 ng/L for men, hs-troponin I ≥ 10 ng/L for women and ≥ 12 ng/L for men) or by echocardiographic parameters, with a combination of the 2 indicating highest HF risk. Risk equivalents of subclinical CVD. Very high-risk CKD (stage G4 or G5 CKD or very high risk per KDIGO classification). High predicted 10-year CVD risk |
| Stage 4: Clinical CVD in CKM | Clinical CVD (coronary heart disease, HF, stroke, peripheral artery disease, atrial fibrillation) among individuals with excess/dysfunctional adiposity, other CKM risk factors, or CKD. Stage 4a: No kidney failure. Stage 4b: Kidney failure present |
Several sources including the World Health Organization (WHO) define individuals with a body mass index (BMI) exceeding 25 as overweight, while those with a BMI > 30 fall within the obese category[4]. Those with a BMI below 25 are generally considered lean, with the exception of Asian descent[5]. Some evidence suggested that lean patients experience more favorable outcomes, while other studies report no significant difference between lean and non-lean groups[4]. In contrast, certain research has even found higher mortality rates among lean individuals with MASLD. A systematic review and meta-analysis published a year ago evaluated 10 observational cohort studies and found that lean MASLD patients typically had milder liver histology at diagnosis but experienced higher overall mortality over time[6]. While cardiovascular mortality was lower in this group, liver-related mortality was elevated, though the incidence of liver-related complications such as cirrhosis and liver cancer did not appear to increase. This inconsistency between com
Despite recent updates in fatty liver disease nomenclature, limited evidence exists on how liver conditions and CMD could overlap and progress in terms of outcomes among lean and non-lean individuals with MASLD[7]. The purpose of this study was to perform a meta-analysis on clinical outcomes of liver and cardiovascular metabolic conditions among lean and non-lean MASLD. This research’s final takeaway questions the traditional view relying solely on BMI as a predictor of MASLD and opens new avenues for exploring other parameters, such as body fat percentage, when comparing the outcomes of liver and CMD in the context of lean and non-lean MASLD[8].
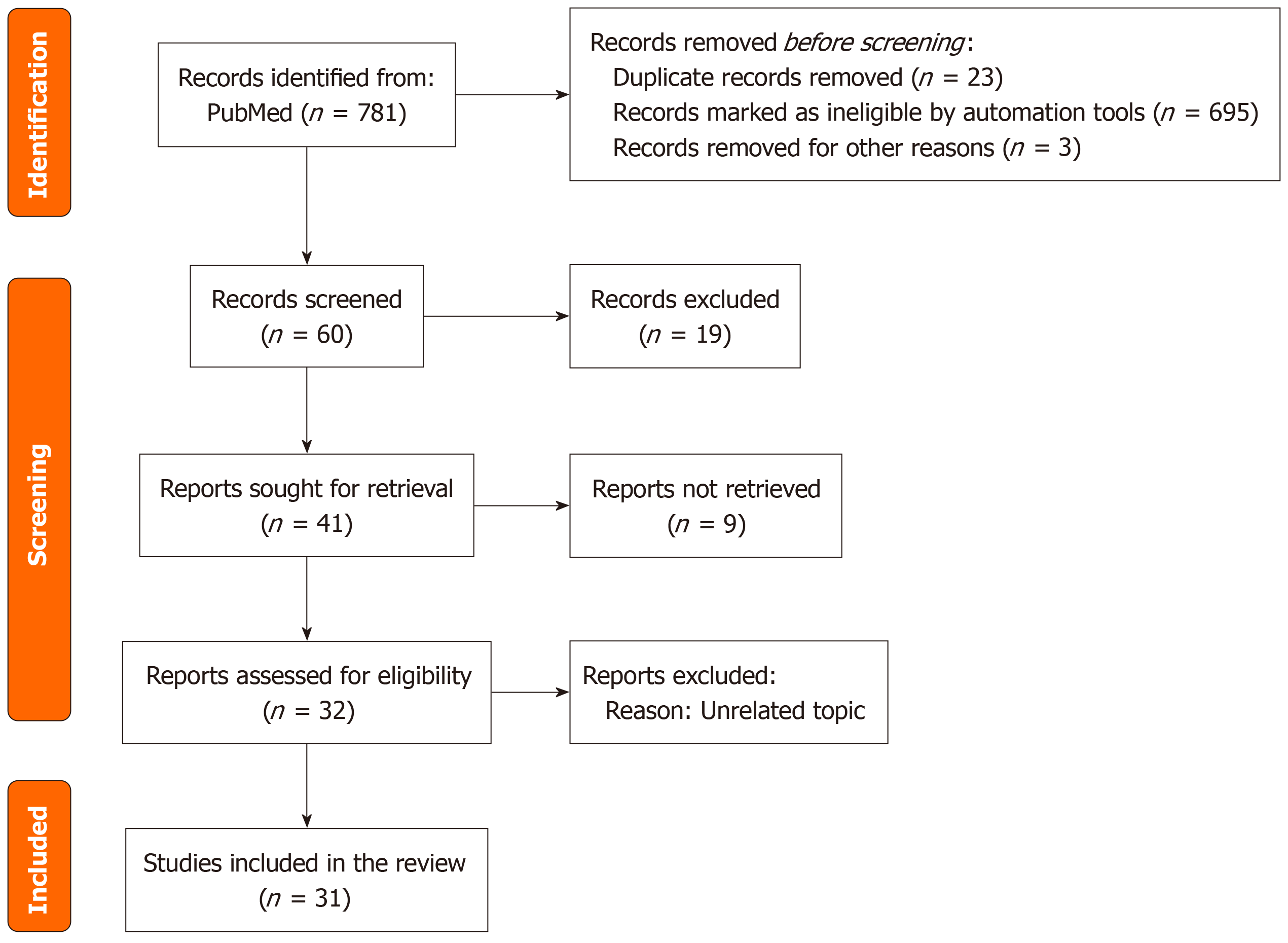
We searched using the PubMed, MEDLINE, Cochrane and EMBASE database to identify articles published over the past 20 years, up to February 2025 (Figure 1). First, we searched independently the terms “cardiometabolic disease”, “liver metabolic disease”, “lean MASLD”, “lean MASH”, including the previous terminologies of non-alcoholic fatty liver disease and nonalcoholic steatohepatitis. Then we searched the combinations of those terms with each of our seven outcomes of interest in the following format: “CMD, liver metabolic disease, lean MASLD, liver fibrosis”, “CMD, liver metabolic disease, lean MASLD, MASH”, “CMD, liver metabolic disease, lean MASLD, steatosis”, “CMD, liver metabolic disease, lean MASLD, cirrhosis”, “CMD, liver metabolic disease, lean MASLD, CVD”, “CMD, liver metabolic disease, lean MASLD, hypertension”, and “CMD, liver metabolic disease, lean MASLD, death”. For the purpose of our study, we adopted the WHO definition of “lean” as individuals with a BMI < 25 kg/m2 for those of non-Asian descent and < 23 kg/m2 for those of Asian descent. “Non-lean” was defined as individuals with any BMI that did not meet the criteria for lean status. inclusion criteria were limited to articles published in English that encompassed meta-analyses, randomized clinical trials, systematic reviews, observational studies, reviews, books, and documents. Preclinical studies were excluded. Two reviewers independently assessed the eligibility of full-text articles following the initial screening of titles and abstracts. Any disagreements related to the search strategy or study selection were resolved through discussion with a third reviewer. Studies that met the inclusion criteria were further evaluated by the third reviewer before being included in the final systematic review and meta-analysis. The ethical approval was not required for this study, as it is a secondary analysis of previously published data.
We evaluated seven outcomes related to liver and CMD in our study. These outcomes included liver fibrosis, steatosis, metabolic dysfunction-associated steatohepatitis (MASH), cirrhosis, cardiovascular disease (CVD), hypertension, and death. CVD was defined broadly and encompassed stroke, acute myocardial infarction, cardiac arrest, transient ischemic attack, cerebral aneurysm, congestive HF, and ischemic heart disease. We did not assess the causes or treatments of these outcomes, but rather focused on their presence among lean and non-lean individuals with MASLD. Cirrhosis was reported regardless of its etiology, whether alcoholic, nonalcoholic, or viral. We ensured that the definition of each outcome was consistent across all eligible studies.
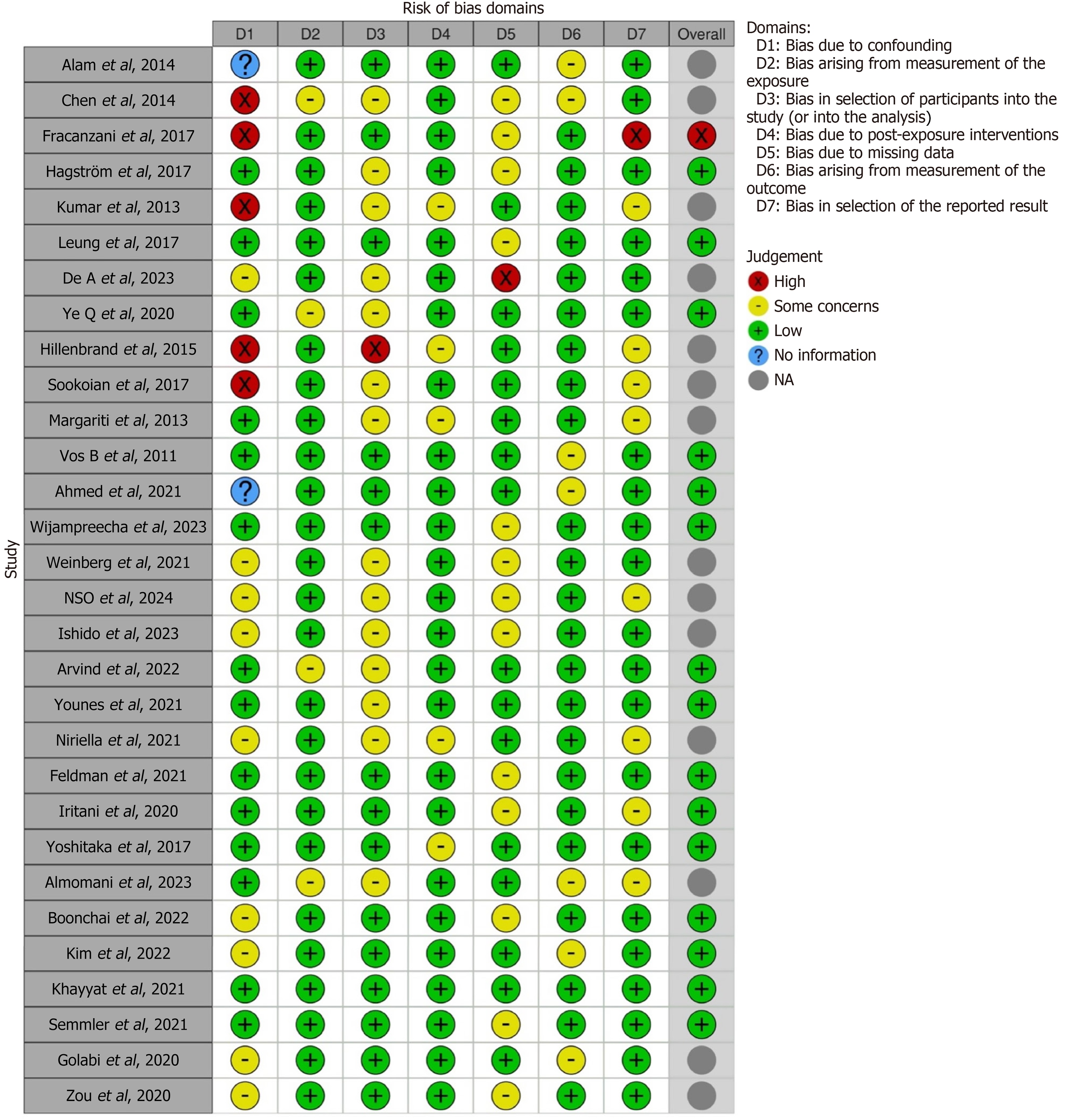
Two authors extracted and entered data from eligible studies into Excel workbooks. The data included mean age, sex, and outcomes of interest along with their respective statistics. After data entry, we used the Comprehensive Meta-Analysis software, version 4.0 (BIOSTAT, Englewood, NJ, United States), to analyze the data, generate tables, and create forest plots comparing outcomes between two groups: Lean and non-lean MASLD. Given the expected variability across international studies, a random-effects model was used to estimate effect sizes, assuming that the effects varied across studies rather than being identical. Specifically, we calculated pooled odds ratios (pORs) for each of the seven outcomes to assess differences between lean and non-lean MASLD groups, with 95% confidence intervals (CIs) and a significance threshold of P < 0.05. Heterogeneity was assessed using both the Cochran Q test and the I² statistic. Our pooled statistics generated the estimates of the variation of effect size across the studies. The Q-statistic provided a test of the null hypothesis that all studies in the analysis shared a common effect size. If all the studies shared the same effect size, the expected value of Q would be equal to the degrees of freedom. A Q test P value below 0.05, combined with an I² value ≥ 50%, was interpreted as indicating moderate heterogeneity. Finally, the publication bias was assessed using the Risk of Bias in Non-randomized Studies-of Exposure tool (Figure 2).
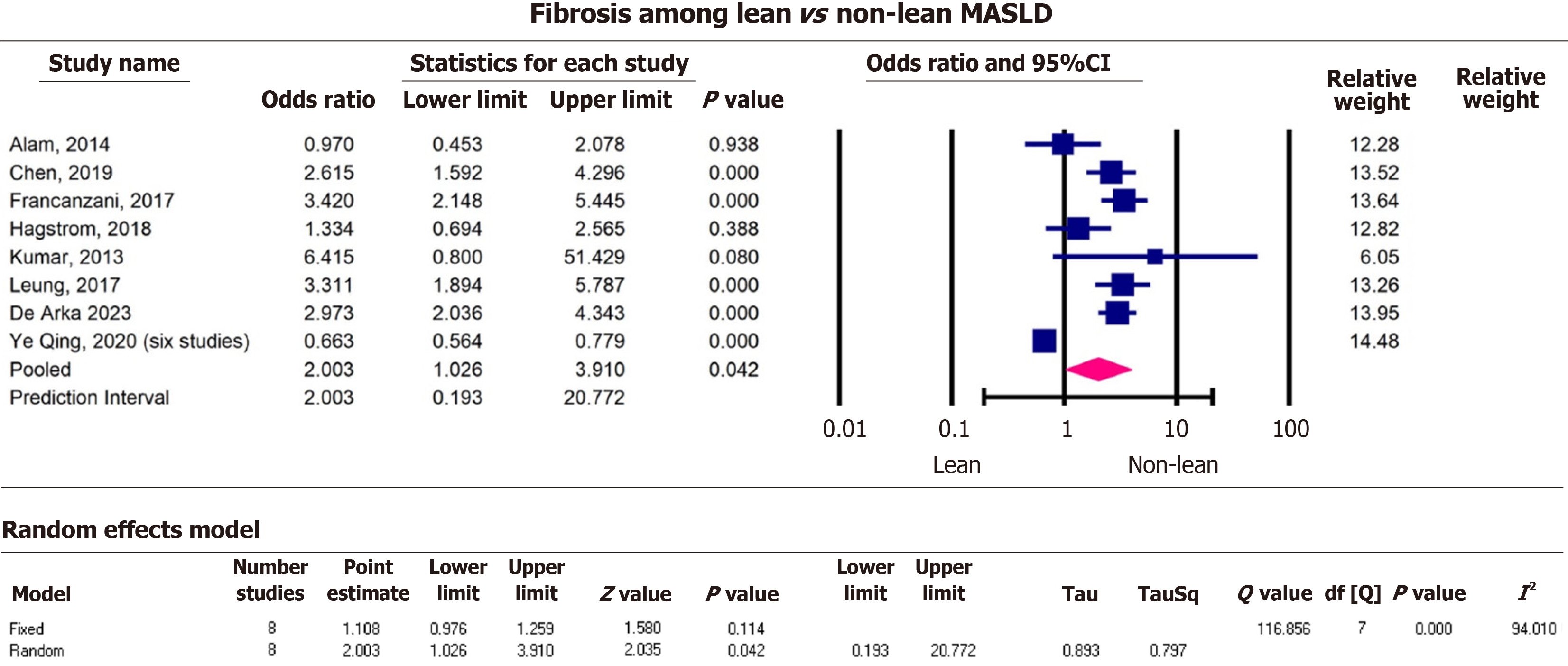
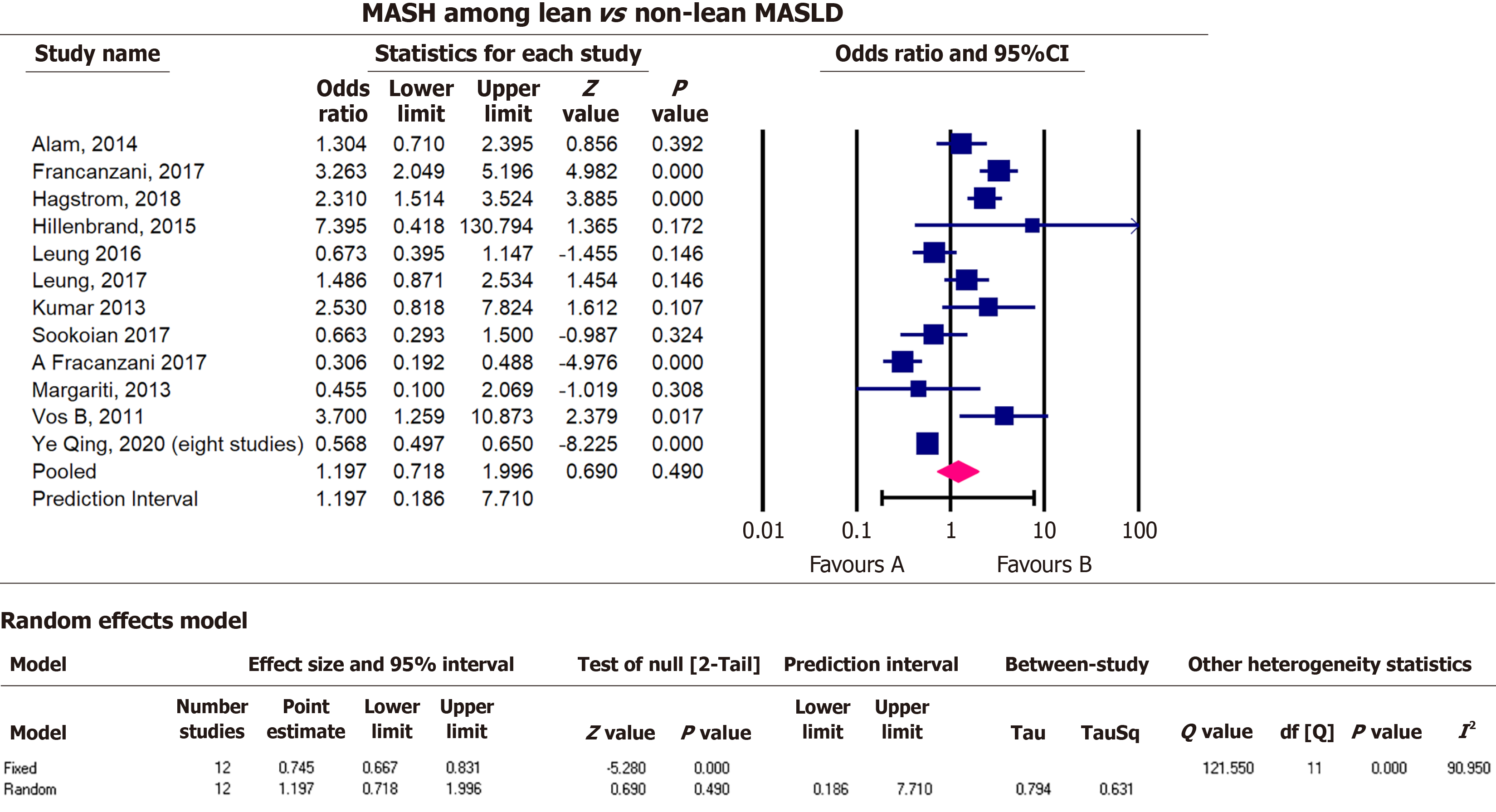
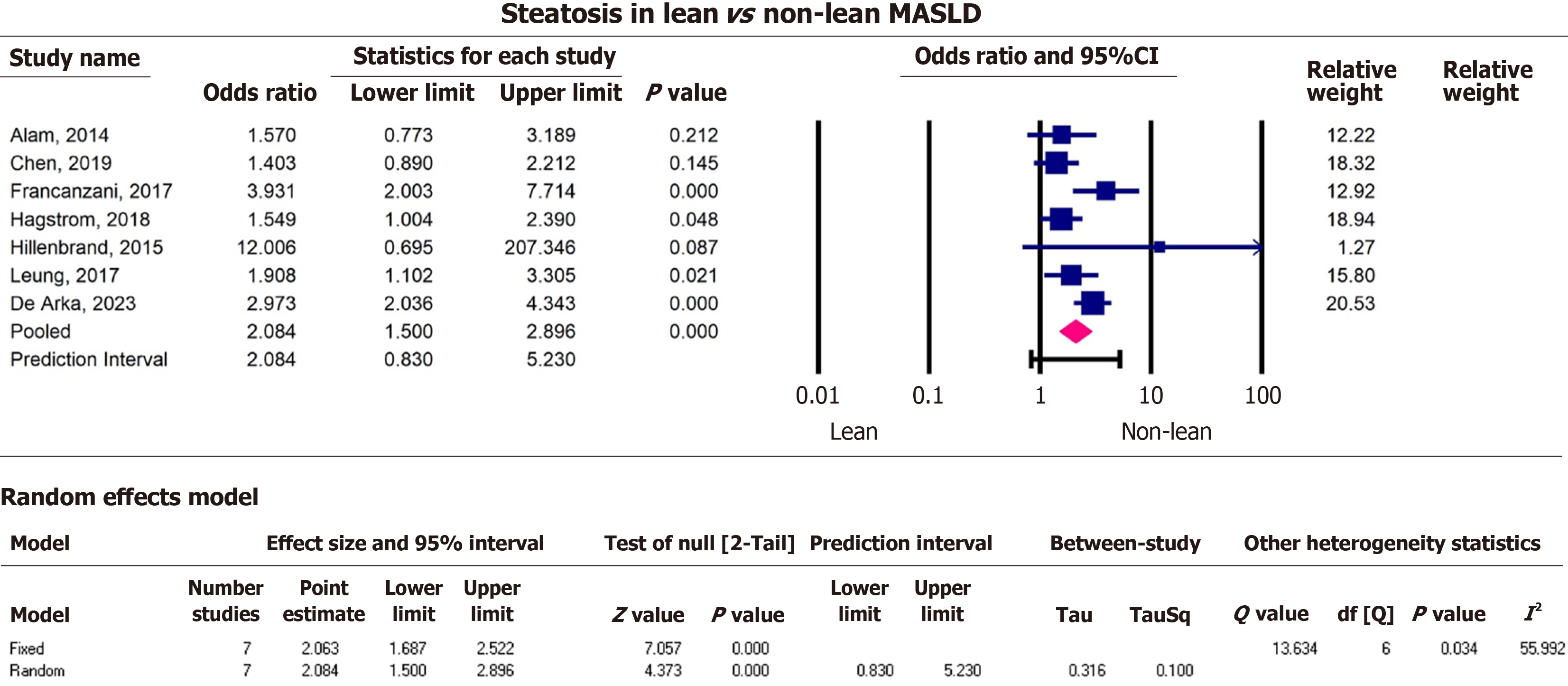
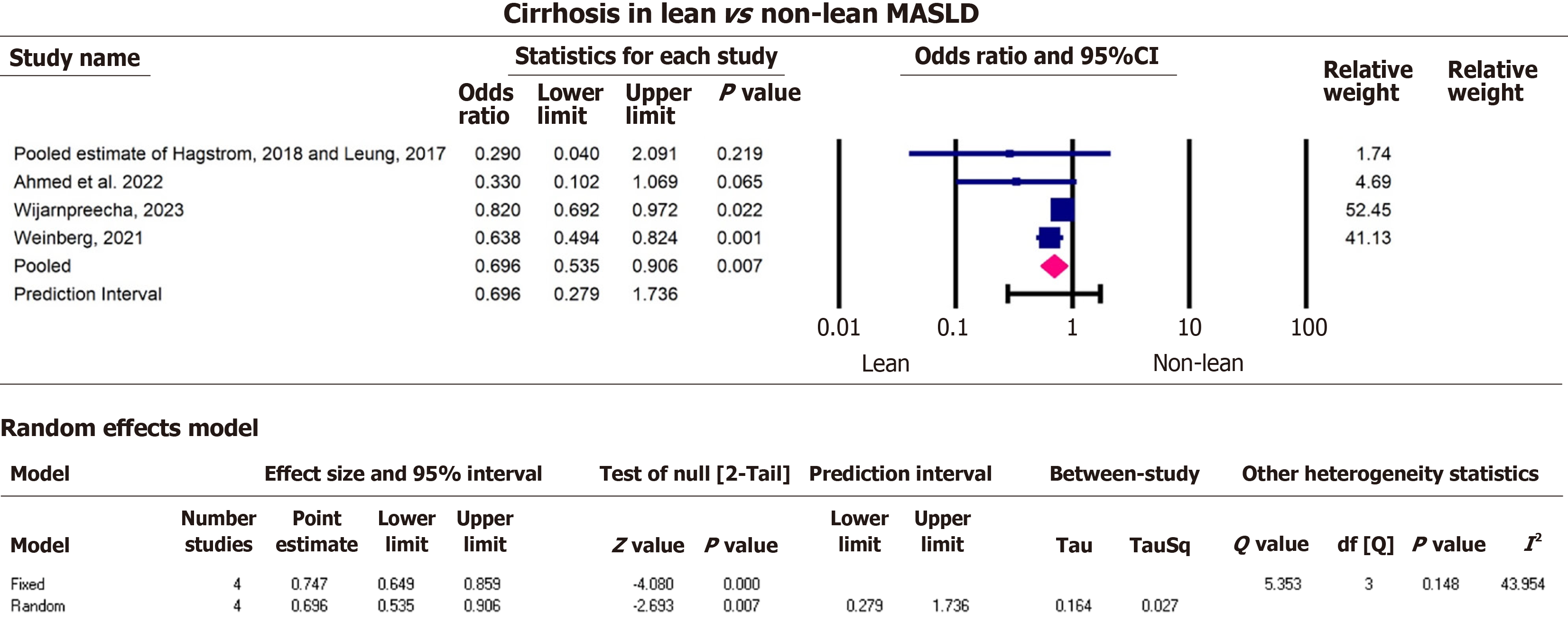
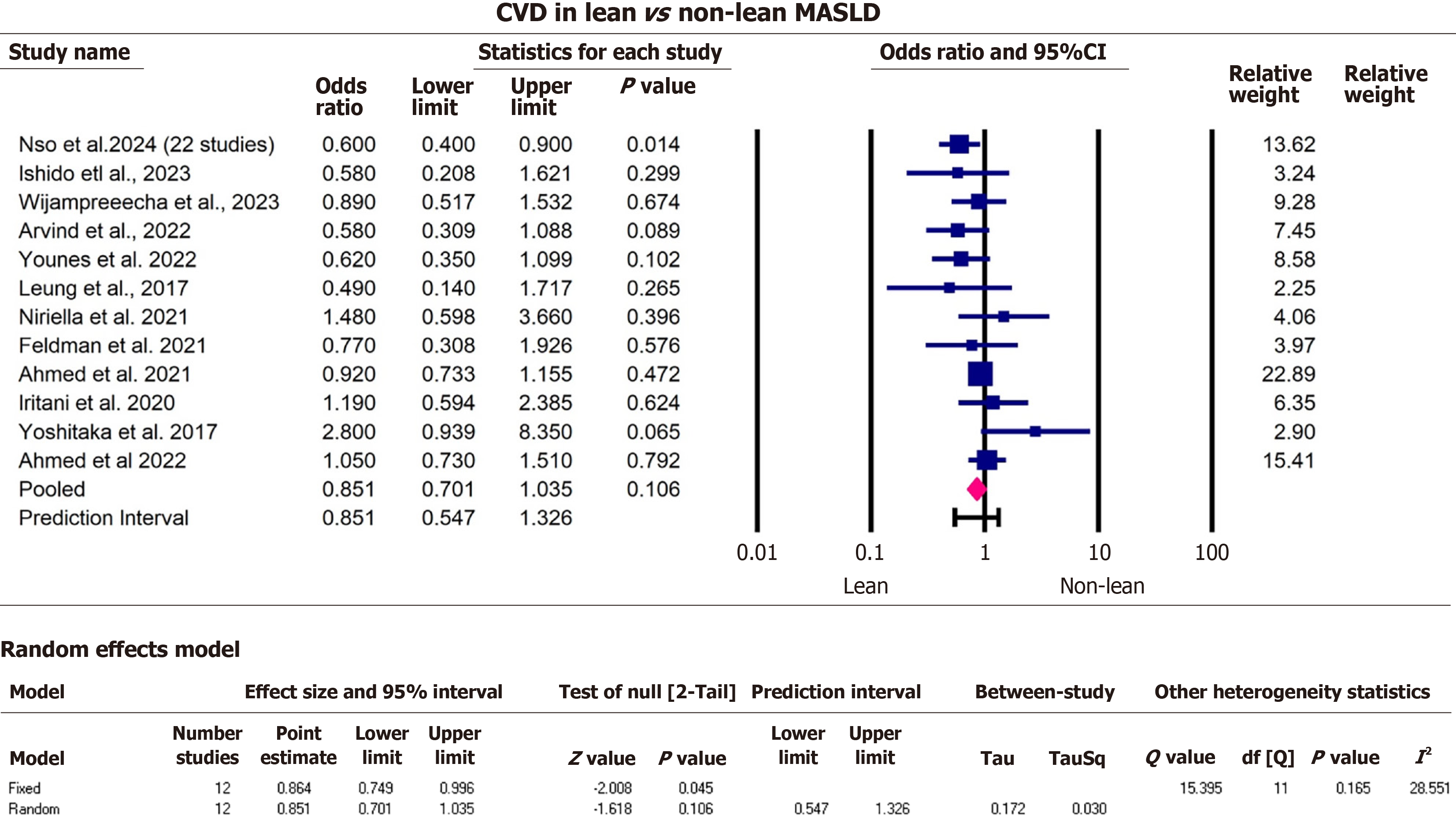
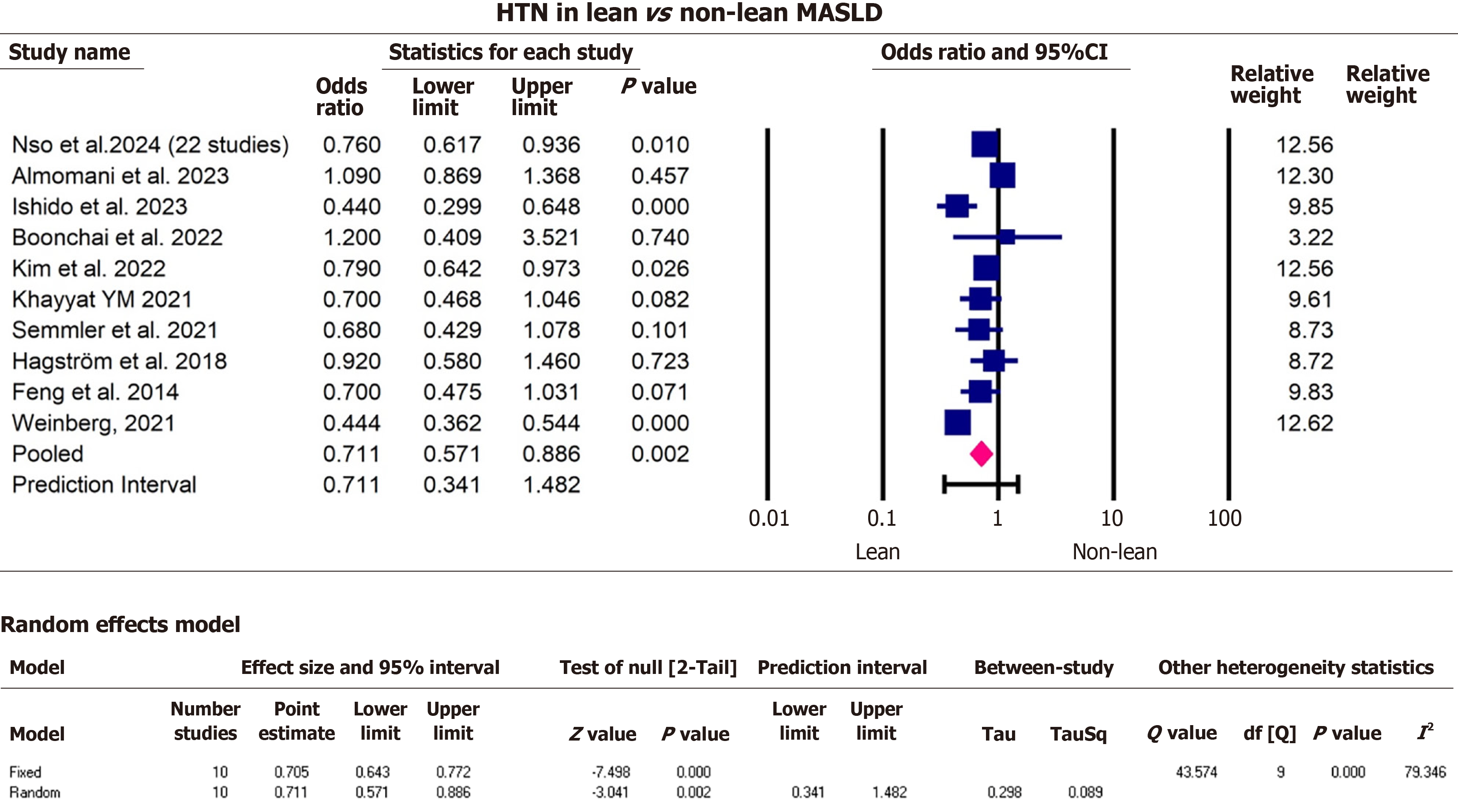
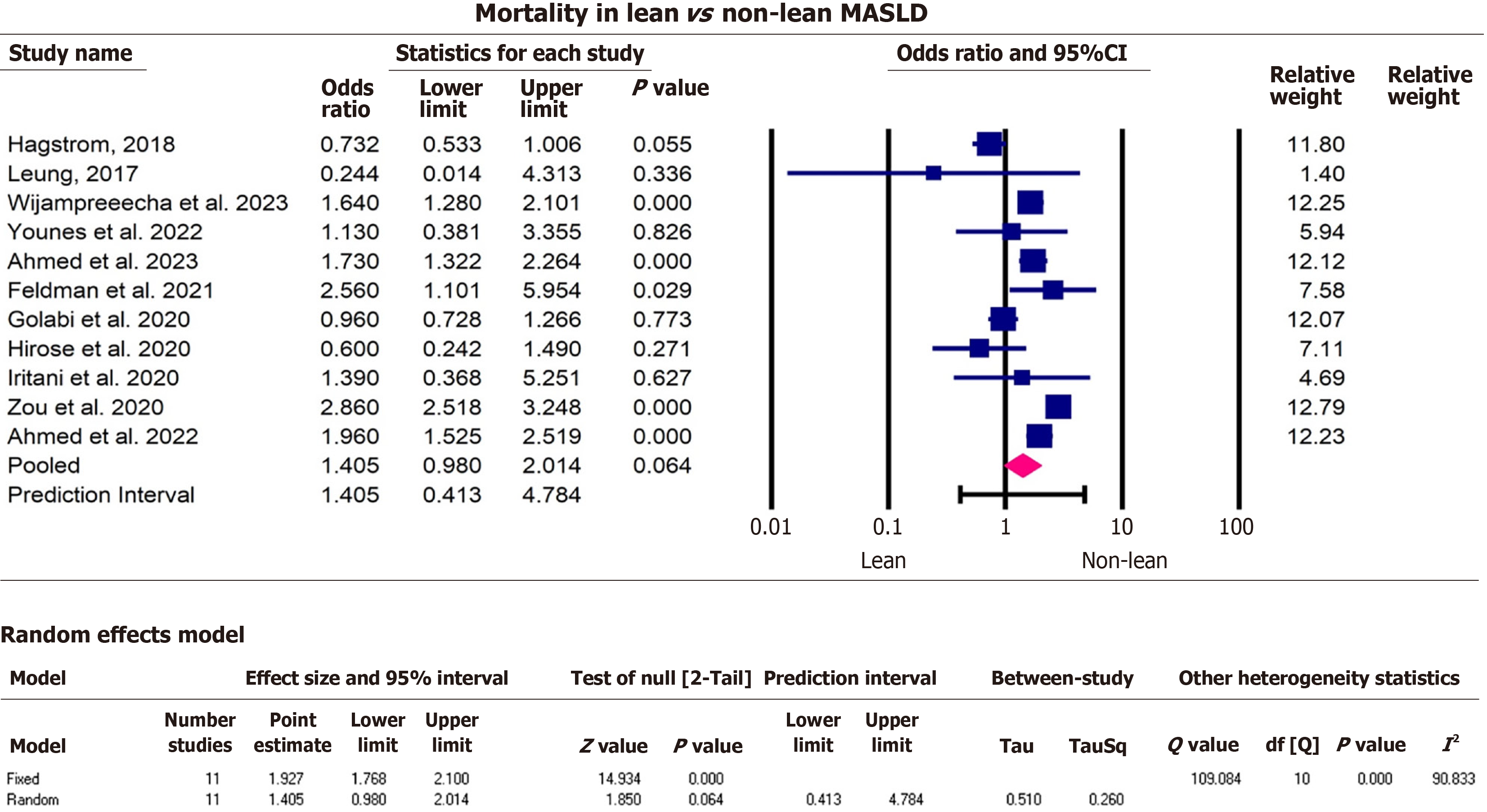
After screening titles and abstracts and removing duplicates, 31 articles met the inclusion criteria. A total of 10735550 participants from all 31 eligible studies were included in our study (Table 2). The average age from studies that consistently reported their data (11 studies) was 52.2 ± 15.0 years. The majority of participants were male 57.4% for 22 studies. Most authors shared the definition of lean from the WHO with a BMI cutoff of < 25 kg/m2, except for six studies that made a distinction for the Asian group, defining lean as a BMI < 23 kg/m2[9-13]. Nine studies were conducted in North America (United States), one in South America (Argentina), eleven in Asia (Bangladesh, Sri Lanka, India, Saudi Arabia, Japan, Thailand, Korea), seven studies in Europe (Italy, Sweden, Greece, Germany, Belgium, Austria), two worldwide and one unspecified study. The seven outcomes of interest were extracted from height studies that reported liver fibrosis[14-21] (Figure 3), twelve reporting MASH[13-15,17-25] (Figure 4), seven reporting hepatic steatosis[9,15-17,19,20,22] (Figure 5), five studies on cirrhosis[17,19,26-28] (Figure 6), twelve studies on CVD[10-13,19,26,27,29-33] (Figure 7), ten studies reporting hypertension[10,12-14,17,28,29,34-36] (Figure 8), and eleven studies on death[17,19,26,27,31-33,37,38] (Figure 9).
| Ref. | Study design | Participants (n) | Age (mean ± SD) | Sex (male/female) | Region (country) | Definition of lean | Outcomes of interest investigated |
| [15] | Cross-sectional | 465 | 40.8 ± 9.9 | 118/283 | Asia (Bangladesh) | Not defined | Steatosis, MASH, and fibrosis |
| [16] | Systematic review and meta-analysis | 54441 | 45.6 | 2320/1592 | Unspecified | BMI < 23 kg/m2 | MASLD occurrence, fibrosis |
| [21] | Retrospective cohort | 669 | Lean (46 ± 13); non-lean (49 ± 12) | Europe (Italy) | BMI < 25 kg/m2 | MASH, steatosis, fibrosis, HTN | |
| [17] | Prospective cohort | 2117 | 48.2 ± 13.7 | 402/244 | Europe (Sweden) | BMI < 25 kg/m2 | MASH, fibrosis, steatosis, HTN, CVD |
| [18] | Case-control | 307 | Asia | BMI < 25 kg/m2 | Fibrosis | ||
| [19] | Prospective cohort | 307 | Asia | Unspecified | Fibrosis, liver failure | ||
| [9] | Retrospective cohort | 1040 | Lean (38.5 ± 12.04); non-lean (41.46 ± 11.14) | 604/436 | Asia (India) | BMI < 23 kg/m2 | MASH, fibrosis, steatosis, HTN |
| [20] | Cross-sectional | 10576383 | 2347669/17345 | Worldwide (24 countries) | BMI < 25 kg/m2 | MASH, fibrosis, steatosis, HTN, CVD | |
| [22] | Cross-sectional | 124 | 69/67 | Europe (Germany) | BMI < 25 kg/m2 | Steatosis, MASH | |
| [23] | Case-control | 390 | Control (47.5 ± 13), MASLD (52 ± 8), MASH (51.7 ± 9) | 141/159 | South America (Argentina) | BMI < 25 kg/m2 | Fibrosis, MASH |
| [24] | Cross-sectional | 162 | All participants 47 ± 14 | 96/66 | Europe (Greece) | BMI < 25 kg/m2 | MASH, fibrosis |
| [25] | Cross-sectional | 1777 | Control (48), lean MASLD (40), obese (49) | 817/960 | Europe (Belgium) | BMI < 25 kg/m2 | HTN, fibrosis, MASH, steatosis |
| [26] | Cross-sectional | 4834 | Lean (51.5 ± 18.0); non-lean [overweight (54.1 ± 15.7), obese (51.0 ± 14.1)] | 2220/2614 | North America (United States) | BMI < 25 kg/m2 | Cirrhosis, death |
| [27] | Retrospective cohort | 18594 | Lean (51.0 ± 36, 63); non-lean [overweight (52.5 ± 41, 63), obese (52.0 ± 40, 61)] | 8767/9827 | North America (United States) | BMI < 25 kg/m2 | Cirrhosis, death, CVD |
| [28] | Cross-sectional | 3386 | 56.9 ± 13 | 1402/1984 | North America (United States) | BMI < 25 kg/m2 | Cirrhosis, CVD |
| [29] | Meta-analysis | 23514 | 53.2 ± 6.8 | 14814/8700 | North America (United States) | BMI < 25 kg/m2 | CVD, death |
| [10] | Retrospective cohort | 581 | Lean (58 ± 12); non-lean (59 ± 11) | 330/251 | Asia (Japan) | BMI < 23 kg/m2 | CVD, HTN |
| [30] | Prospective cohort | 394 | 173/221 | North America (United States) | BMI < 25 kg/m2 | CVD | |
| [31] | Prospective cohort | 1339 | 48 | 2320/1430 | Worldwide | BMI < 25 kg/m2 | Fibrosis, CVD |
| [11] | Prospective cohort | 2985 | 703/1227 (excluded control group) | Asia (Sri Lanka) | BMI < 23 kg/m2 | CVD | |
| [32] | Prospective cohort study | 1312 | MASLD cohort (49.5 ± 13.5), control (52.4 ± 5.8) | 762/550 | Europe (Austria) | BMI ≤ 25 kg/m2 | Steatohepatitis, fibrosis, CVD, death |
| [33] | Retrospective cohort | 446 | 268/178 | Asia (Japan) | BMI < 25 kg/m2 | CVD, HTN | |
| [12] | Retrospective cohort study | 1647 | Non-overweight (50.0 ± 7.9), Overweight (48.8 ± 8.9) | 941/706 | Asia (Japan) | BMI < 23 kg/m2 | CVD, HTN |
| [26] | Cross-sectional | 4834 | Normal weight (51.5 ± 18.0), overweight (54.1 ± 15.7), obese (51.0 ± 14.1) | 2220/2614 | North America (United States) | BMI ≤ 25 kg/m2 | CVD, cirrhosis, death |
| [34] | Cross-sectional | 3410 | Lean (51.5), non-lean (50.0) | 1460/1950 | North America (United States) | BMI ≤ 25 kg/m2 | CVD, HTN |
| [13] | Cross-sectional | 743 | Lean MASLD (75.0 ± 10.7). Non-lean MASLD (66.8 ± 11.9) | 309/434 | Asia (Thailand) | BMI < 23 kg/m2 | Fibrosis, HTN |
| [14] | Cross-sectional | 2144 | 56.1 ± 9.3 | 1359/785 | Asia (Korea) | BMI < 23 kg/m2 | CVD, HTN |
| [35] | Prospective cohort | 1261 | 699/563 | Asia (Saudi Arabia) | BMI ≤ 25 kg/m2 | CVD, HTN | |
| [36] | Cross-sectional | 2238 | Lean without MASLD (56.76 ± 9.9); lean with MASLD (60.36 ± 10.2); overweight MASLD (61.36 ± 9.5); obese MASLD (61.66 ± 9.8) | 1158/1080 | Europe (Austria) | BMI ≤ 25 kg/m2 | CVD, HTN |
| [38] | Cross-sectional | 9341 | Full sample (43.64 ± 0.37) | 4474/4867 | North America (United States) | BMI ≤ 25 kg/m2 | Mortality, CVD |
| [37] | Cross-sectional | 14365 | 6730/7635 | North America (United States) | BMI ≤ 25 kg/m2 | Mortality, CVD |
Before we performed the pooled estimate analysis, most studies reported a higher effect estimate for fibrosis among non-lean MASLD patients, except one with a pooled estimate from 22 studies[20]. The effect sizes for MASH and CVD were distributed approximately equally around the null value. All studies reported higher estimates for steatosis in non-lean patients and lower estimates for cirrhosis among lean MASLD patients. The estimated effects of hypertension were higher among non-lean group. Overall, studies that reported mortality found higher estimates among non-lean MASLD patients.
After performing the pooled estimates of effects size, non-lean MASLD patients had 2.0 times the odds of liver fibrosis (pOR = 2.0, 95%CI: 1.0, 3.9, P = 0.04) among lean MASLD patients with the difference being statistically significant. The Q-value for fibrosis was 116.9 with 11 degrees of freedom and P value < 0.001. We rejected the null hypothesis that the true effect size was identical in all eight studies that reported fibrosis. The I² statistic is 0.94 told us that 94% of variance in observed effects reflected variance in true effects rather than sampling error. The variance of true effect (T2) was 0.8 and the standard deviation of true effect (T) was 0.9 (Figure 3).
Non-lean MASLD patients had 2.1 times the odds of hepatic steatosis (pOR = 2.1, 95%CI: 1.5, 2.9, P < 0.000) heterogeneity test Q = 13.6, df = 6, I2 = 56.0, T2 = 0.1, T = 0.3, P = 0.03, compared to lean MASLD. Furthermore, lean MASLD patients were less likely to have cirrhosis (pOR = 0.7, 95%CI: 0.5, 0.9, P = 0.007), heterogeneity test Q = 5.4, df = 3, I2 = 44.0, T2 = 0.03, T = 0.2, P = 0.1 compared to non-lean MASLD. Lean MASLD had lower odds of hypertension compared to non-lean MASLD (pOR = 0.7, 95%CI: 0.6, 0.9, P = 0.002), heterogeneity test Q = 43.6, df = 9, I2 = 79.3, T2 = 0.09, T = 0.1, P < 0.001, compared to non-lean MASLD patients (Figures 5, 6, and 8). There was no significant statistical difference in mortality between lean and non-lean MASLD groups (pOR = 1.4, 95%CI: 1.0, 2.0, P = 0.06), heterogeneity test Q = 109.1, df = 10, I2 = 90.9, T2 = 0.3, T = 0.5, P < 0.001 (Figures 4, 7, and 9). In addition, there was no significant statistical difference in presence of MASH and CVD among lean vs non-lean MASLD/MASH 1.2 (pOR = 1.2, 95%CI: 0.7, 2.0, P = 0.5), MASH heterogeneity test Q = 121.6, df = 11, I2 = 0.91, T2 = 0.6, T = 0.8, P < 0.001. CVD (pOR = 0.9, 95%CI: 0.7, 1.0, P = 0.1), heterogeneity test Q = 15.4, df = 11, I2 = 28.6, T2 = 0.03, T = 0.2, P = 0.2.
The study aims to assess seven outcomes of liver and CMD among lean and non-lean with MASLD. Those outcomes include liver fibrosis, steatosis, MASH, cirrhosis, CVD, hypertension, and death.
Both groups of lean and non-lean MASLD had substantial long-term liver and non-liver comorbidities, suggesting that obesity should not be the sole criterion for MASLD screening[20,31]. There is a need to develop risk calculators that include variables such as sex, age, race, ethnicity, and vascular adipose tissue concentration, along with other well-known CVD risk factors, to determine the risk of CVD in patients with obesity[39]. In addition, compared with lean individuals, baseline metabolic health in obese individuals is not significantly associated with incident CVD, as we also found in our pooled estimates[40]. Furthermore, previous evidence shows that lean MASLD patients tend to have less severe histological features than overweight or obese MASLD patients, including fibrosis, steatohepatitis, and MASH, as we’ve observed in our pooled estimate[23]. However, the risk of liver-related mortality is higher in lean MASLD patients than in non-lean MASLD patients in some studies published in the literature, but we found evidence of the opposite effect with 40% higher odds of mortality in non-lean adults compared to lean individuals with our P value = 0.06[41]. Lean in
The pathophysiology of the interaction between MASLD and CMD, in which MASLD contributes to CMD development, was initially unclear[44]. However, recent studies have demonstrated that MASLD is a significant risk factor for major adverse cardiovascular events and the leading cause of mortality in adults with MASLD[44,45]. Emerging evidence suggests that these conditions share common pathogenic mechanisms, including visceral obesity, low-grade inflammation, oxidative stress, and insulin resistance[43,44,46]. Liver damage in MASLD leads to the release of various inflammatory biomarkers, including cytokines, reactive oxygen species, tumor necrosis factor-α, transforming growth factor-β, plasminogen activator inhibitor-1, C-reactive protein, and interleukin-6. The imbalance in the secretion of these proinflammatory biomarkers contributes to the development of CMD[43,47]. Additionally, decreased plasma adiponectin levels, primarily secreted by the liver, represent another contributing mechanism[44]. There is limited literature on patient outcomes when CMD overlaps with MASLD. Most studies focus solely on cardiac events[44,48]. Targher et al[44] reported that within the MASLD population, the pooled mortality rate was 12.6 per 1000 person-years (95%CI: 6.7, 23.7) for all-cause mortality, 4.2 per 1000 person-years (95%CI: 1.3, 7.0) for cardiac-specific mortality, 2.8 per 1000 person-years (95%CI: 0.8, 4.9) for extrahepatic cancer-specific mortality, and 0.92 per 1000 person-years (95%CI: 0.0, 2.2) for liver-specific mortality. These cardiovascular complications include nonfatal coronary heart disease, stroke, HF, or car
Management should include screening for and addressing both modifiable and non-modifiable risk factors of CVD in MASLD patients. Non-modifiable risk factors include age, male gender, family history of early-onset CVD, certain ethnic backgrounds (e.g., South Asian), and early menopause (before age 40 years). Modifiable risk factors, such as smoking, high plasma low-density lipoprotein cholesterol levels, sedentary lifestyle, poor dietary habits, and obesity-related conditions (e.g., hypertension, insulin resistance, atherogenic dyslipidemia, and type 2 diabetes mellitus), should be addressed. In patients with established risk factors, statin therapy is recommended for those at intermediate risk (≥ 7.5% 10-year estimated CVD risk)[43]. For patients with established type 2 diabetes mellitus, glucagon-like peptide-1 receptor agonists, sodium-glucose cotransporter 2 inhibitors, or pioglitazone should be added to the treatment plan. Other medications, such as renin-angiotensin system inhibitors, may also be considered. Given the complexity of CMD associated with MASLD, a multidisciplinary approach to management is essential. However, there are currently no specific guidelines addressing both conditions simultaneously, as traditional approaches manage them separately within their respective specialties. Additionally, there is limited research comparing lean and non-lean individuals affected by both conditions concurrently. This highlights the importance of a multidisciplinary team in developing an integrated, individualized management plan for patients with overlapping CMD and MASLD. The core team should include cardiologists, nephrologists, endocrinologists, hepatologists, gastroenterologists, psychologists, and dietitians/nutritionists to optimize patient outcomes. While primary prevention strategies for CMD-MASLD interactions focus on shared risk factors, there is a lack of literature specifically addressing lean patients. Preventive measures are often elaborated for overweight or obese individuals including regular physical activity and a healthy diet, emphasizing vegetables, fruits, and nuts, and minimal consumption of processed whole grains[38-50].
Our study has few limitations. First, we relied on secondary data and published literature that had variability in how MASLD, CMD, liver diseases, and obesity were defined and measured across studies. Many of the studies defined differently lean and non-lean patients based on two distinct cutoffs for BMI: One group of studies considered Asian descent with BMI < 23 kg/m2, and the second group used BMI < 25 kg/m2 which may lead to classification bias and complicating direct comparison. Additionally, some studies included only one sociodemographic group with individuals of European descent and other only Asian populations, with the exception of a few that were conducted worldwide. Some variables in Table 2 were not reported in all studies, resulting in missing data, which we acknowledge as a study limitation. Finally, we acknowledge the higher heterogeneity observed in mortality, MASH, and fibrosis outcomes, which may have influenced the pooled estimates.
Liver complications and CMD occur frequently in patients who are obese or overweight. These two complications can overlap or occur independently. While it is generally known that lean MASLD patients have overall fewer complications than non-lean MASLD patients, this significant difference at the population level does not imply clinical differences, as cases of complications have also been observed among lean MASLD individuals in clinical practice. Therefore, obesity should not be the sole criterion for screening, and there is a need to develop body fat percentage metrics and other parameters for better assessment of lean patients.
| 1. | Chan WK, Chuah KH, Rajaram RB, Lim LL, Ratnasingam J, Vethakkan SR. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): A State-of-the-Art Review. J Obes Metab Syndr. 2023;32:197-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 617] [Cited by in RCA: 527] [Article Influence: 175.7] [Reference Citation Analysis (10)] |
| 2. | Zheng H, Sechi LA, Navarese EP, Casu G, Vidili G. Metabolic dysfunction-associated steatotic liver disease and cardiovascular risk: a comprehensive review. Cardiovasc Diabetol. 2024;23:346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 112] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 3. | Kuchay MS, Martínez-Montoro JI, Choudhary NS, Fernández-García JC, Ramos-Molina B. Non-Alcoholic Fatty Liver Disease in Lean and Non-Obese Individuals: Current and Future Challenges. Biomedicines. 2021;9:1346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 74] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 4. | World Health Organization. Obesity and overweight. May 7, 2025. [cited 3 August 2025]. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. |
| 5. | Jih J, Mukherjea A, Vittinghoff E, Nguyen TT, Tsoh JY, Fukuoka Y, Bender MS, Tseng W, Kanaya AM. Using appropriate body mass index cut points for overweight and obesity among Asian Americans. Prev Med. 2014;65:1-6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 210] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 6. | Wong V. Overview of Lean MASH and MASLD. Gastroenterol Hepatol (N Y). 2024;20:745-747. [PubMed] |
| 7. | Danpanichkul P, Suparan K, Prasitsumrit V, Ahmed A, Wijarnpreecha K, Kim D. Long-term outcomes and risk modifiers of metabolic dysfunction-associated steatotic liver disease between lean and non-lean populations. Clin Mol Hepatol. 2025;31:74-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 51.0] [Reference Citation Analysis (1)] |
| 8. | Potter AW, Chin GC, Looney DP, Friedl KE. Defining Overweight and Obesity by Percent Body Fat Instead of Body Mass Index. J Clin Endocrinol Metab. 2025;110:e1103-e1107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 70] [Article Influence: 70.0] [Reference Citation Analysis (0)] |
| 9. | De A, Mehta M, Singh P, Bhagat N, Mitra S, Das A, Duseja A. Lean Indian patients with non-alcoholic fatty liver disease (NAFLD) have less metabolic risk factors but similar liver disease severity as non-lean patients with NAFLD. Int J Obes (Lond). 2023;47:986-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 10. | Ishido S, Tamaki N, Takahashi Y, Uchihara N, Suzuki K, Tanaka Y, Miyamoto H, Yamada M, Matsumoto H, Nobusawa T, Keitoku T, Takaura K, Tanaka S, Maeyashiki C, Yasui Y, Tsuchiya K, Nakanishi H, Kurosaki M, Izumi N. Risk of cardiovascular disease in lean patients with nonalcoholic fatty liver disease. BMC Gastroenterol. 2023;23:211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 11. | Niriella MA, Ediriweera DS, Kasturiratne A, De Silva ST, Dassanayaka AS, De Silva AP, Kato N, Pathmeswaran A, Wickramasinghe AR, de Silva HJ. Outcomes of NAFLD and MAFLD: Results from a community-based, prospective cohort study. PLoS One. 2021;16:e0245762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 12. | Yoshitaka H, Hamaguchi M, Kojima T, Fukuda T, Ohbora A, Fukui M. Nonoverweight nonalcoholic fatty liver disease and incident cardiovascular disease: A post hoc analysis of a cohort study. Medicine (Baltimore). 2017;96:e6712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 92] [Article Influence: 10.2] [Reference Citation Analysis (3)] |
| 13. | Boonchai P, Kositamongkol C, Jitrukthai S, Phothirat S, Mepramoon E, Nimitpunya P, Srivanichakorn W, Chaisathaphol T, Washirasaksiri C, Auesomwang C, Sitasuwan T, Tinmanee R, Sayabovorn N, Charatcharoenwitthaya P, Phisalprapa P. Clinical Differences and Non-Alcoholic Fatty Liver Disease-Related Factors of Lean and Non-Lean Patients with Metabolic Syndrome. J Clin Med. 2022;11:2445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Kim H, Lee CJ, Ahn SH, Lee KS, Lee BK, Baik SJ, Kim SU, Lee JI. MAFLD Predicts the Risk of Cardiovascular Disease Better than NAFLD in Asymptomatic Subjects with Health Check-Ups. Dig Dis Sci. 2022;67:4919-4928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (2)] |
| 15. | Alam S, Gupta UD, Alam M, Kabir J, Chowdhury ZR, Alam AK. Clinical, anthropometric, biochemical, and histological characteristics of nonobese nonalcoholic fatty liver disease patients of Bangladesh. Indian J Gastroenterol. 2014;33:452-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (4)] |
| 16. | Chen YP, Lu FB, Hu YB, Xu LM, Zheng MH, Hu ED. A systematic review and a dose-response meta-analysis of coffee dose and nonalcoholic fatty liver disease. Clin Nutr. 2019;38:2552-2557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 17. | Hagström H, Nasr P, Ekstedt M, Hammar U, Stål P, Hultcrantz R, Kechagias S. Risk for development of severe liver disease in lean patients with nonalcoholic fatty liver disease: A long-term follow-up study. Hepatol Commun. 2018;2:48-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 244] [Article Influence: 30.5] [Reference Citation Analysis (7)] |
| 18. | Kumar R, Rastogi A, Sharma MK, Bhatia V, Tyagi P, Sharma P, Garg H, Chandan Kumar KN, Bihari C, Sarin SK. Liver stiffness measurements in patients with different stages of nonalcoholic fatty liver disease: diagnostic performance and clinicopathological correlation. Dig Dis Sci. 2013;58:265-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 76] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 19. | Leung JC, Loong TC, Wei JL, Wong GL, Chan AW, Choi PC, Shu SS, Chim AM, Chan HL, Wong VW. Histological severity and clinical outcomes of nonalcoholic fatty liver disease in nonobese patients. Hepatology. 2017;65:54-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 291] [Article Influence: 32.3] [Reference Citation Analysis (3)] |
| 20. | Ye Q, Zou B, Yeo YH, Li J, Huang DQ, Wu Y, Yang H, Liu C, Kam LY, Tan XXE, Chien N, Trinh S, Henry L, Stave CD, Hosaka T, Cheung RC, Nguyen MH. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5:739-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 741] [Cited by in RCA: 672] [Article Influence: 112.0] [Reference Citation Analysis (6)] |
| 21. | Fracanzani AL, Petta S, Lombardi R, Pisano G, Russello M, Consonni D, Di Marco V, Cammà C, Mensi L, Dongiovanni P, Valenti L, Craxì A, Fargion S. Liver and Cardiovascular Damage in Patients With Lean Nonalcoholic Fatty Liver Disease, and Association With Visceral Obesity. Clin Gastroenterol Hepatol. 2017;15:1604-1611.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 179] [Article Influence: 19.9] [Reference Citation Analysis (6)] |
| 22. | Hillenbrand A, Kiebler B, Schwab C, Scheja L, Xu P, Henne-Bruns D, Wolf AM, Knippschild U. Prevalence of non-alcoholic fatty liver disease in four different weight related patient groups: association with small bowel length and risk factors. BMC Res Notes. 2015;8:290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 23. | Sookoian S, Puri P, Castaño GO, Scian R, Mirshahi F, Sanyal AJ, Pirola CJ. Nonalcoholic steatohepatitis is associated with a state of betaine-insufficiency. Liver Int. 2017;37:611-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 24. | Margariti A, Deutsch M, Manolakopoulos S, Tiniakos D, Papatheodoridis GV. The severity of histologic liver lesions is independent of body mass index in patients with nonalcoholic fatty liver disease. J Clin Gastroenterol. 2013;47:280-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (3)] |
| 25. | Vos B, Moreno C, Nagy N, Féry F, Cnop M, Vereerstraeten P, Devière J, Adler M. Lean non-alcoholic fatty liver disease (Lean-NAFLD): a major cause of cryptogenic liver disease. Acta Gastroenterol Belg. 2011;74:389-394. [PubMed] |
| 26. | Ahmed OT, Gidener T, Mara KC, Larson JJ, Therneau TM, Allen AM. Natural History of Nonalcoholic Fatty Liver Disease With Normal Body Mass Index: A Population-Based Study. Clin Gastroenterol Hepatol. 2022;20:1374-1381.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 27. | Wijarnpreecha K, Li F, Lundin SK, Suresh D, Song MW, Tao C, Chen VL, Lok ASF. Higher mortality among lean patients with non-alcoholic fatty liver disease despite fewer metabolic comorbidities. Aliment Pharmacol Ther. 2023;57:1014-1027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (4)] |
| 28. | Weinberg EM, Trinh HN, Firpi RJ, Bhamidimarri KR, Klein S, Durlam J, Watkins S, Reddy KR, Weiss M, Zink RC, Lok AS. Lean Americans With Nonalcoholic Fatty Liver Disease Have Lower Rates of Cirrhosis and Comorbid Diseases. Clin Gastroenterol Hepatol. 2021;19:996-1008.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 29. | Nso N, Mergen D, Ikram M, Macrinici V, Hussain K, Lee K, Ugwendum D, Trimingham M, Balasubramanian S, Sam R, Njei B. Cardiovascular morbidity and mortality in lean vs. non-lean MASLD: A comprehensive meta-analysis. Curr Probl Cardiol. 2024;49:102569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 30. | Arvind A, Henson JB, Osganian SA, Nath C, Steinhagen LM, Memel ZN, Donovan A, Balogun O, Chung RT, Simon TG, Corey KE. Risk of Cardiovascular Disease in Individuals With Nonobese Nonalcoholic Fatty Liver Disease. Hepatol Commun. 2022;6:309-319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Younes R, Govaere O, Petta S, Miele L, Tiniakos D, Burt A, David E, Vecchio FM, Maggioni M, Cabibi D, McLeod D, Pareja MJ, Fracanzani AL, Aller R, Rosso C, Ampuero J, Gallego-Durán R, Armandi A, Caviglia GP, Zaki MYW, Liguori A, Francione P, Pennisi G, Grieco A, Birolo G, Fariselli P, Eslam M, Valenti L, George J, Romero-Gómez M, Anstee QM, Bugianesi E. Caucasian lean subjects with non-alcoholic fatty liver disease share long-term prognosis of non-lean: time for reappraisal of BMI-driven approach? Gut. 2022;71:382-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 151] [Article Influence: 37.8] [Reference Citation Analysis (1)] |
| 32. | Feldman A, Wernly B, Strebinger G, Eder SK, Zandanell S, Niederseer D, Strasser M, Haufe H, Sotlar K, Paulweber B, Datz C, Aigner E. Liver-related Mortality is Increased in Lean Subjects with Non- alcoholic Fatty Liver Disease Compared to Overweight and Obese Subjects. J Gastrointestin Liver Dis. 2021;30:366-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 33. | Iritani S, Akuta N, Kawamura Y, Kajiwara A, Kasuya K, Fujiyama S, Sezaki H, Hosaka T, Kobayashi M, Kobayashi M, Saito S, Suzuki F, Arase Y, Ikeda K, Suzuki Y, Kumada H. Non-invasive predictors of prognosis of Asian patients with histopathologically-confirmed lean nonalcoholic fatty liver disease. BMC Gastroenterol. 2020;20:368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 34. | Almomani A, Kumar P, Onwuzo S, Boustany A, Krishtopaytis E, Hitawala A, Alshaikh D, Albakri A, Hussein L, Hussein E, Asaad I. Epidemiology and prevalence of lean nonalcoholic fatty liver disease and associated cirrhosis, hepatocellular carcinoma, and cardiovascular outcomes in the United States: a population-based study and review of literature. J Gastroenterol Hepatol. 2023;38:269-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (4)] |
| 35. | Khayyat YM. Lean non-alcoholic fatty liver disease and associated metabolic disturbance: A Saudi Arabian cross-sectional study. Physiol Rep. 2021;9:e14949. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 36. | Semmler G, Wernly S, Bachmayer S, Wernly B, Schwenoha L, Huber-Schönauer U, Stickel F, Niederseer D, Aigner E, Datz C. Nonalcoholic Fatty Liver Disease in Lean Subjects: Associations With Metabolic Dysregulation and Cardiovascular Risk-A Single-Center Cross-Sectional Study. Clin Transl Gastroenterol. 2021;12:e00326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 37. | Zou B, Yeo YH, Nguyen VH, Cheung R, Ingelsson E, Nguyen MH. Prevalence, characteristics and mortality outcomes of obese, nonobese and lean NAFLD in the United States, 1999-2016. J Intern Med. 2020;288:139-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 207] [Article Influence: 34.5] [Reference Citation Analysis (4)] |
| 38. | Golabi P, Paik JM, Kumar A, Al Shabeeb R, Eberly KE, Cusi K, GunduRao N, Younossi ZM. Nonalcoholic fatty liver disease (NAFLD) and associated mortality in individuals with type 2 diabetes, pre-diabetes, metabolically unhealthy, and metabolically healthy individuals in the United States. Metabolism. 2023;146:155642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 80] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 39. | Rana MN, Neeland IJ. Adipose Tissue Inflammation and Cardiovascular Disease: An Update. Curr Diab Rep. 2022;22:27-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 110] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 40. | Mongraw-Chaffin M, Foster MC, Anderson CAM, Burke GL, Haq N, Kalyani RR, Ouyang P, Sibley CT, Tracy R, Woodward M, Vaidya D. Metabolically Healthy Obesity, Transition to Metabolic Syndrome, and Cardiovascular Risk. J Am Coll Cardiol. 2018;71:1857-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 369] [Cited by in RCA: 314] [Article Influence: 39.3] [Reference Citation Analysis (1)] |
| 41. | Ha J, Yim SY, Karagozian R. Mortality and Liver-Related Events in Lean Versus Non-Lean Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2023;21:2496-2507.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (3)] |
| 42. | Albhaisi S, Chowdhury A, Sanyal AJ. Non-alcoholic fatty liver disease in lean individuals. JHEP Rep. 2019;1:329-341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 136] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 43. | Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, Mills PR, Keach JC, Lafferty HD, Stahler A, Haflidadottir S, Bendtsen F. Liver Fibrosis, but No Other Histologic Features, Is Associated With Long-term Outcomes of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015;149:389-97.e10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2514] [Cited by in RCA: 2380] [Article Influence: 216.4] [Reference Citation Analysis (5)] |
| 44. | Targher G, Chonchol M, Zoppini G, Abaterusso C, Bonora E. Risk of chronic kidney disease in patients with non-alcoholic fatty liver disease: is there a link? J Hepatol. 2011;54:1020-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 135] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 45. | Adams LA, Anstee QM, Tilg H, Targher G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut. 2017;66:1138-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 987] [Cited by in RCA: 863] [Article Influence: 95.9] [Reference Citation Analysis (9)] |
| 46. | Theodorakis N, Nikolaou M. From Cardiovascular-Kidney-Metabolic Syndrome to Cardiovascular-Renal-Hepatic-Metabolic Syndrome: Proposing an Expanded Framework. Biomolecules. 2025;15:213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 55] [Article Influence: 55.0] [Reference Citation Analysis (1)] |
| 47. | Kronenberg F. Emerging risk factors and markers of chronic kidney disease progression. Nat Rev Nephrol. 2009;5:677-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 110] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 48. | Ix JH, Sharma K. Mechanisms linking obesity, chronic kidney disease, and fatty liver disease: the roles of fetuin-A, adiponectin, and AMPK. J Am Soc Nephrol. 2010;21:406-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 294] [Cited by in RCA: 274] [Article Influence: 17.1] [Reference Citation Analysis (5)] |
| 49. | Simon TG, Roelstraete B, Hagström H, Sundström J, Ludvigsson JF. Non-alcoholic fatty liver disease and incident major adverse cardiovascular events: results from a nationwide histology cohort. Gut. 2022;71:1867-1875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 231] [Article Influence: 57.8] [Reference Citation Analysis (1)] |
| 50. | Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, Himmelfarb CD, Khera A, Lloyd-Jones D, McEvoy JW, Michos ED, Miedema MD, Muñoz D, Smith SC Jr, Virani SS, Williams KA Sr, Yeboah J, Ziaeian B. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:e596-e646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1459] [Cited by in RCA: 2118] [Article Influence: 302.6] [Reference Citation Analysis (3)] |