Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.113325
Revised: November 16, 2025
Accepted: January 8, 2026
Published online: March 21, 2026
Processing time: 174 Days and 0.9 Hours
Icariin (ICA) is a natural compound derived from Epimedium that has shown promise in treating colorectal cancer (CRC) by reducing inflammation, inducing apoptosis, and limiting tumor progression.
To elucidate the specific mechanisms underlying its therapeutic effects on this malignancy.
We examined the impact of different ICA concentrations on the biological be
Our findings indicate that ICA attenuated CRC cell proliferation, invasion, and migration, while suppressing TAN recruitment and promoting apoptosis in a concentration-dependent manner. When small interfering SMAD4 was introduced, the anticancer effects of ICA were diminished. Furthermore, disruption of the CXCL8-CXCR2 axis re
ICA impedes the recruitment of TANs primarily through SMAD4-mediated modulation of the CXCL8-CXCR2 axis, thereby suppressing EMT and other pro-metastatic programs in CRC.
Core Tip: This study elucidates a novel mechanism by which the natural compound icariin (ICA) combats colorectal cancer. We demonstrate that ICA upregulates the tumor suppressor SMAD4, thereby inhibiting the production of C-X-C motif chemokine ligand 8 (CXCL8) and other C-X-C motif chemokine receptor 2 (CXCR2)-associated chemokines. This action predominantly limits the recruitment of tumor-associated neutrophils (TANs) via the CXCL8-CXCR2 axis. Notably, ICA not only reduces neutrophil infiltration but also reprograms TANs toward an antitumor N1 phenotype. By disrupting this SMAD4-CXCL8-CXCR2-TAN axis, ICA effectively restrains epithelial-mesenchymal transition and other pro-metastatic processes, positioning it as a promising therapeutic candidate with a distinct immunomodulatory strategy against colorectal cancer progression.
- Citation: Zhao YH, Chen J, Gong T, Ge HY, Li M. Icariin inhibits tumor-associated neutrophil recruitment via the CXCL8-CXCR2 axis and restrains pro-metastatic programs in colorectal cancer. World J Gastroenterol 2026; 32(11): 113325
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/113325.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.113325
Colorectal cancer (CRC) ranks among the most prevalent and lethal malignancies worldwide[1,2]. Despite significant advances in cancer diagnosis and treatment, the complex pathogenesis, aggressive progression, and high metastatic potential of CRC continue to pose major challenges for both patients and clinicians[3,4]. Understanding the molecular mechanisms that govern CRC metastasis has long been a central focus of cancer research[5], with the tumor microenvironment (TME) now recognized as a key determinant of disease progression[6].
The TME, composed of a complex network of immune cells, fibroblasts, and extracellular matrix components, plays a crucial role in influencing tumor behavior[7]. Among immune cells, tumor-associated neutrophils (TANs) have attracted significant attention because of their diverse and often paradoxical roles in cancer[8]. TANs represent a heterogeneous population of neutrophils that infiltrate tumor sites and interact with both immune and cancer cells[9]. Their precise involvement in cancer is complex, as they can exert either protumorigenic or anti-tumorigenic effects depending on the microenvironment and signaling pathways involved[10]. Within this context, TANs are recruited into the TME by cy
SMAD4 is a critical molecule in the transforming growth factor-β (TGF-β) signaling pathway, which plays a pivotal role in CRC. Patients with high SMAD4 expression exhibit significantly longer overall and disease-free survival compared with those showing low SMAD4 expression, suggesting that SMAD4 may serve as a prognostic marker in CRC[19]. The SMAD4 gene, which is frequently mutated in CRC, has been found to harbor inactivating mutations in a subset of cases[20]. Reduced SMAD4 levels are also considered a key indicator of poor prognosis in CRC[21]. According to a study by Yamamoto et al[22], CRC cells lacking SMAD4 attracted CCR1+ cells, most of which were TANs, thereby enhancing their metastatic potential to the lung. Likewise, Ogawa et al[23] reported that SMAD4 knockdown increased CXCL1 and CXCL8 expression in human CRC cells, promoting neutrophil recruitment to CRC tumors.
Icariin (ICA) is a naturally occurring compound that has drawn considerable scientific interest due to its multiple pharmacological activities, including anti-inflammatory[24], antioxidant[25], and anti-tumor[26] effects. Several studies have shown that ICA can influence the TGF-β/SMAD signaling pathway[27], in which SMAD4 functions as a key signaling mediator[28]. Furthermore, ICA suppresses the production of CXCL8, which may be related to its anti-inflammatory properties, as CXCL8 is commonly produced during inflammatory processes[29]. Collectively, these findings sug
The present study explores ICA as a potential therapeutic agent for CRC. Our research aims to elucidate the underlying mechanisms through which ICA interferes with TAN recruitment, with a particular emphasis on SMAD4-mediated modulation of the CXCL8-CXCR2 axis. The significance of this work is multifaceted, as it addresses a critical gap in understanding the molecular mechanisms driving CRC progression and pro-metastatic processes, which continue to present formidable clinical challenges. The insights gained from this study have the potential to inform and advance therapeutic strategies for CRC and possibly other malignancies.
Human CRC cell lines SW480 (300302, CLS Cell Lines Service GmbH, Eppelheim, Germany) and Caco-2 (CL-0050, Procell, China), the normal human colonic epithelial cell line NCM460 (SNL-519, Wuhan Fine Biological Technology Co., Ltd., Wuhan, Hubei Province, China), and primary human peripheral blood neutrophils (QS-H819, Keycell, China) were purchased for the experiments. SW480 and Caco-2 cells were cultured in low-glucose Dulbecco’s modified Eagle’s medium (M17650, R and D Systems, Shanghai, China) supplemented with 10% foetal bovine serum (FBS; MBS2568758, MyBioSource, San Diego, CA, United States) and 1% penicillin-streptomycin mixture (PSF-1, Zen-Bio, Inc., NC, United States). NCM460 cells were maintained in RPMI-1640 complete medium (PM150110B, Pricella, Wuhan, Hubei Province, China) with 10% FBS and 1% penicillin-streptomycin. The morphology and purity of primary human peripheral blood neutrophils were evaluated by Wright’s staining according to the manufacturer’s instructions (C0135, Beyotime, China). These neutrophils were cultured in RPMI medium (11875093, Thermo Fisher Scientific, MA, United States) containing 10% FBS and 1% penicillin-streptomycin.
Caco-2 cells were cocultured with neutrophils and randomly divided into groups as follows: (1) Different concentrations of ICA (ICA-L/M/H: 25 μmol/L, 50 μmol/L, and 100 μmol/L; 13624, Cayman Chemical, MI, United States) or an equal volume of vehicle (control: DMSO) were added to the cells; and (2) Cells were treated with 100 μmol/L ICA and/or anti-CXCL8 antibody (PA1353, BosterBio, CA, United States), with the control group receiving an equal volume of vehicle (DMSO and immunoglobulin G).
To silence SMAD4 expression, small interfering (si) RNA targeting SMAD4 and a negative control (NC) siRNA were obtained from Shanghai GenePharma (Shanghai, China). Transfection into Caco-2 cells was carried out using Lipo
Total RNA was extracted using the total RNA extraction kit (EP014, ELK Biotechnology, Denver, CO, United States) according to the manufacturer’s protocol. Reverse transcription was performed using the UltraScrip™ complementary DNA synthesis kit (PB30.11-02, PCR Biosystems, London, United Kingdom) to generate complementary DNA, followed by reverse transcription-quantitative polymerase chain reaction to detect target gene expression. Primers were designed and synthesized by Sangon Biotech Co., Ltd. (Shanghai, China), and their sequences are listed in Table 1. mRNA expres
| Name | Forward | Reverse |
| SMAD4 | GCTGAGTACCGAGGAGCAGA | GGAGGAGGGAGAGGAGGAAG |
| CXCL1 | TGGCATTCAAGGAGTGTGAC | GGTGGCTTGAGGTGAGTGGT |
| CXCL8 | TCTGCAGCTCTGTGTGAAGG | AGACAGAGCTGCAGAAATCAG |
| TNF-α | AGCCCATGTTGTAGCAAACC | GGTCTGGGCCATAGAACTGA |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
| CXCL10 | ATCATCCCTGCGAGCCTATCCT | GACCTTTTTTGGCTAAACGCTTTC |
| Icam2 | GACGGTCTCAACTTTTCCTGCC | CCATTTGGTTGTCCTGCATCGG |
| Arg1 | CATTGGCTTGCGAGACGTAGAC | GCTGAAGGTCTCTTCCATCACC |
| MMP9 | GCTGACTACGATAAGGACGGCA | TAGTGGTGCAGGCAGAGTAGGA |
| GAPDH | TGCACCACCAACTGCTTAGC | GGCATGGACTGTGGTCATGAG |
Proteins were extracted from cells or tissues using rapamycin-induced proximity assay buffer (R0278, Merck, Germany), and protein concentrations were determined using the Bradford assay (abx090644, Abbexa Ltd, United Kingdom). The samples were mixed with sodium-dodecyl sulfate gel electrophoresis loading buffer (B7021S, New England Biolabs, MA, United States) and separated by electrophoresis. After transferring the proteins onto membranes, 5% bovine serum albumin (MBS2567133, MyBioSource, San Diego, CA, United States) was used to block nonspecific binding sites. The membranes were then washed and incubated with the following primary antibodies: Matrix metalloproteinase 2 (MMP2; 1:500, A6247, ABclonal, Wuhan, Hubei Province, China), MMP9 (1:500, A0289, ABclonal, Wuhan, Hubei Province, China), E-cadherin (1:500, A3044, ABclonal, Wuhan, Hubei Province, China), α-catenin (1:500, A5635, ABclonal, Wuhan, Hubei Province, China), vimentin (1:2000, A19607, ABclonal, Wuhan, Hubei Province, China), SMAD4 (1:500, A5657, ABclonal, Wuhan, Hubei Province, China), CXCL1 (1:500, A24999, ABclonal, Wuhan, Hubei Province, China), and CXCL8 (1:500, A24736, ABclonal, Wuhan, Hubei Province, China). After incubation with the appropriate secondary antibodies, mem
Neutrophil migration was assessed using Transwell chambers (CLS3422, Merck, Germany). The lower compartment was filled with CRC cell supernatant, and the upper chamber contained neutrophils. Anti-CXCL8 antibody and/or ICA were added to the lower chamber. Neutrophils were treated with or without SB225002 (orb1306985, Biorbyt, United Kingdom) before being seeded into the upper chamber. After incubation, the proportion of CXCR2+ neutrophils that migrated into the lower chamber was quantified using flow cytometry.
Cells collected from the lower chamber of the Transwell system were labeled with CD66b (MBS212531, MyBioSource, San Diego, CA, United States) and CXCR2 (A00455-1, BosterBio, CA, United States) antibodies, incubated in the dark, rinsed with phosphate buffered saline (PBS), and analyzed by flow cytometry. For apoptosis analysis, treated CRC cells were resuspended in PBS and subsequently incubated in binding buffer containing Annexin V-FITC (MBS258045, MyBioSource, San Diego, CA, United States) and propidium iodide (640914, BioLegend, San Diego, CA, United States). After 15 minutes of incubation, cell apoptosis was assessed by flow cytometry.
CRC cells were seeded in 96-well plates, and various concentrations of ICA (0 μmol/L, 25 μmol/L, 50 μmol/L, and 100 μmol/L) were added to each group. After 12 hours, 24 hours, and 48 hours of incubation, cell counting kit-8 reagent (C0037, Beyotime, China) was added to each well and incubated for an additional 2 hours. Absorbance at 450 nm was then measured to assess cell viability.
Transwell assay: Cells were treated according to the method described for 72 hours. After removing the drug-containing culture medium, cells were rinsed with PBS, replaced with fresh complete medium, and cultured for another 12 hours. Supernatants were collected to prepare conditioned medium. CRC cells were then seeded in the upper chamber of Transwell inserts (CLS3422, Merck, Germany) precoated with Matrigel matrix (356255, Corning Life Sciences, MA, United States). The lower chamber was filled with conditioned medium, while the upper chamber contained the CRC cell sus
Scratch assay: CRC cells prepared as a single-cell suspension were seeded in 6-well plates. Once cells reached confluence, a straight scratch was made vertically along a reference line on the back of the well using a 200 μL pipette tip. After rinsing with PBS, cells were incubated in serum-free medium. Scratch closure was observed and photographed under a microscope at 0 hour and 48 hours after scratching.
A prepared suspension of CRC Caco-2 cells was subcutaneously inoculated into the right axilla of nude mice, with each mouse receiving 0.1 mL. After five days of inoculation, flat or oval-shaped masses gradually appeared at the injection site, indicating successful establishment of the subcutaneous xenograft model of CRC. Tumor-bearing BALB/c nude mice (6 weeks old; strain 401, Vital River, Beijing, China) were randomly assigned to either the model group or the ICA-treated group according to a random number table (n = 5 per group). On the seventh day postinoculation, when the diameter of the subcutaneous tumor reached approximately 0.5 cm, drug treatment was initiated. Mice received intraperitoneal injections of DMSO or ICA (9 mg/mL) at a volume of 0.2 mL. Tumor volume in each mouse was measured every four days following administration. Animals were housed in individually ventilated cages (five per cage) under controlled environmental conditions (20-26 °C, 40%-70% relative humidity, 12 hours/12 hours light/dark cycle), with ad libitum access to standard chow and water. On the second day after cessation of treatment, blood was collected from the orbital sinus, and mice in each group were euthanized. Tumor tissues were aseptically dissected and preserved by freezing.
Tumor tissues were processed, dehydrated, embedded, and sectioned. The sections were examined under an optical microscope to evaluate histological alterations in the tissues, which were appropriately stained with hematoxylin and eosin (C0105S, Beyotime, China).
Tumor tissues obtained by aseptic dissection from each group of nude mice were fixed in 10% neutral buffered formalin, and paraffin sections were prepared following dehydration, embedding, and sectioning. Primary antibodies against E-cadherin (1:50, A3044, ABclonal, Wuhan, Hubei Province, China), α-catenin (1:50, A5635, ABclonal, Wuhan, Hubei Province, China), vimentin (1:100, A19607, ABclonal, Wuhan, Hubei Province, China), SMAD4 (1:50, A5657, ABclonal, Wuhan, Hubei Province, China; 1:50, M00074, BosterBio, CA, United States), CD66b (1:25, MBS215571, MyBioSource, San Diego, CA, United States), CXCL1 (2 μg/mL, A00533-2, BosterBio, CA, United States), CXCL8 (1 μg/mL, PB9621, BosterBio, CA, United States), and CXCR2 (5 μg/mL, A00455-2, BosterBio, CA, United States) were applied, followed by incubation with secondary antibodies (1 mg/mL, BA1003, BosterBio, CA, United States) for 60 minutes. DAB (3,3’-diami
Interleukin-1β (ELH-IL1b-1, RayBiotech, MA, United States) and tumor necrosis factor-α (DY211-05, R and D Systems, Shanghai, China) levels were quantified using enzyme-linked immunosorbent assay kits according to the manufacturers’ instructions.
Data analysis was conducted using GraphPad Prism software, and results are presented as mean ± SD. Statistical significance between groups was determined using one-way or two-way ANOVA for multiple group comparisons and t-tests for two-group comparisons. Histological analyses were performed and quantified using ImageJ software.
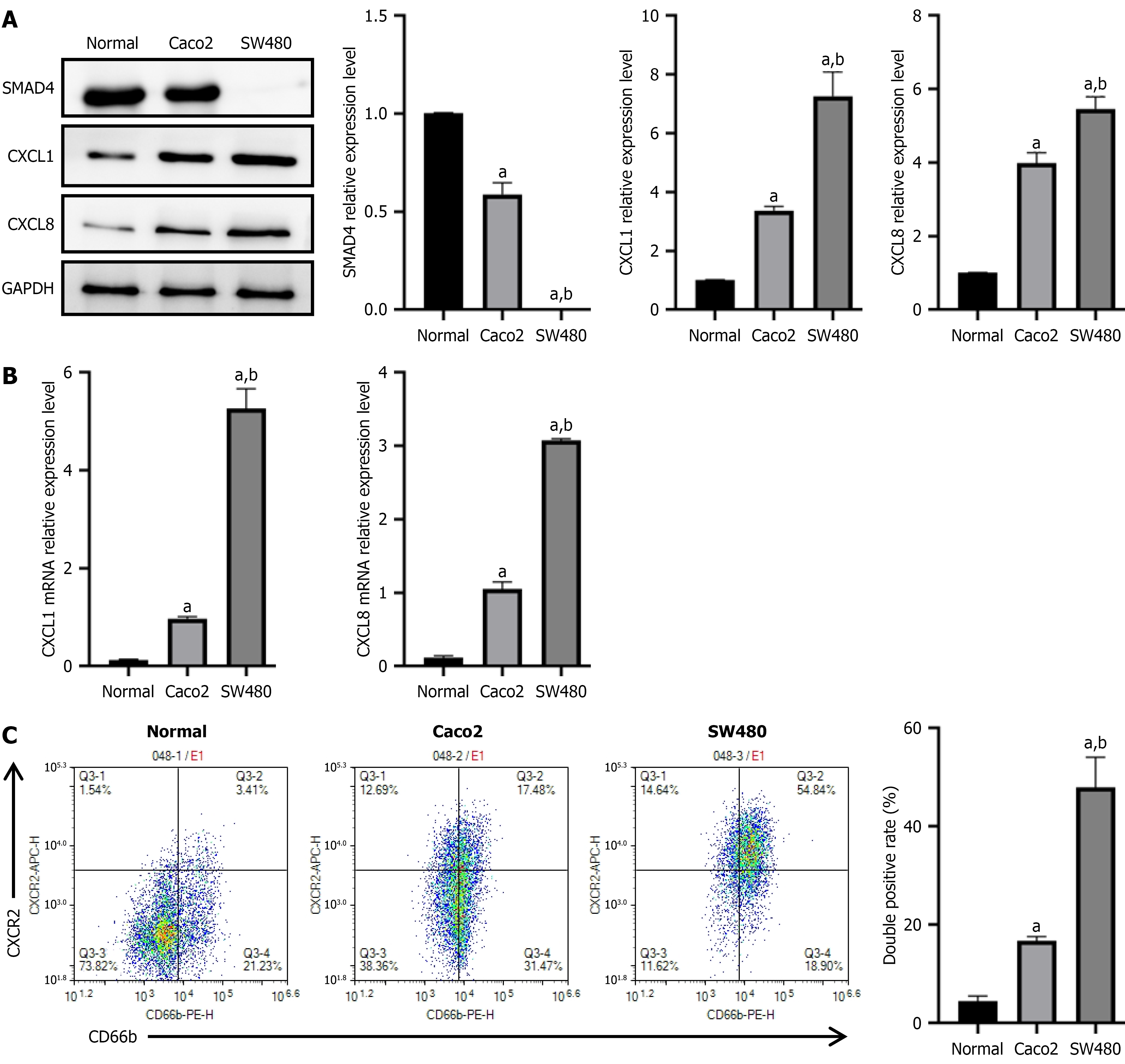
First, we evaluated SMAD4 expression in the human normal colonic epithelial cell line NCM460 (normal) and CRC cell lines Caco-2 and SW480. The results showed that SMAD4 expression was markedly lower in CRC cells than in normal cells, whereas SW480 exhibited no SMAD4 expression at all (Figure 1A). Moreover, we found that CXCL1 and CXCL8 protein and mRNA levels were elevated in cancer cells, particularly in SW480 cells (Figure 1A and B). We then explored the interaction between CRC cells and neutrophils. The upper compartment was filled with neutrophils, while the lower chamber contained CRC cell supernatants. Flow cytometry was used to determine the proportion of CXCR2+ TANs in the bottom chamber. The migratory response of CXCR2+ TANs to SMAD4-deficient cells (SW480) was observed to be more than twice that of the Caco-2 group (Figure 1C).
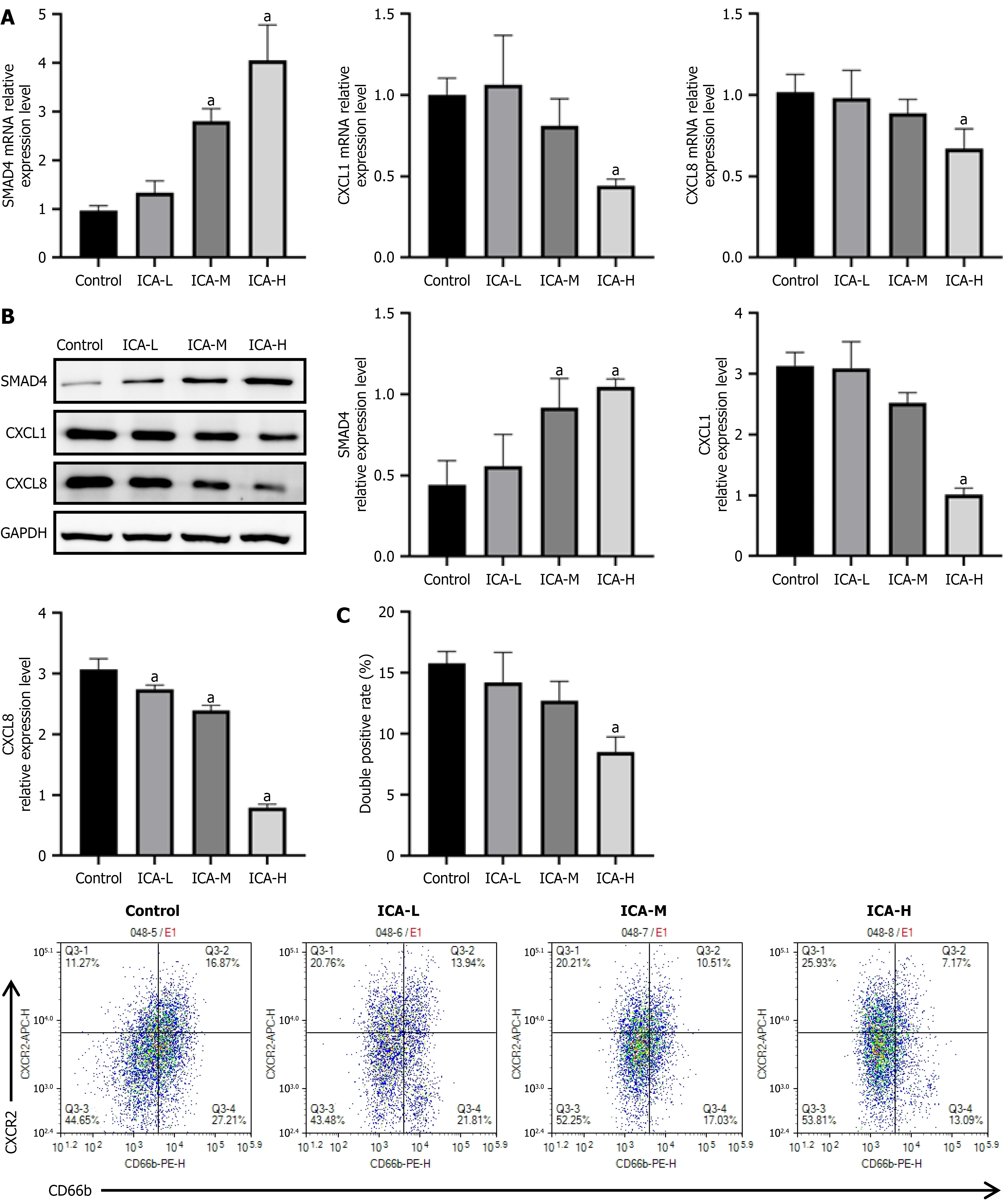
Based on preliminary literature research, we identified the notable antitumor potential of ICA. To specifically examine the antitumor mechanisms of ICA, we tested the effects of different ICA concentrations (ICA-L/M/H: 25 μmol/L, 50 μmol/L, and 100 μmol/L) on gene expression in CRC cells. As shown in Figure 2A and B, ICA reduced CXCL1 and CXCL8 protein and mRNA expression while increasing SMAD4 expression at both levels. Likewise, at high ICA concentrations, CXCR2+ TAN recruitment decreased significantly (Figure 2C).
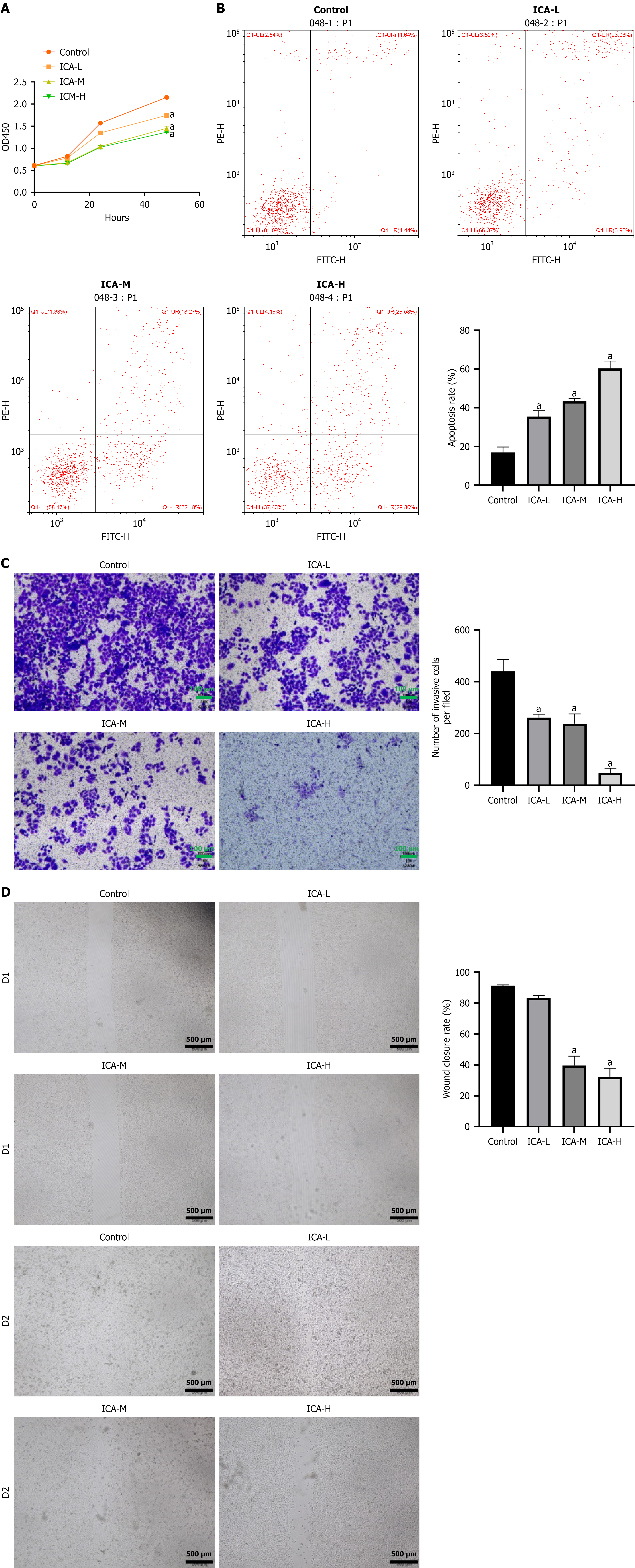
We subsequently investigated the effects of ICA on the biological behaviors of CRC cells by coculturing them with neutrophils. As depicted in Figure 3, ICA suppressed CRC cell proliferation, migration, and invasion, and these effects were most pronounced at the highest ICA concentration. Additionally, ICA promoted apoptosis in CRC cells.
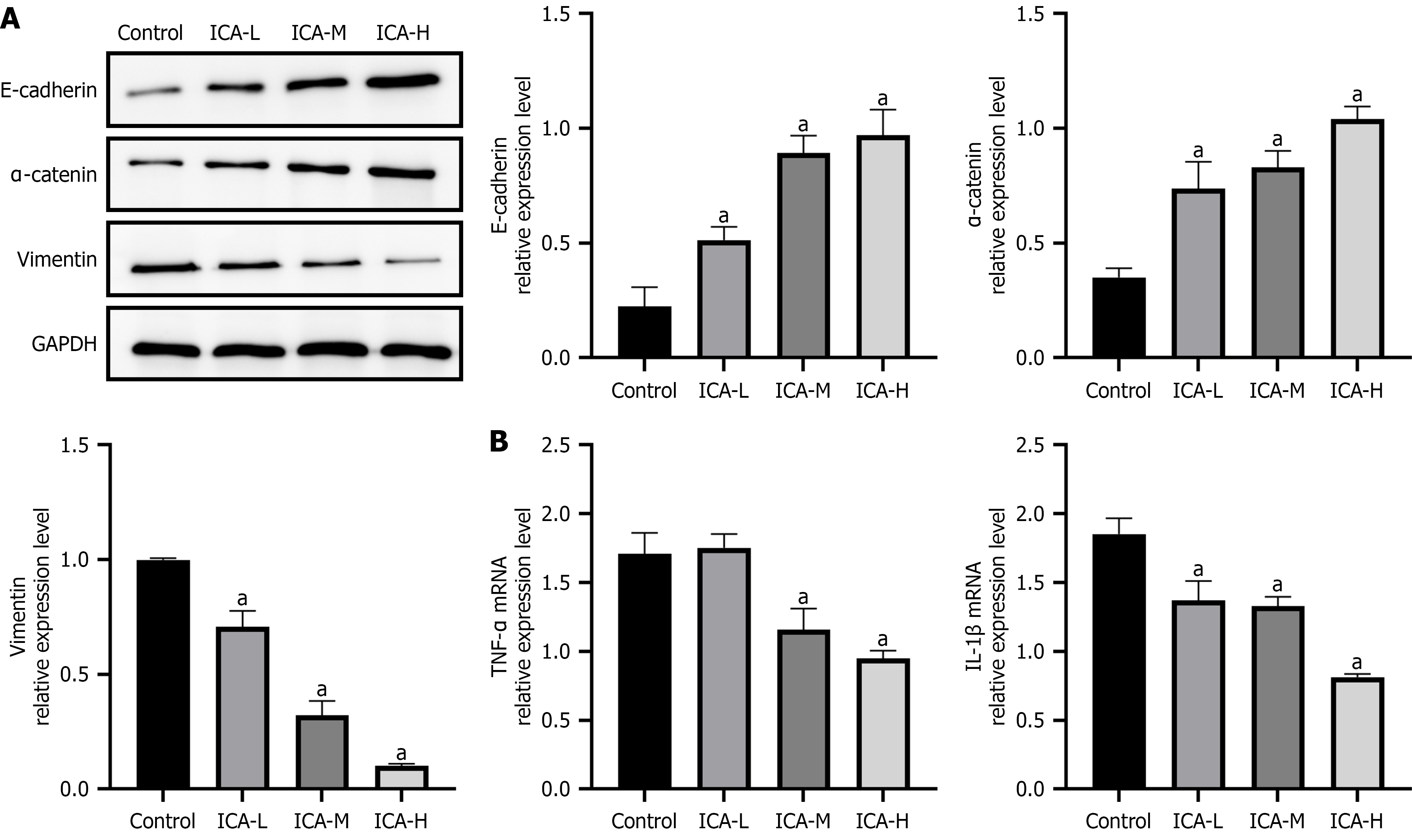
Then, we conducted additional investigations into the effects of different ICA concentrations on the expression of epithelial-mesenchymal transition (EMT)-related proteins in the coculture system of neutrophils and CRC cells. As illustrated in Figure 4A, ICA enhanced the expression of E-cadherin and α-catenin while reducing vimentin expression, suggesting its potential role in suppressing EMT. Moreover, we observed a decrease in the mRNA expression levels of interleukin-1β and tumor necrosis factor-α following ICA treatment (Figure 4B). A correlation was observed between ICA concentration and the aforementioned effects.
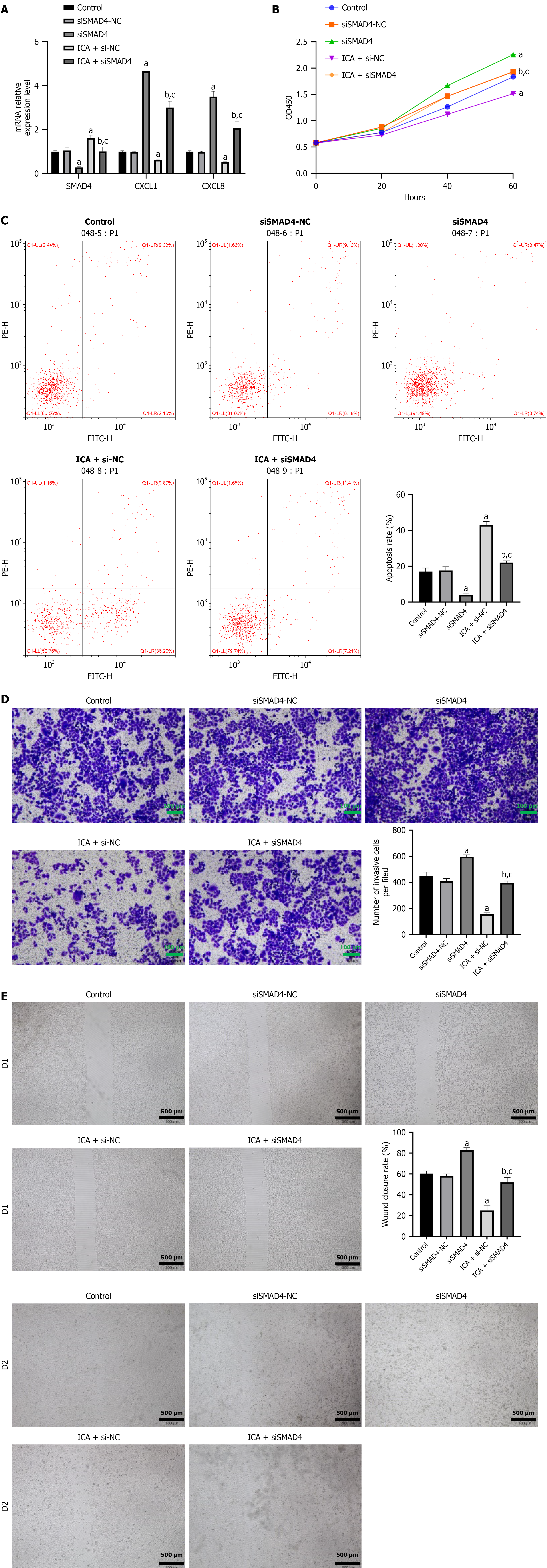
To further explore the role of SMAD4 in mediating the antitumor effects of ICA, we employed siSMAD4 to interfere with the pharmacological activity of ICA. The cocultured cells were divided into control, siSMAD4-NC, siSMAD4, ICA + si-NC, and ICA + siSMAD4 groups. As shown in Figure 5, the mRNA expression of SMAD4 significantly decreased in the siSMAD4 group, attenuating the upregulatory effect of ICA on SMAD4. Furthermore, experimental data indicated that the siSMAD4 NC did not significantly affect the results. In contrast to the siSMAD4-NC group, CXCL1 and CXCL8 mRNA expression was markedly increased in the siSMAD4 group, while ICA administration reduced their expression. However, when combined with siSMAD4, the inhibitory effects of ICA were reversed (Figure 5).
As illustrated in Figure 5, the inhibitory effects of ICA on CRC cell proliferation, migration, and invasion were reversed when SMAD4 expression was downregulated. Similarly, compared with the ICA group, the ICA + siSMAD4 group showed a marked decrease in the apoptotic rate. Therefore, it can be inferred that the antitumor actions of ICA on CRC cells are mediated through SMAD4 upregulation. When SMAD4 expression is suppressed, the therapeutic efficacy of ICA is significantly reduced.
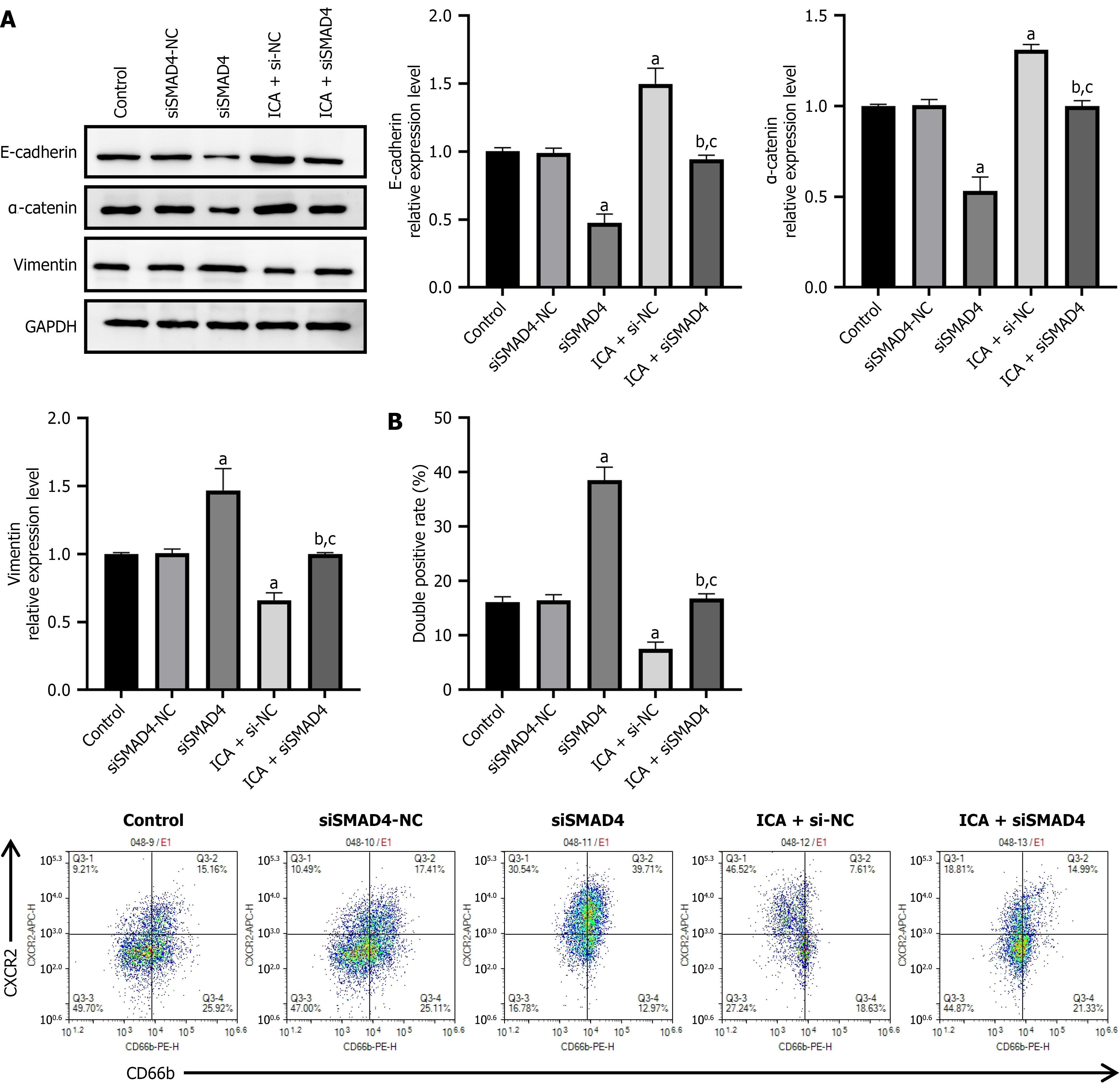
Furthermore, when SMAD4 was silenced, the inhibitory effect of ICA on EMT was reversed, as evidenced by lower expression of E-cadherin and α-catenin and higher levels of vimentin in the ICA + siSMAD4 group compared with the ICA group (Figure 6A). Similarly, the attenuation of CXCR2+ TAN recruitment by ICA was weakened upon SMAD4 downregulation (Figure 6B).
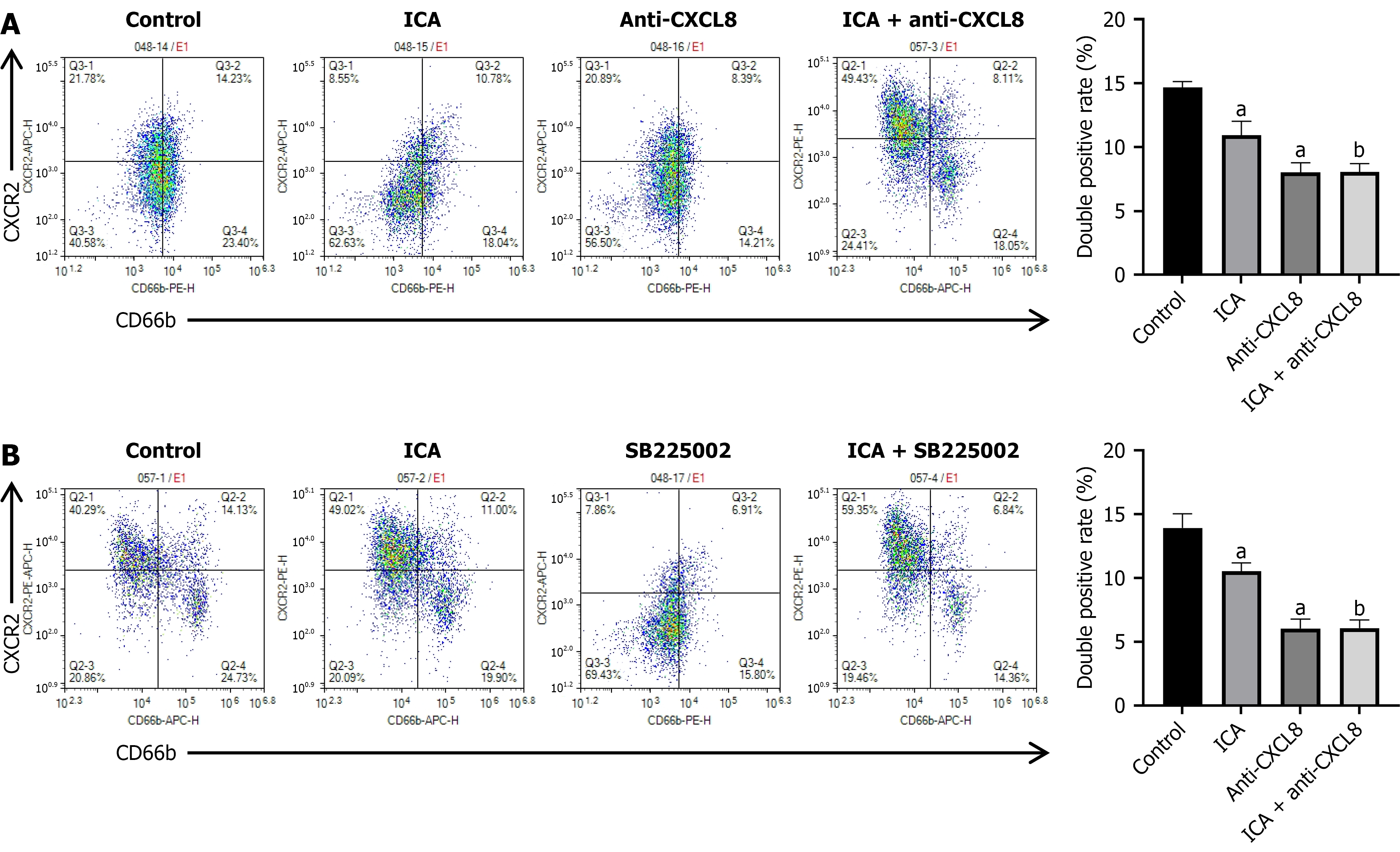
Subsequently, we investigated the combined effects of ICA and a CXCL8 antibody by adding the antibody to the lower chamber. As shown in Figure 7A, the recruitment of CXCR2+ TANs was inhibited in both the ICA and CXCL8 antibody groups. However, the combined treatment did not produce a more pronounced inhibitory effect on CXCR2+ TAN recruitment. Likewise, introducing SB225002 (a CXCR2 inhibitor) into the upper chamber suppressed CXCR2+ TAN recruitment when ICA and SB225002 were administered separately, yet the combined administration did not exhibit an additive inhibitory effect (Figure 7B). Our findings suggest that inhibition of the CXCL8-CXCR2 axis can suppress CXCR2+ TAN recruitment and that the additional application of ICA following CXCL8-CXCR2 axis blockade does not further enhance this inhibitory effect. In conclusion, we propose that ICA suppresses the recruitment of CXCR2+ TANs in CRC cells primarily through the CXCL8-CXCR2 axis.
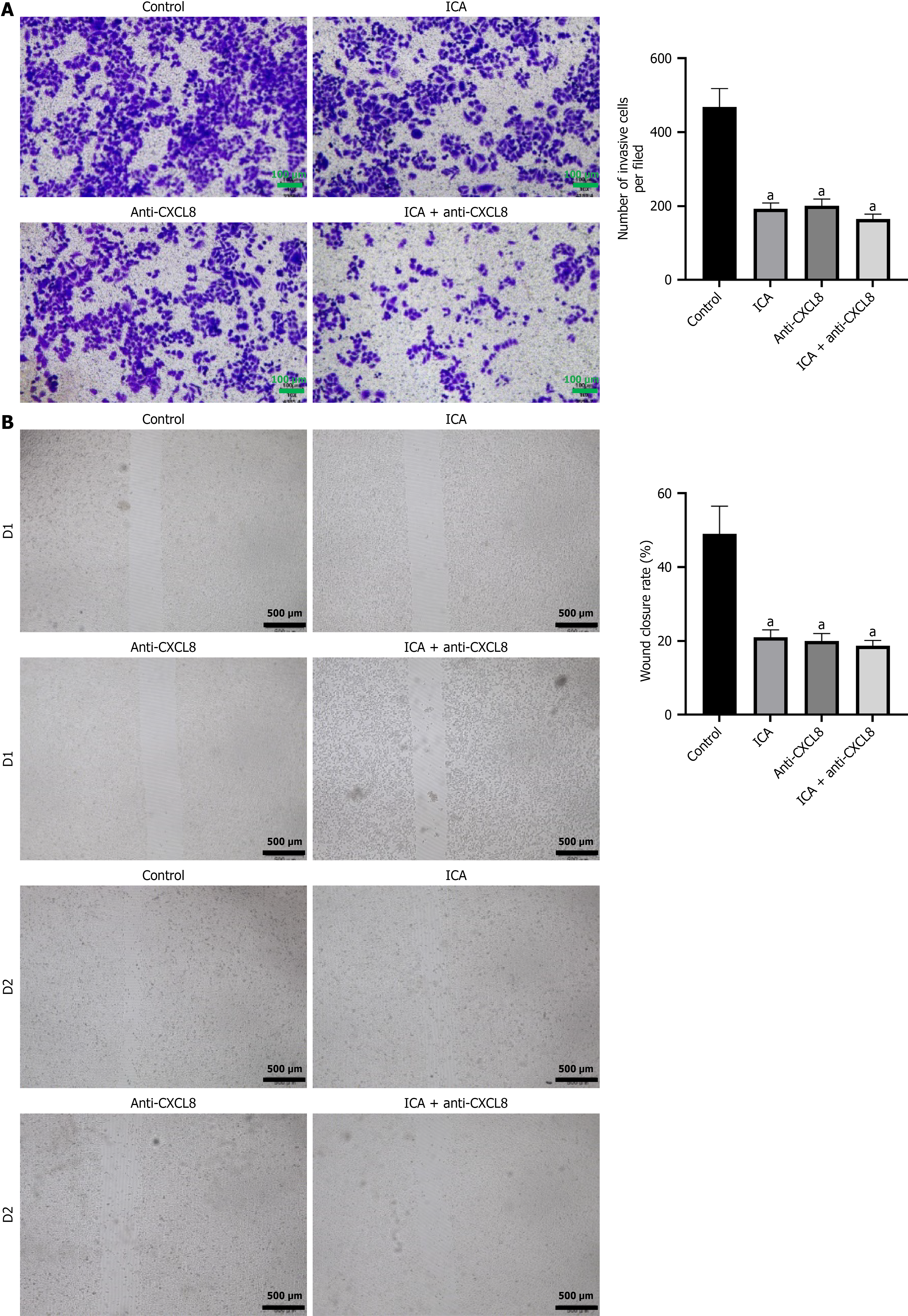
Further results revealed that the CXCL8 antibody could inhibit the migration and invasion of CRC cells. When used in combination, CRC cell migration was slightly further suppressed, while the inhibitory effect on invasion showed no significant change (Figure 8). Additionally, in the above experiments, we observed that ICA reduced CXCL8 protein expression. Thus, we conclude that by downregulating CXCL8 expression, ICA exerts inhibitory effects on CRC cell inva
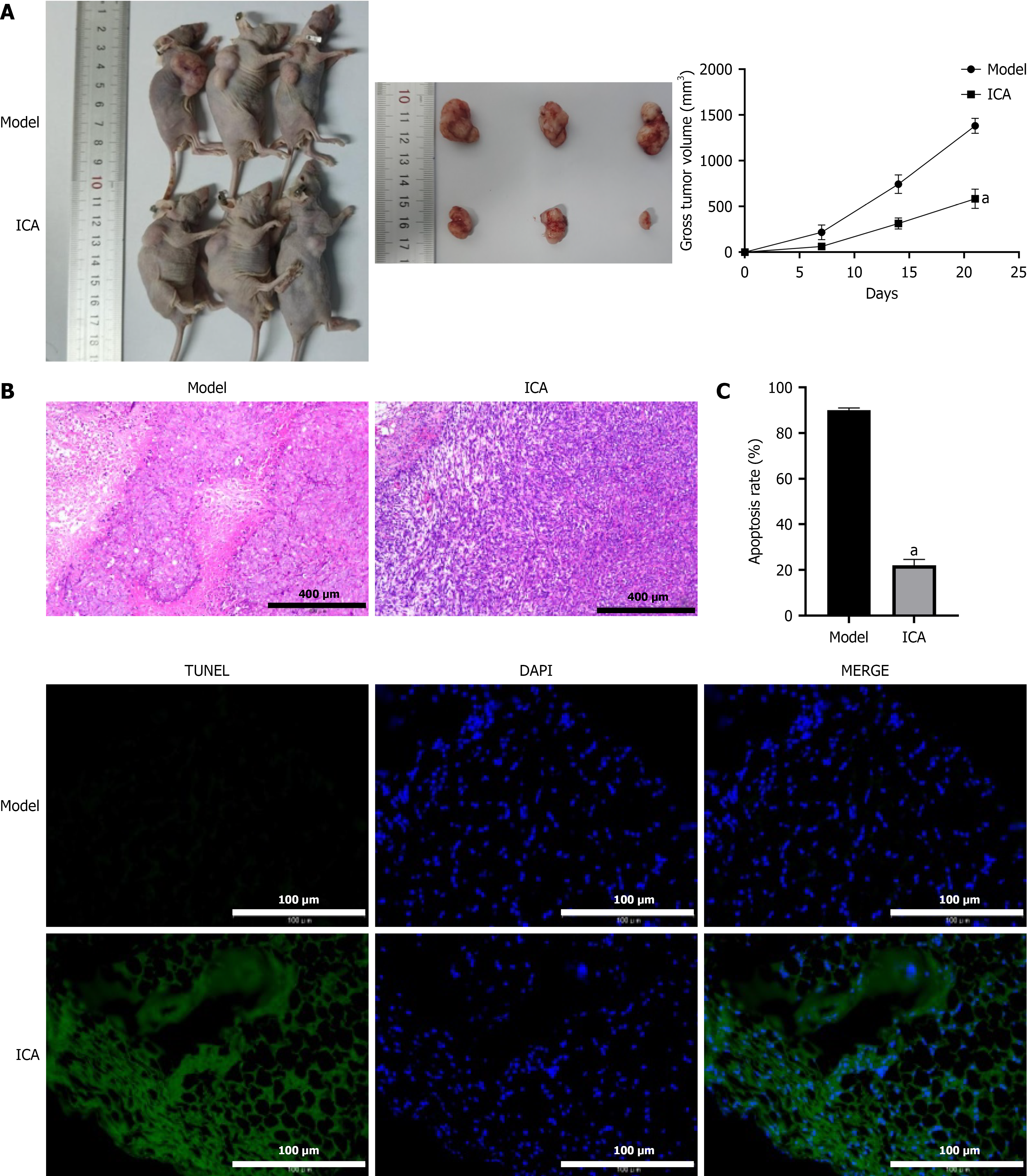
To validate the antitumor efficacy of ICA, we established a subcutaneous tumor model in CRC mice and administered ICA treatment to evaluate its effects. As shown in Figure 9A, ICA treatment significantly inhibited tumor growth. More
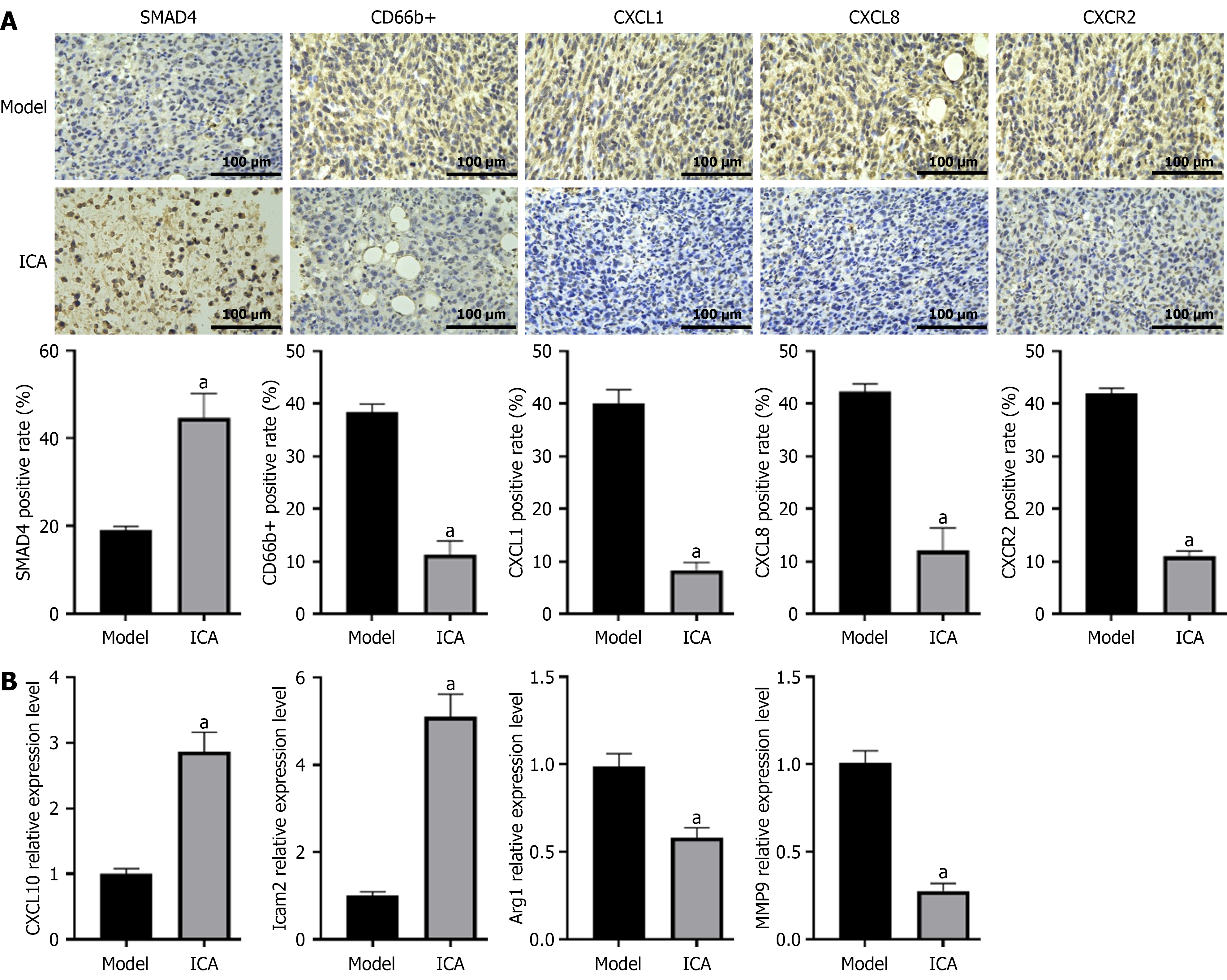
To gain deeper insights into the antitumor mechanisms of ICA, we conducted a comprehensive analysis of neutrophil infiltration-associated protein expression in tumor tissues. As illustrated in Figure 10A, ICA treatment substantially upre
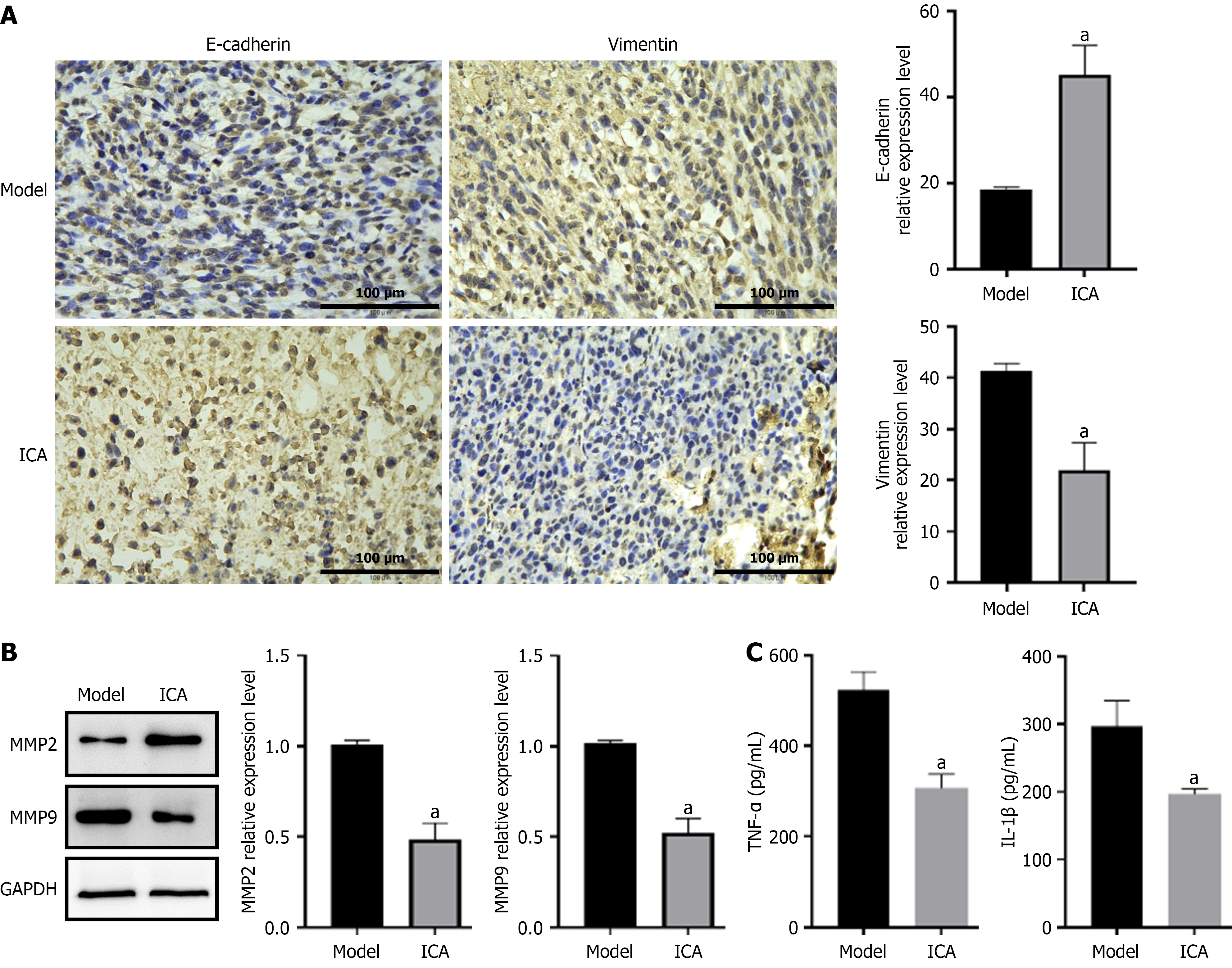
Subsequently, our data demonstrated that ICA therapy increased E-cadherin protein expression, decreased vimentin expression, and suppressed the EMT process. Moreover, ICA therapy reduced the production of MMP2 and MMP9 proteins, as well as the levels of inflammatory markers in tumor tissue (Figure 11).
CRC is a complex and aggressive malignancy involving diverse molecular and cellular processes[30]. In this study, we highlighted the role of SMAD4, a critical mediator of TGF-β signaling in CRC progression, and the potential therapeutic benefits of ICA. Aberrant SMAD4 expression has been linked to CRC development[31], while the TME, particularly neutrophil infiltration, plays an essential role in cancer progression[32]. Under normal circumstances, the TGF-β/SMAD4 signaling pathway plays a crucial role in regulating gene expression and participating in biological processes such as the cell cycle[33], apoptosis[34], and cell differentiation[35], thereby exerting a tumor-suppressive effect[36]. However, in CRC, our findings revealed a significant reduction in SMAD4 expression, particularly in SW480 cells. Concurrently, CXCL1 and CXCL8, two inflammation-related chemokines, showed elevated protein and mRNA expression in cancer cells. CXCR2, a receptor located on the surface of neutrophils, facilitates their migration to sites of inflammation by bin
Importantly, our study provides deeper insight into the immunomodulatory role of ICA, extending beyond its primary function of reducing neutrophil numbers. The in vivo phenotypic analysis of TANs revealed that ICA not only decreased overall infiltration but also markedly altered the polarization state of the remaining cells. As shown in the N1/N2 phenotype characterization, ICA treatment promoted a shift in the TAN population toward an antitumor N1 phenotype while concurrently suppressing the protumor N2 phenotype. This reprogramming indicates that ICA’s beneficial impact within the TME is twofold: It diminishes the overall load of protumorigenic neutrophils. It actively cultivates an antitumor immune milieu by directing TAN polarization toward a tumoricidal state. These findings significantly refine our understanding of ICA’s mechanism of action, highlighting its role as both a modulator of neutrophil function and an inhibitor of their recruitment.
Research indicates that cancer cells attract neutrophils to tumor sites by releasing chemotactic factors such as CXCL1 and CXCL8[40]. Neutrophils, in turn, remodel the extracellular matrix of the TME by releasing proteases, thereby facilitating cancer cell migration[41]. Chemotactic factors activate cancer cell migration pathways by binding to corresponding receptors such as the interleukin-8 receptor CXCR2, enhancing invasiveness[42]. Neutrophils also secrete proteases, including MMPs[43], which degrade matrix components such as collagen and fibronectin, creating an environment conducive to tumor invasion[44]. During EMT, cell-surface adhesion proteins E-cadherin and α-catenin decrease, whereas mesenchymal proteins N-cadherin and vimentin increase[45], altering adhesion and motility and enhancing migratory potential[46]. Additionally, the TGF-β/SMAD signaling pathway plays a key regulatory role in EMT, influencing cellular morphology and function[47]. Consistent with previous reports, our results indicated that ICA exerted a suppressive effect on EMT both in vivo and in vitro, as evidenced by increased E-cadherin and α-catenin levels along with reduced vimentin expression. Moreover, ICA inhibited proliferation, migration, and invasion of CRC cells while promoting apoptosis in vitro and suppressed MMP2 and MMP9 expression in vivo. These findings support our hypothesis that ICA impedes cancer progression and metastasis.
In summary, our research demonstrates that ICA possesses potent antitumor activity in CRC, inhibiting tumor growth and limiting pro-metastatic programs. ICA achieves this by upregulating SMAD4 expression and exerting inhibitory effects on a network of CXCR2 chemokines (including CXCL1 and CXCL8), thereby reducing the migration of CXCR2+ TANs to the tumor site. Consequently, ICA exerts multiple inhibitory effects on tumor metastasis, including the sup
Our results also raise important considerations for clinical translation. A key implication lies in identifying patient subgroups that might derive the greatest benefit from ICA or its derivatives. Given that ICA acts in part by upregulating SMAD4 and counteracting the protumorigenic inflammation associated with SMAD4 deficiency, it is plausible that CRC patients with low SMAD4 expression could respond more favorably to such treatment. This hypothesis aligns with the principles of precision oncology and warrants further validation. Future studies should aim to confirm SMAD4 exp
However, several limitations of this study should be acknowledged. First, our in vivo experiments lacked a positive control group (e.g., a standard chemotherapeutic agent such as 5-fluorouracil or a mechanism-based targeted therapy), which would have better contextualized the relative efficacy and therapeutic potential of ICA within the current treat
In conclusion, ICA predominantly acts through a SMAD4-modulated CXCL8-CXCR2 axis to reduce TAN recruitment and restrain pro-metastatic programs in CRC.
| 1. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14385] [Cited by in RCA: 15438] [Article Influence: 2573.0] [Reference Citation Analysis (4)] |
| 2. | Wei MM, Wang SS, Zheng JL, Chen L, Peng X, Chen JF, An HM, Hu B. Herbal Medicine Teng-Long-Bu-Zhong-Tang Inhibits the Growth of Human RKO Colorectal Cancer by Regulating Apoptosis, Senescence, and Angiogenesis. World J Tradit Chin Med. 2022;8:110-114. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467-1480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4063] [Cited by in RCA: 3540] [Article Influence: 505.7] [Reference Citation Analysis (10)] |
| 4. | Peng P, Luan Y, Sun P, Wang L, Zeng X, Wang Y, Cai X, Ren P, Yu Y, Liu Q, Ma H, Chang H, Song B, Fan X, Chen Y. Prognostic Factors in Stage IV Colorectal Cancer Patients With Resection of Liver and/or Pulmonary Metastases: A Population-Based Cohort Study. Front Oncol. 2022;12:850937. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 5. | Ni Y, Liang Y, Li M, Lin Y, Zou X, Han F, Cao J, Li L. The updates on metastatic mechanism and treatment of colorectal cancer. Pathol Res Pract. 2023;251:154837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 6. | de Visser KE, Joyce JA. The evolving tumor microenvironment: From cancer initiation to metastatic outgrowth. Cancer Cell. 2023;41:374-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3233] [Cited by in RCA: 2668] [Article Influence: 889.3] [Reference Citation Analysis (4)] |
| 7. | Rossi GR, Trindade ES, Souza-Fonseca-Guimaraes F. Tumor Microenvironment-Associated Extracellular Matrix Components Regulate NK Cell Function. Front Immunol. 2020;11:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 8. | Bonavita O, Massara M, Bonecchi R. Chemokine regulation of neutrophil function in tumors. Cytokine Growth Factor Rev. 2016;30:81-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 9. | Maas RR, Soukup K, Fournier N, Massara M, Galland S, Kornete M, Wischnewski V, Lourenco J, Croci D, Álvarez-Prado ÁF, Marie DN, Lilja J, Marcone R, Calvo GF, Santalla Mendez R, Aubel P, Bejarano L, Wirapati P, Ballesteros I, Hidalgo A, Hottinger AF, Brouland JP, Daniel RT, Hegi ME, Joyce JA. The local microenvironment drives activation of neutrophils in human brain tumors. Cell. 2023;186:4546-4566.e27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 151] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 10. | Aghamajidi A, Farhangnia P, Pashangzadeh S, Damavandi AR, Jafari R. Tumor-promoting myeloid cells in the pathogenesis of human oncoviruses: potential targets for immunotherapy. Cancer Cell Int. 2022;22:327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 11. | Masucci MT, Minopoli M, Carriero MV. Tumor Associated Neutrophils. Their Role in Tumorigenesis, Metastasis, Prognosis and Therapy. Front Oncol. 2019;9:1146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 509] [Article Influence: 72.7] [Reference Citation Analysis (0)] |
| 12. | Zhang Y, Guoqiang L, Sun M, Lu X. Targeting and exploitation of tumor-associated neutrophils to enhance immunotherapy and drug delivery for cancer treatment. Cancer Biol Med. 2020;17:32-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 86] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 13. | Hurt B, Schulick R, Edil B, El Kasmi KC, Barnett C Jr. Cancer-promoting mechanisms of tumor-associated neutrophils. Am J Surg. 2017;214:938-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 106] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 14. | Li C, Xu Y, Zhang J, Zhang Y, He W, Ju J, Wu Y, Wang Y. The effect of resveratrol, curcumin and quercetin combination on immuno-suppression of tumor microenvironment for breast tumor-bearing mice. Sci Rep. 2023;13:13278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 44] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 15. | Bordbari S, Mörchen B, Pylaeva E, Siakaeva E, Spyra I, Domnich M, Droege F, Kanaan O, Lang KS, Schadendorf D, Lang S, Helfrich I, Jablonska J. SIRT1-mediated deacetylation of FOXO3a transcription factor supports pro-angiogenic activity of interferon-deficient tumor-associated neutrophils. Int J Cancer. 2022;150:1198-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 16. | Sheng Y, Peng W, Huang Y, Cheng L, Meng Y, Kwantwi LB, Yang J, Xu J, Xiao H, Kzhyshkowska J, Wu Q. Tumor-activated neutrophils promote metastasis in breast cancer via the G-CSF-RLN2-MMP-9 axis. J Leukoc Biol. 2023;113:383-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 17. | Gong L, Cumpian AM, Caetano MS, Ochoa CE, De la Garza MM, Lapid DJ, Mirabolfathinejad SG, Dickey BF, Zhou Q, Moghaddam SJ. Promoting effect of neutrophils on lung tumorigenesis is mediated by CXCR2 and neutrophil elastase. Mol Cancer. 2013;12:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 148] [Article Influence: 11.4] [Reference Citation Analysis (4)] |
| 18. | Liu X, Lan T, Mo F, Yang J, Wei Y, Wei X. Antitumor and Radiosensitization Effects of a CXCR2 Inhibitor in Nasopharyngeal Carcinoma. Front Cell Dev Biol. 2021;9:689613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Alazzouzi H, Alhopuro P, Salovaara R, Sammalkorpi H, Järvinen H, Mecklin JP, Hemminki A, Schwartz S Jr, Aaltonen LA, Arango D. SMAD4 as a prognostic marker in colorectal cancer. Clin Cancer Res. 2005;11:2606-2611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 151] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 20. | Fang T, Liang T, Wang Y, Wu H, Liu S, Xie L, Liang J, Wang C, Tan Y. Prognostic role and clinicopathological features of SMAD4 gene mutation in colorectal cancer: a systematic review and meta-analysis. BMC Gastroenterol. 2021;21:297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 21. | Zhao M, Mishra L, Deng CX. The role of TGF-β/SMAD4 signaling in cancer. Int J Biol Sci. 2018;14:111-123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 221] [Cited by in RCA: 482] [Article Influence: 60.3] [Reference Citation Analysis (1)] |
| 22. | Yamamoto T, Kawada K, Itatani Y, Inamoto S, Okamura R, Iwamoto M, Miyamoto E, Chen-Yoshikawa TF, Hirai H, Hasegawa S, Date H, Taketo MM, Sakai Y. Loss of SMAD4 Promotes Lung Metastasis of Colorectal Cancer by Accumulation of CCR1+ Tumor-Associated Neutrophils through CCL15-CCR1 Axis. Clin Cancer Res. 2017;23:833-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (3)] |
| 23. | Ogawa R, Yamamoto T, Hirai H, Hanada K, Kiyasu Y, Nishikawa G, Mizuno R, Inamoto S, Itatani Y, Sakai Y, Kawada K. Loss of SMAD4 Promotes Colorectal Cancer Progression by Recruiting Tumor-Associated Neutrophils via the CXCL1/8-CXCR2 Axis. Clin Cancer Res. 2019;25:2887-2899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (3)] |
| 24. | Bi Z, Zhang W, Yan X. Anti-inflammatory and immunoregulatory effects of icariin and icaritin. Biomed Pharmacother. 2022;151:113180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 133] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 25. | Martiniakova M, Babikova M, Omelka R. Pharmacological agents and natural compounds: available treatments for osteoporosis. J Physiol Pharmacol. 2020;71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 47] [Reference Citation Analysis (0)] |
| 26. | Song L, Chen X, Mi L, Liu C, Zhu S, Yang T, Luo X, Zhang Q, Lu H, Liang X. Icariin-induced inhibition of SIRT6/NF-κB triggers redox mediated apoptosis and enhances anti-tumor immunity in triple-negative breast cancer. Cancer Sci. 2020;111:4242-4256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 149] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 27. | Yang Y, Nian H, Tang X, Wang X, Liu R. Effects of the combined Herba Epimedii and Fructus Ligustri Lucidi on bone turnover and TGF-β1/Smads pathway in GIOP rats. J Ethnopharmacol. 2017;201:91-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 28. | Liang W, Lin M, Li X, Li C, Gao B, Gan H, Yang Z, Lin X, Liao L, Yang M. Icariin promotes bone formation via the BMP-2/Smad4 signal transduction pathway in the hFOB 1.19 human osteoblastic cell line. Int J Mol Med. 2012;30:889-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 29. | Xiong W, Ma H, Zhang Z, Jin M, Wang J, Xu Y, Wang Z. The protective effect of icariin and phosphorylated icariin against LPS-induced intestinal goblet cell dysfunction. Innate Immun. 2020;26:97-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 30. | Jin L, Chen Y, Cheng D, He Z, Shi X, Du B, Xi X, Gao Y, Guo Y. YAP inhibits autophagy and promotes progression of colorectal cancer via upregulating Bcl-2 expression. Cell Death Dis. 2021;12:457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 98] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 31. | Wu N, Jiang M, Liu H, Chu Y, Wang D, Cao J, Wang Z, Xie X, Han Y, Xu B. LINC00941 promotes CRC metastasis through preventing SMAD4 protein degradation and activating the TGF-β/SMAD2/3 signaling pathway. Cell Death Differ. 2021;28:219-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 131] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 32. | Bellomo G, Rainer C, Quaranta V, Astuti Y, Raymant M, Boyd E, Stafferton R, Campbell F, Ghaneh P, Halloran CM, Hammond DE, Morton JP, Palmer D, Vimalachandran D, Jones R, Mielgo A, Schmid MC. Chemotherapy-induced infiltration of neutrophils promotes pancreatic cancer metastasis via Gas6/AXL signalling axis. Gut. 2022;71:2284-2299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 33. | Guo B, Friedland SC, Alexander W, Myers JA, Wang W, O'Dell MR, Getman M, Whitney-Miller CL, Agostini-Vulaj D, Huber AR, Mello SS, Vertino PM, Land HK, Steiner LA, Hezel AF. Arid1a mutation suppresses TGF-β signaling and induces cholangiocarcinoma. Cell Rep. 2022;40:111253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 34. | Rajasekaran N, Song K, Lee JH, Wei Y, Erkin ÖC, Lee H, Shin YK. Nuclear Respiratory Factor-1, a Novel SMAD4 Binding Protein, Represses TGF-β/SMAD4 Signaling by Functioning as a Transcriptional Cofactor. Int J Mol Sci. 2021;22:5595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Hiwatashi K, Ueno S, Sakoda M, Kubo F, Tateno T, Kurahara H, Mataki Y, Maemura K, Ishigami S, Shinchi H, Natsugoe S. Strong Smad4 expression correlates with poor prognosis after surgery in patients with hepatocellular carcinoma. Ann Surg Oncol. 2009;16:3176-3182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 36. | Mohd Faheem M, Rasool RU, Ahmad SM, Jamwal VL, Chakraborty S, Katoch A, Gandhi SG, Bhagat M, Goswami A. Par-4 mediated Smad4 induction in PDAC cells restores canonical TGF-β/ Smad4 axis driving the cells towards lethal EMT. Eur J Cell Biol. 2020;99:151076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 37. | Korbecki J, Kupnicka P, Chlubek M, Gorący J, Gutowska I, Baranowska-Bosiacka I. CXCR2 Receptor: Regulation of Expression, Signal Transduction, and Involvement in Cancer. Int J Mol Sci. 2022;23:2168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 114] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 38. | Liu L, Wang Y, Yu S, Liu H, Li Y, Hua S, Chen YG. Transforming Growth Factor Beta Promotes Inflammation and Tumorigenesis in Smad4-Deficient Intestinal Epithelium in a YAP-Dependent Manner. Adv Sci (Weinh). 2023;10:e2300708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 39. | Galdiero MR, Marone G, Mantovani A. Cancer Inflammation and Cytokines. Cold Spring Harb Perspect Biol. 2018;10:a028662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 206] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 40. | SenGupta S, Hein LE, Parent CA. The Recruitment of Neutrophils to the Tumor Microenvironment Is Regulated by Multiple Mediators. Front Immunol. 2021;12:734188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 41. | Zhou J, Liu H, Jiang S, Wang W. Role of tumor-associated neutrophils in lung cancer (Review). Oncol Lett. 2023;25:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 42. | Xiong S, Dong L, Cheng L. Neutrophils in cancer carcinogenesis and metastasis. J Hematol Oncol. 2021;14:173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 476] [Cited by in RCA: 436] [Article Influence: 87.2] [Reference Citation Analysis (4)] |
| 43. | Germann M, Zangger N, Sauvain MO, Sempoux C, Bowler AD, Wirapati P, Kandalaft LE, Delorenzi M, Tejpar S, Coukos G, Radtke F. Neutrophils suppress tumor-infiltrating T cells in colon cancer via matrix metalloproteinase-mediated activation of TGFβ. EMBO Mol Med. 2020;12:e10681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 137] [Article Influence: 22.8] [Reference Citation Analysis (4)] |
| 44. | Zhu Y, Huang Y, Ji Q, Fu S, Gu J, Tai N, Wang X. Interplay between Extracellular Matrix and Neutrophils in Diseases. J Immunol Res. 2021;2021:8243378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 45. | Greco A, De Virgilio A, Rizzo MI, Pandolfi F, Rosati D, de Vincentiis M. The prognostic role of E-cadherin and β-catenin overexpression in laryngeal squamous cell carcinoma. Laryngoscope. 2016;126:E148-E155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 46. | Li HJ, Ke FY, Lin CC, Lu MY, Kuo YH, Wang YP, Liang KH, Lin SC, Chang YH, Chen HY, Yang PC, Wu HC. ENO1 Promotes Lung Cancer Metastasis via HGFR and WNT Signaling-Driven Epithelial-to-Mesenchymal Transition. Cancer Res. 2021;81:4094-4109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 100] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 47. | Yao Y, Chen R, Wang G, Zhang Y, Liu F. Exosomes derived from mesenchymal stem cells reverse EMT via TGF-β1/Smad pathway and promote repair of damaged endometrium. Stem Cell Res Ther. 2019;10:225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 204] [Article Influence: 29.1] [Reference Citation Analysis (0)] |