Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.114335
Revised: October 17, 2025
Accepted: January 4, 2026
Published online: March 21, 2026
Processing time: 180 Days and 23.5 Hours
Polyethylene glycol (PEG), a globally prevalent laxative, is extensively applied for bowel preparation prior to colonoscopy or abdominal surgery due to its exce
To investigate whether PEG increases the risk of intestinal infection, and whether Lactobacillus acidophilus (LAC) can mitigate the adverse effects and thereby reduce the risk of subsequent enteric infections.
To investigate this, we employed a murine model infected with Citrobacter roden
The severity of infectious enteritis was assessed, and the prophylactic efficacy of LAC supplementation was evaluated. Our findings indicate that PEG administration significantly elevated CR load, enhanced virulence gene expression, and exacerbated intestinal inflammation post-infection, with the infection window extending approximately 14 days. Through fecal metagenomic analysis and co-housing experiments, we demonstrated that PEG-associated enteritis is critically mediated by gut microbiota dysbiosis. Furthermore, supplementation with LAC was shown to mitigate susceptibility to CR infection.
Our results suggest that PEG administration microbiota-dependently increases intestinal infection susceptibility, highlighting the therapeutic potential of LAC in restoring microbial homeostasis.
Core Tip: Although polyethylene glycol is a relevant compound for clinical applications, recent studies suggest that it could potentially alter the colonic microbiota composition, thereby opening ways for opportunistic pathogen colonization and invasion. Therefore, we aimed to shed light to these questions by infecting a murine model with Citrobacter rodentium, a surrogate for enteropathogenic Escherichia coli and enterohemorrhagic Escherichia coli. Furthermore, we investigated whether Lactobacillus acidophilus administration could potentially protect the colon microbiota against such possible alteration and pathogen invasion.
- Citation: Kou GJ, Shen J, Li LX, Zuo XL, Li YQ. Lactobacillus acidophilus attenuates polyethylene glycol-induced susceptibility to Citrobacter rodentium infection via microbiota modulation. World J Gastroenterol 2026; 32(11): 114335
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/114335.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.114335
Due to its exceptional safety profile, polyethylene glycol (PEG) is widely used for comprehensive bowel preparation preceding colonoscopy or abdominal surgery[1]. PEG consumption reportedly alters significantly the composition of the intestinal microbiota[2]. Specifically, PEG administration modifies the α- and β-diversity of the intestinal microbiota within 6 days, increasing the prevalence of mucus-associated bacteria (e.g., Akkermansia or Bacteroides) and reducing Firmicutes abundance after 14 days[3]. However, another study indicated that gut microbiotic abundance was 34.7-fold below the baseline levels in volunteers upon PEG-induced bowel cleansing, with recovery to optimal levels in approximately two weeks[4]. These findings underscore the necessity of further assessment related to how PEG affects the intestinal microbiota and bacterial recovery rate.
A previously published meta-analysis revealed that laxative-induced alterations in the intestinal flora are comparable to those induced by proton-pump inhibitors[5]. Proton-pump inhibitors reportedly correlated positively with an in
Enteropathogenic Escherichia coli (EPEC) and enterohemorrhagic Escherichia coli (EHEC) are primary pathogenic bacteria responsible for infectious enteritis in humans[7]. Citrobacter rodentium (CR) is a rodent-specific enteric pathogen capable of adhering to the colonic mucosa and forming attachment and effacing lesions, including colonic crypt hy
Lactobacillus acidophilus (LAC) is among the most widely used probiotics with a demonstrated potential for not only ameliorating gut dysbiosis, as proved by reduced Firmicutes-to-Bacteroidetes ratios and endotoxin-bearing Gram-negative bacterial levels, but also maintaining intestinal barrier integrity and reducing inflammation[14]. These properties suggest that LAC might play a protective role in mitigating the adverse effects of PEG-induced microbiota alterations and subsequent enteric infections.
Although PEG is a relevant compound for clinical applications, recent studies suggest that it could potentially alter the colonic microbiota composition, thereby opening ways for opportunistic pathogen colonization and invasion. Despite the relevance of this possibility, the potential of this phenomenon and its underlying mechanisms are poorly understood. Therefore, this study focused on shedding light to these questions by infecting a murine model with CR, a surrogate for EPEC and EHEC, following PEG-induced bowel preparation, and investigating how LAC administration could potentially protect the colon microbiota against such possible alteration and pathogen invasion.
Wild-type C57BL/6 male mice (6-7 weeks old) were obtained from Jicui Biological Technology Co., Ltd. (Nanjing, Jiangsu Province, China). The animals were housed under specific pathogen-free conditions, maintained at a controlled temperature of 23 ± 1 °C and relative humidity of 45% ± 5%. Following a one-week acclimatization period, the mice were used for experimental procedures. All animal experiments were conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee of Qilu Hospital, Shandong University (Approval No. DWLL-2021-058).
Intestinal cleansing was performed using 2.1 mL of PEG electrolyte solution (Heshuang, Wanhe Pharmaceutical, Shenzhen, Guangdong Province, China), containing PEG 4000 as active ingredient. Control groups received 2.1 mL of sterile distilled water. Administration was performed via oral gavage with 300 μL aliquots of the respective solutions at 30-minute intervals, followed by a 4-hour fasting period.
CR strain DBS 100 was cultured overnight in Luria-Bertani broth at 37 °C under aerobic conditions. Bacterial cells were harvested by 10-minute centrifugation at 4000 × g and 4 °C. The pellet was washed twice with phosphate buffered saline (PBS) through sequential centrifugation (4000 × g, 3 minutes) and resuspended in sterile PBS. Mice were infected via oral gavage with 200 μL PBS containing 1 × 109 colony-forming units (CFUs) of CR.
For bacterial quantification, fecal samples were collected at designated time points post-infection and homogenized in PBS. Tissue samples (including those of the colon, mesenteric lymph nodes, spleen, and hepatic parenchyma), were aseptically collected and weighed. Tissue homogenates and fecal suspensions (30-100 μL) were plated on MacConkey agar and incubated at 37 °C for 14-16 hours. CR colonies were enumerated and expressed as CFUs per gram of tissue or fecal material.
Following PEG administration, mice were orally gavaged daily for 3 consecutive days with either 1.86 × 106 CFU of LAC (Hangzhou Grand Pharmaceutical Co., Ltd., Zhejiang Province, China) suspended in 200 μL PBS or 200 μL of PBS alone (designated as the PBS control group). On post-treatment day 3, all animals underwent standardized CR infection protocol as previously described. The animals were divided into a control (n = 5) and a PEG (n = 5) experimental group.
For microbial genomic profiling, freshly voided fecal specimens were aseptically collected in RNase/DNase-free microcentrifuge tubes. Subsequently, the samples were flash-frozen in liquid nitrogen prior to storage at -80 °C. Total bacterial genomic DNA was purified using the Stool Genomic Plus DNA extraction kit (Juhemei Biotechnology Co., Ltd., Guangdong Province, China) following the manufacturer’s instructions.
Concurrently, distal colonic mucosal biopsies were homogenized in an RNA isolation reagent (Vazyme, R401-01, Nanjing, Jiangsu Province, China), with subsequent RNA integrity verification using agarose gel electrophoresis. RNA quantification was performed using a NanoDrop™ 2000 spectrophotometer (Thermo Fisher Scientific, MA, United States). First-strand cDNA synthesis was carried out using 1 μg total RNA with HiScript III RT SuperMix for quantitative real-time polymerase chain reaction (qPCR) (+gDNA wiper) (Vazyme, R323-01, Nanjing, Jiangsu Province, China), incorporating genomic DNA elimination steps.
Quantitative PCR analyses were conducted on a StepOnePlus™ Real-Time PCR System (Thermo Fisher; 4376600, MA, United States) with the following reaction parameters: 95 °C for 60 seconds, followed by 40 cycles of 95 °C for 15 seconds, 60 °C for 15 seconds and 72 °C for 45 seconds. The reaction mixtures (20 μL total volume) contained: 10 μL 2 × AceQ® Universal SYBR qPCR Master Mix (Vazyme, Q511-02, Nanjing, Jiangsu Province, China), 1 μL cDNA template, 0.8 μL of each of the gene-specific forward/reverse primers (Table 1), and 7.4 μL DEPC-treated water. Table 1 summarizes the primers used for qPCR. RrsA (16s RNA) was used as the internal standard in order to normalize target gene expressions. All reactions were performed in technical triplicates, determining relative quantifications by the comparative 2-∆∆Ct method.
| Genes | Forward | Reverse |
| RrsA (16s RNA) | 5’-AGGCCTTCGGGTTGTAAAGT-3’ | 5’-ATTCCGATTAACGCTTGCAC-3’ |
| Ler | 5’-AATATACCTGATGGTGCTCTTG-3’ | 5’-TTCTTCCATTCAATAATGCTTCTT-3’ |
| Tir | 5’-TACACATTCGGTTATTCAGCAG-3’ | 5’-GACATCCAACCTTCAGCATA-3’ |
| Gapdh | 5’-CATGGCCTCCAAGGAGTAAG-3’ | 5’-CCTAGGCCCCTCCTGTTATT-3’ |
| IFNγ | 5’-CAGCAACAGCAAGGCGAAAAAGG-3’ | 5’-TTTCCGCTTCCTGAGGCTGGAT-3’ |
| TNFα | 5’-CAGGCGGTGCCTATGTCTC-3’ | 5’-CGATCACCCCGAAGTTCAGTAG-3’ |
| IL-17A | 5’-CCCTGGACTCTCCACCGCAA-3’ | 5’-TCCCTCCGCATTGACACAGC-3’ |
Approximately 1 cm of distal colon tissue was fixed in 10% neutral buffered formalin at room temperature for 24 hours. Following fixation, the tissue samples underwent a standardized dehydration series, paraffin embedding, and sectioning at a thickness of 4-μm. Sections were stained with hematoxylin and eosin using established protocols. Blinded histopathological scoring was performed by two independent observers, adhering to the criteria outlined in a previously validated scoring system[15].
To assess how PEG affects goblet cell dynamics, mice were euthanized 12 hours post-PEG bowel preparation. Distal colonic tissues were fixed in Carnoy’s fixative (containing 60% methanol, 30% glacial acetic acid, and 10% chloroform) at 4 °C for 24 hours, followed by three washes in 70% ethanol. Paraffin-embedded tissues were sectioned at 5-μm thickness and stained with Periodic Acid-Schiff’s stain to visualize mucin-containing goblet cells. Goblet cells were quantified by analyzing approximately 25 longitudinally oriented crypts per animal using light microscopy (400 × magnification). The data represent biological replicates from 5-7 animals per experimental group.
Fecal pellets (n = 3 per mouse) were collected aseptically and stored at -80 °C until DNA extraction, followed by 16S rRNA sequencing analyses were conducted on the Majorbio Cloud Platform (Shanghai Majorbio Bio-Pharm Technology Co., Ltd., Shanghai, China). Microbial genomic DNA was isolated using the E.Z.N.A.® Soil DNA Kit (Omega Bio-Tek, Norcross, GA, United States). The hypervariable V3-V4 regions of the 16S rRNA gene were PCR-amplified using universal primers 338F (5’-ACTCCTACGGGAGGCAGCAG-3’) and 806R (5’-GGACTACHVGGGTWTCTAAT-3’). Amplicons were sequenced using the Illumina MiSeq platform (2 × 300 bp paired-end; Illumina, San Diego, CA, United States). Raw sequences were processed using UPARSE (v7.1) with a 97% similarity threshold for operational taxonomic unit (OTU) clustering. Taxonomic classification was performed against the Greengenes database.
The data are represented as the mean ± SD. Statistical analyses were conducted using two-tailed unpaired t-tests implemented in GraphPad Prism 8 (GraphPad Software, San Diego, CA, United States). The threshold for the statistical significance between the experimental groups was defined as follows: aP < 0.05, bP < 0.01, and cP < 0.001.
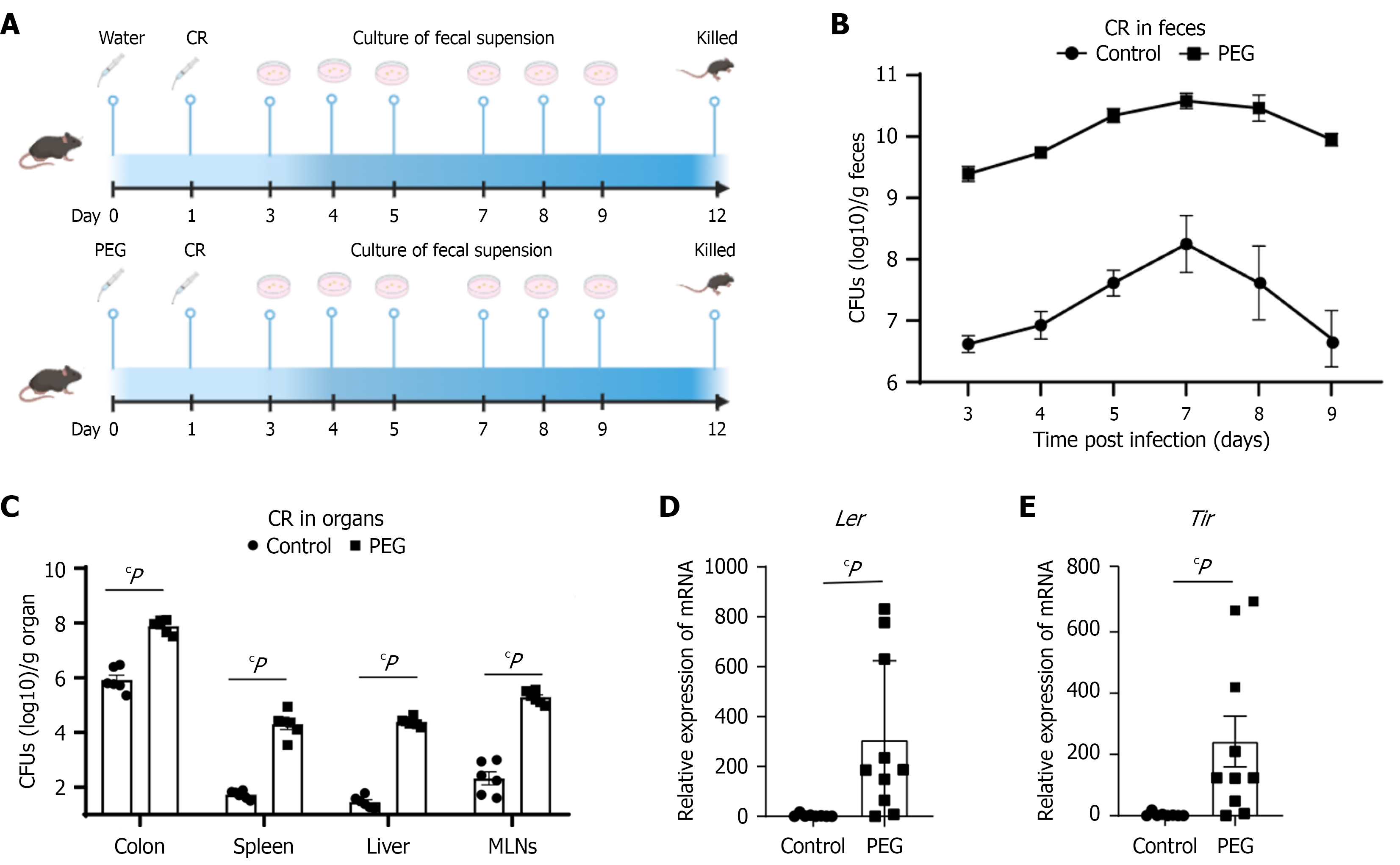
To investigate the impact of PEG electrolyte solution on CR infection, wild-type C57BL/6J mice were orally inoculated with CR following 24-hour pretreatment with PEG. Quantitative analysis revealed that PEG-administered mice maintained significantly elevated fecal CR burdens (> 109 CFU/g) from 3-9 days post-infection (d.p.i.), peaking on day 7 (Figure 1A). In contrast, control animals exhibited approximately 107 CFU/g fecal bacterial loads with a similar peak timing (Figure 1B).
Given the established association between intestinal barrier dysfunction and systemic bacterial translocation during enteric infections, we performed a comprehensive analysis of bacterial colonization patterns across multiple organ systems. PEG-treated mice exhibited significantly increased CR loads in the colonic tissues (P < 0.01), hepatic parenchyma (P < 0.001), splenic tissue (P < 0.01), and mesenteric lymph nodes (P < 0.05) compared to the untreated controls (Figure 1C). To investigate potential PEG-mediated enhancement of CR pathogenicity, we quantified transcriptional expression of key virulence regulators Ler and Tir in fecal samples at 5 d.p.i. using qRT-PCR. Notably, PEG administration induced significant upregulation of both Ler (91.34-fold increase, P < 0.001) and Tir (70.87-fold increase, P < 0.001) gene expression relative to the control (Figure 1D and E).
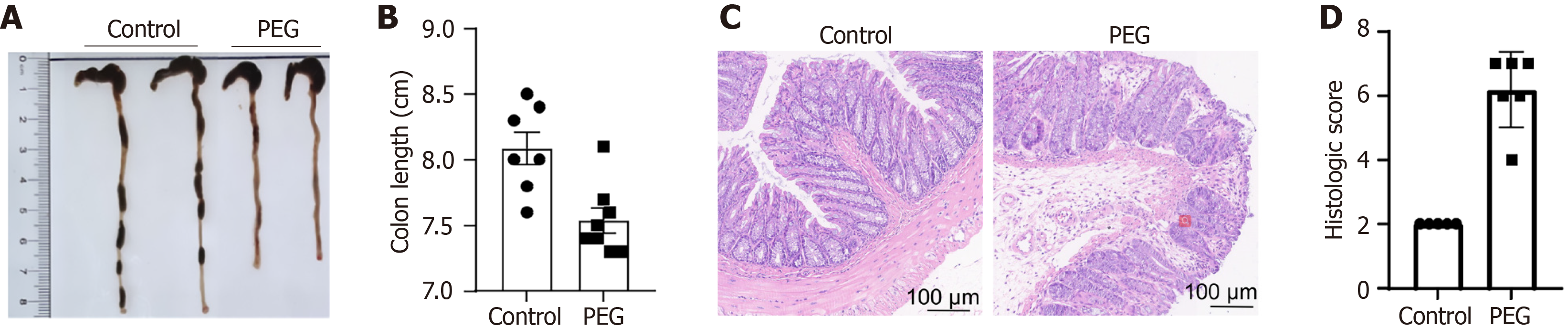
Next, we performed histopathological assessment at 12 d.p.i., revealing pronounced colonic shortening in PEG-treated mice compared to the controls (8.09 ± 0.33 vs 7.54 ± 0.27, P < 0.01; Figure 2A and B). Microscopic evaluation demonstrated exacerbated inflammatory infiltrates and severe crypt architectural disruption in the distal colonic mucosa of PEG-exposed animals, with significantly increased histopathological scores (6.17 ± 1.17 vs 2.00 ± 0.58, P < 0.001; Figure 2C and D).
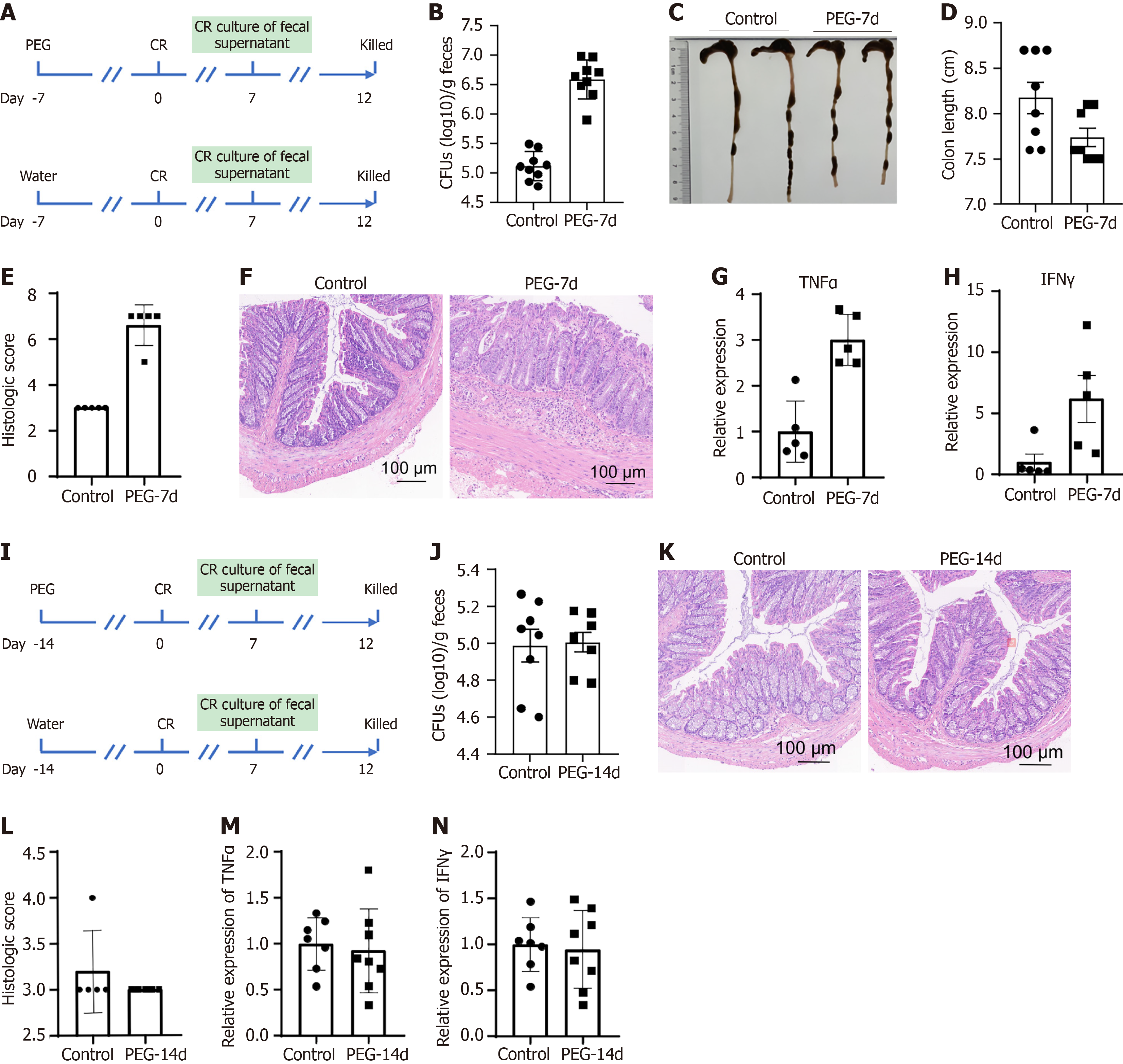
To temporally characterize PEG-induced intestinal vulnerability, we performed experimental CR challenges on days 7 or 14 following PEG-mediated bowel preparation. We assessed bacterial shedding kinetics through fecal CFU counts at 7 d.p.i. and evaluated disease severity parameters (colon morphology, histopathology, and inflammatory markers) at 12 d.p.i. (Figure 3A).
Mice infected at 7 days post-PEG treatment displayed enhanced infection severity, manifesting as significantly increased fecal bacterial loads (P < 0.001; Figure 3B), marked colonic shortening (P < 0.01; Figure 3C and D), and increased histopathological scores (P < 0.001; Figure 3E and F). Quantitative PCR analysis revealed substantial proinflammatory cytokine expression upregulation in the PEG-7-day group, with 3.0-fold (P < 0.001) and 6.2-fold (P < 0.05) increases in tumor necrosis factor-α and interferon-γ mRNA levels, respectively, compared to the controls (Figure 3G and H). These results demonstrate prolonged intestinal susceptibility to CR infection through 7 days post-PEG exposure.
Notably, animals challenged at post-PEG administration day 14 displayed complete infection susceptibility resolution, with no significant differences in fecal bacterial burden (Figure 3I and J), colonic histopathology scores (Figure 3K and L), or cytokine expression profiles (Figure 3M and N) compared to the untreated controls. This temporal pathogen susceptibility resolution suggests the full restoration of intestinal barrier homeostasis and microbial resistance mechanisms by day 14 following PEG-induced bowel preparation.
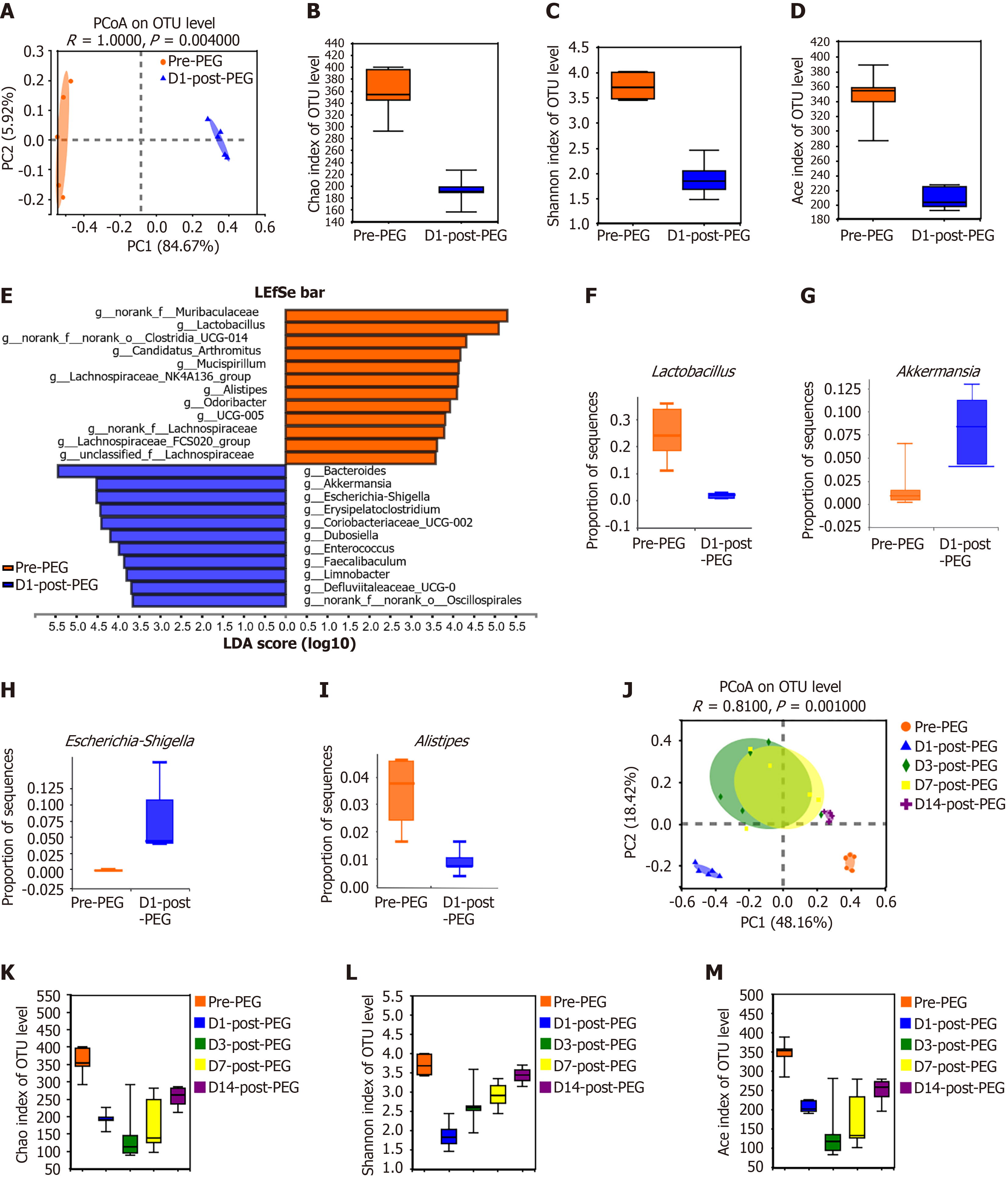
Given that intestinal cleansing demonstrably perturbs gut microbiota, we conducted the comparative analysis of fecal samples collected from mice at the baseline (pre-PEG) and post-PEG day 1 using 16S rRNA gene sequencing. Principal coordinate analysis at the OTU level revealed distinct clustering patterns between the pre- and post-PEG groups (Figure 4A). ANOSIM analysis confirmed significant intergroup differences in the microbial community structure (r = 1.0, P = 0.004). Alpha diversity metrics demonstrated substantial reduction upon PEG administration, with significant reduction in the Chao (P < 0.001), Shannon (P < 0.001), and ACE (P < 0.001) indices at the OTU level (Figure 4B-D).
Linear discriminant analysis Effect Size analysis identified differential taxonomic enrichment between the groups: Pre-PEG microbiota showed significant representation of Muribaculaceae, Clostridia, Mucispirillum, Candidatus arthromitus, Lachnospiraceae, and Alistipes, while post-PEG communities exhibited a predominance of Bacteroides, Akkermansia, Escherichia-Shigella, Erysipelatoclostridium, Coriobacteriaceae, Enterococcus, Faecalibaculum, and Limnobacter (Figure 4E-I). To assess longitudinal recovery of microbial communities, we collected fecal samples at pre-PEG as well as 1-, 3-, 7-, and 14-day post-PEG intervals. Both β- and α-diversity metrics demonstrated gradual temporal restoration, indicating partial microbiota recovery by day 14 (Figure 4J-M).
The macromolecular mucin MUC2, predominantly secreted by goblet cells, constitutes the colonic mucus layer. Our histological evaluation revealed significant goblet cell density increases upon PEG preparation compared to the baseline (P < 0.001; Supplementary Figure 1). This cellular proliferation suggests the potential activation of negative feedback mechanisms to accelerate mucin production and restore mucosal barrier integrity.
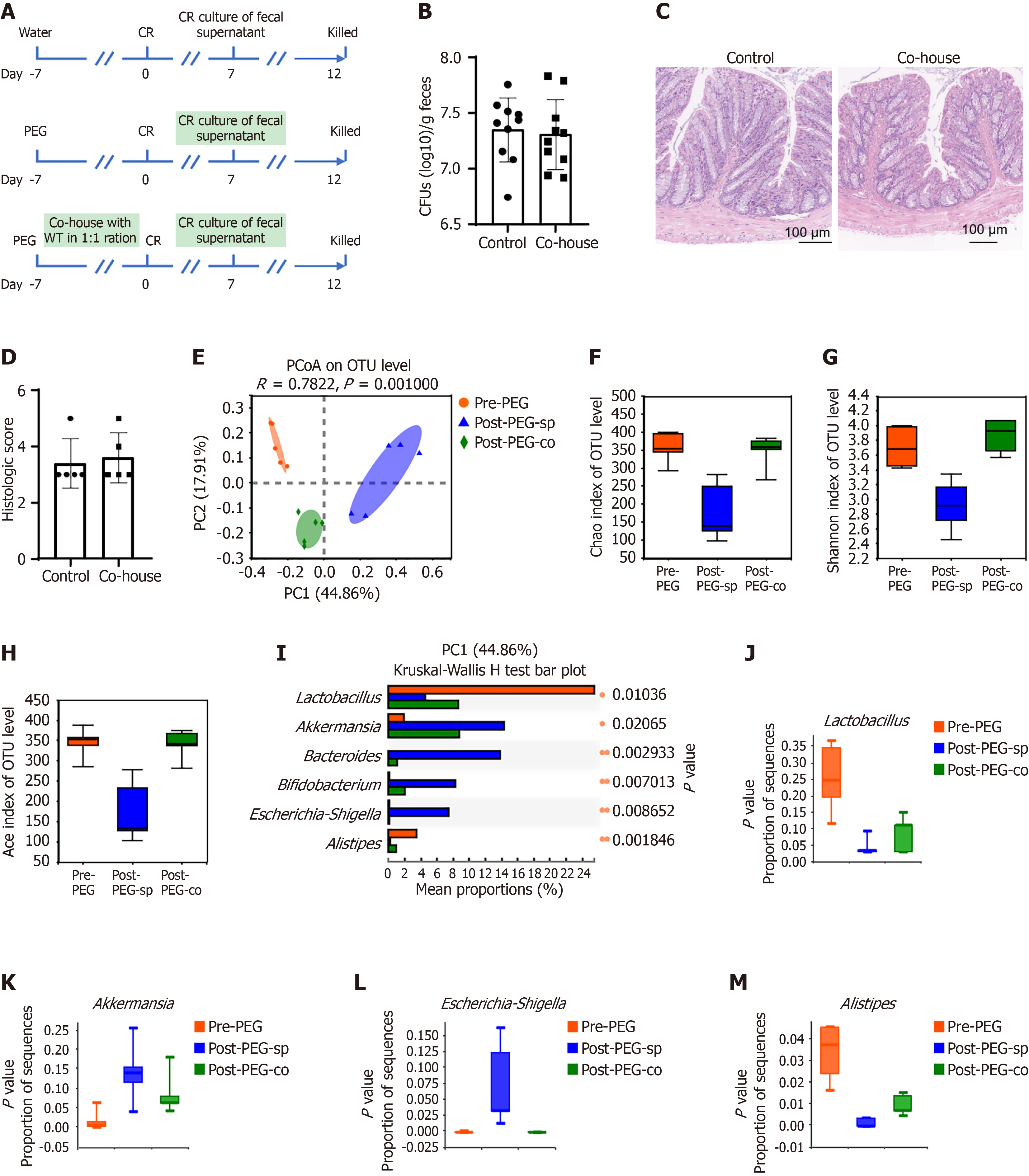
To investigate whether PEG-induced gut microbiota alterations mediate increased CR infection susceptibility, we performed a co-housing experiment of PEG-treated and untreated mice in a 1:1 ratio for 7 days prior to CR challenge (Figure 5A). Notably, co-housed mice exhibited no significant differences in fecal CR colonization (Figure 5B) or colonic histopathological scores (Figure 5C and D) compared to the PEG-treated and control groups.
16S rRNA sequencing analysis of microbial communities revealed distinct clustering among the pre-PEG baseline, post-PEG separately housed, and post-PEG co-housed mouse groups (Figure 5E). Co-housed mice exhibited microbiota profiles that converged toward pre-PEG baseline states compared to the separately housed animals. Alpha diversity metrics including the Shannon, ACE, and Chao indices demonstrated complete restoration of microbial richness in the co-housed mice to pre-PEG levels (Figure 5F-H). Taxonomic analysis identified significant microbiotic remodeling upon co-housing, characterized by increased relative abundance of Lactobacillales (P < 0.01) and Alistipes (P < 0.05), coupled with marked reduction in Escherichia-Shigella populations (P < 0.001; Figure 5I-M).
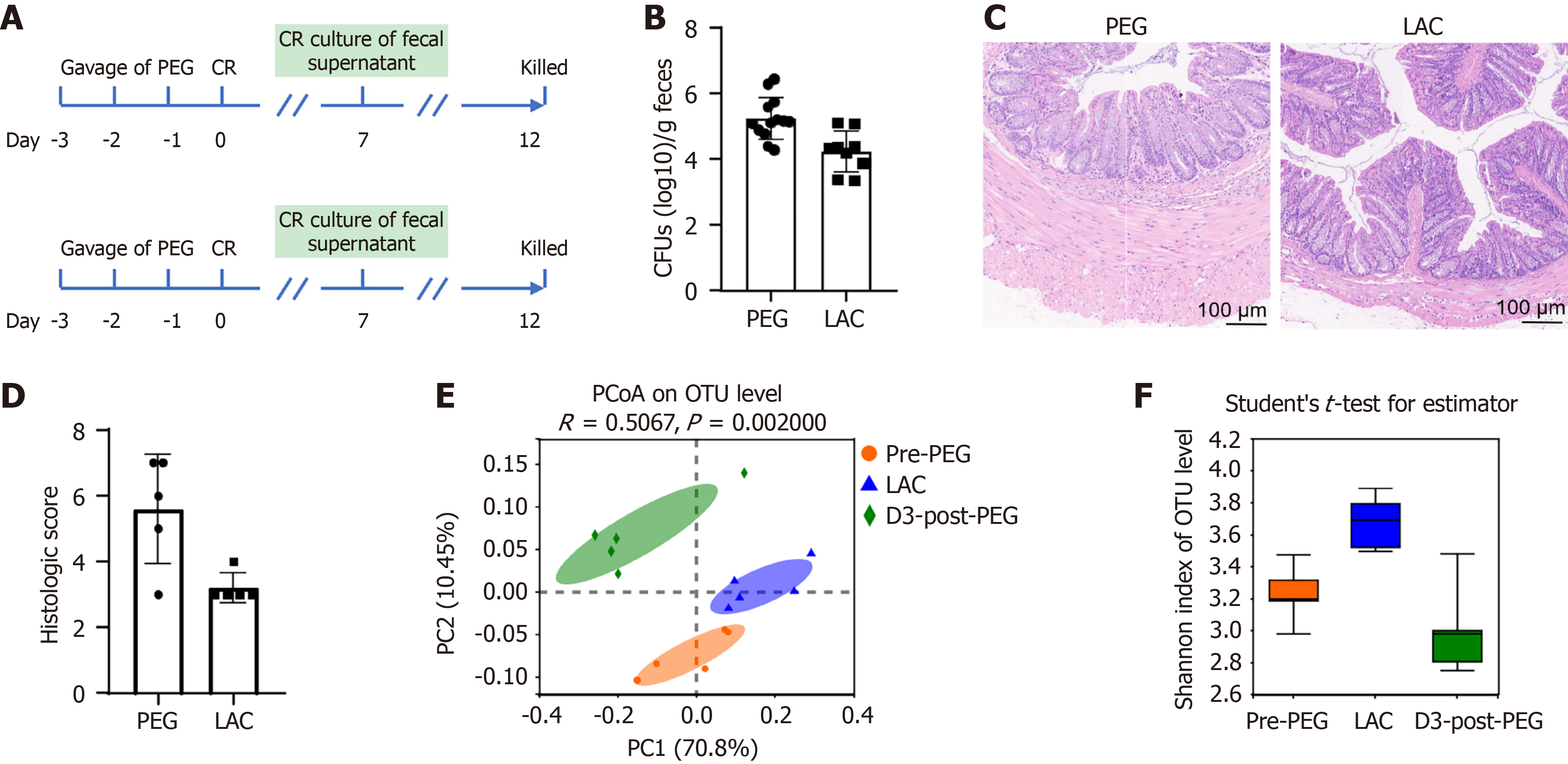
Finally, given the observed depletion of Lactobacillus upon PEG preparation, we investigated whether prophylactic LAC administration could affect CR susceptibility. Mice received 1.86 × 106 CFU/day LAC or PBS control for 3 days prior to PEG treatment (Figure 6A). LAC pretreatment significantly reduced fecal CR burdens compared to the controls (P < 0.001; Figure 6B) and improved colonic inflammation highlighted by the reduced histopathological scores (P < 0.05; Figure 6C and D).
Our microbiota analysis demonstrated distinct cluster separation among the groups, with LAC-treated mice exhibiting a distribution closer to the pre-PEG baseline compared to the PEG-only controls (Figure 6E). In addition, based on the Shannon index (P < 0.001), LAC administration significantly enhanced microbial diversity (Figure 6F). Taken together, LAC supplementation regulates the gut microbiota and mitigates PEG-induced CR infections.
Adequate bowel cleansing remains essential for both diagnostic colonoscopy efficacy and positive surgical outcomes. While PEG-based solutions are widely considered safe and effective for this purpose[1,16], their potential impact on enteric pathogen susceptibility remains poorly understood. In this study, we demonstrated that PEG-induced bowel preparation increases intestinal infection risk through altering the intestinal microbiota as well as impairing intestinal barriers. Notably, prophylactic LAC administration mitigated these effects by restoring microbial homeostasis and reducing inflammatory responses.
Low-dose PEG (0.4 g/kg) might suggestibly inhibit CR adhesion to the colonic epithelium, highlighting its therapeutic potential against enteropathogens such as EPEC and EHEC[17]. In contrast, our results reveal that high-dose PEG administration paradoxically enhances bacterial colonization and virulence expression. Mechanistically, PEG-induced diarrhea-related rapid electrolyte depletion and osmotic imbalance likely disrupt intestinal homeostasis, thereby creating a permissive microenvironment for pathogen proliferation[18]. PEG consumption, washing out the luminal content, led to intestinal fluid and osmolality disturbances. The mucus layer is an important component of intestinal barrier functions. Defects of the mucus layer allow bacteria to reach epithelial cells more often than normal, thereby activating the immune system[3]. It has been established that the application of large amounts of PEG leads to thinning of the mucus layer and requires a longer time for recovery[4]. Taken together, the substantial depletion of intestinal fluids and concomitant reduction in mucus layer integrity were identified as critical determinants of enhanced infection susceptibility.
Consistent with established infection kinetics, both control and PEG-treated cohorts exhibited comparable fecal CR colonization patterns, reaching peak burdens at 7-8 d.p.i. followed by gradual clearance. This temporal similarity suggests that PEG exacerbates infection severity without altering CR pathogenesis. Our microbiota analysis revealed transient but significant PEG-induced dysbiosis, with full compositional restoration by post-treatment day 14. This 14-day “infection vulnerability window” correlated with dynamic fluctuations in mucin-associated taxa. Specifically, we observed transient post-PEG enrichment of Akkermansia, a genus positively associated with goblet cell density and barrier reinforcement in murine models[20-22]. This compensatory response might represent a host-protective mechanism counteracting PEG-induced mucosal damage. Published studies have demonstrated a significant reduction in Akkermansia abundance following a 6-day low-dose PEG administration protocol, with subsequent recovery upon treatment cessation. Notably, in the present study, we identified a marked increase in Akkermansia relative abundance within the gut microbiota 24 hours post-PEG intervention. This microbial shift exhibited a significant positive correlation with increased goblet cell proliferation, suggesting a potential protective mechanism applied by the host organism to maintain intestinal mucosal homeostasis during osmotic challenge.
The therapeutic potential of LAC supplementation deserves particular emphasis. As well-characterized probiotics, Lactobacillus spp. enhance epithelial integrity through multiple mechanisms such as tight junction modulation, antimicrobial peptide induction, and Wnt/β-catenin-mediated epithelial regeneration[23-25]. Our discoveries are in good agreement with these mechanisms, demonstrating that LAC pretreatment reverses PEG-induced Lactobacillus depletion, reduces CR colonization, and attenuates colitis severity. Meanwhile, a controlled clinical study found that the administration of LAC-containing probiotics following colonoscopy led to a significant reduction in the number of days with abdominal pain compared to the placebo group[26]. It is important to note that invasive Lactobacillus infections, though rare, have been well-documented. This risk carries significant weight for immunocompromised hosts, such as individuals with human immunodeficiency virus, prosthetic heart valves, immunosuppressive therapy, and diabetes mellitus are now prevalent among a substantial portion of the general population[27,28].
While murine models provide precious mechanistic insights into the underlying mechanisms, clinical translation requires the validation of such discoveries in human cohorts. We acknowledge the limitations of our study, including the absence of large-scale clinical correlation data: A critical next step given the widespread use of PEG in clinical practice. Nevertheless, our results propose a pragmatic preventive strategy: Short-term probiotic supplementation during bowel preparation protocols might reduce infection risks in vulnerable populations.
In summary, this study establishes that PEG bowel preparation transiently increases intestinal infection susceptibility through microbiota disruption and barrier impairment. The key discoveries of this study are the following: PEG induces a 14-day vulnerability window characterized by dysbiosis and mucosal thinning. Lactobacillus depletion mediates enhanced CR colonization post-PEG. Prophylactic LAC administration restores microbial balance and reduces pathogen burden.
We thank Prof. Shi-Yang Li, Meng-Qi Zheng and Zi-Xiao Zhao for providing guidance and antibodies.
| 1. | Hassan C, Ponchon T, Bisschops R, van Hooft JE, Messmann H, Gralnek IM, Dinis-Ribeiro M. European Society of Gastrointestinal Endoscopy (ESGE) Publications Policy - Update 2020. Endoscopy. 2020;52:123-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (2)] |
| 2. | ASGE Technology Committee; Mamula P, Adler DG, Conway JD, Diehl DL, Farraye FA, Kantsevoy SV, Kaul V, Kethu SR, Kwon RS, Rodriguez SA, Tierney WM. Colonoscopy preparation. Gastrointest Endosc. 2009;69:1201-1209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 46] [Article Influence: 2.7] [Reference Citation Analysis (3)] |
| 3. | Tropini C, Moss EL, Merrill BD, Ng KM, Higginbottom SK, Casavant EP, Gonzalez CG, Fremin B, Bouley DM, Elias JE, Bhatt AS, Huang KC, Sonnenburg JL. Transient Osmotic Perturbation Causes Long-Term Alteration to the Gut Microbiota. Cell. 2018;173:1742-1754.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 188] [Article Influence: 23.5] [Reference Citation Analysis (4)] |
| 4. | Jalanka J, Salonen A, Salojärvi J, Ritari J, Immonen O, Marciani L, Gowland P, Hoad C, Garsed K, Lam C, Palva A, Spiller RC, de Vos WM. Effects of bowel cleansing on the intestinal microbiota. Gut. 2015;64:1562-1568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 198] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 5. | van der Wulp MY, Derrien M, Stellaard F, Wolters H, Kleerebezem M, Dekker J, Rings EH, Groen AK, Verkade HJ. Laxative treatment with polyethylene glycol decreases microbial primary bile salt dehydroxylation and lipid metabolism in the intestine of rats. Am J Physiol Gastrointest Liver Physiol. 2013;305:G474-G482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Park YH, Seong JM, Cho S, Han HW, Kim JY, An SH, Gwak HS. Effects of proton pump inhibitor use on risk of Clostridium difficile infection: a hospital cohort study. J Gastroenterol. 2019;54:1052-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Knutton S, Baldwin T, Williams PH, McNeish AS. Actin accumulation at sites of bacterial adhesion to tissue culture cells: basis of a new diagnostic test for enteropathogenic and enterohemorrhagic Escherichia coli. Infect Immun. 1989;57:1290-1298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 666] [Cited by in RCA: 678] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 8. | Garmendia J, Frankel G, Crepin VF. Enteropathogenic and enterohemorrhagic Escherichia coli infections: translocation, translocation, translocation. Infect Immun. 2005;73:2573-2585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 305] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 9. | Crepin VF, Collins JW, Habibzay M, Frankel G. Citrobacter rodentium mouse model of bacterial infection. Nat Protoc. 2016;11:1851-1876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 87] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 10. | Mundy R, MacDonald TT, Dougan G, Frankel G, Wiles S. Citrobacter rodentium of mice and man. Cell Microbiol. 2005;7:1697-1706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 402] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 11. | Kamada N, Kim YG, Sham HP, Vallance BA, Puente JL, Martens EC, Núñez G. Regulated virulence controls the ability of a pathogen to compete with the gut microbiota. Science. 2012;336:1325-1329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 503] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 12. | Barba J, Bustamante VH, Flores-Valdez MA, Deng W, Finlay BB, Puente JL. A positive regulatory loop controls expression of the locus of enterocyte effacement-encoded regulators Ler and GrlA. J Bacteriol. 2005;187:7918-7930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 119] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 13. | Kamada N, Chen G, Núñez G. A complex microworld in the gut: Harnessing pathogen-commensal relations. Nat Med. 2012;18:1190-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 14. | Kang Y, Kang X, Yang H, Liu H, Yang X, Liu Q, Tian H, Xue Y, Ren P, Kuang X, Cai Y, Tong M, Li L, Fan W. Lactobacillus acidophilus ameliorates obesity in mice through modulation of gut microbiota dysbiosis and intestinal permeability. Pharmacol Res. 2022;175:106020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 177] [Article Influence: 44.3] [Reference Citation Analysis (0)] |
| 15. | Erben U, Loddenkemper C, Doerfel K, Spieckermann S, Haller D, Heimesaat MM, Zeitz M, Siegmund B, Kühl AA. A guide to histomorphological evaluation of intestinal inflammation in mouse models. Int J Clin Exp Pathol. 2014;7:4557-4576. [PubMed] |
| 16. | Pall H, Zacur GM, Kramer RE, Lirio RA, Manfredi M, Shah M, Stephen TC, Tucker N, Gibbons TE, Sahn B, McOmber M, Friedlander J, Quiros JA, Fishman DS, Mamula P. Bowel preparation for pediatric colonoscopy: report of the NASPGHAN endoscopy and procedures committee. J Pediatr Gastroenterol Nutr. 2014;59:409-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (2)] |
| 17. | Qi W, Joshi S, Weber CR, Wali RK, Roy HK, Savkovic SD. Polyethylene glycol diminishes pathological effects of Citrobacter rodentium infection by blocking bacterial attachment to the colonic epithelia. Gut Microbes. 2011;2:267-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Guandalini S. Intestinal ion and nutrient transport in health and infectious diarrhoeal diseases. Drugs. 1988;36 Suppl 4:26-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 19. | Johansson ME, Gustafsson JK, Holmén-Larsson J, Jabbar KS, Xia L, Xu H, Ghishan FK, Carvalho FA, Gewirtz AT, Sjövall H, Hansson GC. Bacteria penetrate the normally impenetrable inner colon mucus layer in both murine colitis models and patients with ulcerative colitis. Gut. 2014;63:281-291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 582] [Cited by in RCA: 797] [Article Influence: 66.4] [Reference Citation Analysis (5)] |
| 20. | Plovier H, Everard A, Druart C, Depommier C, Van Hul M, Geurts L, Chilloux J, Ottman N, Duparc T, Lichtenstein L, Myridakis A, Delzenne NM, Klievink J, Bhattacharjee A, van der Ark KC, Aalvink S, Martinez LO, Dumas ME, Maiter D, Loumaye A, Hermans MP, Thissen JP, Belzer C, de Vos WM, Cani PD. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat Med. 2017;23:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1899] [Cited by in RCA: 1619] [Article Influence: 179.9] [Reference Citation Analysis (6)] |
| 21. | Shin NR, Lee JC, Lee HY, Kim MS, Whon TW, Lee MS, Bae JW. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut. 2014;63:727-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1394] [Cited by in RCA: 1233] [Article Influence: 102.8] [Reference Citation Analysis (2)] |
| 22. | Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci U S A. 2013;110:9066-9071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4060] [Cited by in RCA: 3511] [Article Influence: 270.1] [Reference Citation Analysis (5)] |
| 23. | Hou Q, Ye L, Liu H, Huang L, Yang Q, Turner JR, Yu Q. Lactobacillus accelerates ISCs regeneration to protect the integrity of intestinal mucosa through activation of STAT3 signaling pathway induced by LPLs secretion of IL-22. Cell Death Differ. 2018;25:1657-1670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 353] [Cited by in RCA: 313] [Article Influence: 39.1] [Reference Citation Analysis (0)] |
| 24. | Zheng Y, Zhao J, Nie X, Chitrakar B, Gao J, Sang Y. Mutual adhesion of Lactobacillus spp. to intestinal cells: A review of perspectives on surface layer proteins and cell surface receptors. Int J Biol Macromol. 2024;282:137031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 25. | Fernández-Ciganda S, Fraga M, Zunino P. Probiotic Lactobacilli Administration Induces Changes in the Fecal Microbiota of Preweaned Dairy Calves. Probiotics Antimicrob Proteins. 2022;14:804-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | D'Souza B, Slack T, Wong SW, Lam F, Muhlmann M, Koestenbauer J, Dark J, Newstead G. Randomized controlled trial of probiotics after colonoscopy. ANZ J Surg. 2017;87:E65-E69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 27. | Rossi F, Amadoro C, Gasperi M, Colavita G. Lactobacilli Infection Case Reports in the Last Three Years and Safety Implications. Nutrients. 2022;14:1178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 28. | Haghighat L, Crum-Cianflone NF. The potential risks of probiotics among HIV-infected persons: Bacteraemia due to Lactobacillus acidophilus and review of the literature. Int J STD AIDS. 2016;27:1223-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |