Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.115299
Revised: December 8, 2025
Accepted: January 15, 2026
Published online: April 7, 2026
Processing time: 159 Days and 19.5 Hours
Ulcerative colitis (UC) is a chronic, recurrent inflammatory disease of the gas
To determine whether berberine alleviates dextran sulfate sodium (DSS)-induced UC in mice by enhancing gut microbiota-dependent intestinal barrier function and inhibiting gasdermin D (GSDMD) activation.
An acute colitis mouse model was established by administering 3% DSS. The mice were treated daily with berberine (50 mg/kg and 100 mg/kg), after which body weight, colon length, histological scoring, and intestinal levels of inflammatory cytokines were assessed. Intestinal barrier permeability was evaluated, and 16S ribosomal RNA sequencing was conducted. Fecal microbiota transplantation and cohousing studies were performed to determine the role of the gut microbiota. The inhibitory effects of GSDMD were pharmacologically manipulated to sub
Berberine has shown significant potential for alleviating the severity of DSS-induced acute colitis. Fecal microbiota transplantation and cohousing experiments showed that the gut microbiota is indispensable for the beneficial effects of ber
Berberine alleviates experimental colitis by inhibiting the GSDMD-mediated pathway and increasing gut barrier function via a microbiota-mediated approach, thus offering a new molecular target for colitis therapy.
Core Tip: Berberine alleviates dextran sulfate sodium-induced murine ulcerative colitis through a mechanism that involves gut microbiota-dependent enhancement of intestinal barrier function and suppression of gasdermin D activation. Fecal microbiota trans
- Citation: Yang P, Wang R, Zhou YP, Wen JF, Yang DX. Berberine alleviates experimental colitis by enhancing gut-microbiota-dependent intestinal barrier function and suppressing gasdermin D activation. World J Gastroenterol 2026; 32(13): 115299
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/115299.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.115299
Ulcerative colitis (UC), the most prevalent form of inflammatory bowel disease (IBD), is a chronic, nonspecific inflammatory condition affecting the colon and rectum[1]. It is linked to a decreased quality of life and an increased susceptibility to developing coli
Inflammasomes are pivotal inflammatory protein assemblies that are integral to the innate immune system, and NOD-like receptors are among their essential components. These receptors can detect a wide range of endogenous and exogenous danger signals, triggering the activation of inflammatory responses[5]. The inflammasome is integral to the host’s defense strategies and plays a pivotal role in maintaining the balance of the intestinal immune system[6]. Gasdermin D (GSDMD) serves as a pivotal executor of the pyroptotic cascade and is ac
Traditional Chinese medicine (TCM) has a unique advantage in treating chronic diseases because of its multiple targets and minimal adverse effects[1,14,15]. Berberine is a valuable component of TCM, known for its ability to alleviate sym
DSS was purchased from MP Biochemicals (batch No. 0216011090, CA, United States). Berberine was purchased from Yuanye (batch No. B21379, China). Disulfiram was purchased from Med Chem Express (batch No. HY-B0240, NJ, United States). Anti-claudin-1 and anti-zonula occludens-1 (anti-ZO-1) antibodies were purchased from Proteintech (batch No. 21773-1-AP, 13050-1-AP, Wuhan, Hubei Province, China). Anti-GSDMD, anti-apoptosis-associated speck-like protein containing a caspase-recruitment domain (anti-ASC) and anti-β-actin were purchased from Affinity (batch No. AF4012, DF6304, AF7018, OH, United States). Horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (H + L) and HRP-conjugated goat anti-mouse IgG (H + L) were purchased from Proteintech (batch No. SA00001-2 and SA00001-1, Wuhan, Hubei Province, China). Enzyme-linked immunosorbent assay (ELISA) kit for mouse interleukin (IL)-1β, IL-6 and tumor necrosis factor-α (TNF-α) were purchased from Elabscience (batch No. E-EL-M0037, E-EL-M0044E-EL-M3063, China). Radioimmune precipitation assay lysis buffer was purchased from Beyotime (batch No. P0013B, China). Brilliant SYBR Green QPCR Master Mix was purchased from TransGen Biotech (batch No. AQ211-01, China). TRIzol was purchased from Thermo Fisher (batch No. 15596018, MA, United States). Polyvinylidene fluoride membranes was purchased from Sigma-Aldrich (batch No. IPVH0010, MO, United States). Protease inhibitor mixture was purchased from Aladdin (batch No. C129408, China) (Table 1).
| Key reagent or resource | Source | Identifier | Country |
| Anti-GSDMD | Affinity | AF4012 | United States |
| Anti-ASC | Affinity | DF6304 | United States |
| Anti-ZO-1 | Proteintech | 21773-1-AP | China |
| Anti-claudin-1 | Proteintech | 13050-1-AP | China |
| Anti-β-actin | Affinity | AF7018 | United States |
| HRP-labeled goat anti-rabbit IgG (H + L) | Affinity | S0001 | United States |
| Dextran sulfate sodium | MP Biochemicals | 0216011090 | United States |
| Disulfiram | Med Chem Express | HY-B0240 | United States |
| Berberine | Yuanye | B21379 | China |
| TRIzol | ThermoFisher | 15596018 | United States |
| Brilliant SYBR Green QPCR Master Mix | TransGen | AQ211-01 | China |
| ELISA kit for mouse IL-1β | Elabscience | E-EL-M0037 | China |
| ELISA kit for mouse IL-6 | Elabscience | E-EL-M0044 | China |
| ELISA kit for mouse TNF-α | Elabscience | E-EL-M3063 | China |
| Chemiluminescence (ECL) substrate | NCM | P10300 | China |
| Protease inhibitor mixture | Aladdin | C129408 | China |
| Polyvinylidene fluoride membranes | Sigma-Aldrich | IPVH0010 | United States |
| Multiskan Skyhigh With Touch Screen | Thermo Fisher Scientific | A51119600 | United States |
| Chemiluminescence imaging system | Shanghai Qinxing Scientific Instrument Co., Ltd | ChemiScope 6200 Touch | China |
| MagPure Soil DNA LQ Kit | Magen | D6356-02 | China |
| Western blot electrophoresis system | Bio-Rad Laboratories | 552BR | United States |
| Illumina sequencing adapte | OE Biotech Co., Ltd | China | |
| NanoDrop 2000 spectrophotometer | Thermo Fisher Scientific | United States | |
| Real-time fluorescence quantitative PCR system | Bioer Technology Co., Ltd | China |
Male C57BL/6 mice, 6-8 weeks of age, were employed in this study. These mice were housed within a controlled, enclosed environment. For the specific procedures of mouse euthanasia, please refer to the detailed instructions provided in the Supplementary material.
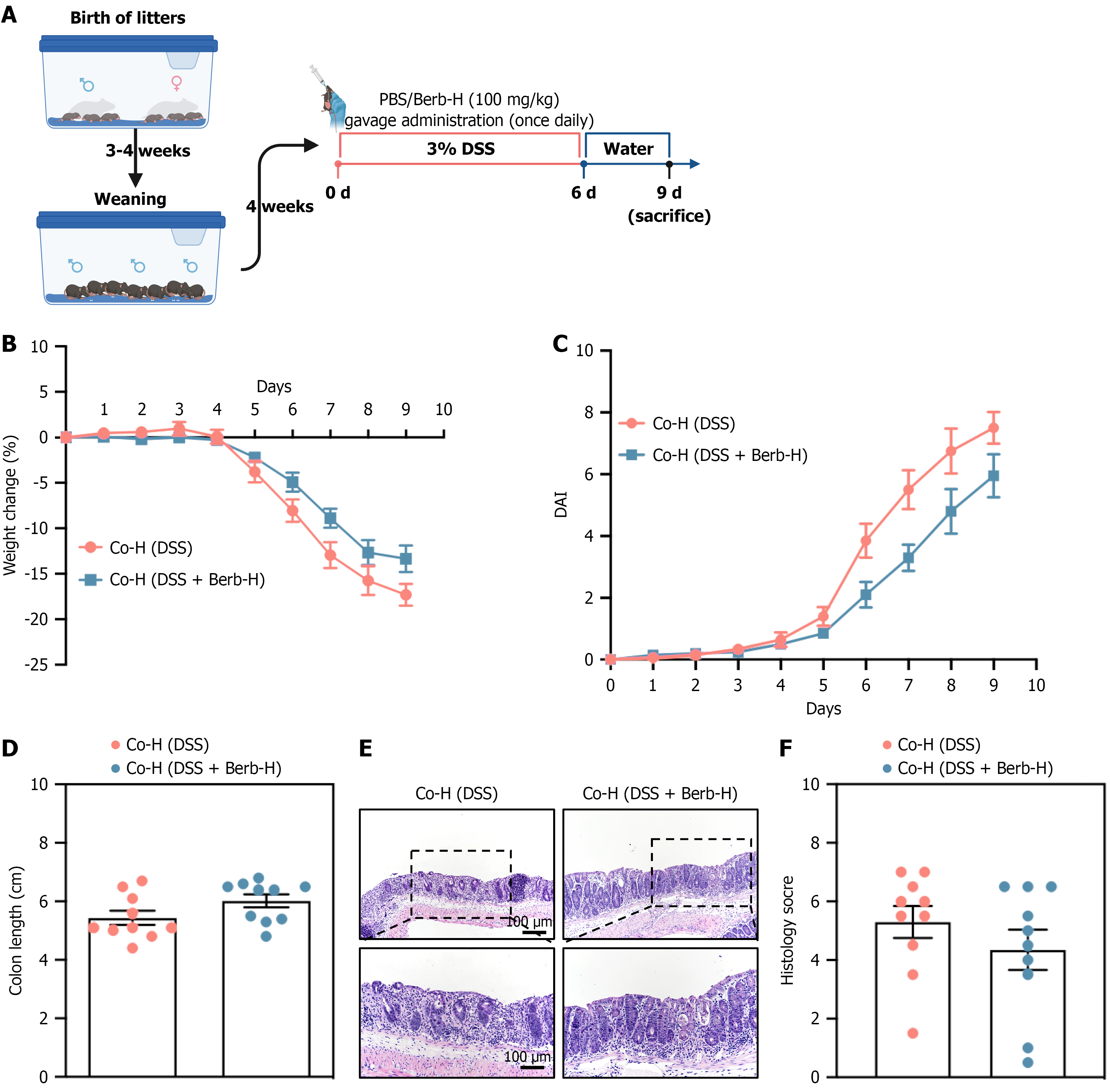
To induce acute experimental colitis, the mice were randomly assigned to four groups: Control group (CON, n = 5), DSS group (n = 8), DSS + 50 mg/kg berberine group (Berb-L, n = 8), and DSS + 100 mg/kg berberine group (Berb-H, n = 8). All mice except control animals were given 3% DSS in their drinking water for 6 days and then returned to regular water until the end of the experiment on day 9. Body weight, stool consistency, and appearance/posture were monitored daily to assess the disease activity index (DAI) in an unbiased manner. The DAI represents an aggregate assessment of weight reduction relative to the starting weight, stool consistency, and physical posture; refer to Supplementary Table 1 for the evaluation criteria.
Fecal microbiota transplantation (FMT) was carried out according to an established protocol[26,27]. Briefly, the donor mice were treated with berberine or vehicle for 6 days. After the feeding phase, the stools were gathered in a sterile set
Four-week-old male C57BL/6J mice were randomly assigned to the Ch-DSS or Ch-DSS-berberine (100 mg/kg) group (n = 8 per group), which were housed in the same cage but received different treatments. After 4 weeks of feeding, the Ch-DSS cohort was given drinking water by oral gavage for a period of seven days, whereas the Ch-DSS-berberine cohort was given berberine by oral gavage for the same duration. A DSS-induced colitis model was established as previously de
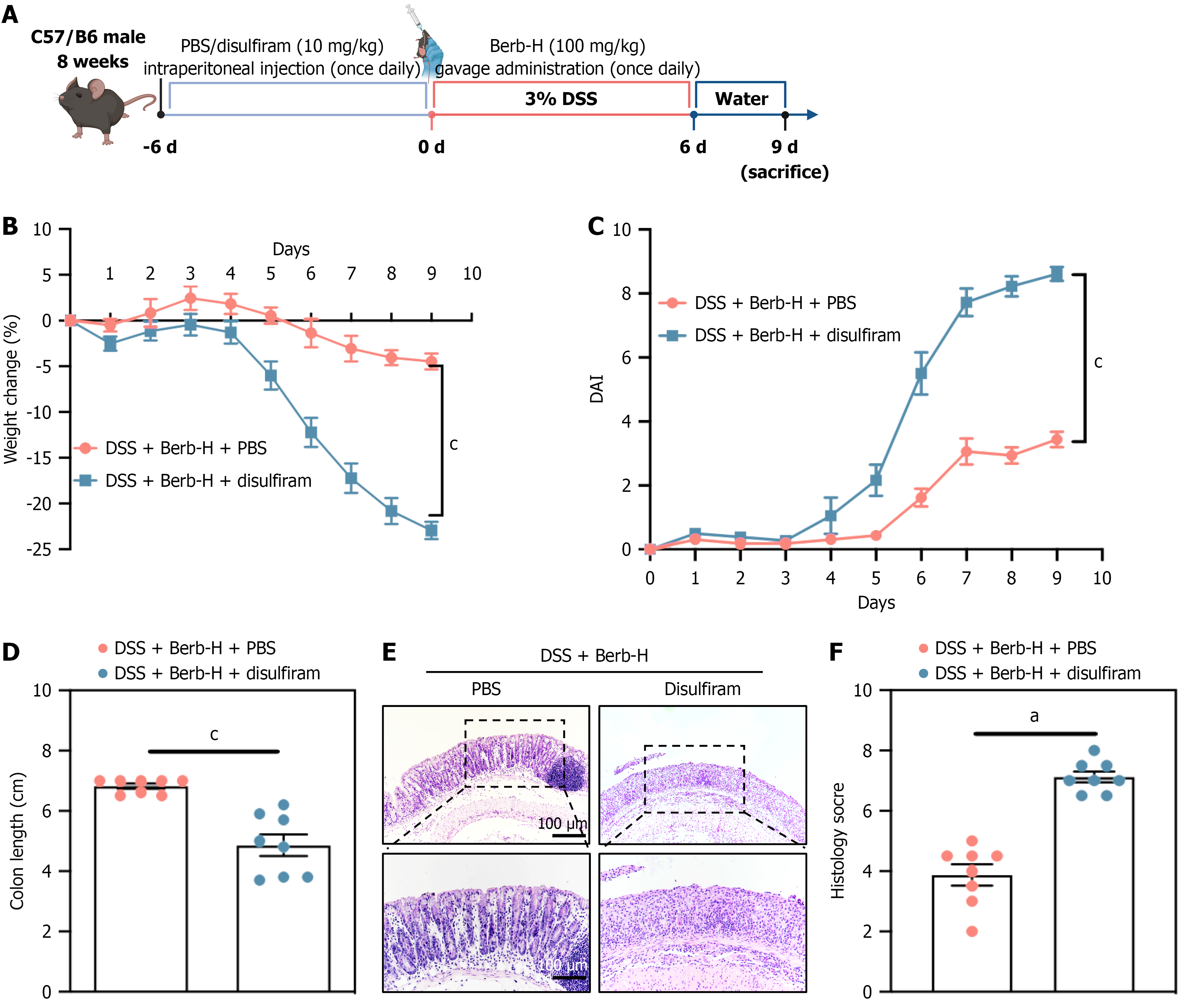
The mice (n = 8 per group) were randomly allocated to either the phosphate-buffered saline (PBS) or the disulfiram (10 mg/kg was intraperitoneally injected) treatment cohort and were treated for seven consecutive days. Berberine (100 mg/kg; designated Berb-H) was dissolved in sterile water and subsequently administered orally via gavage daily. A DSS-induced colitis model was established as previously described.
For histological analysis, colonic tissues were fixed in 4% paraformaldehyde for 48 hours, embedded in paraffin, se
Total RNA was extracted using TRIzol reagent and reverse-transcribed into complementary DNA. The resulting complementary DNA was subjected to quantitative polymerase chain reaction (PCR) on a Bioer fluorescence real-time PCR system using Brilliant SYBR Green QPCR Master Mix, following the manufacturer’s instructions. Quantitative PCR analysis was performed using EvaGreen QPCR MasterMix on a MiniOpticon Real-Time PCR Detection System (Bioer Technology Co., Ltd, China). The sequences of the primers that were used are listed in Table 2.
| Primer | Primer sequence (5’-3’) | Primer sequence (5’-3’) |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
| IL-6 | CTTGGGACTGATGCTGGTGAC | GCCATTGCACAACTCTTTTCTC |
| TNF-α | TACTGAACTTCGGGGTGATCG | TCCTCCACTTGGTGGTTTGC |
| Hprt | GTCCCAGCGTCGTGATTAGC | TGGCCTCCCATCTCCTTCA |
Colons were removed from mice, briefly washed with PBS, and then thoroughly rinsed in cold PBS containing gentamicin (20 μg/mL), penicillin G (200 μg/mL), and streptomycin (200 μg/mL) to eliminate remaining intestinal bacteria. The cleaned colonic tissues were then maintained in enriched culture medium supplemented with penicillin G (200 μg/mL) and streptomycin (200 μg/mL), After 24 hours of incubation at 37 °C, culture supernatant were collected, and the levels of IL-1β, IL-6 and TNF-α were measured using ELISA kits.
The colons were then sectioned at 3 μm and subjected to antigen retrieval in citrate buffer. The paraffin-embedded segments of the colon were methodically decolorized in xylene, after which they were progressively rehydrated in a graded alcohol series culminating in water. The tissue sections were then cooled to 4 °C for an extended overnight incubation period with the following primary antibodies, as per the protocol: Anti-ZO-1 (1:400), anti-claudin-1 (1:400), and anti-GSDMD (1:300). Immunohistochemical analysis was conducted using a biotinylated horse anti-rabbit IgG secondary antibody in conjunction with streptavidin-HRP, followed by the development of signals with diaminobenzidine.
To extract total protein, colon tissues were lysed in radioimmune precipitation assay buffer supplemented with protease and phosphatase inhibitor cocktails. Subsequently, equivalent amounts of protein were resolved by sodium-dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. These protein membranes were probed with specific primary antibodies, including anti-GSDMD (1:1000), anti-ASC (1:1000), and anti-β-actin (1:2000), followed by detection with the corresponding HRP-conjugated secondary antibodies (1:10000). The resulting bands were subsequently treated with an enhanced chemiluminescence substrate manufactured by and visualized within the Syngene fully automated gel imaging analysis system (ChemiScope 6200 Touch, China).
Genomic DNA was extracted from stool samples using the DNeasy PowerSoil kit (D6356-02, Magen, China) according to the manufacturer’s instructions. DNA concentration and purity were assessed with a NanoDrop 2000 spectrophotometer (Thermo Fisher, MA, United States), and integrity was verified by agarose gel electrophoresis. The V3-V4 regions of the bacterial 16S ribosomal RNA (rRNA) gene were amplified via PCR with the following specific primers. 343F: 5’-TACGGRAGGCAGCAG-3’; 798R: 5’-AGGGTATCTAATCCT-3’. The reverse primer was tagged with a unique sample barcode, ensuring that both primers were seamlessly compatible with the Illumina sequencing adapter. Raw sequencing data were obtained in FASTQ format. Paired-end reads were first processed with Cutadapt to remove adapter sequences and then imported into QIIME 2 for quality control. Low-quality reads were trimmed, denoised, merged, and filtered for chimeras using the DADA2 plugin with default parameters, generating amplicon sequence variants (ASVs), representative sequences, and an ASV abundance table. Taxonomic assignment of representative sequences was performed against the Silva database (version 138). Alpha diversity, including Good’s coverage, was calculated to evaluate microbial diversity within samples. Unweighted UniFrac distance matrices generated in QIIME 2 were used for principal analysis. All 16S rRNA gene amplicon sequencing and bioinformatic analyses were conducted by OE Biotech Co., Ltd (Shanghai, China).
We downloaded the GSDMD structure with the ID Q9D8T2 from the UniProt database (https://www.uniprot.org/). The structure of berberine was successfully obtained from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). We utilized AutoDockTools and Open Babel software to perform three-dimensional structural transformation of compounds, perform ligand extraction and format conversion, and prepare proteins for dehydration and hydrogenation using various preprocessing procedures. We used AutoDock for molecular docking simulations and PyMOL for the subsequent visualization of the results.
Molecular dynamics simulations were carried out using GROMACS 2022.3. AmberTools22 was used to preprocess small molecules, with the general amber force field assigned to them. Gaussian 16W was employed to add hydrogen atoms and calculate restrained electrostatic potentials, and the resulting charges were incorporated into the system topology. After completion of the simulations, trajectories were analyzed using the built-in analysis tools in GROMACS. Key parameters, including the root-mean-square deviation, root-mean-square fluctuation (RMSF) of individual residues, and the protein radius of gyration, were calculated. These analyses were combined with molecular mechanics/gene
Data were analyzed using GraphPad Prism (version 8.0.2) and are presented as the mean ± SEM. Statistical comparisons between two groups were performed using a two-tailed unpaired t test, while one-way ANOVA was applied for analyses involving more than two groups. A P value < 0.05 was considered statistically significant.
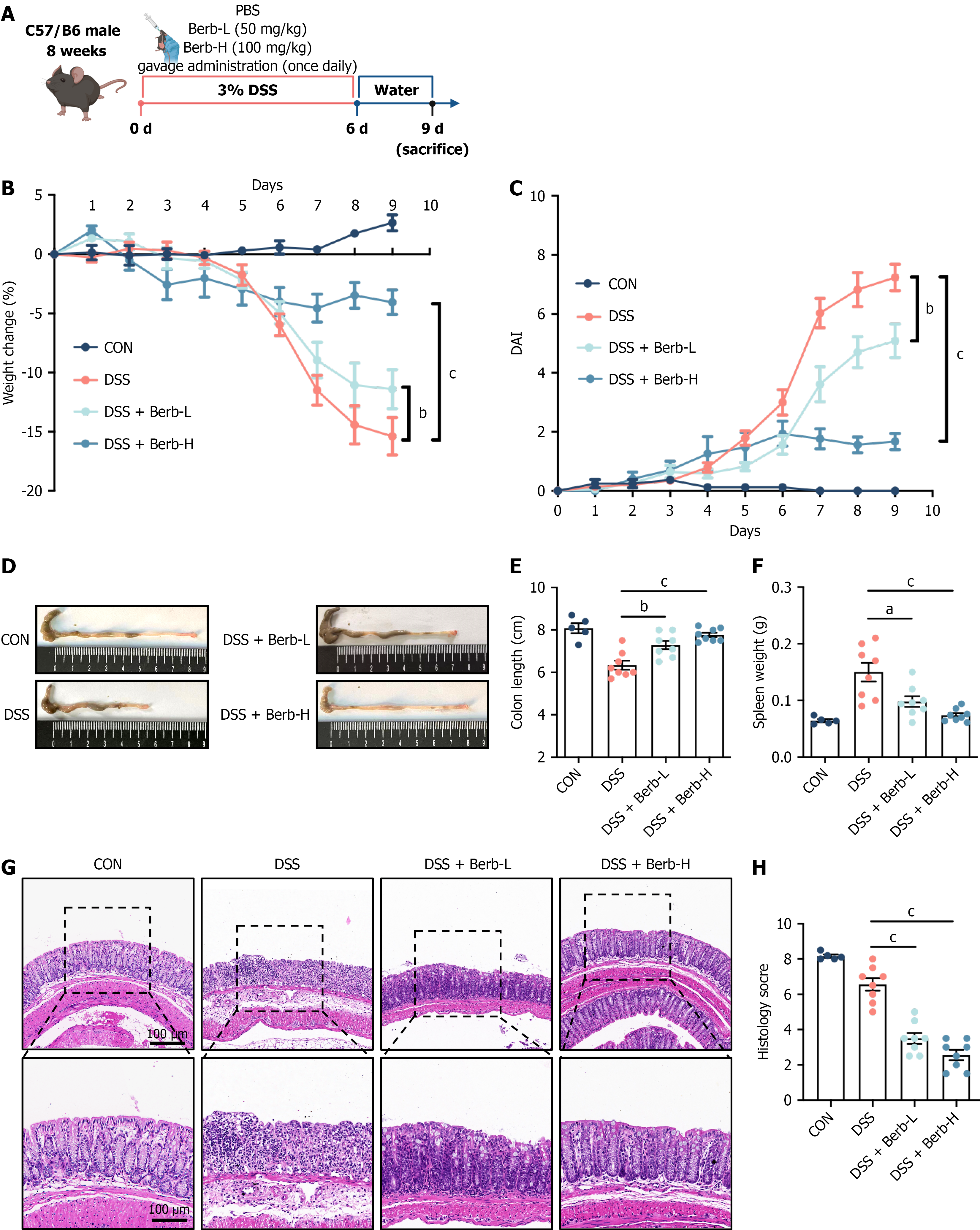
We investigated the ability of berberine to induce anti-inflammatory responses in acute colitis, as depicted in Figure 1A, which outlines the study subgroups and intervention schema. As shown in Figure 1B, the body weights of all groups except for the CON group tended to increase initially but then decrease. The mice exhibited marked intestinal inflammation, manifested as weight loss, diarrhea, and rectal hemorrhage; in contrast to those in the CON group, this inflammation led to a pronounced increase in the DAI score (Figure 1C). Furthermore, the study groups exhibited a significant decrease in colon length (Figure 1D and E) and an increase in spleen weight (Figure 1F). After various oral doses of ber
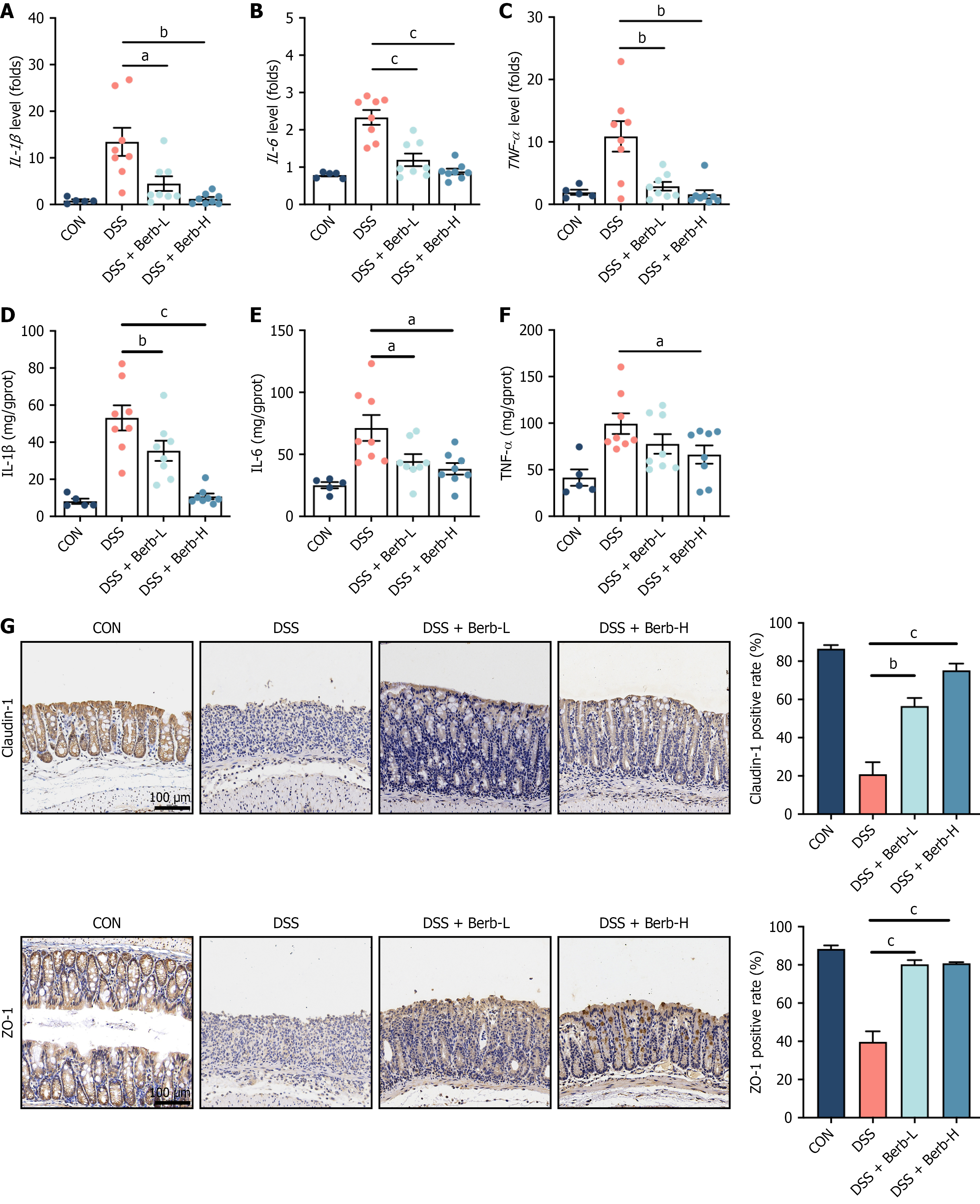
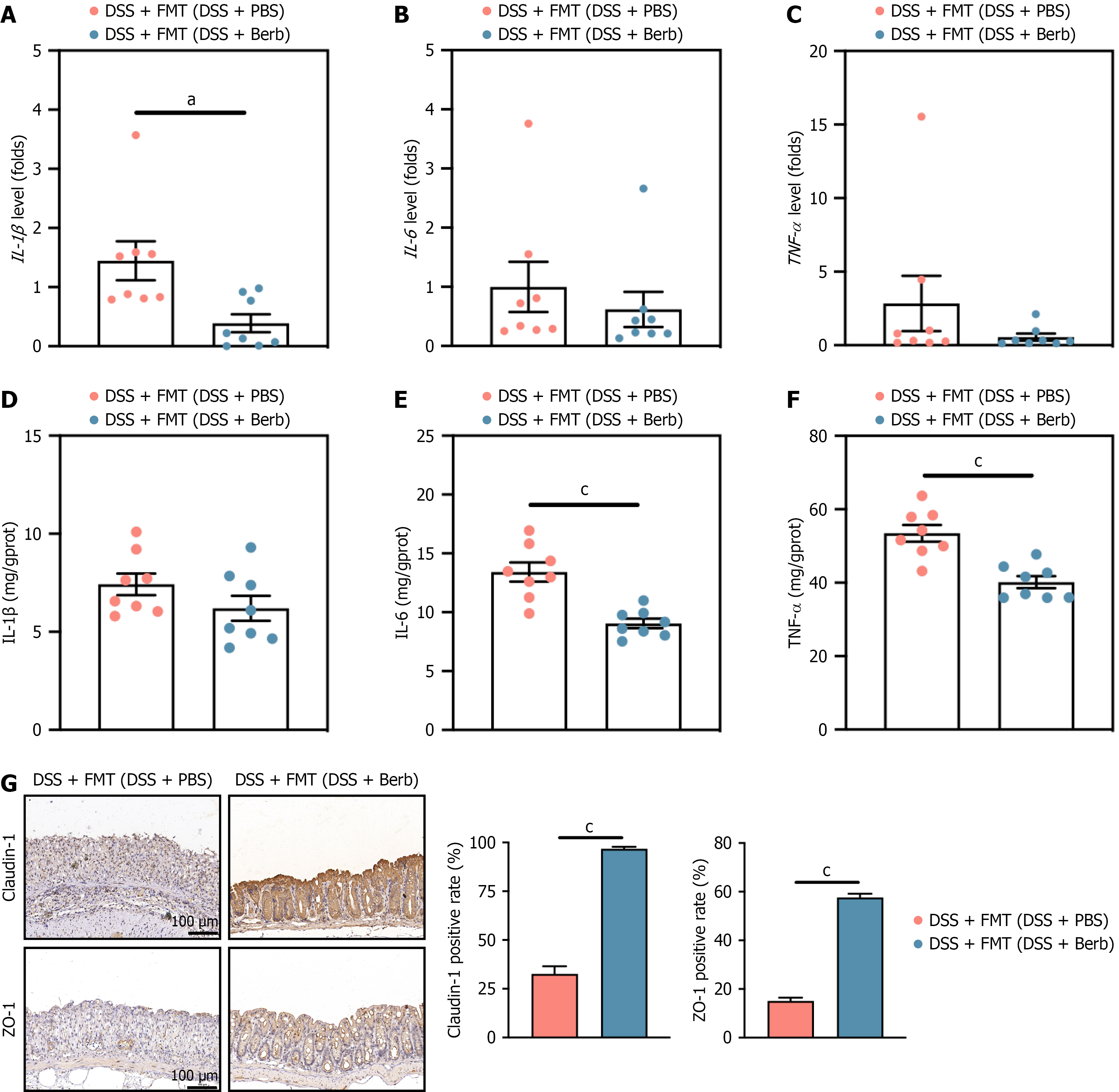
Regulation of cytokines is essential for controlling the inflammatory mechanism of colitis. We analyzed alterations in the levels of inflammation-associated cytokines in colonic tissue. The concentrations of primary proinflammatory cytokines, including IL-1β, IL-6, and TNF-α, were elevated in the colons of mice treated with DSS compared with those in the control group (Figure 2A-F). Berberine treatment normalized cytokine levels, with the Berb-H group demonstrating an especially remarkable improvement. Immunohistochemical analysis demonstrated that compared with the control treatment, DSS treatment reduced the protein expression of claudin-1 and ZO-1 in colonic tissue. Conversely, treatment with berberine increased claudin-1 protein expression (Figure 2G). Changes in ZO-1 levels exhibited a comparable trend (Figure 2G). Berberine attenuated DSS-induced intestinal injury and mucosal inflammation in mice by reducing colonic proinflammatory cytokine levels and enhancing the expression of anti-inflammatory factors in colonic tissue.
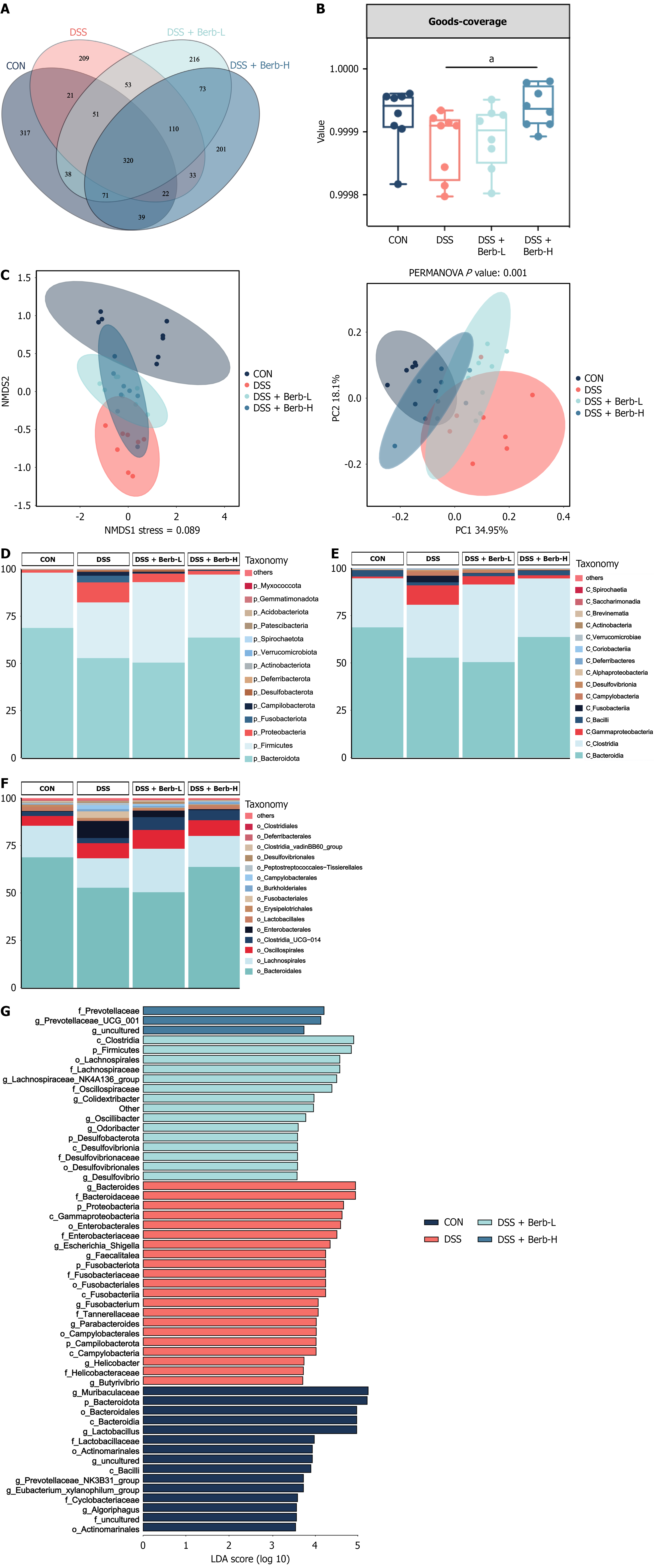
To understand the changes in the enteric microbiome associated with acute colitis following berberine administration, we performed 16S rRNA gene high-throughput sequencing to examine the intestinal microbial profiles of mice from four distinct groups. As illustrated in Supplementary Figure 1A, the curve gently leveled off, indicating that an ample sequ
We examined the species composition of the mouse gut microbiota in further detail for each category. Compared with the CON group, the experimental model group exhibited a notable increase in the prevalence of Fusobacteria and Proteobacteria, whereas the presence of Bacteroidetes decreased (Figure 3D-F). In berberine-treated mice, the relative abundance of Proteobacteria was decreased, whereas Bacteroidetes were enriched (Figure 3D-F). At the order level, the abundances of Enterobacterales, Erysipelotrichales, Campylobacterales, and Fusobacteriales increased in this model group, whereas the abundances of Bacteroidales, Lactobacillus, and Lachnospirales decreased (Figure 3D-F and Supplementary Figure 1B). The results (Figure 3G and Supplementary Figure 1C) indicated that the contributions of Bacteroidales, Muribaculaceae, and Lactobacillus were prominent within the CON group [linear discriminant analysis (LDA) ≥ 4]. Proteobacteria, Escherichia-Shigella, and Helicobacter significantly influenced the model group (LDA > 4), and Prevotellaceae_UCG_001 and Lachnospiraceae significantly affected the berberine group (LDA > 4).
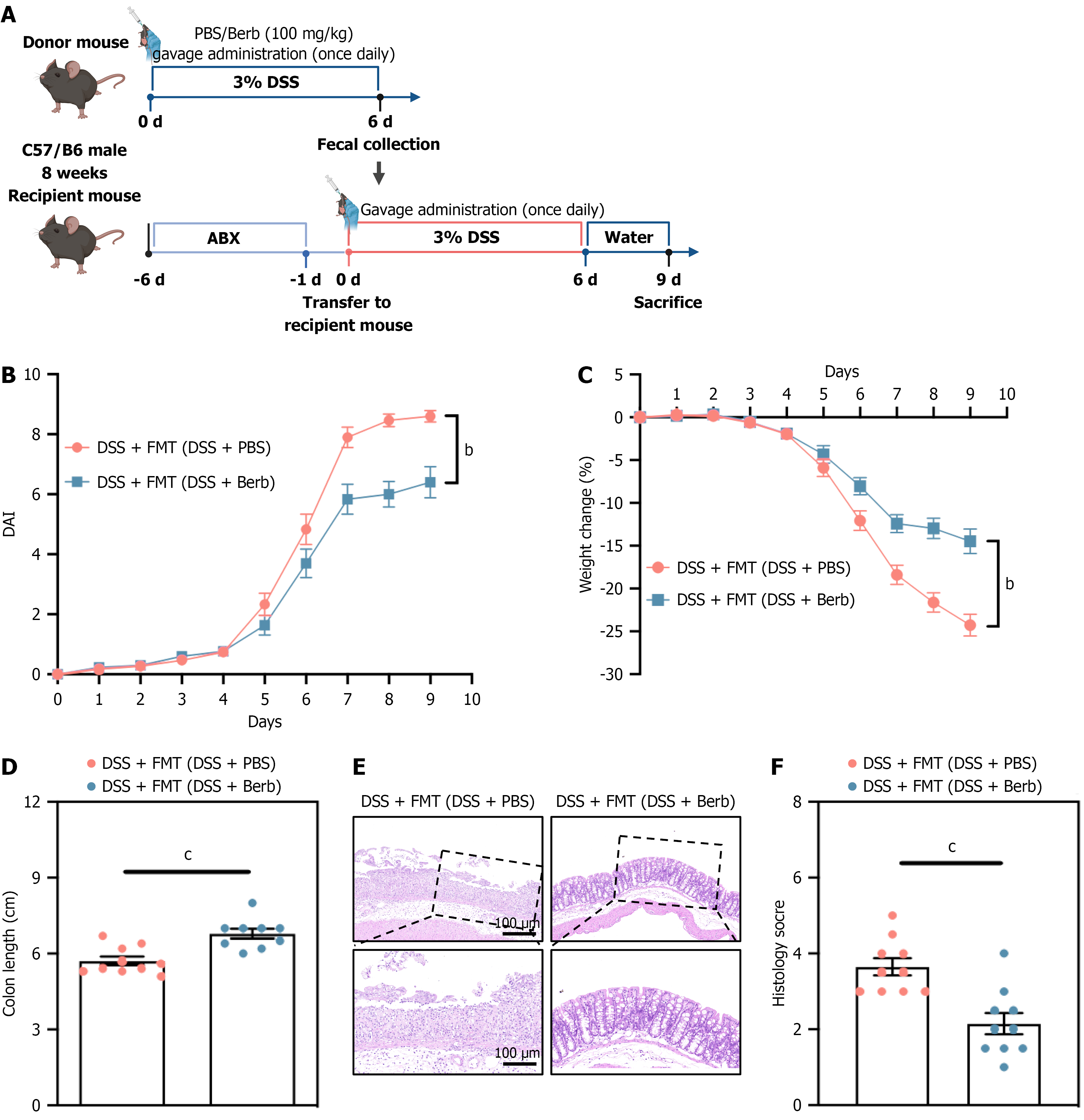
To elucidate the therapeutic potential of gut microbiota modifications induced by berberine for the treatment of colitis, FMT studies were conducted. The transplantation of microbiota from DSS-berberine-treated donor mice to DSS-exposed recipient mice mitigated DSS-induced colitis, as evidenced by increased colon length, decreased DAI scores, and reduced pathological abnormalities (Figure 4). Moreover, IL-1β, IL-6, and TNF-α levels were significantly lower in the recipient group (Figure 5A-F). Moreover, the levels of ZO-1 and claudin-1 were elevated in the FMT group, indicating that the integrity of the mucus layer was restored (Figure 5G). To evaluate the impact of berberine on the gut microbiota, we conducted microbiota transfer experiments by cohousing mice receiving different treatments. The cohousing of lit
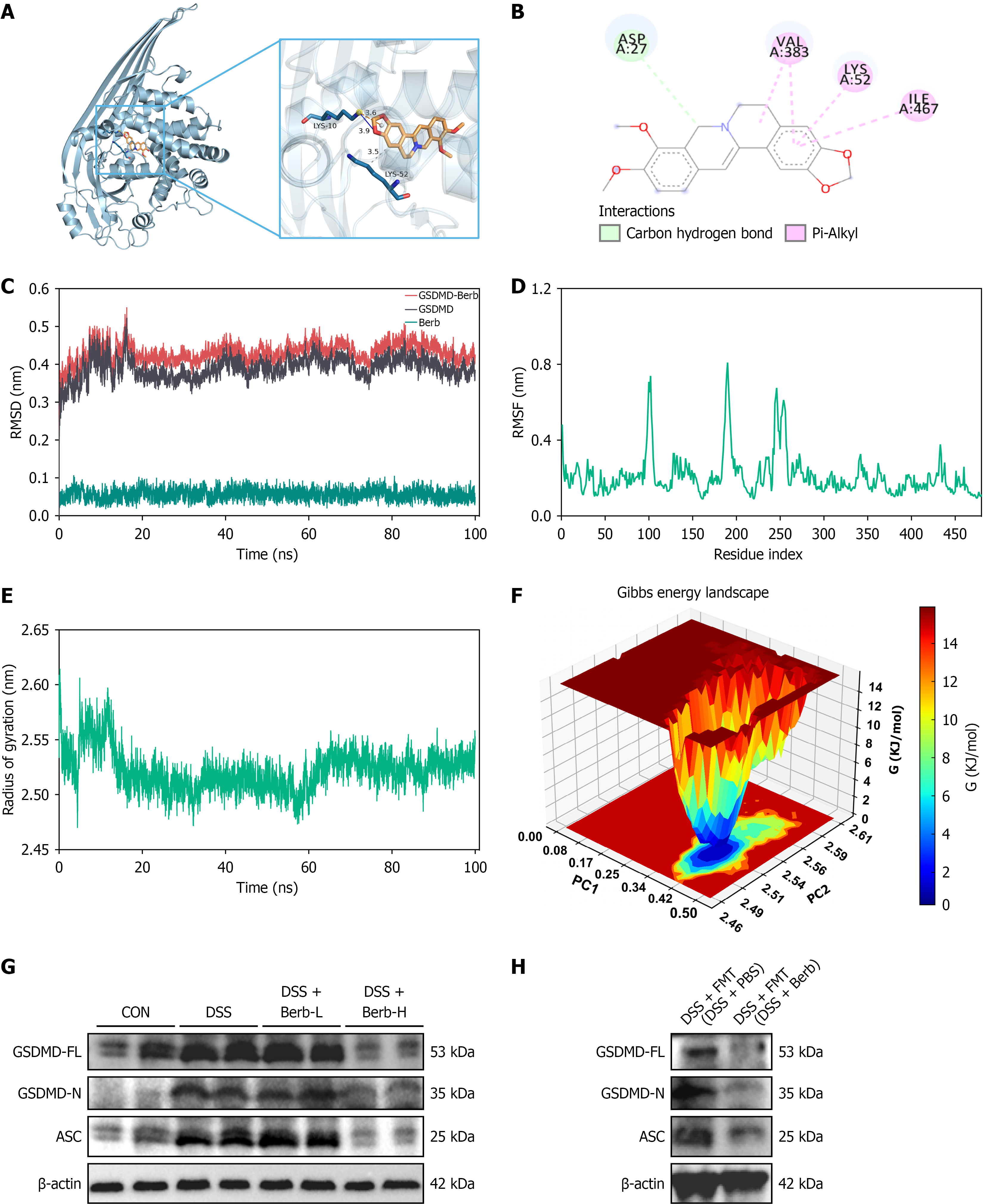
We performed molecular docking analysis to determine the binding affinity of berberine for GSDMD proteins. The optimal docking outcomes are depicted as detailed receptor-ligand visualizations in immersive three-dimensional imagery, with the cartoon representation highlighting the precise locations of the residues involved in the ligand-protein interaction. Molecular docking studies revealed that berberine effectively binds to GSDMD, with a binding energy of -7.3 kcal/mol, suggesting that berberine has a significant affinity for GSDMD (Figure 7A). Dynamic simulation analysis revealed the formation of hydrogen bonds between berberine and the critical amino acid residues VAL383, LYS52, and ILE467 in GSDMD (Figure 7B).
Remained below 0.6 nm throughout the 100 nanoseconds. Conversely, the root-mean-square deviation for the small molecules oscillated around the 18 nanoseconds mark and achieved homeostasis by 20 nanoseconds, indicating that the interaction between the small molecules and the proteins achieved a stable configuration after 20 nanoseconds (Figure 7C). An examination of molecular dynamics simulations revealed that the RMSF of the berberine-GSDMD protein compound system displayed minimal variation throughout the trajectories. Notably, the RMSF values remained below 2.65, confirming the system’s robust stability (Figure 7D). The binding free energy (molecular mechanics/generalized Born surface area) of the last frame, at which the berberine-GSDMD protein compound system achieved homeostasis, was determined, and an energy breakdown analysis was conducted. The data presented in the table reveal a binding free energy of -19.41 kcal/mol, indicating a significant binding affinity. This finding demonstrates that the small-molecule compound forms a robust bond with the protein once a stable state is reached (Figure 7E). The free energy topography showed distinct minimum energy wells, demonstrating a stable binding configuration between berberine and GSDMD (Figure 7F). In addition, western blot and immunohistochemistry analyses revealed that pretreatment with berberine effectively suppressed the DSS-induced upregulation of GSDMD expression (Figure 7G and H). These findings imply that the efficacy of berberine in combating colitis may be partially due to its ability to inhibit GSDMD via direct binding to the protein.
Considering that berberine has the potential to modulate anti-inflammatory responses via GSDMD, we evaluated whether the pharmacological suppression of GSDMD could mitigate the symptoms of colitis. Disulfiram, an inhibitor of GSDMD, effectively blocks oligomerization, pore formation, and the occurrence of pyroptotic cell death. Consequently, we administered disulfiram over a period of seven days after the initiation of berberine therapy in mice with DSS-induced colitis. The administration of disulfiram accelerated the progression of colitis, as indicated by increased manifestations of colitis-associated symptoms, including a more rapid decline in body weight and a higher DAI score (Figure 8). In contrast to the GSDMD inhibitor disulfiram, the NLRP3 inhibitor MCC950 has been shown to protect against UC[26]. These findings underscore the substantial therapeutic potential of targeting NLRP3 and GSDMD in the treatment of UC, collectively suggesting that inhibiting GSDMD represents a promising strategy for IBD management.
Natural products are increasingly being explored as therapeutic options for chronic inflammatory diseases. Berberine, a bioactive alkaloid derived from TCM, has demonstrated substantial efficacy in treating UC despite its limited in vivo bioavailability[28]. Previous studies have shown that berberine alleviates colitis by attenuating intestinal inflammation, modulating the gut microbiota composition, protecting the mucosal barrier, regulating intestinal immune responses, and reducing oxidative stress[29-32]. Nevertheless, the precise targets of berberine and its mechanism of action in the pre
Berberine modulates the gut microbiota-metabolite-immune axis. Numerous studies have demonstrated that the gut microbiota plays a central role in the pathogenesis of UC[33]. Both patients with IBD and experimental colitis models exhibit microbiota dysbiosis characterized by reduced microbial diversity and shifts in beneficial and harmful bacterial populations[34]. Consistent with these findings, our research revealed a significant decrease in gut bacterial diversity, concurrent with an increased prevalence of potentially detrimental bacterial populations, including Proteobacteria, Helicobacter, and Escherichia-Shigella, in mice with colitis. Lactobacillus, a probiotic genus that plays a pivotal role in safe
Microbial metabolites are key modulators of mucosal immunity and host inflammatory responses[35]. In this study, we further investigated FMT, a strategy designed to correct ecological disruption within the gut microbiota and demon
Berberine directly targets GSDMD to alleviate intestinal inflammation. GSDMD, the principal executor of inflammasome-mediated pyroptosis, is essential for maintaining mucosal immune homeostasis and has been implicated in gastrointestinal inflammatory disorders[36]. Previous studies have confirmed that macrophage GSDMD can modulate the cyclic guanosine monophosphate-adenosine monophosphate synthase-mediated inflammatory response, improving the disease progression of DSS-induced UC[11], suggesting that GSDMD represents a promising molecular target for innovative IBD therapies. In our study, we elucidated the direct targeting effect of berberine on GSDMD through multidimensional experiments. In vivo experiments demonstrated that berberine effectively reversed the inhibitory effect of DSS on GSDMD protein expression, restoring its normal regulatory levels. Molecular docking experiments revealed that berberine specifically binds to a key site in the N-terminal pore-forming domain of GSDMD, forming stable binding pat
The therapeutic efficacy of berberine arises from synergistic interactions between microbiota remodeling and direct suppression of GSDMD activation. On the one hand, berberine reshapes the gut microbiota composition by promoting the growth and proliferation of beneficial bacteria such as Lactobacillus and Oscillibacter. These bacteria produce anti-inflammatory metabolites, including short-chain fatty acids, and inhibit the colonization of pathogenic bacteria, thereby restoring microbial balance and reducing excessive immune activation. On the other hand, berberine directly binds to GSDMD and inhibits its activation, blocking inflammasome-mediated pyroptosis, which alleviates damage to intestinal epithelial cells and excessive inflammatory responses. These two pathways are dynamically interconnected: The gut microbiota can influence the activation threshold of GSDMD by regulating the intestinal immune microenvironment, while GSDMD-mediated inflammatory responses can, reciprocally, affect the colonization efficiency and metabolic phenotypes of gut microbes. This synergistic effect significantly enhances the overall ability of berberine to alleviate inflammation and improve intestinal function, positioning it as a highly promising candidate for IBD treatment.
Multidimensional research has provided new theoretical support and practical directions for elucidating the me
Previous studies have demonstrated that berberine exerts anti-inflammatory effects by targeting molecules such as the NEK7-NLRP3 complex and IRGM1. However, the regulatory relationship between GSDMD and these known targets remains to be clarified. Based on the findings, the hierarchical characteristics of the inflammatory regulatory network, and the mechanisms of established targets, we propose that the multitarget action of berberine is centered on “synergistic regulation”, with the following core logic. NEK7 is a key regulator of NLRP3 inflammasome assembly, and NEK7-mediated activation of the NLRP3/caspase-1 pathway is a critical upstream event for GSDMD cleavage and activation. By simultaneously targeting the NEK7-NLRP3 complex and GSDMD, berberine can dually block the pyroptosis pathway and establish a highly efficient cascade-inhibitory effect. IRGM1 mainly attenuates inflammatory injury by promoting autophagic clearance of intracellular pathogens, whereas GSDMD-mediated pyroptosis amplifies immune responses by releasing inflammatory cytokines. Moreover, berberine improves the gut microbiota composition by targeting formate-tetrahydrofolate ligase, and microbiota-derived metabolites may, in turn, indirectly inhibit GSDMD activation. The simultaneous regulation of these targets by berberine achieves functional complementarity, collectively maintaining the homeostasis of the intestinal immune microenvironment.
Despite the promising preclinical potential of berberine, several bottlenecks still impede its clinical translation. The low bioavailability of berberine leads to insufficient drug concentrations at colonic lesion sites, limiting its ability to fully exert targeted effects on GSDMD, NEK7 and other molecules. Nonspecific systemic distribution may induce adverse effects such as gastrointestinal irritation, thereby compromising long-term treatment adherence. In addition, conventional single-component formulations are inadequate for meeting the requirements of multitarget synergy and fail to achieve precise colonic enrichment and sustained release of berberine. Based on the above challenges, we propose that colon-targeted formulations represent a crucial optimization direction, involving the following strategies: (1) Development of colon-targeted delivery systems, such as pH-sensitive microspheres, probiotic-coated nanoparticles, or pectin-chitosan composite carriers, to achieve targeted release of berberine in the colon while reducing systemic exposure; (2) Design of berberine-based combination formulations according to multitarget synergistic mechanisms, for example, coadministration with mesalazine or probiotics, to further enhance therapeutic efficacy; and (3) Improvement of water solubility and bioavailability through structural modification or formulation optimization, thereby increasing target binding efficiency. Previous studies have shown that PEGylated liposomes loaded with berberine can significantly modulate the NLRP3/caspase-1/GSDMD axis[40], suggesting the translational potential of this delivery platform. These optimization strategies are expected to specifically address the current limitations in the clinical application of berberine and provide critical support for advancing it from experimental evidence to clinical practice.
Nevertheless, our research has notable limitations that merit attention. First, the methodology employed in our anti
This study utilized a DSS-induced colitis mouse model and employed a range of experiments, including dose-gradient validation, FMT microbiota intervention, and GSDMD inhibitor treatments, combined with 16S sequencing, molecular docking, and western blot techniques. These approaches elucidate the synergistic therapeutic mechanism of berberine, which targets GSDMD to inhibit pyroptosis and modulate the gut microbiota-immune axis. These findings not only expand the theoretical framework for the actions of active compounds in TCM but also provide new candidate drugs and intervention strategies for the targeted treatment of UC.
| 1. | Wang M, Fu R, Xu D, Chen Y, Yue S, Zhang S, Tang Y. Traditional Chinese Medicine: A promising strategy to regulate the imbalance of bacterial flora, impaired intestinal barrier and immune function attributed to ulcerative colitis through intestinal microecology. J Ethnopharmacol. 2024;318:116879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 90] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 2. | Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023;402:571-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1251] [Cited by in RCA: 1082] [Article Influence: 360.7] [Reference Citation Analysis (5)] |
| 3. | Zeng W, He D, Xing Y, Liu J, Su N, Zhang C, Wang Y, Xing X. Internal connections between dietary intake and gut microbiota homeostasis in disease progression of ulcerative colitis: a review. Food Sci Hum Wellness. 2021;10:119-130. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 4. | Barreiro-de Acosta M, Molero A, Artime E, Díaz-Cerezo S, Lizán L, de Paz HD, Martín-Arranz MD. Epidemiological, Clinical, Patient-Reported and Economic Burden of Inflammatory Bowel Disease (Ulcerative colitis and Crohn's disease) in Spain: A Systematic Review. Adv Ther. 2023;40:1975-2014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (1)] |
| 5. | Martinon F, Mayor A, Tschopp J. The inflammasomes: guardians of the body. Annu Rev Immunol. 2009;27:229-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1989] [Cited by in RCA: 1845] [Article Influence: 108.5] [Reference Citation Analysis (1)] |
| 6. | Yao X, Zhang C, Xing Y, Xue G, Zhang Q, Pan F, Wu G, Hu Y, Guo Q, Lu A, Zhang X, Zhou R, Tian Z, Zeng B, Wei H, Strober W, Zhao L, Meng G. Remodelling of the gut microbiota by hyperactive NLRP3 induces regulatory T cells to maintain homeostasis. Nat Commun. 2017;8:1896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 169] [Article Influence: 18.8] [Reference Citation Analysis (1)] |
| 7. | Shi J, Zhao Y, Wang K, Shi X, Wang Y, Huang H, Zhuang Y, Cai T, Wang F, Shao F. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature. 2015;526:660-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5756] [Cited by in RCA: 5228] [Article Influence: 475.3] [Reference Citation Analysis (3)] |
| 8. | Hu Y, Wang B, Li S, Yang S. Pyroptosis, and its Role in Central Nervous System Disease. J Mol Biol. 2022;434:167379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 74] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 9. | He K, Wan T, Wang D, Hu J, Zhou T, Tao W, Wei Z, Lu Q, Zhou R, Tian Z, Flavell RA, Zhu S. Gasdermin D licenses MHCII induction to maintain food tolerance in small intestine. Cell. 2023;186:3033-3048.e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 10. | Hu Y, Jiang Y, Li S, Ma X, Chen M, Yang R, Wen S, Moynagh PN, Wang B, Hu G, Yang S. The Gasdermin D N-terminal fragment acts as a negative feedback system to inhibit inflammasome-mediated activation of Caspase-1/11. Proc Natl Acad Sci U S A. 2022;119:e2210809119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (2)] |
| 11. | Ma C, Yang D, Wang B, Wu C, Wu Y, Li S, Liu X, Lassen K, Dai L, Yang S. Gasdermin D in macrophages restrains colitis by controlling cGAS-mediated inflammation. Sci Adv. 2020;6:eaaz6717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 143] [Article Influence: 23.8] [Reference Citation Analysis (1)] |
| 12. | Zhang J, Yu Q, Jiang D, Yu K, Yu W, Chi Z, Chen S, Li M, Yang D, Wang Z, Xu T, Guo X, Zhang K, Fang H, Ye Q, He Y, Zhang X, Wang D. Epithelial Gasdermin D shapes the host-microbial interface by driving mucus layer formation. Sci Immunol. 2022;7:eabk2092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 92] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 13. | Li H, Pu J, Yang D, Liu L, Hu Y, Yang S, Wang B. GSDMD protects intestinal epithelial cells against bacterial infections through its N-terminal activity affecting intestinal immune homeostasis. J Biomed Res. 2024;38:585-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 14. | Wu H, Li YL, Wang Y, Wang YG, Hong JH, Pang MM, Liu PM, Yang JJ. Anemoside B4 alleviates ulcerative colitis by attenuating intestinal oxidative stress and NLRP3 inflammasome via activating aryl hydrocarbon receptor through remodeling the gut microbiome and metabolites. Redox Biol. 2025;85:103746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 27.0] [Reference Citation Analysis (3)] |
| 15. | Ling Y, Ding L, Tian Z, Pei L, Wu E. YINDARA-4 relieves visceral hypersensitivity in irritable bowel syndrome rats via regulation of gut microbiota and serotonin levels. Acupunct Herb Med. 2022;2:274-283. [DOI] [Full Text] |
| 16. | Yang Y, Li W, Sun K, Sun S, Zhang Y, Chen L, Ni Y, Hou M, Xu Z, Chen L, Ji M. Berberine ameliorates dextran sulfate sodium -induced colitis through tuft cells and bitter taste signalling. BMC Biol. 2024;22:280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 17. | Huang YQ, Liu JL, Chen GX, Shen DT, Zhu W, Chen XL, Liu FB, Hou QK. Berberine Enhances Intestinal Mucosal Barrier Function by Promoting Vitamin D Receptor Activity. Chin J Integr Med. 2024;30:143-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 18. | Halfvarson J, Brislawn CJ, Lamendella R, Vázquez-Baeza Y, Walters WA, Bramer LM, D'Amato M, Bonfiglio F, McDonald D, Gonzalez A, McClure EE, Dunklebarger MF, Knight R, Jansson JK. Dynamics of the human gut microbiome in inflammatory bowel disease. Nat Microbiol. 2017;2:17004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1052] [Cited by in RCA: 883] [Article Influence: 98.1] [Reference Citation Analysis (6)] |
| 19. | Alizadeh M, Wong U, Siaton BC, France MT, Patil SA, George L, Hudhud D, Motwani K, Scott WH, Raufman JP, von Rosenvinge EC, Cross RK, Ravel J. The intestinal mucosa-associated microbiota in IBD-associated arthritis displays lower relative abundance of Roseburia intestinalis. Gut Microbes. 2025;17:2505114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 20. | Guo M, Li B, Li H, Chen Y, Yuan Q, Shui M, Zhou H, Hao W, Wang S. Colon-Targeted Natural Polysaccharide-Berberine Armored Hydrogel for the Treatment of Colitis. Adv Healthc Mater. 2025;14:e2404908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Ruan H, Zhan YY, Hou J, Xu B, Chen B, Tian Y, Wu D, Zhao Y, Zhang Y, Chen X, Mi P, Zhang L, Zhang S, Wang X, Cao H, Zhang W, Wang H, Li H, Su Y, Zhang XK, Hu T. Berberine binds RXRα to suppress β-catenin signaling in colon cancer cells. Oncogene. 2017;36:6906-6918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 117] [Article Influence: 13.0] [Reference Citation Analysis (2)] |
| 22. | Zeng Q, Deng H, Li Y, Fan T, Liu Y, Tang S, Wei W, Liu X, Guo X, Jiang J, Wang Y, Song D. Berberine Directly Targets the NEK7 Protein to Block the NEK7-NLRP3 Interaction and Exert Anti-inflammatory Activity. J Med Chem. 2021;64:768-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 23. | Wei W, Zeng Q, Wang Y, Guo X, Fan T, Li Y, Deng H, Zhao L, Zhang X, Liu Y, Shi Y, Zhu J, Ma X, Wang Y, Jiang J, Song D. Discovery and identification of EIF2AK2 as a direct key target of berberine for anti-inflammatory effects. Acta Pharm Sin B. 2023;13:2138-2151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 64] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 24. | Yan J, Fang C, Yang G, Li J, Liu Y, Zhang L, Yang P, Fang J, Gu Y, Zhang Y, Jiang W. Identification of FtfL as a novel target of berberine in intestinal bacteria. BMC Biol. 2023;21:280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 25. | Meng G, Li P, Du X, Feng X, Qiu F. Berberine alleviates ulcerative colitis by inhibiting inflammation through targeting IRGM1. Phytomedicine. 2024;133:155909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 40] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 26. | Chassaing B, Koren O, Goodrich JK, Poole AC, Srinivasan S, Ley RE, Gewirtz AT. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature. 2015;519:92-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1758] [Cited by in RCA: 1463] [Article Influence: 133.0] [Reference Citation Analysis (6)] |
| 27. | Ji J, Ge X, Chen Y, Zhu B, Wu Q, Zhang J, Shan J, Cheng H, Shi L. Daphnetin ameliorates experimental colitis by modulating microbiota composition and T(reg)/T(h)17 balance. FASEB J. 2019;33:9308-9322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 28. | Sun S, Yang Y, Xiong R, Ni Y, Ma X, Hou M, Chen L, Xu Z, Chen L, Ji M. Oral berberine ameliorates high-fat diet-induced obesity by activating TAS2Rs in tuft and endocrine cells in the gut. Life Sci. 2022;311:121141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 29. | Li D, Yang C, Zhu JZ, Lopez E, Zhang T, Tong Q, Peng C, Lin LG. Berberine remodels adipose tissue to attenuate metabolic disorders by activating sirtuin 3. Acta Pharmacol Sin. 2022;43:1285-1298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 30. | Li H, Fan C, Lu H, Feng C, He P, Yang X, Xiang C, Zuo J, Tang W. Protective role of berberine on ulcerative colitis through modulating enteric glial cells-intestinal epithelial cells-immune cells interactions. Acta Pharm Sin B. 2020;10:447-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 31. | Jing W, Dong S, Luo X, Liu J, Wei B, Du W, Yang L, Luo H, Wang Y, Wang S, Lu H. Berberine improves colitis by triggering AhR activation by microbial tryptophan catabolites. Pharmacol Res. 2021;164:105358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 149] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 32. | Zhang P, Chen J, Ming Y, Niu Y. Probiotics treatment ameliorated mycophenolic acid-induced colitis by enhancing intestinal barrier function and improving intestinal microbiota dysbiosis in mice. Front Microbiol. 2023;14:1153188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 33. | Zhu W, Winter MG, Byndloss MX, Spiga L, Duerkop BA, Hughes ER, Büttner L, de Lima Romão E, Behrendt CL, Lopez CA, Sifuentes-Dominguez L, Huff-Hardy K, Wilson RP, Gillis CC, Tükel Ç, Koh AY, Burstein E, Hooper LV, Bäumler AJ, Winter SE. Precision editing of the gut microbiota ameliorates colitis. Nature. 2018;553:208-211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 511] [Cited by in RCA: 440] [Article Influence: 55.0] [Reference Citation Analysis (6)] |
| 34. | Wang J, Zhu N, Su X, Gao Y, Yang R. Gut-Microbiota-Derived Metabolites Maintain Gut and Systemic Immune Homeostasis. Cells. 2023;12:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 445] [Cited by in RCA: 386] [Article Influence: 128.7] [Reference Citation Analysis (1)] |
| 35. | Schwarzer R, Jiao H, Wachsmuth L, Tresch A, Pasparakis M. FADD and Caspase-8 Regulate Gut Homeostasis and Inflammation by Controlling MLKL- and GSDMD-Mediated Death of Intestinal Epithelial Cells. Immunity. 2020;52:978-993.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 225] [Article Influence: 37.5] [Reference Citation Analysis (1)] |
| 36. | Wang J, Tan H, Ye Z, Weng S, Shi Y, Xu J, Liu H, Li J, Huang L, Zhai L, Luo H, Lin Z, Zhong C, Tang J, Wang Z, Zhang H, Zhang B, Huang C. Gut microbial Nordihydroguaiaretic acid suppresses macrophage pyroptosis to regulate epithelial homeostasis and inflammation. Gut Microbes. 2025;17:2518338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 37. | Buldukoglu OC, Ocal S, Akca S, Atar GE, Kaya B, Isik MD, Koca O, Harmandar FA, Cekin Y, Cekin AH. A novel inflammatory biomarker in assessing disease activity in ulcerative colitis: gasdermin D. BMC Gastroenterol. 2025;25:418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (3)] |
| 38. | Li Y, Guo B. GSDMD-mediated pyroptosis: molecular mechanisms, diseases and therapeutic targets. Mol Biomed. 2025;6:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 37] [Reference Citation Analysis (1)] |
| 39. | Yang B, Qi Z, Armas Diaz Y, Cassotta M, Grosso G, Cianciosi D, Zhang D, Zou X, Quiles JL, Battino M, Giampieri F. Pyroptosis: A Novel Therapeutic Target for Bioactive Compounds in Human Disease Treatment? A Narrative Review. Nutrients. 2025;17:461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 40. | El Gazzar WB, Farag AA, Samir M, Bayoumi H, Youssef HS, Marei YM, Mohamed SK, Marei AM, Abdelfatah RM, Mahmoud MM, Aboelkomsan EAF, Khalfallah EKM, Anwer HM. Berberine chloride loaded nano-PEGylated liposomes attenuates imidacloprid-induced neurotoxicity by inhibiting NLRP3/Caspase-1/GSDMD-mediated pyroptosis. Biofactors. 2025;51:e2107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |