Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.114563
Revised: November 14, 2025
Accepted: February 2, 2026
Published online: April 7, 2026
Processing time: 184 Days and 18.8 Hours
Invasive aspergillosis (IA) is typically considered an opportunistic pulmonary in
We report a case of a 48-year-old male, who previously underwent thyroidectomy for thyroid cancer but lacked the typical risk factors for IA. He presented to our hospital with refractory diarrhea accompanied by a 6 kg weight loss. Initial colo
For immunocompetent patients with refractory diarrhea, PA should be considered and mNGS-guided antifungals should be initiated.
Core Tip: This case report describes an unusual presentation of refractory diarrhea in a patient who was immunocompetent and had none of the usual risk factors. Despite comprehensive evaluations including endoscopy and histopathology, the etiology remained undetermined. Metagenomic next-generation sequencing (mNGS) identified Aspergillus fumigatus, guiding successful targeted treatment. This highlights the necessity of considering primary intestinal aspergillosis (PA) in certain patients and demonstrates the diagnostic value of mNGS for rare fungal enteritis. Although publication bias may exist, accumulating case reports support considering PA when diagnosing unexplained diarrhea. Furthermore, the use of mNGS is justified when conventional diagnostic methods are inconclusive.
- Citation: Guo T, Wang X, Wang SY, Lin TF. Isolated colonic aspergillosis in an immunocompetent individual diagnosed by metagenomic next-generation sequencing: A case report. World J Gastroenterol 2026; 32(13): 114563
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/114563.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.114563
Invasive aspergillosis (IA) is a life-threatening fungal infection caused by environmental fungi, predominantly Aspergillus fumigatus (A. fumigatus), which are ubiquitous in soil and dust. The incidence of IA has increased with the rise of organ transplantation, chemotherapy, and use of high-dose corticosteroids or immunosuppressants. Infections with other species, such as Aspergillus niger and Aspergillus flavus, are also becoming more common[1,2]. In immunocompromised individuals, airborne Aspergillus species can colonize the sinuses or lungs, leading to invasive sinonasal or pulmonary disease. Intestinal aspergillosis usually results from hematogenous dissemination from a primary pulmonary or alternative fungal focus, specifically affecting a compromised intestinal mucosal barrier. Direct colonization of the compromised intestinal mucosa via ingestion of contaminated food or environmental spores is a rare occurrence[3]. Primary intestinal aspergillosis (PA) in immunocompetent individuals without traditional risk factors is exceptionally rare. Its infrequent occurrence means that is rarely considered in the differential diagnosis of abdominal pain, distension, and diarrhea, rendering clinical diagnosis extremely challenging[4].
The current case study illustrates that PA should be considered in immunocompetent individuals with persistent diarrhea, even without classic risk factors. It also suggests that metagenomic next-generation sequencing (mNGS) is a useful diagnostic tool for cases of unexplained chronic diarrhea. These findings offer new insights for clinical diagnosis and treatment.
A 48-year-old male was admitted to our department due to persistent diarrhea for more than 50 days accompanied by a weight loss of 6 kg.
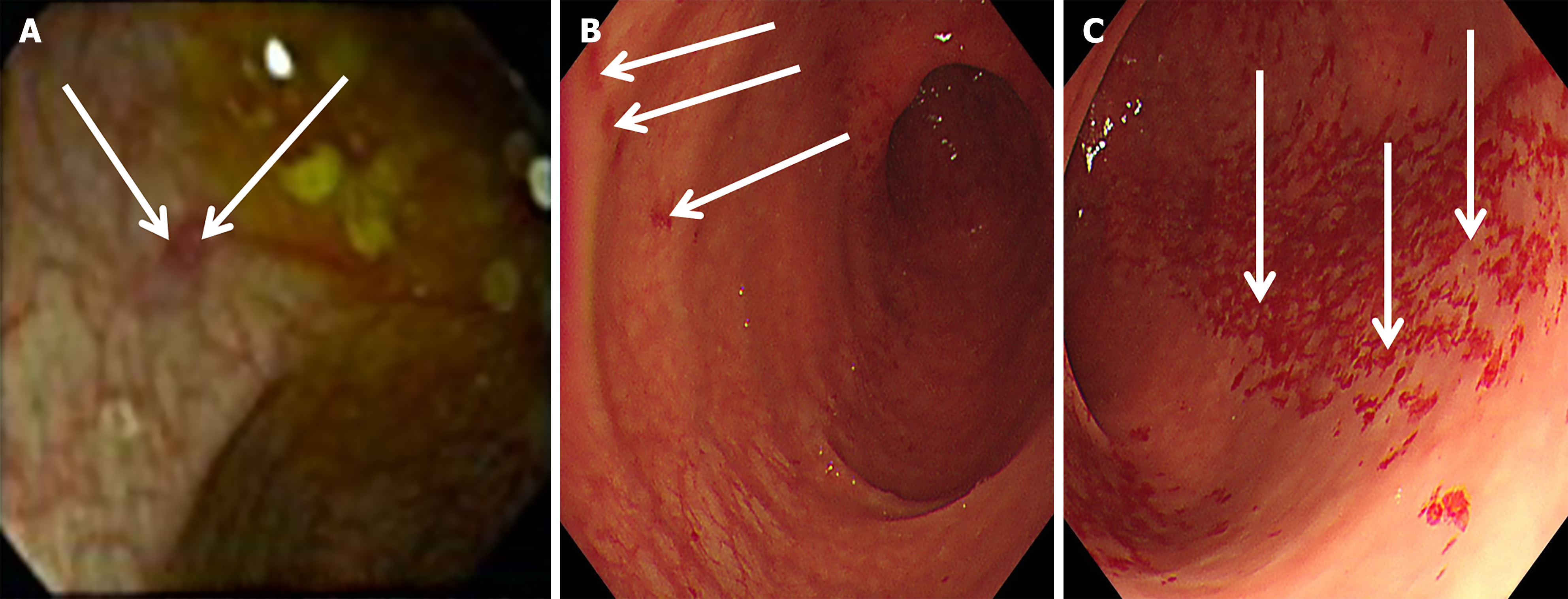
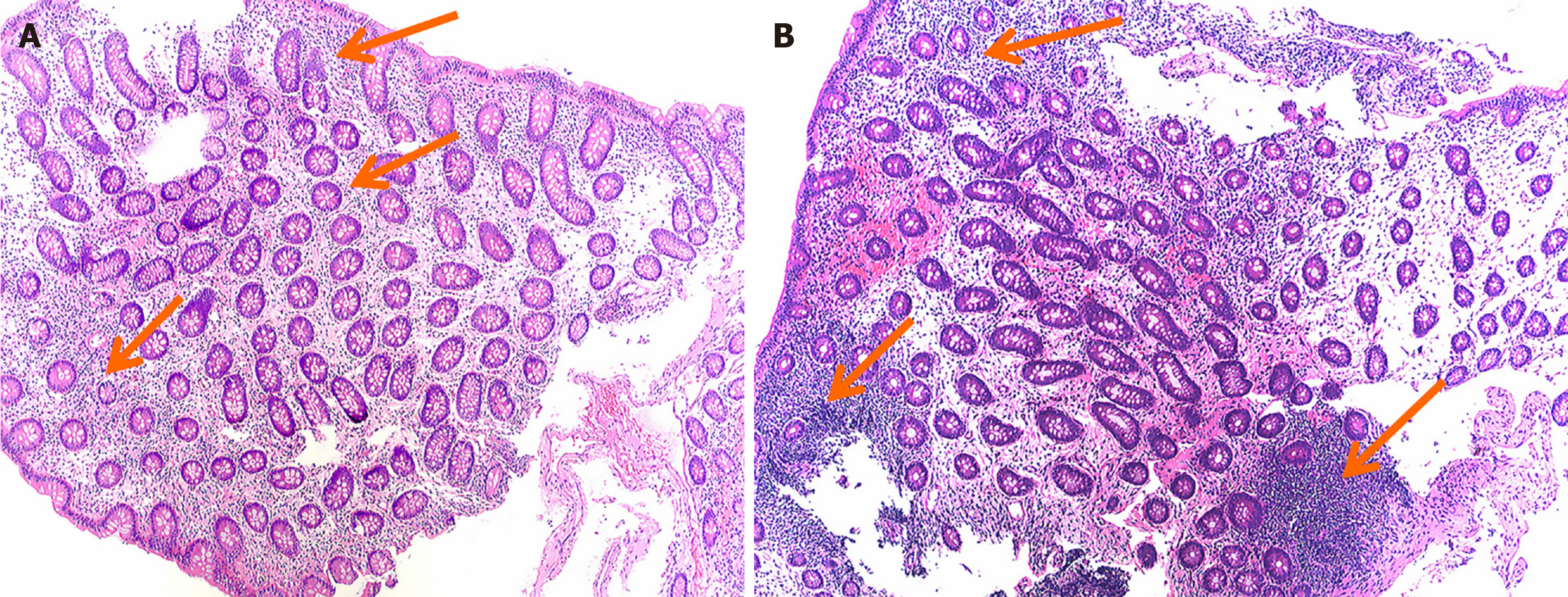
He had been hospitalized in our unit earlier in the disease course for right upper abdominal pain and watery diarrhea (5-7 episodes per day) lasting 1 week. Routine laboratory tests at that time showed no abnormalities; however, colonoscopy revealed erosions and multiple polyps in the transverse colon. Endoscopic and histopathological findings of transverse colon biopsies are presented in the Endoscopy and Pathology section. Consequently, the patient was treated symptomatically and underwent endoscopic polypectomy. He was discharged after partial relief of diarrhea, and was prescribed loperamide, Bacillus licheniformis, and esomeprazole for ongoing abdominal pain and loose stools. Despite this regimen, his diarrhea not only persisted but worsened to over 10 episodes daily, with a total weight loss of 6 kg occurring over more than 2 months. Following unsuccessful management at a local hospital, he was readmitted to our department. Conventional therapy for diarrhea and abdominal pain was provided, and the patient was treated with empirical quinolone antibiotics for more than 3 days; however, his diarrheal symptoms showed no signs of improvement. Despite a repeat colonoscopy and biopsy for histopathological evaluation, the underlying reason for the diarrhea could not be identified. Furthermore, during hospitalization, comprehensive workup confirmed the absence of an underlying immunodeficiency.
The patient had undergone a right radical thyroidectomy 2 years prior to treat papillary thyroid carcinoma and required daily levothyroxine sodium to treat post-operative hypothyroidism. The patient did not have the classic risk factors for IA, including neutropenia, active cancer, high-dose chemotherapy, a history of IA, or a history of allogeneic tran
The patient, a seafood market employee, had no history of unsanitary dietary exposure, no similar symptoms among close contacts, and no recent travel to endemic regions. Additionally, he had no history of smoking or alcohol con
Upon admission, the patient’s vital signs were within normal ranges, and his cardiopulmonary exam was unremarkable. The abdomen was soft, non-distended, and mildly tender to deep palpation of the middle and lower quadrants without rebound tenderness.
Routine laboratory findings were largely unremarkable (Table 1), including complete blood count, procalcitonin, C-reactive protein, liver and renal function markers, tumor markers, immune-related antibodies, and negative human immunodeficiency virus (HIV). A single electrolyte panel showed hypokalemia (potassium 3.47 mmol/L; normal range: 3.5-5.2 mmol/L). The routine stool test showed positivity for white blood cells, suggestive of an inflammatory compo
| Laboratory testing | Results | Reference values | Units |
| Inflammatory markers | |||
| CRP | < 10.0 | 0-10 | mg/mL |
| PCT | < 0.05 | < 0.5 | ng/mL |
| Complete blood count | |||
| WBC | 4.76 | 3.5-9.5 | × 109/L |
| HB | 150 | 130-175 | g/L |
| PLT | 206 | 125-350 | × 109/L |
| NEUT | 2.31 | 1.8-6.3 | × 109/L |
| LYMPH | 1.94 | 1.1-3.2 | × 109/L |
| EO | 0.06 | 0.02-0.52 | × 109/L |
| BASO | 0.03 | 0-0.06 | × 109/L |
| Biochemistry | |||
| ALT | 10.5 | 7-50 | U/L |
| AST | 12.1↓ | 13-40 | U/L |
| Albumin | 48.1 | 40-55 | g/L |
| Creatinine | 62.4 | 53-115 | μmol/L |
| Sodium | 140 | 137-147 | mmol/L |
| Potassium | 3.47↓ | 3.5-5.3 | mmol/L |
| Stool examination | |||
| Parasite | Negative | Negative | - |
| WBC | Positive | 0-1 | - |
| Fungal culture | Negative | Negative | - |
| FOBT | Negative | Negative | - |
| Fungal serology | |||
| (1,3)-β-D-glucan | < 37.55 | < 70 | pg/mL |
| Galactomannan antigen | 0.12 | ≤ 0.5 | S/CO |
| Tumor markers | |||
| CEA | 3.37 | < 5.0 | ng/mL |
| AFP | 1 | < 7 | ng/mL |
| Tuberculosis | |||
| T-SPOT.TB | Positive | Negative | - |
| γ-IFN | 378.0 ↑ | < 14 | pg/mL |
| HIV Ag-Ab | Negative | < 1.00 | AU/mL |
| Auto-antibodies | |||
| ANA | Negative | Negative | - |
| dsDNA | Negative | Negative | - |
| ANAs | Negative | Negative | - |
| Voriconazole trough level | 2.8 | 1-5.5 | μg/mL |
Chest and upper abdominal computed tomography scans were unremarkable. Two sequential colonoscopies de
Because previous examinations failed to identify a cause of the patient's diarrhea, we subsequently analyzed mucosal biopsy tissue from the transverse colon using mNGS during the second colonoscopy. On day 69 of diarrhea, mNGS identified 2958 sequences of Aspergillus (a relatively high abundance), with 383 identified as A. fumigatus with 99% confidence, implicating this species as the causative pathogen. The sequencing results for the strain are available through the National Center for Biotechnology Information (NCBI) repository, registered under the accession number PRJNA1333823 (https://www.ncbi.nlm.nih.gov/sra/PRJNA1333823).
Based on the patient’s comprehensive profile (detailed above), we suspected that he had developed PA.
A biopsy of the transverse colon mucosa revealed a high abundance of A. fumigatus by mNGS. In line with the patient’s clinical profile and the Australasian Antifungal Guidelines Steering Committee’s Consensus guidelines for the diagnosis and management of invasive aspergillosis, 2021[5], we initiated presumptive antifungal therapy with oral voriconazole. The dosing regimen consisted of a loading dose of 6 mg/kg every 12 hours on day 1, followed by a maintenance dose of 4 mg/kg every 12 hours. Trough voriconazole levels were monitored and remained within the recommended therapeutic range at 2.8 μg/mL[6]. The complete cessation of diarrhea within 48 hours strongly indicated A. fumigatus as the causative pathogen rather than a colonizer or contaminant. The patient was discharged on oral voriconazole (4 mg/kg every 12 hours) to ensure continued therapeutic efficacy.
Voriconazole was discontinued 1 week post-discharge during an outpatient visit, as the patient’s abdominal discomfort had resolved and blood tests were normal. Six weeks after cessation of medication, there was no recurrence of abdominal pain or diarrhea, and nutritional status had recovered. Trough voriconazole level at follow-up was 0.1 μg/mL.
The incidence of IA is increasing with the wider use of small molecule-targeted inhibitors and immunotherapies[7]; however, its early diagnosis remains challenging. Although current guidelines regard histopathological examination and culture as the diagnostic gold standard[5,8], histopathology is invasive and obtaining suitable specimens can be difficult. Blood and tissue cultures are time-consuming, lack sensitivity, and offer limited value for early diagnosis. Because intestinal infections have non-specific symptoms and lack distinct imaging findings, fungal stains like PAS or GMS are not routinely ordered during initial histopathological evaluation, which prioritize common conditions such as IBD, intestinal tuberculosis, and polyps. Thus, diagnosing PA in immunocompetent individuals who lack traditional risk factors is challenging. Of note, the diagnosis often relies on evidence from non-culture-based techniques, coupled with a positive response to diagnostic antifungal therapy and absence of relapse during follow-up.
mNGS has emerged as a powerful technology over the past decade, showing promise for closing diagnostic gaps in invasive fungal diseases (IFDs)[9], although it has yet to be incorporated into major IFD management guidelines. To date, only 1 case of intestinal aspergillosis diagnosed by mNGS has been reported[10]. The use of mNGS was a critical and beneficial tool for our case. The diagnostic process was complicated due to several confounding factors. First, the patient was immunocompetent, as evidenced by a normal peripheral lymphocyte count, negative HIV serology, absence of autoantibodies, and no relevant medical or pharmacological history. Although specific immunoglobulin and lymphocyte subset analyses were not tested, the collective clinical data showed no indication of an underlying immunodeficiency. Second, the case presented with atypical symptoms, a lack of relevant imaging findings, and negative results from both culture-based and non-culture-based assays for fungal growth and biomarkers (e.g., serum galactomannan testing). Ultimately, mNGS detected a high level of A. fumigatus, guiding the correct treatment. This supports the conclusion by Ye et al[10] that mNGS should be considered a diagnostic option for IA and other rare IFDs.
Beyond the diagnostic challenge, the pathogenesis occurring in this immunocompetent individual (host) was intriguing. We hypothesize that chronic occupational exposure to A. fumigatus conidia, combined with pre-existing mucosal damage, triggered a localized immunopathological response. Carbohydrates in the fungal cell wall are known to act as intrinsic adjuvants that drive a Th2-biased response[11]. Furthermore, repeated exposure has been shown to induce a complex co-evolution of Th1, Th2, and Th17 responses[12-14]. Thus, the progressive colonic inflammation in our patient had likely resulted from such a dysregulated local immune reaction: An "immune misdirection", whereby the host's response, rather than a fungal invasion, became the primary driver of tissue injury. Nonetheless, the lack of specific immunologic profiling for our case remains a limitation that will need to be included in future investigations of other similar cases, if possible.
To systematically contextualize the rarity and distinctive features of our case, we conducted a structured literature review. We searched PubMed for English-language case reports and series published up to August 2025 using the following query: ("intestinal aspergillosis" OR "colonic aspergillosis" OR "gastrointestinal aspergillosis") AND ("immunocompetent" OR "non-immunocompromised"). We excluded cases with classic immunosuppression (e.g., hematologic malignancy, solid organ transplantation, advanced HIV/acquired immune deficiency syndrome, or prolonged high-dose corticosteroids). This targeted search and screening process identified only two prior cases that met these criteria fully[3,15], unequivocally confirming the exceptional rarity of PA in immunocompetent hosts.
Cha et al[3] reported the first case of lower gastrointestinal aspergillosis in an immunocompetent individual lacking typical risk factors. The authors theorized that the patient’s immune system may have been compromised by their stay in the intensive care unit, use of piperacillin-sulbactam, and mechanical ventilation. They compared these factors to the previously reported non-classic risk factors identified for invasive pulmonary aspergillosis found in critically ill patients prior to 2015. They proposed that these factors, along with hyperglycemia in diabetes, can impair immune responses and contribute to invasive gastrointestinal aspergillosis.
Díaz Alcázar et al[15] also reported the case of a 72-year-old immunocompetent patient who rapidly developed systemic aspergillosis affecting the nervous system, responded poorly to antifungal therapy, and ultimately died. The patient in our case was middle-aged. Despite refractory diarrhea lasting more than 60 days and endoscopic evidence of progressive intestinal involvement, his disease remained confined to the lower gastrointestinal tract. He also lacked any of the non-classic risk factors identified by Díaz Alcázar et al[15] or a history of diabetes. We hypothesize that younger age explains his milder symptoms and localized disease compared to the case reported by Díaz Alcázar et al[15]. We speculate that long-term occupational exposure in a seafood market, an environment conducive to fungal proliferation, represents a non-traditional risk factor for this case. However, this hypothesis requires further validation through more reports or experimental investigations.
A comprehensive 2021 literature review by Yelika et al[4] established that this condition almost invariably occurs as part of a disseminated infection in severely immunocompromised hosts. Furthermore, cases reported since 2021 have continued to adhere to this pattern[10,16-20]. In contrast, our case represents a definitive exception to this established clinical paradigm, being characterized by the absence of any underlying immunodeficiency or evidence of extraintestinal dissemination - underscoring its unique status as a localized primary gastrointestinal infection in an immunocompetent individual.
The management of IA rests on three pillars: Antifungal therapy (prophylactic, empirical, or targeted), surgical intervention (e.g., drainage), and reversal of predisposing conditions. For patients without immunosuppression, suc
Although our patient did not develop disseminated disease or severe sequelae, it is important to note that prolonged diarrhea can lead to shock, electrolyte imbalances, acid-base disturbances, and even life-threatening complications. Therefore, we identified several periods for prescribing empirical antifungal therapy before making a definitive diagnosis. First, following the early initiation of symptom-based treatment and resolution of potentially confusing coexisting conditions (e.g., intestinal polyp removal), the patient’s diarrhea briefly improved, only to then undergo dramatic exacerbation. Second, another key consideration was the patient's long-term occupational exposure in a fungus-prone environment and endoscopic evidence of mucosal barrier damage. Third, there was a lack of improvement of diarrhea after more than 3 days of empirical antibiotic therapy. Even though such cases are exceedingly rare for phy
For our case, the duration of antifungal therapy to treat IA was notably shorter than the courses reported for other cases in the existing literature[3,10], where treatment was continued until complete resolution of symptoms and normalization of laboratory parameters. Currently, no definitive guidelines are established for the optimal treatment duration and management is individualized based on the patient’s clinical response and immune status[5,10]. In our patient, who was immunocompetent, the abdominal symptoms resolved completely within 48 hours and did not recur, eliminating the need for a prolonged therapy aiming to support immune recovery or manage comorbidities. Thus, the shorter course was deemed sufficient. Regardless, future case studies are needed to establish more definitive evidence for treatment duration.
The preferred first-line treatment for the vast majority of patients with IA is voriconazole (intravenous or oral), being in accordance with guidelines[5,23-25]. Although oral voriconazole demonstrated remarkable efficacy in our case, alternative strategies may be considered for others in similar clinical scenarios. For example, patients with severe disease, oral intolerance, or concerns regarding drug absorption, should be provided the option of intravenous formulations for induction therapy; of note, intravenous voriconazole remains the primary recommended agent[5]. Alternative intra
This case highlights that A. fumigatus can cause persistent diarrhea even in immunocompetent individuals without classic risk factors. Occupational exposure and mucosal injury are potential contributing factors. mNGS proved transformative in diagnosing this rare fungal enteritis. For unexplained diarrhea, tissue mNGS should be considered early when routine tests are negative and empirical antibiotic therapy fails. Empirical antifungal therapy should be started in a timely fashion when diarrhea remains refractory despite conventional diagnostic and therapeutic approaches.
We would like to extend our sincere gratitude and appreciation to all of the medical personnel of the 900th Hospital of PLA Joint Logistic Support Force who contributed to the care of this patient. We would also thank the patient and his family for allowing us to share in his experience for the betterment of all those who come after him in facing a similar health crisis.
| 1. | Bilal H, Zhang D, Shafiq M, Khan MN, Chen C, Khan S, Cai L, Khan RU, Hu H, Zeng Y. Epidemiology and antifungal susceptibilities of clinically isolated Aspergillus species in South China. Epidemiol Infect. 2023;151:e184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 2. | Belalmi NEH, Sid N, Sedrati T, Ouhida S, Bennoune O, Özmen Ö. Multisystemic aspergillosis with unusual vertebral osteomyelitis in a turkey flock in Bordj Bou Arreridj Province, Algeria. Open Vet J. 2025;15:2009-2015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 3. | Cha SA, Kim MH, Lim TS, Kim HH, Chang KY, Park HS, Kim HW, Wie SH, Jin DC. Invasive Primary Colonic Aspergillosis in the Immunocompetent Host without Classical Risk Factors. Yonsei Med J. 2015;56:1453-1456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 4. | Yelika SB, Tumati A, Denoya P. Intestinal Aspergillosis: Systematic Review on Patterns of Clinical Presentation and Management. Surg Infect (Larchmt). 2021;22:326-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 5. | Douglas AP, Smibert OC, Bajel A, Halliday CL, Lavee O, McMullan B, Yong MK, van Hal SJ, Chen SC; Australasian Antifungal Guidelines Steering Committee. Consensus guidelines for the diagnosis and management of invasive aspergillosis, 2021. Intern Med J. 2021;51 Suppl 7:143-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 6. | Zheng C, Zhang X, Ma Y, Zhang Y. Voriconazole in the management of invasive pulmonary aspergillosis in patients with severe liver disease: balancing efficacy and hepatotoxicity. J Mycol Med. 2025;35:101549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 7. | Maschmeyer G, De Greef J, Mellinghoff SC, Nosari A, Thiebaut-Bertrand A, Bergeron A, Franquet T, Blijlevens NMA, Maertens JA; European Conference on Infections in Leukemia (ECIL). Infections associated with immunotherapeutic and molecular targeted agents in hematology and oncology. A position paper by the European Conference on Infections in Leukemia (ECIL). Leukemia. 2019;33:844-862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 144] [Article Influence: 20.6] [Reference Citation Analysis (1)] |
| 8. | Donnelly JP, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE, Clancy CJ, Wingard JR, Lockhart SR, Groll AH, Sorrell TC, Bassetti M, Akan H, Alexander BD, Andes D, Azoulay E, Bialek R, Bradsher RW, Bretagne S, Calandra T, Caliendo AM, Castagnola E, Cruciani M, Cuenca-Estrella M, Decker CF, Desai SR, Fisher B, Harrison T, Heussel CP, Jensen HE, Kibbler CC, Kontoyiannis DP, Kullberg BJ, Lagrou K, Lamoth F, Lehrnbecher T, Loeffler J, Lortholary O, Maertens J, Marchetti O, Marr KA, Masur H, Meis JF, Morrisey CO, Nucci M, Ostrosky-Zeichner L, Pagano L, Patterson TF, Perfect JR, Racil Z, Roilides E, Ruhnke M, Prokop CS, Shoham S, Slavin MA, Stevens DA, Thompson GR, Vazquez JA, Viscoli C, Walsh TJ, Warris A, Wheat LJ, White PL, Zaoutis TE, Pappas PG. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367-1376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2335] [Cited by in RCA: 2166] [Article Influence: 361.0] [Reference Citation Analysis (3)] |
| 9. | Wei H, Thammasit P, Amsri A, Pruksaphon K, Deng F, Nosanchuk JD, Youngchim S. An overview of rapid non-culture-based techniques in various clinical specimens for the laboratory diagnosis of Talaromyces marneffei. Front Cell Infect Microbiol. 2025;15:1591429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 10. | Ye J, Jin S, Li Y, Gao W, Zheng C, Zuo H, Zhang C, Song M, Hao J, Liu Y, Feng Z, Zhang H, Zhao Z, Guo Y, Zhang L. Scedosporium boydii pulmonary infection in an immunocompetent patient with COPD confirmed by next-generation metagenomic sequencing and culture: a case report. BMC Infect Dis. 2025;25:938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 11. | Yamashita Y, Okano M, Yoshino T, Hattori H, Yamamoto T, Watanabe T, Takishita T, Akagi T, Nishizaki K. Carbohydrates expressed on Aspergillus fumigatus induce in vivo allergic Th2-type response. Clin Exp Allergy. 2002;32:776-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 12. | Murdock BJ, Shreiner AB, McDonald RA, Osterholzer JJ, White ES, Toews GB, Huffnagle GB. Coevolution of TH1, TH2, and TH17 responses during repeated pulmonary exposure to Aspergillus fumigatus conidia. Infect Immun. 2011;79:125-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 89] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 13. | Poddighe D, Mathias CB, Freyschmidt EJ, Kombe D, Caplan B, Marseglia GL, Oettgen HC. Basophils are rapidly mobilized following initial aeroallergen encounter in naïve mice and provide a priming source of IL-4 in adaptive immune responses. J Biol Regul Homeost Agents. 2014;28:91-103. [PubMed] |
| 14. | Shen S, Prame Kumar K, Stanley D, Moore RJ, Van TTH, Wen SW, Hickey MJ, Wong CHY. Invariant Natural Killer T Cells Shape the Gut Microbiota and Regulate Neutrophil Recruitment and Function During Intestinal Inflammation. Front Immunol. 2018;9:999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 15. | Díaz Alcázar MDM, Ruiz Escolano E, Casado Caballero FJ, Cervilla Sáez de Tejada E. Invasive aspergillosis of gastrointestinal debut without apparent respiratory involvement in an immunocompetent host. Rev Esp Enferm Dig. 2020;112:332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 16. | Bae NY, Byun JM, Kang CK, Choe PG, Kim NJ, Kim MS, Park KJ, Yoon SS. Successful treatment of angioinvasive aspergillosis causing diaphragmatic rupture with bowel perforation and cerebral aspergillosis in a patient with FLT3-mutated acute myeloid leukemia: A case report. Medicine (Baltimore). 2022;101:e28700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 17. | Dargenio VN, Cristofori F, Francavilla R. An Unexpected Guest in a Patient With Ulcerative Colitis. Gastroenterology. 2022;163:e1-e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 18. | Li ZM, Kuang YK, Zheng YF, Xu PH, Wang JY, Gan RJ, Li HX, Bai LH, Xie CM, Tang KJ. Gut-derived fungemia due to Kodamaea ohmeri combined with invasive pulmonary aspergillosis: a case report. BMC Infect Dis. 2022;22:903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 19. | Groenewegen B, Terveer EM, Joosse A, Barnhoorn MC, Zwittink RD. Fecal Microbiota Transplantation for Immune Checkpoint Inhibitor-Induced Colitis Is Safe and Contributes to Recovery: Two Case Reports. J Immunother. 2023;46:216-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 20. | Li YK, Tan B, Qin L, Fan SY, Guo XB, Zhao J, Wang MZ, Xu Y. [Fatal opportunistic infection occurred during the treatment of immune checkpoint inhibitor-associated colitis:case report]. Zhonghua Jie He He Hu Xi Za Zhi. 2025;48:272-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 21. | Li E, Hussein H, Todiwala A, Kirby R. Primary gut aspergillosis in a patient with acute myeloid leukaemia: the importance of early suspicion and definitive treatment. BMJ Case Rep. 2014;2014:bcr2013202316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Kulas J, Mirkov I, Tucovic D, Zolotarevski L, Glamoclija J, Veljovic K, Tolinacki M, Golic N, Kataranovski M. Pulmonary Aspergillus fumigatus infection in rats affects gastrointestinal homeostasis. Immunobiology. 2019;224:116-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 23. | Patterson TF, Thompson GR 3rd, Denning DW, Fishman JA, Hadley S, Herbrecht R, Kontoyiannis DP, Marr KA, Morrison VA, Nguyen MH, Segal BH, Steinbach WJ, Stevens DA, Walsh TJ, Wingard JR, Young JA, Bennett JE. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;63:e1-e60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2370] [Cited by in RCA: 2070] [Article Influence: 207.0] [Reference Citation Analysis (5)] |
| 24. | Warris A, Lehrnbecher T, Roilides E, Castagnola E, Brüggemann RJM, Groll AH. ESCMID-ECMM guideline: diagnosis and management of invasive aspergillosis in neonates and children. Clin Microbiol Infect. 2019;25:1096-1113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 129] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 25. | Walsh TJ, Anaissie EJ, Denning DW, Herbrecht R, Kontoyiannis DP, Marr KA, Morrison VA, Segal BH, Steinbach WJ, Stevens DA, van Burik JA, Wingard JR, Patterson TF; Infectious Diseases Society of America. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2191] [Cited by in RCA: 1791] [Article Influence: 99.5] [Reference Citation Analysis (1)] |