Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.115846
Revised: December 2, 2025
Accepted: January 14, 2026
Published online: March 21, 2026
Processing time: 140 Days and 17.5 Hours
The coexistence of gastrointestinal and respiratory diseases is a widespread phenomenon, featuring complex bidirectional interactions that seriously threaten human health and increase the difficulty of clinical management. The gut-lung axis is an emerging field in the study of organ-organ interactions in recent years, revealing the complex bidirectional regulatory network between gastrointestinal and respiratory diseases. The potential mechanisms of comorbidities of gastro
Core Tip: The gut-lung axis represents a bidirectional communication network that links the gastrointestinal and respiratory systems through microbial, immune, and metabolic pathways. This review synthesized recent advances in understanding how gut dysbiosis, microbial translocation, immune dysregulation, and epigenetic modifications contribute to gastro
- Citation: Huang HJ, Liu PP, Dong DF. Research progress on comorbidity between gastrointestinal and pulmonary diseases from the perspective of the gut-lung axis. World J Gastroenterol 2026; 32(11): 115846
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/115846.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.115846
Most research on the impact of the gut microbiome on disease has focused on intestinal disorders[1]. However, over the past decade numerous significant studies have demonstrated associations between the gut microbiota and diseases beyond the gut. For instance, the influence of the gut microbiota extends to distant sites such as the heart, lungs, brain, pancreas, and liver[2-6]. Specific microbes present in different microenvironments have been implicated in a range of disorders including autoimmune diseases, metabolic disorders, neurological diseases, and various cancers[7-10]. Moreover, diseases in these distant organs can in turn influence the composition of gut microbes, underscoring the bidi
In the context of the lungs, this phenomenon is termed the gut-lung axis. The gut-lung axis represents an emerging research frontier, referring to the interactive system involving the gut microbiota, intestinal barrier function, and pulmonary immune responses[11]. This connection between the lungs and gut is mediated not only by direct neural and immune pathways but also by factors such as gut microbiota and metabolites achieved through the migration of gut microbes[12]. The bacterial composition and metabolites in the gut and lungs can modulate systemic and local immunity with specific microbial communities influencing the development of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and respiratory infections[13]. The concept of the gut-lung axis was first proposed by Pugin et al[14] in 1991 in which they systematically revealed that gut microbiota may cause pulmonary infections through bacterial ectopic migration. Currently, the gut-lung axis has become a hotspot in cross-organ microbiome research. Its “bidirectional communication” has been progressively demonstrated to play a crucial role in inflammation, infection, and tumorigenesis and has been incorporated into multiple international research initiatives[15,16]. This indicates that research into the comorbidity of gastrointestinal and respiratory diseases caused by gut-lung axis dysregulation has advanced from describing epidemiological patterns to investigating underlying mechanisms, attracting increasing attention from scholars.
The co-occurrence of gastrointestinal and pulmonary diseases is widespread[17-19]. For instance, the prevalence of inflammatory bowel disease (IBD) among patients with COPD is significantly higher than in the general population[20]. Approximately 85% of patients with COPD experience at least one gastrointestinal symptom[21], and intestinal permeability is elevated in patients with COPD compared with individuals without COPD[22]. COPD promotes IBD by causing damage to the intestinal mucosal structure[23]. Patients with IBD face a significantly increased risk of developing respiratory-related diseases after IBD diagnosis[13,24] with both lines of evidence indicating a “bidirectional exacerbation” relationship between pulmonary disease and intestinal symptoms.
Dysregulation of the gut-lung axis driven by gut microbiota imbalance is associated with poorer clinical outcomes in respiratory diseases. Conversely, acute exacerbations of pulmonary disease can also worsen intestinal symptoms[25]. Compared with monopathology, comorbid conditions typically yield poorer health outcomes[26,27]. For instance, among patients with COPD those with IBD exhibit higher mortality risk than those without IBD[28]. Furthermore, among patients with asthma those with concomitant gastroesophageal reflux disease (GORD) exhibit significantly increased risks of anxiety and depression compared with those without GORD[29,30]. Therefore, gaining a deeper understanding of the mechanisms underlying gastrointestinal-pulmonary disease comorbidity is critically important.
This review discussed the central role of the gut-lung axis in gastrointestinal and pulmonary disease comorbidity. We examined the bidirectional regulatory mechanisms of the gut-lung axis, the pathophysiological basis of gastrointestinal-pulmonary comorbidity, immunomodulation, microbial translocation and dysbiosis, clinical subtypes of comorbidities, and intervention strategies. This exploration further delineated the regulatory network and clinical translation pathways of the gut-lung axis as a pivotal hub in cross-organ pathophysiological connections, providing a foundation for early identification, precise classification, and personalized treatment of gastrointestinal-pulmonary comorbidities.
Traditionally, the gut has been primarily regarded as a digestive organ while the lungs have been considered responsible for gas exchange and respiratory functions[31]. However, accumulating basic research evidence and epidemiological data in recent years have highlighted critical interactions between the gut microbiota and the lungs[32]. These interactions involve not only host-microbe relationships but also crosstalk between different microbial communities, influencing local and systemic immune responses as well as airway homeostasis. The gut-lung axis exerts its effects through both local regulatory mechanisms and long-distance effects, influencing the progression of respiratory diseases by modulating immune and inflammatory responses. This can compromise intestinal barrier integrity, which is associated with bacterial translocation, persistent inflammation, and pulmonary fibrosis[33-36].
Although the intestinal mucosa and pulmonary mucosa reside in distant organs, they exhibit high structural homology and close functional coordination in terms of immune mechanisms and microecological regulation. From an embryonic developmental perspective, the lungs and trachea develop from the foregut of the intestine while respiratory epithelial cells and membrane structures differentiate from the endoderm of the gastrula. The lungs share developmental homology with the colon and ileum in embryonic tissue formation[37]. Both the lungs and intestines are organs directly exposed to the external environment. They share a typical mucosal structure composed of epithelium and lamina propria, belonging to the common mucosal immune system. The secretion of secretory immunoglobulin A by both pulmonary and intestinal mucosae forms a crucial molecular biological basis for the “lung-gut axis”[38]. The mucosal immune network serves as a bridge connecting the lungs and intestines. The lungs and intestines can function as induction sites and effector sites, respectively, establishing a shared mucosal defense mechanism through mucosal lymphocyte homing[39].
The gastrointestinal vagus nerve serves as a core neural pathway connecting the intestines to the lungs. Through neural, endocrine, and immune mechanisms, it achieves remote regulation of pulmonary function[40]. Krzyzaniak et al[41] found that vagus nerve electrical stimulation significantly reduced post-burn pulmonary permeability and interleukin (IL)-6/IL-17 levels. Liu et al[42] demonstrated that short-chain fatty acids (SCFAs), metabolites of the lung microbiota, upregulate brainstem serotonin via the vagus nerve, indirectly inhibiting pulmonary calcitonin gene-related peptide release and airway inflammation. Activation of the gastrointestinal vagus nerve is often accompanied by reduced pulmonary neutrophil infiltration, thereby suppressing excessive inflammation[43]. Consequently, modulating vagal tone has been proposed as a potential therapeutic strategy for respiratory inflammatory diseases. However, the reliability and safety of this method needs to be carefully evaluated.
Increasing evidence indicates that SCFAs, derived from the fermentation of indigestible foods by gut microbiota, represent the most prominent immunomodulatory metabolites in the gastrointestinal tract and the most thoroughly studied bacterial metabolites exerting their effects along the gut-lung axis[44]. The effects of microbiota-derived SCFAs extend beyond the intestinal lumen. SCFAs diffuse from the gut into the bloodstream, reaching the bone marrow where they promote hematopoiesis, thereby alleviating airway inflammation and maintaining pulmonary homeostasis[32]. Furthermore, since SCFAs produced in the gut can enter the circulation, these metabolites may exert direct effects on the lungs and may also be indirectly mediated by the pulmonary microbiota[45]. Compared with healthy individuals, patients with COPD exhibit lower fecal SCFA concentrations with COPD severity negatively correlated to SCFA levels[46]. This suggests that COPD may represent a downstream consequence of disrupted gut microbiota. Increasing SCFA concentrations and the abundance of beneficial gut microbiota through dietary fiber intake may represent one potential solution for mitigating COPD and its associated complications.
In summary, the gut-lung axis is not merely an anatomical notion, it is a trans-organ homeostatic system built from ontogenically related mucosal architectures, a common mucosal immune network, bidirectional immune-cell trafficking, neuroendocrine-immune multiroute signaling, and long-range control by microbial metabolites.
Immune homeostasis depends on the microbiome, providing cues for the proper maturation and activation of the immune system, including microbial components and metabolites[47]. In humans environmental factors such as diet, antibiotic treatment, and stress can shift the gut microbiota toward reduced numbers of beneficial bacteria, accompanied by increased abundance of non-beneficial species[48]. The epithelial surfaces of the gastrointestinal and respiratory tracts are exposed to diverse microorganisms. Ingested microbes can enter both sites, and microbiota from the gastrointestinal tract can reach the lungs via respiration[13].
Current research is evaluating the impact of the gut microbiota on the lungs and the use of probiotics and prebiotics for preventing and treating acute and chronic pulmonary diseases. For example, a positive correlation exists between the presence of “beneficial” bacteria in the gut (such as Bifidobacterium longum) and lower asthma incidence[49]. Additionally, segmented filamentous bacteria in the gut stimulate pulmonary helper T cell 17 (Th17) cell responses and reduce Streptococcus pneumoniae infections and associated mortality[50].
Disruption of gut microbial composition and function, termed dysbiosis, compromises tissue and immune homeostasis and is associated with various inflammatory diseases both within and outside the gastrointestinal tract[51]. For instance, disruption of gut-lung crosstalk is linked to increased susceptibility to airway diseases and infections, including allergies[52]. The significance of the gut-lung axis is evident in patients with chronic gastrointestinal diseases and pulmonary disorders, such as irritable bowel syndrome and IBD, which exhibit a higher prevalence of pulmonary diseases[52-54]. Inflammatory mediators such as IL-17 and interferon-γ released from pulmonary infections or injuries can also reach the gut via the bloodstream or the gut-lung axis, compromising intestinal barrier integrity and disrupting microbiota homeostasis, thereby triggering or amplifying gut immune responses[55]. Compared with healthy individuals, dysbiosis of the gut microbiota is more commonly observed in patients with COPD[56]. Close monitoring of gut microbiota status holds dual value for diagnosing both gastrointestinal and pulmonary diseases.
Beyond bacterial components emerging evidence highlights the critical yet underappreciated role of the gut myco
The gut microbiota constitutes the largest microbial ecosystem in the human body, and its stability is crucial for maintaining intestinal barrier function and immune homeostasis. Under pathological conditions factors such as smoking, particulate matter exposure, or intestinal inflammation can disrupt the intestinal epithelial barrier, leading to microbial translocation and inducing or exacerbating pulmonary inflammatory responses[58-60]. For instance, exposure to cigarette smoke significantly reduces gut microbiota diversity, diminishes beneficial bacteria, and increases the proportion of Gram-negative bacteria. This, in turn, elevates serum lipopolysaccharide levels, activates alveolar macrophages, and induces the release of inflammatory mediators such as tumor necrosis factor-α (TNF-α) and IL-6, contributing to the development of diseases like COPD and acute respiratory distress syndrome (ARDS). PM2.5 exposure can also alter gut microbiota composition, reduce SCFA levels, and weaken their role in maintaining intestinal barrier function and anti-inflammatory effects, further promoting dysbiosis and systemic inflammation[61].
Impaired intestinal barrier function can lead to the translocation of gut microbiota and their metabolites to the lungs, inducing or exacerbating pulmonary diseases[61,62]. For instance, in models of acute lung injury or ARDS, gut-derived bacteria can accumulate in the lungs, altering the pulmonary microbiome structure and further amplifying inflammatory responses[62]. Gut microbiota dysbiosis and barrier disruption mark the onset of gut-lung axis dysfunction. Microbiota translocation not only directly activates pulmonary immune responses but also remotely modulates inflammatory states in the lungs via metabolic byproducts, representing a key mechanism underlying gastrointestinal-pulmonary disease comorbidity.
Based on existing research, dysbiosis of the gut microbiome is significantly associated with the onset and progression of certain pulmonary diseases as summarized in Table 1[62-65].
| Diseases | Details | Ref. |
| COPD | Fecal microbiological profiles were analyzed using 16S rRNA gene sequencing. The results showed a distinct difference in the bacterial community composition between the AECOPD, COPD, and healthy control groups. The COPD and AECOPD groups had higher levels of Firmicutes but lower levels of Bacteroidetes compared with the healthy control group at the phylum level. At the genus level, there was an increased abundance of Lachnoclostridium, Alistipes, Streptococcus, and Prevotella in patients with COPD and AECOPD. Increasing levels of Lachnoclostridium and Prevotella may indicate an acute exacerbation of COPD | [62] |
| Asthma | Gut microbial species and metabolites primarily SCFAs may either worsen or reduce airway inflammation by regulating the balance of Th1/Th2 and other immune mediators | [63] |
| ARDS | The major characteristic of the intestinal flora in patients with ARDS/CAP was higher abundances of Gram-negative bacteria than normal controls. The gut microbiota derived from patients with ARDS/CAP promoted neuroinflammation and behavioral dysfunction in mice. Mice who underwent fecal transplant from patients with ARDS/CAP had increased systemic LPS, systemic inflammation, and increased colonic barrier permeability | [64] |
| VAP | The fungal genus Alternaria spp. is associated with the occurrence of VAP. The composition of the gut microbiota differs between patients who are critically ill patients and subsequently develop VAP and those who do not | [65] |
The gut microbiota plays a central role in the development and regulation of the immune system, particularly in maintaining the balance of T cell subsets such as regulatory T cells (Treg) and Th17 cells. Dysbiosis can disrupt this equilibrium, inducing immune dysregulation and subsequently affecting the pulmonary immune microenvironment[66-68]. Studies indicate that gut microbiota disruption suppresses Treg cell differentiation while promoting Th17 cell expansion, leading to elevated proinflammatory factors like IL-17 and IL-6. These factors can migrate via the bloodstream to the lungs, activate local immune cells, and induce neutrophil infiltration and lung tissue damage[69].
In bronchopulmonary dysplasia models, perinatal antibiotic exposure disrupts the gut microbiota, downregulates immune-related gene expression, and induces marked inflammatory and fibrotic changes in lung tissue[70]. Additionally, gut microbiota metabolites such as SCFAs and Tryptophan (Trp) metabolites can regulate immune processes including alveolar macrophage polarization and T cell differentiation by activating signaling pathways like AhR and G Protein-Coupled Receptors, thereby maintaining pulmonary immune homeostasis[71-73]. The gut microbiota participates in sustaining pulmonary immune homeostasis by modulating Treg/Th17 balance, immune cell differentiation, and cytokine networks. Microbiota dysbiosis-induced immune imbalance represents one of the core mechanisms mediating comorbidity through the gut-lung axis.
Metabolites produced by gut microbiota not only act as immunomodulators but also serve as key regulators of inflammatory signaling pathways. Dysbiosis can lead to sustained activation of inflammatory signaling cascades, triggering systemic metabolic dysregulation such as insulin resistance, hyperglycemia, and lipid metabolism abnormalities. These metabolic abnormalities, in turn, exacerbate pulmonary inflammation and injury[74]. For instance, SCFAs exert potent anti-inflammatory effects by activating G Protein-Coupled Receptors signaling and inhibiting histone deacetylase activity[75,76]. Dysbiosis reduces SCFA levels, weakening their inhibitory effect on inflammatory pathways like nuclear factor kappa-B and promoting sustained release of inflammatory mediators such as IL-1β and TNF-α, thereby forming an inflammatory cascade[77-79]. In disease models like bronchopulmonary dysplasia and COPD, impaired glucose meta
Butyrate derived from the gut microbiota promotes the production of proresolving mediators by alveolar macrophages, accelerating inflammation resolution. Oral sodium butyrate administration alleviates acute exacerbations in COPD mice[82,83]. The gut microbiota participates in the onset and progression of pulmonary inflammation by regulating inflammatory signaling pathways and metabolic homeostasis. The inflammatory cascade and metabolic dysregulation induced by dysbiosis form a crucial bridge in the gut-lung axis-mediated comorbidity. Therefore, supplementing the gut microbiota or its metabolites may represent an effective therapeutic strategy for treating inflammation-related pulmonary diseases.
Based on existing research, the primary clinical patterns of gastrointestinal and pulmonary disease comorbidity from the gut-lung axis perspective have been preliminarily categorized as follows: Asthma and GORD[84-87]; COPD and IBD[88]; acute pancreatitis and ARDS[89]; and chronic bacterial pneumonia and antibiotic-associated diarrhea[90].
A nationwide cohort study from Denmark indicated an increased risk of obstructive pulmonary disease in IBD[24]. Smoking and genetics are common risk factors for both conditions. Dysbiosis of the gut microbiota can cause inflammatory cells circulating in the body to migrate to the lungs. Multiple studies indicate that the incidence of ARDS among hospitalized patients with severe acute pancreatitis ranges from 18.6% to 46.6%[91,92]. Severe acute pancreatitis leads to the release of trypsin and phospholipase A2 into the bloodstream, which directly digest pulmonary surfactant and induce alveolar collapse[93]. Massive release of TNF-α, IL-6, IL-8, and platelet-activating factor increases pulmonary capillary permeability, leading to neutrophil accumulation in the lungs[94]. Disruption of the intestinal barrier causes bacterial translocation or endotoxin migration, elevating circulating lipopolysaccharides and reactivating alveolar macrophages, forming a “second hit”[95]. Tissue factor release forms microthrombi, exacerbating ventilation-perfusion mismatch[96]. Compared with patients with acute pancreatitis alone, those concurrently suffering from acute pancreatitis and ARDS exhibit higher mechanical ventilation requirements, longer intensive care unit stays, increased mortality rates, and impaired long-term pulmonary function[91]. Patients with bacterial pneumonia receiving antibiotic therapy exhibit a high incidence of antibiotic-associated diarrhea, and combined drug regimens can elevate this risk[97,98]. Antibiotics disrupt the intestinal anaerobic microbiota, impairing bile acid deconjugation and reducing SCFAs, leading to osmotic or secretory diarrhea.
Despite the identification of numerous gastrointestinal-pulmonary disease comorbidities, significant diagnostic and therapeutic challenges persist in this interdisciplinary field. Although the concept of the gut-lung axis has been proposed, its specific immune pathways and the mechanisms of action involving microbial metabolites remain incompletely elucidated, making it difficult to precisely identify therapeutic targets.
Gastrointestinal bleeding, diarrhea, cough, and dyspnea frequently co-occur in patients in the intensive care unit or who are elderly, but symptom overlap complicates disease prioritization. It is more challenging when therapeutic anti-TNF-α biologics for IBD reactivate latent tuberculosis, exacerbating pulmonary infection. Conversely, long-term inhaled corticosteroids in patients with COPD elevate the risk of Clostridium difficile infection[99]. Furthermore, physicians across different departments often prioritize different disease aspects. For instance, pulmonologists rarely assess intestinal symptoms[100,101] while gastroenterologists may overlook chronic cough or airway obstruction[102,103]. Establishing large-scale multi-omics cohorts, developing cross-organ risk prediction algorithms, and conducting adaptive platform trials targeting gut-lung synchronous endpoint are essential to overcome the current fragmented treatment paradigm focused solely on single-disease management.
Regulating the gut microbiota to indirectly improve pulmonary disease phenotypes represents one of the most promising translational directions. In a COPD mouse model, fecal microbiota transplantation significantly reduced alveolar destruction and inflammatory cell infiltration, suggesting its potential application in advanced COPD[99]. Influenza infection causes damage to the lung and intestinal tissues, disrupts the intestinal microbiota and metabolites, and affects the Th17/Treg balance. Antibiotic treatment exacerbates these symptoms. Supplementation of tryptophan and Lactobacillus improves lung and intestinal health[104]. Moreover, alterations in pulmonary and intestinal Trp metabolites correlate strongly with pulmonary microbiota dysbiosis, demonstrating optimal discriminatory power for diagnosing and grading neonatal ARDS[105]. These findings reveal that Trp and its intestinal derivatives can remotely regulate the pulmonary immune microenvironment via the gut-lung axis, influencing inflammation severity and tissue repair. However, clinical translation remains constrained by critical challenges including donor standardization and long-term safety evaluation.
Oral sustained-release butyrate capsules improve Forced Expiratory Volume in 1 second and 6-minute walk distance in patients with COPD; related phase II trials are ongoing[44]. Metabolite supplementation is emerging as a promising therapeutic approach for gastrointestinal and pulmonary comorbidities. It is important to note that the clinical translation of such effects is likely influenced by a multitude of host and environmental factors beyond a single metabolite.
A high-fiber diet has demonstrated potential in improving symptoms among patients with coronavirus disease 2019[106]. The Mediterranean dietary pattern is significantly associated with reduced frequency of COPD exacerbations, potentially achieved by decreasing gut proinflammatory bacteria abundance[107]. Among smokers, smoking cessation combined with inulin supplementation reverses gut dysbiosis and lowers airway IL-8 levels[108,109]. Dietary lifestyle interventions centered on high-fiber intake, Mediterranean-style eating, smoking cessation, and prebiotic supplementation can achieve cross-organ regulation of pulmonary inflammation and susceptibility to infection by reshaping the gut microbiota. This offers a safe, feasible, and cost-effective clinical pathway for non-pharmacological adjunctive therapy in chronic respiratory diseases and respiratory infections. These associations while promising highlight the need to consider the multifaceted nature of disease progression in which diet is one interacting component among genetic, immunological, and comorbid factors.
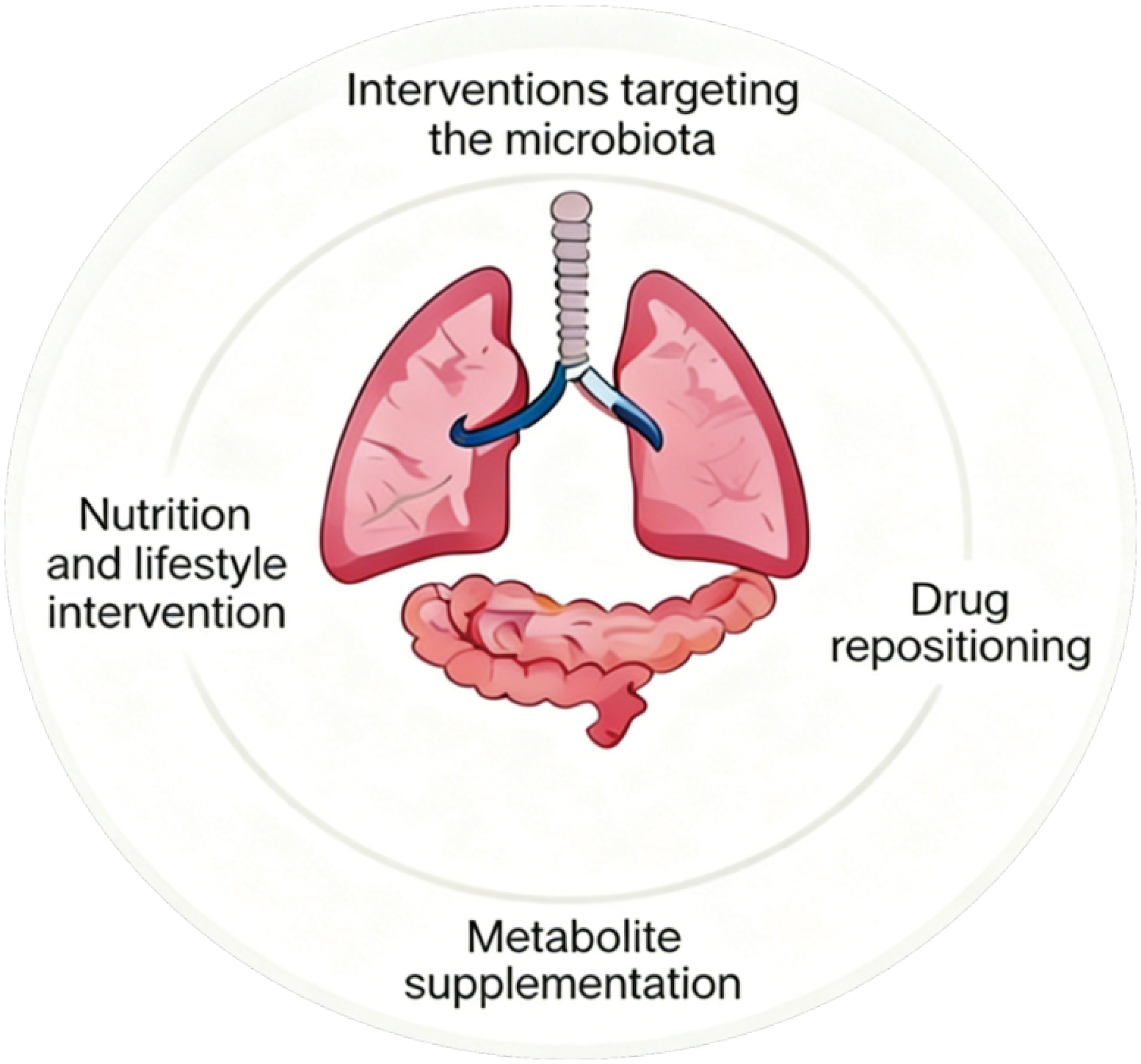
Rifaximin, as a gut-targeted antibiotic, reduces the gut Lipopolysaccharide-producing bacterial load and lowers systemic inflammatory markers in patients with COPD[110]. Curcumin reduces the risk of IL-6 storms in patients with coronavirus disease 2019 by inhibiting the nuclear factor kappa-B pathway while modulating gut microbiota composition to increase Faecalibacterium abundance[111-113]. It is recommended to develop targeted microbiota regulation strategies based on microbial profiles. Drugs like rifaximin and curcumin exert multifaceted effects through the gut-lung axis, providing a new microbiota-dependent mechanism for drug repurposing. This suggests that future clinical applications could screen for beneficiary populations based on gut microbiota, enabling low-cost, high-efficiency drug utilization. We present these strategies in Figure 1.
Comorbidity between gastrointestinal and pulmonary diseases may lead to misdiagnosis due to overlapping symp
Although a growing body of research supports the notion that gut microbiota can influence lung diseases via the gut-lung axis, direct evidence is lacking to establish whether microbial migration constitutes a causal relationship. Most studies remain at the correlational level, making it difficult to rule out confounding factors or reverse causality. For instance, some investigations in patients with ARDS or bronchiectasis have identified gut-derived bacterial genera enriched in the lungs, suggesting potential microbial migration, yet it remains unclear whether this serves as a driving factor in disease development[118]. Furthermore, whether the microbial migration observed in animal models applies to humans requires further validation.
Current intervention studies on the gut-lung axis primarily focus on probiotics, prebiotics, or fecal microbiota transplantation. However, the optimal timing for intervention, target microbial populations, and suitable populations remain unclear[119]. For instance, in chronic lung diseases like COPD or asthma, gut microbiota dysbiosis often occurs during the early stages or acute exacerbations of the disease. There is no unified standard on whether interventions should be implemented during the predisease phase, stable phase, or acute phase[120,121]. Furthermore, different disease phenotypes may correspond to distinct microbial profiles. Precisely identifying key bacterial genera and developing personalized intervention strategies remain significant challenges in current research.
Future research should integrate multi-omics technologies such as metagenomics, metabolomics, and transcriptomics to systematically elucidate the specific mechanisms by which the gut microbiota influence pulmonary diseases through metabolic products and immune regulatory pathways. For instance, previous studies using metagenomic sequencing have identified potential causal relationships between specific gut bacterial genera and pulmonary embolism risk[122], suggesting that multi-omics data can be leveraged to identify potential intervention targets.
Existing animal models are largely confined to specific bacterial strains or single time points, making it difficult to simulate the complex microbiota-host interactions observed in humans. Future efforts should focus on developing more humanized models, such as human microbiota-colonized mice and organ-on-a-chip systems[123-126], combined with dynamic sampling techniques. This approach will enable the simulation of the temporal relationship between microbiota changes and pulmonary immune responses across different disease stages, thereby providing stronger evidence for causal inference.
The application prospects of artificial intelligence and machine learning algorithms in microbiome data analysis are vast. By constructing disease-microbiome association maps and integrating clinical phenotype data, early prediction of pulmonary disease risk and personalized intervention strategies can be achieved[127,128]. For instance, bidirectional causal analysis based on Mendelian randomization methods has been employed to uncover potential causal chains linking gut microbiota to eight respiratory diseases, providing genetic evidence for clinical translation[129,130].
The gut-lung axis, serving as a vital bridge linking intestinal and pulmonary diseases, has achieved preliminary breakthroughs at the mechanistic level. However, challenges remain in causal inference and intervention strategies. Through technological integration, model optimization, and algorithm-driven approaches, future efforts are expected to advance its translation from basic research to precision medicine clinical applications. This holds promise for overcoming the limitations of traditional single-organ therapies, enabling integrated early warning, classification, and personalized regulation of gastrointestinal and respiratory comorbidities.
The gut-lung axis reveals the mechanisms of gastrointestinal and pulmonary comorbidities through multidimensional perspectives including shared immunity, the microbiome, and its metabolites, laying the foundation for cross-organ precision medicine. Current limitations include single-organ therapies, crude microbiome interventions, and the absence of biomarkers. Furthermore, most current evidence stems from cross-sectional studies or animal models, leaving insufficient robust evidence to establish causal relationships in specific clinical contexts or to determine optimal timing and targets for interventions. Research on the gut-lung axis is at a pivotal juncture, transitioning from mechanism exploration toward precision medicine. By focusing on generating robust evidence within specific clinical scenarios and developing targeted intervention tools and predictive models, we can overcome the limitations of current single organ diagnostics and therapies. This approach holds promise for achieving early warning, precise classification, and coordinated management of gastrointestinal-pulmonary comorbidities.
We appreciate the constructive comments provided by the anonymous reviewers to greatly improve the quality of this manuscript.
| 1. | Zhang F, Aschenbrenner D, Yoo JY, Zuo T. The gut mycobiome in health, disease, and clinical applications in association with the gut bacterial microbiome assembly. Lancet Microbe. 2022;3:e969-e983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 322] [Cited by in RCA: 271] [Article Influence: 67.8] [Reference Citation Analysis (0)] |
| 2. | Aykut B, Pushalkar S, Chen R, Li Q, Abengozar R, Kim JI, Shadaloey SA, Wu D, Preiss P, Verma N, Guo Y, Saxena A, Vardhan M, Diskin B, Wang W, Leinwand J, Kurz E, Kochen Rossi JA, Hundeyin M, Zambrinis C, Li X, Saxena D, Miller G. The fungal mycobiome promotes pancreatic oncogenesis via activation of MBL. Nature. 2019;574:264-267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 755] [Cited by in RCA: 675] [Article Influence: 96.4] [Reference Citation Analysis (0)] |
| 3. | Enaud R, Vandenborght LE, Coron N, Bazin T, Prevel R, Schaeverbeke T, Berger P, Fayon M, Lamireau T, Delhaes L. The Mycobiome: A Neglected Component in the Microbiota-Gut-Brain Axis. Microorganisms. 2018;6:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 80] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 4. | Szóstak N, Figlerowicz M, Philips A. The emerging role of the gut mycobiome in liver diseases. Gut Microbes. 2023;15:2211922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 5. | Chacón MR, Lozano-Bartolomé J, Portero-Otín M, Rodríguez MM, Xifra G, Puig J, Blasco G, Ricart W, Chaves FJ, Fernández-Real JM. The gut mycobiome composition is linked to carotid atherosclerosis. Benef Microbes. 2018;9:185-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 6. | Zou Y, Ge A, Lydia B, Huang C, Wang Q, Yu Y. Gut mycobiome dysbiosis contributes to the development of hypertension and its response to immunoglobulin light chains. Front Immunol. 2022;13:1089295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 7. | Shah S, Locca A, Dorsett Y, Cantoni C, Ghezzi L, Lin Q, Bokoliya S, Panier H, Suther C, Gormley M, Liu Y, Evans E, Mikesell R, Obert K, Salter A, Cross AH, Tarr PI, Lovett-Racke A, Piccio L, Zhou Y. Alterations of the gut mycobiome in patients with MS. EBioMedicine. 2021;71:103557. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 8. | Zong Z, Zhou F, Zhang L. The fungal mycobiome: a new hallmark of cancer revealed by pan-cancer analyses. Signal Transduct Target Ther. 2023;8:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 9. | Forbes JD, Bernstein CN, Tremlett H, Van Domselaar G, Knox NC. A Fungal World: Could the Gut Mycobiome Be Involved in Neurological Disease? Front Microbiol. 2018;9:3249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 10. | Wang L, Zhang K, Zeng Y, Luo Y, Peng J, Zhang J, Kuang T, Fan G. Gut mycobiome and metabolic diseases: The known, the unknown, and the future. Pharmacol Res. 2023;193:106807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 11. | Wu B, Tang Y, Zhao L, Gao Y, Shen X, Xiao S, Yao S, Qi H, Shen F. Integrated network pharmacological analysis and multi-omics techniques to reveal the mechanism of polydatin in the treatment of silicosis via gut-lung axis. Eur J Pharm Sci. 2025;207:107030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 12. | Anand S, Mande SS. Diet, Microbiota and Gut-Lung Connection. Front Microbiol. 2018;9:2147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 338] [Cited by in RCA: 298] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 13. | Budden KF, Gellatly SL, Wood DL, Cooper MA, Morrison M, Hugenholtz P, Hansbro PM. Emerging pathogenic links between microbiota and the gut-lung axis. Nat Rev Microbiol. 2017;15:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1311] [Cited by in RCA: 1150] [Article Influence: 127.8] [Reference Citation Analysis (1)] |
| 14. | Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM. Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and nonbronchoscopic "blind" bronchoalveolar lavage fluid. Am Rev Respir Dis. 1991;143:1121-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 828] [Cited by in RCA: 678] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 15. | Xia Q, Chen G, Ren Y, Zheng T, Shen C, Li M, Chen X, Zhai H, Li Z, Xu J, Gu A, Jin M, Fan L. Investigating efficacy of "microbiota modulation of the gut-lung Axis" combined with chemotherapy in patients with advanced NSCLC: study protocol for a multicenter, prospective, double blind, placebo controlled, randomized trial. BMC Cancer. 2021;21:721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 16. | Druszczynska M, Sadowska B, Kulesza J, Gąsienica-Gliwa N, Kulesza E, Fol M. The Intriguing Connection Between the Gut and Lung Microbiomes. Pathogens. 2024;13:1005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 17. | You D, Wu Y, Lu M, Shao F, Tang Y, Liu S, Liu L, Zhou Z, Zhang R, Shen S, Lange T, Xu H, Ma H, Yin Y, Shen H, Chen F, Christiani DC, Jin G, Zhao Y. A genome-wide cross-trait analysis characterizes the shared genetic architecture between lung and gastrointestinal diseases. Nat Commun. 2025;16:3032. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 18. | Wang L, Cai Y, Garssen J, Henricks PAJ, Folkerts G, Braber S. The Bidirectional Gut-Lung Axis in Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med. 2023;207:1145-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 146] [Article Influence: 48.7] [Reference Citation Analysis (1)] |
| 19. | Freuer D, Linseisen J, Meisinger C. Asthma and the risk of gastrointestinal disorders: a Mendelian randomization study. BMC Med. 2022;20:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 20. | Brassard P, Vutcovici M, Ernst P, Patenaude V, Sewitch M, Suissa S, Bitton A. Increased incidence of inflammatory bowel disease in Québec residents with airway diseases. Eur Respir J. 2015;45:962-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 21. | Rutten EP, Spruit MA, Franssen FM, Buurman WA, Wouters EF, Lenaerts K. GI symptoms in patients with COPD. Chest. 2014;145:1437-1438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Rutten EPA, Lenaerts K, Buurman WA, Wouters EFM. Disturbed intestinal integrity in patients with COPD: effects of activities of daily living. Chest. 2014;145:245-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 140] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 23. | Keely S, Hansbro PM. Lung-gut cross talk: a potential mechanism for intestinal dysfunction in patients with COPD. Chest. 2014;145:199-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Jacobsen HA, Karachalia Sandri A, Weinreich UM, Jess T, Larsen L. Increased risk of obstructive lung disease in inflammatory bowel disease: A population-based cohort study. United European Gastroenterol J. 2024;12:477-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 25. | Narayana JK, Aliberti S, Mac Aogáin M, Jaggi TK, Ali NABM, Ivan FX, Cheng HS, Yip YS, Vos MIG, Low ZS, Lee JXT, Amati F, Gramegna A, Wong SH, Sung JJY, Tan NS, Tsaneva-Atanasova K, Blasi F, Chotirmall SH. Microbial Dysregulation of the Gut-Lung Axis in Bronchiectasis. Am J Respir Crit Care Med. 2023;207:908-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 82] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 26. | Song X, Dou X, Chang J, Zeng X, Xu Q, Xu C. The role and mechanism of gut-lung axis mediated bidirectional communication in the occurrence and development of chronic obstructive pulmonary disease. Gut Microbes. 2024;16:2414805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 27. | Mackintosh JA, Desai SR, Adamali H, Patel K, Chua F, Devaraj A, Kouranos V, Kokosi M, Margaritopoulos G, Renzoni EA, Wells AU, Molyneaux PL, Kumar S, Maher TM, George PM. In patients with idiopathic pulmonary fibrosis the presence of hiatus hernia is associated with disease progression and mortality. Eur Respir J. 2019;53:1802412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 28. | Vutcovici M, Bitton A, Ernst P, Kezouh A, Suissa S, Brassard P. Inflammatory bowel disease and risk of mortality in COPD. Eur Respir J. 2016;47:1357-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 29. | Cheung TK, Lam B, Lam KF, Ip M, Ng C, Kung R, Wong BCY. Gastroesophageal reflux disease is associated with poor asthma control, quality of life, and psychological status in Chinese asthma patients. Chest. 2009;135:1181-1185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 30. | Liou A, Grubb JR, Schechtman KB, Hamilos DL. Causative and contributive factors to asthma severity and patterns of medication use in patients seeking specialized asthma care. Chest. 2003;124:1781-1788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, Delhaes L. The Gut-Lung Axis in Health and Respiratory Diseases: A Place for Inter-Organ and Inter-Kingdom Crosstalks. Front Cell Infect Microbiol. 2020;10:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 633] [Cited by in RCA: 551] [Article Influence: 91.8] [Reference Citation Analysis (2)] |
| 32. | Dang AT, Marsland BJ. Microbes, metabolites, and the gut-lung axis. Mucosal Immunol. 2019;12:843-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 854] [Cited by in RCA: 738] [Article Influence: 105.4] [Reference Citation Analysis (1)] |
| 33. | Wang H, Zhang M, Li J, Liang J, Yang M, Xia G, Ren Y, Zhou H, Wu Q, He Y, Yin J. Gut microbiota is causally associated with poststroke cognitive impairment through lipopolysaccharide and butyrate. J Neuroinflammation. 2022;19:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 100] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 34. | Bradley CP, Teng F, Felix KM, Sano T, Naskar D, Block KE, Huang H, Knox KS, Littman DR, Wu HJ. Segmented Filamentous Bacteria Provoke Lung Autoimmunity by Inducing Gut-Lung Axis Th17 Cells Expressing Dual TCRs. Cell Host Microbe. 2017;22:697-704.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 168] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 35. | Wang G, Hu YX, He MY, Xie YH, Su W, Long D, Zhao R, Wang J, Dai C, Li H, Si ZP, Cheng X, Li RM, Li Z, Yang X. Gut-Lung Dysbiosis Accompanied by Diabetes Mellitus Leads to Pulmonary Fibrotic Change through the NF-κB Signaling Pathway. Am J Pathol. 2021;191:838-856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 36. | Boesch M, Baty F, Albrich WC, Flatz L, Rodriguez R, Rothschild SI, Joerger M, Früh M, Brutsche MH. Local tumor microbial signatures and response to checkpoint blockade in non-small cell lung cancer. Oncoimmunology. 2021;10:1988403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 37. | Lou Z, Zhao H, Lyu G. [Mechanism and intervention of mucosal immune regulation based on "lung and large intestine being interior-exteriorly related" theory of traditional Chinese medicine]. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020;49:665-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 38. | Zhao M, Shao F, Yu D, Zhang J, Liu Z, Ma J, Xia P, Wang S. Maturation and specialization of group 2 innate lymphoid cells through the lung-gut axis. Nat Commun. 2022;13:7600. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 39. | Du B, Fu Y, Han Y, Sun Q, Xu J, Yang Y, Rong R. The lung-gut crosstalk in respiratory and inflammatory bowel disease. Front Cell Infect Microbiol. 2023;13:1218565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 40. | Tao J, Campbell JN, Tsai LT, Wu C, Liberles SD, Lowell BB. Highly selective brain-to-gut communication via genetically defined vagus neurons. Neuron. 2021;109:2106-2115.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 41. | Krzyzaniak MJ, Peterson CY, Cheadle G, Loomis W, Wolf P, Kennedy V, Putnam JG, Bansal V, Eliceiri B, Baird A, Coimbra R. Efferent vagal nerve stimulation attenuates acute lung injury following burn: The importance of the gut-lung axis. Surgery. 2011;150:379-389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 42. | Liu B, Huang C, Li X, Yu H, Xia Y, Liu K, You X, Wu J. The Lung Microbiome Modulates Pain-Like Behavior Via the Lung-Brain Axis in a Nitroglycerin-Induced Chronic Migraine Mouse Model. Adv Sci (Weinh). 2025;12:e2416348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 43. | Wang X, Zhang Y, Chang Y, Zhu Y, Xiang G, Chen K, Feng C, Hang Z, Li X, Zhang D. Alternating magnetic field-responsive engineered probiotics for anxiety therapy via gut-brain axis modulation. J Nanobiotechnology. 2025;23:463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 44. | Qu L, Cheng Q, Wang Y, Mu H, Zhang Y. COPD and Gut-Lung Axis: How Microbiota and Host Inflammasome Influence COPD and Related Therapeutics. Front Microbiol. 2022;13:868086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 45. | Parada Venegas D, De la Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, Harmsen HJM, Faber KN, Hermoso MA. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front Immunol. 2019;10:277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2852] [Cited by in RCA: 2563] [Article Influence: 366.1] [Reference Citation Analysis (7)] |
| 46. | Li N, Dai Z, Wang Z, Deng Z, Zhang J, Pu J, Cao W, Pan T, Zhou Y, Yang Z, Li J, Li B, Ran P. Gut microbiota dysbiosis contributes to the development of chronic obstructive pulmonary disease. Respir Res. 2021;22:274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 140] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 47. | Rooks MG, Garrett WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. 2016;16:341-352. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2930] [Cited by in RCA: 2506] [Article Influence: 250.6] [Reference Citation Analysis (1)] |
| 48. | Hakansson A, Molin G. Gut microbiota and inflammation. Nutrients. 2011;3:637-682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 383] [Cited by in RCA: 325] [Article Influence: 21.7] [Reference Citation Analysis (1)] |
| 49. | Akay HK, Bahar Tokman H, Hatipoglu N, Hatipoglu H, Siraneci R, Demirci M, Borsa BA, Yuksel P, Karakullukcu A, Kangaba AA, Sirekbasan S, Aka S, Mamal Torun M, Kocazeybek BS. The relationship between bifidobacteria and allergic asthma and/or allergic dermatitis: a prospective study of 0-3 years-old children in Turkey. Anaerobe. 2014;28:98-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 50. | Gauguet S, D'Ortona S, Ahnger-Pier K, Duan B, Surana NK, Lu R, Cywes-Bentley C, Gadjeva M, Shan Q, Priebe GP, Pier GB. Intestinal Microbiota of Mice Influences Resistance to Staphylococcus aureus Pneumonia. Infect Immun. 2015;83:4003-4014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 161] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 51. | Shreiner AB, Kao JY, Young VB. The gut microbiome in health and in disease. Curr Opin Gastroenterol. 2015;31:69-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1495] [Cited by in RCA: 1190] [Article Influence: 108.2] [Reference Citation Analysis (1)] |
| 52. | Keely S, Talley NJ, Hansbro PM. Pulmonary-intestinal cross-talk in mucosal inflammatory disease. Mucosal Immunol. 2012;5:7-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 309] [Article Influence: 22.1] [Reference Citation Analysis (1)] |
| 53. | Wang H, Liu JS, Peng SH, Deng XY, Zhu DM, Javidiparsijani S, Wang GR, Li DQ, Li LX, Wang YC, Luo JM. Gut-lung crosstalk in pulmonary involvement with inflammatory bowel diseases. World J Gastroenterol. 2013;19:6794-6804. [PubMed] [DOI] [Full Text] |
| 54. | Yazar A, Atis S, Konca K, Pata C, Akbay E, Calikoglu M, Hafta A. Respiratory symptoms and pulmonary functional changes in patients with irritable bowel syndrome. Am J Gastroenterol. 2001;96:1511-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 89] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 55. | Wang YH, Yan ZZ, Luo SD, Hu JJ, Wu M, Zhao J, Liu WF, Li C, Liu KX. Gut microbiota-derived succinate aggravates acute lung injury after intestinal ischaemia/reperfusion in mice. Eur Respir J. 2023;61:2200840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 92] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 56. | Marsland BJ, Trompette A, Gollwitzer ES. The Gut-Lung Axis in Respiratory Disease. Ann Am Thorac Soc. 2015;12 Suppl 2:S150-S156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 423] [Article Influence: 38.5] [Reference Citation Analysis (1)] |
| 57. | Jiang TT, Shao TY, Ang WXG, Kinder JM, Turner LH, Pham G, Whitt J, Alenghat T, Way SS. Commensal Fungi Recapitulate the Protective Benefits of Intestinal Bacteria. Cell Host Microbe. 2017;22:809-816.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 262] [Cited by in RCA: 222] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 58. | Charitos IA, Scacco S, Cotoia A, Castellaneta F, Castellana G, Pasqualotto F, Venneri M, Ferrulli A, Aliani M, Santacroce L, Carone M. Intestinal Microbiota Dysbiosis Role and Bacterial Translocation as a Factor for Septic Risk. Int J Mol Sci. 2025;26:2028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 59. | Nian F, Chen Y, Xia Q, Zhu C, Wu L, Lu X. Gut microbiota metabolite trimethylamine N-oxide promoted NAFLD progression by exacerbating intestinal barrier disruption and intrahepatic cellular imbalance. Int Immunopharmacol. 2024;142:113173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 34] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 60. | Dmytriv TR, Storey KB, Lushchak VI. Intestinal barrier permeability: the influence of gut microbiota, nutrition, and exercise. Front Physiol. 2024;15:1380713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 131] [Article Influence: 65.5] [Reference Citation Analysis (0)] |
| 61. | Cheng TY, Chang CC, Luo CS, Chen KY, Yeh YK, Zheng JQ, Wu SM. Targeting Lung-Gut Axis for Regulating Pollution Particle-Mediated Inflammation and Metabolic Disorders. Cells. 2023;12:901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 62. | Ye F, Li L, Wang J, Yang H. Advances in gut-lung axis research: clinical perspectives on pneumonia prevention and treatment. Front Immunol. 2025;16:1576141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 63. | Liu Y, Dai J, Zhou G, Chen R, Bai C, Shi F. Innovative Therapeutic Strategies for Asthma: The Role of Gut Microbiome in Airway Immunity. J Asthma Allergy. 2025;18:257-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 64. | Zheng H, Zhao Q, Chen J, Lu J, Li Y, Gao H. Gastrointestinal microbiome of ARDS patients induces neuroinflammation and cognitive impairment in mice. J Neuroinflammation. 2023;20:166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 65. | Orieux A, Enaud R, Imbert S, Boyer P, Begot E, Camino A, Boyer A, Berger P, Gruson D, Delhaes L, Prevel R. The gut microbiota composition is linked to subsequent occurrence of ventilator-associated pneumonia in critically ill patients. Microbiol Spectr. 2023;11:e0064123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 66. | Wang J, Hou Y, Mu L, Yang M, Ai X. Gut microbiota contributes to the intestinal and extraintestinal immune homeostasis by balancing Th17/Treg cells. Int Immunopharmacol. 2024;143:113570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 67. | Cheng H, Guan X, Chen D, Ma W. The Th17/Treg Cell Balance: A Gut Microbiota-Modulated Story. Microorganisms. 2019;7:583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 129] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 68. | Liu YJ, Tang B, Wang FC, Tang L, Lei YY, Luo Y, Huang SJ, Yang M, Wu LY, Wang W, Liu S, Yang SM, Zhao XY. Parthenolide ameliorates colon inflammation through regulating Treg/Th17 balance in a gut microbiota-dependent manner. Theranostics. 2020;10:5225-5241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 301] [Cited by in RCA: 260] [Article Influence: 43.3] [Reference Citation Analysis (1)] |
| 69. | Ziaka M, Exadaktylos A. Gut-derived immune cells and the gut-lung axis in ARDS. Crit Care. 2024;28:220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 70. | Willis KA, Siefker DT, Aziz MM, White CT, Mussarat N, Gomes CK, Bajwa A, Pierre JF, Cormier SA, Talati AJ. Perinatal maternal antibiotic exposure augments lung injury in offspring in experimental bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2020;318:L407-L418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 71. | Wang J, Zhu N, Su X, Gao Y, Yang R. Gut-Microbiota-Derived Metabolites Maintain Gut and Systemic Immune Homeostasis. Cells. 2023;12:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 445] [Cited by in RCA: 373] [Article Influence: 124.3] [Reference Citation Analysis (1)] |
| 72. | Samuelson DR, Gu M, Shellito JE, Molina PE, Taylor CM, Luo M, Welsh DA. Pulmonary immune cell trafficking promotes host defense against alcohol-associated Klebsiella pneumonia. Commun Biol. 2021;4:997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 73. | Lv J, Zhang Y, Liu S, Wang R, Zhao J. Gut-lung axis in allergic asthma: microbiota-driven immune dysregulation and therapeutic strategies. Front Pharmacol. 2025;16:1617546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 74. | Hosseinkhani F, Heinken A, Thiele I, Lindenburg PW, Harms AC, Hankemeier T. The contribution of gut bacterial metabolites in the human immune signaling pathway of non-communicable diseases. Gut Microbes. 2021;13:1-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 165] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 75. | Du Y, He C, An Y, Huang Y, Zhang H, Fu W, Wang M, Shan Z, Xie J, Yang Y, Zhao B. The Role of Short Chain Fatty Acids in Inflammation and Body Health. Int J Mol Sci. 2024;25:7379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 213] [Article Influence: 106.5] [Reference Citation Analysis (1)] |
| 76. | Albaladejo-Riad N, El Qendouci M, Cuesta A, Esteban MÁ. Ability of short-chain fatty acids to reduce inflammation and attract leucocytes to the inflamed skin of gilthead seabream (Sparus aurata L.). Sci Rep. 2024;14:31404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 77. | Nireeksha, Maniangat Luke A, Kumari N S, Hegde MN, Hegde NN. Metabolic interplay of SCFA's in the gut and oral microbiome: a link to health and disease. Front Oral Health. 2025;6:1646382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 78. | Liu XF, Shao JH, Liao YT, Wang LN, Jia Y, Dong PJ, Liu ZZ, He DD, Li C, Zhang X. Regulation of short-chain fatty acids in the immune system. Front Immunol. 2023;14:1186892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 266] [Cited by in RCA: 218] [Article Influence: 72.7] [Reference Citation Analysis (0)] |
| 79. | Cheng J, Hu H, Ju Y, Liu J, Wang M, Liu B, Zhang Y. Gut microbiota-derived short-chain fatty acids and depression: deep insight into biological mechanisms and potential applications. Gen Psychiatr. 2024;37:e101374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 96] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 80. | Li Z, Chen SX, Jiang S, Yang YN, Yan XC. Unlocking the secrets of glucose metabolism reprogramming: the role in pulmonary diseases. Front Pharmacol. 2025;16:1551452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 81. | Wang Y, Wang X, Du C, Wang Z, Wang J, Zhou N, Wang B, Tan K, Fan Y, Cao P. Glycolysis and beyond in glucose metabolism: exploring pulmonary fibrosis at the metabolic crossroads. Front Endocrinol (Lausanne). 2024;15:1379521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 82. | Corrêa RO, Castro PR, Moser R, Ferreira CM, Quesniaux VFJ, Vinolo MAR, Ryffel B. Butyrate: Connecting the gut-lung axis to the management of pulmonary disorders. Front Nutr. 2022;9:1011732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 83. | Pradana A, Sari DK, Rusda M, Tarigan AP, Wiyono WH, Soeroso NN, Eyanoer PC, Amin MM. Probiotics-derived butyric acid may suppress systemic inflammation in a murine model of chronic obstructive pulmonary disease (COPD). Narra J. 2025;5:e1332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 84. | Ahmed Z, Habib Ur Rehman M, Abdul Rehman F, Arshad U, Zeeshan M, Khan K, Jabeen M, Khar A. Association Between Gastroesophageal Reflux Disease and Chronic Respiratory Symptoms: A Systematic Review of Recent Clinical Evidence and Therapeutic Implications. Cureus. 2025;17:e91372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 85. | Yan F, Gao W, Quan X, Chen X, Ning L. Cough variant asthma with coexisting gastroesophageal reflux disease: A case report. Exp Ther Med. 2025;30:189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 86. | Wang X, Wang Y, Bu Y, Liu Y, Gong S, Che G. Association of gastroesophageal reflux disease with the incidence of pulmonary disease. Front Cell Dev Biol. 2025;13:1552126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 87. | Mallah N, Turner JM, González-Barcala FJ, Takkouche B. Gastroesophageal reflux disease and asthma exacerbation: A systematic review and meta-analysis. Pediatr Allergy Immunol. 2022;33:e13655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 88. | De Nuccio F, Piscitelli P, Toraldo DM. Gut-lung Microbiota Interactions in Chronic Obstructive Pulmonary Disease (COPD): Potential Mechanisms Driving Progression to COPD and Epidemiological Data. Lung. 2022;200:773-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 89. | Mederos MA, Reber HA, Girgis MD. Acute Pancreatitis: A Review. JAMA. 2021;325:382-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 822] [Cited by in RCA: 669] [Article Influence: 133.8] [Reference Citation Analysis (1)] |
| 90. | Schuijt TJ, Lankelma JM, Scicluna BP, de Sousa e Melo F, Roelofs JJ, de Boer JD, Hoogendijk AJ, de Beer R, de Vos A, Belzer C, de Vos WM, van der Poll T, Wiersinga WJ. The gut microbiota plays a protective role in the host defence against pneumococcal pneumonia. Gut. 2016;65:575-583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 724] [Cited by in RCA: 646] [Article Influence: 64.6] [Reference Citation Analysis (0)] |
| 91. | Lin F, Lu R, Han D, Fan Y, Zhang Y, Pan P. A prediction model for acute respiratory distress syndrome among patients with severe acute pancreatitis: a retrospective analysis. Ther Adv Respir Dis. 2022;16:17534666221122592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 92. | Shao BX, He B, Liu HM, Shao DB, Sun ZR, Lin JF, Zhang W, Ren Y, Nie SN. [Multivariate regression analysis for severe acute pancreatitis patients with ARDS]. 2016 China Poisoning Treatment Capital Forum and the 8th National Academic Conference on Poisoning and Critical Care Treatment; 2016; Beijing, China. China: Chinese Society of Toxicology, 2016: 267-270. [DOI] [Full Text] |
| 93. | Hite RD, Grier BL, Waite BM, Veldhuizen RA, Possmayer F, Yao LJ, Seeds MC. Surfactant protein B inhibits secretory phospholipase A2 hydrolysis of surfactant phospholipids. Am J Physiol Lung Cell Mol Physiol. 2012;302:L257-L265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 94. | Zhou K, Lu J. Progress in cytokine research for ARDS: A comprehensive review. Open Med (Wars). 2024;19:20241076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 95. | Wang Z, Li F, Liu J, Luo Y, Guo H, Yang Q, Xu C, Ma S, Chen H. Intestinal Microbiota - An Unmissable Bridge to Severe Acute Pancreatitis-Associated Acute Lung Injury. Front Immunol. 2022;13:913178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 96. | Xu H, Sheng S, Luo W, Xu X, Zhang Z. Acute respiratory distress syndrome heterogeneity and the septic ARDS subgroup. Front Immunol. 2023;14:1277161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 88] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 97. | Takedani Y, Nakamura T, Fukiwake N, Imada T, Mashino J, Morimoto T. Clinical characteristics and factors related to antibiotic-associated diarrhea in elderly patients with pneumonia: a retrospective cohort study. BMC Geriatr. 2021;21:317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 98. | Slimings C, Riley TV. Antibiotics and healthcare facility-associated Clostridioides difficile infection: systematic review and meta-analysis 2020 update. J Antimicrob Chemother. 2021;76:1676-1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 99. | Lin X, Yu Z, Liu Y, Li C, Hu H, Hu JC, Liu M, Yang Q, Gu P, Li J, Nandakumar KS, Hu G, Zhang Q, Chen X, Ma H, Huang W, Wang G, Wang Y, Huang L, Wu W, Liu NN, Zhang C, Liu X, Zheng L, Chen P. Gut-X axis. Imeta. 2025;4:e270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 56] [Article Influence: 56.0] [Reference Citation Analysis (2)] |
| 100. | Hawkings MJ, Vaselli NM, Charalampopoulos D, Brierley L, Elliot AJ, Buchan I, Hungerford D. A Systematic Review of the Prevalence of Persistent Gastrointestinal Symptoms and Incidence of New Gastrointestinal Illness after Acute SARS-CoV-2 Infection. Viruses. 2023;15:1625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 101. | Zhang J, Garrett S, Sun J. Gastrointestinal symptoms, pathophysiology, and treatment in COVID-19. Genes Dis. 2021;8:385-400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 102. | Georgakopoulou VE, Tarantinos K, Papalexis P, Spandidos DA, Damaskos C, Gkoufa A, Chlapoutakis S, Sklapani P, Trakas N, Mermigkis D. Role of pulmonary function testing in inflammatory bowel diseases (Review). Med Int (Lond). 2022;2:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 103. | Ge H, Hong K, Fan C, Zhang J, Li X, Zhang H, Qiu A. Knowledge, attitude, and practice of healthcare providers on chronic refractory cough: A cross-sectional study. Heliyon. 2024;10:e27564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 104. | Zhang Y, Wan Y, Xin X, Qiao Y, Qiao W, Ping J, Su J. Signals from intestinal microbiota mediate the crosstalk between the lung-gut axis in an influenza infection mouse model. Front Immunol. 2024;15:1435180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 105. | Yang J, He Y, Ai Q, Liu C, Ruan Q, Shi Y. Lung-Gut Microbiota and Tryptophan Metabolites Changes in Neonatal Acute Respiratory Distress Syndrome. J Inflamm Res. 2024;17:3013-3029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 106. | Alves MCS, Rego MS, Silva RCCD, Silva RA, Arruda IES, Paiva-Júnior SSL, Balbino VQ. Gut Microbiota and COVID-19: Unraveling the Gut-Lung Axis and Immunomodulatory Therapies. ACS Infect Dis. 2025;11:1844-1853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 107. | Arslan S, Bozkurt C, Bulut H. High Adherence To The Mediterranean Diet Could Prevent COPD Symptoms And Frailty In Elderly. Clin Nutr ESPEN. 2023;54:501-502. [DOI] [Full Text] |
| 108. | Budden KF, Shukla SD, Bowerman KL, Vaughan A, Gellatly SL, Wood DLA, Lachner N, Idrees S, Rehman SF, Faiz A, Patel VK, Donovan C, Alemao CA, Shen S, Amorim N, Majumder R, Vanka KS, Mason J, Haw TJ, Tillet B, Fricker M, Keely S, Hansbro N, Belz GT, Horvat J, Ashhurst T, van Vreden C, McGuire H, Fazekas de St Groth B, King NJC, Crossett B, Cordwell SJ, Bonaguro L, Schultze JL, Hamilton-Williams EE, Mann E, Forster SC, Cooper MA, Segal LN, Chotirmall SH, Collins P, Bowman R, Fong KM, Yang IA, Wark PAB, Dennis PG, Hugenholtz P, Hansbro PM. Faecal microbial transfer and complex carbohydrates mediate protection against COPD. Gut. 2024;73:751-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 109. | Amadieu C, Coste V, Neyrinck AM, Thijssen V, Leyrolle Q, Bindels LB, Piessevaux H, Stärkel P, de Timary P, Delzenne NM, Leclercq S. Restoring an adequate dietary fiber intake by inulin supplementation: a pilot study showing an impact on gut microbiota and sociability in alcohol use disorder patients. Gut Microbes. 2022;14:2007042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 110. | Broekhuizen R, Wouters EF, Creutzberg EC, Schols AM. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax. 2006;61:17-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 263] [Cited by in RCA: 249] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 111. | Zhu J, He L. The Modulatory Effects of Curcumin on the Gut Microbiota: A Potential Strategy for Disease Treatment and Health Promotion. Microorganisms. 2024;12:642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 112. | Servida S, Piontini A, Gori F, Tomaino L, Moroncini G, De Gennaro Colonna V, La Vecchia C, Vigna L. Curcumin and Gut Microbiota: A Narrative Overview with Focus on Glycemic Control. Int J Mol Sci. 2024;25:7710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 113. | Hu J, Lang H, Fan D, Wen T, Shi J, Xiao C, Li Y, Kang C, Shi P, Shen L, Lin N. Curcumin supplementation accelerates high-altitude acclimatization, prevents polycythemia and modulates gut microbiota in male Han population: a randomized controlled trial. Front Nutr. 2025;12:1572376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 114. | He Y, Wen Q, Yao F, Xu D, Huang Y, Wang J. Gut-lung axis: The microbial contributions and clinical implications. Crit Rev Microbiol. 2017;43:81-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 218] [Article Influence: 24.2] [Reference Citation Analysis (1)] |
| 115. | Watanabe T, Fujiwara Y, Chan FKL. Current knowledge on non-steroidal anti-inflammatory drug-induced small-bowel damage: a comprehensive review. J Gastroenterol. 2020;55:481-495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 116. | Utzeri E, Usai P. Role of non-steroidal anti-inflammatory drugs on intestinal permeability and nonalcoholic fatty liver disease. World J Gastroenterol. 2017;23:3954-3963. [PubMed] [DOI] [Full Text] |
| 117. | Dora D, Szőcs E, Soós Á, Halasy V, Somodi C, Mihucz A, Rostás M, Mógor F, Lohinai Z, Nagy N. From bench to bedside: an interdisciplinary journey through the gut-lung axis with insights into lung cancer and immunotherapy. Front Immunol. 2024;15:1434804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 118. | Zhu J, Huang Z, Lin Y, Zhu W, Zeng B, Tang D. Intestinal-pulmonary axis: a 'Force For Good' against respiratory viral infections. Front Immunol. 2025;16:1534241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 119. | Manoharan S, Iyappan O, Prabahar A, Bhasuran B, Raja K. Gut-lung axis microbiome: Towards precision medicine in respiratory disorders (Review). World Acad Sci J. 2025;7:1-15. [DOI] [Full Text] |
| 120. | Guo J, Yang L. Regulation effect of the intestinal flora and intervention strategies targeting the intestinal flora in alleviation of pulmonary fibrosis development. Biosci Microbiota Food Health. 2024;43:293-299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 121. | Jang YO, Lee SH, Choi JJ, Kim DH, Choi JM, Kang MJ, Oh YM, Park YJ, Shin Y, Lee SW. Fecal microbial transplantation and a high fiber diet attenuates emphysema development by suppressing inflammation and apoptosis. Exp Mol Med. 2020;52:1128-1139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 90] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 122. | Cen L, Qin L, Chen W, Wei L, Tang C, Teng X, Tian Z. Causal Relationship between Gut Microbiota and Pulmonary Embolism: An Analysis Using Mendelian Randomization. Pol J Microbiol. 2025;74:153-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 123. | Ballerini M, Galiè S, Tyagi P, Catozzi C, Raji H, Nabinejad A, Macandog ADG, Cordiale A, Slivinschi BI, Kugiejko KK, Freisa M, Occhetta P, Wargo JA, Ferrucci PF, Cocorocchio E, Segata N, Vignati A, Morgun A, Deleidi M, Manzo T, Rasponi M, Nezi L. A gut-on-a-chip incorporating human faecal samples and peristalsis predicts responses to immune checkpoint inhibitors for melanoma. Nat Biomed Eng. 2025;9:967-984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 34] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 124. | Miller CP, Shin W, Ahn EH, Kim HJ, Kim DH. Engineering Microphysiological Immune System Responses on Chips. Trends Biotechnol. 2020;38:857-872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 125. | Quan Y, Yin Z, Chen S, Lang J, Han L, Yi J, Zhang L, Yue Q, Tian W, Chen P, Du S, Wang J, Dai Y, Hua H, Zeng J, Li L, Zhao J. The gut-lung axis: Gut microbiota changes associated with pulmonary fibrosis in mouse models induced by bleomycin. Front Pharmacol. 2022;13:985223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 126. | Eain MMG, Baginska J, Greenhalgh K, Fritz JV, Zenhausern F, Wilmes P. Engineering Solutions for Representative Models of the Gastrointestinal Human-Microbe Interface. Engineering. 2017;3:60-65. [DOI] [Full Text] |
| 127. | McDowell A, Kang J, Yang J, Jung J, Oh YM, Kym SM, Shin TS, Kim TB, Jee YK, Kim YK. Machine-learning algorithms for asthma, COPD, and lung cancer risk assessment using circulating microbial extracellular vesicle data and their application to assess dietary effects. Exp Mol Med. 2022;54:1586-1595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 128. | Dickson RP, Schultz MJ, van der Poll T, Schouten LR, Falkowski NR, Luth JE, Sjoding MW, Brown CA, Chanderraj R, Huffnagle GB, Bos LDJ; Biomarker Analysis in Septic ICU Patients (BASIC) Consortium. Lung Microbiota Predict Clinical Outcomes in Critically Ill Patients. Am J Respir Crit Care Med. 2020;201:555-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 256] [Article Influence: 42.7] [Reference Citation Analysis (1)] |
| 129. | Zhou X, Shen S, Wang Z. Genetic evidence of bidirectional mendelian randomization study on the causality between gut microbiome and respiratory diseases contributes to gut-lung axis. Sci Rep. 2024;14:25550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 130. | Jin Q, Ren F, Dai D, Sun N, Qian Y, Song P. The causality between intestinal flora and allergic diseases: Insights from a bi-directional two-sample Mendelian randomization analysis. Front Immunol. 2023;14:1121273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 82] [Article Influence: 27.3] [Reference Citation Analysis (0)] |