Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.115809
Revised: December 24, 2025
Accepted: January 16, 2026
Published online: March 21, 2026
Processing time: 141 Days and 20.6 Hours
Gastric syphilis (GS) is a rare infection caused by Treponema pallidum (T. pallidum) and may mimic neoplasia or be overlooked. Co-infection with Helicobacter pylori
A 17-year-old woman presented with upper abdominal pain for two weeks. A large irregular-shaped ulcer on the antrum was identified by endoscopy and the C-14 breath test was positive. Quadruple therapy was given to eradicate H. pylori, but her symptoms persisted after treatment. No significant improvement in the antral ulcer was found on secondary gastroscopy. Gynecologic examination obser
GS should be considered in any patient with an atypical or therapy-refractory gastric ulcer, particularly when serological syphilis positivity exists. IHC is a reliable method for confirming intra-gastric T. pallidum. Clinical doctors need to collaborate with pathologists to clarify diagnosis.
Core Tip: We describe the first case of gastric syphilis, Helicobacter pylori (H. pylori) and human immunodeficiency virus triple infection. The report highlights that syphilis can produce severe “tumor-like” gastric ulceration; moreover, co-infection must be identified when H. pylori eradication fails.
- Citation: Zhang JL, Wei N, Chen TM, Qiu W. Triple infection of gastric syphilis, Helicobacter pylori and human immunodeficiency virus: A case report. World J Gastroenterol 2026; 32(11): 115809
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/115809.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.115809
Syphilis, caused by Treponema pallidum (T. pallidum), is a sexually transmitted infection, which has re-emerged globally in the past decade worldwide[1,2]. Gastric syphilis (GS) remains exceptionally rare, even in China where the incidence of syphilis is high[3]. Non-specific epigastric pain, nausea or occult bleeding frequently leads to misdiagnosis as peptic disease, malignancy or lymphoma[4,5]. To date, no report has documented the simultaneous presence of GS, Helicobacter pylori (H. pylori) and human immunodeficiency virus (HIV). Through integrated endoscopic, pathological, and serological analysis, this case highlights the importance of considering syphilis in atypical gastric manifestations.
A 17-year-old Tibetan woman complained of persistent abdominal pain for approximately 2 weeks.
The patient developed progressive epigastric pain unresponsive to traditional medicines. She denied diarrhea, fever or hematemesis.
The patient had pulmonary tuberculosis 5 years earlier (completed treatment, documented cure). No prior sexually transmitted diseases.
The patient stated that there was no family history of gastric tumors.
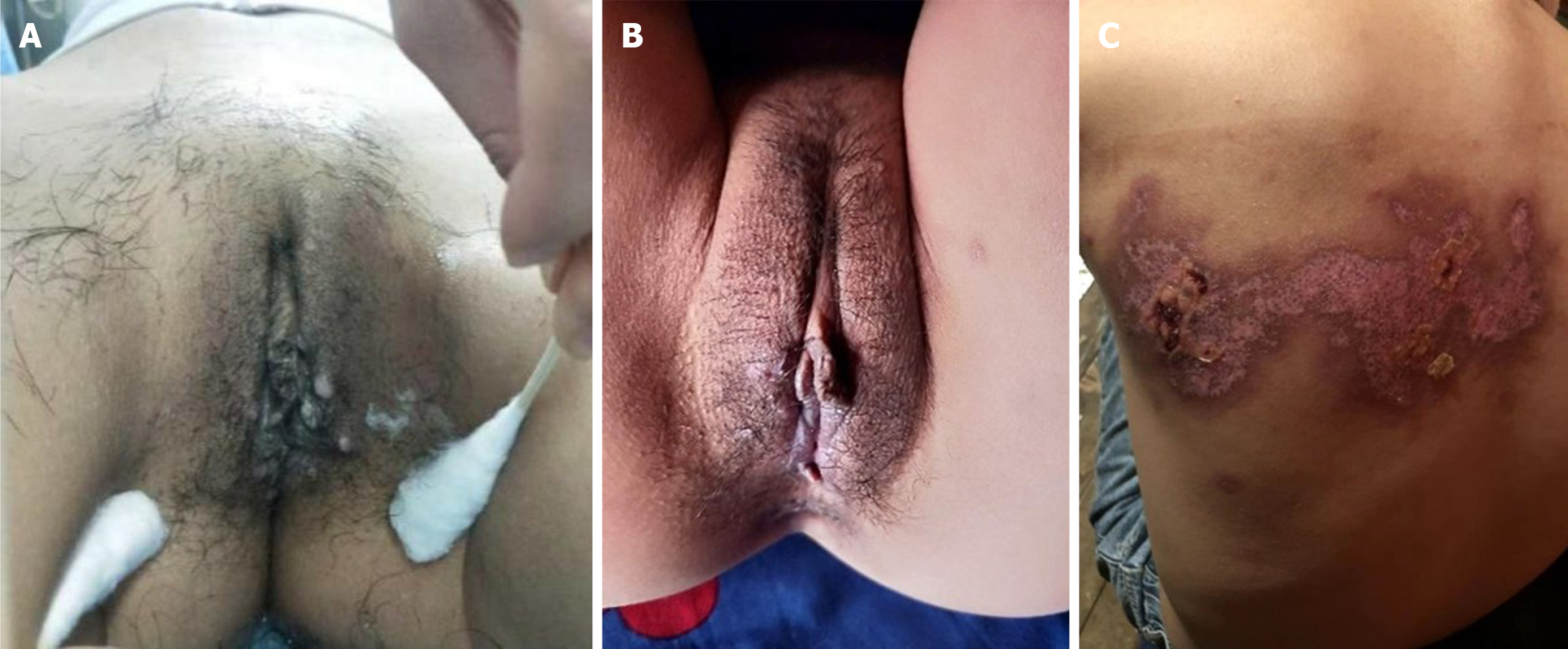
Vital signs were stable. Soft abdomen with localized epigastric tenderness and no guarding was observed. Gynecological inspection revealed painless, indurated, moist papules on the labia majora (Figure 1A).
Laboratory test results included hemoglobin 113 g/L, C-14 urea breath test was positive, serological tests revealed positive results for T. pallidum (rapid plasma reagin 1:128) and HIV. Eight months later, serological testing showed that T. pallidum specific antibody was also positive.
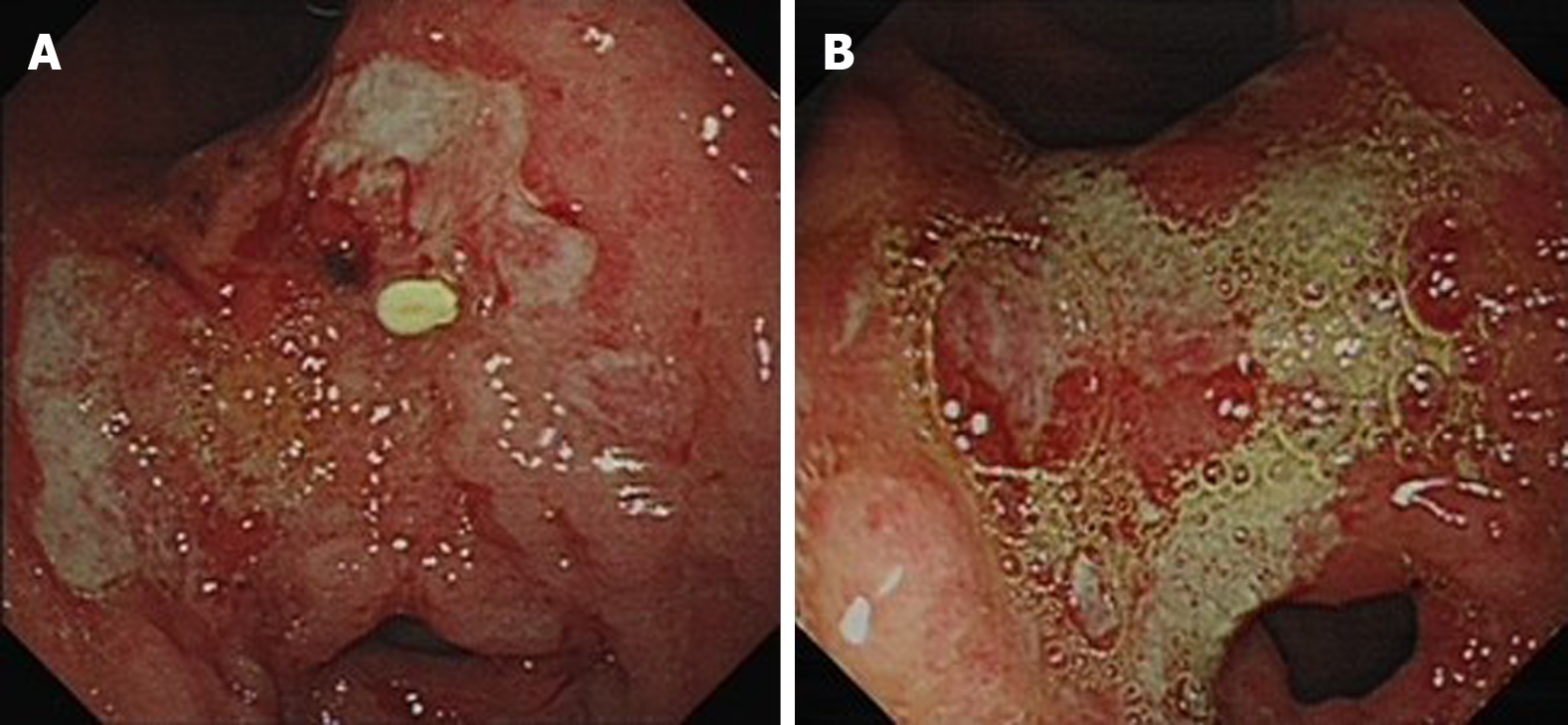
Gastroscopic inspection revealed extensive mucosal erosion, and a large irregular-shaped ulcer with bleeding in the antrum, which was approximately 3 cm × 2 cm in size, the bottom of the ulcer was uneven and the mucosa surrounding the ulcer was thickened with a nodular appearance. The manifestations of the ulcer were similar to that of a gastric tumor (Figure 2A). Quadruple therapy (lansoprazole 30 mg bid; bismuth pectin 200 mg bid; furazolidone 0.1 g bid; clarithromycin 0.5 g bid) was given to eradicate H. pylori. However, her symptoms persisted after treatment and a second gastroscopy showed an unchanged antral ulcer with reduced mucosal swelling (Figure 2B). Following anti-syphilis therapy, she declined repeat endoscopy.
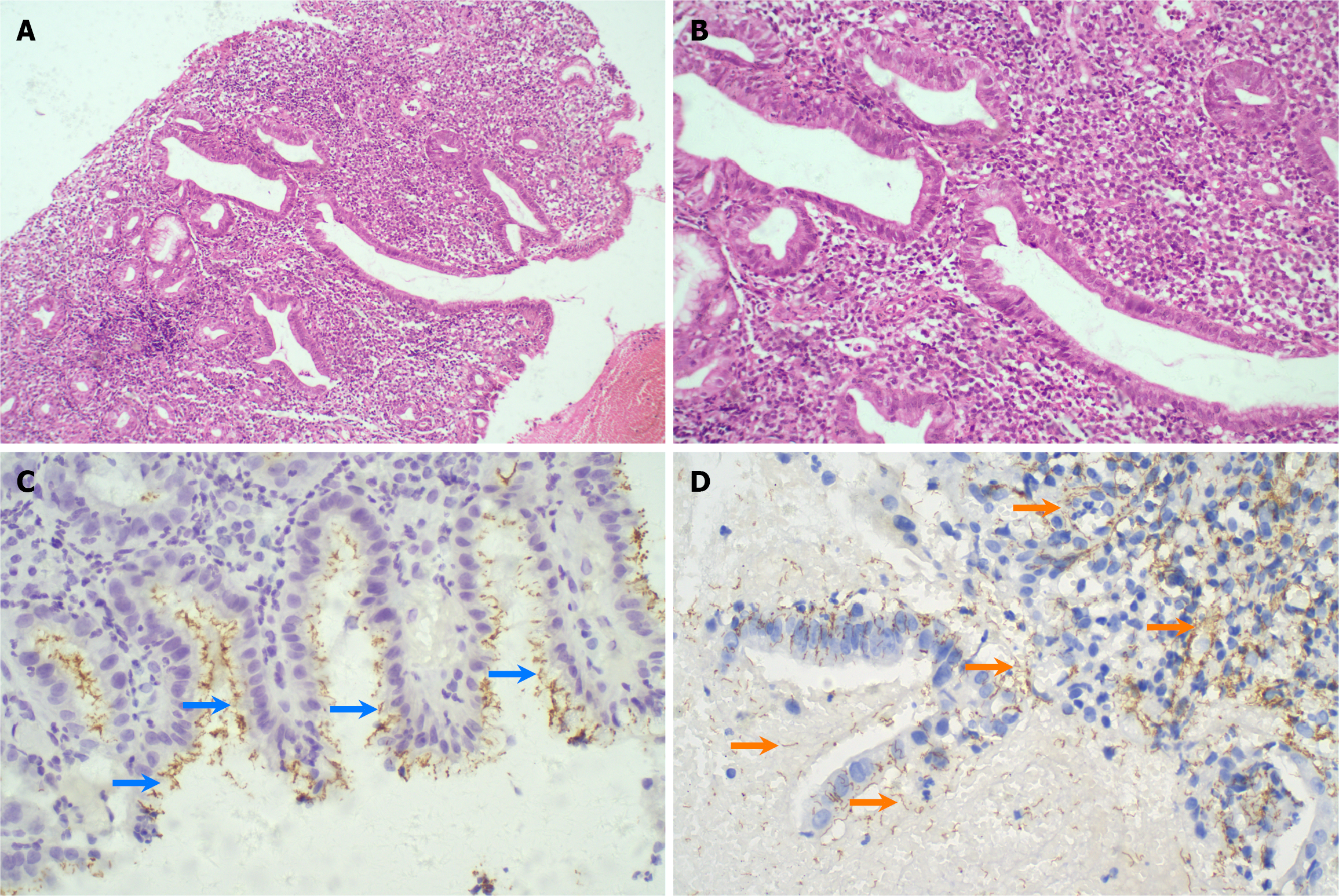
Hematoxylin-eosin staining revealed inflammatory infiltration predominantly consisting of dense lymphocytes and plasma cells, gland destruction, granulation tissue and follicular hyperplasia (Figure 3A and B). Immunohistochemistry (IHC) revealed scattered rod-shaped H. pylori on surface epithelium (Figure 3C) and numerous spiral T. pallidum within the mucosa, muscularis mucosae and exudate (Figure 3D).
The patient had a large irregular ulcer in the gastric antrum, and the C-14 urea breath test was positive. The gastric lesion extended beyond the typical presentation of H. pylori gastritis and the treatment for H. pylori eradication was ineffective. The non-specific antibody titer for syphilis was 1:128, with vulvar syphilis, suggesting a current syphilis infection. The histopathological examination revealed non-specific inflammatory changes, mucosal erosion, and dense inflammatory cell infiltration, with predominantly lymphocytes and plasma cells. In this case, the possibility of GS was suggested. Sub
The final diagnosis was triple infection of GS, H. pylori and HIV.
The patient initially received H. pylori eradication therapy without relief of her symptoms. Following the diagnosis of GS, she was treated with doxycycline (100 mg, bid, 30 days) due to penicillin allergy.
After anti-syphilis treatment, her gastric symptoms disappeared entirely, and the vulva papules gradually disappeared (Figure 1B). The patient suffered a serious herpes zoster virus infection with a large ulcer on her back 8 months later (Figure 1C), and it was speculated that this might have been related to HIV infection. Gastroscopy was declined by the patient at the 8-month visit and she remained asymptomatic regarding the upper-gastrointestinal tract. The patient was subsequently lost to follow-up. The timeline of the patient's progression is shown in Figure 4.
GS is a rare disease that can mimic malignancy, lymphoma, or peptic ulcer disease, often leading to missed or delayed diagnosis[6,7]. The reported age of GS patients ranges from 21 years to 78 years[6,7]. In this case, the large solitary antral ulcer with rolled margins and surrounding nodularity initially raised oncological suspicion. Co-existence of H. pylori, found in up to 58% of gastric ulcer patients, further obscured the underlying syphilitic etiology, particularly in a young, immunocompromised woman without prior history of previous sexually transmitted infections[7,8].
To date, this is the first reported triple infection of GS, H. pylori and HIV in an adolescent. Isolated dual infections have been reported, but never both concurrently[9,10]. In addition, 8 months later she developed herpes-zoster eruption, which might have been related to immune reconstitution. Early antiretroviral therapy can substantially reduce the risk of herpes zoster among individuals living with HIV[11]. At 17 years old, our patient is the youngest reported with GS, under
Endoscopically, GS is termed the “great imitator” owing to deep ulcers, nodularity, thickened folds or linitis-like rigi
Histopathology of GS is non-specific, chronic active gastritis shows dense lymphocyte and plasma cell infiltration[6]. Histological biopsy is prone to miss the diagnosis. GS should be differentiated from diseases such as H. pylori infection, cytomegalovirus infection, fungal infection and lymphoma, which are shown in Table 1. Diagnosis requires detection of T. pallidum in gastric biopsy and IHC staining has higher sensitivity and specificity[13,14]. Moreover, H. pylori colonizes the mucosal surface, whereas T. pallidum invades the full mucosal thickness. Thus, in this case, inflammation persisted after H. pylori eradication, and the patient’s symptoms were not relieved.
| Diseases | Endoscopic characteristics | Histopathological characteristics |
| Gastric syphilis[6,17,18] | Common site is the antrum with multiple ulcers or giant ulcers. Nodular mucosal elevations and thickening of gastric folds that are similar to gastric carcinoma. Fibrotic narrowing and rigidity occur in the late-stage | Non-specific inflammation mainly plasma cells and lymphocytes. IHC staining for T. pallidum in all layers of mucosa |
| H. pylori-associated gastritis[19] | Common site is the junction of the gastric antrum and body. Congestion, edema, erythema, and ulcers appear on the gastric mucosa, usually superficial | Non-specific inflammation mainly neutrophils and lymphocytes on the surface of gastric mucosa. Specific IHC staining for H. pylori |
| Gastric carcinoma[20,21] | Mostly shows as a huge ulcer that is stained with moss adhered to the surface of the ulcer, and the edge of the ulcer is raised like a dam | Hyperplasia of atypical epithelial cells, with a glandular, patchy, or scattered distribution, with special markers, such as CK, P53, and Ki67 |
| Gastric lymphoma[22,23] | Common site is the antrum. Mucosal erosion, ulceration, mucosal nodules or thickening, presenting as flat or polypoid elevations | Large sheets of monotonous cells within the mucosa; specific IHC staining helps the diagnosis |
| Gastric tuberculosis[24] | Common sites are the gastric antrum and lesser curvature. Nodular hypertrophic mass may mimic carcinoma, some present with antrum deformation and obstruction | Granulomatous inflammation with stenosis, perforation, or fistula, and positive staining for acid fast bacilli |
| Gastric cytomegalovirus[25,26] | Common site is the antrum. Punched-out ulcers with partial mucosal thickening and inflammation, which are similar to gastric carcinoma | Eosinophilic inclusions visible in epithelium or stroma. Specific IHC staining for cytomegalovirus |
At present, there is no conclusive evidence to confirm a clear correlation between HIV infection and gastric lesions. It was reported that gastrointestinal opportunistic infections exist in a small number of late-stage HIV infected individuals receiving highly active antiretroviral therapy[15]. Yet the incidence of chronic active gastritis does not differ between HIV-positive patients with or without H. pylori infection[16].
In our case, missed diagnosis might be attributable to: (1) The rarity of GS; (2) Young age and the absence of a sexually transmitted infection history; (3) Antral predilection and nonspecific endoscopy; (4) Positive C-14 breath test diverting attention; and (5) Non-specific histology. Thus, GS is easily overlooked by both gastroenterologists and pathologists. Multidisciplinary evaluation of symptoms, history, serology, endoscopy and histology reduced the misdiagnosis of GS.
Therefore, we propose a stepwise method for patients with refractory gastric ulcers and HIV or high-risk sexual history. Firstly, initial workup includes serology for syphilis and HIV, and the C-13/14 breath test. Secondly, if the patient is H. pylori-negative or treatment fails, then repeat endoscopy with multipoint biopsies and targeted IHC for T. pallidum, cytomegalovirus, Epstein-Barr virus, tuberculosis, and fungal stains should be carried out. Moreover, if syphilis serology is positive, penicillin or doxycycline therapy can be initiated. Finally, clinical symptoms and serology for syphi
This report has a few limitations, as it was a single case that lacked generalizability. However, we hope that this case may contribute to the recognition and management of GS in the context of multiple concurrent infections, and enhance clinical awareness of this rare condition.
This case highlights the diagnostic complexity of triple infection in an immunocompromised woman. GS should be considered in patients with atypical or refractory gastric ulcers, especially after failed H. pylori eradication. IHC staining is a convenient and low-cost method to confirm T. pallidum. Finally, multidisciplinary collaboration between gastroenterologists, infectious-disease specialists and pathologists is essential to avoid misdiagnosis.
| 1. | Goldmeier D, Hay P. Acquired syphilis in adults. N Engl J Med. 1992;327:959-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 2. | Lan YM, Yang SW, Dai MG, Ye B, He FY. Gastric syphilis mimicking gastric cancer: A case report. World J Clin Cases. 2021;9:7798-7804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 3. | Wu Y, Zhu W, Sun C, Yue X, Zheng M, Fu G, Gong X. Prevalence of syphilis among people living with HIV and its implication for enhanced coinfection monitoring and management in China: A meta-analysis. Front Public Health. 2022;10:1002342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 4. | Su R, Liu Y, Shan D, Li P, Ge L, Li D. Prevalence of HIV/syphilis co-infection among men who have sex with men in China: a systematic review and meta-analysis. BMC Public Health. 2025;25:1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 5. | Yang H, Zhang M, Li H, Huang Z, Sun Y, Li W, Li C, Qin X, Wang Y, Zhang X, Zhao Z, Wang L, Wang L, Qian J. Prevalence of common upper gastrointestinal diseases in Chinese adults aged 18-64 years. Sci Bull (Beijing). 2024;69:3889-3898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 6. | Mylona EE, Baraboutis IG, Papastamopoulos V, Tsagalou EP, Vryonis E, Samarkos M, Fanourgiakis P, Skoutelis A. Gastric syphilis: a systematic review of published cases of the last 50 years. Sex Transm Dis. 2010;37:177-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 7. | Navarro-Martínez S, Payá-Llorente C, Planells Roig MV. Gastric syphilis and its importance for a differential diagnosis in HIV patients. Rev Esp Enferm Dig. 2020;112:578-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 8. | Okamoto K, Hatakeyama S, Umezawa M, Hayashi S. Gastric syphilis: The great imitator in the stomach. IDCases. 2018;12:97-98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Ávila-Nieto C, Pedreño-López N, Mitjà O, Clotet B, Blanco J, Carrillo J. Syphilis vaccine: challenges, controversies and opportunities. Front Immunol. 2023;14:1126170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 10. | Osias E, Hung P, Giacani L, Stafylis C, Konda KA, Vargas SK, Reyes-Díaz EM, Comulada WS, Haake DA, Haynes AM, Caceres CF, Klausner JD. Investigation of syphilis immunology and Treponema pallidum subsp. pallidum biology to improve clinical management and design a broadly protective vaccine: study protocol. BMC Infect Dis. 2020;20:444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 11. | Dauby N, Motet C, Libois A, Martin C. The value of herpes zoster prevention in people aging with HIV: A narrative review. HIV Med. 2023;24:1190-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 12. | Choi YL, Han JJ, Lee DK, Cho MH, Kwon GY, Ko YH, Park CK, Ahn G. Gastric syphilis mimicking adenocarcinoma: a case report. J Korean Med Sci. 2006;21:559-562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 13. | Hoang MP, High WA, Molberg KH. Secondary syphilis: a histologic and immunohistochemical evaluation. J Cutan Pathol. 2004;31:595-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 125] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 14. | Liu X, Hameed O. Treponema pallidum immunostain distinguishing syphilitic gastritis from Helicobacter pylori-associated gastritis. Hum Pathol. 2010;41:617-9; author reply 619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 15. | Werneck-Silva AL, Prado IB. Gastroduodenal opportunistic infections and dyspepsia in HIV-infected patients in the era of Highly Active Antiretroviral Therapy. J Gastroenterol Hepatol. 2009;24:135-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 16. | Lv FJ, Luo XL, Meng X, Jin R, Ding HG, Zhang ST. A low prevalence of H pylori and endoscopic findings in HIV-positive Chinese patients with gastrointestinal symptoms. World J Gastroenterol. 2007;13:5492-5496. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Yu HJ, Kim SJ, Oh HH, Im CM, Han B, Myung E, Yun SJ, Lee KH, Joo YE. Case report of gastric syphilis in Korea: Clinical features, pathology, management, and prognosis. Medicine (Baltimore). 2021;100:e28212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (2)] |
| 18. | Souza Varella Frazão M, Guimarães Vilaça T, Olavo Aragão Andrade Carneiro F, Toma K, Eliane Reina-Forster C, Ryoka Baba E, Cheng S, Ferreira de Souza T, Guimarães Hourneaux de Moura E, Sakai P. Endoscopic aspects of gastric syphilis. Case Rep Med. 2012;2012:646525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 19. | Hall R, Bruce-Brand C, Mudini W, Aldera AP. Role of Helicobacter pylori Immunohistochemistry in the Histopathological Assessment of Inflamed Endoscopic Gastric Biopsies. Korean J Helicobacter Up Gastrointest Res. 2024;24:45-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 20. | Huang Q, Cheng YQ, Hu KW, Ding Y. Gastric Cardiac Carcinoma: Recent Progress in Clinicopathology, Prognosis, and Early Diagnosis. J Dig Dis. 2025;26:22-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Umeda Y, Tanaka K, Ikenoyama Y, Hamada Y, Yukimoto H, Yamada R, Tsuboi J, Nakamura M, Katsurahara M, Horiki N, Ogura T, Tamaru S, Nakagawa H, Tawara I. The usefulness of image-enhanced endoscopy to distinguish gastric carcinoma in tumors initially diagnosed as adenomas by endoscopic biopsy: A retrospective study. Medicine (Baltimore). 2023;102:e32881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 22. | Bai Z, Zhou Y. A systematic review of primary gastric diffuse large B-cell lymphoma: Clinical diagnosis, staging, treatment and prognostic factors. Leuk Res. 2021;111:106716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 23. | Violeta Filip P, Cuciureanu D, Sorina Diaconu L, Maria Vladareanu A, Silvia Pop C. MALT lymphoma: epidemiology, clinical diagnosis and treatment. J Med Life. 2018;11:187-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 24. | Chaudhary P, Khan AQ, Lal R, Bhadana U. Gastric tuberculosis. Indian J Tuberc. 2019;66:411-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 25. | Kang S, Kim NH. Cytomegalovirus-induced Oral, Esophageal, Gastric and Colonic Ulcers in an Immunocompetent Patient. Korean J Gastroenterol. 2025;85:389-394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 26. | Yeh PJ, Chiu CT, Lai MW, Wu RC, Kuo CJ, Hsu JT, Su MY, Le PH. Cytomegalovirus gastritis: Clinicopathological profile. Dig Liver Dis. 2021;53:722-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |