Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.114243
Revised: November 21, 2025
Accepted: January 8, 2026
Published online: March 21, 2026
Processing time: 183 Days and 0.4 Hours
Metabolically dysfunction-associated steatotic liver disease (MASLD) has emer
To determine the prevalence of MASLD, identify risk factors, and evaluate genetic influences in the Egyptian population.
In this multicenter observational cross-sectional study (2019-2024), 1060 adults from 22 governorates underwent a comprehensive assessment, including anthropometrics, metabolic profiling, dietary evaluation (validated Food Frequency Questionnaire), abdominal ultrasonography, vibration-controlled transient elastography (FibroScan®), and PNPLA3 rs738409 genotyping. MASLD was diagnosed according to the European Association for the Study of the Liver 2024 guidelines.
An alarmingly high prevalence of MASLD (67.7%) was revealed. Obesity confer
Egypt faces an epidemic of MASLD reflecting complex interactions among me
Core Tip: Egypt faces the world’s highest reported metabolic dysfunction associated steatotic liver disease prevalence (67.7%), driven by synergistic metabolic, dietary, and genetic factors. Obesity (27.5 times the risk), insulin resistance, high intake of refined carbohydrates, and PNPLA3 variants (present in 48% of cases) create a perfect meta
- Citation: Hegazy M, Fathy M, Ashoush O, Abdelshafy S, Abdelfatah D, Saad S, Abd El-Moniem S, Ibrahim W, Ashraf O, Saleh S, Ahmed T, Adly R, Abdel-Wahab A, Ahmed A, Hussein S, Othman A, Abdelghani A. Egypt’s metabolic dysfunction associated steatotic liver disease: Genetic and dietary nexus, shaping artificial intelligence driven healthier future. World J Gastroenterol 2026; 32(11): 114243
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/114243.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.114243
Metabolically dysfunction-associated steatotic liver disease (MASLD) is the most prevalent chronic liver disease globally, affecting over 30% of the population as of 2023. Its incidence is rising in parallel with epidemics of type 2 diabetes, obesity, and cardiometabolic dysfunction. MASLD significantly increases the risk of progressive liver disease, including liver failure and hepatocellular carcinoma, as well as extra-hepatic complications like cardiovascular events and chronic kidney disease[1]. While approximately 10%-30% of isolated steatosis cases progress to steatohepatitis, this risk escalates dramatically to 65% in patients with type 2 diabetes, underscoring MASLD as a critical public health challenge[2].
Globally, MASLD prevalence is estimated at 25%-30%, but significant geographic, ethnic, and socioeconomic disparities exist. Middle Eastern and North African populations, in particular, are frequently underrepresented in large epidemiological studies, obscuring the true burden and unique drivers of the disease in these regions[3]. In Egypt, a perfect storm of dietary, lifestyle, and genetic factors has converged to create a high-risk environment. The primary causes of MASLD are deeply rooted in a shift towards physical inactivity and diets high in refined carbohydrates and added sugars, compounded by a high prevalence of obesity, which currently affects approximately 7.09 million males and 12.7 million females[4]. Specifically, common dietary practices that drive MASLD include the high consumption of refined grains (e.g., Egyptian traditional baladi bread), sugar-sweetened beverages, and traditional sweets, which create a persistent state of positive energy balance and de novo lipogenesis.
This metabolic burden is unevenly distributed. Egypt’s unique ethnic diversity encompassing groups such as Abazas, Turks, Greeks, Bedouin Arabs, and Dom clans suggests a potential role for genetic predisposition. Furthermore, poverty is concentrated in Upper Egypt (27.8% poverty, 28.7% at risk), raising critical questions about how MASLD prevalence varies across governorates, socioeconomic classes, and genetic backgrounds. Key genetic variants in genes such as PNPLA3, TM6SF2, and GCKR are known to modulate hepatic fat accumulation and fibrosis risk[5].
This is where genetic testing holds significant preventive potential. By identifying individuals with high-risk genetic profiles, healthcare providers can implement stratified prevention strategies. For a patient carrying a risky PNPLA3 variant, for instance, even a moderate degree of overweight or sugar consumption may warrant earlier and more intensive lifestyle intervention, personalized dietary counseling, and vigilant monitoring. This proactive approach allows for the precise targeting of resources to those who would benefit most, moving beyond a one-size-fits-all public health model.
Therefore, this study aims to elucidate the genetic-dietary nexus of MASLD in Egypt by mapping prevalent dietary practices against genetic risk factors to provide a foundational dataset for developing artificial intelligence (AI)-driven, personalized prevention strategies to mitigate the disease’s escalating burden.
This cross-sectional observational multicenter study was conducted across 22 Egyptian governorates from March 2019 to February 2024, stratified into Upper Egypt, Middle Egypt, and Lower Egypt to ensure socioeconomic diversity. The study enrolled asymptomatic adults (18-70 years) with body mass index (BMI) ≥ 18.5 kg/m2, excluding alcohol consumers, pregnant women, and individuals with other chronic liver disease (like Wilson’s, autoimmune hepatitis)[6]. Participants included medical personnel, university staff and students, and their relatives from four university hospitals and Ministry of Health facilities.
This study was approved by Cairo University’s Ethics Committee (No. N-41-2019) and registered on ClinicalTrials.gov (No. NCT04081571). Regarding data collection, the collected information included anthropometrics [height, weight, waist circumference (WC), and BMI], demographics (age and gender), lifestyle factors assessed using the International Physical Activity Questionnaire[7], and dietary history evaluated with a validated, semi-quantitative Food Frequency Ques
VCTE using FibroScan® Mini+ 430 was employed to assess both liver stiffness and steatosis. Liver stiffness mea
Fasting blood samples (10 mL) were processed for multiple analyses. Serum analysis (4 mL) included metabolic markers [cholesterol, high-density lipoprotein (HDL), triglycerides (TGs), glucose, and insulin for Homeostasis Model Assessment of Insulin Resistance (HOMA-IR)][14], liver enzymes (aspartate aminotransferase, alanine aminotransferase, gamma-glutamyl transferase, and bilirubin), inflammatory marker [C-reactive protein (CRP)], infectious markers (hepatitis C virus antibody and hepatitis B surface antigen), and thyroid hormones [thyrotropin (TSH), free thyroxine (FT) 3, and FT4]. Glycated hemoglobin (HbA1c) was measured from 2 mL EDTA blood using the Tosoh HLC-723G8 HPLC system[15]. Enzyme-linked immunosorbent assay assays were performed on 2 mL serum to determine L-carnitine (Sunlong Biotech) and lipoprotein lipase (LPL) (Sunred Biological Technology) levels. Genomic DNA was extracted from 2 mL EDTA blood using the EasyPure® kit, and purity was confirmed with NanoDrop™ (260/280 ratio = 1.8)[16]. The PNPLA3 rs738409 (I148M) variant was genotyped, a well-established genetic risk factor for MASLD severity and progression[17].
To assess the prevalence of MASLD in Egypt, a prospective cross-sectional observational study was conducted by a certified biomedical statistician. Based on previous studies, the expected prevalence was 37%. For a 99.9% confidence interval with a margin of error of ± 5% from the expected proportion, the required sample size was calculated to be 1010. To account for a 5% non-response rate, the final sample size was increased to 1060. Epi Info software was used for sample size estimation[18].
Statistical Package for the Social Sciences v27 was used for analysis. Numerical data were expressed as mean ± SD or median (range), and categorical data as n (%). Normality was tested via the Kolmogorov-Smirnov and Shapiro-Wilk tests. Comparisons were conducted as follows: (1) Categorical variables were analyzed using the χ2 test or Fisher’s exact test; and (2) Continuous variables were analyzed using the t-test or Mann-Whitney U test for two groups and analysis of variance or the Kruskal-Wallis test for more than two groups, with Bonferroni post hoc analysis.
Spearman’s correlation coefficients were calculated to assess the associations between non-normally distributed numerical variables (r value ranges from -1 to 1, indicating the strength and direction of the relationship). Variables with P < 0.10 in univariate analysis were entered into a stepwise logistic regression model to evaluate the independent effect of factors on MASLD risk. Logistic regression was employed to estimate adjusted odds ratios and 95%CI to quantify the association between risk factors and MASLD. The 95%CI not including 1.0 was considered statistically significant. All tests were two-tailed, and a P value ≤ 0.05 was considered significant[19].
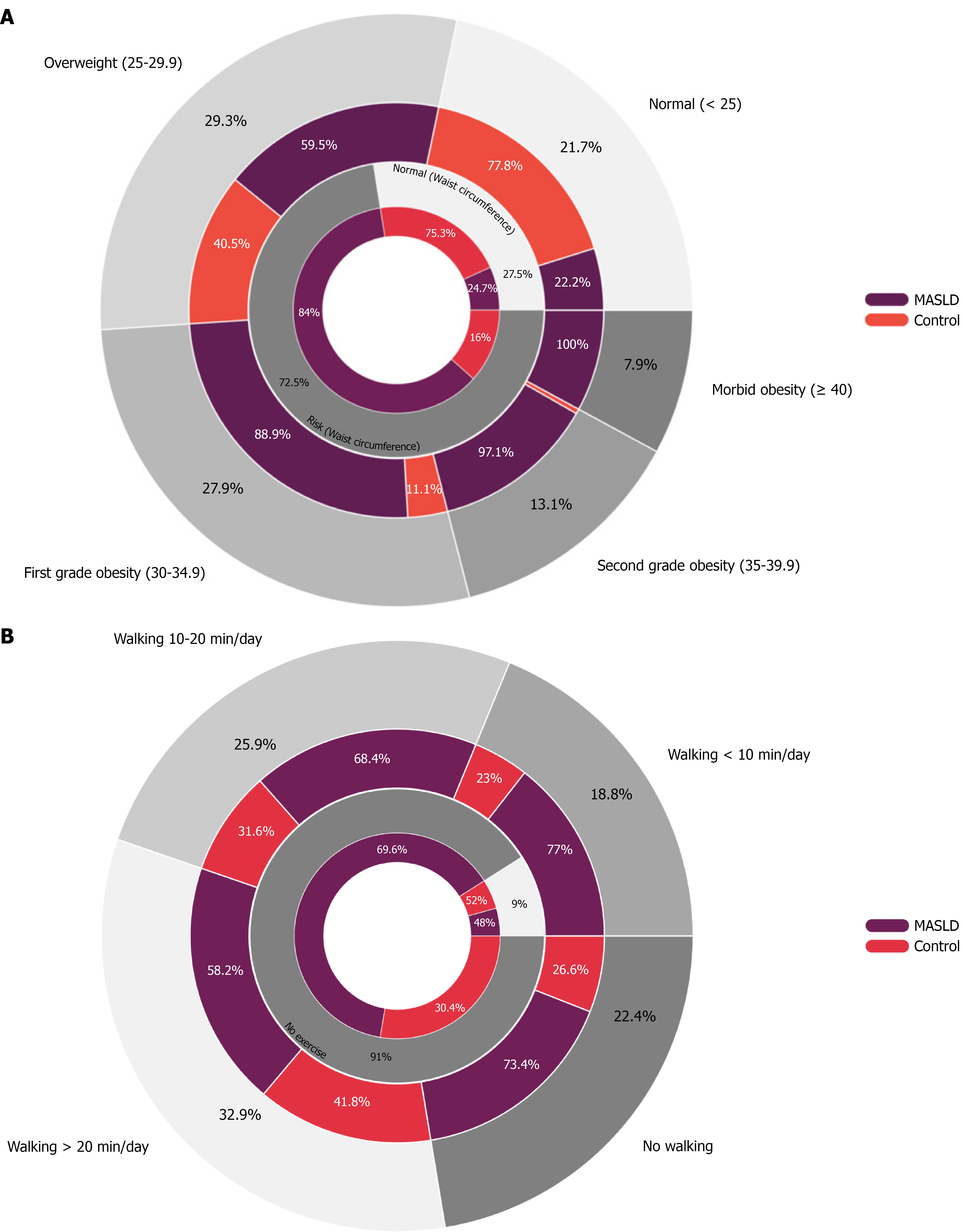
The study included 1060 participants (545 females, 515 males; mean age 38 ± 11 years), of whom 718 (67.7%) were diagnosed with MASLD. MASLD patients were older (41 ± 11 years vs 32 ± 9 years, P < 0.001) and had higher BMIs (30.2 ± 6.3 kg/m2) and WCs (96 ± 14 cm) compared with healthy individuals. Overall, 78.3% were overweight or obese, including 7.9% with morbid obesity. The geographic distribution of MASLD across Egypt was not significant (P = 0.809). Both BMI and WC were significantly higher among MASLD participants, as illustrated in Figure 1A.
Physical activity was predominantly walking (77.6%), while only 9% engaged in regular sports. Healthy participants were more active (14.3% sports, 42.7% walking ≥ 20 minute/day) than MASLD patients (6.8% sports, 28.3% walking, P < 0.001), as illustrated in Figure 1B.
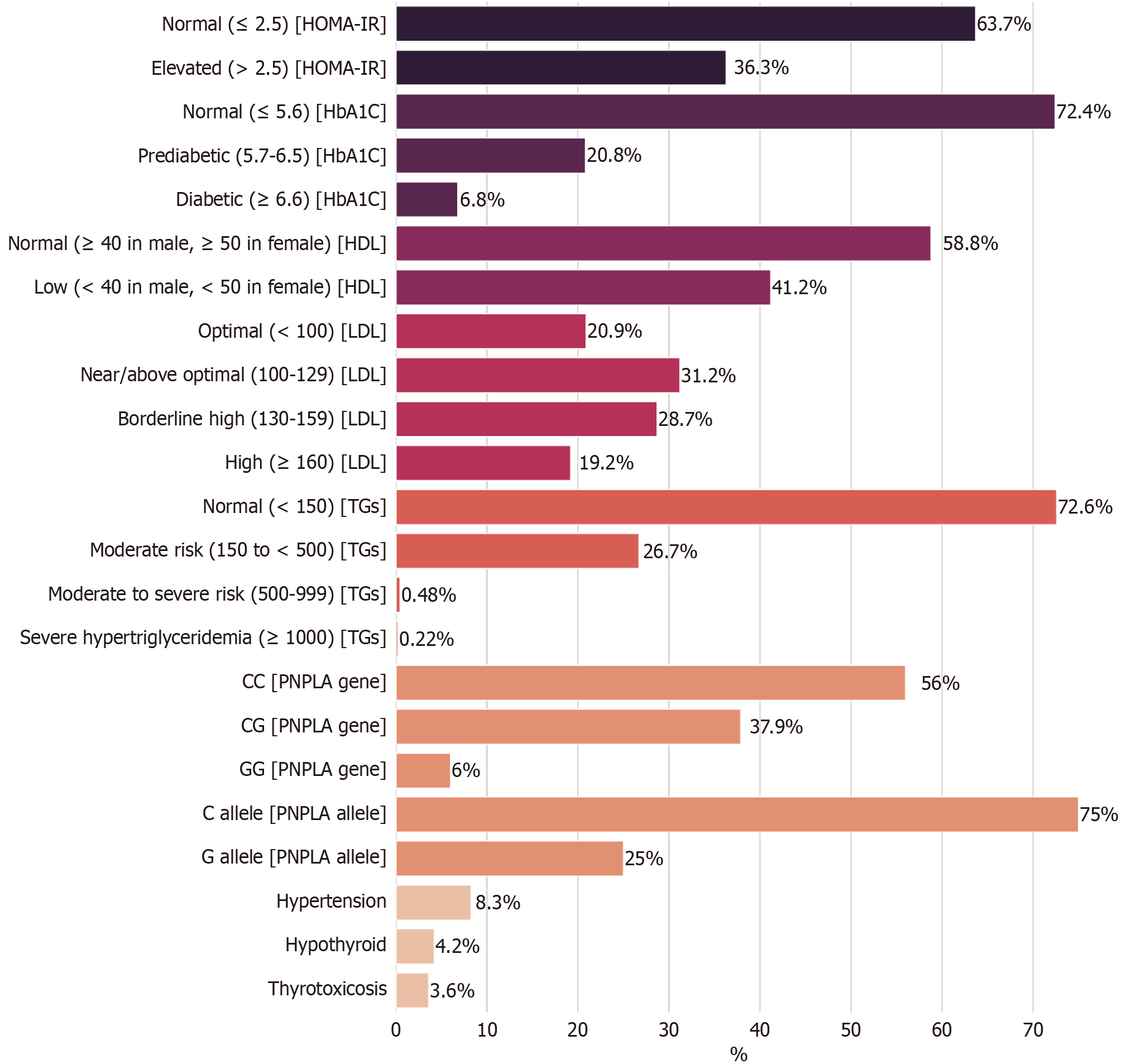
Metabolic abnormalities included prediabetes/diabetes (27.6%), insulin resistance (36.3%), hypertension (8.3%), and thyroid dysfunction (7.8%). The PNPLA3 gene variant was present in 46% of the participants, and 12.3% of the par
MetS was present in 41.2% of MASLD participants, and 54.9% had one or two cardiometabolic risk factors. MASLD was highly prevalent among individuals with metabolic abnormalities: (1) 44.4% with insulin resistance (HOMA-IR > 2.5); (2) 84.6% of prediabetics; (3) 94.4% of diabetics; (4) 94.3% of hypertensive individuals; (5) 79.9% with low HDL-cholesterol (HDL-C); and (6) 86.6% with high TGs.
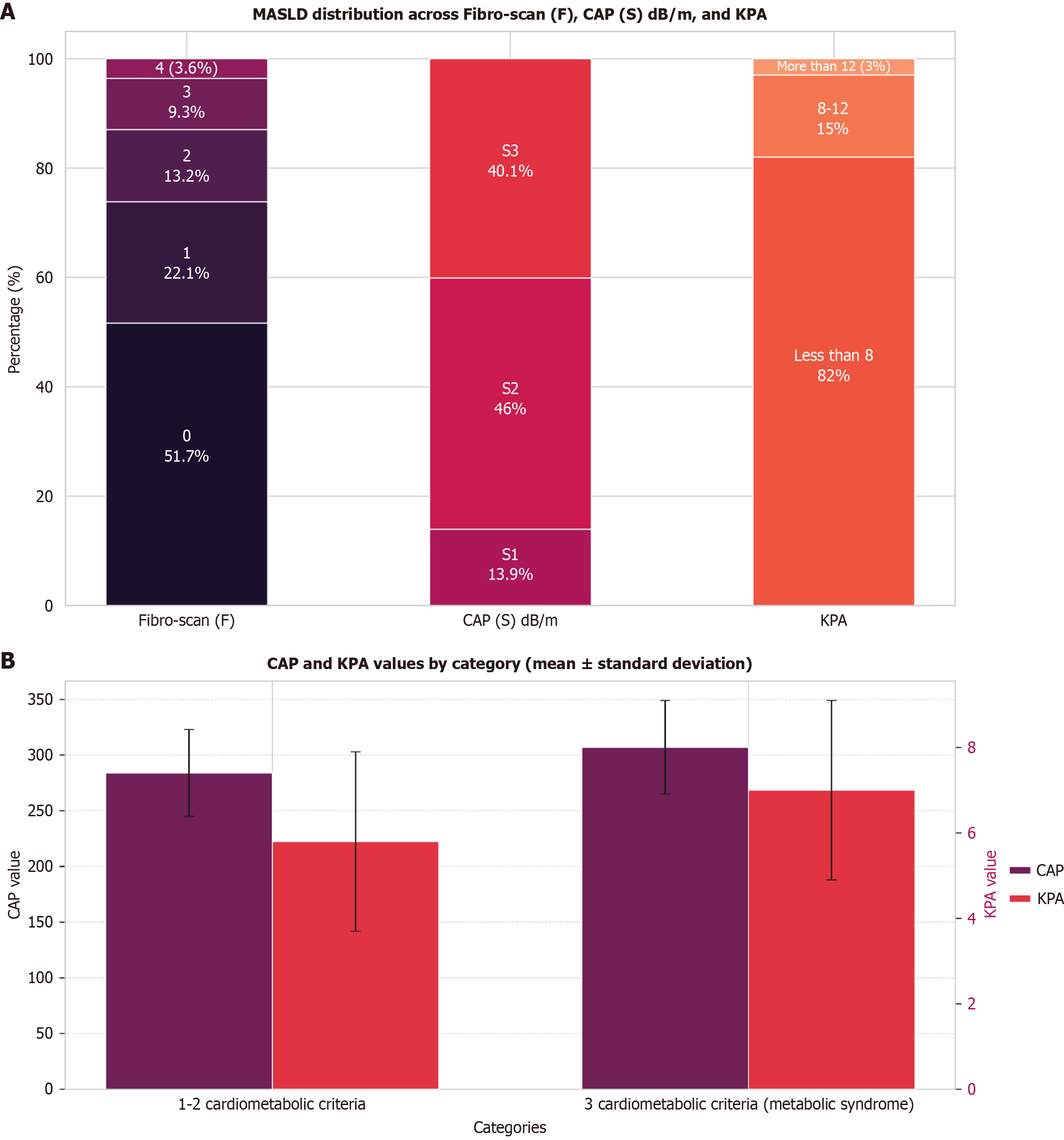
Liver ultrasound confirmed steatosis in 67.7% of participants, including four cases of cirrhosis, while 24 had gallstones and 36 had splenomegaly. VCTE revealed a mean liver stiffness of 6.3 ± 2.5 kPa, as depicted in Figure 3A, with substantially more severe steatosis and fibrosis in MetS patients (P < 0.001), as illustrated in Figure 3B.
Smoking prevalence was similar between MASLD (12.35%) and healthy (12.28%) participants, with no significant difference in steatosis (P = 0.201). However, smokers demonstrated slightly higher liver stiffness (6.7 ± 2.7 kPa vs 6.2 ± 2.5 kPa, P = 0.06).
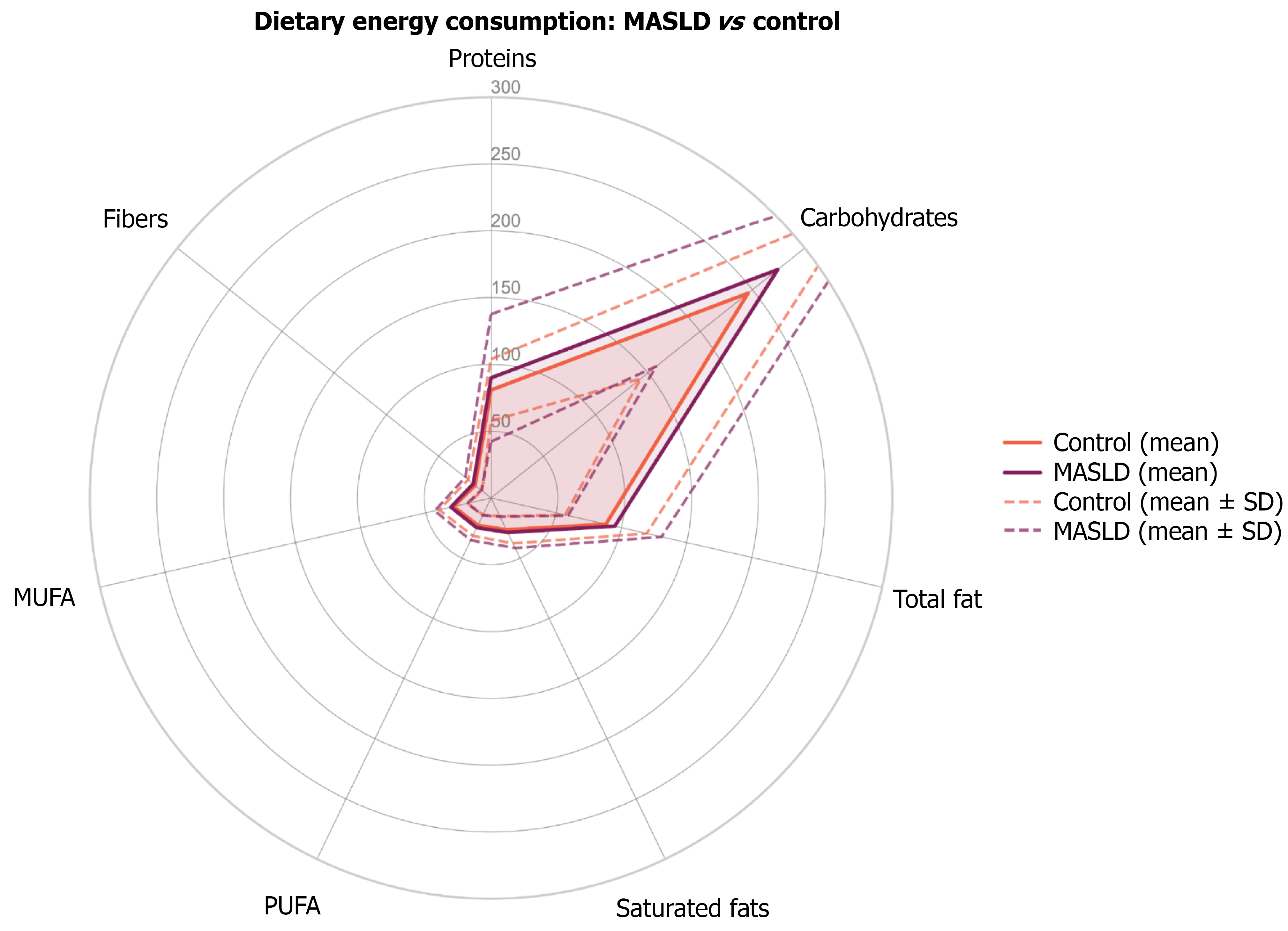
Nutritional analysis revealed that MASLD participants consumed more calories (2293.2 ± 818.5 kcal/day vs 2099.2 ± 818.5 kcal/day, P < 0.001), along with a higher glycemic index (952.5 ± 451.3 vs 863.1 ± 401.7, P = 0.002) and glycemic load (166.6 ± 73.9 vs 155.4 ± 82, P = 0.033). All dietary components were significantly elevated in MASLD patients, as illustrated in Figure 4.
MASLD is strongly associated with disturbances in lipid and glucose metabolism, as evidenced by elevated TGs, low density-lipoproteins (LDL), very LDL (VLDL), fasting glucose, HbA1c, insulin, HOMA-IR, liver enzymes, and CRP levels. Healthy participants had higher FT4 levels. Analysis of the PNPLA3 rs738409 genetic variant revealed its significant role in the Egyptian MASLD population. The variant was highly prevalent, present in 46% of the entire cohort. While its frequency was not drastically different between MASLD patients (48.1%) and healthy controls (43.6%), its impact was modified by body weight and specific dietary factors, pointing to important gene-environment interactions as exhibited in Table 1.
| Metabolically dysfunction-associated steatotic liver disease | Yes | No | P value |
| Lipoprotein lipase | 15 (0.5-185) | 18 (0.5-214) | 0.062 |
| L-carnitine | 12.3 (0.1-63) | 13 (0.6-58) | 0.099 |
| C-reactive protein | 4 (0.3-117) | 2 (0.2-39.2) | < 0.001 |
| Aspartate aminotransferase | 20 (4.5-236) | 18 (9-103) | < 0.001 |
| Alanine aminotransferase | 20 (4-258) | 14 (3-82) | < 0.001 |
| Gamma-glutamyl transferase | 23 (4-595) | 15 (4-137) | < 0.001 |
| Bilirubin | 0.4 (0.1-2) | 0.4 (0-54) | 0.066 |
| International normalized ratio | 1 (0.9-10) | 1.1 (1-1.1) | 0.302 |
| Homeostasis Model Assessment of Insulin Resistance | 2.2 (0.1-73) | 1.4 (0-20.2) | < 0.001 |
| Fasting insulin | 9.9 (0.3-279.4) | 7 (0.3-45) | < 0.001 |
| Triglycerides | 120 (3.9-1086) | 74 (28-409) | < 0.001 |
| Very LDL | 24 (3.1-217.2) | 14.8 (5.8-81.8) | < 0.001 |
| Thyrotropin | 1.6 (0.01-100) | 1.5 (0.01-9.7) | 0.227 |
| Albumin | 4.7 ± 2.4 | 4.7 ± 0.4 | 0.876 |
| Fasting blood sugar | 98.3 ± 33.6 | 88.9 ± 23.4 | < 0.001 |
| Glycated hemoglobin (%) | 5.7 ± 1.1 | 5.2 ± 0.7 | < 0.001 |
| Total cholesterol | 205.9 ± 44.7 | 193.1 ± 40.2 | < 0.001 |
| High-density lipoprotein | 46.4 ± 11.9 | 52.1 ± 13 | < 0.001 |
| LDL | 132.1 ± 39 | 122.8 ± 34.4 | < 0.001 |
| FT3 | 3.3 ± 0.5 | 3.4 ± 1.5 | 0.196 |
| FT4 | 1.3 ± 0.3 | 1.4 ± 0.5 | 0.003 |
| PNPLA gene | |||
| CC | 373 (65.9) | 193 (34.1) | 0.649 |
| CG | 261 (68.1) | 122 (31.9) | |
| GG | 43 (70.5) | 18 (29.5) | |
| PNPLA alle | |||
| C alle | 1007 (66.5) | 508 (33.5) | 0.382 |
| G alle | 347 (68.7) | 158 (31.3) |
Key predictors of MASLD include age, BMI, monounsaturated fatty acid (MUFA) intake, HOMA-IR, and VLDL levels. Overweight individuals have a 4.5-fold higher risk, which escalates to 27.5-fold in obesity. Elevated HOMA-IR increased risk by 3.4 times, independent of BMI. Aging raised MASLD risk by 6% annually in normal-weight individuals and 13% in obese individuals. Predictors varied by BMI: (1) Lean individuals were more affected by male gender (4-fold risk); (2) Low HDL-C; and (3) Insulin resistance. Each gram increase in MUFA intake raised MASLD risk by 3%, while a higher glycemic load increased risk by 1%, particularly in individuals with obesity, as exhibited in Table 2.
| Logistic regression of significant variables | B | SE | OR | 95%CI for OR | P value |
| BMI groups (reference normal BMI) | < 0.001 | ||||
| BMI (overweight vs normal BMI) | 1.5 | 0.3 | 4.5 | 2.5-7.9 | < 0.001 |
| BMI (obese vs normal BMI) | 3.3 | 0.3 | 27.5 | 14.6-51.7 | < 0.001 |
| HOMA-IR (elevated) | 0.9 | 0.2 | 2.7 | 1.6-4.1 | < 0.001 |
| Age | 0.1 | 0.01 | 1.06 | 1.03-1.08 | < 0.001 |
| Monounsaturated fatty acid | 0.03 | 0.01 | 1.03 | 1.01-1.05 | 0.003 |
| Very low-density lipoproteins | 0.02 | 0.01 | 1.02 | 1.01-1.04 | 0.024 |
| Normal BMI | |||||
| Sex (males) | 1.4 | 0.4 | 4.1 | 1.8-9.1 | 0.001 |
| HOMA-IR groups (elevated) | 1.2 | 0.4 | 3.4 | 1.5-7.6 | 0.003 |
| High-density lipoprotein groups (low) | 1.0 | 0.4 | 2.7 | 1.3-5.6 | 0.009 |
| Overweight participants | |||||
| Metabolic syndrome | 1.0 | 0.5 | 2.7 | 1.1-6.8 | 0.033 |
| Patients with obesity | |||||
| HOMA-IR (elevated) | 1.2 | 0.5 | 3.4 | 1.2-9.5 | 0.017 |
| Age | 0.1 | 0.02 | 1.13 | 1.08-1.2 | < 0.001 |
| Glycemic load | 0.01 | 0.004 | 1.01 | 1.005-1.02 | 0.001 |
Liver steatosis, measured via CAP, significantly increased with BMI (259 ± 20 dB/m in normal weight, 303 ± 36 dB/m in obesity, P < 0.001), correlating with liver stiffness (5.4 ± 1.5 kPa vs 6.6 ± 2.7 kPa, P < 0.001).
The study observed that the PNPLA3 gene variant was more common in overweight (53.41%) and obese (42.7%) MASLD participants compared to lean individuals (35.3%), with a significant P value of 0.019.
The median age of participants was 38 years, with 61.8% of MASLD patients being older and 73.7% of healthy controls being younger. MASLD prevalence was higher in older individuals (83%) compared with younger ones (52%), with younger males demonstrating a significantly higher risk (P = 0.029).
BMI and WC were significantly higher in older participants (P < 0.001), with mean BMIs of 31.4 ± 5.9 kg/m2 in younger and 33.4 ± 5.6 kg/m2 in older individuals. Low physical activity was a risk factor for younger MASLD patients (P = 0.012), and higher carbohydrate intake was significant in older MASLD participants.
Metabolic markers varied by age: Total cholesterol and LDL-cholesterol were higher in younger MASLD patients, whereas TGs, VLDL, HbA1c, fasting insulin, HOMA-IR, liver enzymes, and CRP were elevated across all MASLD groups. TSH levels were significantly higher only in younger MASLD participants. MetS was more prevalent in older MASLD patients (51.1%) than in younger ones (25.2%). The laboratory results for different age groups are presented in Table 3.
| MASLD | Non-MASLD | P value | MASLD | Non-MASLD | P value | |
| Lipoprotein lipase | 16 (0.5-185) | 19 (2-204) | 0.300 | 13 (0.5-154) | 16 (0.5-214) | 0.817 |
| L-carnitine | 12.4 (1-58) | 14 (1.2-58) | 0.113 | 12.2 (0.1-63) | 12.8 (0.6-43) | 0.899 |
| C-reactive protein | 3.6 (0.3-56) | 2 (0.3-33) | < 0.001 | 4.3 (0.3-117) | 2.8 (0.2-39.2) | < 0.001 |
| Aspartate aminotransferase | 19 (8-114) | 18 (9-103) | < 0.001 | 20 (4.5-236) | 19 (10-42) | 0.045 |
| Alanine aminotransferase | 21 (4-156) | 14 (4-82) | < 0.001 | 20 (5-258) | 15 (3-53) | < 0.001 |
| Gamma-glutamyl transferase | 21 (7-242) | 13.5 (4-99) | < 0.001 | 24 (4-595) | 17.5 (6-137) | < 0.001 |
| Bilirubin | 0.4 (0.1-1.9) | 0.4 (0.1-54) | 0.098 | 0.4 (0.1-2) | 0.4 (0-1.2) | 0.149 |
| Homeostasis Model Assessment of Insulin Resistance | 2.2 (0.1-73) | 1.4 (0-10.7) | < 0.001 | 2.2 (0.1-31.9) | 1.4 (0.1-20.2) | < 0.001 |
| Fasting insulin | 9.9 (0.3-279.4) | 7 (0.3-45) | < 0.001 | 9.9 (0.3-109.7) | 6.7 (0.3-39.1) | < 0.001 |
| Triglycerides | 104 (36-354) | 68.5 (28-392) | < 0.001 | 130 (3.9-1086) | 96.5 (43-409) | < 0.001 |
| Very low-density lipoproteins | 20.8 (7-70.8) | 13.7 (5.8-78.4) | < 0.001 | 26 (3.1-217.2) | 19.6 (8.6-81.8) | < 0.001 |
| Thyrotropin | 1.7 (0.01-100) | 1.5 (0.01-9.7) | 0.007 | 1.5 (0.01-16) | 1.5 (0.01-6.6) | 0.650 |
PNPLA3 gene analysis revealed comparable wild-type prevalence in both age groups (53.3% in younger and 51.1% in older participants). Although the GG polymorphism was associated with higher MASLD rates (64.7% in younger and 77.8% in older patients), these differences were not statistically significant (P = 0.299 and P = 0.55, respectively).
Age significantly influenced disease severity (P < 0.001), with older MASLD patients exhibiting higher liver steatosis (CAP: 300 ± 41 dB/m vs 283 ± 41 dB/m) and fibrosis (liver stiffness measurement: 6.6 ± 2.8 kPa vs 5.6 ± 1.7 kPa).
Multivariate analysis identified distinct MASLD risk factors across age groups as shown in Table 4. In younger individuals, BMI, HOMA-IR, and WC were significant predictors, whereas HbA1c played a stronger role in older par
| Young population < 38 years | B | SE | OR | 95%CI for OR | P value |
| BMI (reference normal BMI) | < 0.001 | ||||
| BMI (overweight vs normal BMI) | 3.2 | 0.9 | 24 | 4.2-134 | < 0.001 |
| BMI (obese vs normal BMI) | 3.5 | 0.9 | 32 | 5.5-183 | < 0.001 |
| Waist circumference (> 80 in female, > 94 in males) | 1.2 | 0.6 | 3.4 | 1.1-11 | 0.042 |
| HOMA-IR (elevated) | 1.7 | 0.5 | 5.6 | 1.906 | 0.002 |
| Old population ≥ 38 years | |||||
| BMI (reference normal BMI) | < 0.001 | ||||
| BMI (overweight vs normal BMI) | 1.3 | 0.5 | 3.8 | 1.5-9.8 | 0.006 |
| BMI (obese vs normal BMI) | 4.2 | 0.6 | 64.4 | 21-197.1 | < 0.001 |
| Elevated HOMA-IR | 1.1 | 0.4 | 3.1 | 1.4-6.8 | 0.006 |
| HbA1c (reference: Normal HBA1c ≤5.6) | 0.045 | ||||
| HbA1c (prediabetic vs normal HbA1c) | 1.3 | 0.5 | 3.6 | 1.3-9.9 | 0.013 |
| HbA1c (diabetics vs normal HbA1c) | 0.0 | 0.9 | 1.0 | 0.2-5.6 | 0.958 |
BMI and WC were significantly higher in MASLD participants with elevated HbA1c (P < 0.001). Diabetic and pre
Liver steatosis and fibrosis were more severe in diabetic (287 ± 39 dB/m, 5.9 ± 2.2 kPa) and prediabetic (304 ± 44 dB/m, 6.9 ± 2.8 kPa) MASLD patients compared with non-diabetics (P < 0.001). BMI, WC, and HOMA-IR were the strongest predictors in prediabetic/diabetic individuals, with obese diabetics facing the highest risk (17.6-fold).
Both wild-type and variant PNPLA3 gene carriers with MASLD exhibited significantly elevated glucose and lipid markers, liver enzymes, CRP, and FT4 compared with healthy controls, with no additional risk attributable to gene type. High glycemic index, glycemic load, and fructose intake were associated with the PNPLA3 variant in patients with MASLD. Although MUFA intake increased the risk of MASLD, this effect was consistent across all gene types (P = 0.272).
Key predictors of MASLD differed by PNPLA3 status. In wild-type carriers, BMI, WC, high HOMA-IR, and low HDL were primary risk factors. In variant carriers, age (11% increased risk per year), MetS (4.6-fold higher risk), and polyunsaturated fatty acid (PUFA) intake (10% increased risk per gram) were the main drivers of MASLD.
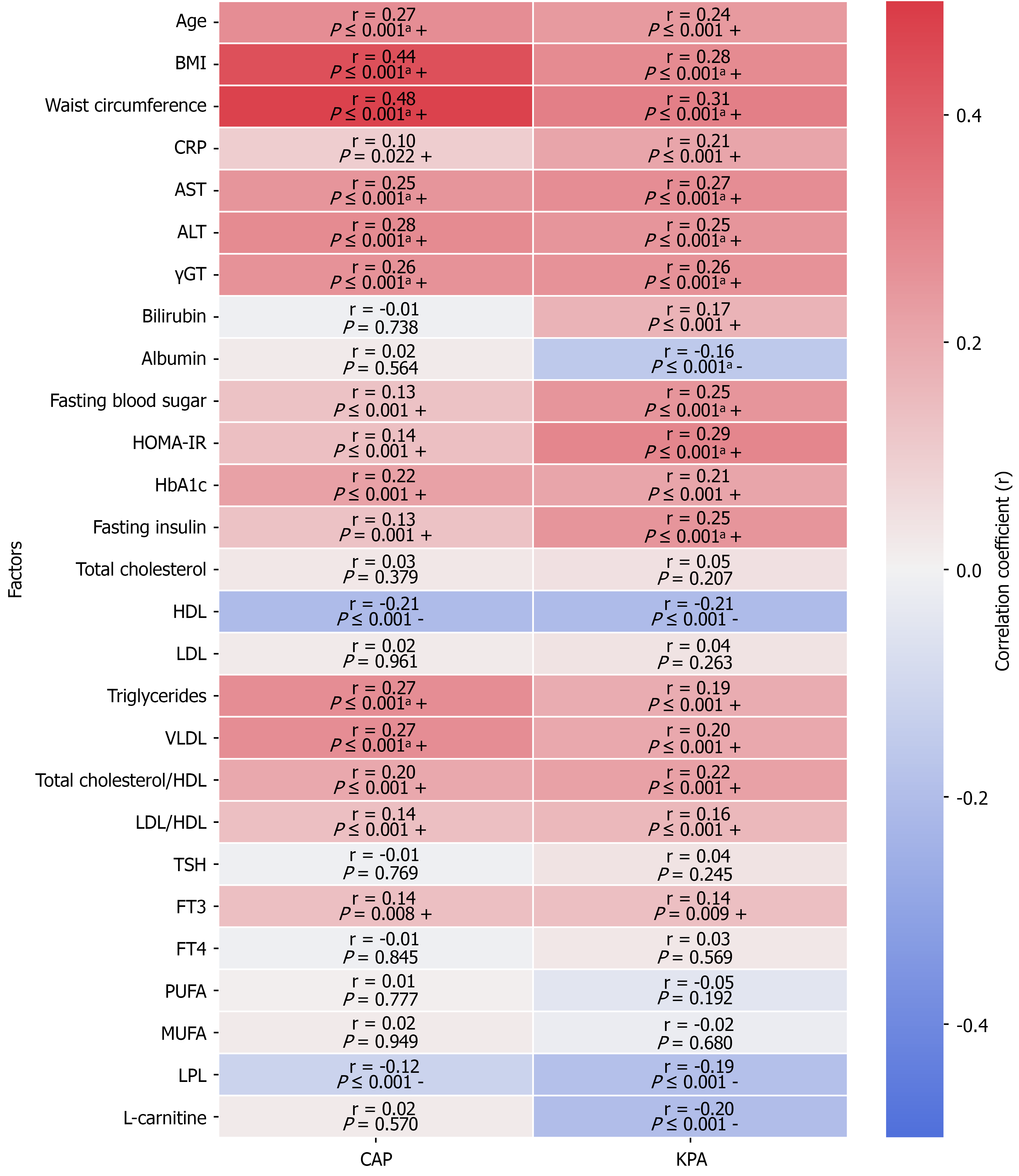
Liver steatosis correlated weakly with CRP, glucose markers, and FT3 (positive) and with HDL and LPL (negative), while revealing moderate correlations with age, BMI, WC, liver enzymes, TGs, and VLDL. Liver fibrosis had weak associations with age, CRP, bilirubin, HbA1c, and TGs/VLDL (positive), as well as with LPL, albumin, and HDL (negative). Still, it correlated moderately with BMI, WC, liver enzymes, fasting glucose/insulin, and HOMA-IR, as displayed in Figure 5.
Egypt carries a significantly higher burden of MASLD compared with global rates. The recent study identified a striking prevalence of 67.7% among Egyptian adults, substantially exceeding both the previously reported national rate of 47.5% and the global average of 25%. This alarming trend persists despite limited awareness among both Egyptian physicians and patients regarding the disease’s severity and long-term consequences.
The escalating obesity epidemic in Egypt (affecting 49.51% of women and 29.53% of men according to 2024 data)[20] appears to be a major driver of MASLD development. The present findings demonstrated a clear weight-dependent progression: 59.5% of overweight individuals and 100% of morbidly obese participants had MASLD, while even 22.2% of lean individuals were affected. Obesity conferred a 27.5-fold higher MASLD risk, and this relationship directly correlated with age. Distinct demographic patterns were additionally observed: (1) Men exhibited a fourfold higher risk compared with women; and (2) Prevalence increased markedly from 52% in younger adults (< 38 years) to 83% in older par
Physical activity demonstrated protective effects, aligning with the American College of Sports Medicine’s 2009 guidelines, which recommend 150-250 minutes of moderate exercise weekly for MASLD prevention[21]. Healthy controls in the current study significantly exceeded MASLD patients in meeting this threshold (P < 0.001), particularly through more than 140 minutes of weekly walking. Notably, inactivity specifically elevated risk in younger but not older MASLD patients, suggesting age-modified protective mechanisms.
These findings underscore the urgent need for targeted interventions addressing Egypt’s unique MASLD risk profile, with a focus on weight management in youth and metabolic health in older adults.
Evidence suggested that atherogenic dyslipidemia, marked by high TGs, low HDL-C, and elevated LDL-cholesterol, is predominant in MASLD participants[22]. The present results demonstrated that participants with MASLD had significantly elevated CRP, liver enzymes, and metabolic markers, with low HDL-C tripling the MASLD risk, and each unit increase in VLDL raising the risk by 2%. Insulin resistance (HOMA-IR ≥ 2.5) strongly predicted MASLD among Egyptians, raising the risk 3.4-fold per unit rise, even in normoglycemic individuals, highlighting its central role in MASLD pathogenesis, as previously documented[23].
Egyptian studies suggest that there is higher consumption of refined carbohydrates traditional baladi bread, white bread and polished rice, sugar sweetened beverages, as well as frequent consumption of deep-fried dishes with low intake of fruits and vegetables that directly contributed to the higher MASLD prevalence among Egyptians[24].
Dietary analysis revealed that a 1-gram increase in MUFAs raised the risk of MASLD by 3%, while a higher glycemic load increased the risk by 1%. Carbohydrate intake was significantly higher in older MASLD patients, whereas younger patients exhibited elevated levels across all dietary components analyzed.
Hepatic steatosis, the hallmark of MASLD, was observed in 67.7% of participants in the recent study, with 40.1% exhibiting severe steatosis, 15% having significant fibrosis (≥ 8 kPa), and 3% having cirrhosis (≥ 12 kPa). These results align with a prior Egyptian study, which reported 57.9% severe steatosis (S3) and 5% with fibrosis (F2-F3) on elas
Per European Association for the Study of the Liver-European Association for the Study of Diabetes-European Association for the Study of Obesity guidelines, non-invasive fibrosis assessment is recommended for patients with abnormal liver enzymes, cardiometabolic risks, or radiological steatosis, as these factors promote steatosis via free fatty acids, inflammation, and oxidative stress[9]. In the current cohort, the prevalence of MetS was higher in older MASLD participants (51.1%) than in younger ones (25.2%), supporting aging and metabolic dysfunction as key predictors of MASLD[26]. Additionally, participants fulfilling all MetS criteria had more severe steatosis and fibrosis than those with 1-2 criteria. This corroborates previous findings that CAP scores alone detect MetS with 0.79 accuracy, improving to 0.904 when combined with machine learning (ML)[27].
Younger individuals with MASLD exhibited significantly higher total cholesterol and LDL levels, while other markers of lipid and glucose metabolism were elevated across all age groups. Prior research identified baseline systolic blood pressure, along with increases in TGs and fasting blood glucose, as risk factors for MASLD in individuals aged ≥ 65 years[28]. Additionally, MASLD patients with diabetes or prediabetes had more severe liver steatosis and fibrosis than non-diabetic MASLD patients, irrespective of BMI.
In the present study, healthy participants displayed marginally higher levels of L-carnitine and LPL. LPL, a TG hydrolase, facilitates lipoprotein uptake via LPL receptors and influences adipocyte differentiation. L-carnitine helps mitigate excess mitochondrial acetyl-CoA, promotes fatty acid oxidation, and reduces hepatic free fatty acid accumulation[29]. Liver steatosis and fibrosis demonstrated a weak negative correlation with LPL and L-carnitine (P < 0.001), supporting their potential role as risk factors for MASLD in the Egyptian population.
Studies from 2012 to 2021 reported an inverse relationship between serum free T4 and fatty liver in euthyroid individuals, with no association observed for TSH or free T3[30,31]. The recent findings align with this: TSH was elevated only in younger MASLD patients, while FT4 was higher in healthy participants. Liver steatosis and fibrosis were weakly correlated with FT3, consistent with research by Van den Berg et al[32], which linked higher FT3, lower FT4, and an elevated FT3/FT4 ratio to euthyroid patients with MASLD. This likely reflects central adiposity, as iodothyronine deiodinase activity increases in obese individuals and high-fat diet models, thereby enhancing the conversion of FT4 to FT3.
Even under isocaloric conditions, the type of dietary fat and carbohydrate influences liver fat accumulation through distinct molecular and metabolic pathways that enhance de novo lipogenesis. Saturated fats and fructose, unlike unsaturated fats, drive the most significant increases in intrahepatic TGs, insulin resistance, and ceramide accumulation, thereby exacerbating the risk of MASLD[33].
In the present study, a 1-gram increase in MUFA and PUFA intake was associated with a 3% and 10% higher risk of MASLD, respectively, while a 1-unit rise in glycemic load was only linked to a 1% increased risk. This apparent paradox, where MUFA and PUFA fats typically considered “healthy” are linked to higher disease risk, likely reflects the specific dietary context in Egypt. The primary sources of these fat are not from whole foods like nuts, avocados, or olive oil used in salad dressings, but rather from oils repeatedly used for deep-frying traditional foods (e.g., falafel, fried fish, and feteer meshaltet) and from hydrogenated oils in commercially produced baked goods and sweets. These processing methods, particularly deep-frying at high temperatures, can lead to the formation of toxic lipid peroxidation products and trans fatty acids, which promote hepatic inflammation, oxidative stress, and insulin resistance[34,35]. Furthermore, a high intake of omega-6 PUFAs without a balanced intake of anti-inflammatory omega-3 PUFAs, a common feature of modern diets, can exacerbate pro-inflammatory eicosanoid production. Therefore, the observed association likely signifies a diet high in energy-dense, processed, and fried foods a pattern that overwhelms any potential benefits of MUFA/PUFA and directly contributes to hepatic lipid accumulation and metabolic dysfunction[35].
The PNPLA3 gene mutation was detected in 43.6% of healthy participants and 48.1% of MASLD participants, aligning with previous reports that PNPLA3 polymorphisms (normal, heterozygous, and homozygous) are highly prevalent in MASLD patients[36]. Nearly half of the present cohort carried this variant, with MASLD participants predominantly exhibiting either the wild-type “CC” or variant “CG/GG” genotypes. The variant was significantly more frequent in overweight (53.41%) and obese (42.7%) individuals with MASLD compared to lean MASLD cases.
Dietary and metabolic associations differed by genotype: In wild-type PNPLA3 carriers, BMI, WC, high HOMA-IR, and low HDL predicted MASLD, whereas mutant gene carriers were influenced by age, PUFA intake, and MetS. Earlier research corroborates that the PNPLA3 genotype modulates MASLD progression, with high intake of carbohydrates, isoflavones, and methionine exacerbating the risk of hepatic fibrosis[37]. These findings underscore the importance of investigating the roles of nutrigenomics and epigenetics in the Egyptian population.
These results establish MASLD as a critical public health priority in Egypt, highlighting the complicated relationship between metabolic, genetic, and lifestyle factors in disease pathogenesis. The key strengths of this study include being the most extensive MASLD study in North Africa, featuring comprehensive metabolic and genetic profiling, standardized imaging protocols across centers, and representative geographic sampling.
While this study provides valuable insights into MASLD epidemiology in Egypt, its cross-sectional design limits the ability to make causal inferences. The reliance on imaging rather than histology, potential selection bias, and unmeasured confounders (e.g., environmental toxins and other genetic variants) underscore the need for longitudinal, biopsy-confirmed studies that incorporate broader risk assessments to validate these findings and inform personalized interventions.
The complex interplay of metabolic, genetic, and lifestyle factors in MASLD pathogenesis makes it a prime candidate for advanced analytical approaches. AI and ML have the potential to improve risk stratification by integrating the predictors identified in this study (e.g., PNPLA3 variants, glycemic load, HOMA-IR) with imaging biomarkers to develop personalized prognostic models. For instance, previous studies have demonstrated that ML models can predict fibrosis progression from ultrasound elastography with high accuracy when combined with metabolic data[38]. In resource-constrained settings like Egypt, AI-powered tools could potentially aid in early detection within primary care, while generative AI might help tailor dietary interventions to local food patterns.
However, the development and deployment of such AI solutions face significant challenges. These include the requirement for large, high-quality, and well-annotated datasets for training, and the critical need to ensure generalizability across diverse populations to mitigate algorithmic bias. The comprehensive, multi-center dataset generated by this study represents a valuable initial step toward building such robust, region-specific models. Future work should focus on validating AI algorithms in the Egyptian and other Middle Eastern and North African populations to ensure their clinical utility and equity.
While this study provides comprehensive insights into the MASLD epidemic in Egypt, several limitations should be considered. First, the cross-sectional design precludes the determination of causality or the assessment of disease progression over time. Second, the diagnosis of MASLD and steatosis grading relied on ultrasonography and VCTE rather than liver histology, the gold standard, which may underestimate the prevalence of steatohepatitis (metabolic dysfunction-associated steatohepatitis) and early fibrosis. Third, although geographically diverse, our sample was one of convenience, potentially limiting its generalizability to all Egyptian subpopulations. Further-more, we did not perform comprehensive serological testing to exclude all secondary causes of steatotic liver disease, such as Wilson’s disease, autoimmune hepatitis, or monogenic lipid disorders. Finally, while we identified significant genetic-dietary interactions, other potential confounders, including environmental toxins, detailed socioeconomic factors, and the gut microbiome, were not assessed and warrant investigation in future studies.
The findings of this research underscore the urgent need for nationwide MASLD screening programs, tailored lifestyle interventions addressing Egypt’s unique dietary patterns, and genetic risk stratification, particularly for individuals carrying the PNPLA3 variant. Physician education is crucial for enhancing disease recognition and management. Future longitudinal studies should investigate progression rates and evaluate targeted prevention strategies in this high-risk population. Furthermore, future research should investigate AI-driven precision medicine approaches for managing MASLD in resource-constrained settings.
This extensive multicenter study reveals an alarmingly high prevalence of MASLD (67.7%) among Egyptian adults, far exceeding global averages and establishing Egypt as a critical hotspot for this condition. The epidemic is driven by a confluence of powerful factors, including a high prevalence of obesity (conferring a 27.5-fold increased risk), insulin resistance, a sedentary lifestyle, and a diet high in refined carbohydrates, and fats. Furthermore, the high population frequency of the PNPLA3 rs738409 variant, which interacts with dietary intake, creates a potent genetic-dietary nexus that modulates disease severity. These findings underscore the urgent public health imperative to implement national screening programs and culturally tailored lifestyle interventions, aligning with recent global calls to action for a coordinated response to the MASLD pandemic.
The authors would like to acknowledge Ashraf Gayel Ahmed (consultant Clinical Nutritionin Road Elfarag Hospital, Egypt), Manal Farouk Ahmed Abdelhafeez, and Hala Wassef Abdallah (consultants of clinical pathology in El-Helal Hospital, Egypt); for their participation in the data collection in this study.
| 1. | Karlsen TH, Sheron N, Zelber-Sagi S, Carrieri P, Dusheiko G, Bugianesi E, Pryke R, Hutchinson SJ, Sangro B, Martin NK, Cecchini M, Dirac MA, Belloni A, Serra-Burriel M, Ponsioen CY, Sheena B, Lerouge A, Devaux M, Scott N, Hellard M, Verkade HJ, Sturm E, Marchesini G, Yki-Järvinen H, Byrne CD, Targher G, Tur-Sinai A, Barrett D, Ninburg M, Reic T, Taylor A, Rhodes T, Treloar C, Petersen C, Schramm C, Flisiak R, Simonova MY, Pares A, Johnson P, Cucchetti A, Graupera I, Lionis C, Pose E, Fabrellas N, Ma AT, Mendive JM, Mazzaferro V, Rutter H, Cortez-Pinto H, Kelly D, Burton R, Lazarus JV, Ginès P, Buti M, Newsome PN, Burra P, Manns MP. The EASL-Lancet Liver Commission: protecting the next generation of Europeans against liver disease complications and premature mortality. Lancet. 2022;399:61-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 542] [Cited by in RCA: 484] [Article Influence: 121.0] [Reference Citation Analysis (3)] |
| 2. | En Li Cho E, Ang CZ, Quek J, Fu CE, Lim LKE, Heng ZEQ, Tan DJH, Lim WH, Yong JN, Zeng R, Chee D, Nah B, Lesmana CRA, Bwa AH, Win KM, Faulkner C, Aboona MB, Lim MC, Syn N, Kulkarni AV, Suzuki H, Takahashi H, Tamaki N, Wijarnpreecha K, Huang DQ, Muthiah M, Ng CH, Loomba R. Global prevalence of non-alcoholic fatty liver disease in type 2 diabetes mellitus: an updated systematic review and meta-analysis. Gut. 2023;72:2138-2148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 233] [Article Influence: 77.7] [Reference Citation Analysis (1)] |
| 3. | Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease. Lancet. 2021;397:2212-2224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2350] [Cited by in RCA: 2041] [Article Influence: 408.2] [Reference Citation Analysis (0)] |
| 4. | Osei Bonsu E, Addo IY. Prevalence and correlates of overweight and obesity among under-five children in Egypt. Front Public Health. 2022;10:1067522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 5. | Sinha N, Lara Ibarra G, Kim YS, Fayez RNS, Elassiouty SMM, Germiniasi A, Saavedra Facusse TB, Seo J. Understanding Poverty and Inequality in Egypt (English). Washington: World Bank Group, 2019. |
| 6. | Gan C, Yuan Y, Shen H, Gao J, Kong X, Che Z, Guo Y, Wang H, Dong E, Xiao J. Liver diseases: epidemiology, causes, trends and predictions. Signal Transduct Target Ther. 2025;10:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 134] [Article Influence: 134.0] [Reference Citation Analysis (1)] |
| 7. | Craig CL, Marshall AL, Sjöström M, Bauman AE, Booth ML, Ainsworth BE, Pratt M, Ekelund U, Yngve A, Sallis JF, Oja P. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;35:1381-1395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11293] [Cited by in RCA: 14529] [Article Influence: 631.7] [Reference Citation Analysis (1)] |
| 8. | Watson EO, Heath AL, Taylor RW, Mills VC, Barris AC, Skidmore PM. Relative validity and reproducibility of an FFQ to determine nutrient intakes of New Zealand toddlers aged 12-24 months. Public Health Nutr. 2015;18:3265-3271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1231] [Article Influence: 615.5] [Reference Citation Analysis (2)] |
| 10. | Javed H, Oyibo SO, Alfuraih AM. Variability, Validity and Operator Reliability of Three Ultrasound Systems for Measuring Tissue Stiffness: A Phantom Study. Cureus. 2022;14:e31731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Eddowes PJ, Sasso M, Allison M, Tsochatzis E, Anstee QM, Sheridan D, Guha IN, Cobbold JF, Deeks JJ, Paradis V, Bedossa P, Newsome PN. Accuracy of FibroScan Controlled Attenuation Parameter and Liver Stiffness Measurement in Assessing Steatosis and Fibrosis in Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2019;156:1717-1730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1232] [Cited by in RCA: 1105] [Article Influence: 157.9] [Reference Citation Analysis (2)] |
| 12. | Mikolasevic I, Orlic L, Franjic N, Hauser G, Stimac D, Milic S. Transient elastography (FibroScan(®)) with controlled attenuation parameter in the assessment of liver steatosis and fibrosis in patients with nonalcoholic fatty liver disease - Where do we stand? World J Gastroenterol. 2016;22:7236-7251. [PubMed] [DOI] [Full Text] |
| 13. | Wong VW, Vergniol J, Wong GL, Foucher J, Chan HL, Le Bail B, Choi PC, Kowo M, Chan AW, Merrouche W, Sung JJ, de Lédinghen V. Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology. 2010;51:454-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1098] [Cited by in RCA: 987] [Article Influence: 61.7] [Reference Citation Analysis (11)] |
| 14. | Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26058] [Cited by in RCA: 24786] [Article Influence: 604.5] [Reference Citation Analysis (1)] |
| 15. | Yuan B, Yang W, Zhang N, Shi H, Dong S. Evaluation of newly-developed glycated hemoglobin clinical analytic reagents and chromatography column on Tosoh HLC-723 G8 Analyzer. Pract Lab Med. 2023;37:e00338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 16. | Sloan S, Jenvey CJ, Piedrafita D, Preston S, Stear MJ. Comparative evaluation of different molecular methods for DNA extraction from individual Teladorsagia circumcincta nematodes. BMC Biotechnol. 2021;21:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 17. | Eslam M, Valenti L, Romeo S. Genetics and epigenetics of NAFLD and NASH: Clinical impact. J Hepatol. 2018;68:268-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 736] [Article Influence: 92.0] [Reference Citation Analysis (4)] |
| 18. | Dean AG, Dean JA, Burton AH, Dicker RC. Epi Info: a general-purpose microcomputer program for public health information systems. Am J Prev Med. 1991;7:178-182. [PubMed] |
| 19. | Hart PD. Physical Activity and Body Mass Index (BMI) as Predictors of Health-related Quality of Life in Montana Adults. J Phys Act Res. 2021;6:135-141. [DOI] [Full Text] |
| 20. | Esmat G, Zaid H, Hassany M, Abdel-Razek W, El-Serafy M, El Akel W, Salah A, Kamal E, Elshishiney G, Ammar I, Kabil K, Abdallah M, Saeed R, Saad T, Omar Y, Dabbous H, El-Sayed MH, El Shazly Y, Doss W, Waked I. Obesity prevalence in adults and patients with hepatitis C: Results from screening a population of 50 million in Egypt. Egypt Liver J. 2024;14:22. [DOI] [Full Text] |
| 21. | Fock KM, Khoo J. Diet and exercise in management of obesity and overweight. J Gastroenterol Hepatol. 2013;28 Suppl 4:59-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 193] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 22. | Kaul U, Parmar D, Manjunath K, Shah M, Parmar K, Patil KP, Jaiswal A. New dual peroxisome proliferator activated receptor agonist-Saroglitazar in diabetic dyslipidemia and non-alcoholic fatty liver disease: integrated analysis of the real world evidence. Cardiovasc Diabetol. 2019;18:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 107] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 23. | Loomba R, Friedman SL, Shulman GI. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell. 2021;184:2537-2564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1672] [Cited by in RCA: 1507] [Article Influence: 301.4] [Reference Citation Analysis (1)] |
| 24. | AlFaris NA, Alshwaiyat NM, Alkhalidy H, AlTamimi JZ, Alagal RI, Alsaikan RA, Alsemari MA, BinMowyna MN, AlKehayez NM. Sugar-sweetened beverages consumption in a multi-ethnic population of middle-aged men and association with sociodemographic variables and obesity. Front Nutr. 2022;9:987048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 25. | Tomah S, Hamdy O, Abuelmagd MM, Hassan AH, Alkhouri N, Al-Badri MR, Gardner H, Eldib AH, Eid EA. Prevalence of and risk factors for non-alcoholic fatty liver disease (NAFLD) and fibrosis among young adults in Egypt. BMJ Open Gastroenterol. 2021;8:e000780. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 26. | Liu YX, Liu X, Cen C, Li X, Liu JM, Ming ZY, Yu SF, Tang XF, Zhou L, Yu J, Huang KJ, Zheng SS. Comparison and development of advanced machine learning tools to predict nonalcoholic fatty liver disease: An extended study. Hepatobiliary Pancreat Dis Int. 2021;20:409-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 27. | Yu CS, Chang SS, Lin CH, Lin YJ, Wu JL, Chen RJ. Identify the Characteristics of Metabolic Syndrome and Non-obese Phenotype: Data Visualization and a Machine Learning Approach. Front Med (Lausanne). 2021;8:626580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | Lin Y, Feng X, Cao X, Miao R, Sun Y, Li R, Ye J, Zhong B. Age patterns of nonalcoholic fatty liver disease incidence: heterogeneous associations with metabolic changes. Diabetol Metab Syndr. 2022;14:181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 29. | Li N, Zhao H. Role of Carnitine in Non-alcoholic Fatty Liver Disease and Other Related Diseases: An Update. Front Med (Lausanne). 2021;8:689042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 30. | Ittermann T, Haring R, Wallaschofski H, Baumeister SE, Nauck M, Dörr M, Lerch MM, Meyer zu Schwabedissen HE, Rosskopf D, Völzke H. Inverse association between serum free thyroxine levels and hepatic steatosis: results from the Study of Health in Pomerania. Thyroid. 2012;22:568-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 31. | Guo W, Qin P, Li XN, Wu J, Lu J, Zhu WF, Diao QQ, Xu NZ, Zhang Q. Free Triiodothyronine Is Associated With Hepatic Steatosis and Liver Stiffness in Euthyroid Chinese Adults With Non-Alcoholic Fatty Liver Disease. Front Endocrinol (Lausanne). 2021;12:711956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | van den Berg EH, van Tienhoven-Wind LJ, Amini M, Schreuder TC, Faber KN, Blokzijl H, Dullaart RP. Higher free triiodothyronine is associated with non-alcoholic fatty liver disease in euthyroid subjects: the Lifelines Cohort Study. Metabolism. 2017;67:62-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 82] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 33. | Hydes T, Alam U, Cuthbertson DJ. The Impact of Macronutrient Intake on Non-alcoholic Fatty Liver Disease (NAFLD): Too Much Fat, Too Much Carbohydrate, or Just Too Many Calories? Front Nutr. 2021;8:640557. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 34. | Bazina N, Ahmed T, Almdaaf M, Abu Hallalah HMO, Jibia S. Chemical Changes in Deep-Fat Frying: Reaction Mechanisms, Oil Degradation, and Health Implications. Food Sci Nutr. 2025;13:e70969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 35. | Dhibi M, Brahmi F, Mnari A, Houas Z, Chargui I, Bchir L, Gazzah N, Alsaif MA, Hammami M. The intake of high fat diet with different trans fatty acid levels differentially induces oxidative stress and non alcoholic fatty liver disease (NAFLD) in rats. Nutr Metab (Lond). 2011;8:65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 36. | Zhang L, You W, Zhang H, Peng R, Zhu Q, Yao A, Li X, Zhou Y, Wang X, Pu L, Wu J. PNPLA3 polymorphisms (rs738409) and non-alcoholic fatty liver disease risk and related phenotypes: a meta-analysis. J Gastroenterol Hepatol. 2015;30:821-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 37. | Vilar-Gomez E, Pirola CJ, Sookoian S, Wilson LA, Belt P, Liang T, Liu W, Chalasani N. Impact of the Association Between PNPLA3 Genetic Variation and Dietary Intake on the Risk of Significant Fibrosis in Patients With NAFLD. Am J Gastroenterol. 2021;116:994-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 38. | Zhu G, Song Y, Lu Z, Yi Q, Xu R, Xie Y, Geng S, Yang N, Zheng L, Feng X, Zhu R, Wang X, Huang L, Xiang Y. Machine learning models for predicting metabolic dysfunction-associated steatotic liver disease prevalence using basic demographic and clinical characteristics. J Transl Med. 2025;23:381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |