Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.115167
Revised: December 5, 2025
Accepted: January 16, 2026
Published online: March 14, 2026
Processing time: 143 Days and 23.3 Hours
Early postoperative edema and ascites after liver resection are common; however, the endocrine drivers of water retention are not fully defined. Arginine vaso
To examine postoperative changes in plasma AVP and plasma aldosterone con
We conducted a prospective cohort study of adults undergoing elective liver resection at a tertiary center. Blood samples were collected preoperatively, im
AVP increased sharply immediately after resection and remained above the preoperative baseline through POD 3, showing the most pronounced and sustained elevation after major liver resection. In contrast, PAC showed a transient postoperative increase that returned to near-baseline levels by POD 2. The period of elevated AVP closely matched the time frame during which early postoperative fluid retention was most evident, as indicated by greater short-term weight gain and reduced urine output. These patterns were consistent across sensitivity analyses and showed similar directional trends in subgroup comparisons based on resection extent.
AVP remains elevated longer than aldosterone and coincides with early fluid retention, particularly after major resection. Vasopressin-driven antidiuresis may be important in postoperative water retention.
Core Tip: This study prospectively assessed postoperative changes in arginine vasopressin (AVP) and plasma aldosterone concentration (PAC) after liver resection and found that AVP elevation persisted longer than PAC elevation, particularly after major resections. The duration of elevated AVP closely matched the period of early postoperative fluid retention, highlighting the important role of AVP beyond the renin-angiotensin-aldosterone system. These findings suggest that targeting the V2 signaling pathway may offer strategies for optimizing fluid management and reducing complications in high-risk patients undergoing liver resection. A detailed understanding of postoperative hormonal changes is crucial for future clinical trials and therapeutic decisions.
- Citation: Aoki Y, Kawano Y, Ga R, Endo K, Ueda J, Shimizu T, Yoshida H. Vasopressin and fluid retention after liver resection: Comparison with the renin-angiotensin-aldosterone system by surgical extent and liver function. World J Gastroenterol 2026; 32(10): 115167
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/115167.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.115167
Liver resection has been established as a curative surgery for primary or metastatic liver tumors and is an important treatment option for certain benign diseases. Advances in surgical techniques and improvements in perioperative management have increased surgical safety; however, postoperative complication rates (Clavien-Dindo classification[1] ≥ IIIa) remain relatively high at approximately 8.5%-11%, with a mortality rate of 1.3%-5%[2-5].
Fluid retention, such as ascites and edema, is a frequent postoperative complication that poses a challenge for postoperative management. In particular, a postoperative weight gain of 3.5 kg or more is considered an independent risk factor for postoperative complications[6]. In Japan, anti-aldosterone drugs that suppress the renin-angiotensin-aldosterone system (RAAS), such as canrenoate and spironolactone, have been empirically used since the 1980s to prevent excessive postoperative fluid retention[7]. It has long been reported that the RAAS is activated in patients with ascites caused by liver cirrhosis[8]. Furthermore, Kobayashi et al[9] reported that in cases complicated by liver cirrhosis, the postoperative plasma aldosterone concentration (PAC) increases significantly and that anti-aldosterone drugs improve the urinary sodium-to-potassium ratio and reduce fluid retention.
This clinical experience has also been reflected in recent guidelines for liver cirrhosis issued by the Japan Society of Gastroenterology, which designate spironolactone as the first-line drug[10]. Consistently, major international guidelines for cirrhosis including the American Association for the Study of Liver Diseases Practice Guidance[11] and the European Association for the Study of the Liver Clinical Practice Guidelines[12] also recommend aldosterone antagonists, particularly spironolactone, as the first-line diuretic therapy for cirrhotic ascites. However, some patients exhibit insufficient diuretic response or develop adverse effects such as renal impairment or hyperkalemia when treated with mineralocorticoid receptor antagonists. It has long been suggested that, in addition to the RAAS, arginine vasopressin (AVP) contributes to fluid retention associated with liver cirrhosis[13,14], and inappropriate AVP secretion has also been reported in animal models[15]. These findings indicate that both the RAAS and vasopressin axes are intricately involved in body fluid regulation in cirrhosis and may influence postoperative water balance after liver resection.
Classical studies have reported that plasma vasopressin levels transiently increase during laparotomy, thoracotomy, and laparoscopic surgery, reflecting the magnitude of surgical stress[16,17]. However, little is known about AVP dynamics during liver resection or its association with postoperative fluid retention. This study aimed to elucidate the postoperative hormonal dynamics of AVP and PAC and clarify their relationship with postoperative fluid retention following liver resection.
We included 56 consecutive patients who underwent hepatic resection at our hospital between March 2019 and October 2019. This non-interventional prospective study was approved by the Ethics Committee of Nippon Medical School Hospital[18] (approval No. 30-01-1070). Written informed consent was obtained from all patients, and data were collected prospectively.
Perioperative management followed the standard institutional protocol. The exclusion criteria included emergency surgery, dialysis-dependent renal failure, and cases requiring renal replacement therapy during the perioperative period. The primary endpoints were temporal changes in PAC and AVP levels, whereas the secondary endpoints were postoperative body weight gain, urine output, and drain discharge volume. Immediately after surgery, patients were admitted to the intensive care unit (ICU) for fluid management under strict monitoring. If there were no abnormalities, the patients were transferred to the general ward the following day. Fluid infusion was generally maintained at 1500-2000 mL/day. Oral intake was initiated on postoperative day (POD) 2, followed by gradual reduction or discontinuation of intravenous infusion based on the intake volume.
According to the standard protocol, 200 mg of potassium canrenoate was administered intravenously in the morning immediately after surgery and again on POD 1 and 2. Oral spironolactone (25 mg/day) was initiated on POD 1.
Body weight, urine output (mL/kg), and drain discharge volume (mL/kg) were recorded daily. The patient background characteristics (age, sex, underlying disease, liver function indices, and surgical procedures) are presented in the results section. Maximum postoperative weight gain (%) was defined as 100 × (maximum postoperative body weight - preoperative body weight)/preoperative body weight. Intraoperative fluid balance (mL/kg/hour) was calculated as (total infused fluids - blood loss - urine output)/body weight/operative time.
PAC and AVP were measured preoperatively (baseline), immediately after surgery, and in the morning on POD 1, 2, 3, and 5 after at least 10 minutes of rest in the supine position. Measurements were outsourced to BML Inc. (Tokyo, Japan). PAC was measured by chemiluminescent enzyme immunoassay (pg/mL); AVP, double-antibody radioimmunoassay (RIA) (pg/mL); and plasma renin activity (PRA), enzyme immunoassay (ng/mL/hour).
Stratification by liver function was based on the indocyanine green retention rate at 15 minutes (ICGR15), with patients with values < 10% and ≥ 10% defined as the normal liver (NL) and impaired liver (IL) groups, respectively.
Following conventions used in literature referencing the Brisbane 2000 terminology[19], major liver resection (MajLR) was defined as resection of ≥ 3 Couinaud segments. In this cohort, the MajLR group included patients who underwent right or left hemihepatectomy and left trisectionectomy but did not undergo central bisectionectomy. Minor liver re
Statistical analyses were performed using JMP Pro 18.0.2 (SAS Institute, Cary, NC, United States). Hormone levels over time were compared using the Wilcoxon signed-rank test for differences between preoperative values and each postoperative time point, and the Mann-Whitney U test was used for intergroup comparisons. Continuous variables were expressed as mean ± SD or median (interquartile range), depending on the distribution. Body weights, which followed a normal distribution, were compared using a paired t-test. Post-hoc urine output on POD 1 and POD 2 was compared between patients with and without hypervasopressinemia on POD 1 using the unpaired Student t-test as the data were normally distributed. A two-sided P value < 0.05 was considered statistically significant. Given the exploratory nature of this study, no adjustments for multiple comparisons were made. The cut-off for hypervasopressinemia (AVP ≥ 4.0 pg/mL) was determined according to the reference range specified by the assay provider (double-antibody RIA). Missing data were handled using complete-case analysis, and no outlier exclusion was performed.
This study included 56 patients (37 men and 19 women) with a median age of 69 years. Primary indications for liver resection were colorectal liver metastasis (CRLM) (n = 25) and hepatocellular carcinoma (HCC) (n = 20). Based on liver function, there were 29 and 27 patients in the IL group (ICGR15 ≥ 10%) and NL group (ICGR15 < 10%), respectively. Based on the surgical procedure, there were 11 and 45 cases of MajLR and 45 MinLR, respectively. A laparoscopic approach was used in 22 patients. Extrahepatic bile duct resection was performed in four cases, and concomitant colorectal resection was performed in three cases. The median intraoperative fluid balance was 8.1 mL/kg/hour, and the median operative time was 295 minutes (Table 1). Regarding medications that could influence RAAS or water balance, spironolactone was taken preoperatively by 9 patients, all of whom belonged to the IL group. Angiotensin II receptor blockers were used by 15 patients (27%), with no notable differences among the IL, NL, MajLR, or MinLR groups. Two patients in the IL group were taking angiotensin-converting enzyme inhibitors. No patients were receiving tolvaptan preoperatively.
| Characteristics | All patients (n = 56) |
| Age (years), median (IQR) | 69 (64-75) |
| Sex (men/women) | 37 (66.0)/19 (33.9) |
| Indication of liver resection | |
| Colorectal liver metastasis | 25 (44.6) |
| Hepatocellular carcinoma | 20 (35.7) |
| Biliary tract cancer | 5 (8.9) |
| Others | 6 (10.7) |
| Indocyanine green retention rate at 15 minutes (%), median (IQR) | 10.2 (7.2-14.5) |
| Kinetic indocyanine green elimination rate constant, median (IQR) | 0.155 (0.129-0.177) |
| Fibrosis-4 index, median (IQR) | 1.82 (1.21-3.06) |
| Albumin-bilirubin scoring | -2.58 ± 0.42 |
| Modified ALBI grade (1/2a/2b/3) | 26 (46.4)/18 (32.1)/12 (21.4)/0 (0) |
| Number of complete resected Couinaud segments, median (IQR) | 0 (0-2) |
| Laparoscopic surgery | 22 (39.3) |
| Extrahepatic bile duct resection | 4 (7.1) |
| Concomitant colorectal resection | 3 (5.4) |
| Operative duration (minutes), median (IQR) | 295 (224-379) |
| Intraoperative blood loss (mL), median (IQR) | 333 (100-600) |
| Intraoperative red blood cell transfusion | 11 (19.6) |
| Intraoperative fluid balance (mL/kg/hour), median (IQR) | 8.1 (6.4-11.9) |
| Preoperative body weight (kg), median IQR | 58.2 (52.6-66.4) |
| Maximum postoperative weight gain (%), median (IQR) | 2.7 (1.1-6.4) |
| Complications (Clavien-Dindo grade ≥ IIIa) (%) | 12 (21.4) |
Postoperatively, tolvaptan was administered in 4 patients, all of whom were in the IL group; two of these were in the MajLR group. Furosemide was used postoperatively in 12 patients (21%), with no significant differences between MajLR and MinLR (22% vs 18%) or between the IL and NL groups (28% vs 15%). Postoperative complications of Clavien-Dindo grade ≥ IIIa occurred in 12 patients (21%), half of whom were in the MajLR group.
When examining the overall time course for all cases, body weight increased significantly from a preoperative value of 60.3 ± 12.0 kg to a postoperative peak of 62.3 ± 11.8 kg (P < 0.001). However, by POD 5, this difference disappeared (P = 0.264), with a peak in weight gain observed between POD 2 and 3. Urine output also increased from POD 2 (33.4 ± 14.9 mL/kg body weight) to POD 3 (34.1 ± 13.6 mL/kg body weight) and then gradually decreased. Serum sodium concentration showed a transient rise from 140.4 ± 2.6 mEq/L preoperatively to 142.0 ± 2.5 mEq/L immediately after surgery (P < 0.001) and then decreased to a minimum at POD 2 (139.5 ± 3.0 mEq/L, P = 0.015). On POD 3 and 5, serum sodium levels generally returned to the preoperative values. The lowest and highest serum sodium concentration levels observed postoperatively were 130 mEq/L and 148 mEq/L, respectively. No cases with severe electrolyte abnormalities were identified. Urinary sodium concentration decreased postoperatively, reaching its lowest level at POD 2 (69.9 ± 47.5 mEq/L vs 113.5 ± 50.0 mEq/L preoperatively; P < 0.001), and by POD 5, it was no longer different from the preoperative values. Serum osmolality also increased from 284 mOsm/L (282-287 mOsm/L) preoperatively to 293 mOsm/L (289-296 mOsm/L) immediately after surgery (P < 0.001) and returned to baseline by POD 2 at 285 mOsm/L (280-287 mOsm/L) (P = 0.963).
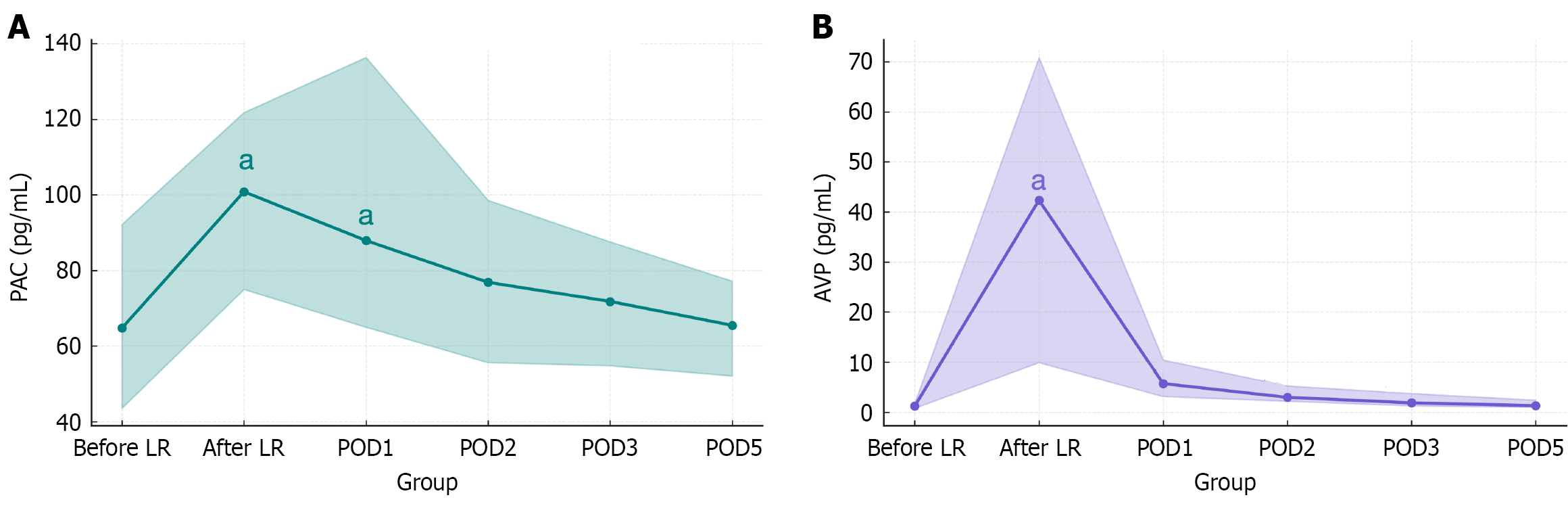
Regarding hormonal dynamics (Figure 1), PAC was significantly elevated immediately after surgery and on POD 1 compared with preoperative values (both P < 0.001), but it returned to baseline by POD 2 (P = 0.072), with no significant differences thereafter. In contrast, AVP peaked immediately after surgery and remained significantly elevated until POD 3 compared with preoperative levels (all P < 0.001), returning to baseline by POD 5 (P = 0.926).
PRA was 64.8 ng/mL/hour (42.6-93.8 ng/mL/hour) preoperatively and peaked at 100.8 ng/mL/hour (74.6-122.5 ng/mL/hour) immediately after surgery (P < 0.001). Although the PRA gradually decreased thereafter, it remained elevated at POD 5 at 65.5 ng/mL/hour (52.0-78.0 ng/mL/hour) compared with baseline (P < 0.001). PAC showed a similar pattern but returned to levels not significantly different from baseline by POD 2.
Serum creatinine levels decreased slightly to 0.68 mg/dL (0.55-0.76 mg/dL) on POD 3 compared with the preoperative value of 0.69 mg/dL (0.59-0.81 mg/dL) (P = 0.040) and returned to 0.69 mg/dL (0.58-0.76 mg/dL) on POD 5 (P = 0.077), indicating no postoperative renal impairment.
These clinical changes namely postoperative weight gain, transient hyponatremia, and reduced urinary sodium excretion around POD 2 were temporally consistent with the sustained elevation of AVP, suggesting predominant free water retention in the early postoperative period.
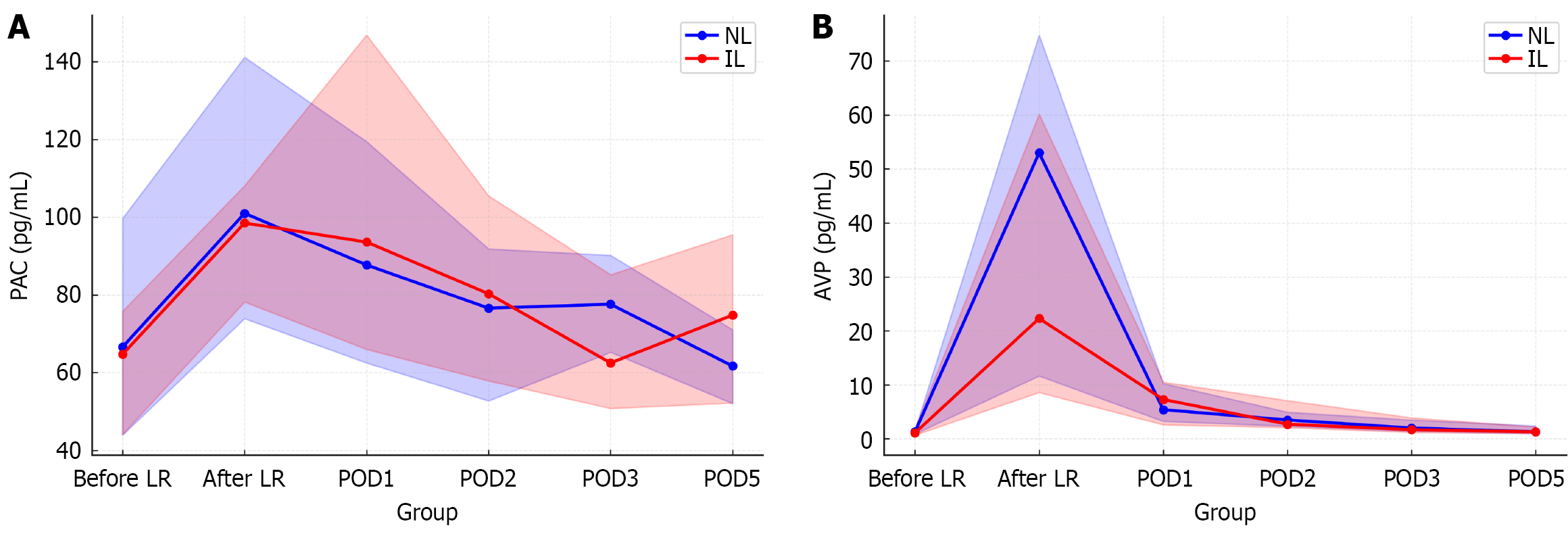
When comparing liver function, the median age was higher in the IL group [71 years (68-77 years) vs 66 years (60-74 years), P = 0.034], and this group included a greater proportion of patients with HCC. There were no significant differences in sex or distribution of surgical procedures between the groups. Regarding liver function indicators, the IL group showed a significantly higher fibrosis-4 (FIB-4) index[20] and worse modified albumin-bilirubin (ALBI) grade[21] compared with the NL group, whereas the ALBI score[22] tended to be higher but did not reach statistical significance (Table 2). In both the NL and IL groups, AVP levels were higher than preoperative values immediately after liver resection, and on POD 1 and POD 2 (all P < 0.001), with the elevation persisting on POD 3 (IL, P = 0.006; NL, P = 0.008). No significant differences were observed between the groups at any time point. For PAC, the IL group showed a marked increase immediately after surgery and on POD 1 (both P < 0.001). In the NL group, PAC increased only immediately after surgery (P < 0.001), with no significant differences thereafter. At all time points, there were no significant differences in PAC levels between the groups (Figure 2). No difference was observed in urine output per kilogram of body weight or in drainage output based on the presence or absence of liver dysfunction.
| NL group (n = 27) | IL group (n = 29) | P value | |
| Age (years), median (IQR) | 66 (60-74) | 71 (68-77) | 0.034a |
| Sex (men/women) | 15 (55.6)/12 (44.4) | 22 (75.9)/7 (24.1) | 0.109 |
| Indication of liver resection | 0.003b | ||
| Colorectal liver metastasis | 16 (59.3) | 9 (31.0) | |
| Hepatocellular carcinoma | 3 (11.1) | 17 (55.6) | |
| Biliary tract cancer | 4 (14.8) | 1 (3.5) | |
| Others | 4 (14.8) | 2 (6.9) | |
| Indocyanine green retention rate at 15 minutes (%), median (IQR) | 7.2 (6.2-7.8) | 14.1 (11.7-18.8) | < 0.001b |
| Kinetic indocyanine green elimination rate constant, median (IQR) | 0.175 (0.170-0.186) | 0.131 (0.111-0.143) | < 0.001b |
| Fibrosis-4 index, median (IQR) | 1.46 (1.13-2.38) | 2.48 (1.37-4.35) | 0.014a |
| Albumin-bilirubin scoring | -2.69 ± 0.08 | -2.49 ± 0.08 | 0.082 |
| Modified ALBI grade (1/2a/2b) | 15 (55.6)/10 (37.0)/2 (7.4) | 11(37.9)/8 (27.6)/10 (34.5) | 0.047a |
| Number of complete resected Couinaud segments, median (IQR) | 1 (0-2) | 0 (0-2) | 0.419 |
| Laparoscopic surgery | 10 (37.0) | 12 (41.1) | 0.740 |
| Extrahepatic bile duct resection | 2 (7.4) | 2 (6.9) | 0.941 |
| Concomitant colorectal resection | 2 (7.4) | 1 (3.5) | 0.511 |
| Operative duration (minute), median (IQR) | 297 (218-328) | 288 (224-384) | 0.850 |
| Intraoperative blood loss (mL), median (IQR) | 388 (100-641) | 290 (83.5-523) | 0.774 |
| Intraoperative red blood cell transfusion | 5 (18.5) | 6 (20.7) | 0.838 |
| Intraoperative fluid balance (mL/kg/hour), median (IQR) | 8.1 (6.4-12.0) | 8.2 (6.6-12.4) | 0.844 |
| Preoperative body weight (kg), median (IQR) | 57.7 (51.9-71.8) | 59.7 (53.3-64.2) | 1.000 |
| Maximum postoperative weight gain (%), median (IQR) | 2.4 (1.0-6.1) | 3.1 (1.5-7.5) | 0.403 |
| Complications (Clavien-Dindo grade ≥ IIIa) (%) | 4 (14.8) | 8 (27.6) | 0.245 |
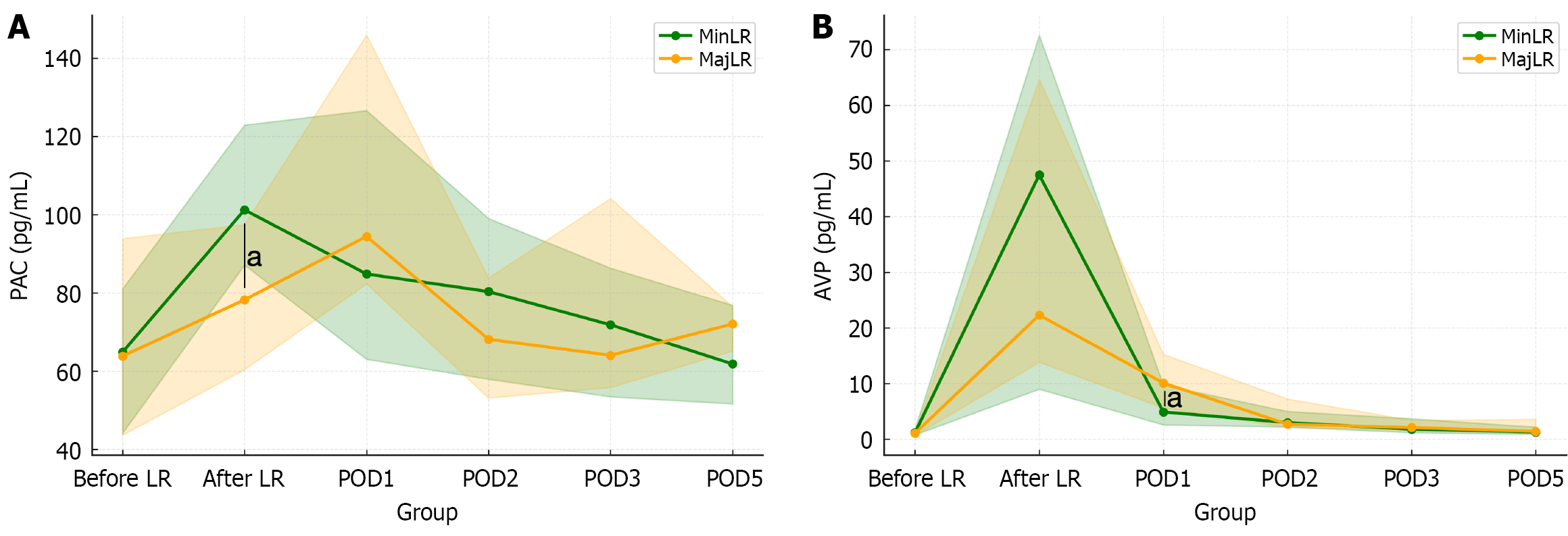
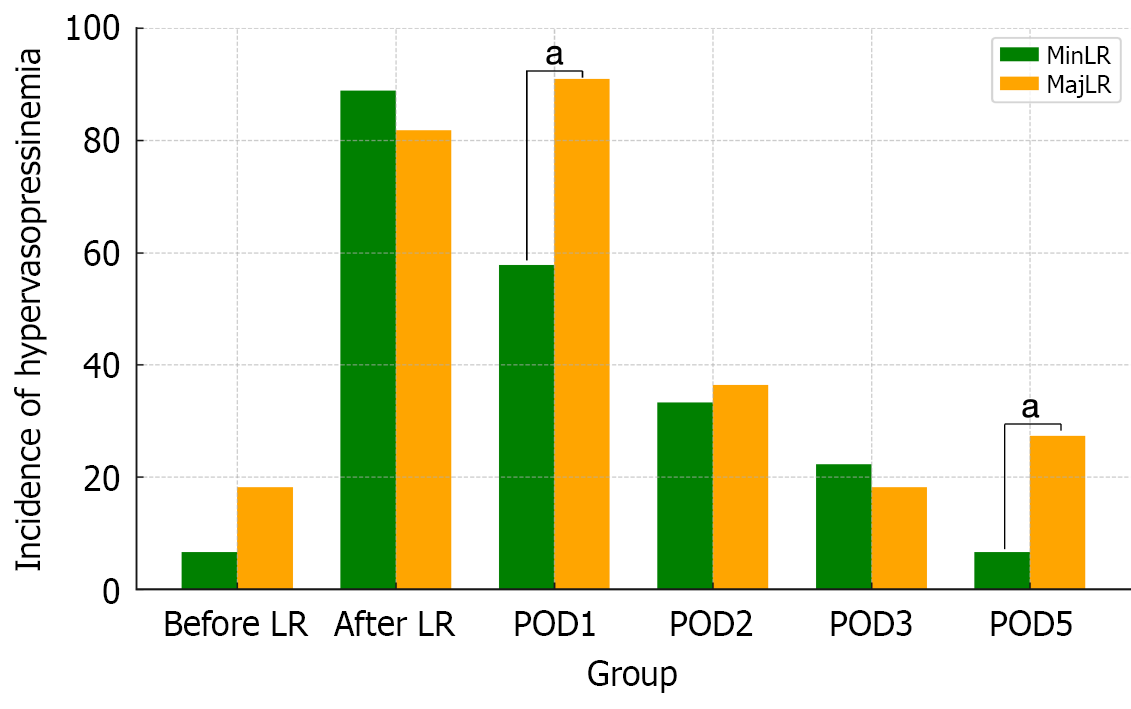
When comparing the surgical procedures, there were no significant differences in age, sex, preoperative body weight, or liver function indices. The MajLR group had a significantly longer operative time and greater blood loss. In addition, biliary tract cancer requiring extrahepatic bile duct resection was more frequently observed in this group. Although the difference was not statistically significant, the intraoperative water balance per unit body weight per hour was generally lower in the MajLR group (Table 3). Rates of body weight change and urine output were comparable between the two groups; however, drain output was significantly higher in the MajLR group: POD 1 [5.1 mL/kg (2.3-7.1 mL/kg) vs 2.0 mL/kg (1.2-3.6 mL/kg) body weight, P = 0.004], POD 2 [2.9 mL/kg (0.9-6.8 mL/kg) vs 1.2 mL/kg (0.6-2.6 mL/kg) body weight, P = 0.055], and POD 3 [3.8 mL/kg (1.3-11.7 mL/kg) vs 0.8 mL/kg (0.4-1.9 mL/kg) body weight, P = 0.005]. PAC was higher in the MinLR group than in the MajLR group immediately after surgery [101.2 pg/mL (81.7-132.2 pg/mL) vs 78.2 pg/mL (54.5-100.6 pg/mL), P = 0.037], but this difference disappeared by POD 1 and did not reappear thereafter. Both groups showed peaks in AVP immediately after surgery, with no difference between them at that time; however, on POD 1, AVP was significantly higher in the MajLR group [10.1 pg/mL (5.4-15.7 pg/mL) vs 4.9 pg/mL (2.6-9.8 pg/mL), P = 0.036] (Figure 3). From POD 2 onward, AVP gradually decreased in both groups, with no significant group differences. When AVP ≥ 4.0 pg/mL was defined as hypervasopressinemia, its occurrence was significantly more frequent in the MajLR group on POD 1 (91% vs 58% in the MinLR group, P = 0.04), and this trend persisted on POD 5 (27% vs 7%, P = 0.05) (Figure 4). There was no difference in this tendency based on the surgical approach (laparoscopic or open).
| MinLR group (n = 45) | MajLR group (n = 11) | P value | |
| Age (years), median (IQR) | 69 (65-75) | 69 (64-77) | 0.942 |
| Sex (men/women) | 31 (68.9)/14 (31.1) | 6 (54.6)/5 (45.5) | 0.368 |
| Indication of liver resection | 0.053 | ||
| Colorectal liver metastasis | 23 (51.1) | 2 (18.2) | |
| Hepatocellular carcinoma | 15 (33.3) | 5 (45.5) | |
| Biliary tract cancer | 2 (4.4) | 3 (27.3) | |
| Others | 5 (11.1) | 1 (9.1) | |
| Indocyanine green retention rate at 15 minutes (%), median (IQR) | 10.0 (7.4-17.2) | 10.4 (6.2-14.1) | 0.458 |
| Kinetic indocyanine green elimination rate constant, median (IQR) | 0.155 (0.118-0.173) | 0.165 (0.131-0.186) | 0.265 |
| Number of complete resected Couinaud segments, median (IQR) | 0 (0-1) | 3 (3-4) | < 0.001b |
| Laparoscopic surgery | 19 (42.2) | 3 (27.3) | 0.363 |
| Extrahepatic bile duct resection | 0 (0.0) | 4 (36.4) | < 0.001b |
| Concomitant colorectal resection | 3 (6.7) | 0 (0.0) | 0.379 |
| Operative duration (minute), median (IQR) | 278 (207-337) | 382 (301-665) | 0.002b |
| Intraoperative blood loss (mL), median (IQR) | 290 (67-484) | 1124 (150-2188) | 0.015a |
| Intraoperative red blood cell transfusion | 7 (15.6) | 4 (36.4) | 0.119 |
| Intraoperative fluid balance (mL/kg/hour), median (IQR) | 8.9 (6.8-12.5) | 6.9 (4.5-8.8) | 0.058 |
| Preoperative body weight (kg), median (IQR) | 58.1 (52.2-68.0) | 59.7 (54.0-65.0) | 0.845 |
| Maximum postoperative weight gain (%), median (IQR) | 2.6 (1.2-6.3) | 3.5 (1.0-6.4) | 0.918 |
| Complications (Clavien-Dindo grade ≥ IIIa) (%) | 6 (13.3) | 6 (54.6) | 0.003b |
In summary, PAC rose transiently from immediately after surgery to POD 1 and normalized by POD 2, whereas AVP remained elevated until at least POD 3; the prevalence of AVP ≥ 4.0 pg/mL was significantly higher after MajLR. In particular, MajLR was associated with prolonged elevation of AVP and a greater frequency of hypervasopressinemia, suggesting a role for AVP in fluid retention management. When hypervasopressinemia was observed on POD 1, urine output per kilogram of body weight was significantly lower on POD 1 (23.7 ± 1.5 mL/kg vs 32.0 ± 2.5 mL/kg body weight, P = 0.010) and POD 2 (30.3 ± 2.4 mL/kg vs 38.9 ± 3.2 mL/kg body weight, P = 0.036).
In this study, we observed the following findings after liver resection: (1) A transient increase in body weight, peaking at POD 2-3; (2) PAC levels that rose immediately after surgery and returned to baseline by POD 2; and (3) AVP levels that peaked immediately after surgery and remained elevated until at least POD 3, with a particularly high frequency of elevated vasopressin (AVP ≥ 4.0 pg/mL) in MajLR cases. Interestingly, patients who underwent MajLR had higher postoperative AVP levels despite appropriate fluid replacement in the ICU. During liver resection, intraoperative fluid infusion is intentionally restricted as part of a low central venous pressure strategy to minimize hepatic venous bleeding[23-25], which transiently reduces effective circulating volume and may stimulate AVP secretion. In addition, a longer operative time and greater surgical stress during MajLR may further enhance vasopressin release. The persistence of elevated AVP levels even after adequate postoperative rehydration suggests that the vasopressin response reflects not only transient hemodynamic changes but also the magnitude of surgical stress characteristic of MajLR. Although both AVP levels and the incidence of Clavien-Dindo grade ≥ IIIa complications were higher in the MajLR group, multiple confounding factors including surgical complexity, operative duration, and perioperative fluid shifts limit the ability to draw causal inferences. Nevertheless, these findings suggest that extensive liver resection is associated with a variety of physiological stimuli that can lead to elevated AVP levels. The surgical approach (laparoscopic vs open) did not affect the occurrence of postoperative hypervasopressinemia, suggesting that surgical invasiveness determined by the extent of resection is a more important factor.
The time course of these changes was consistent with decreases in urinary sodium, increases in drain output, and increases in body weight. These findings suggest that, in addition to the RAAS, the AVP axis contributes to early post
In contrast, AVP levels remained elevated until POD 3, even after their initial postoperative peak, and this pattern was more pronounced in MajLR cases. The sustained elevation of AVP may result not only from postoperative non-osmotic triggers (such as effective hypovolemia, inflammation, pain, nausea, and hemodynamic changes) but also from factors specific to liver resection: (1) Decreased metabolic clearance due to reduced liver parenchymal mass; (2) Relative defi
Spironolactone and canrenoic acid, administered to all patients as part of the standard postoperative protocols, competitively block aldosterone action by binding to its receptors in the distal renal tubules. Although these agents may exert a feedback effect on the RAAS and potentially influence postoperative PAC levels, they do not directly suppress aldosterone secretion. Therefore, an early increase in PAC cannot be fully attributed to the pharmacological effects of aldosterone antagonists. Notably, in the IL group, PAC remained elevated until POD 1, a pattern consistent with the well-described phenomenon of heightened RAAS reactivity in impaired livers. This suggests that the observed PAC response largely reflects physiological activation induced by surgical stress rather than solely medication-related effects. The divergence observed in this study specifically, that “PAC normalizes early, whereas AVP remains elevated until POD 3” may indicate that although sodium retention mediated by the RAAS is pharmacologically attenuated, free water reabsorption through the AVP axis relatively predominates. Nevertheless, because PAC declined even under anti-aldosterone therapy, these findings suggest that AVP plays a key role in postoperative water retention after liver resection. These hormonal changes were accompanied by clinical indicators of water retention, including postoperative weight gain, a decline in serum and urinary sodium around POD 2, and increased drain output after major resection. These findings parallel the sustained elevation of AVP, supporting the predominance of AVP-mediated free water retention in the early postoperative period. These mechanistic interpretations should be regarded as hypothesis-gene
Liver function analysis showed that an increase in PAC was evident immediately after liver resection until POD 2 in the IL group, whereas it was only present immediately after surgery in the NL group. Although the intergroup difference was not statistically significant, a trend was observed that supports previous findings of heightened RAA reactivity in damaged livers[9]. Analysis by surgical procedure revealed that the MajLR group exhibited greater drain output and significantly elevated AVP levels on POD 1, suggesting strong AVP activation in cases experiencing greater surgical stress. In our institution, intraoperative fluid management typically follows a low-central venous pressure strategy during hepatic transection to minimize bleeding; however, detailed hemodynamic targets were not strictly standardized and may have varied among cases. However, the higher proportion of patients with elevated AVP in the MajLR group supports the coexistence of excessive free water reabsorption and third-space fluid shifts. Clinically, these findings suggest that perioperative fluid management should not be guided solely by the RAAS; instead, the AVP axis should be considered from an early stage. Additionally, V2 receptor antagonists are now the standard therapy for refractory ascites in liver cirrhosis[26-28], and a postoperative weight gain exceeding 3.5 kg after liver resection is regarded as an inde
In Japan, diuretic therapy after liver resection has traditionally been guided by ICGR15, which remains a practical and widely accepted indicator when assessing hepatic functional reserve and determining postoperative fluid management strategies. Accordingly, patients in this study were stratified into NL and IL groups based on ICGR15, reflecting real-world clinical practice. Although ICGR15 was the primary indicator used for stratification, additional liver function parameters such as the FIB-4 index and the ALBI score were included to provide a more comprehensive characterization of baseline hepatic reserve. Notably, the IL group exhibited a significantly higher FIB-4 index and worse modified ALBI grade compared with the NL group, indicating that this ICGR15-based stratification was consistent with more advanced underlying liver injury. The concordance of these liver function markers supports the validity of the IL/NL grouping used in this study.
The strength of this study lies in the parallel measurement of PAC and AVP from the preoperative period to POD 5, according to a standardized protocol at a single institution, with stratified analyses based on liver function and surgical procedures. However, this study has some limitations: (1) It was a single-center, small-scale study with limited statistical power; (2) No corrections were applied for multiple comparisons in the exploratory analyses. This approach was considered acceptable given the hypothesis-generating nature of the hormonal time-course evaluation; (3) Measurement of AVP has inherent pre-analytical limitations. AVP is unstable because of its short half-life and susceptibility to degradation during sample handling, and thus absolute values obtained by RIA should be interpreted with caution. Nevertheless, all samples were processed under strictly uniform conditions using the same pre-analytical procedures and RIA platform, ensuring consistency across time points; (4) Several covariates including fluid balance, analgesic methods, opioid use, and osmolality were insufficiently controlled. In particular, perioperative opioid dosage was not uniformly documented, limiting the assessment of its potential effect on AVP secretion. Baseline tumor pathology (e.g., HCC vs CRLM) may also influence surgical stress and endocrine responses, although the study was not powered to adjust for these differences; (5) The use of RAAS-related medications and diuretics varied according to real-world clinical practice. Although these agents may have influenced the absolute values of PAC or AVP, this study focused on the temporal changes in hormonal dynamics rather than cross-sectional comparisons. Therefore, although their potential effects cannot be completely excluded, they are unlikely to have materially altered the interpretation of the characteristic postoperative patterns observed; (6) Clinical outcomes, including surgical site infection and respiratory complications, were not directly examined; and (7) Finally, correlations between AVP levels and postoperative complications could not be assessed because of the limited number of events. Although postoperative weight gain > 3.5 kg has been reported as a predictor of complications[6], the number of events in this cohort was insufficient for a meaningful analysis.
Nevertheless, no systematic reports have examined the postoperative course of AVP after liver resection, and we believe that this study provides important foundational data for developing future strategies regarding diuretic use after liver resection. Patients with persistent AVP elevation, particularly those undergoing MajLR, also demonstrated lower urine output and greater drain output, both of which clinically reflect increased third-space fluid accumulation. These findings suggest that V2 receptor antagonists may be beneficial in cases with early postoperative hypervasopressinemia. Optimal perioperative fluid management algorithms are expected to be established through prospective trials that investigate risk stratification based on surgical method and hormone levels, as well as the early introduction of V2 receptor antagonists in high-risk patients undergoing liver resection.
Both PAC and AVP levels increased after liver resection. However, PAC showed a transient fluctuation limited to the immediate postoperative period through POD 1, whereas AVP remained elevated until POD 3, with elevations particularly frequent after MajLR. These observational findings provide fundamental data on postoperative hormonal regulation and may serve as a basis for future investigations evaluating AVP-targeted (V2) therapeutic strategies.
We thank BML Inc., for the hormone assays. BML was not involved in the study design, analysis, interpretation, or decision to submit the manuscript.
| 1. | Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18949] [Reference Citation Analysis (0)] |
| 2. | He J, Amini N, Spolverato G, Hirose K, Makary M, Wolfgang CL, Weiss MJ, Pawlik TM. National trends with a laparoscopic liver resection: results from a population-based analysis. HPB (Oxford). 2015;17:919-926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 3. | Khan AH, Mahmud O, Fatimi AS, Ahmed S, Wiener AA, Nishtala MV, Stahl CC, Christensen L, Khan MR, Schwartz PB, Zafar SN. A Systematic Review and Meta-Analysis of Oncologic Liver Resections in Low- and Middle-Income Countries: Opportunities to Improve Evidence and Outcomes. J Surg Oncol. 2025;131:865-878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 4. | Marubashi S, Takahashi A, Kakeji Y, Hasegawa H, Ueno H, Eguchi S, Endo I, Goi T, Saiura A, Sasaki A, Takiguchi S, Takeuchi H, Tanaka C, Hashimoto M, Hiki N, Horiguchi A, Masaki T, Yoshida K, Gotoh M, Konno H, Yamamoto H, Miyata H, Seto Y, Kitagawa Y; National Clinical Database. Surgical outcomes in gastroenterological surgery in Japan: Report of the National Clinical Database 2011-2019. Ann Gastroenterol Surg. 2021;5:639-658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 106] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 5. | Orimo T, Hirakawa S, Taketomi A, Tachimori H, Oshikiri T, Miyata H, Kakeji Y, Shirabe K. Risk model for morbidity and mortality following liver surgery based on a national Japanese database. Ann Gastroenterol Surg. 2024;8:896-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 6. | Labgaa I, Joliat GR, Grass F, Jarrar G, Halkic N, Demartines N, Hübner M. Impact of postoperative weight gain on complications after liver surgery. HPB (Oxford). 2020;22:744-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Tagami N, Yoshimoto N, Kawasaki O, Yukioka H, Nishimura K, Fujimori M, Hirohashi K, Kinoshita H. Clinical evaluation of an aldosterone antagonist (Soldactone®) as a diuretic for patients with cirrhosis of the liver after hepatectomy for hepatoma. ICU and CCU. 1985;9:53-58. |
| 8. | Arroyo V, Bosch J, Mauri M, Viver J, Mas A, Rivera F, Rodes J. Renin, aldosterone and renal haemodynamics in cirrhosis with ascites. Eur J Clin Invest. 1979;9:69-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 76] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 9. | Kobayashi S, Hirasawa H, Kobayashi H, Tabata Y, Ohkawa M, Soeda K, Oda S, Murotani N, Odaka M, Satoh H. The Postoperative Change in Blood Aldosterone Level and Its Effect on Sodium Retention in Cirrhotic Patients. Nihon Shokaki Geka Gakkai Zasshi. 1984;17:1421-1425. [DOI] [Full Text] |
| 10. | Yoshiji H, Nagoshi S, Akahane T, Asaoka Y, Ueno Y, Ogawa K, Kawaguchi T, Kurosaki M, Sakaida I, Shimizu M, Taniai M, Terai S, Nishikawa H, Hiasa Y, Hidaka H, Miwa H, Chayama K, Enomoto N, Shimosegawa T, Takehara T, Koike K. Evidence-based clinical practice guidelines for Liver Cirrhosis 2020. J Gastroenterol. 2021;56:593-619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 331] [Cited by in RCA: 274] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 11. | Biggins SW, Angeli P, Garcia-Tsao G, Ginès P, Ling SC, Nadim MK, Wong F, Kim WR. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 690] [Cited by in RCA: 586] [Article Influence: 117.2] [Reference Citation Analysis (0)] |
| 12. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2259] [Cited by in RCA: 2061] [Article Influence: 257.6] [Reference Citation Analysis (6)] |
| 13. | Bichet D, Szatalowicz V, Chaimovitz C, Schrier RW. Role of vasopressin in abnormal water excretion in cirrhotic patients. Ann Intern Med. 1982;96:413-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 207] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 14. | Burmeister P, Schölmerich J, Diener W, Gerok W. Renin, aldosterone and arginine vasopressin in patients with liver cirrhosis: the influence of ascites retransfusion. Eur J Clin Invest. 1986;16:117-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Carreño FR, Ji LL, Cunningham JT. Altered central TRPV4 expression and lipid raft association related to inappropriate vasopressin secretion in cirrhotic rats. Am J Physiol Regul Integr Comp Physiol. 2009;296:R454-R466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 16. | Bormann B, Weidler B, Dennhardt R, Sturm G, Scheld HH, Hempelmann G. Influence of epidural fentanyl on stress-induced elevation of plasma vasopressin (ADH) after surgery. Anesth Analg. 1983;62:727-732. [PubMed] |
| 17. | Solis Herruzo JA, Castellano G, Larrodera L, Morillas JD, Moreno Sanchez D, Provencio R, Muñoz-Yagüe MT. Plasma arginine vasopressin concentration during laparoscopy. Hepatogastroenterology. 1989;36:499-503. [PubMed] |
| 18. | Otsuka T, Matsuyama K. Nippon Medical School's Ethical Review Processes for Studies Involving Human Subjects. J Nippon Med Sch. 2024;91:136-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 19. | Strasberg S, Belghiti J, Clavien P, Gadzijev E, Garden J, Lau W, Makuuchi M, Strong R. The Brisbane 2000 Terminology of Liver Anatomy and Resections. HPB. 2000;2:333-339. [DOI] [Full Text] |
| 20. | Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J, S Sulkowski M, Torriani FJ, Dieterich DT, Thomas DL, Messinger D, Nelson M; APRICOT Clinical Investigators. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4179] [Cited by in RCA: 3906] [Article Influence: 195.3] [Reference Citation Analysis (7)] |
| 21. | Hiraoka A, Michitaka K, Kumada T, Izumi N, Kadoya M, Kokudo N, Kubo S, Matsuyama Y, Nakashima O, Sakamoto M, Takayama T, Kokudo T, Kashiwabara K, Kudo M. Validation and Potential of Albumin-Bilirubin Grade and Prognostication in a Nationwide Survey of 46,681 Hepatocellular Carcinoma Patients in Japan: The Need for a More Detailed Evaluation of Hepatic Function. Liver Cancer. 2017;6:325-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 284] [Article Influence: 31.6] [Reference Citation Analysis (5)] |
| 22. | Johnson PJ, Berhane S, Kagebayashi C, Satomura S, Teng M, Reeves HL, O'Beirne J, Fox R, Skowronska A, Palmer D, Yeo W, Mo F, Lai P, Iñarrairaegui M, Chan SL, Sangro B, Miksad R, Tada T, Kumada T, Toyoda H. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol. 2015;33:550-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2296] [Cited by in RCA: 2230] [Article Influence: 202.7] [Reference Citation Analysis (3)] |
| 23. | Melendez JA, Arslan V, Fischer ME, Wuest D, Jarnagin WR, Fong Y, Blumgart LH. Perioperative outcomes of major hepatic resections under low central venous pressure anesthesia: blood loss, blood transfusion, and the risk of postoperative renal dysfunction. J Am Coll Surg. 1998;187:620-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 348] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 24. | Zatloukal J, Pradl R, Kletecka J, Skalicky T, Liska V, Benes J. Comparison of absolute fluid restriction versus relative volume redistribution strategy in low central venous pressure anesthesia in liver resection surgery: a randomized controlled trial. Minerva Anestesiol. 2017;83:1051-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Ye H, Wu H, Li B, Zuo P, Chen C. Application of cardiovascular interventions to decrease blood loss during hepatectomy: a systematic review and meta-analysis. BMC Anesthesiol. 2023;23:89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Namba M, Hiramatsu A, Aikata H, Kodama K, Uchikawa S, Ohya K, Morio K, Fujino H, Nakahara T, Murakami E, Yamauchi M, Kawaoka T, Tsuge M, Imamura M, Chayama K. Management of refractory ascites attenuates muscle mass reduction and improves survival in patients with decompensated cirrhosis. J Gastroenterol. 2020;55:217-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 27. | Tahara T, Mori K, Mochizuki M, Ishiyama R, Noda M, Hoshi H, Lefor AK, Shinozaki S. Tolvaptan is effective in treating patients with refractory ascites due to cirrhosis. Biomed Rep. 2017;7:558-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 28. | Yatsuhashi H, Sano H, Hirano T, Shibasaki Y. Real-world hospital mortality of liver cirrhosis inpatients in Japan: a large-scale cohort study using a medical claims database: Prognosis of liver cirrhosis. Hepatol Res. 2021;51:682-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 29. | Kobayashi Y, Shindoh J, Kojima K, Akabane M, Kobayashi M, Okubo S, Matsumura M, Hashimoto M. Efficacy and safety of postoperative preemptive use of tolvaptan for patients with cirrhosis undergoing hepatectomy for hepatocellular carcinoma. Langenbecks Arch Surg. 2023;408:381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 30. | Iida H, Maehira H, Mori H, Nitta N, Maekawa T, Takebayashi K, Kaida S, Miyake T, Tani M. Effect of early administration of tolvaptan on pleural effusion post-hepatectomy. Langenbecks Arch Surg. 2023;408:406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 31. | Namba Y, Kobayashi T, Kuroda S, Hashimoto M, Takei D, Fukuhara S, Oshita K, Matsubara K, Honmyo N, Nakano R, Sakai H, Tahara H, Ohira M, Ide K, Ohdan H. Protocol to evaluate the efficacy and safety of tolvaptan in patients with refractory ascites after liver resection: an open-label, single-arm phase I/II study. Int J Surg Protoc. 2024;28:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/