Published online Mar 14, 2019. doi: 10.3748/wjg.v25.i10.1248
Peer-review started: January 2, 2019
First decision: January 18, 2019
Revised: February 14, 2019
Accepted: February 15, 2019
Article in press: February 16, 2019
Published online: March 14, 2019
Processing time: 74 Days and 12.2 Hours
Linked color imaging (LCI) is a method of endoscopic imaging that emphasizes slight differences in red mucosal color.
To evaluate LCI in diagnostic endoscopy of early gastric cancer and to compare LCI and pathological findings.
Endoscopic images were obtained for 39 patients (43 lesions) with early gastric cancer. Three endoscopists evaluated lesion recognition with white light imaging (WLI) and LCI. Color values in Commission Internationale de l'Eclairage (CIE) 1976 L*a*b* color space were used to calculate the color difference (ΔE) between cancer lesions and non-cancer areas. After endoscopic submucosal dissection, blood vessel density in the surface layer of the gastric epithelium was evaluated pathologically. The identical region of interest was selected for analyses of endoscopic images (WLI and LCI) and pathological analyses.
LCI was superior for lesion recognition (P < 0.0001), and ΔE between cancer and non-cancer areas was significantly greater with LCI than WLI (29.4 vs 18.6, P < 0.0001). Blood vessel density was significantly higher in cancer lesions (5.96% vs 4.15%, P = 0.0004). An a* cut-off of ≥ 24 in CIE 1976 L*a*b* color space identified a cancer lesion using LCI with sensitivity of 76.7%, specificity of 93.0%, and accuracy of 84.9%.
LCI is more effective for recognition of early gastric cancer compared to WLI as a result of improved visualization of changes in redness. Surface blood vessel density was significantly higher in cancer lesions, and this result is consistent with LCI image analysis.
Core tip: This study showed the utility of linked color imaging (LCI) for screening endoscopy of early gastric cancer and the concordant change in blood vessel density with redness in LCI image. The key results of the study were as follows: LCI gave better contrast than white light imaging for the color difference between cancer lesions and surrounding non-cancer tissue; an a* cutoff ≥ 24 for the value in Commission Internationale de l'Eclairage 1976 L*a*b* color space had good sensitivity and specificity for diagnosis of early gastric cancer; and surface blood vessel density in cancer lesions was significantly higher than that in non-cancer areas.
- Citation: Fujiyoshi T, Miyahara R, Funasaka K, Furukawa K, Sawada T, Maeda K, Yamamura T, Ishikawa T, Ohno E, Nakamura M, Kawashima H, Nakaguro M, Nakatochi M, Hirooka Y. Utility of linked color imaging for endoscopic diagnosis of early gastric cancer. World J Gastroenterol 2019; 25(10): 1248-1258
- URL: https://www.wjgnet.com/1007-9327/full/v25/i10/1248.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i10.1248
Core tip: This study showed the utility of linked color imaging (LCI) for screening endoscopy of early gastric cancer and the concordant change in blood vessel density with redness in LCI image. The key results of the study were as follows: LCI gave better contrast than white light imaging for the color difference between cancer lesions and surrounding non-cancer tissue; an a* cutoff ≥ 24 for the value in Commission Internationale de l'Eclairage 1976 L*a*b* color space had good sensitivity and specificity for diagnosis of early gastric cancer; and surface blood vessel density in cancer lesions was significantly higher than that in non-cancer areas.
The mortality of gastric cancer has been decreasing worldwide, but there are still more than 800000 deaths due to this disease. Gastric cancer is the third leading cause of death, followed by lung and colorectal cancer, in overall mortality worldwide[1]. In Japan, the morbidity of gastric cancer ranks first in males and third after breast and colorectal cancer in females[2]. Since advanced gastric cancer has a poorer prognosis, early detection and treatment is required to overcome the disease.
Endoscopy is an essential modality for detection of early gastric cancer. Recent advances and innovations in endoscopic devices have provided scopes with a high pixel count, a smaller diameter, and various optical technology. In particular, image-enhanced endoscopy (IEE) that uses imaging and analysis of wavelengths of visible light with illumination has a key role in detection of early gastrointestinal cancer. Among IEE methods, narrow-band imaging (NBI) and blue light imaging (BLI) are commonly used. NBI is effective for observation of capillaries on mucosal surfaces through emission of narrow band light[3-5] and can be used for qualitative diagnosis (differentiation of cancer from non-cancer tissues, and the extent of cancer)[6-8] in the oropharynx and hypopharynx[9], esophagus[10,11] and stomach. BLI allows observation of surface capillaries[12], in magnified observation, there is little difference in the diagnostic performances of BLI and NBI[13].
A new IEE method, referred to as linked color imaging (LCI), with an endoscopic light source was recently introduced by Fujifilm Corp. (Tokyo, Japan). LCI observation is interchangeable with white light imaging (WLI)/BLI with just one push of a button. LCI images are created by emission of white light and narrow-band short-wave light, and processing images to make a well-separated red area. The most important wavelengths for diagnosis of normal mucosa and atrophic mucosa are concentrated around the red color. LCI emphasizes slight differences in the “red” color of the mucosa by image processing, including enhancement of differences in chroma and hue of the red mucosal color. As a result, red mucosa is visualized as redder and white mucosa as whiter. LCI has a color shade that is closer to WLI than NBI and BLI and processes images to separate the mucosal color more effectively; therefore, LCI is a suitable imaging method to identify early gastric cancer.
The utility of LCI for diagnosis of Helicobacter pylori gastritis has been well described[14], but there are few studies of LCI for diagnosis of gastrointestinal cancer and, to our knowledge, parallel analyses of LCI, WLI, and pathology samples have not been reported. Therefore, the objectives of this study are to evaluate the utility of LCI for endoscopic diagnosis of early gastric cancer and to examine if pathological findings can explain the changes in red shade in LCI.
The subjects were 39 patients (43 lesions) with early gastric cancer who underwent endoscopic submucosal dissection (ESD) of the stomach. WLI and LCI at the same time point were used for comparison of endoscopic images. All procedures were performed in the Department of Gastroenterology and Hepatology, Nagoya University Graduate School of Medicine from September 2016 to January 2018. This study was conducted after approval by the ethics committee of Nagoya University, and was performed in accord with the Helsinki Declaration. All subjects gave informed consent to participation after receiving a written explanation of the study.
Equipment and setting: An ELUXEO™ system (video processor VP-7000, light source BL-7000) and a Video Endoscope EG-760Z (Fujifilm) were used in the study. In WLI, the structure emphasis was set at H+1+3 (SE = Hi, DH = +1, DL = +3). In LCI, the structure emphasis was set at A6 and the color emphasis was set at C1. Endoscopic images were stored as uncompressed TIFF files. Images were 1280 × 1024 pixels and RGB 24-bit color.
Image assessment by endoscopists: WLI and LCI images were taken before endoscopic therapy and not magnified in the study. Lesions were imaged with WLI and LCI at the same timepoint, and the images were placed in a file in random order. These images were then evaluated by three endoscopists who routinely perform endoscopic therapy and are board-certified fellows of the Japan Gastroenterological Endoscopy Society. The evaluators were blinded to clinical data and pathological results. Each evaluator viewed one WLI and one LCI image simultaneously and assessed which image was superior for lesion recognition using a 5-point Likert scale: +2, very much; +1, somewhat; 0, undecided; -1, not really; -2, not at all. The same display was used for assessment. The evaluators assessed images alone without discussion with other evaluators.
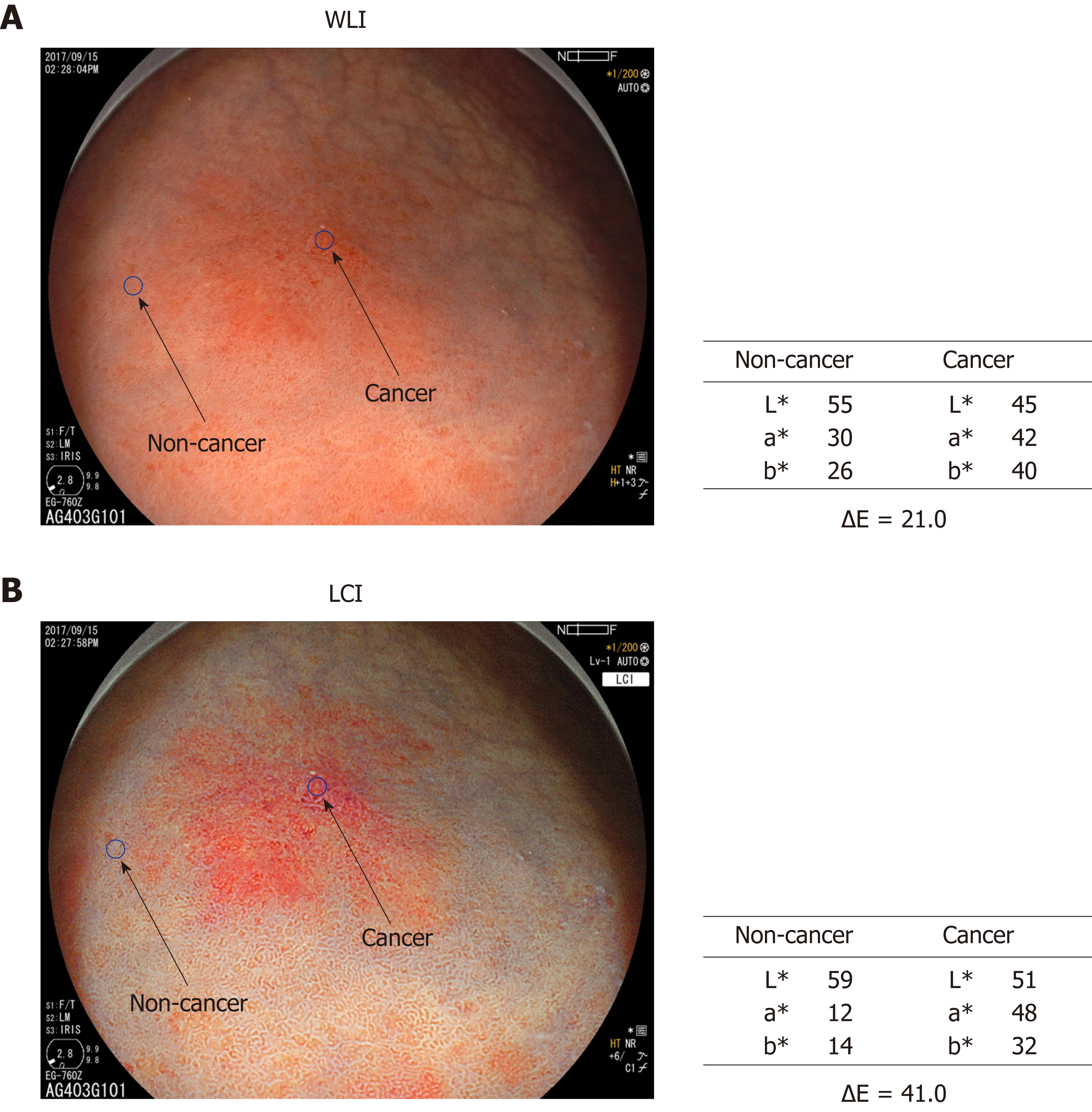
Color assessment of endoscopic images: Two regions of interest (ROI, diameter 24 pixels) were picked from each lesion. One was from the cancer lesion and the other from surrounding non-cancer mucosa. Non-cancer mucosa was normal mucosa without ulceration or other specific changes. After ESD, the identical ROI was marked in the ESD resected sample and evaluated pathologically. ROI color in the endoscopic image was assessed using Commission Internationale de l'Eclairage (CIE) 1976 L*a*b* color space. This is a three-dimensional space for presenting a color with axes of L* (from black to white; white is highest), a* (from green to red; red is highest), and b* (from blue to yellow; yellow is highest). The difference between two colors (color difference) was approximated by the distance in the CIE 1976 L*a*b*color space[15]. L*a*b* values of the ROI in cancer and non-cancer tissues were measured using Adobe Photoshop CS4 (Adobe Systems Inc., San Jose, CA, United States) to estimate the color difference (Figure 1).
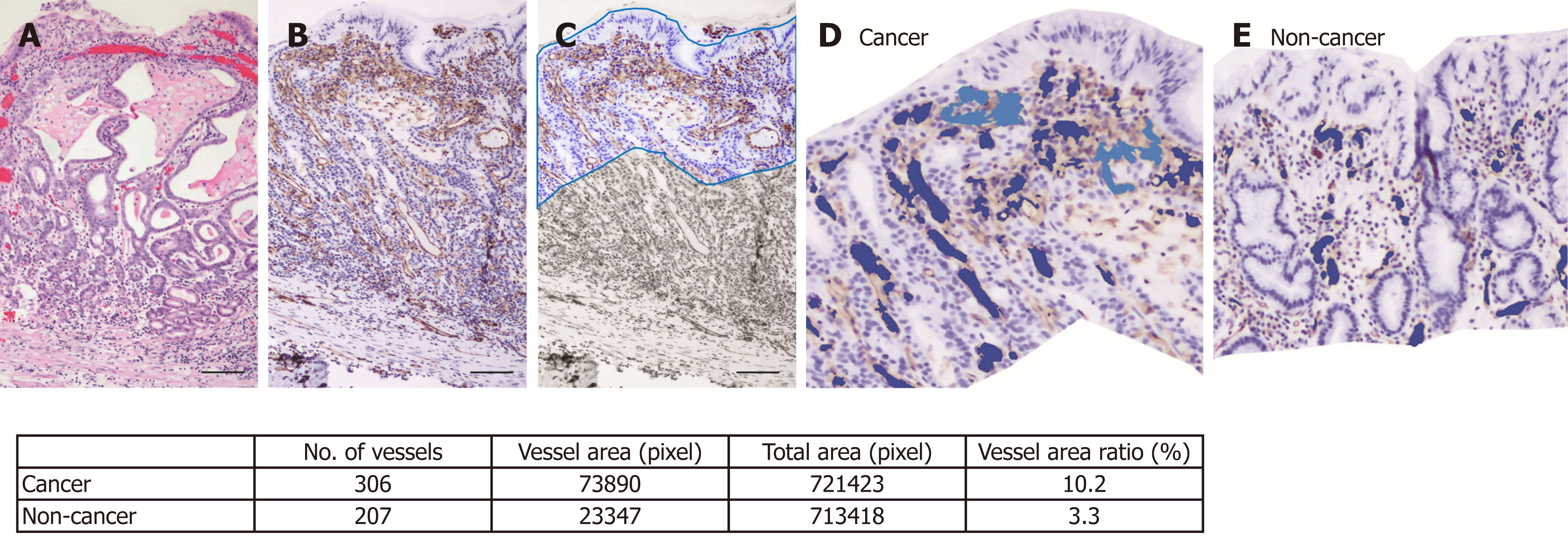
Histopathological assessment: Resected gastric tissue was fixed in 10% neutral buffered formalin and embedded in paraffin. Routine hematoxylin and eosin staining (Figure 2A) and immunohistochemical staining were performed using 3-μm sections. CD31 immunohistochemistry (Figure 2B) was used to highlight the blood vessel wall, using a commercial primary antibody (clone JC/70A, 1:1200; Dako, Glostrup, Denmark). The blood vessel area and its proportion of the total area were analyzed based on photomicrographs for each ROI at ×100 magnification. An area of 350 μm depth from the surface of the mucosa was trimmed on the photomicrograph (Figure 2C) and the blood vessel area was defined as the sum of the CD31-positive area and the area encircled by CD31-positive endothelial cells (Figure 2D and E). Recognition and calculation of the positive area was performed automatically using image analysis software (WinRoof v.3.9.0, Mitani Co., Tokyo, Japan).
Statistical analysis: Statistical analysis were performed using SPSS v.22.0 for Windows (IBM Japan, Tokyo, Japan) and SAS 9.4 (SAS Institute, Inc.). Variables are expressed as medians (range). Differences in paired continuous variables were compared by Wilcoxon signed-rank test, and differences in independent continuous variables were compared by Wilcoxon rank sum test. ROC curves were created with values in CIE 1976 L*a*b* color space to differentiate between cancer and non-cancer lesions. Diagnostic performance was estimated based on the area under the curve (AUC), cut-off values, sensitivity and specificity. The significance level was set at a two-tailed P value < 0.05.
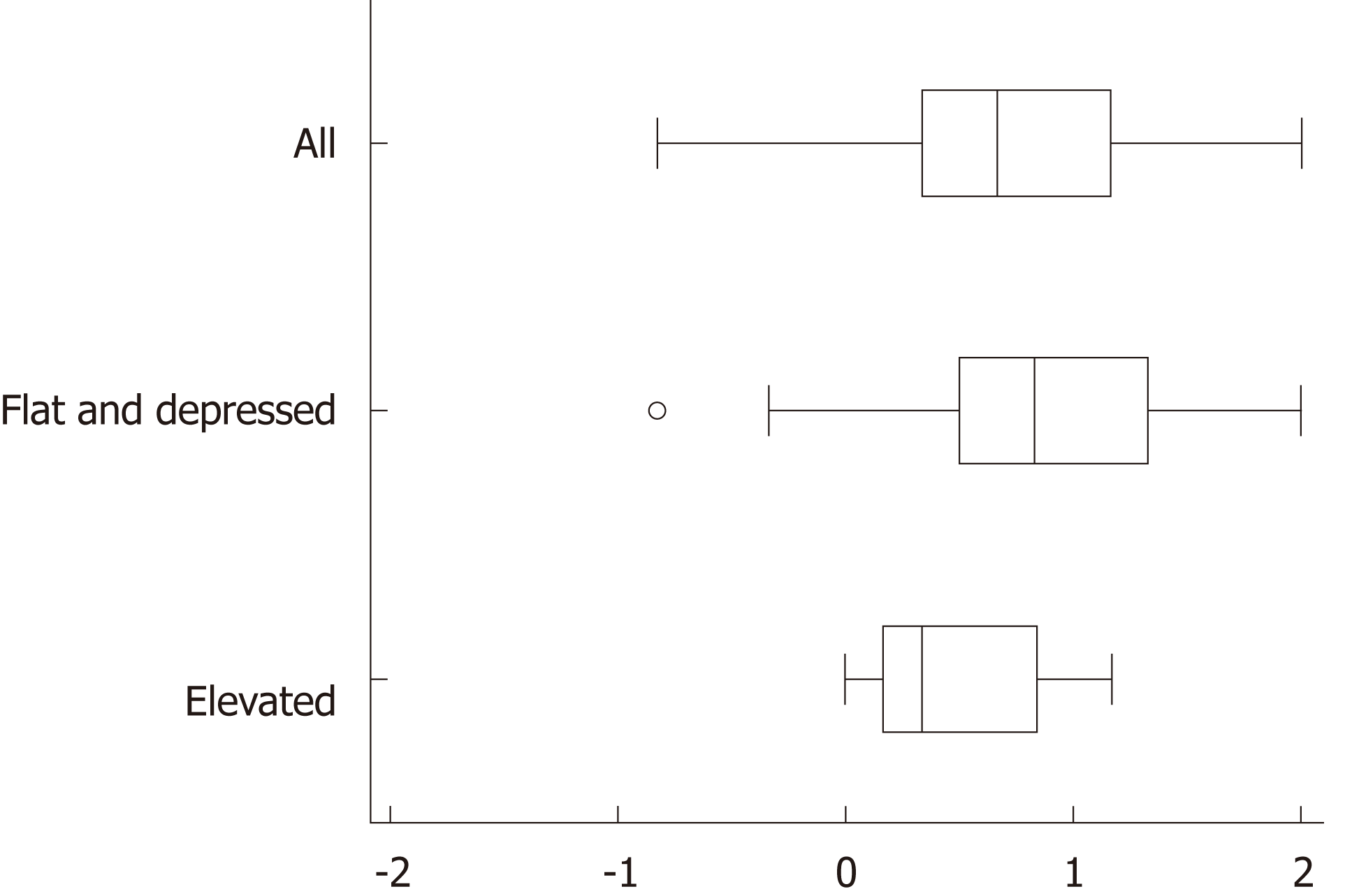
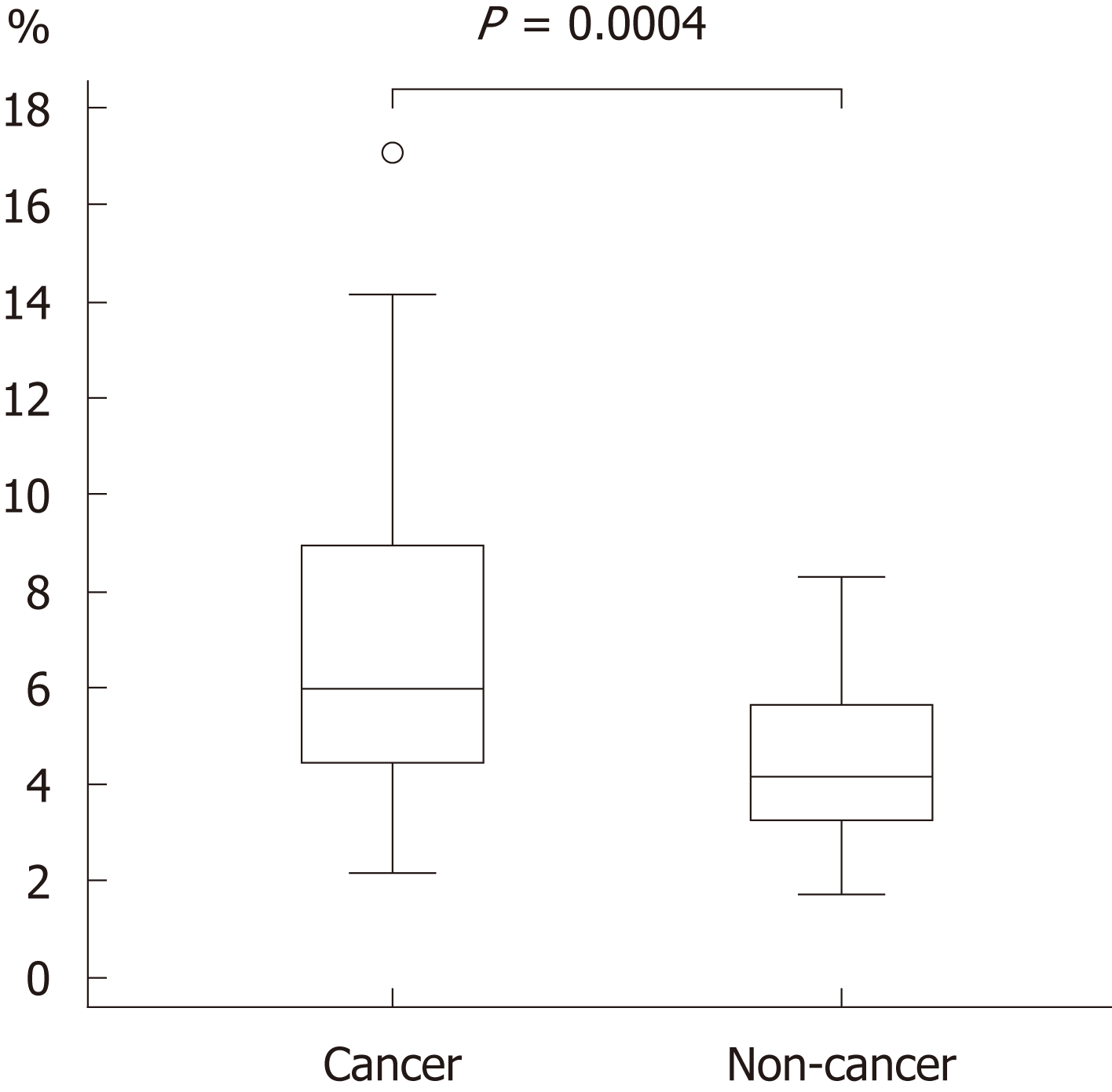
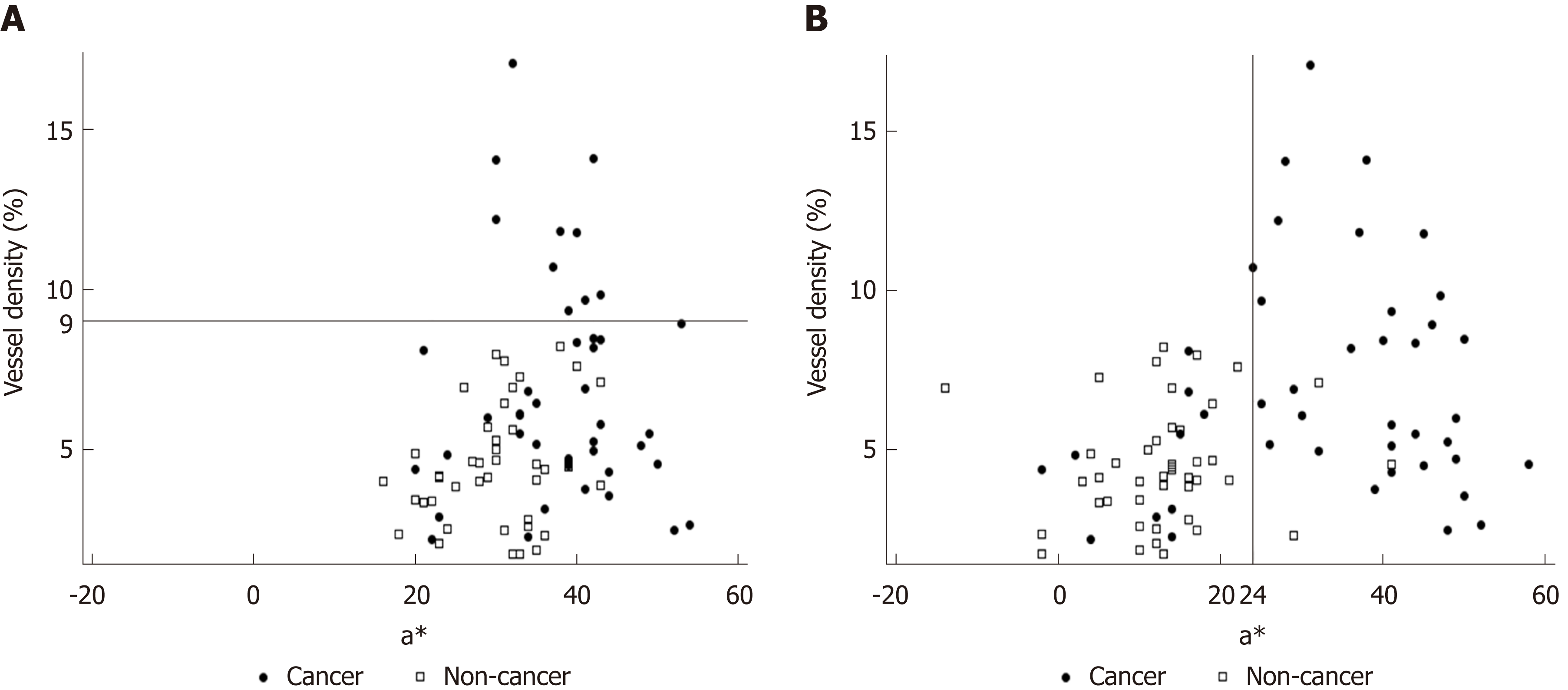
The background and clinicopathological characteristics of the patients and lesions are shown in Table 1. Most lesions were differentiated tissue type (well or mod) and intramucosal carcinoma. All cases were completely resected with a free margin. Evaluation by three endoscopists indicated that LCI was superior to WLI for recognition of lesions of early gastric cancer (P < 0.0001). This finding being particularly strong for flat/depressed lesions compared to elevated lesions (Figure 3). The color difference (ΔE) between cancer and non-cancer areas was significantly higher in LCI than in WLI [29.4 (9.4-63.9) vs 18.6 (5.1-38.4), P < 0.0001] (Table 2) and thus LCI could differentiate a cancer lesion from a non-cancer area better than WLI. In LCI, using a cut-off from ROC curves of ≥ 24 for detection of a cancer lesion, the diagnostic performance had sensitivity of 76.7%, specificity of 93.0%, positive predictive value (PPV) of 91.7%, negative predictive value (NPV) of 80.0%, and accuracy of 84.9%. In pathological assessment, the blood vessel density in the surface layer was significantly higher in cancer lesions than in non-cancer areas [5.96% (2.17-17.08) vs 4.15% (1.71-8.22), P = 0.0004] (Figure 4). Surface blood vessel density exceeding 9% was only found in cancer lesions. The relationship between the value in CIE 1976 L*a*b* color space and blood vessel density in WLI and LCI is shown in Figure 5.
| Item | Number | Range or Percentage | ||
| Cases/lesions | 39/43 | |||
| Age (yr): median (range) | 74 | (57-88) | ||
| Sex: male/female | 27/12 | |||
| Tumor major axis (mm): median (range) | 15 | (6-40) | ||
| Location (%) | U (Fundus) | 5 | (11.6) | |
| M (Corpus) | 7 | (16.3) | ||
| L (Antrum and Pylorus) | 30 | (69.8) | ||
| Remnant Stomach | 1 | (2.3) | ||
| Macroscopic type (%) | Elevated | Type 0-I | 2 | (4.6) |
| Type 0-IIa | 9 | (20.9) | ||
| Type 0-IIa+IIc | 2 | (4.7) | ||
| Flat | Type 0-IIb | 7 | (16.3) | |
| Depressed | Type 0-IIc | 23 | (53.5) | |
| Color of tumor in WLI | Red | 20 | (46.5) | |
| White | 2 | (4.7) | ||
| Isochromatic | 21 | (48.8) | ||
| Infection with Helicobacter pylori | Positive | 14 | (32.6) | |
| Depth of invasion (%) | M | 36 | (83.7) | |
| SM1 | 4 | (9.3) | ||
| SM2 | 3 | (7.0) | ||
| Histological type (%) | Differentiated (well or mod) | 40 | (93.0) | |
| Undifferentiated (poor or sig) | 3 | (7.0) |
| WLI | LCI | P value | |
| Color differences ΔE1 [median (min-max)] | 18.6 (5.1-38.4) | 29.4 (9.4-63.9) | < 0.0001 |
| Elevated | 15.0 (5.8-34.4) | 28.5 (11.0-40.2) | 0.0327 |
| Flat and depressed | 20.6 (5.1-38.4) | 30.9 (9.4-63.9) | < 0.0001 |
The results of this study show the utility of LCI for screening endoscopy of early gastric cancer and the concordant change in blood vessel density with redness in the LCI image. To our knowledge, this is the first study to compare objective color evaluation of early gastric cancer using the CIE 1976 L*a*b* color space in LCI with a pathological analysis of blood vessel density. This study showed that the large color difference in LCI may be due to the surface blood vessel density. The key results of the study were as follows: LCI recognized early gastric cancer lesions more effectively than WLI; LCI gave better contrast than WLI for the color difference between cancer lesions and the surrounding non-cancer area; an a* cutoff ≥ 24 for the value in CIE 1976 L*a*b* color space had good sensitivity and specificity for diagnosis of early gastric cancer; and surface blood vessel density in cancer lesions was significantly higher than that in non-cancer areas.
Based on an assessment by three endoscopists, LCI recognized early gastric cancer lesions more easily than WLI. A previous study also indicated that LCI gave images that were easier to review than WLI for both experts and non-experts in > 70% of patients with early gastric cancer[16]. Endoscopists attempt to find a lesion using the ruggedness of the mucosa and changes in color shade when observing inside the stomach, but this requires appropriate technology to show slight differences in color shade. Most lesions with slight differences in color shade can be detected by WLI using innovations such as adjusted air volume and progression in lesions. However, it is difficult to recognize a flat lesion without mucosal ruggedness or a slightly recessed lesion. To recognize such lesions, a change in color shade between a cancer lesion and a non-cancer area is important.
Subjective descriptions of color are commonly used in conventional endoscopic findings, and consequently, it is difficult to make an objective evaluation. In this study, CIE 1976 L*a*b* color space was used to assess the color objectively. The results showed that LCI gave better contrast than WLI for the color difference between cancer and non-cancer areas. These results are consistent with the assessment that LCI is a better imaging method than WLI for recognition of early gastric cancer, especially for flat and recessed lesions, which are likely to be assessed based on color difference, rather than on mucosal ruggedness.
The relationship of cancer and the microvascular architecture has been widely examined[17-20], with reports of an irregular pattern and heterogeneity of the vasculature, and microvascular dilatation. In our study, we used non-magnified endoscopy to analyze the gross vascular effect, rather than the single vessel architecture. Since irregular running and vessel dilation can increase the blood vessel density, our results are closely related to previous studies using magnified endoscopy. The endoscopic color of early gastric cancer is widely distributed from strong reddening to fading. Some previous studies have mentioned the role of the number of capillaries in defining the color of the mucosa[21-23]. An increase in number, as might be assumed, can increase the blood vessel density. Furukawa et al[24] also mentioned the significance of the blood vessel density. Given the depth that endoscopic light can reach, we examined blood vessel density from the surface to a depth of 350 μm[25]. For as objective an evaluation as possible, the blood vessel density was assessed using pathological imaging analysis software (WinRoof v.3.9.0). This analysis showed that the blood vessel density was significantly higher in cancer lesions than in non-cancer areas, and exceeded 9% in all cancer lesions in the study. These results are consistent with findings showing that the blood vessel density in cancer lesions is higher than that in non-cancer areas in differentiated gastric cancer[24].
Changes in redness between cancer and non-cancer areas in pathological images were assessed objectively by estimating a value in CIE 1976 L*a*b* color space. In LCI, ROC curves drawn using this value estimated an AUC of 0.86 at an a* cut-off value of 24. Values of ≥ 24 and < 24 indicated cancer and non-cancer areas, respectively, with a diagnostic sensitivity of 76.7%, specificity of 93.0%, PPV of 91.7%, NPV of 80.0%, and accuracy of 84.9%. We believe that our finding can lead to improved diagnosis of cancer lesions, based on objective evaluation using CIE 1976 L*a*b* color space. If these color values are evaluable in real time, artificial intelligence may permit automatic recognition and diagnosis of lesions.
This study has several limitations. First, it was a single-center and small-scale study. Second, the subjects were patients with early gastric cancer who underwent ESD, and consequently, most had differentiated intramucosal carcinoma. To include patients with undifferentiated cancer and early gastric cancer invading the submucosa, patients who underwent surgical resection would need to be added. Third, in this study, we focused only on cancer, and we did not evaluate inflammation or adenoma. Fourth, because recognition of WLI or LCI is obvious for expert endoscopist, bias is inevitable in the evaluation. To overcome this bias, we used objective color evaluation based on CIE 1976 L*a*b* color space.
In conclusion, LCI was useful for recognition of early gastric cancer lesions because this method provides good contrast in color differences between lesions and surrounding tissue. Blood vessel density from the surface to a depth of 350 μm was higher in cancer lesions than in non-cancer areas, and LCI clearly shows this feature as a change in redness.
Linked color imaging (LCI) emphasizes slight differences in the “red” color of the mucosa by image processing, including enhancement of differences in chroma and hue of the red mucosal color.
The utility of LCI for diagnosis of Helicobacter pylori gastritis has been well described, but there are few studies of LCI for diagnosis of gastrointestinal cancer and, to our knowledge, parallel analyses of LCI, white light imaging (WLI), and pathology samples have not been reported.
The objectives of this study are to evaluate the utility of LCI for endoscopic diagnosis of early gastric cancer and to examine if pathological findings can explain the changes in red shade in LCI.
Three endoscopists evaluated lesion recognition with WLI and LCI. Color values in Commission Internationale de l'Eclairage (CIE) 1976 L*a*b* color space were used to calculate the color difference (ΔE) between cancer lesions and non-cancer areas. After endoscopic submucosal dissection, blood vessel density in the surface layer of the gastric epithelium was evaluated pathologically. The identical region of interest was selected for analyses of endoscopic images (WLI and LCI) and pathological analyses.
LCI gave better contrast than WLI for the color difference between cancer lesions and surrounding non-cancer tissue; an a* cutoff ≥ 24 for the value in CIE 1976 L*a*b* color space had good sensitivity and specificity for diagnosis of early gastric cancer; and surface blood vessel density in cancer lesions was significantly higher than that in non-cancer areas.
LCI was useful for recognition of early gastric cancer lesions because this method provides good contrast in color differences between lesions and surrounding tissue. Blood vessel density from the surface to a depth of 350 μm was higher in cancer lesions than in non-cancer areas, and LCI clearly shows this feature as a change in redness.
If these color values are evaluable in real time, artificial intelligence may permit automatic recognition and diagnosis of lesions.
| 1. | Global Burden of Disease Cancer Collaboration; Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H, Dicker DJ, Chimed-Orchir O, Dandona R, Dandona L, Fleming T, Forouzanfar MH, Hancock J, Hay RJ, Hunter-Merrill R, Huynh C, Hosgood HD, Johnson CO, Jonas JB, Khubchandani J, Kumar GA, Kutz M, Lan Q, Larson HJ, Liang X, Lim SS, Lopez AD, MacIntyre MF, Marczak L, Marquez N, Mokdad AH, Pinho C, Pourmalek F, Salomon JA, Sanabria JR, Sandar L, Sartorius B, Schwartz SM, Shackelford KA, Shibuya K, Stanaway J, Steiner C, Sun J, Takahashi K, Vollset SE, Vos T, Wagner JA, Wang H, Westerman R, Zeeb H, Zoeckler L, Abd-Allah F, Ahmed MB, Alabed S, Alam NK, Aldhahri SF, Alem G, Alemayohu MA, Ali R, Al-Raddadi R, Amare A, Amoako Y, Artaman A, Asayesh H, Atnafu N, Awasthi A, Saleem HB, Barac A, Bedi N, Bensenor I, Berhane A, Bernabé E, Betsu B, Binagwaho A, Boneya D, Campos-Nonato I, Castañeda-Orjuela C, Catalá-López F, Chiang P, Chibueze C, Chitheer A, Choi JY, Cowie B, Damtew S, das Neves J, Dey S, Dharmaratne S, Dhillon P, Ding E, Driscoll T, Ekwueme D, Endries AY, Farvid M, Farzadfar F, Fernandes J, Fischer F, G/Hiwot TT, Gebru A, Gopalani S, Hailu A, Horino M, Horita N, Husseini A, Huybrechts I, Inoue M, Islami F, Jakovljevic M, James S, Javanbakht M, Jee SH, Kasaeian A, Kedir MS, Khader YS, Khang YH, Kim D, Leigh J, Linn S, Lunevicius R, El Razek HMA, Malekzadeh R, Malta DC, Marcenes W, Markos D, Melaku YA, Meles KG, Mendoza W, Mengiste DT, Meretoja TJ, Miller TR, Mohammad KA, Mohammadi A, Mohammed S, Moradi-Lakeh M, Nagel G, Nand D, Le Nguyen Q, Nolte S, Ogbo FA, Oladimeji KE, Oren E, Pa M, Park EK, Pereira DM, Plass D, Qorbani M, Radfar A, Rafay A, Rahman M, Rana SM, Søreide K, Satpathy M, Sawhney M, Sepanlou SG, Shaikh MA, She J, Shiue I, Shore HR, Shrime MG, So S, Soneji S, Stathopoulou V, Stroumpoulis K, Sufiyan MB, Sykes BL, Tabarés-Seisdedos R, Tadese F, Tedla BA, Tessema GA, Thakur JS, Tran BX, Ukwaja KN, Uzochukwu BSC, Vlassov VV, Weiderpass E, Wubshet Terefe M, Yebyo HG, Yimam HH, Yonemoto N, Younis MZ, Yu C, Zaidi Z, Zaki MES, Zenebe ZM, Murray CJL, Naghavi M. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017;3:524-548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3015] [Cited by in RCA: 3010] [Article Influence: 334.4] [Reference Citation Analysis (1)] |
| 2. | Hori M, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H; Japan Cancer Surveillance Research Group. Cancer incidence and incidence rates in Japan in 2009: a study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 2015;45:884-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 508] [Cited by in RCA: 468] [Article Influence: 42.5] [Reference Citation Analysis (1)] |
| 3. | Yao K, Yao T, Iwashita A. Determining the horizontal extent of early gastric carcinoma: two modern techniques based of differences in the mucosal microvascular architecture and density between carcinomatous and non-carcinomatous mucosa. Dig Endosc. 2002;14:83-87. [DOI] [Full Text] |
| 4. | Yao K, Oishi T, Matsui T, Yao T, Iwashita A. Novel magnified endoscopic findings of microvascular architecture in intramucosal gastric cancer. Gastrointest Endosc. 2002;56:279-284. [PubMed] |
| 5. | Gono K, Obi T, Yamaguchi M, Ohyama N, Machida H, Sano Y, Yoshida S, Hamamoto Y, Endo T. Appearance of enhanced tissue features in narrow-band endoscopic imaging. J Biomed Opt. 2004;9:568-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 727] [Cited by in RCA: 608] [Article Influence: 27.6] [Reference Citation Analysis (3)] |
| 6. | Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy. 2009;41:462-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 341] [Article Influence: 20.1] [Reference Citation Analysis (1)] |
| 7. | Nagahama T, Yao K, Maki S, Yasaka M, Takaki Y, Matsui T, Tanabe H, Iwashita A, Ota A. Usefulness of magnifying endoscopy with narrow-band imaging for determining the horizontal extent of early gastric cancer when there is an unclear margin by chromoendoscopy (with video). Gastrointest Endosc. 2011;74:1259-1267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 137] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 8. | Yao K, Nagahama T, Hirai F, Sou S, Matsui T, Tanabe H, Iwashita A, Ragunath K. Clinical application of magnification endoscopy with NBI in the stomach and the duodenum. Cohen J, editor. Comprehensive atlas of high-resolution endoscopy. Oxford: Blackwell Publishing 2007; 83-103. [DOI] [Full Text] |
| 9. | Muto M, Nakane M, Katada C, Sano Y, Ohtsu A, Esumi H, Ebihara S, Yoshida S. Squamous cell carcinoma in situ at oropharyngeal and hypopharyngeal mucosal sites. Cancer. 2004;101:1375-1381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 277] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 10. | Muto M, Minashi K, Yano T, Saito Y, Oda I, Nonaka S, Omori T, Sugiura H, Goda K, Kaise M, Inoue H, Ishikawa H, Ochiai A, Shimoda T, Watanabe H, Tajiri H, Saito D. Early detection of superficial squamous cell carcinoma in the head and neck region and esophagus by narrow band imaging: a multicenter randomized controlled trial. J Clin Oncol. 2010;28:1566-1572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 547] [Article Influence: 34.2] [Reference Citation Analysis (1)] |
| 11. | Goda K, Tajiri H, Ikegami M, Urashima M, Nakayoshi T, Kaise M. Usefulness of magnifying endoscopy with narrow band imaging for the detection of specialized intestinal metaplasia in columnar-lined esophagus and Barrett's adenocarcinoma. Gastrointest Endosc. 2007;65:36-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 116] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 12. | Osawa H, Yamamoto H. Present and future status of flexible spectral imaging color enhancement and blue laser imaging technology. Dig Endosc. 2014;26 Suppl 1:105-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 112] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 13. | Yoshida N, Hisabe T, Inada Y, Kugai M, Yagi N, Hirai F, Yao K, Matsui T, Iwashita A, Kato M, Yanagisawa A, Naito Y. The ability of a novel blue laser imaging system for the diagnosis of invasion depth of colorectal neoplasms. J Gastroenterol. 2014;49:73-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 103] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 14. | Dohi O, Yagi N, Onozawa Y, Kimura-Tsuchiya R, Majima A, Kitaichi T, Horii Y, Suzuki K, Tomie A, Okayama T, Yoshida N, Kamada K, Katada K, Uchiyama K, Ishikawa T, Takagi T, Handa O, Konishi H, Naito Y, Itoh Y. Linked color imaging improves endoscopic diagnosis of active Helicobacter pylori infection. Endosc Int Open. 2016;4:E800-E805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 96] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 15. | Commission Internationale de l’Eclairage. Colorimetry, 3rd Edition Publication CIE 15: 2004. Available from: http://www.cie.co.at/publications/colorimetry-3rd-edition. |
| 16. | Yoshifuku Y, Sanomura Y, Oka S, Kurihara M, Mizumoto T, Miwata T, Urabe Y, Hiyama T, Tanaka S, Chayama K. Evaluation of the visibility of early gastric cancer using linked color imaging and blue laser imaging. BMC Gastroenterol. 2017;17:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 17. | Kanesaka T, Uedo N, Yao K, Ezoe Y, Doyama H, Oda I, Kaneko K, Kawahara Y, Yokoi C, Sugiura Y, Ishikawa H, Kato M, Takeuchi Y, Muto M, Saito Y. A significant feature of microvessels in magnifying narrow-band imaging for diagnosis of early gastric cancer. Endosc Int Open. 2015;3:E590-E596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Ohashi A, Niwa Y, Ohmiya N, Miyahara R, Itoh A, Hirooka Y, Goto H. Quantitative analysis of the microvascular architecture observed on magnification endoscopy in cancerous and benign gastric lesions. Endoscopy. 2005;37:1215-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Kaise M, Kato M, Urashima M, Arai Y, Kaneyama H, Kanzazawa Y, Yonezawa J, Yoshida Y, Yoshimura N, Yamasaki T, Goda K, Imazu H, Arakawa H, Mochizuki K, Tajiri H. Magnifying endoscopy combined with narrow-band imaging for differential diagnosis of superficial depressed gastric lesions. Endoscopy. 2009;41:310-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 104] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 20. | Araki Y, Sasaki Y, Hanabata N, Yoshimura T, Sawaya M, Hada R, Fukuda S. Morphometry for microvessels in early gastric cancer by narrow band imaging-equipped magnifying endoscopy. Dig Endosc. 2011;23:233-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Adachi Y, Mori M, Enjoji M, Sugimachi K. Microvascular architecture of early gastric carcinoma. Microvascular-histopathologic correlates. Cancer. 1993;72:32-36. [PubMed] |
| 22. | Takemura S, Iwashita A, Yao K, Yao T. The correlation between the quantified microvascular density and the endoscopic color in elevated type of gastric neoplastic lesions. Gastroenterol Endosc. 2002;44:745-754. [DOI] [Full Text] |
| 23. | Honmyo U, Misumi A, Murakami A, Mizumoto S, Yoshinaka I, Maeda M, Yamamoto S, Shimada S. Mechanisms producing color change in flat early gastric cancers. Endoscopy. 1997;29:366-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 24. | Furukawa K, Yao K, Iwashita A, Matsui T, Yao T. Microvascular architecture of depressed type early gastric cancer -quantified vascular density using computer analysis with special reference to the color of endoscopic findings. Gastroenterol Endosc. 1997;39:1358-1369. [DOI] [Full Text] |
| 25. | Yamaguchi H, Saito T, Shiraishi Y, Arai F, Morimoto Y, Yuasa A. Quantitative study on appearance of microvessels in spectral endoscopic imaging. J Biomed Opt. 2015;20:036005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See:
P- Reviewer: Kim YJ, Petrucciani N S- Editor: Ma RY L- Editor: A E- Editor: Yin SY